Study and Mathematical Model of the Chemical Composition and Structure of the Compound Sb2(S1−xSex)3 Based on a Correlation of Data Obtained Through XRD and XPS Characterization
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Methodology
2.2. Deposition of Sb2Se3 Precursor Thin Films
2.3. Incorporation of Sulfur into Sb2Se3 Precursor Thin Films
2.4. Characterization
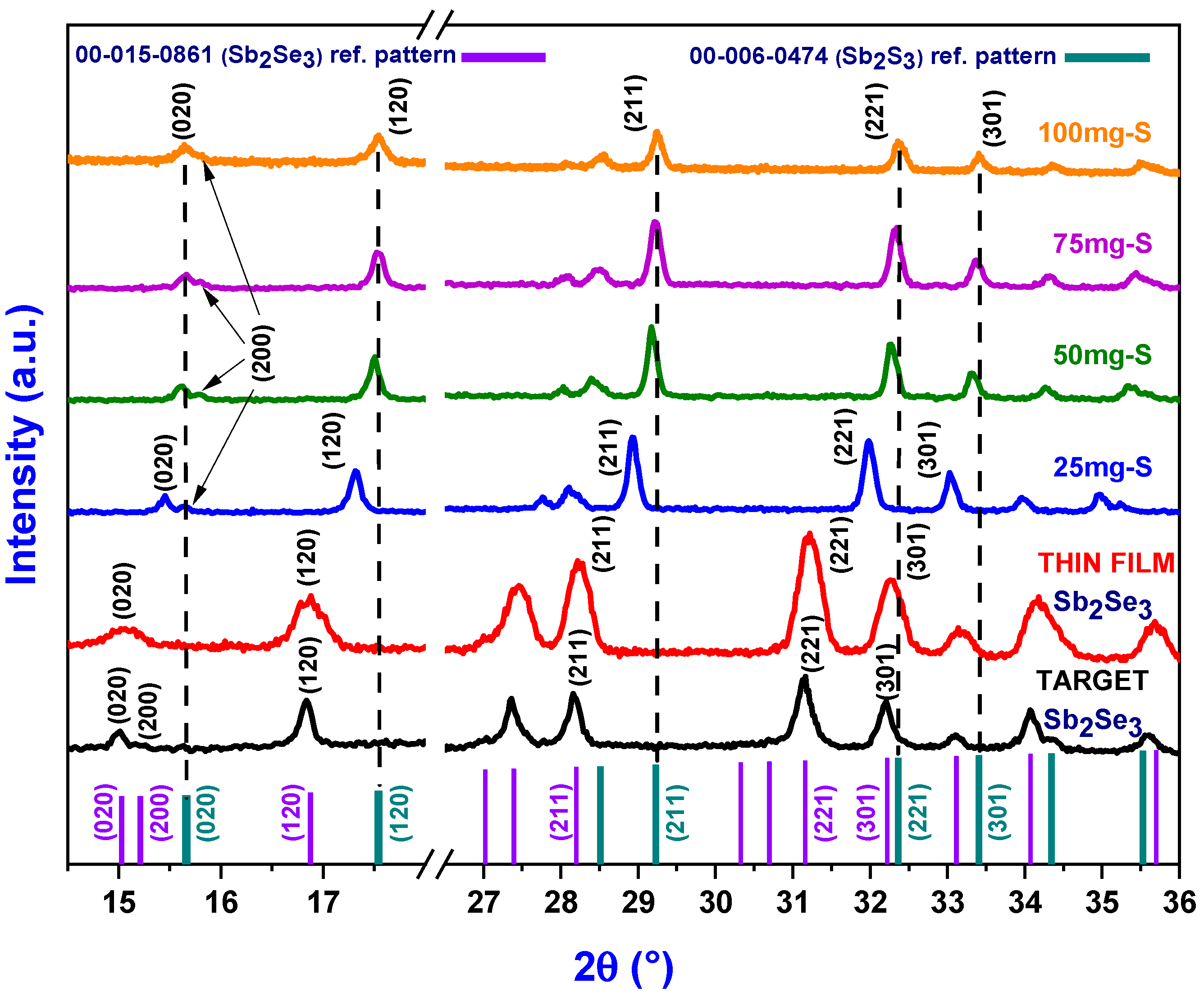
2.4.1. XRD
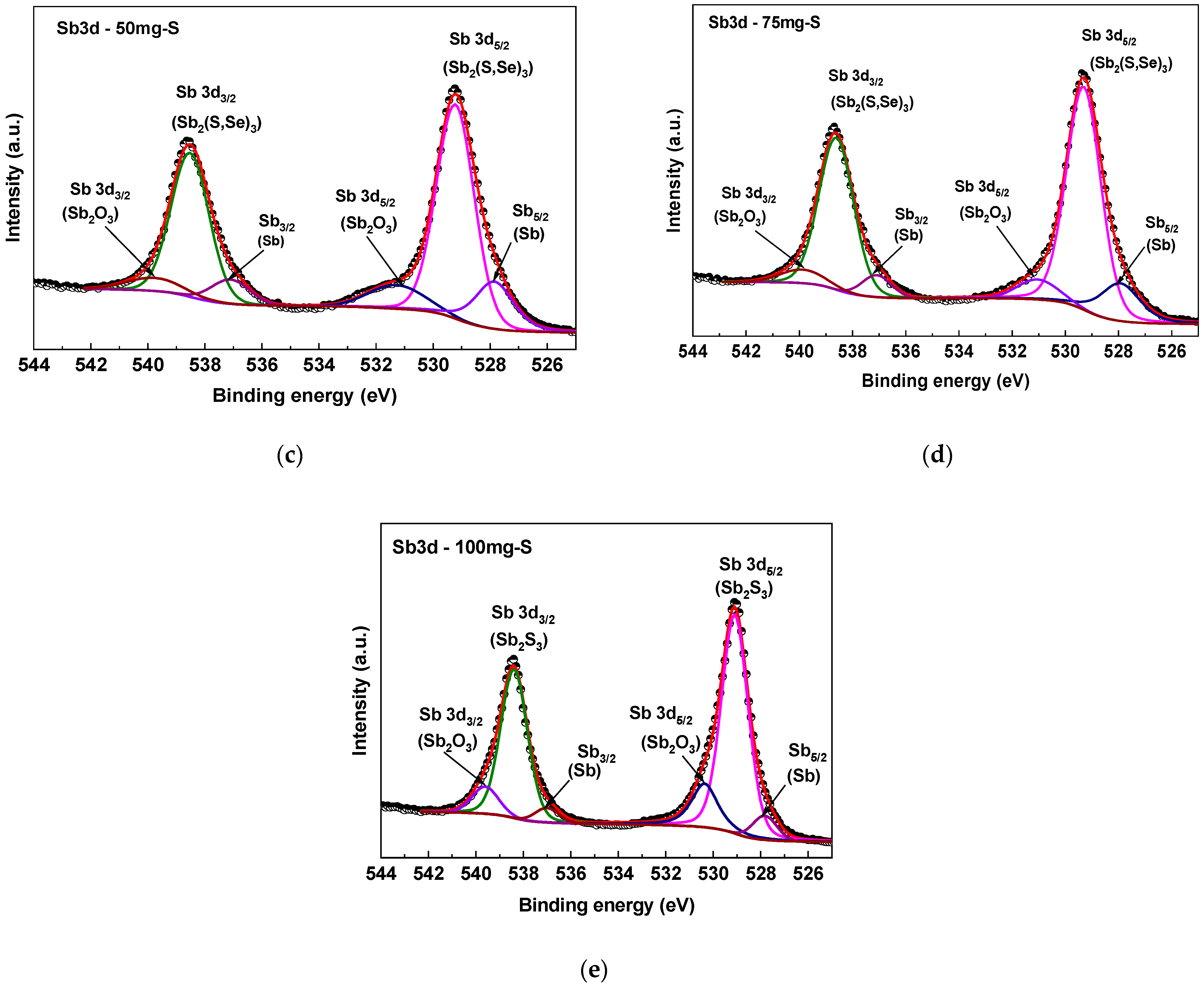
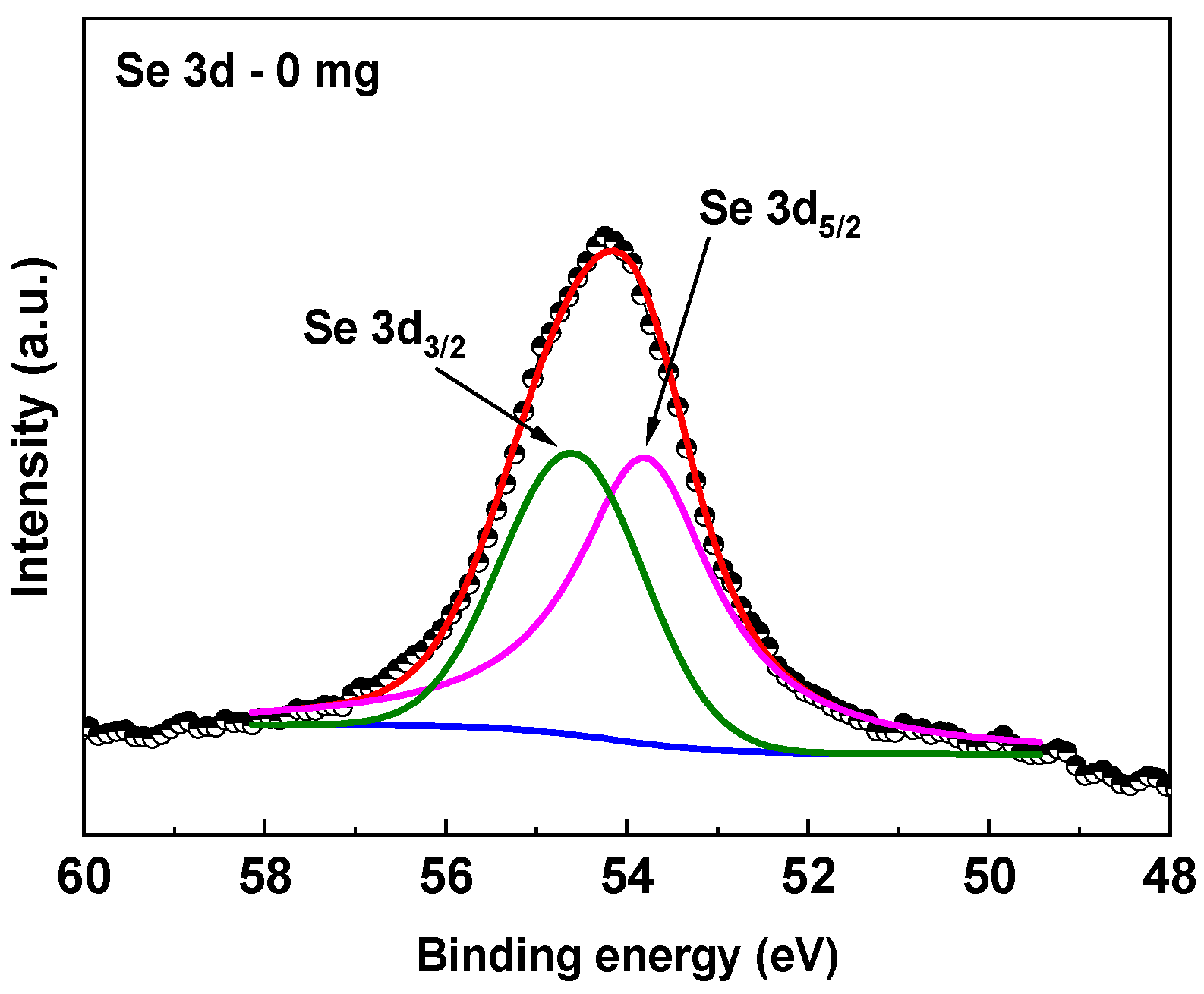
2.4.2. XPS
3. Results
3.1. Structural Characterization of Sb2(S1−xSex)3 Films
3.2. XPS Compositional Analysis of the Films
4. Discussion
4.1. Correlation of XRD and XPS Results

4.1.1. Selenium Losses in Sb2Se3 Thin Films Caused by the Sputtering Deposition Process
4.1.2. Trend Curve Using Only the Points of the References Patterns
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lu, S.; Zhao, Y.; Wen, X.; Xue, D.-J.; Chen, C.; Li, K.; Kondrotas, R.; Wang, C.; Tang, J. Sb2(Se1−x, Sx)3 thin-film solar cells fabricated by single-source vapor transport deposition. Solar RRL 2019, 3, 1800280. [Google Scholar] [CrossRef]
- Nicolás-Marín, M.M.; González-Castillo, J.R.; Vigil-Galán, O.; Courel, M. The state of the art of Sb2(S, Se)3 thin film solar cells: Current progress and future prospect. J. Phys. D Appl. Phys. 2022, 55, 303001. [Google Scholar] [CrossRef]
- Sajitha, D.R.; Stephen, B.; Nakamura, A.; Selvaraj, M.; Salammal, S.T.; Hussain, S. The emergence of chalcogenides: A new era for thin film solar absorbers. Prog. Solid State Chem. 2024, 76, 100490. [Google Scholar] [CrossRef]
