Structure–Property Relationships and Thermal Degradation Mechanism of Terpene Methacrylate-Styrene Copolymers
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. The Ultraviolet (UV) Polymerization
2.3. Methods
2.3.1. The Fourier Transform Infrared Spectroscopy (FTIR)
2.3.2. The Solubility
2.3.3. The Chemical Stability
2.3.4. The Differential Scanning Calorimetry (DSC)
2.3.5. The Thermogravimetric/Derivative Thermogravimetric/Differential Scanning Calorimetry Analysis (TG/DTG/DSC)
2.3.6. The Simultaneous Thermogravimetric Analysis Coupled with the Fourier Transform Infrared Spectroscopy (TG-FTIR)
3. Results and Discussion
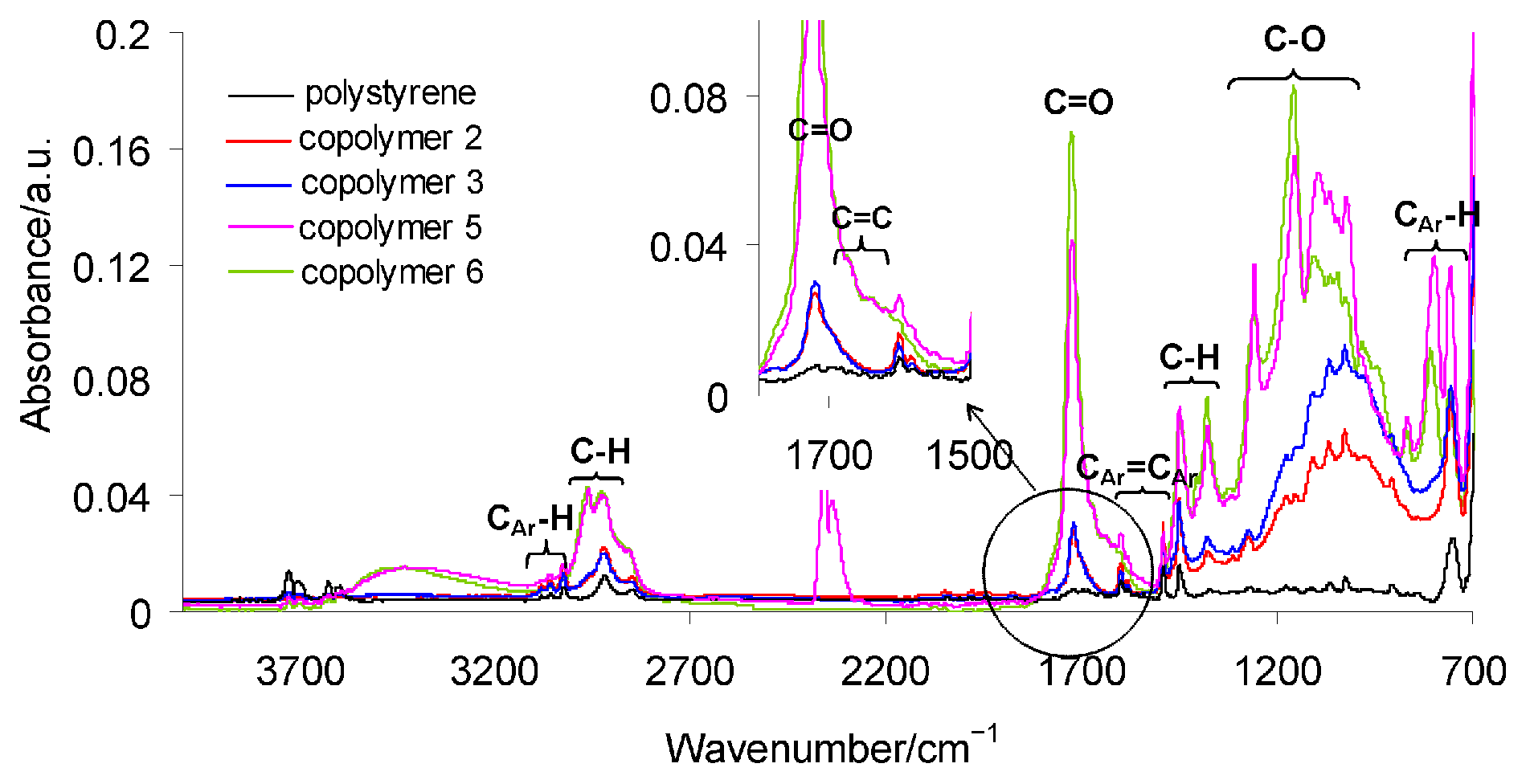
3.1. The Fourier Transform Infrared Spectroscopy (FTIR) Results
3.2. The Solubility of the Tested Polymers Results
3.3. The Chemical Stability Results
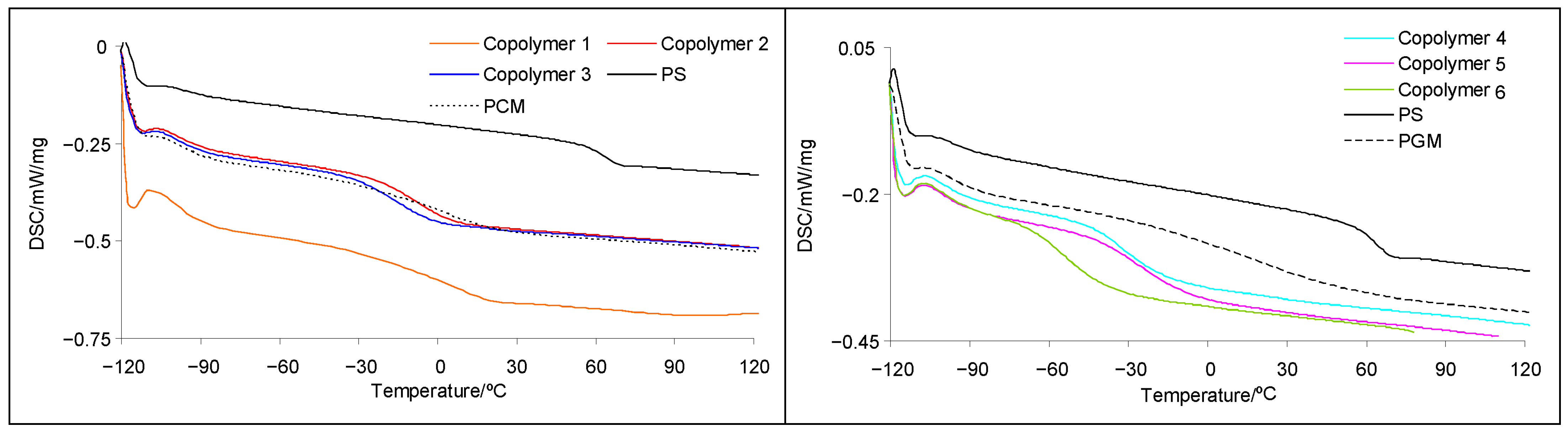
3.4. The Differential Scanning Calorimetry (DSC) Results
3.5. The Thermogravimetric/Derivative Thermogravimetric/Differential Scanning Calorimetry (TG/DTG/DSC) Results (Inert Conditions)
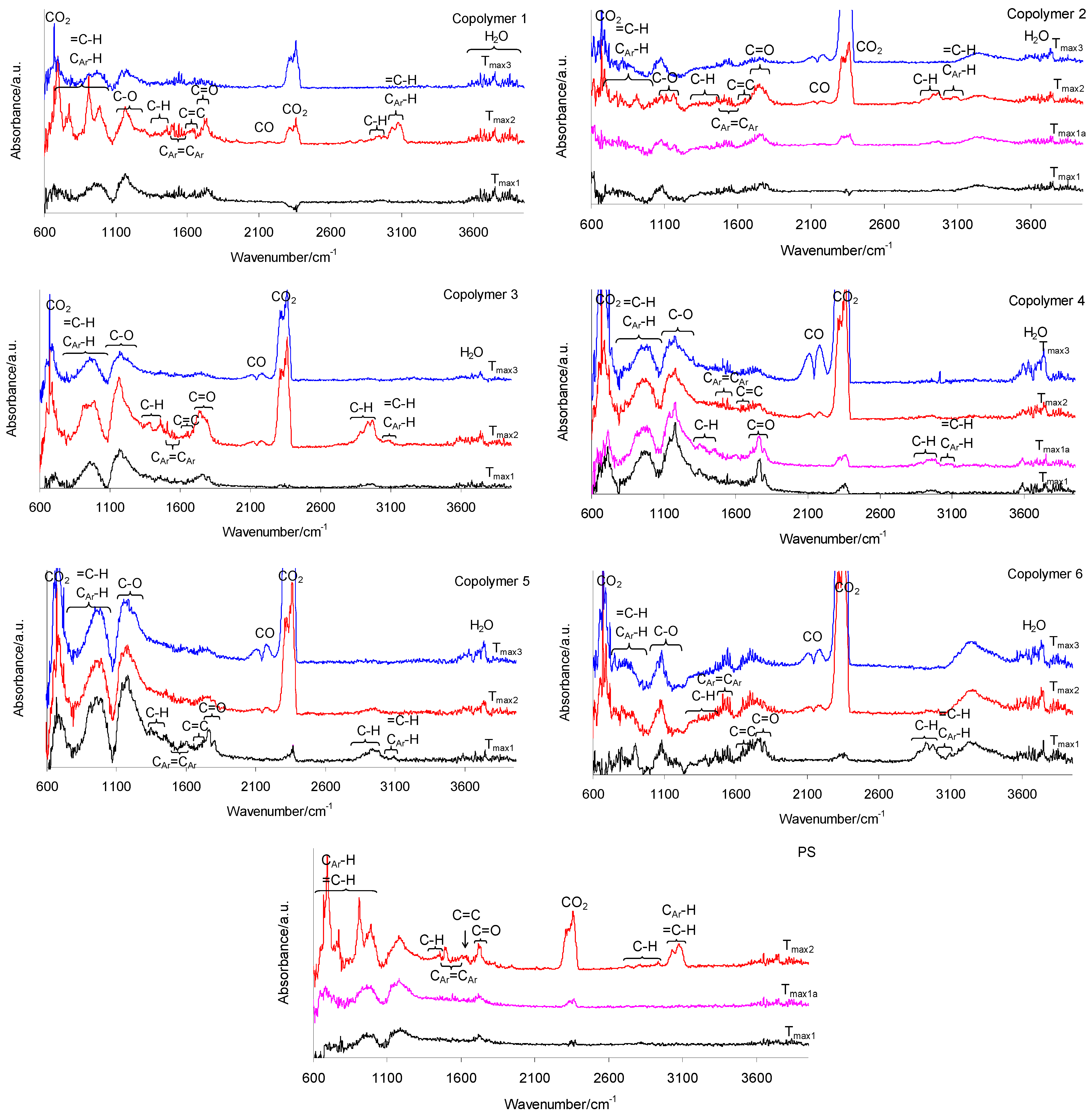
3.6. The Simultaneous Thermogravimetric Analysis Coupled with the Fourier Transform Infrared Spectroscopy (TG/FTIR) Results (Inert Conditions)
3.7. The Thermogravimetric/Derivative Thermogravimetric/Differential Scanning Calorimetry (TG/DTG/DSC) Results (Oxidative Conditions)
3.8. The Simultaneous Thermogravimetric Analysis Coupled with the Fourier Transform Infrared Spectroscopy (TG/FTIR) Results (Oxidative Conditions)
4. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Singh, A.; Chauhan, A.; Gaur, R. A comprehensive review on the synthesis, properties, environmental impacts, and chemiluminescence applications of polystyrene (PS). Discov. Chem. 2025, 2, 47. [Google Scholar] [CrossRef]
- Kreutz, J.C.; de Souza, P.R.; Benetii, V.P.; dos Reis Freitas, A.; Bittencourt, P.R.S.; Gaffo, L. Mechanical and thermal properties of polystyrene and medium density fiberboard composites. Polimeros 2021, 31, e2021013. [Google Scholar] [CrossRef]
- Carswell, T.S.; Hayes, R.F.; Nason, H.K. Physical properties of polystyrene as influenced by temperature. Ind. Eng. Chem. 1942, 34, 454–457. [Google Scholar] [CrossRef]
- Dan, O.; Trofirmova, L. A study of the properties of forming mixtures containing polystyrene wastes. J. Cast. Mater. Eng. 2021, 5, 14–19. [Google Scholar] [CrossRef]
- Hylova, L.; Mizera, A.; Mizera, M.; Grund, R.; Ovsik, M. Mechanical properties study of high impact polystyrene under impact and static tests. IOP Conf. Ser. Mater. Sci. Eng. 2018, 448, 012044. [Google Scholar] [CrossRef]
- Csernica, J.; Brown, A. Effect of plasticizers on the properties of polystyrene films. J. Chem. Educ. 1999, 76, 1526. [Google Scholar] [CrossRef]
- Church, J.M.; Berenson, C. Properties of styrene-polyester copolymers. Ind. Eng. Chem. 1955, 47, 2456–2462. [Google Scholar] [CrossRef]
