Phase Diagrams and Thermal Properties of Fatty Acid Ternary Eutectic Mixtures for Latent Heat Thermal Energy
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
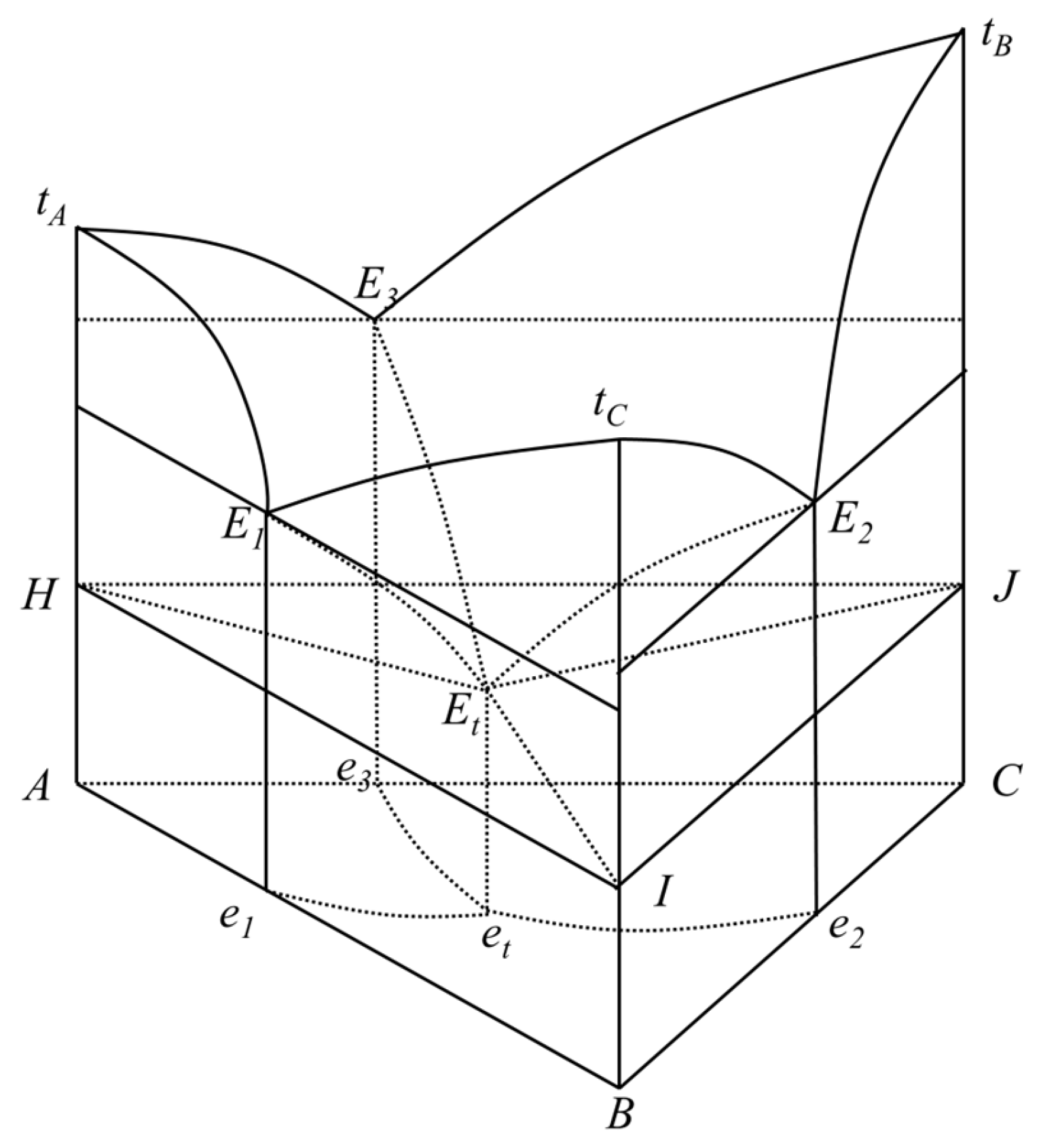
2.2. Theoretical Prediction of Eutectic Properties
2.3. Preparation of Ternary Fatty Acid Mixtures
2.4. Characterization
3. Results and Discussion
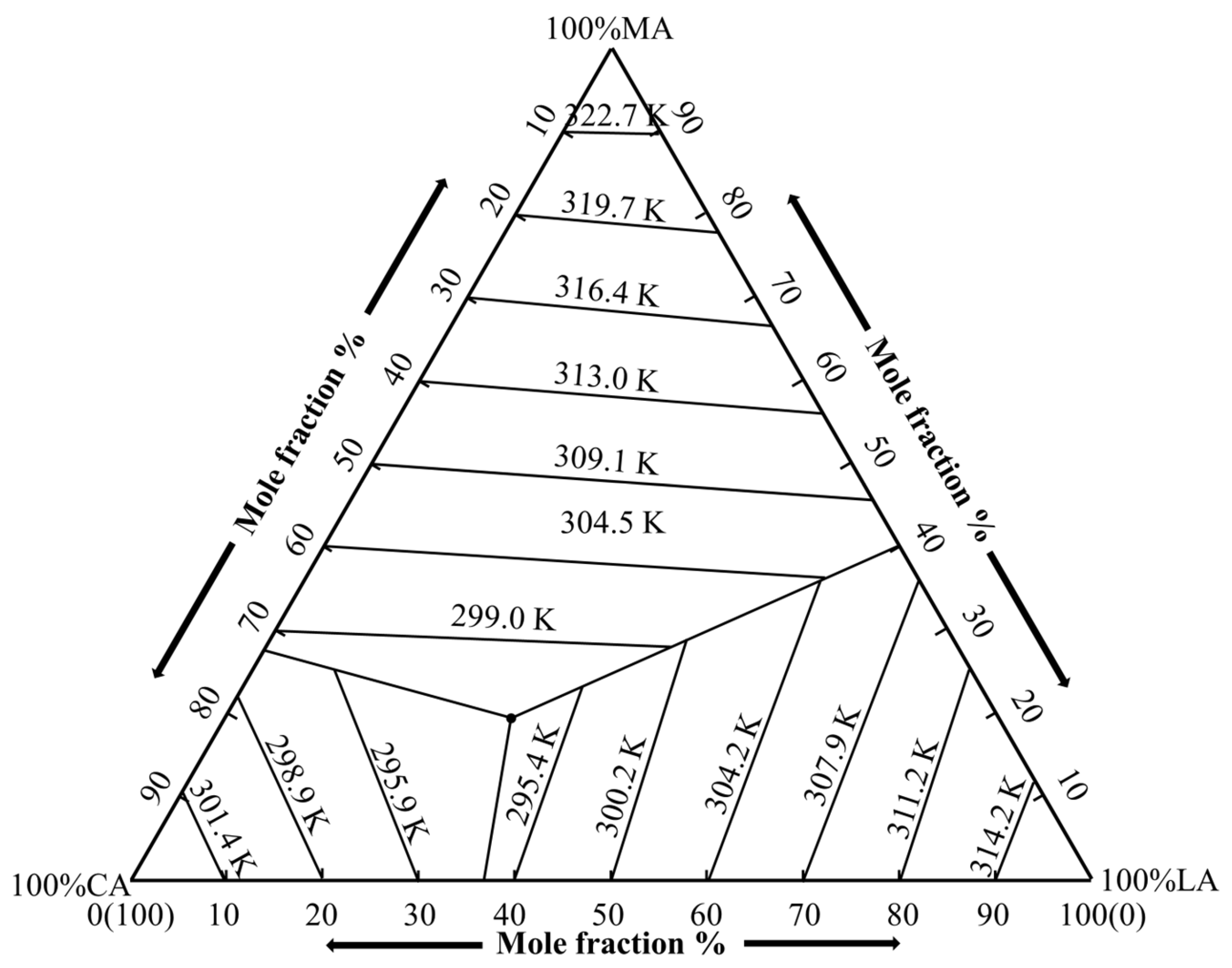
3.1. Phase Diagram of the Ternary Eutectic System
3.2. Determination of Eutectic Point of Ternary Fatty Acid Mixtures by the Schroder Equation
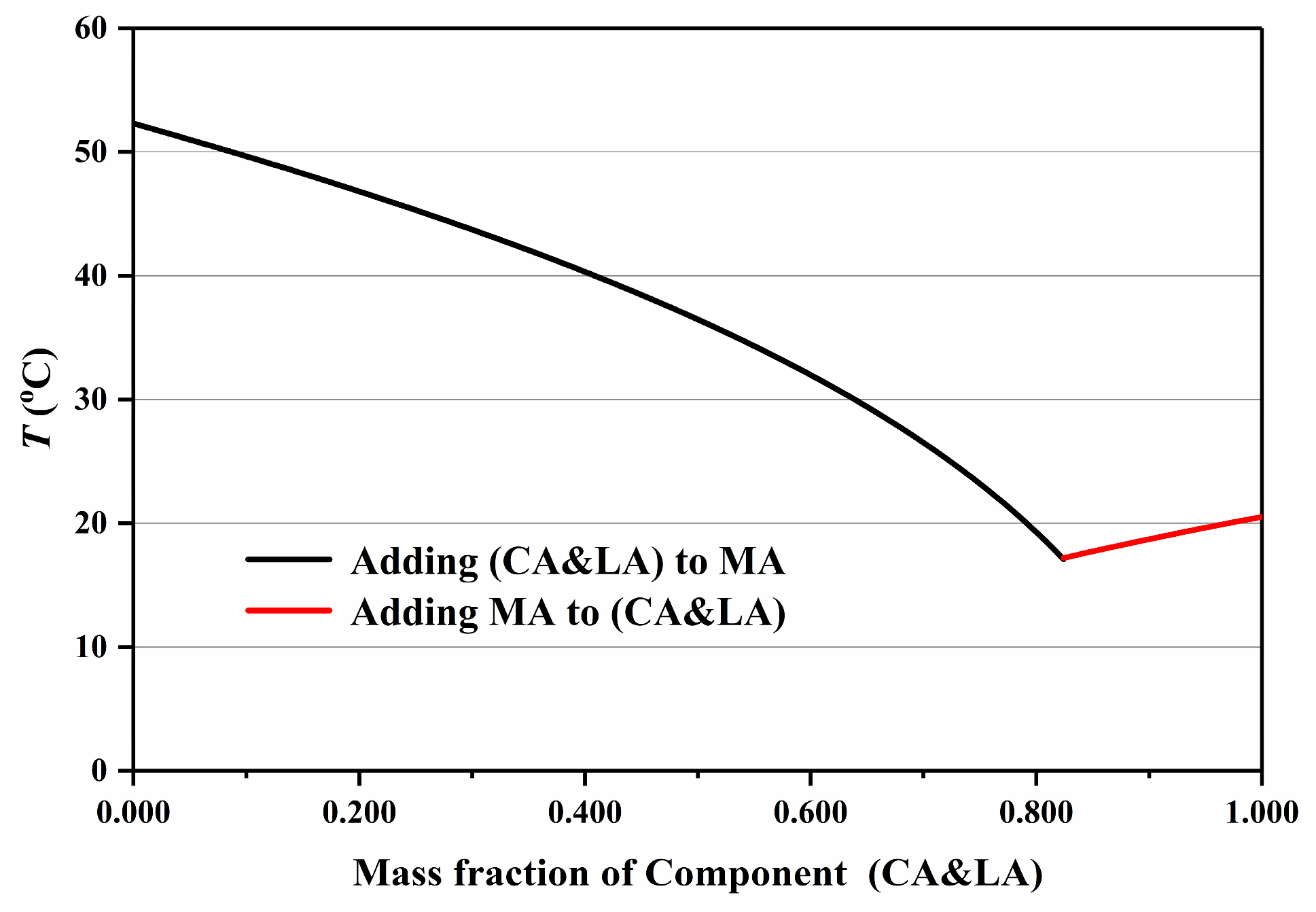
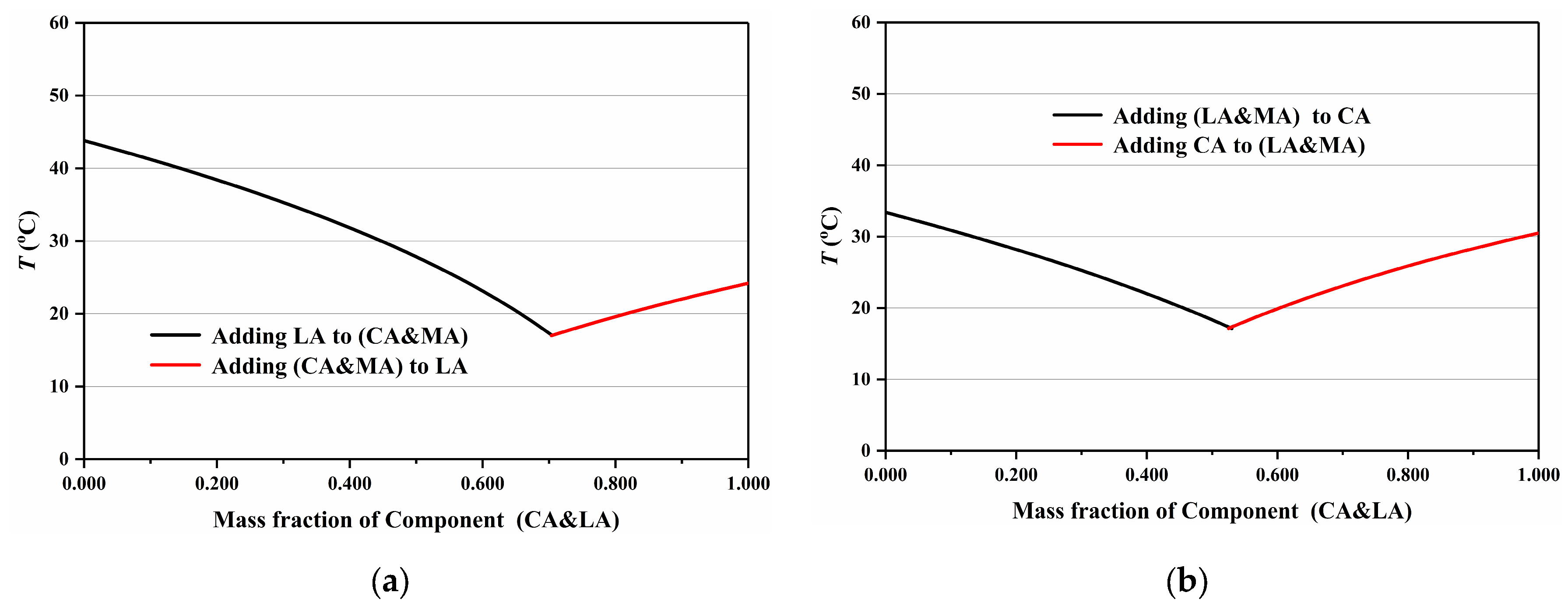
3.3. Determination of Eutectic Point of Ternary Fatty Acid Mixtures by the “Pseudo-Binary” Method
3.4. Thermal Properties of Ternary Fatty Acid Mixtures
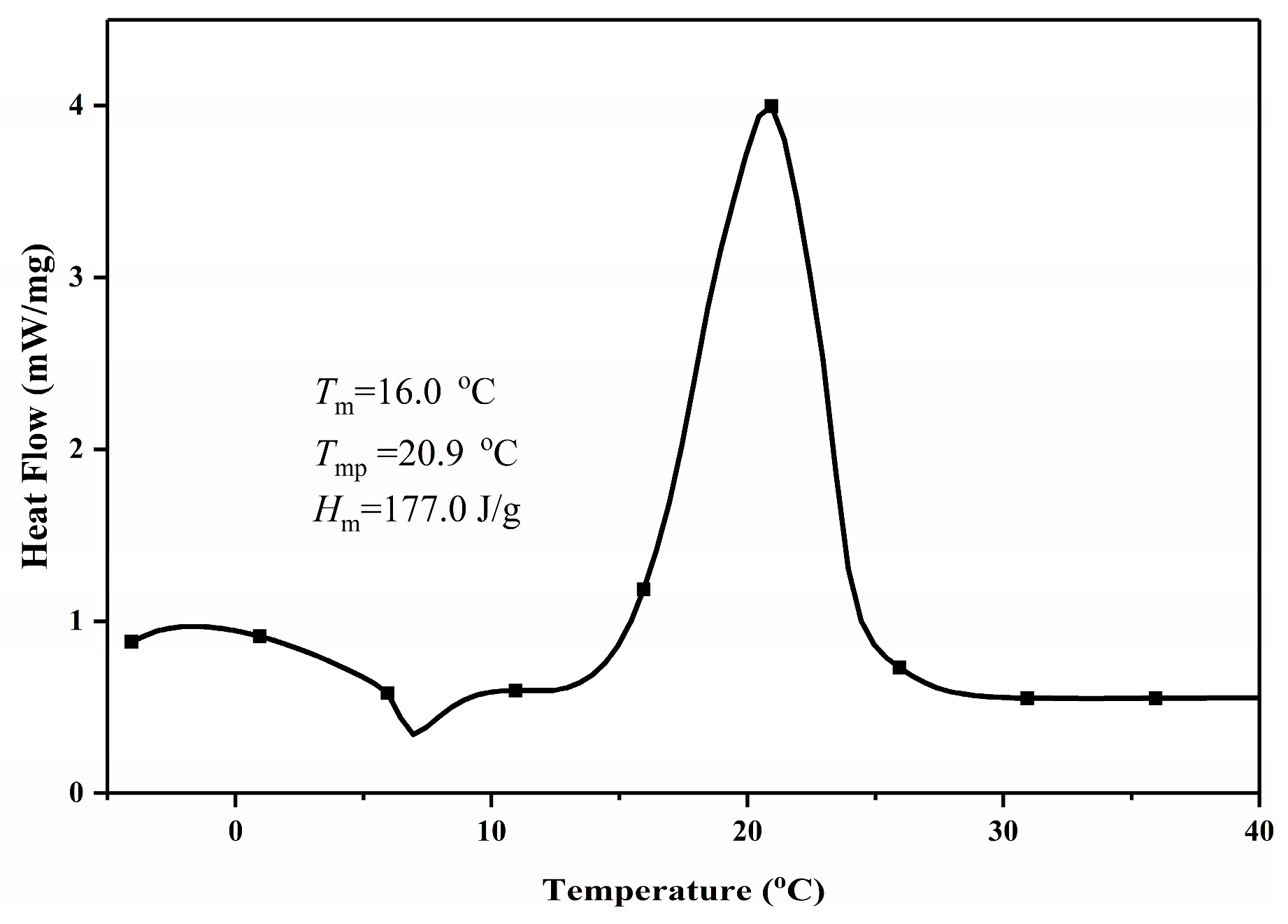
3.4.1. DSC Analysis
3.4.2. FT-IR Spectroscopy Analysis
3.4.3. Thermal Stability Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Abolfazl, G.; Alireza, A.; Roohollah, F. Optimal allocation of energy storage systems, wind turbines and photovoltaic systems in distribution network considering flicker mitigation. Appl. Energy 2022, 319, 119253. [Google Scholar] [CrossRef]
- Levin, T.; Bistline, J.; Sioshansi, R.; Cole, W.J.; Kwon, J.; Burger, S.P.; Crabtree, G.W.; Jenkins, J.D.; O’nEil, R.; Korpås, M.; et al. Energy storage solutions to decarbonize electricity through enhanced capacity expansion modeling. Nat. Energy 2023, 8, 1199–1208. [Google Scholar] [CrossRef]
- International Energy Agency (IEA). Renewables 2024; IEA: Paris, France, 2024; Available online: https://www.iea.org/reports/renewables-2024 (accessed on 27 December 2025).
