Porous Refractories Synthesized Using Rice Husk and Rice Husk Processing Products
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Mix Design
- -
- Content, wt.%: Al2O3—at least 30. The fraction was 0.06–2.0 mm.
- -
- Sodium liquid glass, GOST 13078-2021 [33]. Content, wt.%: SiO2—24.8–36.7; Na2O—8.1–13.3.
- -
- Technical liquid lignosulfonate, TU 2455-028-00279580-2014. Content, wt.%: dry substance—50; sodium salt of lignosulfonic acid—32; pH—4.6.
- -
- Magnesium sulfate 7-hydrate, GOST 4523-77 [34]. Crystalline powder. Content, wt.%: MgSO4·7H2O—99.
- -
- PAP-1 grade aluminum powder, GOST 5494-2022 [35]. Impurity content, wt.%: Fe ≤ 0.5; Si ≤ 0.4; Cu ≤ 0.05; Mn ≤ 0.05. The fraction was <8 μm.
2.2. Analytical Methods
2.3. Sample Preparation
- -
- Dried, Series 1: 1-AlMgC, 1-AlMgC-SS8.5, 1-AlMgC-RH2.4, and 1-AlMgC-RH14.4.
- -
- Fired, Series 2: 2-AlMgC, 2-AlMgC-SS8.5, 2-AlMgC-RH2.4, and 2-AlMgC-RH14.4.
- -
- Destroyed during the thermal shock resistance test (description is given in the Section 2.4), Series 3: 3-AlMgC, 3-AlMgC-SS8.5, 3-AlMgC-RH2.4, and 3-AlMgC-RH14.4.
2.4. Testing and Analysis
3. Results and Discussion
3.1. X-Ray Diffraction Analysis
3.2. Thermal Analysis
3.3. Scanning Electron Microscopy
3.4. Performance Properties
3.4.1. Physical and Mechanical Properties
3.4.2. Thermal Properties
3.5. Comparative Analysis of the Properties of Porous High-Temperature Materials
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| A | Refractory clay |
| Ai | Bond strength, MPa |
| AlMgC | Composition of control sample of high-temperature material containing (wt.%): MgSO4·7H2O—4.5; Al powder—2; refractory clay—59; liquid glass—8.5; lignosulfonate—2; chamotte—24 |
| 1-AlMgC | AlMgC sample dried (Series 1) |
| 2-AlMgC | AlMgC sample fired (Series 2) |
| 3-AlMgC | AlMgC sample destroyed during the thermal shock resistance test (Series 3) |
| AlMgC-RH2.4 | Composition of experimental sample of high-temperature material containing (wt.%): MgSO4·7H2O—4.5; Al powder—2; refractory clay—59; sodium silicate solution from rice husk—8.5; organic condensate from rice husk pyrolysis—2; rice husk—2.4; chamotte—21.6 |
| 1-AlMgC-RH2.4 | AlMgC-RH2.4 sample dried (Series 1) |
| 2-AlMgC-RH2.4 | AlMgC-RH2.4 sample fired (Series 2) |
| 3-AlMgC-RH2.4 | AlMgC-RH2.4 sample destroyed during the thermal shock resistance test (Series 3) |
| AlMgC-RH14.4 | Composition of experimental sample of high-temperature material containing (wt.%): MgSO4·7H2O—5; Al powder—3; refractory clay—67.1; organic condensate from rice husk pyrolysis—10.5; rice husk—14.4 |
| 1-AlMgC-RH14.4 | AlMgC-RH14.4 sample dried (Series 1) |
| 2-AlMgC-RH14.4 | AlMgC-RH14.4 sample fired (Series 2) |
| 3-AlMgC-RH14.4 | AlMgC-RH14.4 sample destroyed during the thermal shock resistance test (Series 3) |
| AlMgC-SS8.5 | Composition of experimental sample of high-temperature material containing (wt.%): MgSO4·7H2O—4.5; Al powder—2; refractory clay—59; silicate solution from rice husk—8.5; organic condensate from rice husk pyrolysis—2; chamotte—24 |
| 1-AlMgC-SS8.5 | AlMgC-SS8.5 sample dried (Series 1) |
| 2-AlMgC-SS8.5 | AlMgC-SS8.5 sample fired (Series 2) |
| 3-AlMgC-SS8.5 | AlMgC-SS8.5 sample destroyed during the thermal shock resistance test (Series 3) |
| B | Bentonite |
| C | AlMgC sample prepared without MgSO4·7H2O and Al powder |
| 1-C | C sample dried (Series 1) |
| C-RH14.4 | AlMgC-RH14.4 sample prepared without MgSO4·7H2O and Al powder |
| 1-C-RH14.4 | C-RH14.4 sample dried (Series 1) |
| DE | Diatomite |
| DS | Industrial diatomaceous silica |
| EDS | Energy dispersive spectroscopy |
| Ex | Extrusion method |
| GC-MS | Gas chromatography–mass spectrometry |
| KC | Kaolin |
| OC-RH | Organic condensate prepared by rice husk pyrolysis |
| PAP-1 | Grade aluminum powder |
| Pr | Pressing method |
| PS1 | Polysaccharide |
| PVA | Polyvinyl alcohol |
| RAC | Red anthill clay |
| RHA | Rice husk ash |
| RHS | Rice husk silica |
| Rtb | Tensile strength in bending, MPa |
| S | Rice husk ash |
| SCC | Silicon carbon composite |
| SCC-V | SCC sample activated with water steam at 850 °C for 30 min |
| SCC-VA | SCC-V sample activated by alkali |
| SD | Sawdust |
| SF | Steel fibers |
| SHMP | Sodium hexametaphosphate |
| Sol | Sol derived from rice husk ash |
| SRC | Silicon carbide |
| W | Wollastonite |
| WS | Waste sediment |
| σ | Ultimate compressive strength, MPa |
| λ | Thermal conductivity, W (m·K)−1 |