- Butzelaar, A.J.; Röring, P.; Mach, T.P.; Hoffmann, M.; Jeschull, F.; Wilhelm, M.; Winter, M.; Brunklaus, G.; Théato, P. Styrene-based poly(ethylene oxide) side-chain block copolymers as solid polymer electrolytes for high-voltage lithium-metal batteries. ACS Appl. Mater. Interfaces 2021, 13, 39257–39270. [Google Scholar] [CrossRef] [PubMed]
- Zaharescu, T.; Banciu, C. Packaging materials based on styrene-isoprene-styrene rriblock copolymer modified with graphene. Polymers 2023, 15, 353. [Google Scholar] [CrossRef] [PubMed]
- Haile, L. Study of recycling of waste high impact polystyrene (PS-HI) and polystyrene with flame retardant additives (PS-FR) co-polymers. J. Text. Sci. Fash. Technol. 2021, 7, 1–11. [Google Scholar] [CrossRef]
- Srivastava, H.; Lade, H.; Paul, D.; Arthanareeswaran, G.; Kweon, J.H. Styrene-based copolymer for polymer membrane modifications. Appl. Sci. 2016, 6, 159. [Google Scholar] [CrossRef]
- Li, F.; Larock, R.C. New soybean oil–styrene–divinylbenzene thermosetting copolymers. II. Dynamic mechanical properties. J. Polym. Sci. Part B Polym. Phys. 2000, 38, 2721–2738. [Google Scholar] [CrossRef]
- Xavier, T.; Samuel, J.; Kurian, T. Thermoplastic ionomers based on “styrene grafted natural rubber”. Int. J. Polym. Mater. Polym. Biomater. 2003, 52, 251–264. [Google Scholar] [CrossRef]
- Galdi, N.; Buonerba, A.; Oliwa, L. Olefin–styrene copolymers. Polymers 2016, 8, 405. [Google Scholar] [CrossRef]
- Schué, E.; Rickertsen, D.R.L.; Korpusik, A.B.; Adili, A.; Seidel, D.; Sumerlin, B.S. Alternating styrene–propylene and styrene–ethylene copolymers prepared by photocatalytic decarboxylation. Chem. Sci. 2023, 14, 11228–11236. [Google Scholar] [CrossRef] [PubMed]
- Bumbac, M.; Zaharescu, T.; Nicolescu, C.M.; Borbath, T.; Borbath, I. Chemiluminescence-based evaluation of styrene block copolymers’ recyclability. Sci. Rep. 2024, 14, 28221. [Google Scholar] [CrossRef]
- Francisco-Vieira, L.; Benavides, R.; Cuara-Diaz, E.; Morales-Acosta, D. Styrene-co-butyl acrylate copolymers with potential application as membranes in PEM fuel cell. Int. J. Hydrogen Energy 2019, 44, 12492–12499. [Google Scholar] [CrossRef]
- Zhan, P.; Chen, J.; Zheng, A.; Shi, H.; Wu, T.; Wei, D.; Xu, X.; Guan, Y. Methyl methacrylate-styrene copolymers prepared by utilizing the Trommsdorff effect in an inverted two-stage extruder. Mater. Res. Express 2020, 7, 095305. [Google Scholar] [CrossRef]
- Rahlwes, D.; Roovers, J.E.L.; Bywater, S. Synthesis and characterization of poly(styrene-g-isoprene) copolymers. Macromolecules 1977, 10, 604–609. [Google Scholar] [CrossRef]
- Tsarevsky, N.V.; Sarbu, T.; Göbelt, B.; Matyjaszewski, K. Synthesis of styrene-acrylonitrile copolymers and related block copolymers by atom transfer radical polymerization. Macromolecules 2002, 35, 6142–6148. [Google Scholar] [CrossRef]
