Recent Developments in Cassava (Manihot esculenta) Based Biocomposites and Their Potential Industrial Applications: A Comprehensive Review
Abstract
Highlights
- Recent advances in the research on cassava (Manihot esculenta) biopolymers and fibers, and their potential industrial applications, were discussed.
- Properties of starch, fibers, polymers, and composites derived from cassava were discussed.
- Efforts to enhance the properties of cassava composites were brought into focus.
- Detailed reports on macro and nano-sized cassava fibers and starch, and their fabrication as blend polymers, biocomposites, and hybrid composites, were reviewed.
Abstract
1. Introduction: Natural Fiber and Starch Biopolymer
2. Natural Fiber and Natural Fiber Reinforced Composites (NFCs)
3. Cassava (Manihot esculenta)
3.1. History of Cassava
3.2. Cassava Plant Parts
3.3. Cassava as Multipurpose Plant
3.4. Bio-Products from Cassava
4. Cassava Starch
Key Features of Cassava Starch
5. Cassava Fiber
5.1. Macro-Size Cassava Fiber
5.2. Nano-Size Cassava Fiber (Preparation and Characterization of Nanocellulose from Cassava)
6. Development of Biopolymer from Cassava Starch
6.1. Natural Fiber-Reinforced Cassava Starch Biopolymer
| Polymer Matrix | Fiber | Reference |
|---|---|---|
| Cassava starch | Green coconut fibers | [144] |
| Cassava starch | Coconut fibers | [145] |
| Cassava starch | Cassava bagasse | [147] |
| Cassava starch | Cellulose cassava bagasse nanofibrils (CBN) | [152] |
| Cassava starch | Cassava bagasse-kraft paper | [149] |
| Cassava starch | Cellulose nanocrystals from kenaf fibers | [150] |
| Native cassava starch | Cinnamon essential oil/clove essential oil/sucrose ester of fatty acids/sugar | [151] |
| Cassava starch | Kapok fiber | [153] |
| Cassava starch | Jute fiber | [153] |
| Cassava starch | Kaolinite-rich clay | [154] |
| Cassava starch | The exploitation of chitosan as a compatible malt | [155] |
| Cassava starch | Malt bagasse | [156] |
| Cassava starch | Blended with zein, gluten, soy protein, kraft fiber, and palm | [157] |
| Cassava flour (CF)/wheat flour (WF) | Cassava stillage residue (CSR) | [158] [159] |
| Poly(vinyl chloride) (PVC) | [160] | |
| Final egg albumen: Cassava starch: sunflower oil | [161] | |
| Cassava stillage residue (CSR) | Self-reinforced | [141] |
| Cassava Starch | Cassava peel/cassava bagasse | [156] |
| Cassava Starch | The remaining fibrous residue of cassava starch extraction | [162] |
| Cassava starch | Cassava nanofiber | [143] |
| Cassava starch | Cassava/sugar palm fiber | [71] |
| Cassava starch | Cassava bagasse cellulose nanofibrils | [163] |
| Cassava starch | Microcrystalline Cellulose Avicel PH101 | [164] |
| Cassava starch | Rice husk fiber | [165] |
| Cassava starch | Rice husk fiber nanocrystalline cellulose | [165] |
| Cassava starch | Cassava root | [166] |
| Cassava starch | Cassava bagasse | [166] |
| Cassava starch | Cassava bagasse lignocellulose nanofibers (LCNF)/nanoclay (Nclay) | [143] |
| Cassava starch | Cassava bagasse/kraft paper | [149] |
| Cassava starch | Waxy starch nanocrystal | [167] |
| Cassava starch | Cassava bagasse | [31] |
| Cassava starch | Zinc oxide nanofiller | [168] |
| Cassava starch | Acetobacter xylinum bacterial cellulose (BC) | [169] |
| Cassava starch | Recycled newspaper pulp fiber | [170] |
| Cassava starch | Kenaf cellulose nanocrystals (CNCs) | [148] |
| Cassava starch | Montmorillonite | [171] |
| Cassava starch | Bamboo nanofibers | [171] |
| Cassava starch | Bacterial cellulose | [172] |
| Cassava starch | ZnO/bacterial cellulose | [173] |
| Cassava starch | Carnauba wax/cashew tree gum-based films | [174] |
| Cassava starch | Concentrated natural rubber latex/cotton fiber | [175] |
| Cassava starch | Cellulose pulp fibers modified with deposition of silica (SiO2) nanoparticles | [176] |
| Cassava starch | Cellulose fiber | [177] |
| Cassava starch | Cassava bagasse (CB) | [178] |
| Cassava starch | Cassava peel (CP) | [178] |
| Cassava starch | Coconut nanocellulose | [179] |
| Cassava starch | Licuri nanocellulose | [179] |
| Cassava starch | Corn stover nanocellulose | [179] |
| Cassava starch | Pulp of eucalyptus commercial nanocellulose | [179] |
| Cassava starch | Cassava peel | [180] |
| Cassava starch | Kenaf cellulose nanocrystals | [181] |
| Cassava starch | Oregano essential oil/sugarcane bagasse | [182] |
| Cassava starch | Nanoclay | [183] |
| Cassava starch | Zein oil | [184] |
| Cassava starch | Gluten oil | [184] |
| Cassava starch | Soy protein oil | [184] |
| Cassava starch | Kraft fiber oil | [184] |
| Cassava starch | Palm oil | [184] |
| Cassava starch | Kenaf nanocrystalline cellulose | [148] |
| Cassava starch | Brazilian coconut fiber | [144] |
| Cassava starch | Eucalyptus cellulose nanocrystals | [185] |
| Cassava starch | Nanoclay | [186] |
| Cassava starch | Cassava roots bagasse | [187] |
| Cassava starch | Cellulose fiber/nanoclay | [188] |
| Cassava starch | Sugarcane bagasse fibers/montmorillonite | [189] |
| Cassava starch | Sisal cellulose nanofibers | [190] |
| Cassava starch | Banana fibers | [191] |
| Cassava starch | Pineapple shell fiber | [192] |
| Cassava starch | Soybean hulls fiber | [193] |
| Cassava starch | Soybean hulls microcrystalline cellulose | [193] |
| Cassava starch | Polylactic acid | [194] |
| Cassava starch | Ramie fibers CNF/nano PCC tapioca starch | [195] |
| Cassava starch | Zeolite | [151] |
| Cassava starch | Beidellite | [151] |
| Cassava starch | Starch nanocrystals (SNCs) | [196] |
| Cassava starch | Nanofiber straw/ZnO | [197] |
| Cassava starch | Pectin particles | [198] |
| Cassava starch | Cotton fibers | [198] |
| Cassava starch | Ramie cellulose microfibrils | [199] |
| Cassava starch | Coconut fiber nanocellulose | [200] |
| Cassava starch | Bamboo fiber, lime juice, epoxidized waste cooking oil | [201] |
| Cassava starch | Cassava nanofibril | [138] |
| Cassava starch | Carboxymethylcellulose/lactic acid bacteria | [202] |
| Cassava starch | ZnO nanorods/PVA electrospun mats/rosemary extract | [203] |
| Cassava starch | Polyaniline | [204] |
| Cassava starch | Nanosilica (SiO2) | [205] |
| Cassava starch | Montmorillonite | [206] |
| Cassava starch | Silica | [207] |
| Cassava starch | Sisal fiber | [208] |
| Cassava starch | Cassava bagasse | [209] |
| Cassava starch | Coconut residue fiber | [210] |
| Cassava starch | Cassava cellulose nanocrystals | [211] |
| Cassava starch | Grape stalks | [212] |
| Cassava starch | Carboxymethylcellulose/turmeric oil | [213] |
| Cassava starch | Sisal fiber/carnauba wax | [214] |
| Cassava starch | Coconut fibers | [215] |
| Cassava starch | Kaolinite | [216] |
6.2. Thermoplastic Cassava Starch Biopolymer Blend-Reinforced Natural Fibers
6.3. Cassava Starch Hybrid Polymer Composites
7. Cassava Fiber-Reinforced Polymer Composites
8. Potential Applications
8.1. Automotive
8.2. Packaging
8.3. Food Coating
8.4. Renewable Energy (Bioethanol)
9. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pandey, A.; Soccol, C.R.; Nigam, P.; Soccol, V.T.; Vandenberghe, L.P.S.; Mohan, R. Biotechnological potential of agro-industrial residues. II: Cassava bagasse. Bioresour. Technol. 2000, 74, 81–87. [Google Scholar] [CrossRef]
- Ilyas, R.A.; Sapuan, S.M.; Ishak, M.R. Isolation and characterization of nanocrystalline cellulose from sugar palm fibres (Arenga pinnata). Carbohydr. Polym. 2018, 181, 1038–1051. [Google Scholar] [CrossRef] [PubMed]
- Edhirej, A.; Sapuan, S.M.; Jawaid, M.; Zahari, N.I. Cassava: Its polymer, fiber, composite, and application. Polym. Compos. 2017, 38, 555–570. [Google Scholar] [CrossRef]
- Mateo, S.; Peinado, S.; Morillas-Gutiérrez, F.; La Rubia, M.D.; Moya, A.J. Nanocellulose from agricultural wastes: Products and applications—A review. Processes 2021, 9, 1594. [Google Scholar] [CrossRef]
- Abotbina, W.; Sapuan, S.M.; Sultan, M.T.H.; Alkbir, M.F.M.; Ilyas, R.A. Extraction, Characterization, and Comparison of Properties of Cassava Bagasse and Black Seed Fibers. J. Nat. fibers 2022, 1–14. [Google Scholar] [CrossRef]
- Ilyas, R.A.; Sapuan, S.M.; Ishak, M.R.; Zainudin, E.S. Development and characterization of sugar palm nanocrystalline cellulose reinforced sugar palm starch bionanocomposites. Carbohydr. Polym. 2018, 202, 186–202. [Google Scholar] [CrossRef] [PubMed]
- Ilyas, R.A.; Sapuan, S.M.; Ibrahim, R.; Abral, H.; Ishak, M.R.; Zainudin, E.S.; Atikah, M.S.N.; Mohd Nurazzi, N.; Atiqah, A.; Ansari, M.N.M.; et al. Effect of sugar palm nanofibrillated cellulose concentrations on morphological, mechanical and physical properties of biodegradable films based on agro-waste sugar palm (Arenga pinnata (Wurmb.) Merr) starch. J. Mater. Res. Technol. 2019, 8, 4819–4830. [Google Scholar] [CrossRef]
- Ilyas, R.A.; Sapuan, S.M.; Ibrahim, R.; Abral, H.; Ishak, M.R.; Zainudin, E.S.; Atiqah, A.; Atikah, M.S.N.; Syafri, E.; Asrofi, M.; et al. Thermal, Biodegradability and Water Barrier Properties of Bio-Nanocomposites Based on Plasticised Sugar Palm Starch and Nanofibrillated Celluloses from Sugar Palm Fibres. J. Biobased Mater. Bioenergy 2020, 14, 234–248. [Google Scholar] [CrossRef]
- Farhat, W.; Venditti, R.; Ayoub, A.; Prochazka, F.; Fernández-de-Alba, C.; Mignard, N.; Taha, M.; Becquart, F. Towards thermoplastic hemicellulose: Chemistry and characteristics of poly-(ε-caprolactone) grafting onto hemicellulose backbones. Mater. Des. 2018, 153, 298–307. [Google Scholar] [CrossRef]
- Haq, F.; Yu, H.; Wang, L.; Teng, L.; Haroon, M.; Khan, R.U.; Mehmood, S.; Amin, B.U.; Ullah, R.S.; Khan, A.; et al. Advances in chemical modifications of starches and their applications. Carbohydr. Res. 2019, 476, 12–35. [Google Scholar] [CrossRef]
- Famá, L.M.; Pettarin, V.; Goyanes, S.N.; Bernal, C.R. Starch/multi-walled carbon nanotubes composites with improved mechanical properties. Carbohydr. Polym. 2011, 83, 1226–1231. [Google Scholar] [CrossRef]
- Asrofi, M.; Sapuan, S.M.; Ilyas, R.A.; Ramesh, M. Characteristic of composite bioplastics from tapioca starch and sugarcane bagasse fiber: Effect of time duration of ultrasonication (Bath-Type). Mater. Today Proc. 2020, 46, 1626–1630. [Google Scholar] [CrossRef]