- Yao, X.; Yi, B.; Yu, Y.; Fan, Y.; Zhu, L. Economic analysis of grid integration of variable solar and wind power with conventional power system. Appl. Energy 2020, 264, 114706. [Google Scholar] [CrossRef]
- Zhou, D.; Xiao, S.; Liu, Y. Preparation and characterization of innovative cement mortar incorporating fatty acid/expanded graphite composite phase change material for thermal energy storage. Sci. Rep. 2024, 14, 67573. [Google Scholar] [CrossRef]
- Duzcan, A.; Kara, Y.A. Optimization of a multi-generation renewable energy supply system for a net-zero energy building with pcm-integrated trombe wall. J. Energy Storage 2025, 134, 117966. [Google Scholar] [CrossRef]
- Zhou, D.; Xiao, S.; Xiao, X.; Liu, Y. Preparation, phase diagrams and characterization of fatty acids binary eutectic mixtures for latent heat thermal energy storage. Separations 2023, 10, 49. [Google Scholar] [CrossRef]
- Miró, L.; Gasia, J.; Cabeza, L. Thermal energy storage (tes) for industrial waste heat (iwh) recovery: A review. Appl. Energy 2016, 179, 284–301. [Google Scholar] [CrossRef]
- Zhang, N.; Yuan, Y.; Cao, X.; Du, Y.; Zhang, Z.; Gui, Y. Latent heat thermal energy storage systems with solid–liquid phase change materials: A review. Adv. Eng. Mater. 2018, 20, 1700753. [Google Scholar] [CrossRef]
- Chalivendula, S.R.; Tarigonda, H. Recent advances in organic phase change materials for thermal energy storage: A review on sustainable development applications. Int. J. Thermophys. 2025, 46, 88. [Google Scholar] [CrossRef]
