From the Problem of Corrosion to Green Solutions: The Role of Biosurfactants as Anti-Corrosion Agents
Abstract
1. Introduction
2. Corrosion
2.1. Mechanisms of Occurrence
2.1.1. Electrochemical Corrosion
2.1.2. Chemical Corrosion
2.2. Corrosion Mechanisms
2.2.1. Erosion Corrosion
2.2.2. Stress Corrosion Cracking (SSC)
2.3. Morphology or Appearance of Corrosion Type
2.3.1. Uniform Corrosion
2.3.2. Bimetallic or Galvanic Corrosion
2.3.3. Pitting Corrosion (PC)
2.3.4. Crevice Corrosion
- (1)
- Oxidation: M → Mn+ + n e−
- (2)
- Reduction: O2 + 2 H2O + 4 e− → 4 OH−
- (3)
- Mn+ + H2O → MOH(n−1)+ + H+
2.3.5. Intergranular Corrosion (IGC)
2.4. Industrial Segments and Corrosion Process
2.4.1. Petroleum, Gas and Petrochemical Sectors
2.4.2. Shipbuilding and Maritime Industry
2.4.3. Power Generation Industry (Solar, Nuclear and Wind)
2.4.4. Construction Industry
2.5. Corrosion Inhibitors
2.5.1. Organic Corrosion Inhibitors
2.5.2. Inorganic Corrosion Inhibitors
2.5.3. Green Corrosion Inhibitors
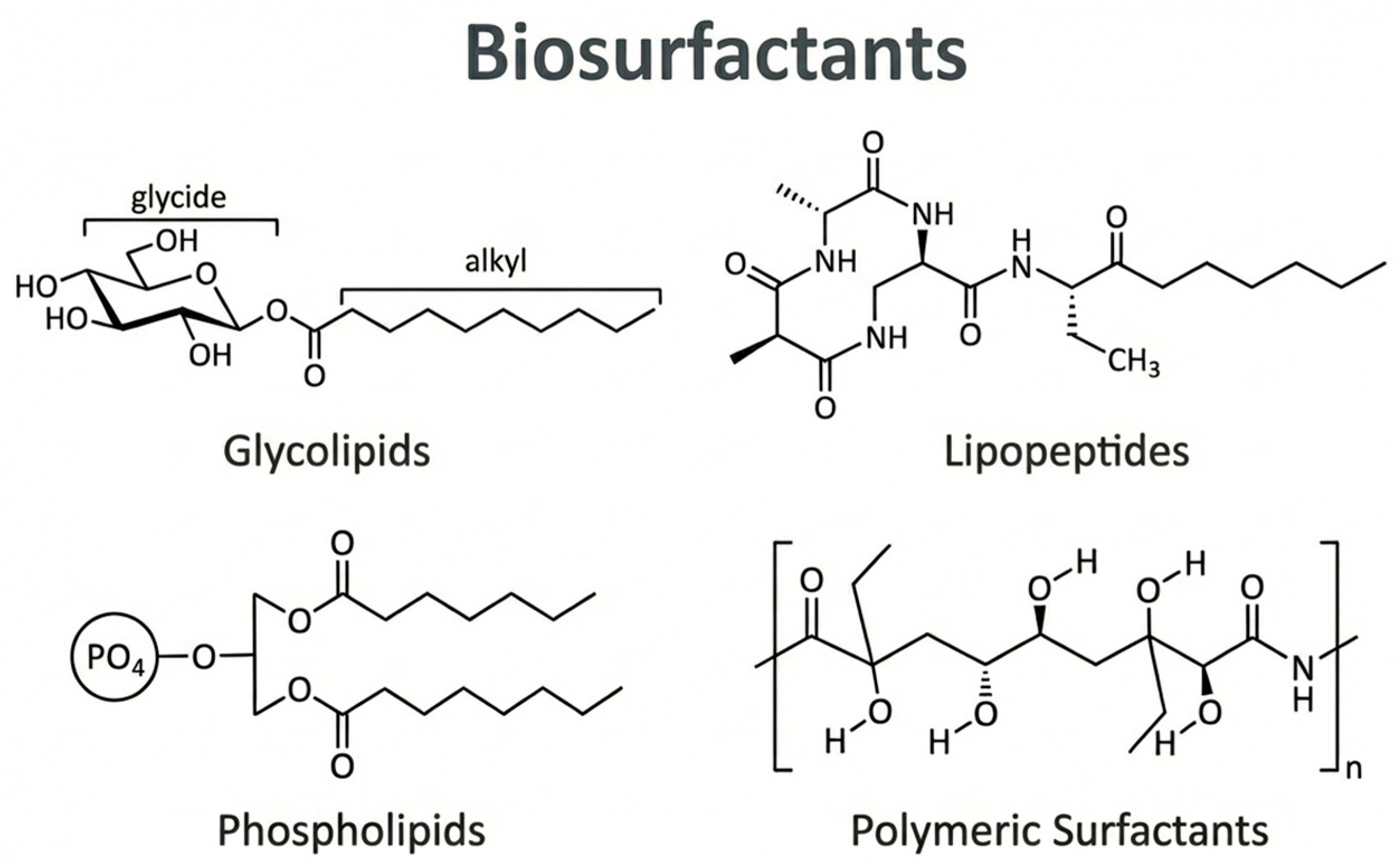
3. Biosurfactants
3.1. Classification of Biosurfactants
3.1.1. Glycolipids
3.1.2. Lipopeptides
3.1.3. Phospholipids
3.1.4. Polymeric Biosurfactants
3.2. Properties of Biosurfactants
- Reduction in Surface Tension (ST): The ability to form micelles, i.e., clusters of amphiphilic molecules, is the main property of BS. A progressive increase in BS concentration reduces ST to a minimum value at which micelles form [203,204]. Thanks to their amphipathic structure, BS enable adsorption at the liquid interface, resulting in the formation of a monolayer that, reducing the molecular attraction between liquids, acts as a thin, elastic membrane; therefore, ST is reduced. BS can then lower ST between two substances, e.g., a gas and a liquid (surface tension of water), two liquids (water and oil), or a liquid and a solid (water and dirt particles) [205].
- Biodegradation is a removal mechanism that can be limited by low bioavailability of hydrophobic contaminants, such as petroleum hydrocarbons. However, bacteria can produce BS that can improve the bioremediation of hydrophobic compounds. A typical example is a microbial consortium that produced a sophorolipid that improved the bioaccessibility of petroleum hydrocarbons for bacterial degradation [206].
- Emulsification is a stable heterogenous disequilibrium process involving an immiscible liquid dispersed, in the form of small droplets, within another liquid. In agriculture, cosmetics, and pharmaceutical sectors, BS are usually employed as emulsifiers. Their emulsifying activity, which depends on substrate, allows their use to enhance bioremediation and remove petroleum from contaminated sites [207,208]. It is typically determined over time (e.g., 24, 96 and 168 h) to assess emulsion stability. The ratio of emulsion height to total liquid height is used as the emulsion index [209].
- Anti-biofilm activity: The use of BS as an alternative to prevent biofilm formation has been largely studied. The wettability and amphiphilic properties of BS enable them to affect corrosion by acting on the adhesivity of microbial cells. The biosurfactant lichenysin from Bacillus licheniformis demonstrated anti-biofilm activity either before or after treatment. Another example is provided by rhamnolipids, which exerted an anti-adhesive and anti-biofilm effect against Gram-positive and Gram-negative bacteria, inhibiting biofilm formation on polystyrene and stainless-steel surfaces [210].
- Antimicrobial action: Biosurfactants demonstrate antibacterial effects by disrupting cell membranes. Unlike traditional antibiotics, BS utilize alternative mechanisms to eliminate target microorganisms [211]. Green surfactants are notable for being low in toxicity and highly biodegradable, primarily because they are made up of simple sugars, fatty acids or polypeptides. Such features guarantee the safety of drug formulations, lower the risk of undesired side effects, and maintain the efficacy of bioactive compounds [212].
- Dispersion happens when the cohesive force among similar particles is reduced. Small quantities of dispersants like BS can be added to a suspension to avoid insoluble particle aggregation. For instance, BS can be used to remove lipophilic compounds from rock surface, making them more motile and facilitating their recover during petroleum extraction. Dispersion also plays a key role in diminishing or entirely suppressing the formation of undesired microbe biofilms [213].
- pH and temperature have little influence on the interface activity of most BS. The activity of lichenysin from B. licheniformis JF-2 was not affected in the pH range of 4.5 to 9.0 and at temperatures up to 50 °C. Likewise, a Bacillus subtilis LB5a lipopeptide kept its emulsifying activity for half a year at 121 °C, pH 4–10 and high salinity [10% (w/v) NaCl] [214].
