3.1. Chemical Composition and Sulfur Retention
All synthesized glasses appeared visually homogeneous and exhibited a very dark burgundy coloration. Their amorphous nature was confirmed by X-ray diffraction, which revealed only a broad diffuse halo without any sharp Bragg reflections (
Figure 1). Minor differences in coloration were observed between the Zn-free and Zn-containing glasses, suggesting compositional or structural variations induced by Zn addition. The chemical compositions determined by XRF, together with the nominal batch compositions for comparison, are summarized in
Table 1.
XRF analysis revealed that partial substitution of MgO by ZnO led to a higher sulfur content retained in the glass structure. Glasses containing ZnO consistently exhibited higher measured SO3 contents than their Zn-free counterparts. This enhanced sulfur retention is accompanied by a reduction in the measured P2O5 content and by a pronounced loss of ZnO during melting.
In the Zn-free glass series, the SiO
2 content decreases systematically from 43.7 to 40.2 mol%, while P
2O
5 increases from 3.8 to 6.1 mol% across the 39-5S to 37-5S compositions, reflecting the intended substitution of SiO
2 by P
2O
5 network formers. The MgO content remains close to the nominal 28 mol%, confirming its role as the dominant alkaline-earth modifier. In all Zn-free glasses, the measured SO
3 contents are slightly below the nominal value of 5 mol%, which is consistent with partial sulfur volatilization under reducing melting conditions [
22].
In contrast, the Zn-containing glasses exhibit markedly lower ZnO concentrations than the nominal values, indicating substantial zinc loss during melting. This behavior is consistent with the high volatility of zinc under reducing conditions, where ZnO can be partially reduced to metallic Zn and subsequently evaporate. Thermodynamic and kinetic studies have demonstrated that ZnO reduction and zinc volatilization are strongly promoted at elevated temperatures in carbon-rich or reducing environments, particularly in multicomponent silicate melts [
15,
23,
24]. The reduced ZnO concentrations measured by XRF therefore indicate that a considerable fraction of zinc was lost during melting rather than incorporated into the final glass structure.
XRF analysis also revealed the presence of several minor oxide components at concentrations typically below 1–2 mol%, in addition to the major glass-forming and modifying oxides. These minor constituents originate from raw-material impurities, crucible corrosion, and high-temperature processing effects. In particular, the presence of Al
2O
3 is attributed to partial dissolution of alumina crucibles during melting, with slightly higher Al
2O
3 contents observed in Zn-containing glasses. While Al
2O
3 incorporation can influence glass structure and crystallization behavior, its concentration in the present compositions remains sufficiently low that it does not obscure the intended compositional trends or affect the interpretation of the main thermal, structural, and electrical results [
24,
25].
3.2. Thermal Behavior of the Glasses (DSC)
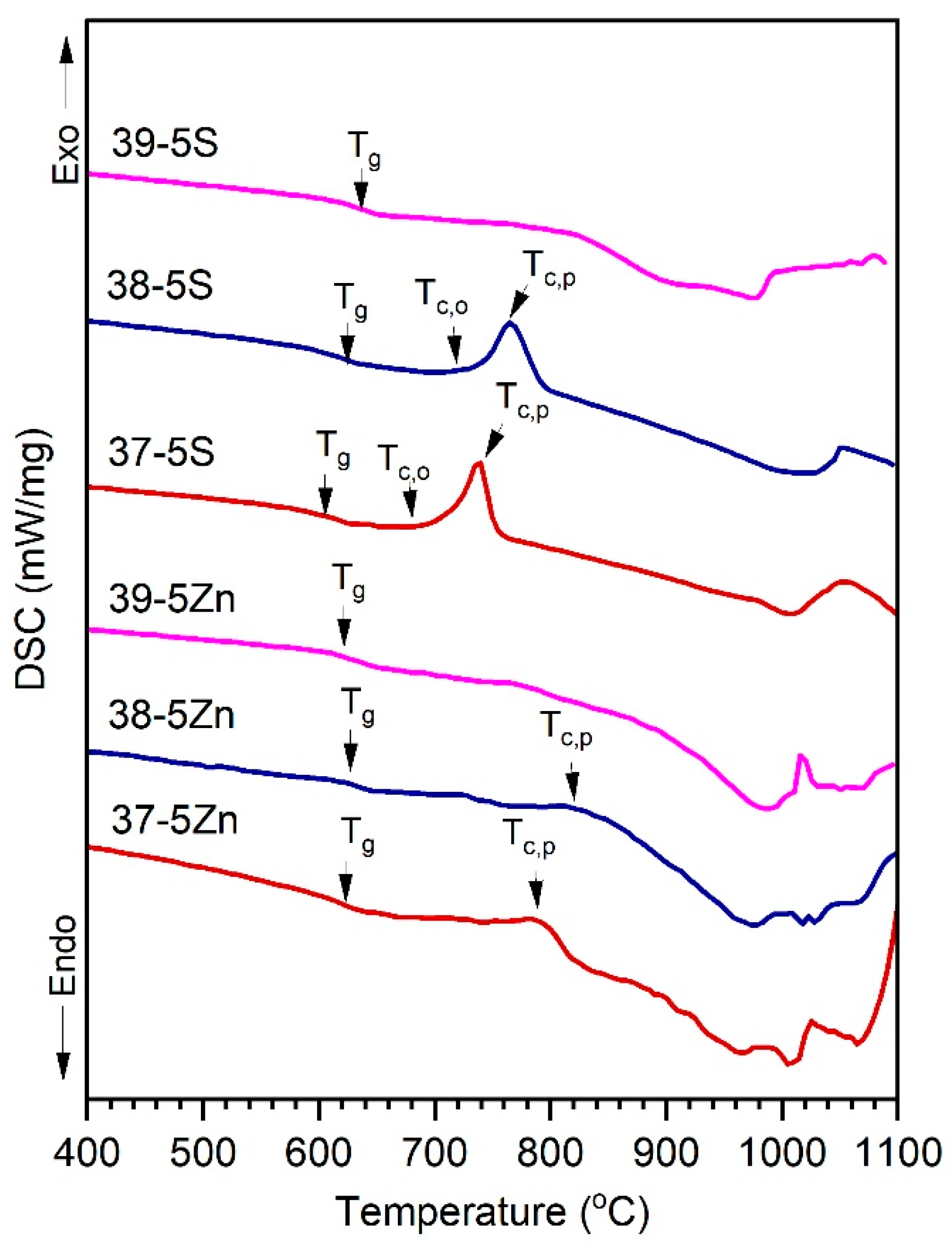
The DSC curves of the Zn-free and Zn-containing glasses are shown in
Figure 2, and the characteristic thermal parameters are summarized in
Table 2. All compositions exhibit a distinct endothermic step associated with the glass transition. In the Zn-free series, the glass transition temperature (T
g) decreases systematically from 626 to 597 °C with increasing P
2O
5. This behavior is characteristic of silicate–phosphate glasses and reflects progressive network depolymerization caused by the incorporation of phosphate units at the cost of silicate units and the associated increase in non-bridging oxygens coordinated to modifier cations [
