Comparison of Mechanical and Corrosion Properties Between Coarse-Grained and Ultrafine-Grained High-Strength Aluminum Alloys
Highlights
- A 7A52 aluminum alloy was processed by multi-axial forging and aging.
- The fine-grained alloy shows higher strength, elongation and corrosion resistance.
- The strengthening is due to dislocation multiplication and grain size reduction.
- The improvement in grain boundary precipitates enhances corrosion resistance.
- This study provides a feasible strategy to improve the comprehensive properties of high-strength aluminum alloys
- The severe plastic deformation and heat treatment optimizes the microstructure of alloys, thus enhances their overall performance.
- Multi-axial forging and aging treatment features excellent engineering applicability and can be extended to industrial production of high-performance aluminum alloy components.
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
4. Conclusions
- (1)
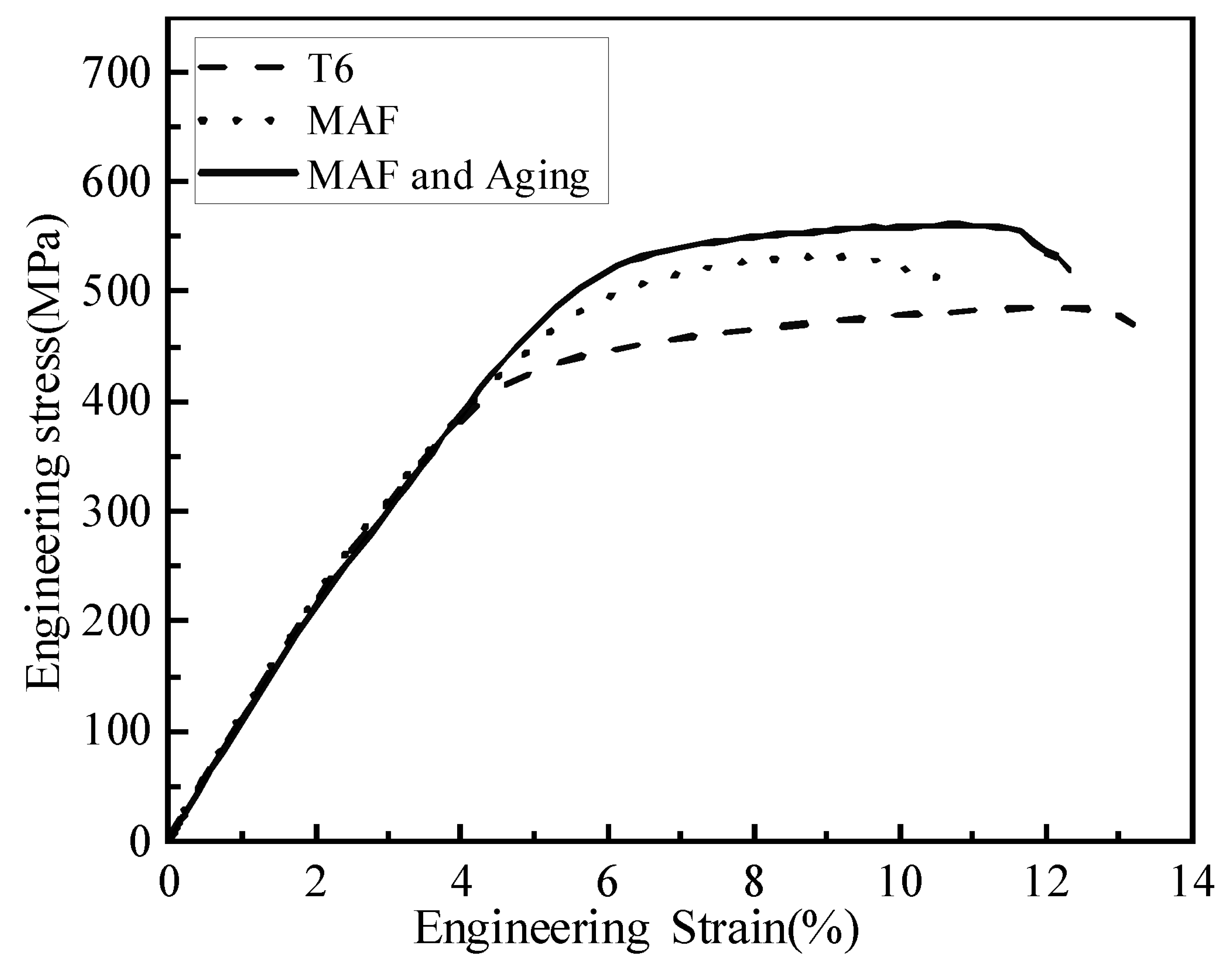
- After MAF, the strength and elongation reached 532 MPa and 10.6%. The strength greatly increased and the elongation sharply decreased in the alloy. The strength was improved because of the dislocation multiplication and the grain size reduction.
