Biocomposites with Epoxy Resin Matrix Modified with Ingredients of Natural Origin
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
- Microcrystalline cellulose from manufacturer Sigma-Aldrich (Saint Louis, MO, USA), in the form of powder.
- Short linen fibers made of Safilin roving (tex 2000).
- Oak wood flour and American walnut wood flour, waste from the carpentry workshop, not contaminated with other types of wood.
- Chemically treated oak wood flour.
- Field horsetail, dried and powdered leaves and stems, harvested from rural areas (own cultivation, Krakow, Poland).
- Powdered field horsetail subjected to pyrolytic treatment (own cultivation, Krakow, Poland).
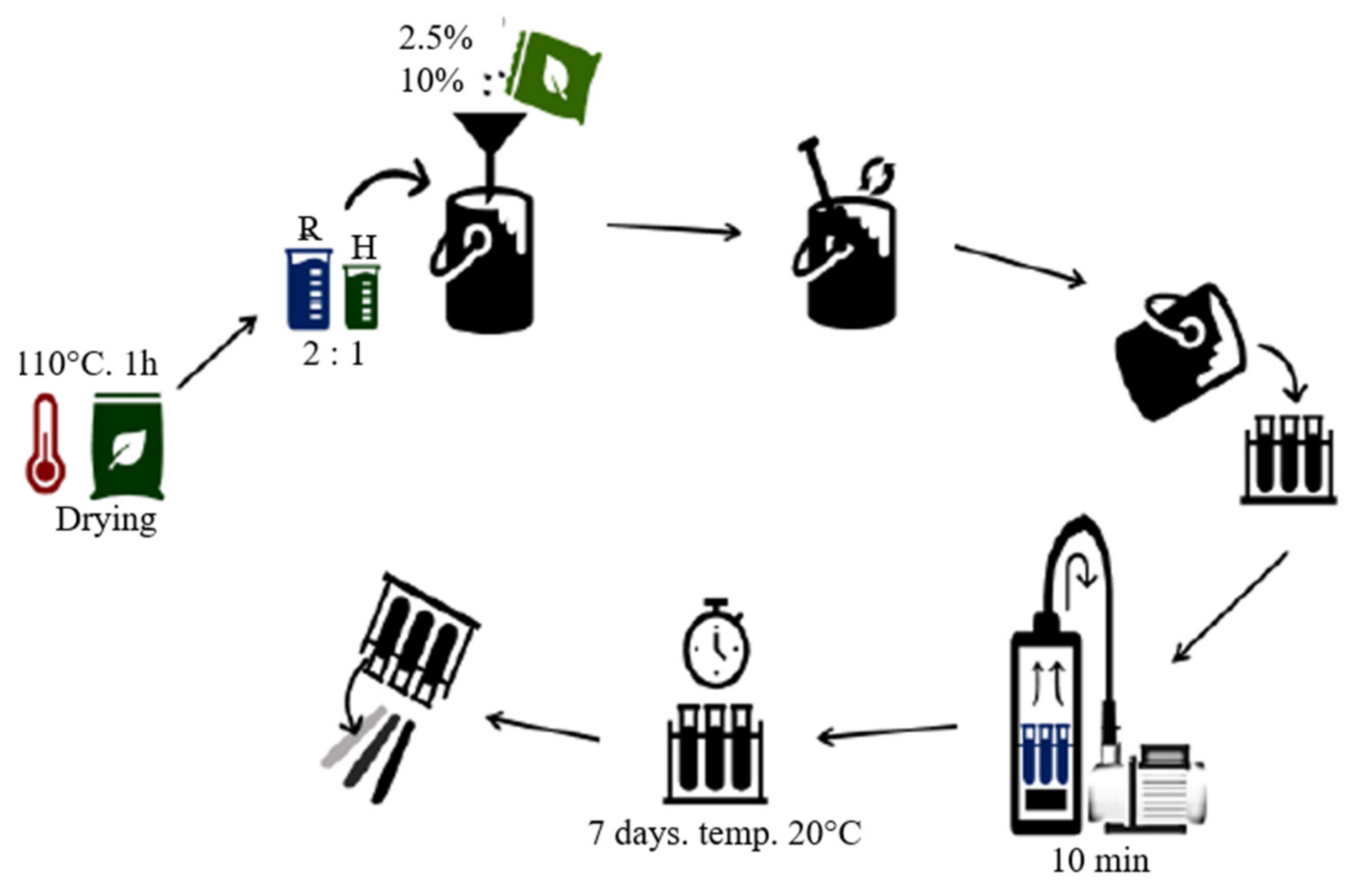
2.2. Production
3. Results and Discussion
3.1. Characteristics of Natural Additives Based on the Microstructure and EDS Research
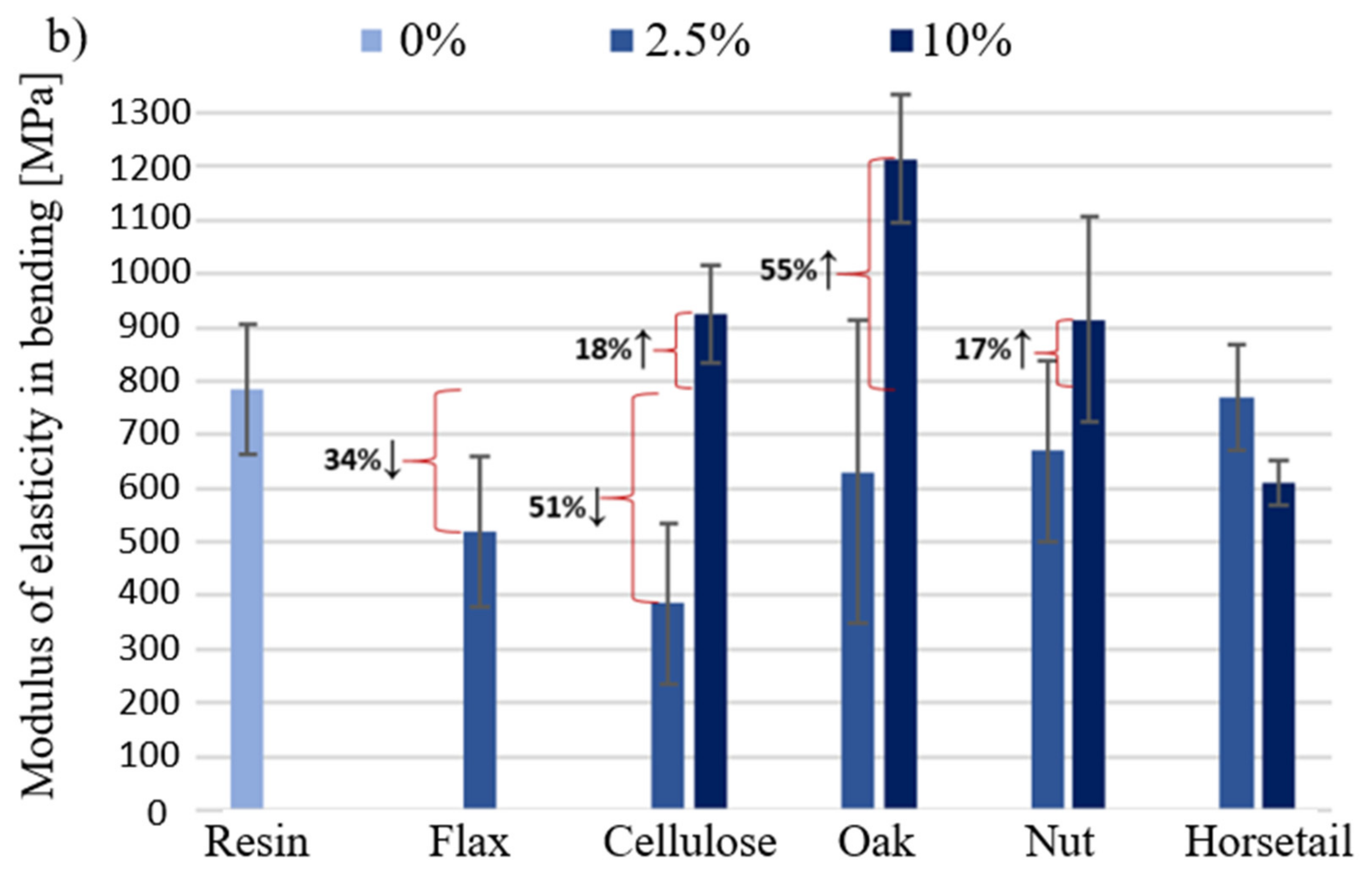
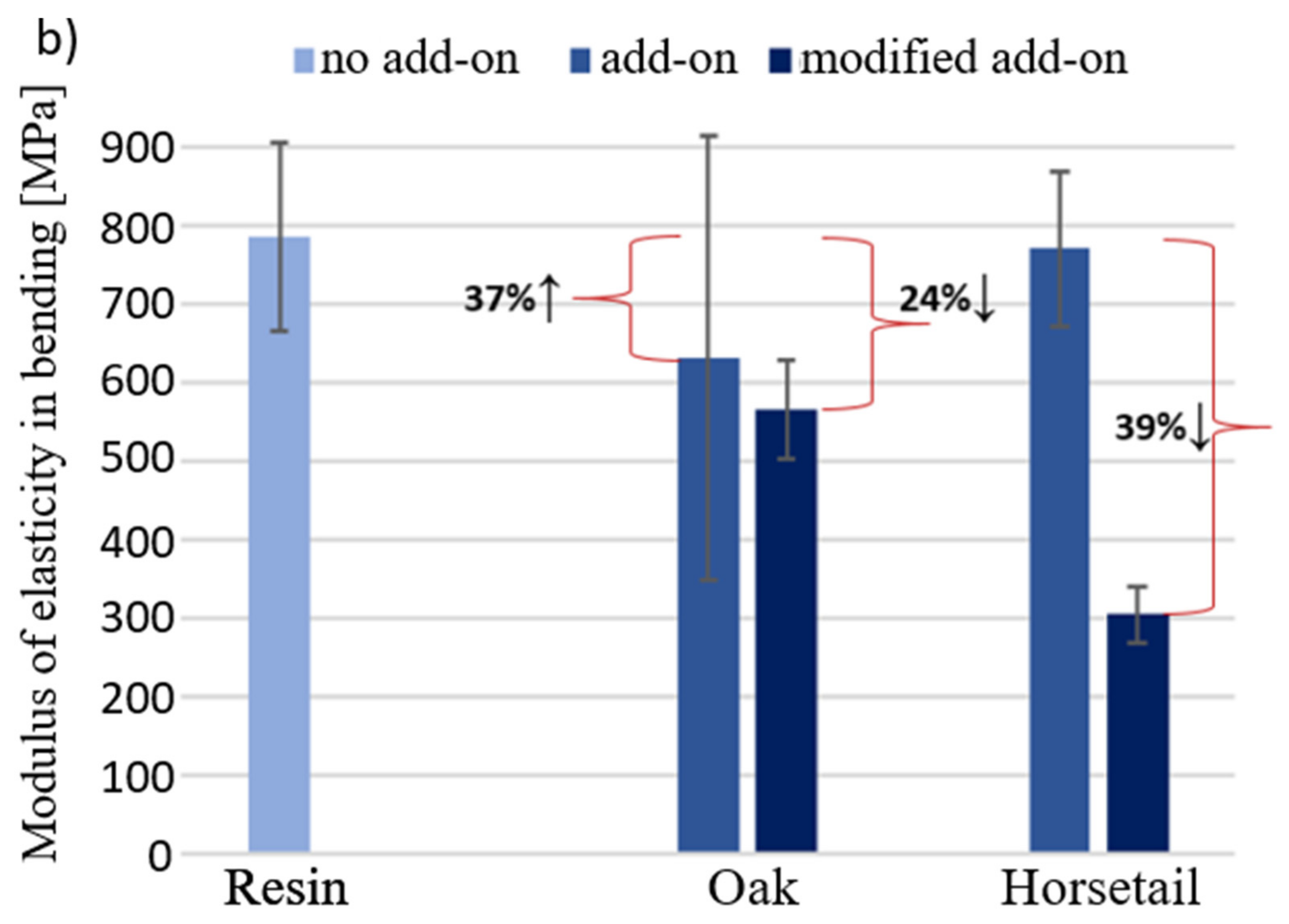
3.2. Flexural Strength and Modulus of Elasticity in Bending
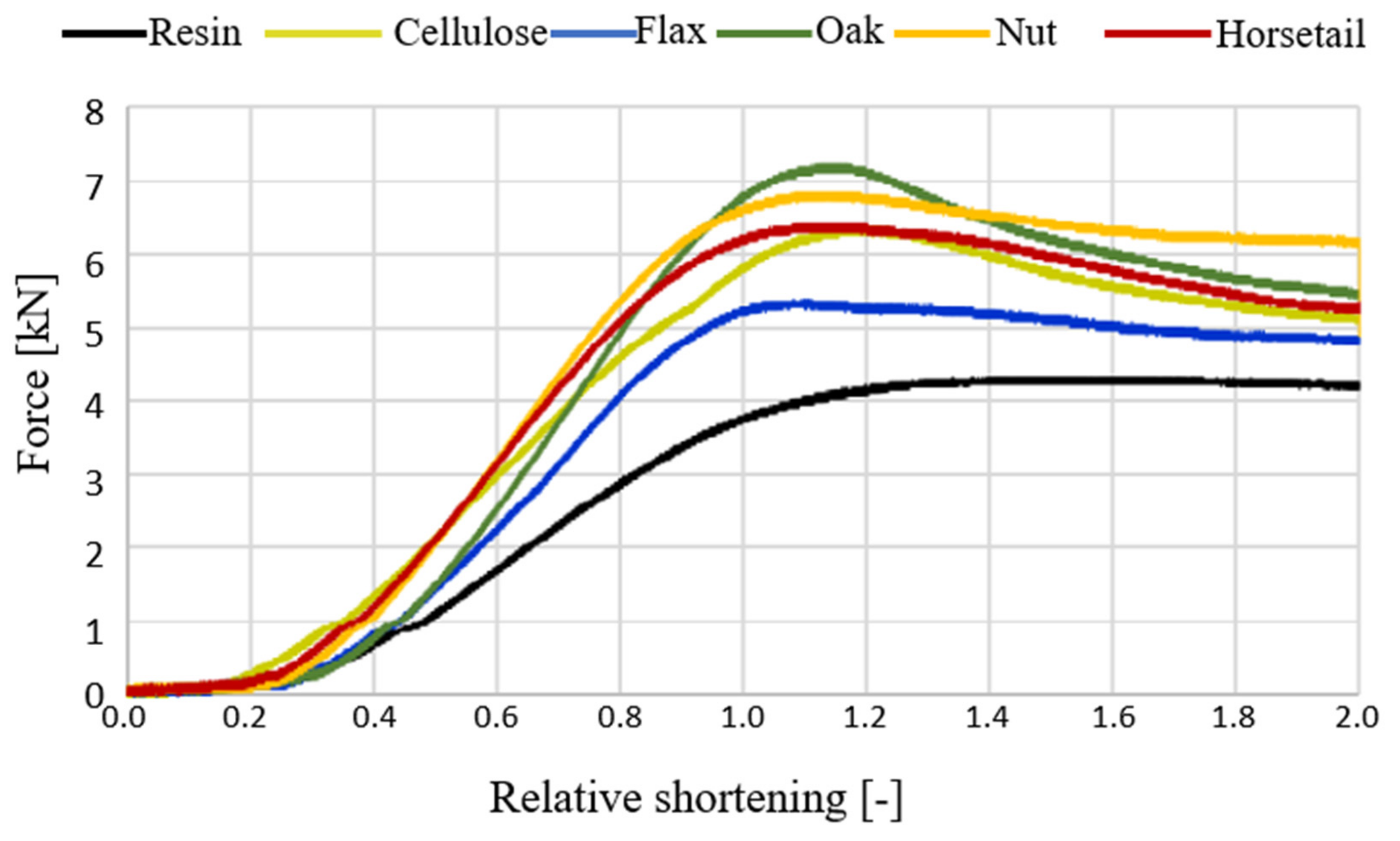