- Radha, K.; Shobana, K.; Mohan, D.; Narayana, Y.L. Synthesis and characterization of styrene-acrylonitrile copolymer blend ultrafiltration membranes. Desalination Water Treat. 2009, 12, 114–126. [Google Scholar] [CrossRef]
- Ohishi, H.; Kishimoto, S.; Ikehara, T.; Nishi, T. Synthesis of styrene–acrylonitrile random copolymers (SAN) and polyarylate block copolymers and the control of their mechanical properties by morphology generation. J. Polym. Sci. Part B Polym. Phys. 2000, 38, 127–137. [Google Scholar] [CrossRef]
- Hill, D.J.T.; O’DOnnell, J.H.; O’SUllivan, P.W. Analysis of the mechanism of copolymerization of styrene and acrylonitrile. Macromolecules 1982, 15, 960–966. [Google Scholar] [CrossRef]
- Ko, E.B.; Lee, D.E.; Yoon, K.B. Electrically conductive nanocomposites composed of styrene-acrylonitrile copolymer and rGO via free-radical polymerization. Polymers 2020, 12, 1221. [Google Scholar] [CrossRef]
- Wu, M.M. Acrylonitrile polymers, survey and styrene-scrylonitrile (SAN). In Kirk-Othmer Encyclopedia of Chemical Technology; Wiley: Hoboken, NJ, USA, 2001. [Google Scholar]
- Tian, M.; You, J.; Qiu, J.; Li, M.; Xing, H.; Xue, J.; Jiang, Z.; Tang, T. Unexpected super anti-compressive styrene-acrylonitrile co-polymer/polyurea nanocomposite foam with excellent solvent resistance, re-processability and shape memory performance. Compos. Part B Eng. 2023, 264, 110908. [Google Scholar] [CrossRef]
- López-García, J.; Primc, G.; Junkar, I.; Lehocký, M.; Mozetič, M. On the hydrophilicity and water resistance effect of styrene-acrylonitrile copolymer treated by CF4 and O2 plasmas. Plasma Process. Polym. 2015, 12, 1075–1084. [Google Scholar] [CrossRef]
- Migahed, M.D.; El-Khodary, A.; Hammam, M.; Shaban, A.; Hafiz, H.R. Compensation effect in styrene-acrylonitrile copolymer and acrylonitrile-butadiene-styrene terpolymer as explored by thermally stimulated depolarization measurements. J. Mater. Sci. 1990, 25, 2795–2799. [Google Scholar] [CrossRef]
- McKeen, L.W. Chapter 5—Styrenic Plastics, Film Properties of Plastics and Elastomers (Third Edition); Plastics Design Library: New York, NY, USA, 2012; pp. 81–89. [Google Scholar]
- Demirors, M. Styrene-Butadiene Copolymers, Encyclopedia of Polymer Science and Technology; Wiley Online Library: Hoboken, NJ, USA, 2001. [Google Scholar]
- Velásquez, E.; Leal, M.; García, L.; Oliva, H. Influence of styrene-b-butadiene copolymer types on phase morphology and polymer partitioning between demixed-macrophases from HIPS-mimicking unstable blends. J. Polym. Res. 2021, 28, 443. [Google Scholar] [CrossRef]
- McClements, J.; Buffone, C.; Shaver, M.P.; Sefiane, K.; Koutsos, V. Poly(styrene-co-butadiene) random copolymer thin films and nanostructures on a mica surface: Morphology and contact angles of nanodroplets. Soft Matter 2017, 13, 6152–6166. [Google Scholar] [CrossRef] [PubMed]
- Canto, L.; Mantovani, G.; Deazevedo, E.; Bonagamba, T.; Hage, E.; Pessan, L. Molecular characterization of styrene-butadiene-styrene block copolymers (SBS) by GPC, NMR, and FTIR. Polym. Bull. 2006, 57, 513–524. [Google Scholar] [CrossRef]
