Structure and Corrosion Resistance of Fe40Al5Cr0.2TiB Alloy After Casting and After Homogenization Annealing
Abstract
1. Introduction
2. Materials and Methods
- Examination of the alloy’s microstructure after casting (sample 1) and following homogenization annealing (sample 2) utilized a light microscope, specifically the Olympus GX51, prior to conducting corrosion tests. The phase analysis of the FeAl alloys before the corrosion test was performed using a JEOL JDX-7S diffractometer (Jeol, Tokyo, Japan) with copper radiation. Phase identification was performed using PCSIWIN software (version 2.0) and the JCPDS card database (ICDD, 2000). Transmission electron microscopy studies of the substructure were performed on a Hitachi HD-2300A microscope (STEM)( Tokyo, Japan). The quantitative characterization of the FeAl alloy microstructure was performed using the quantitative image analysis program MetIlo (version 9.07);
- Evaluation of the corrosion resistance of the FeAl alloys was carried out in a 5% NaCl solution employing potentiodynamic polarization and electrochemical impedance spectroscopy (EIS). These electrochemical analyses were conducted with the AUTOLAB system (PGSTAT30, Metrohm Autolab B.V., Utrecht, The Netherlands). A saturated calomel electrode (SCE) was used as the reference electrode, while a platinum grid served as the auxiliary electrode. Potentiodynamic curves were registered within a potential range of ±250 mV relative to the open circuit potential at a rate of 1 mV·s⁻¹. EIS studies were conducted at the corrosion potential across a frequency range from 50 kHz to 0.005 Hz, utilizing a sinusoidal wave with a 10 mV amplitude and recording 10 measurement points per frequency decade. All electrochemical tests were conducted at room temperature;
- The surface condition of the samples after corrosion testing was examined using a Hitachi S-4200 scanning electron microscope (SEM). The chemical composition was analyzed using an energy-dispersive X-ray spectrometer (EDS) by ThermoNoran (System Seven) (Madison, WI, USA), operated at an electron beam accelerating voltage of 15 keV. The spectrometer is combined with the microscope, as mentioned above. Backscattered electron diffraction was performed using the EBSD INCA HKL detector Nordlys II (Channel 5)—(Hobro, Denmark), which was equipped with the Hitachi S-3400N microscope. The phase analysis of the FeAl alloys after the corrosion test was performed similarly to the alloys before the corrosion test using a JEOL JDX-7S diffractometer.
3. Results and Discussion
