The Fabrication and Bonding of Thermoplastic Microfluidics: A Review
Abstract
1. Introduction
2. Forming the Microchannel Geometry
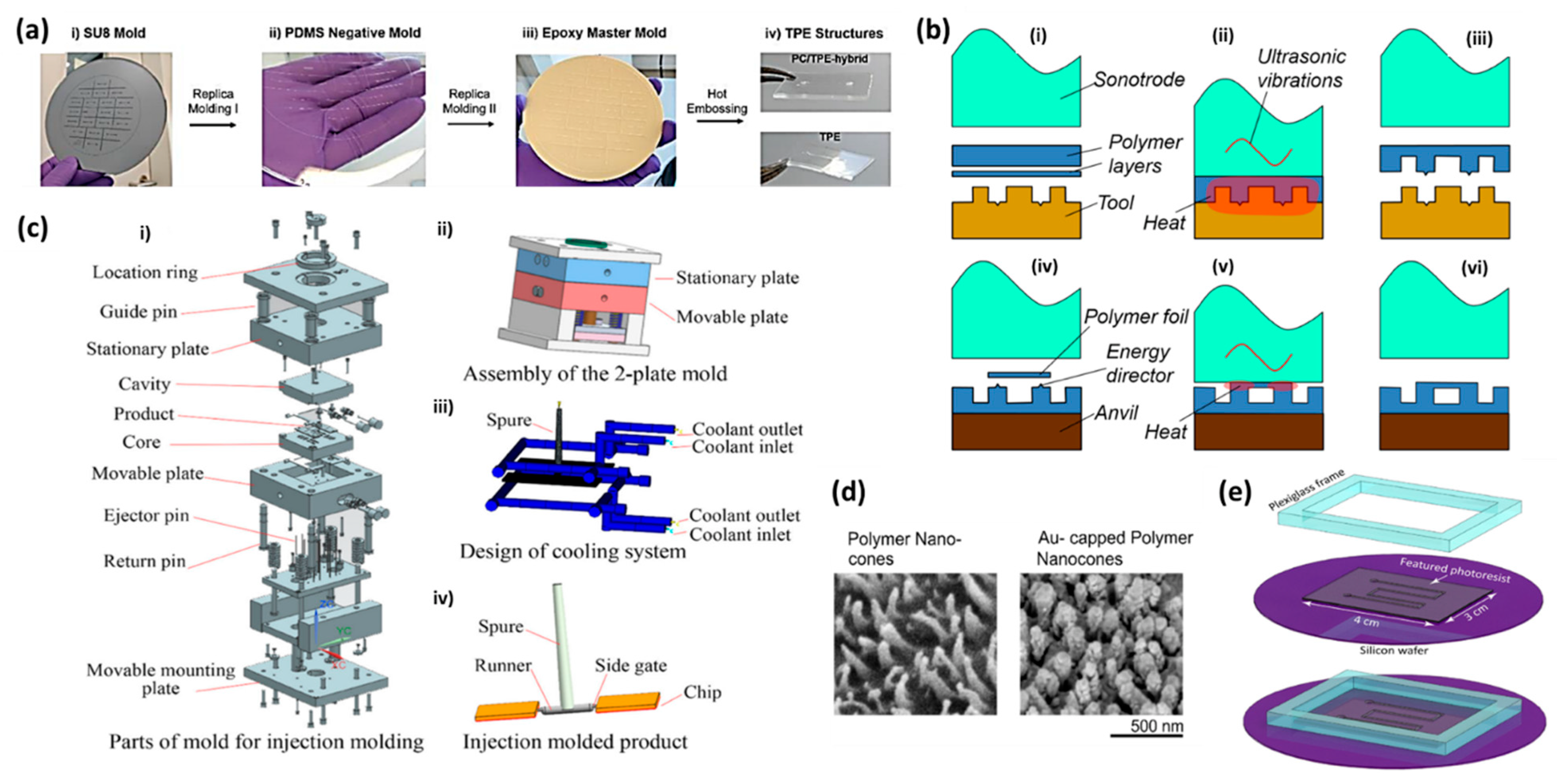
2.1. Hot Embossing
2.2. Injection Molding
2.3. Master Mold Fabrication
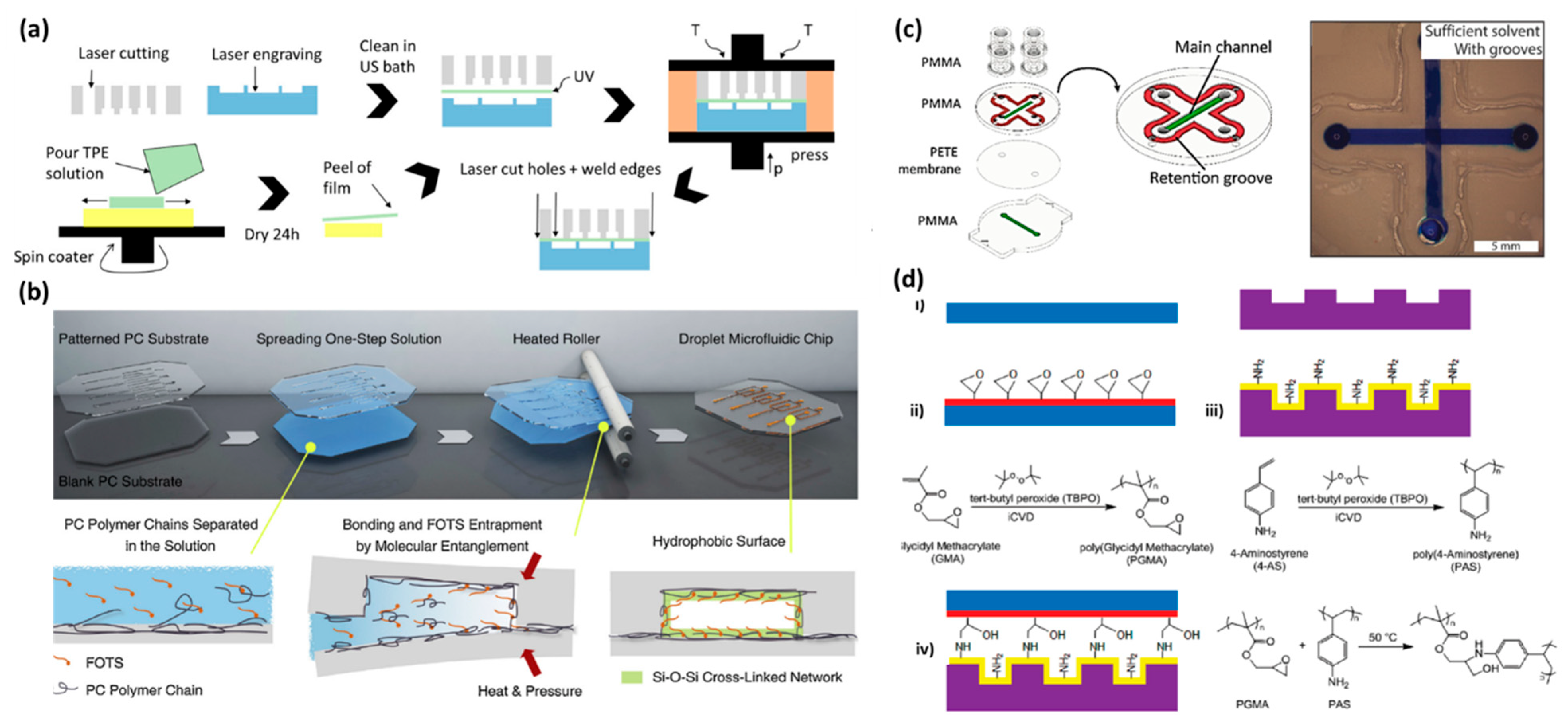
2.4. Laser Ablation
2.5. Additive Manufacturing
2.6. Other Methods

3. Bonding
3.1. Thermal Fusion Bonding
3.2. Solvent Bonding
3.3. Chemical Bonding
3.4. Ultrasonic Welding
3.5. Laser Welding
3.6. Adhesive Layers
4. Conclusions and Future Trend
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shakeri, A.; Rahmani, S.; Imani, S.M.; Osborne, M.; Yousefi, H.; Didar, T.F. 26-Biofunctional interfaces for cell culture in microfluidic devices. In Bioelectronics and Medical Devices: From Materials to Devices-Fabrication, Applications and Reliability; Pal, K., Kraatz, H.-B., Khasnobish, A., Bag, S., Banerjee, I., Kuruganti, U., Eds.; Woodhead Publishing: Cambridge, MA, USA, 2019; pp. 635–699. ISBN 978-0-08-102420-1. [Google Scholar]
- Khan, S.; Burciu, B.; Filipe, C.D.M.; Li, Y.; Dellinger, K.; Didar, T.F. DNAzyme-Based Biosensors: Immobilization Strategies, Applications, and Future Prospective. ACS Nano 2021, 15, 13943–13969. [Google Scholar] [CrossRef] [PubMed]
- Fatanat Didar, T.; Tabrizian, M. Generating multiplex gradients of biomolecules for controlling cellular adhesion in parallel microfluidic channels. Lab Chip 2012, 12, 4363–4371. [Google Scholar] [CrossRef]
- Didar, T.F.; Bowey, K.; Almazan, G.; Tabrizian, M. A miniaturized multipurpose platform for rapid, label-free, and simultaneous separation, patterning, and in vitro culture of primary and rare cells. Adv. Healthc. Mater. 2014, 3, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Shakeri, A.; Sun, N.; Badv, M.; Didar, T.F. Generating 2-dimensional concentration gradients of biomolecules using a simple microfluidic design. Biomicrofluidics 2017, 11, 044111. [Google Scholar] [CrossRef] [PubMed]
- Shakeri, A.; Imani, S.M.; Chen, E.; Yousefi, H.; Shabbir, R.; Didar, T.F. Plasma-induced covalent immobilization and patterning of bioactive species in microfluidic devices. Lab Chip 2019, 19, 3104–3115. [Google Scholar] [CrossRef] [PubMed]
- Shakeri, A.; Khan, S.; Didar, T.F. Conventional and emerging strategies for the fabrication and functionalization of PDMS-based microfluidic devices. Lab Chip 2021, 21, 3053–3075. [Google Scholar] [CrossRef] [PubMed]
- Shakeri, A.; Jarad, N.A.; Leung, A.; Soleymani, L.; Didar, T.F. Biofunctionalization of Glass- and Paper-Based Microfluidic Devices: A Review. Adv. Mater. Interfaces 2019, 6, 1900940. [Google Scholar] [CrossRef]
- Gokaltun, A.; Yarmush, M.L.; Asatekin, A.; Usta, O.B. Recent advances in nonbiofouling PDMS surface modification strategies applicable to microfluidic technology. Technology 2017, 5, 1–12. [Google Scholar] [CrossRef]
- Park, J.Y.; Yoo, S.J.; Lee, E.-J.; Lee, D.H.; Kim, J.Y.; Lee, S.-H. Increased poly(dimethylsiloxane) stiffness improves viability and morphology of mouse fibroblast cells. BioChip J. 2010, 4, 230–236. [Google Scholar] [CrossRef]
- Shakeri, A.; Jarad, N.A.; Khan, S.; Didar, T.F. Bio-functionalization of microfluidic platforms made of thermoplastic materials: A review. Anal. Chim. Acta 2022, 1209, 339283. [Google Scholar] [CrossRef]
- Tsao, C.-W. Polymer Microfluidics: Simple, Low-Cost Fabrication Process Bridging Academic Lab Research to Commercialized Production. Micromachines 2016, 7, 225. [Google Scholar] [CrossRef]
- Badv, M.; Weitz, J.I.; Didar, T.F. Lubricant-Infused PET Grafts with Built-In Biofunctional Nanoprobes Attenuate Thrombin Generation and Promote Targeted Binding of Cells. Small 2019, 15, 1905562. [Google Scholar] [CrossRef]
- Shakeri, A.; Jarad, N.A.; Terryberry, J.; Khan, S.; Leung, A.; Chen, S.; Didar, T.F. Antibody Micropatterned Lubricant-Infused Biosensors Enable Sub-Picogram Immunofluorescence Detection of Interleukin 6 in Human Whole Plasma. Small 2020, 16, 2003844. [Google Scholar] [CrossRef]
- Giri, K.; Tsao, C.-W. Recent Advances in Thermoplastic Microfluidic Bonding. Micromachines 2022, 13, 486. [Google Scholar] [CrossRef]
- Gale, B.K.; Jafek, A.R.; Lambert, C.J.; Goenner, B.L.; Moghimifam, H.; Nze, U.C.; Kamarapu, S.K. A Review of Current Methods in Microfluidic Device Fabrication and Future Commercialization Prospects. Inventions 2018, 3, 60. [Google Scholar] [CrossRef]
- Ye, M.Y.; Yin, X.F.; Fang, Z.L. DNA separation with low-viscosity sieving matrix on microfabricated polycarbonate microfluidic chips. Anal. Bioanal. Chem. 2005, 381, 820–827. [Google Scholar] [CrossRef]
- Liu, Y.; Ganser, D.; Schneider, A.; Liu, R.; Grodzinski, P.; Kroutchinina, N. Microfabricated polycarbonate CE devices for DNA analysis. Anal. Chem. 2001, 73, 4196–4201. [Google Scholar] [CrossRef]
- Faure, K.; Albert, M.; Dugas, V.; Crétier, G.; Ferrigno, R.; Morin, P.; Rocca, J.L. Development of an acrylate monolith in a cyclo-olefin copolymer microfluidic device for chip electrochromatography separation. Electrophoresis 2008, 29, 4948–4955. [Google Scholar] [CrossRef]