| ρ | True density, g·cm−3 |
References
- Kolesnikov, A.S.; Zhakipbaev, B.Y.; Zhanikulov, N.N.; Kolesnikova, O.G.; Аkhmetova, Е.K.; Kuraev, R.M.; Shal, A.L. Review of technogenic waste and methods of its processing for the purpose of complex utilization of tailings from the enrichment of non-ferrous metal ores as a component of the raw material mixture in the production of cement clinker. Rasayan J. Chem. 2021, 14, 997–1005. [Google Scholar] [CrossRef]
- Satbaev, B.N.; Koketaev, A.I.; Aimbetova, E.O.; Berdikulova, F.A.; Shalabaev, N.T.; Satbaev, A.B. Production of chemically resistant refractory concrete mixes from metallurgical waste and their physical and chemical properties. Refract. Ind. Ceram. 2021, 61, 484–486. [Google Scholar] [CrossRef]
- Kolesnikov, A.; Fediuk, R.; Kolesnikova, O.; Zhanikulov, N.; Zhakipbayev, B.; Kuraev, R.; Akhmetova, E.; Shal, A. Processing of waste from enrichment with the production of cement clinker and the extraction of zinc. Materials 2022, 15, 324. [Google Scholar] [CrossRef] [PubMed]
- Zharmenov, A.A.; Satbaev, B.N.; Kazhikenova, S.S.; Nurkenov, O.A. Development of new refractory materials by SHS-technology based on Kazakhstan Republic raw material resources. Refract. Ind. Ceram. 2012, 53, 199–205. [Google Scholar] [CrossRef]
- Satbaev, B.N.; Nukhuly, A.; Sviderskiy, A.K.; Nurkenov, O.A.; Filosin, A.I. New Refractory SHS-Materials and Their Application; PGPI: Pavlodar, Kazakhstan, 2008. [Google Scholar]
- Thang, N.H. Novel porous refractory synthesized from diatomaceous earth and rice husk ash. J. Polym. Compos. 2020, 8, 4295. [Google Scholar] [CrossRef]
- Arthur, E.K.; Gikunoo, E. Property analysis of thermal insulating materials made from Ghanaian anthill clay deposits. Cogent Eng. 2020, 7, 1827493. [Google Scholar] [CrossRef]
- Efremova, S.V. Rice hull as a renewable raw material and its processing routes. Russ. J. Gen. Chem. 2012, 82, 999–1005. [Google Scholar] [CrossRef]
- Gonçalves, M.R.F.; Bergmann, C.P. Thermal insulators made with rice husk ashes: Production and correlation between properties and microstructure. Con. Build. Mater. 2007, 21, 2059–2065. [Google Scholar] [CrossRef]
- Vassilev, S.V.; Baxter, D.; Vassileva, C.G. An overview of the behaviour of biomass during combustion: Part I. Phase-mineral transformations of organic and inorganic matter. Fuel 2013, 112, 391–449. [Google Scholar] [CrossRef]
- Novembre, D.; Gimeno, D.; Marinangeli, L.; Tangari, A.C.; Rosatelli, G.; Ciulla, M.; di Profio, P. Synthesis and Characterization of Na-P1 (GIS) Zeolite Using Rice Husk. Molecules 2024, 29, 5596. [Google Scholar] [CrossRef]
- Yefremova, S.; Kablanbekov, A.; Satbaev, B.; Zharmenov, A. Rice Husk-Based Adsorbents for Removal of Metals from Aqueous Solutions. Materials 2023, 16, 7353. [Google Scholar] [CrossRef]
- Hossain, S.K.S.; Lakshya, M.; Roy, P.K. Rice husk/rice husk ash as an alternative source of silica in ceramics: A review. J. Asian Ceram. Soc. 2018, 6, 299–313. [Google Scholar] [CrossRef]
- Hossain, S.S.; Roy, P.K. Sustainable ceramics derived from solid wastes: A review. J. Asian Ceram. Soc. 2020, 8, 984–1009. [Google Scholar] [CrossRef]
- Ugheoke, B.I.; Mamat, O.; Ariwahjoedi, B. A brief survey of the literature on silica refractory research and development: The case for nanostructured silica obtained from rice husk ash (RHA). Defect Diffus. Forum 2010, 307, 53–62. [Google Scholar] [CrossRef]
- Chandrasekhar, S.; Pramada, P.N.; Praveen, L. Effect of organic acid treatment on the properties of rice husk silica. J. Mater. Sci. 2005, 40, 6535–6544. [Google Scholar] [CrossRef]
- Lu, P.; Hsieh, Y.-L. Highly pure amorphous silica nano-disks from rice straw. Powd. Technol. 2012, 225, 149–155. [Google Scholar] [CrossRef]
- Rozainee, M.; Ngo, S.P.; Salema, A.A.; Tan, K.G.; Ariffin, M.; Zainura, Z.N. Effect of fluidising velocity on the combustion of rice husk in a bench-scale fluidised bed combustor for the production of amorphous rice husk ash. Bioresour. Technol. 2008, 99, 703–713. [Google Scholar] [CrossRef]
- Sankar, S.; Sharma, S.K.; Kaur, N.; Lee, B.; Kim, D.Y.; Lee, S.; Jung, H. Biogenerated silica nanoparticles synthesized from sticky, red, and brown rice husk ashes by a chemical method. Ceram. Int. 2016, 4, 4875–4885. [Google Scholar] [CrossRef]
- Carmona, V.B.; Oliveira, R.M.; Silva, W.T.L.; Mattoso, L.H.C.; Marconcini, J.M. Nanosilica from rice husk: Extraction and characterization. Ind. Crops Prod. 2013, 43, 291–296. [Google Scholar] [CrossRef]
