Finite Element Analysis of Anterior Odontoid Screw Fixation for Type II Odontoid Fractures
Abstract
1. Introduction
2. Materials and Methods
2.1. Population and Image Acquisition
2.2. Segmentation and 3D Modeling
2.3. FEM Model Preparation
2.4. Boundary Conditions and Applied Loads
2.5. Evaluated Parameters
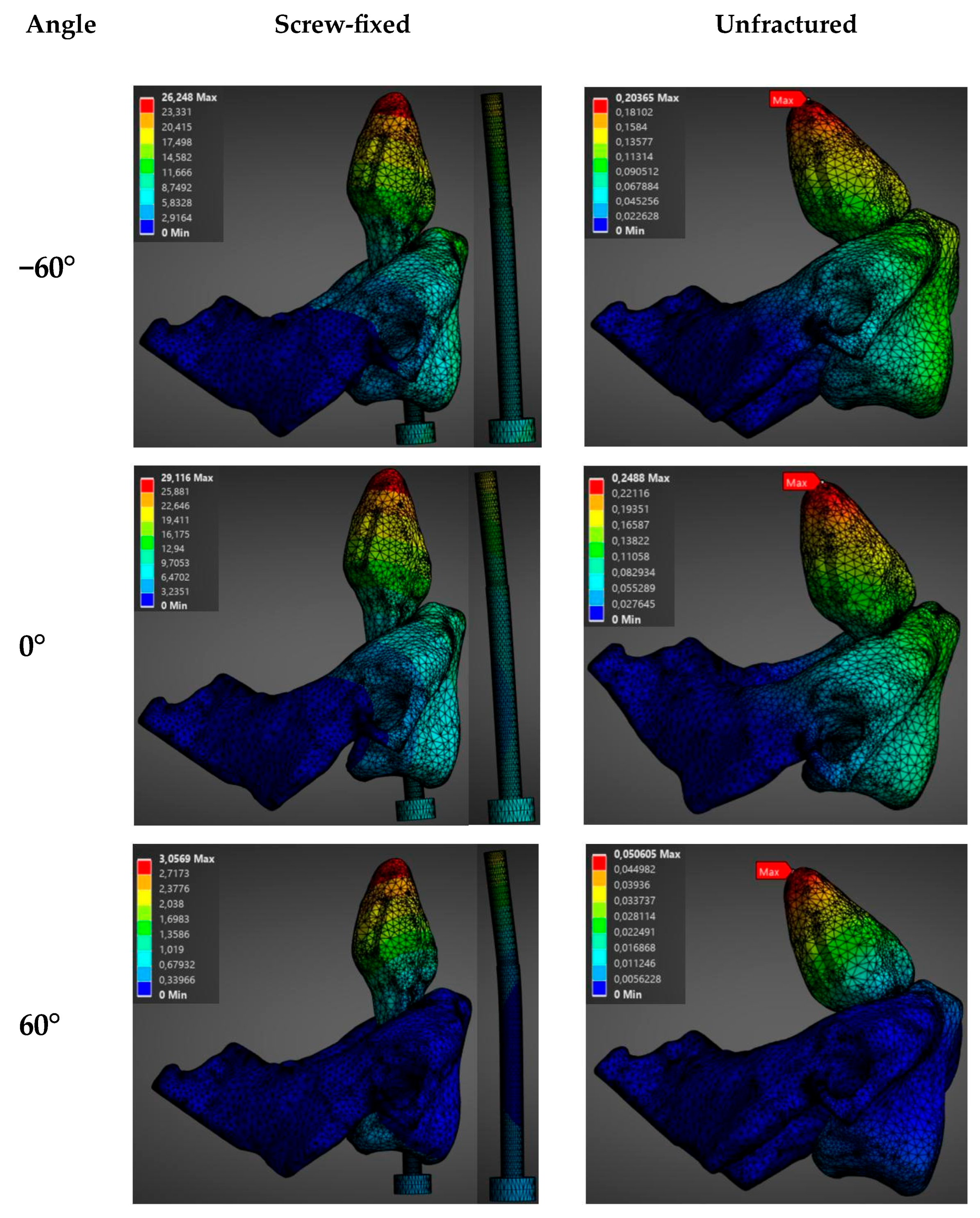
- Deformation: relative displacements between the atlas (C1) and the axis (C2) in the anteroposterior direction. The average total deformation in the odontoid was obtained by averaging the stress values computed by the finite element solver over the odontoid region.
