Thermal Kinetics of Monocationic and Dicationic Pyrrolidinium-Based Ionic Liquids
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Instrumentations
2.2.1. Structural Characterization
2.2.2. Elemental Analysis
2.2.3. Thermal Pyrolysis of ILs
2.3. Experimental
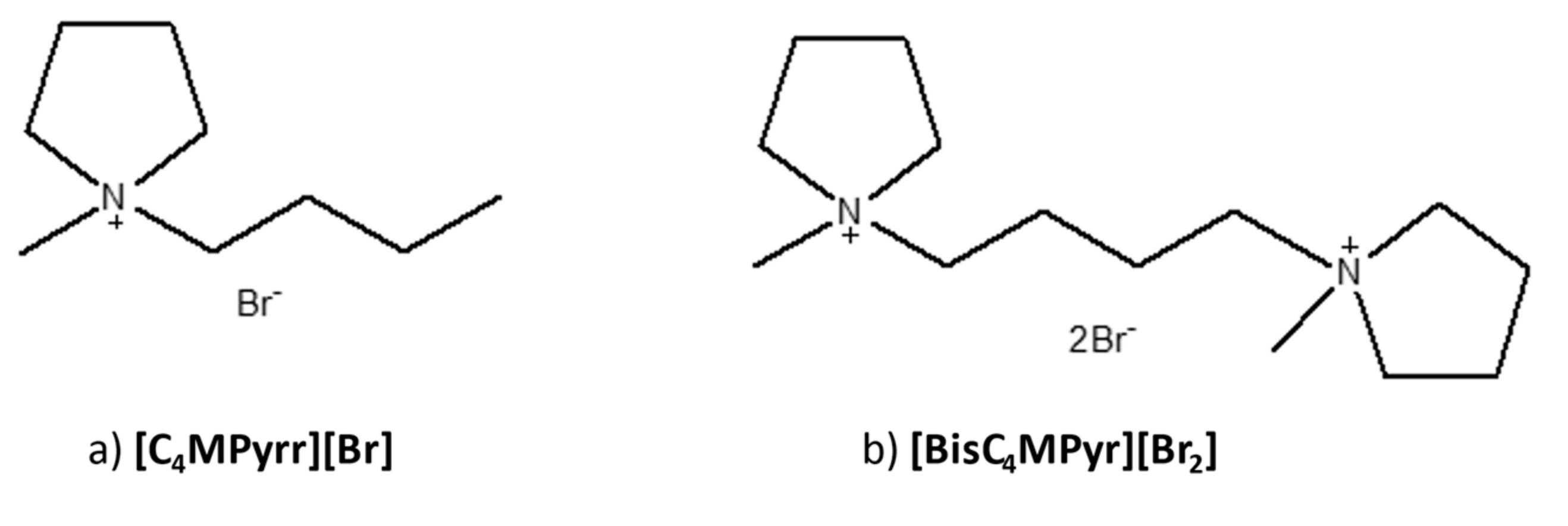
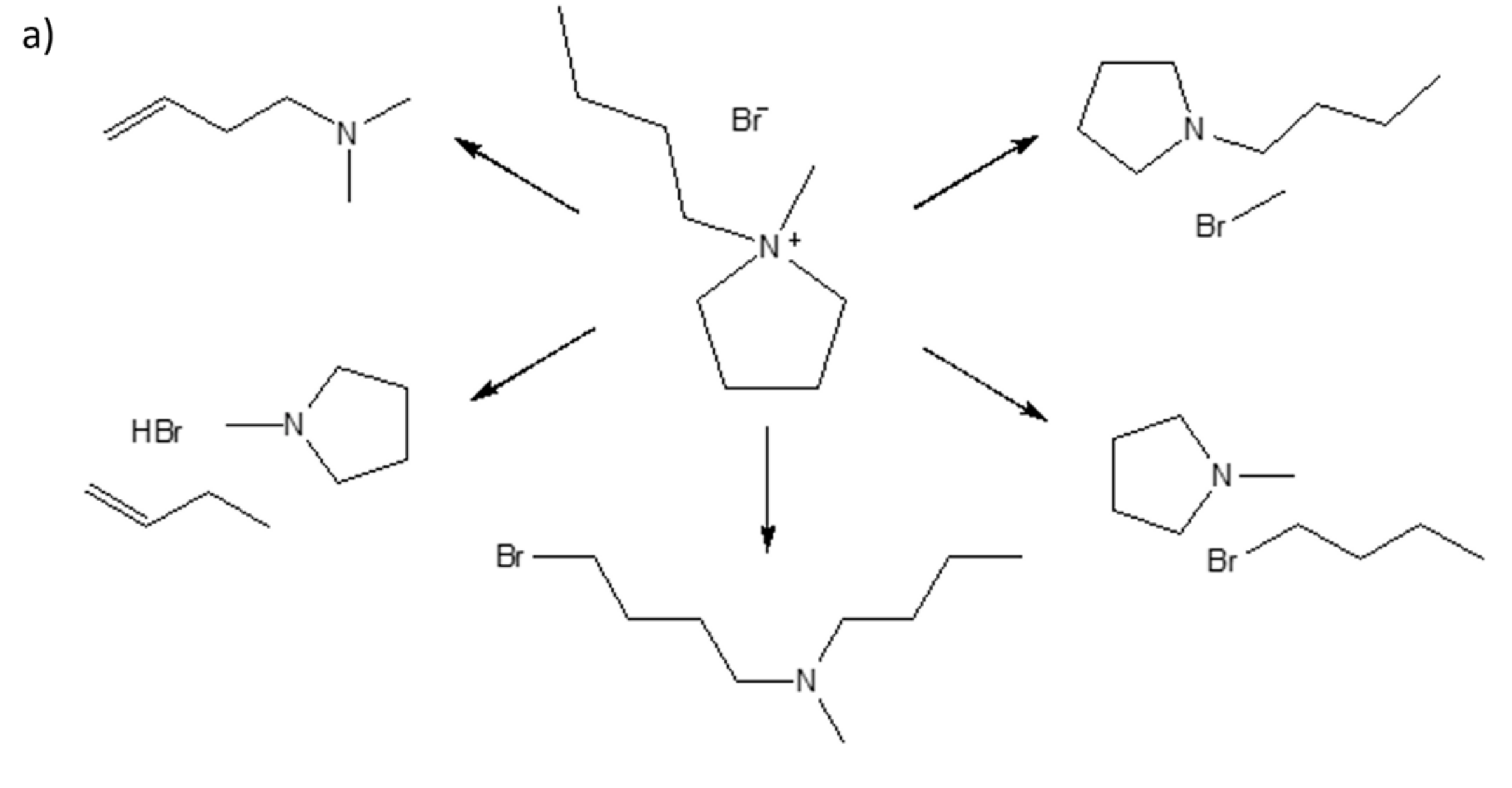
2.3.1. Synthesis of Monocationic IL; 1-butyl-1-methylpyrrolidinium bromide [C4MPyr][Br]
2.3.2. Synthesis of Dicationic IL; 1,4-bis(1-methylpyrrolidinium-1-yl)butane dibromide [BisC4MPyr][Br2]
2.4. Kinetic Thermal Decomposition
3. Results and Discussions
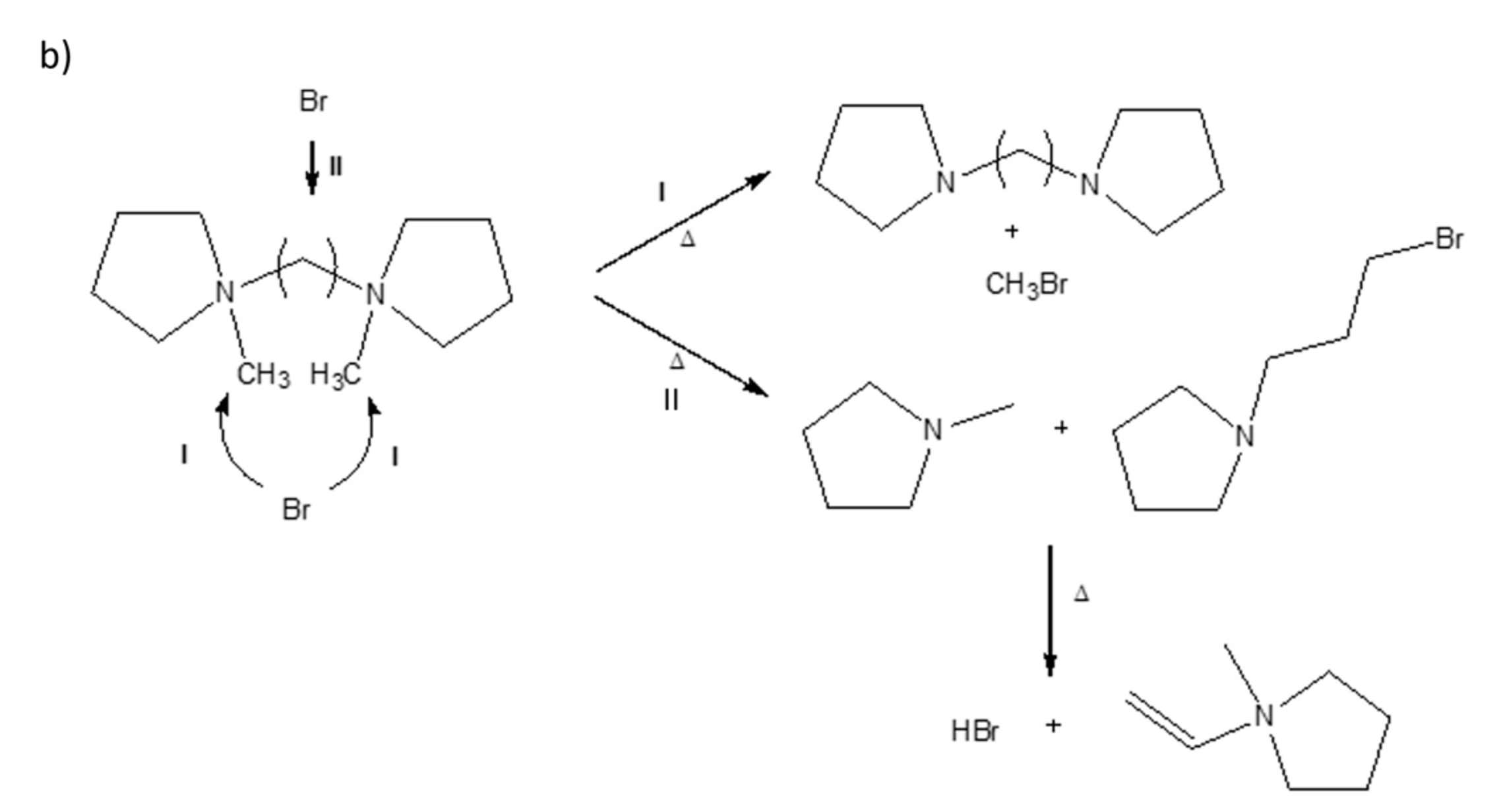
3.1. Thermal Decomposition Analysis
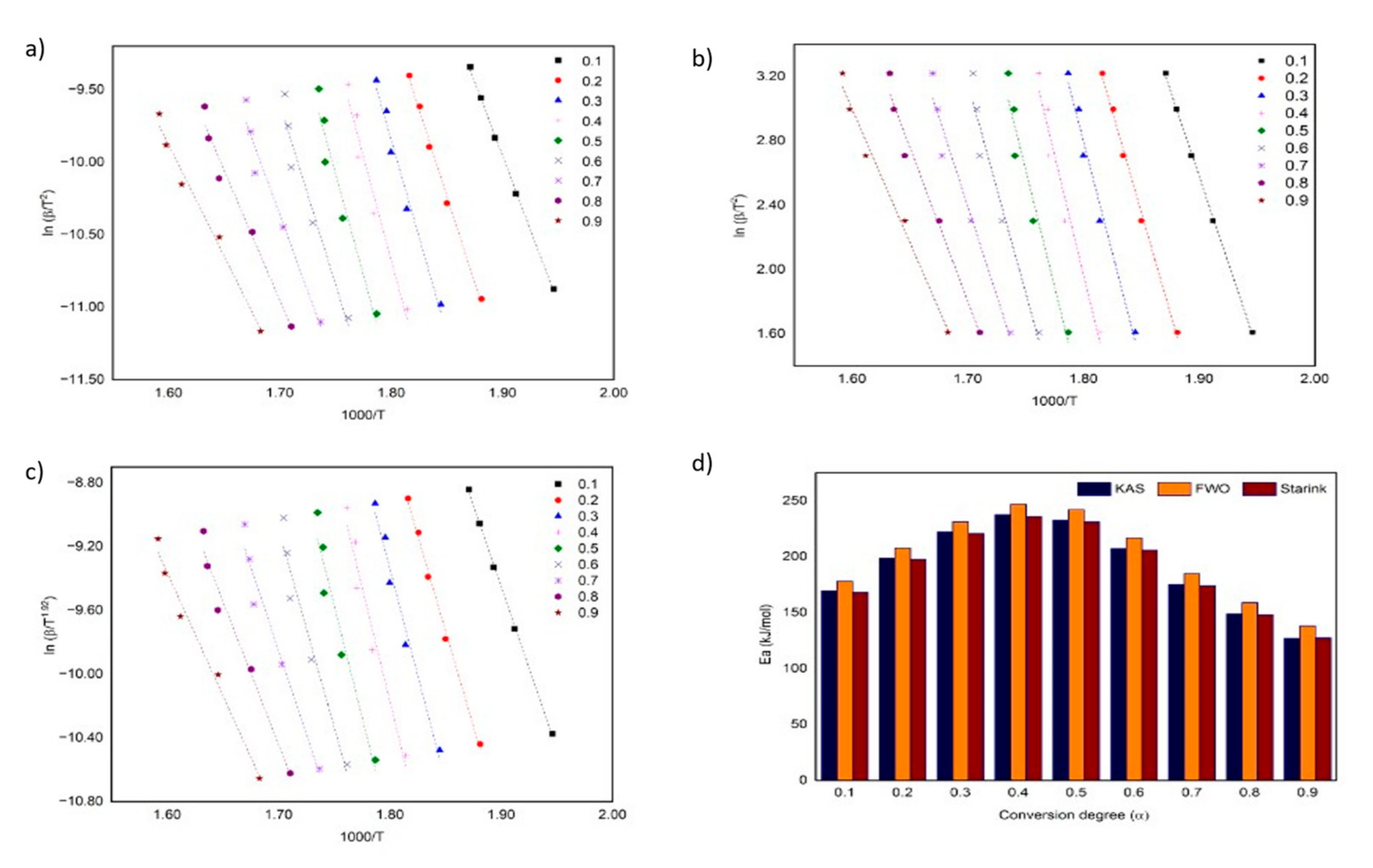
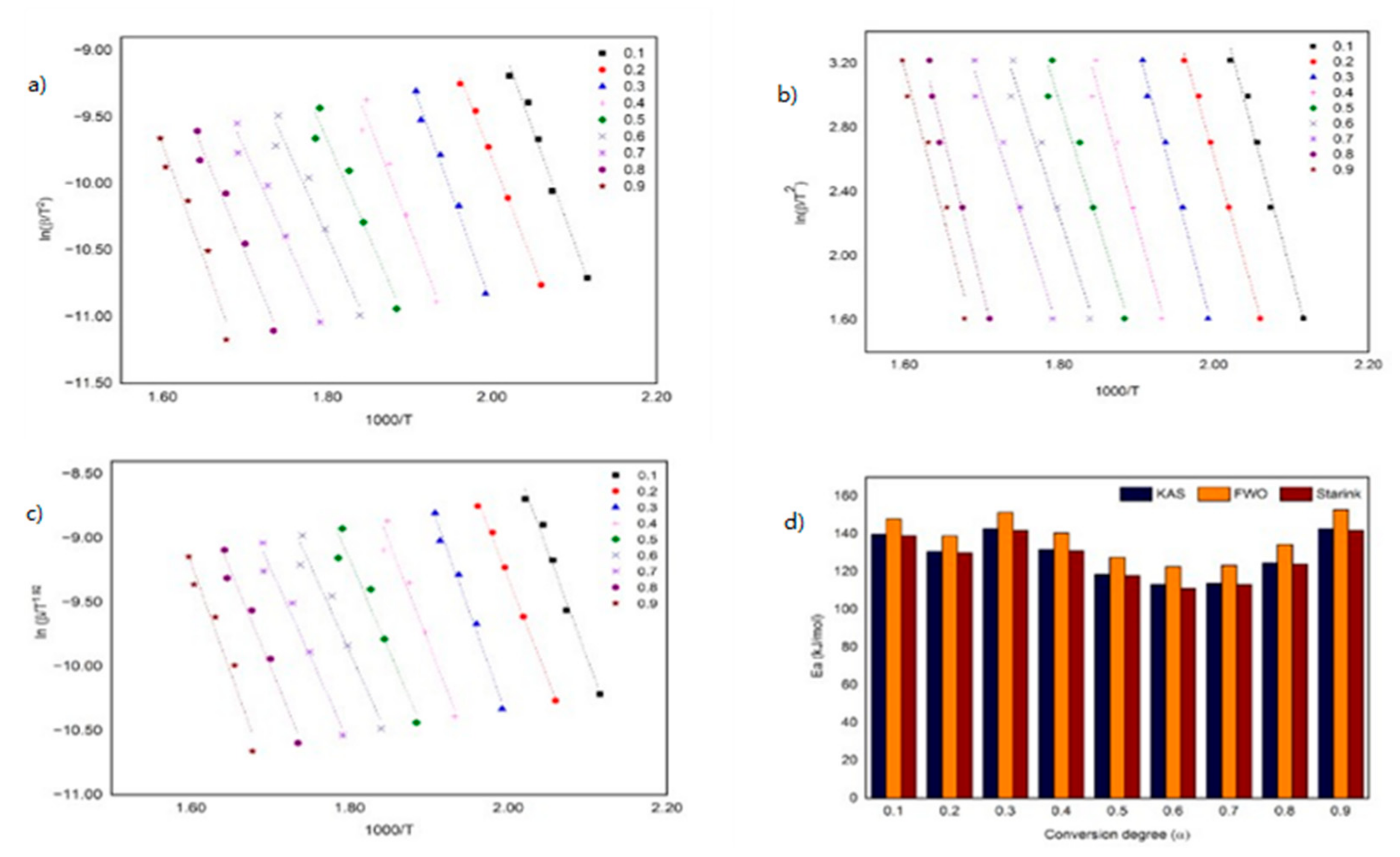
3.2. Kinetic of Thermal Decomposition
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Welton, T. Ionic liquids: A brief history. Biophys. Rev. 2018, 10, 691–706. [Google Scholar] [CrossRef] [Green Version]
- Hurley, F.H.; Wier, T.P., Jr. Electrodeposition of metals from fused quaternary ammonium salts. J. Electrochem. Soc. 1951, 98, 203. [Google Scholar] [CrossRef] [Green Version]
- Robinson, J.; Osteryoung, R. An electrochemical and spectroscopic study of some aromatic hydrocarbons in the room temperature molten salt system aluminum chloride-n-butylpyridinium chloride. J. Am. Chem. Soc. 1979, 101, 323–327. [Google Scholar] [CrossRef]
- Dharaskar, S.A.; Varma, M.N.; Shende, D.Z.; Yoo, C.K.; Wasewar, K.L. Synthesis, characterization and application of 1-butyl-3 methylimidazolium chloride as green material for extractive desulfurization of liquid fuel. Sci. World J. 2013, 2013, 395274. [Google Scholar] [CrossRef]