- Lee, J.H.; Kim, S.K.; Park, H.H.; Kim, T.S. TiO2 coated microfluidic devices for recoverable hydrophilic and hydrophobic patterns. J. Micromech. Microeng. 2015, 25, 035032. [Google Scholar] [CrossRef]
- Young, E.W.K.; Berthier, E.; Guckenberger, D.J.; Sackmann, E.; Lamers, C.; Meyvantsson, I.; Huttenlocher, A.; Beebe, D.J. Rapid Prototyping of Arrayed Microfluidic Systems in Polystyrene for Cell-Based Assays. Anal. Chem. 2011, 83, 1408–1417. [Google Scholar] [CrossRef]
- Jackson, J.M.; Witek, M.A.; Hupert, M.L.; Brady, C.; Pullagurla, S.; Kamande, J.; Aufforth, R.D.; Tignanelli, C.J.; Torphy, R.J.; Yeh, J.J.; et al. UV activation of polymeric high aspect ratio microstructures: Ramifications in antibody surface loading for circulating tumor cell selection. Lab Chip 2014, 14, 106–117. [Google Scholar] [CrossRef] [PubMed]
- Chien, R. Hot embossing of microfluidic platform. Int. Commun. Heat Mass Transf. 2006, 33, 645–653. [Google Scholar] [CrossRef]
- Jena, R.K.; Yue, C.Y. Cyclic olefin copolymer based microfluidic devices for biochip applications: Ultraviolet surface grafting using 2-methacryloyloxyethyl phosphorylcholine. Biomicrofluidics 2012, 6, 012822. [Google Scholar] [CrossRef] [PubMed]
- Su, S.; Jing, G.; Zhang, M.; Liu, B.; Zhu, X.; Wang, B.; Fu, M.; Zhu, L.; Cheng, J.; Guo, Y. One-step bonding and hydrophobic surface modification method for rapid fabrication of polycarbonate-based droplet microfluidic chips. Sens. Actuators B Chem. 2019, 282, 60–68. [Google Scholar] [CrossRef]
- Aghvami, S.A.; Opathalage, A.; Zhang, Z.K.; Ludwig, M.; Heymann, M.; Norton, M.; Wilkins, N.; Fraden, S. Rapid prototyping of cyclic olefin copolymer (COC) microfluidic devices. Sens. Actuators B Chem. 2017, 247, 940–949. [Google Scholar] [CrossRef]
- Becker, H.; Gärtner, C. Polymer microfabrication methods for microfluidic analytical applications. Electrophoresis 2000, 21, 12–26. [Google Scholar] [CrossRef]
- Yi, L.; Xiaodong, W.; Fan, Y. Microfluidic chip made of COP (cyclo-olefin polymer) and comparion to PMMA (polymethylmethacrylate) microfluidic chip. J. Mater. Process. Technol. 2008, 208, 63–69. [Google Scholar] [CrossRef]
- Lachaux, J.; Alcaine, C.; Gómez-Escoda, B.; Perrault, C.M.; Duplan, D.O.; Wu, P.-Y.J.; Ochoa, I.; Fernandez, L.; Mercier, O.; Coudreuse, D.; et al. Thermoplastic elastomer with advanced hydrophilization and bonding performances for rapid (30 s) and easy molding of microfluidic devices. Lab Chip 2017, 17, 2581–2594. [Google Scholar] [CrossRef]
- Busek, M.; Nøvik, S.; Aizenshtadt, A.; Amirola-Martinez, M.; Combriat, T.; Grünzner, S.; Krauss, S. Thermoplastic elastomer (TPE)–poly(methyl methacrylate) (PMMA) hybrid devices for active pumping pdms-free organ-on-a-chip systems. Biosensors 2021, 11, 162. [Google Scholar] [CrossRef]
- Schneider, S.; Brás, E.J.S.; Schneider, O.; Schlünder, K.; Loskill, P. Facile patterning of thermoplastic elastomers and robust bonding to glass and thermoplastics for microfluidic cell culture and organ-on-chip. Micromachines 2021, 12, 575. [Google Scholar] [CrossRef]
- Feyssa, B.; Liedert, C.; Kivimaki, L.; Johansson, L.S.; Jantunen, H.; Hakalahti, L. Patterned Immobilization of Antibodies within Roll-to-Roll Hot Embossed Polymeric Microfluidic Channels. PLoS ONE 2013, 8, e68918. [Google Scholar] [CrossRef]
- Runge, T.; Sackmann, J.; Karl, W.; Lars, S.; Blank, M. Ultrasonically manufactured microfluidic device for yeast analysis. Microsyst. Technol. 2017, 23, 2139–2144. [Google Scholar] [CrossRef]
- Lee, U.N.; Su, X.; Guckenberger, D.J.; Dostie, A.M.; Zhang, T.; Berthier, E.; Theberge, A.B. Fundamentals of rapid injection molding for microfluidic cell-based assays. Lab Chip 2018, 18, 496–504. [Google Scholar] [CrossRef]
- Attia, U.M.; Marson, S.; Alcock, J.R. Micro-injection moulding of polymer microfluidic devices. Microfluid. Nanofluid. 2009, 7, 1–28. [Google Scholar] [CrossRef]
- Roy, S.; Yue, C.Y.; Venkatraman, S.S.; Ma, L.L. Fabrication of smart COC chips: Advantages of N-vinylpyrrolidone (NVP) monomer over other hydrophilic monomers. Sens. Actuators B Chem. 2013, 178, 86–95. [Google Scholar] [CrossRef]
- Büttner, H.; Maradia, U.; Suarez, M.; Stirnimann, J.; Wegener, K. Development of Process Chain for Micro-Injection Molding. Procedia CIRP 2020, 95, 584–589. [Google Scholar] [CrossRef]
- Ogorodnyk, O.; Martinsen, K. Monitoring and control for thermoplastics injection molding A review. Procedia CIRP 2018, 67, 380–385. [Google Scholar] [CrossRef]
- Wang, Y.; Yu, K.-M.; Wang, C.C.L. Spiral and conformal cooling in plastic injection molding. Comput. Des. 2015, 63, 1–11. [Google Scholar] [CrossRef]
- Rahim, S.Z.A.; Sharif, S.; Zain, A.M.; Nasir, S.M.; Mohd Saad, R. Improving the Quality and Productivity of Molded Parts with a New Design of Conformal Cooling Channels for the Injection Molding Process. Adv. Polym. Technol. 2016, 35. [Google Scholar] [CrossRef]
- He, B.; Ying, L.; Li, X.; Hu, P. Optimal design of longitudinal conformal cooling channels in hot stamping tools. Appl. Therm. Eng. 2016, 106, 1176–1189. [Google Scholar] [CrossRef]
