Recent Advances in Chitosan-Based Applications—A Review
Abstract
1. Introduction
2. Synthesis of Chitosan Nanoparticles
3. Importance of Crosslinking
4. Applications of Chitosan
4.1. Drug Delivery
4.1.1. Drug Delivery to Proliferating Tissues
4.1.2. Posterior Segment Ophthalmic Drug Delivery
4.1.3. Controlled Release Topical Ophthalmic Delivery
4.1.4. Nanocarriers Based on Chitosan for Ophthalmic Use
4.2. Gene Delivery
4.3. Bioimaging
4.4. Wound Dressing
4.5. Tissue Engineering Applications of Chitosan
4.5.1. Fibre Based Scaffolds
4.5.2. Hydrogels
4.5.3. Chitosan Microspheres
4.5.4. Chitosan Membrane
4.6. Dentistry
4.7. Ophthalmology
4.7.1. Evaluation of Precorneal Retention and Tolerance
4.7.2. Translational Ophthalmic Applications
4.8. Chitosan as Nutraceuticals
4.9. Cosmetics and Cosmeceuticals
4.9.1. Cosmetic and Pharmaceutical Uses of Chitosan
4.9.2. Chitosan Hybrids for Cosmetic Uses
4.10. Use of Chitosan-Based Derivatives in Food Industry
4.10.1. Food Additives
4.10.2. Additive against Obesity
4.10.3. Additive for Shelf Life Extension of Food
4.10.4. Chitosan as an Antimicrobial Agent
4.10.5. As an Emulsifier
4.10.6. As a Flocculant
4.10.7. As a Functional Food Ingredient
4.10.8. Food Packaging
4.10.9. Chitosan and Blends with Other Biopolymers
4.10.10. Active and Intelligent Films Based on Chitosan
4.10.11. Chitosan Film in Food Packaging
4.10.12. Flexible Packaging Films
4.10.13. Films Infused with Natural Oils and Extracts
4.10.14. Films Infused with Antimicrobial Agents
4.10.15. Films Infused with Plasticizers and Cross-Linkers
4.10.16. Films Infused with Clays
4.10.17. Films Infused with Polysaccharide Particles, Fibers, and Whiskers
4.10.18. Edible Coatings
4.11. Wastewater Purification
4.11.1. Removal of Heavy Metal Ions
4.11.2. Removal of Dyes
4.11.3. Removal of Pesticides
4.11.4. Removal of Pharmaceuticals
4.12. Controlled Release Fertilizers
4.13. Dye Sensitised Solar Cells
4.14. Water Availability in Plants
4.15. Preventing of the Abiotic Stress in Flora
4.16. Controlling Salinity Stress
4.17. Controlling Heat and Drought Stress
4.18. Controlling Heavy Metal Toxicity
5. Toxicity of Chitosan
6. Key Challenges and Future Prospects
7. Conclusions
Funding
Conflicts of Interest
References
- Huq, T.; Khan, A.; Brown, D.; Dhayagude, N.; He, Z.; Ni, Y. Sources, Production and Commercial Applications of Fungal Chitosan: A Review. J. Bioresour. Bioprod. 2022, 7, 85–98. [Google Scholar] [CrossRef]
- Seenuvasan, M.; Sarojini, G.; Dineshkumar, M. Recovery of Chitosan from Natural Biotic Waste. Curr. Dev. Biotechnol. Bioeng. Resour. Recover. Wastes 2020, 115–133. [Google Scholar] [CrossRef]
- No, H.K.; Hur, E.Y. Control of Foam Formation by Antifoam during Demineralization of Crustacean Shell in Preparation of Chitin. J. Agric. Food Chem. 1998, 46, 3844–3846. [Google Scholar] [CrossRef]
- Percot, A.; Viton, C.; Domard, A. Characterization of Shrimp Shell Deproteinization. Biomacromolecules 2003, 4, 1380–1385. [Google Scholar] [CrossRef]
- Said Al Hoqani, H.A.; AL-Shaqsi, N.; Hossain, M.A.; Al Sibani, M.A. Isolation and Optimization of the Method for Industrial Production of Chitin and Chitosan from Omani Shrimp Shell. Carbohydr. Res. 2020, 492, 108001. [Google Scholar] [CrossRef]
- Hisham, F.; Maziati Akmal, M.H.; Ahmad, F.B.; Ahmad, K. Facile Extraction of Chitin and Chitosan from Shrimp Shell. Mater. Today Proc. 2021, 42, 2369–2373. [Google Scholar] [CrossRef]
- Trung, T.S.; Tram, L.H.; Van Tan, N.; Van Hoa, N.; Minh, N.C.; Loc, P.T.; Stevens, W.F. Improved Method for Production of Chitin and Chitosan from Shrimp Shells. Carbohydr. Res. 2020, 489, 107913. [Google Scholar] [CrossRef]
- Pakizeh, M.; Moradi, A.; Ghassemi, T. Chemical Extraction and Modification of Chitin and Chitosan from Shrimp Shells. Eur. Polym. J. 2021, 159, 110709. [Google Scholar] [CrossRef]
- Eddya, M.; Tbib, B.; EL-Hami, K. A Comparison of Chitosan Properties after Extraction from Shrimp Shells by Diluted and Concentrated Acids. Heliyon 2020, 6, e03486. [Google Scholar] [CrossRef]
- Li, K.; Guo, Z.; Chen, X.; Wang, J.; El-Araby, A.; El Ghadraoui, L.; Errachidi, F. Physicochemical Properties and Functional Characteristics of Ecologically Extracted Shrimp Chitosans with Different Organic Acids during Demineralization Step. Molecules 2022, 27, 8285. [Google Scholar] [CrossRef]
- Pohling, J.; Dave, D.; Liu, Y.; Murphy, W.; Trenholm, S. Two-Step Demineralization of Shrimp (Pandalus borealis) Shells Using Citric Acid: An Environmentally Friendly, Safe and Cost-Effective Alternative to the Traditional Approach. Green Chem. 2022, 24, 1141–1151. [Google Scholar] [CrossRef]
- Younes, I.; Rinaudo, M.; Harding, D.; Sashiwa, H. Chitin and Chitosan Preparation from Marine Sources. Structure, Properties and Applications. Mar. Drugs 2015, 13, 1133–1174. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.C.; Zhao, D.; Xue, C.; Mao, X. An Efficient Method for Chitin Production from Crab Shells by a Natural Deep Eutectic Solvent. Mar. Life Sci. Technol. 2022, 4, 384–388. [Google Scholar] [CrossRef]
- Admassu, H.; Gasmalla, M.A.A.; Yang, R.; Zhao, W. Bioactive Peptides Derived from Seaweed Protein and Their Health Benefits: Antihypertensive, Antioxidant, and Antidiabetic Properties. J. Food Sci. 2018, 83, 6–16. [Google Scholar] [CrossRef]
- Zamora-Sillero, J.; Gharsallaoui, A.; Prentice, C. Peptides from Fish By-Product Protein Hydrolysates and Its Functional Properties: An Overview. Mar. Biotechnol. 2018, 20, 118–130. [Google Scholar] [CrossRef]
- Santos, V.P.; Marques, N.S.S.; Maia, P.C.S.V.; de Lima, M.A.B.; de Franco, L.O.; de Campos-Takaki, G.M. Seafood Waste as Attractive Source of Chitin and Chitosan Production and Their Applications. Int. J. Mol. Sci. 2020, 21, 4290. [Google Scholar] [CrossRef]
- Casadidio, C.; Peregrina, D.V.; Gigliobianco, M.R.; Deng, S.; Censi, R.; Di Martino, P. Chitin and Chitosans: Characteristics, Eco-Friendly Processes, and Applications in Cosmetic Science. Mar. Drugs 2019, 17, 369. [Google Scholar] [CrossRef]
- Mhamdi, S.; Ktari, N.; Hajji, S.; Nasri, M.; Sellami Kamoun, A. Alkaline Proteases from a Newly Isolated Micromonospora Chaiyaphumensis S103: Characterization and Application as a Detergent Additive and for Chitin Extraction from Shrimp Shell Waste. Int. J. Biol. Macromol. 2017, 94, 415–422. [Google Scholar] [CrossRef]
- Hamdi, M.; Hammami, A.; Hajji, S.; Jridi, M.; Nasri, M.; Nasri, R. Chitin Extraction from Blue Crab (Portunus segnis) and Shrimp (Penaeus kerathurus) Shells Using Digestive Alkaline Proteases from P. Segnis Viscera. Int. J. Biol. Macromol. 2017, 101, 455–463. [Google Scholar] [CrossRef]
- Jantzen da Silva Lucas, A.; Quadro Oreste, E.; Leão Gouveia Costa, H.; Martín López, H.; Dias Medeiros Saad, C.; Prentice, C. Extraction, Physicochemical Characterization, and Morphological Properties of Chitin and Chitosan from Cuticles of Edible Insects. Food Chem. 2021, 343, 128550. [Google Scholar] [CrossRef]
- Valdez-Peña, A.U.; Espinoza-Perez, J.D.; Sandoval-Fabian, G.C.; Balagurusamy, N.; Hernandez-Rivera, A.; de-la-Garza-Rodriguez, I.M.; Contreras-Esquivel, J.C. Screening of Industrial Enzymes for Deproteinization of Shrimp Head for Chitin Recovery. Food Sci. Biotechnol. 2010, 19, 553–557. [Google Scholar] [CrossRef]
- Lee, D.H.; Doan, C.T.; Tran, T.N.; Nguyen, V.B.; Nguyen, A.D.; Wang, C.L.; Wang, S.L. Proteases Production and Chitin Preparation from the Liquid Fermentation of Chitinous Fishery By-Products by Paenibacillus Elgii. Mar. Drugs 2021, 19, 477. [Google Scholar] [CrossRef]
- Younes, I.; Ghorbel-Bellaaj, O.; Nasri, R.; Chaabouni, M.; Rinaudo, M.; Nasri, M. Chitin and Chitosan Preparation from Shrimp Shells Using Optimized Enzymatic Deproteinization. Process Biochem. 2012, 47, 2032–2039. [Google Scholar] [CrossRef]
- Younes, I.; Hajji, S.; Frachet, V.; Rinaudo, M.; Jellouli, K.; Nasri, M. Chitin Extraction from Shrimp Shell Using Enzymatic Treatment. Antitumor, Antioxidant and Antimicrobial Activities of Chitosan. Int. J. Biol. Macromol. 2014, 69, 489–498. [Google Scholar] [CrossRef]
- Lamarque, G.; Viton, C.; Domard, A. Comparative Study of the First Heterogeneous Deacetylation of α- and β-Chitins in a Multistep Process. Biomacromolecules 2004, 5, 992–1001. [Google Scholar] [CrossRef]
- Harmsen, R.A.G.; Tuveng, T.R.; Antonsen, S.G.; Eijsink, V.G.H.; Sørlie, M. Can We Make Chitosan by Enzymatic Deacetylation of Chitin? Molecules 2019, 24, 3862. [Google Scholar] [CrossRef]
- Yang, G.; Hou, X.; Lu, J.; Wang, M.; Wang, Y.; Huang, Y.; Liu, Q.; Liu, S.; Fang, Y. Enzymatic Modification of Native Chitin and Chitin Oligosaccharides by an Alkaline Chitin Deacetylase from Microbacterium Esteraromaticum MCDA02. Int. J. Biol. Macromol. 2022, 203, 671–678. [Google Scholar] [CrossRef]
- Kaczmarek, M.B.; Struszczyk-Swita, K.; Li, X.; Szczęsna-Antczak, M.; Daroch, M. Enzymatic Modifications of Chitin, Chitosan, and Chitooligosaccharides. Front. Bioeng. Biotechnol. 2019, 7, 243. [Google Scholar] [CrossRef]
- El Knidri, H.; Belaabed, R.; Addaou, A.; Laajeb, A.; Lahsini, A. Extraction, Chemical Modification and Characterization of Chitin and Chitosan. Int. J. Biol. Macromol. 2018, 120, 1181–1189. [Google Scholar] [CrossRef]
- Sullivan, D.J.; Cruz-Romero, M.; Collins, T.; Cummins, E.; Kerry, J.P.; Morris, M.A. Synthesis of Monodisperse Chitosan Nanoparticles. Food Hydrocoll. 2018, 83, 355–364. [Google Scholar] [CrossRef]
- Othman, N.; Masarudin, M.J.; Kuen, C.Y.; Dasuan, N.A.; Abdullah, L.C.; Jamil, S.N.A.M. Synthesis and Optimization of Chitosan Nanoparticles Loaded with L-Ascorbic Acid and Thymoquinone. Nanomaterials 2018, 8, 920. [Google Scholar] [CrossRef]
- Ghadi, A.; Mahjoub, S.; Tabandeh, F.; Talebnia, F. Synthesis and Optimization of Chitosan Nanoparticles: Potential Applications in Nanomedicine and Biomedical Engineering. Casp. J. Intern. Med. 2014, 5, 156. [Google Scholar]
- Liu, G.; Shao, L.; Ge, F.; Chen, J. Preparation of Ultrafine Chitosan Particles by Reverse Microemulsion. China Particuology 2007, 5, 384–390. [Google Scholar] [CrossRef]
- Asgari, S.; Saberi, A.H.; McClements, D.J.; Lin, M. Microemulsions as Nanoreactors for Synthesis of Biopolymer Nanoparticles. Trends Food Sci. Technol. 2019, 86, 118–130. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, Y.; Luo, G.; Dai, Y. Effect of Solvents and Precipitant on the Properties of Chitosan Nanoparticles in a Water-in-Oil Microemulsion and Its Lipase Immobilization Performance. Bioresour. Technol. 2010, 101, 841–844. [Google Scholar] [CrossRef]
- Pandey, P.; Dua, K.; Dureja, H. Erlotinib Loaded Chitosan Nanoparticles: Formulation, Physicochemical Characterization and Cytotoxic Potential. Int. J. Biol. Macromol. 2019, 139, 1304–1316. [Google Scholar] [CrossRef]
- Yurtdaş Kirimlioğlu, G.; Öztürk, A.A. Levocetirizine Dihydrochloride-Loaded Chitosan Nanoparticles: Formulation and In Vitro Evaluation. TURKISH J. Pharm. Sci. 2020, 17, 27–35. [Google Scholar] [CrossRef]
- Gover Antoniraj, M.; Maria Leena, M.; Moses, J.A.; Anandharamakrishnan, C. Cross-Linked Chitosan Microparticles Preparation by Modified Three Fluid Nozzle Spray Drying Approach. Int. J. Biol. Macromol. 2020, 147, 1268–1277. [Google Scholar] [CrossRef]
- Orellano, M.S.; Longo, G.S.; Porporatto, C.; Correa, N.M.; Falcone, R.D. Role of Micellar Interface in the Synthesis of Chitosan Nanoparticles Formulated by Reverse Micellar Method. Colloids Surfaces A Physicochem. Eng. Asp. 2020, 599, 124876. [Google Scholar] [CrossRef]
- Orellano, M.S.; Isaac, P.; Breser, M.L.; Bohl, L.P.; Conesa, A.; Falcone, R.D.; Porporatto, C. Chitosan Nanoparticles Enhance the Antibacterial Activity of the Native Polymer against Bovine Mastitis Pathogens. Carbohydr. Polym. 2019, 213, 1–9. [Google Scholar] [CrossRef]
- El-Naggar, N.E.A.; Saber, W.E.I.A.; Zweil, A.M.; Bashir, S.I. An Innovative Green Synthesis Approach of Chitosan Nanoparticles and Their Inhibitory Activity against Phytopathogenic Botrytis Cinerea on Strawberry Leaves. Sci. Rep. 2022, 12, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Gadkari, R.R.; Suwalka, S.; Yogi, M.R.; Ali, W.; Das, A.; Alagirusamy, R. Green Synthesis of Chitosan-Cinnamaldehyde Cross-Linked Nanoparticles: Characterization and Antibacterial Activity. Carbohydr. Polym. 2019, 226, 115298. [Google Scholar] [CrossRef]
- Galan, J.; Trilleras, J.; Zapata, P.A.; Arana, V.A.; Grande-Tovar, C.D. Optimization of Chitosan Glutaraldehyde-Crosslinked Beads for Reactive Blue 4 Anionic Dye Removal Using a Surface Response Methodology. Life 2021, 11, 85. [Google Scholar] [CrossRef] [PubMed]
- Duraisamy, N.; Dhayalan, S.; Shaik, M.R.; Shaik, A.H.; Shaik, J.P.; Shaik, B. Green Synthesis of Chitosan Nanoparticles Using of Martynia annua L. Ethanol Leaf Extract Their Antibacterial Activity. Crystals 2022, 12, 1550. [Google Scholar] [CrossRef]
- Saravanakumar, K.; Chelliah, R.; MubarakAli, D.; Jeevithan, E.; Oh, D.H.; Kathiresan, K.; Wang, M.H. Fungal Enzyme-Mediated Synthesis of Chitosan Nanoparticles and Its Biocompatibility, Antioxidant and Bactericidal Properties. Int. J. Biol. Macromol. 2018, 118, 1542–1549. [Google Scholar] [CrossRef]