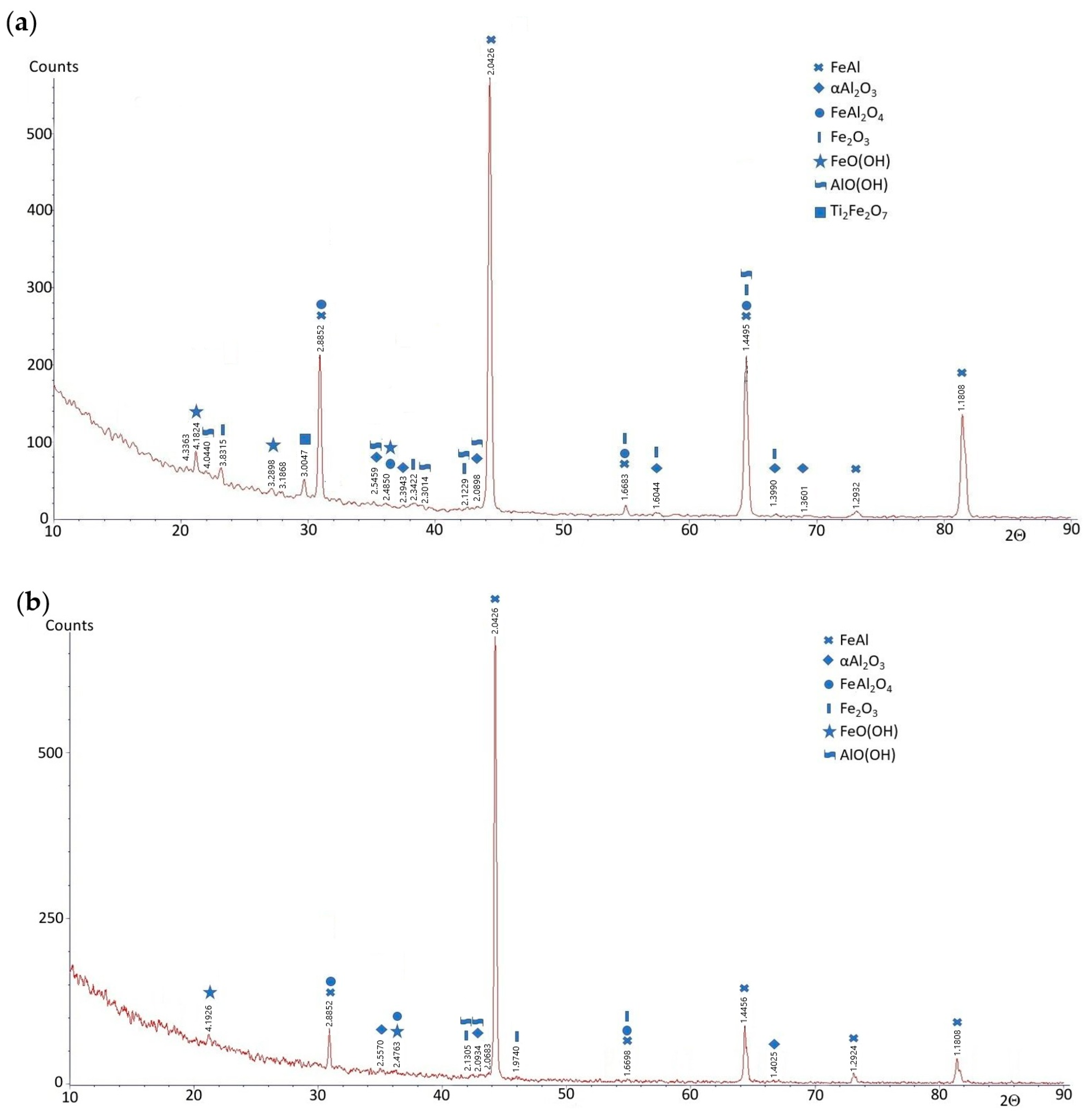
3.1. Characterization Before Corrosion Tests
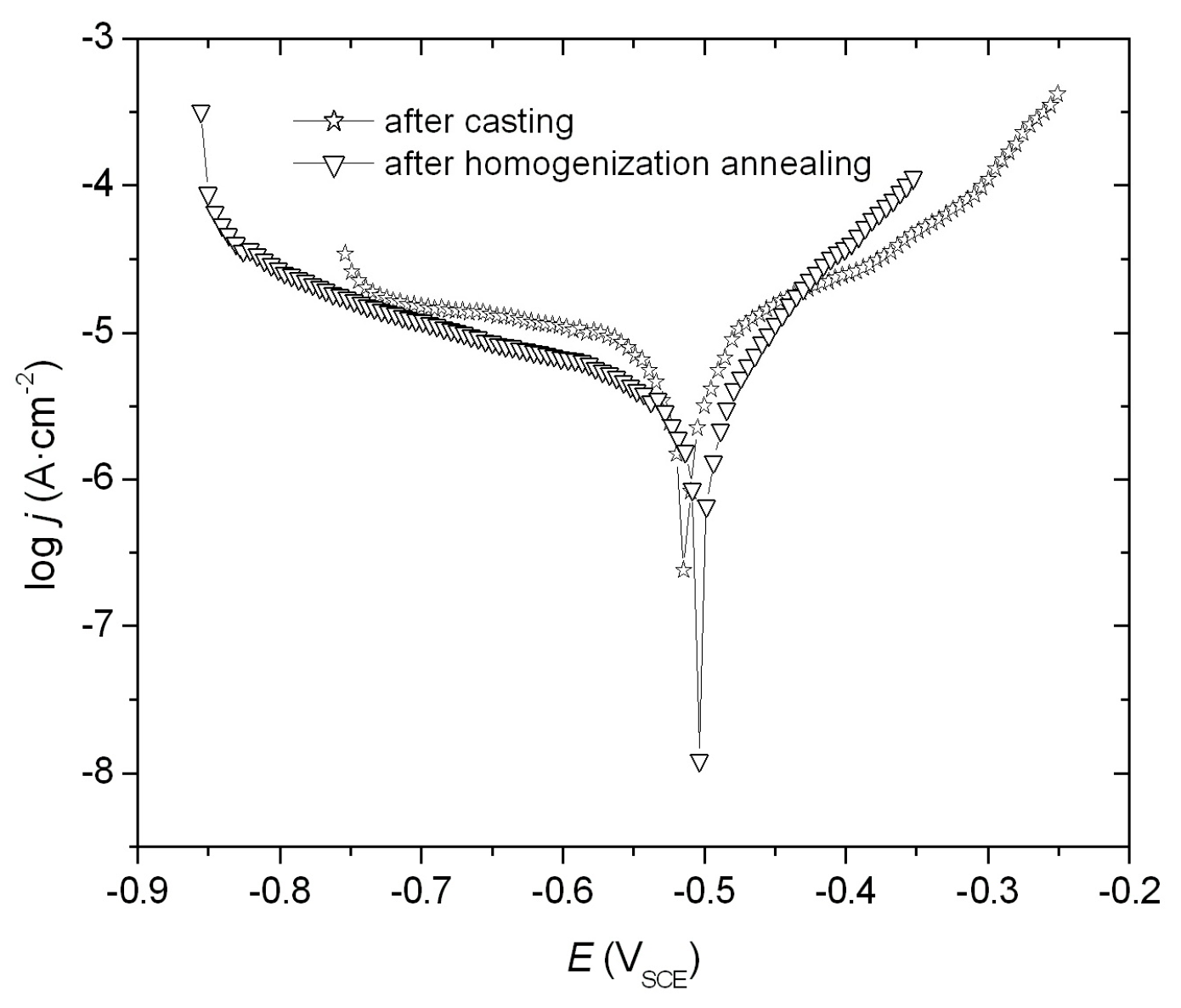
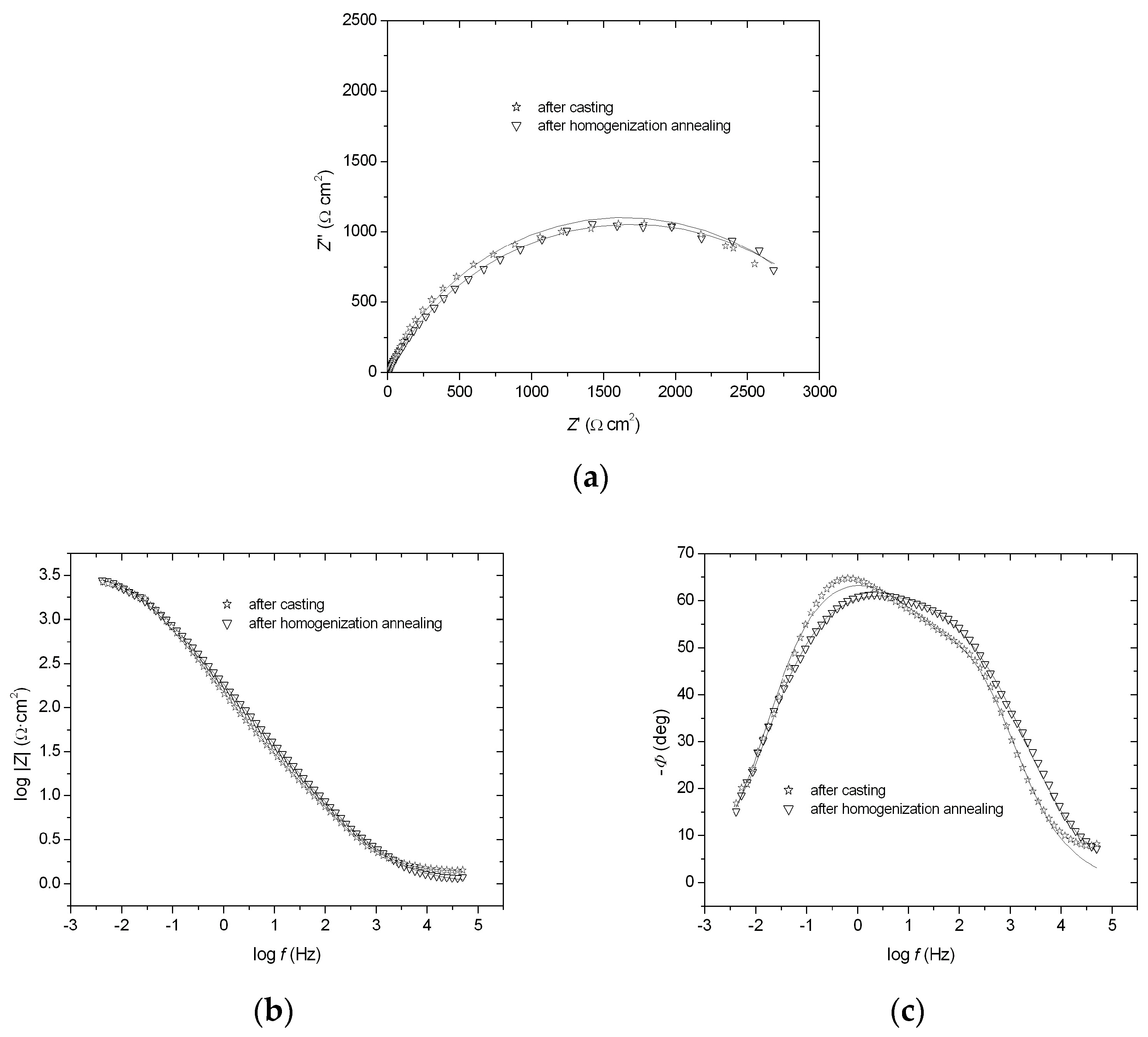
3.2. Corrosion Resistance Tests
3.3. Characterization After Corrosion Tests
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Panek, J.; Karolus, M. Structural studies on Ti-Cu alloy obtained by high-energy ball milling. Mater. Sci. Technol. 2020, 9, 978–981. [Google Scholar] [CrossRef]
- Meinhold, V.; Höhlich, D.; Mehner, T.; Lampke, T. Electrodeposition of Thick and Crack-Free Fe-Cr-Ni Coatings from a Cr (III) Electrolyte. Coatings 2022, 12, 56. [Google Scholar] [CrossRef]
- Popczyk, M. The hydrogen evolution reaction on electrolytic nickel—Based coatings containing metallic molybdenum. Mater. Sci. Forum 2010, 636–637, 1036–1041. [Google Scholar] [CrossRef]
- Kovalska, N.; Mulone, A.; Sort, J.; Klement, U.; Blugan, G.; Hansal, W.; Kautek, W. Electrodeposition of Soft Magnetic Fe-W-P Alloy Coatings from an Acidic Electrolyte. Coatings 2023, 13, 801. [Google Scholar] [CrossRef]
- Bojar, Z.; Przetakiewicz, W. Metal Materials Containing Intermetallic Phases; BEL Studio: Warsaw, Poland, 2006; ISBN 83-89968-03-7. [Google Scholar]
- Leyens, C.; Peters, M. (Eds.) Titanium and Titanium Alloys; Wiley-VCH Verlag GmbH: Weinheim, Germany, 2003. [Google Scholar]