12,
26]. Similar reductions in T
g with increasing phosphate concentration have been widely reported for multicomponent oxide glasses containing alkaline and alkaline-earth modifiers [
12]. In contrast, the Zn-containing glasses exhibit a different T
g response to increasing P
2O
5 content. Although the lowest T
g is observed for the 39-5Zn composition, T
g increases with further phosphate addition. This reversal of the T
g–P
2O
5 trend indicates a change in the structural role of phosphate units in the presence of Zn
2+. Zinc is known to act as an intermediate oxide in silicate and phosphate glasses, forming more directional Zn–O bonds and participating in intermediate or mixed network-forming structural units that locally reinforce the network and counteract depolymerization effects [
27,
28].
The crystallization behavior also differs markedly between the two series. The Zn-free glasses 38-5S and 37-5S display sharp and well-defined crystallization exotherms, indicative of relatively high atomic mobility and diffusion-controlled crystal growth, as commonly observed in modifier-rich glass networks [
29]. In contrast, the corresponding Zn-containing glasses exhibit broadened and weak crystallization features that are shifted to higher temperatures. Such suppression and broadening of crystallization exotherms are characteristic of glasses with increased network rigidity and reduced long-range diffusion, as previously reported for Zn-modified oxide and oxysulfide glasses [
30].
The processing window (ΔT) calculated from the DSC data confirms that Zn-containing glasses possess enhanced kinetic stability and a reduced tendency toward crystallization compared to their Zn-free counterparts. The change in heat capacity at the glass transition (ΔC
p) does not show a strictly monotonic compositional dependence; however, the generally lower ΔC
p values observed in the Zn-containing glasses indicate reduced configurational entropy and restricted local structural rearrangements near T
g, consistent with the thermodynamic interpretation of the glass transition in rigid glass networks [
30,
31]. At higher temperatures, both glass series exhibit endothermic effects associated with melting or thermal decomposition. The upward shift in these effects observed for the Zn-containing glasses reflects the higher cohesive energy of Zn-containing structural units compared to Mg- and K-modified environments, which requires additional thermal energy to disrupt the glass network [
24].
The observed reduction in configurational heat capacity (ΔCp), broadening and weakening of crystallization exotherms, and increase in melting-related endothermic temperatures with increasing ZnO content can be attributed to the intermediate role of Zn
2+ in the glass network [
27,
28]. Zinc is known to form relatively strong and directional Zn–O bonds and to participate in mixed or intermediate structural units in silicate–phosphate glasses, which locally reinforce the glass structure and reduce the number of accessible configurational states near the glass transition [
27,
28]. This increased network rigidity limits long-range atomic diffusion and slows crystallization kinetics, leading to broadened and weaker crystallization exotherms shifted to higher temperatures [
18]. At higher temperatures, the enhanced cohesive energy associated with Zn-containing structural units requires additional thermal energy to disrupt the network, resulting in elevated melting or decomposition temperatures [
24]. Overall, the DSC results demonstrate that partial substitution of MgO by ZnO enhances the thermal stability of silicate–phosphate oxysulfide glasses by increasing network rigidity and slowing crystallization kinetics, while exerting only a moderate influence on the glass transition temperature.
