1. Introduction
Cement is considered one of the most important construction materials worldwide. The cement production process accounts for approximately 5–8% of global carbon dioxide (CO
2) emissions [
1]. During production, gaseous particulates are released into the environment, which can reduce visibility, deteriorate air quality, and contaminate water resources. Workers in cement plants and nearby residents may be exposed to various health risks, particularly respiratory diseases. They can be exposed to nitrogen oxides (NO
x), sulfur dioxide (SO
2), carbon monoxide (CO), volatile organic compounds, ammonia, chlorine, and hydrogen chloride [
2]. High temperatures are required in rotary kilns for chemical reactions to occur, which are achieved through the consumption of substantial amounts of fossil fuels. In addition, processes such as raw material preparation and clinker grinding also consume significant amounts of energy [
3].
With the rapid urbanization process, increasing construction activities have led to a significant rise in cement production. However, cement production is associated with several adverse impacts, including high energy consumption, greenhouse gas emissions, and environmental pollution [
4]. In order to mitigate these environmental effects, research on alternative binder materials that can partially or completely replace cement has gained considerable momentum in recent years. In this context, geopolymers have emerged as a potential alternative to conventional cement due to their high compressive strength and superior durability [
5].
Various industrial by-products and wastes have been investigated as partial replacements for cement or aggregates in concrete and mortar production. Commonly used materials include fly ash, silica fume, ground granulated blast furnace slag, and natural stone powders. Natural stones such as marble, granite, and basalt are often processed into fine powders and used as fillers in mortars or concrete; this has been shown to improve particle packing and contribute to sustainable material use [
6]. In addition, the literature reports studies in which specific materials, such as used foundry sand, have been employed as fine aggregate substitutes [
7].
Irmak Er & Yazıcıoğlu [
8] investigated the feasibility of using granite waste sludge and sepiolite as partial cement replacement materials in self-compacting mortars. In the mortar mixes prepared with different replacement ratios, the fresh-state workability was evaluated, while the hardened mortars were comprehensively assessed in terms of mechanical strength, density, porosity, and water absorption. The findings indicate that the use of granite waste sludge and sepiolite at certain proportions can enhance the mechanical and durability properties of hardened mortars without adversely affecting the performance of fresh mortars. These results suggest that industrial by-products such as granite and sepiolite can be employed as sustainable and technically viable filler materials.
Benjeddou & Alwetaishi [
9] investigated the feasibility of using waste sludge from the marble processing industry, processed into fine powder, as a mineral filler material. The authors reported that the powder consists primarily of calcium carbonate, exhibits inert behavior, and does not participate in cement hydration. Physical and chemical characterization tests indicated that the powder is very fine and well-graded, while hydraulic tests and cement–powder blend experiments demonstrated that mechanical strength was not adversely affected. Consequently, marble sludge powder can be safely utilized in concrete and mortars as a partial cement replacement or filler, offering a sustainable and environmentally friendly material option.
Hameed et al. [
10] investigated the feasibility of using waste marble powder as an inert filler in self-compacting concrete. Mixes containing various dosages of marble powder were compared with a control mix without marble powder, and the fresh-state properties, including flow, viscosity, and passing ability, as well as the hardened-state properties, including compressive and flexural strengths, were evaluated. The study demonstrated that marble powder, up to certain dosages, maintained the fresh concrete performance and did not significantly compromise mechanical properties, indicating its potential as a sustainable inert filler material.
Karakurt & Dumangöz [
11] comprehensively investigated the effects of using marble powder and blast furnace slag as filler materials on the fresh and hardened properties of self-compacting concrete. The study holistically evaluated the rheological behavior and durability characteristics, revealing that the incorporation of approximately 10% marble powder and 30% blast furnace slag preserved the workability of fresh concrete while improving mechanical and durability properties. The findings indicate that these two waste materials can be considered sustainable and technically viable filler materials.
