Fabrication of a Fluorocarbon Low Surface Energy Coating for Anti-Stain Applications
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.3. Characterization
2.4. Thermal and Chemical Stability
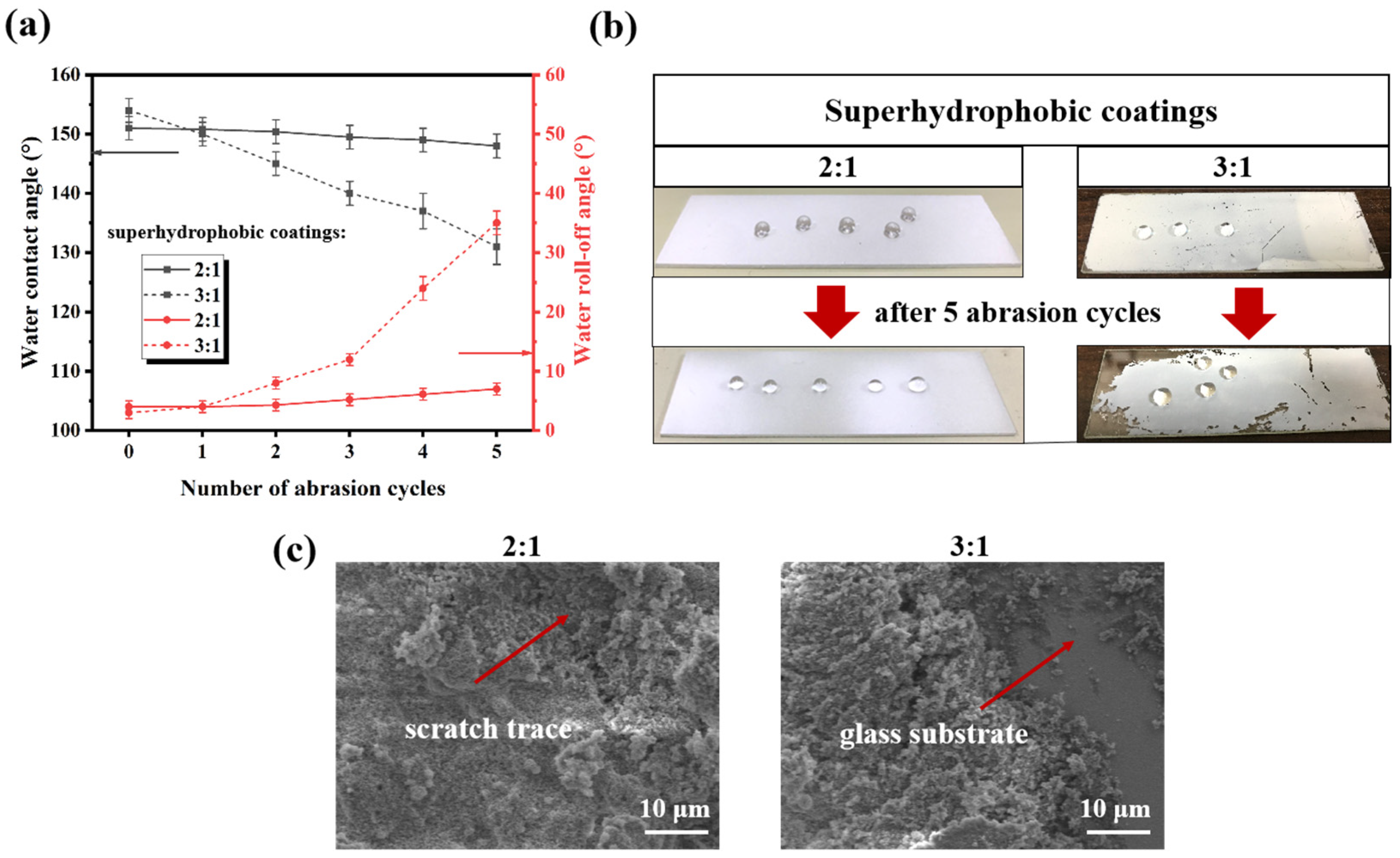
2.5. Abrasion Persistence
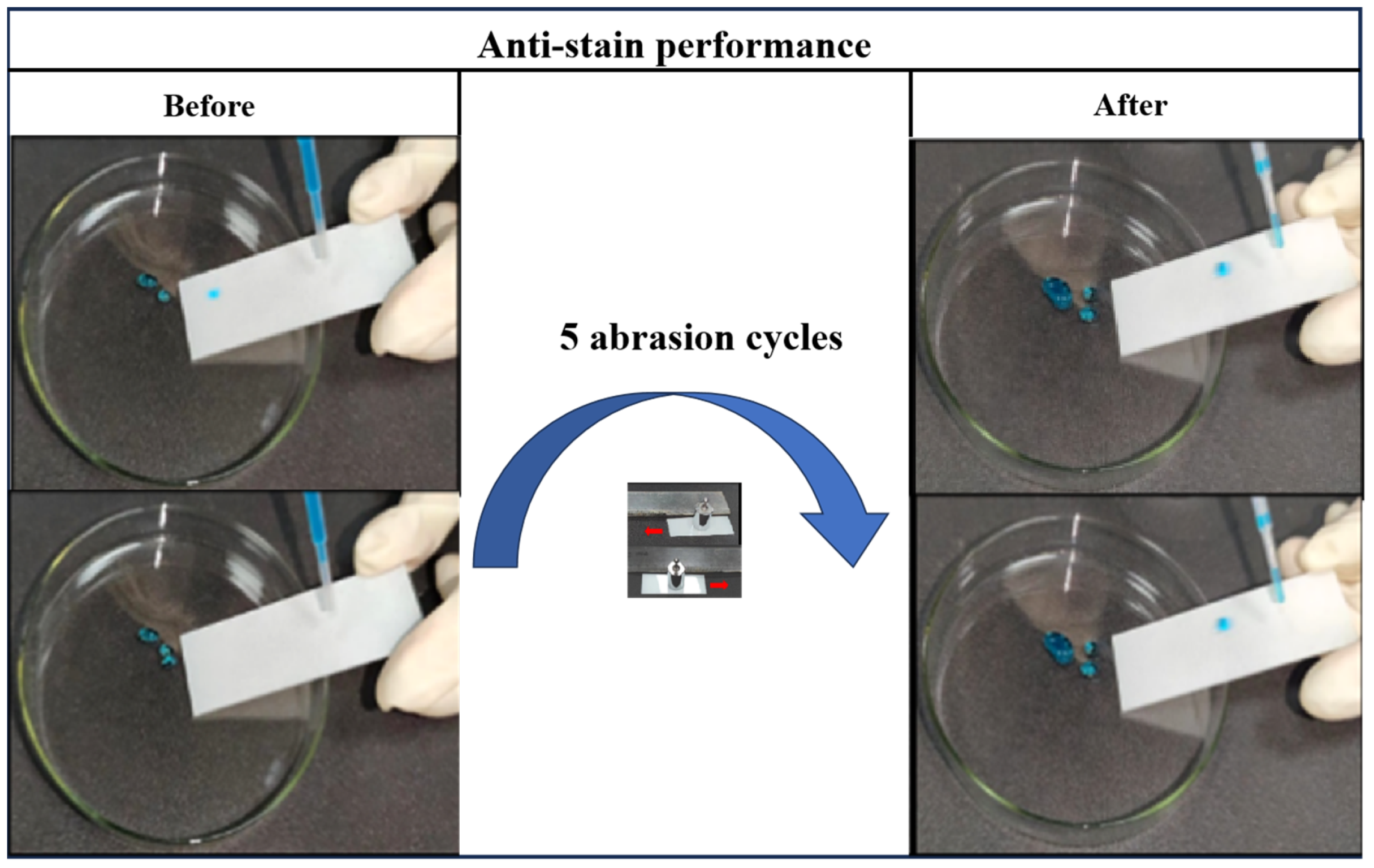
2.6. Anti-Stain Performance
3. Results and Discussion
3.1. Surface Characteristics of Fluorocarbon Resin/SiO2 Nanocomposite Coatings
3.2. Stability of Fluorocarbon Resin/SiO2 Nanocomposite Superhydrophobic Coatings
3.3. Anti-Stain Performance of Optimum Formulation Superhydrophobic Coating
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hari, R.; Mohana Rao, M. Characterization of nano-additive filled epoxy resin composites (ERC) for high voltage gas insulated switchgear (GIS) applications. Int. J. Emerg. Electr. Power Syst. 2022, 23, 47–57. [Google Scholar] [CrossRef]
- Tan, W.; Zhou, W. Research and application of anti-fouling flash method for transmission lines. Guangdong Electr. Power 2013, 26, 81–85. [Google Scholar] [CrossRef]
- Thennakoon, C.A.; Rajapakshe, R.B.S.D.; Rajapakse, R.M.G.; Rajapakse, S. Anti-stain and durable superhydrophobic/antistatic dual functionality surface for fabric materials based on F-ZnO/TiO2 composite. J. Sol-Gel Sci. Technol. 2022, 101, 529–538. [Google Scholar] [CrossRef]
- Latthe, S.S.; Sutar, R.S.; Kodag, V.S.; Bhosale, A.K.; Kumar, A.M.; Kumar Sadasivuni, K.; Xing, R.; Liu, S. Self–cleaning superhydrophobic coatings: Potential industrial applications. Prog. Org. Coat. 2019, 128, 52–58. [Google Scholar] [CrossRef]
- Seyedmehdi, S.A.; Ebrahimi, M. Superhydrophobic modified-polyurethane coatings for bushing of power transformers: From material to fabrication, mechanical and electrical properties. Prog. Org. Coat. 2018, 123, 134–137. [Google Scholar] [CrossRef]