3.3. Characteristics of Composites Behavior under Compressive Force
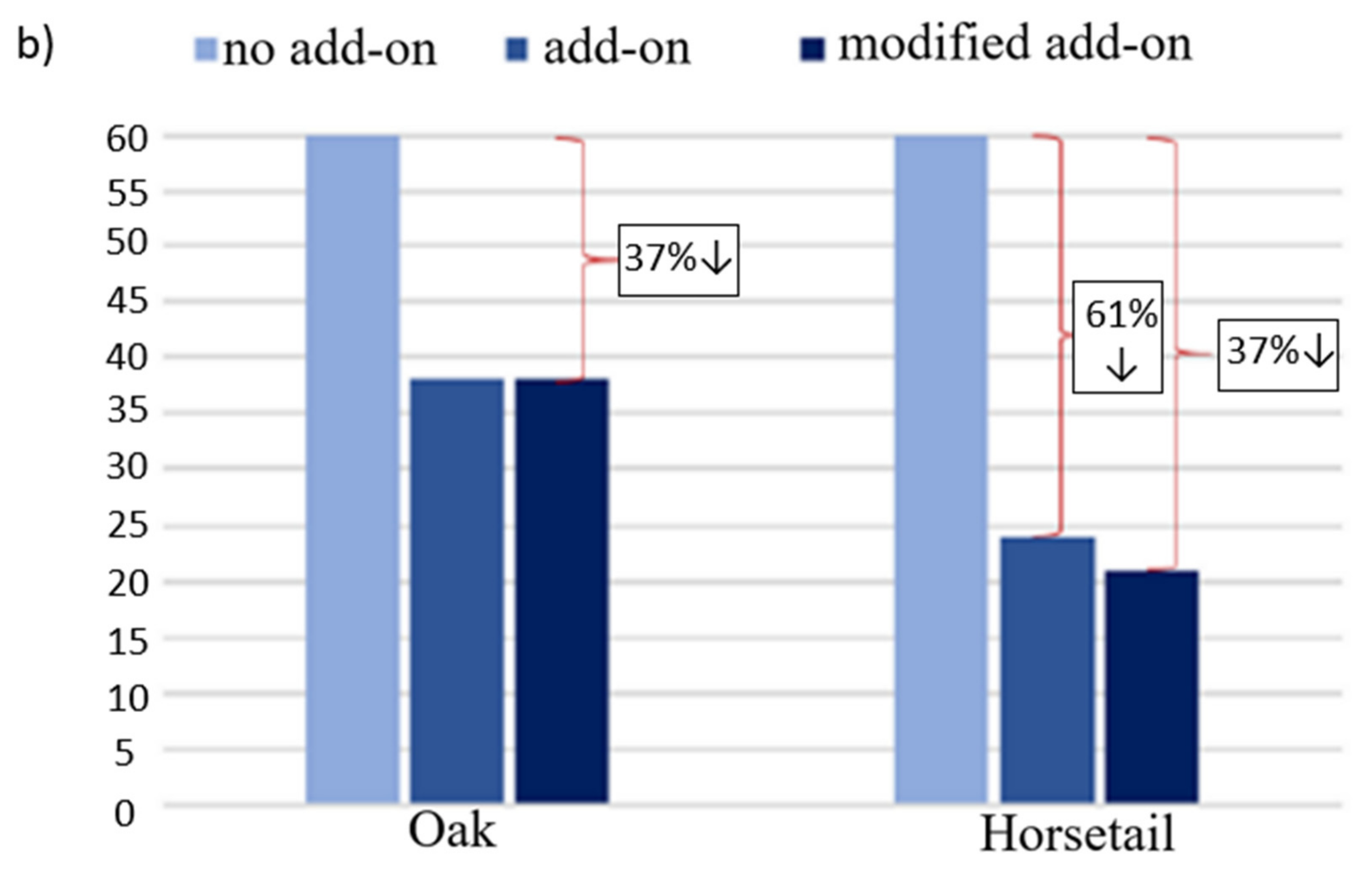
3.4. Strength under Dynamic Loads
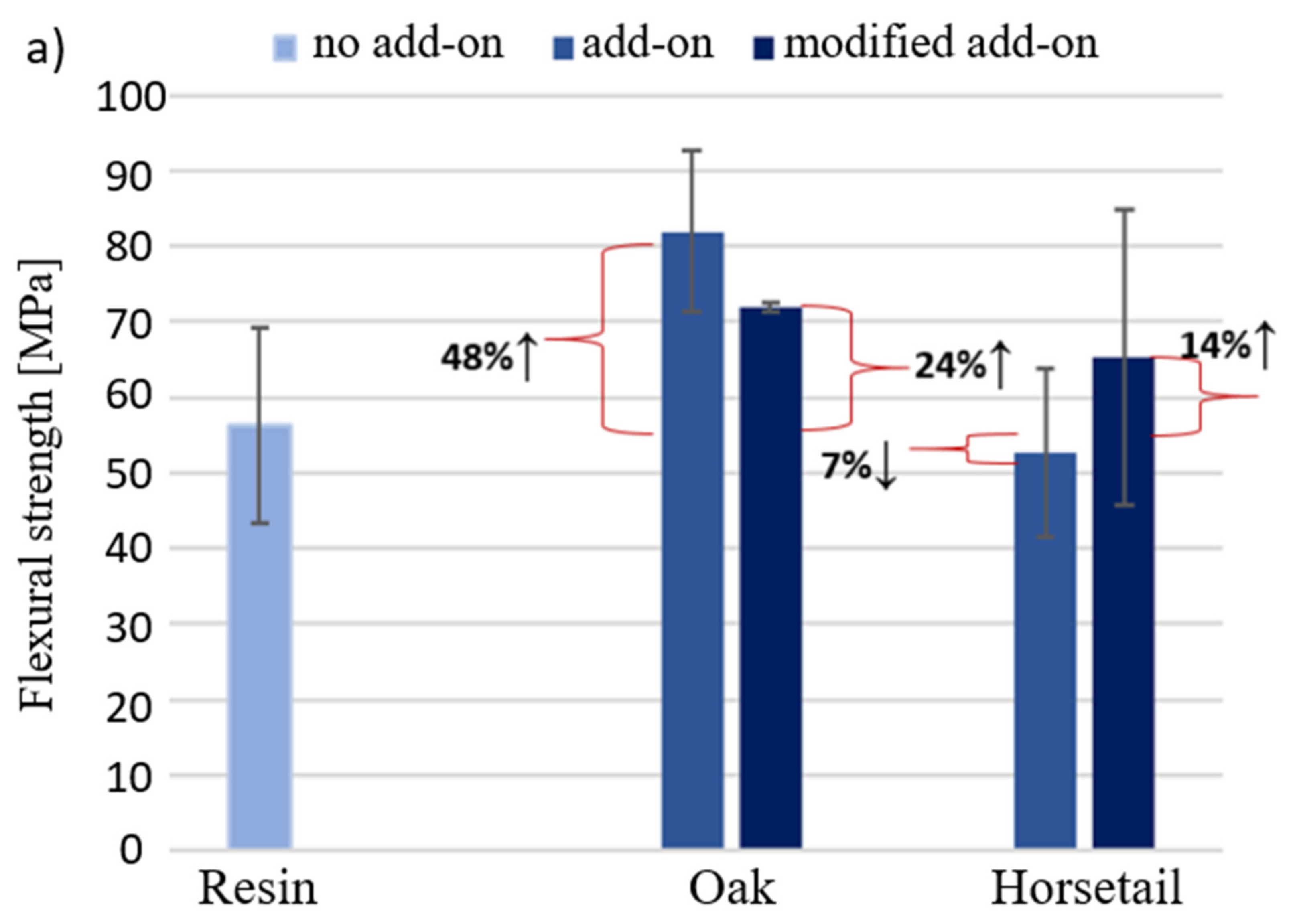
3.5. Results of Mechanical Tests for Samples with Modified Additives
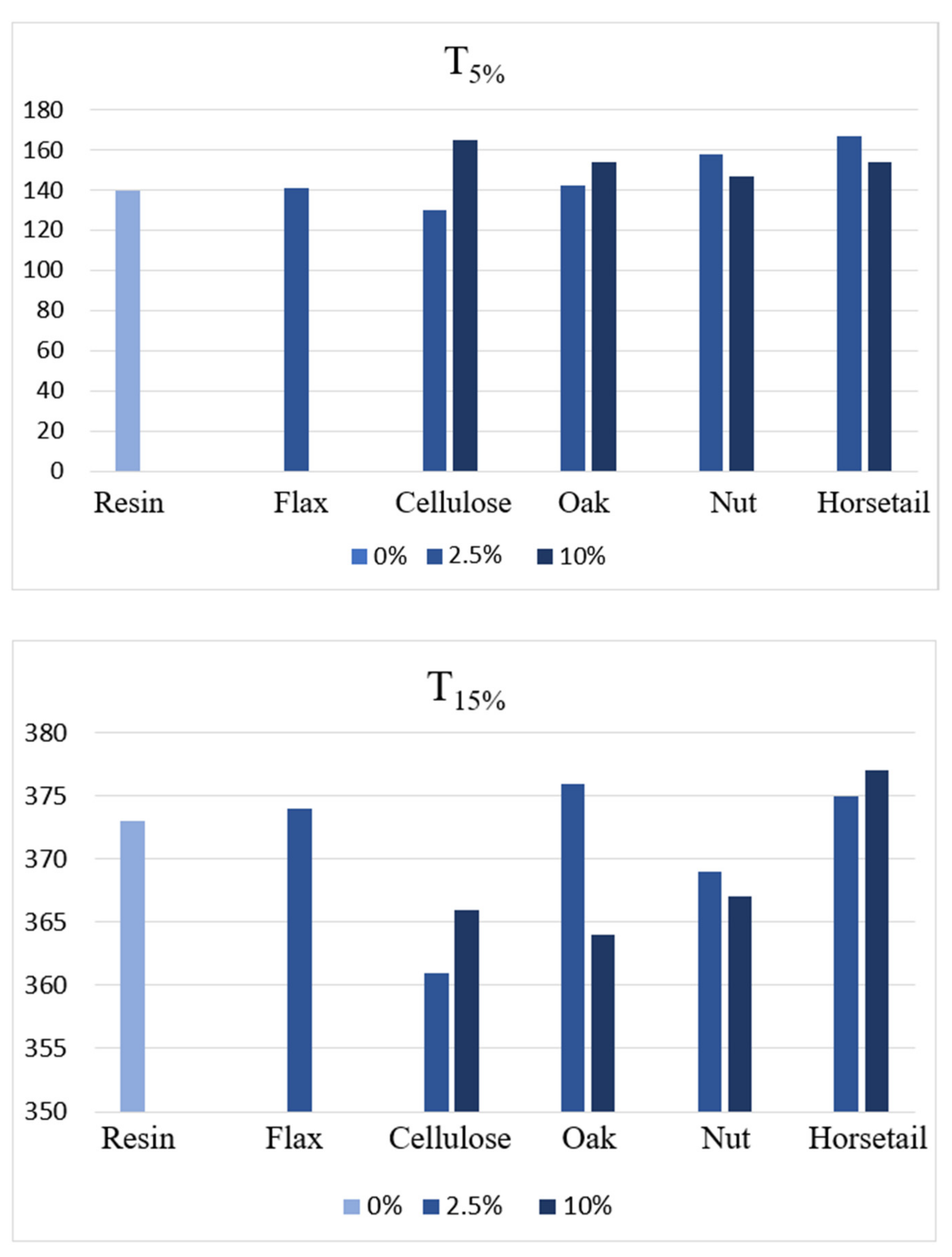
3.6. Thermal Analysis-Thermal Stability and Mechanism of Thermal Degradation
3.7. Determination of the Density and Porosity of the Material
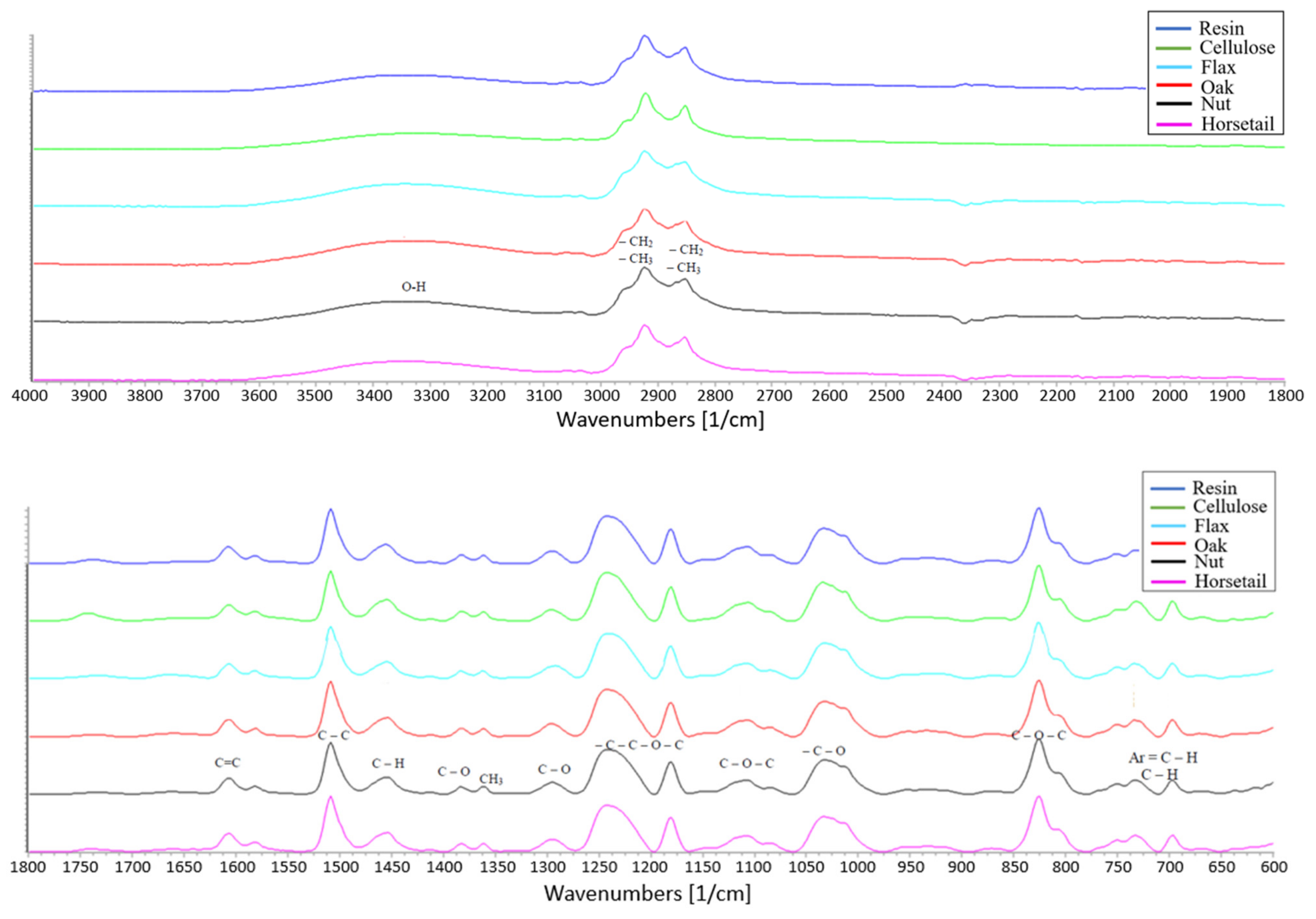
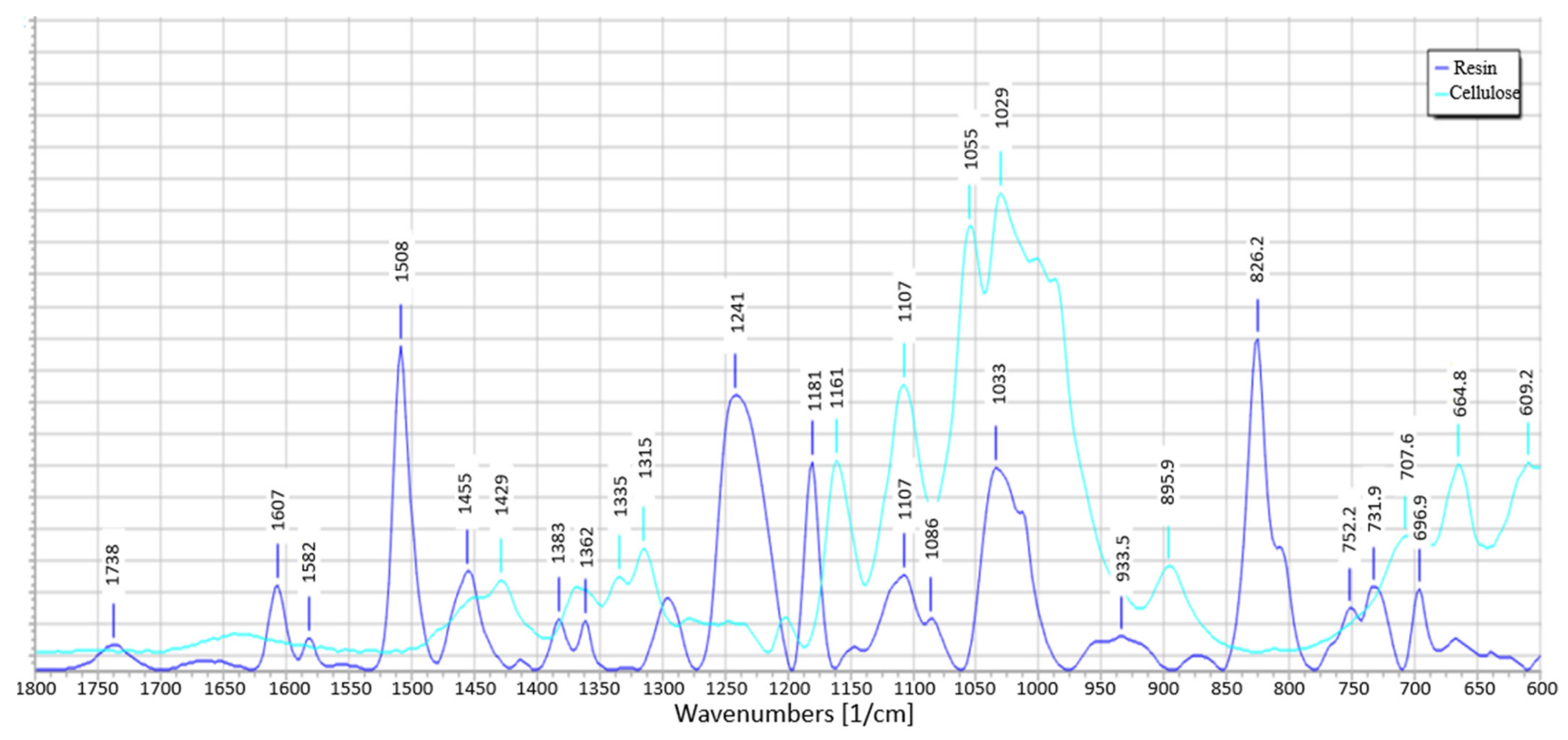
3.8. IR Spectra Analysis
4. Conclusions
- Among the natural additives selected to modify the polymer (duroplastic) matrix-microcrystalline cellulose, flax fibers, oak and American walnut wood flour and horsetail—the most favorable characteristics of the biocomposite in terms of bending and compressive strength—were obtained using unmodified oak wood flour. However, it lowered impact resistance of the composite.
- The greatest impact on thermal stability was noted for composites with horsetail, which may be the result of the presence of silica.
