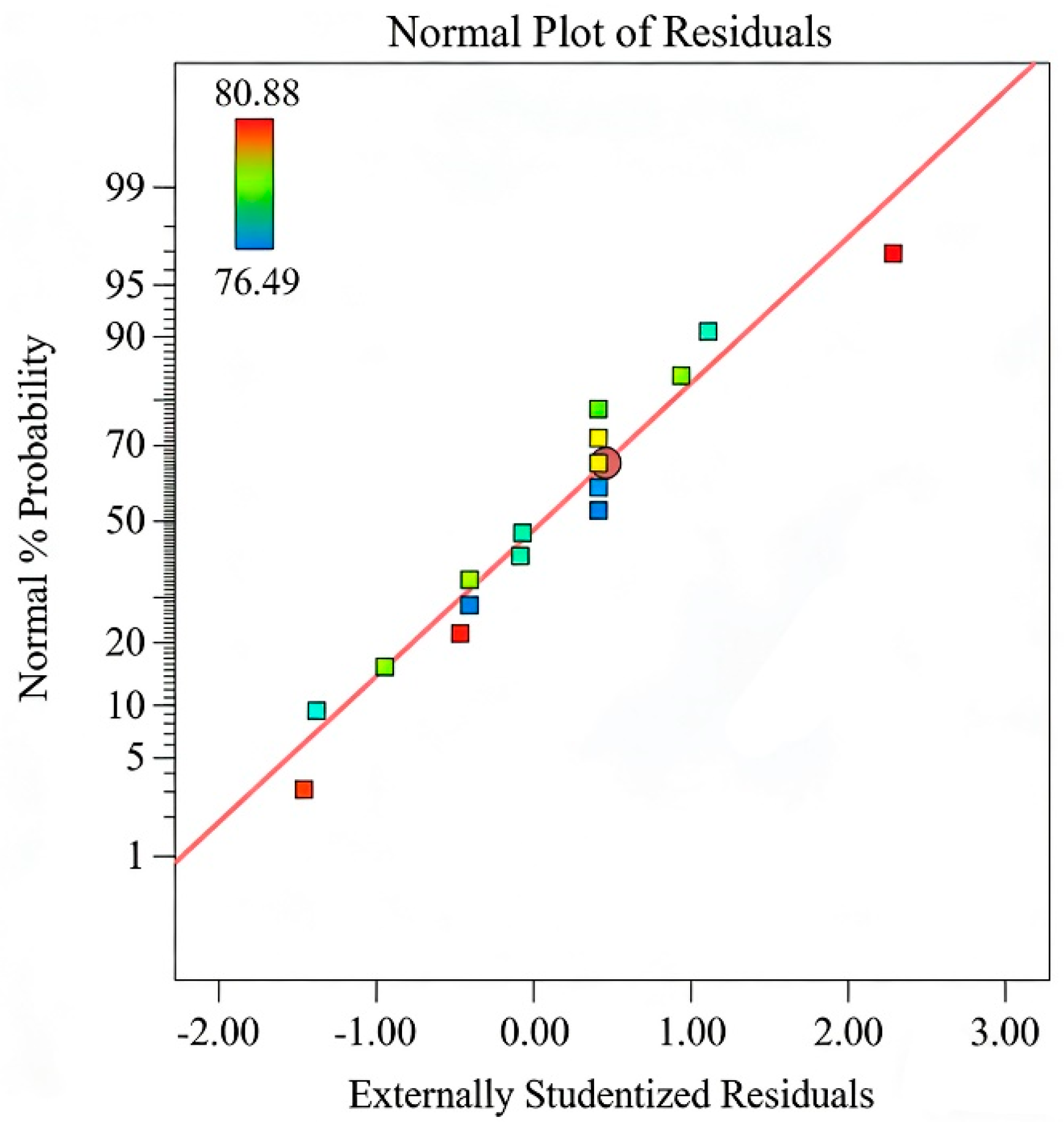
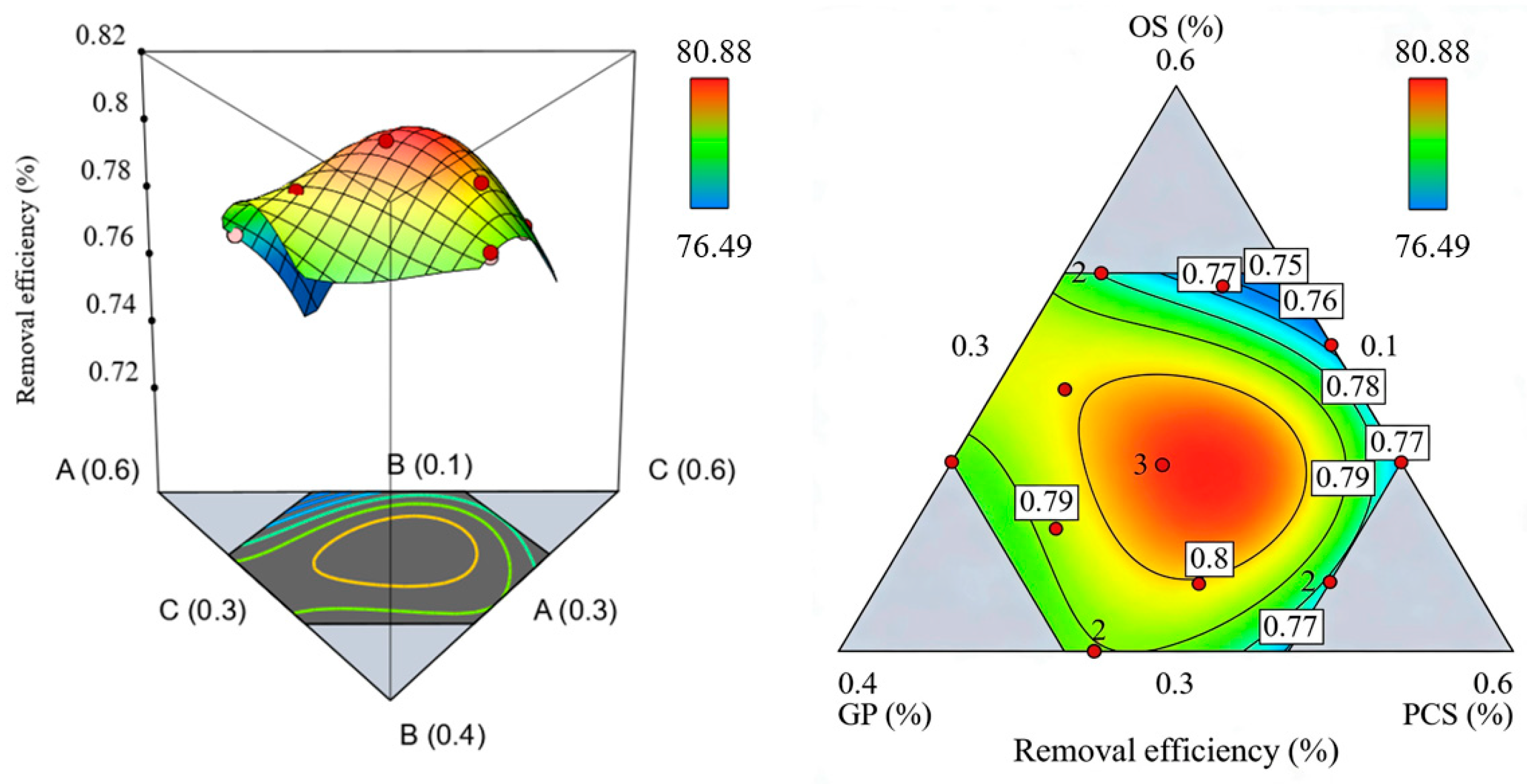
3.4.1. Comparison of Adsorption Performance Between BC and BC@La
To delineate the contribution of the lanthanum modification, the adsorption capacities of the unmodified base ceramsite (BC) and the modified BC@La are directly compared across concentrations. The optimally formulated BC (OS:PCS:GP = 39.8:39.5:20.7) reached an adsorption capacity of ≈1.6 mg/g at a low phosphate concentration of 2 mg/L, with a removal efficiency of 81% (
Table A1 (see
Appendix A for details)). This value likely represents the saturation capacity (
) of the ceramic support itself, as its limited, non-specific sites are easily saturated.
