A Green P–N–Al Synergistic System for Eco-Friendly Flame-Retardant Polystyrene
Highlights
- Developed a green flame-retardant strategy using bio-based PA and halogen-free components for PS
- Achieved 28.5% LOI, UL-94 V-0, 73.8% pHRR reduction, and 78.4% tensile strength retention
- Revealed cooperative condensed- and gas-phase flame-retardant mechanisms via in situ P-N-Al network
- Offers an eco-friendly strategy for high-performance flame-retardant PS materials
- Provides a new approach to overcome the flame-retardancy/mechanics trade-off in polymers
- Demonstrates a design strategy for multifunctional composites with balanced safety and performance
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of PA-TETA Flame Retardant
2.3. Preparation of ADP Solution
2.4. Preparation of PS/ADP/PA-TETA Powder
2.5. Preparation of Flame-Retardant PS/ADP/PA-TETA Composites
2.6. Characterization
3. Results
3.1. Chemical Structure and Microstructure Characterization
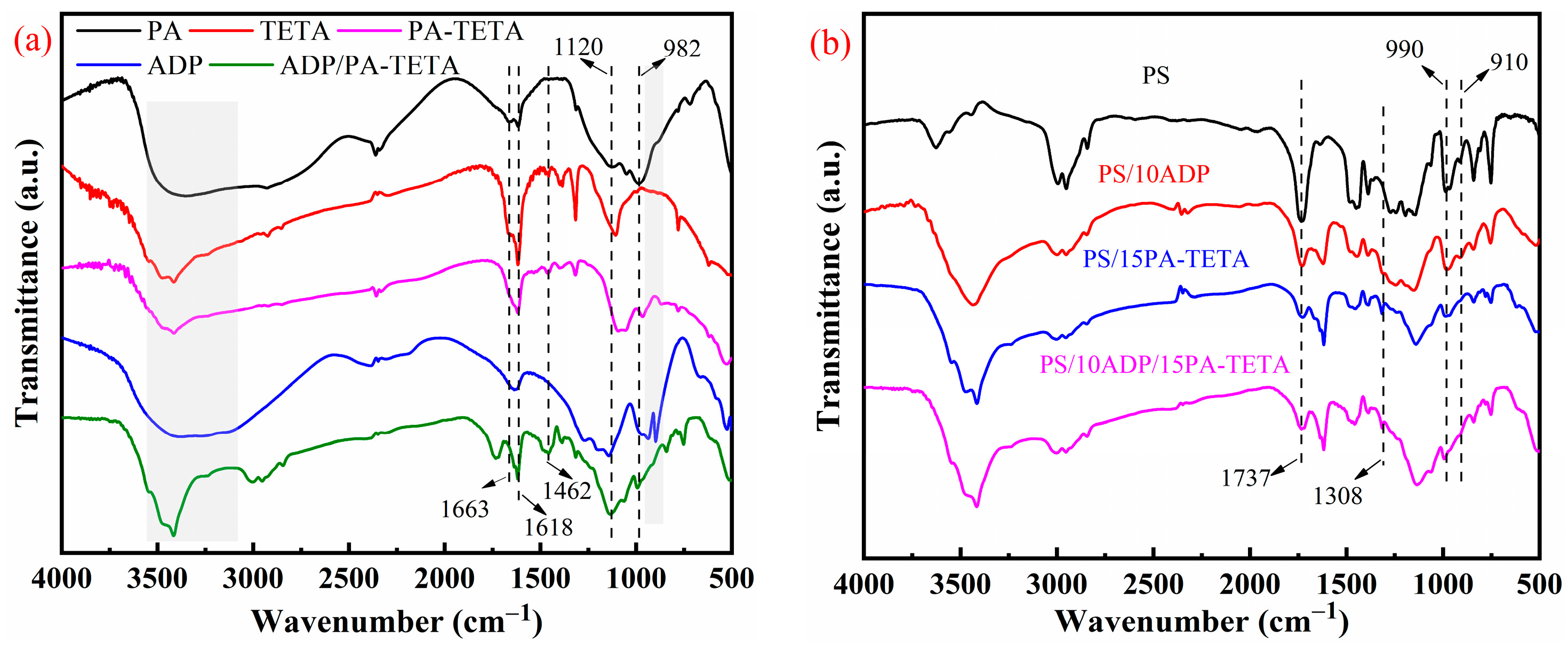
3.1.1. FTIR Analysis of Flame-Retardant PS Composites
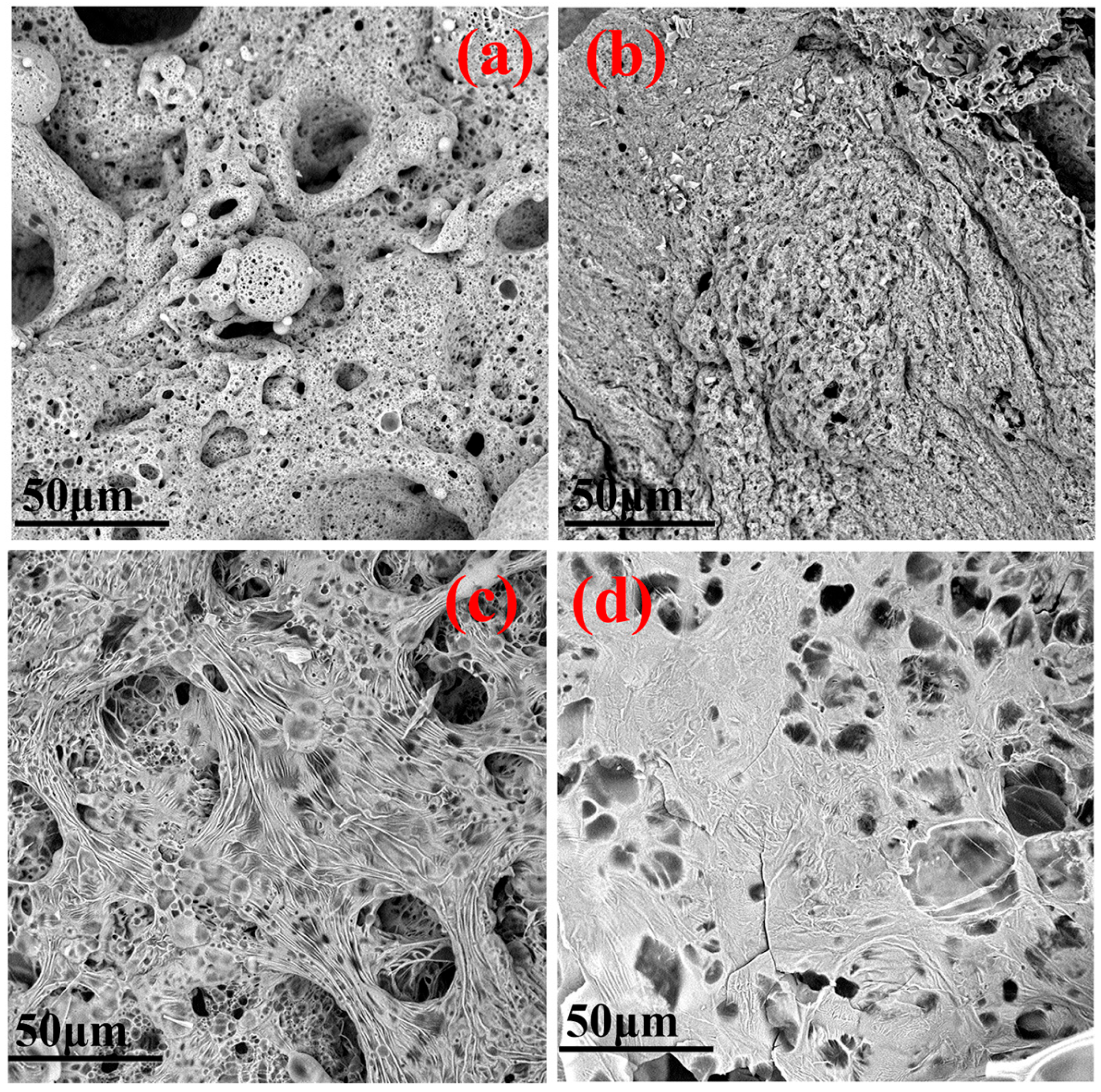
3.1.2. Surface Micromorphology of Flame-Retardant PS Composites
3.2. Thermal Stability of Flame-Retardant PS Composites
3.3. Flame-Retardant Performance of Flame-Retardant PS Composites
3.3.1. Limiting Oxygen Index and Vertical Burning Tests
3.3.2. Analysis of Char Residue
3.4. X-Ray Diffraction Analysis of Char Structure Evolution
3.5. Burning Behavior of Flame-Retardant PS Composites
3.6. Flame-Retardant Mechanism
3.7. Mechanical Properties of Flame-Retardant PS/ADP/PA-TETA Composites
3.8. Comparison of Flame-Retardant Properties with Literature Reports
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Luo, W.; Chen, M.-J.; Wang, T.; Feng, J.-F.; Fu, Z.-C.; Deng, J.-N.; Yan, Y.-W.; Wang, Y.-Z.; Zhao, H.-B. Catalytic polymer self-cleavage for CO2 generation before combustion empowers materials with fire safety. Nat. Commun. 2024, 15, 2726. [Google Scholar] [CrossRef]
