3.1. The Morphology and Mechanical Properties of Modified MoS2 Powder and Its Interface Formation with the Matrix
The application of Cu and Ni coatings on MoS
2 alters the particle morphology compared to uncoated MoS
2. Consequently, prior to experimentation, a comprehensive and in-depth analysis of the characteristics of MoS
2 powders with different coatings is essential.
Figure 4 illustrates the typical morphological features of MoS
2 with different coatings. It can be observed from the figure that both types of MoS
2 particles exhibit complex irregular shapes. The surfaces of the MoS
2 particles (MoS
2@Cu and MoS
2@Ni), however, were fully covered by a relatively dense metallic coating. The observed particle morphology is a direct consequence of the preparation process used. MoS
2 is mostly fine powder prepared by chemical methods. To achieve a specific particle size, granulation is often employed to agglomerate the fine powder, resulting in larger particles of a uniform size. Therefore, the particles usually show complex shapes. Typically, agglomerated MoS
2 particles possess a rougher surface and a higher specific surface area. This characteristic is beneficial for the deposition of a metal coating during the subsequent coating process, which in turn facilitates the formation of MoS
2 particles with a smooth and continuous metallic layer. Consequently, this enhances the bonding strength between MoS
2 and the matrix.
As shown in
Figure 5a, the black phase corresponds to MoS
2-Cu, which appears to be well-integrated within the Cu matrix at this magnification.
Figure 5b illustrates the microstructure of MoS
2@Ni, showing that MoS
2@Ni also forms a stable interface with the matrix.
Figure 5c,d show the microstructures of Cu-MoS
2@Cu and Cu-MoS
2@Ni, respectively. The modified MoS
2 particles are dispersed throughout the Cu matrix, although some degree of agglomeration and local variation in particle density can be observed, which is typical for powder metallurgy processed composites.
Figure 6 presents the microhardness values of the Cu-MoS
2@Cu and Cu-MoS
2@Ni samples, as well as their respective Cu matrix. The two materials exhibited relatively low and comparable microhardness. It is noteworthy that the copper matrix in the Cu-MoS
2@Ni material demonstrated higher hardness than that in the Cu-MoS
2@Cu material. This difference is attributed to the solid solution strengthening effect of nickel, which forms an infinite solid solution with copper. In contrast, MoS
2 reacts with copper during high-temperature sintering to form brittle phases [
30]. Importantly, nickel does not react with MoS
2 under the same conditions. This indicates that the incorporation of MoS
2@Ni not only suppresses interfacial reactions but also increases the hardness of the Cu matrix and enhances the interfacial bonding strength.
Figure 7 and
Figure 8 illustrate the detailed cross-section of Cu-MoS
2 with different coatings and its interface with the matrix. The MoS
2 with a Ni coating formed a tighter interfacial bond with the matrix. However, the MoS
2@Cu seemed to have reacted with the matrix, led to the formation of some flocculent interface products. Further, EDS line scanning was employed to analyze the elemental changes at the interface between MoS
2@Cu or MoS
2@Ni with the matrix. For the MoS
2@Ni/Cu interface, the concentrations of Cu and Ni elements at the interface shows a gradually increased tendency, indicating that there is a significant diffusion of metal elements at the interface. Although the MoS
2@Cu/Cu interface also exhibits a subtle gradient change in interface metal elements, when scanning areas close to the interface reaction region, severe fluctuations in element concentration commence to occur. This very phenomenon proved the occurrence of the interface reaction. EDS analysis also indicated that the Ni coating contained P, suggesting that the deposited layer was a typical Ni–P alloy coating. The presence of P in the Ni–P coating may enhance hardness and corrosion resistance, which contributes to improved interfacial load-bearing capacity during micro-friction testing. In contrast, the Cu coating remains relatively soft and chemically active toward MoS
2, facilitating interfacial reactions during sintering.
To further explore the decomposition behavior of the two kinds of MoS
2 after sintering, the phase composition of the Cu-MoS
2@Cu and Cu-MoS
2@Ni materials was characterized by XRD analysis, as shown in
Figure 9. It is observed that after sintering, the sample containing MoS
2@Cu exhibits a slight decomposition of MoS
2, resulting in the formation of new sulfide phases, such as Cu1.96S and Cu5.40Mo18S24. In contrast, for the sample containing MoS
2@Ni, the phase composition remains largely unchanged before and after sintering.
