A Comprehensive Review of Biodegradable Polymer-Based Films and Coatings and Their Food Packaging Applications
Abstract
1. Introduction
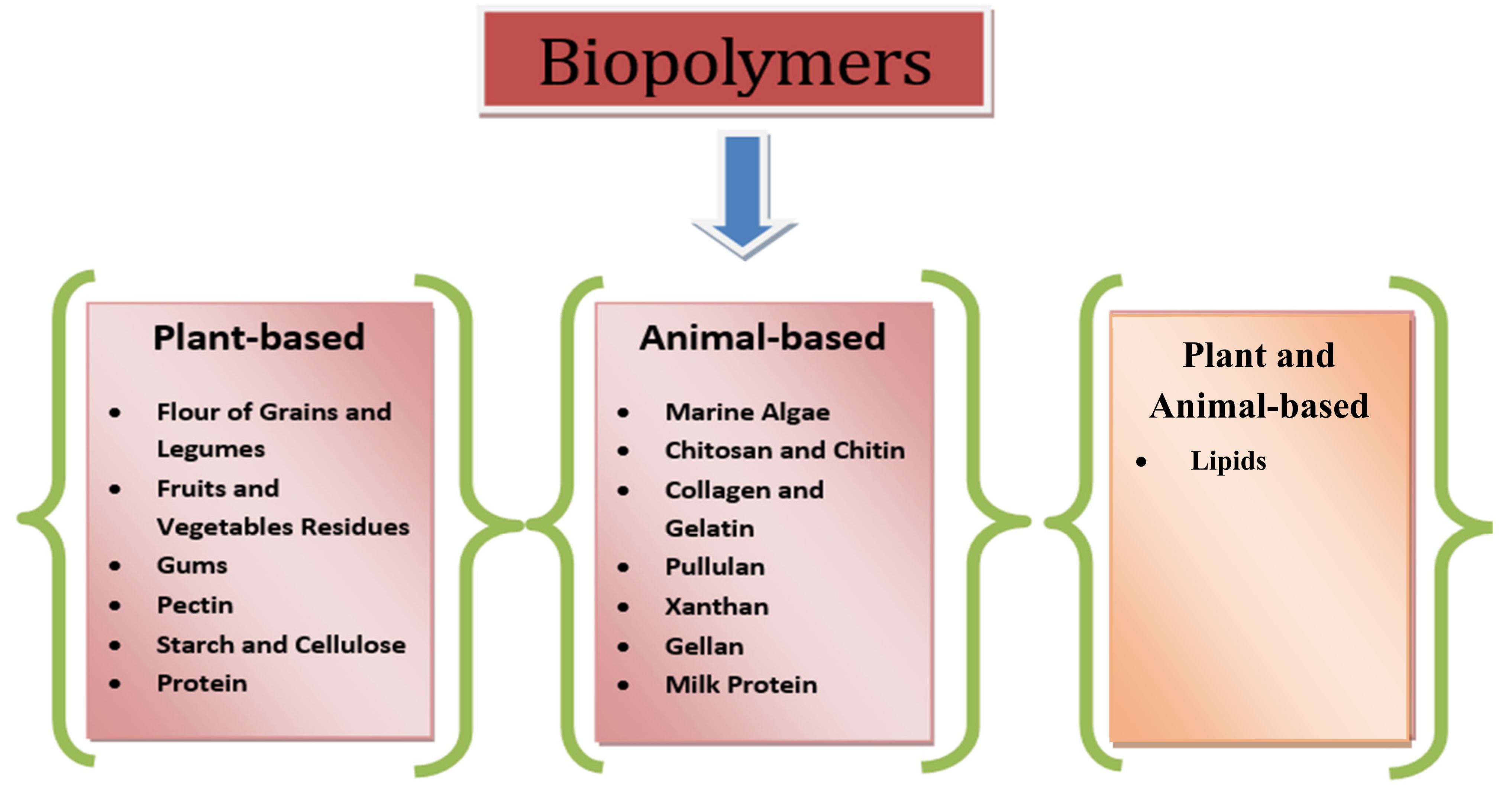
2. Sources of Biodegradable Material
2.1. Plant-Based Material
2.1.1. Flour of Grains and Legumes
2.1.2. Fruits and Vegetables Residues
2.1.3. Gums
2.1.4. Pectin
2.1.5. Starch and Cellulose
2.1.6. Proteins
2.2. Animal-Based Material
2.2.1. Marine Algae
2.2.2. Chitosan and Chitin
2.2.3. Collagen and Gelatin
2.2.4. Pullulan
2.2.5. Xanthan
2.2.6. Gellan
2.2.7. Milk Protein
2.3. Other Materials
Lipids
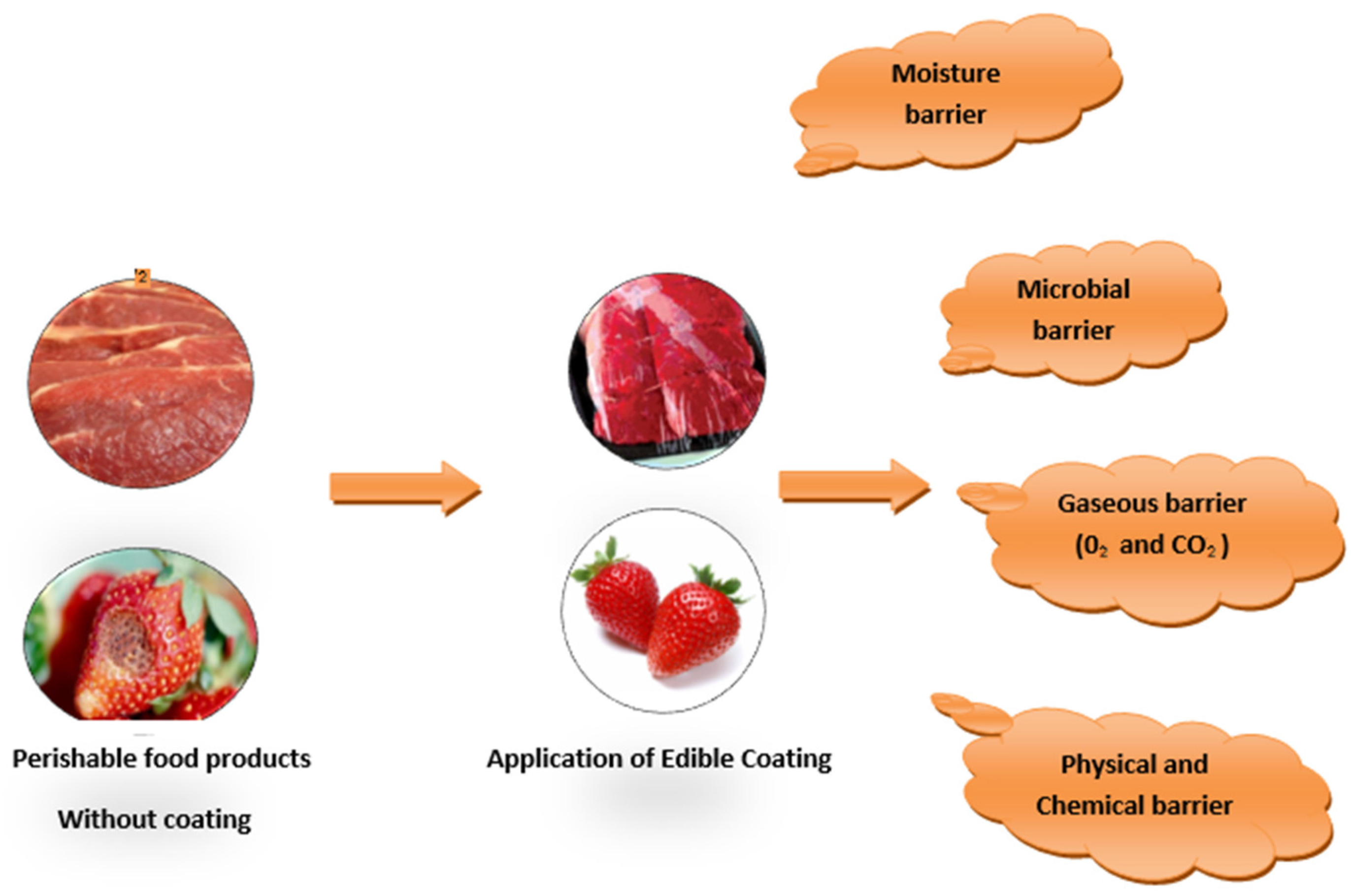
3. Functional Material for Edible Films and Coatings
3.1. Nanomaterials
3.2. Natural Bioactive Material
3.3. Essential Oils (EOs)
4. Fabrication of Edible Packaging Materials
| Biomaterial | Source/Derivative | Properties | Applications | References |
|---|---|---|---|---|
| Flour of grains and legumes | Corn starch, chickpeas, lentils, etc. | High content of starch and protein results in good film-forming properties. | Used as an edible film for wrapping foods. | [13] |
| Fruit and vegetable residues | Peel, pomace, seed fraction, etc. | Films are malleable, water soluble, improved mechanical and barrier properties, enhanced mechanical resistance. | Can be used as edible films and coatings for perishable food items. | [13] |
| Plant gums | Gum arabic, gum karaya, gum ghatti, mesquite, etc. | Potential material for edible film formation, exhibits good physical, chemical, biological properties. | Edible films and coatings on fruits such as strawberries, tomatoes, and pecan nuts. | [13] |
| Pectin | Cell wall of plants | Acts as a natural plasticizer, good mechanical properties, poor barrier properties. | Edible coating for fruits, vegetables, cheese, and meat products. | [24] |
| Starch | Corn, potato, wheat, rice, etc. | Tasteless, clear, O2 and CO2 barrier, not soluble in water, poor mechanical properties. | Wrapper and coatings after some modifications. | [174] |
| Cellulose | Carboxymethyl cellulose (CMC), cellulose acetate (CA), methyl cellulose, ethyl cellulose, hydroxypropyl, hydroxyethyl cellulose, cellophane, etc. | Not moisture resistant, good mechanical properties. | Used for packaging of meat products, confectionary, and cheese. | [77] |
| Protein | Wheat gluten and corn zein, soy protein | Water solubility, opacity, and mechanical and barrier properties, Not soluble in water, but absorb water when they are submerged. | Can be used after modifications, used in glues, dyes, and paper coatings. | [24,76] |
| Marine algae | Alginate, agar, carrageenan | Natural binder in coating. Poor water resistance, good barrier for oxygen, lipid, and oxidation of fat. | Edible coatings for fruits, vegetables, cheese, and meat products. | [77] |
| Chitosan | Exoskeleton of crustaceans, fungal cell walls, and other animal sources | Good mechanical properties, good barriers for oxygen transition, antimicrobial properties. | Used in fruit coatings, cellophane packaging. | [77] |
| Collagen and gelatin | Hair, skin, nails, bones, and ligaments of beef or fish | Translucent films, permeable to moisture. | Used in the coating of sausage and other meat products. | [29] |
| Pullulan | Microbiological source | Thermal and oil resistance, low gas barrier. | Used for food coatings. | [175] |
| Xanthan | Microbiological source | Excellent solubility, good rheological properties and stability. | Used in coatings for fruits such as strawberries, garden berries, and grapes. | [176] |
| Gellan | Microbiological source | Exhibits good gelling, colloidal, and antimicrobial properties, exhibits good water solubility. | Used in edible films and coatings for fruits and vegetables. | [176] |
| Milk proteins | Caseinate and whey protein | Casein-based films are opaque and water insoluble, but they absorb water, and have good mechanical properties; expensive. | Can be used to coat highly perishable foods such as meat products. | [177,178,179,180] |
| Lipid | Acetoglycerides, beeswax, surfactants, triglycerides, fatty acids | Weak mechanical properties, good barrier against moisture migration. | Edible coatings for fruit and meat products. | [77,181,182] |
| Natural bioactive material | Fruit waste, agricultural waste (antioxidant and antimicrobial compound) | Exhibits excellent antioxidant and antimicrobial properties. | Can be used as an active agent in edible films such as agar-based films. | [29] |
5. Fabrication of Composite Films and Coatings
5.1. Film Fabrication Methods
- Simple coacervation: This occurs when a hydrocolloid dispersed in water precipitates or changes phase after solvent evaporation (drying), after the addition of a hydro-soluble non-electrolyte in which the hydrocolloid is insoluble (e.g., ethanol), or after pH adjustment and the addition of an electrolyte that causes salting out or cross-linking.
- Complex coacervation: When two hydrocolloid solutions with opposite electron charges are combined, the polymer complex interacts and precipitates.
- Gelation or thermal coagulation: This is a process in which a macromolecule is heated, creating denaturation, and then gelatin (e.g., proteins such as ovalbumin) or precipitation, or even the cooling of a hydrocolloid dispersion, causing gelation (e.g., gelatin or agar).