- Ghunem, R.A.; Cherney, E.A.; Farzaneh, M.; Momen, G.; Illias, H.A.; Malagón, G.A.M.; Peesapati, V.; Yin, F. Development and Application of Superhydrophobic Outdoor Insulation: A Review. IEEE Trans. Dielectr. Electr. Insul. 2022, 29, 1392–1399. [Google Scholar] [CrossRef]
- Hooda, A.; Goyat, M.S.; Pandey, J.K.; Kumar, A.; Gupta, R. A review on fundamentals, constraints and fabrication techniques of superhydrophobic coatings. Prog. Org. Coat. 2020, 142, 105557. [Google Scholar] [CrossRef]
- Parvate, S.; Dixit, P.; Chattopadhyay, S. Superhydrophobic Surfaces: Insights from Theory and Experiment. J. Phys. Chem. B 2020, 124, 1323–1360. [Google Scholar] [CrossRef]
- Ahmad, I.; Kan, C.-W. A Review on Development and Applications of Bio-Inspired Superhydrophobic Textiles. Materials 2016, 9, 892. [Google Scholar] [CrossRef]
- Goharshenas Moghadam, S.; Parsimehr, H.; Ehsani, A. Multifunctional superhydrophobic surfaces. Adv. Colloid Interface Sci. 2021, 290, 102397. [Google Scholar] [CrossRef]
- Gateman, S.M.; Page, K.; Halimi, I.; Nascimento, A.R.C.; Savoie, S.; Schulz, R.; Moreau, C.; Parkin, I.P.; Mauzeroll, J. Corrosion of One-Step Superhydrophobic Stainless-Steel Thermal Spray Coatings. ACS Appl. Mater. Interfaces 2020, 12, 1523–1532. [Google Scholar] [CrossRef] [PubMed]
- Ouhib, F.; Dirani, A.; Aqil, A.; Glinel, K.; Nysten, B.; Jonas, A.M.; Jerôme, C.; Detrembleur, C. Transparent superhydrophobic coatings from amphiphilic-fluorinated block copolymers synthesized by aqueous polymerization-induced self-assembly. Polym. Chem. 2016, 7, 3998–4003. [Google Scholar] [CrossRef]
- Hashjin, R.N.R.; Ranjbar, Z.; Yari, H.; Momen, G. Tuning up sol-gel process to achieve highly durable superhydrophobic coating. Surf. Interfaces 2022, 33, 102282. [Google Scholar] [CrossRef]
