Surface Engineering of Nanomaterials with Polymers, Biomolecules, and Small Ligands for Nanomedicine
Abstract
1. Introduction
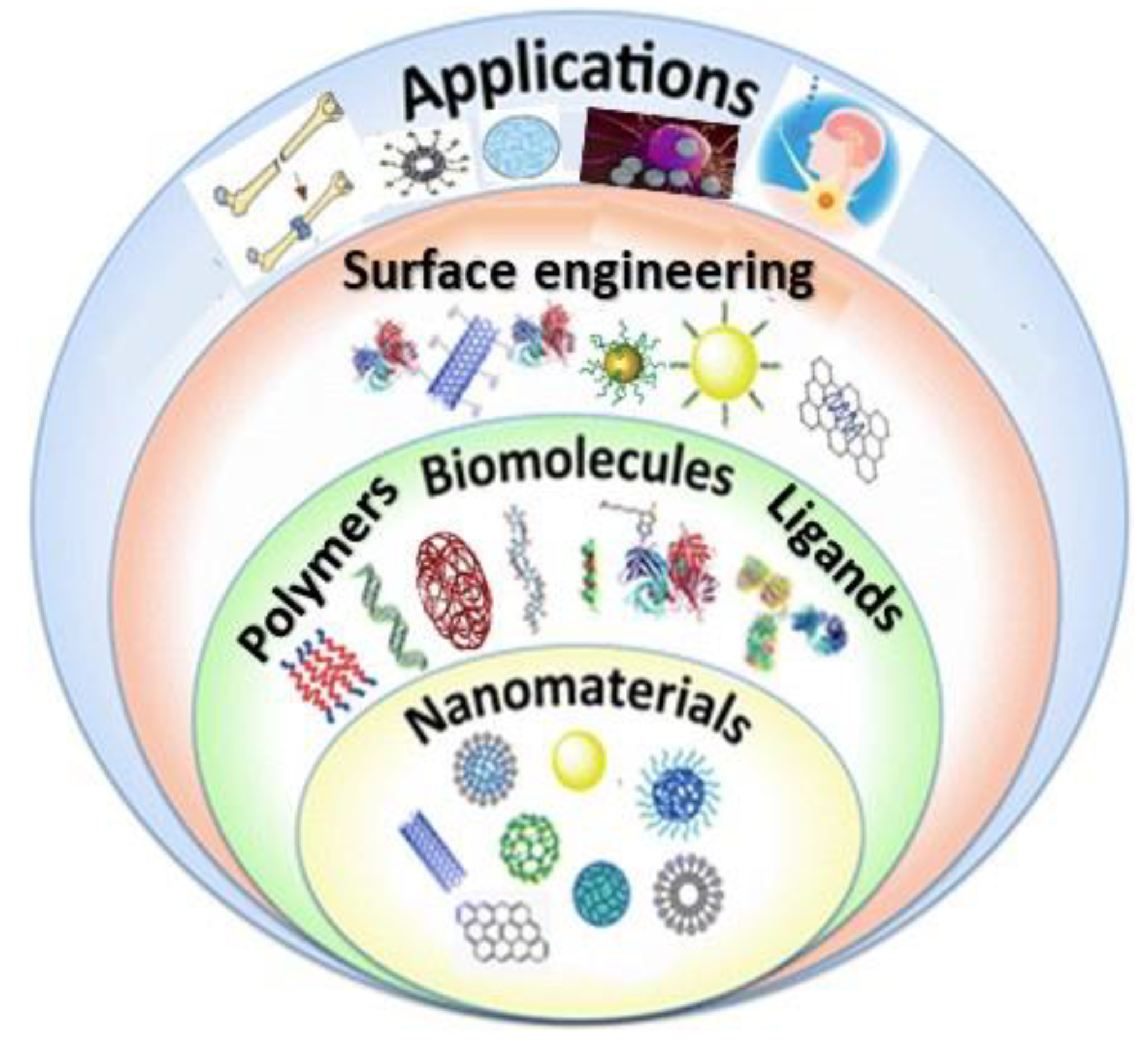
2. Strategies for Surface Functionalization of Nanomaterials
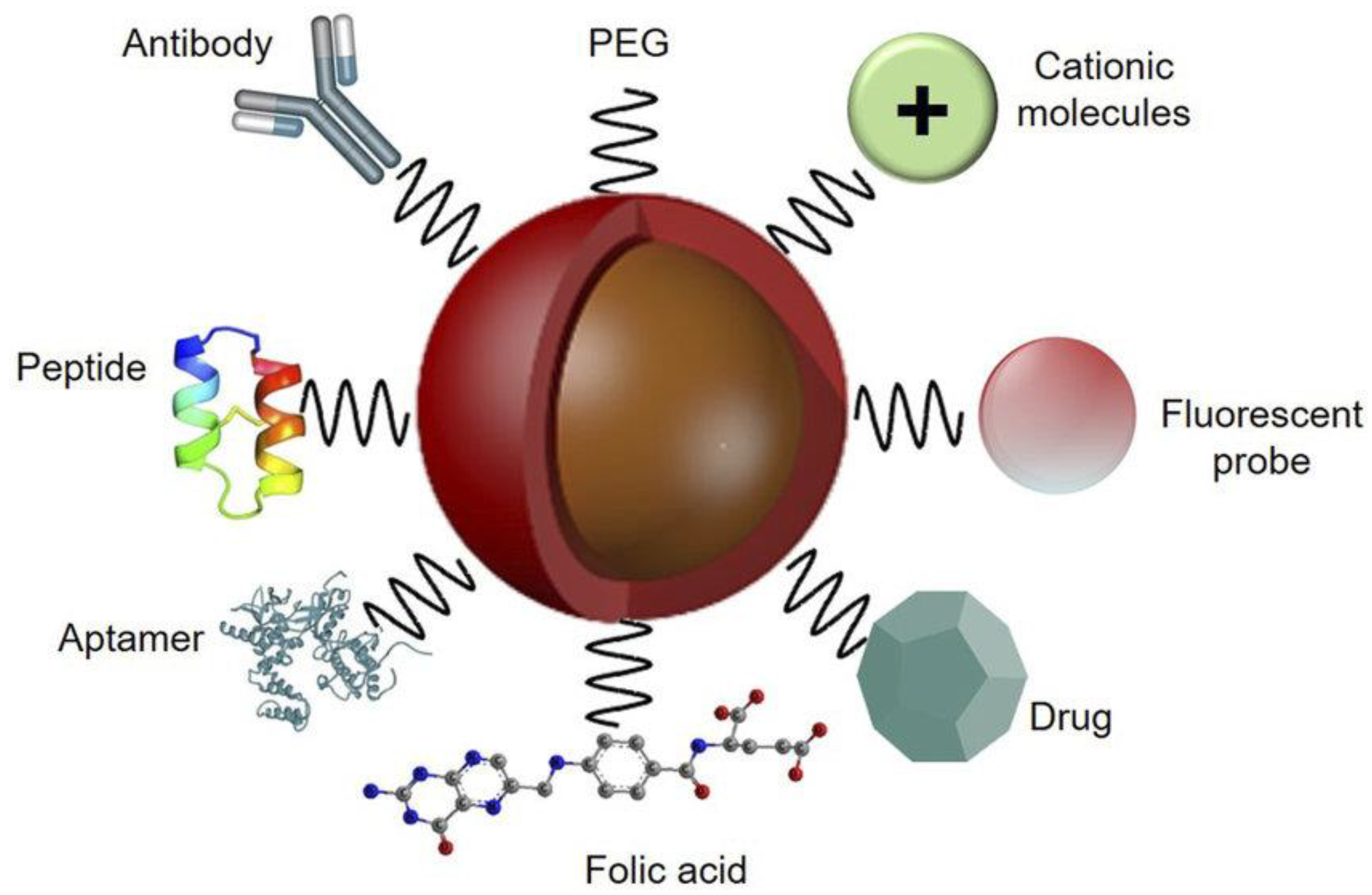
2.1. Functionalization by Covalent Conjugation
2.2. Functionalization by Noncovalent Conjugation
2.3. Functionalization by Biomolecules
2.4. Functionalization by Polymers
2.5. Functionalization by Small Ligands
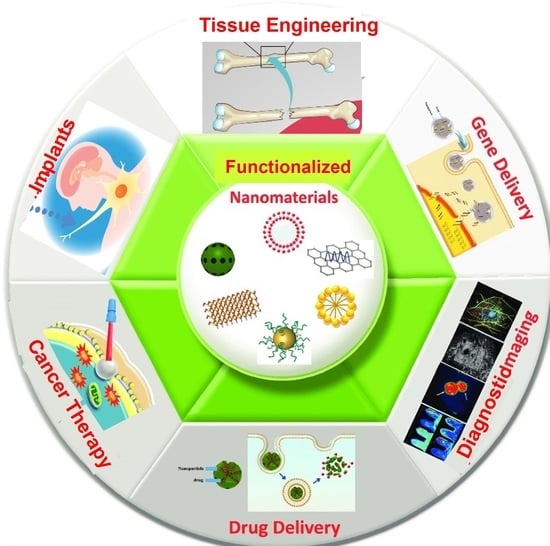
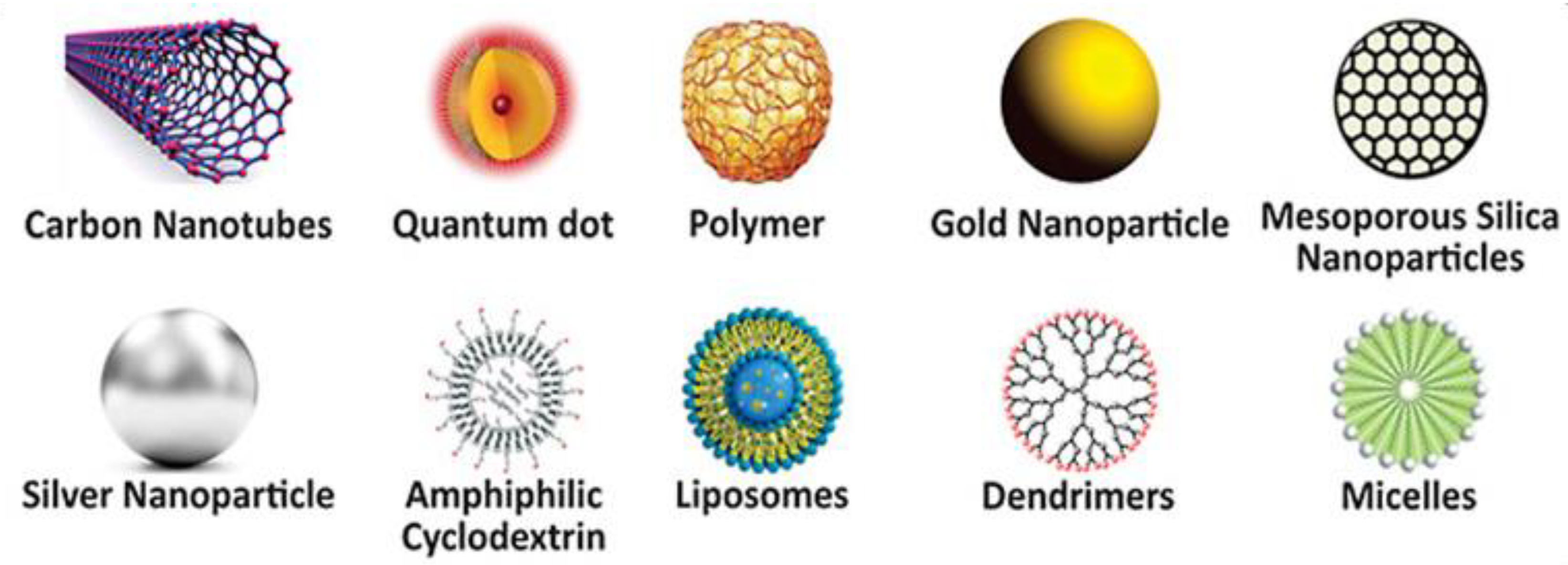
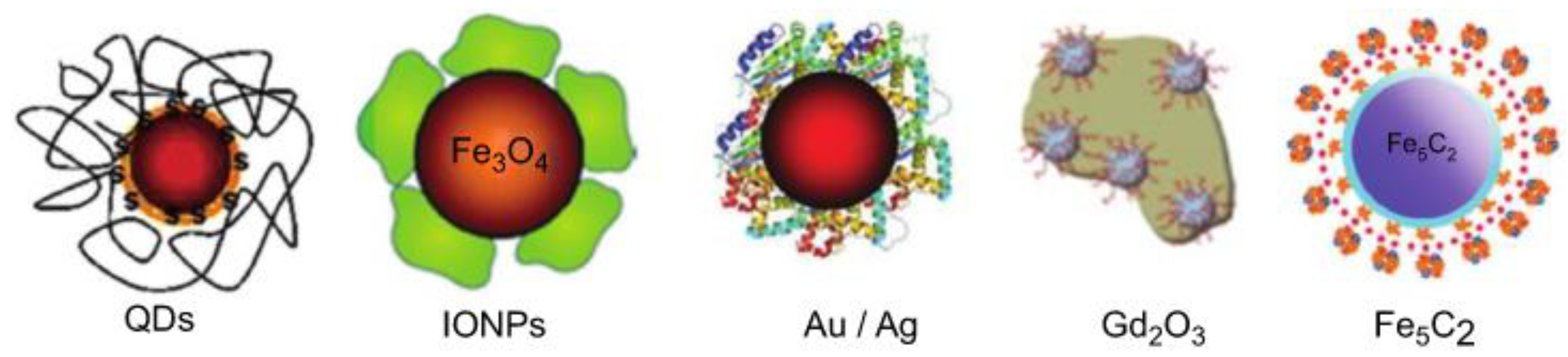
3. Functionalized Nanomaterials
3.1. Metallic Nanoparticles
3.2. Metal Oxide-Based Nanomaterials
3.3. Ceramic-Based Nanomaterials
3.4. Carbon-Based Nanomaterials
3.5. Polymeric Nanomaterials
4. Cytotoxicity: The Role of Functionalization
5. Cost-Effective Functionalization
6. Applications of Functionalized Nanomaterials in Biomedicine
6.1. Diagnostic Implications of Functionalized Nanomaterials
6.2. Therapeutic Applications of Functionalized Nanomaterials
7. Functionalized Nanomaterials: Drug/Gene Delivery
8. Functionalized Nanomaterials: Regenerative Medicine
9. Functionalized Nanomaterials: Cancer Therapy
10. Functionalized Nanomaterials: Medical Implants
11. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| aniline functionalized graphene quantum dots | a-GQDs |
| aptameric functionalized materials | AFMs |
| carbon nanotubes | CNTs |
| carbon quantum dots | CQDs |
| cationic β-cyclodextrin-based polymer | CCD/P |
| docetaxel-loaded liposomes functionalized with transferrin | LIP-DTX-TF |
| dopamine | DOPA |
| dopamine-polyethylene glycol-carboxylic acid | DPA-PEG-COOH |
| enhanced permeability and retention | EPR |
| extra-cellular matrix | ECM |
| folate receptor | FR |
| folic acid | FA |
| folic acid-coated gold nanoparticles conjugated with fluorophore | FA-Au-FITC |
| functionalized nanoparticles | FNPs |
| gold nanoparticles | AuNPs |
| graphene oxide | GO |
| hyaluronic acid | HLA |
| hydroxyapatite | HDA |
| lactate dehydrogenase | LDH |
| poly(lactic-co-glycolic acid) | PLGA |
| magnetic nanoparticles | MNPs |
| magnetic resonance imaging | MRI |
| mesoporous silica nanoparticles | MSN |
| methacryl polyhedral oligomeric silsesquioxane | M-POSS |
| nanoparticles | NPs |
| near-infrared | NIR |
| nitrogen-doped carbon quantum dots | NCQDs |
| nitrogen-doped graphene quantum dots | N-GQDs |
| oleic acid-coated iron oxide NPs | Fe3O4-OA |
| phenyl boronic acid | PBA |
| phenylethylamine | PhEA |
| photodynamic therapy | PDT |
| photothermal | PT |
| piperine | PIP |
| poly(acrylic acid) | PAA |
| polyethylene glycol | PEG |
| polyethylene glycol with thiol end group | thiol-PEG |
| polyethylene glycol-gelatin-chitosan-hyaluronidase-5-fluorouracil | CS-HYL-5-FU-PEG-G |
| positron emission tomography | PET |
| quantum dots | QDs |
| receptor-mediated endocytosis | RME |
| reticulum endothelial system | RES |
| sodium alginate (SA)–polyvinyl alcohol (PVA)–bovin serum albumin | SA-PVA-BSA |
| sodium alginate/polyethylene glycol (vinyl alcohol) | SA/PVA/Ca |
| superparamagnetic iron oxide NPs | SPIONs |
| tetraethylorthosilicate | TEOS |
| ultra-small superparamagnetic iron oxide NP | USPIONs |
References
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; del Pilar Rodriguez-Torres, M.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef] [PubMed]
- Bilal, M.; Qindeel, M.; Raza, A.; Mehmood, S.; Rahdar, A. Stimuli-responsive nanoliposomes as prospective nanocarriers for targeted drug delivery. J. Drug Deliv. Sci. Technol. 2021, 66, 102916. [Google Scholar] [CrossRef]
- Rauf, A.; Tabish, T.A.; Ibrahim, I.M.; Hassan, M.R.U.; Tahseen, S.; Sandhu, M.A.; Shahnaz, G.; Rahdar, A.; Cucchiarini, M.; Pandey, S. Design of Mannose-Coated Rifampicin nanoparticles modulating the immune response and Rifampicin induced hepatotoxicity with improved oral drug delivery. Arab. J. Chem. 2021, 14, 103321. [Google Scholar] [CrossRef]
- Hong, E.J.; Choi, D.G.; Shim, M.S. Targeted and effective photodynamic therapy for cancer using functionalized nanomaterials. Acta Pharm. Sin. B 2016, 6, 297–307. [Google Scholar] [CrossRef]
- Rahdar, A.; Hajinezhad, M.R.; Hamishekar, H.; Ghamkhari, A.; Kyzas, G.Z. Copolymer/graphene oxide nanocomposites as potential anticancer agents. Polym. Bull. 2021, 78, 4877–4898. [Google Scholar] [CrossRef]
- Zhang, G.; Khan, A.A.; Wu, H.; Chen, L.; Gu, Y.; Gu, N. The Application of Nanomaterials in Stem Cell Therapy for Some Neurological Diseases. Curr. Drug Targets 2018, 19, 279–298. [Google Scholar] [CrossRef]
- Theus, A.S.; Ning, L.; Jin, L.; Roeder, R.K.; Zhang, J.; Serpooshan, V. Nanomaterials for bioprinting: Functionalization of tissue-specific bioinks. Essays Biochem. 2021, 65, 429–439. [Google Scholar] [CrossRef]
- Lloyd, J.R.; Byrne, J.M.; Coker, V.S. Biotechnological synthesis of functional nanomaterials. Curr. Opin. Biotechnol. 2011, 22, 509–515. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M. Hot Topics in Macromolecular Science. Macromol 2021, 1, 173–176. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M. Nanoparticle reinforced polymers. Polymers 2019, 11, 625. [Google Scholar] [CrossRef]
- Kobayashi, K.; Wei, J.; Iida, R.; Ijiro, K.; Niikura, K. Surface engineering of nanoparticles for therapeutic applications. Polym. J. 2014, 46, 460–468. [Google Scholar] [CrossRef]
- Sanità, G.; Carrese, B.; Lamberti, A. Nanoparticle Surface Functionalization: How to Improve Biocompatibility and Cellular Internalization. Front. Mol. Biosci. 2020, 7, 587012. [Google Scholar] [CrossRef] [PubMed]
- Díez-Pascual, A.M. Effect of Graphene Oxide on the Properties of Poly(3-Hydroxybutyrate-co-3-Hydroxyhexanoate. Polymers 2021, 13, 2233. [Google Scholar] [CrossRef] [PubMed]
- Razzaq, S.; Rauf, A.; Raza, A.; Akhtar, S.; Tabish, T.A.; Sandhu, M.A.; Zaman, M.; Ibrahim, I.M.; Shahnaz, G.; Rahdar, A.; et al. Multifunctional Polymeric Micelle for Targeted Delivery of Paclitaxel by the Inhibition of the P-Glycoprotein Transporters. Nanomaterials 2021, 11, 2858. [Google Scholar] [CrossRef]
- Rahdar, A.; Hasanein, P.; Bilal, M.; Beyzaei, H.; Kyzas, G.Z. Quercetin-loaded F127 nanomicelles: Antioxidant activity and protection against renal injury induced by gentamicin in rats. Life Sci. 2021, 276, 119420. [Google Scholar] [CrossRef]
- Er, S.; Laraib, U.; Arshad, R.; Sargazi, S.; Rahdar, A.; Pandey, S.; Thakur, V.K.; Díez-Pascual, A.M. Amino Acids, Peptides, and Proteins: Implications for Nanotechnological Applications in Biosensing and Drug/Gene Delivery. Nanomaterials 2021, 11, 3002. [Google Scholar] [CrossRef]
- Mrówczyński, R.; Grześkowiak, B.F. Biomimetic Catechol-Based Nanomaterials for Combined Anticancer Therapies. Nanoeng. Biomater. Biomed. Appl. 2022, 2, 145–180. [Google Scholar] [CrossRef]
- Jamir, M.; Islam, R.; Pandey, L.M.; Borah, J. Effect of surface functionalization on the heating efficiency of magnetite nanoclusters for hyperthermia application. J. Alloy. Compd. 2021, 854, 157248. [Google Scholar] [CrossRef]
- Nejati, K.; Dadashpour, M.; Gharibi, T.; Mellatyar, H.; Akbarzadeh, A. Biomedical Applications of Functionalized Gold Nanoparticles: A Review. J. Clust. Sci. 2021, 33, 1–16. [Google Scholar] [CrossRef]
- Sonju, J.J.; Dahal, A.; Singh, S.S.; Jois, S.D. Peptide-functionalized liposomes as therapeutic and diagnostic tools for cancer treatment. J. Control. Release 2021, 329, 624–644. [Google Scholar] [CrossRef]
- Jazayeri, M.H.; Amani, H.; Pourfatollah, A.A.; Pazoki-Toroudi, H.; Sedighimoghaddam, B. Various methods of gold nanoparticles (GNPs) conjugation to antibodies. Sens. Bio-Sens. Res. 2016, 9, 17–22. [Google Scholar] [CrossRef]
- Li, H.; Wang, Q.; Liang, G. Phase Transfer of Hydrophobic Nanoparticles Functionalized with Zwitterionic Bisphosphonate Ligands for Renal-Clearable Imaging Nanoprobes. ACS Appl. Nano Mater. 2021, 4, 2621–2633. [Google Scholar] [CrossRef]
- Karthik, V.; Selvakumar, P.; Kumar, P.S.; Vo, D.-V.N.; Gokulakrishnan, M.; Keerthana, P.; Elakkiya, V.T.; Rajeswari, R. Graphene-based materials for environmental applications: A review. Environ. Chem. Lett. 2021, 19, 3631–3644. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Diez-Vicente, A. L Antibacterial SnO2 nanorods as efficient fillers of poly(propylene fumarate-co-ethylene glycol) biomaterials. Mater. Sci. Eng. C 2017, 78, 806–816. [Google Scholar] [CrossRef]
- Alshamrani, M. Broad-Spectrum Theranostics and Biomedical Application of Functionalized Nanomaterials. Polymers 2022, 14, 1221. [Google Scholar] [CrossRef]
