State of the Art in Development of Heat Exchanger Geometry Optimization and Different Storage Bed Designs of a Metal Hydride Reactor
Abstract
1. Introduction
2. Heat Transfer Issues
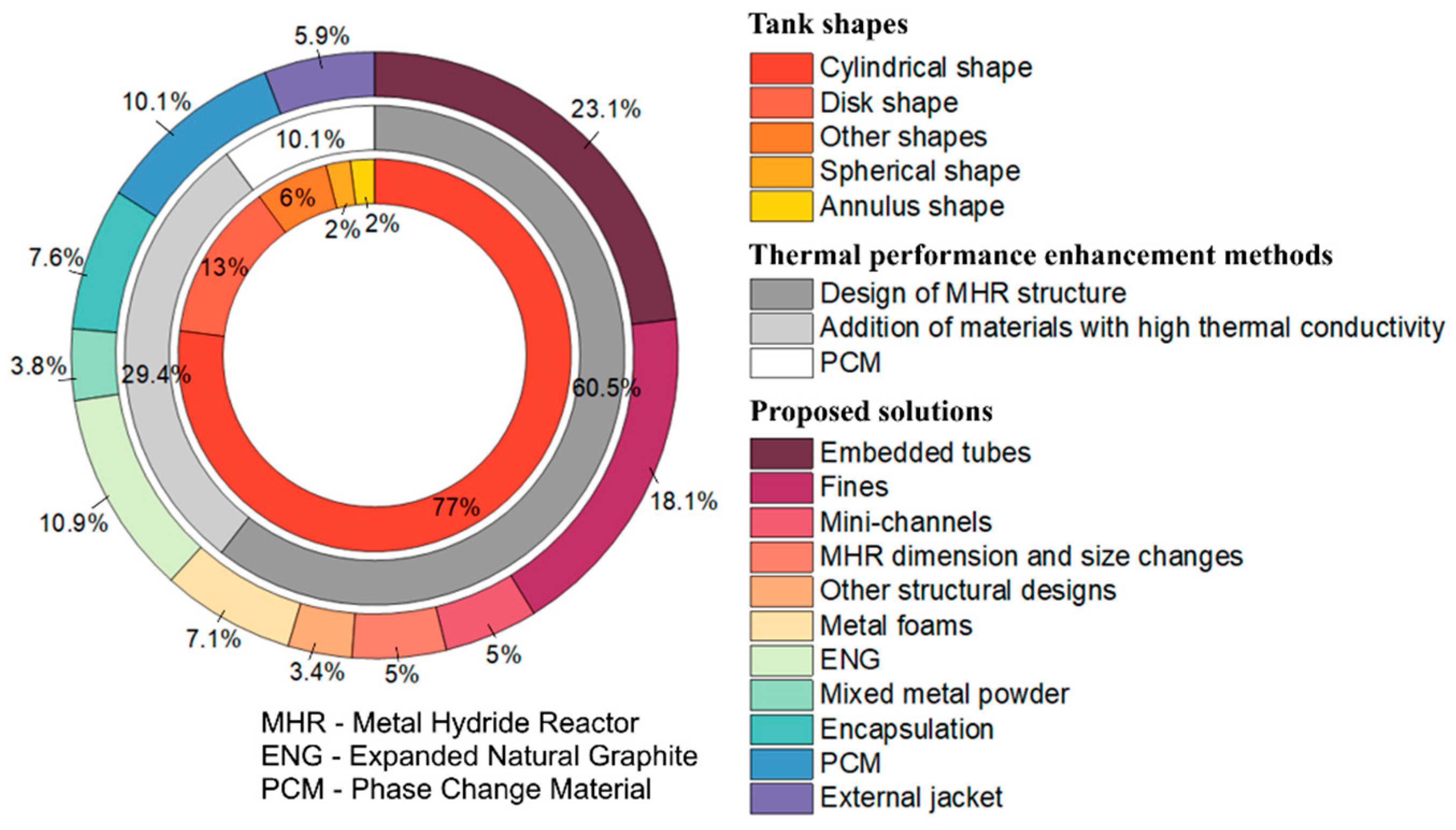
3. Metal Hydride Reactor Types and Shapes
4. Metal Hydride Bed Modification
5. Heat Exchangers
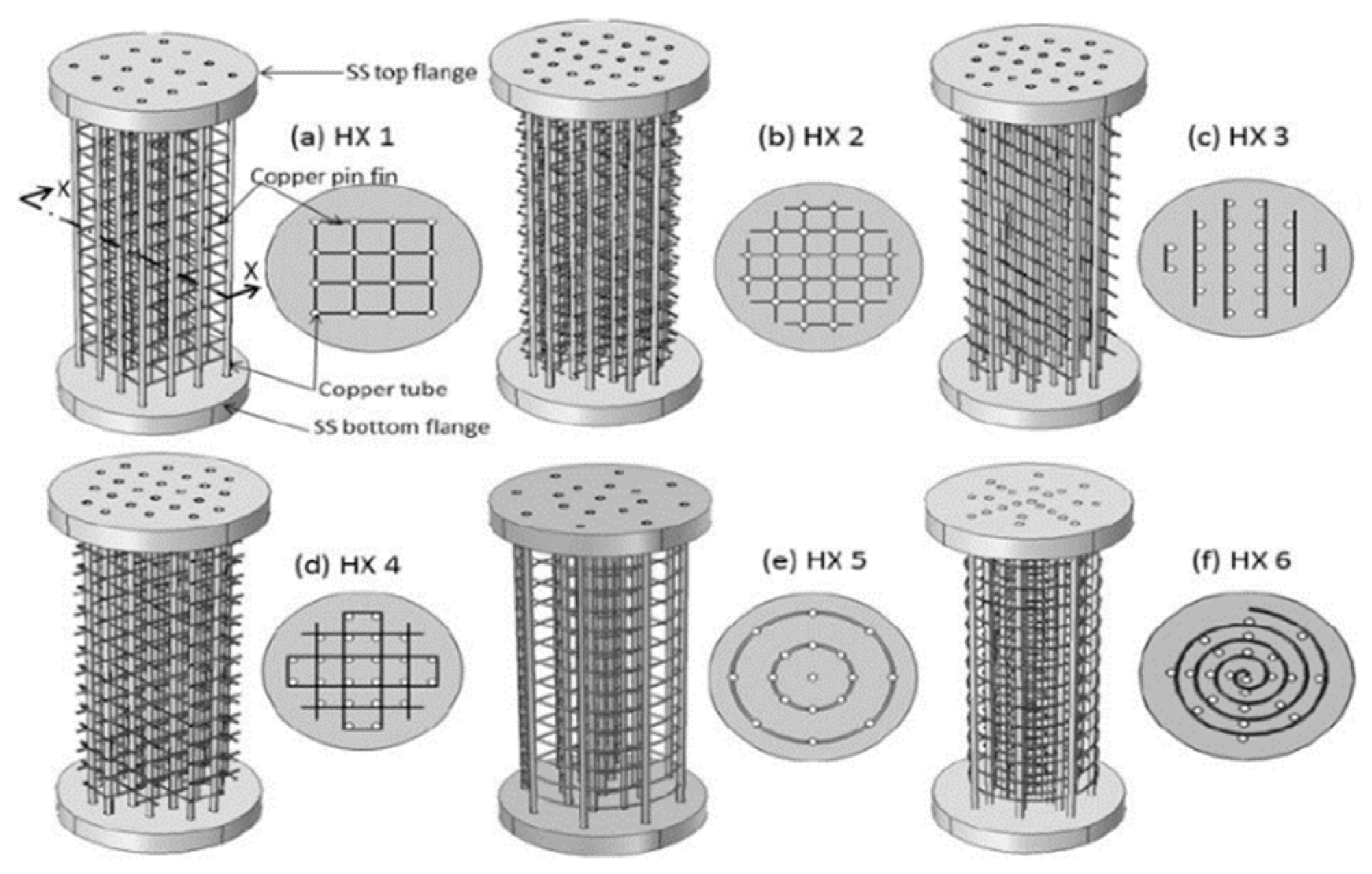
5.1. Design and Number of Cooling Tubes
5.2. Design and Number of Fins
5.3. Other Heat Exchanger Designs
6. Additive Manufacturing for Heat Exchangers
7. Summary
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lee, S.Y.; Lee, J.H.; Kim, Y.H.; Kim, J.W.; Lee, K.J.; Park, S.J. Recent Progress Using Solid-State Materials for Hydrogen Storage: A Short Review. Processes 2022, 10, 304. [Google Scholar] [CrossRef]
- Usman, M.R. Hydrogen Storage Methods: Review and Current Status. Renew. Sustain. Energy Rev. 2022, 167, 112743. [Google Scholar] [CrossRef]
- Rodríguez-Segade, M.; Hernández, S.; Díaz, J. Multi-Bubble Scheme and Structural Analysis of a Hypersonic Stratospheric Flight Vehicle. Aerosp. Sci. Technol. 2022, 124, 107514. [Google Scholar] [CrossRef]
- Barthelemy, H.; Weber, M.; Barbier, F. Hydrogen Storage: Recent Improvements and Industrial Perspectives. Int. J. Hydrogen Energy 2017, 42, 7254–7262. [Google Scholar] [CrossRef]
- Tarhan, C.; Çil, M.A. A Study on Hydrogen, the Clean Energy of the Future: Hydrogen Storage Methods. J. Energy Storage 2021, 40, 102676. [Google Scholar] [CrossRef]
- Valenti, G. Hydrogen Liquefaction and Liquid Hydrogen Storage. Compend. Hydrog. Energy 2016, 2, 27–51. [Google Scholar] [CrossRef]
- Biniwale, R.; Rayalu, S.; Devotta, S.; Ichikawa, M. Chemical Hydrides: A Solution to High Capacity Hydrogen Storage and Supply. Int. J. Hydrogen Energy 2008, 33, 360–365. [Google Scholar] [CrossRef]
- Mohan, M.; Sharma, V.K.; Kumar, E.A.; Gayathri, V. Hydrogen Storage in Carbon Materials—A Review. Energy Storage 2019, 1, e35. [Google Scholar] [CrossRef]
- Rowsell, J.L.C.; Yaghi, O.M. Strategies for Hydrogen Storage in Metal-Organic Frameworks. Angew. Chem. Int. Ed. 2005, 44, 4670–4679. [Google Scholar] [CrossRef]
- Dong, J.; Wang, X.; Xu, H.; Zhao, Q.; Li, J. Hydrogen Storage in Several Microporous Zeolites. Int. J. Hydrogen Energy 2007, 32, 4998–5004. [Google Scholar] [CrossRef]
- Lyu, J.; Elman, R.R.; Svyatkin, L.A.; Kudiiarov, V.N. Theoretical and Experimental Research of Hydrogen Solid Solution in Mg and Mg-Al System. Materials 2022, 15, 1667. [Google Scholar] [CrossRef] [PubMed]
- Sakintuna, B.; Lamaridarkrim, F.; Hirscher, M. Metal Hydride Materials for Solid Hydrogen Storage: A Review. Int. J. Hydrogen Energy 2007, 32, 1121–1140. [Google Scholar] [CrossRef]
- Todorova, S.; Abrashev, B.; Rangelova, V.; Mihaylov, L.; Vassileva, E.; Petrov, K.; Spassov, T. Hydrogen Gas Phase and Electrochemical Hydriding of LaNi5−xMx (M = Sn, Co, Al) Alloys. Materials 2020, 14, 14. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Peng, Y.; Zhang, Q. Growth Kinetics of MgH2 Nanocrystallites Prepared by Ball Milling. J. Mater. Sci. Technol. 2020, 50, 178–183. [Google Scholar] [CrossRef]
- Schlapbach, L.; Züttel, A. Hydrogen-Storage Materials for Mobile Applications. Nature 2001, 414, 353–358. [Google Scholar] [CrossRef]
- Hou, Q.; Yang, X.; Zhang, J. Review on Hydrogen Storage Performance of MgH2: Development and Trends. ChemistrySelect 2021, 6, 1589–1606. [Google Scholar] [CrossRef]
- Zhang, X.L.; Liu, Y.F.; Zhang, X.; Hu, J.J.; Gao, M.X.; Pan, H.G. Empowering Hydrogen Storage Performance of MgH2 by Nanoengineering and Nanocatalysis. Mater. Today Nano 2020, 9, 100064. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, J.; Hou, Q.; Guo, X.; Xu, G. Improvement of Mg-Based Hydrogen Storage Materials by Metal Catalysts: Review and Summary. ChemistrySelect 2021, 6, 8809–8829. [Google Scholar] [CrossRef]
- Guo, Z.X.; Shang, C.; Aguey-Zinsou, K.F. Materials Challenges for Hydrogen Storage. J. Eur. Ceram. Soc. 2008, 28, 1467–1473. [Google Scholar] [CrossRef]
- Kikkinides, E.; Georgiadis, M.; Stubos, A. On the Optimization of Hydrogen Storage in Metal Hydride Beds. Int. J. Hydrogen Energy 2006, 31, 737–751. [Google Scholar] [CrossRef]
- Shafiee, S.; McCay, M.H. Different Reactor and Heat Exchanger Configurations for Metal Hydride Hydrogen Storage Systems—A Review. Int. J. Hydrogen Energy 2016, 41, 9462–9470. [Google Scholar] [CrossRef]
- Afzal, M.; Mane, R.; Sharma, P. Heat Transfer Techniques in Metal Hydride Hydrogen Storage: A Review. Int. J. Hydrogen Energy 2017, 42, 30661–30682. [Google Scholar] [CrossRef]
- Cui, Y.; Zeng, X.; Xiao, J.; Kou, H. The Comprehensive Review for Development of Heat Exchanger Configuration Design in Metal Hydride Bed. Int. J. Hydrogen Energy 2022, 47, 2461–2490. [Google Scholar] [CrossRef]
- Nguyen, H.Q.; Shabani, B. Review of Metal Hydride Hydrogen Storage Thermal Management for Use in the Fuel Cell Systems. Int. J. Hydrogen Energy 2021, 46, 31699–31726. [Google Scholar] [CrossRef]
- Dong, Z.; Wang, Y.; Wu, H.; Zhang, X.; Sun, Y.; Li, Y.; Chang, J.; He, Z.; Hong, J. A Design Methodology of Large-Scale Metal Hydride Reactor Based on Schematization for Hydrogen Storage. J. Energy Storage 2022, 49, 104047. [Google Scholar] [CrossRef]
- Miao, G.; Li, P.; Liu, C.; Liu, Y.; Zhang, H.; Lin, F.; Qu, X. Review of Thermal Management Technology for Metal Hydride Reaction Beds. Sustain. Energy Fuels 2023, 7, 2025–2041. [Google Scholar] [CrossRef]
- Katamala, M.; Dutta, P.; Srinivasa Murthy, S. Applications of Metal Hydride Based Thermal Systems: A Review. SSRN Electron. J. 2022, 229. [Google Scholar] [CrossRef]
- Kukkapalli, V.K.; Kim, S.; Thomas, S.A. Thermal Management Techniques in Metal Hydrides for Hydrogen Storage Applications: A Review. Energies 2023, 16, 3444. [Google Scholar] [CrossRef]
- Ho, J.Y.; Leong, K.C.; Wong, T.N. Additively-Manufactured Metallic Porous Lattice Heat Exchangers for Air-Side Heat Transfer Enhancement. Int. J. Heat Mass Transf. 2020, 150, 119262. [Google Scholar] [CrossRef]
- Chekurov, S.; Kajaste, J.; Saari, K.; Kauranne, H.; Pietola, M.; Partanen, J. Additively Manufactured High-Performance Counterflow Heat Exchanger. Prog. Addit. Manuf. 2018, 4, 55–61. [Google Scholar] [CrossRef]
- Roccetti Campagnoli, M.; Galati, M.; Saboori, A. On the Processability of Copper Components via Powder-Based Additive Manufacturing Processes: Potentials, Challenges and Feasible Solutions. J. Manuf. Process. 2021, 72, 320–337. [Google Scholar] [CrossRef]
- McDonough, J.R. A Perspective on the Current and Future Roles of Additive Manufacturing in Process Engineering, with an Emphasis on Heat Transfer. Therm. Sci. Eng. Prog. 2020, 19, 100594. [Google Scholar] [CrossRef]
- Niknam, S.A.; Mortazavi, M.; Li, D. Additively Manufactured Heat Exchangers: A Review on Opportunities and Challenges. Int. J. Adv. Manuf. Technol. 2020, 112, 601–618. [Google Scholar] [CrossRef]