- Liu, J.; Min, X.; Zhang, X.; Zhu, X.; Wang, Z.; Wang, T.; Fan, X. A novel synthetic strategy for styrene–butadiene–styrene tri-block copolymer with high cis-1,4 units via changing catalytic active centres. R. Soc. Open Sci. 2019, 6, 190536. [Google Scholar] [CrossRef]
- Chen, X.; Ma, Z.; Zhou, J.; Wang, J.; Zhang, X.; Zhao, R.; Tong, J. Thermal degradation characteristics of styrene-butadiene-styrene copolymer asphalt binder filled with an inorganic flame-retarding agent. Polymers 2022, 14, 3761. [Google Scholar] [CrossRef] [PubMed]
- Lu, J.; Liang, H.; Li, A.; Cheng, Q. Synthesis of block and graft copolymers of β-pinene and styrene by transformation of living cationic polymerization to atom transfer radical polymerization. Eur. Polym. J. 2004, 40, 397–402. [Google Scholar] [CrossRef]
- Lu, J.; Kamigaito, M.; Sawamoto, M.; Higashimura, T.; Deng, Y.-X. Living cationic isomerization polymerization of β-pinene. 2. Synthesis of block and random copolymers with styrene or p-methylstyrene. Macromolecules 1997, 30, 27–31. [Google Scholar] [CrossRef]
- Hahn, C.; Göttker-Schnetmann, I.; Tzourtzouklis, I.; Wagner, M.; Müller, A.H.E.; Floudas, G.; Mecking, S.; Frey, H. Nopadiene: A pinene-derived cyclic diene as a styrene substitute for fully biobased thermoplastic elastomers. J. Am. Chem. Soc. 2023, 145, 26688–26698. [Google Scholar] [CrossRef] [PubMed]
- Garrido Barros, M. Potentially biodegradable polymers based on α- or β-pinene and sugar derivatives or styrene, obtained under normal conditions and on microwave irradiation. Eur. J. Org. Chem. 2007, 8, 1357–1363. [Google Scholar] [CrossRef]
- Afzal, A.; Kausar, A.; Siddiq, M. Review highlighting physical prospects of styrenic polymer and styrenic block copolymer reinforced with carbon nanotube. Polym. Technol. Eng. 2016, 56, 573–593. [Google Scholar] [CrossRef]
- Sharma, S.; Srivastava, A. Synthesis and characterization of copolymers of limonene with styrene initiated by azobisisobutyronitrile. Eur. Polym. J. 2004, 40, 2235–2240. [Google Scholar] [CrossRef]
- Derdar, H.; Mitchell, G.R.; Chaibedraa, S.; Mahendra, V.S.; Cherifi, Z.; Bachari, K.; Chebout, R.; Touahra, F.; Meghabar, R.; Belbachir, M. Synthesis and characterization of copolymers and nanocomposites from limonene, styrene and organomodified-clay using ultrasonic assisted method. Polymers 2022, 14, 2820. [Google Scholar] [CrossRef]
- Marzocchi, R.; Grimaldi, I.; Ruiz de Ballesteros, O.; Femina, G.; Guida, A.; Ricciardi, R.; Morvillo, P.; Capacchione, C.; Auriemma, F. Synthesis and characterization of β-myrcene-styrene and β-ocimene-styrene copolymers. Macromol. Rapid Commun. 2025, 46, 2400641. [Google Scholar] [CrossRef]
- Hulnik, M.I.; Vasilenko, I.V.; Radchenko, A.V.; Peruch, F.; Ganachaud, F.; Kostjuk, S.V. Aqueous cationic homo- and co-polymerizations of β-myrcene and styrene: A green route toward terpene-based rubbery polymers. Polym. Chem. 2018, 9, 5690–5700. [Google Scholar] [CrossRef]