- Xia, Q.; Huang, J.; Feng, Q.; Chen, X.; Liu, X.; Li, X.; Zhang, T.; Xiao, S.; Li, H.; Zhong, Z.; et al. Size- and cell type-dependent cellular uptake, cytotoxicity and in vivo distribution of gold nanoparticles. Int. J. Nanomed. 2019, 14, 6957–6970. [Google Scholar] [CrossRef]
- Hirayama, H.; Amolegbe, S.A.; Islam, M.S.; Rahman, M.A.; Goto, N.; Sekine, Y.; Hayami, S. Encapsulation and controlled release of an antimalarial drug using surface functionalized mesoporous silica nanocarriers. J. Mater. Chem. B 2021, 9, 5043–5046. [Google Scholar] [CrossRef]
- Guadagnini, R.; Halamoda Kenzaoui, B.; Walker, L.; Pojana, G.; Magdolenova, Z.; Bilanicova, D.; Saunders, M.; Juillerat-Jeanneret, L.; Marcomini, A.; Huk, A. Toxicity screenings of nanomaterials: Challenges due to interference with assay processes and components of classic in vitro tests. Nanotoxicology 2015, 9, 13–24. [Google Scholar] [CrossRef]
- Ellah, N.A.; Abouelmagd, S. Surface functionalization of polymeric nanoparticles for tumor drug delivery: Approaches and challenges. Expert Opin. Drug Deliv. 2017, 14, 201–214. [Google Scholar] [CrossRef]
- Fernandes, M.A.; Eloy, J.O.; Luiz, M.T.; Junior, S.L.R.; Borges, J.C.; De la Fuente, L.R.; Luis, C.O.-D.S.; Marchetti, J.M.; Santos-Martinez, M.J.; Chorilli, M. Transferrin-functionalized liposomes for docetaxel delivery to prostate cancer cells. Colloids Surf. A Physicochem. Eng. Asp. 2021, 611, 125806. [Google Scholar] [CrossRef]
- Rong, L.; Qin, S.-Y.; Zhang, C.; Cheng, Y.-J.; Feng, J.; Wang, S.-B.; Zhang, X.-Z. Biomedical applications of functional peptides in nano-systems. Mater. Today Chem. 2018, 9, 91–102. [Google Scholar] [CrossRef]
- Xie, S.; Ai, L.; Cui, C.; Fu, T.; Cheng, X.; Qu, F.; Tan, W. Functional Aptamer-Embedded Nanomaterials for Diagnostics and Therapeutics. ACS Appl. Mater. Interfaces 2021, 13, 9542–9560. [Google Scholar] [CrossRef] [PubMed]
- Farahavar, G.; Abolmaali, S.S.; Gholijani, N.; Nejatollahi, F. Antibody-guided nanomedicines as novel breakthrough therapeutic, diagnostic and theranostic tools. Biomater. Sci. 2019, 7, 4000–4016. [Google Scholar] [CrossRef] [PubMed]
- Mout, R.; Moyano, D.F.; Rana, S.; Rotello, V.M. Surface functionalization of nanoparticles for nanomedicine. Chem. Soc. Rev. 2012, 41, 2539–2544. [Google Scholar] [CrossRef]
- Mariadoss, A.V.A.; Saravanakumar, K.; Sathiyaseelan, A.; Venkatachalam, K.; Wang, M.-H. Folic acid functionalized starch encapsulated green synthesized copper oxide nanoparticles for targeted drug delivery in breast cancer therapy. Int. J. Biol. Macromol. 2020, 164, 2073–2084. [Google Scholar] [CrossRef]
- Wei, W.; Zhang, X.; Zhang, S.; Wei, G.; Su, Z. Biomedical and bioactive engineered nanomaterials for targeted tumor photothermal therapy: A review. Mater. Sci. Eng. C 2019, 104, 109891. [Google Scholar] [CrossRef]
- Azevedo, C.; Macedo, M.H.; Sarmento, B. Strategies for the enhanced intracellular delivery of nanomaterials. Drug Discov. Today 2018, 23, 944–959. [Google Scholar] [CrossRef]
- Farzin, L.; Shamsipur, M.; Samandari, L.; Sheibani, S. Advances in the design of nanomaterial-based electrochemical affinity and enzymatic biosensors for metabolic biomarkers: A review. Microchim. Acta 2018, 185, 1–25. [Google Scholar] [CrossRef]
- Gravely, M.; Safaee, M.M.; Roxbury, D. Biomolecular Functionalization of a Nanomaterial To Control Stability and Retention within Live Cells. Nano Lett. 2019, 19, 6203–6212. [Google Scholar] [CrossRef]
- Montaseri, H.; Kruger, C.A.; Abrahamse, H. Review: Organic nanoparticle based active targeting for photodynamic therapy treatment of breast cancer cells. Oncotarget 2020, 11, 2120–2136. [Google Scholar] [CrossRef]
- Gole, B.; Sanyal, U.; Banerjee, R.; Mukherjee, P.S. High Loading of Pd Nanoparticles by Interior Functionalization of MOFs for Heterogeneous Catalysis. Inorg. Chem. 2016, 55, 2345–2354. [Google Scholar] [CrossRef] [PubMed]
- Bertella, S.; Luterbacher, J.S. Lignin Functionalization for the Production of Novel Materials. Trends Chem. 2020, 2, 440–453. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M. Chemical Functionalization of Carbon Nanotubes with Polymers: A Brief Overview. Macromol 2021, 1, 64–83. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M. Carbon-Based Nanomaterials. Int. J. Mol. Sci. 2021, 22, 7726. [Google Scholar] [CrossRef] [PubMed]
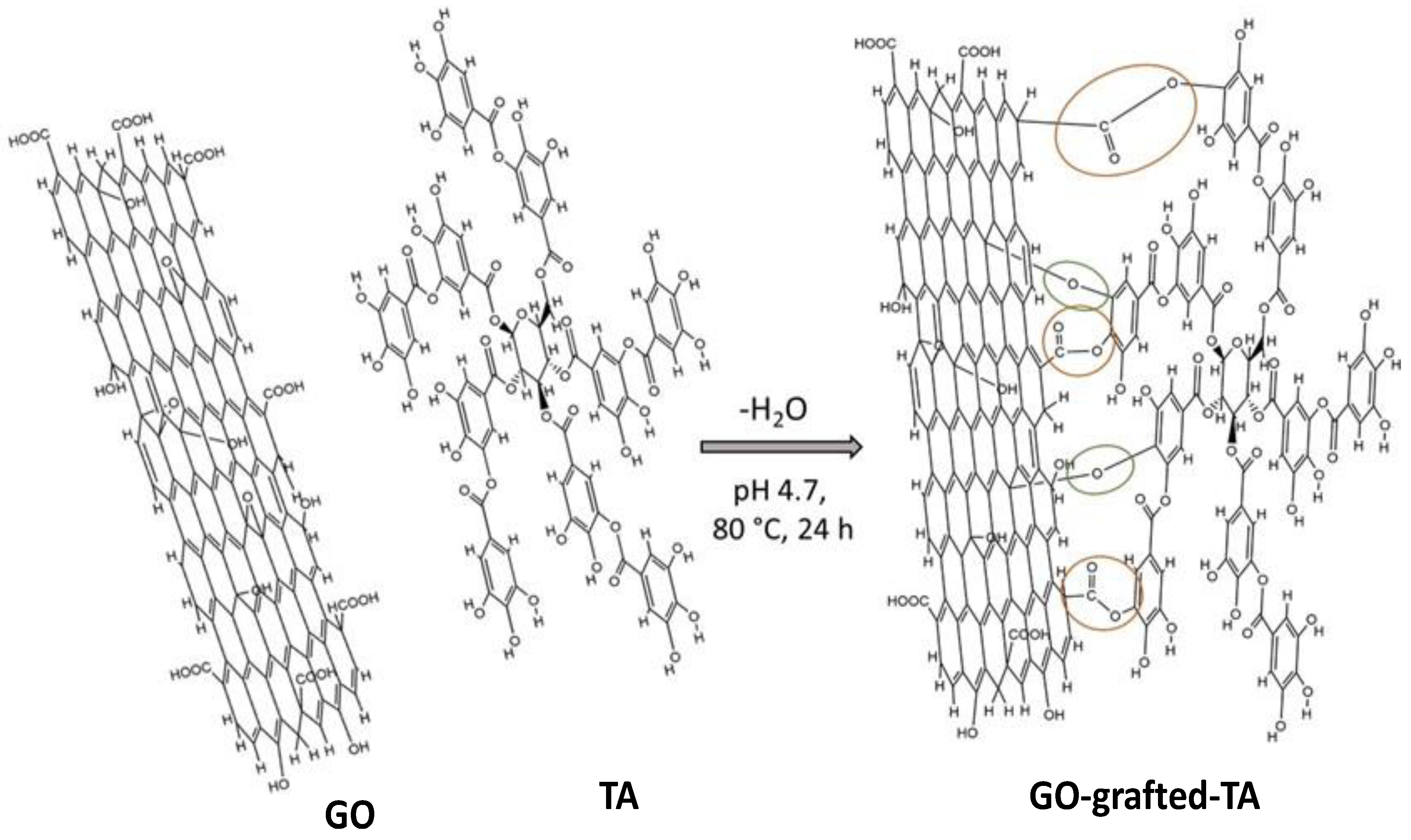
- Sainz-Urruela, C.; Vera-López, S.; Andrés, M.P.S.; Díez-Pascual, A.M. Surface functionalization of graphene oxide with tannic acid: Covalent vs non-covalent approaches. J. Mol. Liq. 2022, 357, 119104. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M. Development of Graphene-Based Polymeric Nanocomposites: A Brief Overview. Polymers 2021, 13, 2978. [Google Scholar] [CrossRef]
- Chen, Q.; Liu, Z. Albumin carriers for cancer theranostics: A conventionalplatform with new promise. Adv. Mater. 2016, 28, 10557–10566. [Google Scholar] [CrossRef]
- Bolaños, K.; Kogan, M.J.; Araya, E. Capping gold nanoparticles with albumin to improve their biomedical properties. Int. J. Nanomed. 2019, 14, 6387–6406. [Google Scholar] [CrossRef]
- Chakraborty, A.; Dhar, P. A review on potential of proteins as an excipient for developing a nano-carrier delivery system. Crit. Rev. Ther. Drug Carr. Syst. 2017, 34, 453–488. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; García-García, D.; Andrés, M.P.S.; Vera, S. Determination of riboflavin based on fluorescence quenching by graphene dispersions in polyethylene glycol. RSC Adv. 2016, 6, 1968. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Díez-Vicente, A.L. Multifunctional poly(glycolic acid-co-propylene fumarate) electrospun fibers reinforced with graphene oxide and hydroxyapatite nanorods. J. Mater. Chem. B 2017, 5, 4084–4096. [Google Scholar] [CrossRef] [PubMed]
- Horo, H.; Bhattacharyya, S.; Mandal, B.; Kundu, L.M. Synthesis of functionalized silk-coated chitosan-gold nanoparticles and microparticles for target-directed delivery of antitumor agents. Carbohydr. Polym. 2021, 258, 117659. [Google Scholar] [CrossRef] [PubMed]
- Díez-Pascual, A.M.; Díez-Vicente, A.L. Electrospun fibers of chitosan-grafted polycaprolactone/poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) blends. J. Mater. Chem. B 2016, 4, 600–612. [Google Scholar] [CrossRef] [PubMed]
- Diez-Pascual, A.M.; Diez-Vicente, A.L. Poly(propylene fumarate)/Polyethylene Glycol-Modified Graphene Oxide Nanocomposites for Tissue Engineering. ACS Appl. Mater. Interfaces 2016, 8, 17902–17914. [Google Scholar] [CrossRef] [PubMed]
- Kleinfeldt, L.; Gädke, J.; Biedendieck, R.; Krull, R.; Garnweitner, G. Spray-Dried Hierarchical Aggregates of Iron Oxide Nanoparticles and Their Functionalization for Downstream Processing in Biotechnology. ACS Omega 2019, 4, 16300–16308. [Google Scholar] [CrossRef]
- Makvandi, P.; Wang, C.Y.; Zare, E.N.; Borzacchiello, A.; Niu, L.N.; Tay, F.R. Metal-Based Nanomaterials in Biomedical Applications: Antimicrobial Activity and Cytotoxicity Aspects. Adv. Funct. Mater. 2020, 30, 1910021. [Google Scholar] [CrossRef]
- Mazzotta, E.; Orlando, C.; Muzzalupo, R. New Nanomaterials with Intrinsic Antioxidant Activity by Surface Functionalization of Niosomes with Natural Phenolic Acids. Pharmaceutics 2021, 13, 766. [Google Scholar] [CrossRef]
- Delfi, M.; Ghomi, M.; Zarrabi, A.; Mohammadinejad, R.; Taraghdari, Z.; Ashrafizadeh, M.; Zare, E.; Agarwal, T.; Padil, V.; Mokhtari, B.; et al. Functionalization of Polymers and Nanomaterials for Biomedical Applications: Antimicrobial Platforms and Drug Carriers. Prosthesis 2020, 2, 117–139. [Google Scholar] [CrossRef]
- Gary-Bobo, M.; Hocine, O.; Brevet, D.; Maynadier, M.; Raehm, L.; Richeter, S.; Charasson, V.; Loock, B.; Morère, A.; Maillard, P.; et al. Cancer therapy improvement with mesoporous silica nanoparticles combining targeting, drug delivery and PDT. Int. J. Pharm. 2012, 423, 509–515. [Google Scholar] [CrossRef]
- Jose, J.; Kumar, R.; Harilal, S.; Mathew, G.E.; Parambi, D.G.T.; Prabhu, A.; Uddin, M.S.; Aleya, L.; Kim, H.; Mathew, B. Magnetic nanoparticles for hyperthermia in cancer treatment: An emerging tool. Environ. Sci. Pollut. Res. 2020, 27, 19214–19225. [Google Scholar] [CrossRef]
- Sharmeen, S.; Rahman, A.M.; Lubna, M.M.; Salem, K.S.; Islam, R.; Khan, M.A. Polyethylene glycol functionalized carbon nanotubes/gelatin-chitosan nanocomposite: An approach for significant drug release. Bioact. Mater. 2018, 3, 236–244. [Google Scholar] [CrossRef]
- Muhammad, M.; Shao, C.S.; Huang, Q. Aptamer-functionalized Au nanoparticles array as the effective SERS biosensor for label-free detection of interleukin-6 in serum. Sens. Actuators B Chem. 2021, 334, 129607. [Google Scholar] [CrossRef]