- Scheithauer, U.; Kordaß, R.; Noack, K.; Eichenauer, M.F.; Hartmann, M.; Abel, J.; Ganzer, G.; Lordick, D. Potentials and Challenges of Additive Manufacturing Technologies for Heat Exchanger. In Advances in Heat Exchangers; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Visaria, M.; Mudawar, I.; Pourpoint, T.; Kumar, S. Study of Heat Transfer and Kinetics Parameters Influencing the Design of Heat Exchangers for Hydrogen Storage in High-Pressure Metal Hydrides. Int. J. Heat Mass Transf. 2010, 53, 2229–2239. [Google Scholar] [CrossRef]
- Mazzucco, A.; Dornheim, M.; Sloth, M.; Jensen, T.R.; Jensen, J.O.; Rokni, M. Bed Geometries, Fueling Strategies and Optimization of Heat Exchanger Designs in Metal Hydride Storage Systems for Automotive Applications: A Review. Int. J. Hydrogen Energy 2014, 39, 17054–17074. [Google Scholar] [CrossRef]
- Dunikov, D.O.; Borzenko, V.I. Heat and Mass Transfer Crisis in a Metal Hydride Accumulator. J. Phys. Conf. Ser. 2020, 1683, 052018. [Google Scholar] [CrossRef]
- Goodell, P.D. Thermal Conductivity of Hydriding Alloy Powders and Comparisons of Reactor Systems. J. Less Common Met. 1980, 74, 175–184. [Google Scholar] [CrossRef]
- Jiao, K.; Li, X.; Yin, Y.; Zhou, Y.; Yu, S.; Du, Q. Effects of Various Operating Conditions on the Hydrogen Absorption Processes in a Metal Hydride Tank. Appl. Energy 2012, 94, 257–269. [Google Scholar] [CrossRef]
- Sreeraj, R.; Aadhithiyan, A.K.; Anbarasu, S. Integration of Thermal Augmentation Methods in Hydride Beds for Metal Hydride Based Hydrogen Storage Systems: Review and Recommendation. J. Energy Storage 2022, 52, 105039. [Google Scholar] [CrossRef]
- Qin, F.; Chen, J.; Lu, M.; Chen, Z.; Zhou, Y.; Yang, K. Development of a Metal Hydride Refrigeration System as an Exhaust Gas-Driven Automobile Air Conditioner. Renew. Energy 2007, 32, 2034–2052. [Google Scholar] [CrossRef]
- Veerraju, C.; Gopal, M.R. Heat and Mass Transfer Studies on Elliptical Metal Hydride Tubes and Tube Banks. Int. J. Hydrogen Energy 2009, 34, 4340–4350. [Google Scholar] [CrossRef]
- Muthukumar, P.; Prakashmaiya, M.; Srinivasamurthy, S. Experiments on a Metal Hydride Based Hydrogen Compressor. Int. J. Hydrogen Energy 2005, 30, 879–892. [Google Scholar] [CrossRef]
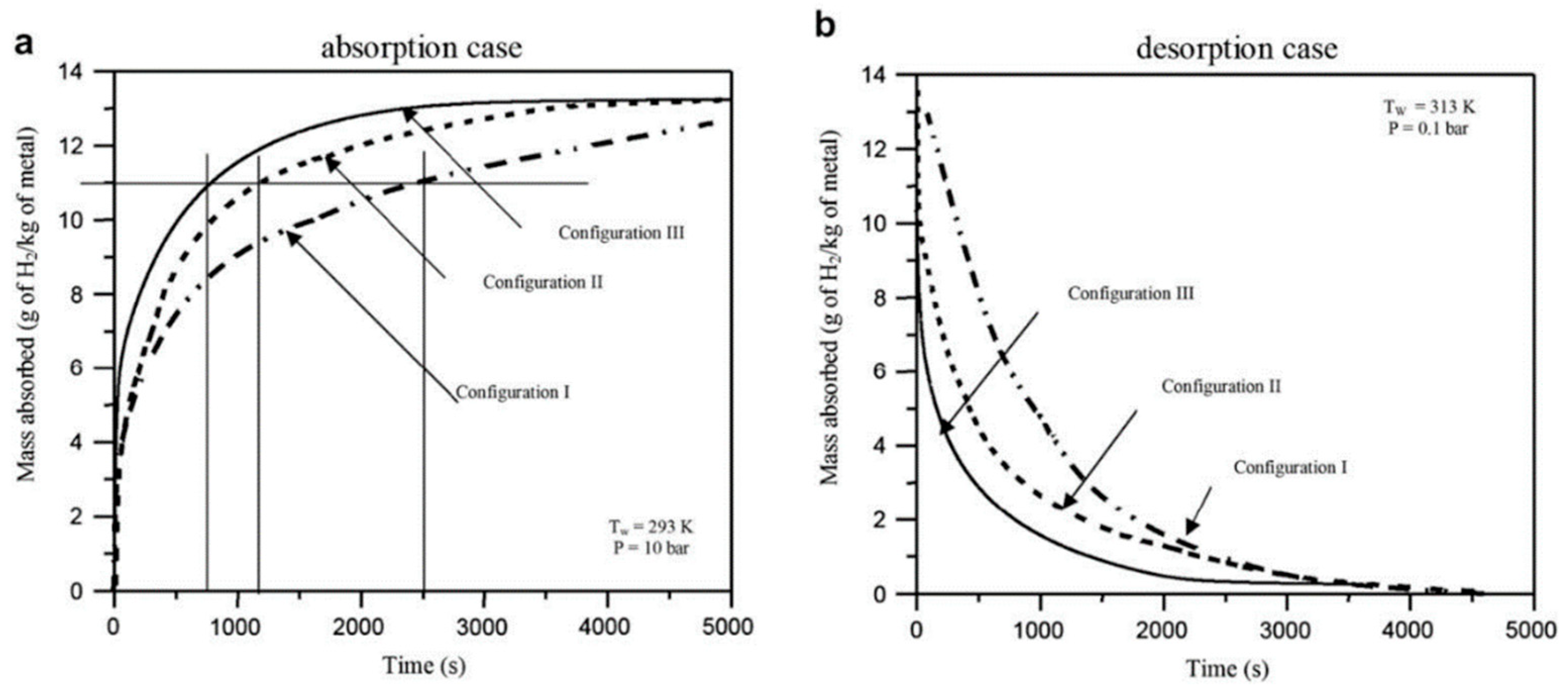
- Lozano, G.A.; Ranong, C.N.; Bellosta von Colbe, J.M.; Bormann, R.; Hapke, J.; Fieg, G.; Klassen, T.; Dornheim, M. Optimization of Hydrogen Storage Tubular Tanks Based on Light Weight Hydrides. Int. J. Hydrogen Energy 2012, 37, 2825–2834. [Google Scholar] [CrossRef]
- Komiya, K.; Daigoro, M.; Shinpei, M.; Norihiko, H.; Kousei, Y.; Shintaro, W.; Keiji, T.; Hidehito, K.; Mituo, M.; Seiichiro, M.; et al. High-Pressure Hydrogen-Absorbing Alloy Tank for Fuel Cell Vehicles. In SAE Technical Paper Series; SAE: Warrendale, PA, USA, 2010. [Google Scholar] [CrossRef]
- Blinov, D.V.; Borzenko, V.I.; Bezdudny, A.V.; Kuleshov, N.V. Prospective Metal Hydride Hydrogen Storage and Purification Technologies. Power Eng. Res. Equip. Technol. 2021, 23, 149–160. [Google Scholar] [CrossRef]
- Lototskyy, M.; Linkov, V. Thermally Driven Hydrogen Compression Using Metal Hydrides. Int. J. Energy Res. 2022, 46, 22049–22069. [Google Scholar] [CrossRef]
- Lototskyy, M.V.; Yartys, V.A.; Pollet, B.G.; Bowman, R.C. Metal Hydride Hydrogen Compressors: A Review. Int. J. Hydrogen Energy 2014, 39, 5818–5851. [Google Scholar] [CrossRef]
- Lototskyy, M.V.; Davids, M.W.; Tolj, I.; Klochko, Y.V.; Sekhar, B.S.; Chidziva, S.; Smith, F.; Swanepoel, D.; Pollet, B.G. Metal Hydride Systems for Hydrogen Storage and Supply for Stationary and Automotive Low Temperature PEM Fuel Cell Power Modules. Int. J. Hydrogen Energy 2015, 40, 11491–11497. [Google Scholar] [CrossRef]
- Mellouli, S.; Ben Khedher, N.; Askri, F.; Jemni, A.; Ben Nasrallah, S. Numerical Analysis of Metal Hydride Tank with Phase Change Material. Appl. Therm. Eng. 2015, 90, 674–682. [Google Scholar] [CrossRef]
- Boukhari, A.; Bessai’h, R. Numerical Investigation of Hydrogen Absorption in an Annulus-Disc Metal Hydride Reactor. In Proceedings of the 2014 5th International Renewable Energy Congress (IREC), Hammamet, Tunisia, 25–27 March 2014. [Google Scholar] [CrossRef]
- Ye, Y.; Ding, J.; Wang, W.; Yan, J. The Storage Performance of Metal Hydride Hydrogen Storage Tanks with Reaction Heat Recovery by Phase Change Materials. Appl. Energy 2021, 299, 117255. [Google Scholar] [CrossRef]
- Zhang, J.; Fisher, T.S.; Ramachandran, P.V.; Gore, J.P.; Mudawar, I. A Review of Heat Transfer Issues in Hydrogen Storage Technologies. J. Heat Transf. 2005, 127, 1391–1399. [Google Scholar] [CrossRef]
- Ron, M.; Gruen, D.; Mendelsohn, M.; Sheet, I. Preparation and Properties of Porous Metal Hydride Compacts. J. Less Common Met. 1980, 74, 445–448. [Google Scholar] [CrossRef]
- Bershadsky, E.; Josephy, Y.; Ron, M. Permeability and Thermal Conductivity of Porous Metallic Matrix Hydride Compacts. J. Less Common Met. 1989, 153, 65–78. [Google Scholar] [CrossRef]
- Kim, K.J.; Lloyd, G.M.; Feldman, K.T.; Razani, A. Thermal Analysis of the Ca0.4Mm0.6Ni5 Metal–Hydride Reactor. Appl. Therm. Eng. 1998, 18, 1325–1336. [Google Scholar] [CrossRef]
- Kim, K. Performance of High Power Metal Hydride Reactors. Int. J. Hydrogen Energy 1998, 23, 355–362. [Google Scholar] [CrossRef]
- Kim, K.J.; Lloyd, G.; Razani, A.; Feldman, K.T. Development of LaNi5/Cu/Sn Metal Hydride Powder Composites. Powder Technol. 1998, 99, 40–45. [Google Scholar] [CrossRef]
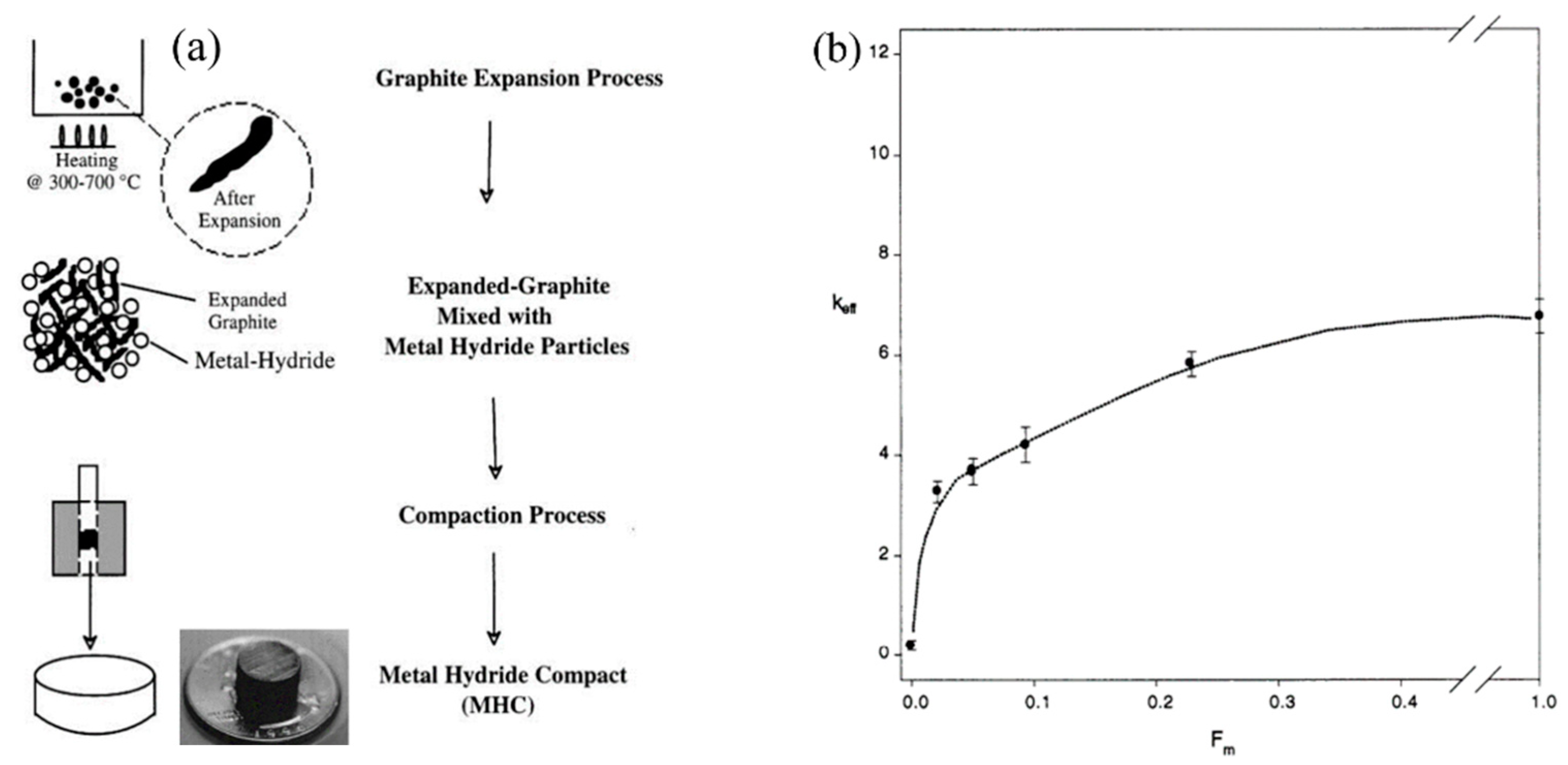
- Sánchez, A.R.; Klein, H.P.; Groll, M. Expanded Graphite as Heat Transfer Matrix in Metal Hydride Beds. Int. J. Hydrogen Energy 2003, 28, 515–527. [Google Scholar] [CrossRef]
- Klein, H.P.; Groll, M. Heat Transfer Characteristics of Expanded Graphite Matrices in Metal Hydride Beds. Int. J. Hydrogen Energy 2004, 29, 1503–1511. [Google Scholar] [CrossRef]
- Deng, C.; Shi, P.; Zhang, S. Effect of Surface Modification on the Electrochemical Performances of LaNi5 Hydrogen Storage Alloy in Ni/MH Batteries. Mater. Chem. Phys. 2006, 98, 514–518. [Google Scholar] [CrossRef]
- Romanov, I.A.; Borzenko, V.I.; Kazakov, A.N. Influence of High Thermal Conductivity Addition on PCT-Isotherms of Hydrogen Storage Alloy. J. Phys. Conf. Ser. 2018, 1128, 012105. [Google Scholar] [CrossRef]
- Atalmis, G.; Demiralp, M.; Yelegen, N.; Kaplan, Y. The Effect of Copper Coated Metal Hydride at Different Ratios on the Reaction Kinetics. Int. J. Hydrogen Energy 2022, 48, 23067–23076. [Google Scholar] [CrossRef]
- Kim, K.J.; Montoya, B.; Razani, A.; Lee, K.-H. Metal Hydride Compacts of Improved Thermal Conductivity. Int. J. Hydrogen Energy 2001, 26, 609–613. [Google Scholar] [CrossRef]
- Pentimalli, M.; Frazzica, A.; Freni, A.; Imperi, E.; Padella, F. Metal Hydride-Based Composite Materials with Improved Thermal Conductivity and Dimensional Stability Properties. In 5th Forum on New Materials Part A; Trans Tech Publications Ltd.: Bäch, Switzerland, 2010. [Google Scholar] [CrossRef]
- Chaise, A.; de Rango, P.; Marty, P.; Fruchart, D.; Miraglia, S.; Olivès, R.; Garrier, S. Enhancement of Hydrogen Sorption in Magnesium Hydride Using Expanded Natural Graphite. Int. J. Hydrogen Energy 2009, 34, 8589–8596. [Google Scholar] [CrossRef]
