Design of CoMoCe-Oxide Nanostructured Composites as Robust Bifunctional Electrocatalyst for Water Electrolysis Overall Efficiency
Abstract
1. Introduction
2. Experimental Section
2.1. Materials
2.2. Synthesis of Trimetallic CoMoCe-Oxide Catalysts
2.3. Characterization and Electrochemical Measurements
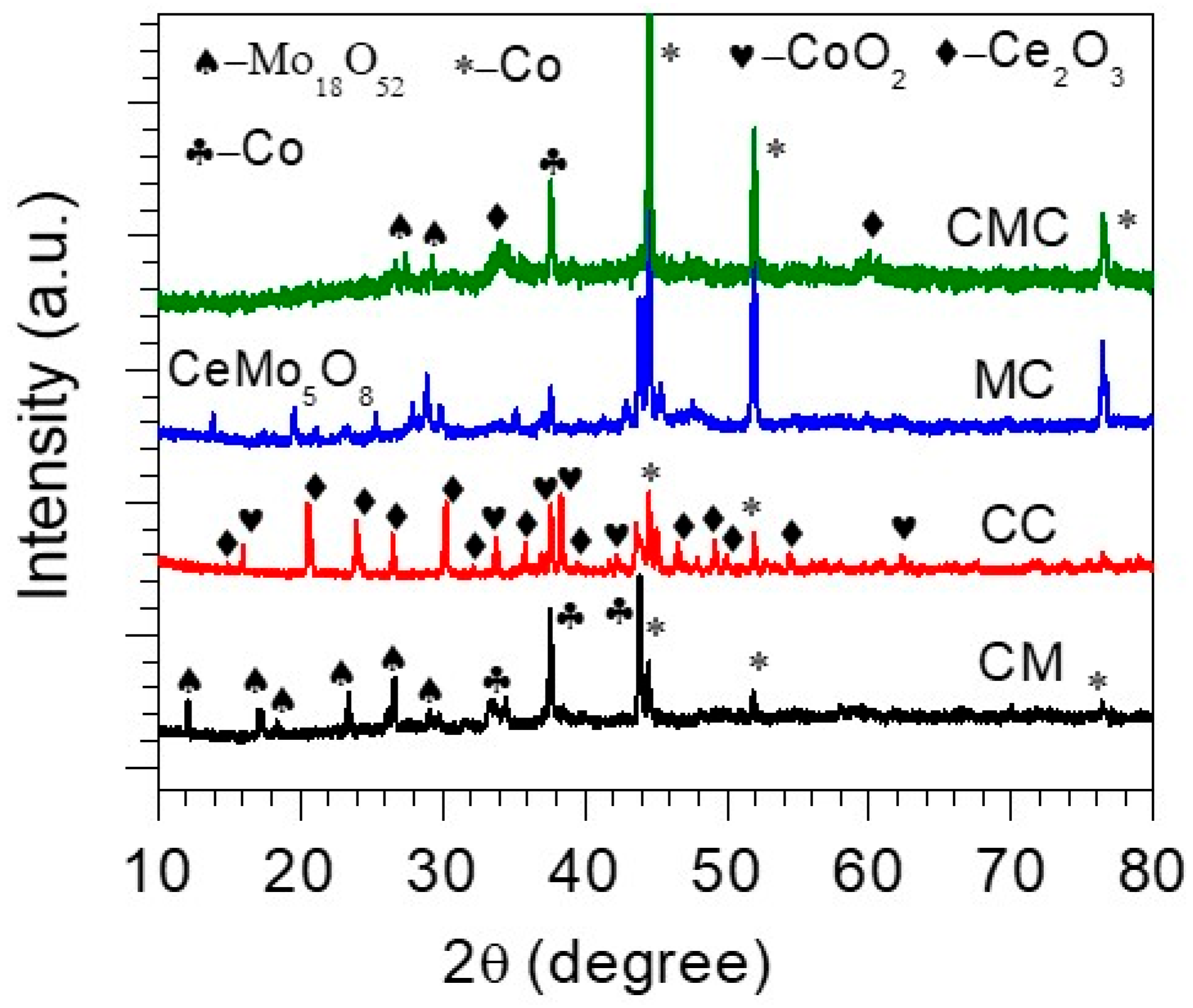
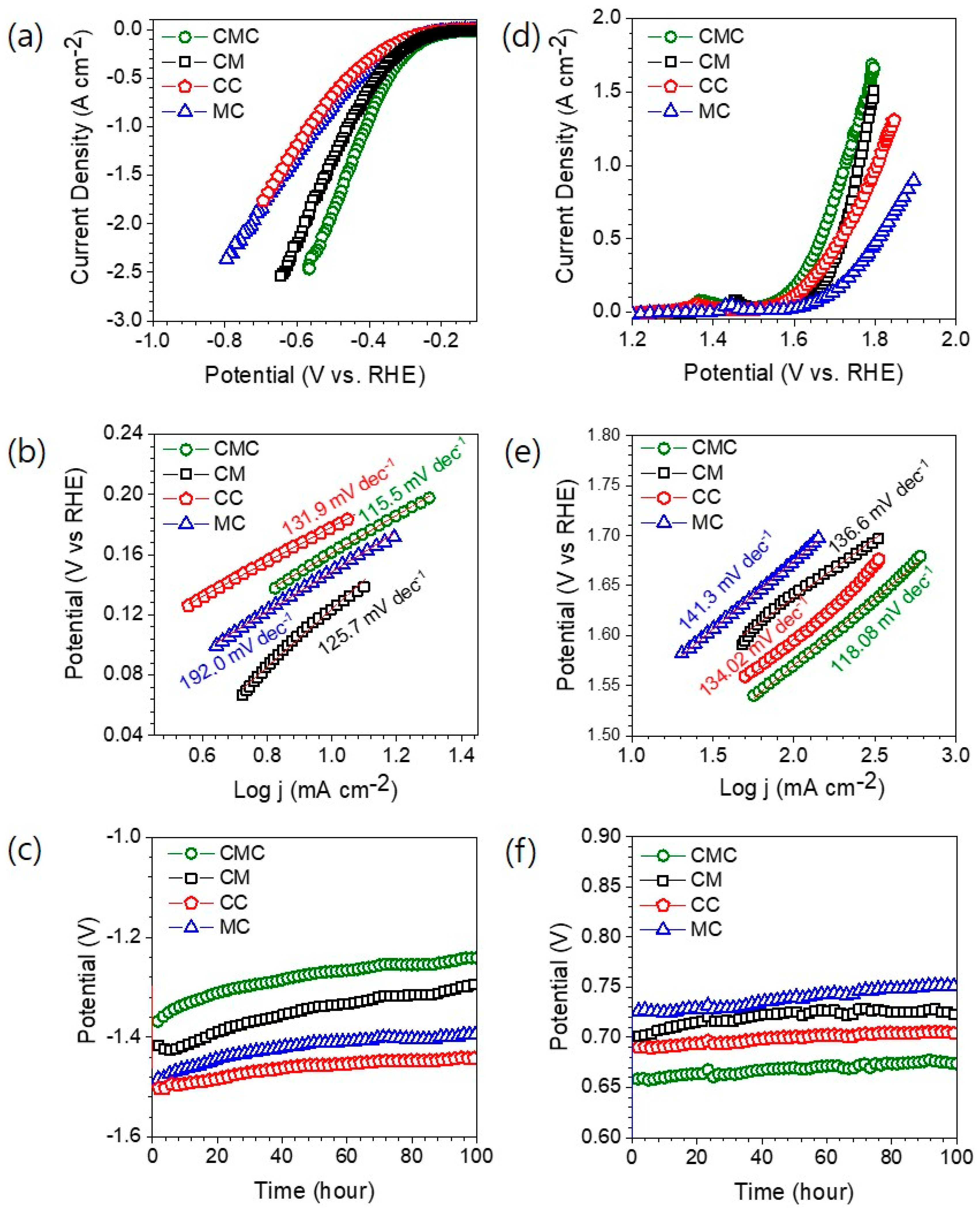
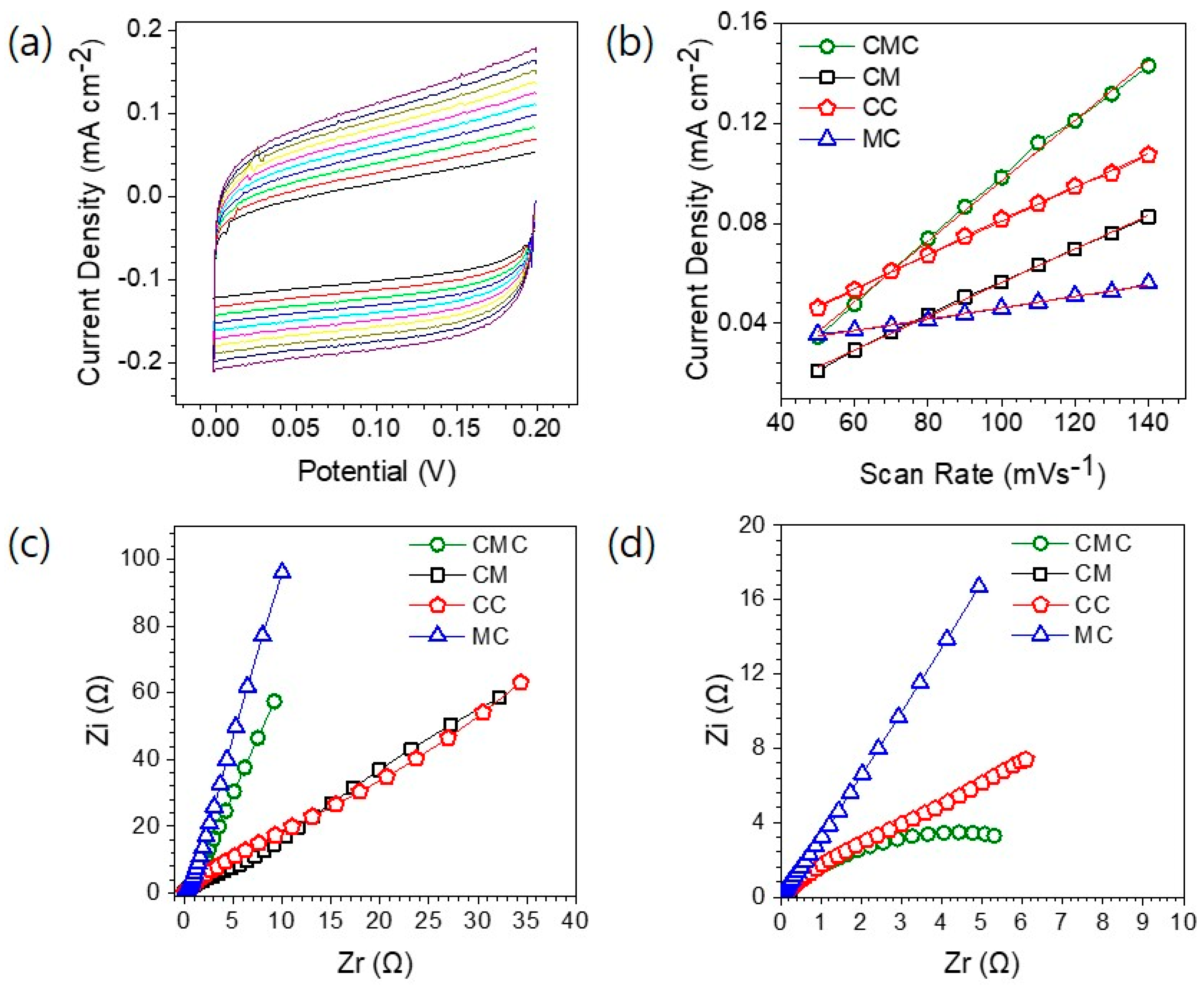
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- IEA. Electricity Market Report 2023; International Energy Agency (IEA): Paris, France, 2023; Available online: https://www.iea.org/reports/electricity-market-report-2023 (accessed on 23 July 2025).
