A Low-Cost Porous Polymer Membrane for Gas Permeation
Abstract
1. Introduction
2. Experimental
2.1. Materials
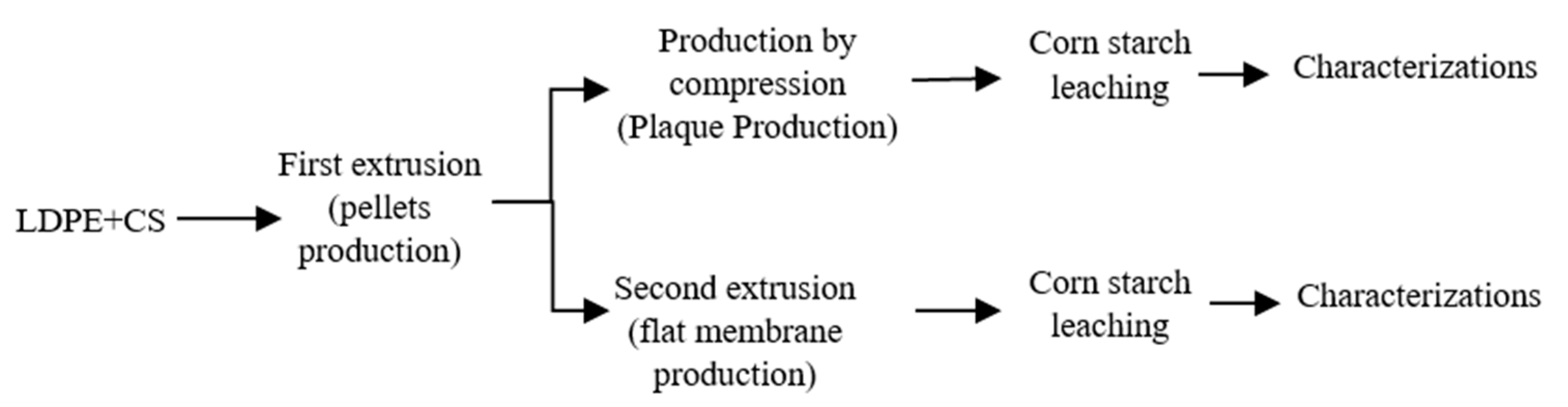
2.2. Membrane Preparation