The results of EDS line analysis and XRD analysis indicate that, during the sintering process, MoS
2@Cu exhibited a higher tendency towards decomposition. This phenomenon can be ascribed to a sequence of reactions occurring between MoS
2 and Cu. The reaction equations are as follows:
The △G of the reactions mentioned above are all consistently less than zero, indicating that these reactions took place spontaneously during the sintering process, with a sintering temperature of 870 °C; this was calculated using the Gibbs-free energy calculation software HSC Chemistry 6.0. This explains why multiple sulfides were detected in the XRD after sintering in the Cu-MoS2@Cu samples.
However, the reaction equation of MoS
2 and Ni is as follows:
The reaction temperatures required for the above reaction equation all exceed 1100 °C (calculated using the Gibbs-free energy calculation software HSC Chemistry 6.0), which means that in this study, the Ni coating is capable of fulfilling a protective function, effectively inhibiting the significant reaction between MoS2 and the matrix. The analysis indicated the formation of two distinct interfacial structures with the matrix: MoS2@Ni exhibited a dense diffusion-bonded interface, as opposed to the defective diffusion-reaction bonded interface observed with MoS2@Cu.
3.2. Micro-Friction Tests
During actual braking, sliding friction occurs between the friction material and the counterpart. Since the Brinell hardness of the counterpart is significantly higher than that of the friction material, the friction process can be regarded as the slippage of microscopic asperities on the counterpart’s surface over the friction material’s surface. Therefore, investigating the micro-friction (micro-scratch) characteristics of the wear constituents and their interfaces with the matrix aims to provide an in-depth understanding of the friction performance and wear mechanisms exhibited by the friction material under real braking conditions. Understanding the micro-friction behavior helps explain the wear mechanisms of the composites and provides insight into the design of braking materials, improving their stability and durability under extreme conditions.
Figure 10 and
Figure 11 show the scratch morphologies of the interfaces after micro-friction testing. In these figures, the black phase represents MoS
2@Cu or Cu-MoS
2@Ni, while the brighter phase corresponds to the Cu matrix. Because MoS
2 appears dark in the SEM images, scratches on MoS
2 are less visible than those on the matrix. An examination of micro-friction morphology on the interface and matrix reveals that as pressure increases, the width of the scratches significantly increases. While both samples exhibit generally similar scratch morphologies, notable differences are observed at the interfaces. However, the MoS
2@Ni matrix interface demonstrates better resistance to damage under the same pressure conditions compared to the MoS
2@Cu matrix interface.
Figure 10 and
Figure 11 reveal the micro-frictional topography at the MoS
2/matrix interfaces in greater detail in their right-hand sections. Under low applied pressure, the MoS
2@Ni matrix interface remained intact, whereas the MoS
2@Cu matrix interface showed signs of debonding and micro-cracking. Comparative analysis indicates that the load-bearing capacity of MoS
2@Cu matrix interface is lower than that of MoS
2@Ni. Specifically, interfacial delamination initiated near the MoS
2@Cu interface at 0.05 N. Upon increasing the pressure to 0.075 N, the MoS
2@Ni interface maintained its integrity, while the MoS
2@Cu interface developed cracks and incipient failure, demonstrating the superior interfacial integrity of the former. As the pressure gradually increases to 0.15 N, MoS
2 at the interface starts to show noticeable laminar slip, and a significant number of MoS
2 flakes begin to form. Under this experimental condition, the delamination of MoS
2 near the MoS
2@Cu matrix became more pronounced. Based on the observed interfacial failure patterns, the MoS
2@Cu interface exhibited significant interfacial debonding, whereas the MoS
2@Ni interface maintained robust interfacial adhesion despite the fracture occurring within the MoS
2.