5.1.1. Solvent Casting Method
5.1.2. Extrusion Method
6. Methods of Coating
6.1. Dip Coating
6.2. Spray Coating
6.3. Electro Spraying
6.3.1. Electrostatic Powder Coating
6.3.2. Electrostatic Liquid Coating
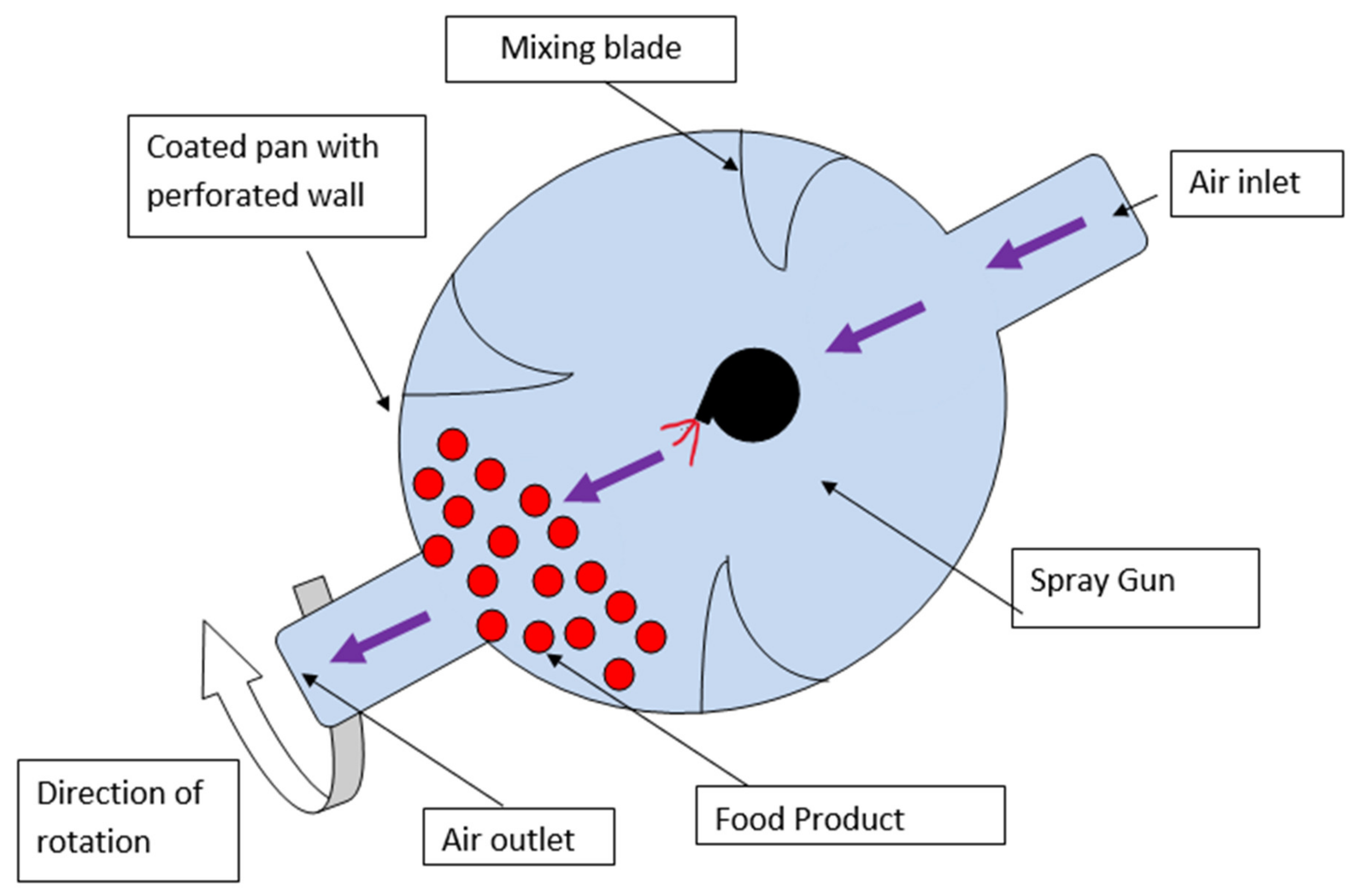
6.4. Panning Method
6.5. Fluidized Bed Processing Method
7. Application of Bio-Based Films and Coatings in Food
7.1. Application of Films
| Biomaterial | Food Product | Method | Key Results | References |
|---|---|---|---|---|
| Starch–alginate films with stearic acid | Ground beef patty | Casting | Improved barrier properties against moisture and helpful in preventing oxidation of lipids. | [135] |
| Chitosan with essential oil | Chicken fillet | Casting | Exhibits antimicrobial and inhibitory activities against spoilage-causing microorganism. | [239] |
| Carrageenan with olive leaf extracts | Lamb meat | Casting | Good antioxidant activity due to presence of phenol, exhibits antimicrobial activity against Escherichia coli, Coliform. Demonstrates lower water vapor permeability which enhances its shelf life. | [240] |
| Chickpea with gallic acid | Highly oxidative food | Casting | Antioxidant activity, phenolic content, demonstrates low water vapor permeability and good mechanical properties. | [36] |
| Gelatin/cellulose nanofibril with Ag nanoparticle and glycerol | Fruits and vegetables | Casting | Good antimicrobial activity against various microorganisms such as E. coli and S. aureus. | [241] |
| Corn starch with glycerol | Mango | Extrusion | Able to maintain physical and chemical qualities of mango for up to 16 days at 12 °C. | [242] |
| Whey protein isolate with oregano essential oil, garlic oil, nisin, natamycin | Kasar cheese | Casting | Effective against Listeria monocytogenes, staphylococcus aureus and Escherichia coli. | [243] |
| Zein/gelatin with tea polyphenol | Fruits and vegetables | Casting | Antimicrobial properties, prevents browning, and controls weight loss in fruits and vegetables. | [244] |
| Tapioca Starch/Chitosan Nanoparticles | Cherry tomatoes | Casting | Antimicrobial property against gram positive bacteria, improves shelf life. | [245] |
| Sodium-caseinate | Extrusion | Good mechanical properties, water vapor permeability, water soluble. | [246] | |
| Sweet potato starch | Baby spinach leaf | Casting | Antimicrobial against E. coli and S. typhimurium | [247] |
7.2. Application of Coatings
| Biopolymer | Active Ingredient | Results | References |
|---|---|---|---|
| Coconut protein and guar gum | Transglutaminase enzyme (oxygen scavenger) | Better barrier properties, improved mechanical properties, water vapor permeability and oxygen transfer rates. | [253] |
| Chitosan | Ascorbic acid | Suppresses browning activity and prolongs microbial and chemical shelf life of freshly cut apples. | [254] |
| Starch | Adiantum capillus-veneris extract | Helps in preservation and microbial treatment of freshly cut apples. | [255] |
| Gelatin, chitosan, and cassava starch | Hibiscus rosa-sinensis (plant mucilage) | Antimicrobial properties. | [256] |
| Carboxymethyl cellulose | Impatiens balsamina L. extract | Coating on “Xinyu” tangerines shows a delay in ripening, decreased decay rate, increased antioxidant activity. | [257] |
| Alginate | Green tea extract | Improves safety in strawberries and raspberries against food-borne pathogens. | [258] |
| Aloe vera extract (pure and diluted) | Applied on tomatoes; pure extract inhibits the growth of bacteria while diluted extract maintains the quality of tomatoes during storage. It also helps to delay ripening. | [259] | |
| Chitosan | Nano emulsion with lemon oil extract | Increased shelf life of food product up to 7 days. | [260] |
| Carrageenan | Ascorbic acid/citric acid/ oxalic acid | Coating applied to apples; possess antifungal and antioxidant activities. Improves color and firmness of product. | [121] |
| Gum Arabic | Lemongrass oil and cinnamon oil | Shows antimicrobial activity on coated fruits such as banana and papaya. | [71] |
| Candelilla wax | Mineral oil | Prevents weight loss and helps in the prevention of loss of color; retain firmness of fruits such as guava and persian lime. | [24] |
| Lipid | Stearic acid, polyglycerol polyricinoleate and butter | Provides excellent barrier against moisture in hygroscopic candy tablets. | [24] |
| Chitosan and pectin | Trans-cinnamaldehyde, beta-cyclodextrin hydrate | Helps in extending shelf life of freshly cut cantaloupe at 4 °C. | [261] |
| Pectin and sodium alginate | Essential oil | Prevents weight loss, demonstrates antioxidant and antimicrobial activity, improves sensory characteristics in fruits such as raspberries and prevents discoloration. | [57] |
| Chitosan and alginate | Resveratrol | Antioxidant and antimicrobial activity, prevents oxidation in smoked sea bass fillets. | [262] |
| Chitosan blended with gelatin | - | Effective antimicrobial activity against S. aureus and demonstrated a considerable decrease in count of aerobic and total coliform in perishable foods such as beef. | [263] |
| Carboxy methyl cellulose | Apple peel (1%) and tartaric acid (0.75%) | Prevents lipid oxidation and shows significant decrease in aerobic plate, yeast, and mold in fresh beef patties. Additionally, no negative impact on sensory characteristics is observed. | [264] |
| Carboxy methyl cellulose | Ascorbic acid | Improved water vapor resistance, significant reduction in polyphenol oxidase activity, able to retain freshness of carrots during storage period. | [265] |
| Gelatin and Glucose | Sorbitol and cysteine | Acts as probiotic coating on hake fish due to presence of Lactobacillus acidophilus and Bifidobacterium bifidum, resulting in enhancing shelf life up to 15 days at refrigerated conditions. | [266] |
| Cassava starch, chitosan and gallic acid | Glycerol | Increased shelf life of ham slices. | [267] |
| K-Carrageenan and chitosan | Glycerol and oriental mustard extract | Reduced contamination of Campylobacter jejuni in fresh chicken breast along with enhancement of shelf life. | [268] |
| Hydroxypropyl methyl cellulose and chitosan | Bergamot essential oil | Observes inhibitory action and control over respiration rate and weight loss on cold-stored grapes. | [269] |
8. Conclusions and Future Perspective
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Díaz-Montes, E.; Castro-Muñoz, R. Edible Films and Coatings as Food-Quality Preservers: An Overview. Foods 2021, 10, 249. [Google Scholar] [CrossRef] [PubMed]
- Versino, F.; Lopez, O.V.; Garcia, M.A.; Zaritzky, N.E. Starch-Based Films and Food Coatings: An Overview. Starch-Staerke 2016, 68, 1026–1037. [Google Scholar] [CrossRef]
- Derraik, J. The Pollution of the Marine Environment by Plastic Debris: A Review. Mar. Pollut. Bull. 2002, 44, 842–852. [Google Scholar] [CrossRef]
- Kumbhar, V.; Masali, P. Biodegradable Cutlery Using MoringaPod Husk: An Alternative to Conventional Plastic Cutlery. Int. J. Innov. Sci. Res. Technol. 2020, 5, 900–903. [Google Scholar]
- Garside, M. Global Plastic Production from 1950 to 2018 (in Million Metric Tons). Statista 2019, 2018. [Google Scholar]
- Porta, R. Plastic Pollution and the Challenge of Bioplastics. J. Appl. Biotechnol. Bioeng. 2017, 2, 111. [Google Scholar] [CrossRef]
- De Souza Machado, A.A.; Kloas, W.; Zarfl, C.; Hempel, S.; Rillig, M.C. Microplastics as an Emerging Threat to Terrestrial Ecosystems. Glob. Chang. Biol. 2018, 24, 1405–1416. [Google Scholar] [CrossRef]
- Li, W.C.; Tse, H.F.; Fok, L. Plastic Waste in the Marine Environment: A Review of Sources, Occurrence and Effects. Sci. Total Environ. 2016, 566–567, 333–349. [Google Scholar] [CrossRef]
- Prashanthi, M.; Rajakumar, S. Integrated Water Manageent in India: Status and Future Prospects for Environmental Sustainability; Springer: Berlin/Heidelberg, Germany, 2016; ISBN 978-3-319-27226-9. [Google Scholar]
- Patil, H.N.; Sinhal, P. A Study on Edible Cutlery: An Alternative for Conventional Ones. Atithya J. Hosp. 2018, 4, 45–51. [Google Scholar]
- United Nations. World Population Prospects: The 2019 Revision, Highlights, Departement of Economic and Social Affairs; United Nations: New York, NY, USA, 2019. [Google Scholar]
- Sarkodie, S.A.; Strezov, V.; Weldekidan, H.; Asamoah, E.F.; Owusu, P.A.; Doyi, I.N.Y. Environmental Sustainability Assessment Using Dynamic Autoregressive-Distributed Lag Simulations—Nexus between Greenhouse Gas Emissions, Biomass Energy, Food and Economic Growth. Sci. Total Environ. 2019, 668, 318–332. [Google Scholar] [CrossRef]
- Galus, S.; Kibar, E.A.A.; Gniewosz, M.; Kraśniewska, K. Novel Materials in the Preparation of Edible Films and Coatings-A Review. Coatings 2020, 10, 674. [Google Scholar] [CrossRef]
- Jooyandeh, H. Whey Protein Films and Coatings: A Review. Pak. J. Nutr. 2011, 10, 296–301. [Google Scholar] [CrossRef]
- Kumar, L.; Ramakanth, D.; Akhila, K.; Gaikwad, K.K. Edible Films and Coatings for Food Packaging Applications: A Review. Environ. Chem. Lett. 2021, 20, 875–900. [Google Scholar] [CrossRef]
- Asgher, M.; Qamar, S.A.; Bilal, M.; Iqbal, H.M.N. Bio-Based Active Food Packaging Materials: Sustainable Alternative to Conventional Petrochemical-Based Packaging Materials. Food Res. Int. 2020, 137, 109625. [Google Scholar] [CrossRef]
- Trajkovska Petkoska, A.; Daniloski, D.; D’Cunha, N.M.; Naumovski, N.; Broach, A.T. Edible Packaging: Sustainable Solutions and Novel Trends in Food Packaging. Food Res. Int. 2021, 140, 109981. [Google Scholar] [CrossRef]
- Saucedo-Pompa, S.; Rojas-Molina, R.; Aguilera-Carbó, A.F.; Saenz-Galindo, A.; de La Garza, H.; Jasso-Cantú, D.; Aguilar, C.N. Edible Film Based on Candelilla Wax to Improve the Shelf Life and Quality of Avocado. Food Res. Int. 2009, 42, 511–515. [Google Scholar] [CrossRef]
- Sogvar, O.B.; Koushesh Saba, M.; Emamifar, A. Aloe Vera and Ascorbic Acid Coatings Maintain Postharvest Quality and Reduce Microbial Load of Strawberry Fruit. Postharvest Biol. Technol. 2016, 114, 29–35. [Google Scholar] [CrossRef]
- Dave, R.K.; Ramana Rao, T.V.; Nandane, A.S. Improvement of Post-Harvest Quality of Pear Fruit with Optimized Composite Edible Coating Formulations. J. Food Sci. Technol. 2017, 54, 3917–3927. [Google Scholar] [CrossRef]
- Tavassoli-Kafrani, E.; Gamage, M.V.; Dumée, L.F.; Kong, L.; Zhao, S. Edible Films and Coatings for Shelf Life Extension of Mango: A Review. Crit. Rev. Food Sci. Nutr. 2022, 62, 2432–2459. [Google Scholar] [CrossRef]