- Chen, S.-C.; Jong, W.-R.; Chang, J.-A. Dynamic mold surface temperature control using induction heating and its effects on the surface appearance of weld line. J. Appl. Polym. Sci. 2006, 101, 1174–1180. [Google Scholar] [CrossRef]
- Wang, G.; Zhao, G.; Li, H.; Guan, Y. Research on a New Variotherm Injection Molding Technology and its Application on the Molding of a Large LCD Panel. Polym. Plast. Technol. Eng. 2009, 48, 671–681. [Google Scholar] [CrossRef]
- Saito, T.; Satoh, I.; Kurosaki, Y. A new concept of active temperature control for an injection molding process using infrared radiation heating. Polym. Eng. Sci. 2002, 42, 2418–2429. [Google Scholar] [CrossRef]
- Ma, X.; Li, R.; Jin, Z.; Fan, Y.; Zhou, X.; Zhang, Y. Injection molding and characterization of PMMA-based microfluidic devices. Microsyst. Technol. 2020, 26, 1317–1324. [Google Scholar] [CrossRef]
- Kim, Y.; Song, J.; Lee, Y.; Cho, S.; Kim, S.; Lee, S.R.; Park, S.; Shin, Y.; Jeon, N.L. High-throughput injection molded microfluidic device for single-cell analysis of spatiotemporal dynamics. Lab Chip 2021, 21, 3150–3158. [Google Scholar] [CrossRef]
- Ko, J.; Lee, Y.; Lee, S.; Lee, S.R.; Jeon, N.L. Human Ocular Angiogenesis-Inspired Vascular Models on an Injection-Molded Microfluidic Chip. Adv. Healthc. Mater. 2019, 8, 1–10. [Google Scholar] [CrossRef]
- Viehrig, M.; Thilsted, A.H.; Matteucci, M.; Wu, K.; Catak, D.; Schmidt, M.S.; Zór, K.; Boisen, A. Injection-Molded Microfluidic Device for SERS Sensing Using Embedded Au-Capped Polymer Nanocones. ACS Appl. Mater. Interfaces 2018, 10, 37417–37425. [Google Scholar] [CrossRef] [PubMed]
- Council, N.R. 3. Manufacturing: Materials and Processing. In Polymer Science and Engineering: The Shifting Research Frontiers; The National Academies Press: Washington, DC, USA, 1994; pp. 65–115. ISBN 978-0-309-07677-7. [Google Scholar]
- McKeen, L.W. Chapter 1-Introduction to Plastics and Polymers. In Plastics Design Library; William Andrew Publishing: Boston, MA, USA, 2012; pp. 1–18. ISBN 978-1-4557-2551-9. [Google Scholar]
- Jena, R.K.; Yue, C.Y.; Lam, Y.C.; Wang, Z.Y. High fidelity hot-embossing of COC microdevices using a one-step process without pre-annealing of polymer substrate. Sens. Actuators B Chem. 2010, 150, 692–699. [Google Scholar] [CrossRef]
- Müller, B.; Sulzer, P.; Walch, M.; Zirath, H.; Buryška, T.; Rothbauer, M.; Ertl, P.; Mayr, T. Measurement of respiration and acidification rates of mammalian cells in thermoplastic microfluidic devices. Sens. Actuators B Chem. 2021, 334, 129664. [Google Scholar] [CrossRef]
- Hupert, M.L.; Guy, W.J.; Llopis, S.D.; Shadpour, H.; Rani, S.; Nikitopoulos, D.E.; Soper, S.A. Evaluation of micromilled metal mold masters for the replication of microchip electrophoresis devices. Microfluid. Nanofluid. 2007, 3, 1–11. [Google Scholar] [CrossRef]
- Perrone, E.; Cesaria, M.; Zizzari, A.; Bianco, M.; Ferrara, F.; Raia, L.; Guarino, V.; Cuscun, M.; Mazzeo, M.; Gigli, G.; et al. Potential of CO2-laser processing of quartz for fast prototyping of microfluidic reactors and templates for 3D cell assembly over large scale. Mater. Today Bio 2021, 12, 100163. [Google Scholar] [CrossRef]
- Chantiwas, R.; Hupert, M.L.; Pullagurla, S.R.; Balamurugan, S.; Tamarit-López, J.; Park, S.; Datta, P.; Goettert, J.; Cho, Y.K.; Soper, S.A. Simple replication methods for producing nanoslits in thermoplastics and the transport dynamics of double-stranded DNA through these slits. Lab Chip 2010, 10, 3255–3264. [Google Scholar] [CrossRef][Green Version]
- Subramanian, B.; Kim, N.; Lee, W.; Spivak, D.A.; Nikitopoulos, D.E.; McCarley, R.L.; Soper, S.A. Surface modification of droplet polymeric microfluidic devices for the stable and continuous generation of aqueous droplets. Langmuir 2011, 27, 7949–7957. [Google Scholar] [CrossRef]
- Mirgissa, A.; Jeon, H.; Park, A.; Yi, K.; Baek, S.; Park, A.; Kim, D. Cavitation-microstreaming-based lysis and DNA extraction using a laser-machined polycarbonate microfluidic chip. Sensors Actuators B. Chem. 2021, 346, 130511. [Google Scholar] [CrossRef]
- Kulsharova, G.; Dimov, N.; Marques, M.P.C.; Szita, N.; Baganz, F. Simplified immobilisation method for histidine-tagged enzymes in poly(methyl methacrylate) microfluidic devices. N. Biotechnol. 2018, 47, 31–38. [Google Scholar] [CrossRef]
- Bhardwaj, T.; Jha, S. Microfluidic Platform for Aptamer based Fluorimetric Analysis of Analytes. In Biodevices; LingYin Information Technology Co., Ltd.: Beijing, China, 2018; pp. 218–223. [Google Scholar]
- Roberts, M.A.; Rossier, J.S.; Bercier, P.; Girault, H. UV laser machined polymer substrates for the development of microdiagnostic systems. Anal. Chem. 1997, 69, 2035–2042. [Google Scholar] [CrossRef]
- Liu, Y.; Zhong, W.; Meng, S.; Kong, J.; Lu, H.; Yang, P.; Girault, H.H.; Liu, B. Assembly-controlled biocompatible interface on a microchip: Strategy to highly efficient proteolysis. Chem.-A Eur. J. 2006, 12, 6585–6591. [Google Scholar] [CrossRef]