- Fu, Y.; Dai, Q.; Tan, H.; Han, X.; Zhang, B.; Xiong, Q.; Yi, W.; Yang, Z.; Li, L.; Liu, K. The influence of myrcene on anionic copoly-merization of 1,3-pentadiene and styrene. Eur. Polym. J. 2024, 221, 113507. [Google Scholar] [CrossRef]
- Wahlen, C.; Blankenburg, J.; von Tiedemann, P.; Ewald, J.; Sajkiewicz, P.; Müller, A.H.E.; Floudas, G.; Frey, H. Tapered multiblock copolymers based on farnesene and styrene: Impact of biobased polydiene architectures on material properties. Macromolecules 2020, 53, 10397–10408. [Google Scholar] [CrossRef]
- Gutarowska, B.; Kotynia, R.; Bieliński, D.; Anyszka, R.; Wręczycki, J.; Piotrowska, M.; Koziróg, A.; Berłowska, J.; Dziugan, P. New sulfur organic polymer-concrete composites containing waste materials: Mechanical characteristics and resistance to biocorrosion. Materials 2019, 12, 2602. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Zhang, M.; He, J.; Ni, P. Synthesis and characterization of graft copolymers with poly(ε-caprolactone) side chain using hydroxylated poly(β-myrcene-co-α-methyl styrene). Molecules 2024, 29, 2363. [Google Scholar] [CrossRef]
- Shukla, A.; Srivastava, A.K. Terpolymerization of linalol, styrene, and methyl methacrylate: Synthesis, characterization, and kinetic study. Polym. Technol. Eng. 2002, 41, 777–793. [Google Scholar] [CrossRef]
- Shukla, A.; Srivastava, A.K. Determination of reactivity ratios and kinetics of free radical copolymerization of linalool with styrene. Polym. Adv. Technol. 2004, 15, 445–452. [Google Scholar] [CrossRef]
- Singh, A.; Kamal, M.; Singh, D.K. Interpenetrating polymer network of poly(linalool-co-MMA) and styrene initiated by selenonium ylide. J. Macromol. Sci. Part A 2015, 52, 193–201. [Google Scholar] [CrossRef]
- Tong, J.D.; Ni, S.; Winnik, M.A. Synthesis of polyisoprene-b-polystyrene block copolymers bearing a fluorescent dye at the junction by the combination of living anionic polymerization and atom transfer radical polymerization. Macromolecules 2000, 33, 1482–1486. [Google Scholar] [CrossRef]
- Srivastava, A.; Pandey, P. Synthesis and characterisation of copolymers containing geraniol and styrene initiated by benzoyl peroxide. Eur. Polym. J. 2002, 38, 1709–1712. [Google Scholar] [CrossRef]
- Min, E.H.; Wong, K.H.; Setijadi, E.; Ladouceur, F.; Straton, M.; Argyros, A. Menthol-based chiral copolymers for polymer optical fibres (POF). Polym. Chem. 2011, 2, 2045–2051. [Google Scholar] [CrossRef]
- Sahu, P.; Bhowmick, A.K.; Kali, G. Terpene based elastomers: Synthesis, properties, and applications. Processes 2020, 8, 553. [Google Scholar] [CrossRef]
- Worzakowska, M. UV polymerization of methacrylates-preparation and properties of novel copolymers. Polymers 2021, 13, 1659. [Google Scholar] [CrossRef] [PubMed]