- Tuorkey, M.; Khedr, Y.; Aborhyem, S.; Xue, X. Green Synthesis of Chicory (Cichorium intybus L.) Chitosan Nanoparticles and Evaluation of Their Anti-Fungal, Anti-Hemolytic, and Anti-Cancer Activities. J. Bioact. Compat. Polym. 2022, 37, 421–436. [Google Scholar] [CrossRef]
- Harish Prashanth, K.V.; Tharanathan, R.N. Chitin/Chitosan: Modifications and Their Unlimited Application Potential—An Overview. Trends Food Sci. Technol. 2007, 18, 117–131. [Google Scholar] [CrossRef]
- Kurita, K.; Koyama, Y.; Taniguchi, A. Studies on Chitin. IX. Crosslinking of Water-soluble Chitin and Evaluation of the Products as Adsorbents for Cupric Ion. J. Appl. Polym. Sci. 1986, 31, 1169–1176. [Google Scholar] [CrossRef]
- Wei, Y.C.; Hudson, S.M.; Mayer, J.M.; Kaplan, D.L. The Crosslinking of Chitosan Fibers. J. Polym. Sci. Part A Polym. Chem. 1992, 30, 2187–2193. [Google Scholar] [CrossRef]
- Li, J.; Cai, C.; Li, J.; Li, J.; Li, J.; Sun, T.; Wang, L.; Wu, H.; Yu, G. Chitosan-Based Nanomaterials for Drug Delivery. Molecules 2018, 23, 2661. [Google Scholar] [CrossRef]
- Gu, Z.; Aimetti, A.A.; Wang, Q.; Dang, T.T.; Zhang, Y.; Veiseh, O.; Cheng, H.; Langer, R.S.; Anderson, D.G. Injectable Nano-Network for Glucose-Mediated Insulin Delivery. ACS Nano 2013, 7, 4194–4201. [Google Scholar] [CrossRef] [PubMed]
- Engkagul, V.; Klaharn, I.Y.; Sereemaspun, A.; Chirachanchai, S. Chitosan Whisker Grafted with Oligo(Lactic Acid) Nanoparticles via a Green Synthesis Pathway: Potential as a Transdermal Drug Delivery System. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 2523–2531. [Google Scholar] [CrossRef] [PubMed]
- Rajitha, P.; Gopinath, D.; Biswas, R.; Sabitha, M.; Jayakumar, R. Chitosan Nanoparticles in Drug Therapy of Infectious and Inflammatory Diseases. Expert Opin. Drug Deliv. 2016, 13, 1177–1194. [Google Scholar] [CrossRef] [PubMed]
- Mao, S.; Sun, W.; Kissel, T. Chitosan-Based Formulations for Delivery of DNA and SiRNA. Adv. Drug Deliv. Rev. 2010, 62, 12–27. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhu, H.; Wang, J.; Fang, Q.; Peng, Z. Enzymatically Disulfide-Crosslinked Chitosan/Hyaluronic Acid Layer-by-Layer Self-Assembled Microcapsules for Redox-Responsive Controlled Release of Protein. ACS Appl. Mater. Interfaces 2018, 10, 33493–33506. [Google Scholar] [CrossRef]
- Bahram, M.; Mohseni, N.; Moghtader, M.; Bahram, M.; Mohseni, N.; Moghtader, M. An Introduction to Hydrogels and Some Recent Applications. Emerg. Concepts Anal. Appl. Hydrogels 2016. [Google Scholar] [CrossRef]
- Tao, F.; Ma, S.; Tao, H.; Jin, L.; Luo, Y.; Zheng, J.; Xiang, W.; Deng, H. Chitosan-Based Drug Delivery Systems: From Synthesis Strategy to Osteomyelitis Treatment–A Review. Carbohydr. Polym. 2021, 251, 117063. [Google Scholar] [CrossRef]
- Shoueir, K.R.; El-Desouky, N.; Rashad, M.M.; Ahmed, M.K.; Janowska, I.; El-Kemary, M. Chitosan Based-Nanoparticles and Nanocapsules: Overview, Physicochemical Features, Applications of a Nanofibrous Scaffold, and Bioprinting. Int. J. Biol. Macromol. 2021, 167, 1176–1197. [Google Scholar] [CrossRef]
- Dong, W.; Ye, J.; Wang, W.; Yang, Y.; Wang, H.; Sun, T.; Gao, L.; Liu, Y. Self-Assembled Lecithin/Chitosan Nanoparticles Based on Phospholipid Complex: A Feasible Strategy to Improve Entrapment Efficiency and Transdermal Delivery of Poorly Lipophilic Drug. Int. J. Nanomed. 2020, 15, 5629–5643. [Google Scholar] [CrossRef]
- Fereig, S.A.; El-Zaafarany, G.M.; Arafa, M.G.; Abdel-Mottaleb, M.M.A. Tacrolimus-Loaded Chitosan Nanoparticles for Enhanced Skin Deposition and Management of Plaque Psoriasis. Carbohydr. Polym. 2021, 268, 118238. [Google Scholar] [CrossRef]
- Walbi, I.A.; Ahmad, M.Z.; Ahmad, J.; Algahtani, M.S.; Alali, A.S.; Alsudir, S.A.; Aodah, A.H.; Albarqi, H.A. Development of a Curcumin-Loaded Lecithin/Chitosan Nanoparticle Utilizing a Box-Behnken Design of Experiment: Formulation Design and Influence of Process Parameters. Polymers 2022, 14, 3758. [Google Scholar] [CrossRef] [PubMed]
- Rukmangathen, R.; Yallamalli, I.M.; Yalavarthi, P.R. Formulation and Biopharmaceutical Evaluation of Risperidone-Loaded Chitosan Nanoparticles for Intranasal Delivery. Drug Dev. Ind. Pharm. 2019, 45, 1342–1350. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.M.; Madni, A.; Tahir, N.; Parveen, F.; Khan, S.; Jan, N.; Ali, A.; Abdurrahim, M.; Farooq, U.; Khan, M.I. Co-Delivery of Curcumin and Cisplatin to Enhance Cytotoxicity of Cisplatin Using Lipid-Chitosan Hybrid Nanoparticles. Int. J. Nanomed. 2020, 15, 2207–2217. [Google Scholar] [CrossRef] [PubMed]
- Burhan, A.M.; Klahan, B.; Cummins, W.; Andrés-Guerrero, V.; Byrne, M.E.; O’reilly, N.J.; Chauhan, A.; Fitzhenry, L.; Hughes, H. Posterior Segment Ophthalmic Drug Delivery: Role of Muco-Adhesion with a Special Focus on Chitosan. Pharmaceutics 2021, 13, 1685. [Google Scholar] [CrossRef]
- Asasutjarit, R.; Theerachayanan, T.; Kewsuwan, P.; Veeranodha, S.; Fuongfuchat, A.; Ritthidej, G.C. Development and Evaluation of Diclofenac Sodium Loaded-N-Trimethyl Chitosan Nanoparticles for Ophthalmic Use. AAPS PharmSciTech 2015, 16, 1013–1024. [Google Scholar] [CrossRef]
- Li, J.; Tian, S.; Tao, Q.; Zhao, Y.; Gui, R.; Yang, F.; Zang, L.; Chen, Y.; Ping, Q.; Hou, D. Montmorillonite/Chitosan Nanoparticles as a Novel Controlled-Release Topical Ophthalmic Delivery System for the Treatment of Glaucoma. Int. J. Nanomed. 2018, 13, 3975–3987. [Google Scholar] [CrossRef]
- Paliwal, R.; Paliwal, S.R.; Sulakhiya, K.; Das Kurmi, B.; Kenwat, R.; Mamgain, A. Chitosan-Based Nanocarriers for Ophthalmic Applications; Elsevier Ltd.: Amsterdam, The Netherlands, 2019; ISBN 9780081025536. [Google Scholar]
- Silva, D.; Pinto, L.F.V.; Bozukova, D.; Santos, L.F.; Serro, A.P.; Saramago, B. Chitosan/Alginate Based Multilayers to Control Drug Release from Ophthalmic Lens. Colloids Surfaces B Biointerfaces 2016, 147, 81–89. [Google Scholar] [CrossRef]
- Nagarwal, R.C.; Singh, P.N.; Kant, S.; Maiti, P.; Pandit, J.K. Chitosan Nanoparticles of 5-Fluorouracil for Ophthalmic Delivery: Characterization, in-Vitro and in-Vivo Study. Chem. Pharm. Bull. (Tokyo) 2011, 59, 272–278. [Google Scholar] [CrossRef]
- Abdelhamid, H.N.; Dowaidar, M.; Langel, Ü. Carbonized Chitosan Encapsulated Hierarchical Porous Zeolitic Imidazolate Frameworks Nanoparticles for Gene Delivery. Microporous Mesoporous Mater. 2020, 302, 110200. [Google Scholar] [CrossRef]
- Jaiswal, S.; Dutta, P.K.; Kumar, S.; Koh, J.; Pandey, S. Methyl Methacrylate Modified Chitosan: Synthesis, Characterization and Application in Drug and Gene Delivery. Carbohydr. Polym. 2019, 211, 109–117. [Google Scholar] [CrossRef]
- Huang, G.; Chen, Q.; Wu, W.; Wang, J.; Chu, P.K.; Bai, H.; Tang, G. Reconstructed Chitosan with Alkylamine for Enhanced Gene Delivery by Promoting Endosomal Escape. Carbohydr. Polym. 2020, 227, 115339. [Google Scholar] [CrossRef]
- Abdelhamid, H.N. Chitosan-Based Nanocarriers for Gene Delivery. Nanoeng. Biomater. 2022, 91–105. [Google Scholar] [CrossRef]
- Rahmani, S.; Hakimi, S.; Esmaeily, A.; Samadi, F.Y.; Mortazavian, E.; Nazari, M.; Mohammadi, Z.; Tehrani, N.R.; Tehrani, M.R. Novel Chitosan Based Nanoparticles as Gene Delivery Systems to Cancerous and Noncancerous Cells. Int. J. Pharm. 2019, 560, 306–314. [Google Scholar] [CrossRef]
- Kwak, S.Y.; Lew, T.T.S.; Sweeney, C.J.; Koman, V.B.; Wong, M.H.; Bohmert-Tatarev, K.; Snell, K.D.; Seo, J.S.; Chua, N.H.; Strano, M.S. Chloroplast-Selective Gene Delivery and Expression in Planta Using Chitosan-Complexed Single-Walled Carbon Nanotube Carriers. Nat. Nanotechnol. 2019, 14, 447–455. [Google Scholar] [CrossRef]
- Cao, Y.; Tan, Y.F.; Wong, Y.S.; Liew, M.W.J.; Venkatraman, S. Recent Advances in Chitosan-Based Carriers for Gene Delivery. Mar. Drugs 2019, 17, 381. [Google Scholar] [CrossRef] [PubMed]
- Harish, R.; Nisha, K.D.; Prabakaran, S.; Sridevi, B.; Harish, S.; Navaneethan, M.; Ponnusamy, S.; Hayakawa, Y.; Vinniee, C.; Ganesh, M.R. Cytotoxicity Assessment of Chitosan Coated CdS Nanoparticles for Bio-Imaging Applications. Appl. Surf. Sci. 2020, 499, 143817. [Google Scholar] [CrossRef]
- Shi, H.; Liang, N.; Liu, J.; Li, S.; Gong, X.; Yan, P.; Sun, S. AIE-Active Polymeric Micelles Based on Modified Chitosan for Bioimaging-Guided Targeted Delivery and Controlled Release of Paclitaxel. Carbohydr. Polym. 2021, 269, 118327. [Google Scholar] [CrossRef]
- Xu, Y.; Liang, N.; Liu, J.; Gong, X.; Yan, P.; Sun, S. Design and Fabrication of Chitosan-Based AIE Active Micelles for Bioimaging and Intelligent Delivery of Paclitaxel. Carbohydr. Polym. 2022, 290, 119509. [Google Scholar] [CrossRef]
- Mujeeb Rahman, P.; Muraleedaran, K.; Mujeeb, V.M.A. Applications of Chitosan Powder with in Situ Synthesized Nano ZnO Particles as an Antimicrobial Agent. Int. J. Biol. Macromol. 2015, 77, 266–272. [Google Scholar] [CrossRef]
- Jones, V.; Grey, J.; Bmj, K.H. Wound Dressings. 2006. Available online: https://www.bmj.com/ (accessed on 21 January 2023).
- Matica, M.A.; Aachmann, F.L.; Tøndervik, A.; Sletta, H.; Ostafe, V. Chitosan as a Wound Dressing Starting Material: Antimicrobial Properties and Mode of Action. Int. J. Mol. Sci. 2019, 20, 5889. [Google Scholar] [CrossRef]
- Liverani, L.; Abbruzzese, F.; Mozetic, P.; Basoli, F.; Rainer, A.; Trombetta, M. Electrospinning of Hydroxyapatite–Chitosan Nanofibers for Tissue Engineering Applications. Asia-Pacific J. Chem. Eng. 2014, 9, 407–414. [Google Scholar] [CrossRef]
- Zhou, Z.; Yan, D.; Cheng, X.; Kong, M.; Liu, Y.; Feng, C.; Chen, X. Biomaterials Based on N,N,N-Trimethyl Chitosan Fibers in Wound Dressing Applications. Int. J. Biol. Macromol. 2016, 89, 471–476. [Google Scholar] [CrossRef]
- Rajabi, M.; McConnell, M.; Cabral, J.; Ali, M.A. Chitosan Hydrogels in 3D Printing for Biomedical Applications. Carbohydr. Polym. 2021, 260, 117768. [Google Scholar] [CrossRef]
- Ma, Y.; Xin, L.; Tan, H.; Fan, M.; Li, J.; Jia, Y.; Ling, Z.; Chen, Y.; Hu, X. Chitosan Membrane Dressings Toughened by Glycerol to Load Antibacterial Drugs for Wound Healing. Mater. Sci. Eng. C 2017, 81, 522–531. [Google Scholar] [CrossRef]
- Güneş, S.; Tıhmınlıoğlu, F. Hypericum Perforatum Incorporated Chitosan Films as Potential Bioactive Wound Dressing Material. Int. J. Biol. Macromol. 2017, 102, 933–943. [Google Scholar] [CrossRef]
- Sivashanmugam, A.; Arun Kumar, R.; Vishnu Priya, M.; Nair, S.V.; Jayakumar, R. An Overview of Injectable Polymeric Hydrogels for Tissue Engineering. Eur. Polym. J. 2015, 72, 543–565. [Google Scholar] [CrossRef]
- Ambrogi, V.; Donnadio, A.; Pietrella, D.; Latterini, L.; Proietti, F.A.; Marmottini, F.; Padeletti, G.; Kaciulis, S.; Giovagnoli, S.; Ricci, M. Chitosan Films Containing Mesoporous SBA-15 Supported Silver Nanoparticles for Wound Dressing. J. Mater. Chem. B 2014, 2, 6054–6063. [Google Scholar] [CrossRef]
- Wu, J.; Su, C.; Jiang, L.; Ye, S.; Liu, X.; Shao, W. Green and Facile Preparation of Chitosan Sponges as Potential Wound Dressings. ACS Sustain. Chem. Eng. 2018, 6, 9145–9152. [Google Scholar] [CrossRef]
- Rajendran, S. Advanced Textiles for Wound Care; Woodhead Publishing: Clarkston, UK, 2009. [Google Scholar]
- Hiranpattanakul, P.; Jongjitpissamai, T.; Aungwerojanawit, S.; Tachaboonyakiat, W. Fabrication of a Chitin/Chitosan Hydrocolloid Wound Dressing and Evaluation of Its Bioactive Properties. Res. Chem. Intermed. 2018, 44, 4913–4928. [Google Scholar] [CrossRef]
- Liu, J.; Shen, H. Clinical Efficacy of Chitosan-Based Hydrocolloid Dressing in the Treatment of Chronic Refractory Wounds. Int. Wound J. 2022, 19, 2012–2018. [Google Scholar] [CrossRef]
- Technology–Axio Biosolutions. Available online: https://axiobio.com/technology/ (accessed on 21 January 2023).
- CHITODERM PLUS Chitosan-Wundauflage | Trusetal Verbandstoffwerk, 8 × 10 cm, PZN 04585606, MIT Haftrand. Available online: https://www.pflegediscount-shop.de/Chitoderm-Plus-mit-Haftrand (accessed on 21 January 2023).
- Maciej, S.; Becker, F.G.; Cleary, M.; Team, R.M.; Holtermann, H.; The, D.; Agenda, N.; Science, P.; Sk, S.K.; Hinnebusch, R.; et al. Synteza i Aktywność Biologiczna Nowych Analogów Tiosemikarbazonowych Chelatorów Żelaza. Uniw. Śląski 2013, 7, 343–354. [Google Scholar] [CrossRef]
- Haemostats & Wound Dressings Quickly Clot Blood | Celox. Available online: https://www.celoxmedical.com/ (accessed on 21 January 2023).
- ChitoClear-Natural, High-Quality Chitosan from Iceland. Available online: https://www.primex.is/products-services/chitoclear/ (accessed on 21 January 2023).
- Opticell Gelling Fiber Wound Dressings | Medline Industries, Inc. Available online: https://punchout.medline.com/product/Opticell-Gelling-Fiber-Wound-Dressings/Gelling-Fiber-Dressings/Z05-PF50473 (accessed on 21 January 2023).
- Shop-Tricol Biomedical. Available online: https://tricolbiomedical.com/allproducts/ (accessed on 21 January 2023).
- Hemostasis-Products. Available online: https://www.anscare.com/en/product/category/Hemostasis (accessed on 21 January 2023).