In stark contrast, the BC@La ceramsite exhibited a dynamic adsorption profile: its capacity increased from 0.88 mg/g at 2 mg/L to 2.78 mg/g at 50 mg/L (under standard conditions: dosage 1 g/L, 24 h equilibrium) (
Section 3.4.2 and
Figure 4b), demonstrating abundant and high-affinity active sites. Most decisively, the lanthanum-specific capacity of BC@La reached 128.0 mg P per gram of La—two orders of magnitude higher than the total adsorption capacity of the BC support (≈1.6 mg/g). This quantitative comparison provides unambiguous evidence that the porous BC matrix primarily serves as a mechanical host, while the surface-immobilized lanthanum species are responsible for the high-capacity, high-affinity phosphate adsorption.
3.4.2. Phosphorus Adsorption Characteristics of BC@La Ceramsite
During adsorption, pH serves as a critical parameter governing the surface charge and speciation of functional groups on the adsorbent.
Figure 4a presents the phosphate removal efficiency of BC@La from simulated wastewater across different initial pH levels. The results indicate that removal efficiency first increases and then decreases as pH varies from 3 to 11. This trend is primarily attributed to the increasing negative charge on phosphate species as pH rises from acidic to neutral. This enhances their electrostatic attraction to the positively charged adsorbent surface, thereby promoting adsorption. With increasing alkalinity, electrostatic repulsion and competitive adsorption by OH
− ions become predominant. Although phosphate ions exist predominantly as PO
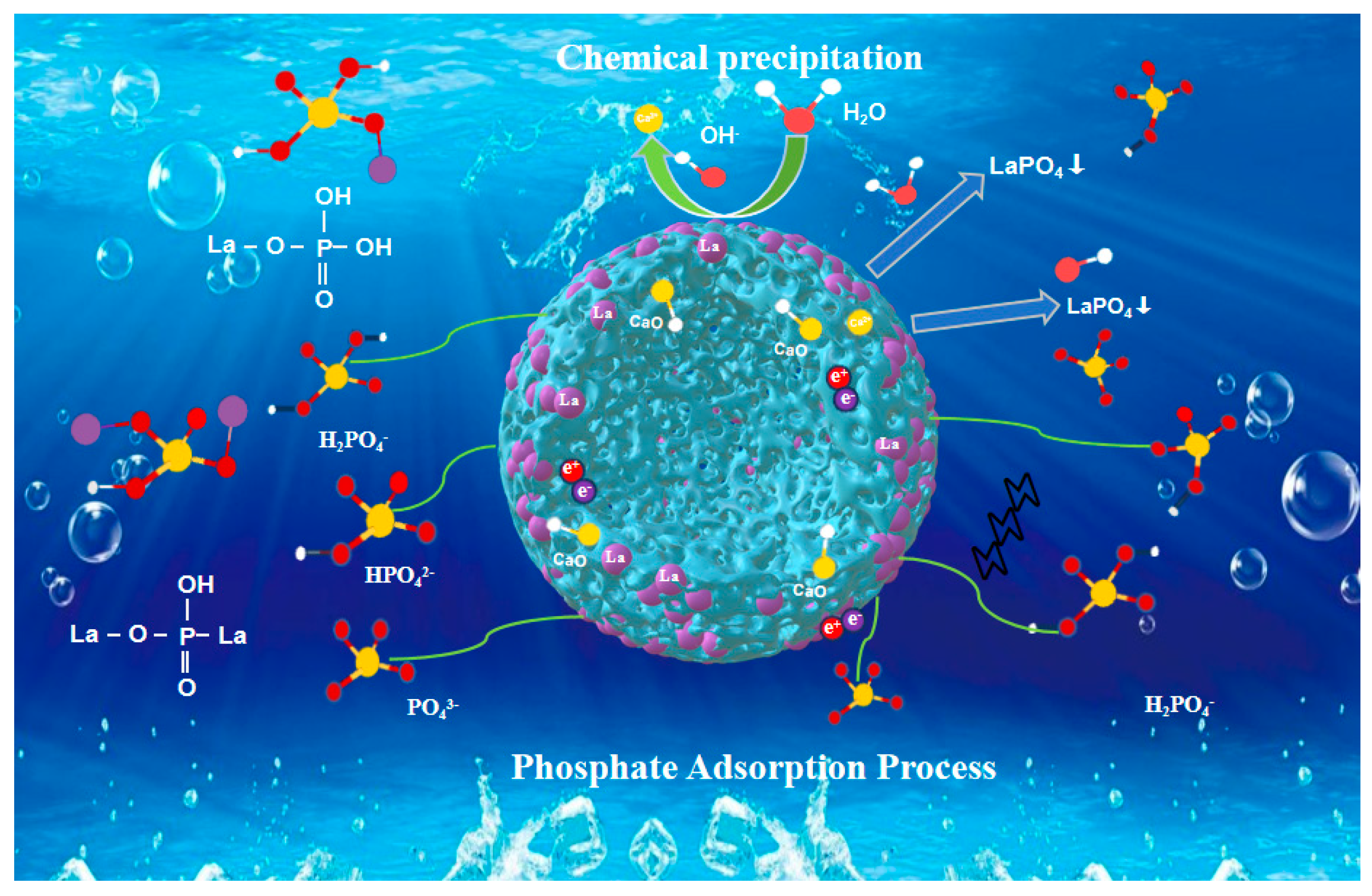
43− with higher negative charge under alkaline conditions, their access to and binding with adsorption sites are hindered, leading to a pronounced reduction in removal efficiency. The plausible chemical interactions between La and P are described by Equations (3)–(5) [
29].
As shown in
Figure 4b, 1 g of ceramsite was added to 1 L of phosphate-containing wastewater with varying initial concentrations. With increasing phosphate concentration, the adsorption uptake (q) of the ceramsite increased progressively, while the removal efficiency exhibited a gradual decline (