- Makowskaya, K.; Jargieło, P.; Worzakowska, M.; Rogulska, M. Thermal properties and the decomposition path of novel UV polymers of terpene-based monomer: Citronellyl methacrylate. J. Therm. Anal. Calorim. 2023, 148, 13349–13362. [Google Scholar] [CrossRef]
- Worzakowska, M. TG/DSC/FTIR/QMS analysis of environmentally friendly poly(citronellyl methacrylate)-co-poly(benzyl methacrylate) copolymers. J. Mater. Sci. 2023, 58, 2005–2024. [Google Scholar] [CrossRef]
- Podgórski, M. Structure–property relationship in new photo-cured dimethacrylate-based dental resins. Dent. Mater. 2012, 28, 398–409. [Google Scholar] [CrossRef] [PubMed]
- Kaith, B.S.; Singha, A.S.; Grupa, S.K. Graft copolymerization of flax fibres with binary vinyl monomer mixtures and evaluation of swelling, moisture absorbance and thermal behavior of the grafted fibres. J. Polym. Mater. 2003, 20, 195–199. [Google Scholar]
- NIST Chemistry Webbook. NIST Standard Reference Data. 2011. Available online: http://webbook.nist.gov (accessed on 1 February 2026).
- Setiano, S.; Panatarani, C.; Singh, D.; Joni, I.M. Semi-empirical infrared spectra simulation of pyrene-like molecules insight for simple analysis of functionalization grapheme quantum dots. Sci. Rep. 2023, 13, 2282. [Google Scholar] [CrossRef]
- Kot, M.; Wojcieszak, R.; Janiszewska, E.; Pietrowski, M.; Zieliński, M. Effect of modification of amorphous silica with ammonium agets on the physicochemical properties and hydrogenation activity of ir/SiO2 catalysts. Materials 2021, 14, 968. [Google Scholar] [CrossRef]
- Feng, Y.; Wang, W.; Wang, Y.; Sun, J.; Zhang, C.; Mao, Y.; Xiqiang, Z.; Song, Z. Experimental study on in-situ decomposition of VOCs using microwave-induced metal discharge. Waste Biomass Valorization 2019, 10, 3921–3929. [Google Scholar] [CrossRef]
- Ece, M.S.; Kutluay, S. Comparative and competitive adsorption of gaseous toluene, ethylbenzene, and xylene onto natural cellulose-modified Fe3O4 nanoparticles. J. Environ. Chem. Eng. 2022, 10, 107389. [Google Scholar] [CrossRef]
- Gonzalez-Aguilar, A.M.; Pérez-García, V.; Riesco-Ávila, J.M. A thermo-catalytic pyrolysis of polystyrene waste review: A systematic, statistical, and bibliometric approach. Polymers 2023, 15, 1582. [Google Scholar] [CrossRef]
- Balema, V.P.; Hlova, I.Z.; Carnahan, S.L.; Seyedi, M.; Dolotko, O.; Rossini, A.J.; Luzinov, I. Depolymerization of polystyrene under ambient conditions. New J. Chem. 2021, 45, 2935–2938. [Google Scholar] [CrossRef]
- Kumar, V.; Khan, A.; Rabnawaz, M. Efficient depolymerization of polystyrene with table salt and oxidized copper. ACS Sustain. Chem. Eng. 2022, 10, 6493–6502. [Google Scholar] [CrossRef]
- Yoshida, E. Vacuum pyrolysis depolymerization of waste polystyrene foam into high-purity styrene using a spirit lamp flame for convenient chemical recycling. RSC Sustain. 2023, 1, 2058–2065. [Google Scholar] [CrossRef]