3.3. Phase Composition of Devitrified Glasses (XRD)
Upon thermal treatment, glasses may undergo devitrification, as crystallization is thermodynamically favored due to the reduction in free energy associated with the transformation from a disordered amorphous structure to an ordered crystalline phase [
1,
2].
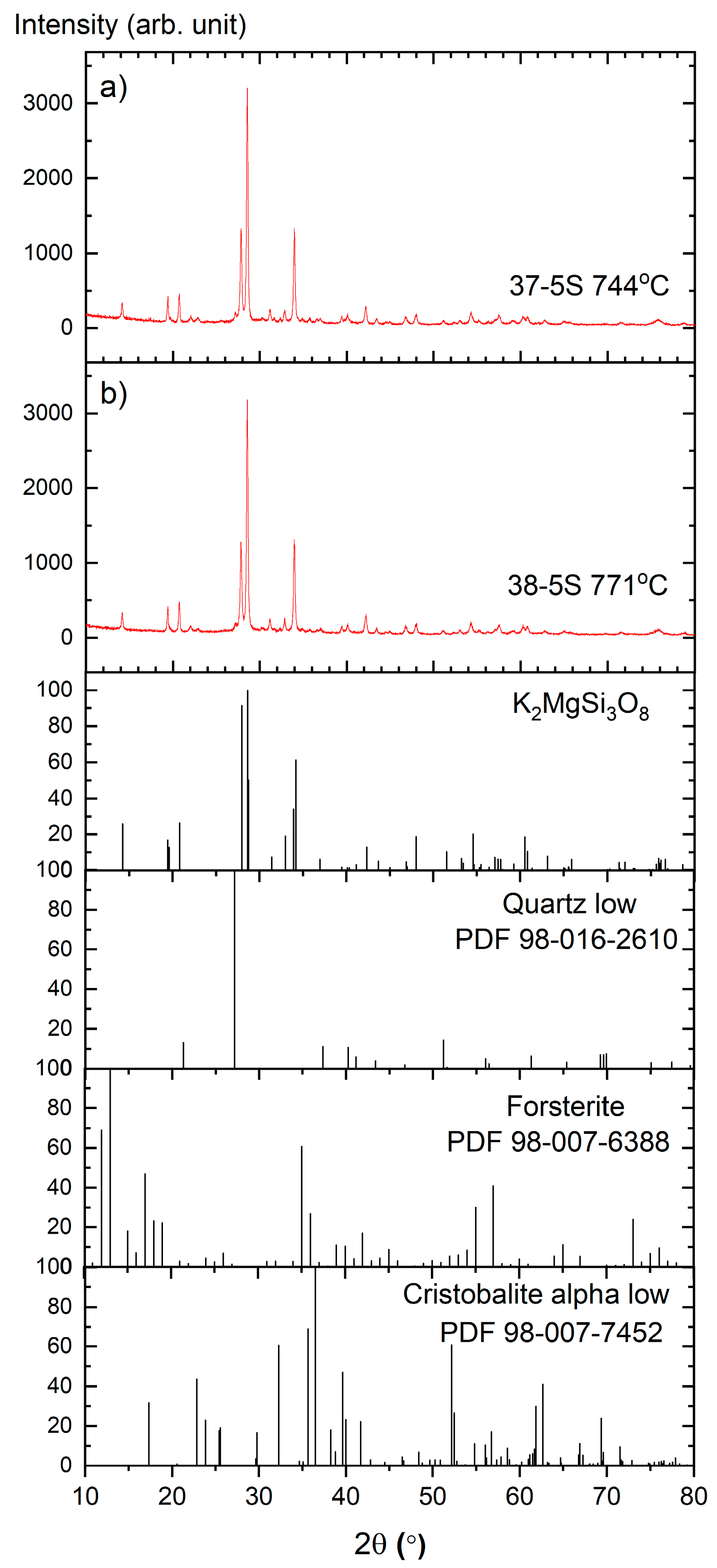
Figure 3 presents the X-ray diffraction (XRD) patterns of the Zn-free glasses after isothermal heat treatment, together with the corresponding Rietveld refinements. The 37-5S glass was annealed at 744 °C for 24 h (
Figure 3a), while the 38-5S glass was treated at 771 °C for 24 h (
Figure 3b). These temperatures were selected based on the crystallization events identified in the DSC curves (
Figure 1). The good agreement between the experimental diffraction patterns and the calculated profiles confirms the reliability of the phase identification and quantitative analysis.
