Hydrophilic CO-Releasing Material of PEGlyated Ruthenium Carbonyl Complex
Abstract
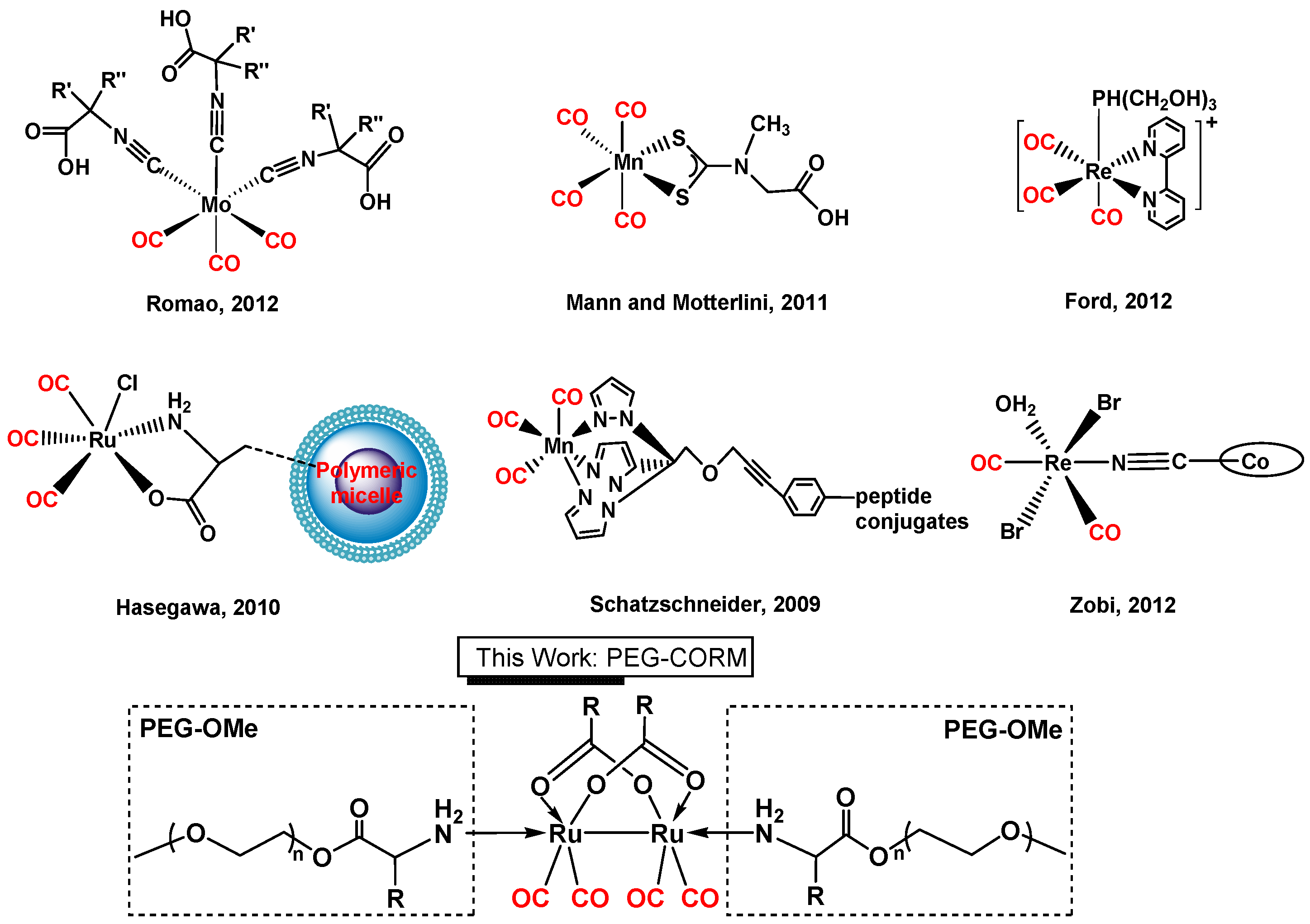
1. Introduction
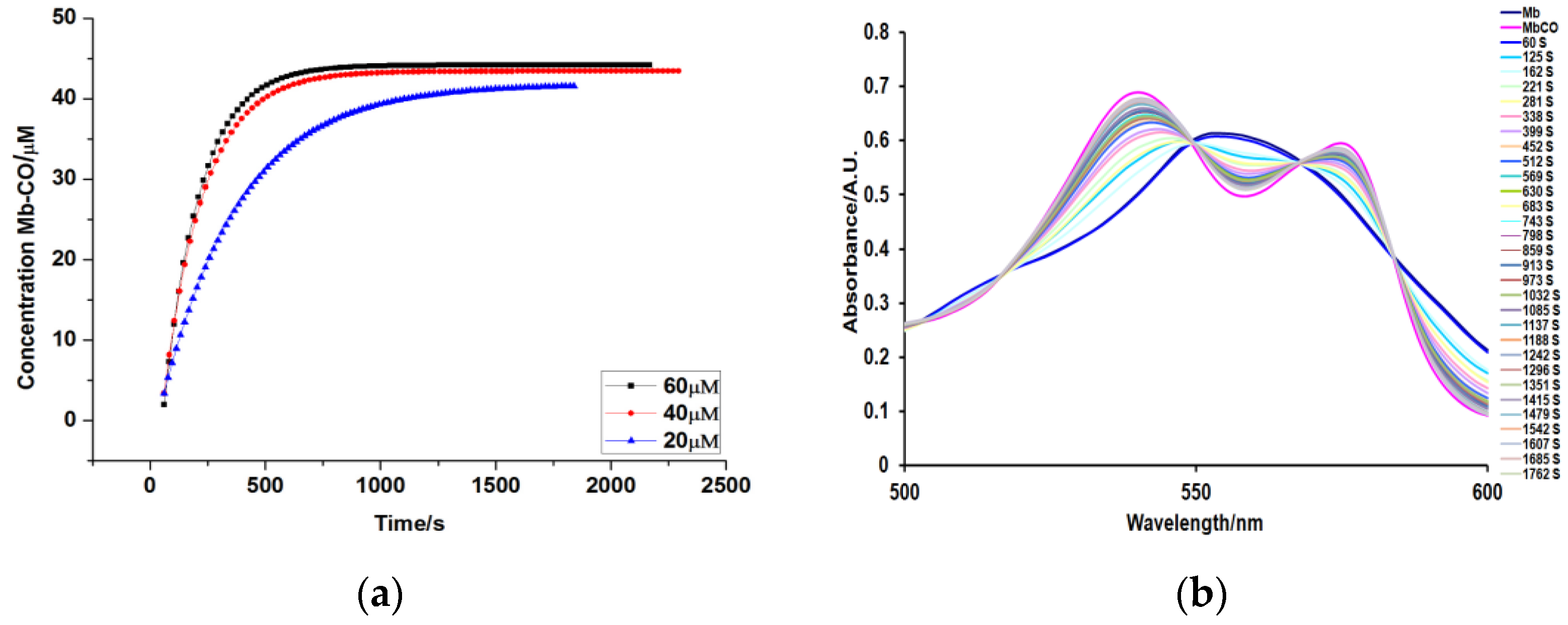
2. Results and Discussion
3. Materials and Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Motterlini, R.; Otterbein, L.E. Selected Reviews of therapeutic application of CO and CO-RM. The therapeutic potential of carbon monoxide. Nat. Rev. Drug Discov. 2010, 9, 728–743. [Google Scholar] [CrossRef] [PubMed]
- Bernardes, G.J.L. Carbon-Monoxide-Releasing Molecules for the Delivery of Therapeutic CO In Vivo. Angew. Chem. Int. Ed. 2014, 53, 9712–9721. [Google Scholar] [CrossRef]
- Ryter, S.W.; Alam, J.; Choi, A.M.K. Heme Oxygenase-1/Carbon Monoxide: From Basic Science to Therapeutic Applications. Physiol. Rev. 2006, 86, 583–650. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Joe, Y.; Yu, J.K.; Chen, Y.; Jeong, S.O.; Mani, N.; Cho, G.J.; Pae, H.; Ryterd, S.W.; Chung, H.T. Carbon monoxide protects against hepatic ischemia/reperfusion injury by modulating the miR-34a/SIRT1 pathway. Biochim. Biophys. Acta 2015, 1852, 1550–1559. [Google Scholar] [CrossRef]
- Wei, Y.; Chen, P.; Bruyn, M.; Zhang, W.; Bremer, E.; Helfrich, W. Carbon monoxide-Releasing Molecule-2 (CO-RM-2) attenuates acute hepatic ischemia reperfusion injury in rats. BMC Gastroenterol. 2010, 10, 42. [Google Scholar] [CrossRef]