Prakash et al. [
12] examined the use of waste marble powder in cement-based composites. They replaced cement with marble powder at replacement levels of 0%, 5%, 10%, 20%, 30%, 40%, and 50%. The researchers analyzed water absorption, porosity, water permeability, compressive strength, splitting tensile strength, flexural strength, and microstructural properties. Their findings showed a slight increase in compressive strength in specimens containing up to 10% marble powder replacement.
Abouelnour et al. [
13] studied the individual and combined use of marble powder, granite powder, and nano-alumina powder as partial replacements for Portland cement. Marble and granite powders were used at replacement levels of 2%, 4%, 6%, 8%, and 10% by weight of cement, while nano-alumina powder was used at 0.25%, 0.5%, 0.75%, 1%, and 1.25%. The study evaluated the physical properties, workability, mechanical performance, and microstructural characteristics of the mixtures. The highest splitting tensile, flexural, and compressive strength values were obtained at 6% replacement for marble and granite powders and at 1% for nano-alumina powder.
Sevinç & Durgun [
14] produced mortar mixtures by partially replacing cement with barite, pumice, and basalt powders at replacement levels of 7.5% and 15%. The mortar specimens were subjected to elevated temperatures of 100, 200, 300, 400, 500, and 600 °C after being cured for 90 days. Two different cooling methods—air and water—were applied following the heating process. Before heating, the unit weight, ultrasonic pulse velocity, and compressive strength of the specimens were measured. After exposure to high temperatures, compressive strength, ultrasonic pulse velocity, and mass loss were determined. Additionally, TGA, SEM, and XRD analyses were performed. The results indicated that the specimens containing basalt were less affected by high temperatures compared to the others.
Huseien et al. [
15] investigated the use of epoxy resin polymer as a self-healing agent in Portland cement-based mortars. The mortars were prepared with different epoxy resin contents (5–20% by weight of cement), and the effects on mechanical performance and microstructure were evaluated. The experimental results showed that 10% epoxy resin addition significantly improved compressive, flexural, and tensile strengths, while also reducing porosity and water absorption. Higher resin contents led to stabilization of the mechanical properties. This study shows that the interaction of epoxy resin with cement hydration, coupled with the self-healing mechanism, improves both mechanical and durability performance of cementitious mortars, adding to the knowledge on polymer-modified cementitious systems.
Abuqasim et al. [
16] investigated the use of porcelain polishing residue (PPR), a fine powder generated during the surface polishing of porcelain tiles, as a partial cement replacement in Portland cement-based mortars. Mortars were prepared with different PPR contents (5–20% by weight of cement) and evaluated in terms of fresh properties, mechanical performance, and microstructure. The results showed that moderate PPR addition improved compressive, flexural, and tensile strengths, reduced porosity, and decreased water absorption, while higher replacement ratios stabilized the mechanical properties. The study demonstrated that PPR interacts with cement hydration to enhance the microstructure and durability of mortars, highlighting its potential as a sustainable, artificial plate filler in cementitious systems.
Bayraktar et al. [
17] investigated the mechanical and durability properties of sustainable cement-based foam concretes incorporating waste ceramic powder (CP) and waste polyester fibers. In the study, ceramic powder was used as a partial cement replacement at 0%, 10%, and 20%, while polyester fibers were added to the mixtures at volume fractions ranging from 0 to 0.6%. The experimental results showed that the use of 10–20% ceramic powder improved the pore structure through a micro-filler effect and positively influenced durability-related properties such as water absorption, whereas the polyester fibers contributed to crack control and enhanced ductility. The study demonstrated that the performance of cementitious systems can be optimized through the appropriate use of ceramic-based waste powders and polymer additives.
Nowadays, as an alternative to plates made from natural stones such as marble and granite, artificial plates are manufactured using various chemical- and mineral-based components. These materials are widely employed in kitchen and bathroom countertops, worktops, as well as wall and ceiling claddings. Due to their smooth surface texture, wide range of colors and patterns, antibacterial properties, high mechanical strength, and inconspicuous joints, the use of artificial plates has been steadily increasing in recent years [
18].