- Liu, B.; Zhao, H.; Wang, Y. Advanced flame-retardant methods for polymeric materials. Adv. Mater. 2022, 34, 2107905. [Google Scholar] [CrossRef]
- Levchik, S.V.; Weil, E.D. New developments in flame retardancy of styrene thermoplastics and foams. Polym. Int. 2008, 57, 431–448. [Google Scholar] [CrossRef]
- Saquib, Q.; Siddiqui, M.A.; Ahmed, J.; Al-Salim, A.; Ansari, S.M.; Faisal, M.; Al-Khedhairy, A.A.; Musarrat, J.; AlWathnani, H.A.; Alatar, A.A.; et al. Hazards of low dose flame-retardants (BDE-47 and BDE-32): Influence on transcriptome regulation and cell death in human liver cells. J. Hazard. Mater. 2016, 308, 37–49. [Google Scholar] [CrossRef] [PubMed]
- Perera, I.; Seneweera, S.; Hirotsu, N. Manipulating the phytic acid content of rice grain toward improving micronutrient bioavailability. Rice 2018, 11, 4. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, A.; Cheng, Y.; Li, M.; Cui, Y.; Li, Z. Recent advances in biomass phytic acid flame retardants. Polym. Test. 2023, 124, 108100. [Google Scholar] [CrossRef]
- Song, W.-M.; Zhang, L.-Y.; Li, P.; Liu, Y. High-Efficient Flame-Retardant Finishing of Cotton Fabrics Based on Phytic Acid. Int. J. Mol. Sci. 2023, 24, 1093. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Z.; Huo, Y.; An, Y.; Liu, W.; Dou, Y. Bionic octopus tentacle structure-inspired engineering of HPP@PBA nanotubes: Towards mechanically reinforced epoxy nanocomposites with outstanding flame retardancy and smoke suppression. Eur. Polym. J. 2024, 221, 113510. [Google Scholar] [CrossRef]
- Liang, Y.; Xu, J.; Ahmed, N.; Shi, J.; Wen, M.; Yan, N. Construction of biomass-based flame-retardant, antimicrobial and hydrophobic wood coatings with POSS organic-inorganic nanoparticles. Eur. Polym. J. 2024, 219, 113370. [Google Scholar] [CrossRef]