- Chemical modification of wood flour did not have a positive effect on the improvement of properties, although it was expected. The test results of the modified additive are not better than that of the unmodified additive. Similar conclusions were drawn from the pyrolytic modification. It allowed for a better crumbling of horsetail and its even distribution in the composite, but the results for the composite are comparable with the unmodified additive.
- Modification of the matrix with components with high cellulose content resulted in improved bending strength. The results of the research indicate that greater improvement was obtained with a lower proportion of plant additives. The introduction of plant additives to the matrix has a positive effect on the improvement of compressive strength. Even a small amount of additive (2.5% by weight of resin + hardener) significantly increased the maximum force that could be resisted by the compressed material (even up to 70% in relation to pure resin). The reinforcement is due to the fact that the particles in the matrix limit the movement of the polymer chains and, thus, the deformation of the matrix.
- The impact strength of composites reinforced with plant particles decreases with the increase in the proportion of the modifying phase. The conducted research shows that the decrease is smaller the more the modifier contains more cellulose, the smaller its fraction and the more even its distribution in the matrix. The exceptions are flax fibers, which contain the most cellulose among plant additives. This is due to the fact that they were introduced in the form of short fibers, in which the homogenization of the components resulted in the accumulation of a significant amount of air, and the production method used turned out to be insufficiently effective for its removal.
- The advantage of introducing natural additives is that it will reduce the formation of toxic substances that are released as a result of the decomposition of the epoxy resin.