- (2)
- After MAF and aging, the strength and elongation reached 561 MPa and 12.3%. The strength was enhanced further due to precipitation strengthening. The elongation was improved by reducing the forging stresses. During aging, recovery and grain growth occur, which also results in higher elongation. Moreover, the precipitates were dispersed within the UFG, which increased the dislocation accumulation and work hardening. Therefore, the elongation could be enhanced as well.
- (3)
- The UFG structure 7A52 aluminum alloy produced by MAF and aging with excellent corrosion resistance was mainly due to GBPs’ coarse and discontinuous distribution.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, J.; Song, B.; Wei, Q.; Bourell, D.; Shi, Y. A review of selective laser melting of aluminum alloys: Processing, microstructure, property and developing trends. J. Mater. Sci. Technol. 2019, 35, 270–284. [Google Scholar] [CrossRef]
- Azarniya, A.; Taheri, A.K.; Taheri, K.K. Recent advances in ageing of 7xxx series aluminum alloys: A physical metallurgy perspective. J. Alloys Compd. 2019, 781, 945–983. [Google Scholar] [CrossRef]
- Ran, F.Q.; Chai, L.H.; Gao, K.Y.; Nie, Z.R.; Chen, Z.Y. Influence of various aging treatments on microstructure, strength and corrosion behaviour of high Zn content Al–Zn–Mg–Cu alloy. Corros. Eng. Sci. Technol. 2014, 49, 712–718. [Google Scholar] [CrossRef]
- He, Z.; Gao, W.; Lu, Z.; Feng, C. Effects of Heat Treatment on Microstructure and Properties of 7A85 Aluminium Alloy. J. Mater. Eng. 2015, 43, 13–18. [Google Scholar] [CrossRef]
- Tsai, T.C.; Chuang, T.H. Role of grain size on the stress corrosion cracking of 7475 aluminum alloys. Mater. Sci. Eng. A 1997, 225, 135–144. [Google Scholar] [CrossRef]
- Oliveira, A.F.; de Barros, M.C.; Cardoso, K.R.; Travessa, D.N. The effect of RRA on the strength and SCC resistance on AA7050 and AA7150 aluminium alloys. Mater. Sci. Eng. A 2004, 379, 321–326. [Google Scholar] [CrossRef]
- Su, R.; Qu, Y.; Li, R. Effect of Aging Treatments on the Mechanical and Corrosive Behaviors of Spray-Formed 7075 Alloy. J. Mater. Eng Perform. 2014, 23, 3842–3848. [Google Scholar] [CrossRef]
- Valiev, R.Z.; Islamgaliev, R.K.; Alexandrov, I.V. Bulk nanostructured materials from severe plastic deformation. Prog. Mater. Sci. 2000, 45, 103–189. [Google Scholar] [CrossRef]
- Cao, H.; Yang, Y.; Zhang, X.; Ma, J.; Zhang, T.; Zhang, Z. Microstructure Evolution of 7075 Aluminum Alloy by Rotary. Materials 2022, 15, 1445. [Google Scholar] [CrossRef]
- Karimi, S.; Fakhar, N.; Faraji, M.; Fereshteh-Saniee, F. Simultaneous improvement of mechanical strength and corrosion resistance in aluminum alloy 5083 via severe plastic deformation. Mater. Chem. Phys. 2024, 313, 128755. [Google Scholar] [CrossRef]
- Eizadjou, M.; Manesh, H.D.; Janghorban, K. Microstructure and mechanical properties of ultra-fine grains (UFGs) aluminum strips produced by ARB process. J. Alloys Compd. 2009, 474, 406–415. [Google Scholar] [CrossRef]
- Kim, Y.-S.; Seok Yu, H.; Hyuk Shin, D. Low sliding-wear resistance of ultrafine-grained Al alloys and steel having undergone severe plastic deformation. Int. J. Mater. Res. 2009, 100, 871–874. [Google Scholar] [CrossRef]
- Padap, A.K.; Chaudhari, G.P.; Nath, S.K.; Pancholi, V. Ultrafine-grained steel fabricated using warm multiaxial forging: Microstructure and mechanical properties. Mater. Sci. Eng. A 2009, 527, 110–117. [Google Scholar] [CrossRef]
- GB/T 7998-2005; Test Method for Intergranular Corrosion of Aluminium Alloy. Standards Press of China: Beijing, China, 2005.
