Recent Progress of Layered Double Hydroxide-Based Materials in Wastewater Treatment
Abstract
:1. Introduction
2. Preparation and Modification of LDHs
2.1. Coprecipitation Method
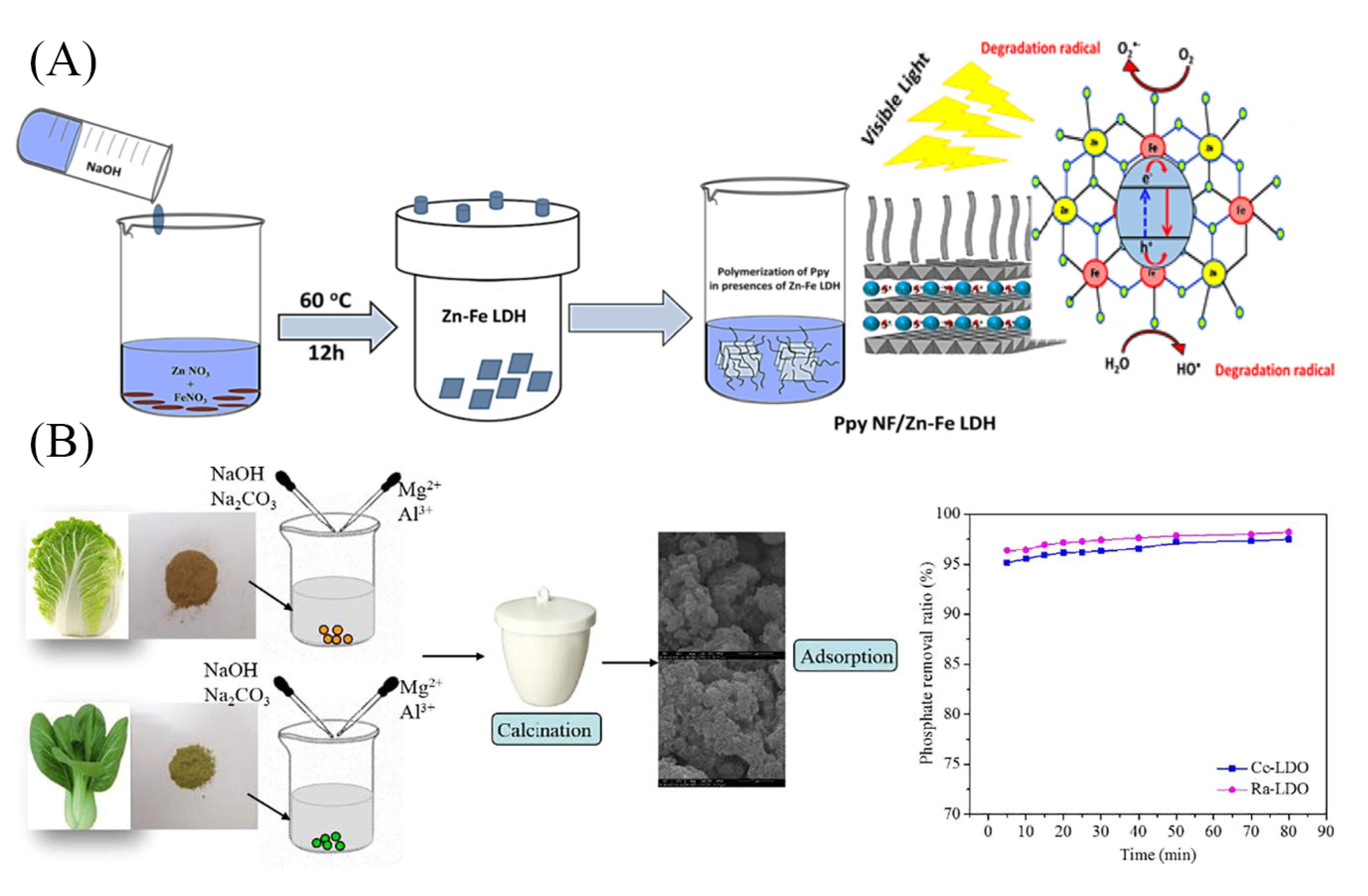
2.2. Hydrothermal Method
2.3. Ion Exchange Method
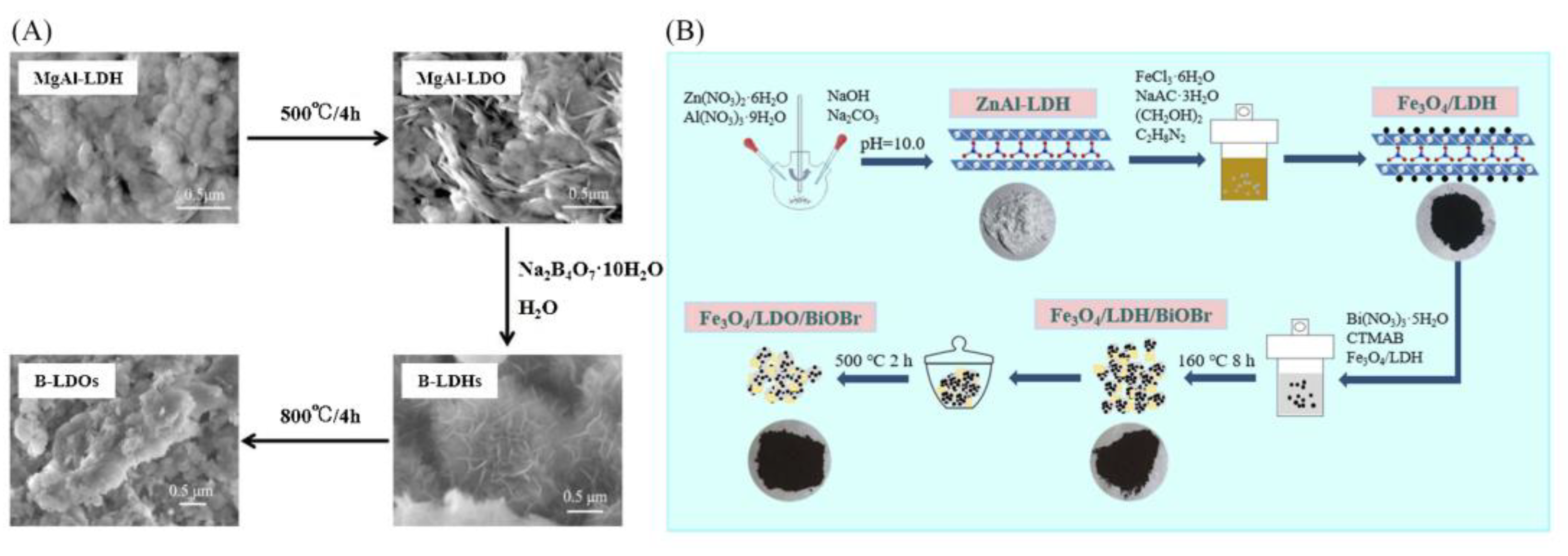
2.4. Calcination Recovery Method
2.5. Sol–Gel Method
3. Adsorptive Removal of Pollutants by LDH-Based Materials
3.1. Inorganic Pollutants
3.1.1. Heavy Metals
| Adsorbent | Heavy Metal Ions | Adsorption Conditions | Theoretical Adsorption Capacity (mg/g) | Adsorption Mechanism | Ref. | ||
|---|---|---|---|---|---|---|---|
| Adsorbent Dosage | pH | Oscillation Time (min) | |||||
| MgAl-CO3-LDH, Fe3O4/MgAl-CO3-LDH | Cd2+ Cd2+ | 0.08 g | 4 | 60 300 | 61.40~70.20 45.60~54.70 | precipitation, surface adsorption, surface complexation | [28] |
| MgAl-Cys-LDH | Cu2+ Pb2+ Cd2+ | 0.05 g | 4 | 90 180 10 | 58.07 186.20 93.11 | precipitation, surface complexation, isomorphous replacement | [29] |
| Fe3O4/LDH-AM | Cd2+ Pb2+ Cu2+ | 0.05 g | 5~6 | 240 180 240 | 74.06 266.60 64.66 | surface complexation, precipitation | [58] |
| Magnetic MgAl-LDO/carbon | Cd2+ Pb2+ Cu2+ | 6 | 90 120 400 | 386.10 359.70 192.70 | precipitation, surface complexation, electrostatic attraction | [59] | |
| CS/MgAl-LDH | Pb2+ Cu2+ | 0.05 g | 6 | 10 60 | 333.30 140.80 | precipitation, surface complexation, isomorphous replacement | [60] |
| CaAl-LDH | Cu2+ Cd2+ | 0.01 g | 5/5.8 | 381.90 1035.40 | precipitation, isomorphous replacement, surface complexation | [57] | |
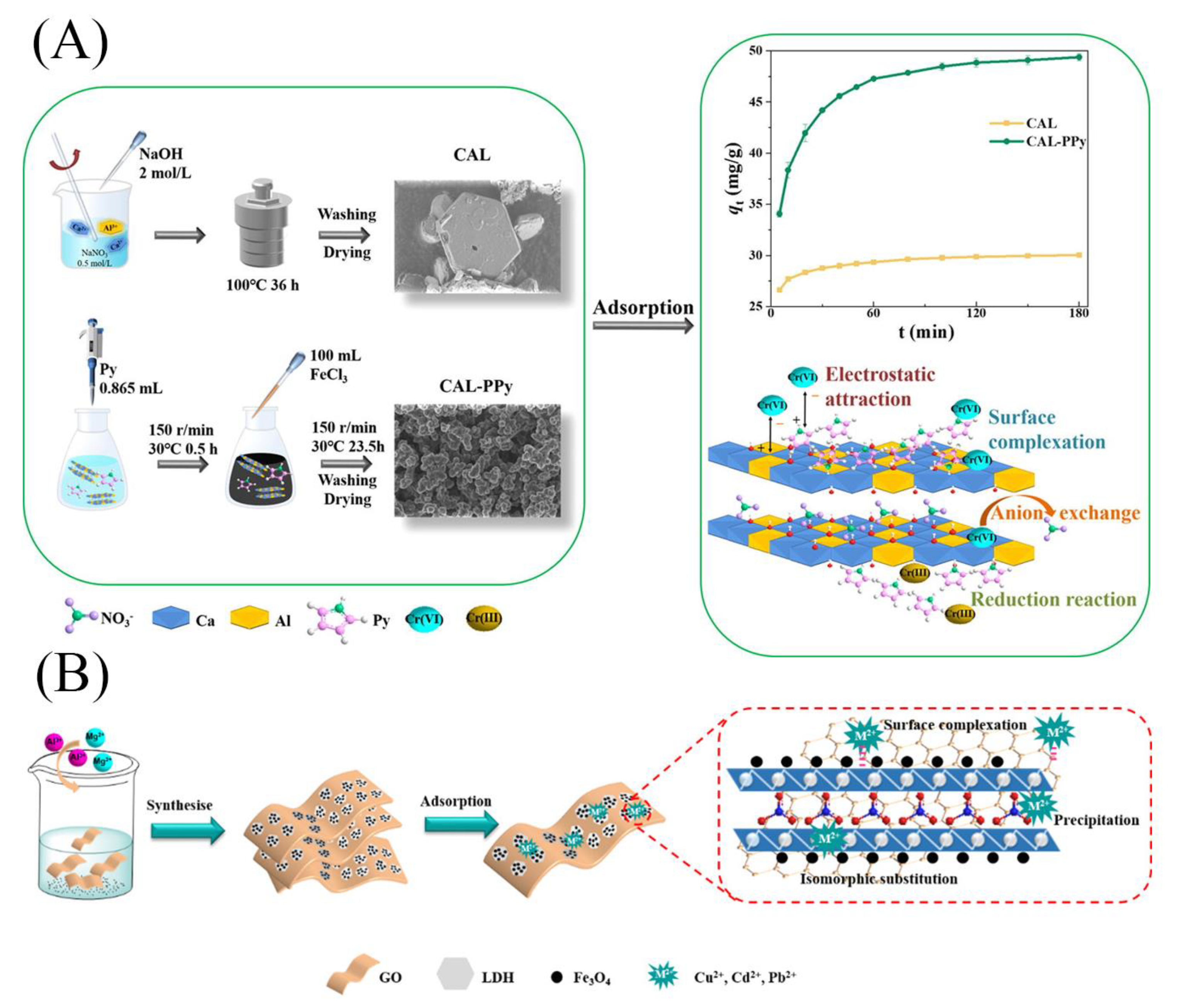
| GO/LDH Fe3O4@GO/LDH | Cu2+ Cd2+ Pb2+ | 20 mg | 240 | 89.26~80.72 76.67~70.26 226.98~213.96 | surface complexation, precipitation, isomorphous replacement | [41] | |
| LDH-EDTA-AM | Cr6+ | 30 | 48.47 | electrostatic attraction, surface complexation, ion exchange, reduction | [30] | ||
| Fe3O4-ZnAl-LDH/TiO2 | Cr6+ | 0.02 g/L | 3 | 480 | electrostatic attraction, ion exchange, photoreduction | [61] | |
| CaAl-LDH CAL-PPy | Cr6+ | 0.03 g | 34.06 66.14 | electrostatic attraction, surface complexation, anion exchange, reduction | [40] | ||

3.1.2. Inorganic Anions
3.2. Organic Pollutants
3.2.1. Dyes
3.2.2. Oil
3.2.3. Persistent Organic Pollutants (POPs)
3.3. Other Pollutants
4. Application of LDHs in Advanced Oxidation Processes
4.1. Fenton-like Reaction
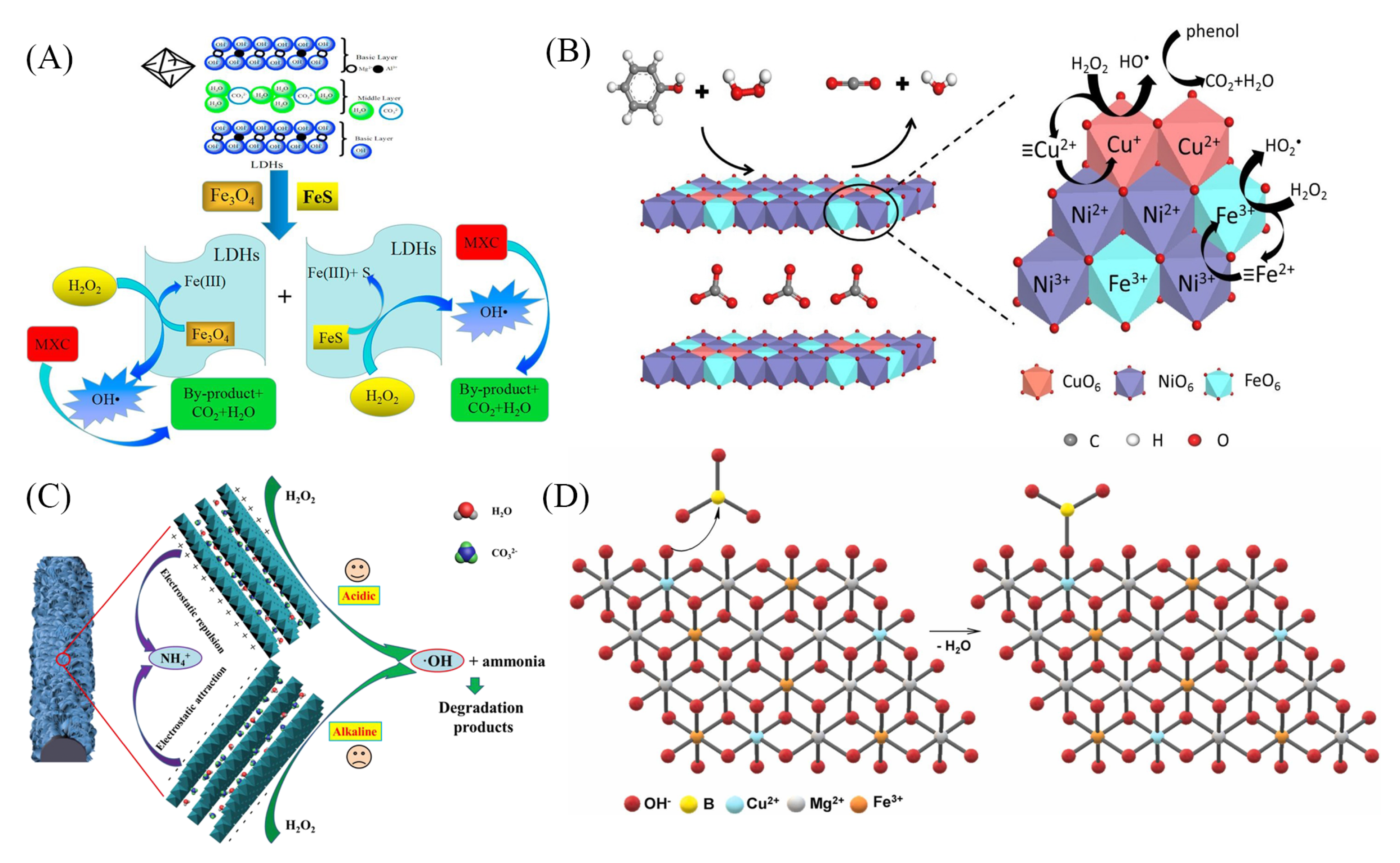
4.2. Persulfate-Based AOPs

4.3. Electrocatalytic System
5. Conclusions and Prospect
- (1)
- In terms of the preparation of LDHs, the method of high-performance LDHs is relatively cumbersome and not conducive to industrial production, which limits the application. In addition, LDHs are mostly in powder form and are difficult to be recovered after the treatment process;
- (2)
- In terms of wastewater treatment using LDH-based materials, most of the studies are still on the laboratory scale. LDHs are often used to remove a single pollutant, while various pollutants usually coexist in the water environment;
- (3)
- In terms of mechanism research, there is a lack of in-depth methods, and the quantitative contribution of each mechanism to the total capacity needs to be further investigated.
- (1)
- Due to the urgent demand for high-efficient and low-cost materials for treating wastewater, there is a need to produce sustainable, low-cost, and industry-scale LDHs. For better recovery of LDHs and improvement of reusability during large-scale use, magnetic or non-powdered LDHs can be designed and synthesized for wastewater treatment;
- (2)
- To make full use of the advantages of LDHs and other nanomaterials, high-performance LDH-based composites can be prepared via various methods. For example, LDHs with multivariate ions and the combination of LDHs with other high-performance materials may be one of the research directions to obtain high-performance catalysts;
- (3)
- More work from laboratory to industrial production is urgently needed. There is also a need to strengthen the research on the performance of modified LDHs in removing specific pollutants selectively from multiple pollutants;
- (4)
