Reduced Graphene Oxide Aerogels Cartridges for Solid Phase Extraction of Benzotriazoles
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis and Characterization of the rGO Aerogels
2.3. Solid Phase Extraction Tests
2.4. Chromatographic Conditions
3. Results and Discussion
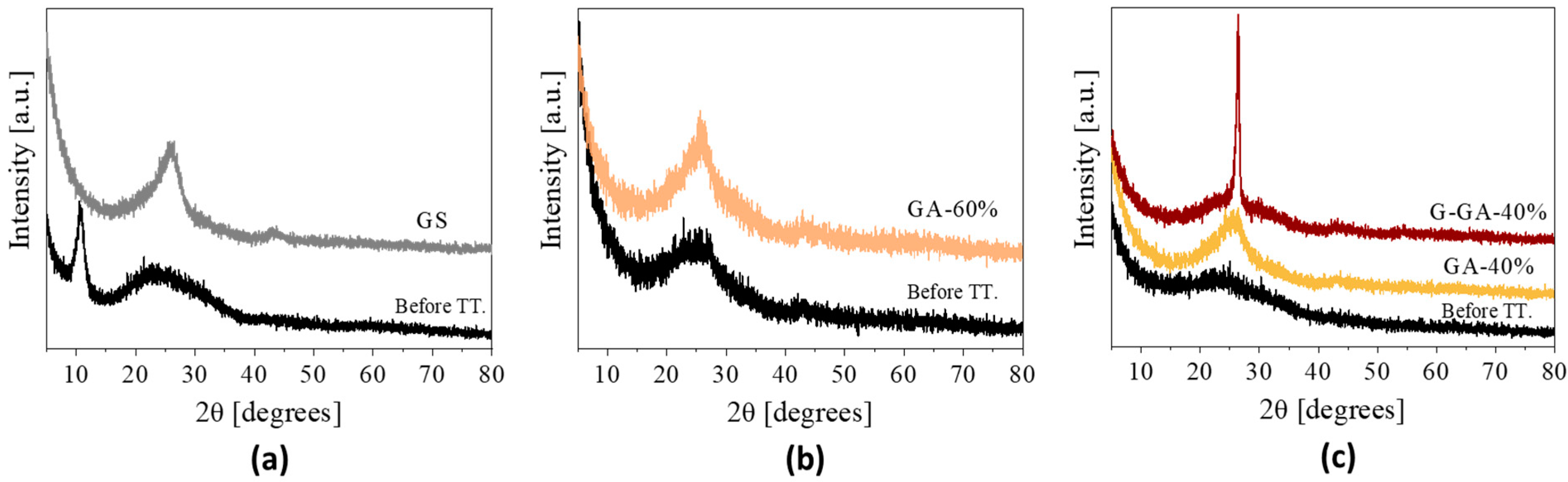
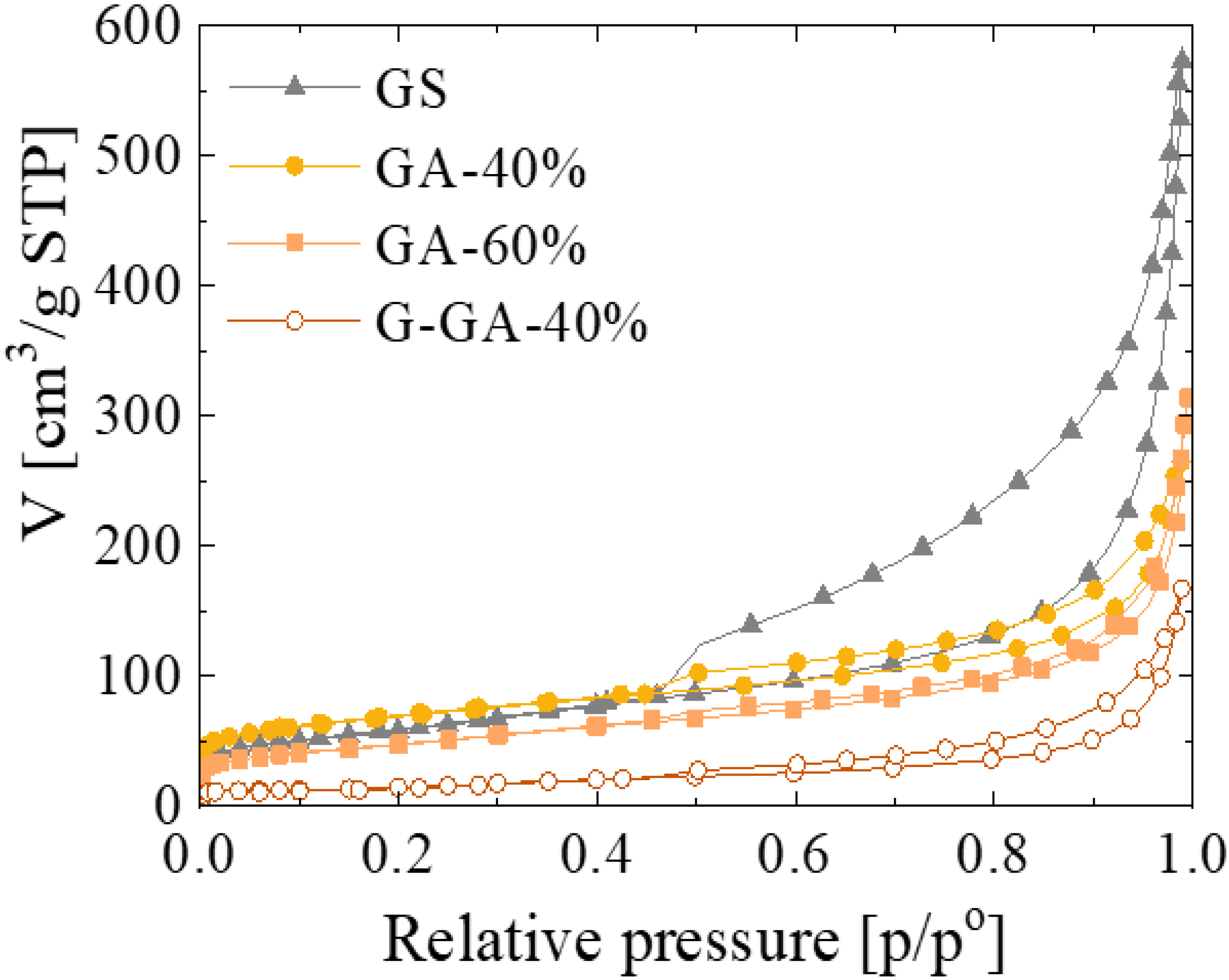
3.1. Properties of the rGO Aerogels
3.2. rGO Aerogels in Solid Phase Extraction
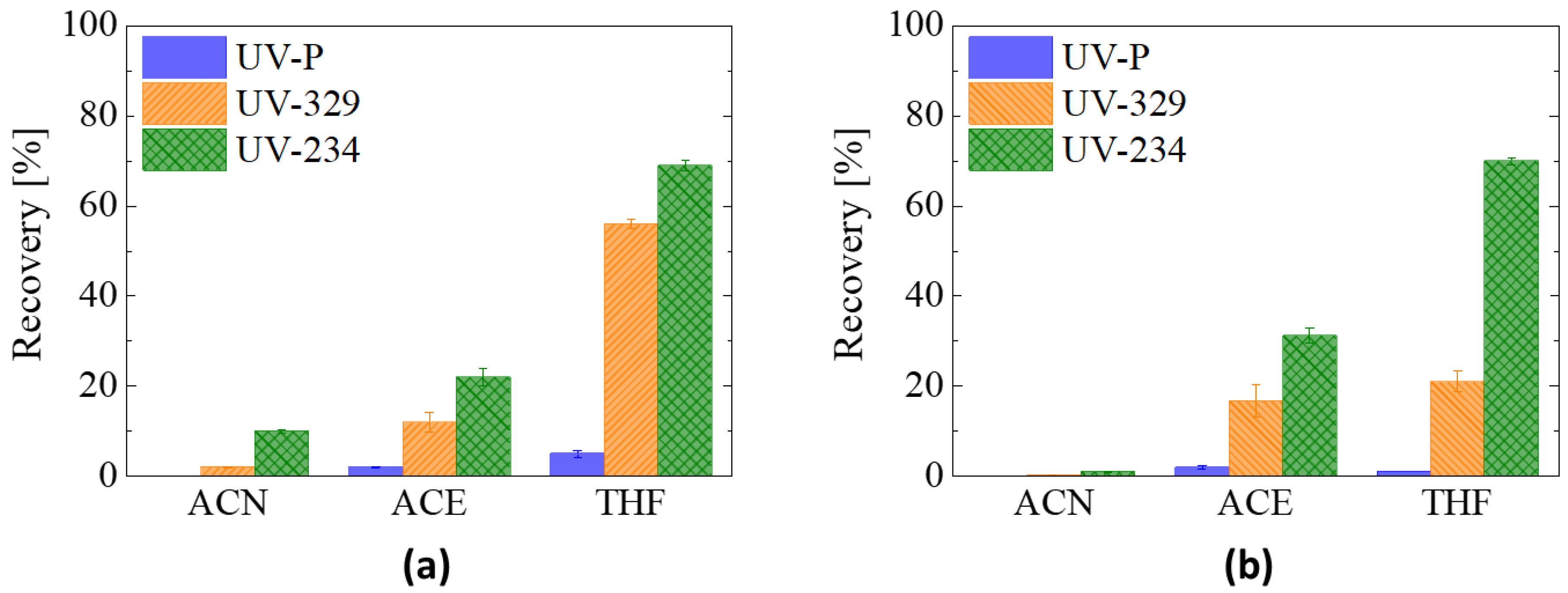
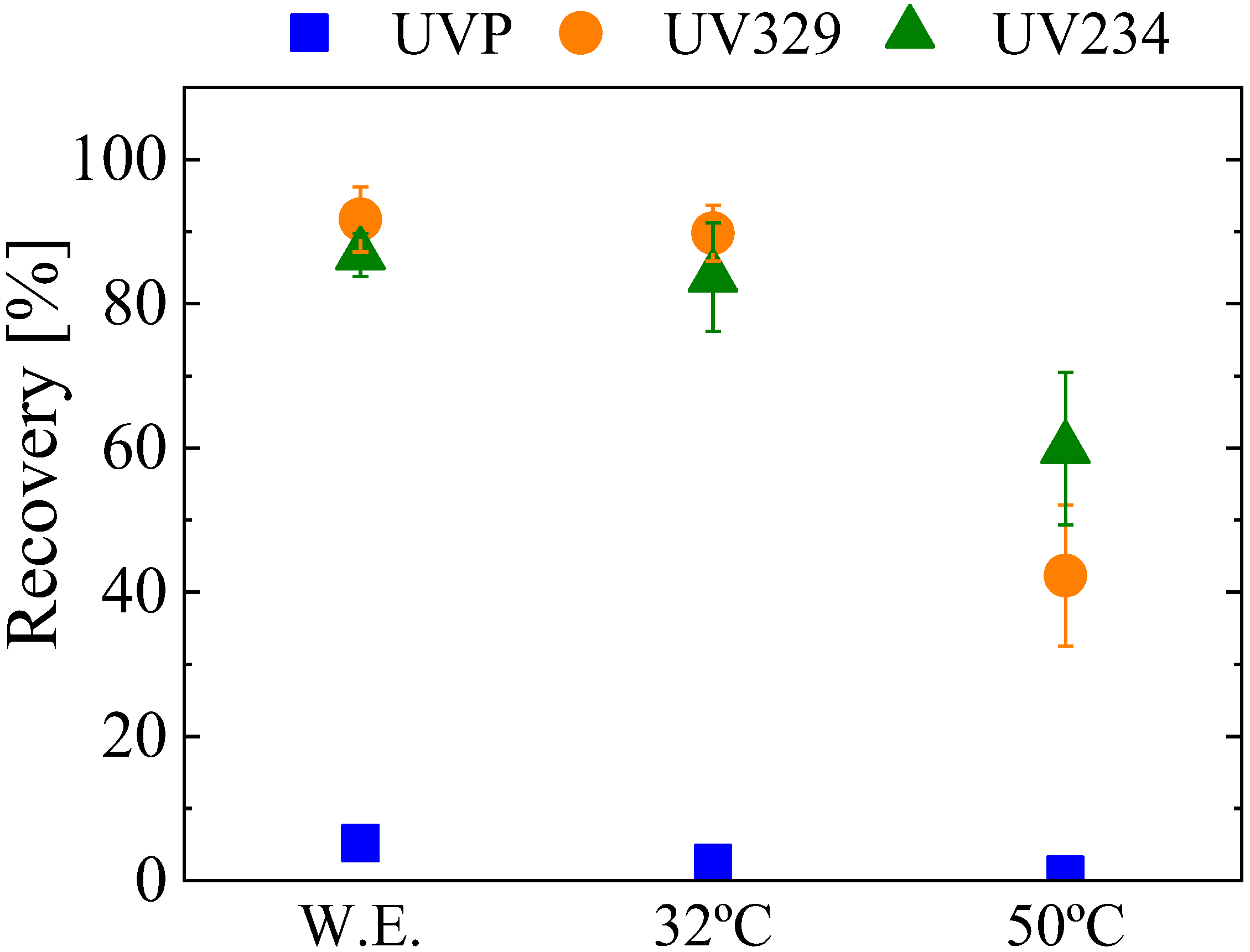
3.2.1. Recovery Test: Elution Solvent and rGO Aerogel Performance
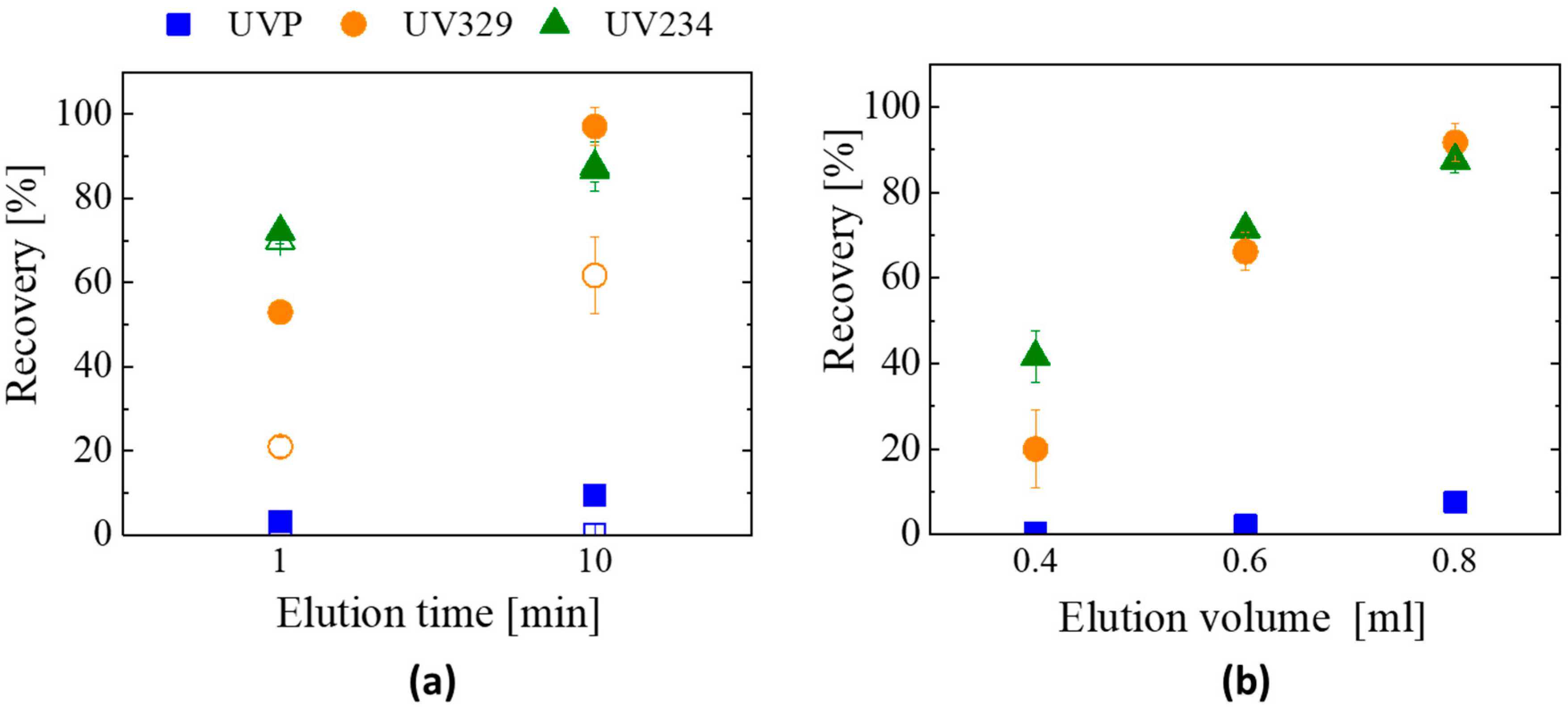
3.2.2. Optimization of the SPE Method: Elution Time and Elution Volume
3.3. Applicability of the SPE Method
3.4. SPE-HPLC Tests with Real (River) Waters
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Dwivedi, A. Researches in Water Pollution: A Review. Int. Res. J. Nat. Appl. Sci. 2017, 4, 118–142. [Google Scholar] [CrossRef]
- Chaudhry, F.; Malik, M. Factors Affecting Water Pollution: A Review. J. Ecosyst. Ecography 2017, 7, 225–231. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, X.; Jiang, J.; Han, J.; Li, W.; Li, X.; Yee Leung, K.M.; Snyder, S.A.; Alvarez, P.J.J. Which Micropollutants in Water Environments Deserve More Attention Globally? Environ. Sci. Technol. 2022, 56, 13–29. [Google Scholar] [CrossRef]
- Speltini, A.; Pastore, M.; Merlo, F.; Maraschi, F.; Sturini, M.; Dondi, D.; Profumo, A. Humic Acids Pyrolyzed onto Silica Microparticles for Solid-Phase Extraction of Benzotriazoles and Benzothiazoles from Environmental Waters. Chromatographia 2019, 82, 1275–1283. [Google Scholar] [CrossRef]