- Rahman, M.K.; Phung, T.H.; Oh, S.; Kim, S.H.; Ng, T.N.; Kwon, K.-S. High-Efficiency Electrospray Deposition Method for Nonconductive Substrates: Applications of Superhydrophobic Coatings. ACS Appl. Mater. Interfaces 2021, 13, 18227–18236. [Google Scholar] [CrossRef] [PubMed]
- Varshney, P.; Mohapatra, S.S.; Kumar, A. Superhydrophobic coatings for aluminium surfaces synthesized by chemical etching process. Int. J. Smart Nano Mater. 2016, 7, 248–264. [Google Scholar] [CrossRef]
- Saji, V.S. Superhydrophobic surfaces and coatings by electrochemical anodic oxidation and plasma electrolytic oxidation. Adv. Colloid Interface Sci. 2020, 283, 102245. [Google Scholar] [CrossRef]
- Sharma, K.; Malik, M.K.; Hooda, A.; Pandey, K.; Sharma, J.; Goyat, M.S. Triethoxyoctylsilane-Modified SiO2 Nanoparticle-Based Superhydrophobic Coating for Corrosion Resistance of Mild Steel. J. Mater. Eng. Perform. 2023, 32, 6329–6338. [Google Scholar] [CrossRef]
- Pakdel, E.; Zhao, H.; Wang, J.; Tang, B.; Varley, R.J.; Wang, X. Superhydrophobic and photocatalytic self-cleaning cotton fabric using flower-like N-doped TiO2/PDMS coating. Cellulose 2021, 28, 8807–8820. [Google Scholar] [CrossRef]
- Sutar, R.S.; Nagappan, S.; Bhosale, A.K.; Sadasivuni, K.K.; Park, K.-H.; Ha, C.-S.; Latthe, S.S. Superhydrophobic Al2O3–Polymer Composite Coating for Self-Cleaning Applications. Coatings 2021, 11, 1162. [Google Scholar] [CrossRef]
- Tu, K.; Kong, L.; Wang, X.; Liu, J. Semitransparent, durable superhydrophobic polydimethylsiloxane/SiO2 nanocomposite coatings on varnished wood. Holzforschung 2016, 70, 1039–1045. [Google Scholar] [CrossRef]
- Rilda, Y.; Safitri, R.; Putri, Y.E.; Refinel, R.; Agustien, A.; Leaw, W.L.; Nur, H. Hexamethyldisiloxane-modified ZnO-SiO2-coated superhydrophobic textiles for antibacterial application. J. Chin. Chem. Soc. 2019, 66, 594–599. [Google Scholar] [CrossRef]