- Pohlmann, C.; Röntzsch, L.; Kalinichenka, S.; Hutsch, T.; Kieback, B. Magnesium Alloy-Graphite Composites with Tailored Heat Conduction Properties for Hydrogen Storage Applications. Int. J. Hydrogen Energy 2010, 35, 12829–12836. [Google Scholar] [CrossRef]
- Madaria, Y.; Anil Kumar, E. Measurement and Augmentation of Effective Thermal Conductivity of La0.8Ce0.2Ni5 Hydride Bed. J. Alloys Compd. 2017, 691, 442–451. [Google Scholar] [CrossRef]
- Delhomme, B.; de Rango, P.; Marty, P.; Bacia, M.; Zawilski, B.; Raufast, C.; Miraglia, S.; Fruchart, D. Large Scale Magnesium Hydride Tank Coupled with an External Heat Source. Int. J. Hydrogen Energy 2012, 37, 9103–9111. [Google Scholar] [CrossRef]
- Singh, U.R.; Bhogilla, S. Performance Analysis of LaNi5 Added with Expanded Natural Graphite for Hydrogen Storage System. Int. J. Hydrogen Energy 2022, 48, 21466–21475. [Google Scholar] [CrossRef]
- Dieterich, M.; Pohlmann, C.; Bürger, I.; Linder, M.; Röntzsch, L. Long-Term Cycle Stability of Metal Hydride-Graphite Composites. Int. J. Hydrogen Energy 2015, 40, 16375–16382. [Google Scholar] [CrossRef]
- Park, C.S.; Jung, K.; Jeong, S.U.; Kang, K.S.; Lee, Y.H.; Park, Y.-S.; Park, B.H. Development of Hydrogen Storage Reactor Using Composite of Metal Hydride Materials with ENG. Int. J. Hydrogen Energy 2020, 45, 27434–27442. [Google Scholar] [CrossRef]
- Bao, Z.; Yan, D.; Zhu, Z.; Wu, D. Performance Investigation of Metal Hydride Reactors Adopting Multilayer Bed with Graded Content of Expanded Natural Graphite for Thermochemical Heat Storage. Appl. Therm. Eng. 2021, 188, 116602. [Google Scholar] [CrossRef]
- Zhu, Z.; Bao, Z.; Wu, D. Optimization of the Content Distribution of Expanded Natural Graphite in a Multilayer Metal Hydride Bed for Thermochemical Heat Storage. Appl. Therm. Eng. 2022, 216, 119115. [Google Scholar] [CrossRef]
- Popilevsky, L.; Skripnyuk, V.M.; Beregovsky, M.; Sezen, M.; Amouyal, Y.; Rabkin, E. Hydrogen Storage and Thermal Transport Properties of Pelletized Porous Mg-2 Wt.% Multiwall Carbon Nanotubes and Mg-2 Wt.% Graphite Composites. Int. J. Hydrogen Energy 2016, 41, 14461–14474. [Google Scholar] [CrossRef]
- Klein, H.-P.; Groll, M. Development of a Two-Stage Metal Hydride System as Topping Cycle in Cascading Sorption Systems for Cold Generation. Appl. Therm. Eng. 2002, 22, 631–639. [Google Scholar] [CrossRef]
- Mayer, U.; Groll, M.; Supper, W. Heat and Mass Transfer in Metal Hydride Reaction Beds: Experimental and Theoretical Results. J. Less Common Met. 1987, 131, 235–244. [Google Scholar] [CrossRef]
- Supper, W.; Groll, M.; Mayer, U. Reaction Kinetics in Metal Hydride Reaction Beds with Improved Heat and Mass Transfer. J. Less Common Met. 1984, 104, 279–286. [Google Scholar] [CrossRef]
- Bjurstrom, H.; Komazaki, Y.; Suda, S. The Dynamics of Hydrogen Transfer in a Metal Hydride Heat Pump. J. Less Common Met. 1987, 131, 225–234. [Google Scholar] [CrossRef]
- Laurencelle, F.; Dehouche, Z.; Morin, F.; Goyette, J. Experimental Study on a Metal Hydride Based Hydrogen Compressor. J. Alloys Compd. 2009, 475, 810–816. [Google Scholar] [CrossRef]
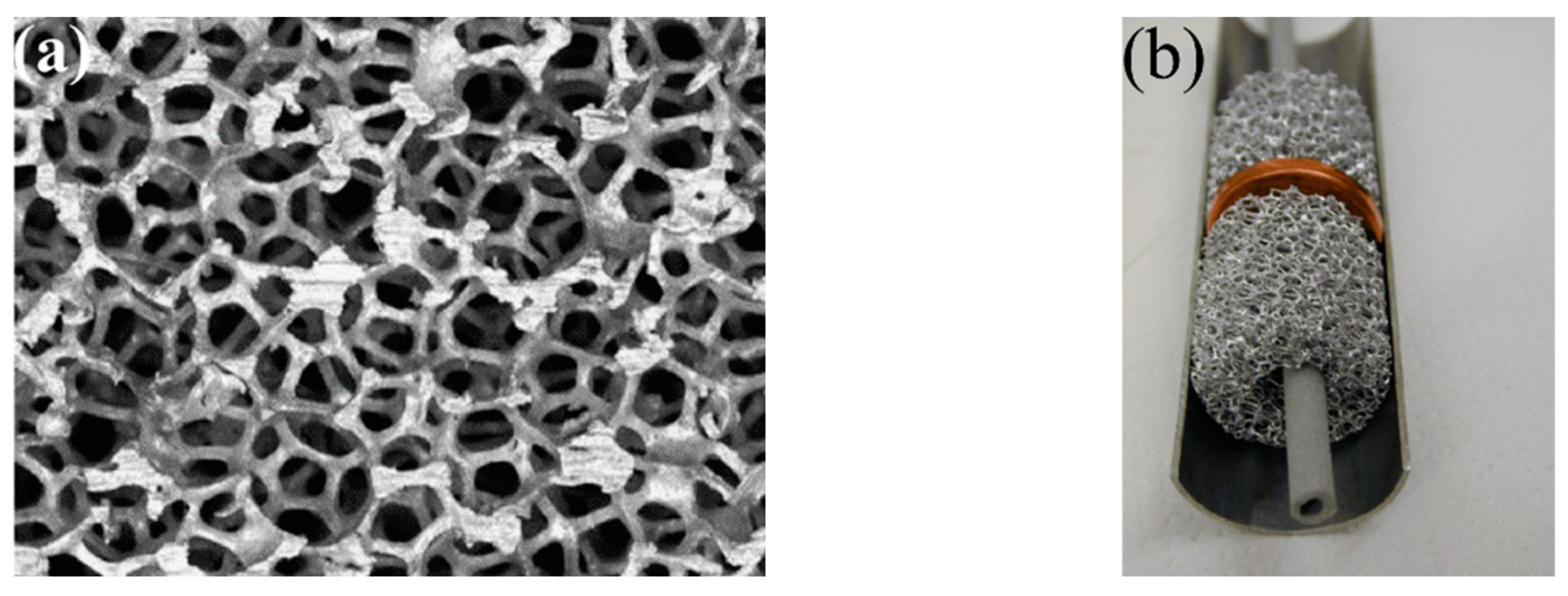
- Laurencelle, F.; Goyette, J. Simulation of Heat Transfer in a Metal Hydride Reactor with Aluminium Foam. Int. J. Hydrogen Energy 2007, 32, 2957–2964. [Google Scholar] [CrossRef]
- Minko, K.B.; Artemov, V.I.; Yan’kov, G.G. Numerical Study of Hydrogen Purification Using Metal Hydride Reactor with Aluminium Foam. Appl. Therm. Eng. 2015, 76, 175–184. [Google Scholar] [CrossRef]
- Suda, S.; Komazaki, Y.; Kobayashi, N. Effective Thermal Conductivity of Metal Hydride Beds. J. Less Common Met. 1983, 89, 317–324. [Google Scholar] [CrossRef]
- Mellouli, S.; Dhaou, H.; Askri, F.; Jemni, A.; Ben Nasrallah, S. Hydrogen Storage in Metal Hydride Tanks Equipped with Metal Foam Heat Exchanger. Int. J. Hydrogen Energy 2009, 34, 9393–9401. [Google Scholar] [CrossRef]
- Tsai, M.L.; Yang, T.S. On the Selection of Metal Foam Volume Fraction for Hydriding Time Minimization of Metal Hydride Reactors. Int. J. Hydrogen Energy 2010, 35, 11052–11063. [Google Scholar] [CrossRef]
- Wang, H.; Prasad, A.K.; Advani, S.G. Hydrogen Storage Systems Based on Hydride Materials with Enhanced Thermal Conductivity. Int. J. Hydrogen Energy 2012, 37, 290–298. [Google Scholar] [CrossRef]
- Ferekh, S.; Gwak, G.; Kyoung, S.; Kang, H.; Chang, M.; Yun, S.; Oh, Y.; Kim, W.; Kim, D.; Hong, T.; et al. Numerical Comparison of Heat-Fin- and Metal-Foam-Based Hydrogen Storage Beds during Hydrogen Charging Process. Int. J. Hydrogen Energy 2015, 40, 14540–14550. [Google Scholar] [CrossRef]
- Nagel, M.; Komazaki, Y.; Suda, S. Effective Thermal Conductivity of a Metal Hydride Bed Augmented with a Copper Wire Matrix. J. Less Common Met. 1986, 120, 35–43. [Google Scholar] [CrossRef]
- Ron, M.; Bershadsky, E.; Josephy, Y. Thermal Conductivity of PMH Compacts, Measurements and Evaluation. Int. J. Hydrogen Energy 1992, 17, 623–630. [Google Scholar] [CrossRef]
- Ishikawa, H.; Oguro, K.; Kato, A.; Suzuki, H.; Ishii, E. Preparation and Properties of Hydrogen Storage Alloys Microencapsulated by Copper. J. Less Common Met. 1986, 120, 123–133. [Google Scholar] [CrossRef]
- Lee, M.; Kim, K.J.; Hopkins, R.R.; Gawlik, K. Thermal Conductivity Measurements of Copper-Coated Metal Hydrides (LaNi5, Ca0.6Mm0.4Ni5, and LaNi4.75Al0.25) for Use in Metal Hydride Hydrogen Compression Systems. Int. J. Hydrogen Energy 2009, 34, 3185–3190. [Google Scholar] [CrossRef]
- Pohlmann, C.; Röntzsch, L.; Weißgärber, T.; Kieback, B. Heat and Gas Transport Properties in Pelletized Hydride–Graphite-Composites for Hydrogen Storage Applications. Int. J. Hydrogen Energy 2013, 38, 1685–1691. [Google Scholar] [CrossRef]
- Bao, Z. Performance Investigation and Optimization of Metal Hydride Reactors for High Temperature Thermochemical Heat Storage. Int. J. Hydrogen Energy 2015, 40, 5664–5676. [Google Scholar] [CrossRef]
- Visaria, M.; Mudawar, I.; Pourpoint, T. Enhanced Heat Exchanger Design for Hydrogen Storage Using High-Pressure Metal Hydride: Part 1. Design Methodology and Computational Results. Int. J. Heat Mass Transf. 2011, 54, 413–423. [Google Scholar] [CrossRef]
- Yuan, J.; Yao, M.; Zhao, B.; Lv, Y.; Huang, H.; Chen, J.; Liu, B.; Zhang, B.; Wu, Y. Influence of heat exchanger structure on hydrogen absorption-desorption performance of hydrogen storage vessel. Prog. Nat. Sci. Mater. Int. 2022, 32, 617–624. [Google Scholar] [CrossRef]
- Mohan, G.; Prakashmaiya, M.; Srinivasamurthy, S. Performance Simulation of Metal Hydride Hydrogen Storage Device with Embedded Filters and Heat Exchanger Tubes. Int. J. Hydrogen Energy 2007, 32, 4978–4987. [Google Scholar] [CrossRef]
- Hardy, B.J.; Anton, D.L. Hierarchical Methodology for Modeling Hydrogen Storage Systems. Part II: Detailed Models. Int. J. Hydrogen Energy 2009, 34, 2992–3004. [Google Scholar] [CrossRef]
- Bhouri, M.; Goyette, J.; Hardy, B.J.; Anton, D.L. Honeycomb Metallic Structure for Improving Heat Exchange in Hydrogen Storage System. Int. J. Hydrogen Energy 2011, 36, 6723–6738. [Google Scholar] [CrossRef]
- Garrison, S.L.; Hardy, B.J.; Gorbounov, M.B.; Tamburello, D.A.; Corgnale, C.; vanHassel, B.A.; Mosher, D.A.; Anton, D.L. Optimization of Internal Heat Exchangers for Hydrogen Storage Tanks Utilizing Metal Hydrides. Int. J. Hydrogen Energy 2012, 37, 2850–2861. [Google Scholar] [CrossRef]