- Kim, M.-K.; Zoh, K.-D. Occurrence and Removals of Micropollutants in Water Environment. Environ. Eng. Res. 2016, 21, 319–332. [Google Scholar] [CrossRef]
- Rogowska, J.; Cieszynska-Semenowicz, M.; Ratajczyk, W.; Wolska, L. Micropollutants in Treated Wastewater. Ambio 2020, 49, 487–503. [Google Scholar] [CrossRef] [PubMed]
- Egambaram, O.P.; Kesavan Pillai, S.; Ray, S.S. Materials Science Challenges in Skin UV Protection: A Review. Photochem. Photobiol. 2020, 96, 779–797. [Google Scholar] [CrossRef] [PubMed]
- Kiejza, D.; Karpińska, J.; Kotowska, U. Degradation of Benzotriazole UV Stabilizers in PAA/d-Electron Metal Ions Systems—Removal Kinetics, Products and Mechanism Evaluation. Molecules 2022, 27, 3349. [Google Scholar] [CrossRef] [PubMed]
- Alotaibi, M.D.; McKinley, A.J.; Patterson, B.M.; Reeder, A.Y. Benzotriazoles in the Aquatic Environment: A Review of Their Occurrence, Toxicity, Degradation and Analysis. Water Air Soil Pollut. 2015, 226, 226. [Google Scholar] [CrossRef]
- Kraševec, I.; Prosen, H. Solid-Phase Extraction of Polar Benzotriazoles as Environmental Pollutants: A Review. Molecules 2018, 23, 2501. [Google Scholar] [CrossRef]
- Nouri, N.; Khorram, P.; Duman, O.; Sibel, T.; Hassan, S. Overview of Nanosorbents Used in Solid Phase Extraction Techniques for the Monitoring of Emerging Organic Contaminants in Water and Wastewater Samples. Trends Environ. Anal. Chem. 2020, 25, e00081. [Google Scholar] [CrossRef]
- Poole, C.F. Core Concepts and Milestones in the Development of Solid-Phase Extraction. In Solid-Phase Extraction; Elsevier: Amsterdam, The Netherlands, 2020; pp. 1–36. ISBN 978-0-12-816906-3. [Google Scholar]