- Mensah, R.A.; Shanmugam, V.; Narayanan, S.; Renner, J.S.; Babu, K.; Neisiany, R.E.; Försth, M.; Sas, G.; Das, O. A review of sustainable and environment-friendly flame retardants used in plastics. Polym. Test. 2022, 108, 107511. [Google Scholar] [CrossRef]
- Mokhena, T.C.; Sadiku, E.R.; Ray, S.S.; Mochane, M.J.; Matabola, K.P.; Motloung, M. Flame retardancy efficacy of phytic acid: An overview. J. Appl. Polym. Sci. 2022, 139, e52495. [Google Scholar] [CrossRef]
- Kang, H.; Yan, H.; Guo, R.; Wu, J.; Wang, Y.; Tian, J.; Zhang, C. Caramelization-inspired bio-based waterborne fire-resistant coating for various substrates. Chem. Eng. J. 2024, 502, 158016. [Google Scholar] [CrossRef]
- Zheng, X.-T.; Dong, Y.-Q.; Liu, X.-D.; Xu, Y.-L.; Jian, R.-K. Fully bio-based flame-retardant cotton fabrics via layer-by-layer self assembly of laccase and phytic acid. J. Clean. Prod. 2022, 350, 131525. [Google Scholar] [CrossRef]
- Yang, W.; Zhang, H.; Hu, X.; Liu, Y.; Zhang, S.; Xie, C. Self-assembled bio-derived microporous nanosheet from phytic acid as efficient intumescent flame retardant for polylactide. Polym. Degrad. Stab. 2021, 191, 109664. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, Q.; Cui, J.; Yan, Y. Ecofriendly Flame-Retardant Polystyrene Composites: Exploiting the Synergistic Effects of Phytic Acid, Polyethyleneimine, and Expandable Graphite. Materials 2025, 18, 4308. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhang, Q.; Wang, Q.; Liu, X.; Cui, J.; Yan, Y. Ecofriendly flame-retardant polystyrene composites containing phytic acid-based flame retardant and montmorillonite nanosheets. Mater. Today Commun. 2025, 48, 113409. [Google Scholar] [CrossRef]
- Zhang, S.; Wang, X.; Ding, M.; Huang, Y.; Li, L.; Wang, M. In-situ incorporation of metal phytates for green and highly efficient flame-retardant wood with excellent smoke-suppression property. Ind. Crop. Prod. 2022, 187, 115287. [Google Scholar] [CrossRef]