- Bhouri, M.; Goyette, J.; Hardy, B.J.; Anton, D.L. Numerical Modeling and Performance Evaluation of Multi-Tubular Sodium Alanate Hydride Finned Reactor. Int. J. Hydrogen Energy 2012, 37, 1551–1567. [Google Scholar] [CrossRef]
- Bhouri, M.; Goyette, J.; Hardy, B.J.; Anton, D.L. Sensitivity Study of Alanate Hydride Storage System. Int. J. Hydrogen Energy 2011, 36, 621–633. [Google Scholar] [CrossRef]
- Mohan, G.; Maiya, M.P.; Srinivasa Murthy, S. A Cell Based Approach to Determine the Minimum Weight of Metal Hydride Hydrogen Storage Devices. In Proceedings of the ASME 2009 3rd International Conference on Energy Sustainability, San Francisco, CA, USA, 19–23 July 2009; Volume 1. [Google Scholar] [CrossRef]
- Menaceur, R.; Boukhari, A.; Laouini, S.E.; Attia, M.E.H. Heat and Mass Transfer Processes of Solid-State Hydrogen Discharging: A CFD Study. J. Fundam. Appl. Sci. 2020, 12, 650–666. [Google Scholar] [CrossRef]
- Krokos, C.A.; Nikolic, D.; Kikkinides, E.S.; Georgiadis, M.C.; Stubos, A.K. Modeling and Optimization of Multi-Tubular Metal Hydride Beds for Efficient Hydrogen Storage. Int. J. Hydrogen Energy 2009, 34, 9128–9140. [Google Scholar] [CrossRef]
- Georgiadis, M.C.; Kikkinides, E.S.; Makridis, S.S.; Kouramas, K.; Pistikopoulos, E.N. Design and Optimization of Advanced Materials and Processes for Efficient Hydrogen Storage. Comput. Chem. Eng. 2009, 33, 1077–1090. [Google Scholar] [CrossRef]
- Gambini, M. Metal Hydride Energy Systems Performance Evaluation. Part A: Dynamic Analysis Model of Heat and Mass Transfer. Int. J. Hydrogen Energy 1994, 19, 67–80. [Google Scholar] [CrossRef]
- Garimella, S.; Richards, D.E.; Christensen, R.N. Experimental Investigation of Heat Transfer in Coiled Annular Ducts. J. Heat Transf. 1988, 110, 329–336. [Google Scholar] [CrossRef]
- Prabhanjan, D.G.; Raghavan, G.S.V.; Rennie, T.J. Comparison of Heat Transfer Rates between a Straight Tube Heat Exchanger and a Helically Coiled Heat Exchanger. Int. Commun. Heat Mass Transf. 2002, 29, 185–191. [Google Scholar] [CrossRef]
- Fleming, W. Effective Heat Transfer in a Metal-Hydride-Based Hydrogen Separation Process. Int. J. Hydrogen Energy 2001, 26, 711–724. [Google Scholar] [CrossRef]
- Keith, M.D.; Kukkapalli, V.K.; Kim, S. Phase Change Cooling of a Metal Hydride Reactor for Rapid Hydrogen Absorption. Energies 2022, 15, 2490. [Google Scholar] [CrossRef]
- Bai, X.S.; Yang, W.W.; Zhang, W.Y.; Yang, F.S.; Tang, X.Y. Hydrogen Absorption Performance of a Novel Cylindrical MH Reactor with Combined Loop-Type Finned Tube and Cooling Jacket Heat Exchanger. Int. J. Hydrogen Energy 2020, 45, 28100–28115. [Google Scholar] [CrossRef]
- Mellouli, S.; Askri, F.; Dhaou, H.; Jemni, A.; Ben Nasrallah, S. A Novel Design of a Heat Exchanger for a Metal-Hydrogen Reactor. Int. J. Hydrogen Energy 2007, 32, 3501–3507. [Google Scholar] [CrossRef]
- Mellouli, S.; Askri, F.; Dhaou, H.; Jemni, A.; Ben Nasrallah, S. Numerical Study of Heat Exchanger Effects on Charge/Discharge Times of Metal–Hydrogen Storage Vessel. Int. J. Hydrogen Energy 2009, 34, 3005–3017. [Google Scholar] [CrossRef]
- Wang, H.; Prasad, A.K.; Advani, S.G. Hydrogen Storage System Based on Hydride Materials Incorporating a Helical-Coil Heat Exchanger. Int. J. Hydrogen Energy 2012, 37, 14292–14299. [Google Scholar] [CrossRef]
- Sekhar, B.S.; Lototskyy, M.; Kolesnikov, A.; Moropeng, M.L.; Tarasov, B.P.; Pollet, B.G. Performance Analysis of Cylindrical Metal Hydride Beds with Various Heat Exchange Options. J. Alloys Compd. 2015, 645, S89–S95. [Google Scholar] [CrossRef]
- Wu, Z.; Yang, F.; Zhang, Z.; Bao, Z. Magnesium Based Metal Hydride Reactor Incorporating Helical Coil Heat Exchanger: Simulation Study and Optimal Design. Appl. Energy 2014, 130, 712–722. [Google Scholar] [CrossRef]
- Wu, Z.; Yang, F.; Zhu, L.; Feng, P.; Zhang, Z.; Wang, Y. Improvement in Hydrogen Desorption Performances of Magnesium Based Metal Hydride Reactor by Incorporating Helical Coil Heat Exchanger. Int. J. Hydrogen Energy 2016, 41, 16108–16121. [Google Scholar] [CrossRef]
- Visaria, M.; Mudawar, I. Coiled-Tube Heat Exchanger for High-Pressure Metal Hydride Hydrogen Storage Systems—Part 1. Experimental Study. Int. J. Heat Mass Transf. 2012, 55, 1782–1795. [Google Scholar] [CrossRef]
- Visaria, M.; Mudawar, I. Coiled-Tube Heat Exchanger for High-Pressure Metal Hydride Hydrogen Storage Systems—Part 2. Computational Model. Int. J. Heat Mass Transf. 2012, 55, 1796–1806. [Google Scholar] [CrossRef]
- Eisapour, A.H.; Naghizadeh, A.; Eisapour, M.; Talebizadehsardari, P. Optimal Design of a Metal Hydride Hydrogen Storage Bed Using a Helical Coil Heat Exchanger along with a Central Return Tube during the Absorption Process. Int. J. Hydrogen Energy 2021, 46, 14478–14493. [Google Scholar] [CrossRef]
- Feng, P.; Liu, Y.; Ayub, I.; Wu, Z.; Yang, F.; Zhang, Z. Optimal Design Methodology of Metal Hydride Reactors for Thermochemical Heat Storage. Energy Convers. Manag. 2018, 174, 239–247. [Google Scholar] [CrossRef]
- Tong, L.; Xiao, J.; Yang, T.; Bénard, P.; Chahine, R. Complete and Reduced Models for Metal Hydride Reactor with Coiled-Tube Heat Exchanger. Int. J. Hydrogen Energy 2019, 44, 15907–15916. [Google Scholar] [CrossRef]
- Dhaou, H.; Souahlia, A.; Mellouli, S.; Askri, F.; Jemni, A.; Ben Nasrallah, S. Experimental Study of a Metal Hydride Vessel Based on a Finned Spiral Heat Exchanger. Int. J. Hydrogen Energy 2010, 35, 1674–1680. [Google Scholar] [CrossRef]
- Mellouli, S.; Askri, F.; Dhaou, H.; Jemni, A.; Ben Nasrallah, S. Numerical Simulation of Heat and Mass Transfer in Metal Hydride Hydrogen Storage Tanks for Fuel Cell Vehicles. Int. J. Hydrogen Energy 2010, 35, 1693–1705. [Google Scholar] [CrossRef]
- Ma, J.; Wang, Y.; Shi, S.; Yang, F.; Bao, Z.; Zhang, Z. Optimization of Heat Transfer Device and Analysis of Heat & Mass Transfer on the Finned Multi-Tubular Metal Hydride Tank. Int. J. Hydrogen Energy 2014, 39, 13583–13595. [Google Scholar] [CrossRef]
- Li, H.; Wang, Y.; He, C.; Chen, X.; Zhang, Q.; Zheng, L.; Yang, F.; Zhang, Z. Design and Performance Simulation of the Spiral Mini-Channel Reactor during H2 Absorption. Int. J. Hydrogen Energy 2015, 40, 13490–13505. [Google Scholar] [CrossRef]
- Tiwari, S.; Sharma, P. Optimization Based Methodology to Design Metal Hydride Reactor for Thermal Storage Application. J. Energy Storage 2021, 41, 102845. [Google Scholar] [CrossRef]
- Wang, D.; Li, S.; Huang, Z.; Liu, Z.; Wang, Y.; Yang, F.; Wu, Z.; Zhang, Z.; Wu, J.; Shi, L.; et al. Design Optimization, Sensitivity Analysis and Operational Comparison of a Duplex Helical Elliptical Tube Metal Hydride Reactor. Sustain. Energy Fuels 2020, 4, 5851–5868. [Google Scholar] [CrossRef]
- Larpruenrudee, P.; Bennett, N.S.; Gu, Y.; Fitch, R.; Islam, M.S. Design Optimization of a Magnesium-Based Metal Hydride Hydrogen Energy Storage System. Sci. Rep. 2022, 12, 13436. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Wang, Y.; Huang, Z.; Yang, F.; Wu, Z.; Zheng, L.; Wu, L.; Zhang, Z. Design Optimization and Sensitivity Analysis of the Radiation Mini-Channel Metal Hydride Reactor. Energy 2019, 173, 443–456. [Google Scholar] [CrossRef]