Artificial plates, commonly referred to as engineered stone materials, differ fundamentally from natural stones in both composition and production process. While natural marble and granite consist solely of mineral phases, engineered stone plates are manufactured by binding mineral aggregates with polymeric resins. This resin-bonded composite structure results in distinct physical, mechanical, and thermal behaviors compared to natural stone materials. In particular, the presence of organic binder phases influences particle characteristics, interaction with cementitious matrices, and degradation mechanisms under elevated temperatures. Therefore, artificial plate waste powders should be evaluated separately from natural stone powders when considering their use as filler materials in cement-based composites.
Similar to natural stone plates, the processing of artificial plates also generates production waste that cannot be reused. These wastes are mostly disposed of in landfill sites alongside other stone plate residues. However, the environmental impact of these artificial plate wastes, which are produced entirely from chemical components and resins, is considerably higher compared to that of natural stone waste.
Therefore, in order to both reduce environmental pollution and recover economic value from these wastes, it is of great importance to incorporate artificial plate waste into production processes through reuse or recycling methods. This approach aligns with sustainable production goals and contributes to increasing material efficiency in the construction sector. Based on the reviewed studies,
Table 1 presents a comparative overview of the use of natural and engineered stone powders as cement replacement materials in SCM and SCC.
However, despite extensive studies on natural stone powders, limited research has focused on resin-containing artificial plate wastes, particularly regarding their behavior in self-compacting mortars under elevated temperatures. Therefore, this study aims to evaluate the feasibility of using artificial plate waste powders as sustainable filler materials in self-compacting mortars. Waste cultured marble (WMP) and quartz-based composite plate powders (WQP) were used as partial cement replacements at various replacement levels, and their effects on fresh properties, mechanical performance, durability, and microstructural characteristics were systematically investigated. In addition, selected specimens were exposed to elevated temperatures of 300 °C, 600 °C, and 900 °C to assess the thermal stability and microstructural evolution of mortars containing resin-bonded artificial plate powders.
3. Results
3.1. Fresh Properties Results
The EFNARC committee recommends that the mini slump flow of self-compacting mortars should fall within the range of 240–260 mm. Since the water content was maintained constant across all mixtures, achieving flow diameters within the specified range was accomplished through the use of varying dosages of high-performance water-reducing admixture (HRWR). As shown in
Table 2, the required HRWR dosage increased with higher replacement ratios. The flow diameters of the fresh mortars are presented in
Figure 9. Based on the data in
Table 2 and
Figure 9, it can be concluded that as the replacement ratio increases, the water demand rises and the workability of the mortars decreases.
The unit weight of mortars decreased with increasing replacement ratios, primarily due to the reduction in high-density cement content. Cement particles possess significantly higher density compared to the replacement materials, and their binding properties help reduce porosity in the matrix. Substituting cement with non-binding materials increases void content, making the observed decrease in unit weight an expected outcome.
Interestingly, mortars prepared with WQP, despite its lower particle density, exhibited higher unit weight than those prepared with WMP. This can be attributed to differences in particle morphology and surface characteristics. WQP particles are smoother and more hydrophobic, which limits water distribution, reduces air entrapment, and promotes denser packing. In contrast, WMP is calcite-based with rougher surfaces, favoring air entrapment and lower unit weight.
3.2. Porosity and Bulk Specific Gravity Test Results
Porosity is a critical parameter that directly influences the durability, permeability, and overall performance of mortar. In general, lower porosity indicates a denser microstructure and consequently improved mechanical properties and durability.
As the replacement ratio increases, the porosity of the mortars increases, while their bulk specific gravity decreases. This trend is primarily attributed to the substitution of high-density cement particles with lower-density powders, which reduces the total amount of solid material per unit volume. Additionally, the increase in replacement ratio leads to a higher number of voids within the mortar. The particle shape and surface roughness of the powders affect the distribution of water within the matrix, thereby contributing to the observed trends in porosity and bulk specific gravity. The porosity and bulk specific gravity results of QP and MP specimens are presented in
Figure 10.