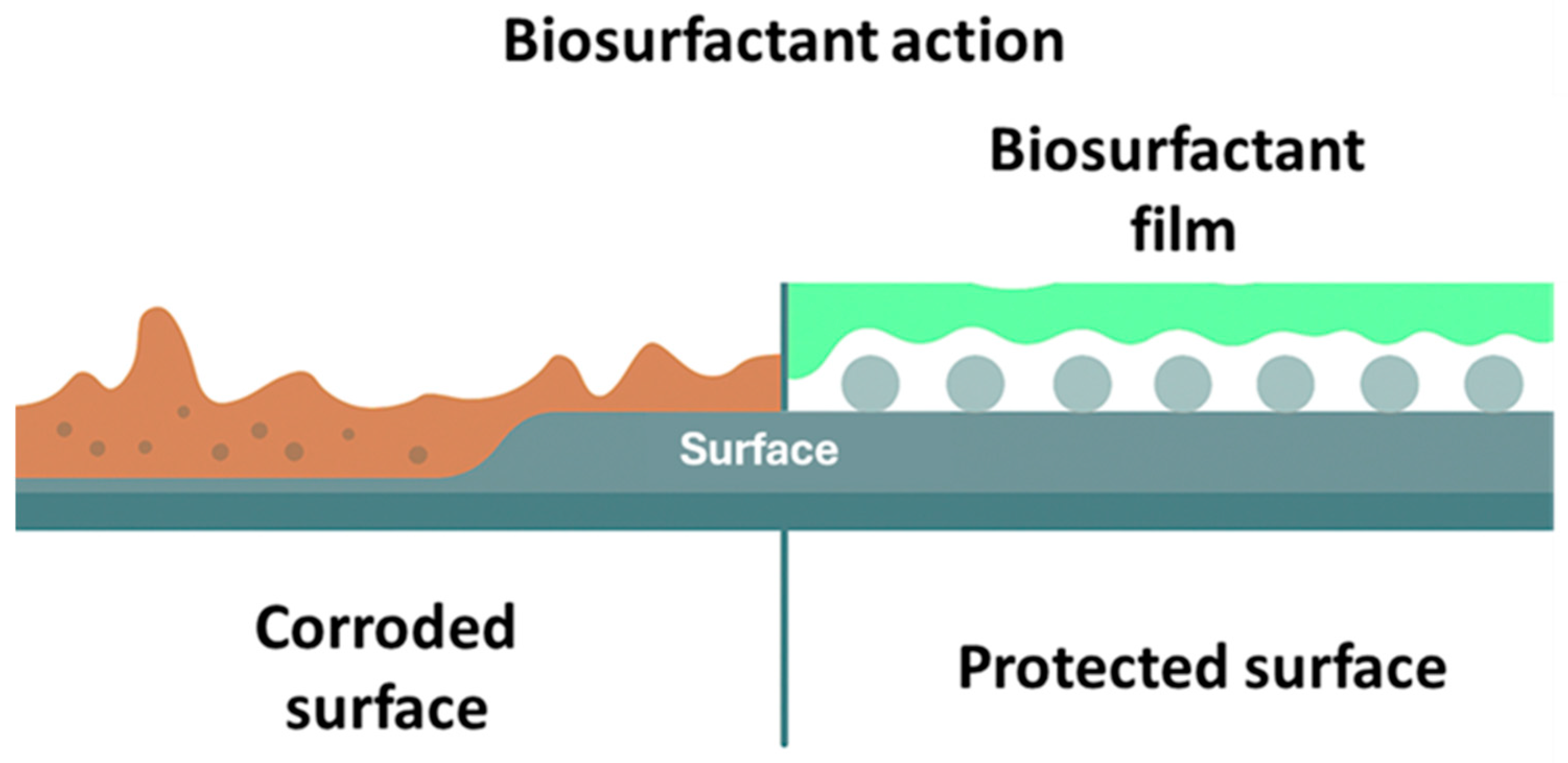
Biosurfactants as Corrosion Inhibitors
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Jimoh, A.A.; Booysen, E.; Van Zyl, L.; Trindade, M. Do biosurfactants as anti-biofilm agents have a future in industrial water systems? Front. Bioeng. Biotechnol. 2023, 11, 1244595. [Google Scholar] [CrossRef]
- Shwetha, K.M.; Praveen, B.M.; Devendra, B.K. A review on corrosion inhibitors: Types, mechanisms, electrochemical analysis, corrosion rate and efficiency of corrosion inhibitors on mild steel in an acidic environment. Results Surf. Interfaces 2024, 16, 100258. [Google Scholar] [CrossRef]
- Al-Moubaraki, A.H.; Obot, I.B. Corrosion challenges in petroleum refinery operations: Sources, mechanisms, mitigation, and future outlook. J. Saudi Chem. Soc. 2021, 25, 101370. [Google Scholar] [CrossRef]
- Selva Filho, A.A.P.; Faccioli, Y.E.S.; Converti, A.; Casazza, A.A.; Soares da Silva, R.d.C.F.; Sarubbo, L.A. The Application of a New Microbial Biosurfactant to Remove Residual Oil from Electric Power Plant and to Inhibit Metal Corrosion in a Salty Environment. Energies 2025, 18, 3359. [Google Scholar] [CrossRef]
- Akpan, E.D.; Singh, A.K.; Lgaz, H.; Quadri, T.W.; Shukla, S.K.; Mangla, B.; Ebenso, E.E. Coordination compounds as corrosion inhibitors of metals: A review. Coord. Chem. Rev. 2024, 499, 215503. [Google Scholar] [CrossRef]
- Aslam, M.; Rani, A.; Singh, R.; Nand, B.; Verma, C.; AlFantazi, A.; Singh, P. Harnessing deep eutectic solvents for advanced corrosion protection: A review. J. Mol. Liq. 2025, 422, 126919. [Google Scholar] [CrossRef]
- Aslam, R.; Mobin, M.; Zehra, S.; Aslam, J. A comprehensive review of corrosion inhibitors employed to mitigate stainless steel corrosion in different environments. J. Mol. Liq. 2022, 364, 119992. [Google Scholar] [CrossRef]
- Verma, C.; Ebenso, E.; Abdellaziz, B.; Hussain, C.M. Recent developments in sustainable corrosion inhibitors: Design, performance, and industrial scale applications. Mater. Adv. 2021, 2, 3806–3850. [Google Scholar] [CrossRef]
- Yang, L. 1—Introduction. In Techniques for Corrosion Monitoring; Yang, L., Ed.; Woodhead Publishing: Sawston, UK, 2008; pp. 1–5. [Google Scholar] [CrossRef]
- Koch, G.; Varney, J.; Thompson, N.; Moghissi, O.; Gould, M.; Payer, J. International Measures of Prevention, Application, and Economics of Corrosion Technologies (IMPACT) Study; NACE International: Houston, TX, USA, 2016; Available online: https://www.zerust.com/wp-content/uploads/2018/10/Nace-International-Report.pdf (accessed on 28 January 2026).
- Wang, D.; Liu, J.; Jia, R.; Dou, W.; Kumseranee, S.; Punpruk, S.; Gu, T. Distinguishing two different microbiologically influenced corrosion (MIC) mechanisms using an electron mediator and hydrogen evolution detection. Corros. Sci. 2020, 177, 108993. [Google Scholar] [CrossRef]
- Gu, T.; Wang, D.; Lekbach, Y.; Xu, D. Extracellular electron transfer in microbial biocorrosion. Curr. Opin. Electrochem. 2021, 29, 100763. [Google Scholar] [CrossRef]
- Bender, R.; Féron, D.; Mills, D.; Ritter, S.; Bäßler, R.; Bettge, D.; Zheludkevich, M. Corrosion challenges towards a sustainable society. Mater. Corros. 2022, 73, 1730–1751. [Google Scholar] [CrossRef]
- Belkheiri, A.; Dahmani, K.; Khattabi, M.; Mzioud, K.; Kharbouch, O.; Galai, M.; Touhami, M.E. Evaluation of 14-(p-tolyl)-14H-dibenzo[a,j]xanthene as a highly efficient organic corrosion inhibitor for mild steel in 1 M HCl: Electrochemical, theoretical, and surface characterization. Int. J. Electrochem. Sci. 2024, 19, 100873. [Google Scholar] [CrossRef]
- Sabiha, M.; Kerroum, Y.; El Hawary, M.; Boudalia, M.; Bellaouchou, A.; Hammani, O.; Amin, H.M. Investigating the adsorption and corrosion protection efficacy and mechanism of marjoram extract on mild steel in HCl medium. Molecules 2025, 30, 272. [Google Scholar] [CrossRef]
- Yeganeh, M.; Heydarie, Y.; Shahryari, Z.; Asadi, N.; Kahkesh, H.; Ramezanalizadeh, H. Corrosion inhibitor of API pipeline steels: A review. J. Bio-Tribo-Corros. 2025, 11, 44. [Google Scholar] [CrossRef]
- Guzmán, E.; Ortega, F.; Rubio, R.G. Exploring the world of rhamnolipids: A critical review of their production, interfacial properties, and potential application. Curr. Opin. Colloid Interface Sci. 2024, 69, 101780. [Google Scholar] [CrossRef]
- Romero Vega, G.; Gallo Stampino, P. Bio-based surfactants and biosurfactants: An overview and main characteristics. Molecules 2025, 30, 863. [Google Scholar] [CrossRef]
- Baccile, N.; Kleinen, J. Biosurfactants and bioamphiphiles, survey, perspectives and applicative potential from a colloid science point of view. Curr. Opin. Colloid Interface Sci. 2025, 75, 101891. [Google Scholar] [CrossRef]
- Sivakumar, D.; Ramasamy, R.; Thiagarajan, Y.R.; Thirumalairaj, B.; Krishnamoorthy, U.; Haque Siddiqui, M.I.; Shah, M.A. Biosurfactants in biocorrosion and corrosion mitigation of metals: An overview. Open Chem. 2024, 22, 20240036. [Google Scholar] [CrossRef]
- Kugaji, M.; Ray, S.K.; Parvatikar, P.; Raghu, A.V. Biosurfactants: A review of different strategies for economical production, their applications and recent advancements. Adv. Colloid Interface Sci. 2025, 337, 103389. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Nayak, J.; Paul, I.; Kumar, R.; Jeon, B.H. Technological advancement in sustainable production of biosurfactant using agro-industrial wastes: A review of enhanced bioprocessing and computational optimization techniques. Crit. Rev. Environ. Sci. Technol. 2025, 55, 1430–1454. [Google Scholar] [CrossRef]
- Thacker, H.; Ram, V. Green corrosion inhibitors derived from plant extracts and drugs for mild steel in acid media: A review. Results Surf. Interfaces 2025, 18, 100364. [Google Scholar] [CrossRef]
- Aljibori, H.; Al-Amiery, A.; Isahak, W.N.R. Advancements in corrosion prevention techniques. J. Bio-Tribo-Corros. 2024, 10, 78. [Google Scholar] [CrossRef]
- Cragnolino, G.A. Corrosion fundamentals and characterization techniques. In Techniques for Corrosion Monitoring; Woodhead Publishing: Cambridge, UK, 2021; Volume 2, pp. 7–42. [Google Scholar] [CrossRef]
- Adarakatti, P.S. Fundamentals of electrochemistry. In Agricultural Electrochemistry; ACS Publications: Washington, DC, USA, 2025; Volume 1496, pp. 17–46. [Google Scholar] [CrossRef]
- Perez, N. Electrochemical corrosion. In Materials Science: Theory and Engineering, 1st ed.; Perez, N.S., Ed.; Springer Nature: Cham, Switzerland, 2024; Volume 1, pp. 835–898. [Google Scholar] [CrossRef]
- Harsimran, S.; Santosh, K.; Rakesh, K. Overview of corrosion and its control: A critical review. Proc. Eng. Sci. 2021, 3, 13–24. [Google Scholar] [CrossRef]
- Liu, X.; Gong, M.; Deng, S.; Zhao, T.; Shen, T.; Zhang, J.; Wang, D. Transforming damage into benefit: Corrosion engineering enabled electrocatalysts for water splitting. Adv. Funct. Mater. 2021, 31, 2009032. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, R.; Duan, J.; Shi, X.; Zhang, Y.; Guan, F.; Hou, B. Extracellular polymeric substances and biocorrosion/biofouling: Recent advances and future perspectives. Int. J. Mol. Sci. 2022, 23, 5566. [Google Scholar] [CrossRef]
- Li, Z.; Yang, J.; Lu, S.; Dou, W.; Gu, T. Stress corrosion cracking failure of X80 carbon steel U-bend caused by Desulfovibrio vulgaris biocorrosion. J. Mater. Sci. Technol. 2024, 174, 95–105. [Google Scholar] [CrossRef]
- Knisz, J.; Eckert, R.; Gieg, L.M.; Koerdt, A.; Lee, J.S.; Silva, E.R.; Wade, S.A. Microbiologically influenced corrosion—More than just microorganisms. FEMS Microbiol. Rev. 2023, 47, fuad041. [Google Scholar] [CrossRef] [PubMed]
- Verma, C. Basics and theories of corrosion: Thermodynamics and electrochemistry. In Handbook of Science & Engineering of Green Corrosion Inhibitors; Elsevier: Amsterdam, The Netherlands, 2022; Volume 3, pp. 21–30. [Google Scholar] [CrossRef]
- Zeng, C.; Neils, A.; Lesko, J.; Post, N. Machine learning accelerated discovery of corrosion-resistant high-entropy alloys. Comput. Mater. Sci. 2024, 237, 112925. [Google Scholar] [CrossRef]
- Tan, Q.; Atrens, A.; Mo, N.; Zhang, M.-X. Oxidation of magnesium alloys at high temperatures in air: A review. Corros. Sci. 2016, 112, 734–759. [Google Scholar] [CrossRef]
- De la Fuente, D. Corrosion of aluminum, aluminum alloys, and composites. In Encyclopedia of Materials: Metals and Alloys; Caballero, F.G., Ed.; Elsevier: Amsterdam, The Netherlands, 2022; pp. 160–169. [Google Scholar] [CrossRef]
- Xu, W.; Zhang, B.; Addison, O.; Wang, X.; Hou, B.; Yu, F. Mechanically-assisted crevice corrosion and its effect on materials degradation. Corros. Commun. 2023, 11, 23–32. [Google Scholar] [CrossRef]
- Rao, P.; Mulky, L. Erosion-corrosion of materials in industrial equipment: A review. ChemElectroChem 2023, 10, 202300152. [Google Scholar] [CrossRef]
- Wang, Z.B.; Zheng, Y.G. Critical flow velocity phenomenon in erosion-corrosion of pipelines: Determination methods, mechanisms and applications. J. Pipeline Sci. Eng. 2021, 1, 63–73. [Google Scholar] [CrossRef]
