Macrocyclic Compounds: Metal Oxide Particles Nanocomposite Thin Films Deposited by MAPLE
Abstract
1. Introduction
2. Experimental Section
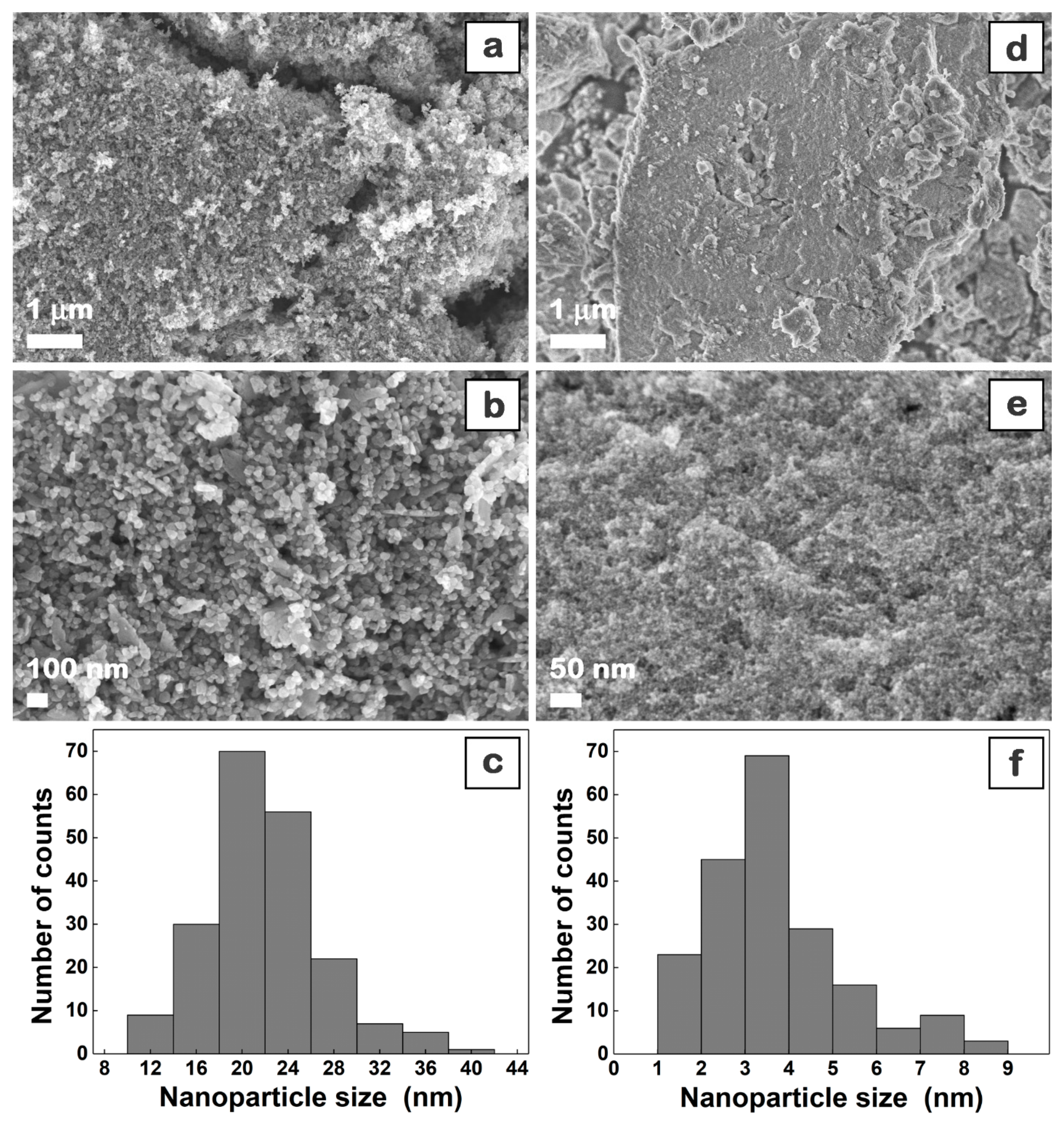
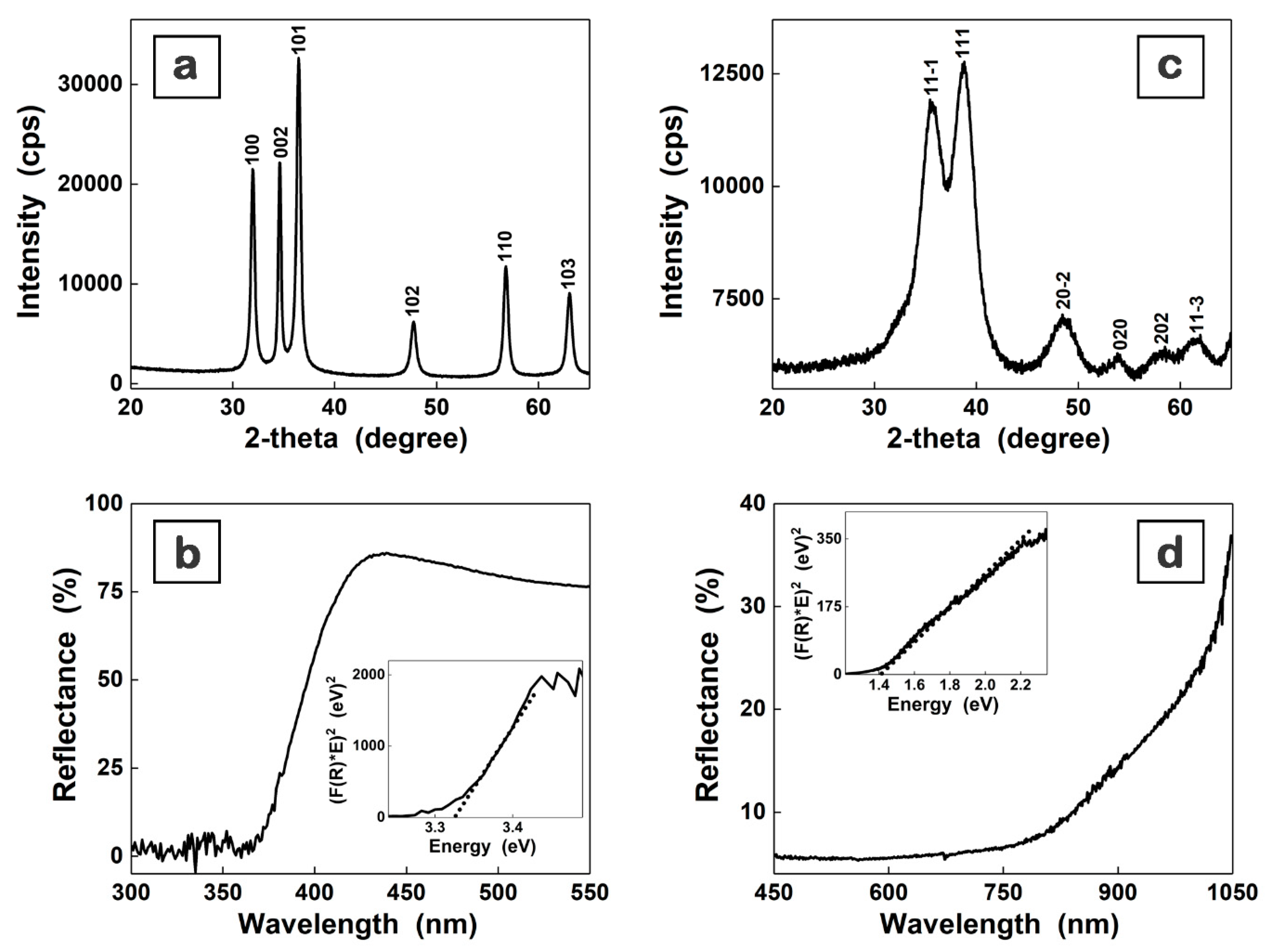
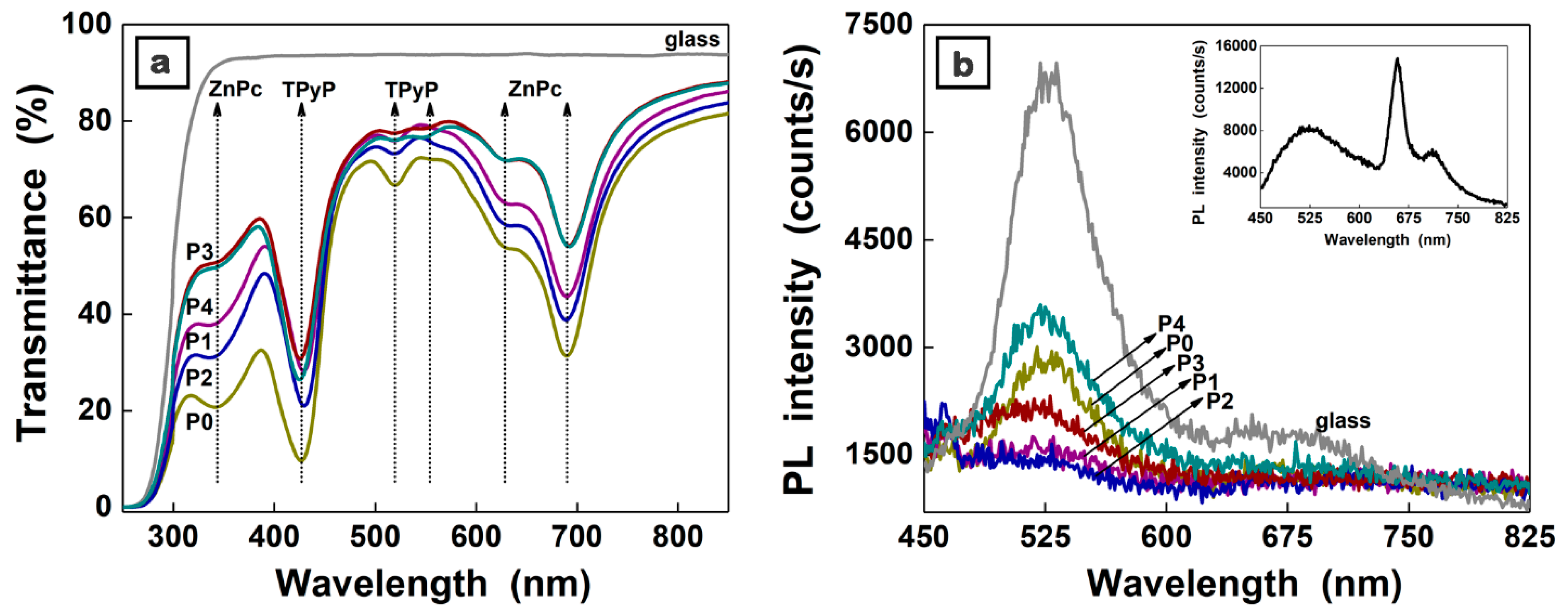
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Park, S.B.; Han, J.W.; Kim, J.H.; Wibowo, A.F.; Prameswati, A.; Park, J.; Lee, J.; Moon, M.-W.; Kim, M.-S.; Kim, Y.H. Multifunctional Stretchable Organic–Inorganic Hybrid Electronics with Transparent Conductive Silver Nanowire/Biopolymer Hybrid Films. Adv. Opt. Mater. 2021, 9, 2002041. [Google Scholar] [CrossRef]
- Saveleva, M.S.; Eftekhari, K.; Abalymov, A.; Douglas, T.E.L.; Volodkin, D.; Parakhonskiy, B.V.; Skirtach, A.G. Hierarchy of Hybrid Materials—The Place of Inorganics-in-Organics in it, Their Composition and Applications. Front. Chem. 2019, 7, 179. [Google Scholar] [CrossRef] [PubMed]
- Atta-Eyison, A.A.; Anukwah, G.D.; Zugle, R. Photocatalysis using zinc oxide-zinc phthalocyanine composite for effective mineralization of organic pollutants. Catal. Commun. 2021, 160, 106357. [Google Scholar] [CrossRef]
- Raucci, M.G.; Demitri, C.; Soriente, A.; Fasolino, I.; Sannino, A.; Ambrosio, L. Gelatin/nano-hydroxyapatite hydrogel scaffold prepared by sol-gel technology as filler to repair bone defects. J. Biomed. Mater. Res. Part A 2018, 106, 2007–2019. [Google Scholar] [CrossRef]
- Vuoriluoto, M.; Hokkanen, A.; Mäkelä, T.; Harlin, A.; Orelma, H. Optical properties of an organic-inorganic hybrid film made of regenerated cellulose doped with light-scattering TiO2 particles. Opt. Mater. 2022, 123, 111882. [Google Scholar] [CrossRef]
- Ikake, H.; Hara, S.; Shimizu, S. Skillful Control of Dispersion and 3D Network Structures: Advances in Functional Organic–Inorganic Nano-Hybrid Materials Prepared Using the Sol-Gel Method. Polymers 2022, 14, 3247. [Google Scholar] [CrossRef]
- Abbas, K.F.; Abdulameer, A.F. Blending Ratio Effect of ZnPc/ZnO Hybrid Nanocomposite on Surface Morphology and Structural Properties. J. Phys. Conf. Ser. 2021, 2114, 012015. [Google Scholar] [CrossRef]
- Lee, S.H.; Kang, B.S.; Kwak, M.K. Magneto-Responsive Actuating Surfaces with Controlled Wettability and Optical Transmittance. ACS Appl. Mater. Interfaces 2022, 14, 14721–14728. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Díaz, M.V.; de la Torre, G.; Torres, T. Lighting porphyrins and phthalocyanines for molecular photovoltaics. Chem. Commun. 2010, 46, 7090–7108. [Google Scholar] [CrossRef]