3.3. Sorptivity Test Results
Sorptivity is a crucial durability parameter that characterizes the rate at which a material absorbs water through capillary suction. Due to their superior workability, self-compacting mortars have the potential to develop a denser and more homogeneous microstructure compared to conventional mortars. This theoretically results in lower porosity and permeability. However, any degradation in the microstructure or an increase in the capillary pore system can adversely affect the expected superior durability performance of self-compacting mortars.
The sorptivity test results for 28-day cured prism mortar specimens are presented in
Figure 11. Among all groups, REF exhibited the lowest mass/area ratio, indicating the lowest capillary water absorption rate and therefore the highest resistance to external water ingress. As the WP replacement ratio increased, the capillary water absorption curves deviated further from that of the control mix, reaching higher mass/area ratios. This indicates that the incorporation of WP significantly increases both the capacity and rate of capillary water absorption in the mortars.
3.4. Flexural Strength Test Results
Figure 12a presents the flexural strength results at 7, 28, and 56 days. Both WQP and WMP exhibit favorable performance at low replacement levels in terms of flexural strength. However, higher replacement ratios generally lead to a reduction in flexural strength, with this decrease being more pronounced in the QP series specimens. At low replacement levels, the waste powders enhance particle packing density by filling the fine pores within the mortar matrix through their micro-filling effect. This reduces the void volume between cement particles, resulting in a more compact microstructure and more efficient stress transfer. The powders may also impart slight flexibility to the matrix, reducing brittleness and contributing to the observed strength increase.
At higher replacement ratios, however, the reduction in cement content leads to a decrease in binding capacity, a reduction in the quantity of cement hydration products, and the formation of a greater number of voids within the mortar structure. These factors collectively contribute to the observed decline in flexural strength.
The results clearly indicate that there exists an optimal replacement range for both waste materials, beyond which flexural strength begins to decrease. This optimal level is approximately 10% for WQP and may reach up to 15% for WMP.
The temperature-dependent flexural strength results are presented in
Figure 12b. For the REF and QP5 specimens, a slight increase in flexural strength was observed at 300 °C compared to ambient temperature. This enhancement can be attributed to the partial release of bound water at lower temperatures, which may promote the partial closure of micro-voids and result in a denser matrix. Similar trends have been reported in the literature, where certain modified mortars demonstrated increased flexural strength around 300 °C due to filler effects and improved matrix integrity [
26]. Nevertheless, for both specimen groups, an overall reduction in flexural strength was noted with increasing replacement ratio and exposure temperature.
Examination of
Figure 13 indicates a moderate correlation between both porosity and flexural strength, as well as replacement ratio and flexural strength. Second-order polynomial regressions were applied to both relationships, with R
2 values of 0.59 and 0.62 for flexural strength versus porosity and replacement ratio, respectively, indicating moderate correlations. These findings suggest that while porosity and replacement ratio influence strength to a certain extent, the variation in strength is largely governed by other factors. The incorporation of inert mineral powders can limit cement hydration, thereby reducing the formation of binding phases. Although porosity exhibits certain trends, the particle characteristics, binding capacity, and microstructural interactions of the replacement materials play a more dominant role in determining flexural strength. Furthermore, the filler effect of the mineral powders and their interaction with the matrix contribute to the nonlinearity of the relationship between replacement ratio and strength.
3.5. Compressive Strength Test Results
The average compressive strengths of the mortar specimens at 7, 28, and 56 days are presented in
Figure 14a. In all mortar mixes, compressive strength generally increased with curing time, indicating the continued hydration of cement and the progressive development of strength. The highest compressive strength was observed in the control specimen. As the replacement ratio increased, the compressive strength generally decreased in both the QP and MP groups. However, at each replacement level, the MP specimens exhibited lower strength compared to the corresponding QP specimens, indicating a more favorable performance of the QP group.