- Liu, Y.; Xue, Y.; Ji, J.; Chen, X.; Kong, J.; Yang, P.; Girault, H.H.; Liu, B. Gold nanoparticle assembly microfluidic reactor for efficient on-line proteolysis. Mol. Cell. Proteom. 2007, 6, 1428–1436. [Google Scholar] [CrossRef]
- Liu, K.; Xiang, J.; Ai, Z.; Zhang, S.; Fang, Y.; Chen, T.; Zhou, Q.; Li, S.; Wang, S.; Zhang, N. PMMA microfluidic chip fabrication using laser ablation and low temperature bonding with OCA film and LOCA. Microsyst. Technol. 2017, 23, 1937–1942. [Google Scholar] [CrossRef]
- Liu, S.; Fan, Y.; Gao, K.; Zhang, Y. Fabrication of Cyclo-olefin polymer-based microfluidic devices using CO2 laser ablation. Mater. Res. Express 2018, 5, 095305. [Google Scholar] [CrossRef]
- Paoli, R.; Di Giuseppe, D.; Badiola-Mateos, M.; Martinelli, E.; Lopez-Martinez, M.J.; Samitier, J. Rapid manufacturing of multilayered microfluidic devices for organ on a chip applications. Sensors 2021, 21, 1382. [Google Scholar] [CrossRef] [PubMed]
- Persson, H.; Park, S.; Mohan, M.; Cheung, K.K.; Simmons, C.A.; Young, E.W.K. Rapid assembly of PMMA microfluidic devices with PETE membranes for studying the endothelium. Sensors Actuators B Chem. 2022, 356, 131342. [Google Scholar] [CrossRef]
- Ongaro, A.E.; Di Giuseppe, D.; Kermanizadeh, A.; Crespo, A.M.; Mencattini, A.; Ghibelli, L.; Mancini, V.; Wlodarczyk, K.L.; Hand, D.P.; Martinelli, E.; et al. Polylactic is a Sustainable, Low Absorption, Low Auto fluorescence Alternative to Other Plastics for Microfluidic and Organ-on-Chip Applications. Anal. Biochem. 2020, 92, 6693–6701. [Google Scholar] [CrossRef]
- Demuru, S.; Haque, R.; Joho, M.O.; Bionaz, A.; van der Wal, P.; Briand, D. 3D-Integration of Printed Electrochemical Sensors in PET Microfluidics for Biochemical Sensing. In Proceedings of the 2019 20th International Conference on Solid-State Sensors, Actuators and Microsystems & Eurosensors XXXIII (TRANSDUCERS & EUROSENSORS XXXIII), Berlin, Germany, 23–27 June 2019; IEEE: Piscataway, NJ, USA, 2019; pp. 2464–2467. [Google Scholar]
- Jensen, M.F.; Noerholm, M.; Christensen, L.H.; Geschke, O. Microstructure fabrication with a CO2 laser system: Characterization and fabrication of cavities produced by raster scanning of the laser beam. Lab Chip 2003, 3, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Li, L.; Jiang, J.; Zhang, Y.; Chen, G.; Fan, Y. Reversible bonding for microfluidic devices with UV release tape. Microfluid. Nanofluid. 2022, 26, 23. [Google Scholar] [CrossRef]
- Vidya, S.; Wattal, R.; Singh, L.; Mathiyalagan, P. CO2 Laser Micromachining of Polymethyl Methacrylate (PMMA): A Review. In Advances in Manufacturing and Industrial Engineering; Singari, R.M., Mathiyazhagan, K., Kumar, H., Eds.; Springer: Singapore, 2021; pp. 939–945. [Google Scholar]
- Chai, M.; Cui, R.; Liu, J.; Zhang, Y.; Fan, Y. Polyformaldehyde-based microfluidics and application in enhanced oil recovery. Microsyst. Technol. 2021, 28, 947–954. [Google Scholar] [CrossRef]
- Chung, C.K.; Lin, Y.C.; Huang, G.R. Bulge formation and improvement of the polymer in CO2 laser micromachining. J. Micromech. Microeng. 2005, 15, 1878–1884. [Google Scholar] [CrossRef]
- Rossi, S.; Puglisi, A.; Benaglia, M. Additive Manufacturing Technologies: 3D Printing in Organic Synthesis. ChemCatChem 2018, 10, 1512–1525. [Google Scholar] [CrossRef]
- Padash, M.; Enz, C.; Carrara, S. Microfluidics by Additive Manufacturing for Wearable Biosensors: A Review. Sensors 2020, 20, 4236. [Google Scholar] [CrossRef]
- Stansbury, J.W.; Idacavage, M.J. 3D printing with polymers: Challenges among expanding options and opportunities. Dent. Mater. 2016, 32, 54–64. [Google Scholar] [CrossRef]
- Bhattacharjee, N.; Urrios, A.; Kang, S.; Folch, A. The upcoming 3D-printing revolution in microfluidics. Lab Chip 2016, 16, 1720–1742. [Google Scholar] [CrossRef]
- Gonzalez, G.; Roppolo, I.; Pirri, C.F.; Chiappone, A. Current and emerging trends in polymeric 3D printed microfluidic devices. Addit. Manuf. 2022, 55, 102867. [Google Scholar] [CrossRef]
- He, Y.; Wu, Y.; Fu, J.; Gao, Q.; Qiu, J. Developments of 3D Printing Microfluidics and Applications in Chemistry and Biology: A Review. Electroanalysis 2016, 28, 1658–1678. [Google Scholar] [CrossRef]
- Aschenbrenner, D.; Friedrich, O.; Gilbert, D.F. 3D Printed Lab-on-a-Chip Platform for Chemical Stimulation and Parallel Analysis of Ion Channel Function. Micromachines 2019, 10, 548. [Google Scholar] [CrossRef]
- Yeh, C.; Chen, Y.; Shie, M.; Fang, H. Poly(Dopamine)-Assisted Immobilization of Xu Duan on 3D Printed Poly(Lactic Acid) Scaffolds to Up-Regulate Osteogenic and Angiogenic Markers of Bone Marrow Stem Cells. Materials 2015, 8, 4299–4315. [Google Scholar] [CrossRef]
- Doronin, F.A.; Rudyak, Y.V.; Rytikov, G.O.; Evdokimov, A.G.; Nazarov, V.G. 3D-printed planar microfluidic device on oxyfluorinated PET-substrate. Polym. Test. 2021, 99, 107209. [Google Scholar] [CrossRef]
- Duong, L.H.; Chen, P. Simple and low-cost production of hybrid 3D-printed microfluidic devices. Biomicrofluidics 2019, 13, 024108. [Google Scholar] [CrossRef]