- Schmidt, A.M.; Calvete, M.J.F. Phthalocyanines: An Old Dog Can Still Have New (Photo)Tricks! Molecules 2021, 26, 2823. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Breazu, C.; Stanculescu, A.; Costas, A.; Stanculescu, F.; Girtan, M.; Gherendi, F.; Popescu Pelin, G.; Socol, G. Flexible organic heterostructures obtained by MAPLE. Appl. Phys. A 2018, 124, 602. [Google Scholar] [CrossRef]
- Wang, J.; Dong, W.; Si, Z.; Cui, X.; Duan, Q. Synthesis and enhanced nonlinear optical performance of phthalocyanine indium polymers with electron-donating group porphyrin by efficient energy transfer. Dyes Pigments 2022, 198, 109985. [Google Scholar] [CrossRef]
- Walter, M.G.; Rudine, A.B.; Wamser, C.C. Porphyrins and phthalocyanines in solar photovoltaic cells. J. Porphyr. Phthalocyanines 2010, 14, 759–792. [Google Scholar] [CrossRef]
- Hohnholz, D.; Steinbrecher, S.; Hanack, M. Applications of phthalocyanines in organic light emitting devices. Mol. Struct. 2000, 521, 231–237. [Google Scholar] [CrossRef]
- Josefsen, L.B.; Boyle, R.W. Unique diagnostic and therapeutic roles of porphyrins and phthalocyanines in photodynamic therapy, imaging and theranostics. Theranostics 2012, 2, 916–966. [Google Scholar] [CrossRef]
- Tian, J.; Huang, B.; Nawaz, M.H.; Zhang, W. Recent Advances of Multi-Dimensional Porphyrin-Based Functional Materials in Photodynamic Therapy. Coord. Chem. Rev. 2020, 420, 213410. [Google Scholar] [CrossRef]
- Neelgund, G.; Oki, A. Cobalt phthalocyanine-sensitized graphene−ZnO composite: An efficient near-infrared-active photothermal agent. ACS Omega 2019, 4, 5696–5704. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N. Hybrid Nanocomposite Thin Films for Photovoltaic Applications: A Review. Nanomaterials 2021, 11, 1117. [Google Scholar] [CrossRef]
- Kołodziejczak-Radzimska, A.; Jesionowski, T. Zinc Oxide—From Synthesis to Application: A Review. Materials 2014, 7, 2833–2881. [Google Scholar] [CrossRef]
- Vivek, P.; Rekha, M.; Suvitha, A.; Kowsalya, M.; Stephen, A. Diamond morphology CuO nanomaterial’s elastic properties, ADMET, optical, structural studies, electrical conductivity and antibacterial activities analysis. Inorg. Nano-Met. Chem. 2022, 52, 1243–1255. [Google Scholar] [CrossRef]
- Preda, N.; Enculescu, M.; Enculescu, I. Polysaccharide-assisted crystallization of ZnO micro/nanostructures. Mater. Lett. 2014, 115, 256–260. [Google Scholar] [CrossRef]
- Preda, N.; Costas, A.; Enculescu, M.; Enculescu, I. Biomorphic 3D fibrous networks based on ZnO, CuO and ZnO–CuO composite nanostructures prepared from eggshell membranes. Mater. Chem. Phys. 2020, 240, 122205. [Google Scholar] [CrossRef]
- Kim, I.; Viswanathan, K.; Kasi, G.; Thanakkasaranee, S.; Sadeghi, K.; Seo, J. ZnO nanostructures in active antibacterial food packaging: Preparation methods, antimicrobial mechanisms, safety issues, future prospects, and challenges. Food Rev. Int. 2020, 38, 537–565. [Google Scholar] [CrossRef]