- Mahmoudpour, M.; Ding, S.; Lyu, Z.; Ebrahimi, G.; Du, D.; Dolatabadi, J.E.N.; Torbati, M.; Lin, Y. Aptamer functionalized nanomaterials for biomedical applications: Recent advances and new horizons. Nano Today 2021, 39, 101177. [Google Scholar] [CrossRef]
- Mahmoudpour, M.; Karimzadeh, Z.; Ebrahimi, G.; Hasanzadeh, M.; Ezzati Nazhad Dolatabadi, J. Synergizing Functional Nanomaterials with Aptamers Based on Electrochemical Strategies for Pesticide Detection: Current Status and Perspectives. Crit. Rev. Anal. Chem. 2021, 1–28. [Google Scholar] [CrossRef] [PubMed]
- Hassanisaadi, M.; Bonjar GH, S.; Rahdar, A.; Pandey, S.; Hosseinipour, A.; Abdolshahi, R. Environmentally Safe Biosynthesis of Gold Nanoparticles Using Plant Water Extracts. Nanomaterials 2021, 11, 2033. [Google Scholar] [CrossRef] [PubMed]
- Simon, S.; Ciceo-Lucacel, R.; Radu, T.; Baia, L.; Ponta, O.; Iepure, A.; Simon, V. Gold nanoparticles developed in sol–gel derived apatite—bioactive glass composites. J. Mater. Sci. Mater. Med. 2012, 23, 1193–1201. [Google Scholar] [CrossRef]
- Qingling, F.; Wei, J.; Aifantis, K.E.; Fan, Y.; Feng, Q.; Cui, F.-Z.; Watari, F. Current investigations into magnetic nanoparticles for biomedical applications. J. Biomed. Mater. Res. Part A 2016, 104, 1285–1296. [Google Scholar] [CrossRef]
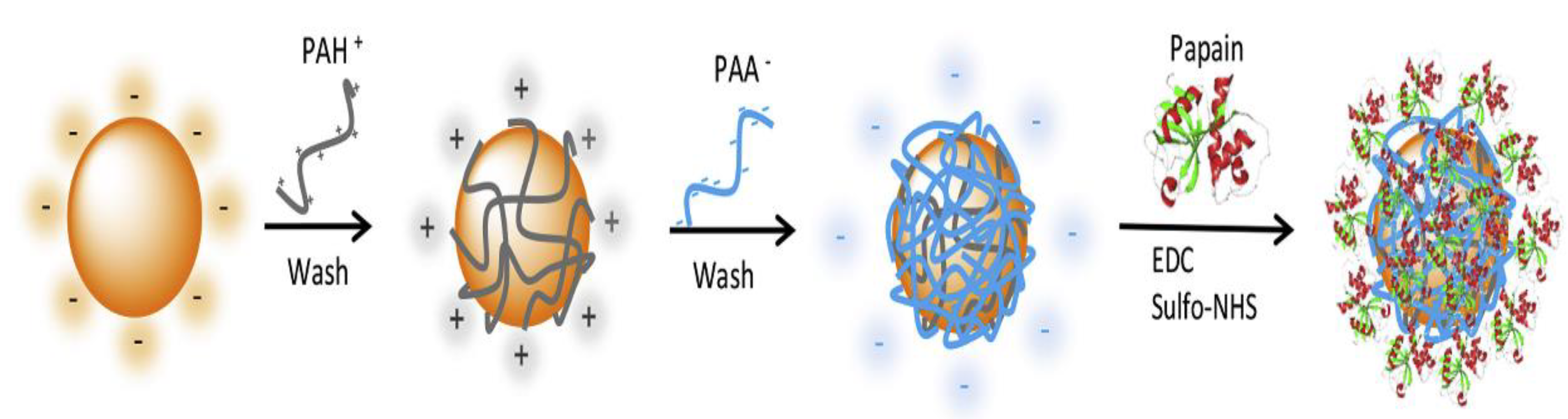
- Liu, S.; Höldrich, M.; Sievers-Engler, A.; Horak, J.; Lämmerhofer, M. Papain-functionalized gold nanoparticles as heterogeneous biocatalyst for bioanalysis and biopharmaceuticals analysis. Anal. Chim. Acta 2017, 963, 33–43. [Google Scholar] [CrossRef]
- Pourjavadi, A.; Bagherifard, M.; Doroudian, M. Synthesis of micelles based on chitosan functionalized with gold nanorods as a light sensitive drug delivery vehicle. Int. J. Biol. Macromol. 2020, 149, 809–818. [Google Scholar] [CrossRef]
- Tiwari, P.M.; Vig, K.; Dennis, V.A.; Singh, S.R. Functionalized gold nanoparticles and their biomedical applications. Nanomaterials 2011, 1, 31–63. [Google Scholar] [CrossRef]
- Donoso-González, O.; Lodeiro, L.; Aliaga, Á.E.; Laguna-Bercero, M.A.; Bollo, S.; Kogan, M.J.; Yutronic, N.; Sierpe, R. Functionalization of gold nanostars with cationic β-cyclodextrin-based polymer for drug co-loading and SERS monitoring. Pharmaceutics 2021, 13, 261. [Google Scholar] [CrossRef] [PubMed]
- Shon, Y.S.; Choo, H. [60]Fullerene-linked gold nanoparticles: Synthesis and layer-by-layer growth on a solid surface. Chem. Commun. 2002, 21, 2560–2561. [Google Scholar] [CrossRef]
- Sudeep, P.K.; Ipe, B.I.; Thomas, K.G.; George, M.V.; Barazzouk, S.; Hotchandani, S.; Kamat, P.V. Fullerene-functionalized gold nanoparticles. A self-assembled photoactive antenna-metal nanocore assembly. Nano Lett. 2002, 2, 29–35. [Google Scholar] [CrossRef]
- Yaseen, M.; Humayun, M.; Khan, A.; Usman, M.; Ullah, H.; Tahir, A.; Ullah, H. Preparation, functionalization, modification, and applications of nanostructured gold: A critical review. Energies 2021, 14, 1278. [Google Scholar] [CrossRef]
- Liz-Marzán, L.M.; Giersig, M.; Mulvaney, P. Synthesis of nanosized gold−silica core−shell particles. Langmuir 1996, 12, 4329–4335. [Google Scholar] [CrossRef]
- Bhargava, A.; Dev, A.; Mohanbhai, S.J.; Pareek, V.; Jain, N.; Choudhury, S.R.; Panwar, J.; Karmakar, S. Pre-coating of protein modulate patterns of corona formation, physiological stability and cytotoxicity of silver nanoparticles. Sci. Total Environ. 2021, 772, 144797. [Google Scholar] [CrossRef]
- Matsuo, T. Functionalization of Ruthenium Olefin-Metathesis Catalysts for Interdisciplinary Studies in Chemistry and Biology. Catalysts 2021, 11, 359. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Díez-Vicente, A.L. Nano-TiO2 Reinforced PEEK/PEI Blends as Biomaterials for Load-Bearing Implant Applications. ACS Appl. Mater. Interfaces 2015, 7, 5561–5573. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Díez-Vicente, A.L. Effect of TiO2 Nanoparticles on the Performance of Polyphenysulfone Biomaterial for Orthopaedic Implants. J. Mater. Chem. B 2014, 2, 7502–7514. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Diez-Vicente, A.L. High-Performance Aminated Poly(phenylene sulfide)/ZnO Nanocomposites for Medical Applications. ACS Appl. Mater. Interfaces 2014, 6, 10132–101045. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Diez-Vicente, A.L. Antimicrobial and sustainable food packaging based on poly(butylene adipate-co-terephthalate) and electrospun chitosan nanofibers. RSC Adv. 2015, 5, 93095. [Google Scholar] [CrossRef]
- Jalalian, S.H.; Taghdisi, S.M.; Hamedani, N.S.; Kalat, S.A.M.; Lavaee, P.; ZandKarimi, M.; Ghows, N.; Jaafari, M.R.; Naghibi, S.; Danesh, N.M.; et al. Epirubicin loaded super paramagnetic iron oxide nanoparticle-aptamer bioconjugate for combined colon cancer therapy and imaging in vivo. Eur. J. Pharm. Sci. 2013, 50, 191–197. [Google Scholar] [CrossRef] [PubMed]
- Wei, R.; Xu, Y.; Xue, M. Hollow iron oxide nanomaterials: Synthesis, functionalization, and biomedical applications. J. Mater. Chem. B 2021, 9, 1965–1979. [Google Scholar] [CrossRef] [PubMed]
- Cheah, P.; Brown, P.; Qu, J.; Tian, B.; Patton, D.L.; Zhao, Y. Versatile Surface Functionalization of Water-Dispersible Iron Oxide Nanoparticles with Precisely Controlled Sizes. Langmuir 2021, 37, 1279–1287. [Google Scholar] [CrossRef] [PubMed]
- Nayeem, J.; Al-Bari, A.A.; Mahiuddin; Rahman, A.; Mefford, O.T.; Ahmad, H.; Rahman, M. Silica coating of iron oxide magnetic nanoparticles by reverse microemulsion method and their functionalization with cationic polymer P(NIPAm-co-AMPTMA) for antibacterial vancomycin immobilization. Colloids Surf. A Physicochem. Eng. Asp. 2021, 611, 125857. [Google Scholar] [CrossRef]
- Sun, C.; Lee, J.S.H.; Zhang, M. Magnetic nanoparticles in MR imaging and drug delivery. Adv. Drug Deliv. Rev. 2008, 60, 1252–1265. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Díez-Vicente, A.L. Development of Linseed Oil/TiO2 Green Nanocomposites as Antimicrobial Coatings. J. Mater. Chem. B 2015, 3, 4458–4471. [Google Scholar] [CrossRef]
- Kundu, M.; Sadhukhan, P.; Ghosh, N.; Chatterjee, S.; Manna, P.; Das, J.; Sil, P.C. pH-responsive and targeted delivery of curcumin via phenylboronic acid-functionalized ZnO nanoparticles for breast cancer therapy. J. Adv. Res. 2019, 18, 161–172. [Google Scholar] [CrossRef]
- Al-Harbi, N.; Mohammed, H.; Al-Hadeethi, Y.; Bakry, A.S.; Umar, A.; Hussein, M.A.; Abbassy, M.A.; Vaidya, K.G.; Berakdar, G.A.; Mkawi, E.M.; et al. Silica-Based Bioactive Glasses and Their Applications in Hard Tissue Regeneration: A Review. Pharmaceuticals 2021, 20, 75. [Google Scholar] [CrossRef]
- Filho, O.P.; La Torre, G.P.; Hench, L.L. Effect of crystallization on apatite-layer formation of bioactive glass 45S5. J. Biomed. Mater. Res. Off. J. Soc. Biomater. Jpn. Soc. Biomater. 1996, 30, 509–514. [Google Scholar] [CrossRef]
- Aina, V.; Cerrato, G.; Martra, G.; Bergandi, L.; Costamagna, C.; Ghigo, D.; Malavasi, G.; Lusvardi, G.; Menabue, L. Gold-containing bioactive glasses: A solid-state synthesis to produce alternative biomaterials for bone implantations. J. R. Soc. Interface 2013, 10, 20121040. [Google Scholar] [CrossRef] [PubMed]
- Naffakh, M.; Diez-Pascual, A.M. Thermoplastic Polymer Nanocomposites Based on Inorganic Fullerene-like Nanoparticles and Inorganic Nanotubes. Inorganics 2014, 2, 291–312. [Google Scholar] [CrossRef]
- Gibson, I.R.; Bonfield, W. Novel synthesis and characterization of an AB-type carbonate-substituted hydroxyapatite. J. Biomed. Mater. Res. Off. J. Soc. Biomater. Jpn. Soc. Biomater. Aust. Soc. Biomater. Korean Soc. Biomater. 2002, 59, 697–708. [Google Scholar] [CrossRef] [PubMed]
- Ursino, H.L.; James, B.D.; Ludtka, C.M.; Allen, J.B. Bone tissue engineering. In Tissue Engineering Using Ceramics and Polymers; Elsevier: Amsterdam, The Netherlands, 2022; pp. 587–644. [Google Scholar]
- Díez-Pascual, A.M.; Rahdar, A. LbL Nano-Assemblies: A Versatile Tool for Biomedical and Healthcare Applications. Nanomaterials 2022, 12, 949. [Google Scholar] [CrossRef] [PubMed]
- Díez-Pascual, A.M. State of the Art in the Antibacterial and Antiviral Applications of Carbon-Based Polymeric Nanocomposites. Int. J. Mol. Sci. 2021, 22, 10511. [Google Scholar] [CrossRef]
- Schnorr, J.M.; Swager, T.M. Emerging applications of carbon nanotubes. Chem. Mater. 2011, 23, 646–657. [Google Scholar] [CrossRef]