The scratch characteristics of the interfaces between MoS
2 and matrix exhibit a strong correlation with the structural properties of MoS
2 and the performance of the interface between MoS
2 and the Cu matrix. MoS
2@Ni forms a stronger diffusion-bonded interface with the Cu matrix, leading to such interfaces being able to maintain better adhesion during the micro-friction tests. The strong interface of the MoS
2@Ni matrix formed due to two main factors. First, at an 870 °C sintering temperature, Ni does not react with MoS
2, delaying the MoS
2-Cu reaction and reducing interfacial products. Second, the Ni-Cu diffusion rate is faster than Cu-Cu, promoting a high-strength diffusion bonding interface between the Ni coating and the matrix [
39]. Thus, the Cu-MoS
2@Ni interface shows no significant damage under various pressures. Nevertheless, MoS
2@Cu formed a diffusion–reaction mixed bonding interface with the matrix. Due to the presence of interface defects, the bonding strength of this interface was relatively low. Consequently, interfacial debonding began to occur even under a test pressure of 0.075 N. Additionally, the relatively low strength of MoS
2 and its layered structure render MoS
2 highly susceptible to damage during micro-friction tests. This may even result in failure prior to the debonding of the interface. Therefore, during the micro-friction tests, the inability of MoS
2 constituents and the interface debonding might occur simultaneously, collectively contributing to the interface failure. Notably, owing to the occurrence of interfacial reactions, the MoS
2@Cu and MoS
2@Cu matrix interface is more susceptible to damage in comparison with MoS
2@Ni, leading to the damage of the interface even under the lowest pressure. Overall, with increasing pressure, the failure mechanism of the MoS
2@Cu interface transitions from localized interfacial debonding to failure within the interfacial reaction layer and delamination of MoS
2. For MoS
2@Ni, the interfacial failure mechanism is characterized by delamination of MoS
2 near the interface under high test pressure.
Figure 12 displays the variation in residual depth (hrd) and penetration depth (hpd) with sliding distance for the interface regions A, B, and C in Cu-MoS
2@Cu and Cu-MoS
2@Ni samples under different pressure conditions. In micro-friction testing, both residual depth (hrd) and penetration depth (hpd) are critical indicators for evaluating hardness and deformation behavior (hpd refers to the maximum depth the indenter penetrates into the material’s surface under applied load, typically used to assess the material’s stiffness and hardness). Harder materials generally show smaller penetration depths. On the other hand, hrd represents the residual indentation depth remaining after unloading with elevated test pressures; however, the variation trend of the indentation depth exhibits distinct patterns depending on the specific characteristics of the test region. The permanent deformation depth left after the indenter is removed from the material’s surface during the unloading process reflects the extent of deformation, with softer materials typically leaving deeper residual depths. In the figures, A, B, and C represent three distinct test regions, referring to Cu-MoS
2@Cu or the Cu-MoS
2@Ni phase region, the interface region, and the matrix, respectively.
Figure 12 presents the variations in hrd and hpd with sliding distance under different pressure conditions, as shown in
Figure 10 and
Figure 11. As illustrated, with an increasing sliding distance, the hrd and hpd show a discernible decreasing trend. In addition, the hrd and hpd consistently increase with elevated test pressures; however, the variation trend of the hrd and hpd exhibit distinct patterns depending on the specific characteristics of the test region. Owing to its relatively low hardness and propensity for interlayer delamination failure, the MoS
2 region always exhibits a greater indentation depth and residual depth during mechanical testing. Under identical testing conditions, the indentation depth and residual depth in the MoS
2@Cu phase exhibited relatively higher values. Particularly at a testing load of 0.15 N, the maximum indentation depth reached 8500 nm, and the residual depth reached 6300 nm. Conversely, the matrix region exhibits relatively lower indentation depth and residual depth due to its high hardness and toughness. The interfacial region, serving as a bridge connecting MoS
2 and the substrate, exhibited a progressive reduction in both indentation depth and residual depth. The reduction in these depths became progressively smaller. Furthermore, the decrease in hpd and hrd at the MoS
2@Ni/matrix interface was more gradual compared to that of MoS
2@Cu.
In micro-scratch hardness testing, both hpd and hpd are associated with the material’s hardness and deformation characteristics, respectively. The indicator hpd refers to the scratch depth at which the indenter penetrates the material’s surface under applied loads, and is typically used to describe the actual insertion depth during the testing process. Harder materials generally show smaller penetration depths. On the other hand, hrd, representing the residual depth, is employed to reflect the extent of material deformation after the test. MoS2 exhibits relatively low hardness and is prone to tangential deformation during testing, resulting in increased hpd and hpd, whereas the relatively harder Cu-matrix shows a comparatively lower hpd and hrd.