Rietveld refinement revealed dipotassium magnesium trisilicate, K2MgSi3O8, as the dominant crystalline phase in both devitrified samples, accounting for 85.4% in the 37-5S glass and 83.7% in the 38-5S glass. Forsterite, Mg2SiO4 (ICDD PDF 98-007-6388), was identified as a secondary crystalline phase, with phase fractions of 10.9% and 12.5% for the 37-5S and 38-5S samples, respectively. Minor crystalline phases include low-temperature α-cristobalite, SiO2 (ICDD PDF 98-007-7452), present at 1.6% in the 37-5S glass and 1.8% in the 38-5S glass, as well as low-temperature quartz, SiO2 (ICDD PDF 98-016-2610), accounting for 1.9% and 2.1% in the respective samples. No sulfur-containing crystalline phases were detected within the detection limits of the XRD measurements.
The observed phase assemblage suggests that, upon devitrification, modifier cations are not uniformly distributed within the silicate–phosphate network but tend to segregate into localized regions or clusters. Such structural heterogeneity may lead to the formation of preferential pathways that facilitate ionic transport, in agreement with previous reports on mixed-modifier glass systems [
32].
The DSC curves of Zn-containing glasses exhibit weaker and broader crystallization effects shifted to higher temperatures, indicating reduced crystallization kinetics; therefore, devitrification was not achieved for these compositions under the heat-treatment conditions applied in this study.
3.5. Electrical and Dielectric Properties
Examining how impedance varies with frequency and temperature offers important insight into changes in the intrinsic (bulk) resistance of the glass samples. The impedance is a complex function, where the real component Z′ reflects the resistive behavior, and the imaginary component Z″ represents the capacitive response of the material. In electrochemical impedance spectroscopy (EIS) studies of glass, parameters such as bulk resistance, relaxation time, and activation energy can be extracted from the Nyquist plot by fitting equivalent electrical circuits. The bulk resistance is obtained from the high-frequency intercept on the real axis, the relaxation time from the characteristic frequency of the semicircle, and the activation energy from the temperature dependence of conductivity or relaxation time. The activation energy E
a from conductivity is related to long-range ion transport through the glass network, while the activation energy from relaxation time reflects localized ion hopping or short-range motion. Nyquist plots for 38-5S and 38-5Zn glasses are shown in
Figure 6a and
Figure 6b, respectively.
For glass 38-5S, the Nyquist plot contains two semicircular arcs and a Warburg element (
Figure 6a). The high-frequency arc corresponds to bulk processes, whereas the low-frequency arc is associated with the electrode–glass interface At higher temperatures, the Warburg-type response becomes more evident in the low-frequency region, reflecting enhanced ionic diffusion at the glass–electrode interface. The bulk resistance is determined from the high-frequency intercept of the first semicircle with the Z′ axis, while the local minimum in Z″ between the two semicircles marks the transition between bulk and interfacial processes.
In contrast, the Zn-containing 38-5Zn glass (
Figure 6b) exhibits a single, depressed semicircular arc without a detectable Warburg element. The absence of diffusion-controlled impedance suggests suppression of long-range ionic transport, consistent with restricted modifier mobility caused by the formation of stronger Zn–O/S structural units. This interpretation is in agreement with the reduced crystallization tendency observed by DSC [
27]. The semicircles do not originate at the Z′ = 0 point but are shifted away from the origin, indicating the presence of a series resistance and/or unresolved high-frequency processes, while the depressed shape reflects non-Debye relaxation associated with structural heterogeneity of the glass.
A suitable fitted EEC (Electrical Equivalent Circuit) model is essential for accurately capturing the electrical response of the different regions within the material. The fitted curves reproduce the experimental data closely. Each semicircle in the Nyquist diagram is modeled using a parallel R–C element. However, in both cases, the capacitor is replaced by a constant-phase element (CPE) to account for the depressed, non-ideal semicircles whose centers lie below the real axis. This behavior is characteristic of glassy and crystalline materials exhibiting non-Debye relaxation. In the classical Debye model, a single relaxation time generates a perfect semicircle in the complex impedance plane, with its center positioned directly on the real (Z′) axis. The semicircular arcs progressively contract with increasing temperature for both types of glasses. Elevated temperatures generate more space-charge carriers, enhance electron hopping, and thus confirm the semiconducting nature of the materials. The 38-5Zn glass shows significantly lower impedance values (both capacity and resistance) compared to the 38-5S glass.
Figure 7 shows the dependence of the electrical resistance of 38-5Zn and 38-5S glasses on temperature. Sample 38-5S exhibits significantly higher resistance values than 38-5Zn. The slope of the temperature dependence is constant, which suggests that only one electrical conduction mechanism is present in this temperature region.
All fitted data were validated using the Kramers–Kronig (K–K) test, confirming the physical consistency, linearity, and stability of the measured impedance spectra. The selected equivalent circuit models show R2 values show deviations below 10%.