- Jeya, J.; Chandrasekaran, M.; Venkatesan, S.P.; Sriram, V.; Britto, J.G.; Mageshwaran, G.; Durairaj, R.B. Scaling up Difficulties and Commercial Aspects of Edible Films for Food Packaging: A Review. Trends Food Sci. Technol. 2020, 100, 210–222. [Google Scholar] [CrossRef]
- Bizymis, A.P.; Tzia, C. Edible Films and Coatings: Properties for the Selection of the Components, Evolution through Composites and Nanomaterials, and Safety Issues. Crit. Rev. Food Sci. Nutr. 2021. [Google Scholar] [CrossRef] [PubMed]
- Hassan, B.; Chatha, S.A.S.; Hussain, A.I.; Zia, K.M.; Akhtar, N. Recent Advances on Polysaccharides, Lipids and Protein Based Edible Films and Coatings: A Review. Int. J. Biol. Macromol. 2018, 109, 1095–1107. [Google Scholar] [CrossRef] [PubMed]
- Raghav, K.; Agarwal, N.; Saini, M. Edible Coating of Fruits and Vegetables: A Review. Int. J. Sci. Res. Mod. Educ. 2016, 1, 188–204. [Google Scholar]
- Artiga-Artigas, M.; Acevedo-Fani, A.; Martín-Belloso, O. Improving the Shelf Life of Low-Fat Cut Cheese Using Nanoemulsion-Based Edible Coatings Containing Oregano Essential Oil and Mandarin Fiber. Food Control 2017, 76, 1–12. [Google Scholar] [CrossRef]
- Jridi, M.; Abdelhedi, O.; Salem, A.; Kechaou, H.; Nasri, M.; Menchari, Y. Physicochemical, Antioxidant and Antibacterial Properties of Fish Gelatin-Based Edible Films Enriched with Orange Peel Pectin: Wrapping Application. Food Hydrocoll. 2020, 103, 105688. [Google Scholar] [CrossRef]
- Valencia-Chamorro, S.A.; Palou, L.; Delŕio, M.A.; Pérez-Gago, M.B. Antimicrobial Edible Films and Coatings for Fresh and Minimally Processed Fruits and Vegetables: A Review. Crit. Rev. Food Sci. Nutr. 2011, 51, 872–900. [Google Scholar] [CrossRef]
- Chiralt, A.; Menzel, C.; Hernandez-García, E.; Collazo, S.; Gonzalez-Martinez, C. Use of By-Products in Edible Coatings and Biodegradable Packaging Materials for Food Preservation. In Sustainability of the Food System: Sovereignty, Waste, and Nutrients Bioavailability; Academic Press: Cambridge, MA, USA, 2020. [Google Scholar]
- Cano, A.; Chafer, M.; Chiralt, A.; Gonzalez-Martinez, C. Strategies to Improve the Functionality of Starch-Based Films. In Handbook of Composites from Renewable Materials; John Wiley & Sons: Hoboken, NJ, USA, 2017; Volumes 1–8. [Google Scholar]
- Tóth, A.; Halász, K. Characterization of Edible Biocomposite Films Directly Prepared from Psyllium Seed Husk and Husk Flour. Food Packag. Shelf Life 2019, 20, 100299. [Google Scholar] [CrossRef]
- Nouraddini, M.; Esmaiili, M.; Mohtarami, F. Development and Characterization of Edible Films Based on Eggplant Flour and Corn Starch. Int. J. Biol. Macromol. 2018, 120, 1639–1645. [Google Scholar] [CrossRef]
- Montalvo-Paquini, C.; Avila-Sosa, R.; López-Malo, A.; Palou, E. Preparation and Characterization of Proteinaceous Films from Seven Mexican Common Beans (Phaseolus vulgaris L.). J. Food Qual. 2018, 2018, 9782591. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, Y. Comparison of Physicochemical and Mechanical Properties of Edible Films Made from Navy Bean and Corn Starches. J. Sci. Food Agric. 2021, 101, 1538–1545. [Google Scholar] [CrossRef]
- Ochoa-Yepes, O.; Medina-Jaramillo, C.; Guz, L.; Famá, L. Biodegradable and Edible Starch Composites with Fiber-Rich Lentil Flour to Use as Food Packaging. Starch-Staerke 2018, 70, 1700222. [Google Scholar] [CrossRef]
- Kocakulak, S.; Sumnu, G.; Sahin, S. Chickpea Flour-Based Biofilms Containing Gallic Acid to Be Used as Active Edible Films. J. Appl. Polym. Sci. 2019, 136, 47704. [Google Scholar] [CrossRef]
- Roberta, M.S.A.; Mariana, S.L.F.; E’dira, C.B.A.G. Functional Capacity of Flour Obtained from Residues of Fruit and Vegetables. Int. Food Res. J. 2014, 21, 1675–1681. [Google Scholar]
- Pelissari, F.M.; Andrade-Mahecha, M.M.; do Amaral Sobral, P.J.; Menegalli, F.C. Comparative Study on the Properties of Flour and Starch Films of Plantain Bananas (Musa paradisiaca). Food Hydrocoll. 2013, 30, 681–690. [Google Scholar] [CrossRef]
- Andrade, R.M.S.; Ferreira, M.S.L.; Gonçalves, É.C.B.A. Development and Characterization of Edible Films Based on Fruit and Vegetable Residues. J. Food Sci. 2016, 81, E412–E418. [Google Scholar] [CrossRef]
- McHugh, T.H.; Senesi, E. Apple Wraps: A Novel Method to Improve the Quality and Extend the Shelf Life of Fresh-Cut Apples. J. Food Sci. 2000, 65, 480–485. [Google Scholar] [CrossRef]
- Sothornvit, R.; Pitak, N. Oxygen Permeability and Mechanical Properties of Banana Films. Food Res. Int. 2007, 40, 365–370. [Google Scholar] [CrossRef]
- Wang, X.; Kong, D.; Ma, Z.; Zhao, R. Effect of Carrot Puree Edible Films on Quality Preservation of Fresh-Cut Carrots. Irish J. Agric. Food Res. 2015, 54, 64–71. [Google Scholar] [CrossRef]
- Rangel-Marrón, M.; Mani-López, E.; Palou, E.; López-Malo, A. Effects of Alginate-Glycerol-Citric Acid Concentrations on Selected Physical, Mechanical, and Barrier Properties of Papaya Puree-Based Edible Films and Coatings, as Evaluated by Response Surface Methodology. LWT 2019, 101, 83–91. [Google Scholar] [CrossRef]
- Ayquipa-Cuellar, E.; Salcedo-Sucasaca, L.; Azamar-Barrios, J.A.; Chaquilla-Quilca, G. Assessment of Prickly Pear Peel Mucilage and Potato Husk Starch for Edible Films Production for Food Packaging Industries. Waste Biomass Valorization 2021, 12, 321–331. [Google Scholar] [CrossRef]
- Motlagh, S.; Ravines, P.; Karamallah, K.A.; Ma, Q. The Analysis of Acacia Gums Using Electrophoresis. Food Hydrocoll. 2006, 20, 848–854. [Google Scholar] [CrossRef]
- Patel, S.; Goyal, A. Applications of Natural Polymer Gum Arabic: A Review. Int. J. Food Prop. 2015, 18, 986–998. [Google Scholar] [CrossRef]
- Mahfoudhi, N.; Hamdi, S. Use of Almond Gum and Gum Arabic as Novel Edible Coating to Delay Postharvest Ripening and to Maintain Sweet Cherry (Prunus avium) Quality during Storage. J. Food Process. Preserv. 2015, 39, 1499–1508. [Google Scholar] [CrossRef]
- Bashir, M.; Haripriya, S. Assessment of Physical and Structural Characteristics of Almond Gum. Int. J. Biol. Macromol. 2016, 93, 476–482. [Google Scholar] [CrossRef]
- Aydogdu, A.; Radke, C.J.; Bezci, S.; Kirtil, E. Characterization of Curcumin Incorporated Guar Gum/Orange Oil Antimicrobial Emulsion Films. Int. J. Biol. Macromol. 2020, 148, 110–120. [Google Scholar] [CrossRef]
- Banegas, R.S.; Zornio, C.F.; De Borges, A.M.G.; Porto, L.C.; Soldi, V. Preparation, Characterization and Properties of Films Obtained from Cross-Linked Guar Gum. Polimeros 2013, 23, 182–188. [Google Scholar] [CrossRef]
- Arismendi, C.; Chillo, S.; Conte, A.; Del Nobile, M.A.; Flores, S.; Gerschenson, L.N. Optimization of Physical Properties of Xanthan Gum/Tapioca Starch Edible Matrices Containing Potassium Sorbate and Evaluation of Its Antimicrobial Effectiveness. LWT-Food Sci. Technol. 2013, 53, 290–296. [Google Scholar] [CrossRef]
- Ge, L.; Li, X.; Zhang, R.; Yang, T.; Ye, X.; Li, D.; Mu, C. Development and Characterization of Dialdehyde Xanthan Gum Crosslinked Gelatin Based Edible Films Incorporated with Amino-Functionalized Montmorillonite. Food Hydrocoll. 2015, 51, 129–135. [Google Scholar] [CrossRef]
- Guo, J.; Ge, L.; Li, X.; Mu, C.; Li, D. Periodate Oxidation of Xanthan Gum and Its Crosslinking Effects on Gelatin-Based Edible Films. Food Hydrocoll. 2014, 39, 243–250. [Google Scholar] [CrossRef]
- Al-Tayyar, N.A.; Youssef, A.M.; Al-Hindi, R.R. Edible Coatings and Antimicrobial Nanoemulsions for Enhancing Shelf Life and Reducing Foodborne Pathogens of Fruits and Vegetables: A Review. Sustain. Mater. Technol. 2020, 26, e00215. [Google Scholar] [CrossRef]
- Otoni, C.G.; Avena-Bustillos, R.J.; Azeredo, H.M.C.; Lorevice, M.V.; Moura, M.R.; Mattoso, L.H.C.; McHugh, T.H. Recent Advances on Edible Films Based on Fruits and Vegetables—A Review. Compr. Rev. Food Sci. Food Saf. 2017, 16, 1151–1169. [Google Scholar] [CrossRef] [PubMed]
- Robles-Flores, G.D.C.; Abud-Archila, M.; Ventura-Canseco, L.M.C.; Meza-Gordillo, R.; Grajales-Lagunes, A.; Ruiz-Cabrera, M.A.; Gutiérrez-Miceli, F.A. Development and Evaluation of a Film and Edible Coating Obtained from the Cajanus Cajan Seed Applied to Fresh Strawberry Fruit. Food Bioprocess Technol. 2018, 11, 2172–2181. [Google Scholar] [CrossRef]
- Daudt, R.M.; Avena-Bustillos, R.J.; Williams, T.; Wood, D.F.; Külkamp-Guerreiro, I.C.; Marczak, L.D.F.; McHugh, T.H. Comparative Study on Properties of Edible Films Based on Pinhão (Araucaria angustifolia) Starch and Flour. Food Hydrocoll. 2016, 60, 279–287. [Google Scholar] [CrossRef]
- Chen, Y.; Xu, L.; Wang, Y.; Chen, Z.; Zhang, M.; Chen, H. Characterization and Functional Properties of a Pectin/Tara Gum Based Edible Film with Ellagitannins from the Unripe Fruits of Rubus Chingii Hu. Food Chem. 2020, 325, 126964. [Google Scholar] [CrossRef] [PubMed]
- Dhall, R.K. Advances in Edible Coatings for Fresh Fruits and Vegetables: A Review. Crit. Rev. Food Sci. Nutr. 2013, 53, 435–450. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W. Fabrication of Pectin/Agar Blended Functional Film: Effect of Reinforcement of Melanin Nanoparticles and Grapefruit Seed Extract. Food Hydrocoll. 2021, 118, 106823. [Google Scholar] [CrossRef]
- Jahromi, M.; Niakousari, M.; Golmakani, M.T.; Mohammadifar, M.A. Physicochemical and Structural Characterization of Sodium Caseinate Based Film-Forming Solutions and Edible Films as Affected by High Methoxyl Pectin. Int. J. Biol. Macromol. 2020, 165, 1949–1959. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W. Preparation of Pectin/Agar-Based Functional Films Integrated with Zinc Sulfide Nano Petals for Active Packaging Applications. Colloids Surf. B Biointerfaces 2021, 207, 111999. [Google Scholar] [CrossRef]
- Ezati, P.; Roy, S.; Rhim, J.-W. Pectin/Gelatin-Based Bioactive Composite Films Reinforced with Sulfur Functionalized Carbon Dots. Colloids Surf. A Physicochem. Eng. Asp. 2021, 636, 128123. [Google Scholar] [CrossRef]
- Ravishankar, S.; Jaroni, D.; Zhu, L.; Olsen, C.; McHugh, T.; Friedman, M. Inactivation of Listeria Monocytogenes on Ham and Bologna Using Pectin-Based Apple, Carrot, and Hibiscus Edible Films Containing Carvacrol and Cinnamaldehyde. J. Food Sci. 2012, 77, M377–M382. [Google Scholar] [CrossRef]
- Mild, R.M.; Joens, L.A.; Friedman, M.; Olsen, C.W.; Mchugh, T.H.; Law, B.; Ravishankar, S. Antimicrobial Edible Apple Films Inactivate Antibiotic Resistant and Susceptible Campylobacter jejuni Strains on Chicken Breast. J. Food Sci. 2011, 76, M163–M168. [Google Scholar] [CrossRef] [PubMed]