The variation in indentation depth at the interface is associated with the load-bearing capacity of the interface. Generally speaking, the higher the interfacial strength between the two phases, the larger the load-bearing capacity of the interface. When the indenter slides across a high-strength interface, the change in indentation depth is more gradual. Conversely, a low-strength interface may be damaged as the indenter slides across, resulting in a sudden change in indentation depth due to the partial loss of load-bearing capacity. Therefore, micro-scratch tests, combined with post-experiment interfacial morphology characteristics, allow for a qualitative assessment of the interfacial performance between two phases. As shown in
Figure 12, the indentation depth in interfacial region of the Cu-MoS
2@Cu sample is higher compared to the Cu-MoS
2@Ni interface. The change in indentation depth at the MoS
2@Cu and Cu matrix interface initially fast increases and then decreases, indicating that the interface has been compromised under test pressures of 0.075 N and 0.15 N, leading to a deterioration in its load-bearing capacity. This result corresponds with the interfacial debonding phenomenon observed in
Figure 10, clearly demonstrating the inferior mechanical performance of the Cu-MoS
2@Cu matrix interface. This further confirms that the strength of the diffusion–reaction bonding interface in Cu-MoS
2@Cu is lower than that of the diffusion bonding interface in Cu-MoS
2@Ni.
Figure 13 presents the variation in coefficient of friction (COF) with sliding distance for the interfacial regions of Cu-MoS
2@Cu and Cu-MoS
2@Ni samples under different applied pressures. As illustrated, the coefficient of friction demonstrates an overall increasing trend with elevated pressure, exhibiting the most pronounced enhancement when the pressure reaches 0.15 N. A careful observation of the variation in the coefficient of friction with respect to sliding position reveals that the coefficient of friction remains relatively low when the indenter slides on the MoS
2 phase. However, upon crossing the interface, the COF begins to increase, particularly when the applied test pressure reaches 0.15 N, at which a rapid escalation in the COF is observed. A comparative analysis of two distinct interface types indicates that under loading conditions of 0.05 N and 0.075 N, the friction coefficient at the interface maintains relative stability. Nevertheless, when the testing pressure is increased to 0.15 N, the MoS
2@Cu/Cu interface shows a more prominent and abrupt change in the friction coefficient, with the maximum value possibly surpassing 0.6.
Based on the principles of adhesive friction theory [
37], the coefficient of friction is given by:
In the equation, μa represents an adhesive component of COF, and μp represents a plowing component of COF. The adhesive component during the sliding process can be calculated using the following equation [
38]:
In the equation, τ represents the material’s shear strength, and Hv denotes the material’s hardness. Equation (13) indicates that the adhesive friction component of the material during the testing process is only related to the intrinsic properties of the material and is independent of the testing conditions.
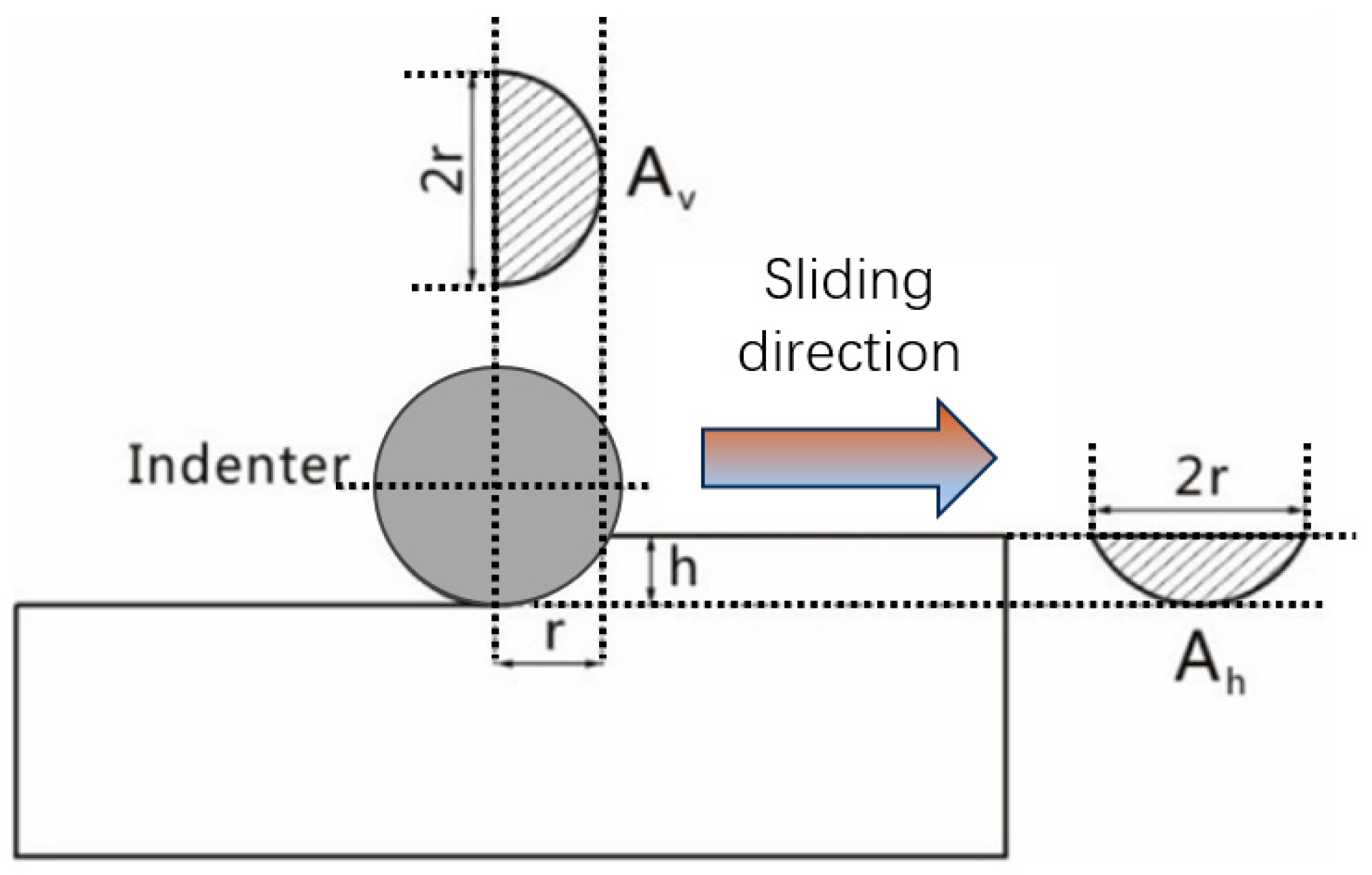