- Caumartin, Y.; Stephen, J.; Deng, J.; Lian, D.; Lan, Z.; Liu, W.; Garcia, B.; Jevnikar, A.M.; Wang, H.; Cepinskas, G.; et al. Carbon monoxide-releasing molecules protect against ischemia-reperfusion injury during kidney transplantation. Kidney Int. 2011, 79, 1080–1089. [Google Scholar] [CrossRef]
- Kretschmer, R.; Gessner, G.; Görls, H.; Heinemann, S.H.; Westerhausen, M. Dicarbonyl-bis(cysteamine)iron(II): A light induced carbon monoxide releasing molecule based on iron(CO-RM-S1). J. Inorg. Biochem. 2011, 105, 6–9. [Google Scholar] [CrossRef]
- Romanski, S.; Kraus, B.; Schatzschneider, U.; Neudörfl, J.M.; Amslinger, S.; Schmalz, H.G. Acyloxybutadiene Iron Tricarbonyl Complexes as Enzyme-Triggered CO-Releasing Molecules (ET-CO-RMs). Angew. Chem. Int. Ed. 2011, 50, 2392–2396. [Google Scholar] [CrossRef]
- Lomont, J.P.; Nguyen, S.C.; Harris, C.B. Exploring the Utility of Tandem Thermal–Photochemical CO Delivery with CORM-2. Organometallic 2014, 33, 6179–6185. [Google Scholar] [CrossRef]
- Johnson, T.R.; Mann, B.E.; Teasdale, I.P.; Adams, H.; Foresti, R.; Green, C.J.; Motterlini, R. Metal carbonyls as pharmaceuticals? [Ru(CO)3Cl(glycinate)], a CO-Releasing molecule with an extensive aqueous solution chemistry. Dalton Trans. 2007, 15, 1500–1508. [Google Scholar] [CrossRef]
- Seixas, J.D.; Mukhopadhyay, A.; Santos-Silva, T.; Otterbein, L.E.; Gallo, D.J.; Rodrigues, S.S.; Guerreiro, B.H.; Gonçalves, A.M.L.; Penacho, N.; Marques, A.R.; et al. Characterization of a versatile organometallic pro-drug (CO-RM) for experimental CO based therapeutics. Dalton Trans. 2013, 42, 5985–5998. [Google Scholar] [CrossRef] [PubMed]