- Zharmenov, A.; Yefremova, S.; Satbaev, B.; Shalabaev, N.; Satbaev, S.; Yermishin, S.; Kablanbekov, A. Production of refractory materials using a renewable source of silicon dioxide. Minerals 2022, 12, 1010. [Google Scholar] [CrossRef]
- Sobrosa, F.Z.; Stochero, N.P.; Marangon, E.; Tier, M.D. Development of refractory ceramics from residual silica derived from rice husk ash. Ceram. Int. 2017, 43, 7142–7146. [Google Scholar] [CrossRef]
- Stochero, N.P.; Marangon, E.; Nunes, A.S.; Tier, M.D. Development of refractory ceramics from residual silica derived from rice husk ash and steel fibres. Ceram. Int. 2017, 43, 13875–13880. [Google Scholar] [CrossRef]
- Silva, D.; Pachla, E.; Marangon, E.; Tier, M.; Garcia, A.P. Effects of rice husk ash and wollastonite incorporation on the physical and thermal properties of refractory ceramic composites. Rev. Matéria 2020, 25, e-12802. [Google Scholar] [CrossRef]
- Hossain, S.S.; Bae, C.J.; Roy, P.K. A replacement of traditional insulation refractory brick by a waste-derived lightweight refractory castable. Int. J. Appl. Ceram. Technol. 2021, 18, 1783–1791. [Google Scholar] [CrossRef]
- Bhardwaj, A.; Hossain, S.K.S.; Majhi, M.R. Preparation and characterization of clay bonded high strength silica refractory by utilizing agriculture waste. Boletín Soc. Española Cerámica Vidr. 2017, 56, 256–262. [Google Scholar] [CrossRef]
- Sembiring, S.; Simanjuntak, W.; Situmeang, R.; Riyanto, A.; Karo-Karo, P. Effect of alumina addition on the phase transformation and crystallisation properties of refractory cordierite prepared from amorphous rice husk silica. J. Asian Ceram. Soc. 2017, 5, 186–192. [Google Scholar] [CrossRef]
- Sembiring, S.; Simanjuntak, W.; Situmeang, R.; Riyanto, A.; Sebayang, K. Preparation of refractory cordierite using amorphous rice husk silica for thermal insulation purposes. Ceram. Int. 2016, 42, 8431–8437. [Google Scholar] [CrossRef]
- Serra, M.F.; Conconi, M.S.; Gauna, M.R.; Suárez, G.; Aglietti, E.F.; Rendtorff, N.M. Mullite (3Al2O3·2SiO2) ceramics obtained by reaction sintering of rice husk ash and alumina, phase evolution, sintering and microstructure. J. Asian Ceram. Soc. 2016, 4, 61–67. [Google Scholar] [CrossRef]
- Satbaev, B.; Yefremova, S.; Zharmenov, A.; Kablanbekov, A.; Yermishin, S.; Shalabaev, N.; Satbaev, A.; Khen, V. Rice Husk Research: From Environmental Pollutant to a Promising Source of Organo-Mineral Raw Materials. Materials 2021, 14, 4119. [Google Scholar] [CrossRef]
- Efremova, S.V.; Korolev, Y.M.; Sukharnikov, Y.I. X-ray diffraction characterization of silicon-carbon nanocomposites produced from rice husk and its derivatives. Dokl. Chem. 2008, 419, 78–81. [Google Scholar] [CrossRef]
- Yefremova, S.; Kablanbekov, A.; Sukharnikov, Y.; Hain, Y.; Zharmenov, A. Porous Carbon Materials: Synthesis, Characterization, Application; Polygrafservice: Almaty, Kazakhstan, 2017. [Google Scholar]
- GOST 13078-2021; Sodium Liquid Glass. Specifications. Russian Institute of Standardization: Moscow, Russia, 2021.
- GOST 4523-77; Reagents. Magnesium sulphate, 7-aqueous. Specifications. Publishing House of Standards: Moscow, Russia, 1978.
- GOST 5494-2022; Pigmentary Aluminium. Specifications. Russian Institute of Standardization: Moscow, Russia, 2022.
- Sukurov, B.; Kvyatkovskiy, S.; Kozhakhmetov, S.; Semenova, A.; Dyussebekova, M.; Kvyatkovskaya, M. Structural Changes in Copper Slags During Slow Cooling. Metals 2024, 14, 1187. [Google Scholar] [CrossRef]
- Sokolovskaya, L.; Kvyatkovskiy, S.; Kozhakhmetov, S.; Semenova, A.; Sukurov, B.; Dyussebekova, M.; Shakhalov, A. Slag after Smelting of Anode Mud: Role of Sulphiding Sintering. Minerals 2024, 14, 781. [Google Scholar] [CrossRef]
- Tastanova, A.; Temirova, S.; Sukurov, B.; Biryukova, A.; Abdykirova, G. Experimental Manufacturing of Ferromanganese Alloy from Man-Made Manganese-Containing Wastes. Processes 2023, 11, 3328. [Google Scholar] [CrossRef]
- GOST 2409-2014; Refractories. Method for Determination of Bulk Density, Apparent and True Porosity, and Water Absorption. Standardinform: Moscow, Russia, 2014.
- GOST 4071.1-2021; Refractory Products with Less than 45% True Porosity. Method for Determination of Compressive Strength at Room Temperature. Russian Institute of Standardization: Moscow, Russia, 2021.
- GOST 31356-2007; Dry Building Mixtures on a Cement Binder. Test Methods. Standardinform: Moscow, Russia, 2008.