- Leusch, F.; Prochazka, E.; Carswell, S.; Escher, B. Optimising Micropollutants Extraction for Analysis of Water Samples: Comparison of Different Solid Phase Materials and Liquid-Liquid Extraction. In Proceedings of the Science Forum and Stakeholder Engagement: Building Linkages, Collaboration and Science Quality, Brisbane, Australia, 28–29 September 2010; pp. 191–195. [Google Scholar]
- Guillot, S.; Kelly, M.T.; Fenet, H.; Larroque, M. Evaluation of Solid-Phase Microextraction as an Alternative to the Official Method for the Analysis of Organic Micro-Pollutants in Drinking Water. J. Chromatogr. A 2006, 1101, 46–52. [Google Scholar] [CrossRef]
- Fernández-Fernández, V.; Ramil, M.; Cela, R.; Rodríguez, I. Solid-Phase Extraction and Fractionation of Multiclass Pollutants from Wastewater Followed by Liquid Chromatography Tandem-Mass Spectrometry Analysis. Anal. Bioanal. Chem. 2022, 414, 4149–4165. [Google Scholar] [CrossRef] [PubMed]
- Čizmić, M.; Babić, S.; Kaštelan-Macan, M. Multi-Class Determination of Pharmaceuticals in Wastewaters by Solid-Phase Extraction and Liquid Chromatography Tandem Mass Spectrometry with Matrix Effect Study. Environ. Sci. Pollut. Res. 2017, 24, 20521–20539. [Google Scholar] [CrossRef] [PubMed]
- Masini, J.C.; do Nascimento, F.H.; Vitek, R. Porous Monolithic Materials for Extraction and Preconcentration of Pollutants from Environmental Waters. Trends Environ. Anal. Chem. 2021, 29, e00112. [Google Scholar] [CrossRef]
- Zheng, X.; Zhao, Y.; Wen, W.; Zheng, H.; Gao, L. Application of Graphene and Its Compounds in Pretreatment of Environmental Samples. IOP Conf. Ser. Earth Environ. Sci. 2021, 687, 012064. [Google Scholar] [CrossRef]