A schematic of the micro-scratch test is presented in
Figure 14. The plowing friction coefficient,
which neglects the effect of elastic recovery on the contact contour, is given by:
In this expression, is defined as the plowing component of the friction force. and are the horizontal and vertical projected areas of the contact zone, respectively, with the latter under plastic deformation. and correspond to the yield/compressive strength of the friction material along the sliding direction and the vertical direction, respectively.
For MoS2, its relatively low hardness results in a higher Ah/Av ratio. However, during the scratching process, the layered structure of MoS2 tends to shear and spread along the sliding direction, leading to extremely low values of both and τ in that direction. With the escalation of test pressure, the enhancement of the / ratio constitutes the main factors leading to growth in the COF. Material properties and structural characteristics of MoS2 facilitate the development of a low COF, thereby enabling superior lubrication performance.
For the friction calculation of the copper matrix, the matrix is a homogeneous material, and the yield stress in all directions is the same. Therefore, the ploughing component of the COF can be simplified as [
21]
In the equation, the R is the radius of indenter. Based on the correlation between indentation depth and hardness [
9],
In the equation, Fn is the normal force. From Equations (13) and (14), it can be observed that the COF on the matrix is primarily governed by the variable plowing component. It is positively correlated with pressure and negatively correlated with hardness. The rapid increase in the COF from MoS2 to the matrix is attributed to two important factors: firstly, the enhancement of the adhesive component of COF due to the improved tangential strength of the surface; secondly, the swift rise in the / ratio. Consequently, even when the indenter operated on the matrix, leading to a reduction in the / ratio, the COF still exhibits a significant elevation.
The friction coefficient variation at the interface exhibits a more complex pattern. The variation in the COF correlates with the area distribution of the indenter’s contact with the material during the micro-friction tests, when the interface exhibits adequate load-bearing capacity and maintains structural integrity. The COF can be calculated as follows:
In the formula, represents the coverage area of MoS2 on the contact surface. Under these circumstances, the variation in the COF should exhibit an approximately linear relationship with the sliding movement of the indenter. Under the operational conditions of 0.05 N and 0.075 N for both samples, the variation in the COF at the interface distinctly adheres to the pattern delineated by Equation (13). However, when the applied pressure was increased to 0.15 N, severe interfacial failure occurred at the Cu-MoS2@Cu interface, resulting in a rapid increase in indenter penetration depth. This led to an elevation in the Ah/Av ratio, causing a steep rise in the COF. Subsequently, as the indenter gradually slid onto the substrate, the friction coefficient began to decrease slowly. As a result, the COF at the Cu-MoS2@Cu interface formed a characteristic peak in the COF curve.