- Liu, K.; Zhou, J.; Liu, Y.; Mao, N.; Sun, L.; Wang, Y.; Xu, L.; Zhang, K.; Wang, C.; Yang, H. Flame retardant wood-based phase change materials with inorganic hydrated salt for thermal energy storage. Ind. Crops Prod. 2025, 227, 120825. [Google Scholar] [CrossRef]
- Nazir, H.; Batool, M.; Ali, M.; Kannan, A.M. Fatty acids based eutectic phase change system for thermal energy storage applications. Appl. Therm. Eng. 2018, 142, 466–475. [Google Scholar] [CrossRef]
- Liu, M.; Qian, R.; Yang, Y.; Lu, X.; Huang, L.; Zou, D. Modification of phase change materials for electric-thermal, photo-thermal, and magnetic-thermal conversions: A comprehensive review. Adv. Funct. Mater. 2024, 34, 2400038. [Google Scholar] [CrossRef]
- Alkan, C.; Sar, A.; Karaipekli, A.; Uzun, O. Preparation, characterization, and thermal properties of microencapsulated phase change material for thermal energy storage. Sol. Energy Mater. Sol. Cells 2009, 93, 143–147. [Google Scholar] [CrossRef]
- Sernaglia, M.; Blanco, D.; Battez, A.H.; González, R.; Fernández-González, A.; Bartolomé, M. Two fatty acid anion-based ionic liquids—Part II: Effectiveness as an additive to a polyol ester. J. Mol. Liq. 2025, 310, 113158. [Google Scholar] [CrossRef]
- Abass, P.J.; Muthulingam, S. Selection and thermophysical assessment of phase change materials (pcm) for space cooling applications in buildings. Numer. Heat Transf. Part A Appl. 2025, 86, 2423–2445. [Google Scholar] [CrossRef]