- IEA. Electricity 2024-Analysis; International Energy Agency (IEA): Paris, France, 2024; Available online: https://www.iea.org/reports/electricity-2024 (accessed on 23 July 2025).
- Statistical Review of World Energy 2020, 69th ed. Available online: https://www.bp.com/content/dam/bp/business-sites/en/global/corporate/pdfs/energy-economics/statistical-review/bp-stats-review-2020-full-report.pdf (accessed on 23 July 2025).
- U.S. Energy Information Administration. Hydrogen explained, Independent Statistics and Analysis. 23 June 2023. Available online: https://www.eia.gov/energyexplained/hydrogen/ (accessed on 23 July 2025).
- Inamdar, A.I.; Salunke, A.S.; Shrestha, N.K.; Im, H. Heterogeneous oxide/sulfide materials as superior bifunctional electrocatalysts for carbon-neutral green hydrogen production: A short review. Appl. Phys. Rev. 2024, 11, 041310. [Google Scholar] [CrossRef]
- Ibrahim, M.M.; Mezni, A.; Alsawat, M.; Kumeria, T.; Das, M.R.; Alzahly, S.; Aldalbahi, A.; Gornicka, K.; Ryl, J.; Amin, M.A.; et al. Enhanced hydrogen evolution reaction on highly stable titania-supported PdO and Eu2O3 nanocomposites in a strong alkaline solution. Int. J. Energy Res. 2019, 43, 5367–5383. [Google Scholar] [CrossRef]
- Zuben, T.; Moreira, D.; Germscheidt, R.; Yoshimura, R.; Dorretto, D.; Araujo, A.; Salles, A.; Bonacin, J. Is Hydrogen Indispensable for a Sustainable World? A Review of H2 Applications and Perspectives for the Next Years. J. Braz. Chem. Soc. 2022, 33, 824–843. [Google Scholar] [CrossRef]
- Inamdar, A.I.; Chavan, H.; Hou, B.; Lee, C.; Lee, S.; Cha, S.; Kim, H.; Im, H. A Robust Nonprecious CuFe Composite as a Highly Efficient Bifunctional Catalyst for Overall Electrochemical Water Splitting. Small 2020, 16, 1905884. [Google Scholar] [CrossRef]
- Inamdar, A.I.; Salunke, A.S.; Seok, J.H.; Chavan, H.S.; Shrestha, N.K.; Lee, S.U.; Cho, S.; Im, H. Cobalt oxide/cerium oxide heterogeneous interfaces as advanced durable and bifunctional electrocatalysts for robust industrially relevant overall water splitting. J. Mater. Chem. A 2024, 12, 31362–31374. [Google Scholar] [CrossRef]
- Chavan, H.S.; Lee, C.; Inamdar, A.I.; Han, J.; Park, S.; Cho, S.; Shreshta, N.K.; Lee, S.U.; Hou, B.; Im, H.; et al. Designing and Tuning the Electronic Structure of Nickel–Vanadium Layered Double Hydroxides for Highly Efficient Oxygen Evolution Electrocatalysis. ACS Catal. 2022, 12, 3821–3831. [Google Scholar] [CrossRef]
- Shrestha, N.K.; Patil, S.A.; Seok, J.H.; Salunke, A.S.; Cho, S.; Inamdar, A.I.; Park, Y.; Lee, S.U.; Kim, H.; Im, H. Cerium guided site-selective crystal disorder engineering of MIL-88B(Ni) frameworks for electrocatalysis offering high-performance water oxidation. Mater. Today Phys. 2023, 38, 101252. [Google Scholar] [CrossRef]