- Shi, Z.; Li, Q.; Xu, D.; Huai, Q.; Zhang, H. Graphene-Based Pipette Tip Solid-Phase Extraction with Ultra-High Performance Liquid Chromatography and Tandem Mass Spectrometry for the Analysis of Carbamate Pesticide Residues in Fruit Juice: Sample Preparation. J. Sep. Sci. 2016, 39, 4391–4397. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.; Liang, Q.; Zhang, X.; Yang, L.; Ding, M. Graphene Aerogel Based Monolith for Effective Solid-Phase Extraction of Trace Environmental Pollutants from Water Samples. J. Chromatogr. A 2016, 1447, 39–46. [Google Scholar] [CrossRef]
- Wang, X.; Wang, J.; Du, T.; Kou, H.; Du, X.; Lu, X. Determination of Six Benzotriazole Ultraviolet Filters in Water and Cosmetic Samples by Graphene Sponge-Based Solid-Phase Extraction Followed by High-Performance Liquid Chromatography. Anal. Bioanal. Chem. 2018, 410, 6955–6962. [Google Scholar] [CrossRef] [PubMed]
- Pena-Pereira, F.; Romero, V.; de la Calle, I.; Lavilla, I.; Bendicho, C. Graphene-Based Nanocomposites in Analytical Extraction Processes. TrAC Trends Anal. Chem. 2021, 142, 116303. [Google Scholar] [CrossRef]
- Zhang, Q.; You, L.; Chen, B.; He, M.; Hu, B. Reduced Graphene Oxide Coated Nickel Foam for Stir Bar Sorptive Extraction of Benzotriazole Ultraviolet Absorbents from Environmental Water. Talanta 2021, 231, 122332. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Jiang, H.-L.; Wang, X.; Wang, X.; Xu, G.; Zhang, B.; Wang, L.; Zhao, R.-S.; Lin, J.-M. Recent Advances in Graphene-Based Magnetic Composites for Magnetic Solid-Phase Extraction. TrAC Trends Anal. Chem. 2018, 102, 60–74. [Google Scholar] [CrossRef]