- Greer, A.J.; Jacquemin, J.; Hardacre, C. Industrial Applications of Ionic Liquids. Molecules 2020, 25, 5207. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Qin, L.; Mu, T.; Xue, Z.; Gao, G. Are ionic liquids chemically stable? Chem. Rev. 2017, 117, 7113–7131. [Google Scholar] [CrossRef]
- Dong, K.; Zhang, S.; Wang, J. Understanding the hydrogen bonds in ionic liquids and their roles in properties and reactions. Chem. Commun. 2016, 52, 6744–6764. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Cheng, Z. Thermal Stability of Ionic Liquids: Current Status and Prospects for Future Development. Processes 2021, 9, 337. [Google Scholar] [CrossRef]
- Xue, Z.; Qin, L.; Jiang, J.; Mu, T.; Gao, G. Thermal, electrochemical and radiolytic stabilities of ionic liquids. Phys. Chem. Chem. Phys. 2018, 20, 8382–8402. [Google Scholar] [CrossRef]
- Cao, Y.; Mu, T. Comprehensive investigation on the thermal stability of 66 ionic liquids by thermogravimetric analysis. Ind. Eng. Chem. Res. 2014, 53, 8651–8664. [Google Scholar] [CrossRef]
- Sbirrazzuoli, N. Interpretation and physical meaning of kinetic parameters obtained from isoconversional kinetic analysis of polymers. Polymers 2020, 12, 1280. [Google Scholar] [CrossRef] [PubMed]
- Al-Sallami, W.; Parsaeian, P.; Neville, A. An appraisal of the thermal decomposition mechanisms of ILs as potential lubricants. Lubr. Sci. 2019, 31, 229–238. [Google Scholar] [CrossRef]
- Akcay, A.; Balci, V.; Uzun, A. Structural Factors Controlling Thermal Stability of Imidazolium Ionic Liquids with 1-n-butyl-3-methylimidazolium cation on y-Al2O3. Thermochim. Acta 2014, 589, 131–136. [Google Scholar] [CrossRef]