- Chen, S.; Zhao, Y.; Chang, C.; Wang, X.; Hou, L.; Jin, J.; Gao, F. Construction of heterogeneous core–shell CoFeMo polymetallic sulfide/N-rich carbon skeleton composite catalysts based on stepwise displacement of MOF for overall water splitting. Fuel 2024, 365, 131230. [Google Scholar] [CrossRef]
- Inamdar, A.I.; Chavan, H.S.; Pawar, S.M.; Kim, H.; Im, H. NiFeCo oxide as an efficient and sustainable catalyst for the oxygen evolution reaction. Int. J. Energy Res. 2020, 44, 1789–1797. [Google Scholar] [CrossRef]
- Inamdar, A.I.; Chavan, H.; Seok, J.; Lee, C.; Shin, G.; Park, S.; Yeon, S.; Cho, S.; Park, Y.; Shrestha, N.; et al. Optimal rule-of-thumb design of NiFeMo layered double hydroxide nanoflakes for highly efficient and durable overall water-splitting at large currents. J. Mater. Chem. A 2022, 10, 20497–20508. [Google Scholar] [CrossRef]
- Dalai, N.; Mohanty, B.; Mitra, A.; Jena, B. Highly Active Ternary Nickel–Iron oxide as Bifunctional Catalyst for Electrochemical Water Splitting. Chem. Sel. 2019, 4, 7791–7796. [Google Scholar] [CrossRef]
- Pickrahn, K.L.; Garg, A.; Bent, S.F. ALD of Ultrathin Ternary Oxide Electrocatalysts for Water Splitting. ACS Catal. 2015, 5, 1609–1616. [Google Scholar] [CrossRef]
- Xu, M.; Li, W.; Zhong, M.; Yang, J.; Gao, M.; Pinna, N.; Lu, X. Trimetallic FeNiMo Nanofibers as High-Efficiency Electrocatalyst for Robust Oxygen Evolution. ACS Mater. Lett. 2024, 6, 3548–3556. [Google Scholar] [CrossRef]
- Chai, X.; Yuan, C.; Gao, R.T.; Wang, L. MoOx/CoMo on Cu Nanowires as an Efficiently Bifunctional Electrocatalyst for Overall Water Splitting. ChemCatChem 2024, 16, e202400582. [Google Scholar] [CrossRef]
- Yan, X.; Tian, L.; Atkins, S.; Liu, Y.; Murowchick, J.; Chen, X. Converting CoMoO4 into CoO/MoOx for Overall Water Splitting by Hydrogenation. ACS Sustain. Chem. Eng. 2016, 4, 3743–3749. [Google Scholar] [CrossRef]
- Bao, J.; Wang, Z.; Xie, J.; Xu, L.; Lei, F.; Guan, M.; Huang, Y.; Zhao, Y.; Xia, J.; Li, H. The CoMo-LDH ultrathin nanosheet as a highly active and bifunctional electrocatalyst for overall water splitting. Inorg. Chem. Front. 2018, 5, 2964–2970. [Google Scholar] [CrossRef]
- Sangamithirai, M.; Vijayarangan, M.; Gayathri, A.; Muthamildevi, M.; Jayabharathi, J. Design strategy of encapsulated nanoplates and nanorods (ID-CoMo): Enhanced catalytic activity and sustainability for overall & solar cell water splitting. Sustain. Energy Fuels 2024, 8, 3124–3135. [Google Scholar]
- Gholampour, N.; Zhao, Y.; Devred, F.; Sassoye, C.; Casale, S.; Debecker, D.P. CO2 Methanation over Cobalt Nanoparticles Embedded in ZIF-L–Derived Porous Carbon. ChemCatChem 2023, 15, e202201338. [Google Scholar] [CrossRef]