- Spoială, A.; Ilie, C.-I.; Trușcă, R.-D.; Oprea, O.-C.; Surdu, V.-A.; Vasile, B.Ș.; Ficai, A.; Ficai, D.; Andronescu, E.; Dițu, L.-M. Zinc Oxide Nanoparticles for Water Purification. Materials 2021, 14, 4747. [Google Scholar] [CrossRef]
- Luo, X.; Xu, L.; Xu, B.; Li, F. Electrodeposition of zinc oxide/tetrasulfonated copper phthalocyanine hybrid thin film for dye-sensitized solar cell application. Appl. Surf. Sci. 2011, 257, 6908–6911. [Google Scholar] [CrossRef]
- Mattioli, G.; Filippone, F.; Alippi, P.; Giannozzi, P.; Bonapasta, A.A. A hybrid zinc phthalocyanine/zinc oxide system for photovoltaic devices: A DFT and TDDFPT theoretical investigation. J. Mater. Chem. 2012, 22, 440–446. [Google Scholar] [CrossRef]
- Seddigi, Z.S.; Ahmed, S.A.; Sardar, S.; Pal, S.K. Ultrafast dynamics at the zinc phthalocyanine/zinc oxide nanohybrid interface for efficient solar light harvesting in the near red region. Sol. Energy Mater. Sol. Cells 2015, 143, 63–71. [Google Scholar] [CrossRef]
- Novotný, M.; Marešová, E.; Fitl, P.; Vlček, J.; Bergmann, M.; Vondráček, M.; Yatskiv, R.; Bulır, J.; Hubık, P.; Hruska, P.; et al. The properties of samarium-doped zinc oxide/phthalocyanine structure for optoelectronics prepared by pulsed laser deposition and organic molecular evaporation. Appl. Phys. A 2015, 122, 225. [Google Scholar] [CrossRef]
- Ekrami, M.; Magna, G.; Emam-djomeh, Z.; Saeed Yarmand, M.; Paolesse, R.; Di Natale, C. Porphyrin-Functionalized Zinc Oxide Nanostructures for Sensor Applications. Sensors 2018, 18, 2279. [Google Scholar] [CrossRef]
- Choi, S.; Lee, J.; Lee, K.; Yoon, S.M.; Yoon, M. Porphyrin-decorated ZnO nanowires as nanoscopic injectors for phototheragnosis of cancer cells. New J. Chem. 2022, 46, 13465–13474. [Google Scholar] [CrossRef]
- Yu, Q.; Xu, J. Structure and surface properties of fluorinated organic–inorganic hybrid films. J. Sol-Gel Sci. Technol. 2012, 61, 243–248. [Google Scholar] [CrossRef]
- Zheng, Q.; Zhang, B.; Lin, X.; Shen, X.; Yousefi, N.; Huang, Z.-D.; Li, Z.; Kim, J.-K. Highly transparent and conducting ultralarge graphene oxide/single-walled carbon nanotube hybrid films produced by Langmuir–Blodgett assembly. J. Mater. Chem. 2012, 22, 25072. [Google Scholar] [CrossRef]
- Sharma, G.D.; Kumar, R.; Sharma, S.K.; Roy, M.S. Charge generation and photovoltaic properties of hybrid solar cells based on ZnO and copper phthalocyanines (CuPc). Sol. Energy Mater. Sol. Cells 2006, 90, 933–943. [Google Scholar] [CrossRef]