- Further in-depth studies on the mechanisms of LDHs for various pollutants can be carried out by means of spectroscopic characterization, interfacial chemical methods, and theoretical calculations. The contribution of each mechanism should be conducted to understand the interfacial process.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mishra, G.; Dash, B.; Pandey, S. Layered double hydroxides: A brief review from fundamentals to application as evolving biomaterials. Appl. Clay Sci. 2018, 153, 172–186. [Google Scholar] [CrossRef]
- Ma, L.; Wang, Q.; Islam, S.M.; Liu, Y.; Ma, S.; Kanatzidis, M.G. Highly selective and efficient removal of heavy metals by layered double hydroxide intercalated with the MoS42− ion. J. Am. Chem. Soc. 2016, 138, 2858–2866. [Google Scholar] [CrossRef]
- Li, C.; Wei, M.; Evans, D.G.; Duan, X. Layered double hydroxide-based nanomaterials as highly efficient catalysts and adsorbents. Small 2014, 10, 4469–4486. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, X.; An, C.; Wang, S. Electrochemistry-enhanced peroxymonosulfate activation by CoAl-LDH@biochar for simultaneous treatment of heavy metals and PAHs. Sep. Purif. Technol. 2023, 311, 123341. [Google Scholar] [CrossRef]
- Mohapatra, L.; Parida, K. A review on the recent progress, challenges and perspective of layered double hydroxides as promising photocatalysts. J. Mater. Chem. A 2016, 4, 10744–10766. [Google Scholar] [CrossRef]
- Boumeriame, H.; Da Silva, E.S.; Cherevan, A.S.; Chafik, T.; Faria, J.L.; Eder, D. Layered double hydroxide (LDH)-based materials: A mini-review on strategies to improve the performance for photocatalytic water splitting. J. Energy Chem. 2022, 64, 406–431. [Google Scholar] [CrossRef]
- Guan, X.; Yuan, X.; Zhao, Y.; Bai, J.; Li, Y.; Cao, Y.; Chen, Y.; Xiong, T. Adsorption behaviors and mechanisms of Fe/Mg layered double hydroxide loaded on bentonite on Cd (II) and Pb (II) removal. J. Colloid Interface Sci. 2022, 612, 572–583. [Google Scholar] [CrossRef]
- Zhang, G.; Zhang, X.; Meng, Y.; Pan, G.; Ni, Z.; Xia, S. Layered double hydroxides-based photocatalysts and visible-light driven photodegradation of organic pollutants: A review. Chem. Eng. J. 2020, 392, 123684. [Google Scholar] [CrossRef]
- Nerud, F.; Baldrian, P.; Gabriel, J.; Ogbeifun, D. Decolorization of synthetic dyes by the Fenton reagent and the Cu/pyridine/H2O2 system. Chemosphere 2001, 44, 957–961. [Google Scholar] [CrossRef]
- Pan, D.; Ge, S.; Tian, J.; Shao, Q.; Guo, L.; Liu, H.; Wu, S.; Ding, T.; Guo, Z. Research progress in the field of adsorption and catalytic degradation of sewage by hydrotalcite-derived materials. Chem. Rec. 2020, 20, 355–369. [Google Scholar] [CrossRef]
- Dong, Y.; Kong, X.; Luo, X.; Wang, H. Adsorptive removal of heavy metal anions from water by layered double hydroxide: A review. Chemosphere 2022, 303, 134685. [Google Scholar] [CrossRef]
- Keyikoglu, R.; Khataee, A.; Yoon, Y. Layered double hydroxides for removing and recovering phosphate: Recent advances and future directions. Adv. Colloid Interface 2022, 300, 102598. [Google Scholar] [CrossRef]
- Dias, A.C.; Ferreira Fontes, M.P. Arsenic (V) removal from water using hydrotalcites as adsorbents: A critical review. Appl. Clay Sci. 2020, 191, 105615. [Google Scholar] [CrossRef]
- Mittal, J. Recent progress in the synthesis of layered double hydroxides and their application for the adsorptive removal of dyes: A review. J. Environ. Manag. 2021, 295, 113017. [Google Scholar] [CrossRef]
- He, X.; Qiu, X.; Hu, C.; Liu, Y. Treatment of heavy metal ions in wastewater using layered double hydroxides: A review. J. Dispers. Sci. Technol. 2018, 39, 792–801. [Google Scholar] [CrossRef]
- Tang, Z.; Qiu, Z.; Lu, S.; Shi, X. Functionalized layered double hydroxide applied to heavy metal ions absorption: A review. Nanotechnol. Rev. 2020, 9, 800–819. [Google Scholar] [CrossRef]
- Taoufik, N.; Sadiq, M.H.; Abdennouri, M.; Qourzal, S.; Khataee, A.; Sillanpaa, M.; Barka, N. Recent advances in the synthesis and environmental catalytic applications of layered double hydroxides-based materials for degradation of emerging pollutants through advanced oxidation processes. Mater. Res. Bull. 2022, 154, 111924. [Google Scholar] [CrossRef]