2.2.1. Production of the Membrane by Extrusion
2.2.2. Production of the Membrane by Compression
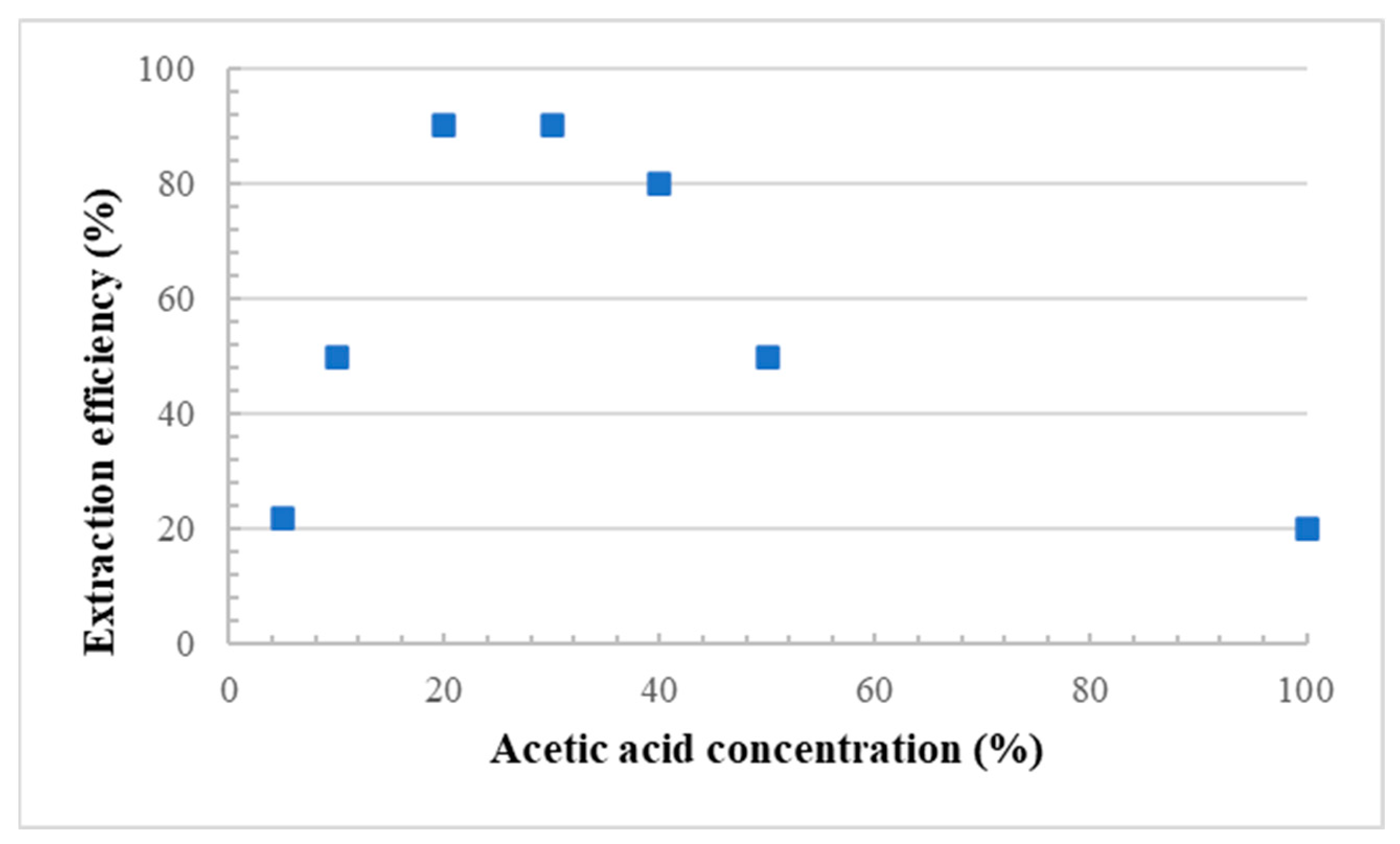