- dos Santos-Gómez, L.; García, J.R.; Montes-Morán, M.A.; Menéndez, J.A.; García-Granda, S.; Arenillas, A. Ultralight-Weight Graphene Aerogels with Extremely High Electrical Conductivity. Small 2021, 17, 2103407. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; An, J.; Potts, J.R.; Velamakanni, A.; Murali, S.; Ruoff, R.S. Hydrazine-Reduction of Graphite- and Graphene Oxide. Carbon 2011, 49, 3019–3023. [Google Scholar] [CrossRef]
- Huh, S.H. Thermal Reduction of Graphene Oxide. In Physics and Applications of Graphene-Experiments; IntechOpen: London, UK, 2011; Volume 19, pp. 73–90. [Google Scholar]
- Chatzimitakos, T.G.; Stalikas, C.D. Sponges and Sponge-Like Materials in Sample Preparation: A Journey from Past to Present and into the Future. Molecules 2020, 25, 3673. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of Gases, with Special Reference to the Evaluation of Surface Area and Pore Size Distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Moreno, A.H.; Arenillas, A.; Calvo, E.G.; Bermúdez, J.M.; Menéndez, J.A. Carbonisation of Resorcinol–Formaldehyde Organic Xerogels: Effect of Temperature, Particle Size and Heating Rate on the Porosity of Carbon Xerogels. J. Anal. Appl. Pyrolysis 2013, 100, 111–116. [Google Scholar] [CrossRef]
- Rey-Raap, N.; Angel Menéndez, J.; Arenillas, A. Simultaneous Adjustment of the Main Chemical Variables to Fine-Tune the Porosity of Carbon Xerogels. Carbon 2014, 78, 490–499. [Google Scholar] [CrossRef]
- Canal-Rodríguez, M.; Ramírez-Montoya, L.A.; Villanueva, S.F.; Flores-López, S.L.; Angel Menéndez, J.; Arenillas, A.; Montes-Morán, M.A. Multiphase Graphitisation of Carbon Xerogels and Its Dependence on Their Pore Size. Carbon 2019, 152, 704–714. [Google Scholar] [CrossRef]