- Xie, Z.; Zhou, H.; He, C.; Pan, Z.; Yao, G.; Lai, B. Synthesis, application and catalytic performance of layered double hydroxide based catalysts in advanced oxidation processes for wastewater decontamination: A review. Chem. Eng. J. 2021, 414, 128713. [Google Scholar] [CrossRef]
- Chen, J.; Yuan, H.; Yu, J.; Yan, M.; Yang, Y.; Lin, S. Regulating lithium extraction based on intercalated SO42− in Li/Al-LDHs. J. Colloid Interface Sci. 2023, 649, 694–702. [Google Scholar] [CrossRef]
- Li, K.; Li, S.; Li, Q.; Liu, H.; Yao, W.; Wang, Q.; Chai, L. Design of a high-performance ternary LDHs containing Ni, Co and Mn for arsenate removal. J. Hazard. Mater. 2022, 427, 127865. [Google Scholar] [CrossRef]
- Enkhtuvshin, E.; Kim, K.M.; Kim, Y.K.; Mihn, S.; Kim, S.J.; Jung, S.Y.; Thao, N.T.T.; Ali, G.; Akbar, M.; Chung, K.Y.; et al. Stabilizing oxygen intermediates on redox-flexible active sites in multimetallic Ni-Fe-Al-Co layered double hydroxide anodes for excellent alkaline and seawater electrolysis. J. Mater. Chem. A 2021, 9, 27332–27346. [Google Scholar] [CrossRef]
- Li, C.F.; Xie, L.J.; Zhao, J.W.; Gu, L.F.; Wu, J.Q.; Li, G.R. Interfacial electronic modulation by Fe2O3/NiFe-LDHs heterostructures for efficient oxygen evolution at high current density. Appl. Catal. B-Environ. 2022, 306, 121097. [Google Scholar] [CrossRef]
- Li, H.; Sun, Y.; Wang, J.; Liu, Y.; Li, C. Nanoflower-branch LDHs and CoNi alloy derived from electrospun carbon nanofibers for efficient oxygen electrocatalysis in microbial fuel cells. Appl. Catal. B-Environ. 2022, 307, 121136. [Google Scholar] [CrossRef]
- Chen, Y.; Li, J.; Zhai, B.; Liang, Y. Enhanced photocatalytic degradation of RhB by two-dimensional composite photocatalyst. Colloid Surf. A 2019, 568, 429–435. [Google Scholar] [CrossRef]
- Tang, S.; Yao, Y.; Chen, T.; Kong, D.; Shen, W.; Lee, H.K. Recent advances in the application of layered double hydroxides in analytical chemistry: A review. Anal. Chim. Acta 2020, 1103, 32–48. [Google Scholar] [CrossRef]
- Lu, L.; Li, J.; Ng, D.H.L.; Yang, P.; Song, P.; Zuo, M. Synthesis of novel hierarchically porous Fe3O4@MgAl-LDH magnetic microspheres and its superb adsorption properties of dye from water. J. Ind. Eng. Chem. 2017, 46, 315–323. [Google Scholar] [CrossRef]
- Mohamed, F.; Abukhadra, M.R.; Shaban, M. Removal of safranin dye from water using polypyrrole nanofiber/Zn-Fe layered double hydroxide nanocomposite (PPy NF/Zn-Fe LDH) of enhanced adsorption and photocatalytic properties. Sci. Total Environ. 2018, 640, 352–363. [Google Scholar] [CrossRef]
- Shan, R.; Yan, L.; Yang, K.; Hao, Y.; Du, B. Adsorption of Cd(II) by Mg-Al-CO3- and magnetic Fe3O4/Mg-Al-CO3-layered double hydroxides: Kinetic, isothermal, thermodynamic and mechanistic studies. J. Hazard. Mater. 2015, 299, 42–49. [Google Scholar] [CrossRef]
- Zhang, X.; Yan, L.; Li, J.; Yu, H. Adsorption of heavy metals by L-cysteine intercalated layered double hydroxide: Kinetic, isothermal and mechanistic studies. J. Colloid Interface Sci. 2020, 562, 149–158. [Google Scholar] [CrossRef]
- Li, J.; Yu, H.; Zhang, X.; Zhu, R.; Yan, L. Crosslinking acrylamide with EDTA-intercalated layered double hydroxide for enhanced recovery of Cr(VI) and Congo red: Adsorptive and mechanistic study. Front. Environ. Sci. Eng. 2020, 14, 52. [Google Scholar] [CrossRef]
- Shan, R.; Yan, L.; Yang, K.; Yu, S.; Hao, Y.; Yu, H.; Du, B. Magnetic Fe3O4/MgAl-LDH composite for effective removal of three red dyes from aqueous solution. Chem. Eng. J. 2014, 252, 38–46. [Google Scholar] [CrossRef]
- Su, X.; Chen, Y.; Li, Y.; Li, J.; Song, W.; Li, X.; Yan, L. Enhanced adsorption of aqueous Pb(II) and Cu(II) by biochar loaded with layered double hydroxide: Crucial role of mineral precipitation. J. Mol. Liq. 2022, 357, 119083. [Google Scholar] [CrossRef]
- Zhang, Z.; Yan, L.; Yu, H.; Yan, T.; Li, X. Adsorption of phosphate from aqueous solution by vegetable biochar/layered double oxides: Fast removal and mechanistic studies. Bioresour. Technol. 2019, 284, 65–71. [Google Scholar] [CrossRef]
- Chaillot, D.; Bennici, S.; Brendle, J. Layered double hydroxides and LDH-derived materials in chosen environmental applications: A review. Environ. Sci. Pollut. Res. 2021, 28, 24375–24405. [Google Scholar] [CrossRef]
