Phase Transitions and Electric Properties of PbBr2 under High Pressure: A First-Principles Study
Abstract
1. Introduction
2. Materials and Methods
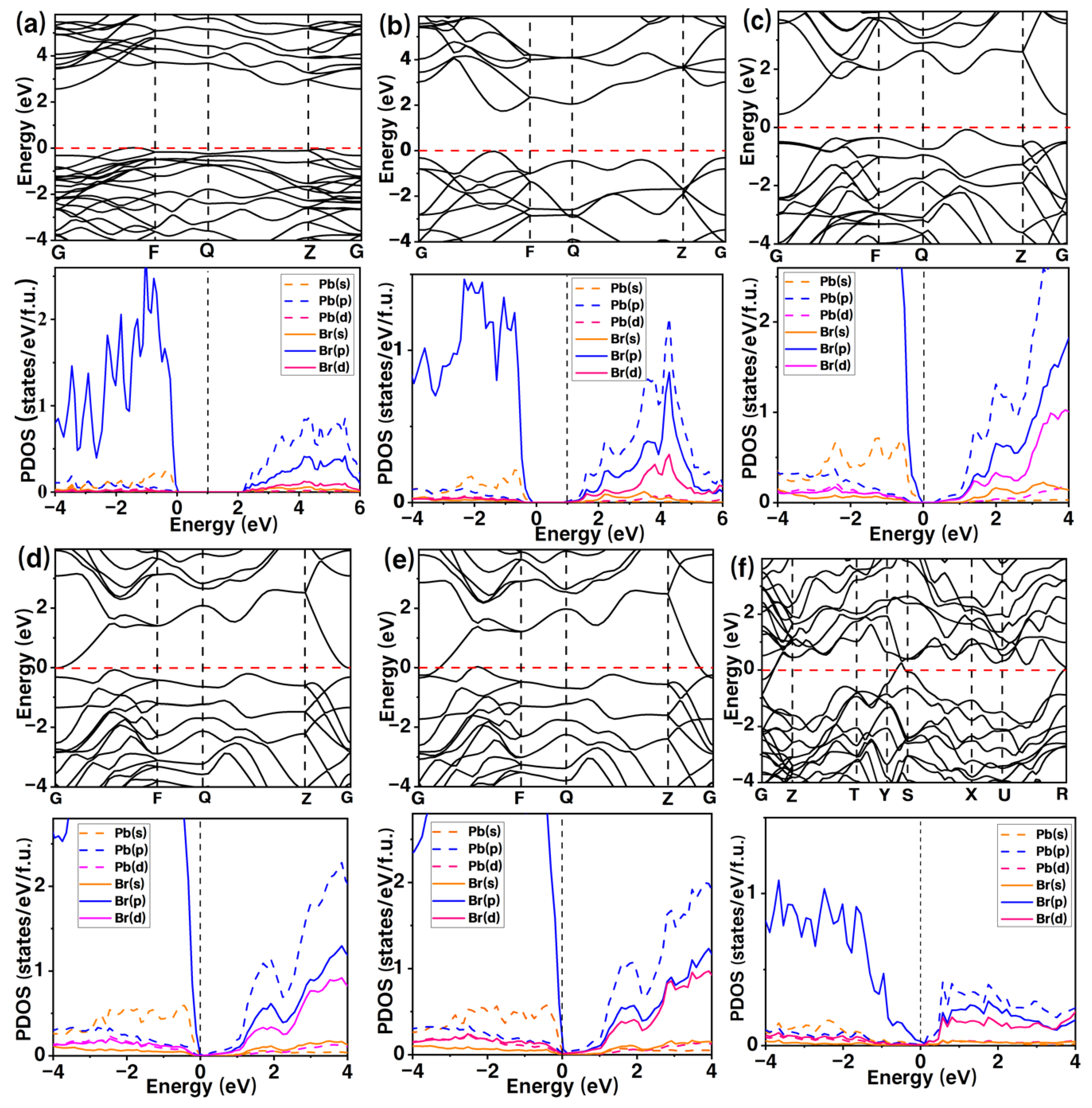
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yanagida, T.; Fujimoto, Y.; Yoshikawa, A.; Yokota, Y.; Kamada, K.; Pejchal, J.; Chani, V.; Kawaguchi, N.; Fukuda, K.; Uchiyama, K.; et al. Development and Performance Test of Picosecond Pulse X-ray Excited Streak Camera System for Scintillator Characterization. Appl. Phys. Express 2010, 3, 056202. [Google Scholar] [CrossRef]
- Torres, O.G.; Gordillo, G.; Plazas, M.C.; Téllez, D.A.L.; Roa-Rojas, J. Optical features of PbBr2 semiconductor thin films for radiation attenuation application. J. Mater. Sci. Mater. Electron. 2021, 32, 16937–16944. [Google Scholar] [CrossRef]
- Nikl, M. Scintillation detectors for X-rays. Meas. Sci. Technol. 2006, 17, R37–R54. [Google Scholar] [CrossRef]
- Tubbs, M.R. The optical properties and chemical decomposition of halides with layer structures. I. crystal structures, optical properties, and electronic structure. Phys. Status Solidi 1972, 49, 11–50. [Google Scholar] [CrossRef]
- Kusumoto, H.; Kaito, T.; Yanagiya, S.-I.; Mori, A.; Inoue, T. Growth of single crystals of PbBr2 in silica gel. J. Cryst. Growth 2005, 277, 536–540. [Google Scholar] [CrossRef]
- Kaito, T.; Yanagiya, S.-I.; Mori, A.; Kurumada, M.; Kaito, C.; Inoue, T. Characteristic nanocrystallite growth of PbBr2 in a magnetic field in gel. J. Cryst. Growth 2006, 294, 407–410. [Google Scholar] [CrossRef]
- Kaito, T.; Inoue, T.; Yanagiya, S.; Mori, A.; Masaki, K. Melt growth and characterization of PbBr2 single crystals. J. Cryst. Growth 2005, 275, e721–e726. [Google Scholar] [CrossRef]
- Ahmad, Z.; Mishra, A. Growth of PbBr2 microrods with unique structure and surface morphology. J. Mater. Sci. Mater. Electron. 2020, 31, 4672. [Google Scholar] [CrossRef]
- Girgis, S.Y.; Mady, K.A. Electrical conductivity of vacuum-deposited lead bromide layers. J. Mater. Sci. Lett. 1986, 5, 1091–1094. [Google Scholar] [CrossRef]