- Anderson, J.L.; Ding, R.; Ellern, A.; Armstrong, D.W. Structure and properties of high stability geminal dicationic ionic liquids. J. Am. Chem. Soc. 2005, 127, 593–604. [Google Scholar] [CrossRef] [Green Version]
- Masri, A.N.; MI, A.M.; Leveque, J.-M. A review on dicationic ionic liquids: Classification and application. Ind. Eng. Manag. 2016, 5, 197–204. [Google Scholar]
- Shirota, H.; Mandai, T.; Fukazawa, H.; Kato, T. Comparison between dicationic and monocationic ionic liquids: Liquid density, thermal properties, surface tension, and shear viscosity. J. Chem. Eng. Data 2011, 56, 2453–2459. [Google Scholar] [CrossRef]
- Medjahed, N.; Debdab, M.; Haddad, B.; Belarbi, E.; KIBOU, Z.; Berrichi, A.; Bachir, R.; Choukchou-Braham, N. Synthesis and Structural Characterization of Imidazolium-Based Dicationic Ionic Liquids. Chem. Proc. 2021, 3, 80. [Google Scholar] [CrossRef]
- Qi, M.; Armstrong, D.W. Dicationic ionic liquid stationary phase for GC-MS analysis of volatile compounds in herbal plants. Anal. Bioanal.Chem. 2007, 388, 889–899. [Google Scholar] [CrossRef]
- Boumediene, M.; Haddad, B.; Paolone, A.; Assenine, M.A.; Villemin, D.; Rahmouni, M.; Bresson, S. Synthesis, conformational studies, vibrational spectra and thermal properties, of new substituted dicationic based on biphenylenedimethylene linked bis-1-methylimidazolium ionic liquids. J. Mol. Struct. 2020. [Google Scholar] [CrossRef]
- Khan, A.S.; Man, Z.; Bustam, M.A.; Kait, C.F.; Ullah, Z.; Sarwono, A.; Wilfred, C.D. Pyrolysis kinetics of 1-propyronitrile imidazolium trifluoroacetate ionic liquid using thermogravimetric analysis. Procedia Eng. 2016, 148, 1332–1339. [Google Scholar] [CrossRef] [Green Version]