- Poh, H.T.; Sim, B.T.; Chwee, T.S.; Leong, W.K.; Fan, W.Y. The Dithiolate-Bridged Diiron Hexacarbonyl Complex Na2[(μ-SCH2CH2COO)Fe(CO)3]2 as a Water-Soluble PhotoCO-RM. Organometallics 2014, 33, 959–963. [Google Scholar] [CrossRef]
- Kianfar, E.; Monkowius, U.; Portenkirchner, E.; Knöer, G.; Naturforsch, Z. Synthesis and Characterization of Novel Re(BIAN)(CO)(3)Cl Derivatives Including the First Example of a Water-soluble Tricarbonyl Rhenium(I) Complex with Bis(imino)acenaphthene Ligands. Z. Naturforsch. B 2014, 69, 691–698. [Google Scholar] [CrossRef]
- Mede, R.; Klein, M.; Claus, R.A.; Krieck, S.; Quickert, S.; Görls, H.; Neugebauer, U.; Schmitt, M.; Gessner, G.; Heinemann, S.H.; et al. CO-RM-EDE1: A Highly Water-Soluble and Nontoxic Manganese-Based photoCO-RM with a Biogenic Ligand Sphere. Inorg. Chem. 2016, 55, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Atkin, A.J.; Fairlamb, I.J.S.; Whitwood, A.C.; Lynam, J.M. Synthesis and Reactivity of Molybdenum Complexes Containing Functionalized Alkynyl Ligands: A Photochemically Activated CO-Releasing Molecule (PhotoCO-RM). Organometallics 2011, 30, 4643–4654. [Google Scholar] [CrossRef]
- Marques, A.R.; Kromer, L.; Bento, I.; Otterbein, L.E.; Blattler, W.A.; Romao, C.C. Generation of Carbon Monoxide Releasing Molecules(CO-RMs) as Drug Can didates for the Treatment of Acute Liver Injury: Targeting of CO-RMs to the Liver. Organometallics 2012, 31, 5810–5822. [Google Scholar] [CrossRef]