- Naffakh, M.; Díez-Pascual, A.M.; Gómez-Fatou, M.A. New hybrid nanocomposites containing carbon nanotubes, inorganic fullerene-like WS2 nanoparticles and poly(ether ether ketone) (PEEK). J. Mater. Chem. 2011, 21, 7425. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Martínez, G.; González-Domínguez, J.M.; Ansón, A.; Martínez, M.T.; Gómez, M.A. Grafting of a hydroxylated poly(ether ether ketone) to the surface of single-walled carbon nanotubes. J. Mater. Chem. 2020, 20, 8285. [Google Scholar] [CrossRef]
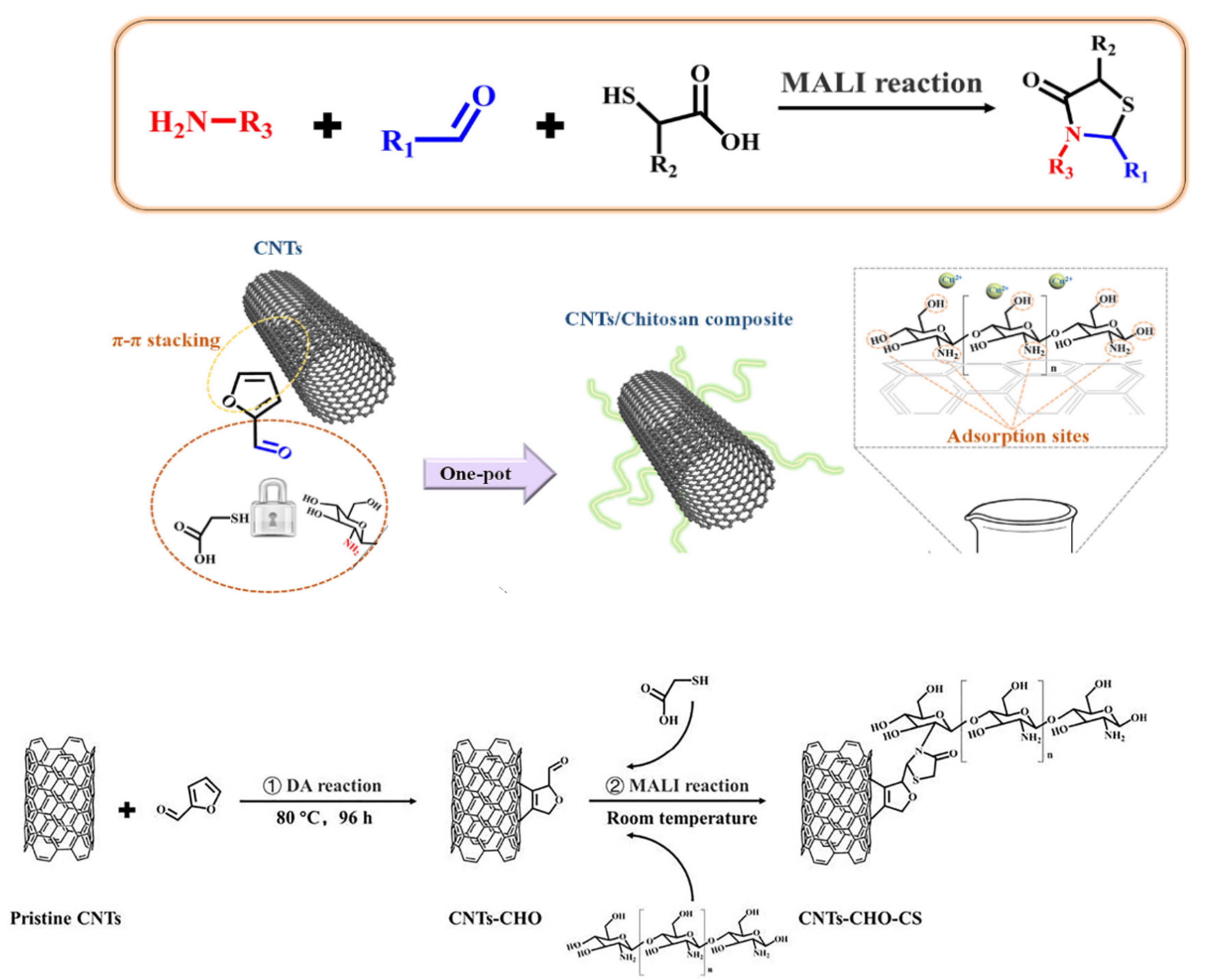
- Dou, J.; Gan, D.; Huang, Q.; Liu, M.; Chen, J.; Deng, F.; Zhu, X.; Wen, Y.; Zhang, X.; Wei, Y. Functionalization of carbon nanotubes with chitosan based on MALI multicomponent reaction for Cu2+ removal. Int. J. Biol. Macromol. 2019, 136, 476–485. [Google Scholar] [CrossRef]
- Sainz-Urruela, C.; Vera-López, S.; San Andrés, M.P.; Díez-Pascual, A.M. Graphene-Based Sensors for the Detection of Bioactive Compounds: A Review. Int. J. Mol. Sci. 2021, 22, 3316. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M. Antibacterial Action of Nanoparticle Loaded Nanocomposites Based on Graphene and Its Derivatives: A Mini-Review. Int. J. Mol. Sci. 2020, 21, 3563. [Google Scholar] [CrossRef] [PubMed]
- Innocenzi, P.; Stagi, L. Carbon-based antiviral nanomaterials: Graphene, C-dots, and fullerenes. A perspective. Chem. Sci. 2020, 11, 6606–6622. [Google Scholar] [CrossRef] [PubMed]
- Xin, Q.; Shah, H.; Nawaz, A.; Xie, W.; Akram, M.Z.; Batool, A.; Tian, L.; Jan, S.U.; Boddula, R.; Guo, B.; et al. Antibacterial carbon-based nanomaterials. Adv. Mater. 2019, 31, e1804838. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Mu, L.; Wen, J.; Zhou, Q. Covalently synthesized graphene oxide-aptamer nanosheets for efficient visible-light photocatalysis of nucleic acids and proteins of viruses. Carbon 2012, 50, 2772–2781. [Google Scholar] [CrossRef]
- Van Tam, T.; Hur, S.H.; Chung, J.S.; Choi, W.M. Novel paper- and fiber optic-based fluorescent sensor for glucose detection using aniline-functionalized graphene quantum dots. Sens. Actuators B Chem. 2021, 329, 129250. [Google Scholar] [CrossRef]
- Seifi, T.; Kamali, A.R. Antiviral performance of graphene-based materials with emphasis on COVID-19: A review. Med. Drug Discov. 2021, 11, 100099. [Google Scholar] [CrossRef]
- Bai, J.; Chen, L.; Zhu, Y.; Wang, X.; Wu, X.; Fu, Y. A novel luminescence sensor based on porous molecularly imprinted polymer-ZnS quantum dots for selective recognition of paclitaxel. Colloids Surf. A Physicochem. Eng. Asp. 2020, 610, 125696. [Google Scholar] [CrossRef]
- Soleymani, J.; Hasanzadeh, M.; Somi, M.H.; Ozkan, S.A.; Jouyban, A. Targeting and sensing of some cancer cells using folate bioreceptor functionalized nitrogen-doped graphene quantum dots. Int. J. Biol. Macromol. 2018, 118, 1021–1034. [Google Scholar] [CrossRef]
- Banerjee, A.; Pons, T.; Lequeux, N.; Dubertret, B. Quantum dots–DNA bioconjugates: Synthesis to applications. Interface Focus 2016, 6, 20160064. [Google Scholar] [CrossRef]
- Sun, D.; Gang, O. DNA-Functionalized Quantum Dots: Fabrication, Structural, and Physicochemical Properties. Langmuir 2013, 29, 7038–7046. [Google Scholar] [CrossRef]
- Wang, G.; Li, Z.; Luo, X.; Yue, R.; Shen, Y.; Ma, N. DNA-templated nanoparticle complexes for photothermal imaging and labeling of cancer cells. Nanoscale 2018, 10, 16508–16520. [Google Scholar] [CrossRef] [PubMed]
- Hajikarimi, Z.; Khoei, S.; Khoee, S.; Mahdavi, S.R. Evaluation of the cytotoxic effects of PLGA coated iron oxide nanoparticles as a carrier of 5-fluorouracil and mega-voltage X-ray radiation in DU145 prostate cancer cell line. IEEE Trans. Nanobioscience 2014, 13, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Thamake, S.I.; Raut, S.2.; Ranjan, A.P.; Gryczynski, Z.; Vishwanatha, J.K. Surface functionalization of PLGA nanoparticles by non-covalent insertion of a homo-bifunctional spacer for active targeting in cancer therapy. Nanotechnology 2010, 22, 035101. [Google Scholar] [CrossRef] [PubMed]
- Du, H.; Parit, M.; Liu, K.; Zhang, M.; Jiang, Z.; Huang, T.-S.; Zhang, X.; Si, C. Multifunctional Cellulose Nanopaper with Superior Water-Resistant, Conductive, and Antibacterial Properties Functionalized with Chitosan and Polypyrrole. ACS Appl. Mater. Interfaces 2021, 13, 32115–32125. [Google Scholar] [CrossRef] [PubMed]
- Sofla, R.L.M.; Rezaei, M.; Babaie, A. Investigation of the effect of graphene oxide functionalization on the physical, mechanical and shape memory properties of polyurethane/reduced graphene oxide nanocomposites. Diam. Relat. Mater. 2019, 95, 195–205. [Google Scholar] [CrossRef]
- Yan, S.; Wang, W.; Li, X.; Ren, J.; Yun, W.; Zhang, K.; Li, G.; Yin, J. Preparation of mussel-inspired injectable hydrogels based on dual-functionalized alginate with improved adhesive, self-healing, and mechanical properties. J. Mater. Chem. B 2018, 6, 6377–6390. [Google Scholar] [CrossRef] [PubMed]
- Saifi, M.A.; Khan, W.; Godugu, C. Cytotoxicity of Nanomaterials: Using Nanotoxicology to Address the Safety Concerns of Nanoparticles. Pharm. Nanotechnol. 2018, 6, 3–16. [Google Scholar] [CrossRef]
- Srivastava, V.; Gusain, D.; Sharma, Y.C. Critical Review on the Toxicity of Some Widely Used Engineered Nanoparticles. Ind. Eng. Chem. Res. 2015, 54, 6209–6233. [Google Scholar] [CrossRef]
- Madani, S.Y.; Mandel, A.; Seifalian, A.M. A concise review of carbon nanotube’s toxicology. Nano Rev. 2013, 4, 21521. [Google Scholar] [CrossRef]
- Yang, H.; Liu, C.; Yang, D.; Zhang, H.; Xi, Z. Comparative study of cytotoxicity, oxidative stress and genotoxicity induced by four typical nanomaterials: The role of particle size, shape and composition. J. Appl. Toxicol. 2009, 29, 69–78. [Google Scholar] [CrossRef]
- Katsumiti, A.; Berhanu, D.; Howard, K.T.; Arostegui, I.; Oron, M.; Reip, P.; Valsami-Jones, E.; Cajaraville, M. Cytotoxicity of TiO2 nanoparticles to mussel hemocytes and gill cells in vitro: Influence of synthesis method, crystalline structure, size and additive. Nanotoxicology 2015, 9, 543–553. [Google Scholar] [CrossRef] [PubMed]
- Sato, S.; Nakamura, R.; Abe, S. Visible-light sensitization of TiO2 photocatalysts by wet-method N doping. Appl. Catal. A Gen. 2005, 284, 131–137. [Google Scholar] [CrossRef]
- Sukhanova, A.; Bozrova, S.; Sokolov, P.; Berestovoy, M.; Karaulov, A.; Nabiev, I. Dependence of Nanoparticle Toxicity on Their Physical and Chemical Properties. Nanoscale Res. Lett. 2018, 13, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Yao, H.; Shi, X. Cooperative entry of nanoparticles into the cell. J. Mech. Phys. Solids 2014, 73, 151–165. [Google Scholar] [CrossRef]
- Elrahman, A.A.; Mansour, F. Targeted magnetic iron oxide nanoparticles: Preparation, functionalization and biomedical application. J. Drug Deliv. Sci. Technol. 2019, 52, 702–712. [Google Scholar] [CrossRef]
- Kheirallah, D.A.M.; El-Samad, L.M.; Abdel-Moneim, A.M. DNA damage and ovarian ultrastructural lesions induced by nickel oxide nano-particles in Blaps polycresta (Coleoptera: Tenebrionidae). Sci. Total Environ. 2021, 753, 141743. [Google Scholar] [CrossRef] [PubMed]
- Du, Y.; Jin, J.; Liang, H.; Jiang, W. Structural and physicochemical properties and biocompatibility of linear and looped polymer-capped gold nanoparticles. Langmuir 2019, 35, 8316–8324. [Google Scholar] [CrossRef]
- Sen, G.T.; Ozkemahli, G.; Shahbazi, R.; Erkekoglu, P.; Ulubayram, K.; Kocer-Gumusel, B. The effects of polymer coating of gold nanoparticles on oxidative stress and DNA damage. Int. J. Toxicol. 2020, 39, 328–340. [Google Scholar] [CrossRef]
- Wang, R.; Bowling, I.; Liu, W. Cost effective surface functionalization of gold nanoparticles with a mixed DNA and PEG monolayer for nanotechnology applications. RSC Adv. 2017, 7, 3676–3679. [Google Scholar] [CrossRef]
- Singh, A.; Amiji, M.M. Application of nanotechnology in medical diagnosis and imaging. Curr. Opin. Biotechnol. 2022, 74, 241–246. [Google Scholar] [CrossRef]
- El-Sayed, A.; Kamel, M. Advances in nanomedical applications: Diagnostic, therapeutic, immunization, and vaccine production. Environ. Sci. Pollut. Res. 2020, 27, 19200–19213. [Google Scholar] [CrossRef] [PubMed]
- Mi, Y.; Shao, Z.; Vang, J.; Kaidar-Person, O.; Wang, A.Z. Application of nanotechnology to cancer radiotherapy. Cancer Nanotechnol. 2016, 7, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Aithal, P.S. Nanotechnology Innovations & Business Opportunities: A Review. Int. J. Manag. IT Eng. 2016, 6, 182–204. [Google Scholar]