- Muhammad, A. Thermal and kinetic analysis of pure and contaminated ionic liquid: 1-butyl-2.3-dimethylimidazolium chloride (BDMIMCl). Pol. J. Chem. Technol. 2016, 18, 122–125. [Google Scholar] [CrossRef] [Green Version]
- Quraishi, K.S.; Bustam, M.A.; Krishnan, S.; Aminuddin, N.F.; Azeezah, N.; Ghani, N.A.; Uemura, Y.; Lévêque, J.M. Ionic liquids toxicity on fresh water microalgae, Scenedesmus quadricauda, Chlorella vulgaris & Botryococcus braunii; selection criterion for use in a two-phase partitioning bioreactor (TPPBR). Chemosphere 2017, 184, 642–651. [Google Scholar] [PubMed]
- Lombardo, M.; Easwar, S.; Pasi, F.; Trombini, C.; Dhavale, D.D. Protonated arginine and lysine as catalysts for the direct asymmetric aldol reaction in ionic liquids. Tetrahedron 2008, 64, 9203–9207. [Google Scholar] [CrossRef]
- Burrell, A.K.; Del Sesto, R.E.; Baker, S.N.; McCleskey, T.M.; Baker, G.A. The large scale synthesis of pure imidazolium and pyrrolidinium ionic liquids. Green Chem. 2007, 9, 449–454. [Google Scholar] [CrossRef]
- Montalbán, M.G.; Collado-González, M.; Díaz-Baños, F.G.; Víllora, G. Predicting Density and Refractive Index of Ionic Liquids. In Ionic Liquids: Progress and Developments in; IntechOpen Limited: London, UK, 2017; p. 339. [Google Scholar]
- Masri, A.; Mutalib, M.A.; Yahya, W.; Aminuddin, N.; Leveque, J. Thermokinetics of SO3H-functionalized dicationic ionic liquids: Effect of anions. IOP Conf. Ser. Mater. Sci. Eng. 2018, 458, 012072. [Google Scholar] [CrossRef]