- GOST 10180-2012; Concretes. Methods for Strength Determination Using Reference Specimens. Standardinform: Moscow, Russia, 2018.
- GOST 30256-94; Building Materials and Products. Method for Determination of Thermal Conductivity by Cylindrical Probe. Publishing House of Standards: Moscow, Russia, 1996.
- GOST 4070-2000; Refractory Products. Method for Determination of Refractoriness Under Load. Standardinform: Moscow, Russia, 2006.
- Ivanova, V.P. Thermal Analysis of Minerals and Rocks; Nedra: Leningrad, Russia, 1974. [Google Scholar]
- Sanewirush, U.S.; Saewong, P. Synthesis of Ca-Al-Si-O compounds from local wastes. In Materials Science Forum; Kim, H., Yang, J.F., Sekino, T., Lee, S.W., Eds.; Trans Tech Publications Ltd.: Baech, Switzerland, 2009; Volume 620–622, pp. 121–124. [Google Scholar] [CrossRef]
- Azat, S.; Mansurov, Z.A. Wastewater treatment using carbon sorbent. Chem. Bull. Kazakh Nat. Univ. 2011, 1, 166–169. [Google Scholar]
- Aitugan, A.; Tanirbergenova, S.; Tileuberdi, Y.; Yucel, O.; Tugelbayeva, D.; Mansurov, Z.; Ongarbayev, Y. A carbonized cobalt catalyst supported by acid-activated clay for the selective hydrogenation of acetylene. React. Kinet. Mech. Catal. 2021, 133, 277–292. [Google Scholar] [CrossRef]










| Sample | SiO2 | Al2O3 | P | Ca | Mg | TiO2 | Mn | Fe | Cu | Pb | Zn | K | Na |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| RH | 20.4 | N.D. | 0.14 | 1.86 | 0.42 | 0.33 | 0.034 | 0.68 | 0.005 | 0.006 | 0.006 | 0.091 | 0.052 |
| SCC | 36.8 | N.D. | 0.10 | 0.94 | 0.21 | 0.08 | 0.029 | 0.33 | 0.0005 | 0.0085 | 0.010 | 0.094 | 0.018 |
| Sample | C | N | H | S |
|---|---|---|---|---|
| RH | 41.59 | 1.05 | 4.32 | 0.17 |
| SCC | 49.02 | 0.30 | 0.21 | 0.44 |
| Element | Content, wt.% |
|---|---|
| Si | 18.36 |
| P | 0.047 |
| S | 0.046 |
| K | 0.324 |
| Ca | 0.285 |
| Ti | 0.595 |
| V | 0.016 |
| Cr | 0.015 |
| Fe | 2.08 |
| Zn | 0.002 |
| Sr | 0.004 |
| Zr | 0.036 |
| Nb | 0.003 |
| Ba | 0.013 |
| Pb | 0.002 |
| Bi | 0.002 |
| Al | 5.23 |
| Balance | 72.95 |