- Li, M.; Li, J.; Yu, L.; Zhang, Y.; Dai, Y.; Chen, R.; Huang, W. Trap-filling of ZnO buffer layer for improved efficiencies of organic solar cells. Front. Chem. 2020, 8, 399. [Google Scholar] [CrossRef] [PubMed]
- Mishra, A.; Bhatt, N.; Bajpai, A. Nanostructured superhydrophobic coatings for solar panel applications. In Nanomaterials-Based Coatings; Elsevier: Amsterdam, The Netherlands, 2019; pp. 397–424. [Google Scholar] [CrossRef]
- Yilbas, B.S.; Al-Sharafi, A.; Ali, H. Surfaces for Self-Cleaning. In Self-Cleaning of Surfaces and Water Droplet Mobility; Yilbas, B.S., Al-Sharafi, A., Ali, H., Eds.; Elsevier: Amsterdam, The Netherlands, 2019; pp. 45–98. [Google Scholar] [CrossRef]
- Na, J.; Kang, B.; Sin, D.; Cho, K.; Park, Y.D. Understanding Solidification of Polythiophene Thin Films during Spin-Coating: Effects of Spin-Coating Time and Processing Additives. Sci. Rep. 2015, 5, 13288. [Google Scholar] [CrossRef]
- Kamanyi, A.E., Jr.; Ngwa, W.; Luo, W.; Grill, W. Effects of solvent vapor pressure and spin-coating speed on morphology of thin polymer blend films. Proc. SPIE 2008, 6935, 69351X. [Google Scholar] [CrossRef]
- Strawhecker, K.E.; Kumar, S.K.; Douglas, J.F.; Karim, A. The Critical Role of Solvent Evaporation on the Roughness of Spin-Cast Polymer Films. Macromolecules 2001, 34, 4669–4672. [Google Scholar] [CrossRef]
- Visan, A.I.; Popescu-Pelin, G.; Gherasim, O.; Grumezescu, V.; Socol, M.; Zgura, I.; Florica, C.; Popescu, R.C.; Savu, D.; Holban, A.M.; et al. Laser Processed Antimicrobial Nanocomposite Based on Polyaniline Grafted Lignin Loaded with Gentamicin-Functionalized Magnetite. Polymers 2019, 11, 283. [Google Scholar] [CrossRef]
- Dong, B.X.; Strzalka, J.; Jiang, Z.; Li, H.; Stein, G.E.; Green, P.F. Crystallization Mechanism and Charge Carrier Transport in MAPLE-Deposited Conjugated Polymer Thin Films. ACS Appl. Mater. Interfaces 2017, 9, 44799–44810. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Socol, G. Organic Thin Films Deposited by Matrix-Assisted Pulsed Laser Evaporation (MAPLE) for Photovoltaic Cell Applications: A Review. Coatings 2021, 11, 1368. [Google Scholar] [CrossRef]
- Caricato, A.P.; Ge, W.; Stiff-Roberts, A.D. UV- and RIR-MAPLE: Fundamentals and Applications, 1st ed.; Springer Series in Materials Science; Springer Nature Switzerland: Cham, Switzerland, 2018; pp. 275–308. [Google Scholar] [CrossRef]
- Ge, W.; Li, N.K.; McCormick, R.D.; Lichtenberg, E.; Yingling, Y.G.; Stiff-Roberts, A.D. Emulsion-Based RIR-MAPLE Deposition of Conjugated Polymers: Primary Solvent Effect and Its Implications on Organic Solar Cell Performance. ACS Appl. Mater. Interfaces 2016, 8, 19494–19506. [Google Scholar] [CrossRef] [PubMed]