- Islam, M.A.; Farhat, Z. Erosion-corrosion mechanism and comparison of erosion-corrosion performance of API steels. Wear 2017, 376, 533–541. [Google Scholar] [CrossRef]
- Mainardi, V.A.; Cardoso, R.P.; Brunatto, S.F.; Scheuer, C.J. Slurry and liquid impingement erosion behavior of low-temperature plasma carburized AISI 420 martensitic stainless steel. Surf. Coat. Technol. 2024, 477, 130390. [Google Scholar] [CrossRef]
- Liu, C.; Tong, S.; Yue, Y.; Wang, H.; Song, J.; Li, Y.; Wang, Z. Laser-based fabrication of superwetting titanium alloy with enhanced corrosion and erosion-corrosion resistance. Colloids Surf. A Physicochem. Eng. Asp. 2024, 688, 133648. [Google Scholar] [CrossRef]
- Maher, S.D.; Sarvghad, M.; Olivares, R.; Ong, T.C.; Will, G.; Steinberg, T.A. Critical components in supercritical CO2 Brayton cycle power blocks for solar power systems: Degradation mechanisms and failure consequences. Sol. Energy Mater. Sol. Cells 2022, 242, 111768. [Google Scholar] [CrossRef]
- Alamri, A.H. Application of machine learning to stress corrosion cracking risk assessment. Egypt. J. Pet. 2022, 31, 11–21. [Google Scholar] [CrossRef]
- Ross, C.T. A conceptual design of an underwater vehicle. Ocean Eng. 2006, 33, 2087–2104. [Google Scholar] [CrossRef]
- Kadhim, A.; Al-Amiery, A.A.; Alazawi, R.; Al-Ghezi, M.K.S.; Abass, R.H. Corrosion inhibitors. A review. Int. J. Corros. Scale Inhib. 2021, 10, 54–67. [Google Scholar] [CrossRef]
- Cruz, J.P.B.; Veruz, E.G.; Aoki, I.V.; Schleder, A.M.; de Souza, G.F.M.; Vaz, G.L.; Martins, M.R. Uniform corrosion assessment in oil and gas pipelines using corrosion prediction models—Part 1: Models performance and limitations for operational field cases. Process Saf. Environ. Prot. 2022, 167, 500–515. [Google Scholar] [CrossRef]
- Ibrahimi, B.E.; Nardeli, J.V.; Guo, L. Sustainable Corrosion Inhibitors I: Fundamentals, Methodologies, and Industrial Applications; American Chemical Society: Washington, DC, USA, 2021; Volume 1403, pp. 1–19. [Google Scholar] [CrossRef]
- Kolesnikov, A.; Kolek, M.; Dohmann, J.F.; Horsthemke, F.; Börner, M.; Bieker, P.; Stan, M.C. Galvanic corrosion of lithium-powder-based electrodes. Adv. Energy Mater. 2020, 10, 2000017. [Google Scholar] [CrossRef]
- Moraes, C.V.; Kelly, R.G. A comparison of FEM results from the use of different governing equations in a galvanic cell part I: In the presence of a supporting electrolyte. Electrochim. Acta 2023, 469, 143146. [Google Scholar] [CrossRef]
- Ruiz-Garcia, A.; Esquivel-Peña, V.; Godínez, F.A.; Montoya, R. Corrosion modeling of aluminum alloys: A brief review. ChemElectroChem 2024, 11, 202300712. [Google Scholar] [CrossRef]
- Montoya, M.; Genesca, J.; Montoya, R. The AA7075–CS1018 galvanic couple under evaporating droplets. Corros. Mater. Degrad. 2024, 5, 92–108. [Google Scholar] [CrossRef]
- Wang, R. On the effect of pit shape on pitted plates, Part II: Compressive behavior due to random pitting corrosion. Ocean Eng. 2021, 236, 108737. [Google Scholar] [CrossRef]
- Shojai, S.; Schaumann, P.; Brömer, T. Probabilistic modelling of pitting corrosion and its impact on stress concentrations in steel structures in the offshore wind energy. Mar. Struct. 2022, 84, 103232. [Google Scholar] [CrossRef]
- Gibson, J.; Karney, B. A 30-year review of copper pitting corrosion and pinhole leaks: Achievements and research gaps. AWWA Water Sci. 2021, 3, 1221. [Google Scholar] [CrossRef]
- Sahu, S.; Frankel, G.S. Phase field modeling of crystallographic corrosion pits. J. Electrochem. Soc. 2022, 169, 020557. [Google Scholar] [CrossRef]
- Costa, E.M.; Dedavid, B.A.; Santos, C.A.; Lopes, N.F.; Fraccaro, C.; Pagartanidis, T.; Lovatto, L.P. Crevice corrosion on stainless steels in oil and gas industry: A review of techniques for evaluation, critical environmental factors and dissolved oxygen. Eng. Fail. Anal. 2023, 144, 106955. [Google Scholar] [CrossRef]
- Kelly, R.G.; Lee, J.S. Localized corrosion: Crevice corrosion. In Encyclopedia of Interfacial Chemistry; Elsevier: Amsterdam, The Netherlands, 2018; Volume 7, pp. 291–301. [Google Scholar] [CrossRef]
- Popoola, L.T.; Grema, A.S.; Latinwo, G.K.; Gutti, B.; Balogun, A.S. Corrosion problems during oil and gas production and its mitigation. Int. J. Ind. Chem. 2013, 4, 35. [Google Scholar] [CrossRef]
- Cramer, S.D.; Covino, B.S. A Corrosion: Fundamentals, testing, and protection. Mater. Park OH ASM Int. 2003, 13, 1135. [Google Scholar] [CrossRef]
- Kim, Y.J.; Bahn, C.B.; Baek, S.H.; Choi, W.; Song, G.D. Crevice chemistry and corrosion in high temperature water: A review. Nucl. Eng. Technol. 2024, 56, 3112–3122. [Google Scholar] [CrossRef]
- Birbilis, N.; Hinton, B. Corrosion and corrosion protection of aluminium. In Fundamentals of Aluminium Metallurgy, 1st ed.; Lumley, R., Ed.; Woodhead Publishing: Sawston, UK, 2011; Volume 1, pp. 574–604. [Google Scholar] [CrossRef]
- Makhlouf, A.S.H. Intelligent stannate-based coatings of self-healing functionality for magnesium alloys. In Intelligent Coatings for Corrosion Control, 1st ed.; Tiwari, A., Rawlins, J., Hihara, L.H., Eds.; Woodhead Publishing: Sawston, UK, 2015; Volume 1, pp. 537–555. [Google Scholar] [CrossRef]
- Qin, J.; Zhao, W.; Yan, Z.; Wang, R.; Yu, Z.; Shi, Z.; Wang, B. Intergranular corrosion and stress corrosion cracking properties evaluation and mechanism study of multi-microalloyed 2519 Al alloy. Corros. Sci. 2024, 236, 112288. [Google Scholar] [CrossRef]
- Kruger, J. Corrosion of metals: Overview. In Encyclopedia of Materials: Science and Technology; Pergamon: Oxford, UK, 2001; Volume 11, pp. 1701–1706. [Google Scholar] [CrossRef]
- Hartoyo, F.; Irianti, G.P.; Fatriansyah, J.F.; Ovelia, H.; Mas’ud, I.A.; Digita, F.R.; Anis, M. Weibull distribution optimization for piping risk calculation due to uniform corrosion using Monte Carlo method. Mater. Today Proc. 2023, 80, 1650–1655. [Google Scholar] [CrossRef]
- Liu, M. Effect of uniform corrosion on mechanical behavior of E690 high-strength steel lattice corrugated panel in marine environment: A finite element analysis. Mater. Res. Express 2021, 8, 066510. [Google Scholar] [CrossRef]
- Cheng, T.; Huang, H.; Huang, G. Galvanic corrosion behavior between ADC12 aluminum alloy and copper in 3.5 wt% NaCl solution. J. Electroanal. Chem. 2022, 927, 116984. [Google Scholar] [CrossRef]
- Gao, Y.; Kainuma, S.; Yang, M.; Ishihara, S.; Inoue, D. Galvanic corrosion behavior of hot-dip Al and 55Al–Zn coatings applied to steel bolted joints in atmospheric environments. Constr. Build. Mater. 2023, 401, 132694. [Google Scholar] [CrossRef]
- Liu, Y.; Miao, Z.; Yuan, H.; Geng, X.; Hogan, L.; Zhu, M. Experimental study of tensile behaviour of rebars with simulated pitting corrosion. Constr. Build. Mater. 2024, 445, 137952. [Google Scholar] [CrossRef]
- Xiao, Y.; Lin, B.; Tang, J.; Zheng, H.; Wang, Y.; Zhang, H.; Sun, X. Effect of elastic tensile stress on the pitting corrosion mechanism and passive film of 2205 duplex stainless steel. Electrochim. Acta 2024, 477, 143765. [Google Scholar] [CrossRef]
- Pu, Y.; Guo, Z.; Hou, S.; Hou, Y.; Zhu, C.; Feng, F.; Chen, S. Toward understanding the microbiologically induced crevice corrosion of 90/10 Cu-Ni alloy in the presence of Desulfovibrio vulgaris. Corros. Sci. 2025, 248, 112802. [Google Scholar] [CrossRef]
- Zhao, M.; Wang, J.; Ma, Y.; Xu, Y.; Zhang, X.; Li, Z.; Wang, F. Electroactive Shewanella algae accelerates the crevice corrosion of X70 pipeline steel in marine environment. Corros. Sci. 2024, 235, 112226. [Google Scholar] [CrossRef]
- Du, H.Y.; Hu, Q.K.; Yue, X.W.; Jia, J.W.; Wei, Y.H.; Hou, L.F.; Liu, X.D. Effect of Mo on intergranular corrosion behavior in super-austenitic stainless steel. Corros. Sci. 2024, 231, 111986. [Google Scholar] [CrossRef]
- Ida, N.; Nishimoto, M.; Muto, I.; Sugawara, Y. Role of MnS in the intergranular corrosion and depassivation of sensitized type 304 stainless steel. npj Mater. Degrad. 2024, 8, 2. [Google Scholar] [CrossRef]
- Yang, Z.; Li, L.; Qiao, Y.; Li, C.; Zhang, L.; Cui, J.; Zheng, Y. Cavitation erosion-corrosion properties of as-cast TC4 and LPBF TC4 in 0.6 mol/L NaCl solution: A comparison investigation. Ultrason. Sonochemistry 2024, 108, 106947. [Google Scholar] [CrossRef]
- Wang, K.; Xu, Q.; Li, Y.; Guo, P.; Jia, Y.; Zhou, H. Influence of sand particle size on the erosion-corrosion resistance of Ni2FeCrMo0. 2 HEA in seawater: Particle-surface-electrochemistry interaction. J. Mater. Res. Technol. 2024, 32, 426–438. [Google Scholar] [CrossRef]
- Zeng, Q.; Chen, Y.; Yang, Z.; Zhang, L.; Wang, Z.; Wang, L.; Wang, J. Effect of grain size and grain boundary type on intergranular stress corrosion cracking of austenitic stainless steel: A phase-field study. Corros. Sci. 2024, 241, 112557. [Google Scholar] [CrossRef]