Figure 4b). At a concentration of 50 mg/L, BC@La achieved a removal efficiency of 88.34, corresponding to a q of 2.78 mg/g under these specific conditions.
Figure 4c illustrates the influence of adsorbent dosage and different initial phosphate concentrations on removal efficiency. It is crucial to distinguish the nature of the values reported here from the standardized q
e. The q values plotted in
Figure 4b,c represent the observed phosphate uptake under the specific, non-standardized conditions of each individual experiment (varying dosage or concentration). These values are presented to illustrate trends and are distinct from the equilibrium adsorption capacity (q
e) obtained under the controlled, standardized conditions of the adsorption isotherm experiments (fixed dosage of 1 g/L, 24 h equilibrium), as detailed in
Section 3.4.3 and
Figure 5.
With increasing adsorbent dosage, the observed uptake decreased for all initial phosphate concentrations tested (5, 10, 20, and 50 mg/L). For the wastewater containing 50 mg/L phosphate, the observed uptake declined gradually when the ceramsite dosage increased from 0.3 to 1 g/L. However, when the dosage was further increased from 1 to 2 g/L, the observed uptake decreased markedly from 2.87 mg/g (obtained at the low, non-standard dosage of 0.3 g/L) to 1.32 mg/g. This value (2.87 mg/g) should not be compared directly with the standardized equilibrium adsorption capacity (qe) of 2.78 mg/g obtained at the optimal dosage of 1 g/L, as they originate from fundamentally different experimental protocols. Similar trends were observed for the other concentration groups. The optimal ceramsite (BC@La) developed in this study demonstrated excellent adsorption performance, highlighting its significant application potential. Therefore, considering both adsorption effectiveness and economic feasibility, a dosage of 1 g/L was selected as the optimal condition for the static adsorption experiments with the ceramsite.