- Asrofi, M.; Sujito; Syafri, E.; Sapuan, S.M.; Ilyas, R.A. Improvement of Biocomposite Properties Based Tapioca Starch and Sugarcane Bagasse Cellulose Nanofibers. Key Eng. Mater. 2020, 849, 96–101. [Google Scholar] [CrossRef]
- Hazrol, M.D.; Sapuan, S.M.; Ilyas, R.A.; Othman, M.L.; Sherwani, S.F.K. Electrical properties of sugar palm nanocrystalline cellulose reinforced sugar palm starch nanocomposites. Polimery 2020, 65, 363–370. [Google Scholar] [CrossRef]
- Ilyas, R.A.; Sapuan, S.M.; Atiqah, A.; Ibrahim, R.; Abral, H.; Ishak, M.R.; Zainudin, E.S.; Nurazzi, N.M.; Atikah, M.S.N.; Ansari, M.N.M.; et al. Sugar palm (Arenga pinnata [ Wurmb.] Merr ) starch films containing sugar palm nanofibrillated cellulose as reinforcement: Water barrier properties. Polym. Compos. 2020, 41, 459–467. [Google Scholar] [CrossRef]
- Atikah, M.S.N.; Ilyas, R.A.; Sapuan, S.M.; Ishak, M.R.; Zainudin, E.S.; Ibrahim, R.; Atiqah, A.; Ansari, M.N.M.; Jumaidin, R. Degradation and physical properties of sugar palm starch/sugar palm nanofibrillated cellulose bionanocomposite. Polimery/Polymers 2019, 64, 680–689. [Google Scholar] [CrossRef]
- Rozilah, A.; Jaafar, C.N.A.; Sapuan, S.M.; Zainol, I.; Ilyas, R.A. The Effects of Silver Nanoparticles Compositions on the Mechanical, Physiochemical, Antibacterial, and Morphology Properties of Sugar Palm Starch Biocomposites for Antibacterial Coating. Polymers 2020, 12, 2605. [Google Scholar] [CrossRef]
- Sanyang, M.L.; Sapuan, S.M.; Jawaid, M.; Ishak, M.R.; Sahari, J. Effect of sugar palm-derived cellulose reinforcement on the mechanical and water barrier properties of sugar palm starch biocomposite films. BioResources 2016, 11, 4134–4145. [Google Scholar] [CrossRef]
- Madhumitha, G.; Fowsiya, J.; Mohana Roopan, S.; Thakur, V.K. Recent advances in starch–clay nanocomposites. Int. J. Polym. Anal. Charact. 2018, 23, 331–345. [Google Scholar] [CrossRef]
- Cyras, V.P.; Manfredi, L.B.; Ton-That, M.-T.; Vázquez, A. Physical and mechanical properties of thermoplastic starch/montmorillonite nanocomposite films. Carbohydr. Polym. 2008, 73, 55–63. [Google Scholar] [CrossRef]
- Wu, Q.; Shao, W.; Xia, N.; Wang, P.; Kong, F. A separable paper adhesive based on the starch―Lignin composite. Carbohydr. Polym. 2020, 229, 115488. [Google Scholar] [CrossRef] [PubMed]
- Nazrin, A.; Sapuan, S.M.; Zuhri, M.Y.M.; Ilyas, R.A.; Syafiq, R.; Sherwani, S.F.K. Nanocellulose Reinforced Thermoplastic Starch (TPS), Polylactic Acid (PLA), and Polybutylene Succinate (PBS) for Food Packaging Applications. Front. Chem. 2020, 8, 213. [Google Scholar] [CrossRef] [PubMed]
- Ilyas, R.A.; Sapuan, S.M.; Kadier, A.; Kalil, M.S.; Ibrahim, R.; Atikah, M.S.N.; Nurazzi, N.M.; Nazrin, A.; Lee, C.H.; Faiz Norrrahim, M.N.; et al. Properties and Characterization of PLA, PHA, and Other Types of Biopolymer Composites. In Advanced Processing, Properties, and Applications of Starch and Other Bio-Based Polymers; Al-Oqla, F., Sapuan, S.M., Eds.; Elsevier: Amsterdam, The Netherlands, 2020; pp. 111–138. [Google Scholar]
- Zaghloul, M.Y.; Zaghloul, M.M.Y.; Zaghloul, M.M.Y. Influence of Stress Level and Fibre Volume Fraction on Fatigue Performance of Glass Fibre-Reinforced Polyester Composites. Polymers 2022, 14, 2662. [Google Scholar] [CrossRef] [PubMed]
- Zaghloul, M.M.Y.; Zaghloul, M.Y.M.; Zaghloul, M.M.Y. Experimental and modeling analysis of mechanical-electrical behaviors of polypropylene composites filled with graphite and MWCNT fillers. Polym. Test. 2017, 63, 467–474. [Google Scholar] [CrossRef]
- Mahmoud Zaghloul, M.Y.; Yousry Zaghloul, M.M.; Yousry Zaghloul, M.M. Developments in polyester composite materials—An in-depth review on natural fibres and nano fillers. Compos. Struct. 2021, 278, 114698. [Google Scholar] [CrossRef]
- Azammi, A.M.N.; Ilyas, R.A.; Sapuan, S.M.; Ibrahim, R.; Atikah, M.S.N.; Asrofi, M.; Atiqah, A. Characterization studies of biopolymeric matrix and cellulose fibres based composites related to functionalized fibre-matrix interface. In Interfaces in Particle and Fibre Reinforced Composites; Elsevier: London, UK, 2020; pp. 29–93. ISBN 9780081026656. [Google Scholar]
- Wenger, J.; Stern, T.; Schöggl, J.-P.; van Ree, R.; De Corato, U.; De Bari, I.; Bell, G.; Stichnothe, H. Natural Fibers and Fiber-Based Materials in Biorefineries; IEA Bioenergy: Graz, Austria, 2018. [Google Scholar]
- Pickering, K.L.; Aruan Efendy, M.G. Preparation and mechanical properties of novel bio-composite made of dynamically sheet formed discontinuous harakeke and hemp fibre mat reinforced PLA composites for structural applications. Ind. Crops Prod. 2016, 84, 139–150. [Google Scholar] [CrossRef]
- Ilyas, R.A.; Sapuan, S.M.; Ishak, M.R.; Zainudin, E.S. Effect of delignification on the physical, thermal, chemical, and structural properties of sugar palm fibre. BioResources 2017, 12, 8734–8754. [Google Scholar] [CrossRef]
- Edhirej, A.; Sapuan, S.M.; Jawaid, M.; Zahari, N.I. Preparation and characterization of cassava bagasse reinforced thermoplastic cassava starch. Fibers Polym. 2017, 18, 162–171. [Google Scholar] [CrossRef]
- Trakulvichean, S.; Chaiprasert, P.; Otmakhova, J. Integrated Economic and Environmental Assessment of Biogas and Bioethanol Production from Cassava Cellulosic Waste. Waste Biomass Valorization 2017, 10, 691–700. [Google Scholar] [CrossRef]
- Rattanachomsri, U.; Tanapongpipat, S.; Eurwilaichitr, L.; Champreda, V. Simultaneous non-thermal saccharification of cassava pulp by multi-enzyme activity and ethanol fermentation by Candida tropicalis. J. Biosci. Bioeng. 2009, 108, 357. [Google Scholar] [CrossRef]
- Suwanasri, K.; Trakulvichean, S.; Grudloyma, U.; Songkasiri, W.; Commins, T.; Chaiprasert, P.; Tanticharoen, M. Biogas—Key Success Factors for Promotion in Thailand. J. Sustain. Energy Environ. 2015, 25, 30. [Google Scholar]
- Kosugi, A.; Kondo, A.; Ueda, M.; Murata, Y.; Vaithanomsat, P.; Thanapase, W.; Arai, T.; Mori, Y. Production of ethanol from cassava pulp via fermentation with a surface-engineered yeast strain displaying glucoamylase. Renew. Energy 2009, 34, 1354–1358. [Google Scholar] [CrossRef]
- Sriroth, K.; Chollakup, R.; Chotineeranat, S.; Piyachomkwan, K.; Oates, C.G. Processing of cassava waste for improved biomass utilization. Bioresour. Technol. 2000, 71, 63–69. [Google Scholar] [CrossRef]
- Virunanon, C.; Ouephanit, C.; Burapatana, V. Cassava pulp enzymatic hydrolysis process as a preliminary step in bio-alcohols production from waste starchy resources. J. Clean. Prod. 2013, 39, 273–279. [Google Scholar] [CrossRef]
- Siregar, J.P. Tensile and Flexural Properties of Arenga pinnata Filament (Ijuk Filament) Reinforced Epoxy. Ph.D. Thesis, Universiti Putra Malaysia, Selangor, MA, USA, 2005. [Google Scholar]
- Asyraf, M.R.M.; Ishak, M.R.; Sapuan, S.M.; Yidris, N.; Ilyas, R.A. Woods and composites cantilever beam: A comprehensive review of experimental and numerical creep methodologies. J. Mater. Res. Technol. 2020, 9, 6759–6776. [Google Scholar] [CrossRef]
- Asyraf, M.R.M.; Ishak, M.R.; Sapuan, S.M.; Yidris, N.; Ilyas, R.A.; Rafidah, M.; Razman, M.R. Evaluation of design and simulation of creep test rig for full-scale cross arm structure. Adv. Civ. Eng. 2020, 2020, 6980918. [Google Scholar] [CrossRef]
- Asyraf, M.R.M.; Ishak, M.R.; Sapuan, S.M.; Yidris, N.; Shahroze, R.M.; Johari, A.N.; Rafidah, M.; Ilyas, R.A. Creep test rig for cantilever beam: Fundamentals, prospects and present views. J. Mech. Eng. Sci. 2020, 14, 6869–6887. [Google Scholar] [CrossRef]
- Mohanty, A.K.; Misra, M.; Drzal, L.T. Natural Fibers, Biopolymers, and Biocomposites; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Lilholt, H.; Lawther, J.M. Natural Organic Fibers. In Comprehensive Composite Materials; Elsevier: Amsterdam, The Netherlands, 2000; pp. 303–325. [Google Scholar]
- Saheb, D.N.; Jog, J.P. Natural fiber polymer composites: A review. Adv. Polym. Technol. 1999, 18, 351–363. [Google Scholar] [CrossRef]
- Li, X.; Tabil, L.G.; Panigrahi, S. Chemical treatments of natural fiber for use in natural fiber-reinforced composites: A review. J. Polym. Environ. 2007, 15, 25–33. [Google Scholar] [CrossRef]
- Jumaidin, R.; Ilyas, R.A.; Saiful, M.; Hussin, F.; Mastura, M.T. Water Transport and Physical Properties of Sugarcane Bagasse Fibre Reinforced Thermoplastic Potato Starch Biocomposite. J. Adv. Res. Fluid Mech. Therm. Sci. 2019, 61, 273–281. [Google Scholar]
- Jumaidin, R.; Saidi, Z.A.S.; Ilyas, R.A.; Ahmad, M.N.; Wahid, M.K.; Yaakob, M.Y.; Maidin, N.A.; Rahman, M.H.A.; Osman, M.H. Characteristics of Cogon Grass Fibre Reinforced Thermoplastic Cassava Starch Biocomposite: Water Absorption and Physical Properties. J. Adv. Res. Fluid Mech. Therm. Sci. 2019, 62, 43–52. [Google Scholar]
- Jumaidin, R.; Khiruddin, M.A.A.; Asyul Sutan Saidi, Z.; Salit, M.S.; Ilyas, R.A. Effect of cogon grass fibre on the thermal, mechanical and biodegradation properties of thermoplastic cassava starch biocomposite. Int. J. Biol. Macromol. 2020, 146, 746–755. [Google Scholar] [CrossRef] [PubMed]
- Aziz, S.H.; Ansell, M.P. The effect of alkalization and fibre alignment on the mechanical and thermal properties of kenaf and hemp bast fibre composites: Part 1—Polyester resin matrix. Compos. Sci. Technol. 2004, 64, 1219–1230. [Google Scholar] [CrossRef]
- Mohd Nurazzi, N.; Khalina, A.; Chandrasekar, M.; Aisyah, H.A.; Ayu Rafiqah, S.; Ilyas, R.A.; Hanafee, Z.M. Effect of fiber orientation and fiber loading on the mechanical and thermal properties of sugar palm yarn fiber reinforced unsaturated polyester resin composites. Polimery 2020, 65, 115–124. [Google Scholar] [CrossRef]
- Nurazzi, N.M.; Khalina, A.; Sapuan, S.; Ilyas, R.; Rafiqah, S.A.; Hanafee, Z. Thermal properties of treated sugar palm yarn/glass fiber reinforced unsaturated polyester hybrid composites. J. Mater. Res. Technol. 2019, 9, 1606–1618. [Google Scholar] [CrossRef]
- Nurazzi, N.; Khalina, K.; Sapuan, S. Mechanical properties of sugar palm yarn/woven glass fiber reinforced unsaturated polyester composites: Effect of fiber loadings and alkaline treatment. Polimery 2019, 64, 665–675. [Google Scholar] [CrossRef]