- Barthwal, S.; Kumar, R.; Pathak, S. Present status and future perspective of antimony chalcogenide (Sb2X3) photovoltaics. ACS Appl. Energy Mater. 2022, 5, 6545–6585. [Google Scholar] [CrossRef]
- Dahmardeh, Z.; Saadat, M. Exploring the potential of standalone and tandem solar cells with Sb2S3 and Sb2Se3 absorbers: A simulation study. Sci. Rep. 2023, 13, 22632. [Google Scholar] [CrossRef] [PubMed]
- Chen, T. Antimony Selenosulfide: An Emerging Solar Material with 10% Efficiency; Springer Nature: Berlin/Heidelberg, Germany, 2021; Available online: https://communities.springernature.com/posts/antimony-selenosulfide-an-emerging-solar-material-with-10-efficiency (accessed on 12 December 2025).
- Xiao, Y.; Feng, L.; Wang, H. A CTL-free homo-heterojunction antimony chalcogenide solar cell: Theoretical study. J. Phys. Chem. Solids 2024, 184, 111742. [Google Scholar] [CrossRef]
- Wang, X.; Tang, R.; Jiang, C.; Lian, W.; Ju, H.; Jiang, G.; Li, Z.; Zhu, C.; Chen, T. Manipulating the electrical properties of Sb2(S,Se)3 film for high-efficiency solar cell. Adv. Energy Mater. 2020, 10, 2002341. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, S.; Jiang, C.; Li, C.; Xiao, P.; Tang, R.; Gong, J.; Chen, G.; Chen, T.; Li, J.; et al. Regulating energy band alignment via alkaline metal fluoride assisted solution post-treatment enabling Sb2(S,Se)3 solar cells with 10.7% efficiency. Adv. Energy Mater. 2022, 12, 2103015. [Google Scholar] [CrossRef]
- Wang, X.; Tang, R.; Wu, C.; Zhu, C.; Chen, T. Development of antimony sulfide–selenide Sb2(S, Se)3-based solar cells. J. Energy Chem. 2018, 27, 713–721. [Google Scholar] [CrossRef]