- Pistone, A.; Scolaro, C.; Celesti, C.; Visco, A. Study of Protective Layers Based on Crosslinked Glutaraldehyde/3-aminopropyltriethoxysilane. Polymers 2022, 14, 801. [Google Scholar] [CrossRef] [PubMed]
- Nishino, T.; Meguro, M.; Nakamae, K.; Matsushita, M.; Ueda, Y. The lowest surface free energy based on -CF3 alignment. Langmuir 1999, 15, 4321–4323. [Google Scholar] [CrossRef]
- Bayer, I.S.; Steele, A.; Loth, E. Superhydrophobic and electroconductive carbon nanotube-fluorinated acrylic copolymer nanocomposites from emulsions. Chem. Eng. J. 2013, 221, 522–530. [Google Scholar] [CrossRef]
- Olkowicz, K.; Buczko, Z.; Nasiłowska, B.; Kowalczyk, K.; Czwartos, J. Superhydrophobic Coating Based on Porous Aluminum Oxide Modified by Polydimethylsiloxane (PDMS). Materials 2022, 15, 1042. [Google Scholar] [CrossRef] [PubMed]
- Mokarian, Z.; Rasuli, R.; Abedini, Y. Facile synthesis of stable superhydrophobic nanocomposite based on multi-walled carbon nanotubes. Appl. Surf. Sci. 2016, 369, 567–575. [Google Scholar] [CrossRef]
- Atta, A.M.; Ezzat, A.O.; El-Saeed, A.M.; Wahby, M.H.; Abdallah, M.M.S. Superhydrophobic organic and inorganic clay nanocomposites for epoxy steel coatings. Prog. Org. Coat. 2020, 140, 105502. [Google Scholar] [CrossRef]
- Wu, X.; Wang, K.; Zhang, J.; Jie, X.; Chen, Z.; Lai, Y. A polyester-silica anti-condensation surface with anti-fouling property. Chem. Eng. J. 2022, 440, 135934. [Google Scholar] [CrossRef]
- Liu, Y.; Zhao, Z.; Shao, Y.; Wang, Y.; Liu, B. Preparation of a superhydrophobic coating based on polysiloxane modified SiO2 and study on its anti-icing performance. Surf. Coat. Technol. 2022, 437, 128359. [Google Scholar] [CrossRef]