- Canulescu, S.; Schou, J.; Fæster, S.; Hansen, K.V.; Conseil, H. Deposition of matrix-free fullerene films with improved morphology by matrix-assisted pulsed laser evaporation (MAPLE). Chem. Phys. Lett. 2013, 588, 119–123. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Costas, A.; Breazu, C.; Stanculescu, A.; Rasoga, O.; Popescu-Pelin, G.; Mihailescu, A.; Socol, G. Hybrid organic-inorganic thin films based on zinc phthalocyanine and zinc oxide deposited by MAPLE. Appl. Surf. Sci. 2020, 503, 144317. [Google Scholar] [CrossRef]
- Ge, W.; Atewologun, A.; Stiff-Roberts, A.D. Hybrid nanocomposite thin films deposited by emulsion-based resonant infrared matrix assisted pulsed laser evaporation for photovoltaic applications. Org. Electron. 2015, 22, 98–107. [Google Scholar] [CrossRef]
- Stanculescu, F.; Rasoga, O.; Catargiu, A.M.; Vacareanu, L.; Socol, M.; Breazu, C.; Preda, N.; Socol, G.; Stanculescu, A. MAPLE prepared heterostructures with arylene based polymer active layer for photovoltaic applications. Appl. Surf. Sci. 2015, 336, 240–248. [Google Scholar] [CrossRef]
- Toftmann, B.; Papantonakis, M.R.; Auyeung, R.C.Y.; Kim, W.; O’Malley, S.M.; Bubb, D.M.; Horwitz, J.S.; Schou, J.; Johansen, R.F.; Haglund, R.F. UV and RIR matrix assisted pulsed laser deposition of organic MEH-PPV films. Thin Solid Films 2004, 453–454, 177–181. [Google Scholar] [CrossRef]
- Gutierrez-Llorente, A. Growth of polyalkylthiophene films by matrix assisted pulsed laser evaporation. Org. Electron. 2004, 5, 29–34. [Google Scholar] [CrossRef]
- Califano, V.; Bloisi, F.; Vicari, L.; Barra, M.; Cassinese, A.; Fanelli, E.; Buzio, R.; Valbusa, U.; Carella, A.; Roviello, A. Substrate temperature dependence of the structure of polythiophene thin films obtained by matrix assisted pulsed laser evaporation (MAPLE). Eur. Phys. J. Appl. Phys. 2009, 48, 10505. [Google Scholar] [CrossRef]
- Stanculescu, A.; Rasoga, O.; Socol, M.; Vacareanu, L.; Grigoras, M.; Socol, G.; Stanculescu, F.; Breazu, C.; Matei, E.; Preda, N.; et al. MAPLE prepared heterostructures with oligoazomethine: Fullerene derivative mixed layer for photovoltaic applications. Appl. Surf. Sci. 2017, 417, 183–195. [Google Scholar] [CrossRef]
- Ghani, F.; Kristen, J.; Riegler, H. Solubility Properties of Unsubstituted Metal Phthalocyanines in Different Types of Solvents. J. Chem. Eng. Data 2012, 57, 439–449. [Google Scholar] [CrossRef]
- Socol, M.; Rasoga, O.; Stanculescu, F.; Girtan, M.; Stanculescu, A. Effect of the morphology on the optical and electrical properties of TPyP thin films deposited by vacuum evaporation. Optoelectron. Adv. Mater. Rapid Commun. 2010, 4, 2032–2038. [Google Scholar]