| Ingredient | Mix Abbreviation | |||||
|---|---|---|---|---|---|---|
| AlMgC | AlMgC-SS8.5 | AlMgC-RH2.4 | AlMgC-RH14.4 | C | C-RH14.4 | |
| MgSO4·7H2SO4 (tech.) | 4.5 | 4.5 | 4.5 | 5 | 0 | 0 |
| Al powder, PAP-1 | 2 | 2 | 2 | 3 | 0 | 0 |
| Refractory clay | 59 | 59 | 59 | 67.1 | 59 | 67.1 |
| Liquid glass Na2SiO3 (liq.) | 8.5 | 0 | 0 | 0 | 8.5 | 0 |
| SS-RH | 0 | 8.5 | 8.5 | 0 | 0 | 0 |
| Lignosulfonate | 2 | 0 | 0 | 0 | 2 | 0 |
| OC-RH | 0 | 2 | 2 | 10.5 | 0 | 10.5 |
| Chamotte | 24 | 24 | 21.6 | 0 | 24 | 0 |
| RH | 0 | 0 | 2.4 | 14.4 | 0 | 14.4 |
| Total | 100 | 100 | 100 | 100 | 93.5 | 92 |
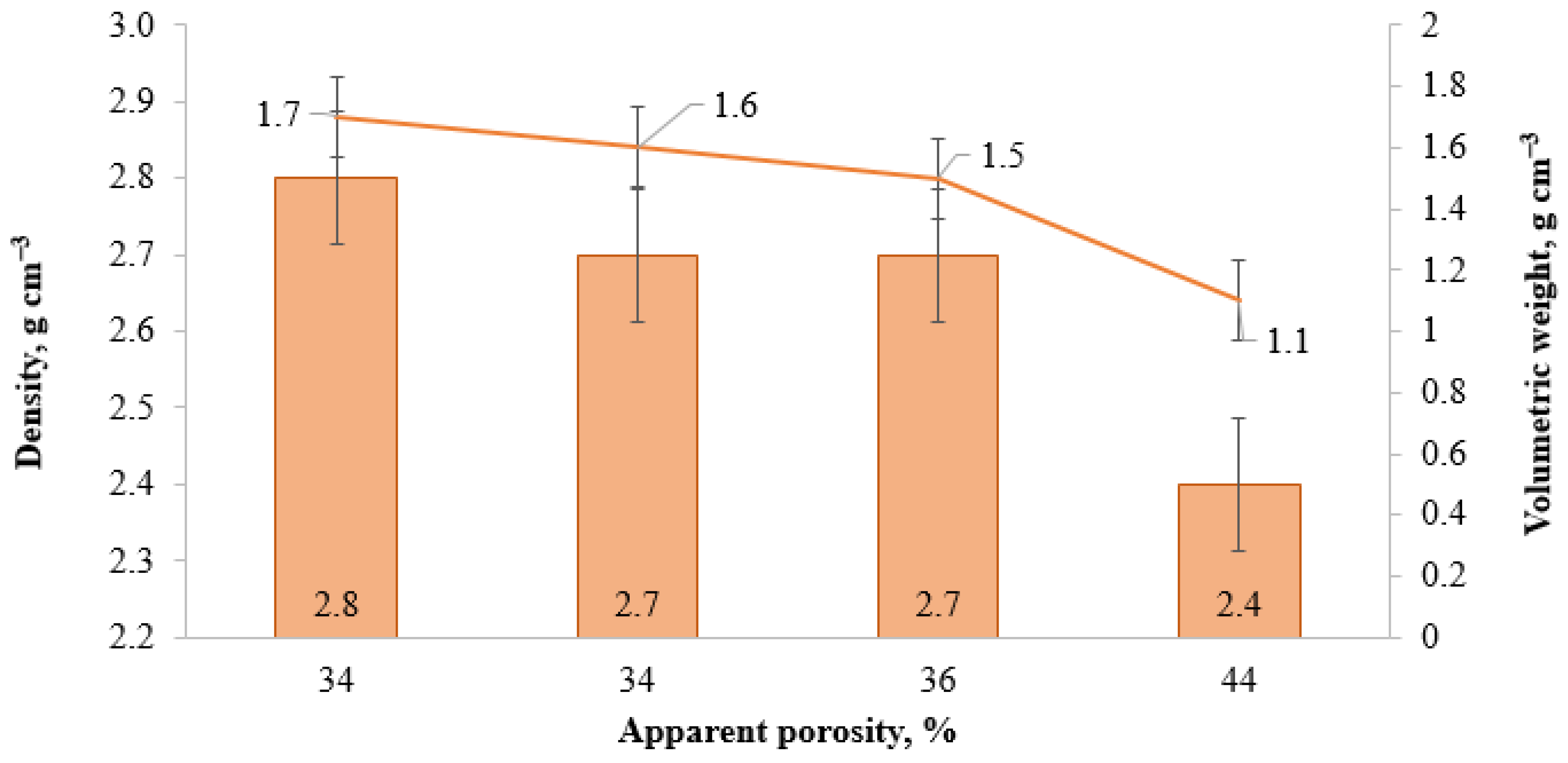
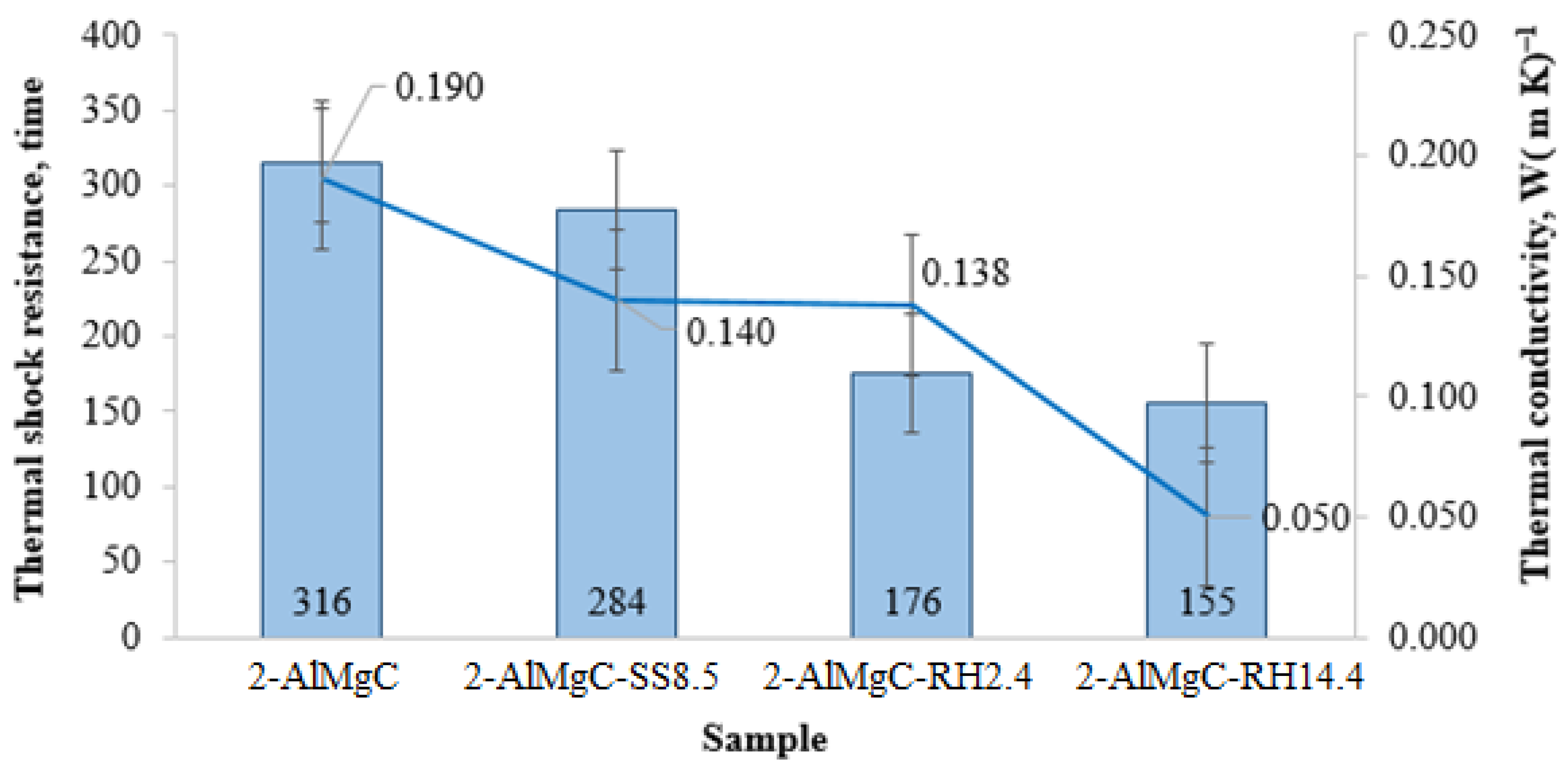
| Sample | Lignosulfonate, wt.% | OC-RH, wt.% | RH, wt.% | Apparent Porosity, % | Density, g·cm−3 | Volumetric Weight, g·cm−3 | Compressive Strength, MPa | Bond Strength, MPa | Tensile Strength in Bending, MPa |
|---|---|---|---|---|---|---|---|---|---|
| 2-AlMgC | 2 | 0 | 0 | 34 | 2.8 | 1.7 | 2.9 | 0.011 | 11.9 |
| 2-AlMgC-SS8.5 | 0 | 2 | 0 | 34 | 2.7 | 1.6 | 7.8 | 0.010 | 14.2 |
| 2-AlMgC-RH2.4 | 0 | 2 | 2.4 | 36 | 2.7 | 1.5 | 4.2 | 0.010 | 8.2 |
| 2-AlMgC-RH14.4 | 0 | 10.5 | 14.4 | 44 | 2.4 | 1.1 | 2.1 | 0.011 | 4.5 |
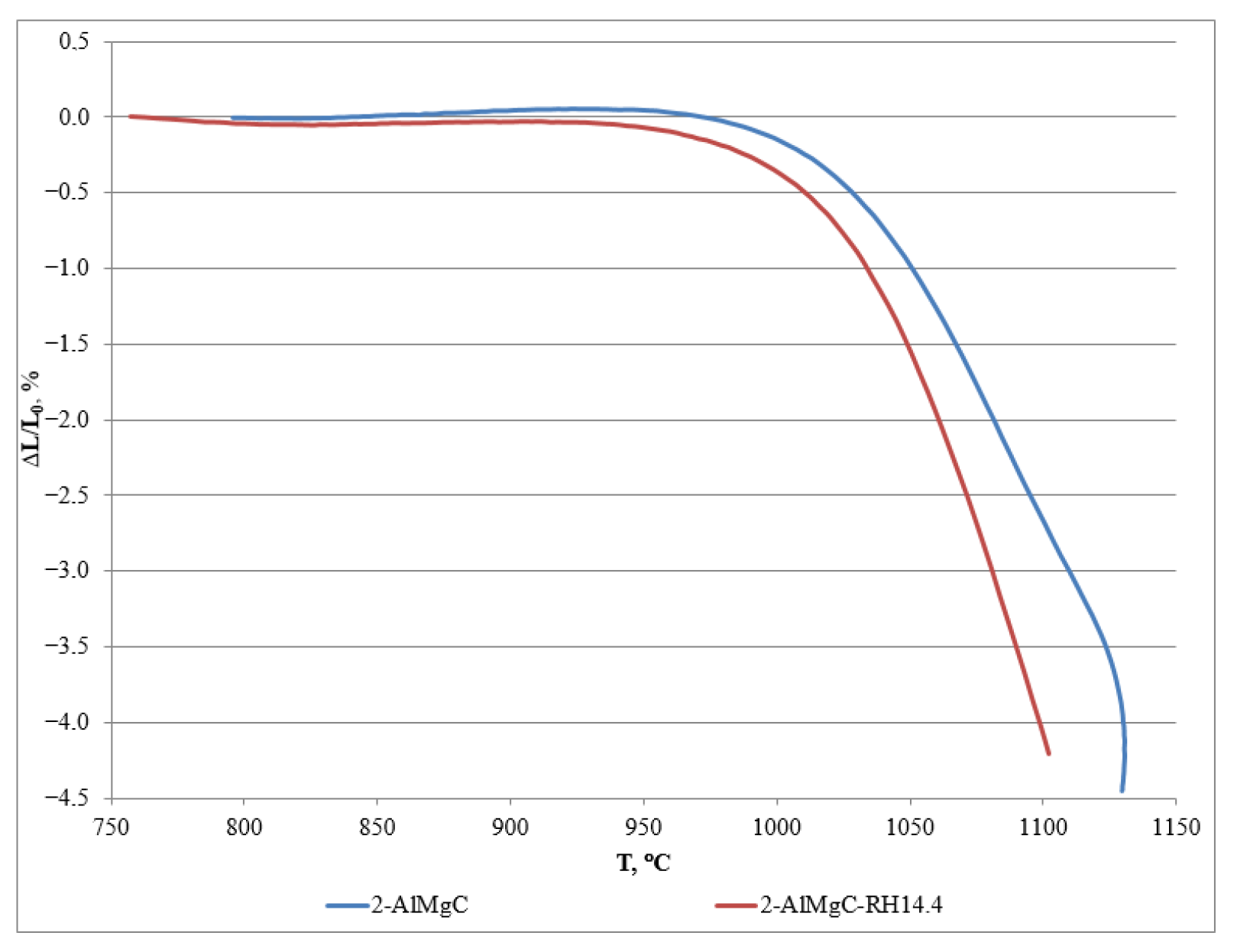
| Sample | T06, °C | T4, °C |
|---|---|---|
| 2-AlMgC | 1030 | 1120 |
| 2-AlMgC-RH14.4 | 1020 | 1100 |
| Sample | Raw Materials | References | ||||||
| DE, wt.% | RHA, wt.% | SD, wt.% | - | - | - | - | ||
| M0 | 50.0 | 50.0 | 0 | - | - | - | - | [6] |
| M5 | 47.5 | 47.5 | 5.0 | - | - | - | - | [6] |
| M10 | 45.0 | 45.0 | 10.0 | - | - | - | - | [6] |
| M15 | 42.5 | 42.5 | 15.0 | - | - | - | - | [6] |
| M20 | 40.0 | 40.0 | 20.0 | - | - | - | - | [6] |
| M25 | 37.5 | 37.5 | 25.0 | - | - | - | - | [6] |
| M30 | 35.0 | 35.0 | 30.0 | - | - | - | - | [6] |
| M35 | 32.5 | 32.5 | 35.0 | - | - | - | - | [6] |
| RAC, wt.% | SD, wt.% | RH, wt.% | - | - | - | - | ||
| Control | 100 | 0 | 0 | - | - | - | - | [7] |
| Anthill-SD5% | 95 | 5 | 0 | - | - | - | - | [7] |
| Anthill-SD10% | 90 | 10 | 0 | - | - | - | - | [7] |