Due to its higher bonding strength, the MoS2@Ni-Cu matrix interface provides better support to the indenter during sliding. Consequently, its variations in indentation depth and COF were more stable compared to those of the MoS2@Cu-Cu matrix interface. The higher bonding strength of the MoS2@Ni-Cu matrix interface compared to the MoS2@Cu-Cu matrix interface, as evidenced by the COF variations indicative of interfacial failure in micro-scratch testing, accounts for its more stable tribological behavior.
3.3. Macroscopic Frictional Behavior of the Cu-Based Composites
Macro-friction tests were conducted on a scaled-down friction tester to investigate the tribological behavior of Cu-based composites containing two modified lubricating components under different braking energy densities. This research explores the characteristics of the modified lubricating components, their interface features with the matrix, and their relationship to friction and wear performance.
Table 4 presents the theoretical density, measured density, and relative density values of Cu-BC-MoS
2@Cu and Cu-BC-MoS
2@Ni materials. As shown in the table, the measured density of Cu-BC-MoS
2@Ni (7.92 g/cm
3) is higher than that of Cu-BC-MoS
2@Cu (7.88 g/cm
3), with both materials exhibiting similar densities. It can be found that, during the sintering process, the reaction between MoS
2 and Cu is more extensive in Cu-BC-MoS
2@Cu than in Cu-BC-MoS
2@Ni, resulting in the formation of more brittle phases. Additionally, Cu and Ni form an infinitely soluble solid solution with fewer internal defects, thus slightly increasing the density of the Cu-BC-MoS
2@Ni material.
Porosity plays a critical role in determining the tribological behavior of composite materials. As presented in
Table 4, both composites exhibit relative densities of approximately 91–92%, corresponding to a porosity level of about 8–9%. Notably, Cu BC MoS
2@Ni demonstrates a slightly higher density (7.92 g/cm
3 vs. 7.88 g/cm
3) and thus marginally lower porosity compared to its counterpart. The presence of porosity exerts a dual influence on tribological performance. On one hand, pores can act as stress concentrators, potentially reducing local mechanical strength and facilitating crack initiation under cyclic loading, particularly under high-energy-density conditions. On the other hand, pores may serve as reservoirs for wear debris and contribute to the mechanical interlocking of tribolayers, which can, in some cases, enhance the adhesion of oxide films. In the present study, for Cu BC MoS
2@Cu, which possesses a weaker interfacial bonding and higher porosity, the synergistic effect of these factors exacerbates subsurface crack propagation, thereby promoting the occurrence of delamination wear at an energy density of 100 J/mm
2. In contrast, the denser matrix (attributed to slightly lower porosity) and stronger interfacial bonding of Cu BC MoS
2@Ni effectively inhibit crack growth, confining wear predominantly to the oxide layer rather than extending into the bulk material. Furthermore, the marginally reduced porosity in Cu BC MoS
2@Ni contributes to its higher microhardness (
Figure 6) and enhanced resistance to plastic deformation during sliding, which aligns well with its more stable friction coefficient and lower wear rate.
Figure 15 shows the variation in the friction coefficients of Cu-BC-MoS
2@Cu and MoS
2@Ni materials with the number of braking cycles. As shown in
Figure 16, under the same velocity conditions, both materials exhibit high stability in their friction coefficients. However, MoS
2@Ni demonstrates a more stable friction coefficient compared to MoS
2@Cu. Furthermore, with an increase in speed, the friction coefficients of both materials tend to decrease, with MoS
2@Ni maintaining good stability.
Figure 15d,e show that the friction curves of both materials generally exhibit a saddle-shaped profile. As the energy density increases, the curvature on both sides of the friction curve becomes more pronounced. The appearance of this saddle-shaped friction curve is primarily related to the contact state during the friction process. During the initial stage of the friction test, the impact generated by the contact of the friction pair is the main factor leading to the initial peak in the braking curve. In the middle stage, the formation of a friction film causes the instantaneous COF to gradually decrease and stabilize. At the final stage, due to the decreasing rotational speed and intensified heat accumulation at the friction interface, the interlocking engagement between the friction material and the counterpart is enhanced, resulting in a rapid increase in the COF. This manifests as a more pronounced “tail-up” phenomenon in the friction termination stage with increasing friction speed.