- Pan, Y.; Sun, B.; Chen, H.; Liu, Z.; Dai, W.; Yang, X.; Li, X. Stress corrosion cracking behavior and mechanism of 2205 duplex stainless steel under applied polarization potentials. Corros. Sci. 2024, 231, 111978. [Google Scholar] [CrossRef]
- Solovyeva, V.A.; Almuhammadi, K.H.; Badeghaish, W.O. Current downhole corrosion control solutions and trends in the oil and gas industry: A review. Materials 2023, 16, 1795. [Google Scholar] [CrossRef]
- Khan, M.A.A.; Hussain, M.; Djavanroodi, F. Microbiologically influenced corrosion in oil and gas industries: A review. Int. J. Corros. Scale Inhib. 2021, 10, 80–106. [Google Scholar] [CrossRef]
- Safian, M.T.U.; Pandian, B.R.; Mohamad Ibrahim, M.N. The importance and evolution of corrosion inhibitors in the drilling fluid for the offshore drilling industry. Corros. Rev. 2024, 42, 399–433. [Google Scholar] [CrossRef]
- Cao, Q.; Brameld, M.; Birbilis, N.; Thomas, S. On the mitigation of corrosion under insulation (CUI) of mild steel using local cathodic protection. Corrosion 2019, 75, 1541–1551. [Google Scholar] [CrossRef]
- Cao, Q.; Pojtanabuntoeng, T.; Esmaily, M.; Thomas, S.; Brameld, M.; Amer, A.; Birbilis, N. A review of corrosion under insulation: A critical issue in the oil and gas industry. Metals 2022, 12, 561. [Google Scholar] [CrossRef]
- Sanni, O.; Iwarere, S.A.; Daramola, M.O. Investigation of eggshell agro-industrial waste as a potential corrosion inhibitor for mild steel in oil and gas industry. Sustainability 2023, 15, 6155. [Google Scholar] [CrossRef]
- Jin, H.; Wang, M.; Xiang, H.; Liu, X.; Wang, C.; Fu, D. A PSO-RBF prediction method on flow corrosion of heat exchanger using the industrial operations data. Process Saf. Environ. Prot. 2024, 183, 11–23. [Google Scholar] [CrossRef]
- Ma, X.; Zhu, G.; Lu, D.; Hou, B.D.J. Corrosion management system in China. Strateg. Study Chin. Acad. Eng. 2002, 24, 190–197. [Google Scholar] [CrossRef]
- Vakili, M.; Koutník, P.; Kohout, J. Addressing hydrogen sulfide corrosion in oil and gas industries: A sustainable perspective. Sustainability 2024, 16, 1661. [Google Scholar] [CrossRef]
- Jasim, Z.I.; Rashid, K.H.; Khadom, A.A. Corrosion and corrosion control of the steel in acidizing oil wells processes: An overview of organic inhibitors. Russ. J. Appl. Chem. 2024, 97, 56–74. [Google Scholar] [CrossRef]
- Castro, J.D.; Evaristo, M.; Carvalho, S. Mechanical and anti-corrosion behaviour of ZrNxOy films deposited by reactive high-power impulse magnetron sputtering for maritime applications. Surf. Coat. Technol. 2023, 473, 129991. [Google Scholar] [CrossRef]
- Imran, M.M.H.; Jamaludin, S.; Ayob, A.F.M.; Ali, A.A.I.M.; Ahmad, S.Z.A.S.; Akhbar, M.F.A.; Mohamed, S.B. Application of artificial intelligence in marine corrosion prediction and detection. J. Mar. Sci. Eng. 2023, 11, 256. [Google Scholar] [CrossRef]
- Beko, M.; Ivošević, Š.; Dlabač, T. Application of sensors in the corrosion monitoring of the ship’s structural parts. In Proceedings of the 2021 25th International Conference on Information Technology (IT), Zabljak, Montenegro, 16–20 February 2021; Volume 1, pp. 1–4. [Google Scholar] [CrossRef]
- Davidson, I.; Cahill, P.; Hinz, A.; Kluza, D.; Scianni, C.; Georgiades, E. A review of biofouling of ships’ internal seawater systems. Front. Mar. Sci. 2021, 8, 761531. [Google Scholar] [CrossRef]
- Vourna, P.; Ktena, A.; Hristoforou, E.V.; Papadopoulos, N.D. Assessment of corrosion in naval steels submerged in artificial seawater utilizing a magnetic non-destructive sensor. Sensors 2025, 25, 5015. [Google Scholar] [CrossRef]
- Buruiana, D.L.; Herbei, E.E.; Ceoromila, A.M.; Busila, M.; Trus, C.; Bogatu, N.; Ghisman, V. Investigation of corrosion of A and E36 naval steels using potentiodynamic polarization and modified prohesion test. Int. J. Electrochem. Sci. 2021, 16, 21111. [Google Scholar] [CrossRef]
- Bouanani, O.; Sandabad, S.; Ech-Chhibat, M.E.H. Innovations in shipbuilding: A comprehensive review of material selection, corrosion management, and environmental considerations. J. King Abdulaziz Univ. Mar. Sci. 2025, 35, 48–63. [Google Scholar] [CrossRef]
- Pourasl, H.H.; Barenji, R.V.; Khojastehnezhad, V.M. Solar energy status in the world: A comprehensive review. Energy Rep. 2023, 10, 3474–3493. [Google Scholar] [CrossRef]
- Maka, A.O.; Alabid, J.M. Solar energy technology and its roles in sustainable development. Clean Energy 2022, 6, 476–483. [Google Scholar] [CrossRef]
- Dos Santos, S.A.A.; Torres, J.P.N.; Fernandes, C.A.; Lameirinhas, R.A.M. The impact of aging of solar cells on the performance of photovoltaic panels. Energy Convers. Manag. X 2021, 10, 100082. [Google Scholar] [CrossRef]
- Kumar, K.R.; Chaitanya, N.K.; Kumar, N.S. Solar thermal energy technologies and its applications for process heating and power generation–A review. J. Clean. Prod. 2021, 282, 125296. [Google Scholar] [CrossRef]
- Walczak, M.; Pineda, F.; Fernandez, A.G.; Mata-Torres, C.; Escobar, R.A. Materials corrosion for thermal energy storage systems in concentrated solar power plants. Renew. Sustain. Energy Rev. 2018, 86, 22–44. [Google Scholar] [CrossRef]
- Zhao, H.Y.; Wang, X.; Wu, J.; Shu, C.; Gao, F.L.; Li, C.; Yu, Z.Z. An intelligent, solar-responsive, and thermally conductive phase-change system toward solar-thermal-electrical conversion featuring daytime blooming for solar energy harvesting and nighttime closing for thermal preservation. Adv. Funct. Mater. 2024, 34, 2406236. [Google Scholar] [CrossRef]
- Rana, Z.; Zamora, P.P.; Soliz, A.; Soler, D.; Reyes Cruz, V.E.; Cobos-Murcia, J.A.; Galleguillos Madrid, F.M. Solar panel corrosion: A review. Int. J. Mol. Sci. 2025, 26, 5960. [Google Scholar] [CrossRef]
- Sadiq, M.; Shinwari, R.; Wen, F.; Usman, M.; Hassan, S.T.; Taghizadeh-Hesary, F. Do globalization and nuclear energy intensify the environmental costs in top nuclear energy-consuming countries? Prog. Nucl. Energy 2023, 156, 104533. [Google Scholar] [CrossRef]
- Mathew, M.D. Nuclear energy: A pathway towards mitigation of global warming. Prog. Nucl. Energy 2022, 143, 104080. [Google Scholar] [CrossRef]
- Zhang, D.H.; Meng, X.C.; Zuo, G.Z.; Huang, M.; Li, L.; Xu, W.; Hu, J.S. Study of the corrosion characteristics of 304 and 316L stainless steel in the static liquid lithium. J. Nucl. Mater. 2021, 553, 153032. [Google Scholar] [CrossRef]
- Fu, J.; Gui, W.; Qin, J.; Lin, J.; Ren, X.; Luan, B. High-speed laser-clad 3533-00 Fe-based cemented carbide coatings on nuclear power valve sealing surface to enhanced wear and corrosion resistance. Surf. Coat. Technol. 2024, 480, 130594. [Google Scholar] [CrossRef]
- Nash, C.; Karve, P.; Adams, D. Diagnosing nuclear power plant pipe wall thinning due to flow accelerated corrosion using a passive, thermal non-destructive evaluation method: Feasibility assessment via numerical experiments. Nucl. Eng. Des. 2022, 386, 111542. [Google Scholar] [CrossRef]
- Liu, S.; Zhang, L.; Lu, J.; Zhang, X.; Wang, K.; Gan, Z.; Wang, H. Advances in urban wind resource development and wind energy harvesters. Renew. Sustain. Energy Rev. 2025, 207, 114943. [Google Scholar] [CrossRef]
- Msigwa, G.; Ighalo, J.O.; Yap, P.S. Considerations on environmental, economic, and energy impacts of wind energy generation: Projections towards sustainability initiatives. Sci. Total Environ. 2022, 849, 157755. [Google Scholar] [CrossRef]
- Kwok, K.C.S.; Hu, G. Wind energy system for buildings in an urban environment. J. Wind Eng. Ind. Aerodyn. 2023, 234, 105349. [Google Scholar] [CrossRef]
- Juhl, M.; Hauschild, M.Z.; Dam-Johansen, K. Sustainability of corrosion protection for offshore wind turbine towers. Prog. Org. Coat. 2024, 186, 107998. [Google Scholar] [CrossRef]
- Shao, W.; Du, X.; Zhang, W.; Shi, D.; Zhang, J.; Tang, P. Corrosion mechanisms, responses, and mitigation strategies for steel piles in offshore wind turbines: A comprehensive review. Mater. Corros. 2025, 76, 957–978. [Google Scholar] [CrossRef]
- Kamde, D.K.; Pillai, R.G. Effect of surface preparation on corrosion of steel rebars coated with cement-polymer-composites (CPC) and embedded in concrete. Constr. Build. Mater. 2020, 237, 117616. [Google Scholar] [CrossRef]
- Han, X.; Yang, D.Y.; Frangopol, D.M. Optimum maintenance of deteriorated steel bridges using corrosion resistant steel based on system reliability and life-cycle cost. Eng. Struct. 2021, 243, 112633. [Google Scholar] [CrossRef]
- Green, W.K. Steel reinforcement corrosion in concrete–an overview of some fundamentals. Corros. Eng. Sci. Technol. 2020, 55, 289–302. [Google Scholar] [CrossRef]
- Mousa, A.A.; Abed, J.M.; Shukur, M.H. Effect of supplementary cementitious materials on corrosion resistance of reinforced concrete. Civ. Environ. Eng. 2025, 21, 1065–1082. [Google Scholar] [CrossRef]
- Balonis, M.; Sant, G.; Isgor, O.B. Mitigating steel corrosion in reinforced concrete using functional coatings, corrosion inhibitors, and atomistic simulations. Cem. Concr. Compos. 2019, 101, 15–23. [Google Scholar] [CrossRef]