- Meng, J.; Pan, Y.; Yang, F.; Wang, Y.; Zheng, Z.; Jiang, J. Thermal Stability and Decomposition Kinetics of 1-Alkyl-2,3-Dimethylimidazolium Nitrate Ionic Liquids: TGA and DFT Study. Materials 2021, 14, 2560. [Google Scholar] [CrossRef] [PubMed]
- Bender, C.R.; Kuhn, B.L.; Farias, C.A.; Ziembowicz, F.I.; Beck, T.S.; Frizzo, C.P. Thermal Stability and Kinetic of Decomposition of Mono-and Dicationic Imidazolium-Based Ionic Liquids. J. Braz. Chem. Soc. 2019, 30, 2199–2209. [Google Scholar] [CrossRef]
- Fareghi-Alamdari, R.; Ghorbani–Zamania, F.; Shekarriz, M. Synthesis and thermal characterization of mono and dicationicimidazolium-pyridinium based ionic liquids. Orient. J. Chem. 2015, 31, 1127. [Google Scholar] [CrossRef]
- Guglielmero, L.; Mezzetta, A.; Guazzelli, L.; Pomelli, C.S.; D’Andrea, F.; Chiappe, C. Systematic synthesis and properties evaluation of dicationic ionic liquids, and a glance into a potential new field. Front. Chem. 2018, 6, 612. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Al-Moftah, A.M.S.; Marsh, R.; Steer, J. Thermal decomposition kinetic study of non-recyclable paper and plastic waste by thermogravimetric analysis. ChemEngineering 2021, 5, 54. [Google Scholar] [CrossRef]
- Chen, Y.; Mu, T. Thermal Stability of Ionic Liquids. In Encyclopedia of Ionic Liquids; Zhang, S., Ed.; Springer: Singapore, 2019; pp. 1–13. [Google Scholar] [CrossRef]
- Patil, R.A.; Talebi, M.; Berthod, A.; Armstrong, D.W. Dicationic ionic liquid thermal decomposition pathways. Anal. Bioanal. Chem. 2018, 410, 4645–4655. [Google Scholar] [CrossRef]
- Wooster, T.J.; Johanson, K.M.; Fraser, K.J.; MacFarlane, D.R.; Scott, J.L. Thermal degradation of cyano containing ionic liquids. Green Chem. 2006, 8, 691–696. [Google Scholar] [CrossRef]