The slightly lower friction coefficient of Cu-BC-MoS2@Ni compared to Cu-BC-MoS2@Cu can be attributed to the formation of a weaker reaction–diffusion interface between MoS2@Ni and the copper substrate. In contrast, MoS2@Cu reacts with the substrate to form a stronger reaction interface, which, although having lower strength, is more easily damaged during friction. This results in the generation of fine abrasive particles that contribute to third-body wear, thereby enhancing friction. MoS2 has a hexagonal close-packed crystal structure, with strong in-plane atomic bonding and relatively weak interlayer bonding. During friction, the interlayer chemical bonds of MoS2 break, forming a lubricating sulfide film that provides excellent lubrication performance. Furthermore, the interface between MoS2@Ni and the substrate generates fewer sulfides and hard phases, preserving more MoS2, thereby enhancing its lubricating effect.
As shown in
Figure 15 and
Figure 16, both Cu-BC-MoS
2@Cu and Cu-BC-MoS
2@Ni exhibit good stability coefficients under low-energy-density conditions. As the energy density increases, the higher hardness of Cu-BC-MoS
2@Ni leads to reduced material stability during friction. However, the continuous lubrication effect of MoS
2 reduces the tendency for adhesion, allowing Cu-BC-MoS
2@Ni to maintain stability at high speeds, with minimal change in stability compared to Cu-BC-MoS
2@Cu. Furthermore, the stronger interfacial bonding between Cu and Ni enhances the material’s resistance to deformation, thereby reducing frictional resistance and lowering the friction coefficient. Furthermore, the high heat capacity of molybdenum (Mo) during its decomposition enables it to absorb a significant amount of frictional heat, lowering the surface temperature and thereby further promoting the stability of the COF.
Figure 17 presents the wear rates of Cu-BC-MoS
2@Cu and Cu-BC-MoS
2@Ni materials, along with their comparative materials, under different energy densities.
Figure 18 illustrates the evolution of the friction surface and the transition of the wear mechanism of Cu-BC-MoS
2@Cu and Cu-BC-MoS
2@Ni composite materials based on copper, at energy densities of 30, 50, and 100 J/mm
2.
At a low energy density of 30 J/mm2, the accumulation of frictional heat is restricted, thereby preventing the formation of a stable lubricating film on the surface. Friction primarily occurs through the mechanical action of micro-protrusions. For Cu-BC-MoS2@Cu, the weak bonding at the Cu-MoS2 interface leads to early consumption of MoS2 through reaction with the copper substrate. This results in the poor retention of the lubricating phase and prevents the formation of an effective lubricating film. This leads to the detachment of hard-phase particles, such as CuMoS2, contributing to third-body wear, during friction. The surface exhibits typical plowing wear morphology, with numerous plowing grooves and pits, and even exposes the Cu substrate. In contrast, Cu-BC-MoS2@Ni forms solid solutions with the Cu substrate, producing solid-solution strengthening and a stronger interface bond. This retains more MoS2 and allows for the formation of an effective lubricating film. As a result, the surface wear is lighter, primarily showing plowing wear with only minor adhesive phenomena.
As the energy density increases to 50 J/mm2, the interface temperature and contact pressure rise significantly, leading to surface oxidation and the formation of a light gray oxide film. For Cu-BC-MoS2@Cu, the increase in oxide film hardness reduces the number of plowing grooves. However, due to the relatively weak bonding at the Cu-MoS2 reaction interface, the oxide film is prone to detachment from the substrate, resulting in flaky spalling pits. This causes the wear mechanism to transition from purely plowing wear to a composite mechanism, including plowing, adhesion, and minor oxidative wear. On the other hand, due to the high-strength diffusion bond interface between Ni and MoS2, Cu-BC-MoS2@Ni forms a more compact oxide film, which is less prone to spalling. The surface integrity is better, and the overall wear rate is lower compared to Cu-BC-MoS2@Cu.
At medium energy density, although Cu-BC-MoS2@Ni forms a more compact oxide film than at low energy density, the film formation rate is slower than its consumption rate during friction. Therefore, at this medium energy density, the wear rate of Cu-BC-MoS2@Ni is the highest. For Cu-BC-MoS2@Cu, the oxide film can form under medium-energy-density friction, whereas Cu-BC-MoS2@Cu cannot generate an oxide film under low energy density and lacks the lubrication from residual MoS2 during the friction process. As a result, the wear rate of Cu-BC-MoS2@Cu is the lowest at medium energy density.