- Varshney, S.; Chi, L.; Singh, C.V.; Nogami, J. Atomic structure of PbBr2 thin films on Ag (111). Solid State Commun. 2022, 343, 114651. [Google Scholar] [CrossRef]
- Mutalikdesai, A.; Ramasesha, S.K. Emerging solar technologies: Perovskite solar cell. Resonance 2017, 22, 1061–1083. [Google Scholar] [CrossRef]
- Cleveland, I.J.; Tran, M.N.; Dey, A.; Aydil, E.S. Vapor deposition of CsPbBr3 thin films by evaporation of CsBr and PbBr2. J. Vac. Sci. Technol. A 2021, 39, 043415. [Google Scholar] [CrossRef]
- Ren, Q.; Ding, L.-Y.; Chen, F.-S.; Cheng, R.-P.; Xu, D. The optical properties of lead bromide crystals. J. Mater. Sci. Lett. 1997, 16, 1247–1248. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, Y.; Yang, Z.; Feng, J.; Xu, Z.; Li, Q.; Hu, M.; Ye, H.; Zhang, X.; Liu, M.; et al. Low-temperature-gradient crystallization for multi-inch high-quality perovskite single crystals for record performance photodetectors. Mater. Today 2019, 22, 67–75. [Google Scholar] [CrossRef]
- Lan, J.; Luo, L.; Wang, M.; Li, F.; Wu, X.; Wang, F. One pot gram-scale synthesis of CsPbBr3 nanocrystals and their application in green LED. J. Lumin. 2019, 210, 464–471. [Google Scholar] [CrossRef]
- Zhao, Y.; Wang, Y.; Liang, X.; Shi, H.; Wang, C.; Fan, J.; Hu, X.; Liu, E. Enhanced photocatalytic activity of Ag-CsPbBr3/CN composite for broad spectrum photocatalytic degradation of cephalosporin antibiotics 7-ACA. Appl. Catal. B Environ. 2019, 247, 57–69. [Google Scholar] [CrossRef]
- Chin, S.-H.; Choi, J.W.; Hu, Z.; Mardegan, L.; Sessolo, M.; Bolink, H.J. Tunable luminescent lead bromide complexes. J. Mater. Chem. C 2020, 8, 15996–16000. [Google Scholar] [CrossRef]
- Yin, J.; Zhang, Y.; Bruno, A.; Soci, C.; Bakr, O.M.; Brédas, J.-L.; Mohammed, O.F. Intrinsic Lead Ion Emissions in Zero-Dimensional Cs4PbBr6 Nanocrystals. ACS Energy Lett. 2017, 2, 2805–2811. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, R.; Wang, Z.; Li, D.; Cui, T. Formation of twelve-fold iodine coordination at high pressure. Nat. Commun. 2022, 13, 412. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Wang, Y.; Lv, J.; Ma, Y. Materials discovery at high pressures. Nat. Rev. Mater. 2017, 2, 17005. [Google Scholar] [CrossRef]