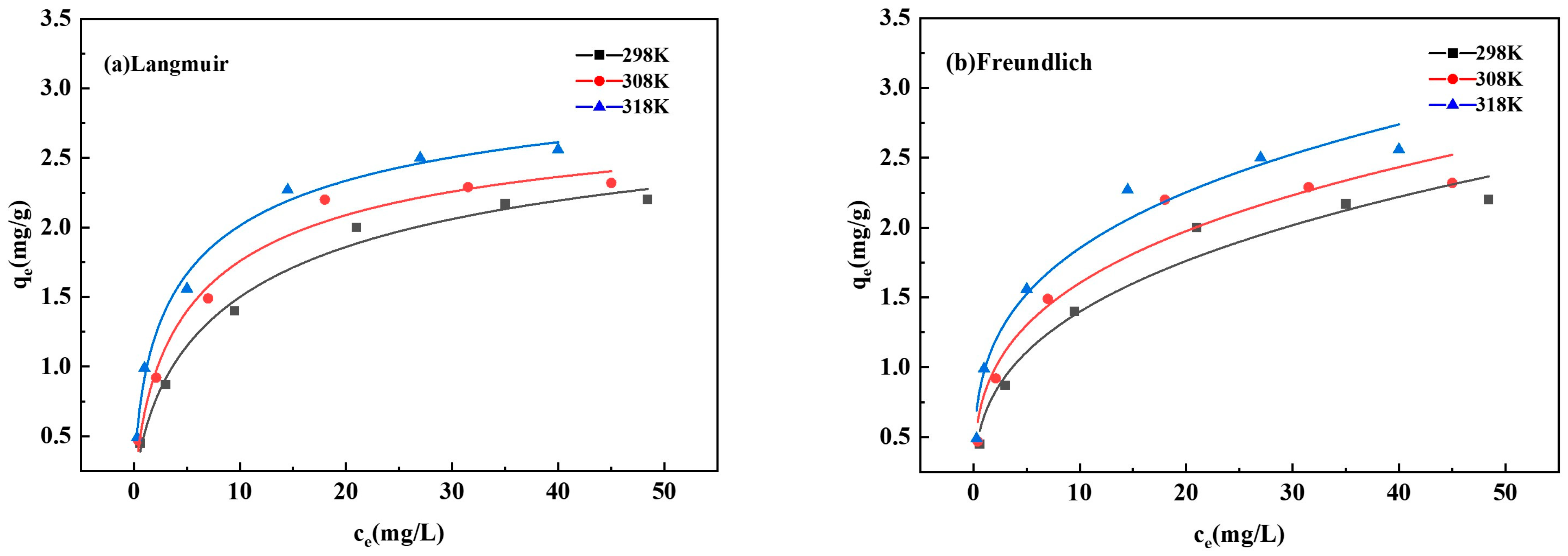
3.4.3. Adsorption Isotherm
To investigate the temperature effect (298, 308, and 318 K) on phosphate adsorption, experiments were conducted under standardized conditions: an initial phosphate concentration range of 0.5–50 mg/L, a constant adsorbent dosage of 1 g/L, and an equilibrium period of 24 h. The equilibrium adsorption capacity (q
e), determined under this fixed-dosage protocol, serves as a robust metric for comparative analysis and modeling. Subsequently, the obtained equilibrium data were analyzed by fitting to both the Langmuir and Freundlich isotherm models (Equations (A1) and (A2) (see
Appendix C for details)).
The fitted curves (
Figure 5a,b) exhibit a characteristic shape: adsorption capacity increased rapidly at low equilibrium concentrations and then approached a plateau, indicating the saturation of available adsorption sites [
30]. To quantitatively compare the models and gain physical insights, key parameters are summarized in
Table 5. The higher correlation coefficients (R
2) for the Langmuir model across all temperatures suggest that it provides a better description of the adsorption data than the Freundlich model [
31]. The maximum monolayer adsorption capacity (
) derived from the Langmuir model increased from 2.20 mg/g at 298 K to 2.56 mg/g at 318 K. Concurrently, the Langmuir constant (
), which reflects adsorption affinity, also increased with temperature. This consistent trend confirms the endothermic nature of the phosphate adsorption process on BC@La [
32].