| Anthill-SD15% | 85 | 15 | 0 | - | - | - | - | [7] |
| Anthill-SD20% | 80 | 20 | 0 | - | - | - | - | [7] |
| Anthill-RH5% | 95 | 0 | 5 | - | - | - | - | [7] |
| Anthill-RH10% | 90 | 0 | 10 | - | - | - | - | [7] |
| Anthill-RH15% | 85 | 0 | 15 | - | - | - | - | [7] |
| Anthill-RH20% | 80 | 0 | 20 | - | - | - | - | [7] |
| Anthill-SD-RH5% | 95 | 2.5 | 2.5 | - | - | - | - | [7] |
| Anthill-SD-RH10% | 90 | 5 | 5 | - | - | - | - | [7] |
| Anthill-SD-RH15% | 85 | 7.5 | 7.5 | - | - | - | - | [7] |
| Anthill-SD-RH20% | 80 | 10 | 10 | - | - | - | - | [7] |
| KC, vol.% | RHS, vol.% | - | - | - | - | - | ||
| KC | 100 | 0 | - | - | - | - | - | [22] |
| KS5 | 95 | 5 | - | - | - | - | - | [22] |
| KS10 | 90 | 10 | - | - | - | - | - | [22] |
| KS20 | 80 | 20 | - | - | - | - | - | [22] |
| KC, vol.% | RHS, vol.% | SF, vol.% | - | - | - | - | ||
| KC | 100 | 0 | 0 | - | - | - | - | [23] |
| K20S | 80 | 20 | 0 | - | - | - | - | [23] |
| K20S3F | 77 | 20 | 3 | - | - | - | - | [23] |
| K20S6F | 74 | 20 | 6 | - | - | - | - | [23] |
| K20S9F | 71 | 20 | 9 | - | - | - | - | [23] |
| A, wt.% | S, wt.% | W, wt.% | - | - | - | - | ||
| A | 100 | 0 | 0 | - | - | - | - | [24] |
| AS10 | 90 | 10 | 0 | - | - | - | - | [24] |
| AS10W5 | 85 | 10 | 5 | - | - | - | - | [24] |
| AS10W10 | 80 | 10 | 10 | - | - | - | - | [24] |
| AS10W20 | 70 | 10 | 20 | - | - | - | - | [24] |
| RHA Unground, >200 μm, wt.% | RHA Ground, <200 μm, wt.% | Sol, Dry Sol, wt.% | SHMP, wt.% | Water added, wt.% | Water from Sol, wt.% | SiO2, wt.% | ||
| s-1, green, 80 °C | 50 | 47.3 | 2.5 | 0.2 | 5 | 5.8 | - | [25] |
| s-2, 900 °C | 50 | 44.8 | 5 | 0.2 | 2 | 11.6 | - | [25] |
| s-3, 1000 °C | 50 | 42.30 | 7.5 | 0.2 | - | 17.5 | - | [25] |
| s-4, 1100 °C | 50 | 39.80 | 10 | 0.2 | - | 23.3 | 95 | [25] |
| s-5, 1200 °C | 50 | 37.30 | 12.5 | 0.2 | - | 29.2 | - | [25] |
| Sample | Physical and Mechanical Properties | Thermal Properties | References | |||||
|---|---|---|---|---|---|---|---|---|