- Li, H.; He, H.; Jiang, T.; Du, Y.; Wu, Z.; Xu, L.; Wang, X.; Liu, X.; Yu, W.; Xue, W. Preparation of Co/S co-doped carbon catalysts for excellent methylene blue degradation. Int. J. Miner. Metall. Mater. 2025, 32, 169–181. [Google Scholar] [CrossRef]
- Cattelan, M.; Yang, J.; Cielo, L.; Nappini, S.; Carlotto, S.; Nalesso, M.; Azcona, I.N.; Yivlialin, R.; Sun, X.; Bussetti, G.; et al. Operando Exploration of CoAl-LDH: Transformations Driving Alkaline Oxygen Evolution Reaction. Small 2025, 21, 2412351. [Google Scholar] [CrossRef]
- Salvò, D.; Mosconi, D.; Neyman, A.; Bar-Sadan, M.; Calvillo, L.; Granozzi, G.; Cattelan, M.; Agnoli, S. Nanoneedles of Mixed Transition Metal Phosphides as Bifunctional Catalysts for Electrocatalytic Water Splitting in Alkaline Media. Nanomaterials 2023, 13, 683. [Google Scholar] [CrossRef]
- Thomas, T.; Jayababu, N.; Shruthi, J.; Mathew, A.; Cerdán-Pasarán, A.; Alejandro Hernández-Magallanes, J.; Sanal, K.C.; Reshmi, R. Room temperature ammonia sensing of α-MoO3 nanorods grown on glass substrates. Thin Solid Film. 2021, 722, 138575. [Google Scholar] [CrossRef]
- Carraro, F.; Calvillo, L.; Cattelan, M.; Favaro, M.; Righetto, M.; Nappini, S.; Pís, I.; Celorrio, V.; Fermín, D.J.; Martucci, A.; et al. Fast One-Pot Synthesis of MoS2/Crumpled Graphene p−n Nanonjunctions for Enhanced Photoelectrochemical Hydrogen Production. ACS Appl. Mater. Interfaces 2015, 7, 25685–25692. [Google Scholar] [CrossRef]
- Bortamuly, R.; Konwar, G.; Boruah, P.K.; Das, M.R.; Mahanta, D.; Saikia, P. CeO2-PANI-HCl and CeO2-PANI-PTSA composites: Synthesis, characterization, and utilization as supercapacitor electrode materials. Ionics 2020, 26, 5747–5756. [Google Scholar] [CrossRef]
- Artiglia, L.; Agnoli, S.; Paganini, M.C.; Cattelan, M.; Granozzi, G. TiO2@CeOx Core−Shell Nanoparticles as Artificial Enzymes with Peroxidase-Like Activity. ACS Appl. Mater. Interfaces 2014, 6, 20130–20136. [Google Scholar] [CrossRef]
- Bêche, E.; Charvin, P.; Perarnau, D.; Abanades, S.; Flamant, G. Ce 3d XPS investigation of cerium oxides and mixed cerium oxide (CexTiyOz). Surf. Interface Anal. 2008, 40, 264–267. [Google Scholar] [CrossRef]
- Paolucci, V.; Basso, M.; Ricci, V.; Colusso, E.; Cattelan, M.; Napolitani, E.; Martucci, A.; Cantalini, C. Pulsed-laser annealed amorphous/crystalline a-V2O5/VO2 heterostructure for selective NO2 and H2 sensing. Sens. Actuators B Chem. 2025, 428, 7235. [Google Scholar] [CrossRef]
- Guo, Y.; Zhou, X.; Tang, J.; Tanaka, S.; Kaneti, Y.V.; Na, J.; Jiang, B.; Yamauchi, Y.; Bando, Y. Multiscale structural optimization: Highly efficient hollow iron-doped metal sulfide heterostructures as bifunctional electrocatalysts for water splitting. Nano Energy 2020, 75, 104913. [Google Scholar] [CrossRef]