- Uzoma, P.C.; Liu, F.; Han, E.-H. Multi-stimuli-triggered and self-repairable fluorocarbon organic coatings with urea-formaldehyde microcapsules filled with fluorosilane. J. Mater. Sci. Technol. 2020, 45, 70–83. [Google Scholar] [CrossRef]
- Ghezal, I.; Moussa, A.; Ben Marzoug, I.; El-Achari, A.; Campagne, C.; Sakli, F. Development and Surface State Characterization of a Spacer Waterproof Breathable Fabric. Fibers Polym. 2020, 21, 910–920. [Google Scholar] [CrossRef]
- Scholberg, H.M.; Guenthner, R.A.; Coon, R.I. Surface Chemistry of Fluorocarbons and their Derivatives. J. Phys. Chem. 1953, 57, 923–925. [Google Scholar] [CrossRef]
- Mazrouei-Sebdani, Z.; Khoddami, A. Alkaline hydrolysis: A facile method to manufacture superhydrophobic polyester fabric by fluorocarbon coating. Prog. Org. Coat. 2011, 72, 638–646. [Google Scholar] [CrossRef]
- Rico, V.; Mora, J.; García, P.; Agüero, A.; Borrás, A.; González-Elipe, A.R.; López-Santos, C. Robust anti-icing superhydrophobic aluminum alloy surfaces by grafting fluorocarbon molecular chains. Appl. Mater. Today 2020, 21, 100815. [Google Scholar] [CrossRef]
- Boinovich, L.B.; Emelyanenko, A.M. The behaviour of fluoro- and hydrocarbon surfactants used for fabrication of superhydrophobic coatings at solid/water interface. Colloids Surf. A Physicochem. Eng. Asp. 2015, 481, 167–175. [Google Scholar] [CrossRef]
- Dong, S.; Li, Y.; Tian, N.; Li, B.; Yang, Y.; Li, L.; Zhang, J. Scalable Preparation of Superamphiphobic Coatings with Ultralow Sliding Angles and High Liquid Impact Resistance. ACS Appl. Mater. Interfaces 2018, 10, 41878–41882. [Google Scholar] [CrossRef]
- Zhang, W.; Wang, D.; Sun, Z.; Song, J.; Deng, X. Robust superhydrophobicity: Mechanisms and strategies. Chem. Soc. Rev. 2021, 50, 4031–4061. [Google Scholar] [CrossRef]