- Lazaridou, A.; Biliaderis, C.G. Edible Films and Coatings with Pectin. In Pectin: Technological and Physiological Properties; Springer: Cham, Switzerland, 2020; pp. 99–123. [Google Scholar] [CrossRef]
- Sucheta; Rai, S.K.; Chaturvedi, K.; Yadav, S.K. Evaluation of Structural Integrity and Functionality of Commercial Pectin Based Edible Films Incorporated with Corn Flour, Beetroot, Orange Peel, Muesli and Rice Flour. Food Hydrocoll. 2019, 91, 127–135. [Google Scholar] [CrossRef]
- Basiak, E.; Lenart, A. Starch Coatings Used in Food Packaging Industry. Zywn. Nauk. Technol. Jakosc/Food. Sci. Technol. Qual. 2013, 20, 21–31. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W. Starch/Agar-Based Functional Films Integrated with Enoki Mushroom-Mediated Silver Nanoparticles for Active Packaging Applications. Food Biosci. 2022, 49, 101867. [Google Scholar] [CrossRef]
- Guimarães, I.C.; dos Reis, K.C.; Menezes, E.G.T.; Rodrigues, A.C.; da Silva, T.F.; de Oliveira, I.R.N.; Vilas Boas, E.V.D.B.V. Cellulose Microfibrillated Suspension of Carrots Obtained by Mechanical Defibrillation and Their Application in Edible Starch Films. Ind. Crops Prod. 2016, 89, 285–294. [Google Scholar] [CrossRef]
- Kocira, A.; Kozłowicz, K.; Panasiewicz, K.; Staniak, M.; Szpunar-Krok, E.; Hortyńska, P. Polysaccharides as Edible Films and Coatings: Characteristics and Influence on Fruit and Vegetable Quality—A Review. Agronomy 2021, 11, 813. [Google Scholar] [CrossRef]
- Krochta, J.M.; Mulder, J.C. Scientific Status Summary: Edible and Biodegradable Polymer Films: Challenges and Opportunities. Food Technol. 1997, 51, 61–74. [Google Scholar]
- Li, H.; Shi, H.; He, Y.; Fei, X.; Peng, L. Preparation and Characterization of Carboxymethyl Cellulose-Based Composite Films Reinforced by Cellulose Nanocrystals Derived from Pea Hull Waste for Food Packaging Applications. Int. J. Biol. Macromol. 2020, 164, 4104–4112. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.W. Carboxymethyl Cellulose-Based Antioxidant and Antimicrobial Active Packaging Film Incorporated with Curcumin and Zinc Oxide. Int. J. Biol. Macromol. 2020, 148, 666–676. [Google Scholar] [CrossRef]
- Pooja Saklani, P.S.; Nath, S.; Kishor Das, S.; Singh, S.M. A Review of Edible Packaging for Foods. Int. J. Curr. Microbiol. Appl. Sci. 2019, 8, 2885–2895. [Google Scholar] [CrossRef]
- Mihalca, V.; Kerezsi, A.D.; Weber, A.; Gruber-Traub, C.; Schmucker, J.; Vodnar, D.C.; Dulf, F.V.; Socaci, S.A.; Fărcaș, A.; Mureșan, C.I.; et al. Protein-Based Films and Coatings for Food Industry Applications. Polymers 2021, 13, 769. [Google Scholar] [CrossRef] [PubMed]
- Grujic, R.; Vukic, M.; Gojkovic, V. Application of Biopolymers in the Food Industry. In Advances in Applications of Industrial Biomaterials; Springer: Cham, Switzerland, 2017. [Google Scholar]
- Lin, D.; Zhao, Y. Innovations in the Development and Application of Edible Coatings for Fresh and Minimally Processed Fruits and Vegetables. Compr. Rev. Food Sci. Food Saf. 2007, 6, 60–75. [Google Scholar] [CrossRef]
- Borchard, W.; Kenning, A.; Kapp, A.; Mayer, C. Phase Diagram of the System Sodium Alginate/Water: A Model for Biofilms. Int. J. Biol. Macromol. 2005, 35, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Dhanapal, A.; Rajamani, L.; Banu, M. Edible Films from Polysaccharides. Food Sci. Qual. 2012, 3, 9–18. [Google Scholar]
- Rhim, J.W. Physical and Mechanical Properties of Water Resistant Sodium Alginate Films. LWT-Food Sci. Technol. 2004, 37, 323–330. [Google Scholar] [CrossRef]
- Tavassoli-Kafrani, E.; Shekarchizadeh, H.; Masoudpour-Behabadi, M. Development of Edible Films and Coatings from Alginates and Carrageenans. Carbohydr. Polym. 2016, 137, 360–374. [Google Scholar] [CrossRef]
- Osorio, F.A.; Molina, P.; Matiacevich, S.; Enrione, J.; Skurtys, O. Characteristics of Hydroxy Propyl Methyl Cellulose (HPMC) Based Edible Film Developed for Blueberry Coatings. Procedia Food Sci. 2011, 1, 287–293. [Google Scholar] [CrossRef]
- Campos, C.A.; Gerschenson, L.N.; Flores, S.K. Development of Edible Films and Coatings with Antimicrobial Activity. Food Bioprocess Technol. 2011, 4, 849–875. [Google Scholar] [CrossRef]
- Roy, S.; Ezati, P.; Rhim, J.-W. Gelatin/Carrageenan-Based Functional Films with Carbon Dots from Enoki Mushroom for Active Food Packaging Applications. ACS Appl. Polym. Mater. 2021, 3, acsapm.1c01175. [Google Scholar] [CrossRef]
- Hambleton, A.; Perpiñan-Saiz, N.; Fabra, M.J.; Voilley, A.; Debeaufort, F. The Schroeder Paradox or How the State of Water Affects the Moisture Transfer through Edible Films. Food Chem. 2012, 132, 1671–1678. [Google Scholar] [CrossRef]
- Schmidt, É.C.; dos Santos, R.; Horta, P.A.; Maraschin, M.; Bouzon, Z.L. Effects of UVB Radiation on the Agarophyte Gracilaria Domingensis (Rhodophyta, Gracilariales): Changes in Cell Organization, Growth and Photosynthetic Performance. Micron 2010, 41, 919–930. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Rhim, J.-W. Gelatin/Agar-Based Functional Film Integrated with Pickering Emulsion of Clove Essential Oil Stabilized with Nanocellulose for Active Packaging Applications. Colloids Surf. A Physicochem. Eng. Asp. 2021, 627, 127220. [Google Scholar] [CrossRef]
- Rhim, J.W. Effect of Clay Contents on Mechanical and Water Vapor Barrier Properties of Agar-Based Nanocomposite Films. Carbohydr. Polym. 2011, 86, 691–699. [Google Scholar] [CrossRef]
- Boziaris, I.S. Novel Food Preservation and Microbial Assessment Techniques; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Zhang, M.; Haga, A.; Sekiguchi, H.; Hirano, S. Structure of Insect Chitin Isolated from Beetle Larva Cuticle and Silkworm (Bombyx Mori) Pupa Exuvia. Int. J. Biol. Macromol. 2000, 27, 99–105. [Google Scholar] [CrossRef]
- Majtán, J.; Bíliková, K.; Markovič, O.; Gróf, J.; Kogan, G.; Šimúth, J. Isolation and Characterization of Chitin from Bumblebee (Bombus Terrestris). Int. J. Biol. Macromol. 2007, 40, 237–241. [Google Scholar] [CrossRef]
- Sajomsang, W.; Gonil, P. Preparation and Characterization of α-Chitin from Cicada Sloughs. Mater. Sci. Eng. C 2010, 30, 357–363. [Google Scholar] [CrossRef]
- Shahid-ul-Islam; Butola, B.S. Recent Advances in Chitosan Polysaccharide and Its Derivatives in Antimicrobial Modification of Textile Materials. Int. J. Biol. Macromol. 2019, 121, 905–912. [Google Scholar] [CrossRef]
- Elsabee, M.Z.; Abdou, E.S. Chitosan Based Edible Films and Coatings: A Review. Mater. Sci. Eng. C 2013, 33, 1819–1841. [Google Scholar] [CrossRef]
- Domard, A.; Domard, M. Chitosan: Structure-Properties Relationship and Biomedical Applications. In Polymeric Biomaterials, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2002. [Google Scholar]
- Youssef, A.M.; Abdel-Aziz, M.S.; El-Sayed, S.M. Chitosan Nanocomposite Films Based on Ag-NP and Au-NP Biosynthesis by Bacillus Subtilis as Packaging Materials. Int. J. Biol. Macromol. 2014, 69, 185–191. [Google Scholar] [CrossRef]
- Chaudhary, S.; Kumar, S.; Kumar, V.; Sharma, R. Chitosan Nanoemulsions as Advanced Edible Coatings for Fruits and Vegetables: Composition, Fabrication and Developments in Last Decade. Int. J. Biol. Macromol. 2020, 152, 154–170. [Google Scholar]
- Pajak, P.; Fortuna, T.; Przetaczek-Roznowska, I. Protein- and Polysaccharide-Based Edible Packagings: Profile and Applications. Zywn. Nauk. Technol. Jakosc/Food. Sci. Technol. Qual. 2013, 20, 5–18. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W. Genipin-Crosslinked Gelatin/Chitosan-Based Functional Films Incorporated with Rosemary Essential Oil and Quercetin. Materials 2022, 15, 3769. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.; Rhim, J.-W. Effect of Chitosan Modified Halloysite on the Physical and Functional Properties of Pullulan/Chitosan Biofilm Integrated with Rutin. Appl. Clay Sci. 2021, 211, 106205. [Google Scholar] [CrossRef]
- Hajji, S.; Younes, I.; Affes, S.; Boufi, S.; Nasri, M. Optimization of the Formulation of Chitosan Edible Coatings Supplemented with Carotenoproteins and Their Use for Extending Strawberries Postharvest Life. Food Hydrocoll. 2018, 83, 375–392. [Google Scholar] [CrossRef]
- Candir, E.; Ozdemir, A.E.; Aksoy, M.C. Effects of Chitosan Coating and Modified Atmosphere Packaging on Postharvest Quality and Bioactive Compounds of Pomegranate Fruit Cv. ‘Hicaznar.’ Sci. Hortic. 2018, 235, 235–243. [Google Scholar] [CrossRef]
- Avila Rodríguez, M.I.; Rodríguez Barroso, L.G.; Sánchez, M.L. Collagen: A Review on Its Sources and Potential Cosmetic Applications. J. Cosmet. Dermatol. 2018, 17, 20–26. [Google Scholar]
- Felician, F.F.; Xia, C.; Qi, W.; Xu, H. Collagen from Marine Biological Sources and Medical Applications. Chem. Biodivers. 2018, 15, e1700557. [Google Scholar]
- Wang, L.F.; Rhim, J.W. Preparation and Application of Agar/Alginate/Collagen Ternary Blend Functional Food Packaging Films. Int. J. Biol. Macromol. 2015, 80, 460–468. [Google Scholar] [CrossRef]
- Wu, X.; Liu, Y.; Liu, A.; Wang, W. Improved Thermal-Stability and Mechanical Properties of Type I Collagen by Crosslinking with Casein, Keratin and Soy Protein Isolate Using Transglutaminase. Int. J. Biol. Macromol. 2017, 98, 292–301. [Google Scholar] [CrossRef]
- Yadav, S.; Mehrotra, G.K.; Bhartiya, P.; Singh, A.; Dutta, P.K. Preparation, Physicochemical and Biological Evaluation of Quercetin Based Chitosan-Gelatin Film for Food Packaging. Carbohydr. Polym. 2020, 227, 115348. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W. Gelatin/Cellulose Nanofiber-Based Functional Films Added with Mushroom-Mediated Sulfur Nanoparticles for Active Packaging Applications. J. Nanostruct. Chem. 2022, 2022, 1–12. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W. Preparation of Gelatin/Carrageenan-Based Color-Indicator Film Integrated with Shikonin and Propolis for Smart Food Packaging Applications. ACS Appl. Bio Mater. 2020, 4, 770–779. [Google Scholar] [CrossRef]
- Hanani, Z.A.N.; Yee, F.C.; Nor-Khaizura, M.A.R. Effect of Pomegranate (Punica granatum L.) Peel Powder on the Antioxidant and Antimicrobial Properties of Fish Gelatin Films as Active Packaging. Food Hydrocoll. 2019, 89, 253–259. [Google Scholar] [CrossRef]
- Uranga, J.; Etxabide, A.; Guerrero, P.; de la Caba, K. Development of Active Fish Gelatin Films with Anthocyanins by Compression Molding. Food Hydrocoll. 2018, 84, 313–320. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W. Fabrication of Copper Sulfide Nanoparticles and Limonene Incorporated Pullulan/Carrageenan-Based Film with Improved Mechanical and Antibacterial Properties. Polymers 2020, 12, 2665. [Google Scholar] [CrossRef]