2.2.3. Formation of the Porous Structure
2.3. Characterizations
2.3.1. Scanning Electron Microscopy (SEM)
2.3.2. Differential Scanning Calorimetry (DSC)
2.3.3. Thermogravimetric Analysis (TGA)
2.3.4. Density
2.3.5. Tensile Properties
2.3.6. Contact Angle
2.3.7. Gas Permeability
3. Results and Discussion
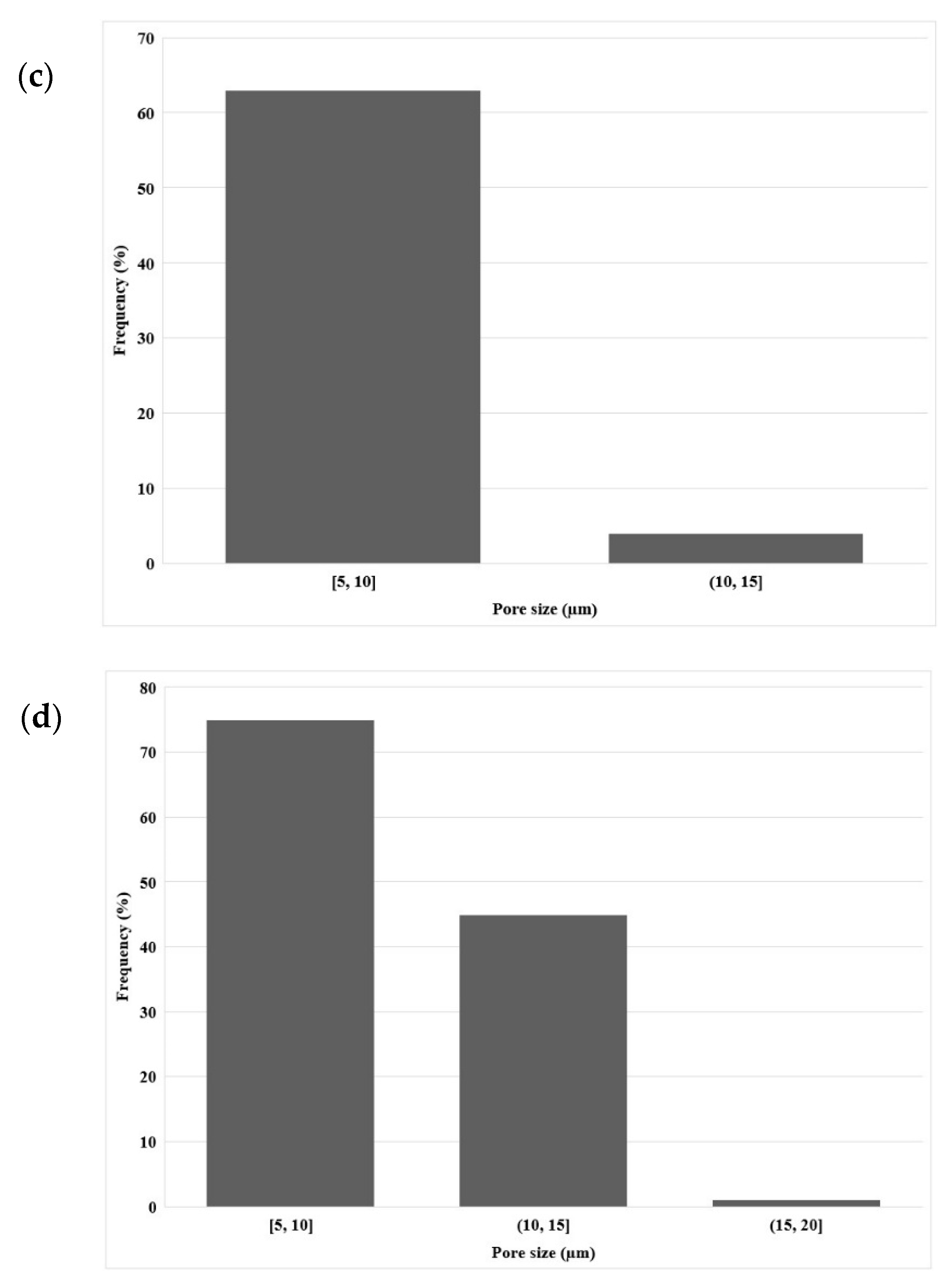