- Yamaguchi, M.; Inui, H.; Ito, K. High-temperature structural intermetallics. Acta Mater. 2000, 48, 307–322. [Google Scholar] [CrossRef]
- Sheng, L.Y.; Xie, Y.; Xi, T.F.; Guo, J.T.; Zheng, Y.F.; Ye, H.Q. Microstructure characteristics and compressive properties of NiAl-based multiphase alloy during heat treatments. Mater. Sci. Eng. A 2011, 528, 8324–8331. [Google Scholar] [CrossRef]
- Frommeyer, G.; Drewes, E.J.; Engl, B. Physical and mechanical properties of iron-aluminium-(Mn, Si) lightweight steels. Metall. Res. Technol. 2000, 97, 1245–1253. [Google Scholar] [CrossRef]
- Czerwinski, F. Current Trends in Automotive Lightweighting Strategies and Materials. Materials 2021, 14, 6631. [Google Scholar] [CrossRef] [PubMed]
- Khaple, S.; Golla, B.R.; Prasad, V.V.S. A review on the current status of Fe-Al based ferritic lightweight steel. Def. Technol. 2023, 26, 1–22. [Google Scholar] [CrossRef]
- Penga, J.; Mosznerb, F.; Rechmanna, J.; Vogela, D.; Palma, M.; Rohwerdera, M. Influence of Al content and pre-oxidationon the aqueous corrosion resistance of binary Fe-Al alloys in sulphuric acid. Corros. Sci. 2019, 149, 123–132. [Google Scholar] [CrossRef]