- Kozerozhets, I.V.; Panasyuk, G.P.; Semenov, E.A.; Avdeeva, V.V.; Danchevskaya, M.N.; Simonenko, N.P.; Vasiliev, M.G.; Kozlova, L.O.; Ivakin, Y.D. Recrystallization of nanosized boehmite in an aqueous medium. Powder Technol. 2023, 413, 118030. [Google Scholar] [CrossRef]
- Gu, P.; Zhang, S.; Li, X.; Wang, X.; Wen, T.; Jehan, R.; Alsaedi, A.; Hayat, T.; Wang, X. Recent advances in layered double hydroxide-based nanomaterials for the removal of radionuclides from aqueous solution. Environ. Pollut. 2018, 240, 493–505. [Google Scholar] [CrossRef]
- Liang, D.; Yue, W.; Sun, G.; Zheng, D.; Ooi, K.; Yang, X. Direct Synthesis of unilamellar MgAl-LDH nanosheets and stacking in aqueous solution. Langmuir 2015, 31, 12464–12471. [Google Scholar] [CrossRef]
- Omwoma, S.; Chen, W.; Tsunashima, R.; Song, Y. Recent advances on polyoxometalates intercalated layered double hydroxides: From synthetic approaches to functional material applications. Coord. Chem. Rev. 2014, 258, 58–71. [Google Scholar] [CrossRef]
- Rathee, G.; Awasthi, A.; Sood, D.; Tomar, R.; Tomar, V.; Chandra, R. A new biocompatible ternary layered double hydroxide adsorbent for ultrafast removal of anionic organic dyes. Sci. Rep. 2019, 9, 16225. [Google Scholar] [CrossRef]
- Yang, D.; Chen, Y.; Li, J.; Li, Y.; Song, W.; Li, X.; Yan, L. Synthesis of calcium-aluminum-layered double hydroxide and a polypyrrole decorated product for efficient removal of high concentrations of aqueous hexavalent chromium. J. Colloid Interface Sci. 2022, 607, 1963–1972. [Google Scholar] [CrossRef]
- Li, J.; Huang, Q.; Yu, H.; Yan, L. Enhanced removal performance and mechanistic study of Cu2+, Cd2+, and Pb2+ by magnetic layered double hydroxide nanosheets assembled on graphene oxide. J. Water Process Eng. 2022, 48, 102893. [Google Scholar] [CrossRef]
- Gonzalez, M.A.; Pavlovic, I.; Barriga, C. Cu(II), Pb(II) and Cd(II) sorption on different layered double hydroxides. A kinetic and thermodynamic study and competing factors. Chem. Eng. J. 2015, 269, 221–228. [Google Scholar] [CrossRef]
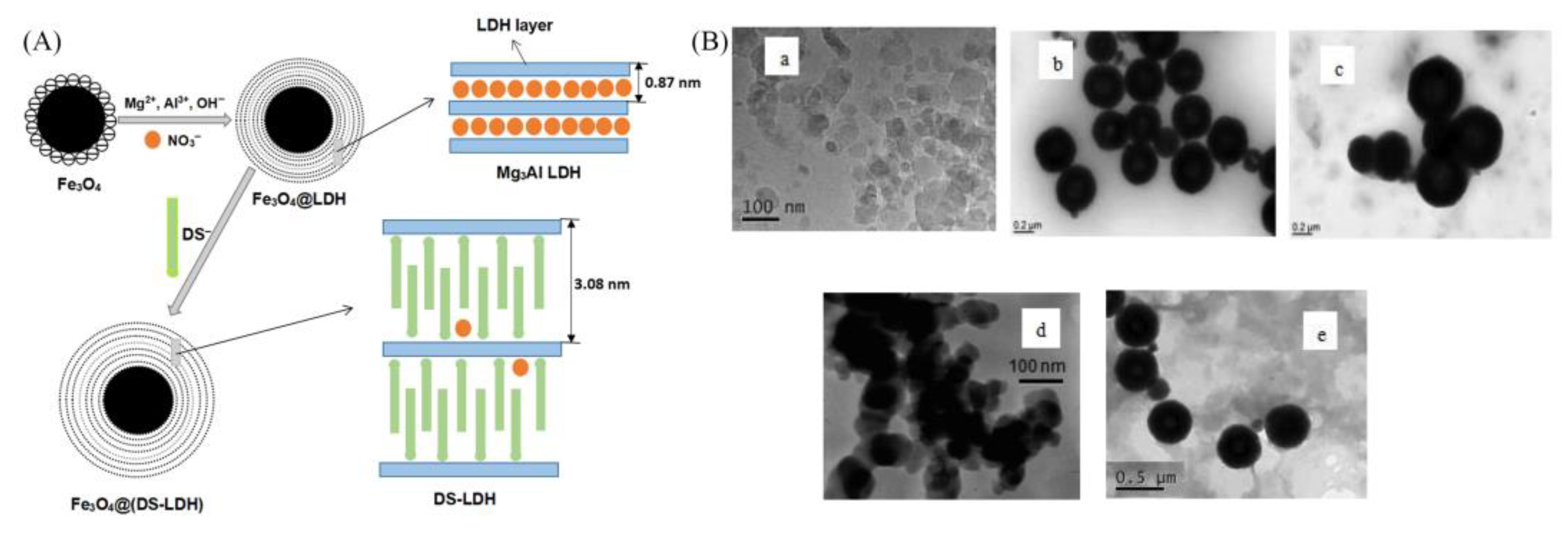
- Li, Y.; Bi, H.; Liang, Y.; Mao, X.; Li, H. A magnetic core-shell dodecyl sulfate intercalated layered double hydroxide nanocomposite for the adsorption of cationic and anionic organic dyes. Appl. Clay Sci. 2019, 183, 105309. [Google Scholar] [CrossRef]
- Jia, Y.; Liu, Z. Preparation of borate anions intercalated MgAl-LDHs microsphere and its calcinated product with superior adsorption performance for Congo red. Colloid Surf. A 2019, 575, 373–381. [Google Scholar] [CrossRef]
- Li, J.; Yu, H.; Yan, T.; Sun, M.; Li, X.; Song, W.; Yan, L. Insight into the enhanced visible-light photoreduction of aqueous Cr(VI) by assembled Fe3O4/LDO/BiOBr composites. Colloid Surf. A 2022, 634, 128021. [Google Scholar] [CrossRef]
- Feng, X.; Long, R.; Wang, L.; Liu, C.; Bai, Z.; Liu, X. A review on heavy metal ions adsorption from water by layered double hydroxide and its composites. Sep. Purif. Technol. 2022, 284, 120099. [Google Scholar] [CrossRef]
- Kameliya, J.; Verma, A.; Dutta, P.; Arora, C.; Vyas, S.; Varma, R.S. Layered double hydroxide materials: A review on their preparation, characterization, and applications. Inorganics 2023, 11, 121. [Google Scholar] [CrossRef]
- Danks, A.E.; Hall, S.R.; Schnepp, Z. The evolution of ‘sol-gel’ chemistry as a technique for materials synthesis. Mater. Horiz. 2016, 3, 91–112. [Google Scholar] [CrossRef]
- Chubar, N.; Gerda, V.; Megantari, O.; Micusik, M.; Omastova, M.; Heister, K.; Man, P.; Fraissard, J. Applications versus properties of Mg-Al layered double hydroxides provided by their syntheses methods: Alkoxide and alkoxide-free sol-gel syntheses and hydrothermal precipitation. Chem. Eng. J. 2013, 234, 284–299. [Google Scholar] [CrossRef]
- Valeikiene, L.; Roshchina, M.; Grigoraviciute-Puroniene, I.; Prozorovich, V.; Zarkov, A.; Ivanets, A.; Kareiva, A. On the reconstruction peculiarities of sol-gel derived Mg2-xMx/Al-1(M = Ca, Sr, Ba) layered double hydroxides. Crystals 2020, 10, 470. [Google Scholar] [CrossRef]
- Luo, Q.; Dong, M.; Li, Q.; Wu, Z.; Liu, Z.; Li, J. Improve the durability of lithium adsorbent Li/Al-LDHs by Fe3+ substitution and nanocomposite of FeOOH. Miner. Eng. 2022, 185, 107717. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, M.; Cheng, Q.; Wang, C.; Li, H.; Han, X.; Fan, Z.; Su, G.; Pan, D.; Li, Z. Research progress of adsorption and removal of heavy metals by chitosan and its derivatives: A review. Chemosphere 2021, 279, 130927. [Google Scholar] [CrossRef]