- Madni, A.; Kousar, R.; Naeem, N.; Wahid, F. Recent Advancements in Applications of Chitosan-Based Biomaterials for Skin Tissue Engineering. J. Bioresour. Bioprod. 2021, 6, 11–25. [Google Scholar] [CrossRef]
- Ressler, A. Chitosan-Based Biomaterials for Bone Tissue Engineering Applications: A Short Review. Polymers 2022, 14, 3430. [Google Scholar] [CrossRef]
- Capuana, E.; Lopresti, F.; Carfì Pavia, F.; Brucato, V.; La Carrubba, V. Solution-Based Processing for Scaffold Fabrication in Tissue Engineering Applications: A Brief Review. Polymers 2021, 13, 2041. [Google Scholar] [CrossRef] [PubMed]
- Rogina, A.; Pribolšan, L.; Hanžek, A.; Gómez-Estrada, L.; Gallego Ferrer, G.; Marijanović, I.; Ivanković, M.; Ivanković, H. Macroporous Poly(Lactic Acid) Construct Supporting the Osteoinductive Porous Chitosan-Based Hydrogel for Bone Tissue Engineering. Polymer 2016, 98, 172–181. [Google Scholar] [CrossRef]
- Deb, P.; Deoghare, A.B.; Borah, A.; Barua, E.; Das Lala, S. Scaffold Development Using Biomaterials: A Review. Mater. Today Proc. 2018, 5, 12909–12919. [Google Scholar] [CrossRef]
- Matthews, J.A.; Wnek, G.E.; Simpson, D.G.; Bowlin, G.L. Electrospinning of Collagen Nanofibers. Biomacromolecules 2002, 3, 232–238. [Google Scholar] [CrossRef]
- Levengood, S.K.L.; Zhang, M. Chitosan-Based Scaffolds for Bone Tissue Engineering. J. Mater. Chem. B 2014, 2, 3161–3184. [Google Scholar] [CrossRef] [PubMed]
- Homayoni, H.; Ravandi, S.A.H.; Valizadeh, M. Electrospinning of Chitosan Nanofibers: Processing Optimization. Carbohydr. Polym. 2009, 77, 656–661. [Google Scholar] [CrossRef]
- Koosha, M.; Raoufi, M.; Moravvej, H. One-Pot Reactive Electrospinning of Chitosan/PVA Hydrogel Nanofibers Reinforced by Halloysite Nanotubes with Enhanced Fibroblast Cell Attachment for Skin Tissue Regeneration. Colloids Surfaces B Biointerfaces 2019, 179, 270–279. [Google Scholar] [CrossRef]
- Zou, P.; Lee, W.H.; Gao, Z.; Qin, D.; Wang, Y.; Liu, J.; Sun, T.; Gao, Y. Wound Dressing from Polyvinyl Alcohol/Chitosan Electrospun Fiber Membrane Loaded with OH-CATH30 Nanoparticles. Carbohydr. Polym. 2020, 232, 115786. [Google Scholar] [CrossRef]
- Mishra, D.; Bhunia, B.; Banerjee, I.; Datta, P.; Dhara, S.; Maiti, T.K. Enzymatically Crosslinked Carboxymethyl–Chitosan/Gelatin/Nano-Hydroxyapatite Injectable Gels for in Situ Bone Tissue Engineering Application. Mater. Sci. Eng. C 2011, 31, 1295–1304. [Google Scholar] [CrossRef]
- Liu, H.; Li, H.; Cheng, W.; Yang, Y.; Zhu, M.; Zhou, C. Novel Injectable Calcium Phosphate/Chitosan Composites for Bone Substitute Materials. Acta Biomater. 2006, 2, 557–565. [Google Scholar] [CrossRef]
- Taghizadeh, M.; Taghizadeh, A.; Yazdi, M.K.; Zarrintaj, P.; Stadler, F.J.; Ramsey, J.D.; Habibzadeh, S.; Hosseini Rad, S.; Naderi, G.; Saeb, M.R.; et al. Chitosan-Based Inks for 3D Printing and Bioprinting. Green Chem. 2022, 24, 62–101. [Google Scholar] [CrossRef]
- Rogina, A.; Ressler, A.; Matić, I.; Gallego Ferrer, G.; Marijanović, I.; Ivanković, M.; Ivanković, H. Cellular Hydrogels Based on PH-Responsive Chitosan-Hydroxyapatite System. Carbohydr. Polym. 2017, 166, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Ji, C.; Annabi, N.; Khademhosseini, A.; Dehghani, F. Fabrication of Porous Chitosan Scaffolds for Soft Tissue Engineering Using Dense Gas CO2. Acta Biomater. 2011, 7, 1653–1664. [Google Scholar] [CrossRef] [PubMed]
- Patil, P.S.; Fathollahipour, S.; Inmann, A.; Pant, A.; Amini, R.; Shriver, L.P.; Leipzig, N.D. Fluorinated Methacrylamide Chitosan Hydrogel Dressings Improve Regenerated Wound Tissue Quality in Diabetic Wound Healing. Adv. Wound Care 2019, 8, 374–385. [Google Scholar] [CrossRef]
- Ding, C.C.; Teng, S.H.; Pan, H. In-Situ Generation of Chitosan/Hydroxyapatite Composite Microspheres for Biomedical Application. Mater. Lett. 2012, 79, 72–74. [Google Scholar] [CrossRef]
- Huang, L.; Xiao, L.; Poudel, A.J.; Li, J.; Zhou, P.; Gauthier, M.; Liu, H.; Wu, Z.; Yang, G. Porous Chitosan Microspheres as Microcarriers for 3D Cell Culture. Elsevier 2018, 202, 611–620. [Google Scholar] [CrossRef]
- Lončarević, A.; Ivanković, M.; Materials, A.R. Electrosprayed Chitosan–Copper Complex Microspheres with Uniform Size. Materials 2021, 14, 5630. [Google Scholar] [CrossRef]
- Zhao, J.; Tian, H.; Shang, F.; Lv, T.; Chen, D.; Feng, J. Injectable, Anti-Cancer Drug-Eluted Chitosan Microspheres against Osteosarcoma. J. Funct. Biomater. 2022, 13, 91. [Google Scholar] [CrossRef] [PubMed]
- Behera, S.S.; Das, U.; Kumar, A.; Bissoyi, A.; Singh, A.K. Chitosan/TiO2 Composite Membrane Improves Proliferation and Survival of L929 Fibroblast Cells: Application in Wound Dressing and Skin Regeneration. Int. J. Biol. Macromol. 2017, 98, 329–340. [Google Scholar] [CrossRef] [PubMed]
- Tamer, T.M.; Valachová, K.; Hassan, M.A.; Omer, A.M.; El-Shafeey, M.; Mohy Eldin, M.S.; Šoltés, L. Chitosan/Hyaluronan/Edaravone Membranes for Anti-Inflammatory Wound Dressing: In Vitro and in Vivo Evaluation Studies. Mater. Sci. Eng. C 2018, 90, 227–235. [Google Scholar] [CrossRef]
- Vivcharenko, V.; Benko, A.; Palka, K.; Wojcik, M.; Przekora, A. Elastic and Biodegradable Chitosan/Agarose Film Revealing Slightly Acidic PH for Potential Applications in Regenerative Medicine as Artificial Skin Graft. Int. J. Biol. Macromol. 2020, 164, 172–183. [Google Scholar] [CrossRef]
- Javed, R.; Rais, F.; Kaleem, M.; Jamil, B.; Ahmad, M.A.; Yu, T.; Qureshi, S.W.; Ao, Q. Chitosan Capping of CuO Nanoparticles: Facile Chemical Preparation, Biological Analysis, and Applications in Dentistry. Int. J. Biol. Macromol. 2021, 167, 1452–1467. [Google Scholar] [CrossRef]
- Zeza, B.; Wohlfahrt, C.; Pilloni, A. Chitosan Brush for Professional Removal of Plaque in Mild Peri-Implantitis. Minerva Stomatol. 2017, 66, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.N.; Koldsland, O.C.; Roos-Jansåker, A.M.; Wohlfahrt, J.C.; Verket, A.; Mdala, I.; Magnusson, A.; Salvesen, E.; Hjortsjö, C. Non-Surgical Treatment of Mild to Moderate Peri-Implantitis Using an Oscillating Chitosan Brush or a Titanium Curette—A Randomized Multicentre Controlled Clinical Trial. Clin. Oral Implants Res. 2022, 33, 1254–1264. [Google Scholar] [CrossRef] [PubMed]
- Wohlfahrt, J.C.; Aass, A.M.; Koldsland, O.C. Treatment of Peri-Implant Mucositis with a Chitosan Brush—A Pilot Randomized Clinical Trial. Int. J. Dent. Hyg. 2019, 17, 170–176. [Google Scholar] [CrossRef] [PubMed]
- Lei, L.; Li, X.; Xiong, T.; Yu, J.; Yu, X.; Song, Z.; Li, X. Covalently Cross-Linked Chitosan Hydrogel Sheet for Topical Ophthalmic Delivery of Levofloxacin. J. Biomed. Nanotechnol. 2018, 14, 371–378. [Google Scholar] [CrossRef]
- Irimia, T.; Dinu-Pîrvu, C.E.; Ghica, M.V.; Lupuleasa, D.; Muntean, D.L.; Udeanu, D.I.; Popa, L. Chitosan-Based in Situ Gels for Ocular Delivery of Therapeutics: A State-of-the-Art Review. Mar. Drugs 2018, 16, 373. [Google Scholar] [CrossRef]
- Braga, M.E.M.; Pato, M.T.V.; Silva, H.S.R.C.; Ferreira, E.I.; Gil, M.H.; Duarte, C.M.M.; de Sousa, H.C. Supercritical Solvent Impregnation of Ophthalmic Drugs on Chitosan Derivatives. J. Supercrit. Fluids 2008, 44, 245–257. [Google Scholar] [CrossRef]
- Vichare, R.; Garner, I.; Paulson, R.J.; Tzekov, R.; Sahiner, N.; Panguluri, S.K.; Mohapatra, S.; Mohapatra, S.S.; Ayyala, R.; Sneed, K.B.; et al. Biofabrication of Chitosan-Based Nanomedicines and Its Potential Use for Translational Ophthalmic Applications. Appl. Sci. 2020, 10, 4189. [Google Scholar] [CrossRef]
- Irimia, T.; Ghica, M.V.; Popa, L.; Anuţa, V.; Arsene, A.L.; Dinu-Pîrvu, C.E. Strategies for Improving Ocular Drug Bioavailability and Cornealwound Healing with Chitosan-Based Delivery Systems. Polymers 2018, 10, 1221. [Google Scholar] [CrossRef]
- Zamboulis, A.; Nanaki, S.; Michailidou, G.; Koumentakou, I.; Lazaridou, M.; Ainali, N.M.; Xanthopoulou, E.; Bikiaris, D.N. Chitosan and Its Derivatives for Ocular Delivery Formulations: Recent Advances and Developments. Polymers 2020, 12, 1519. [Google Scholar] [CrossRef] [PubMed]
- Alonso, M.J.; Sánchez, A. The Potential of Chitosan in Ocular Drug Delivery. J. Pharm. Pharmacol. 2010, 55, 1451–1463. [Google Scholar] [CrossRef]
- Akbari-Alavijeh, S.; Shaddel, R.; Jafari, S.M. Encapsulation of Food Bioactives and Nutraceuticals by Various Chitosan-Based Nanocarriers. Food Hydrocoll. 2020, 105, 105774. [Google Scholar] [CrossRef]
- Patti, A.M.; Katsiki, N.; Nikolic, D.; Al-Rasadi, K.; Rizzo, M. Nutraceuticals in Lipid-Lowering Treatment: A Narrative Review on the Role of Chitosan. Angiology 2015, 66, 416–421. [Google Scholar] [CrossRef]
- Je, J.Y.; Kim, S.K. Chitosan as Potential Marine Nutraceutical, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2012; Volume 65, ISBN 9780124160033. [Google Scholar]
- Abd El-Hack, M.E.; El-Saadony, M.T.; Shafi, M.E.; Zabermawi, N.M.; Arif, M.; Batiha, G.E.; Khafaga, A.F.; Abd El-Hakim, Y.M.; Al-Sagheer, A.A. Antimicrobial and Antioxidant Properties of Chitosan and Its Derivatives and Their Applications: A Review. Int. J. Biol. Macromol. 2020, 164, 2726–2744. [Google Scholar] [CrossRef]
- Kim, S. Competitive Biological Activities of Chitosan and Its Derivatives: Antimicrobial, Antioxidant, Anticancer, and Anti-Inflammatory Activities. Int. J. Polym. Sci. 2018, 2018, 1708172. [Google Scholar] [CrossRef]
- Gomes, L.P.; Paschoalin, V.M.F.; Del Aguila, E.M. Chitosan Nanoparticles: Production, Physicochemical Characteristics and Nutraceutical Applications. Rev. Virtual Quim. 2017, 9, 387–409. [Google Scholar] [CrossRef]
- Guzmán, E.; Ortega, F.; Rubio, R.G. Chitosan: A Promising Multifunctional Cosmetic Ingredient for Skin and Hair Care. Cosmetics 2022, 9, 99. [Google Scholar] [CrossRef]
- Aranaz, I.; Acosta, N.; Civera, C.; Elorza, B.; Mingo, J.; Castro, C.; De los Llanos Gandía, M.; Caballero, A.H. Cosmetics and Cosmeceutical Applications of Chitin, Chitosan and Their Derivatives. Polymers 2018, 10, 213. [Google Scholar] [CrossRef]
- Brigham, C. Chitin and Chitosan: Sustainable, Medically Relevant Biomaterials. Int. J. Biotechnol. Wellness Ind. 2017, 6, 41–47. [Google Scholar] [CrossRef]
- Sionkowska, A.; Kaczmarek, B.; Michalska, M.; Lewandowska, K.; Grabska, S. Preparation and Characterization of Collagen/Chitosan/Hyaluronic Acid Thin Films for Application in Hair Care Cosmetics. Pure Appl. Chem. 2017, 89, 1829–1839. [Google Scholar] [CrossRef]
- Morin-Crini, N.; Lichtfouse, E.; Torri, G.; Crini, G. Applications of Chitosan in Food, Pharmaceuticals, Medicine, Cosmetics, Agriculture, Textiles, Pulp and Paper, Biotechnology, and Environmental Chemistry. Environ. Chem. Lett. 2019, 17, 1667–1692. [Google Scholar] [CrossRef]
- Rejinold, N.S.; Choi, G.; Choy, J.H. Chitosan Hybrids for Cosmeceutical Applications in Skin, Hair and Dental Care: An Update. Emergent Mater. 2021, 4, 1125–1142. [Google Scholar] [CrossRef]
- Ahn, C.-B.; Je, J.-Y. Chitosan and Its Derivatives as Potential Cosmetic Agents: A Review. J. Chitin Chitosan 2015, 20, 287–296. [Google Scholar] [CrossRef]
- Hydamer CMFP-Chitinor. Available online: https://chitinor.com/hydamer-cmfp/ (accessed on 23 January 2023).
- Ovensa Inc. | Aurora, ON, Canada Startup. Available online: https://ovensa.com/ (accessed on 23 January 2023).
- Rita Corporation-Your Source for Specialty Chemicals Worldwide. Available online: https://www.ritacorp.com/ (accessed on 23 January 2023).
- Micellar Shampoo–Kristin Ess Hair. Available online: https://www.kristinesshair.com/products/scalp-purifying-micellar-shampoo (accessed on 23 January 2023).
- Home-CLR Berlin. Available online: https://www.clr-berlin.com/ (accessed on 23 January 2023).
- Buy SEVEN Professional Haircare Products | SEVEN. Available online: https://7haircare.com/shop/ (accessed on 23 January 2023).
- Clariant Specialty Chemicals. Available online: https://www.clariant.com/en/Corporate (accessed on 23 January 2023).
- Hyaluron Plump Conditioner, with Hyaluronic Acid, 591ml l L’Oréal Paris. Available online: https://www.lorealparis.ca/en-ca/hair-expertise/hyaluron-plump/hyaluron-plump-conditioner-591ml (accessed on 23 January 2023).
- Vegetal & Fungal Chitosan Manufactured in Europe-KitoZyme. Available online: https://chitolytic.com/ (accessed on 23 January 2023).
- Set Me Up Max Hold Gel | Herbal Essences. Available online: https://herbalessences.ca/en-ca/our-products/set-me-up/set-me-up-gel/ (accessed on 23 January 2023).
- Home-Personal Care Products Council. Available online: https://www.personalcarecouncil.org/ (accessed on 23 January 2023).
- Luxury Care for Kinky, Coily, Curly, Wavy, Straight and Damaged Hair–VERNON FRANÇOIS®. Available online: https://www.vernonfrancois.com/ (accessed on 23 January 2023).
- Home-Primex Manufacturing. Available online: https://www.primexvents.com/ (accessed on 23 January 2023).
- Murad Skincare | Clinical Skin Care Company. Available online: https://www.muradskincare.ca/ (accessed on 23 January 2023).
- Sino Lion USA. Available online: http://www.sinolion.com/ (accessed on 23 January 2023).
- Ultimate Miracle Worker SPF 30 Moisturizer | Philosophy®. Available online: https://www.philosophy.com/ultimate-miracle-worker-spf-30.html (accessed on 23 January 2023).
- Biocide Specialist at Your Side | Vink Chemicals. Available online: https://www.vink-chemicals.com/ (accessed on 23 January 2023).
- Alpine Minerals Replenishing Body Lotion | St. Ives®. Available online: https://www.stives.com/ca/en/products/replenishing-alpine-minerals-body-lotion (accessed on 23 January 2023).
- Jeen International-Products. Available online: https://www.jeen.com/ (accessed on 23 January 2023).
- Da Silva, S.B.; De Souza, D.; Lacerda, L.D. Food Applications of Chitosan and Its Derivatives. Chitin Chitosan Prop. Appl. 2019, 1, 315–347. [Google Scholar] [CrossRef]
- Harkin, C.; Mehlmer, N.; Woortman, D.V.; Brück, T.B.; Brück, W.M. Nutritional and Additive Uses of Chitin and Chitosan in the Food Industry; Springer: Cham, Switzerland, 2019; ISBN 9783030165819. [Google Scholar]
- Hashemi Gahruie, H.; Niakousari, M. Antioxidant, Antimicrobial, Cell Viability and Enzymatic Inhibitory of Antioxidant Polymers as Biological Macromolecules. Int. J. Biol. Macromol. 2017, 104, 606–617. [Google Scholar] [CrossRef]
- Singh, A.; Mittal, A.; Benjakul, S. Chitosan Nanoparticles: Preparation, Food Applications and Health Benefits. ScienceAsia 2021, 47, 1–10. [Google Scholar] [CrossRef]
- Singh, A.; Mittal, A.; Benjakul, S. Full Utilization of Squid Meat and Its Processing By-Products: Revisit. Food Rev. Int. 2022, 38, 455–479. [Google Scholar] [CrossRef]
- Paquin, F.; Rivnay, J.; Salleo, A.; Stingelin, N.; Silva, C. Multi-Phase Semicrystalline Microstructures Drive Exciton Dissociation in Neat Plastic Semiconductors. J. Mater. Chem. C 2015, 3, 10715–10722. [Google Scholar] [CrossRef]
- Muzzarelli, R.A.A. Chitosan-Based Dietary Foods. Carbohydr. Polym. 1996, 29, 309–316. [Google Scholar] [CrossRef]
- Hirano, S.; Akiyama, Y. Absence of a Hypocholesterolaemic Action of Chitosan in High-serum-cholesterol Rabbits. J. Sci. Food Agric. 1995, 69, 91–94. [Google Scholar] [CrossRef]
- Kabanov, V.L.; Novinyuk, L.V. Chitosan Application in Food Technology: A Review of Rescent Advances. Food Syst. 2020, 3, 10–15. [Google Scholar] [CrossRef]
- Kulawik, P.; Jamróz, E.; Özogul, F. Chitosan Role for Shelf-Life Extension of Seafood. Environ. Chem. Lett. 2020, 18, 61–74. [Google Scholar] [CrossRef]
- Xie, W.; Xu, P.; Liu, Q. Antioxidant Activity of Water-Soluble Chitosan Derivatives. Bioorganic Med. Chem. Lett. 2001, 11, 1699–1701. [Google Scholar] [CrossRef]
- Leceta, I.; Molinaro, S.; Guerrero, P.; Kerry, J.P.; De la Caba, K. Quality Attributes of Map Packaged Ready-to-Eat Baby Carrots by Using Chitosan-Based Coatings. Postharvest Biol. Technol. 2015, 100, 142–150. [Google Scholar] [CrossRef]
- Villafañe, F. Edible Coatings for Carrots. Food Rev. Int. 2016, 33, 84–103. [Google Scholar] [CrossRef]
- Kemboi, V.J.; Kipkoech, C.; Njire, M.; Were, S.; Lagat, M.K.; Ndwiga, F.; Wesonga, J.M.; Tanga, C.M. Biocontrol Potential of Chitin and Chitosan Extracted from Black Soldier Fly Pupal Exuviae against Bacterial Wilt of Tomato. Microorganisms 2022, 10, 165. [Google Scholar] [CrossRef]
- Olawuyi, I.F.; Lee, W. Influence of Chitosan Coating and Packaging Materials on the Quality Characteristics of Fresh-Cut Cucumber. Korean J. Food Preserv. 2019, 26, 371–380. [Google Scholar] [CrossRef]
- Ali, A.; Noh, N.M.; Mustafa, M.A. Antimicrobial Activity of Chitosan Enriched with Lemongrass Oil against Anthracnose of Bell Pepper. Food Packag. Shelf Life 2015, 3, 56–61. [Google Scholar] [CrossRef]
- Suseno, N.; Savitri, E.; Sapei, L.; Padmawijaya, K.S. Improving Shelf-Life of Cavendish Banana Using Chitosan Edible Coating. Procedia Chem. 2014, 9, 113–120. [Google Scholar] [CrossRef]
- Li, H.; Wang, Y.; Liu, F.; Yang, Y.; Wu, Z.; Cai, H.; Zhang, Q.; Wang, Y.; Li, P. Effects of Chitosan on Control of Postharvest Blue Mold Decay of Apple Fruit and the Possible Mechanisms Involved. Sci. Hortic. 2015, 186, 77–83. [Google Scholar] [CrossRef]
- Varasteh, F.; Arzani, K.; Barzegar, M.; Zamani, Z. Pomegranate (Punica Granatum L.) Fruit Storability Improvement Using Pre-Storage Chitosan Coating Technique. 2018. Available online: http://ir.jkuat.ac.ke/handle/123456789/3699 (accessed on 25 January 2023).