- Barcelos, M.C.S.; Vespermann, K.A.C.; Pelissari, F.M.; Molina, G. Current Status of Biotechnological Production and Applications of Microbial Exopolysaccharides. Crit. Rev. Food Sci. Nutr. 2020, 60, 1475–1495. [Google Scholar]
- Diab, T.; Biliaderis, C.G.; Gerasopoulos, D.; Sfakiotakis, E. Physicochemical Properties and Application of Pullulan Edible Films and Coatings in Fruit Preservation. J. Sci. Food Agric. 2001, 81, 988–1000. [Google Scholar] [CrossRef]
- Niu, B.; Shao, P.; Chen, H.; Sun, P. Structural and Physiochemical Characterization of Novel Hydrophobic Packaging Films Based on Pullulan Derivatives for Fruits Preservation. Carbohydr. Polym. 2019, 208, 276–284. [Google Scholar] [CrossRef]
- Li, L.; Sun, J.; Gao, H.; Shen, Y.; Li, C.; Yi, P.; He, X.; Ling, D.; Sheng, J.; Li, J.; et al. Effects of Polysaccharide-Based Edible Coatings on Quality and Antioxidant Enzyme System of Strawberry during Cold Storage. Int. J. Polym. Sci. 2017, 2017, 9746174. [Google Scholar] [CrossRef]
- Radev, R.; Pashova, S. Application of Edible Films and Coatings for Fresh Fruit and Vegetables. Qual.-Access Success 2020, 21, 108–112. [Google Scholar]
- Shit, S.C.; Shah, P.M. Edible Polymers: Challenges and Opportunities. J. Polym. 2014, 2014, 427259. [Google Scholar] [CrossRef]
- Pobiega, K.; Igielska, M.; Włodarczyk, P.; Gniewosz, M. The Use of Pullulan Coatings with Propolis Extract to Extend the Shelf Life of Blueberry (Vaccinium corymbosum) Fruit. Int. J. Food Sci. Technol. 2021, 56, 1013–1020. [Google Scholar] [CrossRef]
- Qi, H.; Hu, W.; Jiang, A.; Tian, M.; Li, Y. Extending Shelf-Life of Fresh-Cut “Fuji” Apples with Chitosan-Coatings. Innov. Food Sci. Emerg. Technol. 2011, 12, 62–66. [Google Scholar] [CrossRef]
- Roy, S.; Priyadarshi, R.; Rhim, J.W. Development of Multifunctional Pullulan/Chitosan-Based Composite Films Reinforced with ZnO Nanoparticles and Propolis for Meat Packaging Applications. Foods 2021, 10, 2789. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, D.; Datta, S.; Biswas, D. Towards a Better Production of Bacterial Exopolysaccharides by Controlling Genetic as Well as Physico-Chemical Parameters. Appl. Microbiol. Biotechnol. 2018, 102, 1587–1598. [Google Scholar]
- Totad, M.G.; Sharma, R.R.; Sethi, S.; Verma, M.K. Effect of Edible Coatings on ‘Misty’ Blueberry (Vaccinium corymbosum) Fruits Stored at Low Temperature. Acta Physiol. Plant. 2019, 41, 183. [Google Scholar] [CrossRef]
- Salehi, F. Edible Coating of Fruits and Vegetables Using Natural Gums: A Review. Int. J. Fruit Sci. 2020, 20, S570–S589. [Google Scholar]
- Saleem, M.S.; Ejaz, S.; Anjum, M.A.; Nawaz, A.; Naz, S.; Hussain, S.; Ali, S.; Canan, İ. Postharvest Application of Gum Arabic Edible Coating Delays Ripening and Maintains Quality of Persimmon Fruits during Storage. J. Food Process. Preserv. 2020, 44, e14583. [Google Scholar] [CrossRef]
- Baraiya, N.S.; Ramana Rao, T.V.; Thakkar, V.R. Composite Coating as a Carrier of Antioxidants Improves the Postharvest Shelf Life and Quality of Table Grapes (Vitis vinifera L. var. Thompson Seedless). J. Agric. Sci. Technol. 2016, 18, 93–107. [Google Scholar]
- Tomadoni, B.; Moreira, M.R.; Pereda, M.; Ponce, A.G. Gellan-Based Coatings Incorporated with Natural Antimicrobials in Fresh-Cut Strawberries: Microbiological and Sensory Evaluation through Refrigerated Storage. LWT 2018, 97, 384–389. [Google Scholar] [CrossRef]
- Mohamed, S.A.A.; El-Sakhawy, M.; El-Sakhawy, M.A.-M. Polysaccharides, Protein and Lipid -Based Natural Edible Films in Food Packaging: A Review. Carbohydr. Polym. 2020, 238, 116178. [Google Scholar] [CrossRef] [PubMed]
- Vanden Braber, N.L.; Di Giorgio, L.; Aminahuel, C.A.; Díaz Vergara, L.I.; Martin Costa, A.O.; Montenegro, M.A.; Mauri, A.N. Antifungal Whey Protein Films Activated with Low Quantities of Water Soluble Chitosan. Food Hydrocoll. 2021, 110, 106156. [Google Scholar] [CrossRef]
- Galus, S.; Kadzińska, J. Whey Protein Edible Films Modified with Almond and Walnut Oils. Food Hydrocoll. 2016, 52, 78–86. [Google Scholar] [CrossRef]
- Umaraw, P.; Munekata, P.E.S.; Verma, A.K.; Barba, F.J.; Singh, V.P.; Kumar, P.; Lorenzo, J.M. Edible Films/Coating with Tailored Properties for Active Packaging of Meat, Fish and Derived Products. Trends Food Sci. Technol. 2020, 98, 10–24. [Google Scholar]
- Rhim, J.W.; Shellhammer, T.H. Lipid-Based Edible Films and Coatings. In Innovations in Food Packaging; Elsevier: Amsterdam, The Netherlands, 2005. [Google Scholar]
- Dehghani, S.; Hosseini, S.V.; Regenstein, J.M. Edible Films and Coatings in Seafood Preservation: A Review. Food Chem. 2018, 240, 505–513. [Google Scholar]
- Parreidt, T.S.; Müller, K.; Schmid, M. Alginate-Based Edible Films and Coatings for Food Packaging Applications. Foods 2018, 7, 170. [Google Scholar] [CrossRef]
- Murrieta-Martínez, C.L.; Soto-Valdez, H.; Pacheco-Aguilar, R.; Torres-Arreola, W.; Rodríguez-Felix, F.; Márquez Ríos, E. Edible Protein Films: Sources and Behavior. Packag. Technol. Sci. 2018, 31, 113–122. [Google Scholar]
- Ribeiro, A.M.; Estevinho, B.N.; Rocha, F. Preparation and Incorporation of Functional Ingredients in Edible Films and Coatings. Food Bioprocess Technol. 2021, 14, 209–231. [Google Scholar]
- Jafarzadeh, S.; Alias, A.K.; Ariffin, F.; Mahmud, S.; Najafi, A.; Sheibani, S. Characterization of a New Biodegradable Edible Film Based on Semolina Loaded with Nano Kaolin. Int. Food Res. J. 2017, 24, 304–309. [Google Scholar]
- Garavand, F.; Cacciotti, I.; Vahedikia, N.; Salara, A.R.; Tarhan, Ö.; Akbari-Alavijeh, S.; Shaddel, R.; Rashidinejad, A.; Nejatian, M.; Jafarzadeh, S.; et al. A Comprehensive Review on the Nanocomposites Loaded with Chitosan Nanoparticles for Food Packaging. Crit. Rev. Food Sci. Nutr. 2020, 62, 1383–1416. [Google Scholar]
- Cakmak, H.; Kumcuoglu, S.; Tavman, S. Production of Edible Coatings with Twin-Nozzle Electrospraying Equipment and the Effects on Shelf-Life Stability of Fresh-Cut Apple Slices. J. Food Process Eng. 2018, 41, e12627. [Google Scholar] [CrossRef]
- Pirozzi, A.; Del Grosso, V.; Ferrari, G.; Donsì, F. Edible Coatings Containing Oregano Essential Oil Nanoemulsion for Improving Postharvest Quality and Shelf Life of Tomatoes. Foods 2020, 9, 1605. [Google Scholar] [CrossRef] [PubMed]
- Dwivany, F.M.; Aprilyandi, A.N.; Suendo, V.; Sukriandi, N. Carrageenan Edible Coating Application Prolongs Cavendish Banana Shelf Life. Int. J. Food Sci. 2020, 2020, 8861610. [Google Scholar] [CrossRef] [PubMed]
- Hermawan, D.; Lai, T.K.; Jafarzadeh, S.; Gopakumar, D.A.; Hasan, M.; Owolabi, F.A.T.; Sri Aprilia, N.A.; Rizal, S.; Khalil, H.P.S.A. Development of Seaweed-Based Bamboo Microcrystalline Cellulose Films Intended for Sustainable Food Packaging Applications. BioResources 2019, 14, 3389–3410. [Google Scholar] [CrossRef]
- Biswas, M.C.; Tiimob, B.J.; Abdela, W.; Jeelani, S.; Rangari, V.K. Nano Silica-Carbon-Silver Ternary Hybrid Induced Antimicrobial Composite Films for Food Packaging Application. Food Packag. Shelf Life 2019, 19, 104–113. [Google Scholar] [CrossRef]
- Sobhan, A.; Muthukumarappan, K.; Wei, L.; Van Den Top, T.; Zhou, R. Development of an Activated Carbon-Based Nanocomposite Film with Antibacterial Property for Smart Food Packaging. Mater. Today Commun. 2020, 23, 101124. [Google Scholar] [CrossRef]
- Dairi, N.; Ferfera-Harrar, H.; Ramos, M.; Garrigós, M.C. Cellulose Acetate/AgNPs-Organoclay and/or Thymol Nano-Biocomposite Films with Combined Antimicrobial/Antioxidant Properties for Active Food Packaging Use. Int. J. Biol. Macromol. 2019, 121, 508–523. [Google Scholar] [CrossRef]
- Saedi, S.; Shokri, M.; Roy, S.; Rhim, J.-W. Silver Loaded Aminosilane Modified Halloysite for the Preparation of Carrageenan-Based Functional Films. Appl. Clay Sci. 2021, 211, 106170. [Google Scholar] [CrossRef]
- Alves, L.; Ferraz, E.; Gamelas, J.A.F. Composites of Nanofibrillated Cellulose with Clay Minerals: A Review. Adv. Colloid Interface Sci. 2019, 272, 101994. [Google Scholar] [CrossRef]
- Abu-Thabit, N.; Saeed Hakeem, A.; Mezghani, K.; Ratemi, E.; Elzagheid, M.; Umar, Y.; Primartomo, A.; Al Batty, S.; Kalam Azad, A.; Al Anazi, S.; et al. Preparation of pH-Indicative and Flame-Retardant Nanocomposite Films for Smart Packaging Applications. Sensors 2020, 20, 5462. [Google Scholar] [CrossRef]
- Alfei, S.; Marengo, B.; Zuccari, G. Nanotechnology Application in Food Packaging: A Plethora of Opportunities versus Pending Risks Assessment and Public Concerns. Food Res. Int. 2020, 137, 109664. [Google Scholar] [CrossRef] [PubMed]
- Raquez, J.M.; Habibi, Y.; Murariu, M.; Dubois, P. Polylactide (PLA)-Based Nanocomposites. Prog. Polym. Sci. 2013, 38, 1504–1542. [Google Scholar] [CrossRef]
- Peters, R.; ten Dam, G.; Bouwmeester, H.; Helsper, H.; Allmaier, G.; vd Kammer, F.; Ramsch, R.; Solans, C.; Tomaniová, M.; Hajslova, J.; et al. Identification and Characterization of Organic Nanoparticles in Food. TrAC-Trends Anal. Chem. 2011, 30, 100–112. [Google Scholar] [CrossRef]
- Roy, S.; Kim, H.C.; Kim, J.W.; Zhai, L.; Zhu, Q.Y.; Kim, J. Incorporation of Melanin Nanoparticles Improves UV-Shielding, Mechanical and Antioxidant Properties of Cellulose Nanofiber Based Nanocomposite Films. Mater. Today Commun. 2020, 24, 100984. [Google Scholar] [CrossRef]
- Łopusiewicz, Ł.; Macieja, S.; Śliwiński, M.; Bartkowiak, A.; Roy, S.; Sobolewski, P. Alginate Biofunctional Films Modified with Melanin from Watermelon Seeds and Zinc Oxide/Silver Nanoparticles. Materials 2022, 15, 2381. [Google Scholar] [CrossRef]
- Jafarzadeh, S.; Ariffin, F.; Mahmud, S.; Alias, A.K.; Hosseini, S.F.; Ahmad, M. Improving the Physical and Protective Functions of Semolina Films by Embedding a Blend Nanofillers (ZnO-Nr and Nano-Kaolin). Food Packag. Shelf Life 2017, 12, 66–75. [Google Scholar] [CrossRef]
- Hasan, M.; Chong, E.W.N.; Jafarzadeh, S.; Paridah, M.T.; Gopakumar, D.A.; Tajarudin, H.A.; Thomas, S.; Khalil, H.P.S.A. Enhancement in the Physico-Mechanical Functions of Seaweed Biopolymer Film via Embedding Fillers for Plasticulture Application-A Comparison with Conventional Biodegradable Mulch Film. Polymers 2019, 11, 210. [Google Scholar] [CrossRef]