- Wu, M.J.; Wu, J.Z.; Zhang, J.; Chen, H.; Zhou, J.Z.; Qian, G.R.; Xu, Z.P.; Du, Z.; Rao, Q.L. A review on fabricating heterostructures from layered double hydroxides for enhanced photocatalytic activities. Catal. Sci. Technol. 2018, 8, 1207–1228. [Google Scholar] [CrossRef]
- El Ashmawy, A.A.; Tada, M.; Yoshimura, C. Weak dehydration enhances the adsorption capacity of boehmite for anionic dyes. Colloid Surf. A 2023, 674, 131954. [Google Scholar] [CrossRef]
- Laipan, M.; Yu, J.; Zhu, R.; Zhu, J.; Smith, A.T.; He, H.; O’Hare, D.; Sun, L. Functionalized layered double hydroxides for innovative applications. Mater. Horiz. 2020, 7, 715–745. [Google Scholar] [CrossRef]
- Ma, S.; Huang, L.; Ma, L.; Shim, Y.; Islam, S.M.; Wang, P.; Zhao, L.-D.; Wang, S.; Sun, G.; Yang, X.; et al. Efficient uranium capture by polysulfide/layered double hydroxide composites. J. Am. Chem. Soc. 2015, 137, 3670–3677. [Google Scholar] [CrossRef]
- Zhang, S.; Chen, Y.; Li, J.; Li, Y.; Song, W.; Li, X.; Yan, L.; Yu, H. Highly efficient removal of aqueous Cu(II) and Cd(II) by hydrothermal synthesized CaAl-layered double hydroxide. Colloid Surf. A 2022, 641, 128584. [Google Scholar] [CrossRef]
- Sun, J.; Chen, Y.; Yu, H.; Yan, L.; Du, B.; Pei, Z. Removal of Cu2+, Cd2+ and Pb2+ from aqueous solutions by magnetic alginate microsphere based on Fe3O4/MgAl-layered double hydroxide. J. Colloid Interface Sci. 2018, 532, 474–484. [Google Scholar] [CrossRef]
- Hou, T.; Yan, L.; Li, J.; Yang, Y.; Shan, L.; Meng, X.; Li, X.; Zhao, Y. Adsorption performance and mechanistic study of heavy metals by facile synthesized magnetic layered double oxide/carbon composite from spent adsorbent. Chem. Eng. J. 2020, 384, 123331. [Google Scholar] [CrossRef]
- Lyu, F.; Yu, H.; Hou, T.; Yan, L.; Zhang, X.; Du, B. Efficient and fast removal of Pb2+ and Cd2+ from an aqueous solution using a chitosan/Mg-Al-layered double hydroxide nanocomposite. J. Colloid Interface Sci. 2019, 539, 184–193. [Google Scholar] [CrossRef]
- Yang, Y.; Li, J.; Yan, T.; Zhu, R.; Yan, L.; Pei, Z. Adsorption and photocatalytic reduction of aqueous Cr(VI) by Fe3O4-ZnAl-layered double hydroxide/TiO2 composites. J. Colloid Interface Sci. 2020, 562, 493–501. [Google Scholar] [CrossRef]
- Hai, N.T.; Dong, T.N.; Giang, T.L.; Tomul, F.; Lima, E.C.; Woo, S.H.; Sarmah, A.K.; Hung, Q.N.; Phuong, T.N.; Dinh, D.N.; et al. Adsorption mechanism of hexavalent chromium onto layered double hydroxides-based adsorbents: A systematic in-depth review. J. Hazard. Mater. 2019, 373, 258–270. [Google Scholar] [CrossRef]
- Poudel, M.B.; Awasthi, G.P.; Kim, H.J. Novel insight into the adsorption of Cr(VI) and Pb(II) ions by MOF derived Co-Al layered double hydroxide @hematite nanorods on 3D porous carbon nanofiber network. Chem. Eng. J. 2021, 417, 129312. [Google Scholar] [CrossRef]
- Maziarz, P.; Matusik, J.; Leiviska, T. Mg/Al LDH enhances sulfate removal and clarification of AMD wastewater in precipitation processes. Materials 2019, 12, 2334. [Google Scholar] [CrossRef]
- Yang, P.; Li, S.; Liu, C.; Liu, X. Interface-constrained layered double hydroxides for stable uranium capture in highly acidic industrial wastewater. ACS Appl. Mater. Interfaces 2021, 13, 17988–17997. [Google Scholar] [CrossRef]
- Yang, L.; Qiao, B.; Zhang, S.; Yao, H.; Cai, Z.; Han, Y.; Li, C.; Li, Y.; Ma, S. Intercalation of salicylaldoxime into layered double hydroxide: Ultrafast and highly selective uptake of uranium from different water systems via versatile binding modes. J. Colloid Interface Sci. 2023, 642, 623–637. [Google Scholar] [CrossRef]
- Mudhoo, A.; Paliya, S.; Goswami, P.; Singh, M.; Lofrano, G.; Carotenuto, M.; Carraturo, F.; Libralato, G.; Guida, M.; Usman, M.; et al. Fabrication, functionalization and performance of doped photocatalysts for dye degradation and mineralization: A review. Environ. Chem. Lett. 2020, 18, 1825–1903. [Google Scholar] [CrossRef]
- Soltani, R.; Marjani, A.; Shirazian, S. A hierarchical LDH/MOF nanocomposite: Single, simultaneous and consecutive adsorption of a reactive dye and Cr(vi). Dalton Trans. 2020, 49, 5323–5335. [Google Scholar] [CrossRef]