- Pokharna, P.P.; Ghantasala, M.K.; Rozhkova, E.A. 3D printed polylactic acid and acrylonitrile butadiene styrene fluidic structures for biological applications: Tailoring bio-material interface via surface modification. Mater. Today Commun. 2021, 27, 102348. [Google Scholar] [CrossRef]
- Khatri, A.; Dhawangale, A.; Mukherji, S. Single step, mould-free fabrication of polymer optical waveguides for localized surface plasmon resonance based sensing platform. Sensors Actuators B Chem. 2019, 280, 243–255. [Google Scholar] [CrossRef]
- Shaffer, S.; Yang, K.; Vargas, J.; Di Prima, M.A.; Voit, W. On reducing anisotropy in 3D printed polymers via ionizing radiation. Polymer 2014, 55, 5969–5979. [Google Scholar] [CrossRef]
- Johnson, B.N.; Lancaster, K.Z.; Hogue, I.B.; Meng, F.; Kong, Y.L.; Enquist, L.W.; McAlpine, M.C. 3D printed nervous system on a chip. Lab Chip 2016, 16, 1393–1400. [Google Scholar] [CrossRef] [PubMed]
- Mi, S.; Du, Z.; Xu, Y.; Sun, W. The crossing and integration between microfluidic technology and 3D printing for organ-on-chips. J. Mater. Chem. B 2018, 6, 6191–6206. [Google Scholar] [CrossRef] [PubMed]
- Dong, T.; Pires, N.M.M. Immunodetection of salivary biomarkers by an optical microfluidic biosensor with polyethylenimine-modified polythiophene-C70 organic photodetectors. Biosens. Bioelectron. 2017, 94, 321–327. [Google Scholar] [CrossRef] [PubMed]
- Shah, P.; Fritz, J.V.; Glaab, E.; Desai, M.S.; Greenhalgh, K.; Frachet, A.; Niegowska, M.; Estes, M.; Jäger, C.; Seguin-Devaux, C.; et al. A microfluidics-based in vitro model of the gastrointestinal human-microbe interface. Nat. Commun. 2016, 7, 11535. [Google Scholar] [CrossRef] [PubMed]
- Kendall, E.L.; Wienhold, E.; Rahmanian, O.D.; Devoe, D.L. Ex situ integration of multifunctional porous polymer monoliths into thermoplastic microfluidic chips. Sens. Actuators B. Chem. 2014, 202, 866–872. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Garg, M.; Christensen, M.G.; Iles, A.; Sharma, A.L.; Singh, S.; Pamme, N. Microfluidic-Based Electrochemical Immunosensing of Ferritin. Biosensors 2020, 10, 91. [Google Scholar] [CrossRef]
- Bruijns, B.; Veciana, A.; Tiggelaar, R.; Gardeniers, H. Cyclic Olefin Copolymer Microfluidic Devices for Forensic Applications. Biosensors 2019, 9, 85. [Google Scholar] [CrossRef]
- Le, N.X.T.; Trinh, K.T.L.; Lee, N.Y. Poly(acrylic acid) as an adhesion promoter for UV-assisted thermoplastic bonding: Application for the in vitro construction of human blood vessels. Mater. Sci. Eng. C 2021, 122, 111874. [Google Scholar] [CrossRef]
- Liu, J.; Sun, X.; Lee, M.L. Adsorption-resistant acrylic copolymer for prototyping of microfluidic devices for proteins and peptides. Anal. Chem. 2007, 79, 1926–1931. [Google Scholar] [CrossRef]
- Horak, J.; Dincer, C.; Bakirci, H.; Urban, G. A disposable dry film photoresist-based microcapillary immunosensor chip for rapid detection of Epstein-Barr virus infection. Sens. Actuators B Chem. 2014, 191, 813–820. [Google Scholar] [CrossRef]
- Kim, P.; Jeong, H.E.; Khademhosseini, A.; Suh, K.Y. Fabrication of non-biofouling polyethylene glycol micro- and nanochannels by ultraviolet-assisted irreversible sealing. Lab Chip 2006, 6, 1432–1437. [Google Scholar] [CrossRef]
- Tian, F.; Lyu, J.; Shi, J.; Tan, F.; Yang, M. A polymeric microfluidic device integrated with nanoporous alumina membranes for simultaneous detection of multiple foodborne pathogens. Sens. Actuators B Chem. 2016, 225, 312–318. [Google Scholar] [CrossRef]
- Tomecka, E.; Zukowski, K.; Jastrzebska, E.; Chudy, M.; Brzozka, Z. Microsystem with micropillar array for three- (gel-embaded) and two-dimensional cardiac cell culture. Sens. Actuators B. Chem. 2018, 254, 973–983. [Google Scholar] [CrossRef]
- Ford, S.M.; Davies, J.; Kar, B.; Qi, S.D.; Mcwhorter, S.; Soper, S.A.; Malek, C.K. Micromachining in Plastics Using X-Ray Lithography for the Fabrication of Micro- Electrophoresis Devices. J. Biomech. Eng. 1999, 121, 13–21. [Google Scholar] [CrossRef]
- Ford, S.M.; Kar, B.; McWhorter, S.; Davies, J.; Soper, S.A.; Klopf, M.; Calderon, G.; Saile, V. Microcapillary Electrophoresis Devices Fabricated Using Polymeric Substrates and X-ray Lithography. J. Microcolumn Sep. 1998, 10, 413–422. [Google Scholar] [CrossRef]
- Chandrasekaran, A.; Kalashnikov, N.; Rayes, R.; Wang, C.; Spicer, J.; Moraes, C. Thermal scribing to prototype plastic microfluidic devices, applied to study the formation of neutrophil extracellular traps. Lab Chip 2017, 17, 2003–2012. [Google Scholar] [CrossRef]
- Roos, M.M.; Winkler, A.; Nilsen, M.; Menzel, S.B.; Strehle, S. Towards Green 3D-Microfabrication of Bio-MEMS Devices Using ADEX Dry Film Photoresists. Int. J. Precis. Eng. Manuf. Technol. 2021, 9, 43–57. [Google Scholar] [CrossRef]
- Xie, X.; Maharjan, S.; Liu, S.; Zhang, Y.S.; Livermore, C. A Modular, Reconfigurable Microfabricated Assembly Platform for Microfluidic Transport and Multitype Cell Culture and Drug Testing. Micromachines 2020, 11, 2. [Google Scholar] [CrossRef]