- Arya, E.K.; Dhanya, B.S. Corrosion control of reinforced concrete structures in construction industry: A review. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2021; Volume 1114, p. 012006. [Google Scholar] [CrossRef]
- Pierott, R.M.R.; Garcia, S.; Kropf, D.; Figueiredo, K.; da Costa, B.B.F.; Amario, M.; Haddad, A. Predictive analysis of corrosion dynamics in prestressed concrete exposed to chloride environments. Infrastructures 2024, 9, 133. [Google Scholar] [CrossRef]
- Qian, R.; Li, Q.; Fu, C.; Zhang, Y.; Wang, Y.; Jin, X. Atmospheric chloride-induced corrosion of steel-reinforced concrete beam exposed to real marine-environment for 7 years. Ocean Eng. 2023, 286, 115675. [Google Scholar] [CrossRef]
- Poursaee, A.; Ross, B. The role of cracks in chloride-induced corrosion of carbon steel in concrete. Corros. Mater. Degrad. 2022, 3, 258–269. [Google Scholar] [CrossRef]
- Klein, N.; Gómez, E.D.; Duffó, G.S.; Farina, S.B. Effect of sulphate on the corrosion of reinforcing steel in concrete. Constr. Build. Mater. 2022, 354, 129214. [Google Scholar] [CrossRef]
- Kanagaraj, B.; Prathana, S.; Joe, A.; Anand, N.; Naser, M.Z.; Lubloy, E. Flexural behavior and performance assessment of corroded geopolymer concrete beams after fire exposure. Case Stud. Chem. Environ. Eng. 2025, 11, 101232. [Google Scholar] [CrossRef]
- Elsayed, M.; Tayeh, B.A.; Taha, Y.; Abd El-Azim, A. Experimental investigation on the behaviour of crumb rubber concrete columns exposed to chloride–sulphate attack. Structures 2022, 46, 246–264. [Google Scholar] [CrossRef]
- Yan, L.; Deng, W.; Wang, N.; Xue, X.; Hua, J.; Chen, Z. Anti-corrosion reinforcements using coating technologies—A review. Polymers 2022, 14, 4782. [Google Scholar] [CrossRef] [PubMed]
- Desai, P.D.; Pawar, C.B.; Avhad, M.S.; More, A.P. Corrosion inhibitors for carbon steel: A review. Vietnam J. Chem. 2023, 61, 15–42. [Google Scholar] [CrossRef]
- Verma, C.; Chauhan, D.S.; Aslam, R.; Banerjee, P.; Aslam, J.; Quadri, T.W.; Rasheed, T. Principles and theories of green chemistry for corrosion science and engineering: Design and application. Green Chem. 2024, 26, 4270–4357. [Google Scholar] [CrossRef]
- Winkler, D.A.; Hughes, A.E.; Özkan, C.; Mol, A.; Würger, T.; Feiler, C.; Lamaka, S.V. Impact of inhibition mechanisms, automation, and computational models on the discovery of organic corrosion inhibitors. Prog. Mater. Sci. 2025, 149, 101392. [Google Scholar] [CrossRef]
- Al-Amiery, A.A.; Al-Azzawi, W.K. Mannich bases as corrosion inhibitors: An extensive review. J. Mol. Struct. 2023, 1294, 136421. [Google Scholar] [CrossRef]
- Al-Amiery, A.A.; Isahak, W.N.R.W.; Al-Azzawi, W.K. Corrosion inhibitors: Natural and synthetic organic inhibitors. Lubricants 2023, 11, 174. [Google Scholar] [CrossRef]
- Olajire, A.A. Recent advances on the treatment technology of oil and gas produced water for sustainable energy industry-mechanistic aspects and process chemistry perspectives. Chem. Eng. J. Adv. 2020, 4, 100049. [Google Scholar] [CrossRef]
- Bijapur, K.; Molahalli, V.; Shetty, A.; Toghan, A.; De Padova, P.; Hegde, G. Recent trends and progress in corrosion inhibitors and electrochemical evaluation. Appl. Sci. 2023, 13, 10107. [Google Scholar] [CrossRef]
- Ahmed E S, J.; Ganesh, G.M. A comprehensive overview on corrosion in rcc and its prevention using various green corrosion inhibitors. Buildings 2022, 12, 1682. [Google Scholar] [CrossRef]
- Tamalmani, K.; Husin, H. Review on corrosion inhibitors for oil and gas corrosion issues. Appl. Sci. 2020, 10, 3389. [Google Scholar] [CrossRef]
- Ahmed, M.A.; Amin, S.; Mohamed, A.A. Current and emerging trends of inorganic, organic and eco-friendly corrosion inhibitors. RSC Adv. 2024, 14, 31877–31920. [Google Scholar] [CrossRef]
- Sahu, B.C. Organic corrosion inhibitors. In Introduction to Corrosion-Basics and Advances, 1st ed.; Singh, A., Ed.; IntechOpen: London, UK, 2023; Volume 1, pp. 1–35. [Google Scholar] [CrossRef]
- Lozano, I.; Mazario, E.; Olivares-Xometl, C.O.; Likhanova, N.V.; Herrasti, P. Corrosion behaviour of API 5LX52 steel in HCl and H2SO4 media in the presence of 1,3-dibencilimidazolio acetate and 1,3-dibencilimidazolio dodecanoate ionic liquids as inhibitors. Mater. Chem. Phys. 2014, 147, 191–197. [Google Scholar] [CrossRef]
- Olasunkanmi, L.O.; Ebenso, E.E. Experimental and computational studies on propanone derivatives of quinoxalin-6-yl-4,5-dihydropyrazole as inhibitors of mild steel corrosion in hydrochloric acid. J. Colloid Interface Sci. 2020, 561, 104–116. [Google Scholar] [CrossRef]
- Hadisaputra, S.; Purwoko, A.A.; Hakim, A.; Prasetyo, N.; Hamdiani, S. Corrosion inhibition properties of phenyl phthalimide derivatives against carbon steel in the acidic medium: DFT, MP2, and Monte Carlo simulation studies. ACS Omega 2022, 7, 33054–33066. [Google Scholar] [CrossRef] [PubMed]
- Bahremand, F.; Shahrabi, T.; Ramezanzadeh, B.; Hosseini, S.A. Sustainable development of an effective anti-corrosion film over the St12-steel surface against seawater attacks using Ce (III) ions/tri-sodium phosphate anions. Sci. Rep. 2023, 13, 12169. [Google Scholar] [CrossRef] [PubMed]
- Al-Amiery, A.A.; Yousif, E.; Isahak, W.N.R.W.; Al-Azzawi, W.K. A review of inorganic corrosion inhibitors: Types, mechanisms, and applications. Tribol. Ind. 2023, 44, 313. [Google Scholar] [CrossRef]
- Mubarak, G.; Verma, C.; Barsoum, I.; Alfantazi, A.; Rhee, K.Y. Internal corrosion in oil and gas wells during casings and tubing: Challenges and opportunities of corrosion inhibitors. J. Taiwan Inst. Chem. Eng. 2023, 150, 105027. [Google Scholar] [CrossRef]
- Amini, M.; Naderi, R.; Mahdavian, M.; Badiei, A. Release of lanthanum cations loaded into piperazine-modified SBA-15 to inhibit the mild steel corrosion. Microporous Mesoporous Mater. 2021, 315, 110908. [Google Scholar] [CrossRef]
- Abdel-Karim, A.M.; El-Shamy, A.M. A review on green corrosion inhibitors for protection of archeological metal artifacts. J. Bio-Tribo-Corros. 2022, 8, 35. [Google Scholar] [CrossRef]
- Fazal, B.R.; Becker, T.; Kinsella, B.; Lepkova, K. A review of plant extracts as green corrosion inhibitors for CO2 corrosion of carbon steel. npj Mater. Degrad. 2022, 6, 5. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, R.; Wu, R.; Luo, Y.; Guo, L.; He, Z. Green and high-efficiency corrosion inhibitors for metals: A review. J. Adhes. Sci. Technol. 2023, 37, 1501–1524. [Google Scholar] [CrossRef]
- Salleh, S.Z.; Yusoff, A.H.; Zakaria, S.K.; Taib, M.A.A.; Seman, A.A.; Masri, M.N.; Ter Teo, P. Plant extracts as green corrosion inhibitor for ferrous metal alloys: A review. J. Clean. Prod. 2021, 304, 127030. [Google Scholar] [CrossRef]
- Miralrio, A.; Espinoza Vázquez, A. Plant extracts as green corrosion inhibitors for different metal surfaces and corrosive media: A review. Processes 2020, 8, 942. [Google Scholar] [CrossRef]
- Kumar, N.; Noor, I.S.M.; Yahya, M.Z.A.; Prakash, S. Botanical corrosion inhibitors in reinforced concrete: Material sustainability assessment and analysis—A review. Zast. Mater. 2025, 66, 40–55. [Google Scholar] [CrossRef]
- Jebali, Z.; Ferkous, H.; Zerroug, M.; Boublia, A.; Delimi, A.; Bouzid, A.; Benguerba, Y. Unveiling the potent corrosion-inhibiting power of Ammophila arenaria aqueous extract for mild steel in acidic environments: An integrated experimental and computational study. J. Environ. Chem. Eng. 2024, 12, 112374. [Google Scholar] [CrossRef]
- Lakikza, I.; Benguerba, Y.; Boublia, A.; Aouni, S.I.; Lahbib, H.; Ferkous, H.; Ernst, B. Comprehensive evaluation of Alysicarpus compactum extract as a natural corrosion inhibitor for St37 carbon steel in acidic media. J. Ind. Eng. Chem. 2025, 147, 161–178. [Google Scholar] [CrossRef]
- Kahkesh, H.; Yeganeh, M.; Shahryari, Z. Algae-based corrosion inhibitors as innovative approaches to corrosion prevention: A review. Algal Res. 2025, 89, 104071. [Google Scholar] [CrossRef]
- Yousif, Q.A.; Bedair, M.A.; Fadel, Z.; Al-Odail, F.; Abuelela, A.M. Evaluating the efficacy of newly synthesized amino acid derivatives as corrosion inhibitors in acidic solutions. Inorg. Chem. Commun. 2024, 164, 112454. [Google Scholar] [CrossRef]
- Al-Amiery, A.; Betti, N.A.; Shaker, L.M. Exploring the effectiveness of 3-chloro-4-morpholin-4-yl-1,2,5-thiadiazole as an eco-friendly corrosion inhibitor for mild steel in HCl solution: Experimental and DFT analysis. Results Eng. 2024, 24, 103014. [Google Scholar] [CrossRef]
- Hussein, R.K.; Abou-Krisha, M.; Yousef, T.A. Theoretical and experimental studies of different amine compounds as corrosion inhibitors for aluminum in hydrochloric acid. Biointerface Res. Appl. Chem. 2021, 11, 9772. [Google Scholar] [CrossRef]
- Belayachi, M.A.; Kharbouch, O.; Dahmani, K.; El Magri, A.; Er-rahmany, N.; Saber, I.; Chaouiki, A. Electrochemical, surface, and theoretical studies on amine-based organic compounds as efficient corrosion inhibitors for mild steel in 1 M HCl. Int. J. Electrochem. Sci. 2025, 20, 100951. [Google Scholar] [CrossRef]