The effect of elevated temperature on compressive strength is shown in
Figure 14b. In all mixes, increasing the replacement ratio led to a significant decrease in compressive strength at elevated temperatures. Nevertheless, a temporary increase in compressive strength was observed at 300 °C, similar to the trend seen in flexural strength for REF, QP5, QP10, QP15, and QP20 specimens.
In contrast, the compressive strength of the MP specimens continuously decreased with increasing temperature. This behavior is associated with the loss of stability of the mineral structure of cultured marble at high temperatures and the decomposition of CaCO
3, which accelerates the formation of microcracks and weakens the matrix integrity, as reported in the literature [
27]. Additionally, at all temperatures, WQP-containing specimens exhibited higher compressive strength than WMP-containing specimens, indicating that WQP provides a more effective filling and binding enhancement within the mortar matrix.
Examination of
Figure 15 indicates a moderate-to-strong correlation between compressive strength and porosity, with an
, and a similar effect observed for the relationship between replacement ratio and compressive strength, with an
. Although the incorporated WP limits cement hydration, variations in porosity and replacement ratio partially account for the observed changes in compressive strength. Furthermore, particle size, filler effect, and microstructural interactions of the mineral powders emerge as additional critical factors influencing strength.
3.6. Abrasion Resistance Test Results
The abrasion mass loss results after exposure to AT, 300, 600, and 900 °C are presented in
Figure 16. In general, abrasion tests conducted under AT conditions showed that mass loss increased with higher replacement ratios in both WQP- and WMP-containing groups. At 300 °C, some specimens exhibited a slight decrease in mass loss, indicating improved wear resistance at this temperature.
At 600 °C and 900 °C, specimens containing WMP exhibited lower wear losses compared to those incorporating WQP. The higher wear rate of WQP at these temperatures may be related to microstructural changes in quartz grains, as reported in previous studies [
28]. Exposure to high temperatures compromised the integrity of the mortar matrix and increased porosity, contributing to the observed increase in abrasion losses. At 900 °C, a sharp increase in wear losses was observed in all specimens.
The general trends of the experimental results obtained in this study relative to the reference mixture are summarized in
Table 5.
3.7. Microstructure Analysis Results
The SEM analysis results of REF specimens subjected to different temperatures are presented below.
The SEM images of REF specimens exposed to different temperatures are presented in
Figure 17. At ambient temperature, the specimen exhibited a compact microstructure with limited porosity and a homogeneous distribution of C–S–H-based hydration products. The aggregate–paste interface remained intact, and a typical self-compacted mortar formation was observed. At 300 °C, the matrix showed signs of degradation; the loss of hydration water led to the formation of fine microcracks and superficial disintegration, while partial decomposition of the C–S–H phase was also observed. Upon exposure to 600 °C, the microcracks widened and significant decomposition of the C–S–H phase occurred, resulting in a looser and more granular binder paste. At 900 °C, the matrix was severely decomposed, porosity increased, and the microstructure lost its ductility, exhibiting a glassy and brittle appearance.
The SEM images of the QP25 specimens exposed to different temperatures are presented in
Figure 18. At ambient temperature, the WQP-containing specimen exhibits a compact and interconnected C–S–H network with dense needle-like ettringite crystals, and the fine-grained mineral additive contributes to reduced porosity. At 300 °C, matrix degradation is observed; however, the matrix integrity is comparatively better preserved than in the control specimen, indicating that the void-filling effect of WQP enhances structural stability. At 600 °C, matrix degradation remains limited. Although dehydration-induced cracks are present, the matrix is still partially preserved, with WQP contributing to maintaining structural stability. At 900 °C, matrix degradation becomes more pronounced, but porosity and overall matrix decomposition are less severe than in the control specimen. Overall, WQP incorporation exerts a partial yet significant positive effect on high-temperature performance.
The SEM analysis results of WMP-containing specimens at different temperatures are presented below.