The activation energy E
a of electrical resistivity was performed using the Arrhenius equation:
where
A is the pre-exponential constant, E
a represents the activation energy,
T is the absolute temperature, and
R is gas constant [
37]
The activation energy Ea was found to be 0.96 eV and 0.91eV for 38-5S and 38-5Zn, respectively, in the investigated temperature range and it was found to be consistent with the observed trend for conductivity of the glass materials. Based on the relatively high activation energy (~1 eV), ionic conduction is identified as the dominant transport mechanism in these materials.
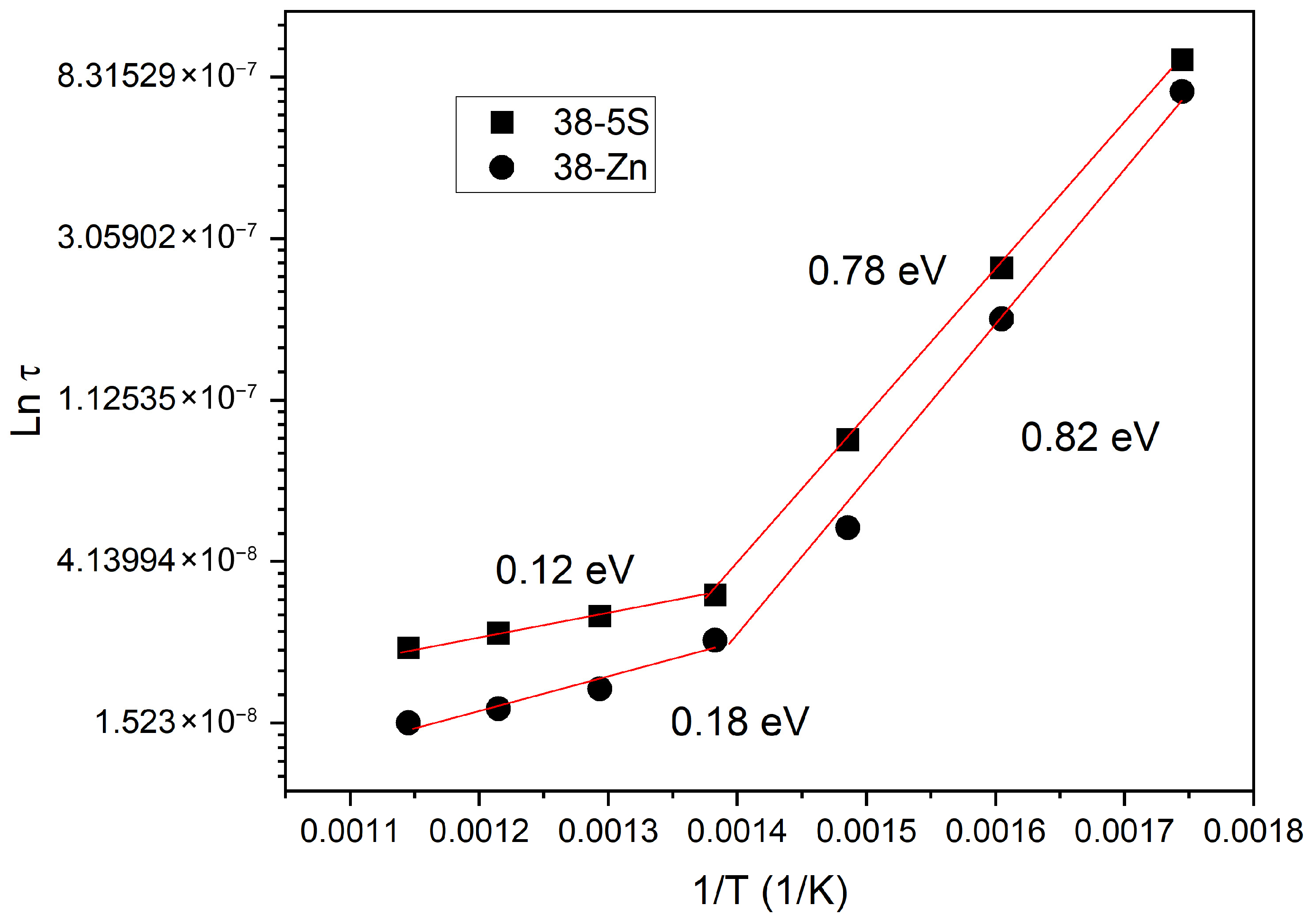
To determine the relaxation time at each temperature, the frequency at which −Z″ reaches its maximum
fmax was identified and present in
Figure 8. The relaxation time τ is calculated from this relaxation frequency using the relationship [
38]:
Figure 8 presents the variation in the relaxation time τ as a function of inverse temperature (1/
T). The analysis was carried out using a modified Arrhenius relation [
37,
39]:
where
τ0 is the pre-exponential constant.