- Huda, M.S.; Drzal, L.T.; Mohanty, A.K.; Misra, M. Chopped glass and recycled newspaper as reinforcement fibers in injection molded poly(lactic acid) (PLA) composites: A comparative study. Compos. Sci. Technol. 2006, 66, 1813–1824. [Google Scholar] [CrossRef]
- Wambua, P.; Ivens, J.; Verpoest, I. Natural fibres: Can they replace glass in fibre reinforced plastics? Compos. Sci. Technol. 2003, 63, 1259–1264. [Google Scholar] [CrossRef]
- Nishino, T.; Hirao, K.; Kotera, M.; Nakamae, K.; Inagaki, H. Kenaf reinforced biodegradable composite. Compos. Sci. Technol. 2003, 63, 1281–1286. [Google Scholar] [CrossRef]
- Kozlowski, R.; Mieleniak, B.; Helwig, M.; Przepiera, A. Flame resistant lignocellulosic-mineral composite particleboards. Polym. Degrad. Stab. 1999, 64, 523–528. [Google Scholar] [CrossRef]
- Avérous, L.; Boquillon, N. Biocomposites based on plasticized starch: Thermal and mechanical behaviour. Carbohydr. Polym. 2004, 56, 111–122. [Google Scholar] [CrossRef]
- Abotbina, W.; Sapuan, S.M.; Sultan, M.T.H.; Alkbir, M.F.M.; Ilyas, R.A. Development and Characterization of Cornstarch-Based Bioplastics Packaging Film Using a Combination of Different Plasticizers. Polymers 2021, 13, 3487. [Google Scholar] [CrossRef] [PubMed]
- Ilyas, R.; Sapuan, S. The Preparation Methods and Processing of Natural Fibre Bio-polymer Composites. Curr. Org. Synth. 2020, 16, 1068–1070. [Google Scholar] [CrossRef] [PubMed]
- Sapuan, S.M.; Mun, N.K.; Hambali, A.; Lok, H.Y.; Fairuz, A.M.; Ishak, M.R. Prototype expert system for material selection of polymeric composite automotive dashboard. Int. J. Phys. Sci. 2011, 6, 5988–5995. [Google Scholar] [CrossRef]
- Al-Oqla, F.M.; Salit, M.S.; Ishak, M.R.; Aziz, N.A. A novel evaluation tool for enhancing the selection of natural fibers for polymeric composites based on fiber moisture content criterion. BioResources 2015, 10, 299–312. [Google Scholar] [CrossRef]
- Faruk, O.; Bledzki, A.K.; Fink, H.-P.; Sain, M. Biocomposites reinforced with natural fibers: 2000–2010. Prog. Polym. Sci. 2012, 37, 1552–1596. [Google Scholar] [CrossRef]
- Kalia, S.; Avérous, L.; Njuguna, J.; Dufresne, A.; Cherian, B.M. Natural fibers, bio-and nanocomposites. Int. J. Polym. Sci. 2011, 2011, 735932. [Google Scholar] [CrossRef]
- Alves, C.; Ferrão, P.M.C.; Silva, A.J.; Reis, L.G.; Freitas, M.; Rodrigues, L.B.; Alves, D.E. Ecodesign of automotive components making use of natural jute fiber composites. J. Clean. Prod. 2010, 18, 313–327. [Google Scholar] [CrossRef]
- Beckermann, G.W.; Pickering, K.L. Engineering and evaluation of hemp fibre reinforced polypropylene composites: Fibre treatment and matrix modification. Compos. Part A Appl. Sci. Manuf. 2008, 39, 979–988. [Google Scholar] [CrossRef]
- Bullions, T.A.; Hoffman, D.; Gillespie, R.A.; Price-O’Brien, J.; Loos, A.C. Contributions of feather fibers and various cellulose fibers to the mechanical properties of polypropylene matrix composites. Compos. Sci. Technol. 2006, 66, 102–114. [Google Scholar] [CrossRef]
- Ilyas, R.A.; Sapuan, S.M.; Ishak, M.R.; Zainudin, E.S. Water transport properties of bio-nanocomposites reinforced by sugar palm (Arenga pinnata) nanofibrillated cellulose. J. Adv. Res. Fluid Mech. Therm. Sci. J. 2018, 51, 234–246. [Google Scholar]
- Ilyas, R.A.; Sapuan, S.M.; Ishak, M.R.; Zainudin, E.S. Sugar palm nanofibrillated cellulose (Arenga pinnata (Wurmb.) Merr): Effect of cycles on their yield, physic-chemical, morphological and thermal behavior. Int. J. Biol. Macromol. 2019, 123, 379–388. [Google Scholar] [CrossRef] [PubMed]
- Ilyas, R.A.; Sapuan, S.M.; Ibrahim, R.; Abral, H.; Ishak, M.R.; Zainudin, E.S.; Asrofi, M.; Atikah, M.S.N.; Huzaifah, M.R.M.; Radzi, A.M.; et al. Sugar palm (Arenga pinnata (Wurmb.) Merr) cellulosic fibre hierarchy: A comprehensive approach from macro to nano scale. J. Mater. Res. Technol. 2019, 8, 2753–2766. [Google Scholar] [CrossRef]
- Ilyas, R.A.; Sapuan, S.M.; Ishak, M.R.; Zainudin, E.S. Sugar palm nanocrystalline cellulose reinforced sugar palm starch composite: Degradation and water-barrier properties. IOP Conf. Ser. Mater. Sci. Eng. 2018, 368, 012006. [Google Scholar] [CrossRef]
- Edhirej, A.; Sapuan, S.M.; Jawaid, M.; Zahari, N.I. Cassava/sugar palm fiber reinforced cassava starch hybrid composites: Physical, thermal and structural properties. Int. J. Biol. Macromol. 2017, 101, 75–83. [Google Scholar] [CrossRef]
- Halimatul, M.J.; Sapuan, S.M.; Jawaid, M.; Ishak, M.R.; Ilyas, R.A. Water absorption and water solubility properties of sago starch biopolymer composite films filled with sugar palm particles. Polimery 2019, 64, 27–35. [Google Scholar] [CrossRef]
- Halimatul, M.J.; Sapuan, S.M.; Jawaid, M.; Ishak, M.R.; Ilyas, R.A. Effect of sago starch and plasticizer content on the properties of thermoplastic films: Mechanical testing and cyclic soaking-drying. Polimery 2019, 64, 32–41. [Google Scholar] [CrossRef]
- Syafri, E.; Sudirman; Mashadi; Yulianti, E.; Deswita; Asrofi, M.; Abral, H.; Sapuan, S.M.; Ilyas, R.A.; Fudholi, A. Effect of sonication time on the thermal stability, moisture absorption, and biodegradation of water hyacinth (Eichhornia crassipes) nanocellulose-filled bengkuang (Pachyrhizus erosus) starch biocomposites. J. Mater. Res. Technol. 2019, 8, 6223–6231. [Google Scholar] [CrossRef]
- Ilyas, R.A.; Sapuan, S.M.; Ibrahim, R.; Atikah, M.S.N.; Atiqah, A.; Ansari, M.N.M.; Norrrahim, M.N.F. Production, Processes and Modification of Nanocrystalline Cellulose from Agro-Waste: A Review. In Nanocrystalline Materials; IntechOpen: London, UK, 2019; pp. 3–32. [Google Scholar]
- Norfarhana, A.S.; Ilyas, R.A.; Ngadi, N. A review of nanocellulose adsorptive membrane as multifunctional wastewater treatment. Carbohydr. Polym. 2022, 291, 119563. [Google Scholar] [CrossRef]
- AL-Oqla, F.M.; Sapuan, S.M. Investigating the Inherent Characteristic/Performance Deterioration Interactions of Natural Fibers in Bio-Composites for Better Utilization of Resources. J. Polym. Environ. 2018, 26, 1290–1296. [Google Scholar] [CrossRef]
- Abral, H.; Basri, A.; Muhammad, F.; Fernando, Y.; Hafizulhaq, F.; Mahardika, M.; Sugiarti, E.; Sapuan, S.M.; Ilyas, R.A.; Stephane, I. A simple method for improving the properties of the sago starch films prepared by using ultrasonication treatment. Food Hydrocoll. 2019, 93, 276–283. [Google Scholar] [CrossRef]
- Abral, H.; Ariksa, J.; Mahardika, M.; Handayani, D.; Aminah, I.; Sandrawati, N.; Pratama, A.B.; Fajri, N.; Sapuan, S.M.; Ilyas, R.A. Transparent and antimicrobial cellulose film from ginger nanofiber. Food Hydrocoll. 2020, 98, 105266. [Google Scholar] [CrossRef]
- Abral, H.; Ariksa, J.; Mahardika, M.; Handayani, D.; Aminah, I.; Sandrawati, N.; Sapuan, S.M.; Ilyas, R.A. Highly transparent and antimicrobial PVA based bionanocomposites reinforced by ginger nanofiber. Polym. Test. 2020, 81, 106186. [Google Scholar] [CrossRef]
- Abral, H.; Atmajaya, A.; Mahardika, M.; Hafizulhaq, F.; Kadriadi; Handayani, D.; Sapuan, S.M.; Ilyas, R.A. Effect of ultrasonication duration of polyvinyl alcohol (PVA) gel on characterizations of PVA film. J. Mater. Res. Technol. 2020, 9, 2477–2486. [Google Scholar] [CrossRef]
- AL-Oqla, F.M.; Sapuan, M.S.; Ishak, M.R.; Abdul Aziz, N. Combined Multi-criteria Evaluation Stage Technique as an Agro Waste Evaluation Indicator for Polymeric Composites: Date Palm Fibers as a Case Study. BioResources 2014, 9, 4608–4621. [Google Scholar] [CrossRef]
- Alaaeddin, M.H.; Sapuan, S.M.; Zuhri, M.Y.M.; Zainudin, E.S.; AL- Oqla, F.M. Physical and mechanical properties of polyvinylidene fluoride—Short sugar palm fiber nanocomposites. J. Clean. Prod. 2019, 235, 473–482. [Google Scholar] [CrossRef]
- Shackelford, G.E.; Haddaway, N.R.; Usieta, H.O.; Pypers, P.; Petrovan, S.O.; Sutherland, W.J. Cassava farming practices and their agricultural and environmental impacts: A systematic map protocol. Environ. Evid. 2018, 7, 30. [Google Scholar] [CrossRef]
- TitleHillocks, R.J.; Thresh, J.M.; Bellotti, A. Cassava: Biology, Production and Utilization; CABI Publishing: Wallingford, UK, 2002. [Google Scholar]
- Parmar, A.; Sturm, B.; Hensel, O. Crops that feed the world: Production and improvement of cassava for food, feed, and industrial uses. Food Secur. 2017, 9, 907–927. [Google Scholar] [CrossRef]
- Westby, A. Cassava utilization, storage and small-scale processing. Cassava Biol. Prod. Util. 2002, 1, 281–300. [Google Scholar] [CrossRef]
- Byju, G.; Suja, G. Mineral Nutrition of Cassava, 1st ed.; Elsevier Inc.: Amsterdam, The Netherlands, 2020; Volume 159, ISBN 9780128204597. [Google Scholar]
- Jarvis, A.; Ramirez-Villegas, J.; Campo, B.V.H.; Navarro-Racines, C. Is Cassava the Answer to African Climate Change Adaptation? Trop. Plant Biol. 2012, 5, 9–29. [Google Scholar] [CrossRef]
- Eke-Okoro, O.N.; Njoku, D.N.; Madu, A.; Ezulike, T.O. Impact of global warming and crop factors on the growth and productivity of four cassava (Manihot esculenta Crantz) cultivars in Nigeria. Sci. Res. Essays 2009, 4, 955–960. [Google Scholar]
- Ding, Z.; Zhang, Y.; Xiao, Y.; Liu, F.; Wang, M.; Zhu, X.; Liu, P.; Sun, Q.; Wang, W.; Peng, M.; et al. Transcriptome response of cassava leaves under natural shade. Sci. Rep. 2016, 6, 31673. [Google Scholar] [CrossRef] [PubMed]
- TitleKatz, S.H.; Weaver, W.W. Encyclopedia of Food and Culture, 1st ed.; Scribner: New York, NY, USA, 2003. [Google Scholar]
- Aregheore, E.; Toxicology, O.A. The toxic effects of cassava (Manihot esculenta grantz) diets on humans: A review. Vet. Hum. Toxicol. 1991, 33, 274–275. [Google Scholar] [PubMed]
- Olsen, K.M.; Schaal, B.A. Evidence on the origin of cassava: Phylogeography of Manihot esculenta. Proc. Natl. Acad. Sci. USA 1999, 96, 5586–5591. [Google Scholar] [CrossRef] [PubMed]
- Sivamani, S.; Chandrasekaran, A.P.; Balajii, M.; Shanmugaprakash, M.; Hosseini-Bandegharaei, A.; Baskar, R. Evaluation of the potential of cassava-based residues for biofuels production. Rev. Environ. Sci. Biotechnol. 2018, 17, 553–570. [Google Scholar] [CrossRef]
- Hannah Ritchie Agricultural Production. Available online: https://ourworldindata.org/agricultural-production (accessed on 8 December 2020).