3.1. Morphology
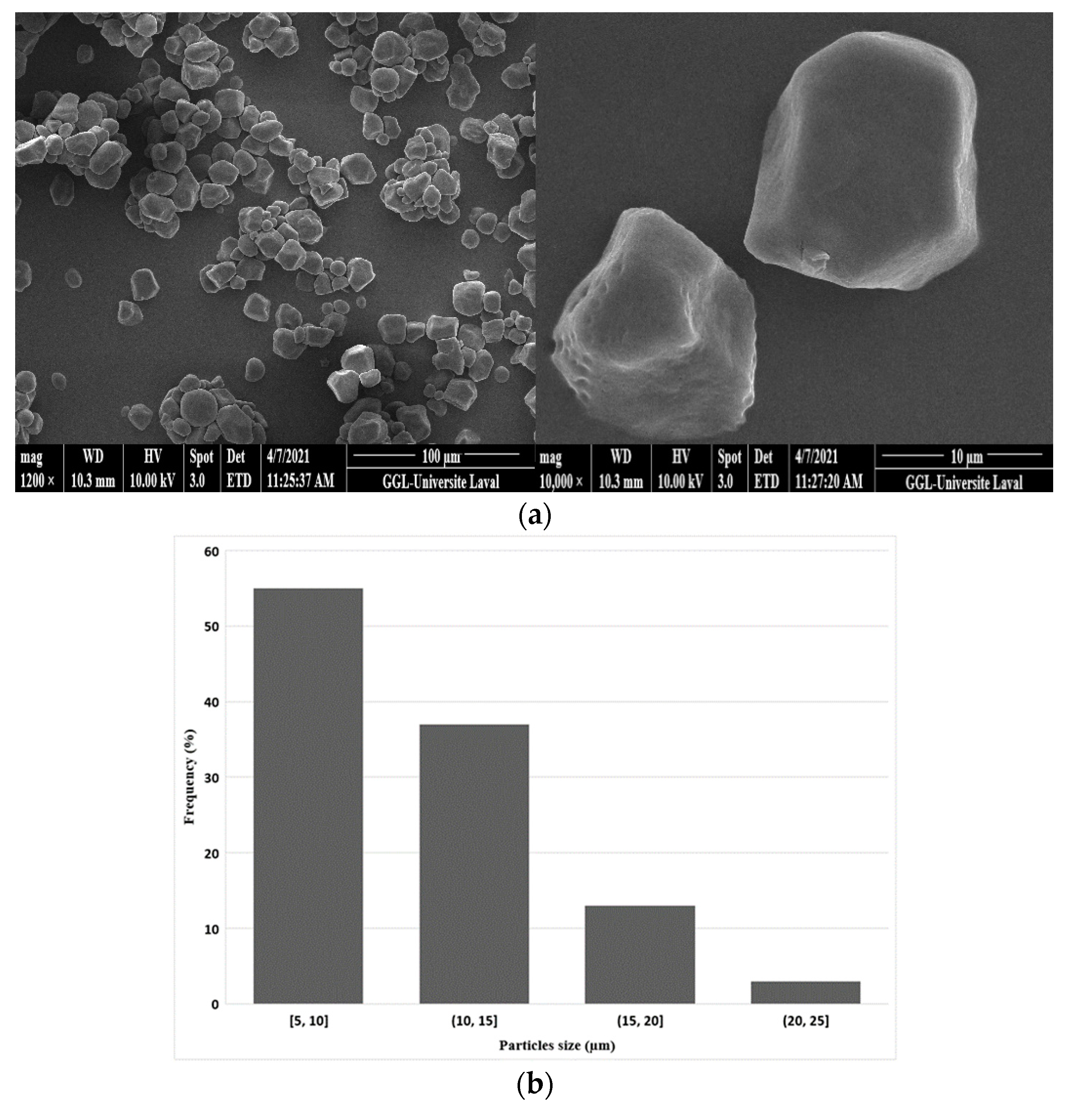
3.1.1. Starch Morphology
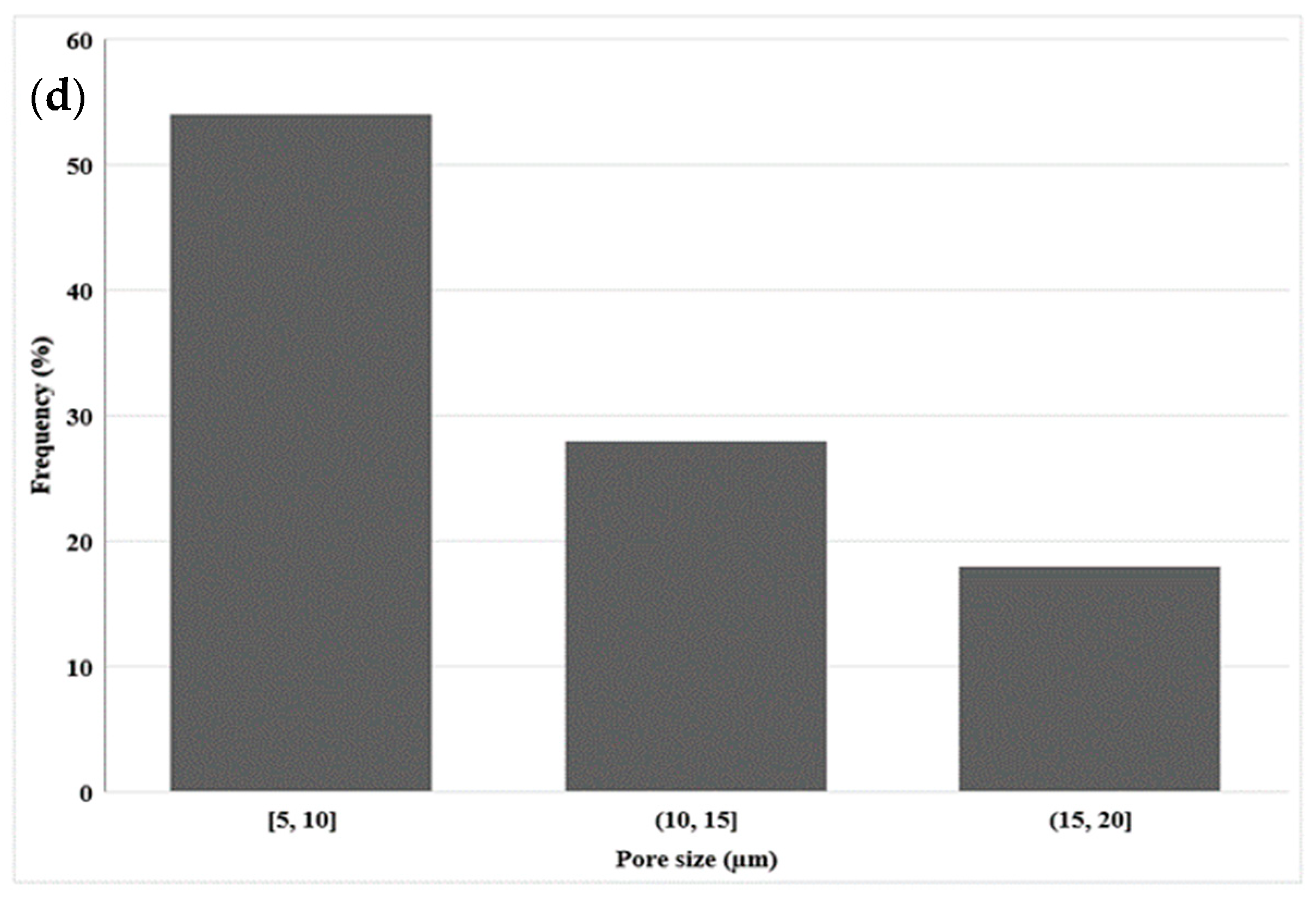
3.1.2. Morphology of LDPE Membrane after Leaching (Extrusion)