- Tian, H.; Wang, Y.; Qu, C.; Liu, R.; Zhao, J.; Long, H. Synthesis of metal coordination polymers based amino trimethylene phosphonic acid and zinc phytate to improve the flame retardancy of polylactic acid. Int. J. Biol. Macromol. 2025, 322, 146922. [Google Scholar] [CrossRef]
- Qin, Z.; Yang, R.; Zhang, W.; Li, D.; Jiao, Q. Synergistic barrier effect of aluminum phosphate on flame retardant polypropylene based on ammonium polyphosphate/dipentaerythritol system. Mater. Des. 2019, 181, 107913. [Google Scholar] [CrossRef]
- Li, Y.; Yang, Z.; Guan, J.; Yan, Q.; Lei, Z. Organic-inorganic hybrid functionalized chitosan/ammonium polyphosphate (APP): A synergistic strategy for flame-retardant and high-mechanical-strength epoxy thermosets. Int. J. Biol. Macromol. 2025, 309, 142579. [Google Scholar] [CrossRef]
- Wan, C.; Fan, Z.; Zhili, G.; Longxiang, T. Effects of Aluminum Phosphate on the Fire-Retardant and Mechanical Properties of Wood Flour/HDPE Composite. Polym. Mater. Sci. Eng. 2017, 33, 111–115. [Google Scholar] [CrossRef]
- Gao, Y.-Y.; Deng, C.; Du, Y.-Y.; Huang, S.-C.; Wang, Y.-Z. A novel bio-based flame retardant for polypropylene from phytic acid. Polym. Degrad. Stab. 2019, 161, 298–308. [Google Scholar] [CrossRef]
- Szadkowski, B.; Rybiński, P.; Sarlin, E.; Marzec, A. Bio-functionalization of fillers with natural phytic acid for sustainable flame-retardant agents for natural rubber composites. Ind. Crop. Prod. 2024, 222, 119504. [Google Scholar] [CrossRef]
- ISO 5660-1:2015; Reaction-to-Fire Tests—Heat Release, Smoke Production and Mass Loss Rate—Part 1: Heat Release Rate (Cone Calorimeter Method) and Smoke Production Rate (Dynamic Measurement). International Organization for Standardization: Geneva, Switzerland, 2015.