- Crook, S.H.; Mann, B.E.; Meijer, A.J.H.M.; Adams, H.; Sawle, P.; Scapens, D.; Motterlini, R. [Mn(CO)4{S2CNMe(CH2CO2H)}], a new water-soluble CO-releasing molecule. Dalton Trans. 2011, 40, 4230–4235. [Google Scholar] [CrossRef] [PubMed]
- Pierri, A.E.; Pallaoro, A.; Wu, G.; Ford, P.C. A Luminescent and Biocompatible PhotoCO-RM. J. Am. Chem. Soc. 2012, 134, 18197–18200. [Google Scholar] [CrossRef]
- Hasegawa, U.; Vlies, A.J.; Simeoni, E.; Wandrey, C.; Hubbell, J.A. Carbon Monoxide-Releasing Micelles for Immunotherapy. J. Am. Chem. Soc. 2010, 132, 18273–18280. [Google Scholar] [CrossRef]
- Pfeiffer, H.; Rojas, A.; Niesel, J.; Schatzschneider, U. Sonogashira and “Click” reactions for the N-terminalandside-chain functionalization of peptides with [Mn(CO)3(tpm)]+-based CO releasing molecules (tpm = tris(pyrazolyl)methane). Dalton Trans. 2009, 22, 4292–4298. [Google Scholar] [CrossRef]
- Zobi, F.; Blacque, O.; Robert, A.; Marcus, J.; Schaubc, C.; Bogdanova, A.Y. 17e− rhenium dicarbonyl CO-releasing molecules on a cobalamin scaffold for biological application. Dalton Trans. 2012, 41, 370. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Liu, H.; Zhao, Q.; Chen, Y.; Liu, B.; Zhang, B.; Zheng, Q. Syntheses and evaluation of drug-like properties of CO-releasing molecules containing ruthenium and group 6 metal. Eur. J. Med. Chem. 2014, 74, 199–215. [Google Scholar] [CrossRef] [PubMed]
- Ringsdorf, H. Structure and properties of pharmacologically active polymers. J. Poly. Sci. 1975, 51, 135–153. [Google Scholar] [CrossRef]