- Liu, R.; Xu, D.; Yao, Z.; Niu, S.; Liu, B. The New High-Pressure Phases of Nitrogen-Rich Ag–N Compounds. Materials 2022, 15, 4986. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Wang, H.; Sun, W.; Lu, C.; Peng, F. Superconductivity in compressed ternary alkaline boron hydrides. Phys. Rev. B 2022, 105, 224107. [Google Scholar] [CrossRef]
- Yang, L.; Qu, X.; Zhong, X.; Wang, D.; Chen, Y.; Lang, J.; Liu, C.; Sun, B.; Yang, J. The unconventionally stoichiometric compounds in the Na–K system at high pressures. Comput. Mater. Sci. 2021, 200, 110818. [Google Scholar] [CrossRef]
- Qu, X.; Yang, L.; Lv, J.; Xie, Y.; Yang, J.; Zhang, Y.; Wang, Y.; Zhao, J.; Chen, Z.; Ma, Y. Particle Swarm Predictions of a SrB8 Monolayer with 12-Fold Metal Coordination. J. Am. Chem. Soc. 2022, 144, 11120–11128. [Google Scholar] [CrossRef]
- Togo, A.; Oba, F.; Tanaka, I. First-principles calculations of the ferroelastic transition between rutile-type and CaCl2-type SiO2 at high pressures. Phys. Rev. B Condens. Matter Mater. Phys. 2008, 78, 134106. [Google Scholar] [CrossRef]
- Raman, A.; Steinfink, H. Crystal chemistry of AB2 structures. Inorg. Chem. 1967, 6, 1789–1791. [Google Scholar] [CrossRef]
- Wang, Y.; Lv, J.; Ma, Y.; Cui, T.; Zou, G. Superconductivity of MgB2 under Ultra high Pressure: A First-Principles Study. Phys. Rev. B 2009, 80, 092505. [Google Scholar] [CrossRef]
- Wang, Y.; Lv, J.; Zhu, L.; Ma, Y. CALYPSO: A Method for Crystal Structure Prediction. Comput. Phys. Commun. 2012, 183, 2063–2070. [Google Scholar] [CrossRef]
- Lv, J.; Wang, Y.; Zhu, L.; Ma, Y. Particle-swarm structure prediction on clusters. J. Chem. Phys. 2012, 137, 084104. [Google Scholar] [CrossRef]
- Lu, W.; Liu, S.; Liu, G.; Hao, K.; Zhou, M.; Gao, P.; Wang, H.; Lv, J.; Gou, H.; Yang, G.; et al. Disproportionation of SO2 at High Pressure and Temperature. Phys. Rev. Lett. 2022, 128, 106001. [Google Scholar] [CrossRef]