3.1.3. Morphology of LDPE Membrane after Leaching (Compression)
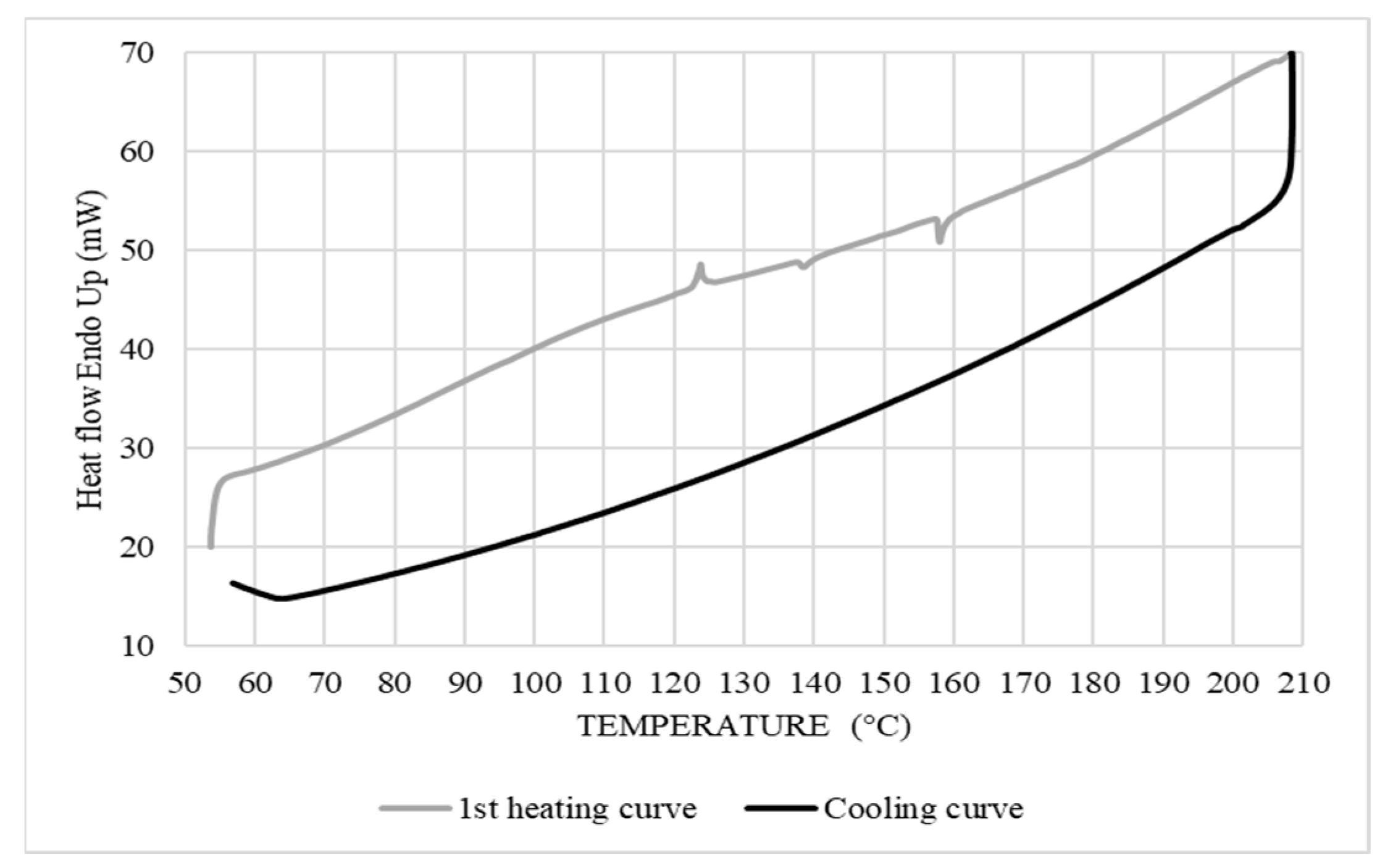
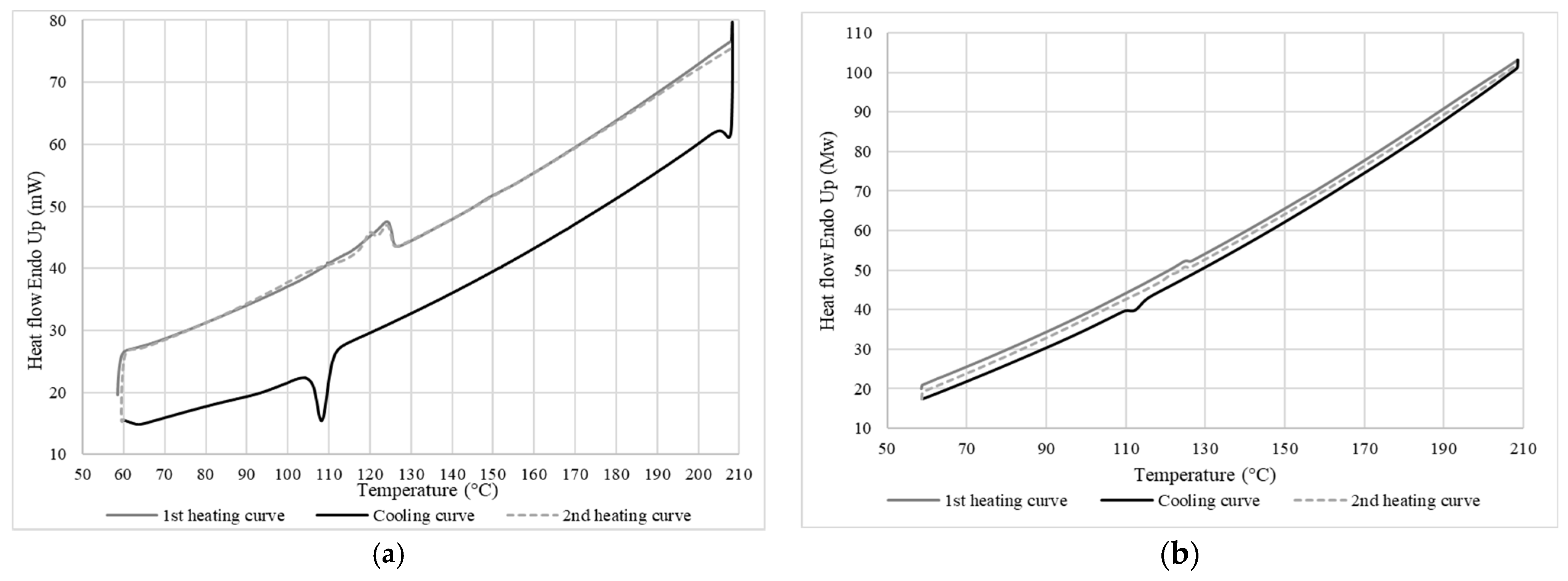