The activation energy associated with the relaxation time () was estimated to be 0.78 eV and 0.82 eV for the 38-5S and 38-5Zn glasses, respectively, in the lower temperature region (≈300–450 °C). These values are characteristic of thermally activated long-range ionic hopping processes, where mobile modifier ions migrate between energetically favorable sites within the disordered glass network. Such activation energies are commonly reported for ion transport in oxide glasses and indicate that relaxation and electrical conduction are governed by the same charge-carrier dynamics in this temperature regime. In contrast, at higher temperatures (≈450–600 °C), the activation energy decreases significantly to 0.12 eV for 38-5S and 0.18 eV for 38-5Zn. This reduction suggests a transition to relaxation mechanisms associated with lower energy barriers, such as localized ionic motions, short-range hopping, or secondary (β-type) relaxation processes. At elevated temperatures, increased thermal energy allows charge carriers to access a broader distribution of pathways with reduced barriers, resulting in enhanced mobility and lower apparent activation energies.
Additionally, the electrical conductivity for 39-5S and 39-5Zn samples was studied at even lower temperatures, spanning from RT to 240 °C, see
Figure S1 in Supplemental Information. Similarly, to the 38-5S and 38-5Zn, a linear temperature dependence of DC conductivity in this temperature range was observed. The corresponding activation energies were estimated to be 0.77 eV and 0.73 eV for the 39-5S and 39-5Zn glasses, respectively. This is in line with thermally activated long-range ionic hopping processes within the disordered glass network.
The observed temperature-dependent change in activation energy reflects the disordered nature of the glass network and the coexistence of multiple relaxation mechanisms. The close correlation between relaxation behavior and electrical conductivity confirms that ionic transport plays a dominant role in both processes across the investigated temperature range.
3.5.1. Electrical Capacitance
Measurements of the electrical capacitance,
Cp, as a function of both frequency and temperature were carried out, and results are presented in
Figure 9. The responses of the two samples were similar, exhibiting a noticeable increase in capacitance only at the lowest frequencies, below 1 kHz, and at elevated temperatures above 400 °C. This behavior indicates that the dielectric response in this regime is strongly frequency- and temperature-dependent, with significant contributions arising primarily under conditions where slow polarization mechanisms or thermally activated processes become dominant. The sample 38-5Zn achieves higher values of
Cp than 38-5S. Such trends are characteristic of materials in which dipolar relaxation or defect- and modifier-related polarization mechanisms are pronounced at low frequencies and high temperatures.
3.5.2. Evolution of the Dielectric Losses
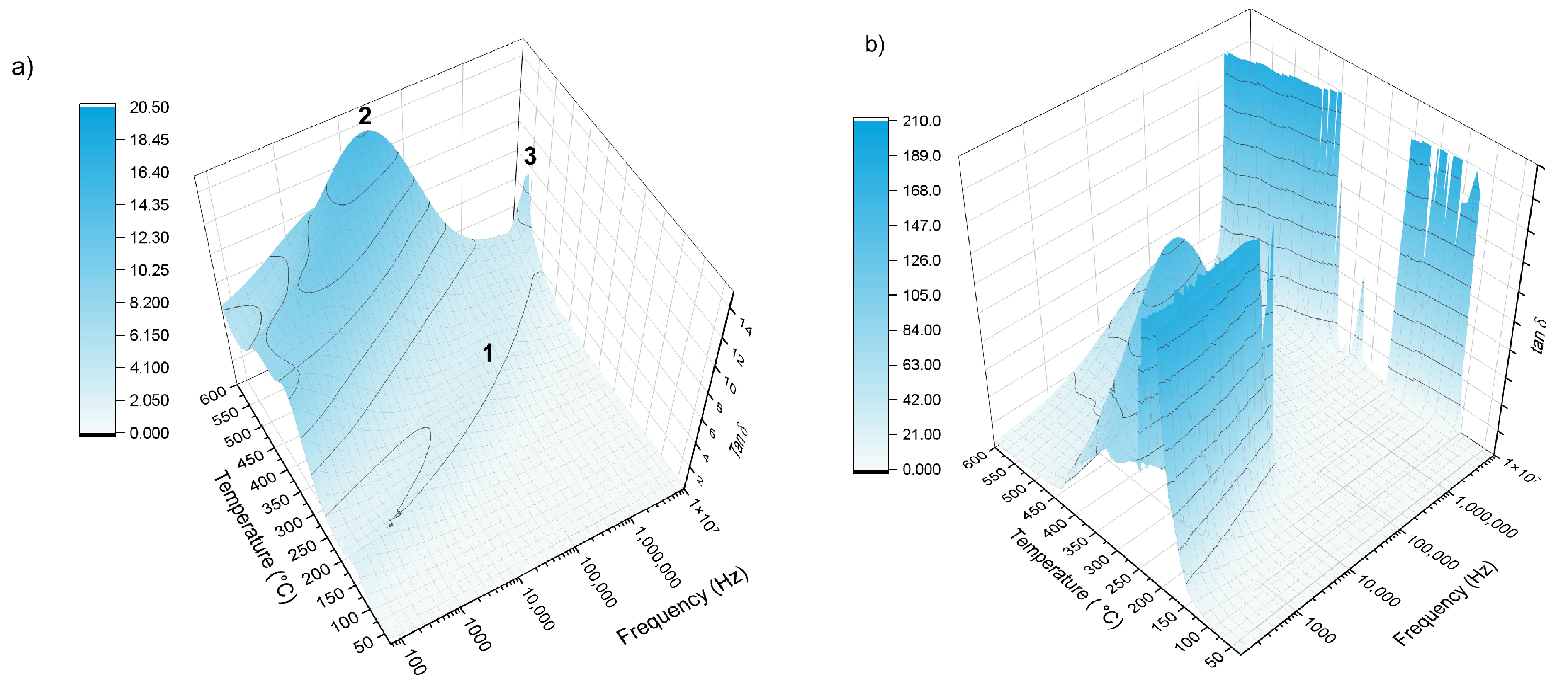
The dielectric losses tan δ (
Figure 10) spectrum represents the ratio of the imaginary to the real part of the permittivity (ε″/ε′) and thus describes how effectively the material dissipates electrical energy [
40]. The observed dependence of tan δ on temperature and frequency arises from the interplay between ionic conduction and dipolar relaxation mechanisms inherent to disordered glass networks [