- Pasut, G.; Sergi, M.; Veronese, F.M. Anti-cancer PEG-enzymes: 30 Years Old, but Still a Current Approach. Adv. Drug Deliv. Rev. 2008, 60, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Crooks, G.R.; Johnson, B.F.G.; Lewis, J.; Williams, I.G.; Gamlen, G. Chemistry of polynuclear compounds. Part XVII. Some carboxylate complexes of ruthenium and osmium carbonyls. J. Chem. Soc. A 1969, 2761–2766. [Google Scholar] [CrossRef]
- Therrien, B.; Suss-Fink, G. Sawhorse-type Diruthenium Tetracarbonyl Complexes. Coord. Chem. Rev. 2009, 253, 2639. [Google Scholar] [CrossRef][Green Version]
- Yang, S.; Chen, M.; Zhou, L.; Zhang, G.; Gao, Z.; Zhang, W. Photo-activated CO-releasing Molecules (PhotoCO-RMs) of Robust Sawhorse Scaffolds [µ2-OOCR1,η1-NH2CHR2(C=O]OCH3,Ru(I)2CO4]. Dalton Trans. 2016, 45, 3727–3733. [Google Scholar] [CrossRef]
- Glomme, A.; März, J.; Dressman, J.B. Comparison of a Miniaturized Shake-Flask Solubility Method with Automated Potentiometric Acid/Base Titrations and Calculated Solubilities. J. Pharm. Sci. 2005, 94, 1–16. [Google Scholar] [CrossRef]
- SAINT. Data Reduction Software; Bruker AXS: Madison, WI, USA, 2009. [Google Scholar]
- Sheldrick, G.M. SADABS, Program for Empirical Absorption Correction of Area Detector Data; University of Gottingen: Gottingen, Germany, 2010. [Google Scholar]
- Dolomanov, O.V.; Bourhis, L.J.; Gildea, R.J.; Howard, J.A.K.; Puschmann, H. A Complete Structure Solution, Refinement and Analysis Program. J. Appl. Cryst. 2009, 42, 339–341. [Google Scholar] [CrossRef]