- Ramgopal, M.; Srinivasamurthy, S. Prediction of Heat and Mass Transfer in Annular Cylindrical Metal Hydride Beds. Int. J. Hydrogen Energy 1992, 17, 795–805. [Google Scholar] [CrossRef]
- Kikkinides, E.; Georgiadis, M.; Stubos, A. Dynamic Modelling and Optimization of Hydrogen Storage in Metal Hydride Beds. Energy 2006, 31, 2428–2446. [Google Scholar] [CrossRef]
- Singh, A.; Prakash Maiya, M.; Srinivasa Murthy, S. Performance of a Solid State Hydrogen Storage Device with Finned Tube Heat Exchanger. Int. J. Hydrogen Energy 2017, 42, 26855–26871. [Google Scholar] [CrossRef]
- Mahmoodi, F.; Rahimi, R. Experimental and Numerical Investigating a New Configured Thermal Coupling between Metal Hydride Tank and PEM Fuel Cell Using Heat Pipes. Appl. Therm. Eng. 2020, 178, 115490. [Google Scholar] [CrossRef]
- Raju, N.N.; Muthukumar, P.; Selvan, P.V.; Malleswararao, K. Design Methodology and Thermal Modelling of Industrial Scale Reactor for Solid State Hydrogen Storage. Int. J. Hydrogen Energy 2019, 44, 20278–20292. [Google Scholar] [CrossRef]
- Kumar, A.; Raju, N.N.; Muthukumar, P.; Selvan, P.V. Experimental Studies on Industrial Scale Metal Hydride Based Hydrogen Storage System with Embedded Cooling Tubes. Int. J. Hydrogen Energy 2019, 44, 13549–13560. [Google Scholar] [CrossRef]
- Anbarasu, S.; Muthukumar, P.; Mishra, S.C. Thermal Modeling of LmNi4.91Sn0.15 Based Solid State Hydrogen Storage Device with Embedded Cooling Tubes. Int. J. Hydrogen Energy 2014, 39, 15549–15562. [Google Scholar] [CrossRef]
- Boukhari, A.; Bessaïh, R. Numerical Heat and Mass Transfer Investigation of Hydrogen Absorption in an Annulus-Disc Reactor. Int. J. Hydrogen Energy 2015, 40, 13708–13717. [Google Scholar] [CrossRef]
- Muthukumar, P.; Singhal, A.; Bansal, G.K. Thermal Modeling and Performance Analysis of Industrial-Scale Metal Hydride Based Hydrogen Storage Container. Int. J. Hydrogen Energy 2012, 37, 14351–14364. [Google Scholar] [CrossRef]
- Gkanas, E.I.; Khzouz, M.; Panagakos, G.; Statheros, T.; Mihalakakou, G.; Siasos, G.I.; Skodras, G.; Makridis, S.S. Hydrogenation Behavior in Rectangular Metal Hydride Tanks under Effective Heat Management Processes for Green Building Applications. Energy 2018, 142, 518–530. [Google Scholar] [CrossRef]
- Bao, Z.; Wu, Z.; Nyamsi, S.N.; Yang, F.; Zhang, Z. Three-Dimensional Modeling and Sensitivity Analysis of Multi-Tubular Metal Hydride Reactors. Appl. Therm. Eng. 2013, 52, 97–108. [Google Scholar] [CrossRef]
- Dehouche, Z.; de Jong, W.; Willers, E.; Isselhorst, A.; Groll, M. Modelling and Simulation of Heating/Air-Conditioning Systems Using the Multi-Hydride-Thermal-Wave Concept. Appl. Therm. Eng. 1998, 18, 457–480. [Google Scholar] [CrossRef]
- Willers, E.; Groll, M. Evaluation of Metal Hydride Machines for Heat Pumping and Cooling Applications. Int. J. Refrig. 1999, 22, 47–58. [Google Scholar] [CrossRef]
- Linder, M.; Kulenovic, R. An Energy-Efficient Air-Conditioning System for Hydrogen Driven Cars. Int. J. Hydrogen Energy 2011, 36, 3215–3221. [Google Scholar] [CrossRef]
- Linder, M.; Mertz, R.; Laurien, E. Experimental Results of a Compact Thermally Driven Cooling System Based on Metal Hydrides. Int. J. Hydrogen Energy 2010, 35, 7623–7632. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, H.; Prasad, A.K.; Advani, S.G. Role of Heat Pipes in Improving the Hydrogen Charging Rate in a Metal Hydride Storage Tank. Int. J. Hydrogen Energy 2014, 39, 10552–10563. [Google Scholar] [CrossRef]
- Dubey, S.K.; Kumar, K.R. Numerical Investigation of Energy Desorption from Magnesium Nickel Hydride Based Thermal Energy Storage System. J. Energy Syst. 2022, 6, 165–175. [Google Scholar] [CrossRef]
- Askri, F.; Bensalah, M.; Jemni, A.; Bennasrallah, S. Optimization of Hydrogen Storage in Metal hydride Tanks. Int. J. Hydrogen Energy 2009, 34, 897–905. [Google Scholar] [CrossRef]
- Kaplan, Y. Effect of Design Parameters on Enhancement of Hydrogen Charging in Metal Hydride Reactors. Int. J. Hydrogen Energy 2009, 34, 2288–2294. [Google Scholar] [CrossRef]
- Bai, X.S.; Yang, W.W.; Tang, X.Y.; Dai, Z.Q.; Yang, F.S. Parametric Optimization of Coupled Fin-Metal Foam Metal Hydride Bed towards Enhanced Hydrogen Absorption Performance of Metal Hydride Hydrogen Storage Device. Energy 2022, 243, 123044. [Google Scholar] [CrossRef]
- Chung, C.A.; Yang, S.-W.; Yang, C.-Y.; Hsu, C.W.; Chiu, P.Y. Experimental Study on the Hydrogen Charge and Discharge Rates of Metal Hydride Tanks Using Heat Pipes to Enhance Heat Transfer. Appl. Energy 2013, 103, 581–587. [Google Scholar] [CrossRef]
- Manai, M.S.; Leturia, M.; Pohlmann, C.; Oubraham, J.; Mottelet, S.; Levy, M.; Saleh, K. Comparative Study of Different Storage Bed Designs of a Solid-State Hydrogen Tank. J. Energy Storage 2019, 26, 101024. [Google Scholar] [CrossRef]
- Nyamsi, S.N.; Wu, Z.; Zhang, Z.; Kolesnikov, A.; Lototskyy, M.V.; Pasupathi, S. Dehydrogenation Performance of Metal Hydride Container Utilising MgH2-Based Composite. Appl. Therm. Eng. 2022, 209, 118314. [Google Scholar] [CrossRef]
- Gkanas, E.I.; Khzouz, M.; Skodras, G.; Makridis, S. Parametric Study on the Heat Management of Metal Hydride Tanks for Sustainable Building Applications. Int. J. Comput. Phys. Ser. 2018, 1, 13–29. [Google Scholar] [CrossRef]
- Afzal, M.; Sharma, P. Design and Computational Analysis of a Metal Hydride Hydrogen Storage System with Hexagonal Honeycomb Based Heat Transfer Enhancements-Part A. Int. J. Hydrogen Energy 2021, 46, 13116–13130. [Google Scholar] [CrossRef]
- Afzal, M.; Gupta, N.; Mallik, A.; Vishnulal, K.S.; Sharma, P. Experimental Analysis of a Metal Hydride Hydrogen Storage System with Hexagonal Honeycomb-Based Heat Transfer Enhancements-Part B. Int. J. Hydrogen Energy 2021, 46, 13131–13141. [Google Scholar] [CrossRef]
- Singh, A.; Maiya, M.P.; Murthy, S.S. Effects of Heat Exchanger Design on the Performance of a Solid State Hydrogen Storage Device. Int. J. Hydrogen Energy 2015, 40, 9733–9746. [Google Scholar] [CrossRef]
- Nyamsi, S.N.; Yang, F.; Zhang, Z. An Optimization Study on the Finned Tube Heat Exchanger Used in Hydride Hydrogen Storage System—Analytical Method and Numerical Simulation. Int. J. Hydrogen Energy 2012, 37, 16078–16092. [Google Scholar] [CrossRef]
- Chandra, S.; Sharma, P.; Muthukumar, P.; Tatiparti, S.S.V. Modeling and Numerical Simulation of a 5 kg LaNi5-Based Hydrogen Storage Reactor with Internal Conical Fins. Int. J. Hydrogen Energy 2020, 45, 8794–8809. [Google Scholar] [CrossRef]
- Ayub, I.; Nasir, M.S.; Liu, Y.; Munir, A.; Wu, Z.; Yang, F.; Zhang, Z. Numerical Modeling and Performance Comparison of High-Temperature Metal Hydride Reactor Equipped with Bakery System for Solar Thermal Energy Storage. Int. J. Hydrogen Energy 2020, 45, 31612–31631. [Google Scholar] [CrossRef]
- Sunku Prasad, J.; Muthukumar, P. Design and Performance Analysis of an Annular Metal Hydride Reactor for Large-Scale Hydrogen Storage Applications. Renew. Energy 2022, 181, 1155–1166. [Google Scholar] [CrossRef]