- Tayel, A.A.; Ibrahim, S.I.A.; Al-Saman, M.A.; Moussa, S.H. Production of Fungal Chitosan from Date Wastes and Its Application as a Biopreservative for Minced Meat. Int. J. Biol. Macromol. 2014, 69, 471–475. [Google Scholar] [CrossRef] [PubMed]
- Kumar-Krishnan, S.; Prokhorov, E.; Hernández-Iturriaga, M.; Mota-Morales, J.D.; Vázquez-Lepe, M.; Kovalenko, Y.; Sanchez, I.C.; Luna-Bárcenas, G. Chitosan/Silver Nanocomposites: Synergistic Antibacterial Action of Silver Nanoparticles and Silver Ions. Eur. Polym. J. 2015, 67, 242–251. [Google Scholar] [CrossRef]
- Raghavendra, G.M.; Jung, J.; Kim, D.; Seo, J. Microwave Assisted Antibacterial Chitosan–Silver Nanocomposite Films. Int. J. Biol. Macromol. 2016, 84, 281–288. [Google Scholar] [CrossRef]
- Yu, W.Z.; Zhang, Y.; Liu, X.; Xiang, Y.; Li, Z.; Wu, S. Synergistic Antibacterial Activity of Multi Components in Lysozyme/Chitosan/Silver/Hydroxyapatite Hybrid Coating. Mater. Des. 2018, 139, 351–362. [Google Scholar] [CrossRef]
- Al-Naamani, L.; Dobretsov, S.; Dutta, J.; Burgess, J.G. Chitosan-Zinc Oxide Nanocomposite Coatings for the Prevention of Marine Biofouling. Chemosphere 2017, 168, 408–417. [Google Scholar] [CrossRef]
- Al-Naamani, L.; Dobretsov, S.; Dutta, J. Chitosan-Zinc Oxide Nanoparticle Composite Coating for Active Food Packaging Applications. Innov. Food Sci. Emerg. Technol. 2016, 38, 231–237. [Google Scholar] [CrossRef]
- Rinaudo, M. Chitin and Chitosan: Properties and Applications. Prog. Polym. Sci. 2006, 31, 603–632. [Google Scholar] [CrossRef]
- Abdelmalek, B.E.; Sila, A.; Haddar, A.; Bougatef, A.; Ayadi, M.A. β-Chitin and Chitosan from Squid Gladius: Biological Activities of Chitosan and Its Application as Clarifying Agent for Apple Juice. Int. J. Biol. Macromol. 2017, 104, 953–962. [Google Scholar] [CrossRef] [PubMed]
- Taştan, Ö.; Baysal, T. Chitosan as a Novel Clarifying Agent on Clear Apple Juice Production: Optimization of Process Conditions and Changes on Quality Characteristics. Food Chem. 2017, 237, 818–824. [Google Scholar] [CrossRef] [PubMed]
- Gassara, F.; Antzak, C.; Ajila, C.M.; Sarma, S.J.; Brar, S.K.; Verma, M. Chitin and Chitosan as Natural Flocculants for Beer Clarification. J. Food Eng. 2015, 166, 80–85. [Google Scholar] [CrossRef]
- Abebe, L.S.; Chen, X.; Sobsey, M.D.; Gray, N.F.; Karanis, P. Chitosan Coagulation to Improve Microbial and Turbidity Removal by Ceramic Water Filtration for Household Drinking Water Treatment. Int. J. Environ. Res. Public Health 2016, 13, 269. [Google Scholar] [CrossRef]
- Ylitalo, R.; Lehtinen, S.; Wuolijoki, E.; Ylitalo, P.; Lehtimäki, T. Cholesterol-Lowering Properties and Safety of Chitosan. Arzneimittel-Forschung/Drug Res. 2002, 52, 1–7. [Google Scholar] [CrossRef]
- Bokura, H.; Kobayashi, S. Chitosan Decreases Total Cholesterol in Women: A Randomized, Double-Blind, Placebo-Controlled Trial. Eur. J. Clin. Nutr. 2003, 57, 721–725. [Google Scholar] [CrossRef]
- Rezaee, M.; Askari, G.; EmamDjomeh, Z.; Salami, M. Effect of Organic Additives on Physiochemical Properties and Anti-Oxidant Release from Chitosan-Gelatin Composite Films to Fatty Food Simulant. Int. J. Biol. Macromol. 2018, 114, 844–850. [Google Scholar] [CrossRef]
- Garavand, F.; Cacciotti, I.; Vahedikia, N.; Rehman, A.; Tarhan, Ö.; Akbari-Alavijeh, S.; Shaddel, R.; Rashidinejad, A.; Nejatian, M.; Jafarzadeh, S.; et al. A Comprehensive Review on the Nanocomposites Loaded with Chitosan Nanoparticles for Food Packaging. Crit. Rev. Food Sci. Nutr. 2022, 62, 1383–1416. [Google Scholar] [CrossRef]
- Priyadarshi, R.; Rhim, J.W. Chitosan-Based Biodegradable Functional Films for Food Packaging Applications. Innov. Food Sci. Emerg. Technol. 2020, 62, 102346. [Google Scholar] [CrossRef]
- Kumar, S.; Mukherjee, A.; Dutta, J. Chitosan Based Nanocomposite Films and Coatings: Emerging Antimicrobial Food Packaging Alternatives. Trends Food Sci. Technol. 2020, 97, 196–209. [Google Scholar] [CrossRef]
- Flórez, M.; Guerra-Rodríguez, E.; Cazón, P.; Vázquez, M. Chitosan for Food Packaging: Recent Advances in Active and Intelligent Films. Food Hydrocoll. 2022, 124, 107328. [Google Scholar] [CrossRef]
- Haghighi, H.; Licciardello, F.; Fava, P.; Siesler, H.W.; Pulvirenti, A. Recent Advances on Chitosan-Based Films for Sustainable Food Packaging Applications. Food Packag. Shelf Life 2020, 26, 100551. [Google Scholar] [CrossRef]
- Cazón, P.; Vázquez, M. Mechanical and Barrier Properties of Chitosan Combined with Other Components as Food Packaging Film. Environ. Chem. Lett. 2020, 18, 257–267. [Google Scholar] [CrossRef]
- Arfat, Y.A.; Ahmed, J.; Hiremath, N.; Auras, R.; Joseph, A. Thermo-Mechanical, Rheological, Structural and Antimicrobial Properties of Bionanocomposite Films Based on Fish Skin Gelatin and Silver-Copper Nanoparticles. Food Hydrocoll. 2017, 62, 191–202. [Google Scholar] [CrossRef]
- Bonilla, J.; Fortunati, E.; Atarés, L.; Chiralt, A.; Kenny, J.M. Physical, Structural and Antimicrobial Properties of Poly Vinyl Alcohol–Chitosan Biodegradable Films. Food Hydrocoll. 2014, 35, 463–470. [Google Scholar] [CrossRef]
- Cazón, P.; Vázquez, M. Applications of Chitosan as Food Packaging Materials. Sustain. Agric. Rev. 2019, 36, 81–123. [Google Scholar] [CrossRef]
- Wyrwa, J.; Barska, A. Innovations in the Food Packaging Market: Active Packaging. Eur. Food Res. Technol. 2017, 243, 1681–1692. [Google Scholar] [CrossRef]
- Yildirim, S.; Röcker, B.; Pettersen, M.K.; Nilsen-Nygaard, J.; Ayhan, Z.; Rutkaite, R.; Radusin, T.; Suminska, P.; Marcos, B.; Coma, V. Active Packaging Applications for Food. Compr. Rev. Food Sci. Food Saf. 2018, 17, 165–199. [Google Scholar] [CrossRef]
- Hafsa, J.; Smach, M.A.; Ben Khedher, M.R.; Charfeddine, B.; Limem, K.; Majdoub, H.; Rouatbi, S. Physical, Antioxidant and Antimicrobial Properties of Chitosan Films Containing Eucalyptus Globulus Essential Oil. LWT-Food Sci. Technol. 2016, 68, 356–364. [Google Scholar] [CrossRef]
- Perdones, Á.; Vargas, M.; Atarés, L.; Chiralt, A. Physical, Antioxidant and Antimicrobial Properties of Chitosan–Cinnamon Leaf Oil Films as Affected by Oleic Acid. Food Hydrocoll. 2014, 36, 256–264. [Google Scholar] [CrossRef]
- Sánchez-González, L.; Cháfer, M.; Chiralt, A.; González-Martínez, C. Physical Properties of Edible Chitosan Films Containing Bergamot Essential Oil and Their Inhibitory Action on Penicillium Italicum. Carbohydr. Polym. 2010, 82, 277–283. [Google Scholar] [CrossRef]
- Sánchez-González, L.; González-Martínez, C.; Chiralt, A.; Cháfer, M. Physical and Antimicrobial Properties of Chitosan–Tea Tree Essential Oil Composite Films. J. Food Eng. 2010, 98, 443–452. [Google Scholar] [CrossRef]
- Ozdemir, M.; Floros, J.D. Active Food Packaging Technologies. Crit. Rev. Food Sci. Nutr. 2010, 44, 185–193. [Google Scholar] [CrossRef]
- Siripatrawan, U.; Kaewklin, P. Fabrication and Characterization of Chitosan-Titanium Dioxide Nanocomposite Film as Ethylene Scavenging and Antimicrobial Active Food Packaging. Food Hydrocoll. 2018, 84, 125–134. [Google Scholar] [CrossRef]
- Wang, Y.C.; Mohan, C.O.; Guan, J.; Ravishankar, C.N.; Gunasekaran, S. Chitosan and Gold Nanoparticles-Based Thermal History Indicators and Frozen Indicators for Perishable and Temperature-Sensitive Products. Food Control 2018, 85, 186–193. [Google Scholar] [CrossRef]
- Singh, S.; Nwabor, O.F.; Syukri, D.M.; Voravuthikunchai, S.P. Chitosan-Poly(Vinyl Alcohol) Intelligent Films Fortified with Anthocyanins Isolated from Clitoria Ternatea and Carissa Carandas for Monitoring Beverage Freshness. Int. J. Biol. Macromol. 2021, 182, 1015–1025. [Google Scholar] [CrossRef]
- Wan, X.; He, Q.; Wang, X.; Liu, M.; Lin, S.; Shi, R.; Tian, J.; Chen, G. Water-Soluble Chitosan-Based Indicator Label Membrane and Its Response Behavior to Carbon Dioxide. Food Control 2021, 130, 108355. [Google Scholar] [CrossRef]
- Sharma, R.K.; Tahiliani, S.; Jain, N.; Priyadarshi, R.; Chhangani, S.; Purohit, S.D.; Joshi, P. Cynodon Dactylon Leaf Extract Assisted Green Synthesis of Silver Nanoparticles and Their Anti-Microbial Activity. Adv. Sci. Eng. Med. 2013, 5, 858–863. [Google Scholar] [CrossRef]
- Priyadarshi, R.; Negi, Y.S. Poly(Vinyl Pyrrolidone)-Mediated Synthesis of Silver Nanowires Decorated with Silver Nanospheres and Their Antimicrobial Activity. Bull. Mater. Sci. 2019, 42, 118. [Google Scholar] [CrossRef]
- Vimala, K.; Yallapu, M.M.; Varaprasad, K.; Reddy, N.N.; Ravindra, S.; Naidu, N.S.; Raju, K.M. Fabrication of Curcumin Encapsulated Chitosan-PVA Silver Nanocomposite Films for Improved Antimicrobial Activity. J. Biomater. Nanobiotechnol. 2011, 2, 55. [Google Scholar] [CrossRef]
- Cárdenas, G.; Díaz, V.J.; Meléndrez, M.F.; Cruzat, C.C.; García Cancino, A. Colloidal Cu Nanoparticles/Chitosan Composite Film Obtained by Microwave Heating for Food Package Applications. Polym. Bull. 2009, 62, 511–524. [Google Scholar] [CrossRef]
- Kim, K.M.; Son, J.H.; Kim, S.K.; Weller, C.L.; Hanna, M.A. Properties of Chitosan Films as a Function of PH and Solvent Type. J. Food Sci. 2006, 71, E119–E124. [Google Scholar] [CrossRef]
- Butler, B.L.; Vergano, P.J.; Testin, R.F.; Bunn, J.M.; Wiles, J.L. Mechanical and Barrier Properties of Edible Chitosan Films as Affected by Composition and Storage. J. Food Sci. 1996, 61, 953–956. [Google Scholar] [CrossRef]