| Volumetric Weight, g·cm−3 | Porosity, % | Bending Strength, MPa | Compressive Strength, MPa | Tensile Strength in Bending, MPa | Thermal Shock Resistance at 1000 °C, Time | Thermal Conductivity, W (m·K)−1 | ||
| M0 | 0.66 | 75 | 1.78 | 17.35 | - | 78 | 0.1035 | [6] |
| M5 | 0.61 | 74 | 1.75 | 17.24 | - | 75 | 0.1024 | [6] |
| M10 | 0.57 | 78 | 1.72 | 17.03 | - | 72 | 0.1003 | [6] |
| M15 | 0.53 | 82 | 1.72 | 16.84 | - | 72 | 0.0984 | [6] |
| M20 | 0.48 | 83 | 1.70 | 16.68 | - | 70 | 0.0968 | [6] |
| M25 | 0.44 | 87 | 1.68 | 16.41 | - | 68 | 0.0941 | [6] |
| M30 | 0.41 | 89 | 1.66 | 16.11 | - | 66 | 0.0911 | [6] |
| M35 | 0.37 | 92 | 1.61 | 15.78 | - | 61 | 0.0878 | [6] |
| Control | 1.86 4 | 25 5 | - | 2.80 | - | 1 3 | 0.55 | [7] |
| Anthill-SD5% | 1.42 4 | 33 5 | - | 1.82 | - | 11 3 | 0.46 | [7] |
| Anthill-SD10% | 1.28 4 | 39 5 | - | 1.38 | - | 16 3 | 0.40 | [7] |
| Anthill-SD15% | 1.14 4 | 43 5 | - | 1.1 | - | 21 3 | 0.35 | [7] |
| Anthill-SD20% | 1.07 4 | 53 5 | - | 0.74 | - | 25 3 | 0.31 | [7] |
| Anthill-RH5% | 1.52 4 | 23 5 | - | 2.32 | - | 8 3 | 0.40 | [7] |
| Anthill-RH10% | 1.36 4 | 34 5 | - | 1.80 | - | 13 3 | 0.33 | [7] |
| Anthill-RH15% | 1.2 4 | 37 5 | - | 1.50 | - | 18 3 | 0.30 | [7] |
| Anthill-RH20% | 1.23 4 | 40 5 | - | 1.05 | - | 20 3 | 0.23 | [7] |
| Anthill-SD-RH5% | 1.5 4 | 31 5 | - | 2.09 | - | 10 3 | 0.44 | [7] |
| Anthill-SD-RH10% | 1.3 4 | 38 5 | - | 1.60 | - | 14 3 | 0.38 | [7] |
| Anthill-SD-RH15% | 1.22 4 | 41 5 | - | 1.12 | - | 19 3 | 0.32 | [7] |
| Anthill-SD-RH20% | 1.15 4 | 43 5 | - | 0.9 | - | 22.5 3 | 0.25 | [7] |
| KC | 2.25 2 | 3 | - | 116.93 | 19.26 | 0.46 3 | - | [22] |
| KS5 | 2.25 2 | 2 | - | 115.0 | 18.75 | 0.47 3 | - | [22] |
| KS10 | 2.25 2 | 1 | - | 129.25 | 24.13 | 0.47 3 | - | [22] |
| KS20 | 2.25 2 | 0.4 | - | 140.06 | 27.98 | 0.73 3 | - | [22] |
| KC | 2.15 2 | 12 | - | - | 24.41 | 1 3 | - | [23] |
| K20S | 2.25 2 | 9 | - | - | 28.34 | 1 3 | - | [23] |
| K20S3F | 2.35 2 | 11 | - | - | 14.00 | 0.25 3 | - | [23] |
| K20S6F | 2.45 2 | 13 | - | - | 13.75 | 0.27 3 | - | [23] |