- Guida, M.; Lanaya, S.; Rbihi, Z.; Hannioui, A. Thermal degradation behaviors of sawdust wood waste: Pyrolysis kinetic and mechanism. J. Mater. Environ. Sci. 2019, 10, 742–755. [Google Scholar]
- Jiang, H.-C.; Lin, W.-C.; Hua, M.; Pan, X.-H.; Shu, C.-M.; Jiang, J.-C. Analysis of thermal stability and pyrolysis kinetic of dibutyl phosphate-based ionic liquid through thermogravimetry, gas chromatography/mass spectrometry, and Fourier transform infrared spectrometry. J. Therm. Anal. Calorim. 2019, 138, 489–499. [Google Scholar] [CrossRef]
- Efimova, A.; Pfützner, L.; Schmidt, P. Thermal stability and decomposition mechanism of 1-ethyl-3-methylimidazolium halides. Thermochim. Acta 2015, 604, 129–136. [Google Scholar] [CrossRef]
- Parajo, J.; Villanueva, M.; Troncoso, J.; Salgado, J. Thermophysical properties of choline and pyridinium based ionic liquids as advanced materials for energy applications. J. Chem. Thermodyn. 2020, 141, 105947. [Google Scholar] [CrossRef]
- Navarro, P.; Larriba, M.; García, J.; Rodríguez, F. Thermal stability and specific heats of {[emim][DCA]+[emim][TCM]} mixed ionic liquids. Thermochim. Acta 2014, 588, 22–27. [Google Scholar] [CrossRef]
- Parajó, J.J.; Villanueva, M.; Salgado, J. 1. Thermal stability of ionic liquids. In Ionic Liquids: Synthesis, Properties, Technologies and Applications; Rasmus, F., Catherine, S., Eds.; De Gruyter: Berlin, Germany, 2019; pp. 1–16. [Google Scholar] [CrossRef]
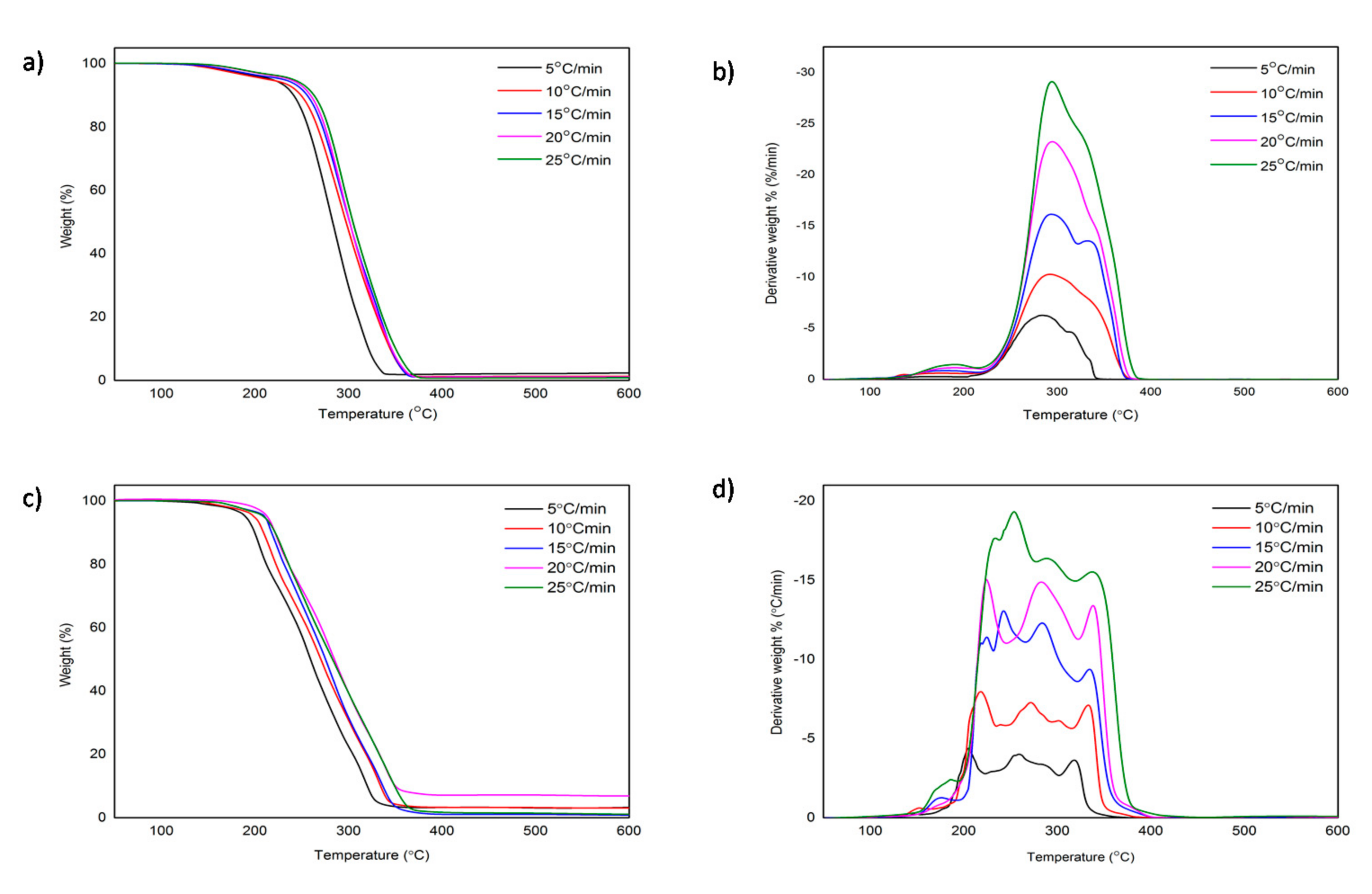
| Heating Rate/°C min−1 | [C4MPyr][Br] | [BisC4MPyr][Br2] | ||
|---|---|---|---|---|