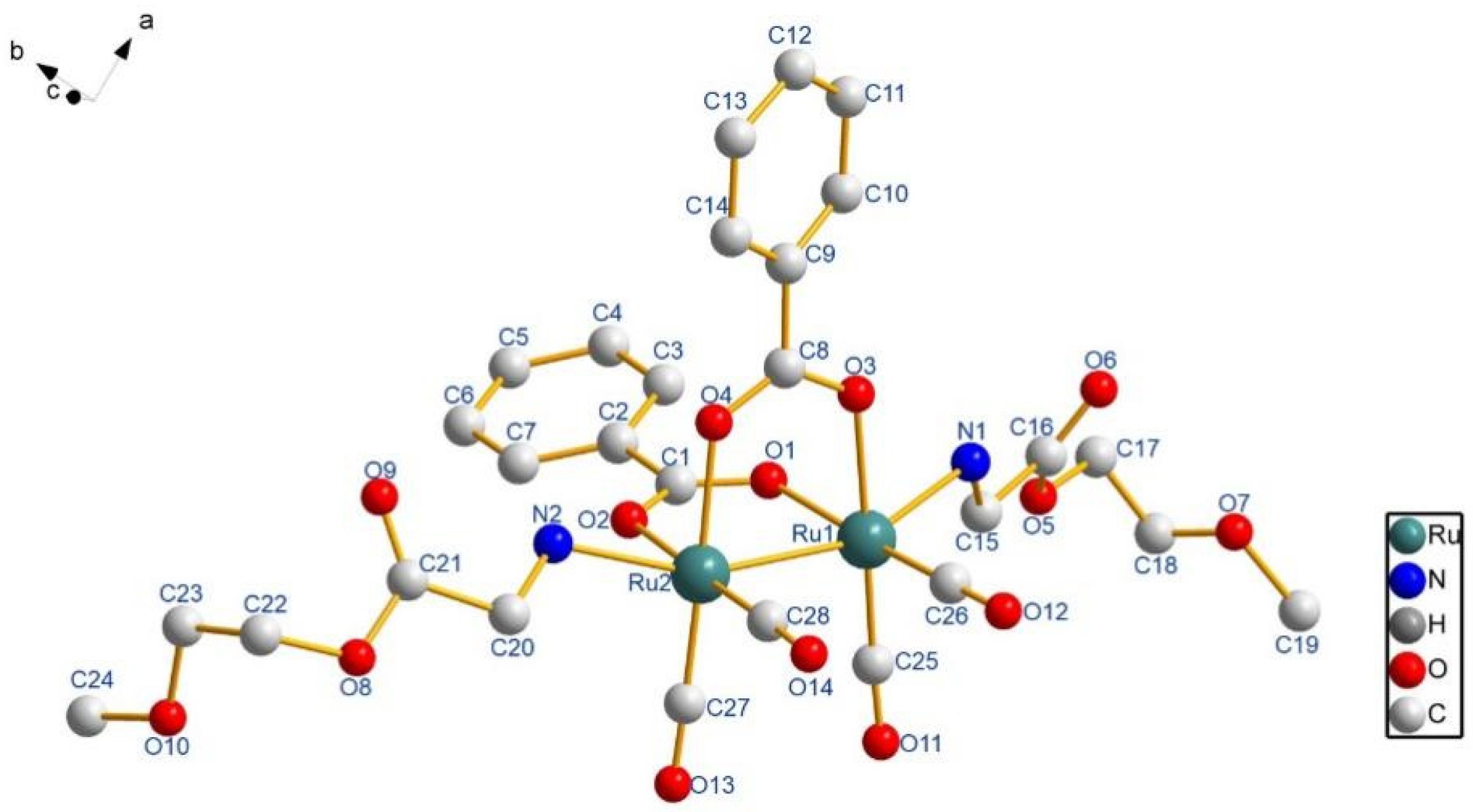

| Entry | Ru(1)–Ru(2) | Ru(1)–N(1) | Ru(2)–N(2) | Ru(1)–Ru(2)–N(2) | Ru(2)–Ru(1)–N(1) |
|---|---|---|---|---|---|
| 8 | 2.6694(10) | 2.239(5) | 2.210(5) | 157.86(15) | 158.27(15) |
| 13 | 2.6634(7) | 2.249(4) | 2.249(4) | 159.09(11) | 160.28(11) |
| 14 | 2.6727(5) | 2.233(4) | 2.240(4) | 158.27(11) | 159.84(10) |
| CO-RM | logP a. | t1/2, 60 µM b. |
|---|---|---|
| 4 | 0.39 | 166 |
| 5 | 1.41 | 276 |
| 6 | 1.17 | 249 |
| 7 | 1.05 | 189 |
| 8 | 1.71 | 1209 |
| 9 | 1.67 | 632 |
| 10 | 1.03 | 962 |
| 11 | 1.06 | 1096 |
| 12 | 1.78 | 1450 |
| 13 | 1.26 | 966 |
| 14 | N. D. | 2699 |
| 15 | N. D. | 2472 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Guo, N.; Yang, S.; Khan, H.; Zhang, W. Hydrophilic CO-Releasing Material of PEGlyated Ruthenium Carbonyl Complex. Materials 2022, 15, 3597. https://doi.org/10.3390/ma15103597
Zhang X, Guo N, Yang S, Khan H, Zhang W. Hydrophilic CO-Releasing Material of PEGlyated Ruthenium Carbonyl Complex. Materials. 2022; 15(10):3597. https://doi.org/10.3390/ma15103597
Chicago/Turabian StyleZhang, Xiao, Nan Guo, Shuhong Yang, Huma Khan, and Weiqiang Zhang. 2022. "Hydrophilic CO-Releasing Material of PEGlyated Ruthenium Carbonyl Complex" Materials 15, no. 10: 3597. https://doi.org/10.3390/ma15103597
APA StyleZhang, X., Guo, N., Yang, S., Khan, H., & Zhang, W. (2022). Hydrophilic CO-Releasing Material of PEGlyated Ruthenium Carbonyl Complex. Materials, 15(10), 3597. https://doi.org/10.3390/ma15103597