- Olasanmi, B.; Akoroda, M.O.; Okogbenin, E.; Egesi, C.; Kahya, S.S.; Onyegbule, O.; Ewa, F.; Guitierrez, J.; Ceballos, H.; Tohme, J.; et al. Bulked segregant analysis identifies molecular markers associated with early bulking in cassava (Manihot esculenta Crantz). Euphytica 2014, 195, 235–244. [Google Scholar] [CrossRef]
- Latif, S.; Müller, J. Potential of cassava leaves in human nutrition: A review. Trends Food Sci. Technol. 2015, 44, 147–158. [Google Scholar] [CrossRef]
- Zhu, W.; Lestander, T.A.; Örberg, H.; Wei, M.; Hedman, B.; Ren, J.; Xie, G.; Xiong, S. Cassava stems: A new resource to increase food and fuel production. GCB Bioenergy 2015, 7, 72–83. [Google Scholar] [CrossRef]
- Williams, H.; Martínez, F.R. Fallacies in the Genetic- Modification Wars, Implications for Developing Countries, and Anthropological Perspectives. Curr. Anthropol. 2002, 43, 611–630. [Google Scholar] [CrossRef]
- Zhang, M.; RONG, M.; LU, X. Fully biodegradable natural fiber composites from renewable resources: All-plant fiber composites. Compos. Sci. Technol. 2005, 65, 2514–2525. [Google Scholar] [CrossRef]
- Martín, C.; Wei, M.; Xiong, S.; Jönsson, L.J. Enhancing saccharification of cassava stems by starch hydrolysis prior to pretreatment. Ind. Crops Prod. 2017, 97, 21–31. [Google Scholar] [CrossRef]
- Djuma’ali, D.; Soewarno, N.; Sumarno, S.; Primarini, D.; Sumaryono, W. Cassava Pulp as a Biofuel Feedstock of an Enzymatic Hydrolysis Proces. Makara J. Technol. 2011, 15, 183. [Google Scholar] [CrossRef]
- Thongchul, N.; Navankasattusas, S.; Yang, S.T. Production of lactic acid and ethanol by Rhizopus oryzae integrated with cassava pulp hydrolysis. Bioprocess Biosyst. Eng. 2009, 33, 407–416. [Google Scholar] [CrossRef] [PubMed]
- Hermiati, E.; Azuma, J.; Mangunwidjaja, D.; Sunarti, T.C.; Suparno, O.; Prasetya, B. Hydrolysis of Carbohydrates in Cassava Pulp and Tapioca Flour under Microwave Irradiation. Indones. J. Chem. 2011, 11, 238–245. [Google Scholar] [CrossRef]
- Raman, N.; Pothiraj, C. Screening of Zymomonas mobilis and Saccharomyces cerevisiae strains for ethanol production from cassava waste. Rasayan J. Chem. 2008, 1, 537–541. [Google Scholar]
- Anbuselvi, S.; Balamurugan, T. Study on ethanol production from cassava leaves and pulp using S. cerevisiae. Res. J. Pharm. Biol. Chem. Sci. 2013, 4, 1755–1761. [Google Scholar]
- Wongskeo, P.; Rangsunvigit, P.; Chavadej, S. Production of glucose from the hydrolysis of cassava residue using bacteria isolates from Thai higher termites. Int. J. Chem. Mol. Eng. 2012, 6, 353–356. [Google Scholar]
- Patle, S.; Lal, B. Investigation of the potential of agro-industrial material as low cost substrate for ethanol production by using Candida tropicalis and Zymomonas mobilis. Biomass Bioenergy 2008, 32, 596–602. [Google Scholar] [CrossRef]
- Johnson, R.; Padmaja, G. Utilization of cassava fibrous residue for the production of glucose and high fructose syrup. Ind. Biotechnol. 2011, 7, 448–455. [Google Scholar] [CrossRef]
- Buddhiporn Sovorawet, J.K. Bioproduction of Ethanol in SHF and SSF from Cassava Stalks. In Biotechnology for Agro-Industrial Residues Utilisation; Springer: Dordrecht, The Netherlands, 2012; Volume 17, pp. 13–33. [Google Scholar] [CrossRef]
- Magesh, A.; Preetha, B.; Viruthagiri, T. Simultaneous Saccharification and fermentation of tapioca stem var. 226 white rose to ethanol by cellulase enzyme and Saccharomyces cerevisiae. Int. J. ChemTech Res. 2011, 3, 1821–1829. [Google Scholar]
- Han, M.; Kim, Y.; Kim, Y.; Chung, B.; Choi, G.W. Bioethanol production from optimized pretreatment of cassava stem. Korean J. Chem. Eng. 2011, 28, 119–125. [Google Scholar] [CrossRef]
- Peláez, H.C. Simultaneous Saccharification and Fermentation of Cassava Stems Sacarificación Y Fermentación Silmutánea De Tallos. Dyna 2013, 80, 97–104. [Google Scholar]
- Selvaraju Sivamania, R.B. Optimization of Bioethanol Production From Cassava Peel Using Statistical Experimental Design. Environ. Prog. Sustain. Energy 2014, 33, 676–680. [Google Scholar] [CrossRef]
- Kongkiattikajorn, J. Production of Glucoamylase from Saccharomycopsis Fibuligera sp. and Hydrolysis of Cassava Peels for Alcohol Production. Int. J. Comput. Internet Manag. 2013, 21, 1–7. [Google Scholar]
- Marx, S.; Nquma, T.Y. Cassava as feedstock for ethanol production in South Africa. Afr. J. Biotechnol. 2013, 12, 4975–4983. [Google Scholar] [CrossRef]
- Pattiya, A.; Sukkasi, S.; Goodwin, V. Fast pyrolysis of sugarcane and cassava residues in a free-fall reactor. Energy 2012, 44, 1067–1077. [Google Scholar] [CrossRef]
- Adetunji, A.; Isadare, D.; Akinluwade, K.; Adewoye, O. Waste-to-Wealth Applications of Cassava—A Review Study of Industrial and Agricultural Applications. Adv. Res. 2015, 4, 212–229. [Google Scholar] [CrossRef]
- Dahniya, M.T. An overview of cassava in Africa. Afr. Crop Sci. J. 1994, 2, 337–343. [Google Scholar]
- Jaramillo, C.M.; González Seligra, P.; Goyanes, S.; Bernal, C.; Famá, L. Biofilms based on cassava starch containing extract of yerba mate as antioxidant and plasticizer. Starch/Staerke 2015, 67, 780–789. [Google Scholar] [CrossRef]
- Padi, R.K.; Chimphango, A. Feasibility of commercial waste biorefineries for cassava starch industries: Techno-economic assessment. Bioresour. Technol. 2019, 297, 122461. [Google Scholar] [CrossRef]
- McKendry, P. Energy production from biomass (part 1): Overview of biomass. Bioresour. Technol. 2002, 83, 37–46. [Google Scholar] [CrossRef]
- Demirbas, A. Potential applications of renewable energy sources, biomass combustion problems in boiler power systems and combustion related environmental issues. Prog. Energy Combust. Sci. 2005, 31, 171–192. [Google Scholar] [CrossRef]
- Banerjee, S.; Mudliar, S.; Sen, R.; Giri, B.; Satpute, D.; Chakrabarti, T.; Pandey, R.A. Commercializing lignocellulosic bioethanol: Technology bottlenecks and possible remedies. Biofuels Bioprod. Biorefining 2010, 6, 246–256. [Google Scholar] [CrossRef]
- Wilkinson, J.; Rocha, R. The Agro-Processing Sector: Empirical Overview, Recent Trends and Development Impacts. Glob. Agro-Ind. Forum 2008, 2008, 1–59. [Google Scholar]
- Lu, Y.; Ding, Y.; Wu, Q. Simultaneous saccharification of cassava starch and fermentation of algae for biodiesel production. J. Appl. Phycol. 2011, 23, 115–121. [Google Scholar] [CrossRef]
- Avérous, L.; Pollet, E. Biodegradable Polymers. Green Energy Technol. 2012, 50, 13–39. [Google Scholar] [CrossRef]
- Glenn, G.M.; Klamczynski, A.P.; Ludvik, C.; Shey, J.; Imam, S.H.; Chiou, B.S.; McHugh, T.; DeGrandi-Hoffman, G.; Orts, W.; Wood, D.; et al. Permeability of starch gel matrices and select films to solvent vapors. J. Agric. Food Chem. 2006, 54, 3297–3304. [Google Scholar] [CrossRef]
- Gunorubon, J.; Kekpugile, K. Journal of Engineering Modification of Cassava Starch for Industrial Uses. Int. J. Eng. Technol. 2012, 2, 913–919. [Google Scholar]
- Soccol, C.R. Biotechnology products from cassava root by solid state fermentation. J. Sci. Ind. Res. 1996, 55, 358–364. [Google Scholar]
- Carta, F.S.; Soccol, C.R.; Ramos, L.P.; Fontana, J.D. Production of fumaric acid by fermentation of enzymatic hydrolysates derived from cassava bagasse. Biomass 1999, 68, 23–28. [Google Scholar] [CrossRef]
- Soccol, C.R.; Marin, B.; Raimbault, M.; Lebeault, J.M. Breeding and growth of Rhizopus in raw cassava by solid state fermentation. Appl. Microbiol. Biotechnol. 1994, 41, 330–336. [Google Scholar] [CrossRef]
- Cereda, M.P.; Takahashi, M. Cassava wastes: Their characterization and uses and treatment in Brazil. Cassava flour and starch: Progress in research and development. Int. Congr. Biotech. 1996, 271, 221–232. [Google Scholar]
- Stertz, S.C. Bioconversão da Farinha de Mandioca Crua (Manihot esculenta, Crantz) por Fungos do Gênero Rhizopus em Fermentação no Estado Sólido; Federal University of Parana (Universidade Federal do Paraná—Ufpr): Curitiba, Brazil, 1997. [Google Scholar]
- Vandenberghe, L.P.S.; Soccol, C.R.; Lebeault, J.M.; Krieger, N. Cassava wastes hydrolysate an alternative carbon source for citric acid production by Candida lipolytica. Int. Congr. Biotech 1998, 98. [Google Scholar]
- Liu, E.K.; He, W.Q.; Yan, C.R. “White revolution” to “white pollution”—Agricultural plastic film mulch in China. Environ. Res. Lett. 2014, 9, 7–10. [Google Scholar] [CrossRef]
- Huang, L.; Zhao, H.; Yi, T.; Qi, M.; Xu, H.; Mo, Q.; Huang, C.; Wang, S.; Liu, Y. Reparation and properties of cassava residue cellulose nanofibril/cassava starch composite films. Nanomaterials 2020, 10, 755. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, B.H.; De Souza Do Prado, K.; Jacinto, A.A.; Da Silva Spinacé, M.A. Influence of sugarcane bagasse fiber size on biodegradable composites of thermoplastic starch. J. Renew. Mater. 2018, 6, 176–182. [Google Scholar] [CrossRef]
- Shuzhen, N.; Liang, J.; Hui, Z.; Yongchao, Z.; Guigan, F.; Huining, X.; Hongqi, D. Enhancing hydrophobicity, strength and UV shielding capacity of starch film via novel co-cross-linking in neutral conditions. R. Soc. Open Sci. 2018, 5, 181206. [Google Scholar] [CrossRef]
- Versino, F.; López, O.V.; García, M.A. Sustainable use of cassava (Manihot esculenta) roots as raw material for biocomposites development. Ind. Crops Prod. 2015, 65, 79–89. [Google Scholar] [CrossRef]
- Kane, S.N.; Mishra, A.; Dutta, A.K. Preparation and Characterization of Cellulose and Nanocellulose from Agro-industrial Waste—Cassava Peel. J. Phys. Conf. Ser. 2017, 755, 012063. [Google Scholar] [CrossRef]
- Travalini, A.P.; Lamsal, B.; Magalhães, W.L.E.; Demiate, I.M. Cassava starch films reinforced with lignocellulose nanofibers from cassava bagasse. Int. J. Biol. Macromol. 2019, 139, 1151–1161. [Google Scholar] [CrossRef]
- Ramírez, M.G.L.; de Muniz, G.I.B.; Satyanarayana, K.G.; Tanobe, V.; Iwakiri, S. Preparation and characterization of biodegradable composites based on Brazilian cassava starch, corn starch and green coconut fibers. Matéria 2010, 15, 330–337. [Google Scholar] [CrossRef]
- Kaisangsri, N.; Kerdchoechuen, O.; Laohakunjit, N. Biodegradable foam tray from cassava starch blended with natural fiber and chitosan. Ind. Crops Prod. 2012, 37, 542–546. [Google Scholar] [CrossRef]
- Lomelí-Ramírez, M.G.; Kestur, S.G.; Manríquez-González, R.; Iwakiri, S.; de Muniz, G.B.; Flores-Sahagun, T.S. Bio-composites of cassava starch-green coconut fiber: Part II—Structure and properties. Carbohydr. Polym. 2014, 102, 576–583. [Google Scholar] [CrossRef] [PubMed]