- Elkodous, M.A.; El-Sayyad, G.S.; Abdelrahman, I.Y.; El-Bastawisy, H.S.; Mohamed, A.E.; Mosallam, F.M.; Nasser, H.; Gobara, M.; Baraka, A.; Elsayed, M.; et al. Therapeutic and diagnostic potential of nanomaterials for enhanced biomedical applications. Colloids Surf. B Biointerfaces 2019, 180, 411–428. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Song, J.; Chen, X.; Yang, H. X-ray-activated nanosystems for theranostic applications. Chem. Soc. Rev. 2019, 48, 3073–3101. [Google Scholar] [CrossRef] [PubMed]
- Gharpure, K.; Wu, S.; Li, C.; Lopez-Berestein, G.; Sood, A.K. Nanotechnology: Future of oncotherapy. Clin. Cancer Res. 2015, 21, 3121–3130. [Google Scholar] [CrossRef]
- Chen, Z.; Peng, Y.; Li, Y.; Xie, X.; Wei, X.; Yang, G.; Zhang, H.; Li, N.; Li, T.; Qin, X. Aptamer-Dendrimer Functionalized Magnetic Nano-Octahedrons: Theranostic Drug/Gene Delivery Platform for Near-Infrared/Magnetic Resonance Imaging-Guided Magnetochemotherapy. ACS Nano 2021, 15, 16683–16696. [Google Scholar] [CrossRef]
- Zeytunluoglu, A.; Arslan, I. Current perspectives on nanoemulsions in targeted drug delivery: An overview. In Handbook of Research on Nanoemulsion Applications in Agriculture, Food, Health, and Biomedical Sciences; IGI Global: Hershey, PA, USA, 2022; pp. 118–140. [Google Scholar]
- Dong, Y.; Wu, X.; Chen, X.; Zhou, P.; Xu, F.; Liang, W. Nanotechnology shaping stem cell therapy: Recent advances, application, challenges, and future outlook. Biomed. Pharmacother. 2021, 137, 111236. [Google Scholar] [CrossRef]
- Pereira, M.N.; Ushirobira, C.Y.; Cunha-Filho, M.S.; Gelfuso, G.M.; Gratieri, T. Nanotechnology advances for hair loss. Ther. Deliv. 2018, 9, 593–603. [Google Scholar] [CrossRef]
- Kumar, S.; Nehra, M.; Kedia, D.; Dilbaghi, N.; Tankeshwar, K.; Kim, K.-H. Nanotechnology-based biomaterials for orthopaedic applications: Recent advances and future prospects. Mater. Sci. Eng. C 2020, 106, 110154. [Google Scholar] [CrossRef]
- Zheng, X.; Zhang, P.; Fu, Z.; Meng, S.; Dai, L.; Yang, H. Applications of nanomaterials in tissue engineering. RSC Adv. 2021, 11, 19041–19058. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Aadil, K.R.; Ranjan, S.; Kumar, V.B. Advances in nanotechnology and nanomaterials based strategies for neural tissue engineering. J. Drug Deliv. Sci. Technol. 2020, 57, 101617. [Google Scholar] [CrossRef]
- Bakopoulou, A.; Papachristou, E.; Bousnaki, M.; Hadjichristou, C.; Kontonasaki, E.; Theocharidou, A.; Papadopoulou, L.; Kantiranis, N.; Zachariadis, G.; Leyhausen, G. Human treated dentin matrices combined with Zn-doped, Mg-based bioceramic scaffolds and human dental pulp stem cells towards targeted dentin regeneration. Dent. Mater. 2016, 32, e159–e175. [Google Scholar] [CrossRef] [PubMed]
- Balakrishnan, B.; Joshi, N.; Jayakrishnan, A.; Banerjee, R. Self-crosslinked oxidized alginate/gelatin hydrogel as injectable, adhesive biomimetic scaffolds for cartilage regeneration. Acta Biomater. 2014, 10, 3650–3663. [Google Scholar] [CrossRef]
- Wang, C.; Zhang, H.; Chen, B.; Yin, H.; Wang, W. Study of the enhanced anticancer efficacy of gambogic acid on Capan-1 pancreatic cancer cells when mediated via magnetic Fe3O4 nanoparticles. Int. J. Nanomed. 2011, 6, 1929. [Google Scholar]
- Haruta, S.; Hanafusa, T.; Fukase, H.; Miyajima, H.; Oki, T. An effective absorption behavior of insulin for diabetic treatment following intranasal delivery using porous spherical calcium carbonate in monkeys and healthy human volunteers. Diabetes Technol. Ther. 2003, 5, 1–9. [Google Scholar] [CrossRef]
- Zazo, H.; Colino, C.I.; Lanao, J.M. Current applications of nanoparticles in infectious diseases. J. Control. Release 2016, 224, 86–102. [Google Scholar] [CrossRef]
- Rudramurthy, G.R.; Swamy, M.K.; Sinniah, U.R.; Ghasemzadeh, A. Nanoparticles: Alternatives against drug-resistant pathogenic microbes. Molecules 2016, 21, 836. [Google Scholar] [CrossRef]
- Smith, B.R.; Gambhir, S.S. Nanomaterials for in vivo imaging. Chem. Rev. 2017, 117, 901–986. [Google Scholar] [CrossRef]
- Imamura, Y.; Yamada, S.; Tsuboi, S.; Nakane, Y.; Tsukasaki, Y.; Komatsuzaki, A.; Jin, T. Near-infrared emitting PbS quantum dots for in vivo fluorescence imaging of the thrombotic state in septic mouse brain. Molecules 2016, 21, 1080. [Google Scholar] [CrossRef]
- Wang, Y.; Wu, H.; Lin, D.; Zhang, R.; Li, H.; Zhang, W.; Liu, W.; Huang, S.; Yao, L.; Cheng, J.; et al. One-dimensional electrospun ceramic nanomaterials and their sensing applications. J. Am. Ceram. Soc. 2022, 105, 765–785. [Google Scholar] [CrossRef]
- Liu, J.; Wang, Z.; Zhao, S.; Ding, B. Multifunctional nucleic acid nanostructures for gene therapies. Nano Res. 2018, 11, 5017–5027. [Google Scholar] [CrossRef]
- Patil-Sen, Y. Advances in nano-biomaterials and their applications in biomedicine. Emerg. Top. Life Sci. 2021, 5, 169–176. [Google Scholar] [CrossRef] [PubMed]
- Wu, G.; Li, P.; Feng, H.; Zhang, X.; Chu, P.K. Engineering and functionalization of biomaterials via surface modification. J. Mater. Chem. B 2015, 3, 2024–2042. [Google Scholar] [CrossRef] [PubMed]
- Singh, T.V.; Shagolsem, L.S. Biopolymer based nano-structured materials and their applications. In Nanostructured Materials and Their Applications; Springer: Singapore, 2021; pp. 337–366. [Google Scholar]
- Sargazi, S.; Mukhtar, M.; Rahdar, A.; Barani, M.; Pandey, S.; Díez-Pascual, A.M. Active Targeted Nanoparticles for Delivery of Poly(ADP-ribose) Polymerase (PARP) Inhibitors: A Preliminary Review. Int. J. Mol. Sci. 2021, 22, 10319. [Google Scholar] [CrossRef] [PubMed]
- Sivasankarapillai, V.S.; Das, S.S.; Sabir, F.; Sundaramahalingam, M.A.; Colmenares, J.C.; Prasannakumar, S.; Rajan, M.; Rahdar, A.; Kyzas, G.Z. Progress in natural polymer engineered biomaterials for transdermal drug delivery systems. Mater. Today Chem. 2021, 19, 100382. [Google Scholar] [CrossRef]
- Bouchoucha, M.; Gaudreault, R.C.; Fortin, M.A.; Kleitz, F. Mesoporous silica nanoparticles: Selective surface functionalization for optimal relaxometric and drug loading performances. Adv. Funct. Mater. 2014, 24, 5911–5923. [Google Scholar] [CrossRef]
- Yoo, J.; Park, C.; Yi, G.; Lee, D.; Koo, H. Active Targeting Strategies Using Biological Ligands for Nanoparticle Drug Delivery Systems. Cancers 2019, 11, 640. [Google Scholar] [CrossRef]
- Huang, Y.; Cao, L.; Parakhonskiy, B.V.; Skirtach, A.G. Hard, Soft, and Hard-and-Soft Drug Delivery Carriers Based on CaCO3 and Alginate Biomaterials: Synthesis, Properties, Pharmaceutical Applications. Pharmaceutics 2022, 14, 909. [Google Scholar] [CrossRef]
- Dizaj, S.M.; Lotfipour, F.; Barzegar-Jalali, M.; Zarrintan, M.-H.; Adibkia, K. Ciprofloxacin HCl-loaded calcium carbonate nanoparticles: Preparation, solid state characterization, and evaluation of antimicrobial effect against Staphylococcus aureus. Artif. Cells Nanomed. Biotechnol. 2016, 45, 535–543. [Google Scholar] [CrossRef]
- Ueno, Y.; Futagawa, H.; Takagi, Y.; Ueno, A.; Mizushima, Y. Drug-incorporating calcium carbonate nanoparticles for a new delivery system. J. Control. Release 2005, 103, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Hood, J.D.; Bednarski, M.; Frausto, R.; Guccione, S.; Reisfeld, R.A.; Xiang, R.; Cheresh, D.A. Tumor regression by targeted gene delivery to the neovasculature. Science 2002, 296, 2404–2407. [Google Scholar] [CrossRef]
- Deng, L.; Li, Q.; Al-Rehili, S.; Omar, H.; Almalik, A.; Alshamsan, A.; Zhang, J.; Khashab, N.M. Hybrid iron oxide–graphene oxide–polysaccharides microcapsule: A micro-matryoshka for on-demand drug release and antitumor therapy in vivo. ACS Appl. Mater. Interfaces 2016, 8, 6859–6868. [Google Scholar] [CrossRef]
- Ye, C.; Combs, Z.A.; Calabrese, R.; Dai, H.; Kaplan, D.L.; Tsukruk, V.V. Robust microcapsules with controlled permeability from silk fibroin reinforced with graphene oxide. Small 2014, 10, 5087–5097. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.-H.; Sun, X.; Chiu, J.-F.; Sun, H.; He, Q.-Y. Transferrin-mediated gold nanoparticle cellular uptake. Bioconjugate Chem. 2005, 16, 494–496. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.; Gu, Y.-J.; Cheng, S.H.; Wong, W.-T. Surface functionalized gold nanoparticles for drug delivery. J. Biomed. Nanotechnol. 2013, 9, 1362–1369. [Google Scholar] [CrossRef]
- Fang, Z.; Li, X.; Xu, Z.; Du, F.; Wang, W.; Shi, R.; Gao, D. Hyaluronic acid-modified mesoporous silica-coated superparamagnetic Fe3O4 nanoparticles for targeted drug delivery. Int. J. Nanomed. 2019, 14, 5785. [Google Scholar] [CrossRef]
- Prabha, G.; Raj, V. Sodium alginate–polyvinyl alcohol–bovin serum albumin coated Fe3O4 nanoparticles as anticancer drug delivery vehicle: Doxorubicin loading and in vitro release study and cytotoxicity to HepG2 and L02 cells. Mater. Sci. Eng. C 2017, 79, 410–422. [Google Scholar] [CrossRef]