- Van Den Broek, L.A.M.; Knoop, R.J.I.; Kappen, F.H.J.; Boeriu, C.G. Chitosan Films and Blends for Packaging Material. Carbohydr. Polym. 2015, 116, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Pelissari, F.M.; Yamashita, F.; Grossmann, M.V.E. Extrusion Parameters Related to Starch/Chitosan Active Films Properties. Int. J. Food Sci. Technol. 2011, 46, 702–710. [Google Scholar] [CrossRef]
- Holley, R.A.; Patel, D. Improvement in Shelf-Life and Safety of Perishable Foods by Plant Essential Oils and Smoke Antimicrobials. Food Microbiol. 2005, 22, 273–292. [Google Scholar] [CrossRef]
- Dorman, H.J.D.; Deans, S.G. Antimicrobial Agents from Plants: Antibacterial Activity of Plant Volatile Oils. J. Appl. Microbiol. 2000, 88, 308–316. [Google Scholar] [CrossRef] [PubMed]
- Canillac, N.; Mourey, A. Antibacterial Activity of the Essential Oil of Picea Excelsa on Listeria, Staphylococcus Aureus and Coliform Bacteria. Food Microbiol. 2001, 18, 261–268. [Google Scholar] [CrossRef]
- Kanatt, S.R.; Chander, R.; Sharma, A. Chitosan and Mint Mixture: A New Preservative for Meat and Meat Products. Food Chem. 2008, 107, 845–852. [Google Scholar] [CrossRef]
- Mari, M.; Bertolini, P.; Pratella, G.C. Non-Conventional Methods for the Control of Post-Harvest Pear Diseases. J. Appl. Microbiol. 2003, 94, 761–766. [Google Scholar] [CrossRef]
- Pessoa, L.M.; Morais, S.M.; Bevilaqua, C.M.L.; Luciano, J.H.S. Anthelmintic Activity of Essential Oil of Ocimum Gratissimum Linn. and Eugenol against Haemonchus Contortus. Vet. Parasitol. 2002, 109, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Abugoch, L.; Tapia, C.; Plasencia, D.; Pastor, A.; Castro-Mandujano, O.; López, L.; Escalona, V.H. Shelf-Life of Fresh Blueberries Coated with Quinoa Protein/Chitosan/Sunflower Oil Edible Film. J. Sci. Food Agric. 2016, 96, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Rambabu, K.; Bharath, G.; Banat, F.; Show, P.L.; Cocoletzi, H.H. Mango Leaf Extract Incorporated Chitosan Antioxidant Film for Active Food Packaging. Int. J. Biol. Macromol. 2019, 126, 1234–1243. [Google Scholar] [CrossRef]
- Imran, M.; Klouj, A.; Revol-Junelles, A.M.; Desobry, S. Controlled Release of Nisin from HPMC, Sodium Caseinate, Poly-Lactic Acid and Chitosan for Active Packaging Applications. J. Food Eng. 2014, 143, 178–185. [Google Scholar] [CrossRef]
- Zimet, P.; Mombrú, Á.W.; Mombrú, D.; Castro, A.; Villanueva, J.P.; Pardo, H.; Rufo, C. Physico-Chemical and Antilisterial Properties of Nisin-Incorporated Chitosan/Carboxymethyl Chitosan Films. Carbohydr. Polym. 2019, 219, 334–343. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Song, M.; Hourston, D.J. Novel Chitosan-Based Films Cross-Linked by Genipin with Improved Physical Properties. Biomacromolecules 2003, 5, 162–168. [Google Scholar] [CrossRef]
- Swain, S.K.; Kisku, S.K.; Sahoo, G. Preparation of Thermal Resistant Gas Barrier Chitosan Nanobiocomposites. Polym. Compos. 2014, 35, 2324–2328. [Google Scholar] [CrossRef]
- Yadav, M.; Behera, K.; Chang, Y.H.; Chiu, F.C. Cellulose Nanocrystal Reinforced Chitosan Based UV Barrier Composite Films for Sustainable Packaging. Polymers 2020, 12, 202. [Google Scholar] [CrossRef]
- Yang, R.; Li, H.; Huang, M.; Yang, H.; Li, A. A Review on Chitosan-Based Flocculants and Their Applications in Water Treatment. Water Res. 2016, 95, 59–89. [Google Scholar] [CrossRef]
- Otoni, C.G.; Avena-Bustillos, R.J.; Azeredo, H.M.C.; Lorevice, M.V.; Moura, M.R.; Mattoso, L.H.C.; McHugh, T.H. Recent Advances on Edible Films Based on Fruits and Vegetables—A Review. Compr. Rev. Food Sci. Food Saf. 2017, 16, 1151–1169. [Google Scholar] [CrossRef] [PubMed]
- Vargas, M.; Albors, A.; Chiralt, A.; González-Martínez, C. Quality of Cold-Stored Strawberries as Affected by Chitosan–Oleic Acid Edible Coatings. Postharvest Biol. Technol. 2006, 41, 164–171. [Google Scholar] [CrossRef]
- Ortiz-Duarte, G.; Pérez-Cabrera, L.E.; Artés-Hernández, F.; Martínez-Hernández, G.B. Ag-Chitosan Nanocomposites in Edible Coatings Affect the Quality of Fresh-Cut Melon. Postharvest Biol. Technol. 2019, 147, 174–184. [Google Scholar] [CrossRef]
- Ojagh, S.M.; Rezaei, M.; Razavi, S.H.; Hosseini, S.M.H. Development and Evaluation of a Novel Biodegradable Film Made from Chitosan and Cinnamon Essential Oil with Low Affinity toward Water. Food Chem. 2010, 122, 161–166. [Google Scholar] [CrossRef]
- Ojagh, S.M.; Rezaei, M.; Razavi, S.H.; Hosseini, S.M.H. Effect of Chitosan Coatings Enriched with Cinnamon Oil on the Quality of Refrigerated Rainbow Trout. Food Chem. 2010, 120, 193–198. [Google Scholar] [CrossRef]
- Priyadarshi, R.; Sauraj; Kumar, B.; Deeba, F.; Kulshreshtha, A.; Negi, Y.S. Chitosan Films Incorporated with Apricot (Prunus Armeniaca) Kernel Essential Oil as Active Food Packaging Material. Food Hydrocoll. 2018, 85, 158–166. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.W. Preparation of Carbohydrate-Based Functional Composite Films Incorporated with Curcumin. Food Hydrocoll. 2020, 98, 105302. [Google Scholar] [CrossRef]
- Yong, H.; Wang, X.; Bai, R.; Miao, Z.; Zhang, X.; Liu, J. Development of Antioxidant and Intelligent PH-Sensing Packaging Films by Incorporating Purple-Fleshed Sweet Potato Extract into Chitosan Matrix. Food Hydrocoll. 2019, 90, 216–224. [Google Scholar] [CrossRef]
- Casariego, A.; Souza, B.W.S.; Cerqueira, M.A.; Teixeira, J.A.; Cruz, L.; Díaz, R.; Vicente, A.A. Chitosan/Clay Films’ Properties as Affected by Biopolymer and Clay Micro/Nanoparticles’ Concentrations. Food Hydrocoll. 2009, 23, 1895–1902. [Google Scholar] [CrossRef]
- Rhim, J.W. Effect of Clay Contents on Mechanical and Water Vapor Barrier Properties of Agar-Based Nanocomposite Films. Carbohydr. Polym. 2011, 86, 691–699. [Google Scholar] [CrossRef]
- Cui, Z.; Beach, E.S.; Anastas, P.T. Modification of Chitosan Films with Environmentally Benign Reagents for Increased Water Resistance. Green Chem. Lett. Rev. 2011, 4, 35–40. [Google Scholar] [CrossRef]
- Mu, H.; Zhang, A.; Zhang, L.; Niu, H.; Duan, J. Inhibitory Effects of Chitosan in Combination with Antibiotics on Listeria Monocytogenes Biofilm. Food Control 2014, 38, 215–220. [Google Scholar] [CrossRef]
- Gomes, L.P.; Souza, H.K.S.; Campiña, J.M.; Andrade, C.T.; Silva, A.F.; Gonçalves, M.P.; Paschoalin, V.M.F. Edible Chitosan Films and Their Nanosized Counterparts Exhibit Antimicrobial Activity and Enhanced Mechanical and Barrier Properties. Molecules 2018, 24, 127. [Google Scholar] [CrossRef]
- Priyadarshi, R.; Negi, Y.S. Effect of Varying Filler Concentration on Zinc Oxide Nanoparticle Embedded Chitosan Films as Potential Food Packaging Material. J. Polym. Environ. 2017, 25, 1087–1098. [Google Scholar] [CrossRef]
- Ahmed, J.; Mulla, M.; Arfat, Y.A.; Thai, T.L.A. Mechanical, Thermal, Structural and Barrier Properties of Crab Shell Chitosan/Graphene Oxide Composite Films. Food Hydrocoll. 2017, 71, 141–148. [Google Scholar] [CrossRef]
- Sun, F.; Cha, H.R.; Bae, K.E.; Hong, S.; Kim, J.M.; Kim, S.H.; Lee, J.; Lee, D. Mechanical Properties of Multilayered Chitosan/CNT Nanocomposite Films. Mater. Sci. Eng. A 2011, 528, 6636–6641. [Google Scholar] [CrossRef]
- Shankar, S.; Rhim, J.W. Preparation of Sulfur Nanoparticle-Incorporated Antimicrobial Chitosan Films. Food Hydrocoll. 2018, 82, 116–123. [Google Scholar] [CrossRef]
- Barra, A.; Ferreira, N.M.; Martins, M.A.; Lazar, O.; Pantazi, A.; Jderu, A.A.; Neumayer, S.M.; Rodriguez, B.J.; Enăchescu, M.; Ferreira, P.; et al. Eco-Friendly Preparation of Electrically Conductive Chitosan-Reduced Graphene Oxide Flexible Bionanocomposites for Food Packaging and Biological Applications. Compos. Sci. Technol. 2019, 173, 53–60. [Google Scholar] [CrossRef]
- Perumal, S.; Atchudan, R.; Yoon, D.H.; Joo, J.; Cheong, I.W. Spherical Chitosan/Gelatin Hydrogel Particles for Removal of Multiple Heavy Metal Ions from Wastewater. Ind. Eng. Chem. Res. 2019, 58, 9900–9907. [Google Scholar] [CrossRef]
- Karimi, F.; Ayati, A.; Tanhaei, B.; Sanati, A.L.; Afshar, S.; Kardan, A.; Dabirifar, Z.; Karaman, C. Removal of Metal Ions Using a New Magnetic Chitosan Nano-Bio-Adsorbent; A Powerful Approach in Water Treatment. Environ. Res. 2022, 203, 111753. [Google Scholar] [CrossRef]
- Tang, S.; Yang, J.; Lin, L.; Peng, K.; Chen, Y.; Jin, S.; Yao, W. Construction of Physically Crosslinked Chitosan/Sodium Alginate/Calcium Ion Double-Network Hydrogel and Its Application to Heavy Metal Ions Removal. Chem. Eng. J. 2020, 393, 124728. [Google Scholar] [CrossRef]
- Surgutskaia, N.S.; Di Martino, A.; Zednik, J.; Ozaltin, K.; Lovecká, L.; Bergerová, E.D.; Kimmer, D.; Svoboda, J.; Sedlarik, V. Efficient Cu2+, Pb2+ and Ni2+ Ion Removal from Wastewater Using Electrospun DTPA-Modified Chitosan/Polyethylene Oxide Nanofibers. Sep. Purif. Technol. 2020, 247, 116914. [Google Scholar] [CrossRef]
- Hernández, R.B.; Yola, O.R.; Mercê, A.L.R. Chemical Equilibrium in the Complexation of First Transition Series Divalent Cations Cu2+, Mn2+ and Zn2+ with Chitosan. J. Braz. Chem. Soc. 2007, 18, 1388–1396. [Google Scholar] [CrossRef]
- Hussain, S.; Kamran, M.; Khan, S.A.; Shaheen, K.; Shah, Z.; Suo, H.; Khan, Q.; Shah, A.B.; Rehman, W.U.; Al-Ghamdi, Y.O.; et al. Adsorption, Kinetics and Thermodynamics Studies of Methyl Orange Dye Sequestration through Chitosan Composites Films. Int. J. Biol. Macromol. 2021, 168, 383–394. [Google Scholar] [CrossRef]