- Singh, A.; Rangarajan, S.; Sammakia, B. Utilizing ragone framework for optimized phase change material-based heat sink design in electronic cooling applications. Int. J. Heat Mass Transf. 2024, 227, 125518. [Google Scholar] [CrossRef]
- Zhao, P.; Yue, Q.; He, H.; Gao, B.; Wang, Y.; Li, Q. Study on phase diagram of fatty acids mixtures to determine eutectic temperatures and the corresponding mixing proportions. Appl. Energy 2014, 115, 483–490. [Google Scholar] [CrossRef]
- Sun, M.; Liu, T.; Sha, H.; Li, M.; Liu, T.; Wang, X.; Chen, G.; Wang, J.; Jiang, D. A review on thermal energy storage with eutectic phase change materials: Fundamentals and applications. J. Energy Storage 2023, 65, 107713. [Google Scholar] [CrossRef]
- Yuan, Y.; Zhang, N.; Li, T.; Cao, X.; Long, W. Thermal performance enhancement of palmitic-stearic acid by adding graphene nanoplatelets and expanded graphite for thermal energy storage: A comparative study. Energy 2016, 97, 488–497. [Google Scholar] [CrossRef]
- Sari, A.; Kaygusuz, K. Thermal energy storage characteristics of myristic and stearic acids eutectic mixture for low temperature heating applications. Chin. J. Chem. Eng. 2006, 14, 6. [Google Scholar] [CrossRef]
- Perween, S.; Ranjan, A. Altered phase behavior of the lauric acid–stearic acid binary mixtures in electrospun pva–pdms mats. Mater. Adv. 2022, 3, 2737–2748. [Google Scholar] [CrossRef]
- Fan, Y.; Zhang, Y.; Li, J. Thermal properties and reliabilities of lauric acid-based binary eutectic fatty acid as a phase change material for building energy conservation. ACS Omega 2022, 7, 16097–16108. [Google Scholar] [CrossRef]