- Rajan, M.; Raj, V.; Al-Arfaj, A.A.; Murugan, A.M. Hyaluronidase enzyme core-5-fluorouracil-loaded chitosan-PEG-gelatin polymer nanocomposites as targeted and controlled drug delivery vehicles. Int. J. Pharm. 2013, 453, 514–522. [Google Scholar] [CrossRef]
- Hua, S.; Ma, H.; Li, X.; Yang, H.; Wang, A. pH-sensitive sodium alginate/poly (vinyl alcohol) hydrogel beads prepared by combined Ca2+ crosslinking and freeze-thawing cycles for controlled release of diclofenac sodium. Int. J. Biol. Macromol. 2010, 46, 517–523. [Google Scholar] [CrossRef]
- Arshad, R.; Tabish, T.A.; Kiani, M.H.; Ibrahim, I.M.; Shahnaz, G.; Rahdar, A.; Kan, M.; Pandey, S. Hyaluronic Acid Functionalized Self-Nano-Emulsifying Drug Delivery System (SNEDDS) for Enhancement in Ciprofloxacin Targeted Delivery against Intracellular Infection. Nanomaterials 2021, 11, 1086. [Google Scholar] [CrossRef] [PubMed]
- Guo, Q.; Jia, Y.; Yuan, M.; Huang, X.; Sui, X.; Tang, F.; Peng, J.; Chen, J.; Lu, S.; Cui, X.; et al. Co-encapsulation of magnetic Fe3O4 nanoparticles and doxorubicin into biodegradable PLGA nanocarriers for intratumoral drug delivery. Int. J. Nanomed. 2012, 7, 1697–1708. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wang, B.; Xu, C.; Xie, J.; Yang, Z.; Sun, S. pH controlled release of chromone from chromone-Fe3O4 nanoparticles. J. Am. Chem. Soc. 2008, 130, 14436–14437. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Xu, C.; Kohler, N.; Hou, Y.; Sun, S. Controlled PEGylation of monodisperse Fe3O4 nanoparticles for reduced non-specific uptake by macrophage cells. Adv. Mater. 2007, 19, 3163–3166. [Google Scholar] [CrossRef]
- Saikia, C.; Hussain, A.; Ramteke, A.; Sharma, H.K.; Maji, T.K. Crosslinked thiolated starch coated Fe3O4 magnetic nanoparticles: Effect of montmorillonite and crosslinking density on drug delivery properties. Starch-Stärke 2014, 66, 760–771. [Google Scholar] [CrossRef]
- Rezaei, A.; Hashemi, E. A pseudohomogeneous nanocarrier based on carbon quantum dots decorated with arginine as an efficient gene delivery vehicle. Sci. Rep. 2021, 11, 1–10. [Google Scholar] [CrossRef]
- Di Marzio, N.; Eglin, D.; Serra, T.; Moroni, L. Bio-Fabrication: Convergence of 3D Bioprinting and Nano-Biomaterials in Tissue Engineering and Regenerative Medicine. Front. Bioeng. Biotechnol. 2020, 8, 326. [Google Scholar] [CrossRef]
- Kapat, K.; Shubhra, Q.T.H.; Zhou, M.; Leeuwenburgh, S. Piezoelectric nano-biomaterials for biomedicine and tissue regeneration. Adv. Funct. Mater. 2020, 30, 1909045. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Diez-Vicente, A.L. Epoxidized Soybean Oil/ZnO Biocomposites for Soft Tissue Applications: Preparation and Characterization. ACS Appl. Mater. Interfaces 2014, 6, 17277–17288. [Google Scholar] [CrossRef]
- Giubilato, E.; Cazzagon, V.; Amorim, M.J.B.; Blosi, M.; Bouillard, J.; Bouwmeester, H.; Costa, A.L.; Fadeel, B.; Fernandes, T.F.; Fito, C.; et al. Risk management framework for nano-biomaterials used in medical devices and advanced therapy medicinal products. Materials 2020, 13, 4532. [Google Scholar] [CrossRef]
- Rana, D.; Ramasamy, K.; Leena, M.; Jiménez, C.; Campos, J.; Ibarra, P.; Haidar, Z.S.; Ramalingam, M. Surface functionalization of nanobiomaterials for application in stem cell culture, tissue engineering, and regenerative medicine. Biotechnol. Prog. 2016, 32, 554–567. [Google Scholar] [CrossRef] [PubMed]
- Labusca, L.; Herea, D.-D.; Mashayekhi, K. Stem cells as delivery vehicles for regenerative medicine-challenges and perspectives. World J. Stem Cells 2018, 10, 43–56. [Google Scholar] [CrossRef] [PubMed]
- Lyons, J.G.; Plantz, M.A.; Hsu, W.K.; Hsu, E.L.; Minardi, S. Nanostructured biomaterials for bone regeneration. Front. Bioeng. Biotechnol. 2020, 8, 922. [Google Scholar] [CrossRef]
- Zhu, L.; Luo, D.; Liu, Y. Effect of the nano/microscale structure of biomaterial scaffolds on bone regeneration. Int. J. Oral Sci. 2020, 12, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Venugopal, J.R.; El-Turki, A.; Ramakrishna, S.; Su, B.; Lim, C.T. Electrospun biomimetic nanocomposite nanofiber of hydroxiapatite/chitosan for bone tissue engineering. Biomaterials 2008, 29, 4314–4322. [Google Scholar] [CrossRef] [PubMed]
- Liang, X.Y.; Duan, P.G.; Gao, J.M.; Guo, R.S.; Qu, Z.H.; Li, X.F.; He, Y.; Yao, H.Q.; Ding, J.D. Bilayered PLGA/PLGA-HAp Composite Scaffold for Osteochondral Tissue Engineering and Tissue Regeneration. ACS Biomater. Sci. Eng. 2018, 4, 3506–3521. [Google Scholar] [CrossRef]
- Naffakh, M.; Diez-Pascual, A.M. WS2 inorganic nanotubes reinforced poly(L-lacticacid)/hydroxyapatite hybrid composite biomaterials. RSC Adv. 2015, 5, 65514. [Google Scholar] [CrossRef]
- Cui, Y.; Li, H.; Li, Y.; Mao, L. Novel insights into nanomaterials for immunomodulatory bone regeneration. Nanoscale Adv. 2022, 4, 334–352. [Google Scholar] [CrossRef]
- McMillan, A.; Nguyen, M.K.; Gonzalez-Fernandez, T.; Ge, P.; Yu, X.; Murphy, W.L.; Kelly, D.; Alsberg, E. Dual non-viral gene delivery from microparticles within 3D high-density stem cell constructs for enhanced bone tissue engineering. Biomaterials 2018, 161, 240–255. [Google Scholar] [CrossRef]
- Perez, J.R.; Kouroupis, D.; Li, D.J.; Best, T.M.; Kaplan, L.; Correa, D. Tissue engineering and cell-based therapies for fractures and bone defects. Front. Bioeng. Biotechnol. 2018, 6, 105. [Google Scholar] [CrossRef]
- Veatch, J.R.; Singhi, N.; Srivastava, S.; Szeto, J.L.; Jesernig, B.; Stull, S.M.; Fitzgibbon, M.; Sarvothama, M.; Yechan-Gunja, S.; James, S.E.; et al. A therapeutic cancer vaccine delivers antigens and adjuvants to lymphoid tissues using genetically modified T cells. J. Clin. Investig. 2021, 131, e144195. [Google Scholar] [CrossRef] [PubMed]
- Acri, T.M.; Laird, N.Z.; Jaidev, L.R.; Meyerholz, D.K.; Salem, A.K.; Shin, K. Nonviral Gene Delivery Embedded in Biomimetically Mineralized Matrices for Bone Tissue Engineering. Tissue Eng. Part A 2021, 27, 1074–1083. [Google Scholar] [CrossRef] [PubMed]
- Ghandforoushan, P.; Hanaee, J.; Aghazadeh, Z.; Samiei, M.; Navali, A.M.; Khatibi, A.; Davaran, S. Novel nanocomposite scaffold based on gelatin/PLGA-PEG-PLGA hydrogels embedded with TGF-β1 for chondrogenic differentiation of human dental pulp stem cells in vitro. Int. J. Biol. Macromol. 2022, 201, 270–287. [Google Scholar] [CrossRef] [PubMed]
- Sirkkunan, D.; Pingguan-Murphy, B.; Muhamad, F. Directing Axonal Growth: A Review on the Fabrication of Fibrous Scaffolds That Promotes the Orientation of Axons. Gels 2022, 8, 25. [Google Scholar] [CrossRef]
- Abdal-Hay, A.; Sheikh, F.A.; Gómez-Cerezo, N.; Alneairi, A.; Luqman, M.; Pant, H.R.; Ivanovski, S. A review of protein adsorption and bioactivity characteristics of poly ε-caprolactone scaffolds in regenerative medicine. Eur. Polym. J. 2022, 162, 110892. [Google Scholar] [CrossRef]
- Joy, J.; Pereira, J.; Aid-Launais, R.; Pavon-Djavid, G.; Ray, A.R.; Letourneur, D.; Meddahi-Pellé, A.; Gupta, B. Gelatin—Oxidized carboxymethyl cellulose blend based tubular electrospun scaffold for vascular tissue engineering. Int. J. Biol. Macromol. 2018, 107, 1922–1935. [Google Scholar] [CrossRef]
- Murugesan, B.; Pandiyan, N.; Arumugam, M.; Sonamuthu, J.; Samayanan, S.; Yurong, C.; Juming, Y.; Mahalingam, S. Fabrication of palladium nanoparticles anchored polypyrrole functionalized reduced graphene oxide nanocomposite for antibiofilm associated orthopedic tissue engineering. Appl. Surf. Sci. 2020, 510, 145403. [Google Scholar] [CrossRef]
- Jie, W.; Song, F.; Li, X.; Li, W.; Wang, R.; Jiang, Y.; Zhao, L.; Fan, Z.; Wang, J.; Liu, B. Enhancing the proliferation of MC3T3-E1 cells on casein phosphopeptide-biofunctionalized 3D reduced-graphene oxide/polypyrrole scaffolds. RSC Adv. 2017, 7, 34415–34424. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M.; Díez-Vicente, A.L. Wound Healing Bionanocomposites Based on Castor Oil Polymeric Films Reinforced with Chitosan-Modified ZnO Nanoparticles. Biomacromolecules 2015, 16, 2631–2644. [Google Scholar] [CrossRef]
- Lee, M.-H.; You, C.; Kim, K.-H. Combined effect of a microporous layer and type I collagen coating on a biphasic calcium phosphate scaffold for bone tissue engineering. Materials 2015, 8, 1150–1161. [Google Scholar] [CrossRef]
- Siddiqi, N.J.; Abdelhalim, M.A.K.; El-Ansary, A.K.; Alhomida, A.S.; Ong, W.Y. Identification of potential biomarkers of gold nanoparticle toxicity in rat brains. J. Neuroinflammation 2012, 9, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Abdelhalim, M.A.K. Exposure to gold nanoparticles produces cardiac tissue damage that depends on the size and duration of exposure. Lipids Health Dis. 2011, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Love, S.A.; Thompson, J.W.; Haynes, C.L. Development of screening assays for nanoparticle toxicity assessment in human blood: Preliminary studies with charged Au nanoparticles. Nanomedicine 2012, 7, 1355–1364. [Google Scholar] [CrossRef] [PubMed]