| K20S9F | 2.60 2 | 13 | - | - | 13.00 | 0.36 3 | - | [23] |
| A | 1.66 1 | 13 | - | - | - | 1 | 0.1659 | [24] |
| AS10 | 1.54 1 | 16 | - | - | - | 1 | 0.1623 | [24] |
| AS10W5 | 1.47 1 | 19 | - | - | - | 1 | 0.1559 | [24] |
| AS10W10 | 1.59 1 | 23 | - | - | - | 1 | 0.1523 | [24] |
| AS10W20 | 1.53 1 | 25 | - | - | - | 1 | 0.1446 | [24] |
| s-1, green, 80 °C | - | - | - | - | - | - | - | [25] |
| s-2, 900 °C | 0.73 2 | 69 5 | - | 4.1 6 | - | - | 0.124 | [25] |
| s-3, 1000 °C | 0.76 2 | 67 5 | - | 5.0 6 | - | - | 0.132 | [25] |
| s-4, 1100 °C | 0.79 2 | 64 5 | - | 5.4 6 | - | - | 0.135 at 30 °C | [25] |
| s-5, 1200 °C | 0.90 2 | 59 5 | - | 6.5 6 | - | - | 0.146 | [25] |
| 2–0 | 1.7/2.8 7 | 34 5 | 0.011 8 | 2.9 | 11.9 | 316 9 | 0.19 | Present study |
| 2–4 | 1.6/2.7 7 | 34 5 | 0.010 8 | 7.8 | 14.2 | 284 9 | 0.14 | Present study |
| 2–9a | 1.5/2.7 7 | 36 5 | 0.010 8 | 4.2 | 8.2 | 176 9 | 0.138 | Present study |
| 2–13 g | 1.1/2.4 7 | 44 5 | 0.011 8 | 2.1 | 4.5 | 155 9 | 0.05 | Present study |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yefremova, S.; Yermishin, S.; Kablanbekov, A.; Satbaev, B.; Shalabaev, N.; Satbaev, S. Porous Refractories Synthesized Using Rice Husk and Rice Husk Processing Products. Materials 2025, 18, 5063. https://doi.org/10.3390/ma18215063
Yefremova S, Yermishin S, Kablanbekov A, Satbaev B, Shalabaev N, Satbaev S. Porous Refractories Synthesized Using Rice Husk and Rice Husk Processing Products. Materials. 2025; 18(21):5063. https://doi.org/10.3390/ma18215063
Chicago/Turabian StyleYefremova, Svetlana, Sergey Yermishin, Askhat Kablanbekov, Baimakhan Satbaev, Nurgali Shalabaev, and Serik Satbaev. 2025. "Porous Refractories Synthesized Using Rice Husk and Rice Husk Processing Products" Materials 18, no. 21: 5063. https://doi.org/10.3390/ma18215063
APA StyleYefremova, S., Yermishin, S., Kablanbekov, A., Satbaev, B., Shalabaev, N., & Satbaev, S. (2025). Porous Refractories Synthesized Using Rice Husk and Rice Husk Processing Products. Materials, 18(21), 5063. https://doi.org/10.3390/ma18215063