- Mallakpour, S.; Radfar, Z.; Feiz, M. Chitosan/tannic acid/ZnFe layered double hydroxides and mixed metal oxides nanocomposite for the adsorption of reactive dyes. Carbohydr. Polym. 2023, 305, 120528. [Google Scholar] [CrossRef]
- Jamaly, S.; Giwa, A.; Hasan, S.W. Recent improvements in oily wastewater treatment: Progress, challenges, and future opportunities. J. Environ. Sci. 2015, 37, 15–30. [Google Scholar] [CrossRef]
- Yaacob, N.; Sean, G.P.; Nazri, N.A.M.; Ismail, A.F.; Abidin, M.N.Z.; Subramaniam, M.N. Simultaneous oily wastewater adsorption and photodegradation by ZrO2-TiO2 heterojunction photocatalysts. J. Water Process Eng. 2021, 39, 101644. [Google Scholar] [CrossRef]
- Ikhsan, S.N.W.; Yusof, N.; Aziz, F.; Ismail, A.F.; Jaafar, J.; Salleh, W.N.W.; Misdan, N. Superwetting materials for hydrophilic-oleophobic membrane in oily wastewater treatment. J. Environ. Manag. 2021, 290, 112565. [Google Scholar] [CrossRef]
- Su, Y.; Li, Z.; Zhou, H.; Kang, S.; Zhang, Y.; Yu, C.; Wang, G. Ni/carbon aerogels derived from water induced self-assembly of Ni-MOF for adsorption and catalytic conversion of oily wastewater. Chem. Eng. J. 2020, 402, 126205. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, B.; Wang, J.; Ren, Y.; Xuan, C.; Liu, C.; Shen, C. Superhydrophobic and superoleophilic porous reduced graphene oxide/polycarbonate monoliths for high-efficiency oil/water separation. J. Hazard. Mater. 2018, 344, 849–856. [Google Scholar] [CrossRef]
- Menino, N.M.; Salla, J.D.S.; do Nascimento, M.S.; Dallago, R.M.; Peralta, R.A.; Moreira, R.F.P.M. High-performance hydrophobic magnetic hydrotalcite for selective treatment of oily wastewater. Environ. Technol. 2021, 44, 2003441. [Google Scholar] [CrossRef]
- Lu, Y.; Sathasivam, S.; Song, J.; Crick, C.R.; Carmalt, C.J.; Parkin, I.P. Robust self-cleaning surfaces that function when exposed to either air or oil. Science 2015, 347, 1132–1135. [Google Scholar] [CrossRef]
- Cui, J.; Zhou, Z.; Xie, A.; Wang, Q.; Liu, S.; Lang, J.; Li, C.; Yan, Y.; Dai, J. Facile preparation of grass-like structured NiCo-LDH/PVDF composite membrane for efficient oil-water emulsion separation. J. Membr. Sci. 2019, 573, 226–233. [Google Scholar] [CrossRef]
- Xie, A.; Cui, J.; Yang, J.; Li, C.; Wang, Y.; Dai, J. Active antifouling carbon cloth@Ni-Co LDH/Ag membrane for efficient oil/water separation. Appl. Clay Sci. 2021, 211, 106161. [Google Scholar] [CrossRef]
- Lartey-Young, G.; Ma, L. Optimization, equilibrium, adsorption behaviour of Cu/Zn/Fe LDH and LDHBC composites towards atrazine reclamation in an aqueous environment. Chemosphere 2022, 293, 133526. [Google Scholar] [CrossRef]
- Mourid, E.H.; Lakraimi, M.; Legrouri, A. Removal and release of the 2,4,5-trichlorophenoxyacetic acid herbicide from wastewater by layered double hydroxides. J. Inorg. Organomet. Polym. Mater. 2021, 31, 2116–2128. [Google Scholar] [CrossRef]
- Gobelius, L.; Hedlund, J.; Duerig, W.; Troger, R.; Lilja, K.; Wiberg, K.; Ahrens, L. Per- and polyfluoroalkyl substances in swedish groundwater and surface water: Implications for environmental quality standards and drinking water guidelines. Environ. Sci. Technol. 2018, 52, 4340–4349. [Google Scholar] [CrossRef]
- Chen, Y.; Georgi, A.; Zhang, W.; Kopinke, F.; Yan, J.; Saeidi, N.; Li, J.; Gu, M.; Chen, M. Mechanistic insights into fast adsorption of perfluoroalkyl substances on carbonate-layered double hydroxides. J. Hazard. Mater. 2021, 408, 124815. [Google Scholar] [CrossRef]
- Min, X.; Huo, J.; Dong, Q.; Xu, S.; Wang, Y. Enhanced sorption of perfluorooctanoic acid with organically functionalized layered double hydroxide. Chem. Eng. J. 2022, 446, 137019. [Google Scholar] [CrossRef]
- Tiwari, E.; Singh, N.; Khandelwal, N.; Monikh, F.A.; Darbha, G.K. Application of Zn/Al layered double hydroxides for the removal of nanoscale plastic debris from aqueous systems. J. Hazard. Mater. 2020, 397, 122769. [Google Scholar] [CrossRef]
- Peng, G.; Xiang, M.; Wang, W.; Su, Z.; Liu, H.; Mao, Y.; Chen, Y.; Zhang, P. Engineering 3D graphene-like carbon-assembled layered double oxide for efficient microplastic removal in a wide pH range. J. Hazard. Mater. 2022, 433, 128672. [Google Scholar] [CrossRef]