- Wang, L.; Guo, Y.; Huang, Y.; Wei, M.; Chen, Z.; Sheng, S.; Jia, H.; Jiang, P. Bio-based phytic acid–L-citrulline–chitosan flame retardant: Synthesis, flame retardancy, smoke suppression, and mechanism in transparent melamine resin coatings for oriented strand board. Polym. Degrad. Stab. 2026, 243, 111755. [Google Scholar] [CrossRef]
- Cheng, B.; Zhou, Q.; Chen, J.; Zhang, X.; Zhu, C.; Wu, M. Vinylated Modification of Biophytic Acid and Flame-Retardant/Crease-Proofing Finishing of Cotton Fabrics via In Situ Copolymerization. Materials 2023, 16, 286. [Google Scholar] [CrossRef]
- Ai, Y.; Zhu, Z.; Liu, W.; Sun, H.; Li, J.; Jiao, R.; Li, A. Aluminum dihydrogen phosphate-melamine modified hollow spheres/epoxy composites: Enhanced flame retardancy, thermal insulation and wave-absorbing properties. Chem. Eng. J. 2025, 524, 169340. [Google Scholar] [CrossRef]
- Muhemmed, A.S.; Aziz, S.B. Structural, spectroscopic, morphological and optical studies of new polymer composite based on polystyrene inserted with natural bitumen. Sci. Rep. 2025, 15, 25978. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.; Xie, Y.; Lan, L.; Wu, F.; Feng, D.; Meng, Y.; Mei, Y.; Xie, D. Synergistic flame retardancy of a bio-based additive and intumescent system for enhanced fire safety in flexible polyurethane foams. J. Polym. Environ. 2025, 33, 3190–3208. [Google Scholar] [CrossRef]
- Xie, R.; Qu, B. Thermo-oxidative degradation behaviors of expandable graphite-based intumescent halogen-free flame retardant LLDPE blends. Polym. Degrad. Stab. 2001, 71, 395–402. [Google Scholar] [CrossRef]
- Wu, Q.; Qu, B. Synergistic effects of silicotungistic acid on intumescent flame-retardant polypropylene. Polym. Degrad. Stab. 2001, 74, 255–261. [Google Scholar] [CrossRef]
- Goj, P.; Handke, B.; Stoch, P. Vibrational characteristics of aluminum–phosphate compounds by an experimental and theoretical approach. Sci. Rep. 2022, 12, 17495. [Google Scholar] [CrossRef] [PubMed]
- van der Meer, H. The crystal structure of a monoclinic form of aluminium metaphosphate, Al(PO3)3. Acta Cryst. 1976, 32, 2423–2426. [Google Scholar] [CrossRef]
- Bemmer, V.; Bowker, M.; Carter, J.H.; Davies, P.R.; Edwards, L.E.; Harris, K.D.M.; Hughes, C.E.; Robinson, F.; Morgan, D.J.; Thomas, M.G. Rationalization of the X-ray photoelectron spectroscopy of aluminium phosphates synthesized from different precursors. RSC Adv. 2020, 10, 8444–8452. [Google Scholar] [CrossRef]
- Wang, J.; Shan, H.; Wu, Y.; Song, L.; Xue, J.; Wang, B.; Wang, C. Room-temperature curing high-strength, water-resistant, flame-retardant adhesive based on aluminum dihydrogen phosphate and gelatinized starch cross-linking. Int. J. Biol. Macromol. 2025, 306, 141441. [Google Scholar] [CrossRef] [PubMed]
- Amiinu, I.S.; Zhang, J.; Kou, Z.; Liu, X.; Asare, O.K.; Zhou, H.; Cheng, K.; Zhang, H.; Mai, L.; Pan, M.; et al. Self-Organized 3D Porous Graphene Dual-Doped with Biomass-Sponsored Nitrogen and Sulfur for Oxygen Reduction and Evolution. ACS Appl. Mater. Interfaces 2016, 8, 29408–29418. [Google Scholar] [CrossRef]