- Arumugam, T.K.; Krishnamoorthy, P.; Rajagopalan, N.R.; Nanthini, S.; Vasudevan, D. Removal of Malachite Green from Aqueous Solutions Using a Modified Chitosan Composite. Int. J. Biol. Macromol. 2019, 128, 655–664. [Google Scholar] [CrossRef]
- Chen, B.; Zhao, H.; Chen, S.; Long, F.; Huang, B.; Yang, B.; Pan, X. A Magnetically Recyclable Chitosan Composite Adsorbent Functionalized with EDTA for Simultaneous Capture of Anionic Dye and Heavy Metals in Complex Wastewater. Chem. Eng. J. 2019, 356, 69–80. [Google Scholar] [CrossRef]
- Le, T.T.N.; Le, V.T.; Dao, M.U.; Nguyen, Q.V.; Vu, T.T.; Nguyen, M.H.; Tran, D.L.; Le, H.S. Preparation of Magnetic Graphene Oxide/Chitosan Composite Beads for Effective Removal of Heavy Metals and Dyes from Aqueous Solutions. Chem. Eng. Commun. 2019, 206, 1337–1352. [Google Scholar] [CrossRef]
- da Rosa Schio, R.; da Rosa, B.C.; Gonçalves, J.O.; Pinto, L.A.A.; Mallmann, E.S.; Dotto, G.L. Synthesis of a Bio–Based Polyurethane/Chitosan Composite Foam Using Ricinoleic Acid for the Adsorption of Food Red 17 Dye. Int. J. Biol. Macromol. 2019, 121, 373–380. [Google Scholar] [CrossRef]
- Mostafa, M.; Bin Jumah, M.N.; Othman, S.I.; Alruhaimi, R.S.; Salama, Y.F.; Allam, A.A.; Abukhadra, M.R. Effective Removal of Different Species of Organophosphorus Pesticides (Acephate, Omthosate, and Methyl Parathion) Using Chitosan/Zeolite-A as Multifunctional Adsorbent. Environ. Technol. Innov. 2021, 24, 101875. [Google Scholar] [CrossRef]
- Motaghi, H.; Arabkhani, P.; Parvinnia, M.; Asfaram, A. Simultaneous Adsorption of Cobalt Ions, Azo Dye, and Imidacloprid Pesticide on the Magnetic Chitosan/Activated Carbon@UiO-66 Bio-Nanocomposite: Optimization, Mechanisms, Regeneration, and Application. Sep. Purif. Technol. 2022, 284, 120258. [Google Scholar] [CrossRef]
- Venkatachalapathy, R.; Packirisamy, A.S.B.; Indira Ramachandran, A.C.; Udhyasooriyan, L.P.; Peter, M.J.; Senthilnathan, K.; Basheer, V.A.; Muthusamy, S. Assessing the Effect of Chitosan on Pesticide Removal in Grape Juice during Clarification by Gas Chromatography with Tandem Mass Spectrometry. Process Biochem. 2020, 94, 305–312. [Google Scholar] [CrossRef]
- Ghimici, L.; Dinu, I.A. Removal of Some Commercial Pesticides from Aqueous Dispersions Using as Flocculant a Thymine-Containing Chitosan Derivative. Sep. Purif. Technol. 2019, 209, 698–706. [Google Scholar] [CrossRef]
- Tzereme, A.; Christodoulou, E.; Kyzas, G.Z.; Kostoglou, M.; Bikiaris, D.N.; Lambropoulou, D.A. Chitosan Grafted Adsorbents for Diclofenac Pharmaceutical Compound Removal from Single-Component Aqueous Solutions and Mixtures. Polymers 2019, 11, 497. [Google Scholar] [CrossRef] [PubMed]
- Mahmoodi, H.; Fattahi, M.; Motevassel, M. Graphene Oxide–Chitosan Hydrogel for Adsorptive Removal of Diclofenac from Aqueous Solution: Preparation, Characterization, Kinetic and Thermodynamic Modelling. RSC Adv. 2021, 11, 36289–36304. [Google Scholar] [CrossRef] [PubMed]
- Chan, K.; Morikawa, K.; Shibata, N.; Zinchenko, A. Adsorptive Removal of Heavy Metal Ions, Organic Dyes, and Pharmaceuticals by Dna–Chitosan Hydrogels. Gels 2021, 7, 112. [Google Scholar] [CrossRef]
- Mojiri, A.; Kazeroon, R.A.; Gholami, A. Cross-Linked Magnetic Chitosan/Activated Biochar for Removal of Emerging Micropollutants from Water: Optimization by the Artificial Neural Network. Water 2019, 11, 551. [Google Scholar] [CrossRef]
- Vo, P.T.; Nguyen, H.T.; Trinh, H.T.; Nguyen, V.M.; Le, A.-T.; Tran, H.Q.; Nguyen, T.T.T. The Nitrogen Slow-Release Fertilizer Based on Urea Incorporating Chitosan and Poly(Vinyl Alcohol) Blend. Environ. Technol. Innov. 2021, 22, 101528. [Google Scholar] [CrossRef]
- Kondal, R.; Kalia, A.; Krejcar, O.; Kuca, K.; Sharma, S.P.; Luthra, K.; Dheri, G.S.; Vikal, Y.; Taggar, M.S.; Abd-Elsalam, K.A.; et al. Chitosan-Urea Nanocomposite for Improved Fertilizer Applications: The Effect on the Soil Enzymatic Activities and Microflora Dynamics in N Cycle of Potatoes (Solanum tuberosum L.). Polymers 2021, 13, 2887. [Google Scholar] [CrossRef]
- Sathisaran, I.; Balasubramanian, M. Physical Characterization of Chitosan/Gelatin-Alginate Composite Beads for Controlled Release of Urea. Heliyon 2020, 6, e05495. [Google Scholar] [CrossRef]
- Dou, Z.; Bini Farias, M.V.; Chen, W.; He, D.; Hu, Y.; Xie, X. Highly Degradable Chitosan-Montmorillonite (MMT) Nano-Composite Hydrogel for Controlled Fertilizer Release. Front. Environ. Sci. Eng. 2023, 17, 1–11. [Google Scholar] [CrossRef]
- Kubavat, D.; Trivedi, K.; Vaghela, P.; Prasad, K.; Vijay Anand, G.K.; Trivedi, H.; Patidar, R.; Chaudhari, J.; Andhariya, B.; Ghosh, A. Characterization of a Chitosan-Based Sustained Release Nanofertilizer Formulation Used as a Soil Conditioner While Simultaneously Improving Biomass Production of Zea mays L. Land Degrad. Dev. 2020, 31, 2734–2746. [Google Scholar] [CrossRef]
- Messa, L.L.; Souza, C.F.; Faez, R. Spray-Dried Potassium Nitrate-Containing Chitosan/Montmorillonite Microparticles as Potential Enhanced Efficiency Fertilizer. Polym. Test. 2020, 81, 106196. [Google Scholar] [CrossRef]
- Chawla, P.; Srivastava, A.; Tripathi, M. Performance of Chitosan Based Polymer Electrolyte for Natural Dye Sensitized Solar Cell. Environ. Prog. Sustain. Energy 2019, 38, 630–634. [Google Scholar] [CrossRef]
- Rahman, N.A.; Hanifah, S.A.; Mobarak, N.N.; Ahmad, A.; Ludin, N.A.; Bella, F.; Su’ait, M.S. Chitosan as a Paradigm for Biopolymer Electrolytes in Solid-State Dye-Sensitised Solar Cells. Polymer 2021, 230, 124092. [Google Scholar] [CrossRef]
- Areerob, Y.; Hamontree, C.; Sricharoen, P.; Limchoowong, N.; Laksee, S.; Oh, W.C.; Pattarith, K. Novel Gamma-Irradiated Chitosan-Doped Reduced Graphene-CuInS 2 Composites as Counter Electrodes for Dye-Sensitized Solar Cells. RSC Adv. 2022, 12, 15427–15434. [Google Scholar] [CrossRef]
- Zulkifli, A.M.; Aqilah Mat Said, N.I.; Aziz, S.B.; Ali Dannoun, E.M.; Hisham, S.; Shah, S.; Bakar, A.A.; Zainal, Z.H.; Tajuddin, H.A.; Hadi, J.M.; et al. Characteristics of Dye-Sensitized Solar Cell Assembled from Modified Chitosan-Based Gel Polymer Electrolytes Incorporated with Potassium Iodide. Molecules 2020, 25, 4115. [Google Scholar] [CrossRef]
- Praveen, E.; John Peter, I.; Muthu Kumar, A.; Ramachandran, K.; Jayakumar, K. Performance of ZnO/ZnS Nanocomposite Based Dye-Sensitized Solar Cell with Chitosan-Polymer Electrolyte. Mater. Today Proc. 2021, 35, 27–30. [Google Scholar] [CrossRef]
- Zheng, S.; Zhang, J.; Deng, H.; Du, Y.; Shi, X. Chitin Derived Nitrogen-Doped Porous Carbons with Ultrahigh Specific Surface Area and Tailored Hierarchical Porosity for High Performance Supercapacitors. J. Bioresour. Bioprod. 2021, 6, 142–151. [Google Scholar] [CrossRef]
- Wei, L.; Deng, W.; Li, S.; Wu, Z.; Cai, J.; Luo, J. Sandwich-like Chitosan Porous Carbon Spheres/MXene Composite with High Specific Capacitance and Rate Performance for Supercapacitors. J. Bioresour. Bioprod. 2022, 7, 63–72. [Google Scholar] [CrossRef]
- Robbins, N.E.; Dinneny, J.R. Growth Is Required for Perception of Water Availability to Pattern Root Branches in Plants. Proc. Natl. Acad. Sci. USA 2018, 115, E822–E831. [Google Scholar] [CrossRef]
- Application of Soil Physics in Environmental Analyses. Appl. Soil Phys. Environ. Anal. 2014, 1, 307–345. [CrossRef]
- Rockström, J.; Barron, J. Water Productivity in Rainfed Systems: Overview of Challenges and Analysis of Opportunities in Water Scarcity Prone Savannahs. Irrig. Sci. 2007, 25, 299–311. [Google Scholar] [CrossRef]
- Farouk, S.; Amany, A. Improving Growth and Yield of Cowpea by Foliar Application of Chitosan under Water Stress. Egypt. J. Biol. 2012, 14, 14–26. [Google Scholar] [CrossRef]
- Abu-Muriefah, S.S. Effect of Chitosan on Common Bean (Phaseolus vulgaris L.) Plants Grown under Water Stress Conditions. Int. Res. J. Agric. Sci. Soil Sci. 2013, 3, 192–199. [Google Scholar]
- Silveira, N.M.; Seabra, A.B.; Marcos, F.C.C.; Pelegrino, M.T.; Machado, E.C.; Ribeiro, R.V. Encapsulation of S-Nitrosoglutathione into Chitosan Nanoparticles Improves Drought Tolerance of Sugarcane Plants. Nitric Oxide-Biol. Chem. 2019, 84, 38–44. [Google Scholar] [CrossRef]
- Almeida, L.G.; Magalhães, P.C.; Karam, D.; da Silva, E.M.; Alvarenga, A.A. Chitosan Application in the Induction of Water Deficit Tolerance in Maize Plants. Acta Sci.-Agron. 2020, 42, 1–10. [Google Scholar] [CrossRef]
- Emam, M.M.; Khattab, H.E.; Helal, N.M.; Deraz, A.E. Effect of Selenium and Silicon on Yield Quality of Rice Plant Grown under Drought Stress. Aust. J. Crop Sci. 2014, 8, 596–605. [Google Scholar]
- Negm, N.A.; Hefni, H.H.H.; Abd-Elaal, A.A.A.; Badr, E.A.; Abou Kana, M.T.H. Advancement on Modification of Chitosan Biopolymer and Its Potential Applications. Int. J. Biol. Macromol. 2020, 152, 681–702. [Google Scholar] [CrossRef]
- Górnik, K.; Grzesik, M.; Romanowska-Duda, B. The Effect of Chitosan on Rooting of Grapevine Cuttings and on Subsequent Plant Growth under Drought and Temperature Stress. J. Fruit Ornam. Plant Res. 2008, 16, 333–343. [Google Scholar]
- Malekpoor, F.; Pirbalouti, A.G.; Salimi, A. Effect of Foliar Application of Chitosan on Morphological and Physiological Characteristics of Basil under Reduced Irrigation. Res. Crop. 2016, 17, 354–359. [Google Scholar] [CrossRef]