- Ge, L.; Shao, B.; Liang, Q.; Huang, D.; Liu, Z.; He, Q.; Wu, T.; Luo, S.; Pan, Y.; Zhao, C.; et al. Layered double hydroxide based materials applied in persulfate based advanced oxidation processes: Property, mechanism, application and perspectives. J. Hazard. Mater. 2022, 424, 127612. [Google Scholar] [CrossRef]
- Huang, Y.; Yang, Y.; Wang, X.; Yuan, X.; Pi, N.; Yuan, H.; Liu, X.; Ni, C. Heterogeneous Fenton-like degradation of methoxychlor in water using two different FeS@hydrotalcites (LHDs) and Fe3O4@LHDs catalysts prepared via an in situ growth method. Chem. Eng. J. 2018, 342, 142–154. [Google Scholar] [CrossRef]
- Ba, Y.; Hu, Z.; Bao, P.; Qiao, M.; Hua, J.; Wu, C.; Wu, J. Ferrous ions accelerate sulfide-induced abiotic dechlorination of DDT in waterlogged paddy soil and in soil solution. J. Soil. Sediment 2011, 11, 1209–1220. [Google Scholar] [CrossRef]
- Yan, J.; Chen, Y.; Gao, W.; Chen, Y.; Qian, L.; Han, L.; Chen, M. Catalysis of hydrogen peroxide with Cu layered double hydrotalcite for the degradation of ethylbenzene. Chemosphere 2019, 225, 157–165. [Google Scholar] [CrossRef]
- Wang, H.; Jing, M.; Wu, Y.; Chen, W.; Ran, Y. Effective degradation of phenol via Fenton reaction over CuNiFe layered double hydroxides. J. Hazard. Mater. 2018, 353, 53–61. [Google Scholar] [CrossRef]
- Dou, L.; Zhang, H. Facile assembly of nanosheet array-like CuMgAl-layered double hydroxide/rGO nanohybrids for highly efficient reduction of 4-nitrophenol. J. Mater. Chem. A 2016, 4, 18990–19002. [Google Scholar] [CrossRef]
- Peng, X.; Wang, M.; Hu, F.; Qiu, F.; Zhang, T.; Dai, H.; Cao, Z. Multipath fabrication of hierarchical CuAl layered double hydroxide/carbon fiber composites for the degradation of ammonia nitrogen. J. Environ. Manag. 2018, 220, 173–182. [Google Scholar] [CrossRef]
- Costa-Serge, N.D.M.; Goncalves, R.G.L.; Ramirez-Ubillus, M.A.; Li, C.; Hammer, P.; Chiron, S.; Nogueira, R.F.P. Effect of the interlamellar anion on CuMgFe-LDH in solar photo-Fenton and Fenton-like degradation of the anticancer drug 5-fluorouracil. Appl. Catal. B-Environ. 2022, 315, 121537. [Google Scholar] [CrossRef]
- Dhakshinamoorthy, A.; Navalon, S.; Alvaro, M.; Garcia, H. Metal nanoparticles as heterogeneous Fenton catalysts. ChemSusChem 2012, 5, 46–64. [Google Scholar] [CrossRef]
- Wang, A.; Chen, Y.; Zheng, Z.; Wang, H.; Li, X.; Yang, Z.; Qiu, R.; Yan, K. In situ N-doped carbon-coated mulberry-like cobalt manganese oxide boosting for visible light driving photocatalytic degradation of pharmaceutical pollutants. Chem. Eng. J. 2021, 411, 128497. [Google Scholar] [CrossRef]
- Yang, Z.; Li, X.; Huang, Y.; Chen, Y.; Wang, A.; Wang, Y.; Li, C.; Hu, Z.; Yan, K. Facile synthesis of cobalt-iron layered double hydroxides nanosheets for direct activation of peroxymonosulfate (PMS) during degradation of fluoroquinolones antibiotics. J. Clean. Prod. 2021, 310, 127584. [Google Scholar] [CrossRef]
- Ren, Y.; Lin, L.; Ma, J.; Yang, J.; Feng, J.; Fan, Z. Sulfate radicals induced from peroxymonosulfate by magnetic ferrospinel MFe2O4 (M = Co, Cu, Mn, and Zn) as heterogeneous catalysts in the water. Appl. Catal. B-Environ. 2015, 165, 572–578. [Google Scholar] [CrossRef]
- Vigneshwaran, S.; Jun, B.M.; Prabhu, S.M.; Elanchezhiyan, S.S.; Ok, Y.S.; Meenakshi, S.; Park, C.M. Enhanced sonophotocatalytic degradation of bisphenol a using bimetal sulfide-intercalated MXenes, 2D/2D nanocomposite. Sep. Purif. Technol. 2020, 250, 117178. [Google Scholar] [CrossRef]
- Gong, C.; Chen, F.; Yang, Q.; Luo, K.; Yao, F.; Wang, S.; Wang, X.; Wu, J.; Li, X.; Wang, D.; et al. Heterogeneous activation of peroxymonosulfate by Fe-Co layered doubled hydroxide for efficient catalytic degradation of Rhoadmine B. Chem. Eng. J. 2017, 321, 222–232. [Google Scholar] [CrossRef]
- Li, X.; Hou, T.; Yan, L.; Shan, L.; Meng, X.; Zhao, Y. Efficient degradation of tetracycline by CoFeLa-layered double hydroxides catalyzed peroxymonosulfate: Synergistic effect of radical and nonradical pathways. J. Hazard. Mater. 2020, 398, 122884. [Google Scholar] [CrossRef]
- Deng, J.; Xiao, L.; Yuan, S.; Wang, W.; Zhan, X.; Hu, Z.-H. Activation of peroxymonosulfate by CoFeNi layered double hydroxide/graphene oxide (LDH/GO) for the degradation of gatifloxacin. Sep. Purif. Technol. 2021, 255, 117685. [Google Scholar] [CrossRef]