- Vallejos, M.E.; Curvelo, A.A.S.; Teixeira, E.M.; Mendes, F.M.; Carvalho, A.J.F.; Felissia, F.E.; Area, M.C. Composite materials of thermoplastic starch and fibers from the ethanol–water fractionation of bagasse. Ind. Crops Prod. 2011, 33, 739–746. [Google Scholar] [CrossRef]
- Zainuddin, S.Y.Z.; Ahmad, I.; Kargarzadeh, H. Cassava starch biocomposites reinforced with cellulose nanocrystals from kenaf fibers. Compos. Interfaces 2013, 20, 189–199. [Google Scholar] [CrossRef]
- Matsui, K.N.; Larotonda FD, S.; Paes, S.S.; Luiz, D.; Pires AT, N.; Laurindo, J.B. Cassava bagasse-Kraft paper composites: Analysis of influence of impregnation with starch acetate on tensile strength and water absorption properties. Carbohydr. Polym. 2004, 55, 237–243. [Google Scholar] [CrossRef]
- Souza, A.C.; Goto, G.E.O.; Mainardi, J.A.; Coelho, A.C.V.; Tadini, C.C. Cassava starch composite films incorporated with cinnamon essential oil: Antimicrobial activity, microstructure, mechanical and barrier properties. LWT-Food Sci. Technol. 2013, 54, 346–352. [Google Scholar] [CrossRef]
- Belibi, P.C.; Daou, T.J.; Ndjaka, J.M.B.; Michelin, L.; Brendlé, J.; Nsom, B.; Durand, B. Tensile and water barrier properties of cassava starch composite films reinforced by synthetic zeolite and beidellite. J. Food Eng. 2013, 115, 339–346. [Google Scholar] [CrossRef]
- Gutiérrez, T.J.; Morales, N.J.; Pérez, E.; Tapia, M.S.; Famá, L. Physico-chemical properties of edible films derived from native and phosphated cush-cush yam and cassava starches. Food Packag. Shelf Life 2015, 3, 1–8. [Google Scholar] [CrossRef]
- Prachayawarakorn, J.; Chaiwatyothin, S.; Mueangta, S.; Hanchana, A. Effect of jute and kapok fibers on properties of thermoplastic cassava starch composites. Mater. Des. 2013, 47, 309–315. [Google Scholar] [CrossRef]
- Chen, G.; Liu, B.; Zhang, B. Characterization of composite hydrocolloid film based on sodium cellulose sulfate and cassava starch. J. Food Eng. 2014, 125, 105–111. [Google Scholar] [CrossRef]
- Mello, L.R.P.F.; Mali, S. Use of malt bagasse to produce biodegradable baked foams made from cassava starch. Ind. Crops Prod. 2014, 55, 187–193. [Google Scholar] [CrossRef]
- Versino, F.; García, M.A. Cassava (Manihot esculenta) starch films reinforced with natural fibrous filler. Ind. Crops Prod. 2014, 58, 305–314. [Google Scholar] [CrossRef]
- Debiagi, F.; Kobayashi, R.K.T.; Nakazato, G.; Panagio, L.A.; Mali, S. Biodegradable active packaging based on cassava bagasse, polyvinyl alcohol and essential oils. Ind. Crops Prod. 2014, 52, 664–670. [Google Scholar] [CrossRef]
- Shittu, T.A.; Raji, A.O.; Sanni, L.O. Bread from composite cassava-wheat flour: I. Effect of baking time and temperature on some physical properties of bread loaf. Food Res. Int. 2007, 40, 280–290. [Google Scholar] [CrossRef]
- Zhang, Y.; Gan, T.; Li, Q.; Su, J.; Lin, Y.; Wei, Y.; Huang, Z.; Yang, M. Mechanical and interfacial properties of poly(vinyl chloride) based composites reinforced by cassava stillage residue with different surface treatments. Appl. Surf. Sci. 2014, 314, 603–609. [Google Scholar] [CrossRef]
- Wongsasulak, S.; Yoovidhya, T.; Bhumiratana, S.; Hongsprabhas, P.; McClements, D.J.; Weiss, J. Thermo-mechanical properties of egg albumen–cassava starch composite films containing sunflower-oil droplets as influenced by moisture content. Food Res. Int. 2006, 39, 277–284. [Google Scholar] [CrossRef]
- Prachayawarakorn, J.; Pomdage, W. Effect of carrageenan on properties of biodegradable thermoplastic cassava starch/low-density polyethylene composites reinforced by cotton fibers. Mater. Des. 2014, 61, 264–269. [Google Scholar] [CrossRef]
- Belibi, P.C.; Daou, T.J.; Ndjaka, J.M.B.; Nsom, B.; Michelin, L.; Durand, B. A Comparative Study of Some Properties of Cassava and Tree Cassava Starch Films. Phys. Procedia 2014, 55, 220–226. [Google Scholar] [CrossRef]
- Teixeira, E.D.M.; Pasquini, D.; Curvelo, A.A.S.S.; Corradini, E.; Belgacem, M.N.; Dufresne, A. Cassava bagasse cellulose nanofibrils reinforced thermoplastic cassava starch. Carbohydr. Polym. 2009, 78, 422–431. [Google Scholar] [CrossRef]
- Siagian, M.; Tarigan, P. Production of Starch Based Bioplastic from Cassava Peel Reinforced with Microcrystalline Celllulose Avicel PH101 Using Sorbitol as Plasticizer. J. Phys. Conf. Ser. 2016, 710, 012012. [Google Scholar]
- Kargarzadeh, H.; Johar, N.; Ahmad, I. Starch biocomposite film reinforced by multiscale rice husk fiber. Compos. Sci. Technol. 2017, 151, 147–155. [Google Scholar] [CrossRef]
- De Morais Teixeira, E.; Da Róz, A.L.; De Carvalho, A.J.F.; Da Silva Curvelo, A.A. Preparation and characterisation of thermoplastic starches from cassava starch, cassava root and cassava bagasse. In Macromolecular Symposia; WILEY-VCH Verlag: Weinheim, Germany, 2005. [Google Scholar]
- Garcia, N.L.; Ribba, L.; Dufresne, A.; Aranguren, M.I.; Goyanes, S. Physico-Mechanical Properties of Biodegradable Starch Nanocomposites. Macromol. Mater. Eng. 2009, 294, 169–177. [Google Scholar] [CrossRef]
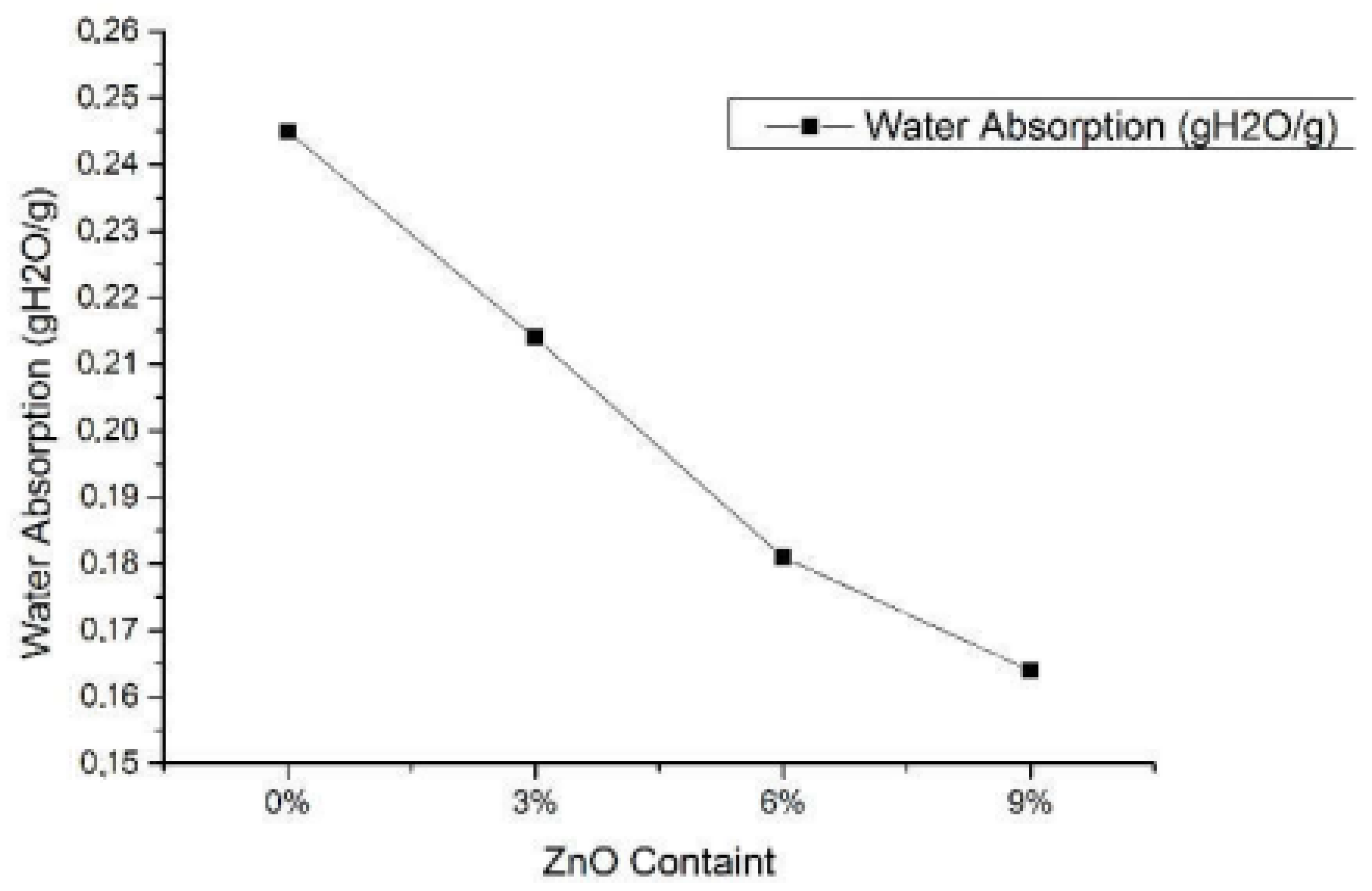
- Harunsyah; Yunus, M.; Fauzan, R. Mechanical properties of bioplastics cassava starch film with Zinc Oxide nanofiller as reinforcement. In Proceedings of the IOP Conference Series: Materials Science and Engineering, Birmingham, UK, 13–15 October 2017. [Google Scholar]
- Woehl, M.A.; Canestraro, C.D.; Mikowski, A.; Sierakowski, M.R.; Ramos, L.P.; Wypych, F. Bionanocomposites of thermoplastic starch reinforced with bacterial cellulose nanofibres: Effect of enzymatic treatment on mechanical properties. Carbohydr. Polym. 2010, 80, 866–873. [Google Scholar] [CrossRef]
- Rahmatiah Al Faruqy, M.S.; Liew, K.C. Properties of Bioplastic Sheets Made from Different Types of Starch Incorporated With Recycled Newspaper Pulp. Trans. Sci. Technol. 2016, 3, 257–264. [Google Scholar]
- Llanos, J.H.; Tadini, C.C. Preparation and characterization of bio-nanocomposite films based on cassava starch or chitosan, reinforced with montmorillonite or bamboo nanofibers. Int. J. Biol. Macromol. 2018, 107, 371–382. [Google Scholar] [CrossRef]
- Da Silva, A.; Nievola, L.M.; Tischer, C.A.; Mali, S.; Faria-Tischer, P.C. Cassava starch-based foams reinforced with bacterial cellulose. J. Appl. Polym. Sci. 2013, 130, 3043–3049. [Google Scholar] [CrossRef]
- Basuki, M.A.; Suryanto, H.; Larasati, A.; Puspitasari, P.; Mujiono. The effect of ZnO addition against crystallinity and water absorption capacity of biofoam based cassava starch reinforced bacterial cellulose. AIP Conf. Proc. 2019, 2120, 050016. [Google Scholar]
- Rodrigues, D.C.; Caceres, C.A.; Ribeiro, H.L.; de Abreu, R.F.A.; Cunha, A.P.; Azeredo, H.M.C. Influence of cassava starch and carnauba wax on physical properties of cashew tree gum-based films. Food Hydrocoll. 2014, 38, 147–151. [Google Scholar] [CrossRef]
- Sanhawong, W.; Banhalee, P.; Boonsang, S.; Kaewpirom, S. Effect of concentrated natural rubber latex on the properties and degradation behavior of cotton-fiber-reinforced cassava starch biofoam. Ind. Crops Prod. 2017, 108, 756–766. [Google Scholar] [CrossRef]
- Raabe, J.; Fonseca, A.D.S.; Bufalino, L.; Ribeiro, C.; Martins, M.A.; Marconcini, J.M.; Mendes, L.M.; Tonoli, G.H.D. Biocomposite of Cassava Starch Reinforced with Cellulose Pulp Fibers Modified with Deposition of Silica (SiO2) Nanoparticles. J. Nanomater. 2015, 2015, 1–9. [Google Scholar] [CrossRef]
- Kaisangsri, N.; Kowalski, R.J.; Kerdchoechuen, O.; Laohakunjit, N.; Ganjyal, G.M. Cellulose fiber enhances the physical characteristics of extruded biodegradable cassava starch foams. Ind. Crops Prod. 2019, 142, 111810. [Google Scholar] [CrossRef]
- Edhirej, A.; Sapuan, S.M.; Jawaid, M.; Zahari, N.I.; Sanyang, M.L. Effect of cassava peel and cassava bagasse natural fillers on mechanical properties of thermoplastic cassava starch: Comparative study. In Proceedings of the AIP Conference Proceedings, Bikaner, India, 24–25 November 2017. [Google Scholar]
- da Silva, J.B.A.; Nascimento, T.; Costa, L.A.S.; Pereira, F.V.; Machado, B.A.; Gomes, G.V.P.; Assis, D.J.; Druzian, J.I. Effect of Source and Interaction with Nanocellulose Cassava Starch, Glycerol and the Properties of Films Bionanocomposites. Mater. Today Proc. 2015, 2, 200–207. [Google Scholar] [CrossRef]
- Edhirej, A.; Sapuan, S.M.; Jawaid, M.; Ismarrubie Zahari, N. Preparation and Characterization of Cassava Starch/Peel Composite Film. Polym. Compos. 2018, 39, 1704–1715. [Google Scholar] [CrossRef]
- Zainuddin, S.Y.Z.; Ahmad, I.; Kargarzadeh, H.; Abdullah, I.; Dufresne, A. Potential of using multiscale kenaf fibers as reinforcing filler in cassava starch-kenaf biocomposites. Carbohydr. Polym. 2013, 92, 2299–2305. [Google Scholar] [CrossRef] [PubMed]
- Ketkaew, S.; Kasemsiri, P.; Hiziroglu, S.; Mongkolthanaruk, W.; Wannasutta, R.; Pongsa, U.; Chindaprasirt, P. Effect of Oregano Essential Oil Content on Properties of Green Biocomposites Based on Cassava Starch and Sugarcane Bagasse for Bioactive Packaging. J. Polym. Environ. 2017, 26, 311–318. [Google Scholar] [CrossRef]
- Wahyuningtiyas, N.E.; Suryanto, H. Properties of Cassava Starch based Bioplastic Reinforced by Nanoclay. J. Mech. Eng. Sci. Technol. 2018, 2, 20–26. [Google Scholar] [CrossRef]
- Kaisangsri, N.; Kerdchoechuen, O.; Laohakunjit, N. Characterization of cassava starch based foam blended with plant proteins, kraft fiber, and palm oil. Carbohydr. Polym. 2014, 110, 70–77. [Google Scholar] [CrossRef]
- Silva, J.B.A.; Pereira, F.V.; Druzian, J.I. Cassava Starch-Based Films Plasticized with Sucrose and Inverted Sugar and Reinforced with Cellulose Nanocrystals. J. Food Sci. 2012, 77, 14–19. [Google Scholar] [CrossRef]
- Suryanto, H.; Hutomo, P.T.; Wanjaya, R.; Puspitasari, P.; Sukarni. The stucture of bioplastic from cassava starch with nanoclay reinforcement. AIP Conf. Proc. 2016, 1778, 030027. [Google Scholar]
- Versino, F.; Garcia, M.A. Particle Size Distribution Effect on Cassava Starch and Cassava Bagasse Biocomposites. ACS Sustain. Chem. Eng. 2018, 7, 1052–1060. [Google Scholar] [CrossRef]
- El Halal, S.L.M.; Bruni, G.P.; Evangelho, J.A.D.; Biduski, B.; Silva, F.T.; Dias, A.R.G.; Zavareze, E.D.R.; Luvielmo, M.D.M. The properties of potato and cassava starch films combined with cellulose fibers and/or nanoclay. Starch-Stärke 2017, 70. [Google Scholar] [CrossRef]
- Vercelheze, A.E.S.; Oliveira, A.; Rezende, M.I.; Müller, C.; Yamashita, F.; Mali, S. Physical Properties, Photo- and Bio-degradation of Baked Foams Based on Cassava Starch, Sugarcane Bagasse Fibers and Montmorillonite. J. Polym. Environ. 2012, 21, 266–274. [Google Scholar] [CrossRef]