- Domínguez, M.; Zarzuela, R.; Moreno-Garrido, I.; Carbú, M.; Cantoral, J.M.; Mosquera, M.J.; Gil, M.L.A. Anti-fouling nano-Ag/SiO2 ormosil treatments for building materials: The role of cell-surface interactions on toxicity and bioreceptivity. Prog. Org. Coat. 2021, 153, 106120. [Google Scholar] [CrossRef]
- Wang, B.; Qian, T.; Zhang, Q.; Zhan, X.; Chen, F. Heat resistance and surface properties of polyester resin modified with fluorosilicone. Surf. Coat. Technol. 2016, 304, 31–39. [Google Scholar] [CrossRef]


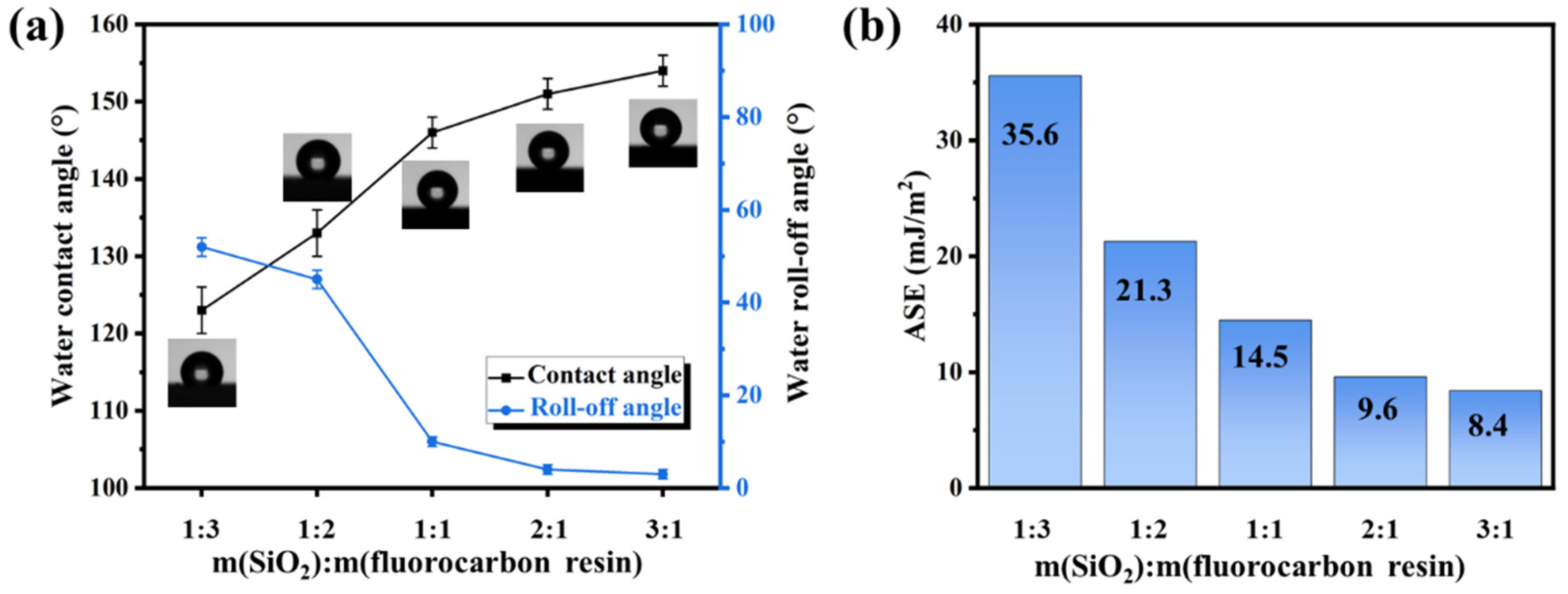


| Sample/m(SiO2):m(Resin) | Rq/nm | Ra/nm | z/µm | /° | /% |
|---|---|---|---|---|---|
| 1:3 | 73 | 62 | 0.44 | 123 ± 3 | 51 |
| 1:2 | 86 | 73 | 0.63 | 133 ± 3 | 66 |
| 1:1 | 106 | 88 | 0.94 | 146 ± 2 | 82 |
| 2:1 | 121 | 109 | 1.36 | 151 ± 2 | 87 |
| 3:1 | 135 | 113 | 1.60 | 154 ± 2 | 89 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pan, S.; Hu, Q.; Zhao, Y.; Wang, Q.; Li, Y.; Qian, Y.; He, C. Fabrication of a Fluorocarbon Low Surface Energy Coating for Anti-Stain Applications. Materials 2023, 16, 7516. https://doi.org/10.3390/ma16247516
Pan S, Hu Q, Zhao Y, Wang Q, Li Y, Qian Y, He C. Fabrication of a Fluorocarbon Low Surface Energy Coating for Anti-Stain Applications. Materials. 2023; 16(24):7516. https://doi.org/10.3390/ma16247516
Chicago/Turabian StylePan, Siwei, Qing Hu, Yaohong Zhao, Qing Wang, Yuanyuan Li, Yihua Qian, and Chunqing He. 2023. "Fabrication of a Fluorocarbon Low Surface Energy Coating for Anti-Stain Applications" Materials 16, no. 24: 7516. https://doi.org/10.3390/ma16247516
APA StylePan, S., Hu, Q., Zhao, Y., Wang, Q., Li, Y., Qian, Y., & He, C. (2023). Fabrication of a Fluorocarbon Low Surface Energy Coating for Anti-Stain Applications. Materials, 16(24), 7516. https://doi.org/10.3390/ma16247516