When the energy density is further increased to 100 J/mm2, the accumulation of frictional heat intensifies, and both materials’ surfaces become covered with a continuous, smooth oxide film. The number of spalling pits increases significantly, and deep pits begin to appear. On Cu-BC-MoS2@Ni, the oxide film is firmly bonded to the substrate, forming a dense oxide layer with localized high-temperature oxidation zones, indicating a more complete oxidation reaction. This further enhances the surface hardness and wear resistance. At this stage, although the wear mechanism primarily involves oxide film fatigue delamination, the degree of spalling is lighter, and the wear rate is lower than that observed at medium energy densities.
For Cu-BC-MoS2@Cu, due to insufficient interface bonding strength, the oxide film undergoes fatigue delamination under cyclic shear stress, with large-scale flaky spalling becoming the predominant wear mechanism. This results in a rapid increase in the wear rate, which reaches its highest value across all three energy densities. In contrast, the oxide film on Cu-BC-MoS2@Ni is strongly bonded to the substrate, featuring a dense oxide layer and localized high-temperature oxidation zones, which suggest a more complete oxidation reaction. These factors contribute to an enhancement in surface hardness and wear resistance. Although the wear mechanism still primarily involves fatigue delamination of the oxide film, the extent of spalling is less severe, and the wear rate is lower than that observed at medium energy densities.
Figure 19 illustrates the evolution of the cross-sectional structure under varying braking energy density (BED) conditions. As the rotational speed increases, distinct subsurface structures gradually emerge.
As shown in
Figure 19a,b, at low BED, neither Cu-BC-MoS
2@Cu nor Cu-BC-MoS
2@Ni exhibit a well-defined subsurface structure. Instead, only discontinuous mechanical mixing and plastic deformation are observed. When the BED increases to 50 J/mm
2, although neither of the two composite materials forms a continuous subsurface structure, Cu-BC-MoS
2@Cu shows a discontinuous mechanical mixing layer with minor plastic deformation, as depicted in
Figure 19c. In contrast, Cu-BC-MoS
2@Ni maintains superior integrity after testing, exhibiting some discontinuous plastic deformation layers.
As the BED increases further, a mechanical mixing layer approximately 20 µm thick, enriched with iron, forms on the wear surface of the Cu-BC-MoS
2@Cu sample. This layer is filled with cracks. However, as shown in
Figure 19f, on the wear surface of the Cu-BC-MoS
2@Ni, a plastic deformation layer of uneven thickness but continuous structure forms. Additionally, incomplete mechanical mixing layers are observed in some regions of this layer. This progression highlights the gradual development of subsurface structures as the BED increases, with Cu-BC-MoS
2@Ni demonstrating better structural stability compared to Cu-BC-MoS
2@Cu under higher energy conditions.
In conclusion, the interface bonding strength and the stability of the oxide film are critical factors determining the wear behavior of these materials at different energy densities. At low energy densities, interface bonding strength governs the retention of the lubricating phase; at medium energy densities, interface bonding influences the spalling tendency of the oxide film; and at high energy densities, interface bonding strength dictates the fatigue life of the oxide film and the overall wear resistance.
As shown in
Figure 20, the analysis of the friction surfaces indicates that for both Cu-BC-MoS
2@Cu and Cu-BC-MoS
2@Ni materials, under low-energy-density conditions, due to the incomplete formation of MoS
2-containing oxide films, a large number of plow grooves and adhesion pits appear due to third-body wear caused by uneven surface protrusions. Under these conditions, both materials primarily experienced abrasive and adhesive wear. With increasing braking energy densities, the progressive formation of a surface oxide layer and debris coverage occurred. The thicker oxide film developed on Cu-BC-MoS
2@Ni, compared to Cu-BC-MoS
2@Cu, provided more effective protection against matrix wear and fracture. During high-energy braking, the formation rate of the oxide film is lower than its consumption rate, and the substances formed by the reaction between MoS
2 and Cu increase the brittleness of the material. The weaker interfacial bonding makes the material more prone to damage during friction, leading to a shift in the primary wear mechanism of Cu-BC-MoS
2@Cu to fatigue delamination of the material. In contrast, Cu-BC-MoS
2@Ni, with its thicker oxide film, primarily experiences fatigue delamination of the oxide film.