- Ye, Q.; Wu, J.; Wu, P.; Wang, J.; Niu, W.; Yang, S.; Chen, M.; Rehman, S.; Zhu, N. Enhancing peroxymonosulfate activation of Fe-Al layered double hydroxide by dissolved organic matter: Performance and mechanism. Water Res. 2020, 185, 116246. [Google Scholar] [CrossRef]
- Ma, Q.; Nengzi, L.C.; Li, B.; Wang, Z.; Liu, L.; Cheng, X. Heterogeneously catalyzed persulfate with activated carbon coated with CoFe layered double hydroxide (AC@CoFe-LDH) for the degradation of lomefloxacin. Sep. Purif. Technol. 2020, 235, 116204. [Google Scholar] [CrossRef]
- Jawad, A.; Lang, J.; Liao, Z.; Khan, A.; Ifthikar, J.; Lv, Z.; Long, S.; Chen, Z.; Chen, Z. Activation of persulfate by CuOx@Co-LDH: A novel heterogeneous system for contaminant degradation with broad pH window and controlled leaching. Chem. Eng. J. 2018, 335, 548–559. [Google Scholar] [CrossRef]
- Zeng, H.; Deng, L.; Zhang, H.; Zhou, C.; Shi, Z. Development of oxygen vacancies enriched CoAl hydroxide@hydroxysulfide hollow flowers for peroxymonosulfate activation: A highly efficient singlet oxygen-dominated oxidation process for sulfamethoxazole degradation. J. Hazard. Mater. 2020, 400, 123297. [Google Scholar] [CrossRef]
- Song, A.; Yang, W.; Yang, W.; Sun, G.; Yin, X.; Gao, L.; Wang, Y.; Qin, X.; Shao, G. Facile synthesis of cobalt nanoparticles entirely encapsulated in slim nitrogen-doped carbon nanotubes as oxygen reduction catalyst. ACS Sustain. Chem. Eng. 2017, 5, 3973–3981. [Google Scholar] [CrossRef]
- Pfeifer, V.; Jones, T.E.; Velez, J.J.V.; Arrigo, R.; Piccinin, S.; Haevecker, M.; Knop-Gericke, A.; Schloegl, R. In situ observation of reactive oxygen species forming on oxygen-evolving iridium surfaces. Chem. Sci. 2017, 8, 2143–2149. [Google Scholar] [CrossRef]
- Zheng, R.; Zhao, C.; Xiong, J.; Teng, X.; Chen, W.; Hu, Z.; Chen, Z. Construction of a hierarchically structured, NiCo-Cu-based trifunctional electrocatalyst for efficient overall water splitting and 5-hydroxymethylfurfural oxidation. Sustain. Energy Fuels 2021, 5, 4023–4031. [Google Scholar] [CrossRef]
- Yang, L.; Liu, Z.; Zhu, S.; Feng, L.; Xing, W. Ni-based layered double hydroxide catalysts for oxygen evolution reaction. Mater. Today Phys. 2021, 16, 100292. [Google Scholar] [CrossRef]
- Liu, X.; Fan, X.; Huang, H.; Lin, H.; Gao, J. Electronic modulation of oxygen evolution on metal doped NiFe layered double hydroxides. J. Colloid Interface Sci. 2021, 587, 385–392. [Google Scholar] [CrossRef]
- Ansari, S.M.; Khan, M.Z.; Anwar, H.; Ikram, M.; Sarfraz, Z.; Alam, N.; Khan, Y. Tungsten oxide-reduced graphene oxide composites for photoelectrochemical water splitting. Arab. J. Sci. Eng. 2021, 46, 813–825. [Google Scholar] [CrossRef]
- Xu, J.; Gai, S.; He, F.; Niu, N.; Gao, P.; Chen, Y.; Yang, P. A sandwich-type three-dimensional layered double hydroxide nanosheet array/graphene composite: Fabrication and high supercapacitor performance. J. Mater. Chem. A 2014, 2, 1022–1031. [Google Scholar] [CrossRef]
- Duan, M.; Qiu, M.; Sun, S.; Guo, X.; Liu, Y.; Zheng, X.; Cao, F.; Kong, Q.; Zhang, J. Intercalating assembly of NiFe LDH nanosheets/CNTs composite as high-performance electrocatalyst for oxygen evolution reaction. Appl. Clay Sci. 2022, 216, 106360. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, Y.; Fu, X.; Song, W.; Li, Y.; Li, X.; Yan, L. Recent Progress of Layered Double Hydroxide-Based Materials in Wastewater Treatment. Materials 2023, 16, 5723. https://doi.org/10.3390/ma16165723
Fu Y, Fu X, Song W, Li Y, Li X, Yan L. Recent Progress of Layered Double Hydroxide-Based Materials in Wastewater Treatment. Materials. 2023; 16(16):5723. https://doi.org/10.3390/ma16165723
Chicago/Turabian StyleFu, Yanli, Xiaoqian Fu, Wen Song, Yanfei Li, Xuguang Li, and Liangguo Yan. 2023. "Recent Progress of Layered Double Hydroxide-Based Materials in Wastewater Treatment" Materials 16, no. 16: 5723. https://doi.org/10.3390/ma16165723
APA StyleFu, Y., Fu, X., Song, W., Li, Y., Li, X., & Yan, L. (2023). Recent Progress of Layered Double Hydroxide-Based Materials in Wastewater Treatment. Materials, 16(16), 5723. https://doi.org/10.3390/ma16165723