- The tests also show that such a combination of components can positively affect the mechanical properties, improving the bending and compressive strength, but lowering the impact toughness.
- The dependence of the improvement/deterioration of properties is a component of several factors, ranging from the modifying additive fraction, its uniformity in the matrix and adhesion between the components, and ending with the type of wood/plant, its chemical composition and microstructure. The next step in the development of duroplastic biocomposites with lignocellulosic additives, especially with a view to assessing the influence of cellulose on properties, could be the introduction of cellulose nanoforms as modifiers. As numerous studies show, even a small addition of nanofibers or cellulose nanocrystals can significantly affect the characteristics of the material.
- The most significant impact on thermal stability was noted for composites with horsetail, which may be the result of the presence of silica.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Correction Statement
References
- Czaja, K.; Korach, Ł.; Barton, J.; Sacher-Majewska, B.; Niemczyk, A. Kompozyty, biokompozyty i nanokompozyty polimerowe. Otrzymywanie, skład, właściwości i kierunki zastosowań. Chemik 2014, 68, 280–287. [Google Scholar]
- Faruk, O.; Bledzki, A.K.; Fink, H.P.; Sain, M. Biocomposites reinforced with natural fibers: 2000–2010. Prog. Polym. Sci. 2012, 37, 1552–1596. [Google Scholar] [CrossRef]
- Zaini, E.S.; Azaman, M.D.; Jamali, M.S.; Ismail, K.A. Synthesis and characterization of natural fiber reinforced polymer composites as core for honeycomb core structure: A review. J. Sandw. Struct. Mater. 2020, 22, 525–550. [Google Scholar] [CrossRef]
- Jeyapragash, R.; Srinivasan, V.; Sathiyamurthy, S. Mechanical properties of natural fiber/particulate reinforced epoxy composites—A review of the literature. Mater. Today Proc. 2020, 22, 1223–1227. [Google Scholar] [CrossRef]
- Brosius, D. Natural Fiber Composites Slowly Take Root. Composites World. 2006. Available online: https://www.compositesworld.com/articles/natural-fiber-composites-slowly-take-root (accessed on 15 May 2022).