- Tideswell, N.W.; Kruse, F.H.; McCullough, J.D. The crystal structure of antimony selenide, Sb2Se3. Acta Crystallogr. 1957, 10, 99–102. [Google Scholar] [CrossRef]
- Salem, M.S.; Shaker, A.; Almurayziq, T.S.; Alshammari, M.T. Prospective efficiency boosting of full-inorganic single-junction Sb2(S, Se)3 solar cell. Sol. Energy Mater. Sol. Cells 2022, 248, 112001. [Google Scholar] [CrossRef]
- Tang, R.; Chen, T.; Ding, L. Engineering microstructures for efficient Sb2(SxSe1−x)3 solar cells. J. Semicond. 2021, 42, 070203. [Google Scholar] [CrossRef]
- Li, K.; Tang, R.; Zhu, C.; Chen, T. Critical review on crystal orientation engineering of antimony chalcogenide thin film for solar cell applications. Adv. Sci. 2023, 11, e2304963. [Google Scholar] [CrossRef] [PubMed]
- Tang, R.; Chen, X.-Y.; Liang, G.-X.; Su, Z.-H.; Luo, J.-T.; Fan, P. Magnetron sputtering deposition and selenization of Sb2Se3 thin film for substrate Sb2Se3/CdS solar cells. Surf. Coat. Technol. 2019, 360, 68–72. [Google Scholar] [CrossRef]
- Liang, X.; Chen, X.; Li, Z.; Li, G.; Chen, J.; Yang, L.; Shen, K.; Xu, Y.; Mai, Y. Effect of deposition pressure on the properties of magnetron sputtering-deposited Sb2Se3 thin-film solar cells. Appl. Phys. A 2019, 125, 381. [Google Scholar] [CrossRef]
- Uslu, M.E.; Kondrotas, R.; Nedzinskas, R.; Volobujeva, O.; Timmo, K.; Kauk-Kuusik, M.; Krustok, J.; Grossberg, M. Study of the optical properties of Sb2(Se1−x, Sx)3 (x = 0–1) solid solutions. Mater. Sci. Semicond. Process. 2022, 144, 106571. [Google Scholar] [CrossRef]