- Santana, J.S.; Rosário, J.M.D.; Pola, C.C.; Otoni, C.G.; FerreiraSoares, N.d.F.; Camilloto, G.P.; Cruz, R.S. Cassava starch-based nanocomposites reinforced with cellulose nanofibers extracted from sisal. J. Appl. Polym. Sci. 2016, 134. [Google Scholar] [CrossRef]
- Mo, X.; Zhong, Y.; Liang, C.; Yu, S. Studies on the properties of banana fibers-reinforced thermoplastic cassava starch composites: Preliminary results. In Advanced Materials Research; Trans Tech Publications Ltd.: Wollerau, Switzerland, 2010. [Google Scholar]
- Cabanillas, A.; Nuñez, J.; Tirado, L.J.P.C.; Vejarano, R.; Tapia-Blácido, D.R.; Arteaga, H.; Siche, R. Pineapple shell fiber as reinforcement in cassava starch foam trays. Polym. Polym. Compos. 2019, 27, 496–506. [Google Scholar] [CrossRef]
- Merci, A.; Marim, R.G.; Urbano, A.; Mali, S. Films based on cassava starch reinforced with soybean hulls or microcrystalline cellulose from soybean hulls. Food Packag. Shelf Life 2019, 20, 100321. [Google Scholar] [CrossRef]
- Ogunrinola, T.M.; Akpan, U. Production of Cassava Starch Bioplastic Film Reinforced with Poly-Lactic Acid (PLA). Int. J. Eng. Res. Adv. Technol. 2018, 4, 56–61. [Google Scholar] [CrossRef]
- Syafri, E.; Kasim, A.; Abral, H.; Sudirman; Sulungbudi, G.T.; Sanjay, M.R.; Sari, N.H. Synthesis and characterization of cellulose nanofibers (CNF) ramie reinforced cassava starch hybrid composites. Int. J. Biol. Macromol. 2018, 120, 578–586. [Google Scholar] [CrossRef]
- Dai, L.; Zhang, J.; Cheng, F. Cross-linked starch-based edible coating reinforced by starch nanocrystals and its preservation effect on graded Huangguan pears. Food Chem. 2019, 311, 125891. [Google Scholar] [CrossRef]
- Amni, C.; Marwan, M.; Mariana, M. The Making of Bioplastic from Cassava Starch Reinforced by Nano Fiber Straw and ZnO. J. Litbang Ind. 2015, 5, 91–99. [Google Scholar] [CrossRef][Green Version]
- Prachayawarakorn, J.; Pattanasin, W. Effect of pectin particles and cotton fibers on properties of thermoplastic cassava starch composites. Songklanakarin J. Sci. Technol. 2016, 38, 129–136. [Google Scholar]
- Syafri, E.; Kasim, A.; Asben, A.; Senthamaraikannan, P.; Sanjay, M. Studies on Ramie cellulose microfibrils reinforced cassava starch composite: Influence of microfibrils loading. J. Nat. Fibers 2018, 17, 122–131. [Google Scholar] [CrossRef]
- Machado, B.A.S.; Nunes, I.L.; Pereira, F.V.; Druzian, J.I. Desenvolvimento e avaliação da eficácia de filmes biodegradáveis de amido de mandioca com nanocelulose como reforço e com extrato de erva-mate como aditivo antioxidante. Ciência Rural 2012, 42, 2085–2091. [Google Scholar] [CrossRef]
- Silviana, S.; Dzulkarom, M.C. Synthesis of Cassava Bagasse Starch-Based Biocomposite Reinforced Woven Bamboo Fibre with Lime Juice as Crosslinker and Epoxidized Waste Cooking Oil (EWCO) as Bioplasticizer. J. Phys. Conf. Ser. 2019, 1295, 012076. [Google Scholar] [CrossRef]
- Li, S.; Ma, Y.; Ji, T.; Sameen, D.E.; Ahmed, S.; Qin, W.; Dai, J.; Li, S.; Liu, Y. Cassava starch/carboxymethylcellulose edible films embedded with lactic acid bacteria to extend the shelf life of banana. Carbohydr. Polym. 2020, 248, 116805. [Google Scholar] [CrossRef] [PubMed]
- Estevez-Areco, S.; Guz, L.; Candal, R.; Goyanes, S. Active bilayer films based on cassava starch incorporating ZnO nanorods and PVA electrospun mats containing rosemary extract. Food Hydrocoll. 2020, 108, 106054. [Google Scholar] [CrossRef]
- Arrieta-Almario, A.; De Sucre, U.; Mendoza-Fandiño, J.M.; Palencia, M. Composite material elaborated from conducting biopolymer cassava starch and polyaniline. Revista Mexicana de Ingeniería Química 2019, 19, 707–715. [Google Scholar] [CrossRef]
- Liu, Y.; Fan, L.; Pang, J.; Tan, D. Effect of tensile action on retrogradation of thermoplastic cassava starch/nanosilica composite. Iran. Polym. J. 2020, 29, 171–183. [Google Scholar] [CrossRef]
- Huang, L.; Han, X.; Chen, H.; An, S.; Zhao, H.; Xu, H.; Huang, C.; Wang, S.; Liu, Y. Preparation and Barrier Performance of Layer-Modified Soil-Stripping/Cassava Starch Composite Films. Polymers 2020, 12, 1611. [Google Scholar] [CrossRef]
- Liu, Y.; Fan, L.; Xiong, J.; Liang, Z. Effect of different particle size of silica on structure, morphology, and properties of thermoplastic cassava starch. Polym. Polym. Compos. 2020, 29, 863–875. [Google Scholar] [CrossRef]
- Liu, Y.; Xiong, J.; Lan, C.; Tan, D.; Liang, Z. Properties of thermoplastic cassava starch/sisal fiber composites in retrogradation. Polym. Polym. Compos. 2020, 30, 09673911221080363. [Google Scholar] [CrossRef]
- Versino, F.; Urriza, M.; García, M.A. Eco-compatible cassava starch films for fertilizer controlled-release. Int. J. Biol. Macromol. 2019, 134, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Nansu, W.; Ross, S.; Ross, G.; Mahasaranon, S. Effect of crosslinking agent on the physical and mechanical properties of a composite foam based on cassava starch and coconut residue fiber. Mater. Today Proc. 2019, 17, 2010–2019. [Google Scholar] [CrossRef]
- Huang, L.; Xu, H.; Zhao, H.; Xu, M.; Qi, M.; Yi, T.; An, S.; Zhang, X.; Li, C.; Huang, C.; et al. Properties of thermoplastic starch films reinforced with modified cellulose nanocrystals obtained from cassava residues. New J. Chem. 2019, 43, 14883–14891. [Google Scholar] [CrossRef]
- Engel, J.B.; Ambrosi, A.; Tessaro, I.C. Development of a Cassava Starch-Based Foam Incorporated with Grape Stalks Using an Experimental Design. J. Polym. Environ. 2019, 27, 2853–2866. [Google Scholar] [CrossRef]
- Mustapha, F.; Jai, J.; Sharif, Z.; Yusof, N. Cassava starch/carboxymethylcellulose biocomposite film for food paper packaging incorporated with turmeric oil. IOP Conf. Ser. Mater. Sci. Eng. 2019, 507, 012008. [Google Scholar] [CrossRef]
- Gomes, V.R.; Leite, R.H.D.L.; Júnior, M.Q.D.S.; Santos, F.K.G.; Aroucha, E.M.M. Influence of Composition on Mechanical Properties of Cassava Starch, Sisal Fiber and Carnauba Wax Biocomposites. Mater. Res. 2019, 22. [Google Scholar] [CrossRef]
- Doumbia, A.; Dable, P.J.M.R. Analysis of the Thermo-Mechanical Behavior of Composite Materials Based on Plasticized Cassava Starch Reinforced with Coconut Fibers. Indian J. Sci. Technol. 2019, 12, 1–10. [Google Scholar] [CrossRef]
- Mbey, J.A.; Hoppe, S.; Thomas, F. Cassava starch-kaolinite composite film. Effect of clay content and clay modification on film properties. Carbohydr. Polym. 2012, 88, 213–222. [Google Scholar] [CrossRef]
- Chotiprayon, P.; Chaisawad, B.; Yoksan, R. Thermoplastic cassava starch/poly(lactic acid) blend reinforced with coir fibres. Int. J. Biol. Macromol. 2020, 156, 960–968. [Google Scholar] [CrossRef]
- Phan The, D.; Debeaufort, F.; Voilley, A.; Luu, D. Biopolymer interactions affect the functional properties of edible films based on agar, cassava starch and arabinoxylan blends. J. Food Eng. 2009, 90, 548–558. [Google Scholar] [CrossRef]
- Brandelero, R.P.H.; Grossmann, M.V.; Yamashita, F. Films of starch and poly(butylene adipate co-terephthalate) added of soybean oil (SO) and Tween 80. Carbohydr. Polym. 2012, 90, 1452–1460. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, E.D.M.; Curvelo, A.A.S.; Corrêa, A.C.; Marconcini, J.M.; Glenn, G.M.; Mattoso, L.H.C. Properties of thermoplastic starch from cassava bagasse and cassava starch and their blends with poly (lactic acid). Ind. Crops Prod. 2012, 37, 61–68. [Google Scholar] [CrossRef]
- Zhang, Y.; Gan, T.; Luo, Y.; Zhao, X.; Hu, H.; Huang, Z.; Huang, A.; Qin, X. A green and efficient method for preparing acetylated cassava stillage residue and the production of all-plant fibre composites. Compos. Sci. Technol. 2014, 102, 139–144. [Google Scholar] [CrossRef]
- Guimarães, M.; Botaro, V.; Novack, K.M.; Teixeira, F.G.; Tonoli, G. Starch/PVA-based nanocomposites reinforced with bamboo nanofibrils. Ind. Crops Prod. 2015, 70, 72–83. [Google Scholar] [CrossRef]
- Mali, S.; Debiagi, F.; Grossmann, M.V.; Yamashita, F. Starch, sugarcane bagasse fibre, and polyvinyl alcohol effects on extruded foam properties: A mixture design approach. Ind. Crops Prod. 2010, 32, 353–359. [Google Scholar] [CrossRef]
- Yin, Q.; Chen, F.; Zhang, H.; Liu, C. Fabrication and characterisation of thermoplastic starch/poly(butylene succinate) blends with maleated poly(butylene succinate) as compatibiliser. Plast. Rubber Compos. 2015, 44, 362–367. [Google Scholar] [CrossRef]
- Abral, H.; Hartono, A.; Hafizulhaq, F.; Handayani, D.; Sugiarti, E.; Pradipta, O. Characterization of PVA/cassava starch biocomposites fabricated with and without sonication using bacterial cellulose fiber loadings. Carbohydr. Polym. 2018, 206, 593–601. [Google Scholar] [CrossRef]
- Moro, T.M.A.; Ascheri, J.L.R.; Ortiz, J.A.R.; Carvalho, C.W.P.; Meléndez-Arévalo, A. Bioplastics of Native Starches Reinforced with Passion Fruit Peel. Food Bioprocess Technol. 2017, 10, 1798–1808. [Google Scholar] [CrossRef]
- Guimarães, M., Jr.; Teixeira, F.G.; Tonoli, G.H.D. Effect of the nano-fibrillation of bamboo pulp on the thermal, structural, mechanical and physical properties of nanocomposites based on starch/poly(vinyl alcohol) blend. Cellulose 2018, 25, 1823–1849. [Google Scholar] [CrossRef]
- Lisdayana, N.; Fahma, F.; Sunarti, T.C.; Iriani, E.S. Thermoplastic Starch–PVA Nanocomposite Films Reinforced with Nanocellulose from Oil Palm Empty Fruit Bunches (OPEFBs): Effect of Starch Type. J. Nat. Fibers 2018, 17, 1069–1080. [Google Scholar] [CrossRef]
- Silviana, S.; Subagio, A. Biocomposite characterization of bagasse starch derived from cassava reinforced by acetylated bamboo cellulose and plasticized by epoxidized waste cooking oil. Rasayan J. Chem. 2019, 12, 1470–1477. [Google Scholar] [CrossRef]
- Subramanya, R.; Prabhakara, S. Surface Modification of Banana Fiber and its Influence on Performance of Biodegradable Banana-Cassava Starch Composites. Appl. Mech. Mater. 2019, 895, 15–20. [Google Scholar] [CrossRef]
- Yi, T.; Qi, M.; Mo, Q.; Huang, L.; Zhao, H.; Liu, D.; Xu, H.; Huang, C.; Wang, S.; Liu, Y. Ecofriendly Preparation and Characterization of a Cassava Starch/Polybutylene Adipate Terephthalate Film. Processes 2020, 8, 329. [Google Scholar] [CrossRef]
- Riyajan, S.-A.; Chantawee, K. Cassava starch composite based films for encapsulated neem: Effect of carboxylated styrene-butadiene rubber coating. Food Packag. Shelf Life 2020, 23, 100438. [Google Scholar] [CrossRef]