- Jarrar, R.; Sawafta, R. Binary and ternary mixtures of eicosane with fatty alcohols and fatty acids as phase change material for building applications. Palest. Tech. Univ. Res. J. 2018, 6, 16–22. [Google Scholar] [CrossRef]
- Du, W.; Fei, H.; He, Q.; Wang, L.; Pan, Y.; Liu, J. Preparation and properties of capric acid–stearic acid-based ternary phase change materials. RSC Adv. 2021, 11, 24938–24948. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Ma, F.; Fu, Z.; Liu, J.; Li, C.; Wen, Y.; Hou, Y.; Shi, K. Optimization and characterization of fatty acids based ternary eutectic phase change system. J. Mol. Liq. 2024, 396, 123975. [Google Scholar] [CrossRef]
- Anand, A.; Shukla, A.; Kumar, A.; Sharma, A. Development and characterization of ternary mixture series of medium and long-chain saturated fatty acids for energy applications. Energy Storage 2019, 2, 10. [Google Scholar] [CrossRef]
- Nazari, M.; Jebrane, M.; Terziev, N. Multicomponent bio-based fatty acid system as phase change material for low temperature energy storage. J. Energy Storage 2021, 39, 102645. [Google Scholar] [CrossRef]
- Zhou, D.; Xiao, S.; Xiao, X. Preparation and thermal performance of fatty acids binary eutectic mixtures/expanded graphite as form-stable phase change materials for thermal energy storage. ACS Omega 2023, 8, 8596–8604. [Google Scholar] [CrossRef]
- Zhou, D.; Yuan, J.; Zhou, Y.; Liu, Y. Preparation and properties of capric–myristic acid/expanded graphite composite phase change materials for latent heat thermal energy storage. Energies 2020, 13, 2462. [Google Scholar] [CrossRef]