- Amjadi, S.; Emaminia, S.; Nazari, M.; Davudian, S.H.; Roufegarinejad, L.; Hamishehkar, H. Application of Reinforced ZnO Nanoparticle-Incorporated Gelatin Bionanocomposite Film with Chitosan Nanofiber for Packaging of Chicken Fillet and Cheese as Food Models. Food Bioprocess Technol. 2019, 12, 1205–1219. [Google Scholar] [CrossRef]
- Jafarzadeh, S.; Rhim, J.W.; Alias, A.K.; Ariffin, F.; Mahmud, S. Application of Antimicrobial Active Packaging Film Made of Semolina Flour, Nano Zinc Oxide and Nano-Kaolin to Maintain the Quality of Low-Moisture Mozzarella Cheese during Low-Temperature Storage. J. Sci. Food Agric. 2019, 99, 2716–2725. [Google Scholar] [CrossRef]
- Alizadeh-Sani, M.; Khezerlou, A.; Ehsani, A. Fabrication and Characterization of the Bionanocomposite Film Based on Whey Protein Biopolymer Loaded with TiO2 Nanoparticles, Cellulose Nanofibers and Rosemary Essential Oil. Ind. Crops Prod. 2018, 124, 300–315. [Google Scholar] [CrossRef]
- Roy, S.; Rhim, J.-W. Anthocyanin Food Colorant and Its Application in PH-Responsive Color Change Indicator Films. Crit. Rev. Food Sci. Nutr. 2020, 61, 2297–2325. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J.; Roy, S.; Rhim, J.-W. Gelatin/Agar-Based Color-Indicator Film Integrated with Clitoria Ternatea Flower Anthocyanin and Zinc Oxide Nanoparticles for Monitoring Freshness of Shrimp. Food Hydrocoll. 2022, 124, 107294. [Google Scholar] [CrossRef]
- Agourram, A.; Ghirardello, D.; Rantsiou, K.; Zeppa, G.; Belviso, S.; Romane, A.; Oufdou, K.; Giordano, M. Phenolic Content, Antioxidant Potential, and Antimicrobial Activities of Fruit and Vegetable by-Product Extracts. Int. J. Food Prop. 2013, 16, 1092–1104. [Google Scholar] [CrossRef]
- Kaya, M.; Ravikumar, P.; Ilk, S.; Mujtaba, M.; Akyuz, L.; Labidi, J.; Salaberria, A.M.; Cakmak, Y.S.; Erkul, S.K. Production and Characterization of Chitosan Based Edible Films from Berberis Crataegina’s Fruit Extract and Seed Oil. Innov. Food Sci. Emerg. Technol. 2018, 45, 287–297. [Google Scholar] [CrossRef]
- Ribeiro-Santos, R.; Andrade, M.; de Melo, N.R.; Sanches-Silva, A. Use of Essential Oils in Active Food Packaging: Recent Advances and Future Trends. Trends Food Sci. Technol. 2017, 61, 132–140. [Google Scholar] [CrossRef]
- Boukaew, S.; Prasertsan, P.; Sattayasamitsathit, S. Evaluation of Antifungal Activity of Essential Oils against Aflatoxigenic Aspergillus flavus and Their Allelopathic Activity from Fumigation to Protect Maize Seeds during Storage. Ind. Crops Prod. 2017, 97, 558–566. [Google Scholar] [CrossRef]
- Anis, A.; Pal, K.; Al-Zahrani, S.M. Essential Oil-Containing Polysaccharide-Based Edible Films and Coatings for Food Security Applications. Polymers 2021, 13, 575. [Google Scholar] [CrossRef]
- Atarés, L.; Chiralt, A. Essential Oils as Additives in Biodegradable Films and Coatings for Active Food Packaging. Trends Food Sci. Technol. 2016, 48, 51–62. [Google Scholar] [CrossRef]
- Cai, C.; Ma, R.; Duan, M.; Deng, Y.; Liu, T.; Lu, D. Effect of Starch Film Containing Thyme Essential Oil Microcapsules on Physicochemical Activity of Mango. LWT 2020, 131, 109700. [Google Scholar] [CrossRef]
- Martins, P.C.; Bagatini, D.C.; Martins, V.G. Oregano Essential Oil Addition in Rice Starch Films and Its Effects on the Chilled Fish Storage. J. Food Sci. Technol. 2021, 58, 1562–1573. [Google Scholar] [CrossRef]
- Nisar, T.; Wang, Z.C.; Yang, X.; Tian, Y.; Iqbal, M.; Guo, Y. Characterization of Citrus Pectin Films Integrated with Clove Bud Essential Oil: Physical, Thermal, Barrier, Antioxidant and Antibacterial Properties. Int. J. Biol. Macromol. 2018, 106, 670–680. [Google Scholar] [CrossRef] [PubMed]
- Kurek, M.; Galus, S.; Debeaufort, F. Surface, Mechanical and Barrier Properties of Bio-Based Composite Films Based on Chitosan and Whey Protein. Food Packag. Shelf Life 2014, 1, 56–67. [Google Scholar] [CrossRef]
- Galus, S.; Lenart, A.; Voilley, A.; Debeaufort, F. Effect of Oxidized Potato Starch on the Physicochemical Properties of Soy Protein Isolate-Based Edible Films. Food Technol. Biotechnol. 2013, 51, 403–409. [Google Scholar]
- Pérez-Gago, M.B.; Rhim, J.W. Edible Coating and Film Materials: Lipid Bilayers and Lipid Emulsions. In Innovations in Food Packaging, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Gutiérrez, T.J.; Tapia, M.S.; Pérez, E.; Famá, L. Edible Films Based on Native and Phosphated 80:20 Waxy:Normal Corn Starch. Starch-Stärke 2015, 67, 90–97. [Google Scholar] [CrossRef]
- Kristo, E.; Biliaderis, C.G. Water Sorption and Thermo-Mechanical Properties of Water/Sorbitol-Plasticized Composite Biopolymer Films: Caseinate-Pullulan Bilayers and Blends. Food Hydrocoll. 2006, 20, 1057–1071. [Google Scholar] [CrossRef]
- Zikmanis, P.; Juhņeviča-Radenkova, K.; Radenkovs, V.; Segliņa, D.; Krasnova, I.; Kolesovs, S.; Orlovskis, Z.; Šilaks, A.; Semjonovs, P. Microbial Polymers in Edible Films and Coatings of Garden Berry and Grape: Current and Prospective Use. Food Bioprocess Technol. 2021, 14, 1432–1445. [Google Scholar] [CrossRef]
- Khwaldia, K.; Ferez, C.; Banon, S.; Desobry, S.; Hardy, J. Milk Proteins for Edible Films and Coatings. Crit. Rev. Food Sci. Nutr. 2004, 44, 239–251. [Google Scholar] [CrossRef]
- Khwaldia, K. Water Vapor Barrier And Mechanical Properties Of Paper-Sodium Caseinate And Paper-Sodium Caseinate-Paraffin Wax Films. J. Food Biochem. 2010, 34, 998–1013. [Google Scholar] [CrossRef]
- Gällstedt, M.; Brottmon, A.; Hedenqvist, M.S. Packaging-Related Properties of Protein- and Chitosan-Coated Paper. Packag. Technol. Sci. 2005, 18, 161–170. [Google Scholar] [CrossRef]
- Sadek, C.; Pauchard, L.; Schuck, P.; Fallourd, Y.; Pradeau, N.; Le Floch-Fouéré, C.; Jeantet, R. Mechanical Properties of Milk Protein Skin Layers after Drying: Understanding the Mechanisms of Particle Formation from Whey Protein Isolate and Native Phosphocaseinate. Food Hydrocoll. 2015, 48, 8–16. [Google Scholar] [CrossRef]
- Skurtys, O.; Acevedo, C.; Pedreschi, F.; Enronoe, J.; Osorio, F.; Aguilera, J.M. Food Hydrocolloid Edible Films and Coatings; Nova Science Publishers: Hauppauge, NY, USA, 2011. [Google Scholar]
- Krochta, J. Proteins as Raw Materials for Films and Coatings. In Protein-Based Films and Coatings; CRC Press: Boca Raton, FL, USA, 2002. [Google Scholar]
- Shahidi, F.; Hossain, A. Preservation of Aquatic Food Using Edible Films and Coatings Containing Essential Oils: A Review. Crit. Rev. Food Sci. Nutr. 2022, 62, 66–105. [Google Scholar] [CrossRef] [PubMed]
- Kouhi, M.; Prabhakaran, M.P.; Ramakrishna, S. Edible Polymers: An Insight into Its Application in Food, Biomedicine and Cosmetics. Trends Food Sci. Technol. 2020, 103, 248–263. [Google Scholar] [CrossRef]
- Redl, A.; Gontard, N.; Guilbert, S. Determination of Sorbic Acid Diffusivity in Edible Wheat Gluten and Lipid Based Films. J. Food Sci. 1996, 61, 116–120. [Google Scholar] [CrossRef]
- Kumari, M.; Mahajan, H.; Joshi, R.; Gupta, M. Development and Structural Characterization of Edible Films for Improving Fruit Quality. Food Packag. Shelf Life 2017, 12, 42–50. [Google Scholar] [CrossRef]
- Suhag, R.; Kumar, N.; Petkoska, A.T.; Upadhyay, A. Film Formation and Deposition Methods of Edible Coating on Food Products: A Review. Food Res. Int. 2020, 136, 109582. [Google Scholar] [CrossRef]
- Rhim, J.W.; Mohanty, A.K.; Singh, S.P.; Ng, P.K.W. Effect of the Processing Methods on the Performance of Polylactide Films: Thermocompression versus Solvent Casting. J. Appl. Polym. Sci. 2006, 101, 3736–3742. [Google Scholar] [CrossRef]
- Jensen, A.; Lim, L.T.; Barbut, S.; Marcone, M. Development and Characterization of Soy Protein Films Incorporated with Cellulose Fibers Using a Hot Surface Casting Technique. LWT-Food Sci. Technol. 2015, 60, 162–170. [Google Scholar] [CrossRef]
- Koide, Y.; Ikake, H.; Muroga, Y.; Shimizu, S. Effect of the Cast-Solvent on the Morphology of Cast Films Formed with a Mixture of Stereoisomeric Poly(Lactic Acids). Polym. J. 2013, 45, 645–650. [Google Scholar] [CrossRef]
- Sherrington, D.C. Introduction to Physical Polymer Science. React. Polym. 1993, 20, 217–218. [Google Scholar] [CrossRef]
- Cha, D.S.; Cooksey, K.; Chinnan, M.S.; Park, H.J. Release of Nisin from Various Heat-Pressed and Cast Films. LWT-Food Sci. Technol. 2003, 36, 209–213. [Google Scholar] [CrossRef]
- Karki, S.; Kim, H.; Na, S.J.; Shin, D.; Jo, K.; Lee, J. Thin Films as an Emerging Platform for Drug Delivery. Asian J. Pharm. Sci. 2016, 11, 559–574. [Google Scholar] [CrossRef]
- Cheng, Y.; Sun, C.; Zhai, X.; Zhang, R.; Zhang, S.; Sun, C.; Wang, W.; Hou, H. Effect of Lipids with Different Physical State on the Physicochemical Properties of Starch/Gelatin Edible Films Prepared by Extrusion Blowing. Int. J. Biol. Macromol. 2021, 185, 1005–1014. [Google Scholar] [CrossRef] [PubMed]
- Peressini, D.; Bravin, B.; Lapasin, R.; Rizzotti, C.; Sensidoni, A. Starch-Methylcellulose Based Edible Films: Rheological Properties of Film-Forming Dispersions. J. Food Eng. 2003, 59, 25–32. [Google Scholar] [CrossRef]
- Krishna, M.; Nindo, C.I.; Min, S.C. Development of Fish Gelatin Edible Films Using Extrusion and Compression Molding. J. Food Eng. 2012, 108, 337–344. [Google Scholar] [CrossRef]
- Liu, H.; Xie, F.; Yu, L.; Chen, L.; Li, L. Thermal Processing of Starch-Based Polymers. Prog. Polym. Sci. 2009, 34, 1348–1368. [Google Scholar] [CrossRef]
- Zhao, Y. Application of Commercial Coatings. In Edible Coatings and Films to Improve Food Quality, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2011; pp. 291–332. [Google Scholar] [CrossRef]
- Senturk Parreidt, T.; Schmid, M.; Müller, K. Effect of Dipping and Vacuum Impregnation Coating Techniques with Alginate Based Coating on Physical Quality Parameters of Cantaloupe Melon. J. Food Sci. 2018, 83, 929–936. [Google Scholar] [CrossRef]
- Andrade, R.D.; Skurtys, O.; Osorio, F.A. Atomizing Spray Systems for Application of Edible Coatings. Compr. Rev. Food Sci. Food Saf. 2012, 11, 323–337. [Google Scholar] [CrossRef]
- Debeaufort, F.; Voilley, A. Lipid-Based Edible Films and Coatings. In Edible Films and Coatings for Food Applications; Springer: Berlin/Heidelberg, Germany, 2009; pp. 135–168. [Google Scholar] [CrossRef]
- Schneller, T.; Waser, R.; Kosec, M.; Payne, D. Chemical Solution Deposition of Functional Oxide Thin Films; Springer: Vienna, Austrua, 2013; pp. 1–796. [Google Scholar] [CrossRef]
- Mastromatteo, M.; Conte, A.; Del Nobile, M.A. Packaging Strategies to Prolong the Shelf Life of Fresh Carrots (Daucus carota L.). Innov. Food Sci. Emerg. Technol. 2012, 13, 215–220. [Google Scholar] [CrossRef]