- Muthukumar, P.; Prakashmaiya, M.; Murthy, S. Experiments on a Metal Hydride-Based Hydrogen Storage Device. Int. J. Hydrogen Energy 2005, 30, 1569–1581. [Google Scholar] [CrossRef]
- Gupta, S.; Sharma, V.K. Design and Analysis of Metal Hydride Reactor Embedded with Internal Copper Fins and External Water Cooling. Int. J. Energy Res. 2020, 45, 1836–1856. [Google Scholar] [CrossRef]
- Parida, A.; Muthukumar, P. Reactor Design and Numerical Study on Metal Hydride Based Finned Reactor Configurations for Hydrogen Compression Application. Int. J. Hydrogen Energy, 2023; in press. [Google Scholar] [CrossRef]
- Corgnale, C.; Hardy, B.; Chahine, R.; Cossement, D. Hydrogen Desorption Using Honeycomb Finned Heat Exchangers Integrated in Adsorbent Storage Systems. Appl. Energy 2018, 213, 426–434. [Google Scholar] [CrossRef]
- George, M.; Mohan, G. Constructal Design of Weight Optimized Metal Hydride Storage Device Embedded with Ribbed Honeycomb. Appl. Therm. Eng. 2023, 219, 119368. [Google Scholar] [CrossRef]
- Zhang, S.; Yang, F.; Zhou, L.; Zhang, Y.; Wu, Z.; Zhang, Z.; Wang, Y. A Novel Multilayer Fin Structure for Heat Transfer Enhancement in Hydride-Based Hydrogen Storage Reactor. Int. J. Energy Res. 2018, 42, 3837–3850. [Google Scholar] [CrossRef]
- Bai, X.S.; Yang, W.W.; Tang, X.Y.; Yang, F.S.; Jiao, Y.H.; Yang, Y. Optimization of Tree-Shaped Fin Structures towards Enhanced Absorption Performance of Metal Hydride Hydrogen Storage Device: A Numerical Study. Energy 2021, 220, 119738. [Google Scholar] [CrossRef]
- Krishna, K.V.; Pandey, V.; Maiya, M.P. Bio-Inspired Leaf-Vein Type Fins for Performance Enhancement of Metal Hydride Reactors. Int. J. Hydrogen Energy 2022, 47, 23694–23709. [Google Scholar] [CrossRef]
- Keshari, V.; Maiya, M.P. Design and Investigation of Hydriding Alloy Based Hydrogen Storage Reactor Integrated with a Pin Fin Tube Heat Exchanger. Int. J. Hydrogen Energy 2018, 43, 7081–7095. [Google Scholar] [CrossRef]
- Raju, M.; Kumar, S. System Simulation Modeling and Heat Transfer in Sodium Alanate Based Hydrogen Storage Systems. Int. J. Hydrogen Energy 2011, 36, 1578–1591. [Google Scholar] [CrossRef]
- Raju, M.; Kumar, S. Optimization of Heat Exchanger Designs in Metal Hydride Based Hydrogen Storage Systems. Int. J. Hydrogen Energy 2012, 37, 2767–2778. [Google Scholar] [CrossRef]
- Bai, X.S.; Yang, W.W.; Tang, X.Y.; Yang, F.S.; Jiao, Y.H.; Yang, Y. Hydrogen Absorption Performance Investigation of a Cylindrical MH Reactor with Rectangle Heat Exchange Channels. Energy 2021, 232, 121101. [Google Scholar] [CrossRef]
- Mudawar, I. Two-Phase Microchannel Heat Sinks: Theory, Applications, and Limitations. J. Electron. Packag. 2011, 133, 041002. [Google Scholar] [CrossRef]
- Lewis, S.D.; Chippar, P. Numerical Investigation of Hydrogen Absorption in a Metal Hydride Reactor with Embedded Embossed Plate Heat Exchanger. Energy 2020, 194, 116942. [Google Scholar] [CrossRef]
- Cui, Y.; Zeng, X.; Kou, H.; Ding, J.; Wang, F. Numerical Modeling of Heat Transfer during Hydrogen Absorption in Thin Double-Layered Annular ZrCo Beds. Results Phys. 2018, 9, 640–647. [Google Scholar] [CrossRef]
- Li, W.; Zeng, X.; Cui, Y.; Xiao, J.; Zhao, H.; Wang, F. Multi-Physical Thermofluidynamic Simulations of Hydrogen Absorption Performance in Double-Layered Cylindrical ZrCo-Based Hydride Beds. J. Appl. Phys. 2021, 130, 155101. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, F.; Zeng, X.; Zhang, K.; Kou, H. A Three-dimensional Heat Transfer Model for Thermal Performance Evaluation of ZrCo-based Hydride Bed with Embedded Circular-shaped Cooling Tubes. Int. J. Energy Res. 2019, 43, 6994–7009. [Google Scholar] [CrossRef]
- Zhang, B.; Wang, F.; Cheng, J.; Zeng, X.; Kou, H.; Sang, G. Multiscale Insights into Hydrogen Charging Behavior in Metal Hydride Reactor Packed with Porous ZrCo Particles. Int. J. Hydrogen Energy 2021, 46, 40217–40238. [Google Scholar] [CrossRef]
- Lin, X.; Yang, H.; Zhu, Q.; Li, Q. Numerical Simulation of a Metal Hydride Tank with LaNi4.25Al0.75 Using a Novel Kinetic Model at Constant Flows. Chem. Eng. J. 2020, 401, 126115. [Google Scholar] [CrossRef]
- Pan, C.; Ziviani, D.; Braun, J.E. A 2-D Reduced Dynamic Model for a Shell-and-Tube Based Metal Hydride Reactor for Geometry and Operation Condition Optimal Design. Appl. Therm. Eng. 2022, 206, 118125. [Google Scholar] [CrossRef]
- Yang, F.S.; Wang, G.X.; Zhang, Z.X.; Meng, X.Y.; Rudolph, V. Design of the Metal Hydride Reactors—A Review on the Key Technical Issues. Int. J. Hydrogen Energy 2010, 35, 3832–3840. [Google Scholar] [CrossRef]
- Pandey, V.; Krishna, K.V.; Maiya, M.P. Numerical Modelling and Heat Transfer Optimization of Large-Scale Multi-Tubular Metal Hydride Reactors. Int. J. Hydrogen Energy 2023, 48, 16020–16036. [Google Scholar] [CrossRef]
- Urbanczyk, R.; Peinecke, K.; Peil, S.; Felderhoff, M. Development of a Heat Storage Demonstration Unit on the Basis of Mg2FeH6 as Heat Storage Material and Molten Salt as Heat Transfer Media. Int. J. Hydrogen Energy 2017, 42, 13818–13826. [Google Scholar] [CrossRef]
- Afzal, M.; Sharma, P. Design of a Large-Scale Metal Hydride Based Hydrogen Storage Reactor: Simulation and Heat Transfer Optimization. Int. J. Hydrogen Energy 2018, 43, 13356–13372. [Google Scholar] [CrossRef]
- Sreeraj, R.; Aadhithiyan, A.K.; Anbarasu, S. Comparison, Advancement, and Performance Evaluation of Heat Exchanger Assembly in Solid-State Hydrogen Storage Device. Renew. Energy 2022, 198, 667–678. [Google Scholar] [CrossRef]
- Tran, T.Q.; Chinnappan, A.; Lee, J.K.Y.; Loc, N.H.; Tran, L.T.; Wang, G.; Kumar, V.V.; Jayathilaka, W.A.D.M.; Ji, D.; Doddamani, M.; et al. 3D Printing of Highly Pure Copper. Metals 2019, 9, 756. [Google Scholar] [CrossRef]
- Zitelli, C.; Folgarait, P.; Di Schino, A. Laser Powder Bed Fusion of Stainless Steel Grades: A Review. Metals 2019, 9, 731. [Google Scholar] [CrossRef]
- Kraiem, N.; Constantin, L.; Mao, A.; Wang, F.; Cui, B.; Silvain, J.-F.; Lu, Y.F. Influence of Aluminum Addition on the Laser Powder Bed Fusion of Copper-Aluminum Mixtures. Addit. Manuf. Lett. 2022, 3, 100080. [Google Scholar] [CrossRef]
- Lindström, V.; Liashenko, O.; Zweiacker, K.; Derevianko, S.; Morozovych, V.; Lyashenko, Y.; Leinenbach, C. Laser Powder Bed Fusion of Metal Coated Copper Powders. Materials 2020, 13, 3493. [Google Scholar] [CrossRef] [PubMed]
- Sharabian, E.; Leary, M.; Fraser, D.; Gulizia, S. Electron Beam Powder Bed Fusion of Copper Components: A Review of Mechanical Properties and Research Opportunities. Int. J. Adv. Manuf. Technol. 2022, 122, 513–532. [Google Scholar] [CrossRef]
- Aversa, A.; Marchese, G.; Saboori, A.; Bassini, E.; Manfredi, D.; Biamino, S.; Ugues, D.; Fino, P.; Lombardi, M. New Aluminum Alloys Specifically Designed for Laser Powder Bed Fusion: A Review. Materials 2019, 12, 1007. [Google Scholar] [CrossRef]
- Kusoglu, I.M.; Gökce, B.; Barcikowski, S. Research Trends in Laser Powder Bed Fusion of Al Alloys within the Last Decade. Addit. Manuf. 2020, 36, 101489. [Google Scholar] [CrossRef]
- Manfredi, D.; Bidulský, R. Laser Powder Bed Fusion of Aluminum Alloys. Acta Metall. Slovaca 2017, 23, 276–282. [Google Scholar] [CrossRef]