- Chaudhari, T.; Rajagopalan, N.; Dam-Johansen, K. Lignin phosphate: A biobased substitute for zinc phosphate in corrosion-inhibiting coatings. ACS Sustain. Chem. Eng. 2024, 12, 7813–7830. [Google Scholar] [CrossRef]
- Mohamed, A.; Martin, U.; Bastidas, D.M. Adsorption and surface analysis of sodium phosphate corrosion inhibitor on carbon steel in simulated concrete pore solution. Materials 2022, 15, 7429. [Google Scholar] [CrossRef]
- Zhao, T.; Wang, Z.; Feng, Y.; Li, Q. Synergistic corrosion inhibition of sodium phosphate and sodium dodecyl sulphate on magnesium alloy AZ91 in 3.5 wt% NaCl solution. Mater. Today Commun. 2022, 31, 103568. [Google Scholar] [CrossRef]
- Kumar, U.P.; He, B.B. Synergistic impact of phytic acid and sodium phosphate on the corrosion inhibition of additively manufactured 304L steel in a 3.5 wt% NaCl medium. J. Mater. Sci. 2025, 60, 11092–11111. [Google Scholar] [CrossRef]
- Gapsari, F.; Setyarini, P.H.; Anam, K.; Hadisaputra, S.; Hidayatullah, S.; Sulaiman, A.M.; Lai, C.W. Efficacy of Andrographis paniculata leaf extract as a green corrosion inhibitor for mild steel in concentrated sulfuric acid: Experimental and computational insights. Results Surf. Interfaces 2025, 18, 100361. [Google Scholar] [CrossRef]
- Ananthkumar, M.; Mini, K.M.; Thilagavathy, P. Eco-friendly corrosion protection of steel in simulated concrete environments using Cassia fistula leaf extract: Electrochemical and surface characterization study. S. Afr. J. Chem. Eng. 2025, 54, 44–49. [Google Scholar] [CrossRef]
- Krishnanjana, K.; Ganesh, G.M. Green corrosion inhibition of Punica granatum peel powder on TMT in salt medium: Outlooks from experiments and artificial neural networks. Green Chem. Lett. Rev. 2025, 18, 2525117. [Google Scholar] [CrossRef]
- Du, P.; Teng, X.; Qiang, Y.; Deng, S.; Li, X.; Xu, D. Structure–mechanism–performance relationships in rubber seed-derived corrosion inhibitors: Toward enhanced steel protection in acidic environments. Colloids Surf. A Physicochem. Eng. Asp. 2025, 727, 138294. [Google Scholar] [CrossRef]
- da Silva Faccioli, Y.E.; da Silva, G.O.; Soares da Silva, R.D.C.F.; Sarubbo, L.A. Application of a biosurfactant from Pseudomonas cepacia CCT 6659 in bioremediation and metallic corrosion inhibition processes. J. Biotechnol. 2022, 351, 109–121. [Google Scholar] [CrossRef]
- Li, Z.; Yuan, X.; Sun, M.; Li, Z.; Zhang, D.; Lei, Y.; Wang, F. Rhamnolipid as an eco-friendly corrosion inhibitor for microbiologically influenced corrosion. Corros. Sci. 2022, 204, 110390. [Google Scholar] [CrossRef]
- Sheydaei, M. The use of plant extracts as green corrosion inhibitors: A review. Surfaces 2024, 7, 380–403. [Google Scholar] [CrossRef]
- Vieira, I.M.M.; Santos, B.L.P.; Ruzene, D.S.; Silva, D.P. An overview of current research and developments in biosurfactants. J. Ind. Eng. Chem. 2021, 100, 1–18. [Google Scholar] [CrossRef]
- Sarubbo, L.A.; Maria da Gloria, C.S.; Durval, I.J.B.; Bezerra, K.G.O.; Ribeiro, B.G.; Silva, I.A.; Banat, I.M. Biosurfactants: Production, properties, applications, trends, and general perspectives. Biochem. Eng. J. 2022, 181, 108377. [Google Scholar] [CrossRef]
- Santos, C.V.M.; Vieira, I.M.M.; Santos, B.L.P.; de Souza, R.R.; Ruzene, D.S.; Silva, D.P. Biosurfactant production from pineapple waste and application of experimental design and statistical analysis. Appl. Biochem. Biotechnol. 2023, 195, 386–400. [Google Scholar] [CrossRef] [PubMed]
- Shaikhah, D.; Loise, V.; Angelico, R.; Porto, M.; Calandra, P.; Abe, A.A.; Caputo, P. New trends in biosurfactants: From renewable origin to green enhanced oil recovery applications. Molecules 2024, 29, 301. [Google Scholar] [CrossRef]
- Hsu, C.Y.; Mahmoud, Z.H.; Hussein, U.A.R.; Abduvalieva, D.; Alsultany, F.H.; Kianfar, E. Biosurfactants: Properties, applications and emerging trends. S. Afr. J. Chem. Eng. 2025, 53, 21–39. [Google Scholar] [CrossRef]
- Okorie, I.K.M.; Ogunjobi, A.A. Characterisation of a unique manganese-containing biosurfactant produced from Pseudomonas aeruginosa strain S16, isolated from soil found in a mixed farm, located in Ibadan, Oyo State, Nigeria. Res. Sq. 2024, 1, 1–13. [Google Scholar] [CrossRef]
- Dini, S.; Bekhit, A.E.D.A.; Roohinejad, S.; Vale, J.M.; Agyei, D. The physicochemical and functional properties of biosurfactants: A review. Molecules 2024, 29, 2544. [Google Scholar] [CrossRef]
- Oliveira, K.W.; Selva Filho, A.A.P.; Faccioli, Y.E.S.; Araújo, G.P.; Converti, A.; Soares da Silva, R.D.C.F.; Sarubbo, L.A. Eco-friendly biosurfactant: Tackling oil pollution in terrestrial and aquatic ecosystems. Fermentation 2025, 11, 199. [Google Scholar] [CrossRef]
- Zhao, F.; Cui, Q.; Su, H.; Li, C.; Dong, M. Biosurfactants are green and versatile enhancers for sustainable treatment of oily sludge: A review. J. Environ. Chem. Eng. 2025, 13, 115087. [Google Scholar] [CrossRef]
- Marquez, R.; Ortiz, M.S.; Barrios, N.; Vera, R.E.; Patiño-Agudelo, Á.J.; Vivas, K.A.; Theiner, E. Surfactants produced from carbohydrate derivatives: Part 2. A review on the value chain, synthesis, and the potential role of artificial intelligence within the biorefinery concept. J. Surfactants Deterg. 2025, 28, 25–76. [Google Scholar] [CrossRef]
- Guzmán, E.; Maestro, A.; Ortega, F.; Rubio, R.G. Association of oppositely charged polyelectrolyte and surfactant in solution: Equilibrium and nonequilibrium features. J. Phys. Condens. Matter 2023, 35, 323001. [Google Scholar] [CrossRef] [PubMed]
- Vučurović, D.; Bajić, B.; Trivunović, Z.; Dodić, J.; Zeljko, M.; Jevtić-Mučibabić, R.; Dodić, S. Biotechnological utilization of agro-industrial residues and by-products—Sustainable production of biosurfactants. Foods 2024, 13, 711. [Google Scholar] [CrossRef] [PubMed]
- Messaoud, G.B. Viscoelastic systems from glycolipid biosurfactants. Curr. Opin. Colloid Interface Sci. 2024, 71, 101805. [Google Scholar] [CrossRef]
- Miao, Y.; To, M.H.; Siddiqui, M.A.; Wang, H.; Lodens, S.; Chopra, S.S.; Lin, C.S.K. Sustainable biosurfactant production from secondary feedstock—Recent advances, process optimization and perspectives. Front. Chem. 2024, 12, 1327113. [Google Scholar] [CrossRef]
- Darwiche, N.; Dufresne, C.; Chartier, A.; Claude, B.; Colas, C.; Fougère, L.; Nehmé, R. Glycolipid and lipopeptide biosurfactants: Structural classes and characterization—Rhamnolipids as a model. Crit. Rev. Anal. Chem. 2024, 1, 1–21. [Google Scholar] [CrossRef]
- Pala, M.; Castelein, M.G.; Dewaele, C.; Roelants, S.L.; Soetaert, W.K.; Stevens, C.V. Tuning the antimicrobial activity of microbial glycolipid biosurfactants through chemical modification. Front. Bioeng. Biotechnol. 2024, 12, 1347185. [Google Scholar] [CrossRef]
- Das, R.P.; Sahoo, S.; Paidesetty, S.K.; Ahmad, I.; Sahoo, B.; Jayabaskaran, C.; Pradhan, A.K. Isolation, characterization, and multimodal evaluation of novel glycolipid biosurfactant derived from Bacillus species: A promising Staphylococcus aureus tyrosyl-tRNA synthetase inhibitor through molecular docking and MD simulations. Int. J. Biol. Macromol. 2024, 261, 129848. [Google Scholar] [CrossRef]
- Thundiparambil Venu, A.; Vijayan, J.; Ammanamveetil, M.H.A.; Kottekkattu Padinchati, K. An insightful overview of microbial biosurfactant: A promising next-generation biomolecule for sustainable future. J. Basic Microbiol. 2024, 64, 2300757. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Fukuoka, T. Sophorolipids, commercialized glycolipid biosurfactants: Derivatives, component analysis, and applications. J. Am. Oil Chem. Soc. 2025, 102, 251–260. [Google Scholar] [CrossRef]
- Li, S.; Jiang, X.; Zhao, C.; Ren, Y.; Luo, L. Evaluation of lipopeptides biosurfactant from Raoultella planticola for bioremediation in n-hexadecane-contaminated soil. J. Environ. Chem. Eng. 2024, 12, 112622. [Google Scholar] [CrossRef]
- Purwasena, I.A.; Fitri, D.K.; Putri, D.M.; Endro, H.; Zakaria, M.N. Lipopeptide biosurfactant as a potential root canal irrigation agent: Antimicrobial and anti-biofilm evaluation. J. Dent. 2024, 144, 104961. [Google Scholar] [CrossRef] [PubMed]
- Marhaendrajana, T.; Widiyaningsih, I.; Kurnia, I.; Sulistyarso, H.B. Fluid-to-fluid and fluid-to-rock interaction on sophorolipids biosurfactant for enhanced oil recovery: A literature review. Sci. Contrib. Oil Gas 2025, 48, 63–76. [Google Scholar] [CrossRef]
- Phulpoto, I.A.; Bobo, W.; Qazi, M.A.; Yu, Z. Lipopeptide biosurfactants enhanced biohydrogen evolution from lignocellulose biomass and shaped the microbial community and diversity. Int. J. Energy Res. 2024, 1, 3084702. [Google Scholar] [CrossRef]
- Sani, A.; Qin, W.Q.; Li, J.Y.; Liu, Y.F.; Zhou, L.; Yang, S.Z.; Mu, B.Z. Structural diversity and applications of lipopeptide biosurfactants as biocontrol agents against phytopathogens: A review. Microbiol. Res. 2024, 278, 127518. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Chen, X.; Chen, B.; Wu, D.; Yang, S. A review on approach for biosurfactants enhance oil recovery technology. Geomicrobiol. J. 2024, 41, 910–920. [Google Scholar] [CrossRef]