- Stresses: Von Mises stress distribution in the screw, cortical bone, and fracture zone. The average stress in the odontoid was obtained by averaging the stress values computed by the finite element solver over the odontoid region.
3. Results
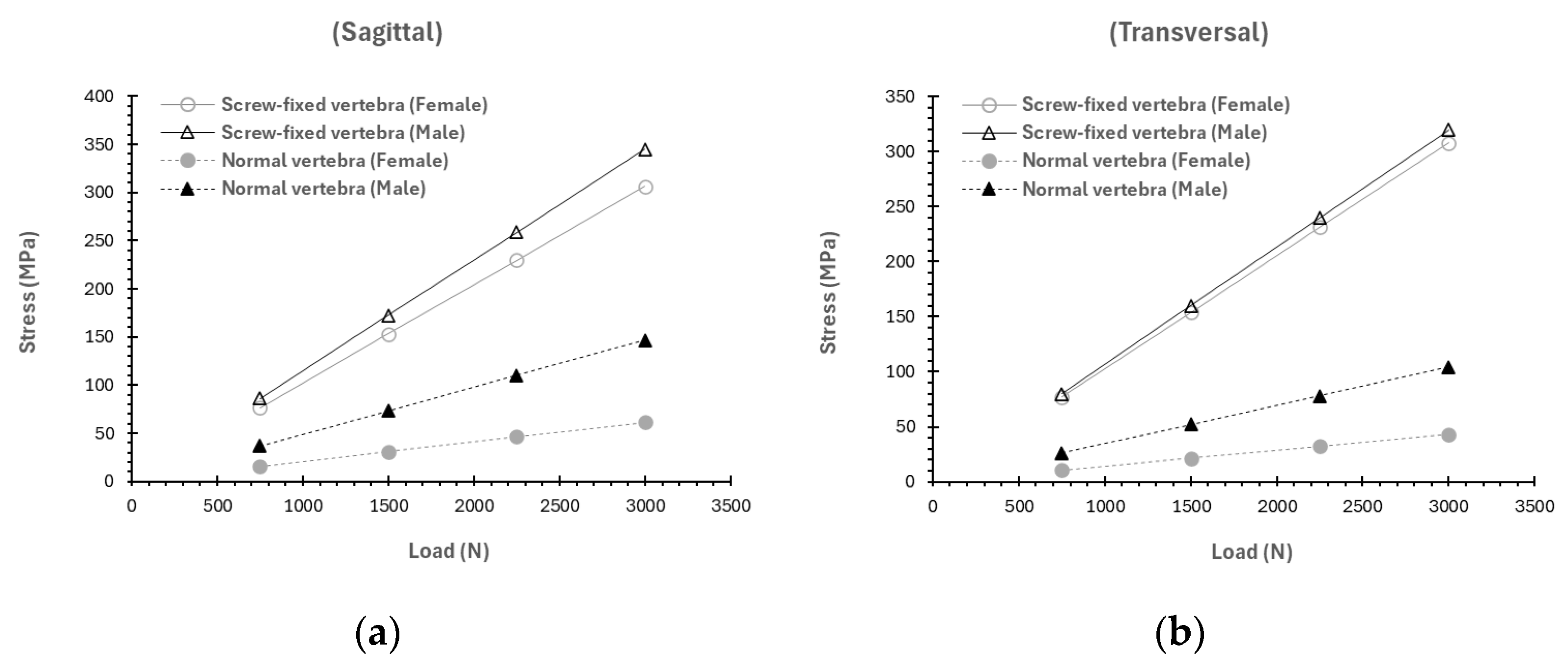
3.1. Influence of the Magnitude of the Force
3.2. Influence of the Orientation of the Load
3.3. Influence of the Sex
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ryan, M.D.; Taylor, T.K. Odontoid Fractures in the Elderly. J. Spinal Disord. 1993, 6, 397–401. [Google Scholar] [CrossRef]
- Bernard, F.; Troude, L.; Isnard, S.; Lemée, J.-M.; Terrier, L.M.; François, P.; Velut, S.; Gay, E.; Fournier, H.-D.; Roche, P.-H. Long Term Surgical Results of 154 Petroclival Meningiomas: A Retrospective Multicenter Study. Neurochirurgie 2019, 65, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.; Sakai, D.; Yamamoto, Y.; Nagai, T.; Sato, M.; Mochida, J. Analysis of Predisposing Factors in Elderly People with Type II Odontoid Fracture. Spine J. 2014, 14, 861–866. [Google Scholar] [CrossRef] [PubMed]
- Kaesmacher, J.; Schweizer, C.; Valentinitsch, A.; Baum, T.; Rienmüller, A.; Meyer, B.; Kirschke, J.S.; Ryang, Y. Osteoporosis Is the Most Important Risk Factor for Odontoid Fractures in the Elderly. J. Bone Miner. Res. 2017, 32, 1582–1588. [Google Scholar] [CrossRef] [PubMed]
- Alluri, R.; Bouz, G.; Solaru, S.; Kang, H.; Wang, J.; Hah, R.J. A Nationwide Analysis of Geriatric Odontoid Fracture Incidence, Complications, Mortality, and Cost. Spine 2021, 46, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Yoganandan, N.; Pintar, F.A. Odontoid Fracture in Motor Vehicle Environments. Accid. Anal. Prev. 2005, 37, 505–514. [Google Scholar] [CrossRef]
- Anderson, L.D.; D’Alonzo, R.T. Fractures of the Odontoid Process of the Axis. J. Bone Jt. Surg. 1974, 56, 1663–1674. [Google Scholar] [CrossRef]
- Hsu, W.K.; Anderson, P.A. Odontoid Fractures: Update on Management. J. Am. Acad. Orthop. Surg. 2010, 18, 383–394. [Google Scholar] [CrossRef]
- Teo, E.C.; Paul, J.P.; Evans, J.H.; Ng, H.W. Experimental Investigation of Failure Load and Fracture Patterns of C2 (Axis). J. Biomech. 2001, 34, 1005–1010. [Google Scholar] [CrossRef]
- Böhler, J. Anterior Stabilization for Acute Fractures and Non-Unions of the Dens. J. Bone Jt. Surg. 1982, 64, 18. [Google Scholar] [CrossRef]