- Cao, Y.; Floehr, J.; Ingebrandt, S.; Schnakenberg, U. Dry Film Resist Laminated Microfluidic System for Electrical Impedance Measurements. Micromachines 2021, 12, 632. [Google Scholar] [CrossRef]
- Trantidou, T.; Regoutz, A.; Voon, X.N.; Payne, D.J.; Ces, O. A “cleanroom-free” and scalable manufacturing technology for the microfluidic generation of lipid-stabilized droplets and cell-sized multisomes. Sens. Actuators B Chem. 2018, 267, 34–41. [Google Scholar] [CrossRef]
- El Fissi, L.; Vandormael, D.; Francis, L.A. Direct assembly of cyclic olefin copolymer microfluidic devices helped by dry photoresist. Sens. Actuators A Phys. 2015, 223, 76–83. [Google Scholar] [CrossRef]
- Leech, P.W.; Wu, N.; Zhu, Y. Application of dry film resist in the fabrication of microfluidic chips for droplet generation. J. Micromech. Microeng. 2009, 19, 65019. [Google Scholar] [CrossRef]
- Paul, D.; Saias, L.; Pedinotti, J.-C.; Chabert, M.; Magnifico, S.; Pallandre, A.; De Lambert, B.; Houdayer, C.; Brugg, B.; Peyrin, J.-M.; et al. A “dry and wet hybrid” lithography technique for multilevel replication templates: Applications to microfluidic neuron culture and two-phase global mixing. Biomicrofluidics 2011, 5, 24102. [Google Scholar] [CrossRef] [PubMed]
- Thompson, B.L.; Ouyang, Y.; Duarte, G.R.M.; Carrilho, E.; Krauss, S.T.; Landers, J.P. Inexpensive, rapid prototyping of microfluidic devices using overhead transparencies and a laser print, cut and laminate fabrication method. Nat. Protoc. 2015, 10, 875–886. [Google Scholar] [CrossRef]
- Welle, A.; Gottwald, E. UV-Based Patterning of Polymeric Substrates for Cell Culture Applications. Biomed. Microdevices 2002, 4, 33–41. [Google Scholar] [CrossRef]
- Didar, T.F.; Li, K.; Tabrizian, M.; Veres, T. High throughput multilayer microfluidic particle separation platform using embedded thermoplastic-based micropumping. Lab Chip 2013, 13, 2615–2622. [Google Scholar] [CrossRef]
- Keller, N.; Nargang, T.M.; Runck, M.; Kotz, F.; Striegel, A.; Sachsenheimer, K.; Klemm, D.; Länge, K.; Worgull, M.; Richter, C.; et al. Tacky cyclic olefin copolymer: A biocompatible bonding technique for the fabrication of microfluidic channels in COC. Lab Chip 2016, 16, 1561–1564. [Google Scholar] [CrossRef]
- Im, S.G.; Bong, K.W.; Lee, C.H.; Doyle, P.S.; Gleason, K.K. A conformal nano-adhesive via initiated chemical vapor deposition for microfluidic devices. Lab Chip 2009, 9, 411–416. [Google Scholar] [CrossRef]
- Xu, J.; Gleason, K.K. Conformal, amine-functionalized thin films by initiated chemical vapor deposition (iCVD) for hydrolytically stable microfluidic devices. Chem. Mater. 2010, 22, 1732–1738. [Google Scholar] [CrossRef]
- Ma, H.L.; Urbaczek, A.C.; de Souza, F.Z.R.; Leão, P.A.G.G.C.; Perussi, J.R.; Carrilho, E. Rapid Fabrication of Microfluidic Devices for Biological Mimicking: A Survey of Materials and Biocompatibility. Micromachines 2021, 12, 346. [Google Scholar] [CrossRef]
- Gleichweit, E.; Baumgartner, C.; Diethardt, R.; Murer, A.; Sallegger, W.; Werkl, D.; Köstler, S. UV/Ozone Surface Treatment for Bonding of Elastomeric COC-Based Microfluidic Devices. Proceedings 2018, 2, 943. [Google Scholar] [CrossRef]
- Yu, S.; Ng, S.P.; Wang, Z.; Tham, C.L.; Soh, Y.C. Thermal bonding of thermoplastic elastomer film to PMMA for microfluidic applications. Surf. Coatings Technol. 2017, 320, 437–440. [Google Scholar] [CrossRef]
- Uba, F.I.; Pullagurla, S.R.; Sirasunthorn, N.; Wu, J.; Park, S.; Chantiwas, R.; Cho, Y.-K.; Shin, H.; Soper, S.A. Surface charge, electroosmotic flow and DNA extension in chemically modified thermoplastic nanoslits and nanochannels. Analyst 2015, 140, 113–126. [Google Scholar] [CrossRef]
- Bianco, M.; Zizzari, A.; Gazzera, L.; Metrangolo, P.; Gigli, G.; Viola, I.; Arima, V. Integrated microfluidic viscometer for edible oil analysis. Sens. Actuators B. Chem. 2018, 265, 91–97. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, L.; Guo, Y.; Zhu, Y.; Qin, J. Engineering stem cell-derived 3D brain organoids in a perfusable organ-on-a-chip system. RSC Adv. 2018, 8, 1677–1685. [Google Scholar] [CrossRef]
- Wan, A.M.D.; Sadri, A.; Young, E.W.K. Liquid phase solvent bonding of plastic microfluidic devices assisted by retention grooves. Lab Chip 2015, 15, 3785–3792. [Google Scholar] [CrossRef]
- Hansen, C.M.; Just, L. Prediction of Environmental Stress Cracking in Plastics with Hansen Solubility Parameters. Ind. Eng. Chem. Res. 2001, 40, 21–25. [Google Scholar] [CrossRef]
- Duong, L.H.; Chen, P. Novel solvent bonding method for creation of a three-dimensional, non-planar, hybrid PLA/PMMA microfluidic chip. Sens. Actuators A Phys. 2018, 280, 350–358. [Google Scholar] [CrossRef]
- Pečar, B.; Možek, M.; Vrtačnik, D. Thermoplastic-PDMS polymer covalent bonding for microfluidic applications. Inf. MIDEM 2017, 47, 147–154. [Google Scholar]
- Sivakumar, R.; Lee, N.Y. Chemically robust succinimide-group-assisted irreversible bonding of poly(dimethylsiloxane)–thermoplastic microfluidic devices at room temperature. Analyst 2020, 145, 6887–6894. [Google Scholar] [CrossRef]