- Simões, C.R.; da Silva, M.W.P.; de Souza, R.F.M.; Hacha, R.R.; Merma, A.G.; Torem, M.L.; Silvas, F.P.C. Biosurfactants: An overview of their properties, production, and application in mineral flotation. Resources 2024, 13, 81. [Google Scholar] [CrossRef]
- Gupta, K.; Sharma, V.K. Cutting-edge perspectives on biosurfactants: Implications for antimicrobial and biomedical applications. 3 Biotech 2024, 14, 297. [Google Scholar] [CrossRef]
- Abhyankar, I.; Hirlekar, S.; Prabhune, A.; Nisal, A. Bridging the gap: An investigation of biosurfactants-polymer systems. Curr. Opin. Colloid Interface Sci. 2024, 72, 101806. [Google Scholar] [CrossRef]
- Kamalesh, T. Advances in stabilization of metallic nanoparticle with biosurfactants-a review on current trends. Heliyon 2024, 10, 9. [Google Scholar] [CrossRef]
- Wang, Y.; Qiao, S.; Niu, Y. Review on current research on biosynthesis of biosurfactants and the regulation influenced by metal ions. J. Microbiol. Biotechnol. 2025, 35, 2503031. [Google Scholar] [CrossRef]
- Bisht, K.; Tyagi, U.; Bajpai Tripathy, D. Biosurfactants—An overview. Macromol. Symp. 2024, 413, 2200203. [Google Scholar] [CrossRef]
- Fu, L.; Ren, Z.; Chen, W.; Liu, Q.; Ye, M.; Qiu, X.; Shao, M. Polyetheramine enhanced biosurfactant/biopolymer flooding for enhanced oil recovery. J. Mol. Liq. 2024, 411, 125757. [Google Scholar] [CrossRef]
- Thakur, V.; Baghmare, P.; Verma, A.; Verma, J.S.; Geed, S.R. Recent progress in microbial biosurfactants production strategies: Applications, technological bottlenecks, and future outlook. Bioresour. Technol. 2024, 408, 131211. [Google Scholar] [CrossRef]
- Silva, M.D.G.C.; Medeiros, A.O.; Converti, A.; Almeida, F.C.G.; Sarubbo, L.A. Biosurfactants: Promising biomolecules for agricultural applications. Sustainability 2024, 16, 449. [Google Scholar] [CrossRef]
- Shaji, A.; Thamarai, P.; Deivayanai, V.C.; Saravanan, A.; Yaashikaa, P.R. Progress in sustainable remediation: Utilizing biosurfactants for eco-friendly contaminant cleanup. Bioresour. Technol. Rep. 2024, 27, 101901. [Google Scholar] [CrossRef]
- Tripathy, D.B. Biosurfactants: Green frontiers in water remediation. ACS EST Water 2024, 4, 4721–4740. [Google Scholar] [CrossRef]
- Patil, H.V.; Badgujar, N.P.; Suresh, S.; Kulkarni, R.D.; Nagaraj, K. Sustainable bio-based surfactants: Advances in green chemistry and environmental applications. Mater. Today Commun. 2025, 48, 113583. [Google Scholar] [CrossRef]
- Wojtowicz, K.; Steliga, T.; Skalski, T.; Kapusta, P. Influence of biosurfactants on the efficiency of petroleum hydrocarbons biodegradation in soil. Sustainability 2025, 17, 6520. [Google Scholar] [CrossRef]
- Al-Sakkaf, M.K.; Onaizi, S.A. Effects of emulsification factors on the characteristics of crude oil emulsions stabilized by chemical and Biosurfactants: A review. Fuel 2024, 361, 130604. [Google Scholar] [CrossRef]
- Ali, A.; Aziz, A.R.A.; Ismael, M.A.; Alqaed, S. Biosurfactants as an alternative eco-friendly solution for water-in-diesel emulsions-A review paper. Heliyon 2024, 10, 17. [Google Scholar] [CrossRef]
- Bjerk, T.R.; Severino, P.; Jain, S.; Marques, C.; Silva, A.M.; Pashirova, T.; Souto, E.B. Biosurfactants: Properties and applications in drug delivery, biotechnology and ecotoxicology. Bioengineering 2021, 8, 115. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.; Siddiqui, A.J.; Hamadou, W.S.; Surti, M.; Awadelkareem, A.M.; Ashraf, S.A.; Adnan, M. Inhibition of bacterial adhesion and antibiofilm activities of a glycolipid biosurfactant from Lactobacillus rhamnosus with its physicochemical and functional properties. Antibiotics 2021, 10, 1546. [Google Scholar] [CrossRef]
- Puyol McKenna, P.; Naughton, P.J.; Dooley, J.S.; Ternan, N.G.; Lemoine, P.; Banat, I.M. Microbial biosurfactants: Antimicrobial activity and potential biomedical and therapeutic exploits. Pharmaceuticals 2024, 17, 138. [Google Scholar] [CrossRef] [PubMed]
- Lourenço, M.; Duarte, N.; Ribeiro, I.A. Exploring biosurfactants as antimicrobial approaches. Pharmaceuticals 2024, 17, 1239. [Google Scholar] [CrossRef]
- Thomas, G.E.; Brant, J.L.; Campo, P.; Clark, D.R.; Coulon, F.; Gregson, B.H.; McKew, B.A. Effects of dispersants and biosurfactants on crude-oil biodegradation and bacterial community succession. Microorganisms 2021, 9, 1200. [Google Scholar] [CrossRef]
- Kashif, A.; Rehman, R.; Fuwad, A.; Shahid, M.K.; Dayarathne, N.; Jamal, A.; Choi, Y. Current advances in the classification, production, properties and applications of microbial biosurfactants—A critical review. Adv. Colloid Interface Sci. 2022, 306, 102718. [Google Scholar] [CrossRef]
- Wang, Q.; Wang, R.; Zhang, Q.; Zhao, C.; Zhou, X.; Zheng, H.; Yan, Z. Application of biomass corrosion inhibitors in metal corrosion control: A review. Molecules 2023, 28, 2832. [Google Scholar] [CrossRef]
- Xiong, Y.; Cao, M. Application of surfactants in corrosion inhibition of metals. Curr. Opin. Colloid Interface Sci. 2024, 73, 101830. [Google Scholar] [CrossRef]
- Abouzeid, F.; Alshammery, S. Surfactants in electrochemical applications: A review. GSC Adv. Res. Rev. 2025, 22, 151–161. [Google Scholar] [CrossRef]
- Lavanya, M.; Machado, A.A. Surfactants as biodegradable sustainable inhibitors for corrosion control in diverse media and conditions: A comprehensive review. Sci. Total Environ. 2024, 908, 168407. [Google Scholar] [CrossRef]
- Zhu, Y.; Free, M.L.; Woollam, R.; Durnie, W. A Review of surfactants as corrosion inhibitors and associated modeling. Prog. Mater. Sci. 2017, 90, 159–223. [Google Scholar] [CrossRef]
- Shaker, L.M.; Al-Amiery, A. Surfactant-based strategies for corrosion protection: Mechanisms, efficiency, and environmental considerations. Next Nanotechnol. 2025, 8, 100294. [Google Scholar] [CrossRef]
- Ostapenko, G.I.; Kalashnikova, N.A. To the nature of surfactant adsorption onto metallic Surfaces: Interaction with metal or hydrophobic effect? adsorption of hexylamine on platinum. J. Electrochem. Soc. 2022, 169, 116502. [Google Scholar] [CrossRef]
- Verma, C.; Hussain, C.M.; Quraishi, M.A.; Alfantazi, A. Green surfactants for corrosion control: Design, performance and applications. Adv. Colloid Interface Sci. 2023, 311, 102822. [Google Scholar] [CrossRef]
- Stanley, G.; Mark, A.V.; David, A.C. Method of Using Biosurfactants as Acid Corrosion Inhibitors in Well Treatment Operations. U.S. Patent 2016237334A1, 18 August 2016. [Google Scholar]
- Li, Z.; Xu, D.; Yuan, X.; Li, Z.; Wang, F. Application of Rhamnolipid as Environment-Friendly Microbial Corrosion Inhibitor. C.N. Patent 114645279A, 21 June 2022. [Google Scholar]
- Petit, M.; Finke, A.; Goyon, V. Use of at least One Amphipatic Biosurfactant as An Alkaline Corrosion Inhibitor. E.P. Patent 4098726A1, 7 December 2022. [Google Scholar]
- Farmer, S.; Alibek, K.; Adams, K.; Karathur, K.N. Multifunctional Composition for Enhanced Oil Recovery, Improved Oil Quality and Prevention of Corrosion. U.S. Patent 12065613B2, 20 August 2024. [Google Scholar]
- Chen, W.; Liu, M.; Jiang, Y. Medical Instrument Antirust Agent Based on Organic Corrosion Inhibitor and Preparation Method of Medical Instrument Antirust Agente. C.N. Patent 117165947A, 5 December 2023. [Google Scholar]
- Ma, X.; Liu, J.; Fang, X.; Shao, J.; Lu, X. Cleaning Agent Containing Sophorolipid as Well as Preparation Method and Application of Cleaning Agent. C.N. Patent 117363429A, 9 January 2024. [Google Scholar]
- Amir, M.; Au, Y.H.C.; Kortney, T.; Richard, A. Biosurfactants for Iron and Zinc Sulfide Scale Remediation and Control. U.S. Patent 2025075118A1, 6 March 2025. [Google Scholar]
- Jing, X.; Fu, D.; Wang, Y. Aviation Engine Carbon Deposit Cleaning Agent and Preparation Method Thereof. C.N. Patent 112680289A, 20 April 2021. [Google Scholar]
- Wu, J.; Liu, Y.; Huang, J.; Yang, J.; Liu, Z.; He, H.; Mei, M.; Zhang, Q.; Wang, H. Bio-Based Efficient Cleaning Rust Remover Material and Preparation Method Thereof. C.N. Patent 119040907A, 29 November 2024. [Google Scholar]
- Olivia, R.; Ang, C.H.; Clotilda, P.; Caroline, M.; Rudy, T.; Joe, N. Corrosion inhibition of mild steel bars by biosurfactant produced by Penicillium citrinum. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2023; Volume 1135, p. 012057. [Google Scholar] [CrossRef]
- Iravani, D.; Farhadian, A.; Seddik, N.B.; Sharifi, R.; Rahimi, A.; Raissouni, I.; Akbarinezhad, E. Optimizing biosurfactant structure: Experimental and theoretical investigation of the influence of hydroxyl groups on sour corrosion mitigation. J. Ind. Eng. Chem. 2025, 144, 625–641. [Google Scholar] [CrossRef]




| Corrosion Type | Material/Environment Studied | Aim of the Study | Reference |