- Wu, Y.; Xia, C.; Cai, L.; Garcia, A.C.; Shi, S.Q. Development of natural fiber-reinforced composite with comparable mechanical properties and reduced energy consumption and environmental impacts for replacing automotive glass-fiber sheet molding compound. J. Clean. Prod. 2018, 184, 92–100. [Google Scholar] [CrossRef]
- Väisänen, T.; Das, O.; Tomppo, L. A review on new bio-based constituents for natural fiber-polymer composites. J. Clean. Prod. 2017, 149, 582–596. [Google Scholar] [CrossRef]
- Urbaniak, M. Biokompozyty epoksydowe wzmacniane naturalnymi włóknami krótkimi oraz mikrowłóknami z łusek zbożowych. Inżynieria Mater. 2015, 1, 5–9. [Google Scholar] [CrossRef]
- Alshammari, B.A.; Alotaibi, M.D.; Alothman, O.Y.; Sanjay, M.R.; Kian, L.K.; Almutairi, Z.; Jawaid, M. A New Study on Characterization and Properties of Natural Fibers Obtained from Olive Tree (Olea europaea L.) Residues. J. Polym. Environ. 2019, 27, 2334–2340. [Google Scholar] [CrossRef]
- Jagadeesh, P.; Puttegowda, M.; Rangappa, S.M.; Siengchin, S. A review on extraction, chemical treatment, characterization of natural fibers and its composites for potential applications. Polym. Compos. 2021, 42, 6239–6264. [Google Scholar] [CrossRef]
- Moghaddam, M.K.; Mortazavi, S.M. Physical and Chemical Properties of Natural Fibers Extracted from Typha Australis Leaves. J. Nat. Fibers 2016, 13, 353–361. [Google Scholar] [CrossRef]
- Jain, J.; Sinha, S.; Jain, S. Compendious Characterization of Chemically Treated Natural Fiber from Pineapple Leaves for Reinforcement in Polymer Composites. J. Nat. Fibers 2021, 18, 845–856. [Google Scholar] [CrossRef]
- Reddy, N.; Yang, Y. Extraction and Characterization of Natural Cellulose Fibers from Common Milkweed Stems Narendra. Polym. Eng. Sci. 2009, 49, 2212–2217. [Google Scholar] [CrossRef]
- Manimaran, P.; Prithiviraj, M.; Saravanakumar, S.S.; Arthanarieswaran, V.P.; Senthamaraikannan, P. Physicochemical, tensile, and thermal characterization of new natural cellulosic fibers from the stems of Sida cordifolia. J. Nat. Fibers 2018, 15, 860–869. [Google Scholar] [CrossRef]
- Mathias, J.D.; Alzina, A.; Grédiac, M.; Michaud, P.; Roux, P.; De Baynast, H.; Delattre, C.; Dumoulin, N.; Faure, T.; Larrey-Lassalle, P.; et al. Upcycling sunflower stems as natural fibers for biocomposite applications. BioResources 2015, 10, 8076–8088. [Google Scholar] [CrossRef]
- Reddy, K.O.; Maheswari, C.U.; Shukla, M.; Song, J.I.; Rajulu, A.V. Composites: Part B Tensile and structural characterization of alkali treated Borassus fruit fine fibers. Compos. Part B 2013, 44, 433–438. [Google Scholar] [CrossRef]
- Nayak, S.; Khuntia, S. Development and study of properties of Moringa oleifera fruit fi bers/polyethylene terephthalate composites for packaging applications. Compos. Commun. 2019, 15, 113–119. [Google Scholar] [CrossRef]
- Binoj, J.S.; Raj, R.E.; Sreenivasan, V.S.; Thusnavis, G.R. Morphological, Physical, Mechanical, Chemical and Thermal Characterization of Sustainable Indian Areca Fruit Husk Fibers (Areca catechu L.) as Potential Alternate for Hazardous Synthetic Fibers. J. Bionic Eng. 2016, 13, 156–165. [Google Scholar] [CrossRef]
- Small, E.; Brookes, B. Temperature and Moisture Content for Storage Maintenance of Germination Capacity of Seeds of Industrial Hemp, Marijuana, and Ditchweed Forms of Cannabis sativa. J. Nat. Fibers 2012, 9, 240–255. [Google Scholar] [CrossRef]
- Ra, I. Effect of Date Palm Seeds on the Tribological Behaviour of Polyester Composites under Different Testing Conditions. J. Sci. Eng. 2015, 4, 1000206. [Google Scholar] [CrossRef]
- Błędzki, A.K.; Jaszkiewicz, A.; Urbaniak, M.; Stankowska-Walczak, D. Biocomposites in the Past and in the Future. Fibres Text. East. Eur. 2012, 20, 15–22. [Google Scholar]
- Hasan, K.M.F.; Horváth, P.G.; Alpár, T. Potential natural fiber polymeric nanobiocomposites: A review. Polymers 2020, 12, 1072. [Google Scholar] [CrossRef] [PubMed]
- Deo, C.; Acharya, S.K. Effect of moisture absorption on mechanical properties of chopped natural fiber reinforced epoxy composite. J. Reinf. Plast. Compos. 2010, 29, 2513–2521. [Google Scholar] [CrossRef]
- Devaraju, A.; Sivasamy, P. Comparative Analysis of Mechanical Characteristics of Sisal Fibre Composite with and without Nano Particles. Mater. Today Proc. 2018, 5, 14362–14366. [Google Scholar] [CrossRef]
- Velusamy, K.; Navaneethakrishnan, P.; Rajeshkumar, G.; Sathishkumar, T.P. The influence of fiber content and length on mechanical and water absorption properties of Calotropis Gigantea fiber reinforced epoxy composites. J. Ind. Text. 2019, 48, 1274–1290. [Google Scholar] [CrossRef]
- Peltola, P. Alternative fibre sources: Paper and wood fibres as reinforcement. In Green Composites: Polymer Composites and the Environment; Woodhead Publishing Series in Composites Science and Engineering; Woodhead Publishing: Sawston, UK, 2004; Volume 5, pp. 81–99. [Google Scholar] [CrossRef]