- Shen, P.Z.; Song, M.; Gao, H.Y.; He, Y.H.; Zou, J.; Xu, N.P.; Huang, B.Y.; Liu, C. Structural characteristics and high-temperature oxidation behavior of porous Fe–40 at.%Al alloy. J. Mater. Sci. 2009, 44, 4413–4421. [Google Scholar] [CrossRef]
- Guilemany, J.M.; Cinca, N.; Dosta, S.; Lima, C. High-temperature oxidation of Fe40Al coatings obtained by HVOF thermal spray. Intermetallics 2007, 15, 1384–1394. [Google Scholar] [CrossRef]
- Kumar, M.; Kant, R.; Chand, S.; Prakash, U.; Sehgal, S.; Saxena, K.K.; Davim, J.P.; Prakash, C. High-Temperature Corrosion Performance of FeAl-Based Alloys Containing Carbon in Molten Salt. Metals 2021, 11, 2040. [Google Scholar] [CrossRef]
- Bednarczyk, I.; Kuc, D.; Niewielski, G. The structure of FeAl and Fe3Al-5%Cr intermetallic phase-based alloys after hot deformation processes. Arch. Mater. Sci. Eng. 2008, 30, 5–8. [Google Scholar]
- Rommerskirchen, I.; Eltester, B.; Grabke, H.J. Oxidation of β-FeAl and Fe-Al alloys. Mater. Corros. 1996, 47, 646–649. [Google Scholar] [CrossRef]
- Huang, D.; Yang, W.Y.; Sun, Z.Q.; Froyen, L. Preparation and mechanical properties of large-ingot Fe3Al-based alloys. J. Mater. Process. Technol. 2004, 146, 175–180. [Google Scholar] [CrossRef]