- Dzung, N.A.; Khanh, V.T.P.; Dzung, T.T. Research on Impact of Chitosan Oligomers on Biophysical Characteristics, Growth, Development and Drought Resistance of Coffee. Carbohydr. Polym. 2011, 84, 751–755. [Google Scholar] [CrossRef]
- Li, Z.; Zhang, Y.; Zhang, X.; Merewitz, E.; Peng, Y.; Ma, X.; Huang, L.; Yan, Y. Metabolic Pathways Regulated by Chitosan Contributing to Drought Resistance in White Clover. J. Proteome Res. 2017, 16, 3039–3052. [Google Scholar] [CrossRef] [PubMed]
- Emami Bistgani, Z.; Siadat, S.A.; Bakhshandeh, A.; Ghasemi Pirbalouti, A.; Hashemi, M. Interactive Effects of Drought Stress and Chitosan Application on Physiological Characteristics and Essential Oil Yield of Thymus Daenensis Celak. Crop J. 2017, 5, 407–415. [Google Scholar] [CrossRef]
- Mahdavi, B.; Sanavy, S.A.M.M.; Aghaalikhani, M.; Sharifi, M.; Dolatabadian, A. Chitosan Improves Osmotic Potential Tolerance in Safflower (Carthamus tinctorius L.) Seedlings. J. Crop Improv. 2011, 25, 728–741. [Google Scholar] [CrossRef]
- Mahdavi, B.; Rahimi, A. Seed Priming with Chitosan Improves the Germination and Growth Performance of Ajowan {Carum Copticum) under Salt Stress. Eurasian J. Biosci. 2013, 7, 69–76. [Google Scholar] [CrossRef]
- Martínez González, L.; Reyes Guerrero, Y.; Falcón Rodríguez, A.; Núñez Vázquez, M. Effect of Seed Treatment with Chitosan on the Growth of Rice (Oryza sativa L.) Seedlings Cv. INCA LP-5 in Saline Medium. Cultiv. Trop. 2015, 36, 143–150. [Google Scholar]
- Ray, S.R.; Bhuiyan, M.J.H.; Hossain, M.A.; Hasan, A.K.; Sharmin, S. Chitosan Ameliorates Growth and Biochemical Attributes in Mungbean Varieties under Saline Condition. Res. Agric. Livest. Fish. 2016, 3, 45–51. [Google Scholar] [CrossRef]
- Al-Tawaha, A.R.; Turk, M.A.; Al-Tawaha, A.R.M.; Alu’datt, M.H.; Wedyan, M.; Al-Ramamneh, E.; Hoang, A.T. Using Chitosan to Improve Growth of Maize Cultivars under Salinity Conditions. Bulg. J. Agric. Sci 2018, 24, 437–442. [Google Scholar]
- Ma, L.; Li, Y.; Yu, C.; Wang, Y.; Li, X.; Li, N.; Chen, Q.; Bu, N. Alleviation of Exogenous Oligochitosan on Wheat Seedlings Growth under Salt Stress. Protoplasma 2012, 249, 393–399. [Google Scholar] [CrossRef]
- Zeng, D.; Luo, X. Physiological Effects of Chitosan Coating on Wheat Growth and Activities of Protective Enzyme with Drought Tolerance. Open J. Soil Sci. 2012, 2, 282–288. [Google Scholar] [CrossRef]
- Jiao, Z.; Li, Y.; Li, J.; Xu, X.; Li, H.; Lu, D.; Wang, J. Effects of Exogenous Chitosan on Physiological Characteristics of Potato Seedlings Under Drought Stress and Rehydration. Potato Res. 2012, 55, 293–301. [Google Scholar] [CrossRef]
- Yang, F.; Hu, J.; Li, J.; Wu, X.; Qian, Y. Chitosan Enhances Leaf Membrane Stability and Antioxidant Enzyme Activities in Apple Seedlings under Drought Stress. Plant Growth Regul. 2009, 58, 131–136. [Google Scholar] [CrossRef]
- Hidangmayum, A.; Dwivedi, P.; Katiyar, D.; Hemantaranjan, A. Application of Chitosan on Plant Responses with Special Reference to Abiotic Stress. Physiol. Mol. Biol. Plants 2019, 25, 313–326. [Google Scholar] [CrossRef] [PubMed]
- Kong, W.; Liu, F.; Zhang, C.; Zhang, J.; Feng, H. Non-Destructive Determination of Malondialdehyde (MDA) Distribution in Oilseed Rape Leaves by Laboratory Scale NIR Hyperspectral Imaging. Sci. Rep. 2016, 6, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, H.C.; Gomes, B.C.R.; Pelegrino, M.T.; Seabra, A.B. Nitric Oxide-Releasing Chitosan Nanoparticles Alleviate the Effects of Salt Stress in Maize Plants. Nitric Oxide 2016, 61, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Sen, S.K.; Chouhan, D.; Das, D.; Ghosh, R.; Mandal, P. Improvisation of Salinity Stress Response in Mung Bean through Solid Matrix Priming with Normal and Nano-Sized Chitosan. Int. J. Biol. Macromol. 2020, 145, 108–123. [Google Scholar] [CrossRef] [PubMed]
- Zayed, M.M.; Elkafafi, S.H.; Zedan, A.M.G.; Dawoud, S.F.M. Effect of Nano Chitosan on Growth, Physiological and Biochemical Parameters of Phaseolus Vulgaris under Salt Stress. J. Plant Prod. 2017, 8, 577–585. [Google Scholar] [CrossRef]
- Hernández-Hernández, H.; Juárez-Maldonado, A.; Benavides-Mendoza, A.; Ortega-Ortiz, H.; Cadenas-Pliego, G.; Sánchez-Aspeytia, D.; González-Morales, S. Chitosan-PVA and Copper Nanoparticles Improve Growth and Overexpress the SOD and JA Genes in Tomato Plants under Salt Stress. Agronomy 2018, 8, 175. [Google Scholar] [CrossRef]
- McKersie, B.D.; Leshem, Y.Y. Stress and Stress Coping in Cultivated Plants. Stress Stress Coping Cultiv. Stress Stress Coping Cultiv. Plants 1994. [Google Scholar] [CrossRef]
- Janmohammadi, M.; Mostafavi, H.; Kazemi, H.; Mahdavinia, G.-R.; Sabaghnia, N. Effect Of Chitosan Application On The Performance Of Lentil Genotypes Under Rainfed Conditions. Acta Technol. Agric. 2014, 17, 86–90. [Google Scholar] [CrossRef]
- Khordadi Varamin, J.; Fanoodi, F.; Sinaki, J.M.; Rezvan, S.; Damavandi, A. Physiological Response of Sesame (Sesamum indicum L.) to Application of Chitosan and Magnesium-Nano Fertilizers under Irrigation Cut-off in a Sustainable Agriculture System. Iran. J. Plant Physiol. 2018, 9, 2629–2639. [Google Scholar] [CrossRef]
- Guan, Y.J.; Hu, J.; Wang, X.J.; Shao, C.X. Seed Priming with Chitosan Improves Maize Germination and Seedling Growth in Relation to Physiological Changes under Low Temperature Stress. J. Zhejiang Univ. Sci. B 2009, 10, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Priyaadharshini, M.; Sritharan, N.; Senthil, A.; Marimuthu, S. Physiological Studies on Effect of Chitosan Nanoemulsion in Pearl Millet under Drought Condition. J. Pharmacogn. Phytochem. 2019, 8, 3304–3307. [Google Scholar]
- Kamari, A.; Pulford, I.D.; Hargreaves, J.S.J. Binding of Heavy Metal Contaminants onto Chitosans–An Evaluation for Remediation of Metal Contaminated Soil and Water. J. Environ. Manag. 2011, 92, 2675–2682. [Google Scholar] [CrossRef] [PubMed]
- Kamari, A.; Pulford, I.D.; Hargreaves, J.S.J. Metal accumulation in lolium perenne and brassica napus as affected by application of chitosans. Int. J. Phytoremediation 2012, 14, 894–907. [Google Scholar] [CrossRef]
- Vasconcelos, M.W. Chitosan and Chitooligosaccharide Utilization in Phytoremediation and Biofortification Programs: Current Knowledge and Future Perspectives. Front. Plant Sci. 2014, 5, 1–4. [Google Scholar] [CrossRef]
- Zong, H.; Li, K.; Liu, S.; Song, L.; Xing, R.; Chen, X.; Li, P. Improvement in Cadmium Tolerance of Edible Rape (Brassica rapa L.) with Exogenous Application of Chitooligosaccharide. Chemosphere 2017, 181, 92–100. [Google Scholar] [CrossRef]
- Kazemi, M.S.; Mohammadi, Z.; Amini, M.; Yousefi, M.; Tarighi, P.; Eftekhari, S.; Rafiee Tehrani, M. Thiolated Chitosan-Lauric Acid as a New Chitosan Derivative: Synthesis, Characterization and Cytotoxicity. Int. J. Biol. Macromol. 2019, 136, 823–830. [Google Scholar] [CrossRef]
- Piri-Gharaghie, T.; Beiranvand, S.; Riahi, A.; Shirin, N.J.; Badmasti, F.; Mirzaie, A.; Elahianfar, Y.; Ghahari, S.; Ghahari, S.; Pasban, K.; et al. Fabrication and Characterization of Thymol-Loaded Chitosan Nanogels: Improved Antibacterial and Anti-Biofilm Activities with Negligible Cytotoxicity. Chem. Biodivers. 2022, 19, e202100426. [Google Scholar] [CrossRef]
- Asghar, M.A.; Yousuf, R.I.; Shoaib, M.H.; Asghar, M.A. Antibacterial, Anticoagulant and Cytotoxic Evaluation of Biocompatible Nanocomposite of Chitosan Loaded Green Synthesized Bioinspired Silver Nanoparticles. Int. J. Biol. Macromol. 2020, 160, 934–943. [Google Scholar] [CrossRef]
- Frigaard, J.; Jensen, J.L.; Galtung, H.K.; Hiorth, M. The Potential of Chitosan in Nanomedicine: An Overview of the Cytotoxicity of Chitosan Based Nanoparticles. Front. Pharmacol. 2022, 13, 1492. [Google Scholar] [CrossRef] [PubMed]
- Dehghan, S.; Kheiri, M.T.; Tabatabaiean, M.; Darzi, S.; Tafaghodi, M. Dry-Powder Form of Chitosan Nanospheres Containing Influenza Virus and Adjuvants for Nasal Immunization. Arch. Pharm. Res. 2013, 36, 981–992. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.; Jo, D.H.; Anower, A.K.M.M.; Islam, S.M.S.; Sohn, S. Chitosan as an Immunomodulating Adjuvant on T-Cells and Antigen-Presenting Cells in Herpes Simplex Virus Type 1 Infection. Mediators Inflamm. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Morin-Crini, N.; Lichtfouse, E.; Torri, G.; Crini, G. Fundamentals and Applications of Chitosan; 2019; ISBN 9783030165383. [Google Scholar]
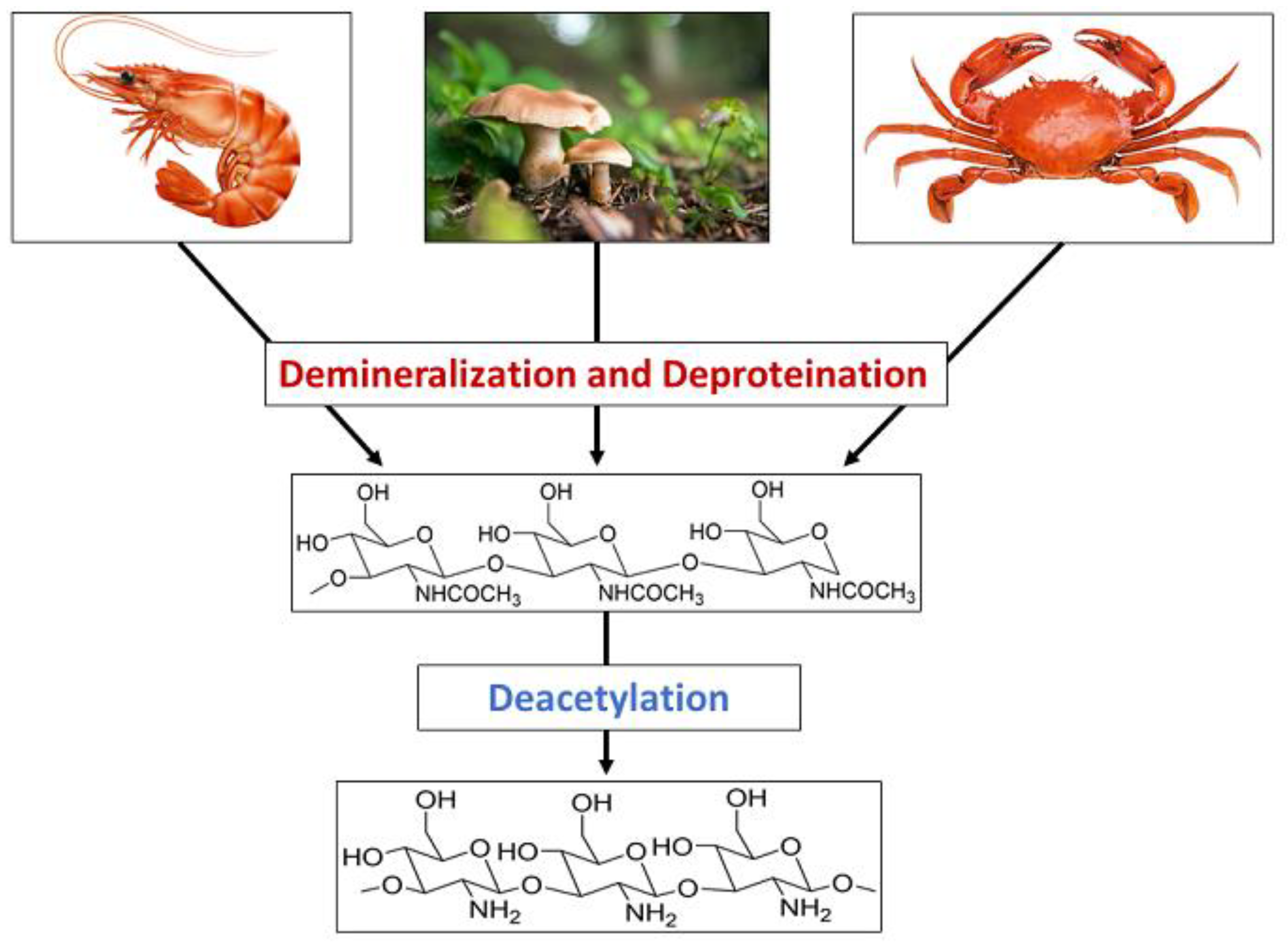
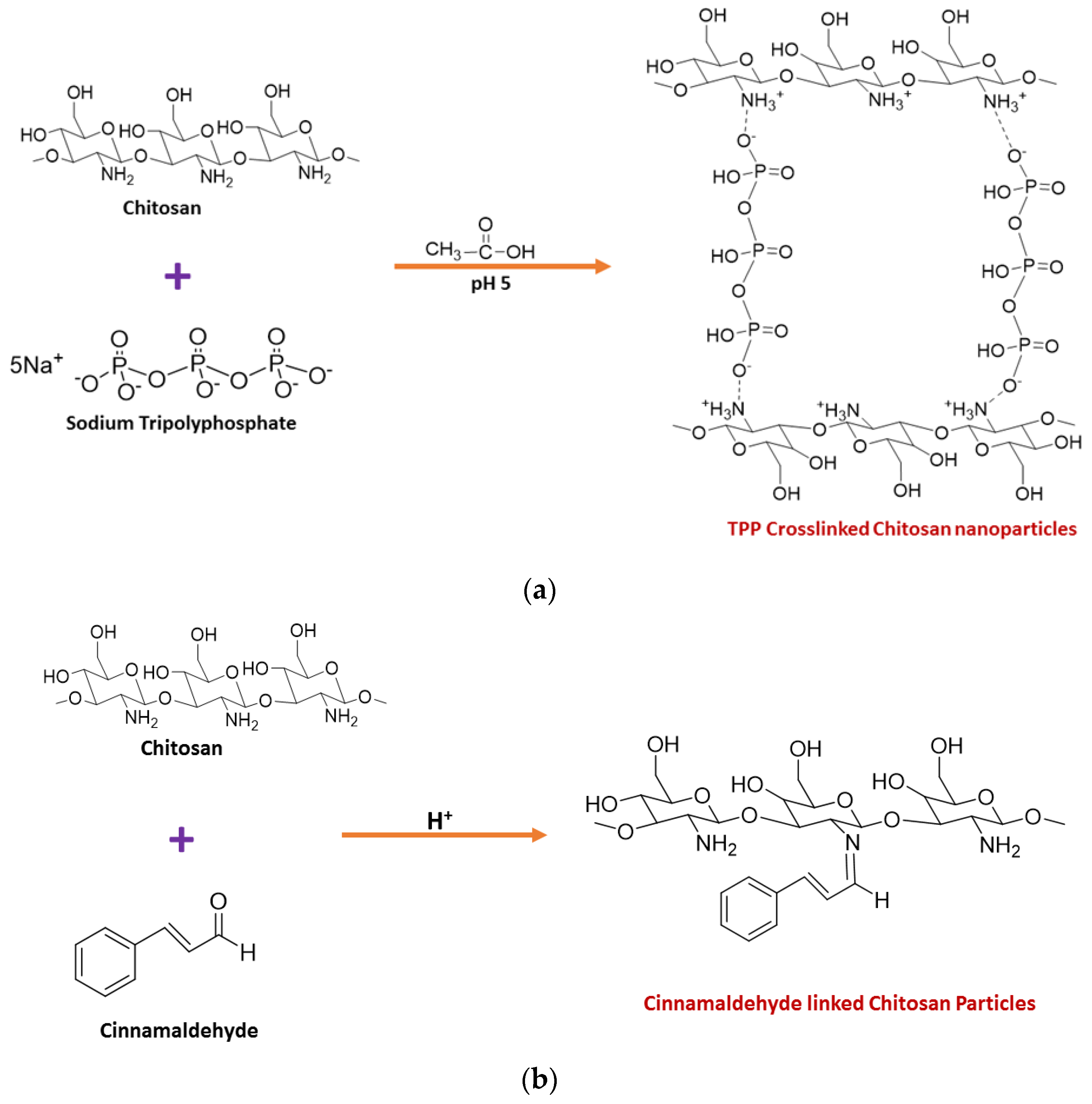
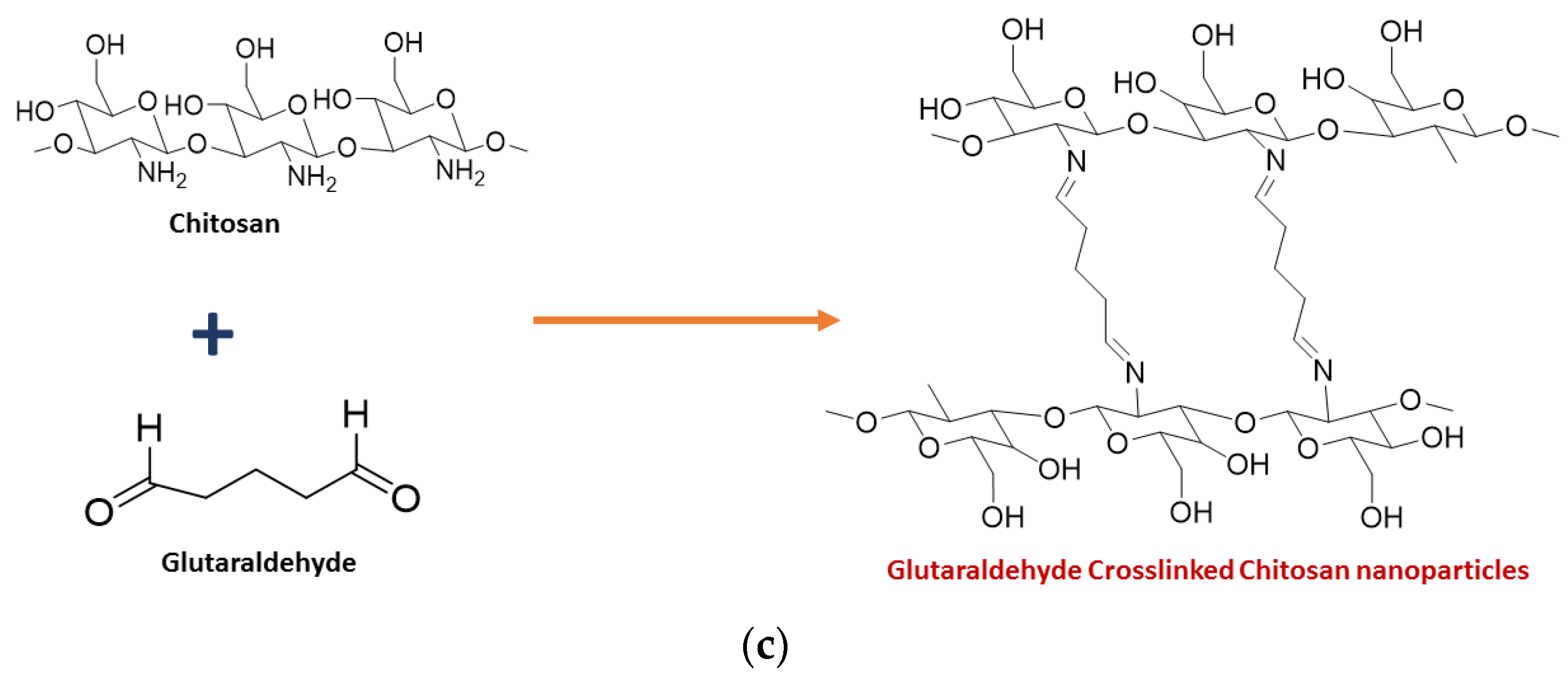
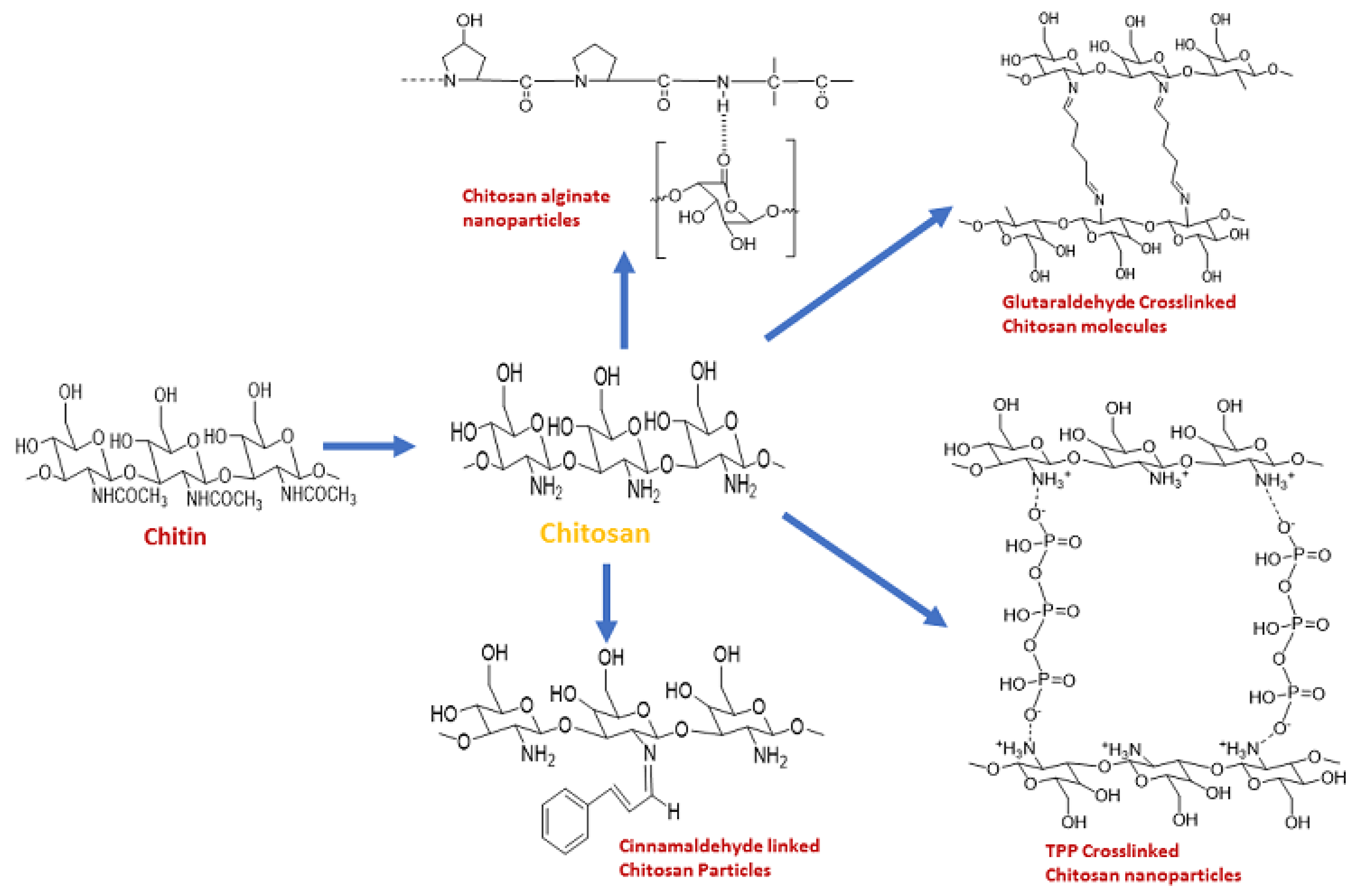
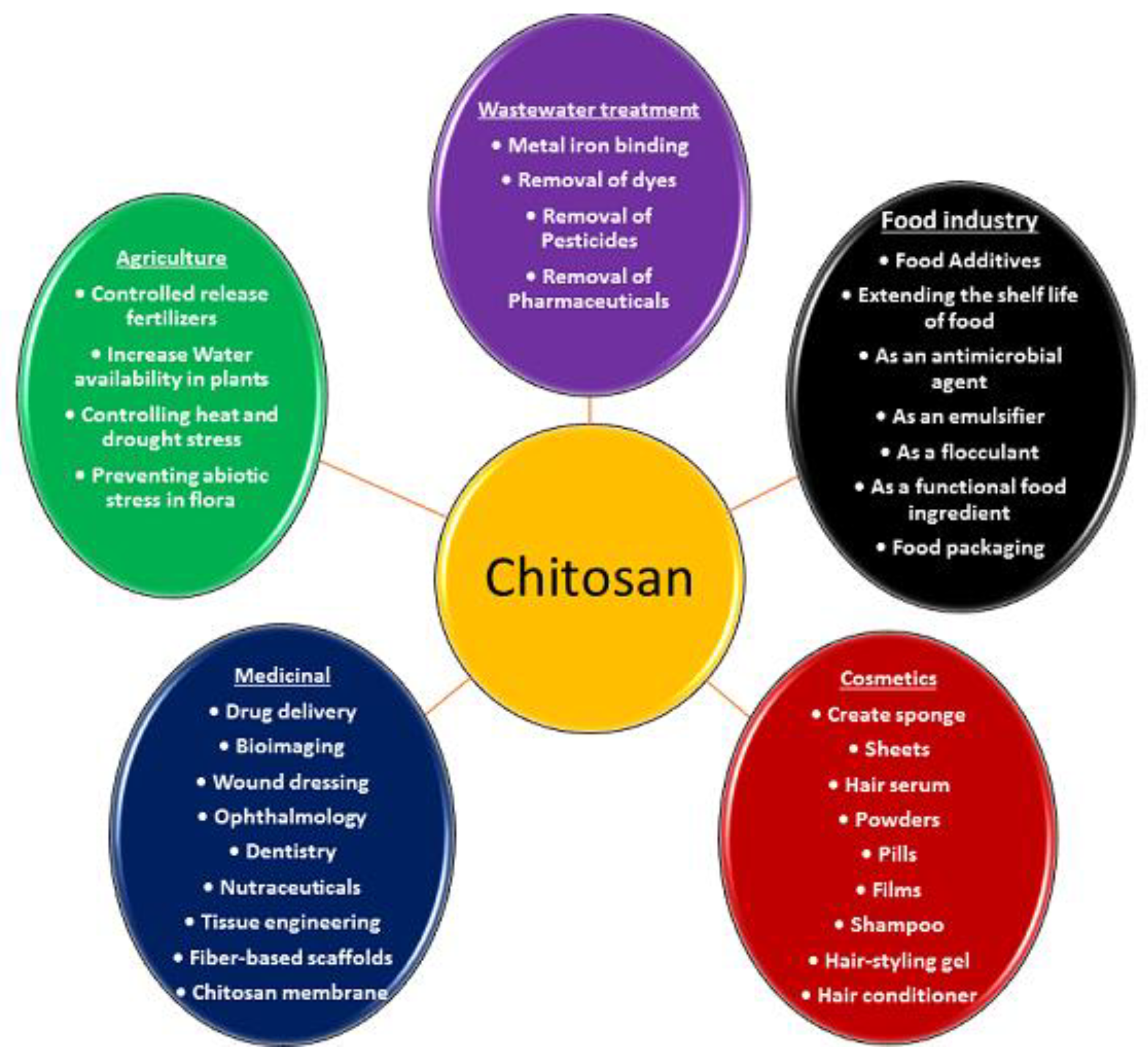
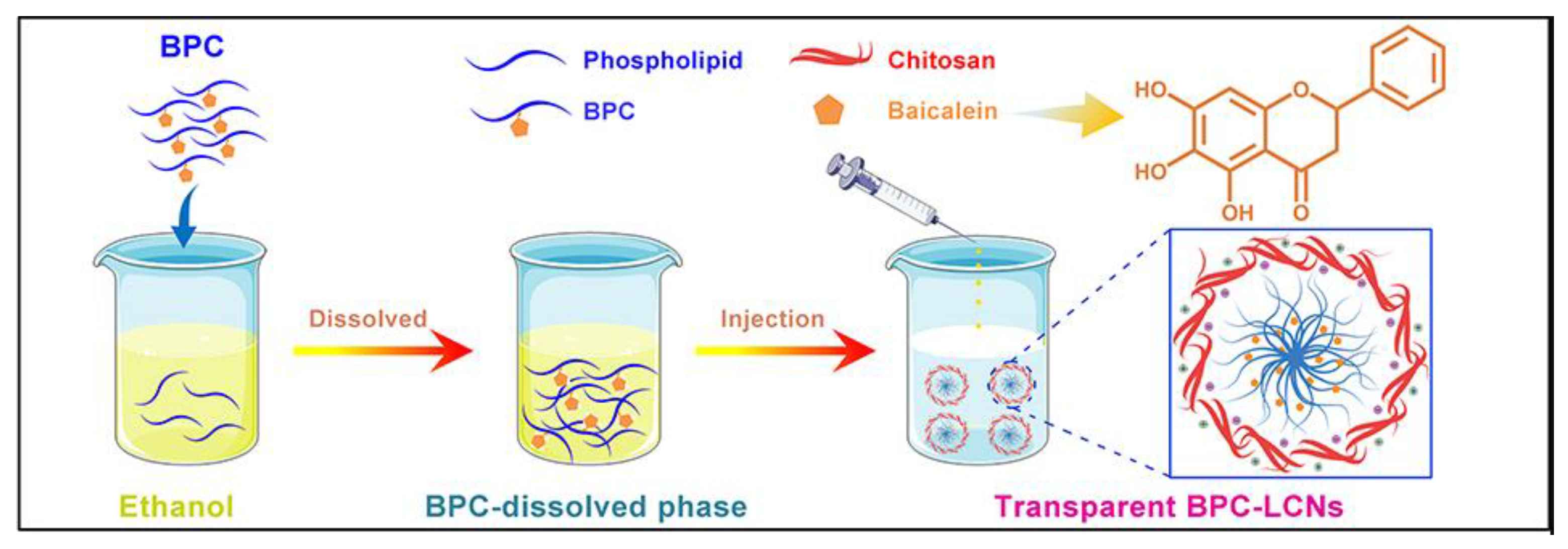

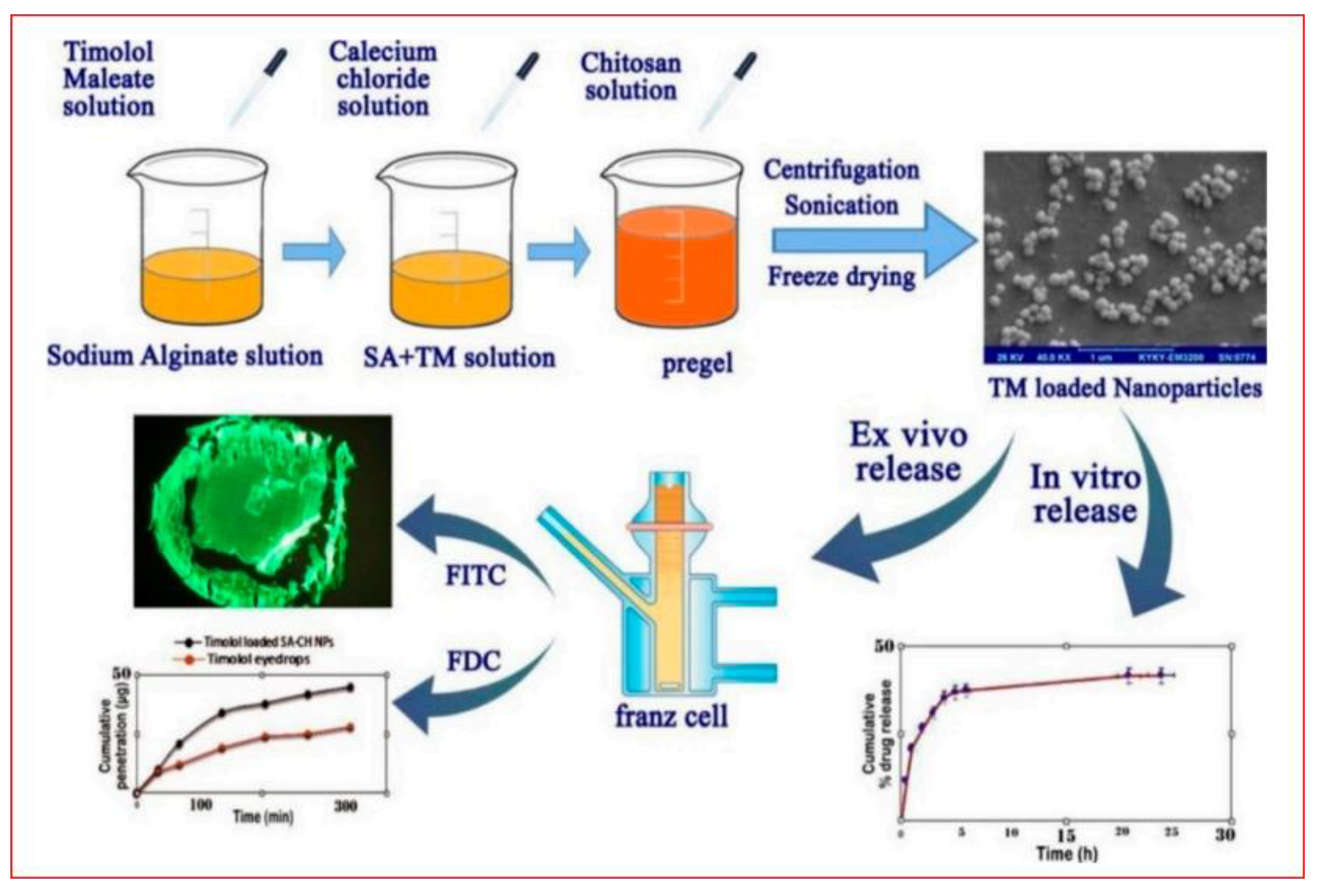

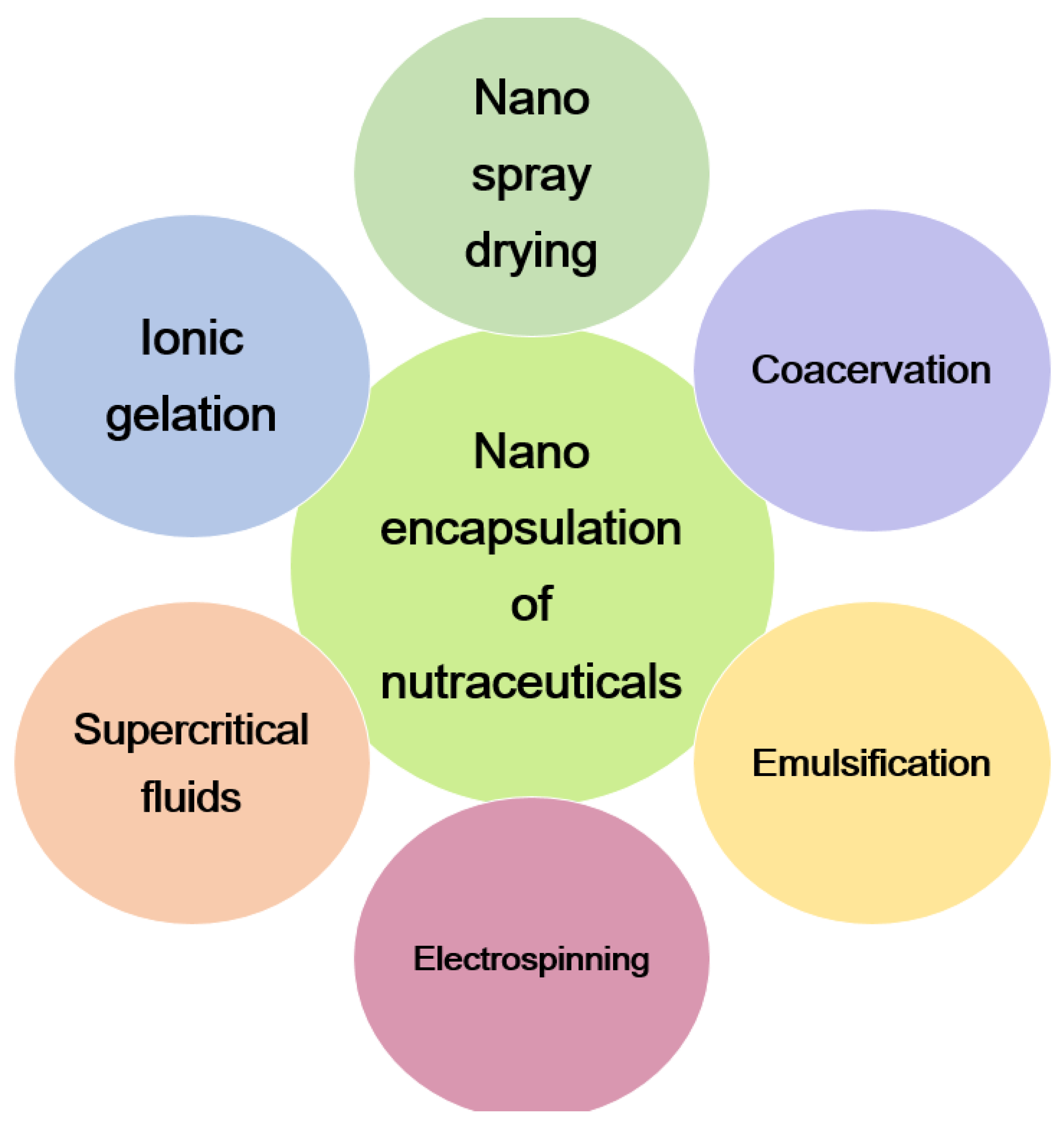
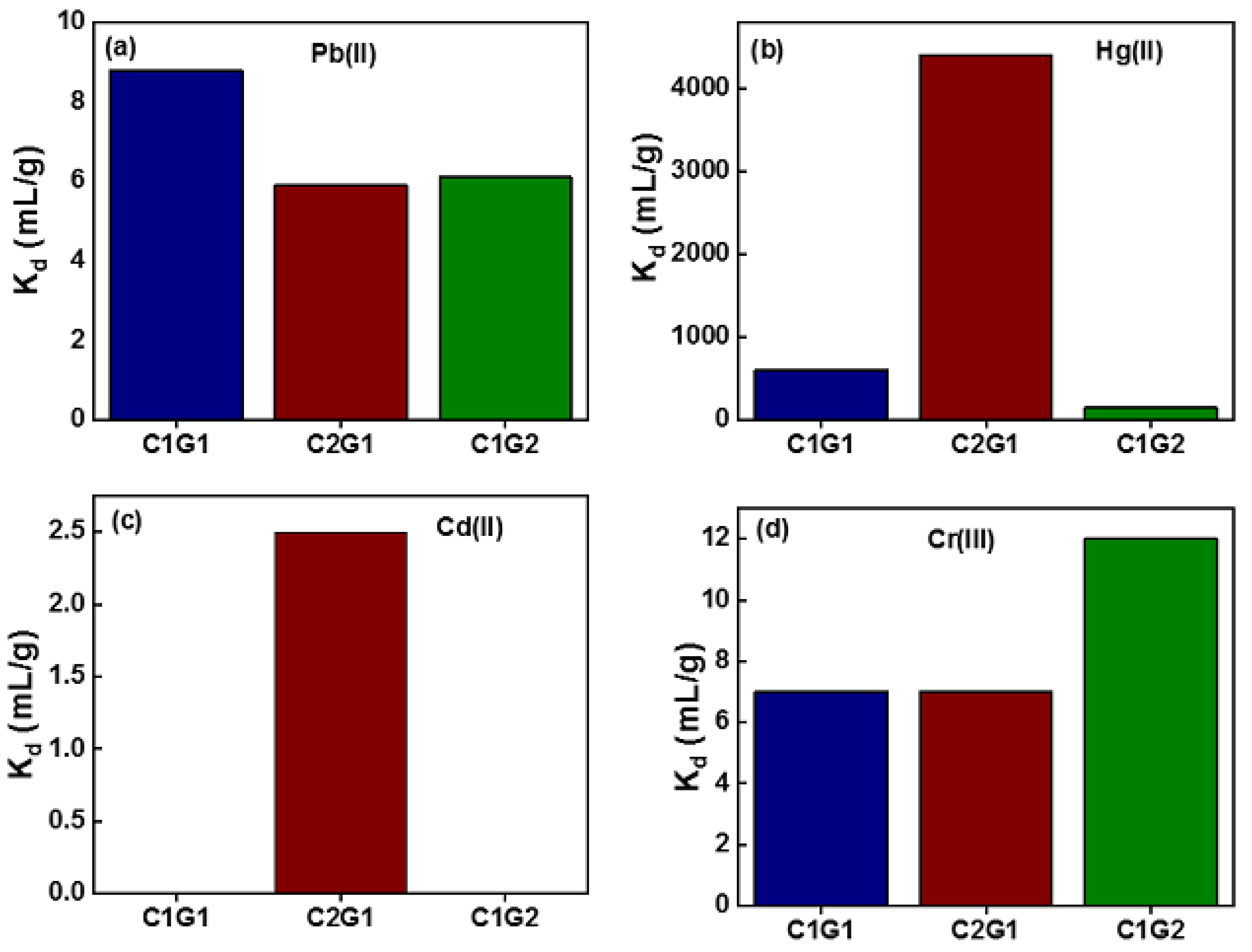
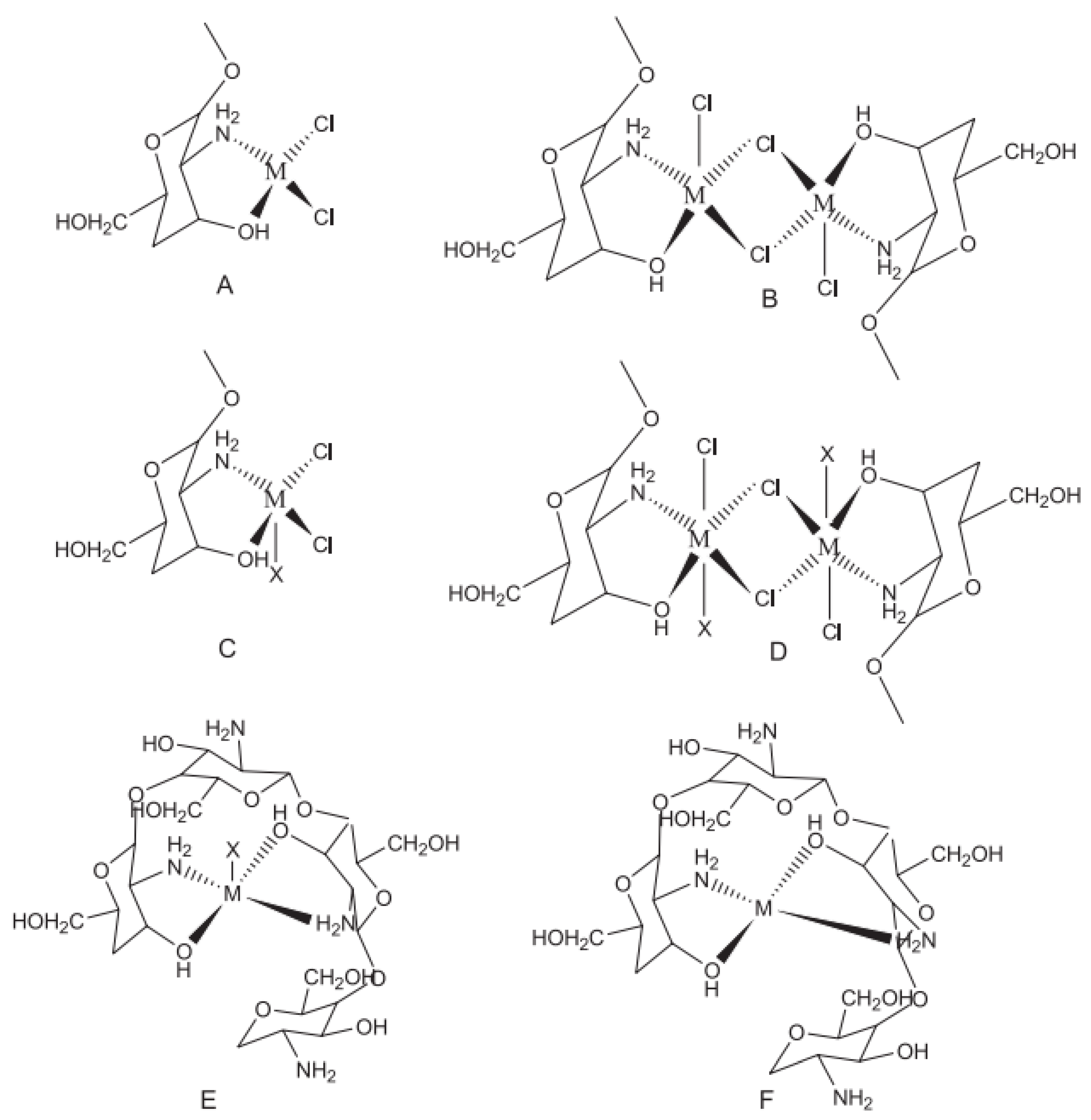
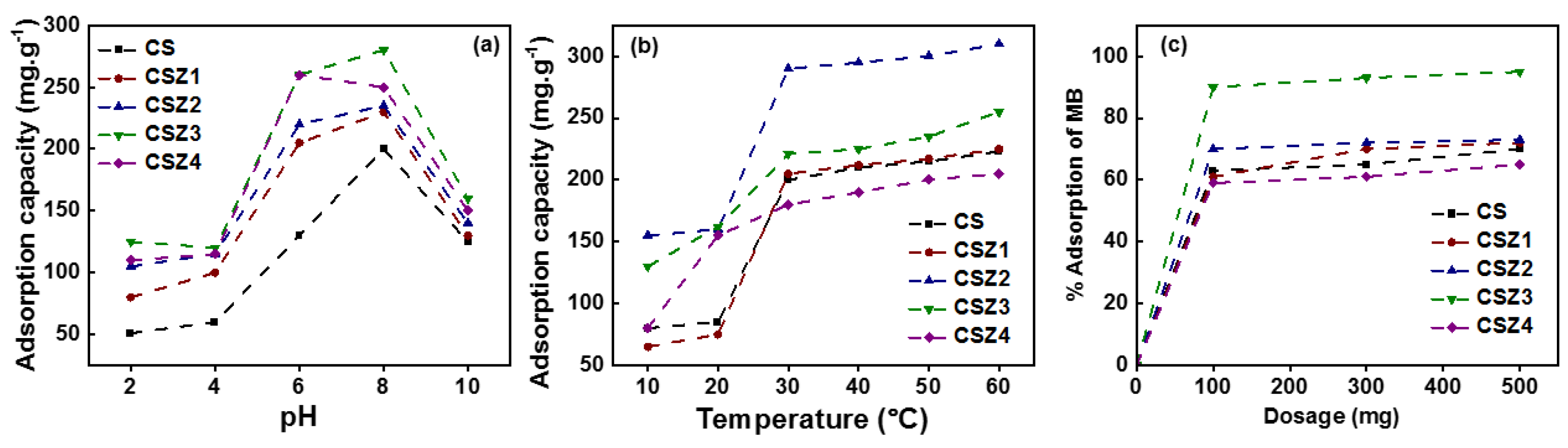
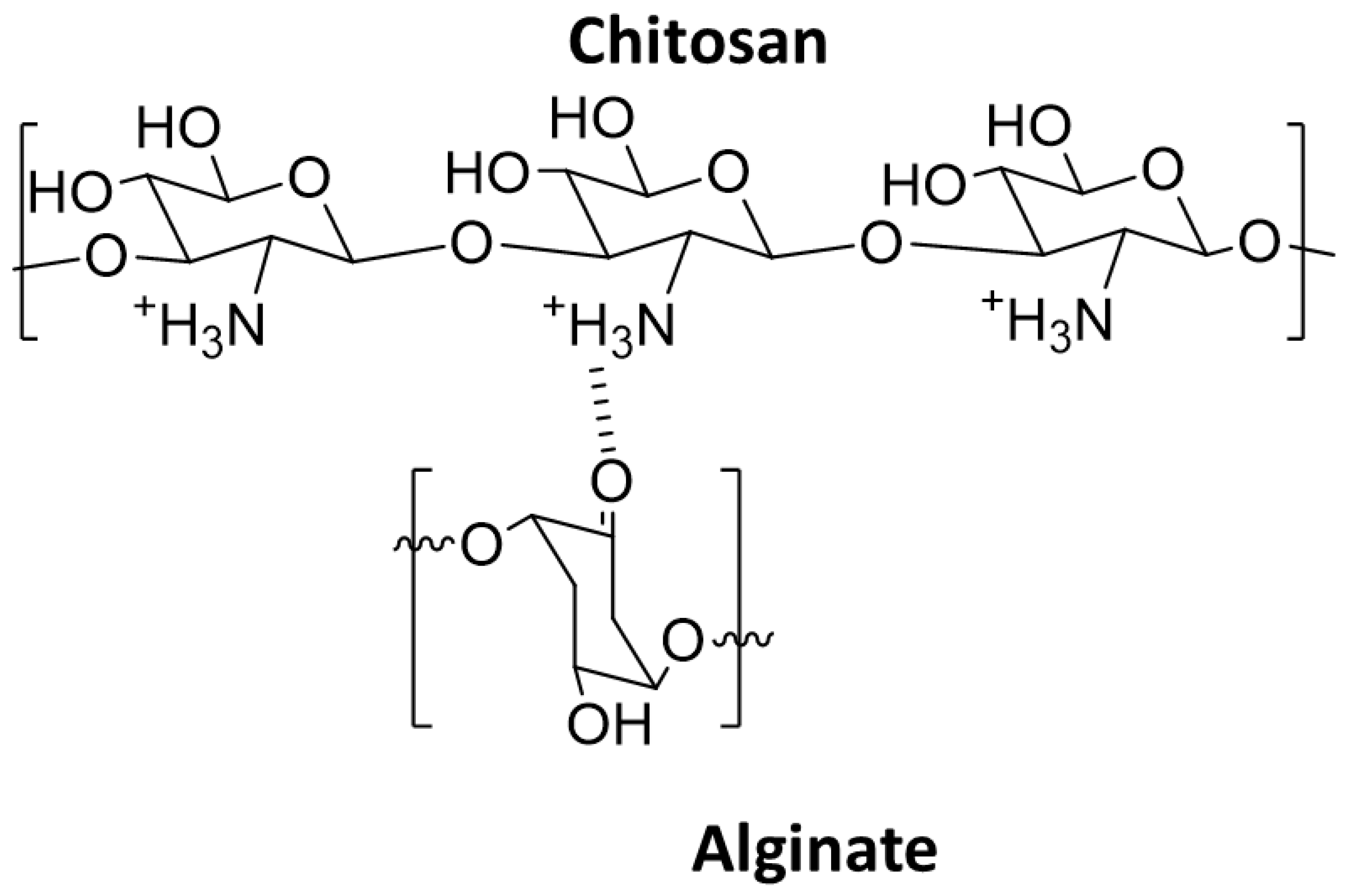
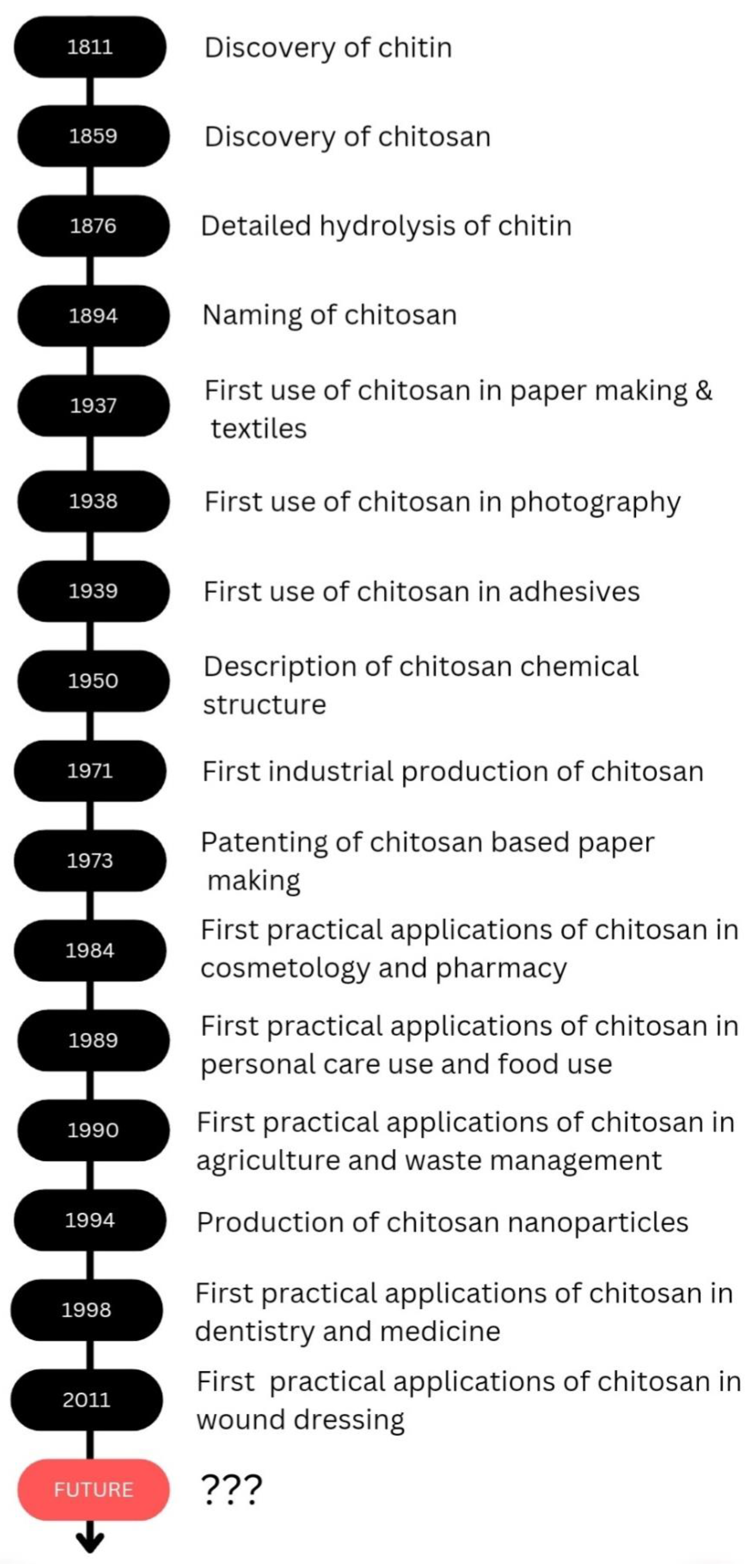
| Commercial Producer | Product | Type | Material | References |
|---|---|---|---|---|
| Axiobio | Axiostat® | Sponge | 100% chitosan | [94] |
| Trusetal | Chitoderm® plus | Superabsorber | Strong superabsorber coated with chitosan | [95] |