- Salazar-Sánchez, M.D.R.; Campo-Erazo, S.D.; Villada-Castillo, H.S.; Solanilla-Duque, J.F. Structural changes of cassava starch and polylactic acid films submitted to biodegradation process. Int. J. Biol. Macromol. 2019, 129, 442–447. [Google Scholar] [CrossRef] [PubMed]
- Aisyah, H.A.; Paridah, M.T.; Sapuan, S.M.; Khalina, A.; Berkalp, O.B.; Lee, S.H.; Lee, C.H.; Nurazzi, N.M.; Ramli, N.; Wahab, M.S.; et al. Thermal Properties of Woven Kenaf/Carbon Fibre-Reinforced Epoxy Hybrid Composite Panels. Int. J. Polym. Sci. 2019, 2019, 1–8. [Google Scholar] [CrossRef]
- Sanjay, M.R.; Madhu, P.; Jawaid, M.; Senthamaraikannan, P.; Senthil, S.; Pradeep, S. Characterization and properties of natural fiber polymer composites: A comprehensive review. J. Clean. Prod. 2018, 172, 566–581. [Google Scholar] [CrossRef]
- Florencia, V.; López, O.V.; García, M.A. Exploitation of by-products from cassava and ahipa starch extraction as filler of thermoplastic corn starch. Compos. Part B Eng. 2020, 182, 107653. [Google Scholar] [CrossRef]
- Nguyen, T.C.; Ruksakulpiwat, C.; Ruksakulpiwat, Y. Effect of cellulose nanofibers from cassava pulp on physical properties of poly(lactic acid) biocomposites. J. Thermoplast. Compos. Mater. 2020, 33, 1094–1108. [Google Scholar] [CrossRef]
- Azlin, M.N.M.; Sapuan, S.M.; Zainudin, E.S.; Zuhri, M.Y.M.; Ilyas, R.A. Natural Polylactic Acid-Based Fiber Composites: A Review. In Advanced Processing, Properties, and Applications of Starch and Other Bio-Based Polymers; Al-Oqla, F.M., Sapuan, S.M., Eds.; Elsevier: Oxford, UK, 2020; pp. 21–34. ISBN 9780128196618. [Google Scholar]
- Ilyas, R.A.; Aisyah, H.A.; Nordin, A.H.; Ngadi, N.; Zuhri, M.Y.M.; Asyraf, M.R.M.; Sapuan, S.M.; Zainudin, E.S.; Sharma, S.; Abral, H.; et al. Natural-Fiber-Reinforced Chitosan, Chitosan Blends and Their Nanocomposites for Various Advanced Applications. Polymers 2022, 14, 874. [Google Scholar] [CrossRef] [PubMed]
- Ilyas, R.A.; Sapuan, S.M.; Harussani, M.M.; Hakimi, M.Y.A.Y.; Haziq, M.Z.M.; Atikah, M.S.N.; Asyraf, M.R.M.; Ishak, M.R.; Razman, M.R.; Nurazzi, N.M.; et al. Polylactic Acid (PLA) Biocomposite: Processing, Additive Manufacturing and Advanced Applications. Polymers 2021, 13, 1326. [Google Scholar] [CrossRef] [PubMed]
- Ilyas, R.A.; Zuhri, M.Y.M.; Norrrahim, M.N.F.; Misenan, M.S.M.; Jenol, M.A.; Samsudin, S.A.; Nurazzi, N.M.; Asyraf, M.R.M.; Supian, A.B.M.; Bangar, S.P.; et al. Natural Fiber-Reinforced Polycaprolactone Green and Hybrid Biocomposites for Various Advanced Applications. Polymers 2022, 14, 182. [Google Scholar] [CrossRef]
- Ilyas, R.A.; Zuhri, M.Y.M.; Aisyah, H.A.; Asyraf, M.R.M.; Hassan, S.A.; Zainudin, E.S.; Sapuan, S.M.; Sharma, S.; Bangar, S.P.; Jumaidin, R.; et al. Natural Fiber-Reinforced Polylactic Acid, Polylactic Acid Blends and Their Composites for Advanced Applications. Polymers 2022, 14, 202. [Google Scholar] [CrossRef] [PubMed]
- Haris, N.I.N.; Hassan, M.Z.; Ilyas, R.; Suhot, M.A.; Sapuan, S.; Dolah, R.; Mohammad, R.; Asyraf, M. Dynamic mechanical properties of natural fiber reinforced hybrid polymer composites: A review. J. Mater. Res. Technol. 2022, 19, 167–182. [Google Scholar] [CrossRef]
- Atiqah, A.; Jawaid, M.; Sapuan, S.M.; Ishak, M.R.; Ansari, M.N.M.; Ilyas, R.A. Physical and thermal properties of treated sugar palm/glass fibre reinforced thermoplastic polyurethane hybrid composites. J. Mater. Res. Technol. 2019, 8, 3726–3732. [Google Scholar] [CrossRef]
- Sanjay, M.R.; Yogesha, B. Studies on Mechanical Properties of Jute/E-Glass Fiber Reinforced Epoxy Hybrid Composites. J. Miner. Mater. Charact. Eng. 2016, 4, 15–25. [Google Scholar] [CrossRef]
- Akampumuza, O.; Wambua, P.M.; Ahmed, A.; Li, W.; Qin, X.-H. Review of the applications of biocomposites in the automotive industry. Polym. Compos. 2016, 38, 2553–2569. [Google Scholar] [CrossRef]
- Hanson., E. How Are Car Manufacturers Adapting to a More Eco-Conscious Climate? Available online: https://www.openaccessgovernment.org/how-are-car-manufacturers-adapting-to-a-more-eco-conscious-climate/96968/ (accessed on 2 December 2020).
- Parker, L. A Whopping 91 Percent of Plastic Isn’t Recycled. Available online: https://www.nationalgeographic.org/article/whopping-91-percent-plastic-isnt-recycled/ (accessed on 3 December 2020).
- Edhirej, A.; Sapuan, S.M.; Jawaid, M.; Zahari, N.I. Effect of various plasticizers and concentration on the physical, thermal, mechanical, and structural properties of cassava-starch-based films. Starch/Staerke 2017, 69, 1500366. [Google Scholar] [CrossRef]
- Syafiq, R.; Sapuan, S.M.; Zuhri, M.Y.M.; Ilyas, R.A.; Nazrin, A.; Sherwani, S.F.K.; Khalina, A. Antimicrobial Activities of Starch-Based Biopolymers and Biocomposites Incorporated with Plant Essential Oils: A Review. Polymers 2020, 12, 2403. [Google Scholar] [CrossRef]
- Norrrahim, M.; Sapuan, S.; Yasim-Anuar, T.A.; Padzil, F.N.; Sharip, N.S.; Ng, L.Y.; Megashah, L.N.; Shazleen, S.S.; Rahim, N.F.; Syafiq, R.; et al. Antimicrobial Studies on Food Packaging Materials. In Food Packaging Advanced Materials, Technologies, and Innovations; Rangappa, S.M., Parameswaranpillai, J., Thiagamani, S.M.K., Krishnasamy, S., Siengchin, S., Eds.; Taylor & Francis Group: London, UK, 2020; pp. 141–170. [Google Scholar]
- Maringgal, B.; Hashim, N.; Mohamed Amin Tawakkal, I.S.; Muda Mohamed, M.T. Recent advance in edible coating and its effect on fresh/fresh-cut fruits quality. Trends Food Sci. Technol. 2020, 96, 253–267. [Google Scholar] [CrossRef]
- Nor, S.M.; Ding, P. Trends and advances in edible biopolymer coating for tropical fruit: A review. Food Res. Int. 2020, 134, 109208. [Google Scholar] [CrossRef]
- Suhag, R.; Kumar, N.; Petkoska, A.T.; Upadhyay, A. Film formation and deposition methods of edible coating on food products: A review. Food Res. Int. 2020, 136, 109582. [Google Scholar] [CrossRef] [PubMed]
- Sondari, D.; Aspiyanto; Amanda, A.S.; Triwulandari, E.; Ghozali, M.; Septiyanti, M.; Iltizam, I. Characterization edible coating made from native and modification cassava starch. AIP Conf. Proc. 2018, 2049, 030013. [Google Scholar] [CrossRef]
- Garcia, L.C.; Pereira, L.M.; de Luca Sarantópoulos, C.I.G.; Hubinger, M.D. Selection of an Edible Starch Coating for Minimally Processed Strawberry. Food Bioprocess Technol. 2010, 3, 834–842. [Google Scholar] [CrossRef]
- Silva, O.A.; Pellá, M.G.; Pellá, M.G.; Caetano, J.; Simões, M.R.; Bittencourt, P.R.S.; Dragunski, D.C. Synthesis and characterization of a low solubility edible film based on native cassava starch. Int. J. Biol. Macromol. 2019, 128, 290–296. [Google Scholar] [CrossRef]
- Chiumarelli, M.; Hubinger, M.D. Stability, solubility, mechanical and barrier properties of cassava starch—Carnauba wax edible coatings to preserve fresh-cut apples. Food Hydrocoll. 2012, 28, 59–67. [Google Scholar] [CrossRef]
- Lago-Vanzela, E.S.; do Nascimento, P.; Fontes, E.A.F.; Mauro, M.A.; Kimura, M. Edible coatings from native and modified starches retain carotenoids in pumpkin during drying. LWT Food Sci. Technol. 2013, 50, 420–425. [Google Scholar] [CrossRef]
- Bierhals, V.S.; Chiumarelli, M.; Hubinger, M.D. Effect of Cassava Starch Coating on Quality and Shelf Life of Fresh-Cut Pineapple (Ananas comosus L. Merril cv “Pérola”). J. Food Sci. 2011, 76, E62–E72. [Google Scholar] [CrossRef]
- Chiumarelli, M.; Pereira, L.M.; Ferrari, C.C.; Sarantópoulos, C.I.G.L.; Hubinger, M.D. Cassava Starch Coating and Citric Acid to Preserve Quality Parameters of Fresh-Cut “Tommy Atkins” Mango. J. Food Sci. 2010, 75, E297–E304. [Google Scholar] [CrossRef]
- Utami, R.; Kawiji; Nurhartadi, E.; Putra, A.Y.T.; Setiawan, A. The effect of cassava starch-based edible coating enriched with kaempferia rotunda and curcuma xanthorrhiza essential oil on refrigerated patin fillets quality. Int. Food Res. J. 2014, 21, 413–419. [Google Scholar]
- Pradyawong, S.; Juneja, A.; Sadiq, M.B.; Noomhorm, A.; Singh, V. Comparison of Cassava Starch with Corn as a Feedstock for Bioethanol Production. Energies 2018, 11, 3476. [Google Scholar] [CrossRef]
- Efeovbokhan, V.E.; Egwari, L.; Alagbe, E.E.; Adeyemi, J.T.; Taiwo, O.S. Production of Bioethanol from Hybrid Cassava Pulp and Peel using Microbial and Acid Hydrolysis. Bioresources 2019, 14, 2596–2609. [Google Scholar]
- Hajar, S.; Azhar, M.; Abdulla, R.; Jambo, S.A.; Marbawi, H.; Azlan, J.; Azifa, A.; Faik, M.; Francis, K. Yeasts in sustainable bioethanol production: A review. Biochem. Biophys. Rep. 2017, 10, 52–61. [Google Scholar] [CrossRef]
- Fournier, J.; Corre, O. Le Bioethanol production from cassava starch by enzymatic hydrolysis, fermentation and ex-situ nanofiltration. Energy Procedia 2017, 138, 883–888. [Google Scholar] [CrossRef]
- Gupta, A.; Prakash, J. Sustainable bio-ethanol production from agro-residues: A review. Renew. Sustain. Energy Rev. 2015, 41, 550–567. [Google Scholar] [CrossRef]
- Popp, J.; Lakner, Z.; Harangi-rákos, M.; Fári, M. The effect of bioenergy expansion: Food, energy, and environment. Renew. Sustain. Energy Rev. 2014, 32, 559–578. [Google Scholar] [CrossRef]
- Okudoh, V.; Trois, C.; Workneh, T.; Schmidt, S. The potential of cassava biomass and applicable technologies for sustainable biogas production in South Africa: A review. Renew. Sustain. Energy Rev. 2014, 39, 1035–1052. [Google Scholar] [CrossRef]
- Witantri, R.G.; Purwoko, T.; Sunarto; Mahajoeno, E. Bioethanol Production By Utilizing Cassava Peels Waste Through Enzymatic And Microbiological Hydrolysis. IOP Conf. Ser. Earth Environ. Sci. 2017, 75, 012014. [Google Scholar] [CrossRef]
- Adetunji, R.O.; Youdeowei, P.K.; Kolawole, O.O. Production of Bio-ethanol from Cassava Peels. In Proceedings of the International Conference on Renewable energy and power, Atlanta, Georgia, 21–23 September 2015. [Google Scholar]
- Kotoka, F.; Tulashie, S.K.; Setsoafia, D.D. Production of bioethanol from liquid waste from cassava dough during gari processing. Biofuels 2017, 10, 493–501. [Google Scholar] [CrossRef]
- Egloso, N.L. The Production of Fuel-Grade Bioethanol From Cassava Starch: A Conceptual The Production of Fuel-Grade Bioethanol From Cassava Starch: A Conceptual Process Design. Int. J. Chem. Environ. Eng. 2018, 5. [Google Scholar]
- Oyeleke, S.B.; Dauda, B.E.N.; Oyewole, O.A.; Okoliegbe, I.N.; Ojebode, T. Production of Bioethanol from Cassava and Sweet Potato Peels. Adv. Environ. Biol. 2011, 5, 3729–3733. [Google Scholar]
- Tanaka, K.; Koyama, M.; Thi, P.; Palenzuela, A.; Habaki, H.; Egashira, R.; Nakasaki, K. International Biodeterioration & Biodegradation Production of high-concentration bioethanol from cassava stem by repeated hydrolysis and intermittent yeast inoculation. Int. Biodeterior. Biodegrad. 2019, 138, 1–7. [Google Scholar] [CrossRef]