- Zhong, X.; Sun, Y.; Iitaka, T.; Xu, M.; Liu, H.; Hemley, R.J.; Chen, C.; Ma, Y. Prediction of Above-Room-Temperature Superconductivity in Lanthanide/Actinide Extreme Superhydrides. J. Am. Chem. Soc. 2022, 144, 13394–13400. [Google Scholar] [CrossRef]
- Zhu, L.; Liu, H.; Pickard, C.J.; Zou, G.; Ma, Y. Reactions of Xenon with Iron and Nickel Are Predicted in the Earth’s Inner Core. Nat. Chem. 2014, 6, 644–648. [Google Scholar] [CrossRef]
- Li, Y.; Hao, J.; Liu, H.; Li, Y.; Ma, Y. The Metallization and Superconductivity of Dense Hydrogen Sulfide. J. Chem. Phys. 2014, 140, 174712. [Google Scholar] [CrossRef]
- Li, Y.; Wang, L.; Liu, H.; Zhang, Y.; Hao, J.; Pickard, C.J.; Nelson, J.R.; Needs, R.J.; Li, W.; Huang, Y.; et al. Dissociation Products and Structures of Solid H2S at Strong Compression. Phys. Rev. B 2016, 93, 020103. [Google Scholar] [CrossRef]
- Li, X.; Zhang, X.; Bergara, A.; Gao, G.; Liu, Y.; Yang, G. Superconducting LaP2H2 with graphenelike phosphorus layers. Phys. Rev. B 2022, 105, 024504. [Google Scholar] [CrossRef]
- Yan, X.; Ding, S.; Zhang, X.; Bergara, A.; Liu, Y.; Wang, Y.; Zhou, X.-F.; Yang, G. Enhanced superconductivity in CuH2 monolayers. Phys. Rev. B 2022, 106, 014514. [Google Scholar] [CrossRef]
- Lv, J.; Wang, Y.; Zhu, L.; Ma, Y. Predicted Novel High–Pressure Phases of Lithium. Phys. Rev. Lett. 2011, 106, 015503. [Google Scholar] [CrossRef]
- Kresse, G.; Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B 1996, 54, 11169. [Google Scholar] [CrossRef]
- Perdew, J.P.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 1996, 77, 3865. [Google Scholar] [CrossRef]
- Blochl, P.E. Projector Augmented–Wave Method. Phys. Rev. B 1994, 50, 17953. [Google Scholar] [CrossRef]
- Momma, K.; Izumi, F. VESTA 3 for three-dimensional visualization of crystal, volumetric and morphology data. J. Appl. Crystallogr. 2011, 44, 1272–1276. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, Y.; Wang, J.; Wang, Y.; Yu, W.W. Pressure-induced phase transitions of lead iodide. RSC Adv. 2016, 6, 84604–84609. [Google Scholar] [CrossRef]