- GB/T 22639-2008; Test Method of Exfoliation Corrosion for Wrought Aluminium and Aluminium Alloys. Standards Press of China: Beijing, China, 2008.
- Valiev, R.Z.; Alexandrov, I.V.; Zhu, Y.T.; Lowe, T.C. Paradox of Strength and Ductility in Metals Processed Bysevere Plastic Deformation. J. Mater. Res. 2002, 17, 5–8. [Google Scholar] [CrossRef]
- Koch, C. Optimization of strength and ductility in nanocrystalline and ultrafine grained metals. Scr. Mater. 2003, 49, 657–662. [Google Scholar] [CrossRef]
- Huang, L.; Chen, K.; Li, S. Influence of grain-boundary pre-precipitation and corrosion characteristics of inter-granular phases on corrosion behaviors of an Al–Zn–Mg–Cu alloy. Mater. Sci. Eng. B 2012, 177, 862–868. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, Y.; Bian, G.; Zhang, Y. Electrochemical behavior and corrosion mechanism of anodized 7B04 aluminum alloy in acid NaCl environments. J. Alloys Compd. 2021, 886, 161231. [Google Scholar] [CrossRef]
- Zuo, J.; Hou, L.; Shi, J.; Cui, H.; Zhuang, L.; Zhang, J. Enhanced plasticity and corrosion resistance of high strength Al-Zn-Mg-Cu alloy processed by an improved thermomechanical processing. J. Alloys Compd. 2017, 716, 220–230. [Google Scholar] [CrossRef]
- Du, S.; Song, Y.; He, Y.; Wei, C.; Chen, R.; Guo, S.; Liang, W.; Lei, S.; Liu, X. Evolution of Microstructure and Mechanical Properties of Ti-6Al-4V Alloy under Heat Treatment and Multi-Axial Forging. Materials 2024, 17, 1060. [Google Scholar] [CrossRef] [PubMed]
- Horita, Z.; Ohashi, K.; Fujita, T.; Kaneko, K.; Langdon, T.G. Achieving High Strength and High Ductility in Precipitation-Hardened Alloys. Adv. Mater. 2005, 17, 1599–1602. [Google Scholar] [CrossRef]
- Zhao, Y.H.; Liao, X.Z.; Cheng, S.; Ma, E.; Zhu, Y.T. Simultaneously Increasing the Ductility and Strength of Nanostructured Alloys. Adv. Mater. 2006, 18, 2280–2283. [Google Scholar] [CrossRef]
- Dieter, G.E. Mechanical Metallurgy, 3rd ed.; McGraw-Hill: New York, NY, USA, 1986; p. 289. [Google Scholar]
- Li, H.; Cao, F.; Guo, S.; Ning, Z.; Liu, Z.; Jia, Y.; Scudino, S.; Gemming, T.; Sun, J. Microstructures and properties evolution of spray-deposited Al-Zn-Mg-Cu-Zr alloys with scandium addition. J. Alloys Compd. 2017, 691, 482–488. [Google Scholar] [CrossRef]
- Wang, D.; Ni, D.R.; Ma, Z.Y. Effect of pre-strain and two-step aging on microstructure and stress corrosion cracking of 7050 alloy. Mater. Sci. Eng. A 2008, 494, 360–366. [Google Scholar] [CrossRef]
- Deschamps, A.; De Geuser, F.; Horita, Z.; Lee, S.; Renou, G. Precipitation kinetics in a severely plastically deformed 7075 aluminium alloy. Acta Mater. 2014, 66, 105–117. [Google Scholar] [CrossRef]
- Kamp, N.; Sinclair, I.; Starink, M.J. Toughness-Strength Relations in the Overaged 7449 Al-Based Alloy. Met. Mater Trans A 2002, 33, 1125–1136. [Google Scholar] [CrossRef]
- Li, Z.; Xiong, B.; Zhang, Y.; Zhu, B.; Wang, F.; Liu, H. Investigation on strength, toughness and microstructure of an Al–Zn–Mg–Cu alloy pre-stretched thick plates in various ageing tempers. J. Mater. Process. Technol. 2009, 209, 2021–2027. [Google Scholar] [CrossRef]