- Yan, Q.; Modigell, M. Mechanical pretreatment of lignocellulosic biomass using a screw press as an essential step in the biofuel production. Chem. Eng. Trans. 2012, 29, 601–606. [Google Scholar] [CrossRef]
- He, Y.; Wildman, R.D.; Tuck, C.J.; Christie, S.D.R.; Edmondson, S. An Investigation of the Behavior of Solvent based Polycaprolactone ink for Material Jetting. Sci. Rep. 2016, 6, 20852. [Google Scholar] [CrossRef]
- Greul, E.; Docampo, P.; Bein, T. Synthesis of Hybrid Tin Halide Perovskite Solar Cells with Less Hazardous Solvents: Methanol and 1,4-Dioxane. Z. Anorg. Allg. Chem. 2017, 643, 1704–1711. [Google Scholar] [CrossRef]
- Bian, S.-W.; Mudunkotuwa, I.A.; Rupasinghe, T.; Grassian, V.H. Aggregation and Dissolution of 4 nm ZnO Nanoparticles in Aqueous Environments: Influence of pH, Ionic Strength, Size, and Adsorption of Humic Acid. Langmuir 2011, 27, 6059–6068. [Google Scholar] [CrossRef] [PubMed]
- Rahnama, A.; Gharagozlou, M. Preparation and properties of semiconductor CuO nanoparticles via a simple precipitation method at different reaction temperatures. Opt. Quantum Electron. 2012, 44, 313–322. [Google Scholar] [CrossRef]
- Ahmad, M.; Zhu, J. ZnO based advanced functional nanostructures: Synthesis, properties and applications. J. Mater. Chem. 2011, 21, 599–614. [Google Scholar] [CrossRef]
- Zoolfakar, A.S.; Rani, R.A.; Morfa, A.J.; O’Mullane, A.P.; Kalantar-Zadeh, K. Nanostructured copper oxide semiconductors: A perspective on materials, synthesis methods and applications. J. Mater. Chem. C 2014, 2, 5247–5270. [Google Scholar] [CrossRef]
- Gaffo, L.; Cordeiro, M.R.; Freitas, A.R.; Moreira, W.C.; Girotto, E.M.; Zucolotto, V. The effects of temperature on the molecular orientation of zinc phthalocyanine films. J. Mater. Sci. 2010, 45, 1366–1370. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Rasoga, O.; Breazu, C.; Stavarache, I.; Stanculescu, F.; Socol, G.; Gherendi, F.; Grumezescu, V.; Stefan, N.; et al. Flexible heterostructures based on metal phthalocyanines thin films obtained by MAPLE. Appl. Surf. Sci. 2016, 374, 403–410. [Google Scholar] [CrossRef]
- Ikram, M.; Imran, M.; Nunzi, J.M.; Bobbara, S.R.; Ali, S.; Islah-u-din. Efficient and low cost inverted hybrid bulk heterojunction solar cells. J. Renew. Sustain. Energy 2015, 7, 043148. [Google Scholar] [CrossRef]
- Socol, M.; Preda, N.; Costas, A.; Borca, B.; Popescu-Pelin, G.; Mihailescu, A.; Socol, G.; Stanculescu, A. Thin films based on cobalt phthalocyanine:C60 fullerene:ZnO hybrid nanocomposite obtained by laser evaporation. Nanomaterials 2020, 10, 468. [Google Scholar] [CrossRef] [PubMed]