- Wei, S.; Liu, H. High-Pressure Structures and Superconductivity of Barium Iodide. Materials 2022, 15, 522. [Google Scholar] [CrossRef]
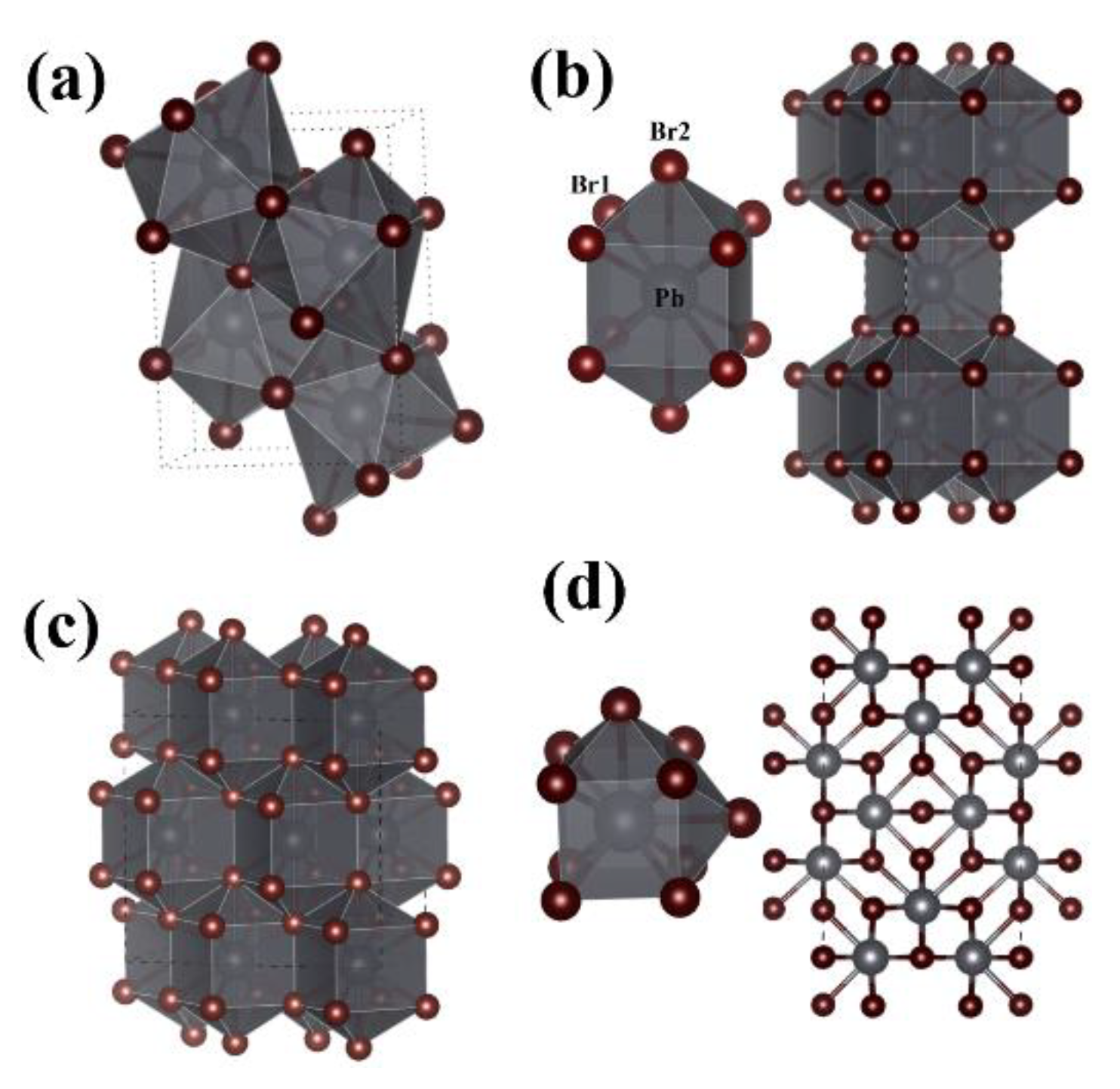



| Phase | Z | Lattice (Å) | Atom | X | Y | Z |
|---|---|---|---|---|---|---|
| Pnma 0 GPa | 4 | a = 8.04246 b = 4.76121 c = 9.66722 | Pb(4c) Br1(4c) Br2(4c) | 0.734 0.516 0.358 | 0.750 0.750 0.250 | 0.910 0.162 0.418 |
| I4/mmm 60 GPa | 2 | a = b = 3.426 c = 8.993 | Pb(2a) Br(4e) | 0.000 0.000 | 0.000 0.000 | 0.000 0.336 |
| Cmca 100 GPa | 8 | a = 8.548 b = 4.679 c = 9.295 α = β = γ = 90 | Pb(8f) Br(16g) | 0.000 −0.166 | 0.790 0.295 | 0.875 0.875 |
| Immm 200 GPa | 8 | a = 3.598 b = 11.177 c = 7.594 α = β = γ = 90 | Pb1(4i) Pb2(4h) Br1(2d) Br2(4g) Br3(8l) Br4(2b) | 0.000 0.000 0.500 0.000 0.500 0.500 | 0.000 0.824 0.000 0.837 0.833 0.000 | 0.241 0.500 0.500 0.000 0.253 0.000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, L.; Zhang, Y.; Chen, Y.; Zhong, X.; Wang, D.; Fan, L.; Lang, J.; Qu, X.; Yang, J. Phase Transitions and Electric Properties of PbBr2 under High Pressure: A First-Principles Study. Materials 2022, 15, 8222. https://doi.org/10.3390/ma15228222
Yang L, Zhang Y, Chen Y, Zhong X, Wang D, Fan L, Lang J, Qu X, Yang J. Phase Transitions and Electric Properties of PbBr2 under High Pressure: A First-Principles Study. Materials. 2022; 15(22):8222. https://doi.org/10.3390/ma15228222
Chicago/Turabian StyleYang, Lihua, Yukai Zhang, Yanli Chen, Xin Zhong, Dandan Wang, Lin Fan, Jihui Lang, Xin Qu, and Jinghai Yang. 2022. "Phase Transitions and Electric Properties of PbBr2 under High Pressure: A First-Principles Study" Materials 15, no. 22: 8222. https://doi.org/10.3390/ma15228222
APA StyleYang, L., Zhang, Y., Chen, Y., Zhong, X., Wang, D., Fan, L., Lang, J., Qu, X., & Yang, J. (2022). Phase Transitions and Electric Properties of PbBr2 under High Pressure: A First-Principles Study. Materials, 15(22), 8222. https://doi.org/10.3390/ma15228222