- Cebulski, J.; Pasek, D.; Bik, M.; Świerczek, K.; Jeleń, P.; Mroczka, K.; Dąbrowa, J.; Zajusz, M.; Wyrwa, J.; Sitarz, M. In-situ XRD investigations of FeAl intermetallic phase-based alloy oxidation. Corros. Sci. 2019, 164, 108344. [Google Scholar] [CrossRef]
- Kowalski, K.; Łosiewicz, B.; Budniok, A.; Kupka, M. Effect of alloying on corrosion resistance of B2 FeAl alloy in aqueous solution of sulfuric acid. Mater. Chem. Phys. 2011, 126, 314–318. [Google Scholar] [CrossRef]
- Hinderliter, B.R.; Croll, S.G.; Tallman, D.E.; Su, Q.; Bierwagen, G.P. Interpretation of EIS data from accelerated exposure of coated metals based on modeling of coating physical properties. Electrochim. Acta 2006, 51, 4505–4515. [Google Scholar] [CrossRef]
- Wykpis, K.; Popczyk, M.; Niedbała, J.; Budniok, A.; Łągiewka, E. Influence of the current density of deposition on the properties of Zn-Ni coatings. Mater. Sci. 2012, 47, 838–847. [Google Scholar] [CrossRef]


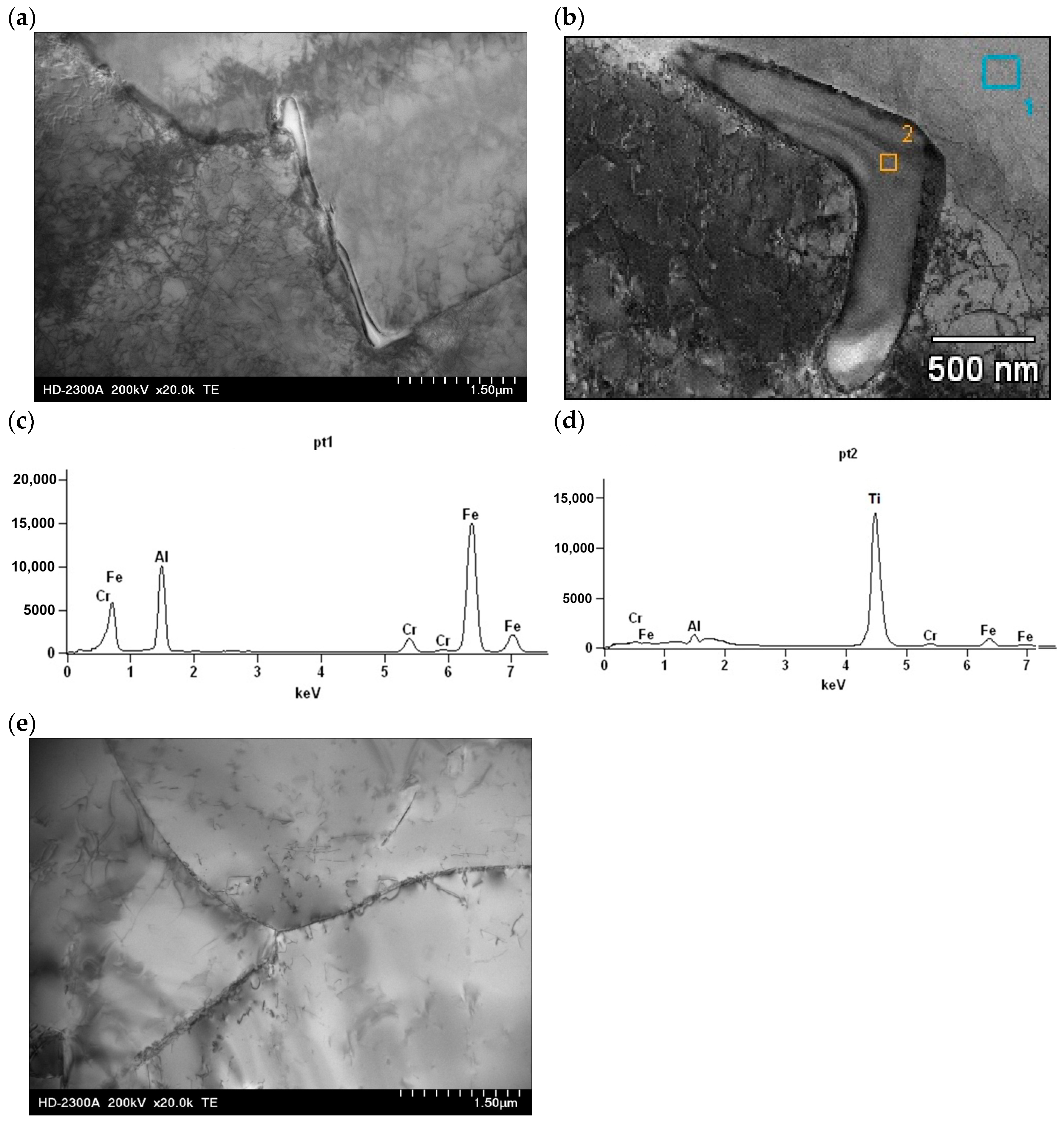

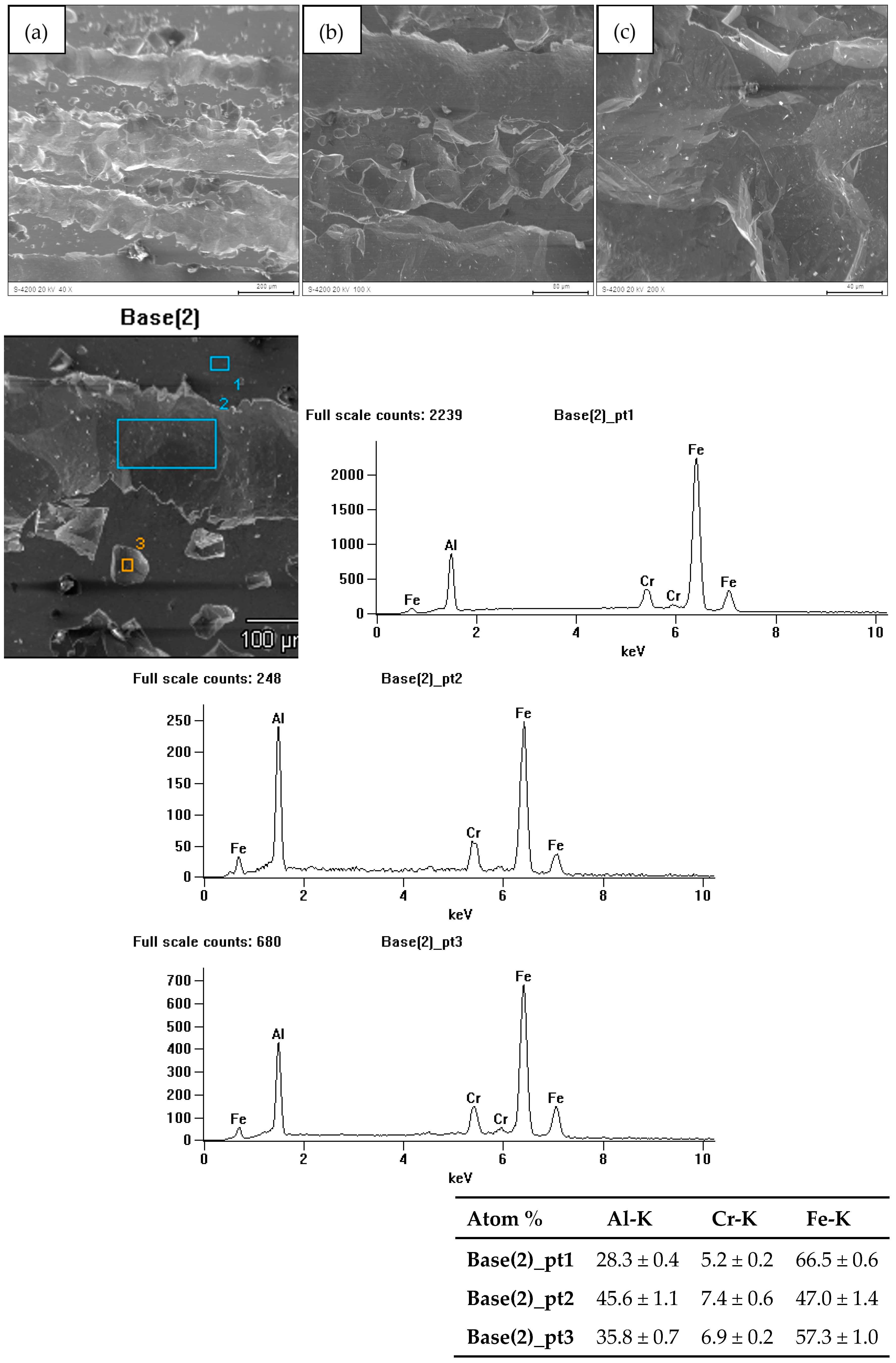


| Compound | Al | Cr | Ti | B | Fe |
|---|---|---|---|---|---|
| % mas. | 24.53 | 5.80 | 0.19 | 0.01 | rest |
| % at. | 40.10 | 4.86 | 0.18 | 0.06 | rest |
| FeAl Alloys | Ecorr [V] | jcorr [μA·cm−2] | Rp [Ω·cm2] |
|---|---|---|---|
| After casting | −0.513 | 5.8 | 4463 |
| After homogenization annealing | −0.503 | 3.8 | 7134 |
| FeAl Alloys | Rp1 (Ω·cm2) | T1 | ϕ1 | Rp2 (Ω·cm2) | T2 | ϕ2 | Rs (Ω·cm2) |