40]. Generally, for glasses, this curve typically rises with temperature, exhibits a peak at a characteristic relaxation frequency, and then declines at higher frequencies [
41]. The low-frequency side of the peak is governed mainly by ionic conduction, while the maximum occurs when the field oscillation rate matches the characteristic reorientation time of dipolar groups within the glass structure, allowing maximum energy absorption. Beyond this relaxation frequency, at higher frequencies, the structural units of the glass cannot reorient fast enough to follow the applied field, leading to a continued decrease in dielectric loss. This behavior reflects the intrinsic relaxation dynamics of disordered glass networks and their limited ability to respond to very rapid electrical perturbations. Both samples exhibit distinct tan δ behaviors as functions of frequency and temperature, and in both cases the dielectric response is relatively complex.
For the first glass composition 38-5S (38SiO
2–9P
2O
5–28MgO–20K
2O–5SO
3), three characteristic regions can be identified in the tan δ spectra. Between 150–500 °C, the tan δ curves display a broad local maximum (marked as “1” in
Figure 10a) that resembles a single relaxation wave: extending across the entire measured frequency range. As the temperature increases, this maximum systematically shifts to higher frequencies, reaching maximum approximately 10 MHz at 500 °C. This behavior is typical of thermally activated dipolar or ionic relaxation, where increased temperature enhances the mobility of network modifiers (e.g., K
+, Mg
2+) and structural units within the mixed SiO
2–P
2O
5 network [
12,
42]. A second region shows global asymmetric maximum (“2”), more pronounced feature appears at around 550 °C, where the tan δ reaches a global maximum of about tan δ = 18 near 500 kHz. The peak shape is notably asymmetric, with the high-frequency side dropping more steeply than the low-frequency side. Such asymmetry often indicates the presence of distributed relaxation times—a signature of structural disorder and the coexistence of multiple relaxation mechanisms (ionic hopping, dipole reorientation, and localized structural rearrangements) within the glass [
42,
43].
The final region exhibits a sharp peak (“3”) was present in the high-temperature and high-frequency region. At still higher temperatures and frequencies, the spectra reveal a sharp, well-defined maximum. This sharp relaxation is commonly associated with fast ionic polarization processes or glass-transition-related structural dynamics, where the glass network becomes sufficiently softened to allow rapid local motion. In mixed-oxide phosphate–silicate glasses, this can involve accelerated mobility of modifier cations (particularly K
+) or rapid reorganization of SO
3-containing structural groups [
44]. For the Zn-doped 38SiO
2–9P
2O
5–28MgO–20K
2O–5SO
3 (38-5Zn) glass, the tan δ response within the same temperature and frequency range differs substantially from that of the undoped sample, showing overall much higher dielectric loss. Although both glasses share general features such as a mid-frequency maximum between approximately 50 kHz and 1 MHz above 450 °C and another maximum in the highest-frequency region at elevated temperatures, the Zn-containing glass displays two particularly strong relaxation peaks at lower temperatures that are absent in the original composition.