- Filippov, A.; Shamarin, N.; Moskvichev, E.; Savchenko, N.; Kolubaev, E.; Khoroshko, E.; Tarasov, S. Heat Input Effect on Microstructure and Mechanical Properties of Electron Beam Additive Manufactured (EBAM) Cu-7.5wt.%Al Bronze. Materials 2021, 14, 6948. [Google Scholar] [CrossRef]
- Aboulkhair, N.T.; Simonelli, M.; Parry, L.; Ashcroft, I.; Tuck, C.; Hague, R. 3D Printing of Aluminium Alloys: Additive Manufacturing of Aluminium Alloys Using Selective Laser Melting. Prog. Mater. Sci. 2019, 106, 100578. [Google Scholar] [CrossRef]
- Moon, H.; Miljkovic, N.; King, W.P. High Power Density Thermal Energy Storage Using Additively Manufactured Heat Exchangers and Phase Change Material. Int. J. Heat Mass Transf. 2020, 153, 119591. [Google Scholar] [CrossRef]
- Yadav, S.; Paul, C.P.; Jinoop, A.N.; Rai, A.K.; Bindra, K.S. Laser Directed Energy Deposition Based Additive Manufacturing of Copper: Process Development and Material Characterizations. J. Manuf. Process. 2020, 58, 984–997. [Google Scholar] [CrossRef]
- Jinoop, A.N.; Paul, C.P.; Mishra, S.K.; Bindra, K.S. Laser Additive Manufacturing Using Directed Energy Deposition of Inconel-718 Wall Structures with Tailored Characteristics. Vacuum 2019, 166, 270–278. [Google Scholar] [CrossRef]
- Liu, X.; Wang, H.; Kaufmann, K.; Vecchio, K. Directed Energy Deposition of Pure Copper Using Blue Laser. J. Manuf. Process. 2023, 85, 314–322. [Google Scholar] [CrossRef]
- Caiazzo, F.; Alfieri, V. Simulation of Laser-Assisted Directed Energy Deposition of Aluminum Powder: Prediction of Geometry and Temperature Evolution. Materials 2019, 12, 2100. [Google Scholar] [CrossRef]
- Ahn, D.-G. Directed Energy Deposition (DED) Process: State of the Art. Int. J. Precis. Eng. Manuf.-Green Technol. 2021, 8, 703–742. [Google Scholar] [CrossRef]
- Gibson, I.; Rosen, D.; Stucker, B.; Khorasani, M. Material Jetting. In Additive Manufacturing Technologies; Springer: Berlin/Heidelberg, Germany, 2020; pp. 203–235. [Google Scholar] [CrossRef]
- Lecis, N.; Mariani, M.; Beltrami, R.; Emanuelli, L.; Casati, R.; Vedani, M.; Molinari, A. Effects of Process Parameters, Debinding and Sintering on the Microstructure of 316L Stainless Steel Produced by Binder Jetting. Mater. Sci. Eng. A 2021, 828, 142108. [Google Scholar] [CrossRef]
- Bai, Y.; Williams, C.B. An Exploration of Binder Jetting of Copper. Rapid Prototyp. J. 2015, 21, 177–185. [Google Scholar] [CrossRef]
- Kumar, A.Y.; Wang, J.; Bai, Y.; Huxtable, S.T.; Williams, C.B. Impacts of Process-Induced Porosity on Material Properties of Copper Made by Binder Jetting Additive Manufacturing. Mater. Des. 2019, 182, 108001. [Google Scholar] [CrossRef]
- Romano, T.; Migliori, E.; Mariani, M.; Lecis, N.; Vedani, M. Densification Behaviour of Pure Copper Processed through Cold Pressing and Binder Jetting under Different Atmospheres. Rapid Prototyp. J. 2022, 28, 1023–1039. [Google Scholar] [CrossRef]
- Im, S.; Ghasri-Khouzani, M.; Muhammad, W.; Batmaz, R.; Esmati, K.; Chakraborty, A.; Natarajan, A.; Martin, É. Evaluation of Different Sintering Agents for Binder Jetting of Aluminum Alloy. J. Mater. Eng. Perform. 2023. [Google Scholar] [CrossRef]
- Gibson, I.; Rosen, D.W.; Stucker, B. Sheet Lamination Processes. In Additive Manufacturing Technologies; Springer: Berlin/Heidelberg, Germany, 2010; pp. 223–252. [Google Scholar] [CrossRef]
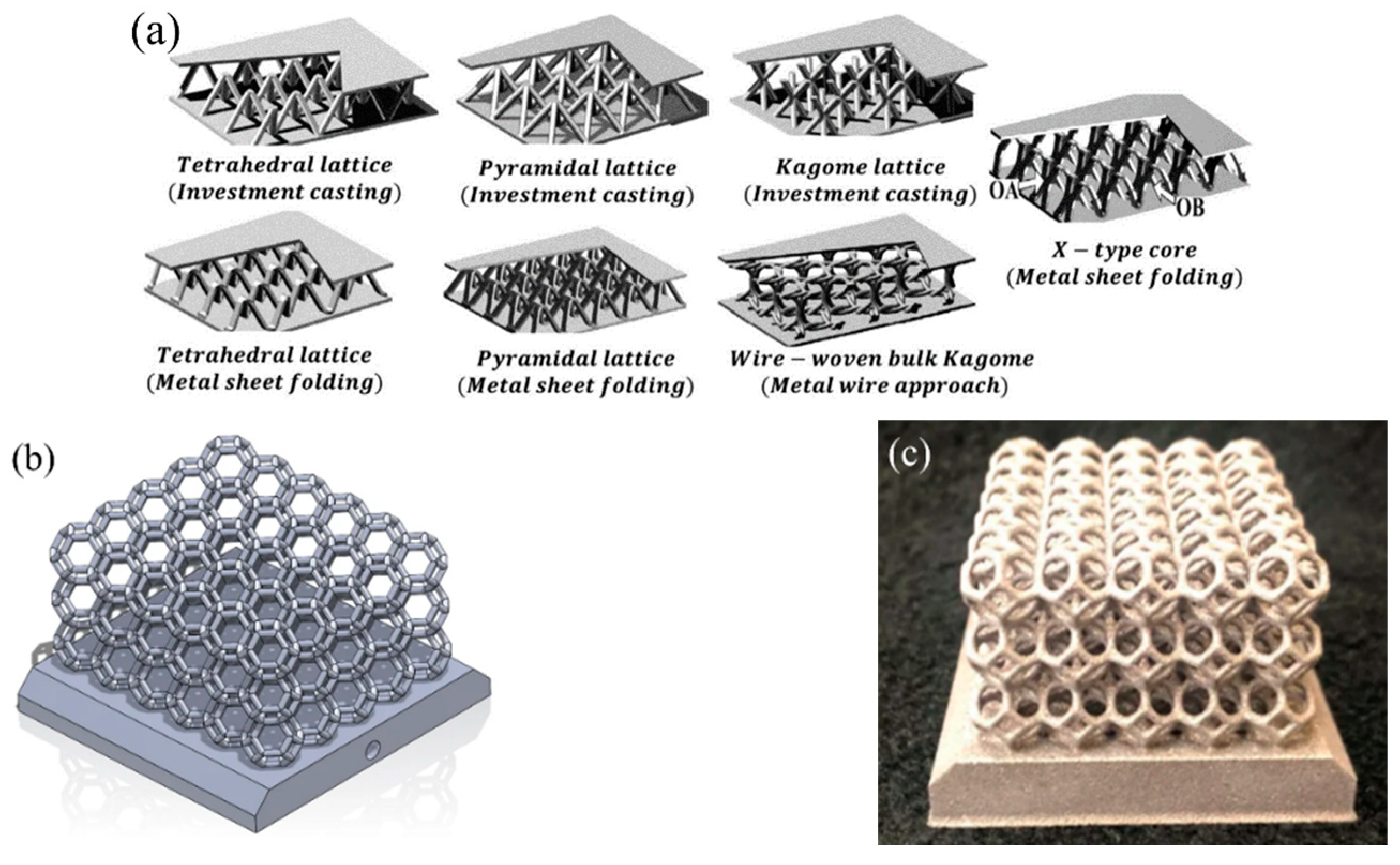
- Yan, H.; Yang, X.; Lu, T.; Xie, G. Convective Heat Transfer in a Lightweight Multifunctional Sandwich Panel with X-Type Metallic Lattice Core. Appl. Therm. Eng. 2017, 127, 1293–1304. [Google Scholar] [CrossRef]
- How To Design a Heat Sink for Additive Manufacturing. Simcenter. PLM Ecosystem. Available online: https://plmes.io/how-to-design-a-heat-sink-for-additive-manufacturing-simcenter/ (accessed on 25 April 2023).
- Kaur, I.; Singh, P. Critical Evaluation of Additively Manufactured Metal Lattices for Viability in Advanced Heat Exchangers. Int. J. Heat Mass Transf. 2021, 168, 120858. [Google Scholar] [CrossRef]
- Keshari, V.; Maiya, M.P. Numerical Simulation of Metal Hydride Hydrogen Storage Device withPin Fin Tube Heat Exchanger. In Proceedings of the World Congress on Mechanical, Chemical, and Material Engineering, Rome, Italy, 8–10 June 2017. [Google Scholar] [CrossRef]
- Martin, J.D. Exploring Additive Manufacturing Processes for Direct 3D Printing of Copper Induction Coils. In Volume 2: Advanced Manufacturing; ASME: New York, NY, USA, 2017. [Google Scholar] [CrossRef]
- Fratalocchi, L.; Groppi, G.; Visconti, C.G.; Lietti, L.; Tronconi, E. Adoption of 3D Printed Highly Conductive Periodic Open Cellular Structures as an Effective Solution to Enhance the Heat Transfer Performances of Compact Fischer-Tropsch Fixed-Bed Reactors. Chem. Eng. J. 2020, 386, 123988. [Google Scholar] [CrossRef]
- Wang, G.; Gu, Y.; Zhao, L.; Xuan, J.; Zeng, G.; Tang, Z.; Sun, Y. Experimental and Numerical Investigation of Fractal-Tree-like Heat Exchanger Manufactured by 3D Printing. Chem. Eng. Sci. 2019, 195, 250–261. [Google Scholar] [CrossRef]
- Comparing Optimization Methods for a Heat Sink Design for 3D Printing. Available online: https://www.comsol.de/blogs/comparing-optimization-methods-for-a-heat-sink-design-for-3d-printing/ (accessed on 26 April 2023).



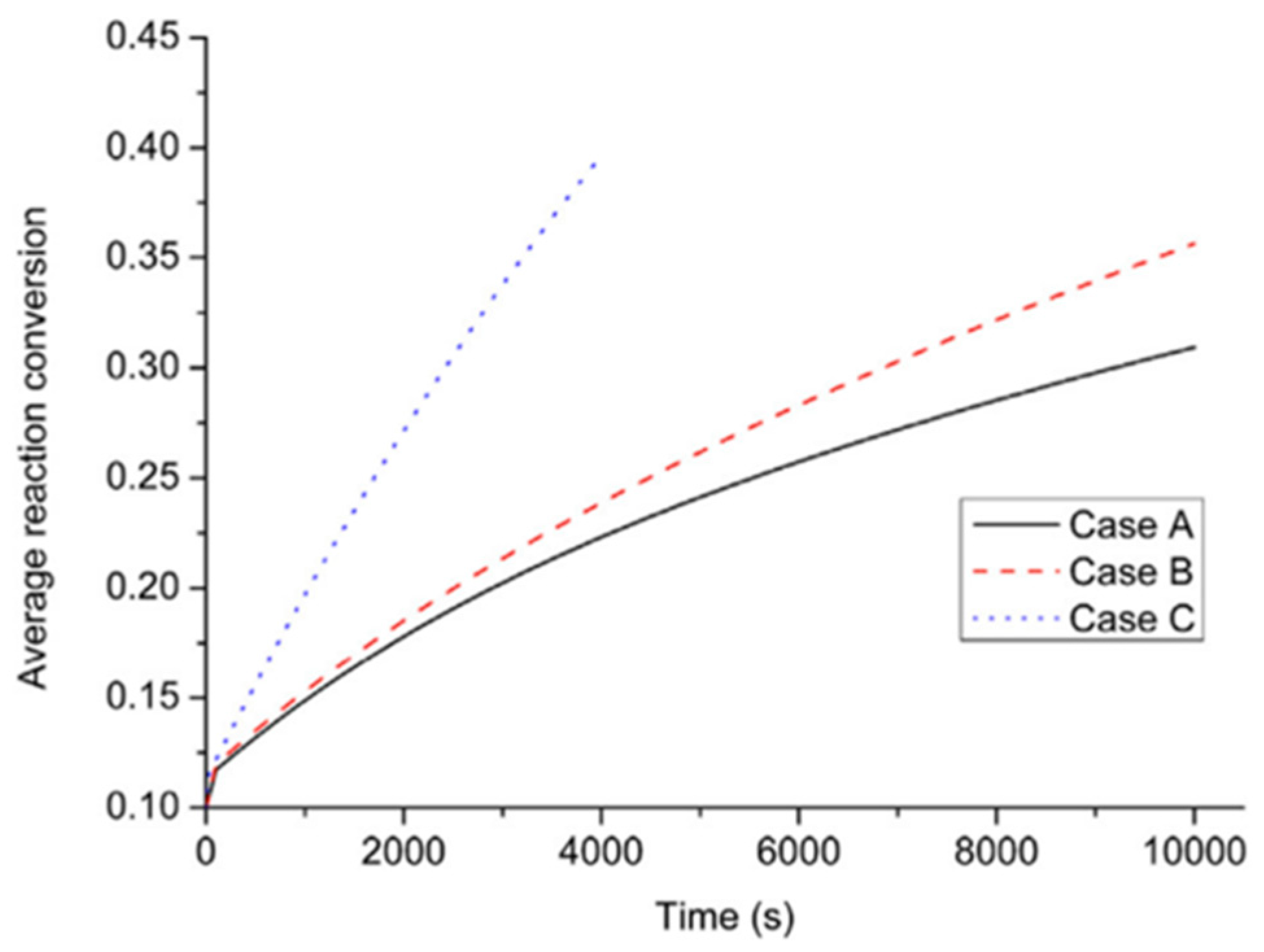
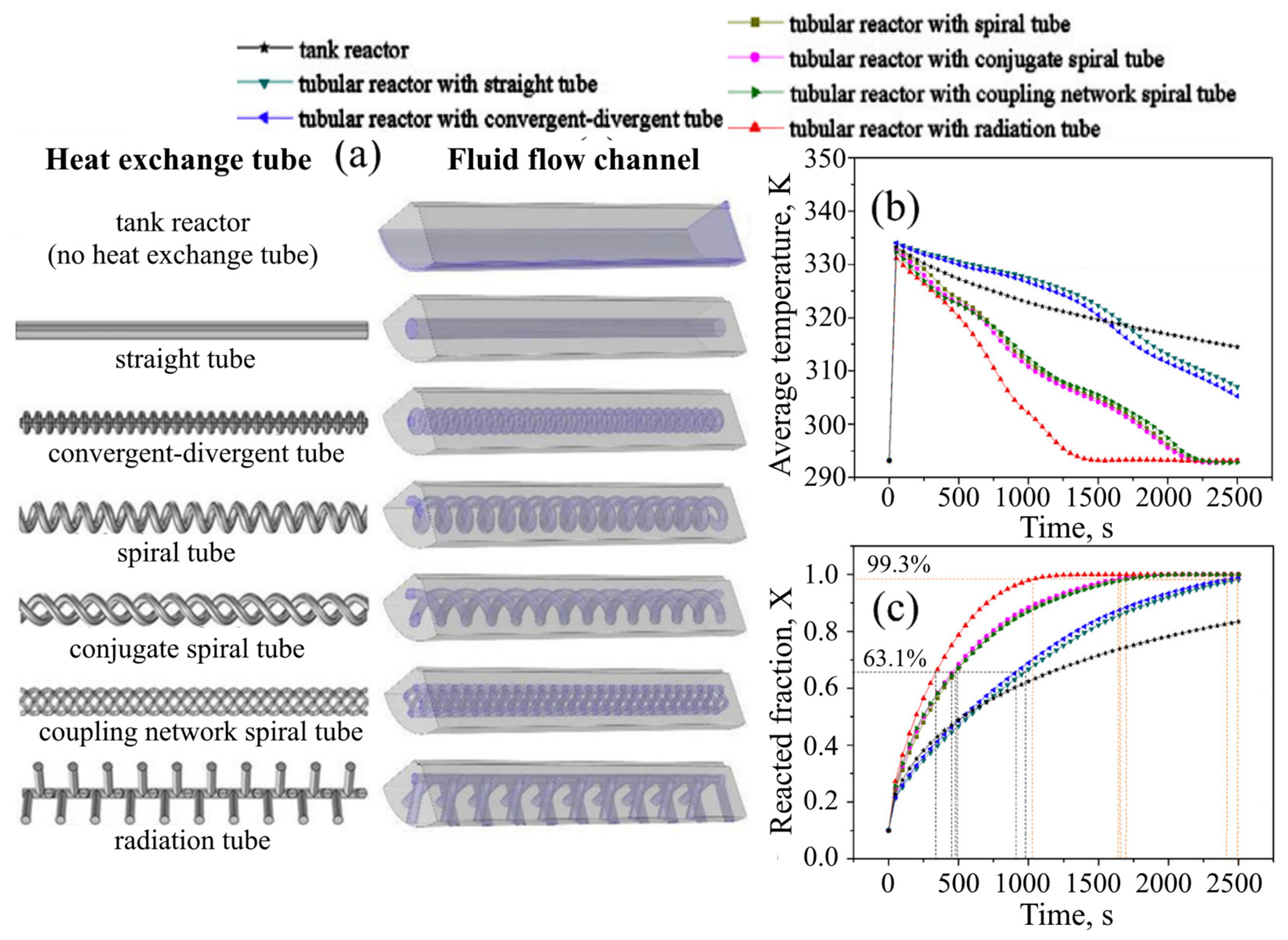

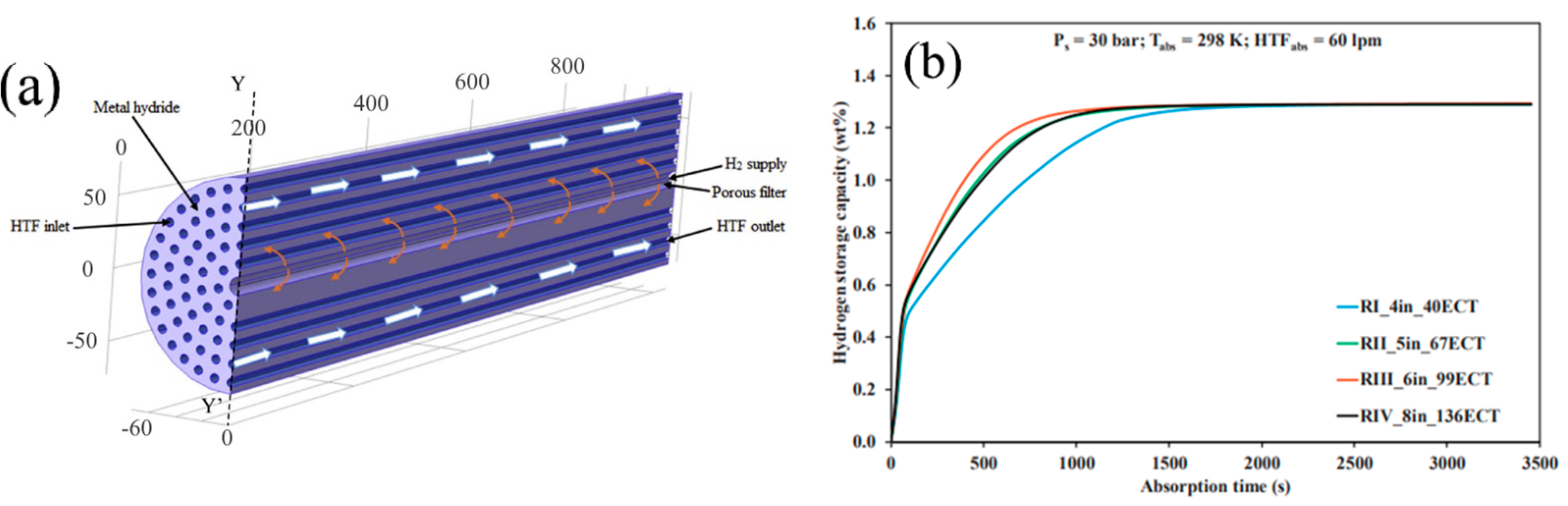

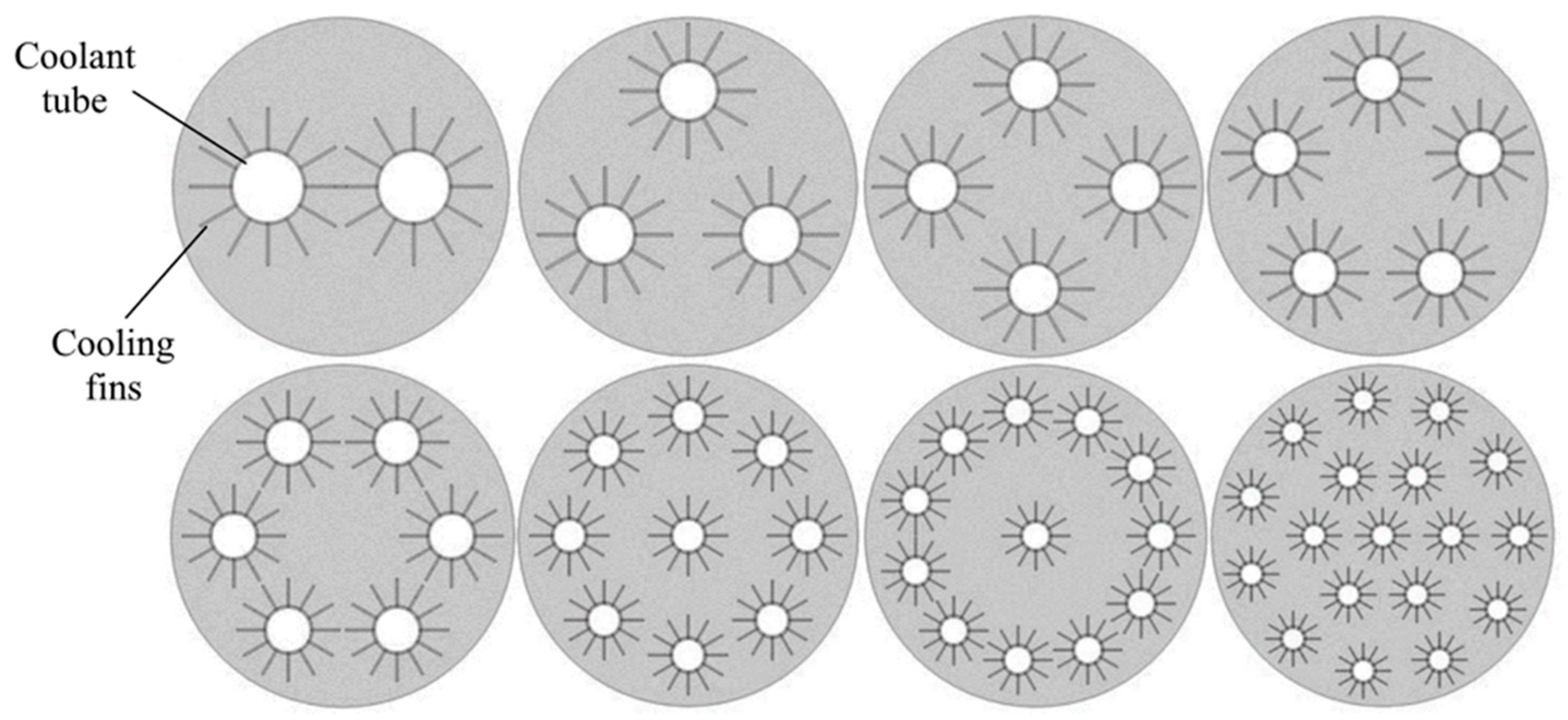

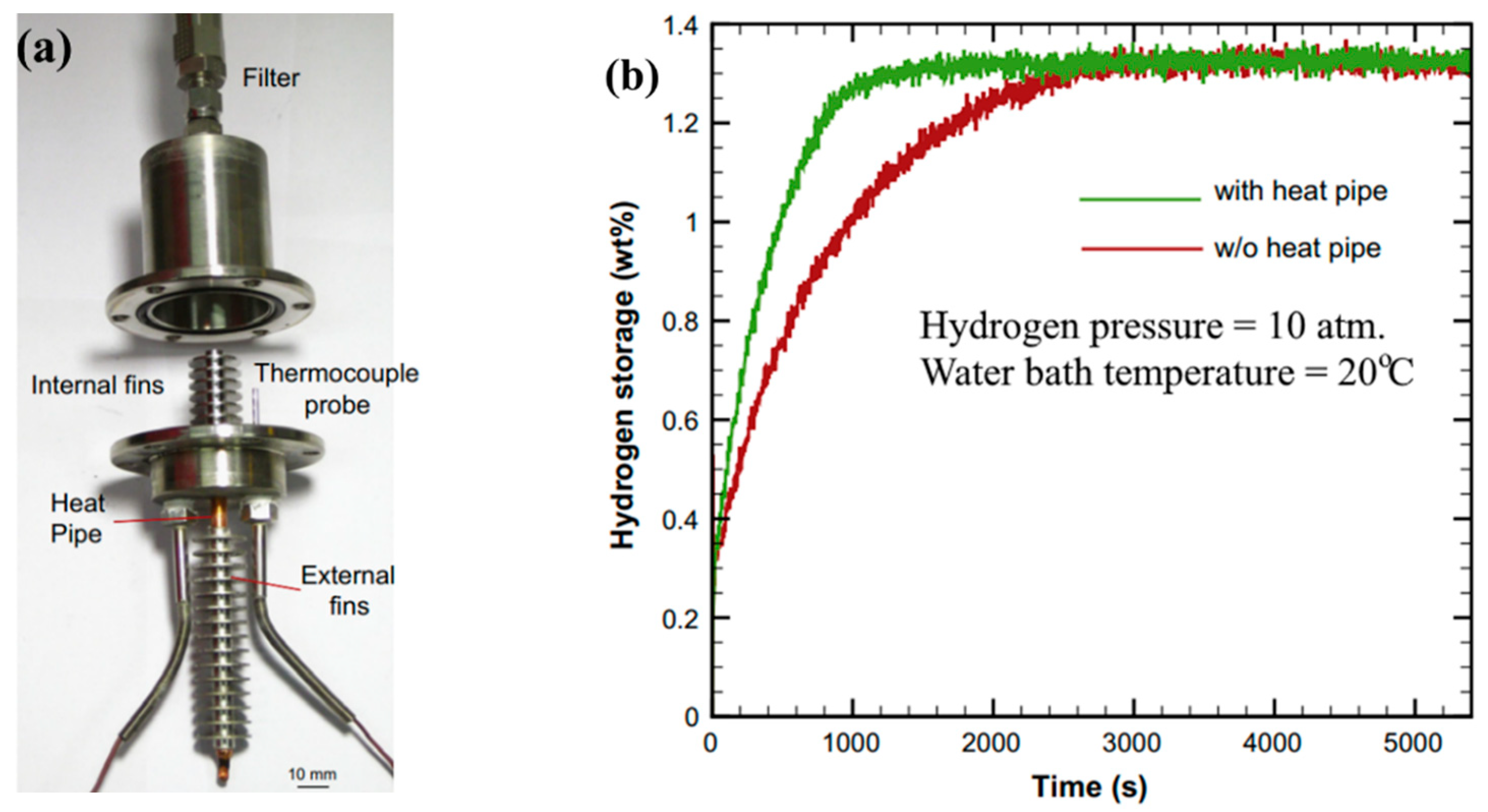
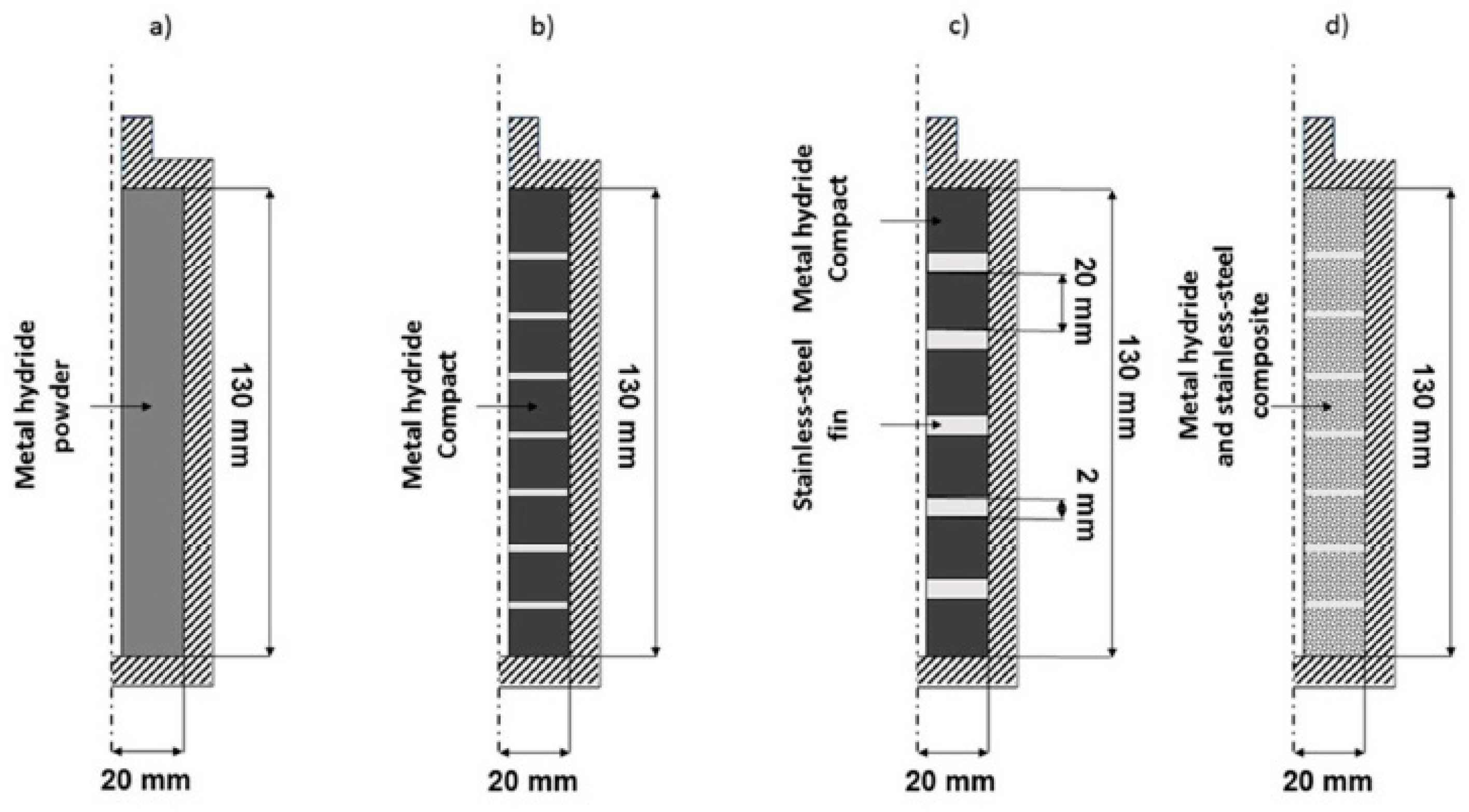
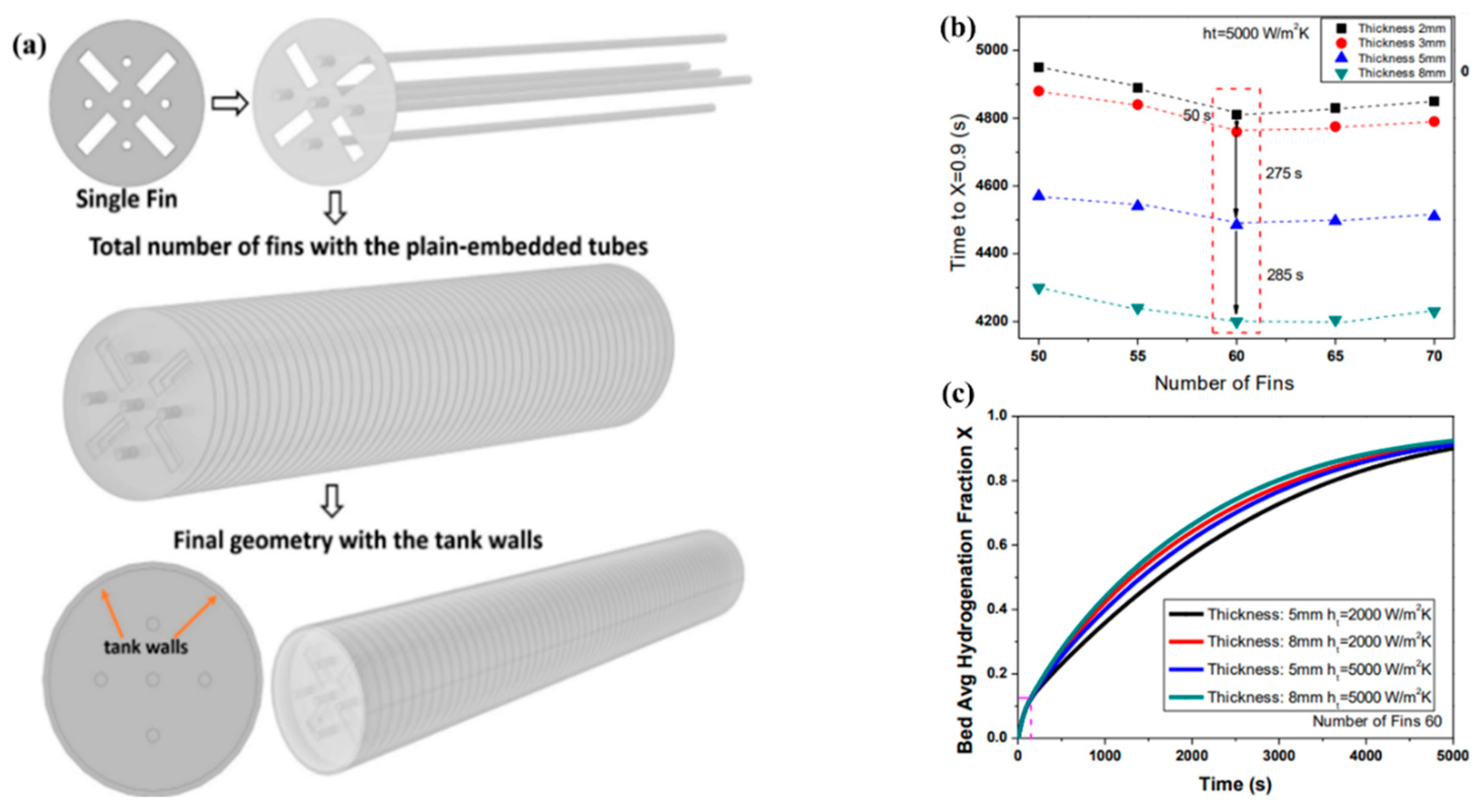

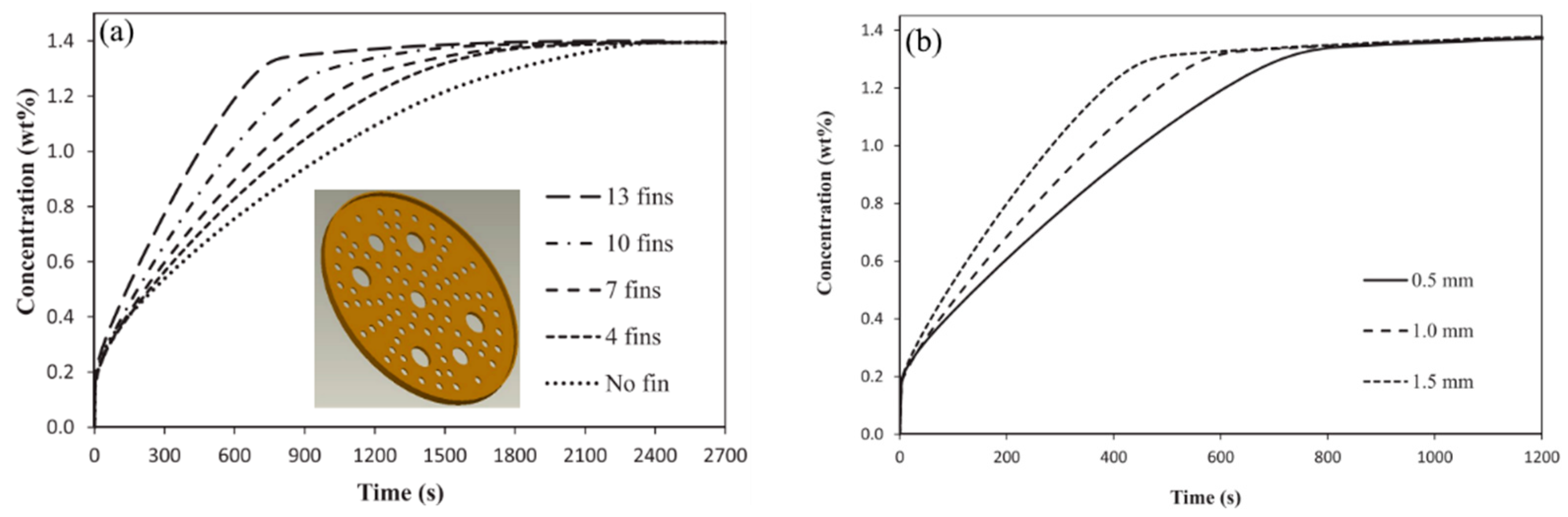
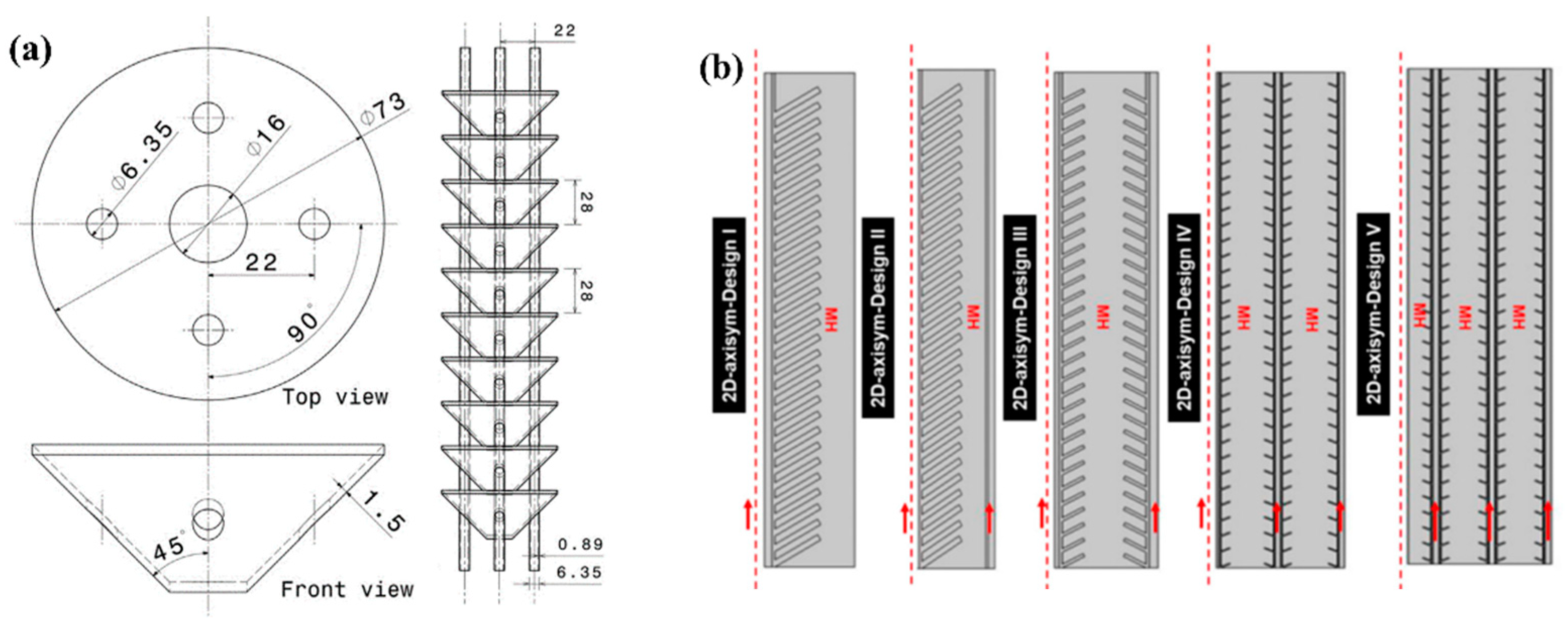
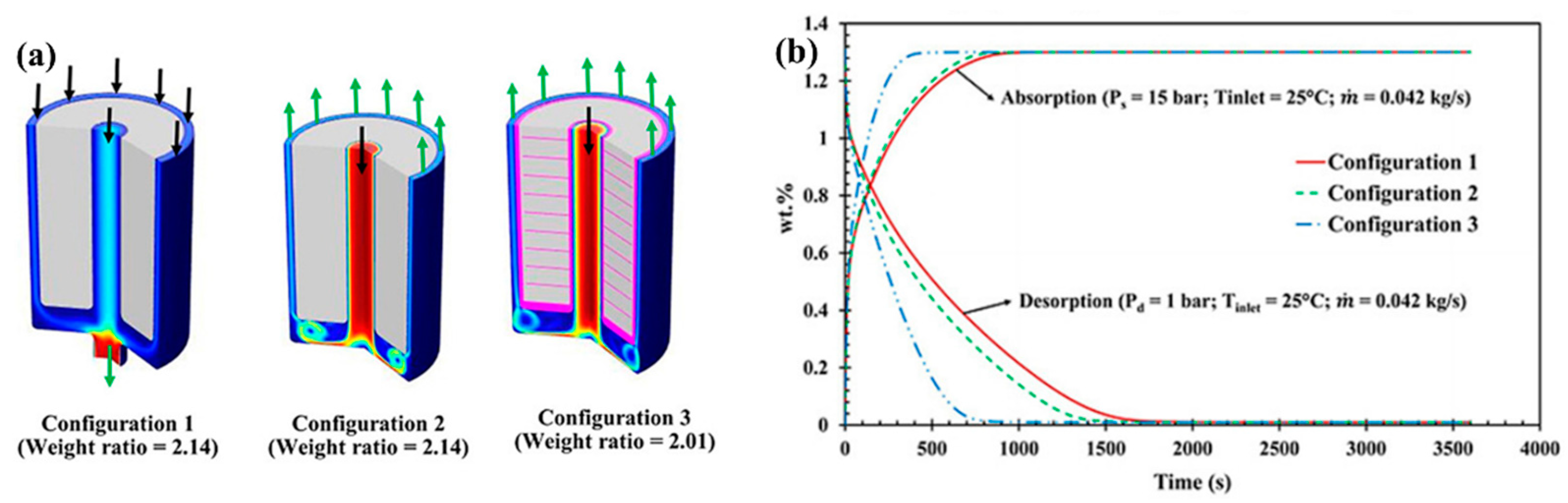

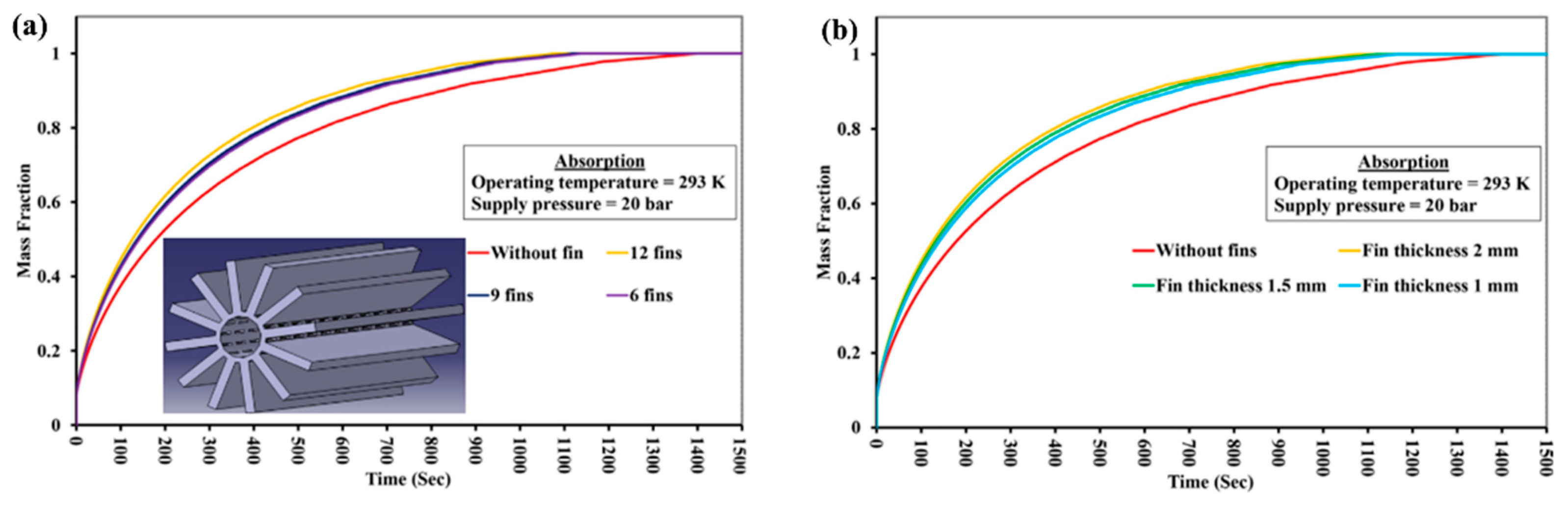

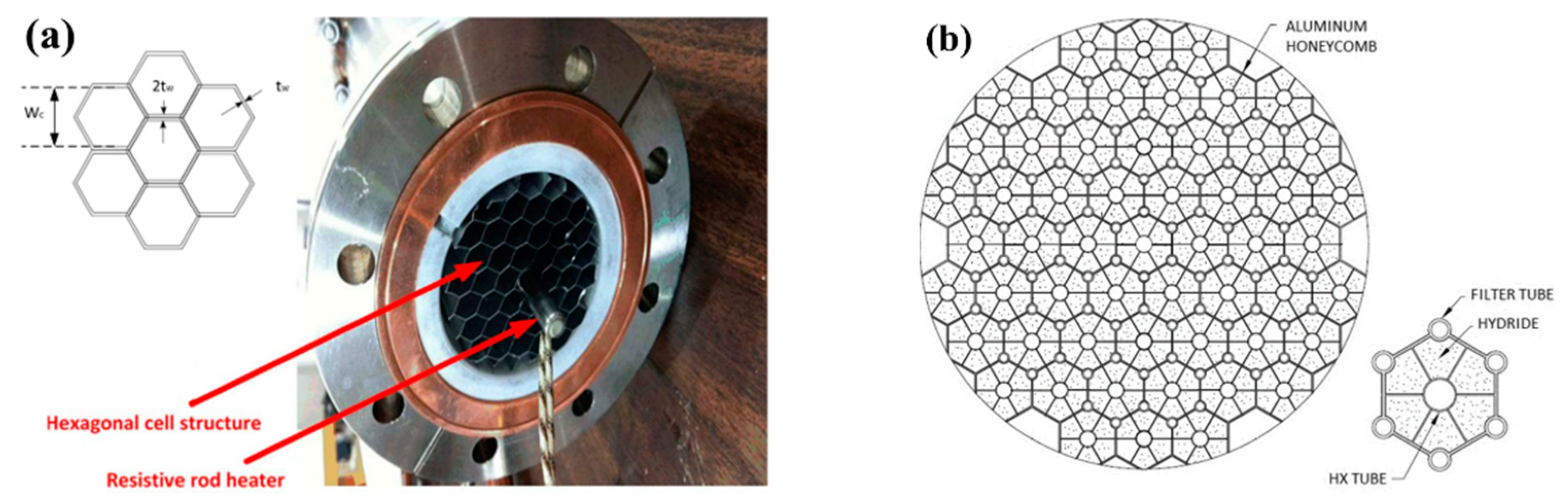
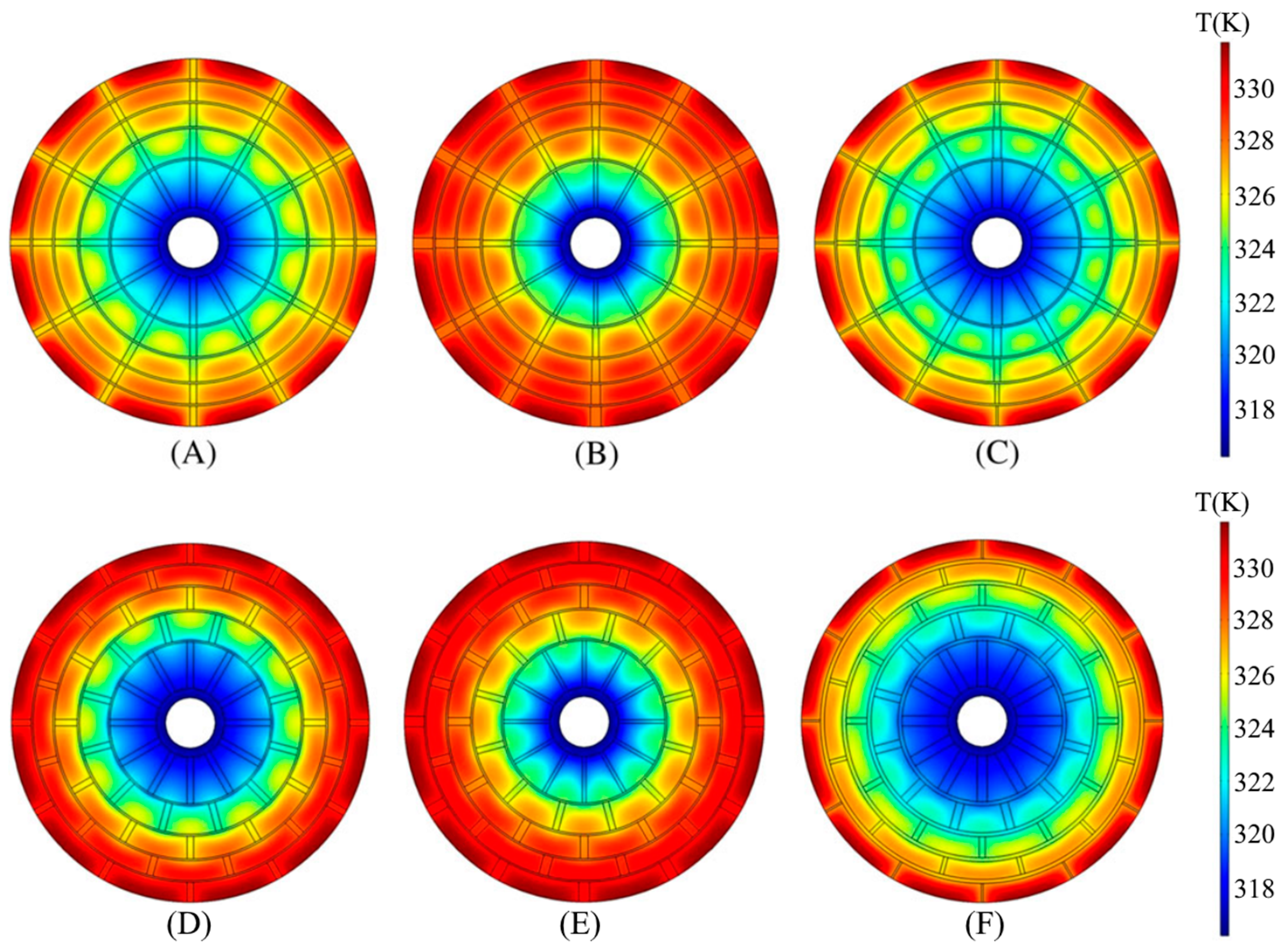





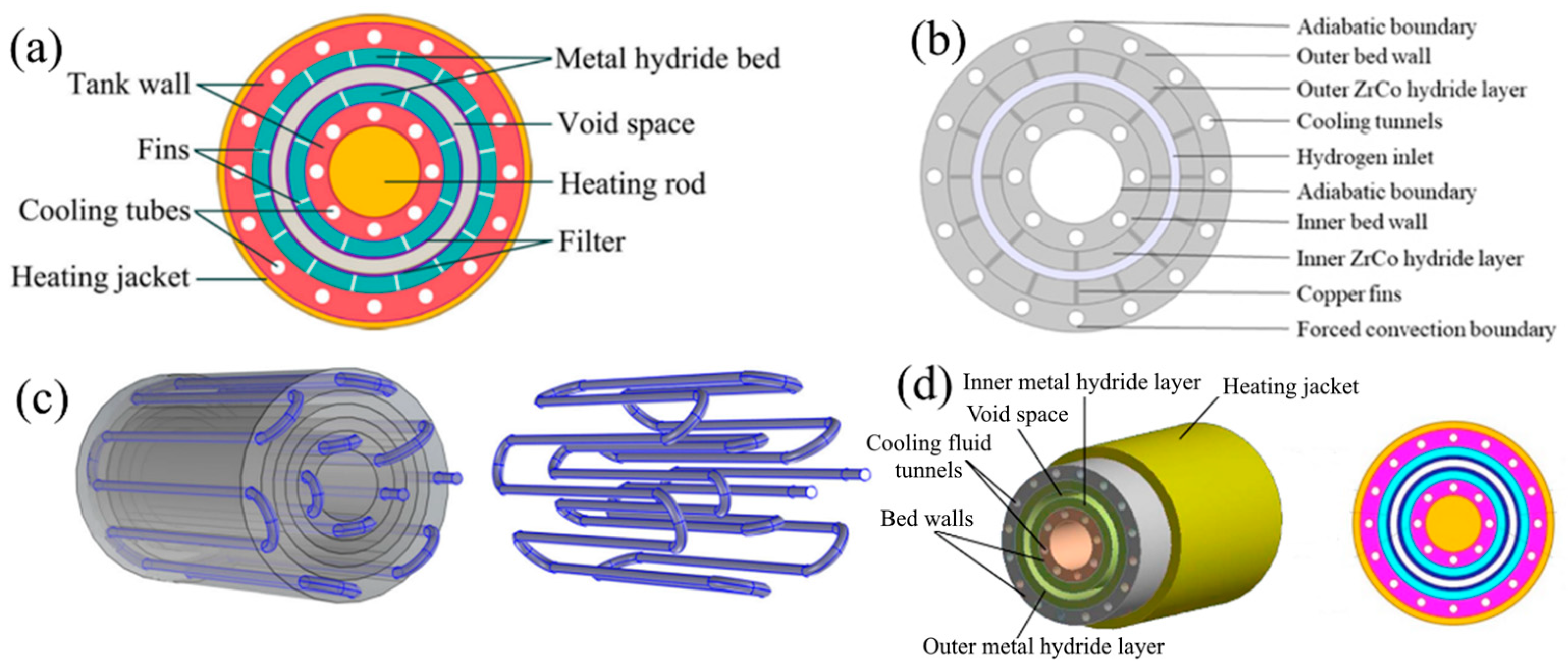


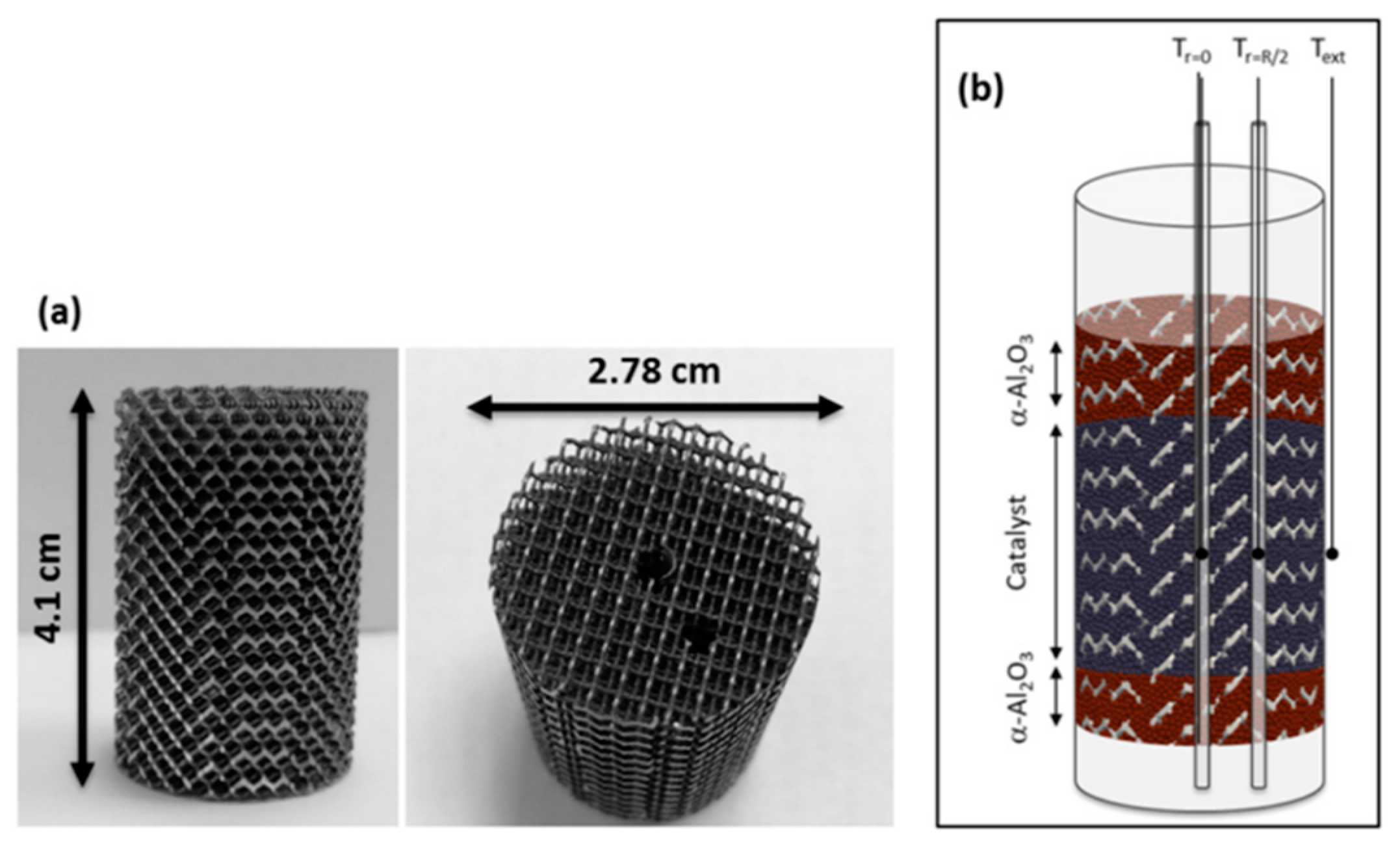

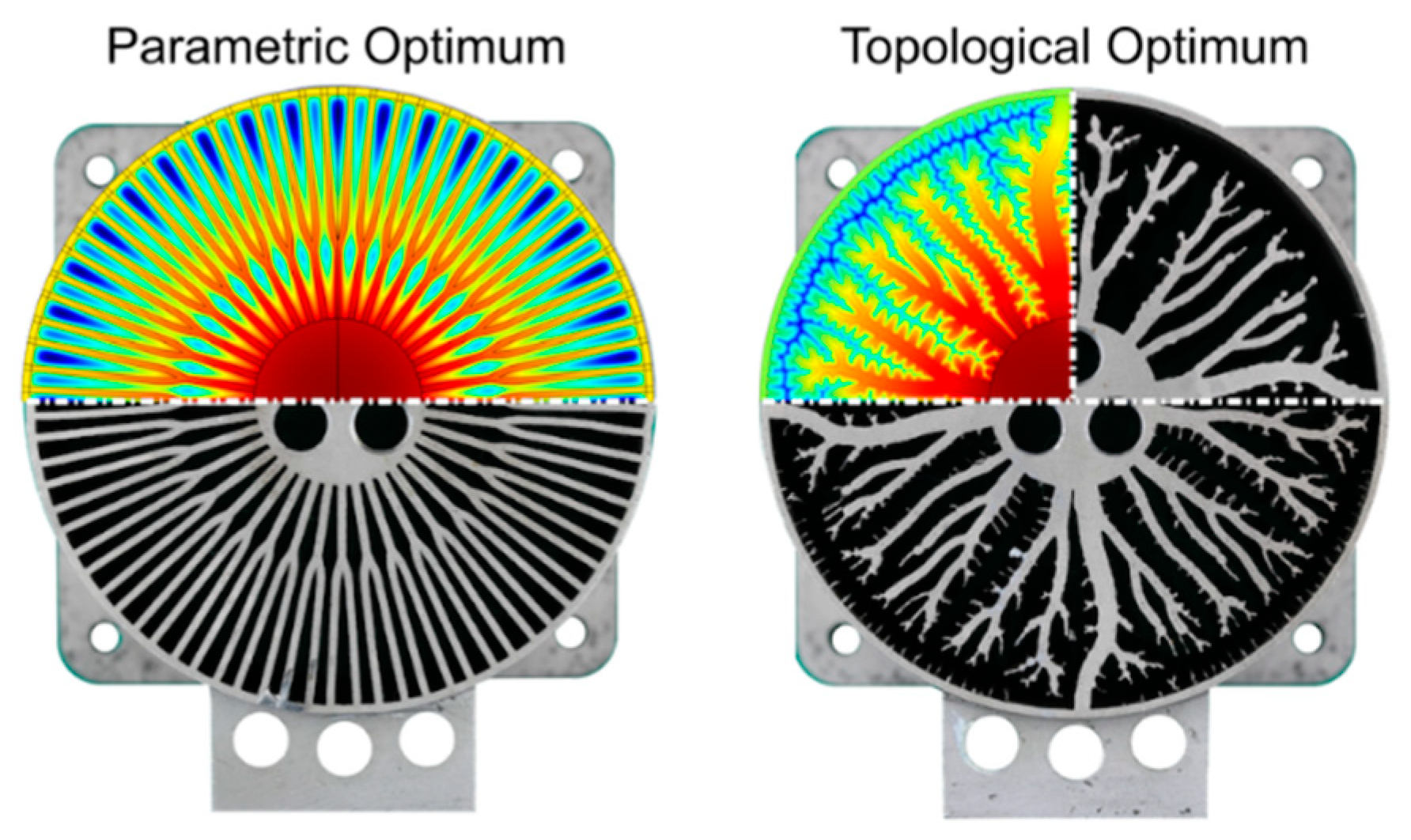
| Metal Hydride Material | Type of Modification | Heat Transfer Coefficient, W/(mK) | References |
|---|---|---|---|
| MmNi4.15Fe0.85 | Aluminum foam | 6.9 | [81] |
| MmNi4.46Al0.54 | Copper wire matrix | 0.5–2.5 | [88] |
| LaNi4.85Sn0.15 | Expanded natural graphite flakes | 19.5 | [59] |
| LaNi5 | Porous metallic-matrix hydride compacts with aluminum | 12.3 | [89] |
| LaNi5 | Copper encapsulation | 3.5 | [90] |
| LaNi4.75Al0.25 | Copper coating | 1.78–4.3 | [91] |
| LaNi5 | Aluminum foam | ≤10.0 | [81] |
| LaNi5 | Copper coating | 6.0–9.0 | [63] |
| LaNi5 | Copper encapsulation | 5.0 | [58] |
| LaNi5 | Copper coating | 2.17–6.6 | [91] |
| Ca0.6Mm0.4Ni5 | Copper coating | 0.8–2.8 | [91] |
| Mg90Ni10 | Pelletized hydride-graphite composites | >10.0 | [92] |
| Hydralloy® C5-based MHC | Metal hydride/ENG compacts | ~10–15 | [71] |
| La0.8Ce0.2Ni5 | Graphite flakes | 4.7 | [68] |
| La0.8Ce0.2Ni5 | Graphite flakes with copper wires | 6.8 | [68] |
| La0.9Ce0.1Ni5 | Metal hydride/ENG compacts | 8.1 | [72] |
| MgH2 | Metal hydride/ENG compacts | 1.0–4.2 | [93] |
| References | Hydrogen Storage Material | Heat Exchanger Design | System Characterization |