|---|---|---|---|---|---|---|---|
| After casting | 1.78 | 0.000347 | 0.65 | 3270 | 0.000206 | 0.75 | 1.05 |
| After homogenization annealing | 1.96 | 0.000335 | 0.47 | 3390 | 0.000097 | 0.70 | 1.03 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cebulski, J.; Pasek, D.; Popczyk, M.; Swinarew, A.; Gabor, J. Structure and Corrosion Resistance of Fe40Al5Cr0.2TiB Alloy After Casting and After Homogenization Annealing. Materials 2025, 18, 308. https://doi.org/10.3390/ma18020308
Cebulski J, Pasek D, Popczyk M, Swinarew A, Gabor J. Structure and Corrosion Resistance of Fe40Al5Cr0.2TiB Alloy After Casting and After Homogenization Annealing. Materials. 2025; 18(2):308. https://doi.org/10.3390/ma18020308
Chicago/Turabian StyleCebulski, Janusz, Dorota Pasek, Magdalena Popczyk, Andrzej Swinarew, and Jadwiga Gabor. 2025. "Structure and Corrosion Resistance of Fe40Al5Cr0.2TiB Alloy After Casting and After Homogenization Annealing" Materials 18, no. 2: 308. https://doi.org/10.3390/ma18020308
APA StyleCebulski, J., Pasek, D., Popczyk, M., Swinarew, A., & Gabor, J. (2025). Structure and Corrosion Resistance of Fe40Al5Cr0.2TiB Alloy After Casting and After Homogenization Annealing. Materials, 18(2), 308. https://doi.org/10.3390/ma18020308