- Sarkar, N.; Ghosh, S.K.; Bannerjee, S.; Aikat, K. Bioethanol production from agricultural wastes: An overview. Renew. Energy 2012, 37, 19–27. [Google Scholar] [CrossRef]
- Principles, M.D.; Alfitri, N.; Hidayat, A.; Putera, Y. Bioethanol Producing Equipment Prototype Using Cassava Peel Using Bioethanol Producing Equipment Prototype Using Cassava Peel Using Microcontroller-Based Destilation Principles. In Proceedings of the International Conference on Applied Sciences, Information and Technology 2019, Yogyakarta, Indonesia, 7–8 August 2019. [Google Scholar]







| Composition | Content g/100 g Dry Pulp | ||||
|---|---|---|---|---|---|
| Rattanachomsri et al. [33] | Suwanasri et al. [34] | Kosugi et al. [35] | Sriroth et al. [36] | Virunanon et al. [37] | |
| Starch | 60.1 ± 0.1 | 60.6 | 68.9 ± 4.0 | 65.4 ± 4.1 | 75.1 |
| Crude fiber | n/d | 28.1 | 27.8 ± 0.2 | 13.2 ± 2.2 | n/d |
| Lignin | 2.8 ± 0.06 | n/d | n/d | n/d | 1.2 |
| Cellulose | 15.6 | n/d | n/d | n/d | 4.1 |
| Protein | n/d | 1.8 | 1.6 ± 0.03 | 2.1 ± 0.25 | n/d |
| Hemicellulose | 4.6 | n/d | n/d | n/d | 4.2 |
| Fat | n/d | n/d | 0.1 ± 0.01 | 0.2 ± 0.06 | n/d |
| Advantages | Disadvantages |
|---|---|
| Low specific weight, higher specific strength | Lower impact strength |
| Renewable, low-energy consumption, low carbon emissions | Sensitive to weather and environment |
| Facile manufacturing process | Poor moisture resistance, fibers easily swell |
| Low-cost manufacturing | Restricted maximum processing temperature |
| Good electrical insulator | Lower durability |
| Good thermal and acoustic insulating properties | Poor thermal resistance |
| Biodegradable | Hydrophilic nature, low wetting with hydrophobic polymers |
| Substrate | Starch (%) | Sugars (%) | Cellulose (%) | Hemicellulose (%) | References |
|---|---|---|---|---|---|
| Bagasse | 65.6 | _ | 8.1 | 2.8 | [103] |
| 60.1 | _ | 15.63 | 4.58 | [33] | |
| 56 | _ | _ | _ | [104] | |
| 79.45 | _ | _ | _ | [105] | |
| 48 | _ | 23 | 9 | [106] | |
| 32.6 | 18 | _ | _ | [107] | |
| 49.66 | _ | 21.47 | 12.97 | [108] | |
| 57.8 | 88.4 | _ | _ | [109] | |
| 64 | _ | _ | 2.11 | [110] | |
| Stem | 8.41 | _ | 21.43 | 11.62 | [111] |
| _ | _ | 56.4 | 20.2 | [112] | |
| _ | _ | 35.2 | 24.3 | [113] | |
| _ | _ | 38.8 | 7.2 | [114] | |
| Peel | 41.85 | 48.22 | _ | _ | [115] |
| 15.82 | _ | 35.86 | 9.27 | [116] | |
| 67 | _ | _ | _ | [117] | |
| Leaf | 28.7 | 29 | _ | _ | [107] |
| Rhizome | _ | _ | 27.82 | 39.67 | [118] |
| Composition | Fresh Weight | Dry Weight |
|---|---|---|
| Calories | 135 | 335 |
| Moisture (%) | 65.5 | 15.7 |
| Proteins (g) | 1.00 | 1.4 |
| Lipids (g) | 0.2 | 0.5 |
| Starch (g) | 32.4 | 80.6 |
| Fibers (g) | 1.1 | 1.2 |
| Ash (g) | 0.9 | 1.8 |
| Calcium (mg) | 26 | 96 |
| Phosphorus (mg) | 32 | 81 |
| Iron (mg) | 0.9 | 7.9 |
| Sodium (mg) | 2 | - |
| Potassium (mg) | 394 | - |
| Vitamin B2 (mg) | 0.04 | 0.06 |
| Vitamin C (mg) | 34 | 0 |
| Niacin (mg) | 0.6 | 0.8 |
| Cyanide (%) | - | 1.6 |
| Composition | Soccol et al. [133] | Cereda et al. [134] | Pandy et al. [1] | Stertz et al. [135] | Vandenberghe et al. [136] |
|---|---|---|---|---|---|
| Moisture | 5.02 | 9.52 | 5.02–11.2 | 10.70 | 11.20 |
| Protein | 1.57 | 0.32 | 0.32–1.61 | 1.60 | 1.61 |
| Lipids | 1.06 | 0.83 | 0.53–1.06 | 0.53 | 0.54 |
| Fibers | 50.55 | 14.88 | 14.88–50.55 | 22.20 | 21.10 |
| Ash | 1.10 | 0.66 | 0.66–1.50 | 1.50 | 1.44 |
| Carbohydrates | 40.50 | 63.85 | 40.50–63.85 | 63.40 | 63.00 |
| Polymer Blend | Fiber | Reference |
|---|---|---|
| Native cassava starch, agar (AG), cassava starch (CAS), and arabinoxylan (AX) | Beta zeolite nanocrystal or Na-beidellite | [218] |
| Cassava flour (CF)/wheat flour (WF) | Cassava stillage residue (CSR) | [158,159] |
| Sodium cellulose sulfate (NaCS)/cassava starch | [219] | |
| Cassava starch/polylactic acid (PLA) | Cassava bagasse | [220] |
| Cassava starch/low-density polyethylene | Cotton fibers | [221] |
| Cassava starch/polyvinyl alcohol (PVA) | Bamboo nanofibrils | [222] |
| Cassava starch/poly(lactic acid) | Coir fiber | [217] |
| Cassava starch/polyvinyl alcohol | Sugarcane bagasse fiber | [223] |
| Cassava starch/poly(butylene succinate) | Maleated poly(butylene succinate) | [224] |
| Cassava starch/polyvinyl alcohol | Bacterial cellulose fiber | [225] |
| Cassava/corn starch | Passion fruit peel | [226] |
| Cassava starch/low-density polyethylene | Cotton fibers | [161] |
| Starch/poly(vinyl alcohol) | Bamboo cellulose nanofiber | [227] |
| Cassava/poly(vinyl alcohol) (PVA) | Oil palm empty fruit bunches (OPEFBs) nanocellulose | [228] |
| Cassava solid waste/bagasse starch (BS) | Bamboo cellulose microfiber (MFC)/epoxidized waste cooking oil (EWCO) | [229] |
| Cassava/banana starch | Banana fiber | [230] |
| Cassava Starch/Polybutylene Adipate Terephthalate (PBAT) | - | [231] |
| Cassava starch/carboxylated styrene-butadiene rubber | Cellulose fiber | [232] |
| Cassava starch/polylactic acid | - | [233] |
| Cassava starch/Chitosan | Kraft fiber | [148] |
| Polymer | Polymer Matrix | Reference |
|---|---|---|
| Cassava starch | Cassava peel/cassava bagasse | [156] |
| Cassava starch | Cassava/sugar palm fiber | [71] |
| Cassava starch | Cassava bagasse lignocellulose nanofibers (LCNF)/nanoclay (Nclay) | [143] |
| Cassava starch | Cassava bagasse/kraft paper | [149] |
| Cassava starch | ZnO/bacterial cellulose | [173] |
| Cassava starch | Carnauba wax/cashew tree gum-based films | [174] |
| Cassava starch | Concentrated natural rubber latex/cotton fiber | [175] |
| Cassava starch | Cellulose pulp fibers modified with deposition of silica (SiO2) nanoparticles | [176] |
| Cassava starch | Oregano essential oil/sugarcane bagasse | [182] |
| Cassava starch | Cellulose fiber/nanoclay | [188] |
| Cassava starch | Sugarcane Bagasse Fibers/montmorillonite | [189] |
| Cassava starch | Ramie fibers CNF/nano PCC tapioca starch | [195] |
| Cassava starch | Nanofiber straw/ZnO | [197] |
| Cassava starch | Bamboo fiber, lime juice, epoxidized waste cooking oil | [201] |
| Cassava starch | Carboxymethylcellulose/lactic acid bacteria | [202] |
| Cassava starch | ZnO nanorods/PVA electrospun mats/rosemary extract | [203] |
| Cassava starch | Carboxymethylcellulose/turmeric oil | [213] |
| Cassava starch | Sisal fiber/carnauba wax | [214] |
| Parameters | Starch | Control | Cellulose | Nanoclay | Cellulose-Nanoclay |
|---|---|---|---|---|---|
| Tensile strength (MPa) | Potato | 12.03 | 12.85 | 14.59 | 17.75 |
| Cassava | 4.68 | 6.87 | 5.78 | 11.72 | |
| Elongation (%) | Potato | 13.81 | 2.12 | 3.50 | 0.94 |
| Cassava | 4.62 | 2.09 | 4.47 | 1.32 |
| Samples | Tensile Strength (MPa) | Elongation at Break (%) |
|---|---|---|
| CS/CMC | 13.29 | 65.72 |
| CS/CMC/LPL-0.5% | 15.69 | 51.40 |
| CS/CMC/LPL-1% | 15.42 | 50.22 |
| CS/CMC/LPL-1.5% | 14.84 | 47.24 |
| CS/CMC/LPL-2% | 12.73 | 45.81 |
| CS/CMC/PPE-0.5% | 13.10 | 47.69 |
| CS/CMC/PPE-1% | 13.04 | 44.30 |
| CS/CMC/PPE-1.5% | 11.21 | 42.26 |
| CS/CMC/PPE-2% | 11.06 | 38.00 |
| Fiber | Polymer Matrix | Reference |
|---|---|---|
| Cassava skin | Polyvinyl alcohol (PVA) | [146] |
| Cassava bagasse | Polyvinyl-alcohol (PVA) incorporated with clove essential oil (CEO) or oregano essential oils (OEO) and cassava | [147] |
| Cassava stillage residue (CSR) | Poly(vinyl chloride) (PVC) | [160] |
| Cassava pulp | Polylactic acid and thermoplastic starch | |
| Cassava skin | Polyvinyl alcohol (PVA) | [146] |
| Cassava stillage residue (CSR) | Poly(vinyl chloride) (PVC) | [160] |
| Cassava and ahipa peels and bagasse | Corn starch | [236] |
| Cassava nanofibers | Poly(lactic acid) | [237] |
| Cassava bagasse | Cassava starch | [147] |
| Cellulose cassava bagasse nanofibrils (CBN) | Cassava starch | [152] |
| Cassava stillage residue (CSR) | Cassava flour (CF)/wheat flour (WF) | [158,159] |
| Poly(vinyl chloride) (PVC) | [160] | |
| Final egg albumen, cassava starch, sunflower oil | [161] | |
| Cassava peel/cassava bagasse | Cassava Starch | [156] |
| The remaining fibrous residue of cassava starch extraction | Cassava Starch | [162] |
| Cassava nanofiber | Cassava starch | [143] |
| Cassava/sugar palm fiber | Cassava starch | [71] |
| Cassava bagasse cellulose nanofibrils | Cassava starch | [163] |
| Cassava bagasse lignocellulose nanofibers (LCNF)/nanoclay (Nclay) | Cassava starch | [143] |
| Cassava bagasse/kraft paper | Cassava starch | [149] |
| Cassava bagasse | Cassava starch | [31] |
| Cassava bagasse (CB) | Cassava starch | [178] |
| Cassava peel (CP) | Cassava starch | [178] |
| Cassava peel | Cassava starch | [180] |
| Cassava roots bagasse | Cassava starch | [187] |
| Cassava nanofibril | Cassava starch | [138] |
| Cassava bagasse | Cassava starch | [209] |
| Cassava cellulose nanocrystals | Cassava starch | [211] |
| TPS | Bagasse Fiber (wt.%) | E(30 °C) (MPa) |
|---|---|---|
| Cassava | 0 | 21.5 |
| 5 | 39.6 | |
| 10 | 95.9 | |
| 15 | 128.5 | |
| Corn | 0 | 18.2 |
| 5 | 59.1 | |
| 10 | 73.0 | |
| 15 | 97.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abotbina, W.; Sapuan, S.M.; Ilyas, R.A.; Sultan, M.T.H.; Alkbir, M.F.M.; Sulaiman, S.; Harussani, M.M.; Bayraktar, E. Recent Developments in Cassava (Manihot esculenta) Based Biocomposites and Their Potential Industrial Applications: A Comprehensive Review. Materials 2022, 15, 6992. https://doi.org/10.3390/ma15196992
Abotbina W, Sapuan SM, Ilyas RA, Sultan MTH, Alkbir MFM, Sulaiman S, Harussani MM, Bayraktar E. Recent Developments in Cassava (Manihot esculenta) Based Biocomposites and Their Potential Industrial Applications: A Comprehensive Review. Materials. 2022; 15(19):6992. https://doi.org/10.3390/ma15196992
Chicago/Turabian StyleAbotbina, Walid, S. M. Sapuan, R. A. Ilyas, M. T. H. Sultan, M. F. M. Alkbir, S. Sulaiman, M. M. Harussani, and Emin Bayraktar. 2022. "Recent Developments in Cassava (Manihot esculenta) Based Biocomposites and Their Potential Industrial Applications: A Comprehensive Review" Materials 15, no. 19: 6992. https://doi.org/10.3390/ma15196992
APA StyleAbotbina, W., Sapuan, S. M., Ilyas, R. A., Sultan, M. T. H., Alkbir, M. F. M., Sulaiman, S., Harussani, M. M., & Bayraktar, E. (2022). Recent Developments in Cassava (Manihot esculenta) Based Biocomposites and Their Potential Industrial Applications: A Comprehensive Review. Materials, 15(19), 6992. https://doi.org/10.3390/ma15196992