The SEM images of the MP25 specimens exposed to different temperatures are presented in
Figure 19. At ambient temperature, the WMP-containing specimen exhibits a compact structure; however, its matrix morphology is more irregular and heterogeneous compared to the WQP-substituted specimen. The filler effect contributes to a reduction in porosity. At 300 °C, matrix degradation is more pronounced than in the WQP-substituted specimen, indicating that the mineralogical composition of WMP limits its thermal stability. At 600 °C, matrix degradation accelerates, the matrix becomes looser, and the pore connectivity increases. Although WMP provides limited improvement compared to the control specimen, it remains less effective than the WQP-substituted specimen. At 900 °C, severe matrix degradation and high porosity are observed. Overall, WMP incorporation has a limited effect on high-temperature performance and does not preserve matrix integrity as effectively as WQP-substituted specimens.
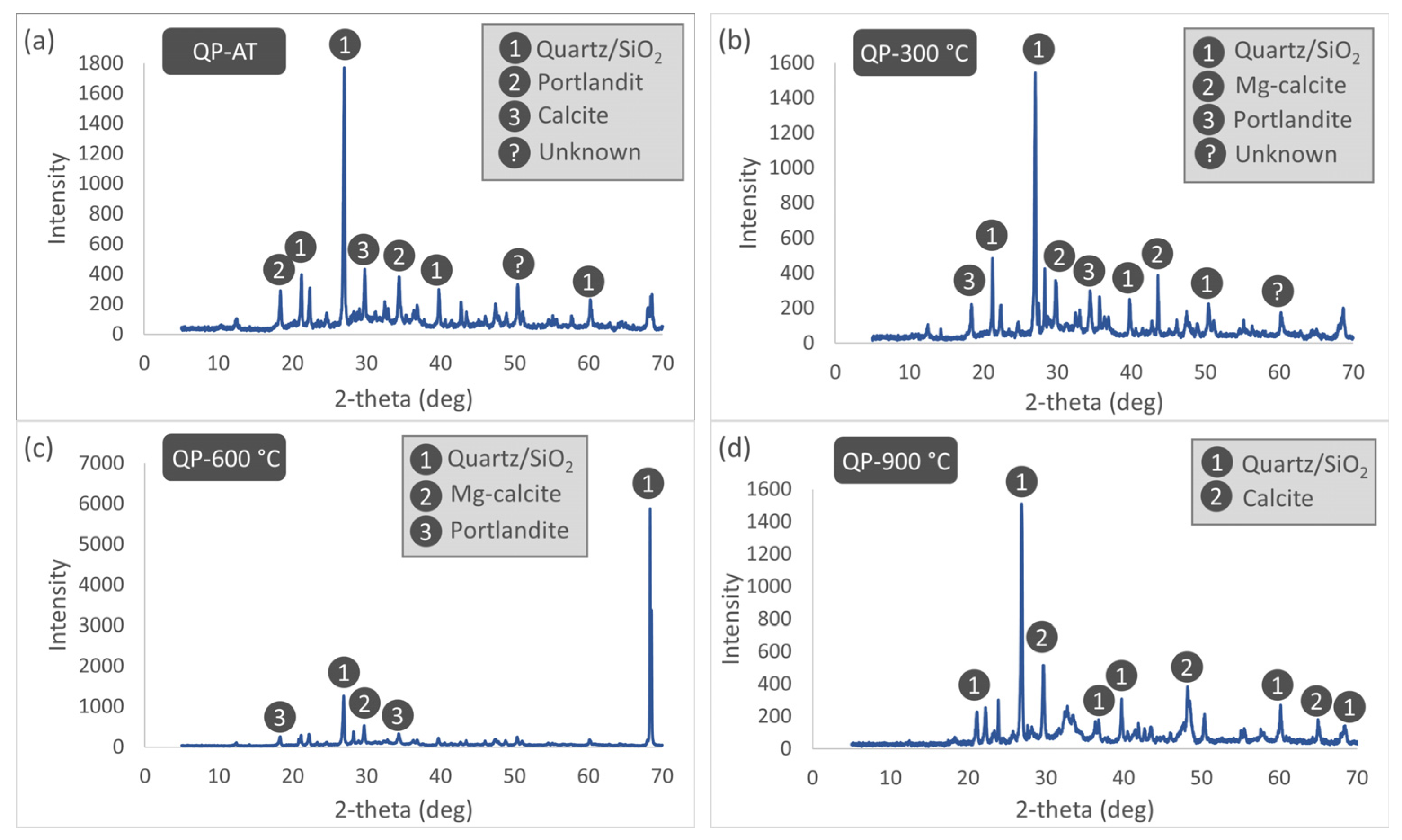
The XRD patterns of the REF specimens subjected to different thermal treatment temperatures are presented in
Figure 20. XRD analyses of the REF specimens under AT conditions show distinct diffraction peaks corresponding to quartz, calcite, portlandite, and kaolinite. When the temperature is increased to 300 °C, the kaolinite peak disappears, whereas a pronounced increase in the intensity of the calcite peak is observed. This increase may be attributed to enhanced crystallinity resulting from extensive dehydration within the mortar matrix or to the acceleration of carbonation reactions under elevated temperature. At 600 °C, the calcite phase within the cement matrix undergoes substantial degradation or becomes completely depleted due to progressive thermal decomposition; at the same temperature, the portlandite peak intensity also decreases markedly and tends toward disappearance. Within this temperature range, the high-temperature polymorph of the clay mineral, nacrite, becomes evident, while the quartz phase retains its structural stability. Finally, at 900 °C, the complete decomposition of portlandite and other hydrated phases results in a mortar matrix composed predominantly of the thermally more stable quartz and calcite phases. The presence of calcite at this temperature—despite its expected calcination—can be explained by the recarbonation of calcium oxide formed during heating through reaction with atmospheric carbon dioxide during the cooling stage.
The XRD patterns of the QP25 specimens subjected to different thermal treatment temperatures are presented in