| To (°C) | Tmax (°C) | To (°C) | Tmax (°C) | |
| 5 | 255 ± 10 | 297 ± 20 | 191 ± 1 | 208 ± 3 |
| 10 | 253 ± 2 | 292 ± 1 | 201 ± 4 | 218 ± 2 |
| 15 | 259 ± 1 | 293. ± 1 | 206 ± 3 | 237 ± 7 |
| 20 | 265 ± 2 | 290 ± 5 | 206 ± 3 | 236 ± 14 |
| 25 | 265 ± 3 | 293 ± 1 | 213 ± 4 | 251 ± 5 |
| Method | α | Ea (kJ/mol) | log A (min−1) | R2 | |||
|---|---|---|---|---|---|---|---|
| [C4MPyr][Br] | [BisC4MPyr][Br2] | [C4MPyr][Br] | [BisC4MPyr][Br2] | [C4MPyr][Br] | [BisC4MPyr][Br2] | ||
| KAS | 0.1 | 169.28 | 139.79 | 15.79 | 14.03 | 0.9991 | 0.9837 |
| 0.2 | 198.85 | 130.61 | 18.45 | 12.88 | 0.9953 | 0.9969 | |
| 0.3 | 222.03 | 142.82 | 20.51 | 13.89 | 0.9778 | 0.9885 | |
| 0.4 | 237.51 | 131.72 | 21.76 | 12.39 | 0.9544 | 0.9594 | |
| 0.5 | 232.59 | 118.52 | 21.03 | 10.80 | 0.941 | 0.9464 | |
| 0.6 | 207.17 | 113.32 | 18.41 | 10.06 | 0.9475 | 0.9585 | |
| 0.7 | 175.08 | 113.77 | 15.22 | 9.86 | 0.9601 | 0.9715 | |
| 0.8 | 148.97 | 124.55 | 12.63 | 10.58 | 0.9739 | 0.9739 | |
| 0.9 | 127.96 | 142.71 | 10.55 | 11.90 | 0.9846 | 0.956 | |
| FWO | 0.1 | 177.99 | 147.83 | 15.77 | 14.10 | 0.9991 | 0.9894 |
| 0.2 | 207.83 | 138.88 | 18.33 | 13.04 | 0.9957 | 0.9973 | |
| 0.3 | 231.19 | 151.35 | 20.30 | 14.00 | 0.9794 | 0.9898 | |
| 0.4 | 246.81 | 140.53 | 21.51 | 12.60 | 0.9576 | 0.9642 | |
| 0.5 | 242.04 | 127.59 | 20.81 | 11.12 | 0.9452 | 0.9535 | |
| 0.6 | 216.76 | 122.63 | 18.30 | 10.43 | 0.9518 | 0.9645 | |
| 0.7 | 184.85 | 123.34 | 15.27 | 10.25 | 0.9640 | 0.9757 | |
| 0.8 | 158.92 | 134.41 | 12.83 | 10.92 | 0.9770 | 0.9776 | |
| 0.9 | 138.12 | 152.87 | 10.90 | 12.16 | 0.9868 | 0.9616 | |
| Starink | 0.1 | 168.28 | 139.00 | 16.83 | 15.07 | 0.9991 | 0.9837 |