- Liu, R.; Ruan, T.; Wang, T.; Song, S.; Guo, F.; Jiang, G. Determination of Nine Benzotriazole UV Stabilizers in Environmental Water Samples by Automated On-Line Solid Phase Extraction Coupled with High-Performance Liquid Chromatography–Tandem Mass Spectrometry. Talanta 2014, 120, 158–166. [Google Scholar] [CrossRef]
- Yu, H.; Di, S.; Su, X.; Wang, J.; Ning, T.; Yang, H.; Zhu, S. Preparation of Beta-Cyclodextrin Based Nanocomposite for Magnetic Solid-Phase Extraction of Organic Ultraviolet Filters. J. Chromatogr. A 2022, 1663, 462765. [Google Scholar] [CrossRef] [PubMed]
- Yang, F.; Li, X.; Meng, D.; Yang, Y. Determination of Ultraviolet Absorbers and Light Stabilizers in Food Packaging Bags by Magnetic Solid Phase Extraction Followed by High-Performance Liquid Chromatography. Food Anal. Methods 2017, 10, 3247–3254. [Google Scholar] [CrossRef]
- Montesdeoca-Esponda, S.; Sosa-Ferrera, Z.; Kabir, A.; Furton, K.G.; Santana-Rodríguez, J.J. Fabric Phase Sorptive Extraction Followed by UHPLC-MS/MS for the Analysis of Benzotriazole UV Stabilizers in Sewage Samples. Anal. Bioanal. Chem. 2015, 407, 8137–8150. [Google Scholar] [CrossRef]
- Zilfidou, E.; Kabir, A.; Furton, K.; Samanidou, V. Fabric Phase Sorptive Extraction: Current State of the Art and Future Perspectives. Separations 2018, 5, 40. [Google Scholar] [CrossRef]
- Canal-Rodríguez, M.; Arenillas, A.; Villanueva, S.F.; Montes-Morán, M.A.; Menénedez, J.A. Graphitized carbon xerogels for lithium-ion batteries. Materials 2020, 13, 119. [Google Scholar] [CrossRef] [PubMed]




| Material | SBET (m2 g−1) | Vtotal (mL g−1) | Vmicro a (mL g−1) |
|---|---|---|---|
| GS | 203 | 0.89 | 0.08 |
| GA-40% | 247 | 0.41 | 0.09 |
| GA-60% | 164 | 0.49 | 0.06 |
| G-GA-40% | 51 | 0.26 | 0.01 |