- Freese, C.; Gibson, M.I.; Klok, H.A.; Unger, R.E.; Kirkpatrick, C.J. Size-and coating-dependent uptake of polymer-coated gold nanoparticles in primary human dermal microvascular endothelial cells. Biomacromolecules 2012, 13, 1533–1543. [Google Scholar] [CrossRef] [PubMed]
- Fu, Q.; Rahaman, M.N.; Bal, B.S.; Brown, R.F.; Day, D.E. Mechanical and in vitro performance of bioactive glass scaffolds prepared by a polymer foam replication technique. Acta Biomater. 2008, 4, 1854–1864. [Google Scholar] [CrossRef]
- Chen, Q.Z.; Thompson, I.D.; Boccaccini, A.R. 45S5 Bioglass®-derived glass–ceramic scaffolds for bone tissue engineering. Biomaterials 2006, 27, 2414–2425. [Google Scholar] [CrossRef]
- Jiang, L.; Chen, D.; Wang, Z.; Zhang, Z.; Xia, Y.; Xue, H.; Liu, Y. Preparation of an electrically conductive graphene oxide/chitosan scaffold for cardiac tissue engineering. Appl. Biochem. Biotechnol. 2019, 188, 952–964. [Google Scholar] [CrossRef]
- Shamekhi, M.A.; Mirzadeh, H.; Mahdavi, H.; Rabiee, A.; Mohebbi-Kalhori, D.; Eslaminejad, M.B. Graphene oxide containing chitosan scaffolds for cartilage tissue engineering. Int. J. Biol. Macromol. 2019, 127, 396–405. [Google Scholar] [CrossRef]
- Nishida, E.; Miyaji, H.; Takita, H.; Kanayama, I.; Tsuji, M.; Akasaka, T.; Sugaya, T.; Sakagami, R.; Kawanami, M. Graphene oxide coating facilitates the bioactivity of scaffold material for tissue engineering. Jpn. J. Appl. Phys. 2014, 53, 06JD04. [Google Scholar] [CrossRef]
- Arshad, R.; Fatima, I.; Sargazi, S.; Rahdar, A.; Karamzadeh-Jahromi, M.; Pandey, S.; Díez-Pascual, A.M.; Bilal, M. Novel Perspectives towards RNA-Based Nano-Theranostic Approaches for Cancer Management. Nanomaterials 2021, 11, 3330. [Google Scholar] [CrossRef]
- Feng, X.; Jiang, D.; Kang, T.; Yao, J.; Jing, Y.; Jiang, T.; Feng, J.; Zhu, Q.; Song, Q.; Dong, N.; et al. Tumor-homing and penetrating peptide-functionalized photosensitizer-conjugated PEG-PLA nanoparticles for chemo-photodynamic combination therapy of drug-resistant cancer. ACS Appl. Mater. Interfaces 2016, 8, 17817–17832. [Google Scholar] [CrossRef] [PubMed]
- Revia, R.A.; Zhang, M. Magnetite nanoparticles for cancer diagnosis, treatment, and treatment monitoring: Recent advances. Mater. Today 2016, 19, 157–168. [Google Scholar] [CrossRef] [PubMed]
- Naidoo, C.; Kruger, C.A.; Abrahamse, H. Photodynamic therapy for metastatic melanoma treatment: A review. Technol. Cancer Res. Treat. 2018, 17. [Google Scholar] [CrossRef] [PubMed]
- Heo, D.N.; Yang, D.H.; Moon, H.-J.; Lee, J.B.; Bae, M.S.; Lee, S.C.; Lee, W.J.; Sun, I.-C.; Kwon, I.K. Gold nanoparticles surface-functionalized with paclitaxel drug and biotin receptor as theranostic agents for cancer therapy. Biomaterials 2012, 33, 856–866. [Google Scholar] [CrossRef]
- Dilnawaz, F.; Singh, A.; Mohanty, C.; Sahoo, S.K. Dual drug loaded superparamagnetic iron oxide nanoparticles for targeted cancer therapy. Biomaterials 2010, 31, 3694–3706. [Google Scholar] [CrossRef]
- Dash, B.; Jose, G.; Lu, Y.-J.; Chen, J.-P. Functionalized reduced graphene oxide as a versatile tool for cancer therapy. Int. J. Mol. Sci. 2021, 22, 2989. [Google Scholar] [CrossRef]
- Shen, J.M.; Gao, F.Y.; Yin, T.; Zhang, H.X.; Ma, M.; Yang, Y.J.; Yue, F. cRGD-functionalized polymeric magnetic nanoparticles as a dual-drug delivery system for safe targeted cancer therapy. Pharmacol. Res. 2013, 70, 102–115. [Google Scholar] [CrossRef]
- Zhang, Q.; Neoh, K.G.; Xu, L.; Lu, S.; Kang, E.T.; Mahendran, R.; Chiong, E. Functionalized mesoporous silica nanoparticles with mucoadhesive and sustained drug release properties for potential bladder cancer therapy. Langmuir 2014, 30, 6151–6161. [Google Scholar] [CrossRef]
- Kim, S.H.; Lee, J.E.; Sharker, S.M.; Jeong, J.H.; In, I.; Park, S.Y. In vitro and in vivo tumor targeted photothermal cancer therapy using functionalized graphene nanoparticles. Biomacromolecules 2015, 16, 3519–3529. [Google Scholar] [CrossRef]
- Wu, Y.-F.; Wu, H.-C.; Kuan, C.-H.; Lin, C.-J.; Wang, L.-W.; Chang, C.-W.; Wang, T.-W. Multi-functionalized carbon dots as theranostic nanoagent for gene delivery in lung cancer therapy. Sci. Rep. 2016, 6, 1–12. [Google Scholar] [CrossRef]
- Xia, Y.; Chen, Y.; Hua, L.; Zhao, M.; Xu, T.; Wang, C.; Li, Y.; Zhu, B. Functionalized selenium nanoparticles for targeted delivery of doxorubicin to improve non-small-cell lung cancer therapy. Int. J. Nanomed. 2018, 13, 6929–6939. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Wu, J.; Williams, G.R.; Niu, S.; Qian, Q.; Zhu, L.-M. Functionalized MoS2-nanosheets for targeted drug delivery and chemo-photothermal therapy. Colloids Surf. B Biointerfaces 2019, 173, 101–108. [Google Scholar] [CrossRef] [PubMed]
- Ammarullah, M.I.; Afif, I.Y.; Maula, M.I.; Winarni, T.I.; Tauviqirrahman, M.; Akbar, I.; Basri, H.; Van der Heide, E.; Jamari, J. Tresca Stress Simulation of Metal-on-Metal Total Hip Arthroplasty during Normal Walking Activity. Materials 2021, 14, 7554. [Google Scholar] [CrossRef] [PubMed]
- Basri, H.; Syahrom, A.; Prakoso, A.T.; Wicaksono, D.; Amarullah, M.I.; Ramadhoni, T.S.; Nugraha, R.D. The Analysis of Dimple Geometry on Artificial Hip Joint to the Performance of Lubrication. J. Phys. Conf. Ser. 2019, 1198, 042012. [Google Scholar] [CrossRef]
- Webster, T.J.; Siegel, R.W.; Bizios, R. Osteoblast adhesion on nanophase ceramics. Biomaterials 1999, 20, 1221–1227. [Google Scholar] [CrossRef]
- Tran, P.A.; Sarin, L.; Hurt, R.H.; Webster, T.J. Opportunities for nanotechnologyenabled bioactive bone implants. J. Mater. Chem. 2009, 19, 2653–2659. [Google Scholar] [CrossRef]
- Zinger, O.; Anselme, K.; Denzer, A.; Habersetzer, P.; Wieland, M.; Jeanfils, J.; Hardouin, P.; Landolt, D. Time-dependent morphology and adhesion of osteoblastic cells on titanium model surfaces featuring scale-resolved topography. Biomaterials 2004, 25, 2695–2711. [Google Scholar] [CrossRef]
- Chu, P.K.; Chen, J.Y.; Wang, L.P.; Huang, N. Plasma-surface modification of biomaterials. Mater. Sci. Eng. R Rep. 2002, 36, 143–206. [Google Scholar] [CrossRef]
- Yu, G.; Hu, L.; Vosgueritchian, M.; Wang, H.; Xie, X.; McDonough, J.R.; Cui, X.; Cui, Y.; Bao, Z. Solution-processed graphene/MnO2 nanostructured textiles for high-performance electrochemical capacitors. Nano Lett. 2011, 11, 2905–2911. [Google Scholar] [CrossRef]
- Rojaee, R.; Fathi, M.; Raeissi, K. Electrophoretic deposition of nanostructured hydroxyapatite coating on AZ91 magnesium alloy implants with different surface treatments. Appl. Surf. Sci. 2013, 285, 664–673. [Google Scholar] [CrossRef]
- Vorobyev, A.Y.; Guo, C. Direct femtosecond laser surface nano/microstructuring and its applications. Laser Photonics Rev. 2013, 7, 385–407. [Google Scholar] [CrossRef]
- Bolelli, G.; Bellucci, D.; Cannillo, V.; Lusvarghi, L.; Sola, A.; Stiegler, N.; Müller, P.; Killinger, A.; Gadow, R.; Altomare, L.; et al. Suspension thermal spraying of hydroxyapatite: Microstructure and in vitro behaviour. Mater. Sci. Eng. C 2014, 34, 287–303. [Google Scholar] [CrossRef] [PubMed]
- Sima, F.; Davidson, P.M.; Dentzer, J.; Gadiou, R.; Pauthe, E.; Gallet, O.; Mihailescu, I.N.; Anselme, K. Inorganic-organic thin implant coatings deposited by lasers. ACS Appl. Mater. Interfaces 2014, 7, 911–920. [Google Scholar] [CrossRef] [PubMed]
- McEntire, B.; Bal, B.S.; Rahaman, M.; Chevalier, J.; Pezzotti, G. Ceramics and ceramic coatings in orthopaedics. J. Eur. Ceram. Soc. 2015, 35, 4327–4369. [Google Scholar] [CrossRef]




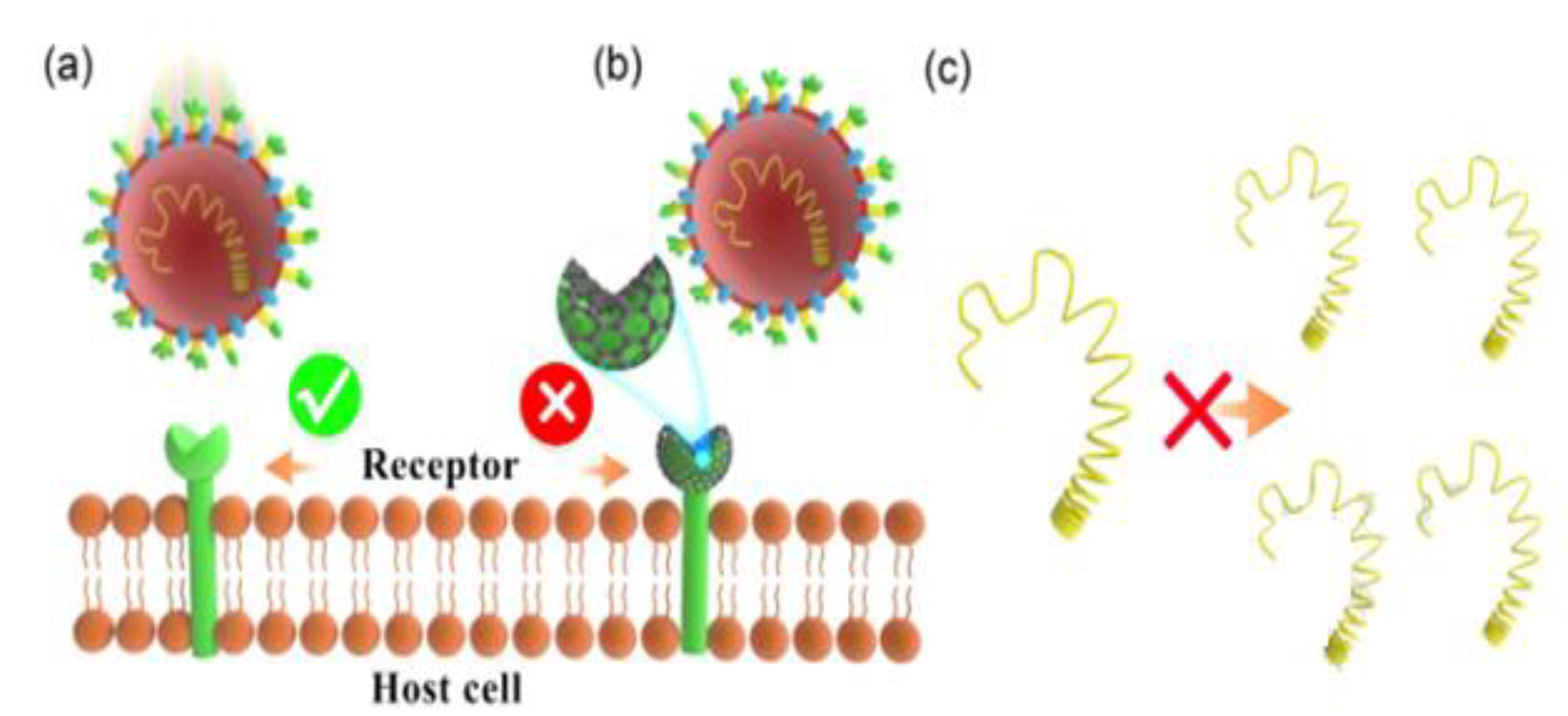
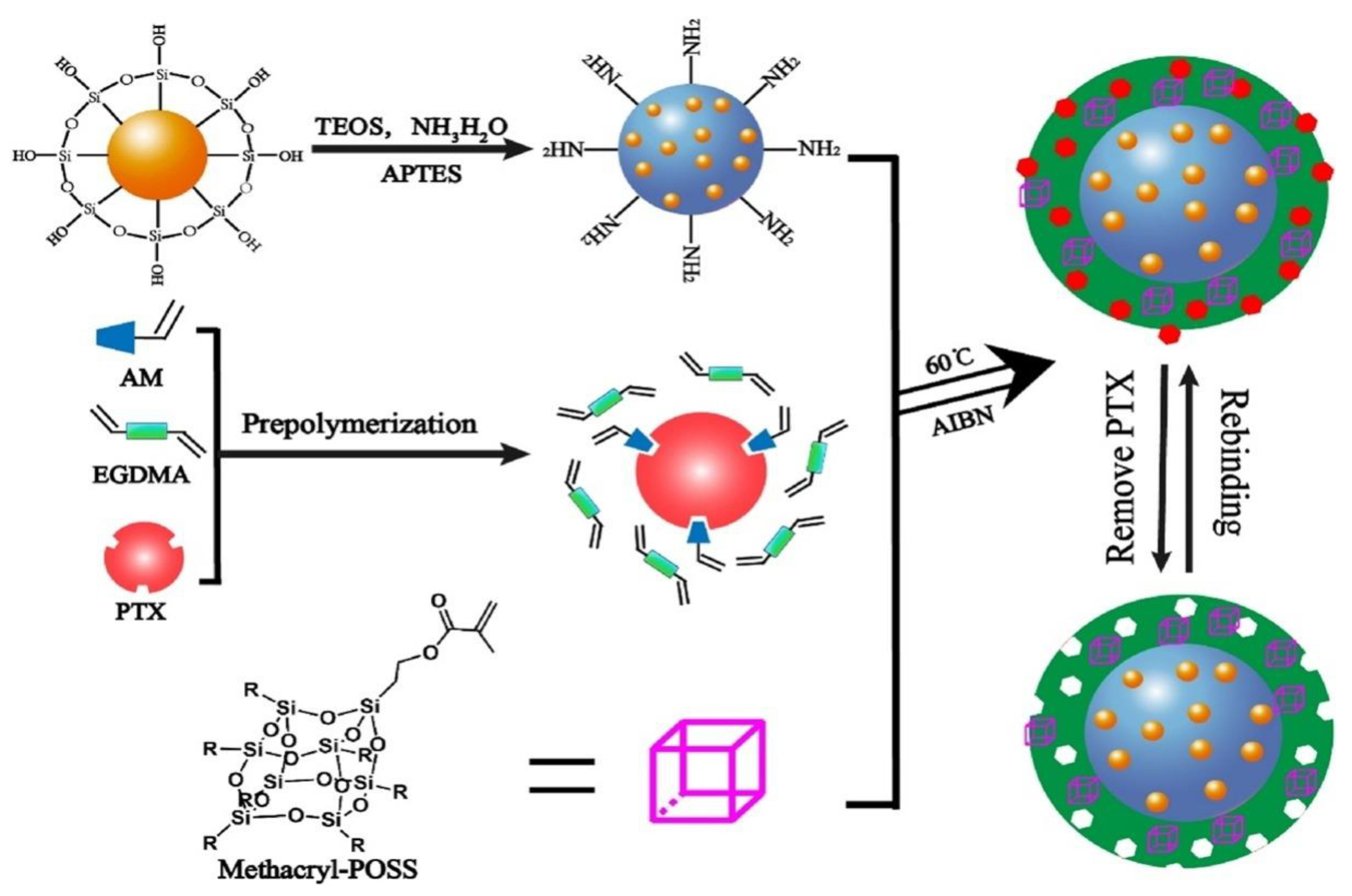

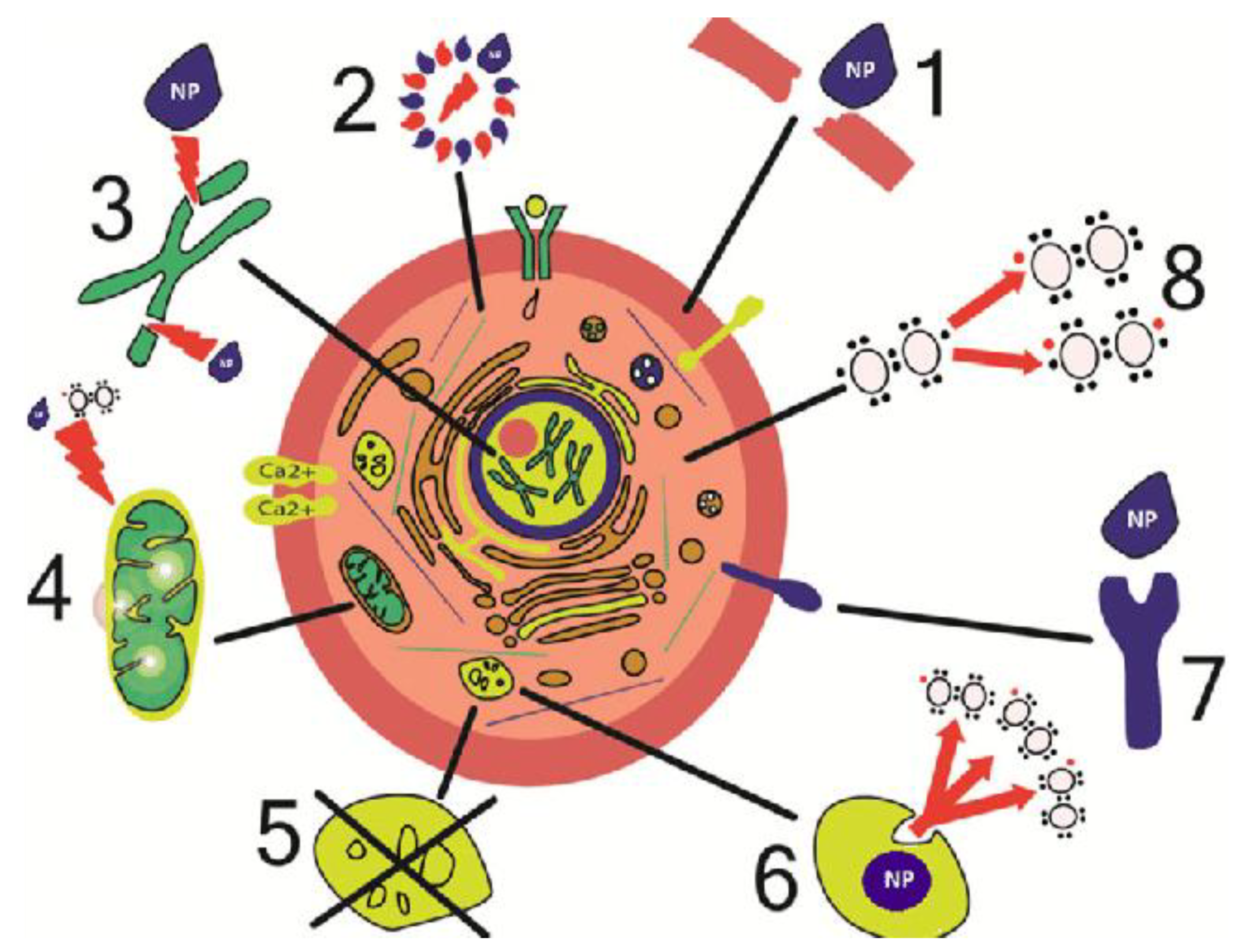

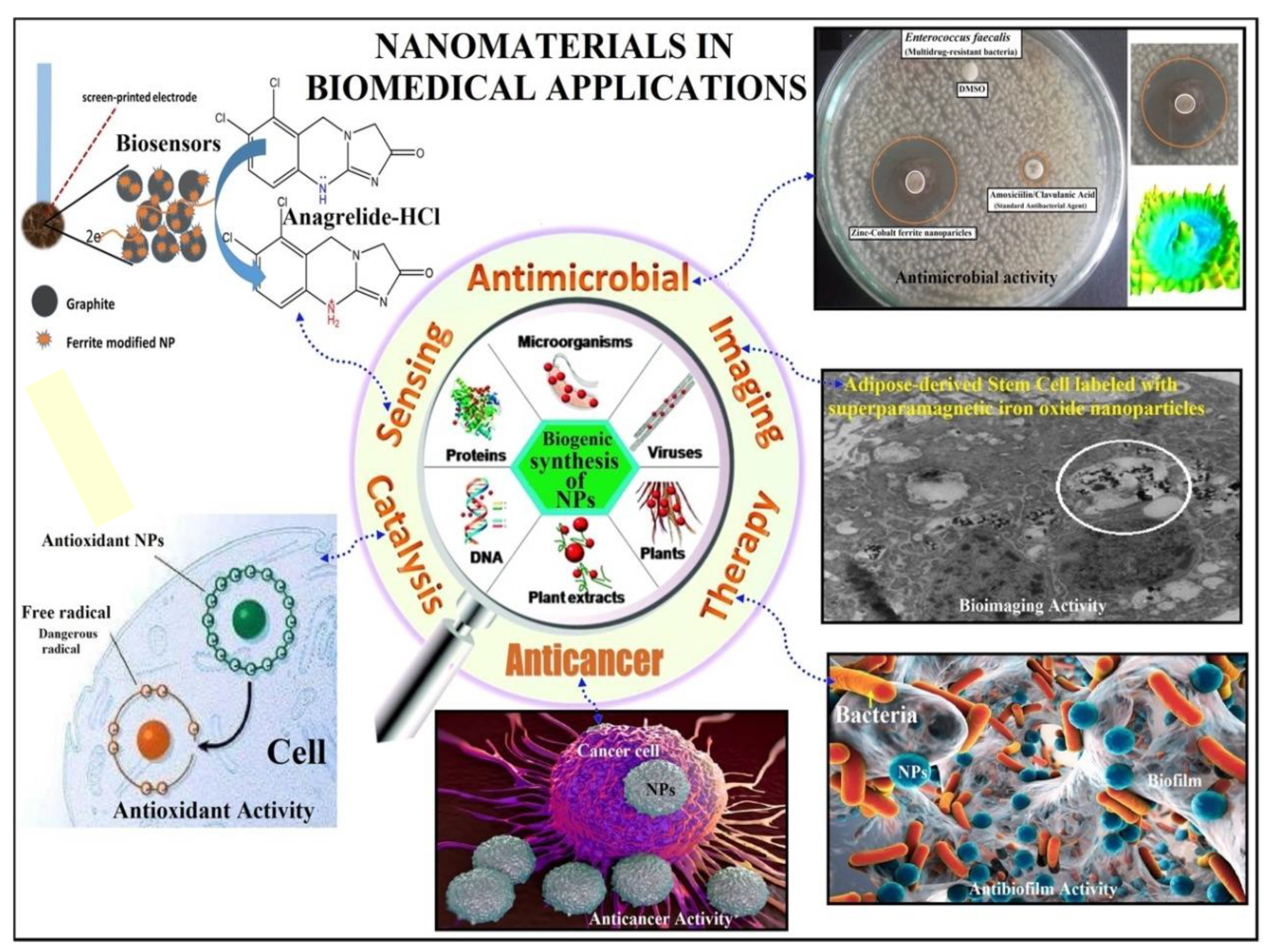
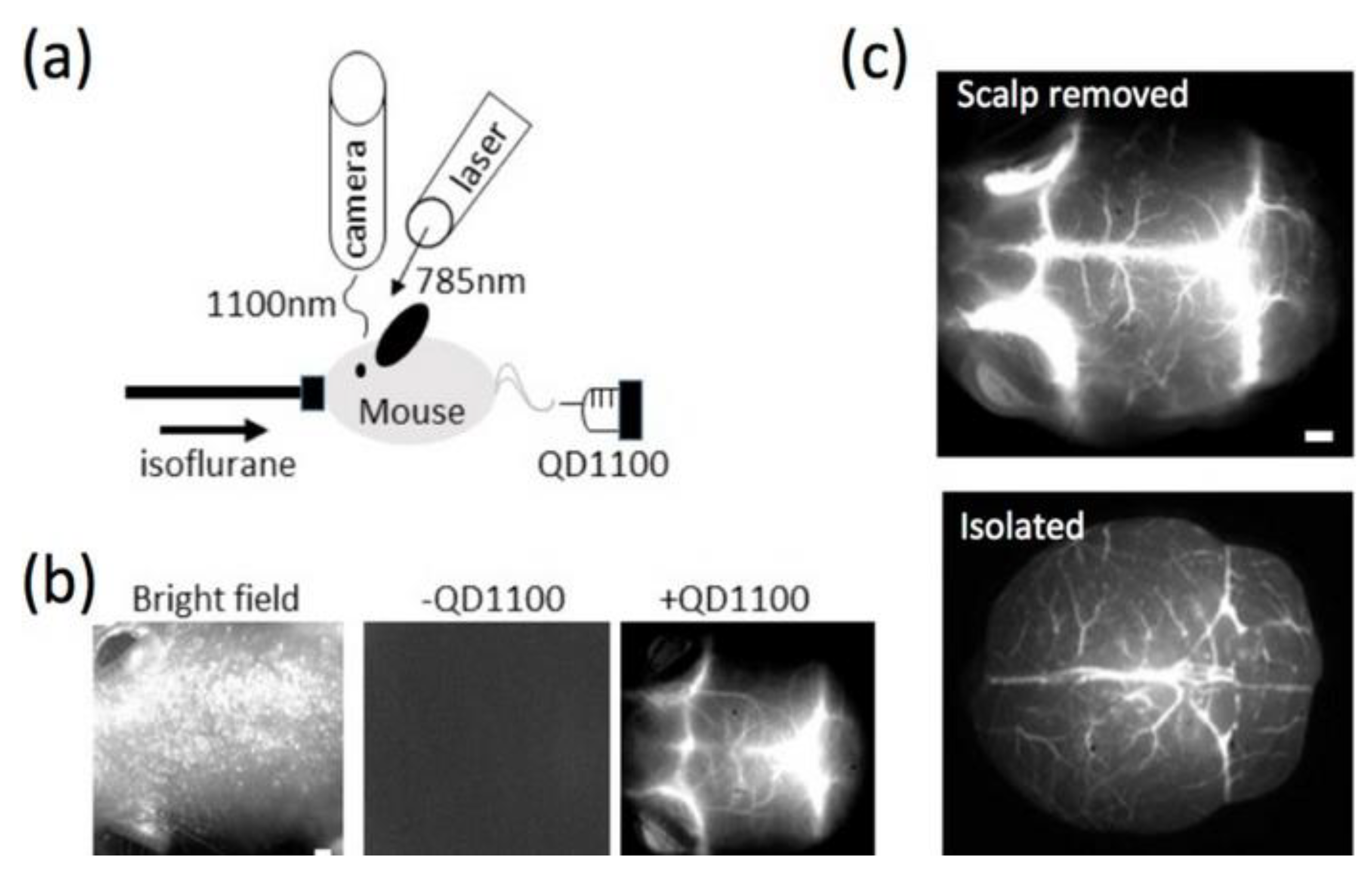

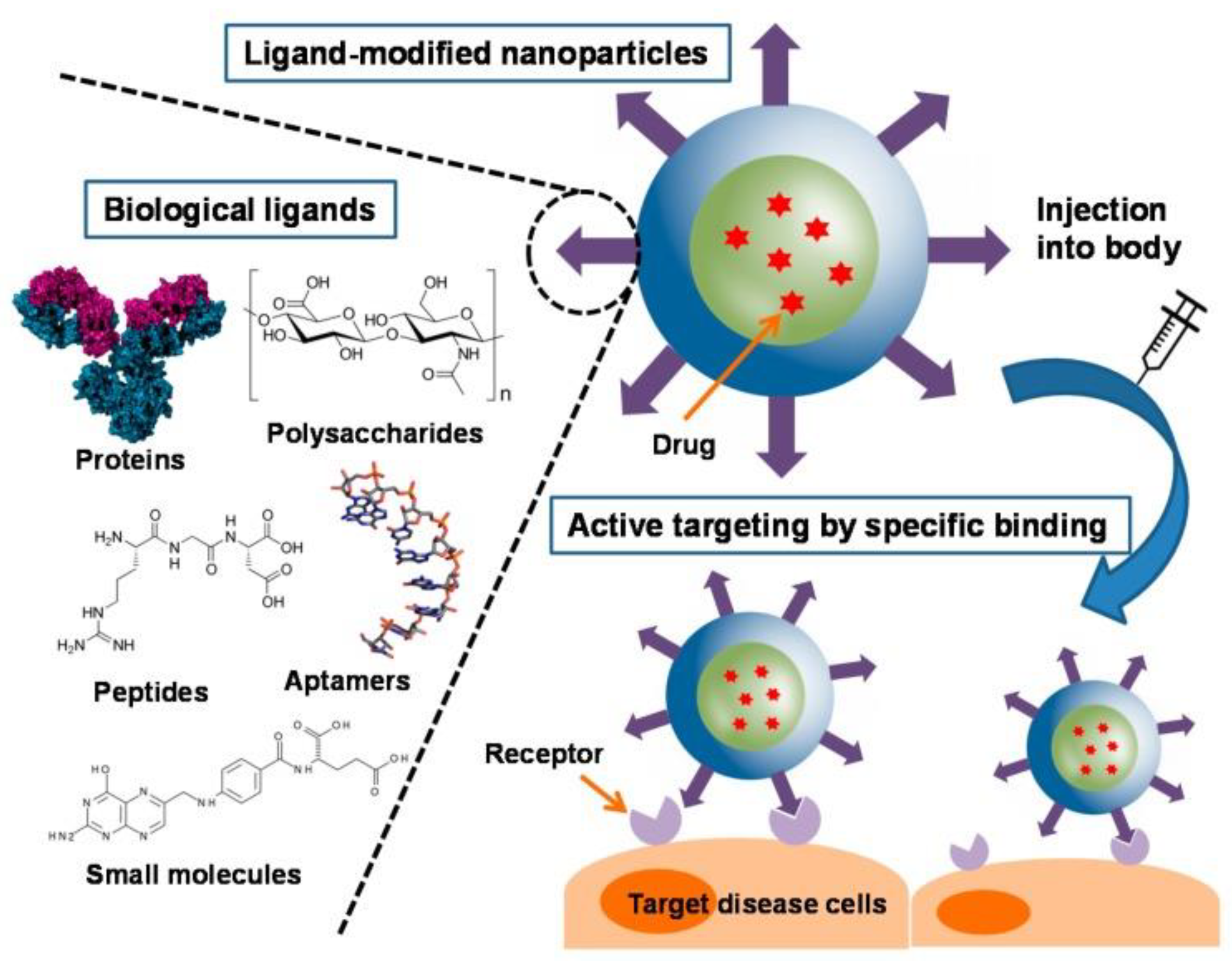
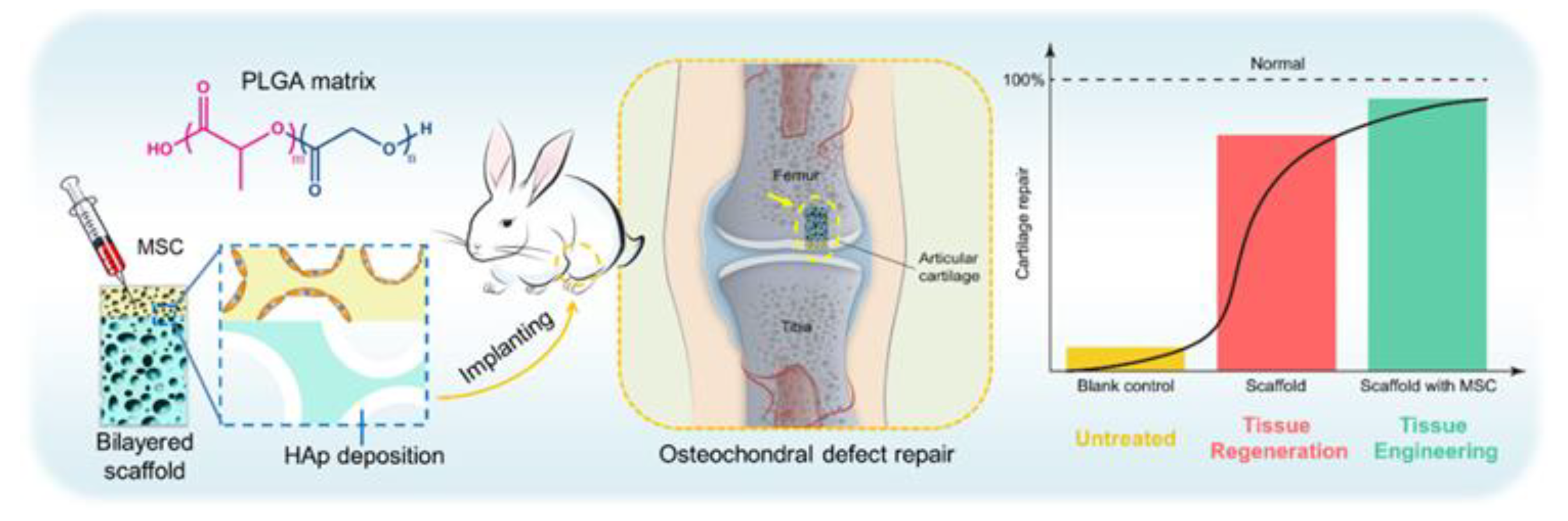
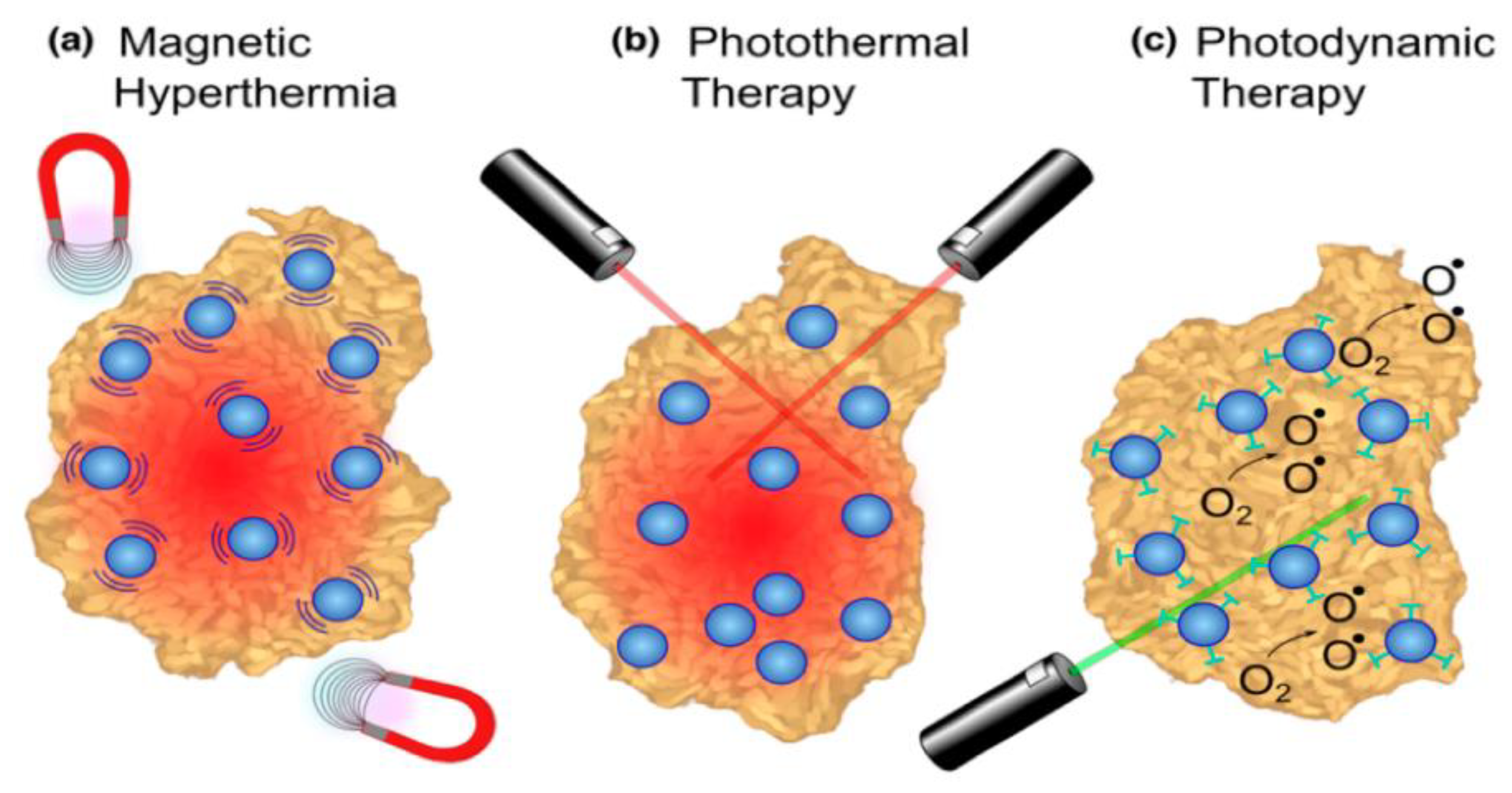
| Application | Example | Ref. |
|---|---|---|
| Diagnostic Imaging | X Ray Tomography Magnetic resonance imaging Photothermal imaging | [136] [137] [138] [112] |
| Therapy | Drug delivery Gene and stem cell therapy Hair growth | [139] [140] [141] |
| Medical implants | Orthopaedic Cardiovascular Neurological Dental | [142] [143] [144] [145] |
| Tissue Engineering | Bone Cartilage | [54] [146] |
| Anticancer | Paclitaxel DOX Docetaxel Gambogic acid | [108] [60] [30] [147] |
| Sensing | Glucose Insulin Metabolic biomarkers | [106] [148] [38] |