- Auwärter, W.; Weber-Bargioni, A.; Riemann, A.; Schiffrin, A.; Gröning, O.; Fasel, R.; Barth, J.V. Self-assembly and conformation of tetrapyridyl-porphyrin molecules on Ag(111). J. Chem. Phys. 2006, 124, 194708. [Google Scholar] [CrossRef]
- Said, A.J.; Poize, G.; Martini, C.; Ferry, D.; Marine, W.; Giorgio, S.; Fages, F.; Hocq, J.; Bouclé, J.; Nelson, J.; et al. Hybrid Bulk Heterojunction Solar Cells Based on P3HT and Porphyrin-Modified ZnO Nanorods. J. Phys. Chem. C 2010, 114, 11273–11278. [Google Scholar] [CrossRef]
- Salim, E.; Bobbara, S.R.; Oraby, A.; Nunzi, J.M. Copper oxide nanoparticle doped bulk-heterojunction photovoltaic devices. Synth. Met. 2019, 252, 21–28. [Google Scholar] [CrossRef]
- Schunemann, C.; Wynands, D.; Wilde, L.; Hein, M.P.; Pfutzner, S.; Elschner, C.; Eichhorn, K.-J.; Leo, K.; Riede, M. Phase separation analysis of bulk heterojunctions in small-molecule organic solar cells using zinc-phthalocyanine and C60. Phys. Rev. B 2012, 85, 245314. [Google Scholar] [CrossRef]
- Roy, D.; Das, N.M.; Shakti, N.; Gupta, P.S. Comparative study of optical, structural and electrical properties of zinc phthalocyanine Langmuir–Blodgett thin film on annealing. RSC Adv. 2014, 4, 42514–42522. [Google Scholar] [CrossRef]
- Ji, W.; Wang, T.-X.; Ding, X.; Lei, S.; Han, B.-H. Porphyrin- and phthalocyanine-based porous organic polymers: From synthesis to application. Coord. Chem. Rev. 2021, 439, 213875. [Google Scholar] [CrossRef]
- Sampaio, R.N.; Gomes, W.R.; Araujo, D.M.S.; Machado, A.E.H.; Silva, R.A.; Marletta, A.; Borissevitch, I.E.; Ito, A.S.; Dinelli, L.R.; Batista, A.A.; et al. Investigation of Ground- and Excited-State Photophysical Properties of 5,10,15,20-Tetra(4-pyridyl)-21H,23H-porphyrin with Ruthenium Outlying Complexes. J. Phys. Chem. A 2011, 116, 18–26. [Google Scholar] [CrossRef]
- Stanculescu, A.; Socol, M.; Rasoga, O.; Breazu, C.; Preda, N.; Stanculescu, F.; Socol, G.; Vacareanu, L.; Girtan, M.; Doroshkevich, A.S. Arylenevinylene Oligomer-Based Heterostructures on Flexible AZO Electrodes. Materials 2021, 14, 7688. [Google Scholar] [CrossRef]
- Ikram, M.; Imran, M.; Nunzi, J.M.; Islah-u-din; Ali, S. Replacement of P3HT and PCBM with metal oxides nanoparticles in inverted hybrid organic solar cells. Synth. Met. 2015, 210, 268–272. [Google Scholar] [CrossRef]
- Jiang, T.; Fu, W. Improved performance and stability of perovskite solar cells with bilayer electron-transporting layers. RSC Adv. 2018, 8, 5897–5901. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Gu, X.; Zhang, Q.; Jiang, J.; Jin, X.; Li, F.; Chen, Z.; Zhao, F.; Li, Q. ZnMgO:ZnO composite films for fast electron transport and high charge balance in quantum dot light emitting diodes. Opt. Mater. Express 2018, 8, 909–918. [Google Scholar] [CrossRef]