- Evans, A.R.; Bakhsheshian, J.; Graffeo, C.S.; Smith, Z.A. Surgical Management of Spinal Pathologies in the Octogenarian: A Narrative Review. Geroscience 2024, 46, 3555–3566. [Google Scholar] [CrossRef] [PubMed]
- Lvov, I.; Grin, A.; Talypov, A.; Godkov, I.; Kordonskiy, A.; Khushnazarov, U.; Smirnov, V.; Krylov, V. The Impact of Odontoid Screw Fixation Techniques on Screw-Related Complications and Fusion Rates: A Systematic Review and Meta-Analysis. Eur. Spine J. 2021, 30, 475–497. [Google Scholar] [CrossRef]
- González-Vargas, P.M.; Caramés, J.; Riveiro, A.; Thenier-Villa, J.L.; Conde, C.; Pou, J. Influence of the Magnitude and Orientation of Forces on the Odontoid Fracture: A Finite Element Model Analyses. Neurocir. (Engl. Ed.) 2025, 36, 500675. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Dong, W.X.; Xu, J.Z.; Yuan, Z.S.; Sun, X.Y.; Zhu, B.K.; Lai, O.J.; Chen, X.G.; Zhao, B.J. Biomechanical study of different kinds of internal fixation for the typeI; Hangman fracture, type II; odontoid fracture and the C(2/3) disc injury: A finite-element analysis. Zhonghua Yi Xue Za Zhi 2018, 98, 2489–2495. [Google Scholar] [CrossRef]
- Shin, D.S.; Lee, K.; Kim, D. Biomechanical Study of Lumbar Spine with Dynamic Stabilization Device Using Finite Element Method. Comput.-Aided Des. 2007, 39, 559–567. [Google Scholar] [CrossRef]
- Li, Q.Y.; Kim, H.-J.; Son, J.; Kang, K.-T.; Chang, B.-S.; Lee, C.-K.; Seok, H.S.; Yeom, J.S. Biomechanical Analysis of Lumbar Decompression Surgery in Relation to Degenerative Changes in the Lumbar Spine–Validated Finite Element Analysis. Comput. Biol. Med. 2017, 89, 512–519. [Google Scholar] [CrossRef]
- Ivancic, P.C. Odontoid Fracture Biomechanics. Spine 2014, 39, E1403–E1410. [Google Scholar] [CrossRef]
- Eremina, G.; Smolin, A.; Xie, J.; Syrkashev, V. Development of a Computational Model of the Mechanical Behavior of the L4–L5 Lumbar Spine: Application to Disc Degeneration. Materials 2022, 15, 6684. [Google Scholar] [CrossRef] [PubMed]
- Muhayudin, N.A.; Basaruddin, K.S.; Ijaz, M.F.; Daud, R. Finite Element Modelling of a Synthetic Paediatric Spine for Biomechanical Investigation. Materials 2023, 16, 4514. [Google Scholar] [CrossRef]
- Graham, R.S.; Oberlander, E.K.; Stewart, J.E.; Griffiths, D.J. Validation and Use of a Finite Element Model of C-2 for Determination of Stress and Fracture Patterns of Anterior Odontoid Loads. J. Neurosurg. Spine 2000, 93, 117–125. [Google Scholar] [CrossRef]