- Borthakur, P.; Boruah, P.K.; Das, M.R.; Ibrahim, M.M.; Altalhi, T.; El-Sheshtawy, H.S.; Szunerits, S.; Boukherroub, R.; Amin, M.A. CoS2 Nanoparticles Supported on rGO, g-C3N4, BCN, MoS2, and WS2 Two-Dimensional Nanosheets with Excellent Electrocatalytic Performance for Overall Water Splitting: Electrochemical Studies and DFT Calculations. ACS Appl. Energy Mater. 2021, 4, 1269–1285. [Google Scholar] [CrossRef]
- Ren, X.; Wu, D.; Ge, R.; Sun, X.; Ma, H.; Yan, T.; Zhang, Y.; Du, B.; Wei, Q.; Chen, L. Self-supported CoMoS4 nanosheet array as an efficient catalyst for hydrogen evolution reaction at neutral pH. Nano Res. 2024, 11, 2024–2033. [Google Scholar] [CrossRef]
- Wang, C.; Wang, F.; Qiu, S.Y.; Gao, J.; Gu, L.-L.; Wang, K.; Zuo, P.-J.; Sun, K.-N.; Zhu, X.-D. Integrating Co3O4 nanoparticles with MnO2 nanosheets as bifunctional electrocatalysts for water splitting. Int. J. Hydrogen Energy 2021, 46, 10356–10365. [Google Scholar] [CrossRef]
- Sultana, U.K.; Riches, J.D.; O’Mullane, A.P. Gold Doping in a Layered Co–Ni Hydroxide System via Galvanic Replacement for Overall Electrochemical Water Splitting. Adv. Funct. Mater. 2018, 28, 1804361. [Google Scholar] [CrossRef]
- Bao, W.L.J.; Guan, M.; Zhao, Y.; Lian, J.; Qiu, J.; Xu, L.; Huang, Y.; Qian, J.; Li, H. Nickel–cobalt-layered double hydroxide nanosheet arrays on Ni foam as a bifunctional electrocatalyst for overall water splitting. Dalton Trans. 2017, 46, 8372. [Google Scholar]
- Guo, P.; Wu, J.; Li, X.; Luo, J.; Lau, W.; Liu, H.; Sun, X.; Liu, L. Micropatterned elastic ionic polyacrylamide hydrogel for low-voltage capacitive and organic thin-film transistor pressure sensors. Nano Energy 2018, 47, 96–104. [Google Scholar] [CrossRef]
- Salunke, A.S.; Patil, R.P.; Shrestha, N.K.; Cho, S.; Im, H.; Inamdar, A.I. Sustainable electrochemical energy generation using ultrathin nanoflakes of mixed phase CoMnMoS as a robust electrocatalyst. J. Power Sources 2025, 642, 236954. [Google Scholar] [CrossRef]
- Mujawar, S.H.; Salunke, A.S.; Deokate, R.J.; Salunke, S.T.; Shrestha, N.K.; Im, H.; Inamdar, A.I. Heterostructured NiMo-sulfide micro-pillar arrays for advanced alkaline electrocatalytic clean hydrogen production via overall water splitting. Appl. Surf. Sci. 2024, 661, 160081. [Google Scholar] [CrossRef]
- Salunke, A.S.; Deokate, R.J.; Salunke, S.T.; Mujawar, S.H.; Shrestha, N.K.; Im, H.; Inamdar, A.I. Iron-hydroxide controlled by cobalt and aluminium as an advanced electrocatalysts for overall water splitting and its temperature dependence. J. Alloys Compd. 2024, 989, 174334. [Google Scholar] [CrossRef]
- Ding, J.; Shao, Q.; Feng, Y.; Huang, X. Ruthenium-nickel sandwiched nanoplates for efficient water splitting electrocatalysis. Nano Energy 2018, 47, 1–7. [Google Scholar] [CrossRef]