- Aran, K.; Sasso, L.A.; Kamdar, N.; Zahn, J.D. Irreversible, direct bonding of nanoporous polymer membranes to PDMS or glass microdevices. Lab Chip 2010, 10, 548–552. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.Y.; McClelland, A.A.; Chen, Z.; Lahann, J. Solventless adhesive bonding using reactive polymer coatings. Anal. Chem. 2008, 80, 4119–4124. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.J.; Pak, K.; Kwak, M.J.; Joo, M.; Kim, B.J.; Oh, M.S.; Baek, J.; Park, H.; Choi, G.; Kim, D.H.; et al. Initiated Chemical Vapor Deposition: A Versatile Tool for Various Device Applications. Adv. Eng. Mater. 2018, 20, 1700622. [Google Scholar] [CrossRef]
- Liang, C.; Meng, F.; Li, J.; Liu, C. Using CO2-laser bugle for ultrasonic bonding of thermoplastic microfluidic devices. J. Mater. Process. Tech. 2018, 252, 25–33. [Google Scholar] [CrossRef]
- Finkbeiner, T.; Soergel, H.L.; Koschitzky, M.P.; Ahrens, R.; Guber, A.E. Ultrasonic welding for the rapid integration of fluidic connectors into microfluidic chips. J. Micromech. Microeng. 2019, 29, 065011. [Google Scholar] [CrossRef]
- Acherjee, B.; Kuar, A.S.; Mitra, S.; Misra, D.; Acharyya, S. Experimental investigation on laser transmission welding of PMMA to ABS via response surface modeling. Opt. Laser Technol. 2012, 44, 1372–1383. [Google Scholar] [CrossRef]
- Juhl, T.B.; Christiansen, J.D.C.; Jensen, E.A. Mechanical testing of polystyrene/polystyrene laser welds. Polym. Test. 2013, 32, 475–481. [Google Scholar] [CrossRef]
- Chen, M.; Zak, G.; Bates, P.J. Effect of carbon black on light transmission in laser welding of thermoplastics. J. Mater. Process. Tech. 2011, 211, 43–47. [Google Scholar] [CrossRef]
- Nguyen, N.; Brosda, M.; Olowinsky, A.; Gillner, A. Absorber-Free Quasi-Simultaneous Laser Welding For Microfluidic Applications. J. Laser Micro/Nanoeng. 2019, 14, 255–261. [Google Scholar] [CrossRef]
- de Pelsmaeker, J.; Graulus, G.; Van Vlierberghe, S.; Thienpont, H.; Van Hemelrijck, D.; Dubruel, P.; Ottevaere, H. Clear to clear laser welding for joining thermoplastic polymers: A comparative study based on physicochemical characterization. J. Mater. Process. Tech. 2018, 255, 808–815. [Google Scholar] [CrossRef]
- Schütte, J.; Freudigmann, C.; Benz, K.; Böttger, J.; Gebhardt, R.; Stelzle, M. A method for patterned in situ biofunctionalization in injection-molded microfluidic devices. Lab Chip 2010, 10, 2551–2558. [Google Scholar] [CrossRef]
- Kratz, S.R.A.; Eilenberger, C.; Schuller, P.; Bachmann, B.; Spitz, S.; Ertl, P.; Rothbauer, M. Characterization of four functional biocompatible pressure-sensitive adhesives for rapid prototyping of cell-based lab-on-a-chip and organ-on-a-chip systems. Sci. Rep. 2019, 9, 9287. [Google Scholar] [CrossRef]
- Badv, M.; Alonso-Cantu, C.; Shakeri, A.; Hosseinidoust, Z.; Weitz, J.I.; Didar, T.F. Biofunctional Lubricant-Infused Vascular Grafts Functionalized with Silanized Bio-Inks Suppress Thrombin Generation and Promote Endothelialization. ACS Biomater. Sci. Eng. 2019, 5, 6485–6496. [Google Scholar] [CrossRef]
- Roy, E.; Galas, J.-C.; Veres, T. Thermoplastic elastomers for microfluidics: Towards a high-throughput fabrication method of multilayered microfluidic devices. Lab Chip 2011, 11, 3193–3196. [Google Scholar] [CrossRef]
- Sathish, S.; Toda-peters, K.; Shen, A.Q. Proof-of-concept modular fluid handling prototype integrated with microfluidic biochemical assay modules for point-of-care. VIEW 2020, 1, e1. [Google Scholar] [CrossRef]
- Darain, F.; Gan, K.L.; Tjin, S.C. Antibody immobilization on to polystyrene substrate-On-chip immunoassay for horse IgG based on fluorescence. Biomed. Microdevices 2009, 11, 653–661. [Google Scholar] [CrossRef]
- Darain, F.; Yager, P.; Gan, K.L.; Tjin, S.C. On-chip detection of myoglobin based on fluorescence. Biosens. Bioelectron. 2009, 24, 1744–1750. [Google Scholar] [CrossRef]
- Tsao, C.; Syu, W. Bonding of thermoplastic microfluidics by using dry adhesive tape. RSC Adv. 2020, 10, 30289–30296. [Google Scholar] [CrossRef]
- Arayanarakool, R.; Le Gac, S.; Van Den Berg, A. Low-temperature, simple and fast integration technique of microfluidic chips by using a UV-curable adhesive. Lab Chip 2010, 10, 2115–2121. [Google Scholar] [CrossRef]
- Jeong, S.; Kim, S.; Buonocore, J.; Park, J.; Welsh, C.J.; Li, J.; Han, A. A Three-Dimensional Arrayed Microfluidic Blood–Brain Barrier Model With Integrated Electrical Sensor Array. IEEE Trans. Biomed. Eng. 2018, 65, 431–439. [Google Scholar] [CrossRef]
- Sun, Y.; Teng, T.; Guo, G.; Wu, G. Ultrasonic bonding method controlled by the characteristic waveform of ultrasonic propagation. Micro Nano Lett. 2019, 14, 547–550. [Google Scholar] [CrossRef]
- Li, S.W.; Xu, J.H.; Wang, Y.J.; Lu, Y.C.; Luo, G. Low-temperature bonding of poly-(methyl methacrylate) microfluidic devices under an ultrasonic field. J. Micromech. Microeng. 2008, 19, 15035. [Google Scholar] [CrossRef]