- Jin, X.; Liu, X.; Li, X.; Yang, C.; Zhang, T.; Lu, Z.; Dong, C. Spider web-inspired multifunctional compound for durable fire safety, smoke suppression and enhanced strength cotton fabrics. Int. J. Biol. Macromol. 2025, 308, 142711. [Google Scholar] [CrossRef]
- Chen, G.; He, F.; Ye, R.; Xie, D.; Chen, F.; Li, S.-N.; Wu, S.; Guo, Y.; Peng, L.; Dai, J.; et al. Facile synthesis of a high-efficiency phosphorus-based flame retardant enabling epoxy resin with superior flame retardancy, mechanical robustness, and transparency. Chem. Eng. J. 2026, 527, 171893. [Google Scholar] [CrossRef]
- Amirabadi, S.; Kheradmandkeysomi, M.; Zandieh, A.; Serles, P.; Tanguy, N.; Filleter, T.; Sain, M.; Park, C.B. Highly tough and flame retardant polystyrene composites by elastomeric nanofibers and hexagonal boron nitride. J. Mater. Sci. Technol. 2024, 198, 208–220. [Google Scholar] [CrossRef]
- Wang, X.; Tu, H.; Xiao, H.; Lu, J.; Xu, J.; Gu, G. A novel halogen-free flame-retardant fabrication for the study of smoke suppression and flame retardancy of polystyrene. Polymer 2023, 283, 126240. [Google Scholar] [CrossRef]
- Li, W.; Qi, L.; Ye, D.; Cai, W.; Xing, W. Facile modification of aluminum hypophosphate and its flame retardancy for polystyrene. Chin. J. Chem. Eng. 2023, 60, 90–98. [Google Scholar] [CrossRef]
- Wang, G.; Bai, S. Synergistic effect of expandable graphite and melamine phosphate on flame-retardant polystyrene. J. Appl. Polym. Sci. 2017, 134, 45474. [Google Scholar] [CrossRef]
- Yan, Y.-W.; Huang, J.-Q.; Guan, Y.-H.; Shang, K.; Jian, R.-K.; Wang, Y.-Z. Flame retardance and thermal degradation mechanism of polystyrene modified with aluminum hypophosphite. Polym. Degrad. Stab. 2014, 99, 35–42. [Google Scholar] [CrossRef]
- Xia, Y.; Mao, Z.; Jin, F.; Guan, Y.; Zheng, A. Synthesis of 1-hydroxy ethylidene-1,1-diphosphonic ammonium and the promise of this ammonium salt as an intumescent flame retardant in polystyrene. Polym. Degrad. Stab. 2014, 102, 186–194. [Google Scholar] [CrossRef]
- Liu, J.; Yu, Z.; Chang, H.; Zhang, Y.; Shi, Y.; Luo, J.; Pan, B.; Lu, C. Thermal degradation behavior and fire performance of halogen-free flame-retardant high impact polystyrene containing magnesium hydroxide and microencapsulated red phosphorus. Polym. Degrad. Stab. 2014, 103, 83–95. [Google Scholar] [CrossRef]











| Samples | ADP (phr) | PA-TETA (phr) * | PS (phr) |
|---|---|---|---|
| PS/10ADP | 10 | 0 | 100 |
| PS/20ADP | 20 | 0 | 100 |
| PS/30ADP | 30 | 0 | 100 |
| PS/5PA-TETA | 0 | 5 | 100 |
| PS/10PA-TETA | 0 | 10 | 100 |
| PS/15PA-TETA | 0 | 15 | 100 |
| PS/20PA-TETA | 0 | 20 | 100 |
| PS/10ADP/10PA-TETA | 10 | 10 | 100 |
| PS/10ADP/15PA-TETA | 10 | 15 | 100 |
| PS/10ADP/20PA-TETA | 10 | 20 | 100 |