- Zhou, D.; Xiao, S.; Liu, Y. The effect of expanded graphite content on the thermal properties of fatty acid composite materials for thermal energy storage. Molecules 2024, 29, 3146. [Google Scholar] [CrossRef]
- Ke, H. Phase diagrams, eutectic mass ratios and thermal energy storage properties of multiple fatty acid eutectics as novel solid-liquid phase change materials for storage and retrieval of thermal energy. Appl. Therm. Eng. 2017, 113, 1319–1331. [Google Scholar] [CrossRef]
- Rubio-Pérez, G.; Muñoz-Rujas, N.; Aguilar, F.; Ravotti, R.; Müller, L.; Montero, E. Evolution of the study of phase diagram of binary and ternary mixtures involving fatty acid esters. Materials 2021, 14, 369. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Su, Y.; Ge, X. Prediction of the Melting Temperature and the Fusion Heat of (Quasi-) Eutectic PCM. J. China Univ. Sci. Technol. 1995, 25, 474–478. (In Chinese) [Google Scholar]
- Hassan, N.; Minakshi, M.; Liew, W.Y.H.; Amri, A.; Jiang, Z.-T. Thermal Characterization of Binary Calcium-Lithium Chloride Salts for Thermal Energy Storage at High Temperature. Energies 2023, 16, 4715. [Google Scholar] [CrossRef]
- Hassan, N.; Minakshi, M.; Ruprecht, J.; Liew, W.Y.H.; Jiang, Z.-T. A Binary Salt Mixture LiCl–LiOH for Thermal Energy Storage. Materials 2023, 16, 1434. [Google Scholar] [CrossRef]






| PCM | Tm (°C) | Tmp (°C) | ΔHm (J·g−1) | Tf (°C) | Tfp (°C) | ΔHf (J·g−1) | Cp,s (J·(g·K)−1) | Cp,l (J·(g·K)−1) |
|---|---|---|---|---|---|---|---|---|
| CA | 30.5 | 32.3 | 176.0 | 30.5 | 28.6 | 172.9 | 1.9 | 2.1 |