It is important to critically discuss the implications of the Langmuir fit. A good fit primarily indicates that the adsorption system conforms to the model’s core assumptions: monolayer coverage and a surface with relatively uniform site energy [
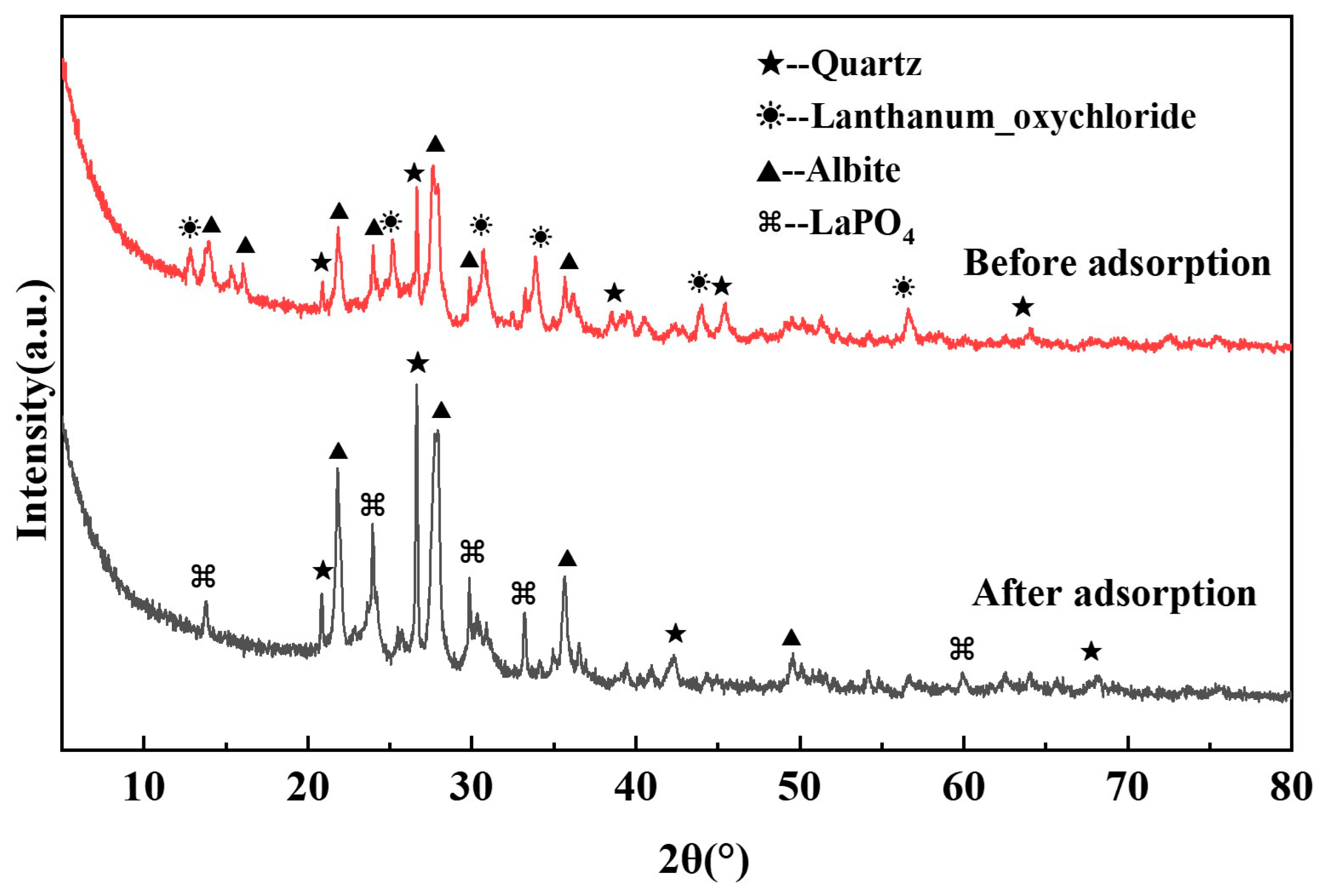
33]. It does not, by itself, exclude the contribution of other simultaneous processes. In our system, the predominance of La
3+ active sites—recovered and uniformly dispersed from waste catalyst—provides a reasonable basis for the “uniform sites” approximation. More importantly, the strong, specific adsorption behavior implied by the Langmuir model is directly corroborated by the formation of LaPO
4 precipitates observed via XRD and the corresponding chemical state changes detected by XPS (
Section 3.5). This multi-technique convergence supports that the adsorption is dominated by site-specific chemisorption, consistent with the physical picture of the Langmuir model.