| Antimicrobial and Antiviral | Streptomycin, penicillin Coronavirus E. coli Airborne viruses | [149] [107] [104] [150] |
| Nanomaterial | Function | Size (nm) | Drug/Gene | Target Organ & Indication | Ref. |
|---|---|---|---|---|---|
| Porous CaCO3 | Intranasal drug carrier | 2000–3200 | Insulin | Postprandial hyperglycemia in diabetes | [162] |
| CaCO3 NPs | Drug/gene delivery | 116 | Ciprofloxacin HCl | S. Aureus | [163] |
| CaCO3 | Drug delivery | 40–200 | Hydrophilic drugs and bioactive proteins (validamycin) | Inflamed region | [164] |
| Cationic NPs | Gene delivery | 50–100 | Raf gene, ATPμ-Raf | Angiogenic blood vessels (tumor-bearing mice) | [165] |
| Fe3O4@GO | Drug release and antitumor therapy | 200–1000 | Hybrid microcapsule | Tumor cells targeting | [166] |
| GO flakes | Drug release | 1000–2000 | DOX microcapsules | - | [167] |
| AuNPs | Drug delivery | 100 | -- | Nasopharyngeal carcinoma cells | [168] |
| FA-Au-FITC 1 | Drug delivery for cancer therapy | 4–7 | DOX | Cytoplasm | [169] |
| HLA 2-Si/Fe3O4 NPs | Drug delivery for cancer therapy | 40–110 | DOX | Tumor tissues | [170] |
| Fe3O4-SA-PVA-BSA 3 | Drug delivery | 240–460 | DOX | Cancer cells | [171] |
| CS-HYL-5-FU-PEG-G 4 | Drug delivery | 300–580 | COLO-205 and HT-29 colon | Cancer cells | [172] |
| SA/PVA/Ca 5 | Drug delivery system | 500–1000 | Diclofenac sodium | - | [173] |
| PLGA 6-Fe3O4 | Drug delivery system | 67 | 5-Fluorouracil | Prostate carcinoma cell | [113] |
| HLA-Nanoemulsion | Drug delivery system | – | Ciprofloxacin | - | [174] |
| Fe3O4 | Drug delivery system | 20 | Gambogic acid | Capan-1 pancreatic cancer cells | [147] |
| PLGA-Fe3O4 NPs | Intratumoral drug delivery | 200–300 | DOX | Murine Lewis lung carcinoma cells | [175] |
| Fe3O4 conjugate oleate/oleylamine | Drug release | 12 | Chromone | HeLa cells | [176] |
| Fe3O4/DPA-PEG-COOH 7 | Drug delivery | 9 | Dextran, PEG | Macrophage Cells | [177] |
| Thiolated starch-coated Fe3O4 | Drug delivery | 40–50 | Isoniazid | Human body cells | [178] |
| Zn-doped Fe3O4 nano-octahedral core | Drug delivery | 10–20 | DOX and HSP70/HSP90 siRNAs | Tumor cells | [138] |
| Arginine-NCQDs 8 | Gene delivery | 6–11 | EGFP gene | Mammalian cells | [179] |
| Nanomaterial | Function | Size (nm) | Tissue | Purpose & Outcomes | Ref. |
|---|---|---|---|---|---|
| PEG-GO | Tissue engineering | 50 | Bone | Improved thermal stability, hydrophilicity, water absorption, biodegradation, mechanical, viscoelastic, and antibacterial properties | [54] |
| Oxidized alginate/ gelatin hydrogel | Tissue regeneration | 100–200 | Cartilage regeneration for the treatment of osteoarthritis | Usefulness of the hydrogel in encouraging cellular migration and proliferation | [146] |
| OCMC 1 | Tissue engineering | 2000–4000 | BALB/c3T3 cells in rates | Biocompatibility, spinnability of hydrogel through electrospinning | [199] |
| Pd/PPy/rGO NC 2 | Tissue engineering | 2–4 | Bone | Biocompatibility, osteoproliferation, and bacterial infection prevention | [200] |
| 3D macro-rGO/PPY | Bone tissue engineering | 100–400 | Backbone | Casein phosphopeptide as bioactive for bone engineering, osteoblastic performance, biological properties | [201] |
| Chitosan-ZnO | Soft tissue engineering | 180 | Improved hydrophilicity, porosity, water absorption, oxygen permeability, biodegradability, antibacterial and wound healing | [202] | |
| Biphasic Calcium Phosphate | Bone tissue engineering | 1–2 | MG63 cells | Micropores and collagen coating influence cellular function, in vitro cellular behavior, scaffold–osteoblast interactions | [203] |
| AuNPs/glass-ceramic matrix | Bone tissue engineering | 5–10 | Bone | In vitro hydroxyapatite synthesis, controlled release of gold species, biocompatibility, and antibacterial activity of AuNPs | [91] |
| AuNPs | Tissue engineering | 20 | Rat brain | AuNP biochemical effects on the rat brain, biomarkers of AuNP toxicity | [204] |
| AuNPs | Tissue Engineering | 10–50 | Cardiac tissue | Effects of AuNPs on the histological deformities of rat heart tissue, toxicity, therapeutic and diagnostic potential of NPs, and their interaction with proteins and other cells | [205] |
| AuNPs | Tissue engineering | 30 nm | Subsets of cells in human organs | NP toxicity in human blood, hemolysis, development of ROS 3, platelet condensation in cell subsets | [206] |
| AuNPs/polymeric coatings | Tissue engineering | 18, 35, 65 | Endothelial cells from human dermis | NP toxicity, uptake behavior, and uptake quantification | [207] |
| Bioactive glass scaffolds | Tissue engineering | 50–100 | Bone repair | Osteoblastic cells for bone reconstruction | [208] |
| Na2Ca2Si3O9 | Bone tissue engineering | 500 | Bone | Bioactive and biodegradable scaffold effects, mechanical support | [209] |
| Bioactive glass- ceramics/apatite | Bone tissue engineering | 8–20 | Bone | Crystallization rate of bioactive glasses on the kinetics of HAD formation | [90] |
| Ca10(PO4)6(OH)2 | Bone tissue engineering | 1000–2000 | Trabecular bone | Extent and nature of carbonate substitution on HDA | [93] |
| GO/Chitosan Scaffold | Cardiac tissue | –– | Cardiac tissue | Investigate cell survival, cell adhesion, development of intercellular networks, genes, and proteins expression | [210] |
| GO/Chitosan Scaffold | Cartilage repair | 35–60 | Cartilage tissue | Nanocomposite effect on human tissue, effects of GO | [211] |
| GO-coated collagen scaffolds | Tissue engineering | –– | Mouse osteoblastic MC3T3-E1 cells | Influence of the GO coating on cell growth and differentiation, biocompatibility and biodegradability of collagen scaffolds, bioactivity studies | [212] |
| Nanocrystalline apatite/AuNPs | Tissue engineering | 2–25 | Bone tissue reconstruction | Toxicity of NPS in simulated physiological fluid | [66] |
| Nanomaterial | Functionalization Agent | Size (nm) | Drug | Purpose & Outcomes | Ref. |
|---|---|---|---|---|---|
| ZnO NPs | PBA | 40 | Curcumin | High drug-loading and release rates, in vitro and in vivo antitumor efficacy | [88] |
| AuNPs | Beta-cyclodextrin with PEG, biotin, PTX, rhodamine B | 30–50 | PTX | Cytocompatibility, stability, and biomolecule binding ease | [217] |
| SPION | 5TR1 Aptamer | 57 | Epirubicin | Magnetic resonance (MR) traceability, nontoxicity, increased permeability, retention effect | [82] |
| Fe3O4 NPS | Glycerol monooleate | 144 | PTX, rapamycin, alone or combined | Intravenous administration of hydrophobic drugs | [218] |
| rGO 1 | Fe3O4 NPs | 54.8 | Camptothecin | pH-responsive drug release profile, good biocompatibility, excellent photodynamic | [219] |
| Fe3O4 MNPs+ PLGA | citric acid | 130–140 | DOX, verapamil | Loading hydrophilic and hydrophobic drugs | [220] |
| MSN 2 | β-cyclodextrin with hydroxyl, amino, and thiol groups | 75.5 | DOX | Higher mucoadhesive on the urothelium | [221] |
| rGO | HA-PEG-g-poly(dimethylaminoethyl methacrylate) | 120−190 | - | Biocompatibility, in vitro cellular uptake sensitive to cancer cells | [222] |
| MSN | Galactose | 277 | Camptothecin | MSN targeting to cancer cells | [59] |
| rPEI- Cdots 3 | FA | 143 | - | Biocompatible, good siRNA gene delivery carrier | [223] |
| PLGA NPs | bis(sulfosuccinimidyl) suberate (BS3) | 184 | Curcumin | Promote the loading of low-soluble drugs and aid in sustained released | [114] |
| ZnO NPs | PBA | 414 | Curcumin | Curcumin distribution to the sialic acid is much easier by PBA conjugation | [88] |
| Se NPs | (Arg–Gly–Asp–d-Phe–Cys [RGDfC]) cyclic peptide | 18 | DOX | Antitumor efficacy in vivo, effective cellular uptake A549 | [224] |
| CuO NPs | FA, starch | 108.83 | Cytochrome C | Antioxidants, anticancer, antimicrobial, drug-carrier | [35] |
| MoS2 | FA, BSA | 133 | DOX | Excellent photothermal conversion ability | [225] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Díez-Pascual, A.M. Surface Engineering of Nanomaterials with Polymers, Biomolecules, and Small Ligands for Nanomedicine. Materials 2022, 15, 3251. https://doi.org/10.3390/ma15093251
Díez-Pascual AM. Surface Engineering of Nanomaterials with Polymers, Biomolecules, and Small Ligands for Nanomedicine. Materials. 2022; 15(9):3251. https://doi.org/10.3390/ma15093251
Chicago/Turabian StyleDíez-Pascual, Ana M. 2022. "Surface Engineering of Nanomaterials with Polymers, Biomolecules, and Small Ligands for Nanomedicine" Materials 15, no. 9: 3251. https://doi.org/10.3390/ma15093251
APA StyleDíez-Pascual, A. M. (2022). Surface Engineering of Nanomaterials with Polymers, Biomolecules, and Small Ligands for Nanomedicine. Materials, 15(9), 3251. https://doi.org/10.3390/ma15093251