- Han, N.; Zhang, X.; Liu, S.; Ke, B.; Xin, X. Effects of pre-stretching and ageing on the strength and fracture toughness of aluminum alloy 7050. Mater. Sci. Eng. A 2011, 528, 3714–3721. [Google Scholar] [CrossRef]
- Chen, S.; Chen, K.; Dong, P.; Ye, S.; Huang, L. Effect of heat treatment on stress corrosion cracking, fracture toughness and strength of 7085 aluminum alloy. Trans. Nonferrous Met. Soc. China 2014, 24, 2320–2325. [Google Scholar] [CrossRef]
- Pan, S.; Yuan, J.; Linsley, C.; Liu, J.; Li, X. Corrosion behavior of nano-treated AA7075 alloy with TiC and TiB2 nanoparticles. Corros. Sci. 2022, 206, 110479. [Google Scholar] [CrossRef]
- Najjar, D.; Magnin, T.; Warner, T.J. Influence of critical surface defects and localized competition between anodic dissolution and hydrogen effects during stress corrosion cracking of a 7050 aluminium alloy. Mater. Sci. Eng. A 1997, 238, 293–302. [Google Scholar] [CrossRef]
- Sarkar, B.; Marek, M.; Starke, E.A. The effect of copper content and heat treatment on the stress corrosion characteristics of Ai-6Zn-2Mg-X Cu alloys. Met. Trans. A 1981, 12, 1939–1943. [Google Scholar] [CrossRef]
- McNaughtan, D.; Worsfold, M.; Robinson, M.J. Corrosion product force measurements in the study of exfoliation and stress corrosion cracking in high strength aluminium alloys. Corros. Sci. 2003, 45, 2377–2389. [Google Scholar] [CrossRef]
- Wloka, J.; Hack, T.; Virtanen, S. Influence of temper and surface condition on the exfoliation behaviour of high strength Al–Zn–Mg–Cu alloys. Corros. Sci. 2007, 49, 1437–1449. [Google Scholar] [CrossRef]









| Zn | Mg | Mn | Cr | Cu | Fe | Zr | Ti | Si | Ni | Al |
|---|---|---|---|---|---|---|---|---|---|---|
| 4.63 | 2.46 | 0.24 | 0.17 | 0.12 | 0.12 | 0.098 | 0.069 | 0.042 | 0.004 | Bal |
| Samples | Ecorr (V) | Icorr (A/cm2) | Rcorr (Ω cm2) |
|---|---|---|---|
| T6 | −0.896 | 6.508 × 10−7 | 15,807.1 |
| MAF + aging | −0.886 | 5.714 × 10−7 | 22,633.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zhao, X.; Shao, Y.; Xu, G.; Liu, T.; Liu, D.; Lin, G. Comparison of Mechanical and Corrosion Properties Between Coarse-Grained and Ultrafine-Grained High-Strength Aluminum Alloys. Materials 2026, 19, 407. https://doi.org/10.3390/ma19020407
Zhao X, Shao Y, Xu G, Liu T, Liu D, Lin G. Comparison of Mechanical and Corrosion Properties Between Coarse-Grained and Ultrafine-Grained High-Strength Aluminum Alloys. Materials. 2026; 19(2):407. https://doi.org/10.3390/ma19020407
Chicago/Turabian StyleZhao, Xiaolian, Yiwen Shao, Guoxiang Xu, Tong Liu, Dong Liu, and Guoqiang Lin. 2026. "Comparison of Mechanical and Corrosion Properties Between Coarse-Grained and Ultrafine-Grained High-Strength Aluminum Alloys" Materials 19, no. 2: 407. https://doi.org/10.3390/ma19020407
APA StyleZhao, X., Shao, Y., Xu, G., Liu, T., Liu, D., & Lin, G. (2026). Comparison of Mechanical and Corrosion Properties Between Coarse-Grained and Ultrafine-Grained High-Strength Aluminum Alloys. Materials, 19(2), 407. https://doi.org/10.3390/ma19020407