The first pronounced maximum appears between about 150 and 250 °C at around 1 kHz and reaches remarkably high tan δ values, indicating that Zn
2+ incorporation strongly modifies the mobility of ions and enhances dipolar polarization. Literature on Zn-modified silicate and phosphate glasses reports that ZnO has been reported to act as an intermediate oxide capable of modifying network connectivity and enhancing localized polarization and hopping processes and dielectric losses [
18]. The second strong maximum, observed between roughly 250 and 370 °C near the same 1 kHz frequency, suggests an additional thermally activated relaxation process involving more substantial structural rearrangements or cooperative dynamics of mixed silicate–phosphate units. Previous studies have shown that Zn-containing glasses can form Zn–O–Si and Zn–O–P linkages, creating localized polar structures that respond slowly to the external field and produce distinct relaxation modes [
12,
27]. These structural effects collectively increase the polarizability of the glass, introduce a more heterogeneous distribution of mobile ions, and generate additional dipolar and ionic relaxation pathways, resulting in a significantly more complex and pronounced tan δ behavior compared with the undoped glass. Although Zn-containing glasses exhibit higher dielectric losses, this does not imply enhanced long-range ionic conductivity. The increased tan δ reflects intensified localized polarization and short-range hopping processes within structurally heterogeneous regions of the glass network associated with Zn
2+ incorporation, while extended ion diffusion remains hindered [
40,
41].
The electrical conductivity mechanisms in the investigated glasses are strongly governed by the nature of modifier cations and their bonding environments within the mixed oxysulfide network. In the Zn-free glasses, charge transport is dominated by thermally activated ionic hopping, primarily involving K
+ ions and, to a lesser extent, Mg
2+ ions. The presence of relatively ionic Mg-O, Mg-S, and K-S bonds creates flexible modifier-rich regions and continuous percolation pathways that facilitate long-range ionic diffusion. This mechanism is supported by the appearance of two semicircular arcs and a Warburg diffusion element in the Nyquist plots, as well as by broad low-frequency tan δ maxima characteristic of ionic conduction in disordered glass networks [
45,
46].
In contrast, substitution of MgO by ZnO markedly alters the conduction mechanism. Zinc behaves as an intermediate oxide that locally increases network rigidity through more directional metal–oxygen interactions and mixed network-forming structural units. These effects reduce free volume and disrupt continuous ionic pathways, thereby suppressing long-range diffusion. As a result, the Zn-containing glasses exhibit a single depressed semicircle without a Warburg contribution, indicating that electrical transport is dominated by localized hopping and dipolar polarization processes rather than extended ionic motion [
12,
26]. Despite their higher dielectric losses, the Zn-containing glasses display increased resistivity, demonstrating that enhanced tan δ arises from localized polarization within Zn-rich structural domains rather than improved macroscopic conductivity [
17].
Overall, Mg-rich glasses favor diffusion-controlled ionic transport, whereas Zn incorporation induces a transition toward polarization-dominated, short-range charge dynamics. This mechanistic distinction is consistent with the DSC results, which show suppressed crystallization in Zn-containing compositions. Taken together, XAS, DSC, and EI spectroscopy consistently demonstrate that Mg promotes structural ordering that facilitates crystallization through ionic Mg-O/S bonding, whereas Zn2+ incorporation stabilizes the glass network by increasing local rigidity and suppressing long-range ionic transport, thereby enhancing thermal stability and promoting localized polarization dynamics.
3.5.3. The Evolution of Dielectric Constant
The real part of the dielectric constant ε′ as a function of frequency and temperature for both glass compositions exhibits a clear increase with rising temperature, particularly in the low-frequency region (
Figure 11). This behavior is typical of ionically conducting glasses and is associated with thermally activated hopping of mobile charge carriers, predominantly K
+ ions, which contribute to space-charge and interfacial polarization at low frequencies. As temperature increases, the enhanced mobility of these charge carriers leads to a pronounced rise in ε′, especially below 1 kHz.
With increasing frequency, ε′ decreases steadily for both compositions. At higher frequencies, charge carriers and dipolar units are unable to reorient rapidly enough to follow the alternating electric field, resulting in a reduced contribution to polarization and a progressive decline in the dielectric constant. This frequency-dependent behavior reflects the limited response time of mobile ions and dipoles in the disordered glass network.
Compared with the Zn-free glass, the Zn-containing composition exhibits systematically lower e′ values over the investigated frequency and temperature ranges. This trend indicates reduced space-charge polarization and diminished long-range ionic mobility upon ZnO incorporation. The decrease in e′ is attributed to increased network rigidity and reduced free volume, which hinder the formation of continuous alkali-ion migration pathways and limit the contribution of mobile charge carriers to dielectric polarization.
Overall, impedance spectroscopy demonstrates that Zn incorporation fundamentally alters the dielectric response of the glasses. While Zn-containing compositions exhibit enhanced dielectric loss and multiple relaxation processes due to increased localized polarization and short-range hopping, long-range ionic diffusion is suppressed, as evidenced by the absence of Warburg-type behavior in the impedance spectra. These electrical characteristics are consistent with the DSC results, which indicate enhanced thermal stability and suppressed crystallization in Zn-containing glasses. Together, the electrical and thermal data confirm that Zn2+ acts as an intermediate oxide that stabilizes the glass network by restricting extended ionic transport while promoting heterogeneous local polarization dynamics.