| LA | 43.8 | 46.5 | 182.0 | 41.9 | 40.0 | 190.6 | 1.7 | 2.3 |
| MA | 52.3 | 54.6 | 188.6 | 51.6 | 49.0 | 193.1 | 1.7 | 2.4 |
| PA | 54.3 | 56.9 | 206.5 | 52.6 | 50.3 | 194.4 | 1.9 | 2.8 |
| SA | 62.2 | 63.5 | 214.3 | 61.0 | 59.2 | 211.2 | 1.6 | 2.2 |
| Ternary Eutectic Fatty Acid Mixtures | Theoretical Eutectic Mass Ratio | Tm (°C) | ΔHm (J·g−1) |
|---|---|---|---|
| CA-LA-MA | 52.58:29.81:17.61 | 17.11 | 167.8 |
| CA-LA-PA | 57.64:31.92:12.44 | 18.43 | 170.4 |
| CA-LA-SA | 59.28:34.36:6.36 | 19.61 | 170.3 |
| CA-MA-PA | 62.12:22.54:15.34 | 21.56 | 173.1 |
| CA-MA-SA | 66.85:24.87:8.28 | 23.00 | 173.1 |
| CA-PA-SA | 71.17:19.37:9.46 | 24.70 | 177.3 |
| LA-MA-PA | 46.44:30.56:23.00 | 29.06 | 177.9 |
| LA-MA-SA | 51.52:34.52:13.96 | 31.13 | 178.0 |
| LA-PA-SA | 54.72:29.52:15.76 | 33.06 | 184.1 |
| MA-PA-SA | 43.35:36.34:20.31 | 37.61 | 189.6 |
| Binary Eutectic Mixtures | Theoretical Eutectic Mass Ratio | Tm (°C) | ΔHm (J·g−1) |
|---|---|---|---|
| CA&LA | 63.1:36.9 | 20.52 | 184.0 |
| CA&MA | 72.2:27.8 | 24.19 | 177.0 |
| LA&MA | 59.4:40.6 | 33.39 | 187.0 |
| Proportion of (CA&LA) (%) | Proportion of MA (%) | Tm (°C) |
|---|---|---|
| 100.0 | 0.0 | 20.52 |
| 92.0 | 8.0 | 19.09 |
| 89.5 | 10.5 | 18.63 |
| 84.8 | 15.2 | 17.68 |
| 82.4 | 17.6 | 17.20 |
| 80.1 | 19.9 | 19.20 |
| 69.9 | 30.1 | 26.57 |
| 54.9 | 45.1 | 34.35 |
| 30.0 | 70.0 | 43.72 |
| 4.8 | 95.2 | 51.04 |
| 0.00 | 100 | 52.30 |
| Fatty Acid | Mass Ratio | Mass Ratio | Tm (°C) | ΔHm (J·g−1) |
|---|---|---|---|---|
| (CA&LA)-MA | (CA&LA):MA = 82.4:17.6 | CA:LA:MA = 52.0:30.4:17.6 | 17.20 | 180.0 |