- Guo, Y.; Chen, W.; Zhang, X.; Zhu, Y.; Zhao, Y. Biomechanical Analysis of Odontoid Fracture with Different Loads by Finite Element Method. In Proceedings of the 2nd International Conference on Bioinformatics and Biomedical Engineering; Association for Computing Machinery: New York, NY, USA, 2008; pp. 866–868. [Google Scholar]
- Güvercin, Y.; Yaylacı, M.; Dizdar, A.; Kanat, A.; Uzun Yaylacı, E.; Ay, S.; Abdioğlu, A.A.; Şen, A. Biomechanical Analysis of Odontoid and Transverse Atlantal Ligament in Humans with Ponticulus Posticus Variation under Different Loading Conditions: Finite Element Study. Injury 2022, 53, 3879–3886. [Google Scholar] [CrossRef]
- Li, L.; Liu, W.-F.; Jiang, H.-K.; Li, Y.-P. Biomechanical Evaluation of Four Different Posterior Screw and Rod Fixation Techniques for the Treatment of the Odontoid Fractures. Int. J. Clin. Exp. Med. 2015, 8, 8571–8580. [Google Scholar]
- Hu, Y.; Dong, W.-X.; Hann, S.; Yuan, Z.-S.; Sun, X.-Y.; Xie, H.; Zhang, M. Construction of Finite Element Model for an Artificial Atlanto-Odontoid Joint Replacement and Analysis of Its Biomechanical Properties. Turk. Neurosurg. 2016, 26, 430–436. [Google Scholar] [CrossRef]
- Liu, C.; Huang, K.; Dai, L.; Huang, X.; Zhang, X. The Biomechanical Study of Different Fixation Techniques for Combination Fractures of Atlas and Axis: A Finite Element Analysis. Eur. J. Med. Res. 2025, 30, 184. [Google Scholar] [CrossRef]
- Paksoy, K.; Avci, I.; Baskurt, O. Comparison of Mechanical Stress on Implants in Anterior Single and Double Screw Fixation of Odontoid Fractures in a Finite Element Model. Turk. Neurosurg. 2025, 35, 395–402. [Google Scholar] [CrossRef]
- Fedorov, A.; Beichel, R.; Kalpathy-Cramer, J.; Finet, J.; Fillion-Robin, J.-C.; Pujol, S.; Bauer, C.; Jennings, D.; Fennessy, F.; Sonka, M.; et al. 3D Slicer as an Image Computing Platform for the Quantitative Imaging Network. Magn. Reson. Imaging 2012, 30, 1323–1341. [Google Scholar] [CrossRef] [PubMed]
- Giorgio, I.; dell’Isola, F.; Andreaus, U.; Misra, A. An orthotropic continuum model with substructure evolution for describing bone remodeling: An interpretation of the primary mechanism behind Wolff’s law. Biomech. Model. Mechanobiol. 2023, 22, 2135–2152. [Google Scholar] [CrossRef] [PubMed]
- Allena, R.; Rémond, Y. How can theramechanics drive bone remodeling? Math. Mech. Complex Syst. 2025, 13, 55–71. [Google Scholar] [CrossRef]