- Chihi, A. Gamma-irradiated stibnite thin films set a remarkable benchmark performance for photoelectrochemical water splitting. RSC Adv. 2024, 14, 12475–12495. [Google Scholar] [CrossRef] [PubMed]
- Murali, K.; Thekkekara, L.; Rahman, M.A.; Sen, S.; Shvedov, V.; Izdebskaya, Y.; Zou, C.; Tawfik, S.A.; Shadrivov, I.; Sriram, S.; et al. Tunability of Sb2Se3 phase change material for multi-domain optoelectronics. Appl. Mater. Today 2024, 40, 102338. [Google Scholar] [CrossRef]
- Cant, D.J.H.; Gorham, J.M.; Clifford, C.A.; Shard, A.G. Standard approaches to XPS and AES quantification—A summary of ISO 18118:2024 on the use of relative sensitivity factors. Surf. Interface Anal. 2025, 57, 148–152. [Google Scholar] [CrossRef]
- Chen, W.; Gao, G.; Zhao, L.; Liu, C.; Qi, J.; Zhu, G. Solid-state selenium diffusion processing to prepare Sb2(S,Se)3 film for planar heterojunction solar cells. Sol. Energy Mater. Sol. Cells 2025, 285, 113495. [Google Scholar] [CrossRef]










| Parameter | 0mg-S | 25mg-S | 50mg-S | 75mg-S | 100mg-S |
|---|---|---|---|---|---|
| 2θ120 (°) | 16.8640 | 17.3160 | 17.5050 | 17.5390 | 17.5400 |
| d120 (Å) | 5.2528 | 5.1167 | 5.0619 | 5.0522 | 5.0519 |
| a (Å) | 11.6944 | 11.3628 | 11.2317 | 11.2342 | 11.2398 |
| 2θ020 (°) | 15.0560 | 15.4490 | 15.6140 | 15.6530 | 15.6560 |
| d020 (Å) | 5.87929 | 5.73060 | 5.6704 | 5.6564 | 5.6553 |
| b (Å) | 11.7586 | 11.4612 | 11.3408 | 11.3128 | 11.3106 |
| 2θ002 (°) | 45.6080 | 46.8109 | 47.1920 | 47.2730 | 47.3370 |
| d002 (Å) | 1.9873 | 1.9390 | 1.9243 | 1.9212 | 1.9187 |
| c (Å) | 3.9747 | 3.8781 | 3.8485 | 3.8423 | 3.8374 |