- Reed, M.R.; Belden, E.R.; Kazantzis, N.K.; Timko, M.T.; Castro-Dominguez, B. Thermodynamic and economic analysis of a deployable and scalable process to recover Monomer-Grade styrene from waste polystyrene. Chem. Eng. J. 2024, 492, 152079. [Google Scholar] [CrossRef]
- Ngohang, F.; Fontaine, G.; Gay, L.; Bourbigot, S. Revisited investigation of fire behavior of ethylene vinyl acetate/aluminum trihydroxide using a combination of mass loss cone, Fourier transform infrared spectroscopy and electrical low pressure impactor. Polym. Degrad. Stab. 2014, 106, 26–35. [Google Scholar] [CrossRef]
- Celina, M.; Ottesen, D.; Gillen, K.; Clough, R. FTIR emission spectroscopy applied to polymer degradation. Polym. Degrad. Stab. 1997, 58, 15–31. [Google Scholar] [CrossRef]
- Xu, L.; Zheng, J.; Che, L.; Wu, X.; Chen, P.; Zhang, L.; Hu, Q.; Huang, G. Synthesis and thermal degradation property study of N-vinylpyrrolidone and acrylamide copolymer. RSC Adv. 2014, 4, 33269–33278. [Google Scholar] [CrossRef]





| Polymer Name | Citronellyl Methacrylate (CM) [g] | Geranyl Methacrylate (GM) [g] | Styrene (S) [g] | Mass Ratio of Monomers (CM or GM to ST) [%] |
|---|---|---|---|---|
| PCM | 3.0 | - | - | 100:0 |
| PS | - | - | 3.0 | 0:100 |
| PGM | - | 3.0 | - | 100:0 |
| Copolymer 1 | 0.6 | - | 2.4 | 20:80 |
| Copolymer 2 | 1.5 | - | 1.5 | 50:50 |
| Copolymer 3 | 2.4 | - | 0.6 | 80:20 |
| Copolymer 4 | - | 0.6 | 2.4 | 20:80 |
| Copolymer 5 | - | 1.5 | 1.5 | 50:50 |
| Copolymer 6 | - | 2.4 | 0.6 | 80:20 |
| α | ||||||||
|---|---|---|---|---|---|---|---|---|
| PCM | PS | PGM | Copolymer 1 | Copolymer 2 | Copolymer 3 | Copolymer 4 | Copolymer 5 | Copolymer 6 |
| 0.90 | 0.93 | 0.88 | 0.92 | 0.94 | 0.93 | 0.85 | 0.85 | 0.86 |
| Polymer Name | ΔwS [%] | ||||||
|---|---|---|---|---|---|---|---|
| Water | Methanol | Butanol | Acetone | Acetic Acid | Ethyl Acetate | Tetrahydrofuran | |
| PCM | 0.4 | 0.3 | 0.3 | 0.5 | 0.9 | 0.7 | 0.6 |
| PS | 0 | 0 | 0 | s | 0 | s | s |
| PGM | 0.6 | 0.7 | 0.9 | 0.4 | 0.8 | 0.8 | 0.7 |
| Copolymer 1 | 0.8 | 0.8 | 0.7 | 0.5 | 0.6 | 0.7 | 0.3 |
| Copolymer 2 | 0.7 | 0.8 | 0.7 | 0.5 | 0.5 | 0.7 | 0.4 |
| Copolymer 3 | 0.7 | 0.7 | 0.8 | 0.6 | 0.4 | 0.7 | 0.5 |
| Copolymer 4 | 0.9 | 0.9 | 0.8 | 0.6 | 0.5 | 0.6 | 0.4 |
| Copolymer 5 | 0.8 | 0.9 | 0.7 | 0.5 | 0.5 | 0.7 | 0.7 |
| Copolymer 6 | 0.7 | 0.8 | 0.7 | 0.4 | 0.4 | 0.6 | 0.5 |
| Polymer Name | ΔwS [%] | ||||||
|---|---|---|---|---|---|---|---|
| Diethyl Ether | Dioxane | Chloroform | CCl4 | Hexane | Cyclohexane | Toluene | |
| PCM | 0.3 | 0.9 | 0.6 | 0.6 | 0.8 | 0.6 | 0.7 |
| PS | 0 | s | s | s | swell | s | s |
| PGM | 0.8 | 0.8 | 0.9 | 0.8 | 0.9 | 0.7 | 1.0 |
| Copolymer 1 | 0.6 | 0.6 | 0.5 | 0.5 | 0.5 | 0.7 | 0.8 |
| Copolymer 2 | 0.6 | 0.5 | 0.5 | 0.4 | 0.6 | 0.6 | 0.9 |
| Copolymer 3 | 0.6 | 0.6 | 0.6 | 0.6 | 0.5 | 0.5 | 0.7 |
| Copolymer 4 | 0.8 | 0.7 | 0.7 | 0.5 | 0.5 | 0.6 | 0.8 |
| Copolymer 5 | 0.8 | 0.6 | 0.7 | 0.6 | 0.4 | 0.5 | 0.8 |