| 0.2 | 197.62 | 129.91 | 19.19 | 13.63 | 0.9953 | 0.9969 | |
| 0.3 | 220.63 | 142.02 | 21.06 | 14.46 | 0.9779 | 0.9885 | |
| 0.4 | 236.00 | 131.02 | 22.18 | 12.84 | 0.9545 | 0.9596 | |
| 0.5 | 231.13 | 117.93 | 21.37 | 11.15 | 0.9412 | 0.9467 | |
| 0.6 | 205.90 | 111.14 | 18.66 | 12.65 | 0.9477 | 0.9588 | |
| 0.7 | 174.08 | 113.25 | 15.42 | 10.07 | 0.9603 | 0.9717 | |
| 0.8 | 148.18 | 123.96 | 12.78 | 10.73 | 0.9741 | 0.9741 | |
| 0.9 | 127.35 | 141.97 | 10.65 | 15.07 | 0.9847 | 0.9563 | |
| Methods | [C4MPyr][Br]/°C | [BisC4MPyr][Br2]/°C |
|---|---|---|
| KAS | 409.73 | 398.29 |
| FWO | 400.73 | 375.67 |
| Starink | 435.42 | 423.68 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ab Rahim, A.H.; Abd Ghani, N.; Hasanudin, N.; Yunus, N.M.; Azman, N.S. Thermal Kinetics of Monocationic and Dicationic Pyrrolidinium-Based Ionic Liquids. Materials 2022, 15, 1247. https://doi.org/10.3390/ma15031247
Ab Rahim AH, Abd Ghani N, Hasanudin N, Yunus NM, Azman NS. Thermal Kinetics of Monocationic and Dicationic Pyrrolidinium-Based Ionic Liquids. Materials. 2022; 15(3):1247. https://doi.org/10.3390/ma15031247
Chicago/Turabian StyleAb Rahim, Asyraf Hanim, Noraini Abd Ghani, Noorhafizah Hasanudin, Normawati M. Yunus, and Ninna Sakina Azman. 2022. "Thermal Kinetics of Monocationic and Dicationic Pyrrolidinium-Based Ionic Liquids" Materials 15, no. 3: 1247. https://doi.org/10.3390/ma15031247
APA StyleAb Rahim, A. H., Abd Ghani, N., Hasanudin, N., Yunus, N. M., & Azman, N. S. (2022). Thermal Kinetics of Monocationic and Dicationic Pyrrolidinium-Based Ionic Liquids. Materials, 15(3), 1247. https://doi.org/10.3390/ma15031247