| PS/10ADP/25PA-TETA | 10 | 25 | 100 |
| Samples | T5% (°C) | Tmax (°C) | Char Residue (%) |
|---|---|---|---|
| PS | 281.1 | 318.8 | 0 |
| PS/10ADP | 288.1 | 302.0 | 2.73 |
| PS/5PA-TETA | 324.8 | 349.5 | 1.33 |
| PS/10PA-TETA | 293.2 | 414.3 | 1.59 |
| PS/15PA-TETA | 296.1 | 412.0 | 2.33 |
| PS/20PA-TETA | 281.1 | 408.4 | 2.67 |
| PS/10ADP/10PA-TETA | 287.5 | 350.5 | 4.11 |
| PS/10ADP/15PA-TETA | 324.0 | 404.2 | 4.72 |
| PS/10ADP/20PA-TETA | 323.5 | 378.1 | 7.22 |
| PS/10ADP/25PA-TETA | 323.1 | 395.0 | 9.80 |
| Samples | LOI (%) | UL94 |
|---|---|---|
| PS | 17.4 | No rating |
| PS/10ADP | 19.3 | No rating |
| PS/20ADP | 20.5 | No rating |
| PS/30ADP | 21.2 | No rating |
| PS/5PA-TETA | 20.4 | No rating |
| PS/10PA-TETA | 21.6 | No rating |
| PS/15PA-TETA | 23.9 | No rating |
| PS/20PA-TETA | 24.8 | No rating |
| PS/10ADP/10PA-TETA | 27.1 | No rating |
| PS/10ADP/15PA-TETA | 28.5 | V0 |
| PS/10ADP/20PA-TETA | 30.0 | V0 |
| PS/10ADP/25PA-TETA | 42.0 | V0 |
| Parameters | PS | PS/10ADP | PS/15PA-TETA | PS/10ADP/15PA-TETA |
|---|---|---|---|---|
| TTI (s) | 36 | 33 | 53 | 54 |
| PHRR (kW/m2) | 716.4 | 391.7 | 481.9 | 187.4 |
| THR (MJ/m2) | 138.2 | 94.0 | 78.8 | 74.4 |
| TSP (m2) | 4.62 | 3.34 | 10.0 | 8.24 |
| Av-EHC (MJ/kg) | 28.6 | 28.5 | 23.9 | 20.8 |
| Time of flameout (s) | 440 | 414 | 459 | 753 |
| Char residue rate (%) | 0 | 10.0 | 5.3 | 19.9 |
| Sample | Loading | LOI (%) | Increase in LOI (%) | UL-94 | Tensile Strength Retention (%) | References |
|---|---|---|---|---|---|---|
| PS/10ADP/15PA-TETA | 25 phr | 28.5 | 63.8 | V-0 | 78.4 | This work |
| PS/PAEI/MMT | 26 phr | 32 | 77.8 | V-0 | 61.9 | [16] |
| PS/PAE/EG | 10 phr | 27.7 | 54 | V-0 | 91.8 | [15] |
| PS/hBN/SBC | 12 wt% | 24 | 33.3 | NR * | NR * | [39] |
| PS/10%PON/10%EG | 20 wt% | 25.8 | 42.5 | NR * | NR * | [40] |
| PS/25%MAHPi | 25 wt% | 24 | 33.3 | V-0 | NR * | [41] |
| PS/MP/EG | 20 wt% | 28 | 55.6 | V-0 | 83 | [42] |
| PS/25%AP | 25 wt% | 25.6 | 42.5 | V-0 | NR * | [43] |
| PS/25%HEDPA/PER/MEL | 25 wt% | 28.3 | 57.5 | V-0 | NR * | [44] |
| HIPS/MH/MRP | 48.7 wt% | 26.6 | 48 | V-0 | NR * | [45] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Li, Z.; Zhang, Q.; Cui, J.; Yan, Y. A Green P–N–Al Synergistic System for Eco-Friendly Flame-Retardant Polystyrene. Materials 2026, 19, 941. https://doi.org/10.3390/ma19050941
Li Z, Zhang Q, Cui J, Yan Y. A Green P–N–Al Synergistic System for Eco-Friendly Flame-Retardant Polystyrene. Materials. 2026; 19(5):941. https://doi.org/10.3390/ma19050941
Chicago/Turabian StyleLi, Zhunzhun, Qimei Zhang, Jian Cui, and Yehai Yan. 2026. "A Green P–N–Al Synergistic System for Eco-Friendly Flame-Retardant Polystyrene" Materials 19, no. 5: 941. https://doi.org/10.3390/ma19050941
APA StyleLi, Z., Zhang, Q., Cui, J., & Yan, Y. (2026). A Green P–N–Al Synergistic System for Eco-Friendly Flame-Retardant Polystyrene. Materials, 19(5), 941. https://doi.org/10.3390/ma19050941