| Copolymer 6 | 0.6 | 0.7 | 0.5 | 0.6 | 0.5 | 0.5 | 0.7 |
| Polymer Name | ΔwS [%] | ||||
|---|---|---|---|---|---|
| 1 M NaOH | 1 M HCl | Buffer (pH 4) | Buffer (pH 7) | Buffer (pH 10) | |
| PCM | 0.4 | 0.0 | 0.0 | 0.1 | 0.1 |
| PS | 0 | −2.0 | −1.0 | 0 | 0 |
| PGM | 0.5 | 0.5 | 0.4 | 0.4 | 0.5 |
| Copolymer 1 | 0.3 | 0.0 | 0.0 | 0.2 | 0.1 |
| Copolymer 2 | 0.2 | 0.1 | 0.1 | 0.3 | 0.2 |
| Copolymer 3 | 0.3 | 0.2 | 0.0 | 0.4 | 0.4 |
| Copolymer 4 | 0.4 | 0.4 | 0.4 | 0.5 | 0.5 |
| Copolymer 5 | 0.4 | 0.3 | 0.4 | 0.4 | 0.5 |
| Copolymer 6 | 0.3 | 0.4 | 0.5 | 0.5 | 0.3 |
| Tg [°C] | ||||||||
|---|---|---|---|---|---|---|---|---|
| PCM | PS | PGM | Copolymer 1 | Copolymer 2 | Copolymer 3 | Copolymer 4 | Copolymer 5 | Copolymer 6 |
| 3.1 | 61.9 | 24.1 | 0.4 | −7.7 | −15.2 | −29.0 | −23.2 | −50.5 |
| Polymer Name | T5% [°C] | Tmax1/Tmax1a [°C] | Δm1 [%] | Tmax2 [°C] | Δm2 [%] | mr [%] |
|---|---|---|---|---|---|---|
| PCM | 251 | 396 | 99.0 | 577 | 1.0 | 0 |
| PS | 200 | 222 | 11.6 | 413 | 88.4 | 0 |
| PGM | 189 | 235 | 54.0 | 434 | 46.0 | 0 |
| Copolymer 1 | 187 | 205 | 17.4 | 412 | 82.6 | 0 |
| Copolymer 2 | 148 | 257 | 38.0 | 407 | 62.0 | 0 |
| Copolymer 3 | 172 | 277 | 34.9 | 403 | 65.1 | 0 |
| Copolymer 4 | 159 | 189/254 | 54.1 | 431 | 35.8 | 10.1 |
| Copolymer 5 | 142 | 180/238 | 56.9 | 424 | 40.9 | 2.2 |
| Copolymer 6 | 144 | 212/276 | 59.4 | 433 | 36.9 | 3.7 |
| Polymer Name | T5% [°C] | Tmax1/Tmax1a [°C] | Δm1 [%] | Tmax2 [°C] | Δm2 [%] | Tmax3 [°C] | Δm3 [%] |
|---|---|---|---|---|---|---|---|
| PCM | 241 | - | - | 292/392 | 93.7 | 505 | - |
| PS | 234 | 167/225 | 7.9 | 391 | 92.1 | - | 0 |
| PGM | 211 | 239 | 44.2 | 427 | 41.4 | 520 | - |
| Copolymer 1 | 181 | 195 | 17.9 | 393 | 78.1 | 464 | 4.0 |
| Copolymer 2 | 163 | 185/246 | 53.0 | 398 | 39.1 | 482 | 7.9 |
| Copolymer 3 | 172 | 273 | 39.2 | 401 | 52.6 | 484 | 8.2 |
| Copolymer 4 | 147 | 182/249 | 75.4 | 416 | 14.2 | 501 | 10.4 |
| Copolymer 5 | 149 | 234 | 57.0 | 414 | 29.4 | 494 | 13.6 |
| Copolymer 6 | 119 | 227 | 65.9 | 419 | 25.0 | 488 | 9.1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Worzakowska, M. Structure–Property Relationships and Thermal Degradation Mechanism of Terpene Methacrylate-Styrene Copolymers. Materials 2026, 19, 974. https://doi.org/10.3390/ma19050974
Worzakowska M. Structure–Property Relationships and Thermal Degradation Mechanism of Terpene Methacrylate-Styrene Copolymers. Materials. 2026; 19(5):974. https://doi.org/10.3390/ma19050974
Chicago/Turabian StyleWorzakowska, Marta. 2026. "Structure–Property Relationships and Thermal Degradation Mechanism of Terpene Methacrylate-Styrene Copolymers" Materials 19, no. 5: 974. https://doi.org/10.3390/ma19050974
APA StyleWorzakowska, M. (2026). Structure–Property Relationships and Thermal Degradation Mechanism of Terpene Methacrylate-Styrene Copolymers. Materials, 19(5), 974. https://doi.org/10.3390/ma19050974