In summary, the isotherm analysis, supported by complementary characterization, indicates that phosphate adsorption onto BC@La is best described as an endothermic, monolayer process driven by strong chemisorption onto La3+ sites, ultimately leading to surface precipitation.
Figure 5a,b show that at lower initial phosphorus concentrations, the adsorption capacity of the ceramsite increases rapidly. As the initial concentration further increases, the curve flattens, and the adsorption capacity approaches saturation. This indicates that at low initial concentrations, the adsorption sites on the ceramsite are not fully occupied, whereas at higher concentrations, the sites become saturated, leading to the plateau in the curve.
Table 5 presents the fitted parameters of the Langmuir and Freundlich models, which describe the adsorption thermodynamics of phosphate onto BC@La. The standard equilibrium adsorption capacity (q
e) increased progressively with temperature, reaching a maximum value of 2.56 mg P per gram of adsorbent at 318 K (
Table A3 (see
Appendix A for details) provides a comparative overview with other adsorbents on this basis). This moderate gravimetric capacity results from the deliberately low lanthanum loading (2 wt.%), sourced from spent catalyst, reflecting a design philosophy focused on the efficient and sustainable utilization of a critical rare-earth resource. To accurately assess the effectiveness of the active phase, the adsorption performance was also evaluated in terms of lanthanum utilization efficiency. In this regard, BC@La exhibits an exceptional lanthanum-specific capacity of 128.0 mg P per gram of La, a metric that surpasses many reported La-based adsorbents when normalized to the mass of the active component. Beyond its intrinsic adsorption metrics, BC@La offers practical engineering advantages: its granular morphology facilitates solid–liquid separation compared to powdered analogues, and its synthesis via solid waste valorization represents a cost-effective and environmentally synergistic materials strategy. Finally, the superior fitting of the Langmuir model (R
2) over the Freundlich model indicates that the adsorption process is predominantly monolayer chemisorption [
34].
3.4.4. Adsorption Kinetics
Table 6 summarizes the fitting parameters of the pseudo-first-order (PFO) and pseudo-second-order (PSO) kinetic models. The PSO model demonstrates superior performance, exhibiting a higher calculated equilibrium adsorption capacity (q
e = 2.64 mg/g) and a more satisfactory correlation coefficient (R
2 = 0.9615) compared to the PFO model. Consequently, the adsorption kinetics of phosphate onto the adsorbent are better described by the PSO model, suggesting a process predominantly governed by chemisorption [
35,
36]. This finding aligns with recent kinetic studies on pharmaceutical adsorption using activated carbon [
37]. A distinct merit of the PSO model is its provision of the initial adsorption rate (h), a parameter with explicit physical significance. As defined by h = k
2q
e2, the calculated value was 0.0313 mg·g
−1·min
−1 (
Table 6). This rate quantitatively characterizes the adsorption velocity at the very beginning of the process (t → 0). The obtained h value corroborates the rapid initial uptake phase evident in the kinetic profile (
Figure 5c), underscoring the high accessibility and efficacy of the active sites on the BC@La surface for phosphate sequestration.
3.4.5. Adsorption Thermodynamics
The temperature-dependent adsorption isotherms (
Figure 5d) were employed to determine the thermodynamic parameters for phosphate adsorption onto the ceramsite, utilizing Equations (A5)–(A7) (see
Appendix C for details). These equations incorporate the distribution coefficient (
KD), the initial and equilibrium phosphate concentrations (c
0 and c
e), the universal gas constant
R (8.314 J·mol
−1·K
−1), and the absolute temperature
T. The derived parameters are summarized in
Table 7. The calculated Gibbs free energy change (Δ
G°) was negative at 298, 308, and 318 K (−2.3687, −2.7368, and −3.0917 kJ·mol
−1, respectively), confirming the spontaneity of the adsorption process. Furthermore, the increasingly negative values of Δ
G° with rising temperature indicate enhanced thermodynamic favorability at higher temperatures. The adsorption was found to be endothermic, as evidenced by a positive enthalpy change (Δ
H° = 8.4481 kJ·mol
−1) [
38]. Additionally, the positive entropy change (Δ
S° = 36.3366 J·mol
−1·K
−1) suggests an increase in the overall disorder of the system following adsorption.