| Material | Concentration (ng mL−1) | EFtheo | UV329 | UV234 | ||
|---|---|---|---|---|---|---|
| Recovery (%) | SD a (%) | Recovery (%) | SD a (%) | |||
| GS | 1 | 50 | 83.6 | 10.4 | 76.7 | 5.0 |
| 100 | 12.5 | 91.0 | 7.8 | 79.7 | 6.8 | |
| GA-60% | 1 | 50 | 70.4 | 8.7 | 84.9 | 2.7 |
| 5 | 12.5 | 56.6 | 5.4 | 88.1 | 4.4 | |
| 100 | 12.5 | 61.7 | 9.1 | 87.6 | 5.8 | |
| G-GA-40% | 1 | 50 | 78.2 | 9.9 | 82.6 | 7.2 |
| 5 | 12.5 | 93.2 | 5.8 | 96.1 | 9.2 | |
| 50 | 12.5 | 80.1 | 8.1 | 83.9 | 3.2 | |
| 100 | 12.5 | 97.1 | 4.5 | 88.6 | 4.2 | |
| Material | UVP | UV329 | UV234 | |||
|---|---|---|---|---|---|---|
| Recovery (%) | SD a (%) | Recovery (%) | SD a (%) | Recovery (%) | SD a (%) | |
| GA-60% | N.D. b | - | 9.7 | 3.2 | 5.2 | 3.9 |
| G-GA-40% | N.D. b | - | N.D. b | - | 4.7 | 2.1 |
| Sample | Analyte | Range (ng mL−1) | Equation | R2 | LOD a (ng mL−1) | LOQ b (ng mL−1) |
|---|---|---|---|---|---|---|
| GA-60% | UV329 | 3–250 | y = 0.215x + 2.329 | 0.9960 | 1.43 | 4.76 |
| UV234 | 3–250 | y = 0.317x + 0.072 | 0.9948 | 1.16 | 3.88 | |
| G-GA-40% | UV329 | 3–250 | y = 0.391x + 0.118 | 0.9981 | 0.95 | 3.17 |
| UV234 | 3–250 | y = 0.340x + 0.338 | 0.9998 | 0.95 | 3.18 |
| Method | Extractant | Performance Parameters | Ref. | |||
|---|---|---|---|---|---|---|
| Material | Time of Synthesis (Days) | Extraction Time (min) | Recovery (%) | EFtheo | ||
| SPE-HPLC-UV | GS | 4.1 | 15 | 89–105 | 6.25 | [21] |
| SBSE-HPLC-DAD | GS/Ni coated | >7 | 60 | 84–112 | 67 | [23] |
| SPE-HPLC-UV | GAs | 3 | 10 | 75–97 | 12.5–50 | This work |
| Material | Spiked (ng mL−1) | UV329 | UV234 | ||||
|---|---|---|---|---|---|---|---|