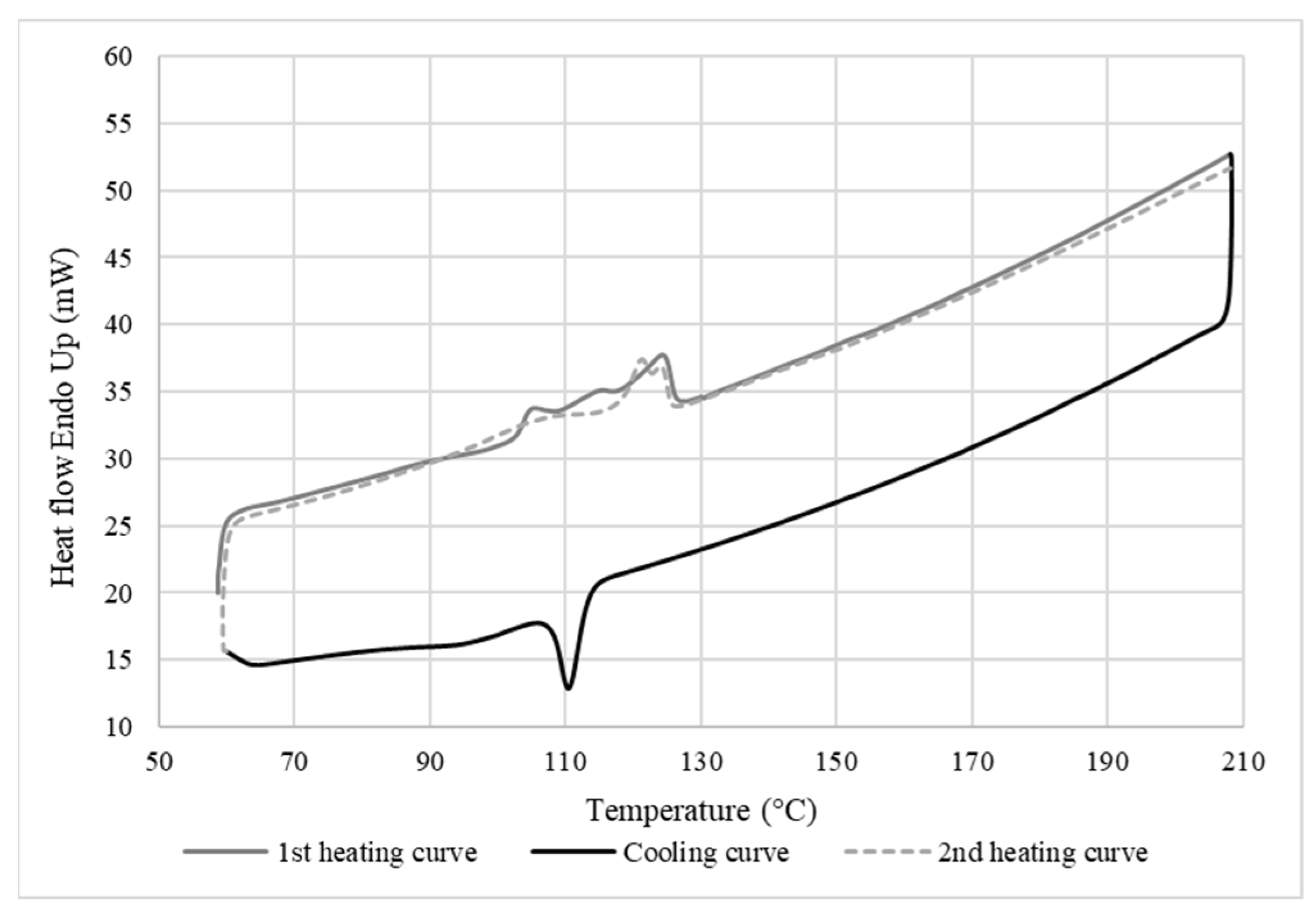
3.2. Differential Scanning Calorimetry
3.2.1. Thermal Properties of Corn Starch
3.2.2. Thermal Properties of LDPE