- Hamzah, H.M.; Osman, A.; Tan, C.P.; Mohamad Ghazali, F. Carrageenan as an Alternative Coating for Papaya (Carica papaya L. Cv. Eksotika). Postharvest Biol. Technol. 2013, 75, 142–146. [Google Scholar] [CrossRef]
- Costa, M.J.; Cerqueira, M.A.; Ruiz, H.A.; Fougnies, C.; Richel, A.; Vicente, A.A.; Teixeira, J.A.; Aguedo, M. Use of Wheat Bran Arabinoxylans in Chitosan-Based Films: Effect on Physicochemical Properties. Ind. Crops Prod. 2015, 66, 305–311. [Google Scholar] [CrossRef]
- Rojas-Graü, M.A.; Soliva-Fortuny, R.; Martín-Belloso, O. Edible Coatings to Incorporate Active Ingredients to Fresh-Cut Fruits: A Review. Trends Food Sci. Technol. 2009, 20, 438–447. [Google Scholar] [CrossRef]
- Soares, N.M.; Fernandes, T.A.; Vicente, A.A. Effect of Variables on the Thickness of an Edible Coating Applied on Frozen Fish—Establishment of the Concept of Safe Dipping Time. J. Food Eng. 2016, 171, 111–118. [Google Scholar] [CrossRef][Green Version]
- Kumar, N. Polysaccharide-Based Component and Their Relevance in Edible Film/Coating: A Review. Nutr. Food Sci. 2019, 49, 793–823. [Google Scholar] [CrossRef]
- Bravin, B.; Peressini, D.; Sensidoni, A. Development and Application of Polysaccharide-Lipid Edible Coating to Extend Shelf-Life of Dry Bakery Products. J. Food Eng. 2006, 76, 280–290. [Google Scholar] [CrossRef]
- Valdés, A.; Ramos, M.; Beltrán, A.; Jiménez, A.; Garrigós, M.C. State of the Art of Antimicrobial Edible Coatings for Food Packaging Applications. Coatings 2017, 7, 56. [Google Scholar] [CrossRef]
- Bergeron, V.; Bonn, D.; Martin, J.Y.; Vovelle, L. Controlling Droplet Deposition with Polymer Additives. Nature 2000, 405, 772–775. [Google Scholar] [CrossRef]
- Khan, M.K.I.; Nazir, A.; Maan, A.A. Electrospraying: A Novel Technique for Efficient Coating of Foods. Food Eng. Rev. 2017, 9, 112–119. [Google Scholar] [CrossRef]
- Bailey, A.G. The Science and Technology of Electrostatic Powder Spraying, Transport and Coating. J. Electrostat. 1998, 45, 85–120. [Google Scholar] [CrossRef]
- Amefia, A.E.; Abu-Ali, J.M.; Barringer, S.A. Improved Functionality of Food Additives with Electrostatic Coating. Innov. Food Sci. Emerg. Technol. 2006, 7, 176–181. [Google Scholar] [CrossRef][Green Version]
- Meng, X.; Zhu, J.; Zhang, H. Influences of Different Powders on the Characteristics of Particle Charging and Deposition in Powder Coating Processes. J. Electrostat. 2009, 67, 663–671. [Google Scholar] [CrossRef]
- Luo, Y.; Zhu, J.; Ma, Y.; Zhang, H. Dry Coating, a Novel Coating Technology for Solid Pharmaceutical Dosage Forms. Int. J. Pharm. 2008, 358. [Google Scholar] [CrossRef] [PubMed]
- Pawar Avinash, S.; Bageshwar Deepak, V.; Khanvilkar Vineeta, V.; Kadam Vilasrao, J. Advances in Pharmaceutical Coatings. Int. J. ChemTech Res. 2010, 2, 16–22. [Google Scholar]
- Sumonsiri, N.; Barringer, S.A. Effect of Powder and Target Properties on Wrap around Effect during Coating. J. Food Sci. 2010, 75, E537–E543. [Google Scholar] [CrossRef]
- Mazumder, M.K.; Sims, R.A.; Biris, A.S.; Srirama, P.K.; Saini, D.; Yurteri, C.U.; Trigwell, S.; De, S.; Sharma, R. Twenty-First Century Research Needs in Electrostatic Processes Applied to Industry and Medicine. Chem. Eng. Sci. 2006, 61, 2192–2211. [Google Scholar] [CrossRef]
- Barringer, S.A.; Sumonsiri, N. Electrostatic Coating Technologies for Food Processing. Annu. Rev. Food Sci. Technol. 2015, 6, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Abu-Ali, J.; Barringer, S.A. Optimization of Liquid Electrostatic Coating. J. Electrostat. 2008, 66, 184–189. [Google Scholar] [CrossRef]
- Watanabe, H.; Matsuyama, T.; Yamamoto, H. Experimental Study on Electrostatic Atomization of Highly Viscous Liquids. J. Electrostat. 2003, 57, 183–197. [Google Scholar] [CrossRef]
- Jaworek, A. Electrospray Droplet Sources for Thin Film Deposition. J. Mater. Sci. 2007, 42, 266–297. [Google Scholar] [CrossRef]
- Wilhelm, O.; Mädler, L.; Pratsinis, S.E. Electrospray Evaporation and Deposition. J. Aerosol Sci. 2003, 34, 815–836. [Google Scholar] [CrossRef]
- Xu, Y.; Hanna, M.A. Electrospray Encapsulation of Water-Soluble Protein with Polylactide. Effects of Formulations on Morphology, Encapsulation Efficiency and Release Profile of Particles. Int. J. Pharm. 2006, 320, 30–36. [Google Scholar] [CrossRef] [PubMed]
- Bock, N.; Dargaville, T.R.; Woodruff, M.A. Controlling Microencapsulation and Release of Micronized Proteins Using Poly(Ethylene Glycol) and Electrospraying. Eur. J. Pharm. Biopharm. 2014, 87, 366–377. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Masiá, R.; López-Nicolás, R.; Periago, M.J.; Ros, G.; Lagaron, J.M.; López-Rubio, A. Encapsulation of Folic Acid in Food Hydrocolloids through Nanospray Drying and Electrospraying for Nutraceutical Applications. Food Chem. 2015, 168, 124–133. [Google Scholar] [CrossRef]
- Sousa, A.M.M.; Souza, H.K.S.; Uknalis, J.; Liu, S.C.; Gonçalves, M.P.; Liu, L. Electrospinning of Agar/PVA Aqueous Solutions and Its Relation with Rheological Properties. Carbohydr. Polym. 2015, 115, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Gomez-Mascaraque, L.G.; Morfin, R.C.; Pérez-Masiá, R.; Sanchez, G.; Lopez-Rubio, A. Optimization of Electrospraying Conditions for the Microencapsulation of Probiotics and Evaluation of Their Resistance during Storage and In-Vitro Digestion. LWT-Food Sci. Technol. 2016, 69, 438–446. [Google Scholar] [CrossRef]
- Laelorspoen, N.; Wongsasulak, S.; Yoovidhya, T.; Devahastin, S. Microencapsulation of Lactobacillus Acidophilus in Zein-Alginate Core-Shell Microcapsules via Electrospraying. J. Funct. Foods 2014, 7, 342–349. [Google Scholar] [CrossRef]
- Pandey, P.; Turton, R.; Joshi, N.; Hammerman, E.; Ergun, J. Scale-up of Apan-Coating Process. AAPS PharmSciTech 2006, 7, E102–E132. [Google Scholar] [CrossRef]
- Dangaran, K.; Tomasula, P.M.; Qi, P. Structure and Function of Protein-Based Edible Films and Coatings. In Edible Films and Coatings for Food Applications; Springer: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Jacquot, M.; Pernetti, M. Spray Coating and Drying Processes. In Fundamentals of Cell Immobilization Biotechnology; Wolters Kluwer: Alphen aan den Rijn, The Netherlands, 2004. [Google Scholar]
- Guignon, B.; Duquenoy, A.; Dumoulin, E.D. Fluid Bed Encapsulation of Particles: Principles and Practice. Dry. Technol. 2002, 20, 419–447. [Google Scholar] [CrossRef]
- Chen, Y.; Yang, J.; Mujumdar, A.; Dave, R. Fluidized Bed Film Coating of Cohesive Geldart Group C Powders. Powder Technol. 2009, 189, 466–480. [Google Scholar] [CrossRef]
- Solís-Morales, D.; Sáenz-Hernández, C.M.; Ortega-Rivas, E. Attrition Reduction and Quality Improvement of Coated Puffed Wheat by Fluidised Bed Technology. J. Food Eng. 2009, 93, 236–241. [Google Scholar] [CrossRef]
- Bertuzzi, M.A.; Slavutsky, A.M. Standard and New Processing Techniques Used in the Preparation of Films and Coatings at the Lab Level and Scale-Up. In Edible Films and Coatings: Fundamentals and Applications; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Cerqueira, M.A.P.R.; Pereira, R.N.C.; da Silva Ramos, O.L.; Teixeira, J.A.C.; Vicente, A.A. (Eds.) Edible Food Packaging: Materials and Processing Technologies; CRC Press: Boca Raton, FL, USA, 2017. [Google Scholar]
- Karimnezhad, F.; Razavilar, V.; Anvar, A.A.; Eskandari, S. Study the Antimicrobial Effects of Chitosan-Based Edible Film Containing the Trachyspermum Ammi Essential Oil on Shelf-Life of Chicken Meat. Microbiol. Res. 2017, 8, 7226. [Google Scholar] [CrossRef]
- Martiny, T.R.; Pacheco, B.S.; Pereira, C.M.P.; Mansilla, A.; Astorga–España, M.S.; Dotto, G.L.; Moraes, C.C.; Rosa, G.S. A Novel Biodegradable Film Based on κ-Carrageenan Activated with Olive Leaves Extract. Food Sci. Nutr. 2020, 8, 3147–3156. [Google Scholar] [CrossRef]
- Li, K.; Jin, S.; Li, J.; Chen, H. Improvement in Antibacterial and Functional Properties of Mussel-Inspired Cellulose Nanofibrils/Gelatin Nanocomposites Incorporated with Graphene Oxide for Active Packaging. Ind. Crops Prod. 2019, 132, 197–212. [Google Scholar] [CrossRef]
- Calderón-Castro, A.; Vega-García, M.O.; de Jesús Zazueta-Morales, J.; Fitch-Vargas, P.R.; Carrillo-López, A.; Gutiérrez-Dorado, R.; Limón-Valenzuela, V.; Aguilar-Palazuelos, E. Effect of Extrusion Process on the Functional Properties of High Amylose Corn Starch Edible Films and Its Application in Mango (Mangifera indica L.) Cv. Tommy Atkins. J. Food Sci. Technol. 2018, 55, 905–914. [Google Scholar] [CrossRef] [PubMed]
- Seydim, A.C.; Sarikus-Tutal, G.; Sogut, E. Effect of Whey Protein Edible Films Containing Plant Essential Oils on Microbial Inactivation of Sliced Kasar Cheese. Food Packag. Shelf Life 2020, 26, 100567. [Google Scholar] [CrossRef]
- Xia, C.; Wang, W.; Wang, L.; Liu, H.; Xiao, J. Multilayer Zein/Gelatin Films with Tunable Water Barrier Property and Prolonged Antioxidant Activity. Food Packag. Shelf Life 2019, 19, 76–85. [Google Scholar] [CrossRef]
- Shapi’i, R.A.; Othman, S.H.; Nordin, N.; Kadir Basha, R.; Nazli Naim, M. Antimicrobial Properties of Starch Films Incorporated with Chitosan Nanoparticles: In Vitro and in Vivo Evaluation. Carbohydr. Polym. 2020, 230, 115602. [Google Scholar] [CrossRef]
- Belyamani, I.; Prochazka, F.; Assezat, G. Production and Characterization of Sodium Caseinate Edible Films Made by Blown-Film Extrusion. J. Food Eng. 2014, 121, 39–47. [Google Scholar] [CrossRef]
- Issa, A.; Ibrahim, S.A.; Tahergorabi, R. Impact of Sweet Potato Starch-Based Nanocomposite Films Activated with Thyme Essential Oil on the Shelf-Life of Baby Spinach Leaves. Foods 2017, 6, 43. [Google Scholar] [CrossRef]
- Kontominas, M.G. Use of Alginates as Food Packaging Materials. Foods 2020, 9, 1440. [Google Scholar] [CrossRef]
- Ramos, Ó.L.; Pereira, J.O.; Silva, S.I.; Fernandes, J.C.; Franco, M.I.; Lopes-da-Silva, J.A.; Pintado, M.E.; Malcata, F.X. Evaluation of Antimicrobial Edible Coatings from a Whey Protein Isolate Base to Improve the Shelf Life of Cheese. J. Dairy Sci. 2012, 95, 6282–6292. [Google Scholar] [CrossRef]
- Mali, S.; Grossmann, M.V.E. Effects of Yam Starch Films on Storability and Quality of Fresh Strawberries (Fragaria Ananassa). J. Agric. Food Chem. 2003, 51, 7005–7011. [Google Scholar] [CrossRef] [PubMed]
- Mushtaq, M.; Gani, A.; Gani, A.; Punoo, H.A.; Masoodi, F.A. Use of Pomegranate Peel Extract Incorporated Zein Film with Improved Properties for Prolonged Shelf Life of Fresh Himalayan Cheese (Kalari/Kradi). Innov. Food Sci. Emerg. Technol. 2018, 48, 25–32. [Google Scholar] [CrossRef]