- Shim, V.P.W.; Yang, L.M.; Liu, J.F.; Lee, V.S. Characterisation of the Dynamic Compressive Mechanical Properties of Cancellous Bone from the Human Cervical Spine. Int. J. Impact Eng. 2005, 32, 525–540. [Google Scholar] [CrossRef]
- Niinomi, M. Mechanical Properties of Biomedical Titanium Alloys. Mater. Sci. Eng. A 1998, 243, 231–236. [Google Scholar] [CrossRef]
- Lütjering, G.; Williams, J.C.; Gysler, A. Microstructure and Mechanical Properties of Titanium Alloys. In Microstructure And Properties of Materials: (Volume 2); World Scientific Publishing Company: Singapore, 2000; pp. 1–77. [Google Scholar]
- Doherty, B.J.; Heggeness, M.H.; Esses, S.I. A Biomechanical Study of Odontoid Fractures and Fracture Fixation. Spine 1993, 18, 178–184. [Google Scholar] [CrossRef] [PubMed]
- Dennison, E.; Eastell, R.; Fall, C.H.D.; Kellingray, S.; Wood, P.J.; Cooper, C. Determinants of Bone Loss in Elderly Men and Women: A Prospective Population-Based Study. Osteoporos. Int. 1999, 10, 384–391. [Google Scholar] [CrossRef]
- Nieves, J.W.; Formica, C.; Ruffing, J.; Zion, M.; Garrett, P.; Lindsay, R.; Cosman, F. Males Have Larger Skeletal Size and Bone Mass than Females, despite Comparable Body Size. J. Bone Miner. Res. 2005, 20, 529–535. [Google Scholar] [CrossRef]
- Julien, T.D.; Frankel, B.; Traynelis, V.C.; Ryken, T.C. Evidence-Based Analysis of Odontoid Fracture Management. Neurosurg. Focus. 2000, 8, e1. [Google Scholar] [CrossRef]
- Rehousek, P.; Jenner, E.; Holton, J.; Czyz, M.; Capek, L.; Henys, P.; Kulvajtova, M.; Krbec, M.; Skala-Rosenbaum, J. Biomechanical Comparison of Cemented versus Non-Cemented Anterior Screw Fixation in Type II Odontoid Fractures in the Elderly: A Cadaveric Study. Spine J. 2018, 18, 1888–1895. [Google Scholar] [CrossRef]
- Magee, W.; Hettwer, W.; Badra, M.; Bay, B.; Hart, R. Biomechanical Comparison of a Fully Threaded, Variable Pitch Screw and a Partially Threaded Lag Screw for Internal Fixation of Type II Dens Fractures. Spine 2007, 32, E475–E479. [Google Scholar] [CrossRef] [PubMed]
- Wright, N.M.; Lauryssen, C. Vertebral Artery Injury in C1-2 Transarticular Screw Fixation: Results of a Survey of the AANS/CNS Section on Disorders of the Spine and Peripheral Nerves. American Association of Neurological Surgeons/Congress of Neurological Surgeons. J. Neurosurg. 1998, 88, 634–640. [Google Scholar] [CrossRef]
- Panjabi, M.M.; Duranceau, J.; Goel, V.; Oxland, T.; Takata, K. Cervical Human Vertebrae. Quantitative Three-Dimensional Anatomy of the Middle and Lower Regions. Spine 1991, 16, 861–869. [Google Scholar] [CrossRef]