- Waliszewska, B.; Prądzyński, W.; Zborowska, M.; Stachowiak-Wencek, A.; Waliszewska, H.; Małysko, E. Chemical composition of black walnut wood. For. Wood Technol. 2015, 91, 179–181. [Google Scholar]
- George, J.; Ivens, J.; Verpoest, I. Mechanical properties of flax fibre reinforced epoxy compositesa. Die Angew. Makromol. Chem. 1999, 272, 41–45. [Google Scholar] [CrossRef]
- Bledzki, A.K.; Gassan, J. Composites reinforced with cellulose based fibres. Prog. Polym. Sci. 1999, 24, 221–274. [Google Scholar] [CrossRef]
- Boopalan, M.; Niranjanaa, M.; Umapathy, M.J. Study on the mechanical properties and thermal properties of jute and banana fiber reinforced epoxy hybrid composites. Compos. Part B Eng. 2013, 51, 54–57. [Google Scholar] [CrossRef]
- Soykeabkaew, N.; Arimoto, N.; Nishino, T.; Peijs, T. All-cellulose composites by surface selective dissolution of aligned ligno-cellulosic fibres. Compos. Sci. Technol. 2008, 68, 2201–2207. [Google Scholar] [CrossRef]
- Barton, J.; Niemczyk, A.; Czaja, K.; Korach, Ł.; Sacher-Majewska, B. Kompozyty, biokompozyty i nanokompozyty i kierunki zastosowań. Chemik 2014, 68, 4–7. [Google Scholar]
- Sgriccia, N.; Hawley, M.C.; Misra, M. Characterization of natural fiber surfaces and natural fiber composites. Compos. Part A Appl. Sci. Manuf. 2008, 39, 1632–1637. [Google Scholar] [CrossRef]
- Georgopoulos, S.T.; Tarantili, P.A.; Avgerinos, E.; Andreopoulos, A.G.; Koukios, E.G. Thermoplastic polymers reinforced with fibrous agricultural residues. Polym. Degrad. Stab. 2005, 90, 303–312. [Google Scholar] [CrossRef]
- Li, X.; Tabil, L.G.; Panigrahi, S. Chemical treatments of natural fiber for use in natural fiber-reinforced composites: A review. J. Polym. Environ. 2007, 15, 25–33. [Google Scholar] [CrossRef]
- Xie, Y.; Hill, C.A.S.; Xiao, Z.; Militz, H.; Mai, C. Silane coupling agents used for natural fiber/polymer composites: A review. Compos. Part A Appl. Sci. Manuf. 2010, 41, 806–819. [Google Scholar] [CrossRef]
- George, J.; Sreekala, M.S.; Thomas, S. A review on interface modification and characterization of natural fiber reinforced plastic composites. Polym. Eng. Sci. 2001, 41, 1471–1485. [Google Scholar] [CrossRef]
- Pach, J.; Kaczmar, J.W. Wpływ chemicznej modyfikacji włókien konopnych na wybrane właściwości mechaniczne kompozytów na osnowie polipropylenowej. Polimery 2011, 56, 385–389. [Google Scholar]
- Sarikaya, E.; Çallioğlu, H.; Demirel, H. Production of epoxy composites reinforced by different natural fibers and their mechanical properties. Compos. Part B Eng. 2019, 167, 461–466. [Google Scholar] [CrossRef]
- Mukherjee, P.S.; Satyanarayana, K.G. Structure and properties of some vegetable fibres—Part 1 Sisal fibre. J. Mater. Sci. 1984, 19, 3925–3934. [Google Scholar] [CrossRef]
















| Addition/Weight Fraction | Cellulose | Flax | Oak | Oak * | Nut | Horsetail | Horsetail * |
|---|---|---|---|---|---|---|---|
| 2.50% | + | + | + | + | + | + | + |
| 10% | + | − | + | − | + | + | − |
| Relative Shortening [-] | Force F (kN) (Force Corresponding to a Specific Relative Shortening) | |||||
|---|---|---|---|---|---|---|
| Resin | Cellulose | Flax | Oak | Nut | Horsetail | |
| 0.04 | 0.7 | 1.3 | 0.8 | 0.8 | 1 | 1.3 |
| 0.06 | 1.7 | 3 | 2.3 | 2.6 | 3.2 | 3.1 |
| 0.08 | 2.9 | 4.6 | 4.1 | 4.9 | 5.4 | 5.1 |
| 0.1 | 3.8 | 5.8 | 5.1 | 6.7 | 6.6 | 6.2 |
| Change (%) to the resin at maximum force | - | 53↑ | 36↑ | 77↑ | 73↑ | 63↑ |
| Additive Content | Composite | Geometric Density (g/cm3) | Apparent Density (g/cm3) | Open Porosity (%) |
|---|---|---|---|---|
| 0% | Resin | 1.1 | 1.12 | 0.08 |
| 5% | Flax | 1.05 | 1.08 | 0.06 |
| Cellulose | 1.12 | 1.13 | 0.03 | |
| Oak | 1.09 | 1.13 | 0.06 | |
| Nut | 1.12 | 1.13 | 0.06 | |
| Horsetail | 1.13 | 1.13 | 0.00 | |
| 10% | Cellulose | 1.11 | 1.15 | 0.06 |
| Oak | 1.12 | 1.13 | 0.03 | |
| Nut | 1.11 | 1.16 | 0.06 | |
| Horsetail | 1.1 | 1.14 | 0.06 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szatkowski, P.; Szatkowska, M.; Gralewski, J.; Czechowski, L.; Kedziora, S. Biocomposites with Epoxy Resin Matrix Modified with Ingredients of Natural Origin. Materials 2022, 15, 7167. https://doi.org/10.3390/ma15207167
Szatkowski P, Szatkowska M, Gralewski J, Czechowski L, Kedziora S. Biocomposites with Epoxy Resin Matrix Modified with Ingredients of Natural Origin. Materials. 2022; 15(20):7167. https://doi.org/10.3390/ma15207167
Chicago/Turabian StyleSzatkowski, Piotr, Martyna Szatkowska, Jacek Gralewski, Leszek Czechowski, and Slawomir Kedziora. 2022. "Biocomposites with Epoxy Resin Matrix Modified with Ingredients of Natural Origin" Materials 15, no. 20: 7167. https://doi.org/10.3390/ma15207167
APA StyleSzatkowski, P., Szatkowska, M., Gralewski, J., Czechowski, L., & Kedziora, S. (2022). Biocomposites with Epoxy Resin Matrix Modified with Ingredients of Natural Origin. Materials, 15(20), 7167. https://doi.org/10.3390/ma15207167