| V(Å3) | 546.5600 | 505.0500 | 490.2083 | 488.3189 | 487.8440 |
| ATi | Sample | Area (u2) |
|---|---|---|
| AT0 | 0mg S | 19,699.3 |
| AT25 | 25mg S | 13,643.0 |
| AT50 | 50mg S | 9955.5 |
| AT75 | 75mg S | 8245.8 |
| Sample | Area Percentage | Calculation of the Amount of Selenium Atoms | Proportion of Selenium Atoms |
|---|---|---|---|
| 0mg S | (19,699.3/19,699.3) × 100 = 100% | (1) × 3 | 3.000 |
| 25mg S | (13,643.0/19,699.3) × 100 = 69.26% | (0.6926) × 3 | 2.077 |
| 50mg S | (9955.5/19,699.3) × 100 = 50.54% | (0.5054) × 3 | 1.516 |
| 75mg S | (8245.8/19,699.3) × 100 = 41.86% | (0.4186) × 3 | 1.255 |
| 100mg S | XPS could not quantify | N/A | ≈0 |
| Sample |
Area (%) |
Se Atoms Without Se Loss Sb2Se3 | Recalculation of Se Atoms (Assuming 2% Loss Sb2Se3−0.06) |
|---|---|---|---|
| 0mg S | 100 | (1) × 3 = 3.000 | (1) × 2.942 = 2.942 |
| 25mg S | 69.26 | (0.6926) × 3 = 2.070 | (0.6926) × 2.942 = 2.036 |
| 50mg S | 50.54 | (0.5054) × 3 = 1.510 | (0.5054) × 2.942 = 1.486 |
| 75mg S | 41.86 | (0.4186) × 3 = 1.250 | (0.4186) × 2.942 = 1.230 |
| 100mg S | XPS could not quantify | N/A ≈ 0 | N/A ≈ 0 |
| Points | Fifth-Degree Equation (Assuming 0% Se Losses) | Fifth-Degree Equation (Assuming 2% Se Losses) | Second-Degree Equation |
|---|---|---|---|
| 1 | (3.000, 31.160°) | (3.000, 31.160°) | (3.000, 31.160°) |
| 1* | (2.940, 31.220°) | (2.926, 31.220°) | (2.942, 31.220°) |
| 2 | (2.077, 31.983°) | (2.036, 31.983°) | (2.006, 31.983°) |
| 3 | (1.516, 32.268°) | (1.486, 32.268°) | (1.428, 32.268°) |
| 4 | (1.255, 32.344°) | (1.230, 32.344°) | (1.191, 32.344°) |