| Found (ng mL−1) | Recovery (SD a) (%) | ME b (SD a) (%) | Found (ng mL−1) | Recovery (SD a) (%) | ME b (SD a) (%) | ||
| GA-60% | 0 | N.D. c | - | - | N.D. c | - | - |
| 25 | 20.05 | 80.0 (9.5) | 98.8 (7.1) | 21.89 | 87.6 (3.02) | 89.8 (1.95) | |
| 50 | 34.74 | 69.5 (8.4) | 97.8 (6.4) | 35.95 | 72.9 (2.26) | 80.8 (1.21) | |
| 100 | 68.55 | 68.6 (5.9) | 88.0 (5.2) | 84.61 | 84.6 (8.2) | 91.9 (8.8) | |
| G-GA-40% | 0 | N.D. c | - | - | N.D. c | - | - |
| 25 | 20.88 | 83.5 (1.5) | 81.4 (1.5) | 21.47 | 85.9 (2.8) | 93.7 (2.6) | |
| 50 | 46.93 | 93.9 (4.9) | 87.4 (4.1) | 51.36 | 102.7 (3.3) | 97.6 (3.2) | |
| 100 | 88.31 | 88.3 (4.9) | 90.6 (4.9) | 81.45 | 81. 5 (9.7) | 83.5 (7.5) | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Flores-López, S.L.; Arenillas, A.; Mikšík, I.; Menéndez, J.A.; Montes-Morán, M.A. Reduced Graphene Oxide Aerogels Cartridges for Solid Phase Extraction of Benzotriazoles. Materials 2023, 16, 2519. https://doi.org/10.3390/ma16062519
Flores-López SL, Arenillas A, Mikšík I, Menéndez JA, Montes-Morán MA. Reduced Graphene Oxide Aerogels Cartridges for Solid Phase Extraction of Benzotriazoles. Materials. 2023; 16(6):2519. https://doi.org/10.3390/ma16062519
Chicago/Turabian StyleFlores-López, Samantha L., Ana Arenillas, Ivan Mikšík, J. Angel Menéndez, and Miguel A. Montes-Morán. 2023. "Reduced Graphene Oxide Aerogels Cartridges for Solid Phase Extraction of Benzotriazoles" Materials 16, no. 6: 2519. https://doi.org/10.3390/ma16062519
APA StyleFlores-López, S. L., Arenillas, A., Mikšík, I., Menéndez, J. A., & Montes-Morán, M. A. (2023). Reduced Graphene Oxide Aerogels Cartridges for Solid Phase Extraction of Benzotriazoles. Materials, 16(6), 2519. https://doi.org/10.3390/ma16062519