Figure 21. XRD analyses of the 25% WQP-substituted specimens under AT conditions reveal the presence of quartz derived from sand and the replacement material, alongside portlandite as a cement hydration product and carbonate-based calcite. When the temperature is increased to 300 °C, extensive dehydration of the mortar begins, resulting in the disappearance of the calcite phase from the diffraction pattern, while portlandite remains present. Additionally, Mg-calcite phases, which are not observed under AT conditions, are detected at this temperature. Upon reaching 600 °C, a pronounced increase in the intensity of the quartz peaks is observed compared to lower temperatures. This significant increase can be attributed to the accelerated calcination of thermally unstable hydrated phases and carbonates, which renders the relatively thermally stable quartz phase dominant in the X-ray diffraction pattern. At this temperature, pure calcite is no longer observed, whereas portlandite and Mg-calcite phases are still detected. At 900 °C, thermal decomposition and phase transformations are largely complete, and the matrix is dominated by quartz and the reformed calcite phase. The reappearance of calcite at this temperature is explained by the recarbonation of calcium oxide, formed at high temperatures, through reaction with atmospheric carbon dioxide during cooling. Overall, WQP substitution enhances the thermal stability of the quartz phase, while the decomposition and reformation behavior of the calcite and Mg-calcite phases exhibits complex temperature-dependent variations.
The XRD patterns of the MP25 specimens subjected to different thermal treatment temperatures are presented in
Figure 22. The XRD analysis of the 25% WMP-substituted mortar at room temperature revealed the presence of quartz originating from the aggregates, portlandite as a cement hydration product, and calcite and kaolinite phases contributed by the WMP substitution and cement additives. Upon heating to 300 °C, intense dehydration of the mortar was observed, leading to the disappearance of the kaolinite phase, while brownmillerite and dolomite phases became pronounced due to high-temperature effects. At 600 °C, although the decomposition of thermally unstable hydrated phases had begun, portlandite, calcite, and kaolinite were still detectable, suggesting that the matrix containing WMP partially delayed the thermal phase transformations. At 900 °C, despite the substantial completion of thermal decomposition, the mortar matrix was dominated by thermally stable aggregate-derived quartz and calcite phases exhibiting high peak intensities. The persistence of calcite as a dominant phase at this elevated temperature may be related to the partial suppression of calcination caused by the release of local CO
2 from organic components within WMP and to the re-carbonation of CaO formed during high-temperature exposure upon cooling. Moreover, the decomposition of other thermally unstable phases at elevated temperatures contributed to the relative dominance of calcite in the XRD patterns.