- Sogut, E.; Seydim, A.C. The Effects of Chitosan and Grape Seed Extract-Based Edible Films on the Quality of Vacuum Packaged Chicken Breast Fillets. Food Packag. Shelf Life 2018, 18, 13–20. [Google Scholar] [CrossRef]
- Sorde, K.L.; Ananthanarayan, L. Effect of Transglutaminase Treatment on Properties of Coconut Protein-Guar Gum Composite Film. LWT 2019, 115, 108422. [Google Scholar] [CrossRef]
- Özdemir, K.S.; Gökmen, V. Effect of Chitosan-Ascorbic Acid Coatings on the Refrigerated Storage Stability of Fresh-Cut Apples. Coatings 2019, 9, 503. [Google Scholar] [CrossRef]
- Hashemi, S.M.B.; Zahabi, N.; Rezaee, Z.; Maherani, Z.; Boghori, P.; Keshavarz, Z. Evaluation of a Starch-Based Edible Film as Carrier of Adiantum Capillus-Veneris Extract to Improve the Shelf Life of Fresh-Cut Pears. J. Food Saf. 2016, 36, 291–298. [Google Scholar] [CrossRef]
- Vignesh, R.M.; Nair, B.R. Improvement of Shelf Life Quality of Tomatoes Using a Novel Edible Coating Formulation. Plant Sci. Today 2019, 6, 84–90. [Google Scholar] [CrossRef]
- Chen, C.; Peng, X.; Zeng, R.; Wan, C.; Chen, M.; Chen, J. Physiological and Biochemical Responses in Cold-Stored Citrus Fruits to Carboxymethyl Cellulose Coating Containing Ethanol Extract of Impatiens balsamina L. Stems. J. Food Process. Preserv. 2017, 41, e12999. [Google Scholar] [CrossRef]
- Falcó, I.; Flores-Meraz, P.L.; Randazzo, W.; Sánchez, G.; López-Rubio, A.; Fabra, M.J. Antiviral Activity of Alginate-Oleic Acid Based Coatings Incorporating Green Tea Extract on Strawberries and Raspberries. Food Hydrocoll. 2019, 87, 611–618. [Google Scholar] [CrossRef]
- García, M.A.; Ventosa, M.; Díaz, R.; Falco, S.; Casariego, A. Effects of Aloe Vera Coating on Postharvest Quality of Tomato. Fruits 2014, 69, 117–126. [Google Scholar] [CrossRef]
- Sessa, M.; Ferrari, G.; Donsì, F. Novel Edible Coating Containing Essential Oil Nanoemulsions to Prolong the Shelf Life of Vegetable Products. Chem. Eng. Trans. 2015, 43, 55–60. [Google Scholar] [CrossRef]
- Martiñon, M.E.; Moreira, R.G.; Castell-Perez, M.E.; Gomes, C. Development of a Multilayered Antimicrobial Edible Coating for Shelf-Life Extension of Fresh-Cut Cantaloupe (Cucumis melo L.) Stored at 4 °C. LWT-Food Sci. Technol. 2014, 56, 341–350. [Google Scholar] [CrossRef]
- Martínez, O.; Salmerón, J.; Epelde, L.; Vicente, M.S.; de Vega, C. Quality Enhancement of Smoked Sea Bass (Dicentrarchus labrax) Fillets by Adding Resveratrol and Coating with Chitosan and Alginate Edible Films. Food Control 2018, 85, 168–176. [Google Scholar] [CrossRef]
- Lagos, M.J.B.; Sobral, P.J.D.A. Application of Active Films with Natural Extract for Beef Hamburger Preservation. Cienc. Rural 2019, 49, e20180797. [Google Scholar] [CrossRef]
- Shin, S.H.; Chang, Y.; Lacroix, M.; Han, J. Control of Microbial Growth and Lipid Oxidation on Beef Product Using an Apple Peel-Based Edible Coating Treatment. LWT-Food Sci. Technol. 2017, 84, 183–188. [Google Scholar] [CrossRef]
- Kowalczyk, D.; Skrzypek, T.; Łupina, K. Effect of Carboxymethyl Cellulose/Candelilla Wax Edible Coating Incorporated with Ascorbic Acid on the Physicochemical and Sensory Qualities of Prepackaged Minimally Processed Carrots (Daucus carota L.) during Cold Storage. J. Food Process. Preserv. 2020, 44, e14713. [Google Scholar] [CrossRef]
- López De Lacey, A.M.; López-Caballero, M.E.; Gómez-Estaca, J.; Gómez-Guillén, M.C.; Montero, P. Functionality of Lactobacillus acidophilus and Bifidobacterium bifidum incorporated to edible coatings and films. Innov. Food Sci. Emerg. Technol. 2012, 16, 277–282. [Google Scholar] [CrossRef]
- Zhao, Y.; Teixeira, J.S.; Gänzle, M.M.; Saldaña, M.D.A. Development of antimicrobial films based on cassava starch, chitosan and gallic acid using subcritical water technology. J. Supercrit. Fluids 2018, 137, 101–110. [Google Scholar] [CrossRef]
- Olaimat, A.N.; Fang, Y.; Holley, R.A. Inhibition of Campylobacter jejuni on fresh chicken breasts by κ-carrageenan/chitosan-based coatings containing allyl isothiocyanate or deodorized oriental mustard extract. Int. J. Food Microbiol. 2014, 187, 77–82. [Google Scholar] [CrossRef]
- Sanchez-Gonzalez, L.; Pastor, C.; Vargas, M.; Chiralt, A.; Gonzalez-Martinez, C.; Chafer, M. Effect of hydroxypropylmethylcellulose and chitosan coatings with and without bergamot essential oil on quality and safety of cold-stored grapes. Postharvest Biol. Technol. 2011, 60, 57–63. [Google Scholar] [CrossRef]
- Calva-Estrada, S.J.; Jiménez-Fernández, M.; Lugo-Cervantes, E. Protein-Based Films: Advances in the Development of Biomaterials Applicable to Food Packaging. Food Eng. Rev. 2019, 11, 78–92. [Google Scholar] [CrossRef]
- Ganiari, S.; Choulitoudi, E.; Oreopoulou, V. Edible and Active Films and Coatings as Carriers of Natural Antioxidants for Lipid Food. Trends Food Sci. Technol. 2017, 68, 70–82. [Google Scholar] [CrossRef]
- Sharma, R.; Ghoshal, G. Emerging Trends in Food Packaging. Nutr. Food Sci. 2018, 48, 764–779. [Google Scholar] [CrossRef]
- Parreidt, T.S.; Lindner, M.; Rothkopf, I.; Schmid, M.; Müller, K. The Development of a Uniform Alginate-Based Coating for Cantaloupe and Strawberries and the Characterization of Water Barrier Properties. Foods 2019, 8, 203. [Google Scholar] [CrossRef]
- Gaikwad, K.K.; Singh, S.; Negi, Y.S.; Lee, Y.S. The Effect of Trans-Polyisoprene/LDPE Based Active Films on Oxidative Stability in Roasted Peanuts. J. Food Meas. Charact. 2020, 14, 1857–1864. [Google Scholar] [CrossRef]
- Ustunol, Z. Edible Films and Coatings for Meat and Poultry. In Edible Films and Coatings for Food Applications; Springer: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Puscaselu, R.; Gutt, G.; Amariei, S. The Use of Edible Films Based on Sodium Alginate in Meat Product Packaging: An Eco-Friendly Alternative to Conventional Plastic Materials. Coatings 2020, 10, 166. [Google Scholar]
- Andevari, G.T.; Rezaei, M. Effect of Gelatin Coating Incorporated with Cinnamon Oil on the Quality of Fresh Rainbow Trout in Cold Storage. Int. J. Food Sci. Technol. 2011, 46, 2305–2311. [Google Scholar] [CrossRef]
- Arshad, R.; Sameen, A.; Huma, N.; Zia, M.A. Exploring the Potential of Active Edible Coating on the Shelf Stability of Dairy Products. Pak. J. Agric. Sci. 2020, 57, 237–244. [Google Scholar] [CrossRef]
- Hashemi, S.M.B.; Raeisi, S. Evaluation of Antifungal and Antioxidant Properties of Edible Coating Based on Apricot (Prunus armeniaca) Gum Containing Satureja Intermedia Extract in Fresh Wild Almond (Amygdalus scoparia) Kernels. J. Food Meas. Charact. 2018, 12, 362–369. [Google Scholar] [CrossRef]
- Mendy, T.K.; Misran, A.; Mahmud, T.M.M.; Ismail, S.I. Application of Aloe Vera Coating Delays Ripening and Extend the Shelf Life of Papaya Fruit. Sci. Hortic. 2019, 246, 769–776. [Google Scholar] [CrossRef]
- Available online: https://www.gminsights.com/industry-analysis/biopolymer-films-market (accessed on 1 August 2022).
- Xiong, S.J.; Pang, B.; Zhou, S.J.; Li, M.K.; Yang, S.; Wang, Y.Y.; Shi, Q.; Wang, S.F.; Yuan, T.Q.; Sun, R.C. Economically competitive biodegradable PBAT/lignin composites: Effect of lignin methylation and compatibilizer. ACS Sustain. Chem. Eng. 2020, 8, 5338–5346. [Google Scholar] [CrossRef]





| Thermoplastic Material | Abbreviation | Packaging Application | Pros and Cons |
|---|---|---|---|
| Polyethylene terephthalate | PET | Water/juice/soft drink bottles, food jars, microwavable containers, plastic films | Good mechanical strength and barrier properties, but low heat resistance and susceptible to oxidation |
| Polypropylene | PP | Drinking bottles for milk, food containers | Good chemical and moisture barrier, difficult to recycle |
| Polyvinyl chloride | PVC | Plastic bags, frozen foods, stretch films, container lids | Flexible, cost effective, difficult to recycle, but low heat resistance as adipates in PVC leach into food |
| Polystyrene | PS | Take-away clamshells, meat trays, bottle caps, straws | Easily recyclable, hard and brittle, but poor chemical resistance, can leach out into food when food is stored |
| Low-density polyethylene | LDP | Disposable cups, plates, and spoons; bread bags | Good chemical resistance, relatively permeable to oxygen but poor odor barrier |
| High-density polyethylene | HDP | Custom packaging, grocery bags, water/milk/juice containers, cereal and snack liners | Good moisture barrier, but poor gas barrier and low heat resistance |
| Sl. No. | Type of Method | Film Formation | Advantage | Disadvantage | Application |
|---|---|---|---|---|---|
| 1. | Solvent casting method (Wet) | Biomaterial is dissolved in suitable solvent (e.g., ethanol, hexane) then cast into a mold followed by drying. | Low cost, ease of operation, fewer defects in film (homogeneous packaging), good optical purity, transparency, excellent flatness, and isotropic orientation. | Limitation of shapes, entrapment of toxic solvent in polymer, long drying period. | Apples, strawberries, quail eggs, cheese slices, etc. |
| 2. | Extrusion method (Dry) | Biomaterial is mixed with solvent and then feed with the help of compressed air; it is then kneaded and heated in the machine, followed by the formation of the finished film. | Short duration of heating, low energy consumption, good mechanical, and optical properties, cheap and highly efficient, no use of solvent. | Can process materials that can tolerate high temperatures; high investment, and maintenance cost. | Mangos, soyabean oil, sausage |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gupta, V.; Biswas, D.; Roy, S. A Comprehensive Review of Biodegradable Polymer-Based Films and Coatings and Their Food Packaging Applications. Materials 2022, 15, 5899. https://doi.org/10.3390/ma15175899
Gupta V, Biswas D, Roy S. A Comprehensive Review of Biodegradable Polymer-Based Films and Coatings and Their Food Packaging Applications. Materials. 2022; 15(17):5899. https://doi.org/10.3390/ma15175899
Chicago/Turabian StyleGupta, Vatsla, Deblina Biswas, and Swarup Roy. 2022. "A Comprehensive Review of Biodegradable Polymer-Based Films and Coatings and Their Food Packaging Applications" Materials 15, no. 17: 5899. https://doi.org/10.3390/ma15175899
APA StyleGupta, V., Biswas, D., & Roy, S. (2022). A Comprehensive Review of Biodegradable Polymer-Based Films and Coatings and Their Food Packaging Applications. Materials, 15(17), 5899. https://doi.org/10.3390/ma15175899