| (CA&MA)-LA | (CA&MA):LA = 70.4:29.6 | CA:LA:MA = 41.8:29.6:28.6 | 16.99 | 181.0 |
| CA-(LA&MA) | CA:(LA&MA) = 52.5:47.5 | CA:LA:MA = 52.5:28.2:19.3 | 17.12 | 182.0 |
| CA-LA-MA | — | CA:LA:MA = 52.58:29.81:17.61 | 17.11 | 167.8 |
| Data Category | Composition (CA:LA:MA, wt %) | Tm (°C) | ΔHm (J·g−1) |
|---|---|---|---|
| This Study—Theoretical | 52.07:30.56:17.37 | 17.11 | 167.8 |
| This Study—Experimental | 52.0:30.0:18.0 | 16.0 | 177.0 |
| Reference [32]—Theoretical | 56.39:26.72:16.89 | 16.5 | 85.76 |
| Reference [32]—Experimental | 66.35:20.62:13.03 | 15.83 | 131.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zhou, D.; Zhou, F.; Yuan, J.; Liu, Z.; Liu, Y. Phase Diagrams and Thermal Properties of Fatty Acid Ternary Eutectic Mixtures for Latent Heat Thermal Energy. Materials 2026, 19, 356. https://doi.org/10.3390/ma19020356
Zhou D, Zhou F, Yuan J, Liu Z, Liu Y. Phase Diagrams and Thermal Properties of Fatty Acid Ternary Eutectic Mixtures for Latent Heat Thermal Energy. Materials. 2026; 19(2):356. https://doi.org/10.3390/ma19020356
Chicago/Turabian StyleZhou, Dongyi, Fanchen Zhou, Jiawei Yuan, Zhifu Liu, and Yicai Liu. 2026. "Phase Diagrams and Thermal Properties of Fatty Acid Ternary Eutectic Mixtures for Latent Heat Thermal Energy" Materials 19, no. 2: 356. https://doi.org/10.3390/ma19020356
APA StyleZhou, D., Zhou, F., Yuan, J., Liu, Z., & Liu, Y. (2026). Phase Diagrams and Thermal Properties of Fatty Acid Ternary Eutectic Mixtures for Latent Heat Thermal Energy. Materials, 19(2), 356. https://doi.org/10.3390/ma19020356