- Kistrup, K.; Poulsen, C.E.; Hansen, M.F.; Wolff, A. Ultrasonic welding for fast bonding of self-aligned structures in lab-on-a-chip systems. Lab Chip 2015, 15, 1998–2001. [Google Scholar] [CrossRef]
- Yu, H.; Tor, S.B.; Loh, N.H. Rapid bonding enhancement by auxiliary ultrasonic actuation for the fabrication of cyclic olefin copolymer (COC) microfluidic devices. J. Micromech. Microeng. 2014, 24, 115020. [Google Scholar] [CrossRef]
- Jiang, X.; Chandrasekar, S.; Wang, C. A laser microwelding method for assembly of polymer based microfluidic devices. Opt. Lasers Eng. 2015, 66, 98–104. [Google Scholar] [CrossRef]
- Roth, G.L.; Esen, C.; Hellmann, R. A new approach to seal polymer microfluidic devices using ultrashort laser pulses. J. Laser Micro Nanoeng. 2019, 14, 49–53. [Google Scholar] [CrossRef]
- Podbiel, D.; Boecking, L.; Bott, H.; Kassel, J.; Czurratis, D.; Laermer, F.; Zengerle, R.; Hoffmann, J. From CAD to microfluidic chip within one day: Rapid prototyping of lab-on-chip cartridges using generic polymer parts. J. Micromech. Microeng. 2020, 30, 115012. [Google Scholar] [CrossRef]
- Bot, V.A.; Shakeri, A.; Weitz, J.I.; Didar, T.F. A Vascular Graft On-a-Chip Platform for Assessing the Thrombogenicity of Vascular Prosthesis and Coatings with Tuneable Flow and Surface Conditions. Adv. Funct. Mater. 2022, 2205078. [Google Scholar] [CrossRef]
- Villegas, M.; Cetinic, Z.; Shakeri, A.; Didar, T.F. Fabricating smooth PDMS microfluidic channels from low-resolution 3D printed molds using an omniphobic lubricant-infused coating. Anal. Chim. Acta 2018, 1000, 248–255. [Google Scholar] [CrossRef]
- Beckwith, A.L.; Borenstein, J.T.; Velásquez-García, L.F. Monolithic, 3D-Printed Microfluidic Platform for Recapitulation of Dynamic Tumor Microenvironments. J. Microelectromech. Syst. 2018, 27, 1009–1022. [Google Scholar] [CrossRef]
- Rajan, S.K.; Kaul, A.; Sarvey, T.; May, G.S.; Bakir, M.S. Design Considerations, Demonstration, and Benchmarking of Silicon Microcold Plate and Monolithic Microfluidic Cooling for 2.5D ICs. In Proceedings of the 2021 IEEE 71st Electronic Components and Technology Conference (ECTC), San Diego, CA, USA, 1–4 June 2021; IEEE: Piscataway, NJ, USA, 2021; pp. 1418–1426. [Google Scholar]
- Lindsay, M.; Bishop, K.; Sengupta, S.; Co, M.; Cumbie, M.; Chen, C.; Johnston, M.L. Heterogeneous Integration of CMOS Sensors and Fluidic Networks Using Wafer-Level Molding. IEEE Trans. Biomed. Circuits Syst. 2018, 12, 1046–1055. [Google Scholar] [CrossRef]

| Method | Experimental Procedure | Effective Parameters | Advantages | Disadvantages | Examples |
|---|---|---|---|---|---|
| Hot embossing |
|
|
|
| PMMA [22,32,53,55,56] COP/COC [19,20,22,24,26,27,28,36,54,55,56] PC [17,18,33,55,56] PS [21] TPEs [29,30,31] |
| Injection molding |
|
|
|
| PMMA [45] PC [25] COC [36,48,52] PS [46,47] |
| Laser ablation |
|
|
|
| PMMA [58,59,63,65,66,69,70,71,73] PC [57,60] PS [60] PET [60,61,62,68,110] PETE [66] COP [64,65] PLA [67] POM [72] |
| Micro milling |
|
|
|
| PMMA [89,92,94,99] PC [90] COC [91,93] |
| UV-curing |
|
|
|
| PEG-based [95,97,98] PMMA [100,101] |
| Thermal scribing |
|
|
|
| PS [102] |
| 3D printing |
|
|
|
| ABS [80,83,84,85] PLA [81,82,84,86] COC [77] Acrylate-based [86] |
| Dry films |
|
|
|
| ADEX [103] SUEX [103,104,105] Ordyl [106] |
| Method | Experimental Procedure | Effective Parameters | Advantages | Disadvantages | Examples |
|---|---|---|---|---|---|
| Thermal fusion bonding |
|
|
|
| PMMA [30,58,89,118,119] PET [61,62,110] COC [20,24,36,117] COP [65] PC [17] PLA [67] TPE [30,118] Dry films [96] Polyester [116] |
| Solvent bonding |
|
|
|
| PMMA [32,59,66,83,124] COC [19,26,93,113,123] PC [25,57] PLA [124] ABS [83] PETE [66] |
| Chemical bonding |
|
|
|
| PMMA [125] COC [26,31,115] TPE [31] PS [31,114] PC [31,65,114,115,125] PET [114,115,126] PES [127] PETE [127] ABS [125] PA [115] PE [115] PTFE [114] |
| Ultrasonic welding |
|
|
|
| PMMA [130,147,148,149] COC [48,150] PC [131,149] PET [131] |
| Laser welding |
|
|
|
| PMMA [132,151] COC [152] PC [153] TPE [153] PS [134] ABS [132] |
| Adhesive layers |
|
|
|
| PMMA [63,65,70,94,141,154] COC [52,137] PET [68] Polyester [72] PTFE [154] POM [72] PS [142,143] PEG [95,97,98] PC [146] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shakeri, A.; Khan, S.; Jarad, N.A.; Didar, T.F. The Fabrication and Bonding of Thermoplastic Microfluidics: A Review. Materials 2022, 15, 6478. https://doi.org/10.3390/ma15186478
Shakeri A, Khan S, Jarad NA, Didar TF. The Fabrication and Bonding of Thermoplastic Microfluidics: A Review. Materials. 2022; 15(18):6478. https://doi.org/10.3390/ma15186478
Chicago/Turabian StyleShakeri, Amid, Shadman Khan, Noor Abu Jarad, and Tohid F. Didar. 2022. "The Fabrication and Bonding of Thermoplastic Microfluidics: A Review" Materials 15, no. 18: 6478. https://doi.org/10.3390/ma15186478
APA StyleShakeri, A., Khan, S., Jarad, N. A., & Didar, T. F. (2022). The Fabrication and Bonding of Thermoplastic Microfluidics: A Review. Materials, 15(18), 6478. https://doi.org/10.3390/ma15186478