| General or uniform corrosion | Pipelines in petroleum and gas sector | Optimization of Weibull distribution for risk calculation | [66] |
| Marine structural steel | Installation of high-strength E690 steel corrugated sandwich panel | [67] | |
| Bimetallic or galvanic corrosion (GC) | ADC12 Aluminum alloy and copper | Study of GC between ADC12 alloy and Cu in a 3.5% (w/w) NaCl solution | [68] |
| Screw joints | Study of GC of an Al coating in contact with a 55Al-Zn coating in an atmospheric environment | [69] | |
| Pitting corrosion (PC) | Reinforcing bars | Evaluation of the tensile behavior of reinforcing bars with simulated pitting corrosion | [70] |
| Duplex stainless steel 2205 | Investigation of the effect of tensile elastic stress on PC mechanism and passive film of steel in a sulfide-containing NaCl solution | [71] | |
| Crevice corrosion (CC) | 90/10 Cu-Ni alloy | Evaluation of alloy susceptibility to CC caused by Desulfovibrio vulgaris | [72] |
| X70 Pipeline steel | Study of electroactive Shewanella sp. in accelerating steel CC in a marine environment | [73] | |
| Intergranular corrosion (IC) | Superaustenitic stainless steel | Study of the effect of molybdenum on steel IC | [74] |
| Sensitized type 304 stainless steel | Study of the role of MnS in steel IC and depassivation | [75] | |
| Erosion corrosion (EC) | As-cast TC4 cast and LPBF TC4 | Study of cavitation erosion corrosion properties of as-cast TC4 and LPBF TC4 in 0.6 mol/L NaCl solution | [76] |
| Ni2FeCrMo0.2HEA | Evaluation of the effect of sand particle size on alloy resistance to EC in seawater | [77] | |
| Stress corrosion cracking (SCC) | Austenitic stainless steel | Study of the influence of grain size and boundary type on steel intergranular SCC | [78] |
| 2205 Duplex stainless steel | Study of the behavior and corrosion mechanism of steel under applied polarization potential | [79] |
| Inhibitors | Compound | Material | Corrosive Medium | Results | Reference |
|---|---|---|---|---|---|
| Organic | Benzimidazole with tryptophan units | Carbon steel | 1.0 M HCl | Inhibition efficiency (IE) achieved 86.68% | [154] |
| 3-Chloro-4-morpholin-4-yl-1,2,5-thiadiazole | Mild steel | 1.0 M HCl | The compound significantly reduced mild steel corrosion, reaching 96% IE | [155] | |
| 2,4-Dinitrophenyl-hydrazine | Aluminum | 1.0 M HCl | The compound proved to be a highly efficient inhibitor, with 88.9% IE | [156] | |
| 11-(2-Chlorophenyl)-3,3-dimethyl-2,3,4,5,10,11-hexahydro-1H-dibenzo[be][1,4]diazepin-1-one | Carbon steel | 1.0 M HCl | IE reached 94.4% | [157] | |
| Inorganic | Lignin phosphate | Carbon steel | 3.5% (w/w) NaCl | The corrosion rate was reduced by approximately 54.2% | [158] |
| Sodium phosphate (Na3PO4) | Carbon steel | 0.6 M Cl− | IE reached 91.7% | [159] | |
| Sodium phosphate and sodium dodecyl sulfate | Magnesium AZ91 | 3.5% (w/w) NaCl | The use of the two compounds together resulted in an initial IE of 99.7% | [160] | |
| Phytic acid and sodium phosphate | SLM 304L steel | 3.5% (w/w) NaCl | The synergistic effect allowed for a maximum IE of 92.35% | [161] | |
| Green | Leaf extract of Andrographis paniculata | Mild steel with the following composition (% by weight): 0.38 Si, 0.42 Cr, 0.12 P, 0.26 Ni, 0.43 Mn, 0.12 C, 0.42 Cu | H2SO4 | The extract, at a concentration of 4000 ppm, ensured an IE of 95.14% | [162] |
| Extract from Cassia fistula leaves | Carbon steel in simulated concrete pore solution | 0.5 mol/L NaCl | The results revealed a maximum IE of 85.21% (from mass loss) and 92.4% (from potentiodynamic polarization) | [163] | |
| Powder from the bark of Punica granatum | Carbon steel bars in reinforced concrete structures | 3.5% (w/w) NaCl | The mass loss test revealed a maximum IE of 83.11% | [164] | |
| Extract from reflux rubber seeds | Cold-rolled steel | 1.0 M HCl | The extract achieved an IE of 95.09% | [165] | |
| Biosurfactant from Pseudomonas cepacia CCT6659 | Carbon steel | Abiotic and biotic systems | The biosurfactant IE was 80 and 87% for abiotic and biotic systems, respectively | [166] | |
| Rhamnolipid | Carbon steel X70 | Simulated seawater | The corrosion rate was reduced by approximately 72.2% | [167] |
| NO. | Patent Number | Patent Title | Description | Year | Reference |
|---|---|---|---|---|---|
| 1 | US2016237334A1 | Method of using biosurfactants as acid corrosion inhibitors in well treatment operations | The corrosive effects resulting from well treatment applications are inhibited and/or prevented by introducing into the well a formulation containing a selected biosurfactant as a corrosion inhibitor | 2016 | [206,223] |
| 2 | CN114645279A | Application of rhamnolipid as environment-friendly microbial corrosion inhibitor | The present invention describes the application of a rhamnolipid as a green biocorrosion inhibitor and belongs to the technical field of microbial corrosion protection. The rhamnolipid is used to inhibit metal material biocorrosion | 2022 | [207,224] |
| 3 | EP4098726A1 | Use of at least one amphipathic biosurfactant as an alkaline corrosion inhibitor | The present invention describes a water-based alkaline preparation containing at least one glycolipid biosurfactant and at least one water-insoluble lubricant, and to its use in processing fluids | 2022 | [225] |
| 4 | US12065613B2 | Multifunctional composition for enhanced oil recovery, improved oil quality and prevention of corrosion | The present invention discloses compositions and methods for concurrently improving oil recovery and quality by reducing sulfur-bearing substances, and hindering or lowering corrosion of oil and gas production equipment. A multifunctional composition is provided, comprising an antimicrobial biosurfactant component, an ammonium salt, and ammonium hydroxide | 2024 | [226] |
| 5 | CN117165947A | Medical instrument antirust agent based on organic corrosion inhibitor and preparation method of medical instrument antirust agent | The invention describes an antirust agent for medical instruments based on an organic corrosion inhibitor and its method of preparation. The rust inhibitor is prepared from an organic corrosion inhibitor (phospholipid), a dispersant, a biosurfactant, an auxiliary agent, and deionized water | 2023 | [227] |
| 6 | CN117363429A | Cleaning agent containing sophorolipid as well as preparation method and application of cleaning agent | The invention describes a sophorolipid-based cleaning agent, with excellent properties such as cleaning power, corrosion inhibition, sterilization, and bacteriostasis, which can perform effective cleaning and protection functions on automotive windshields and similar surfaces | 2024 | [228] |
| 7 | US2025075118A1 | Biosurfactants for iron and zinc sulfide scale remediation and control | The present invention describes scale inhibitors and/or dispersants comprising biosurfactants, as well as methods of their use to remove scale deposits | 2025 | [229] |
| 8 | CN112680289A | Aviation engine carbon deposit cleaning agent and preparation method thereof | The present invention describes a cleaning agent for carbon deposits in aviation engines using a sophorolipid biosurfactant, among other components, to protect all engine assemblies against corrosion, oxidation, and discoloration. At the same time, the agent promises to completely remove carbon deposits and dirt from all engine assemblies, ensuring effective cleaning | 2021 | [230] |
| 9 | CN119040907A | Bio-based efficient cleaning rust remover material and preparation method thereof | The present invention, which belongs to the technical field of metal surface treatment, provides an efficient biologically based rust remover and a method for preparing it | 2024 | [231] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Oliveira, K.W.d.; Faccioli, Y.E.d.S.; de Araújo, G.P.; Converti, A.; da Silva, R.d.C.F.S.; Sarubbo, L.A. From the Problem of Corrosion to Green Solutions: The Role of Biosurfactants as Anti-Corrosion Agents. Materials 2026, 19, 743. https://doi.org/10.3390/ma19040743
Oliveira KWd, Faccioli YEdS, de Araújo GP, Converti A, da Silva RdCFS, Sarubbo LA. From the Problem of Corrosion to Green Solutions: The Role of Biosurfactants as Anti-Corrosion Agents. Materials. 2026; 19(4):743. https://doi.org/10.3390/ma19040743
Chicago/Turabian StyleOliveira, Kaio Wêdann de, Yslla Emanuelly da Silva Faccioli, Gleice Paula de Araújo, Attilio Converti, Rita de Cássia Freire Soares da Silva, and Leonie Asfora Sarubbo. 2026. "From the Problem of Corrosion to Green Solutions: The Role of Biosurfactants as Anti-Corrosion Agents" Materials 19, no. 4: 743. https://doi.org/10.3390/ma19040743
APA StyleOliveira, K. W. d., Faccioli, Y. E. d. S., de Araújo, G. P., Converti, A., da Silva, R. d. C. F. S., & Sarubbo, L. A. (2026). From the Problem of Corrosion to Green Solutions: The Role of Biosurfactants as Anti-Corrosion Agents. Materials, 19(4), 743. https://doi.org/10.3390/ma19040743