- Silva, M.J.; Keaveny, T.M.; Hayes, W.C. Load Sharing between the Shell and Centrum in the Lumbar Vertebral Body. Spine 1997, 22, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Fields, A.J.; Liebenberg, E.C.; Lotz, J.C. Innervation of Pathologies in the Lumbar Vertebral End Plate and Intervertebral Disc. Spine J. 2014, 14, 513–521. [Google Scholar] [CrossRef]
- Brolin, K.; Halldin, P. Development of a Finite Element Model of the Upper Cervical Spine and a Parameter Study of Ligament Characteristics. Spine 2004, 29, 376–385. [Google Scholar] [CrossRef]
- Zhang, Q.H.; Teo, E.C. Finite Element Application in Implant Research for Treatment of Lumbar Degenerative Disc Disease. Med. Eng. Phys. 2008, 30, 1246–1256. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.C.; Wilcox, R.K. Finite Element Analysis of the Spine: Towards a Framework of Verification, Validation and Sensitivity Analysis. Med. Eng. Phys. 2008, 30, 1287–1304. [Google Scholar] [CrossRef] [PubMed]
- Singhal, I.; Harinathan, B.; Warraich, A.; Purushothaman, Y.; Budde, M.D.; Yoganandan, N.; Vedantam, A. Finite Element Modeling of the Human Cervical Spinal Cord and Its Applications: A Systematic Review. N. Am. Spine Soc. J. 2023, 15, 100246. [Google Scholar] [CrossRef] [PubMed]











Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
González-Vargas, P.M.; Millán, A.; Thenier-Villa, J.L.; Badaoui, A.; Conde, C.; Pou, J.; Riveiro, A. Finite Element Analysis of Anterior Odontoid Screw Fixation for Type II Odontoid Fractures. Materials 2026, 19, 825. https://doi.org/10.3390/ma19040825
González-Vargas PM, Millán A, Thenier-Villa JL, Badaoui A, Conde C, Pou J, Riveiro A. Finite Element Analysis of Anterior Odontoid Screw Fixation for Type II Odontoid Fractures. Materials. 2026; 19(4):825. https://doi.org/10.3390/ma19040825
Chicago/Turabian StyleGonzález-Vargas, Pedro Miguel, Antía Millán, José Luis Thenier-Villa, Aida Badaoui, Cesáreo Conde, Juan Pou, and Antonio Riveiro. 2026. "Finite Element Analysis of Anterior Odontoid Screw Fixation for Type II Odontoid Fractures" Materials 19, no. 4: 825. https://doi.org/10.3390/ma19040825
APA StyleGonzález-Vargas, P. M., Millán, A., Thenier-Villa, J. L., Badaoui, A., Conde, C., Pou, J., & Riveiro, A. (2026). Finite Element Analysis of Anterior Odontoid Screw Fixation for Type II Odontoid Fractures. Materials, 19(4), 825. https://doi.org/10.3390/ma19040825