| Sam Medical | ChitoSAM™ 100 | Non-woven chitosan dressing spun directly from chitosan | 100% chitosan | [96] |
| MedTrade | Celox™ | Gauze (Celox Rapid, Celox Gauze) Granules (Celox A, Celox Granules) | Chito-R™ activated chitosan granules | [97] |
| Primex | ChitoClear® | Gel or liquid spray | ChitoClear® positively charged chitosan (the purest chitosan possible) | [98] |
| Medline | Opticell® | Gelling fiber | Primarily composed of chitosan (Cytoform chitosan-based gelling technology) | [99] |
| Tricol Biomedical | ChitoFlex® PRO | Hemostatic dressing active on both sides | chitosan based dressing | [100] |
| ChitoGauze® PRO | Chitosan-coated gauze | |||
| ChitoDot® | Double-sided hemostatic dressing | |||
| HemCon® Bandage PRO | Hemostatic bandage | |||
| HemCon Patch® PRO | Non-invasive hemostatic patch | |||
| HemCon® Strip PRO | Hemostatic bandage | |||
| BenQ Materials | ChitoClot Bandage | non-woven dressing | 100% chitosan-based, non-woven with adhesive back sheet | [101] |
| ChitoClot Pad | sponge | 100% medical-grade chitosan | ||
| ChitoClot Gauze | Gauze |
| Property of Chitosan | Action | Application | References |
|---|---|---|---|
|
| Ocular carrier for topical administration of required medicament. | [133] |
|
| Ocular wound repair and increased permeability | [134] |
|
| Ocular delivery | [69] |
|
| Creation of a formulation for a continuous medication releaseTreatment for glaucoma, age-related macular degeneration, viral and bacterial infections, and diabetic retinopathy | [132] |
|
| Age-related macular degeneration (AMD) and diabetic retinopathy (DR),Posterior eye drug delivery | [64] |
|
| Topical ophthalmic controlled drug deliveryTreatment of glaucoma | [66] |
|
| Increase the ocular bioavailability of diclofenac sodium already available | [65] |
|
| Ocular drug delivery | [135] |
|
| Ocular Delivery Formulations, ocular inflammation | [134] |
| Product/Commercial Name | Manufacturer | Application | References |
|---|---|---|---|
| HydamerTM | Chitinor AS (Tromso, Norway) | Film forming and fixative agent, deodorizing | [149] |
| Triozan | Ovensa Inc. (Aurora, ON, Canada) | Penetration enhancer | [150] |
| Ritachitosan® | Rita Corporation (Crystal Lake, IL, USA) | Film-forming agent | [151] |
| Scalp-purifying micellar shampoo | Kristin Ess Hair (Los Angeles, CA, USA) | Shampoo | [152] |
| CurasanTM | Chemisches Laboratorium Dr. Kurt Richter GmbH (Berlin, Germany) | Film-forming agent | [153] |
| Brazillian Joia strengthening and smoothing shampoo | Sol de Janeiro, Inc. (New York, NY, USA) | Shampoo | [154] |
| ZenvivoTM | Clariant (Muttenz, Switzerland) | Film-forming agent, antimicrobial, deodorizing, moisturizer | [155] |
| Extra gentle conditioner | Kristin Ess Hair (Los Angeles, CA, USA) | Hair conditioner | [156] |
| KIOsmetine® | Kitozyme (Herstal, Belgium) | Film-forming agent, moisturizer | [157] |
| Herbal essences set me up gel | Procter and Gamble (Cincinnati, OH, USA) | Hair-styling gel | [158] |
| Chitosonic® Acid | Personal Care Products Council (Washington DC, USA) | Antimicrobial, moisturizer | [159] |
| Re-vamp mid-length repair cream | Vernom Francois (Los Angeles, CA, USA) | Hair serum | [160] |
| ChitoClearTM | Primex Manufacturing Inc. (Langley, BC, Canada) | Film-forming agent | [161] |
| Anti-aging moisture lotion | Murad LLC (El Segundo, CA, USA) | Skin care | [162] |
| EverquatTM Q50H | Sino Lion (Florham Park, NJ, USA) | Shining agent, antidandruff agent, hair growth promoter, anti-hair-loss agent | [163] |
| Ultimate miracle worker multi-rejuvenating cream | Philosophy (New York, NY, USA) | Skin care | [164] |
| Vinkocos p-6N | Vink Chemicals GmbH & Co. KG (Kakenstorf, Germany) | Film-forming and wetting agent, thickener, stabilizer | [165] |
| St. ives replenishing mineral therapy body lotion | Unilever (London, UK) | Skin moisturizer | [166] |
| Jeen-Chitosan | Jeen International (Fairfield, NJ, USA) | Film-forming agent, moisturizer | [167] |
| Material Type | Additive | Property | References |
|---|---|---|---|
| Chitosan film | Cinnamon essential oils | Antimicrobial activity Mechanical strength Moisture barrier, Food shelf life extension | [246,247] |
| Chitosan film | Apricot kernel essential oils | Moisture barrier Antimicrobial activity Antioxidant activity Food shelf life extension | [248] |
| Chitosan film | Curcumin | Antimicrobial activity Antioxidant activity | [249] |
| Chitosan film | Purple-fleshed sweet potato extract | pH sensing Antioxidant activity | [250] |
| Chitosan film | Micro/nanoclay | Moisture barrier | [251] |
| Chitosan film | Cloisite 30B | Mechanical strength Moisture barrier | [252] |
| Chitosan film | Cloisite 15A | Thermal stability Oxygen barrier | [240] |
| Chitosan film | Citric acid | Cross-linking | [253] |
| Chitosan film | Glycerol | Plasticizing Flexibility | [248] |
| Chitosan film | Genipin | Mechanical strength Water resistance | [239] |
| Chitosan film | Amikacin | Antimicrobial activity | [254] |
| Chitosan coating | Chitosan nanoparticles | Antimicrobial activity | [255] |
| Chitosan coating | Silver-chitosan nanocomposite particles | Extended shelf life Antimicrobial activity | [245] |
| Chitosan coating | Zinc oxide | Moisture barrier Mechanical strength Antimicrobial activity | [256] |
| Chitosan film | Graphene oxide | Moisture barrier Mechanical strength Thermal stability | [257] |
| Chitosan film | Silver | Mechanical strength | [223] |
| Chitosan film | Multiwalled carbon nanotubes | Mechanical strength | [258] |
| Chitosan film | Sulfur | Antimicrobial activity Hydrophobicity Mechanical strength Moisture barrier | [259] |
| Chitosan film | Reduced graphene oxide | Mechanical strength Water resistance Antioxidant activity Electrical conductivity | [260] |
| Plants | Stress | Method of Application | Effects | References |
|---|---|---|---|---|
| Common bean (Phaseolus vulgaris L.) | Drought | Foliar application on seedlings | Increased plant growth and yield | [296] |
| Grapevine (V. vinifera) | Drought | Dipping of stem cutting before planting | Maintained chlorophyll content | [301] |
| Basil (Ocimum basilicum L.) | Drought | Foliar application on seedlings | Enhanced plant growth | [302] |
| Coffee (Coffea canephora var Robusta) | Drought | Foliar application on seedlings | Increased mineral uptake, total chlorophyll content and increased the resistance to drought stress | [303] |
| Cowpea (Vigna ungiculata) | Drought | Foliar application on seedlings | Improved growth and yield | [295] |
| White clover (Trifolium repens) | Drought | Foliar application on seedlings | Enhanced the production of stress-responsive metabolites | [304] |
| Thymus daenensis Celak | Drought | Spraying before the flowering stage | Increased flowering and full bloom and reduced negative impact on dry matter and oil yield | [305] |
| Safflower (Carthamus tinctorius L.) and sunflower (Helianthus annuus L.) | Salt | Seed treatment | Stimulated seedling growth and and tolerance to oxidative stress, reduced enzyme activity and | [306] |
| Plantago ovata | Salt | Seed treatment | Increased roots hoot | [307] |
| Plantago ovate Forsk | Salinity | Seed treatment | increased roots hoot and alleviated salt stress | [307] |
| O. sativa | Salt | Seed treatment | Enhanced catalase and peroxidase enzyme production | [308] |
| Vigna radiata | Salt | Seed treatment | Stimulated morphological parameters | [309] |
| Maize (Zea mays) | Salt | Foliar application | Enhanced all growth critera | [310] |
| Wheat (Triticum aestivum L.) | Salt | Adding to the nutrient solution | Lessened adverse effect of salt stress | [311] |
| Wheat (Triticum aestivum L.) | Drought | Seed treatment | Improved chlorophyll content, growth parameter and antioxidant enzymes | [312] |
| Potato (Solanum tuberosum) var. Favourite | Drought | Foliar application | Alleviated drought stress damage in young potato | [313] |
| Apple (Malus sieversii) | Drought | Foliar application | Improved leaf membrane stability, increased antioxidant enzymes | [314] |
| Fenugreek (Trigonella foenum-graecum L.) | Salinity | Seed treatment | Improved leaf water content, photosynthetic parameters and lessened salt stress | [314] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thambiliyagodage, C.; Jayanetti, M.; Mendis, A.; Ekanayake, G.; Liyanaarachchi, H.; Vigneswaran, S. Recent Advances in Chitosan-Based Applications—A Review. Materials 2023, 16, 2073. https://doi.org/10.3390/ma16052073
Thambiliyagodage C, Jayanetti M, Mendis A, Ekanayake G, Liyanaarachchi H, Vigneswaran S. Recent Advances in Chitosan-Based Applications—A Review. Materials. 2023; 16(5):2073. https://doi.org/10.3390/ma16052073
Chicago/Turabian StyleThambiliyagodage, Charitha, Madara Jayanetti, Amavin Mendis, Geethma Ekanayake, Heshan Liyanaarachchi, and Saravanamuthu Vigneswaran. 2023. "Recent Advances in Chitosan-Based Applications—A Review" Materials 16, no. 5: 2073. https://doi.org/10.3390/ma16052073
APA StyleThambiliyagodage, C., Jayanetti, M., Mendis, A., Ekanayake, G., Liyanaarachchi, H., & Vigneswaran, S. (2023). Recent Advances in Chitosan-Based Applications—A Review. Materials, 16(5), 2073. https://doi.org/10.3390/ma16052073