In general, the use of WQP and WMP influences the microstructure of self-compacting mortars through particle characteristics and filler effects. SEM analyses show that mortars containing WQP exhibit a denser and more homogeneous matrix with fewer microcracks and lower pore connectivity, whereas WMP-containing mortars, particularly at higher replacement levels, display more microcracks and larger pores. XRD results further confirm that the high-temperature behavior is largely determined by the thermal stability of the dominant mineral phases: WQP forms a quartz-rich framework that remains stable at elevated temperatures, while WMP preserves partial matrix integrity due to the slower decomposition of calcite. These observations explain the differences in mechanical performance and durability between the two types of mortars.
4. Conclusions
This study evaluates the potential use of waste powders derived from cultured marble and quartz-based composite plates as partial cement replacements in self-compacting mortars. The effects of these powders on fresh properties, mechanical performance, durability, and microstructural characteristics were systematically investigated, providing mechanistic insights into how filler type and replacement level influence mortar behavior. The findings offer practical guidelines for sustainable SCM production and highlight the contribution of industrial waste materials to environmental and construction sustainability.
The increase in the replacement ratio generally led to higher water demand and reduced workability. Mortars containing WQP exhibited a higher unit weight compared to those containing WMP. This is thought to result from the particle morphology, as observed in SEM images, with WMP particles being more rounded and WQP particles being angular and irregular. The filler efficiency and particle distribution provided by the angular morphology of WQP contributed to a denser and more homogeneous mortar matrix. Porosity remained slightly above the control at low to medium replacement levels (up to ~15%) but increased more noticeably at higher replacement levels. Sorptivity values were higher than the control for all replacement ratios, without a consistent trend with increasing replacement.
Flexural strength showed a slight improvement at low replacement levels due to the filler effect, with performance comparable to the control maintained up to 10% for both WQP and WMP. Beyond this replacement ratio, flexural strength generally decreased. Compressive strength decreased with increasing replacement levels; however, mixtures containing WQP consistently exhibited higher compressive strength than those containing WMP, indicating a more effective filler contribution of WQP. Although compressive strength values remained lower than the control mixture, replacement levels up to 10% for both WQP and WMP maintained acceptable and consistent strength across curing ages and exposure temperatures. Abrasion resistance generally declined with increasing replacement ratios; nevertheless, mixtures with low replacement levels, particularly up to 10% for both WQP and WMP, exhibited abrasion mass losses comparable to or lower than the control, which can be attributed to the micro-filler effect.
SEM analyses revealed that WQP-containing mortars had a more tightly packed and homogeneous microstructure with fewer microcracks and reduced pore connectivity, whereas WMP-containing mortars exhibited higher microcrack density and larger pores, particularly at higher replacement levels. XRD results showed that the high-temperature performance of the mortars was largely determined by the thermal stability of the dominant mineral phases: WQP contributed to a quartz-rich framework that remained stable at elevated temperatures, while WMP maintained partial matrix integrity due to the slower decomposition of calcite, which helped to limit matrix deterioration.
At low replacement levels, the use of WQP and WMP exhibited acceptable performance in terms of compressive strength, flexural strength, porosity, water absorption, and abrasion resistance. These mixtures, at replacement levels up to 10% for both WQP and WMP, are suitable for non-structural applications or areas exposed to normal environmental conditions, providing a practical guideline for construction use. Utilizing these industrial waste powders as replacement materials can contribute to minimizing the environmental impact of cement. Furthermore, incorporating these wastes into mortar production provides a sustainable solution for waste management and helps reduce environmental pollution. It also provides significant potential for producing eco-friendly construction materials.