| 5 | (0.930, 32.400°) | (0.930, 32.400°) | (0.930, 32.400°) |
| 6 | (0.000, 32.364°) | (0.000, 32.364°) | (0.000, 32.364°) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
López-García, M.; Chalé-Lara, F.; Rodríguez-González, E.; González-Castillo, J.R.; López-Oyama, A.B. Study and Mathematical Model of the Chemical Composition and Structure of the Compound Sb2(S1−xSex)3 Based on a Correlation of Data Obtained Through XRD and XPS Characterization. Materials 2026, 19, 1072. https://doi.org/10.3390/ma19061072
López-García M, Chalé-Lara F, Rodríguez-González E, González-Castillo JR, López-Oyama AB. Study and Mathematical Model of the Chemical Composition and Structure of the Compound Sb2(S1−xSex)3 Based on a Correlation of Data Obtained Through XRD and XPS Characterization. Materials. 2026; 19(6):1072. https://doi.org/10.3390/ma19061072
Chicago/Turabian StyleLópez-García, Martín, Fabio Chalé-Lara, Eugenio Rodríguez-González, Jesús Roberto González-Castillo, and Ana B. López-Oyama. 2026. "Study and Mathematical Model of the Chemical Composition and Structure of the Compound Sb2(S1−xSex)3 Based on a Correlation of Data Obtained Through XRD and XPS Characterization" Materials 19, no. 6: 1072. https://doi.org/10.3390/ma19061072
APA StyleLópez-García, M., Chalé-Lara, F., Rodríguez-González, E., González-Castillo, J. R., & López-Oyama, A. B. (2026). Study and Mathematical Model of the Chemical Composition and Structure of the Compound Sb2(S1−xSex)3 Based on a Correlation of Data Obtained Through XRD and XPS Characterization. Materials, 19(6), 1072. https://doi.org/10.3390/ma19061072