- Cuesta, V.; Singh, M.K.; Gutierrez-Fernandez, E.; Martín, J.; Domínguez, R.; de la Cruz, P.; Sharma, G.D.; Langa, F. Gold(III) Porphyrin Was Used as an Electron Acceptor for Efficient Organic Solar Cells. ACS Appl. Mater. Interfaces 2022, 14, 11708–11717. [Google Scholar] [CrossRef] [PubMed]
- Sadoogi, N.; Rostami, A.; Faridpak, B.; Farrokhifar, M. Performance analysis of organic solar cells: Opto-electrical modeling and simulation. Eng. Sci. Technol. 2021, 24, 229–235. [Google Scholar] [CrossRef]
- Elumalai, N.K.; Uddin, A. Open circuit voltage of organic solar cells: An in-depth review. Energy Environ. Sci. 2016, 9, 391–410. [Google Scholar] [CrossRef]
- Wanninayake, A.P.; Gunashekar, S.; Li, S.; Church, B.C.; Abu-Zahra, N. Performance enhancement of polymer solar cells using copper oxide nanoparticles. Semicond. Sci. Technol. 2015, 30, 064004. [Google Scholar] [CrossRef]



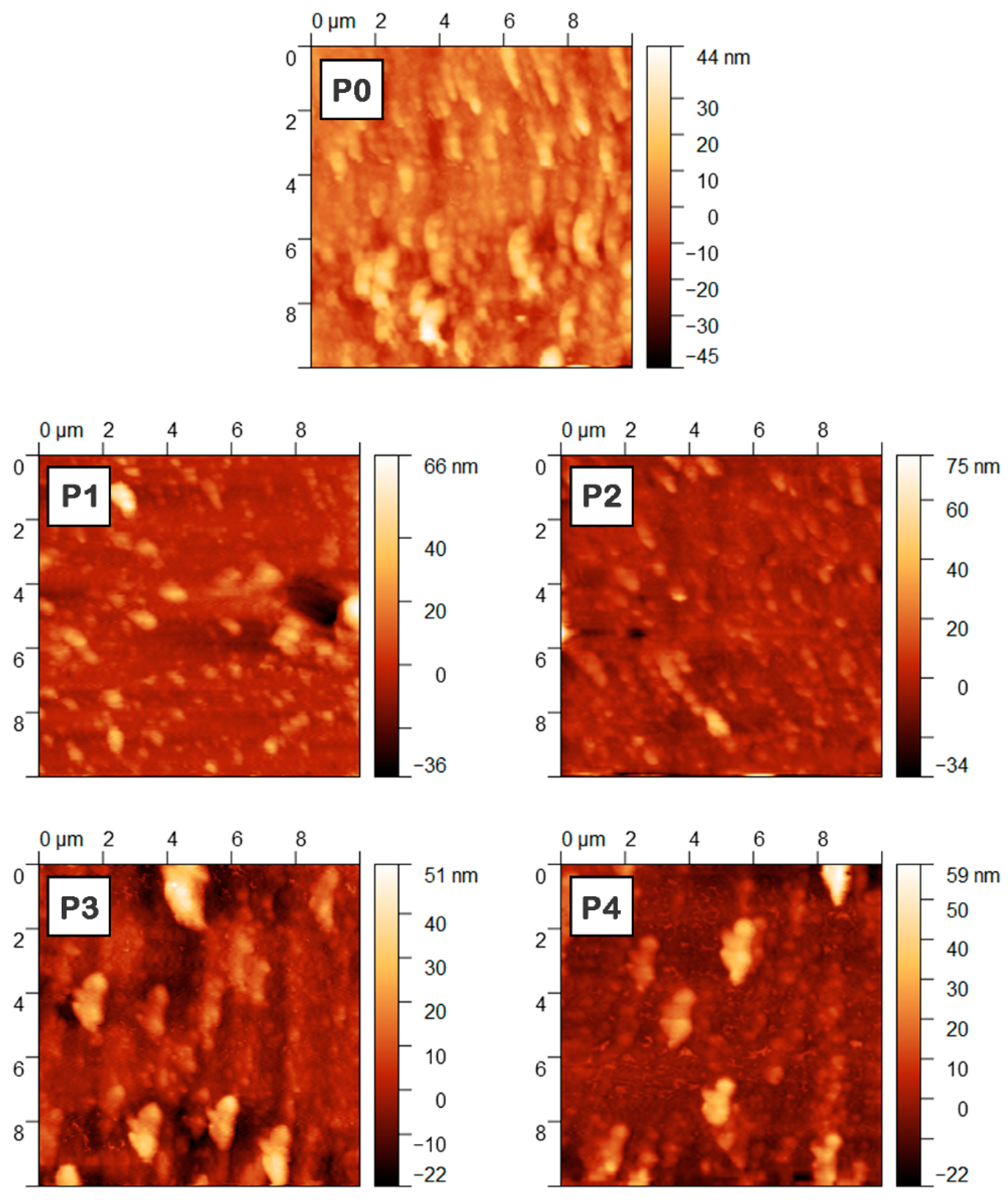
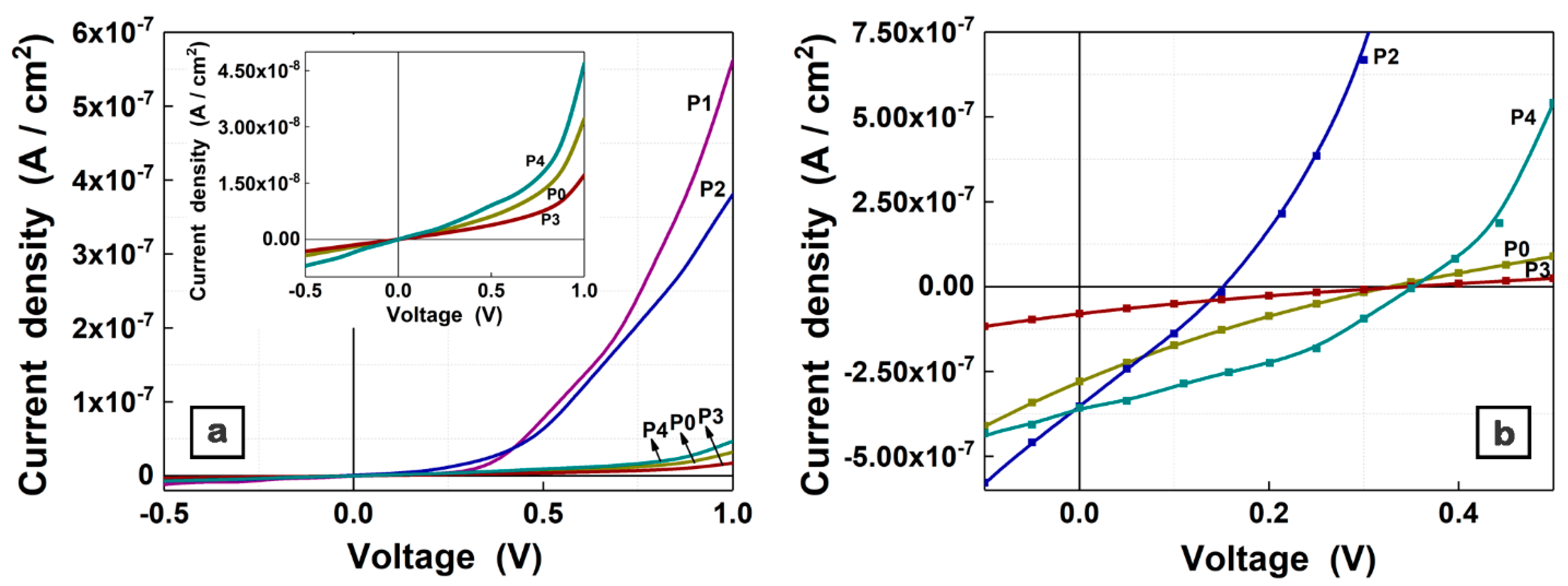
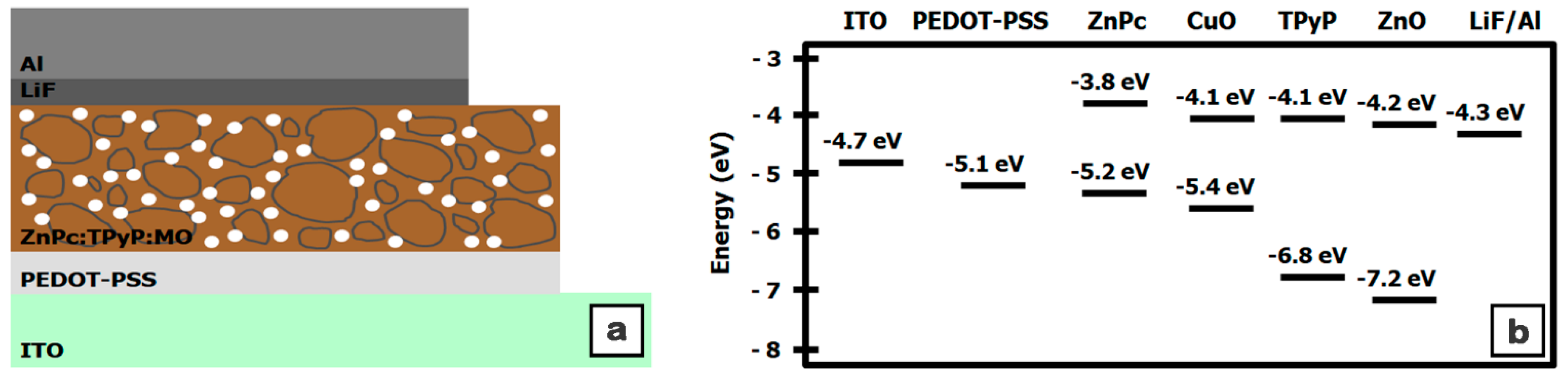
| Sample | Component Ratio ZnPc:TPyP:ZnO:CuO | Thickness (nm) | RMS (nm) | Ra (nm) |
|---|---|---|---|---|
| P0 | 1:1.5:0:0 | 105 | 5.3 | 3.9 |
| P1 | 1:1.275:0.225:0 | 60 | 8.3 | 5.3 |
| P2 | 1:1.125:0.375:0 | 95 | 6.5 | 4.4 |
| P3 | 0.85:1.5:0:0.15 | 95 | 8.8 | 6.2 |
| P4 | 0.75:1.5:0:0.25 | 90 | 8.6 | 5.6 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Socol, M.; Preda, N.; Breazu, C.; Costas, A.; Rasoga, O.; Petre, G.; Popescu-Pelin, G.; Iftimie, S.; Stochioiu, A.; Socol, G.; et al. Macrocyclic Compounds: Metal Oxide Particles Nanocomposite Thin Films Deposited by MAPLE. Materials 2023, 16, 2480. https://doi.org/10.3390/ma16062480
Socol M, Preda N, Breazu C, Costas A, Rasoga O, Petre G, Popescu-Pelin G, Iftimie S, Stochioiu A, Socol G, et al. Macrocyclic Compounds: Metal Oxide Particles Nanocomposite Thin Films Deposited by MAPLE. Materials. 2023; 16(6):2480. https://doi.org/10.3390/ma16062480
Chicago/Turabian StyleSocol, Marcela, Nicoleta Preda, Carmen Breazu, Andreea Costas, Oana Rasoga, Gabriela Petre, Gianina Popescu-Pelin, Sorina Iftimie, Andrei Stochioiu, Gabriel Socol, and et al. 2023. "Macrocyclic Compounds: Metal Oxide Particles Nanocomposite Thin Films Deposited by MAPLE" Materials 16, no. 6: 2480. https://doi.org/10.3390/ma16062480
APA StyleSocol, M., Preda, N., Breazu, C., Costas, A., Rasoga, O., Petre, G., Popescu-Pelin, G., Iftimie, S., Stochioiu, A., Socol, G., & Stanculescu, A. (2023). Macrocyclic Compounds: Metal Oxide Particles Nanocomposite Thin Films Deposited by MAPLE. Materials, 16(6), 2480. https://doi.org/10.3390/ma16062480