- Li, Y.; Zhu, Z.; Zhong, Y.L.; Jin, Y.; Saha, P.; Cheng, Q. Heterogeneous Cr-doped Co3S4/NiMoS4 bifunctional electrocatalyst for efficient overall water splitting. J. Power Source 2024, 614, 234969. [Google Scholar] [CrossRef]
- Hou, J.; Zhang, B.; Li, Z.; Cao, S.; Sun, Y.; Wu, Y.; Gao, Z.; Sun, L. Vertically Aligned Oxygenated-CoS2–MoS2 Heteronanosheet Architecture from Polyoxometalate for Efficient and Stable Overall Water Splitting. ACS Catal. 2018, 8, 4612–4621. [Google Scholar] [CrossRef]
- Huang, L.; Chen, D.; Luo, G.; Lu, Y.R.; Chen, C.; Zou, Y.; Dong, C.L.; Li, Y.; Wang, S. Zirconium-regulation-induced bifunctionality in 3D cobalt–iron oxide nanosheets for overall water splitting. Adv. Mater. 2019, 31, 1901439. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Zhao, C.; Qu, Y.; Zhou, H.; Zhou, F.; Wang, J.; Wu, Y.; Li, Y. Trifunctional self-supporting cobalt-embedded carbon nanotube films for ORR, OER, and HER triggered by solid diffusion from bulk metal. Adv. Mater. 2019, 31, 1808043. [Google Scholar] [CrossRef] [PubMed]
- Li, G.-L.; Qiao, X.-Y.; Miao, Y.-Y.; Wang, T.-Y.; Deng, F. Synergistic Effect of N-NiMoO4/Ni Heterogeneous Interface with Oxygen Vacancies in N-NiMoO4/Ni/CNTs for Superior Overall Water Splitting. Small 2023, 19, 2207196. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Inamdar, A.I.; Salunke, A.S.; Patil, J.V.; Mali, S.S.; Hong, C.K.; Ali, B.; Patil, S.A.; Shrestha, N.K.; Lee, S.; Cho, S. Design of CoMoCe-Oxide Nanostructured Composites as Robust Bifunctional Electrocatalyst for Water Electrolysis Overall Efficiency. Materials 2025, 18, 4052. https://doi.org/10.3390/ma18174052
Inamdar AI, Salunke AS, Patil JV, Mali SS, Hong CK, Ali B, Patil SA, Shrestha NK, Lee S, Cho S. Design of CoMoCe-Oxide Nanostructured Composites as Robust Bifunctional Electrocatalyst for Water Electrolysis Overall Efficiency. Materials. 2025; 18(17):4052. https://doi.org/10.3390/ma18174052
Chicago/Turabian StyleInamdar, Akbar I., Amol S. Salunke, Jyoti V. Patil, Sawanta S. Mali, Chang Kook Hong, Basit Ali, Supriya A. Patil, Nabeen K. Shrestha, Sejoon Lee, and Sangeun Cho. 2025. "Design of CoMoCe-Oxide Nanostructured Composites as Robust Bifunctional Electrocatalyst for Water Electrolysis Overall Efficiency" Materials 18, no. 17: 4052. https://doi.org/10.3390/ma18174052
APA StyleInamdar, A. I., Salunke, A. S., Patil, J. V., Mali, S. S., Hong, C. K., Ali, B., Patil, S. A., Shrestha, N. K., Lee, S., & Cho, S. (2025). Design of CoMoCe-Oxide Nanostructured Composites as Robust Bifunctional Electrocatalyst for Water Electrolysis Overall Efficiency. Materials, 18(17), 4052. https://doi.org/10.3390/ma18174052