|---|---|---|---|
| Gkanas E. I. et al. [154] | MmNi4.6Al0.4 | Combination of cooling tubes with fins; fins with five holes. | The optimum fin number is 60 and fin thickness 5–8 mm. The value of the heat transfer coefficient is about 2000–5000 W/(m2K). |
| Afzal M. and Sharma P. [155,156] | La0.9Ce0.1Ni5 | Hexagonal heat transfer enhancement. | Improves the desorption rates by 20%, from 313 K to 323 K. Improves the absorption performance of the hydride bed by over 30%. |
| Singh A. et al. [157] | LaNi5 | Copper fins with perforation; fin diameter—52 mm; fin thickness—0.5 mm. | The charging time for 10 g of hydrogen: 614 s (Design 1), 560 s (Design 2), 582 s (Design 3) and 604 s (Design 4). |
| Singh A. et al. [133] | LaNi5 | Two “U”-shaped tubes and copper fins with perforation; fin diameter—61 mm. | The required charging time is around 610 s for a storage capacity of 12 g (1.2 wt%). |
| Garrison S. L. et al. [98] | A sodium alanate complex metal hydride | A transverse fin and a longitudinal fin. | Parameters for the optimal design (transverse fin): internal diameter of the cooling tube, 0.085 in.; thickness of the cooling tube, 0.020 in.; length of cooling fin, 0.290 in.; thickness of the cooling fin, 0.004 in. To store 1 kg of hydrogen would require ~97,800 unit cells with 14.9 kg of aluminum cooling tubes and cooling fins and 126 kg of hydride precursor. Parameters for the optimal design (longitudinal fin): internal diameter of the cooling tube, 0.100 in.; thickness of the cooling tube, 0.020 in.; length of cooling fin, 0.340 in.; thickness of the cooling fin, 0.004 in. To store 1 kg of hydrogen would require 537 independent cooling tubes in a 1 m-long tank vessel, with 14.7 kg of aluminum cooling tubes and cooling fins and 126 kg of hydride precursor. |
| Nyamsi S. N. et al. [158] | LaNi5 | Two designs: the baseline design with a fin length of 7.8 mm and the optimized design with a fin length of 15 mm. | The hydrogen charging time is ~600 s when the optimized design is used; Increasing the cooling tube diameter can bring about 25% of hydrogen charging time. |
| Chandra S. et al. [159] | LaNi5-based system | Cylindrical reactor (OD 88.9 mm) with internal conical copper fins and cooling tubes (1/4”, SS 316); 10, 13 and 19 copper fins with 2, 4 and 6 copper cooling tubes. | Conical fins offer enhanced heat transfer. A design with 19 fins with 6 tubes requires 290 and 375 s for 80% and 90% hydrogen saturation level, respectively. |
| Ayub I. et al. [160] | Nano-engineered composite (MgH2 + V2O5) | Annular hollow truncated conical fins (steel 316L). The total length of a metal hydride reactor is 0.64 m, and the radius for all the designs is 0.12 m. | Optimal design: central heat transfer pipe + multiple jackets for MH bed and heat transfer fluid. The reaction time was 15,000 s, value of gravimetric exergy output rate was 1.23 W/kg, and value of exergy output was 0.028 kW. |
| Prasad J. S. and Muthukumar P. [161] | LaNi5 | Cross fins. | Fins occupy about 4.6% of the reactor volume, improving the hydrogen sorption and desorption rate by a factor of 2.07 and 1.92. |
| Visaria M. et al. [94] | High-pressure metal hydride Ti1.1CrMn | A 260.3 mm-long prototype with aluminum plates. The heat exchanger could store 2.65 kg of metal hydride powder. | The design occupies 29% of the pressure vessel volume. The metal hydride was able to store 90% of its maximum hydrogen capacity at the end of 300 s. |
| Gupta S. and Sharma V. K. [163] | La0.9Ce0.1Ni5 | Copper internal longitudinal fins without complex modification and outer water jacket. Reactor of 163 mm length and 33 mm diameter. | The optimum fin structure: 12 fins, fin height 12 mm, fin thickness 2 mm. Reduction in the overall reaction time by almost 500 s; reduction in the rise in average MH bed temperature by 22.3 K during absorption; reduction in the rise in average MH bed temperature by 6.8 K during desorption. |
| Bhouri M. et al. [100] | NaAlH4 + 2% TiCl3 1/3AlCl3 + 0.5%FeCl3 | Each module contains seven finned tubes spaced uniformly and arranged in a triangular array inside a cylindrical shell. | A 41% improvement in hydrogen charge rate after 720 s of charging. |
| Parida A. and Muthukumar P. [164] | MmNi4.6Al0.4 | Three different fin configurations: longitudinal, transverse and spiral fins. | A 0.5 kg reactor can discharge hydrogen at the rate of 2.27 ppm for 2000 s. |
| Corgnale C. et al. [165] | MOF-5 | Longitudinal honeycomb aluminum structure. | The proposed adsorption system was also shown to discharge all available hydrogen in less than 500 s; work in cryogenic conditions; and have a nominal heating power of 100 W. |
| George M. and Mohan G. [166] | TiCl3 catalyzed NaAlH4 | Honeycomb structure. | At the optimum length/thickness ratio, the device, designed to charge 0.01 kg of hydrogen in 10 min, weighs 1.2 kg. |
| Zhang S. et al. [167] | LaNi5 | Configurations with straight fins, fan-shaped fins and quadratic curve-shaped fins. | Reduction in reaction time by 25%. |
| Bai X. S. et al. [168] | LaNi5 | A tree-shaped longitudinal fin. | Compared to the radial fin reactor, the hydrogen absorption time to reach 90% saturation is reduced by almost 20.7% for the optimized tree-shaped fin reactor. For the MHs thermal conductivity of 1.1 W/(mK), 3 W/(mK) and 5 W/(mK), the charging times for the mean bed temperature to reach 300 K are 1645 s, 1434 s and 1355 s, respectively. |
| Krishna K. V. et al. (c) [169] | LaNi5 | Bio-inspired leaf-vein type fins. | The optimized 7° inclination angle design with four keels required 57 s to reach 90% storage capacity and reduced absorption time by 73% compared to a longitudinally finned heat exchanger. |
| Keshari V. and Maiya M. P. [170] | LaNi5 | Copper pin fins and cooling tubes. | Total absorption time of 636 s with maximum storage capacity of 1.4 wt% (15 bar H2 gas supply pressure, heat transfer fluid temperature of 298 K, flow rate of 6.75 L/min). |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kudiiarov, V.; Elman, R.; Pushilina, N.; Kurdyumov, N. State of the Art in Development of Heat Exchanger Geometry Optimization and Different Storage Bed Designs of a Metal Hydride Reactor. Materials 2023, 16, 4891. https://doi.org/10.3390/ma16134891
Kudiiarov V, Elman R, Pushilina N, Kurdyumov N. State of the Art in Development of Heat Exchanger Geometry Optimization and Different Storage Bed Designs of a Metal Hydride Reactor. Materials. 2023; 16(13):4891. https://doi.org/10.3390/ma16134891
Chicago/Turabian StyleKudiiarov, Viktor, Roman Elman, Natalia Pushilina, and Nikita Kurdyumov. 2023. "State of the Art in Development of Heat Exchanger Geometry Optimization and Different Storage Bed Designs of a Metal Hydride Reactor" Materials 16, no. 13: 4891. https://doi.org/10.3390/ma16134891
APA StyleKudiiarov, V., Elman, R., Pushilina, N., & Kurdyumov, N. (2023). State of the Art in Development of Heat Exchanger Geometry Optimization and Different Storage Bed Designs of a Metal Hydride Reactor. Materials, 16(13), 4891. https://doi.org/10.3390/ma16134891