3.2.3. Thermal Properties of the Membrane
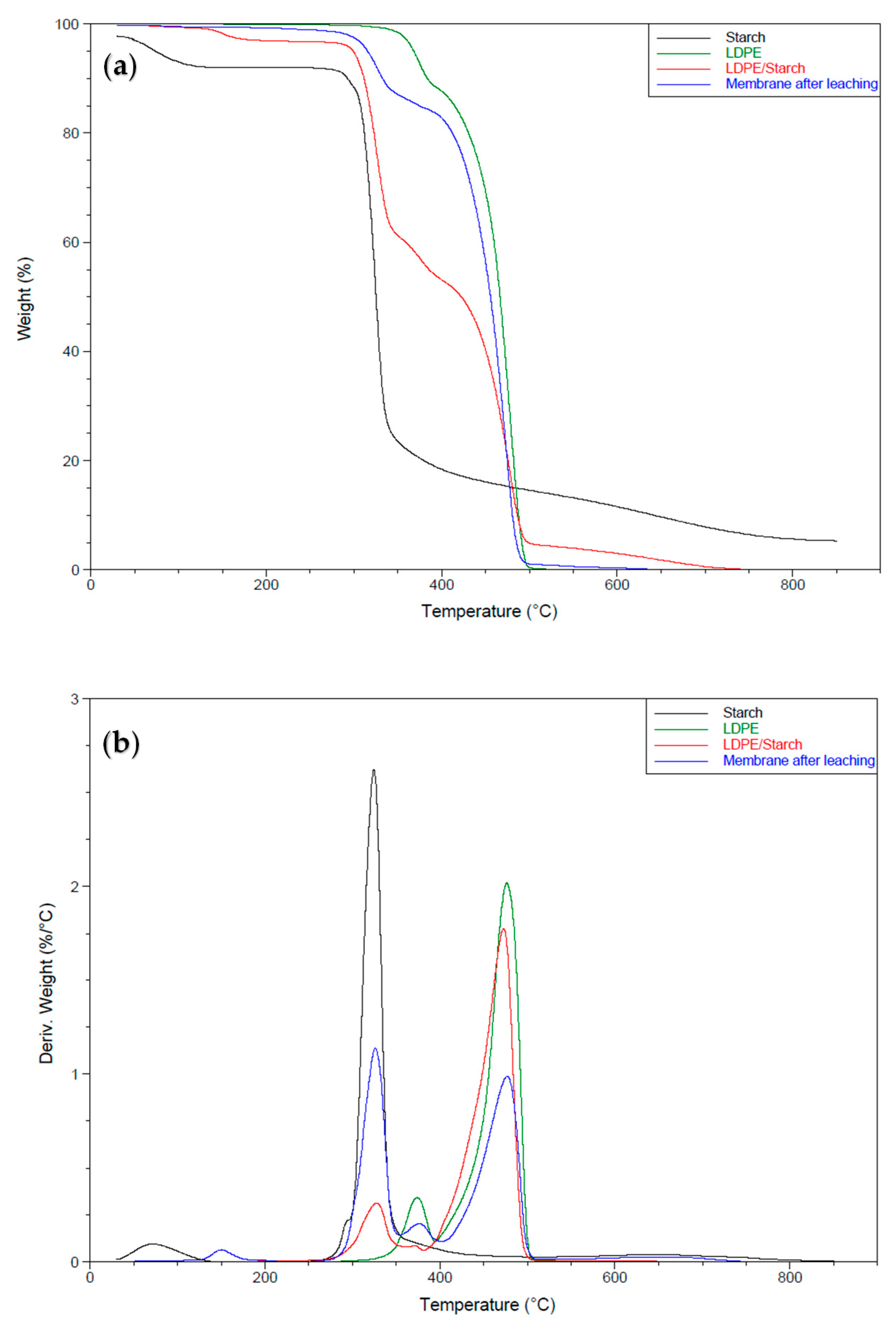
3.3. Thermogravimetric Analysis
Thermal Stability of the LDPE Membrane
3.4. Density
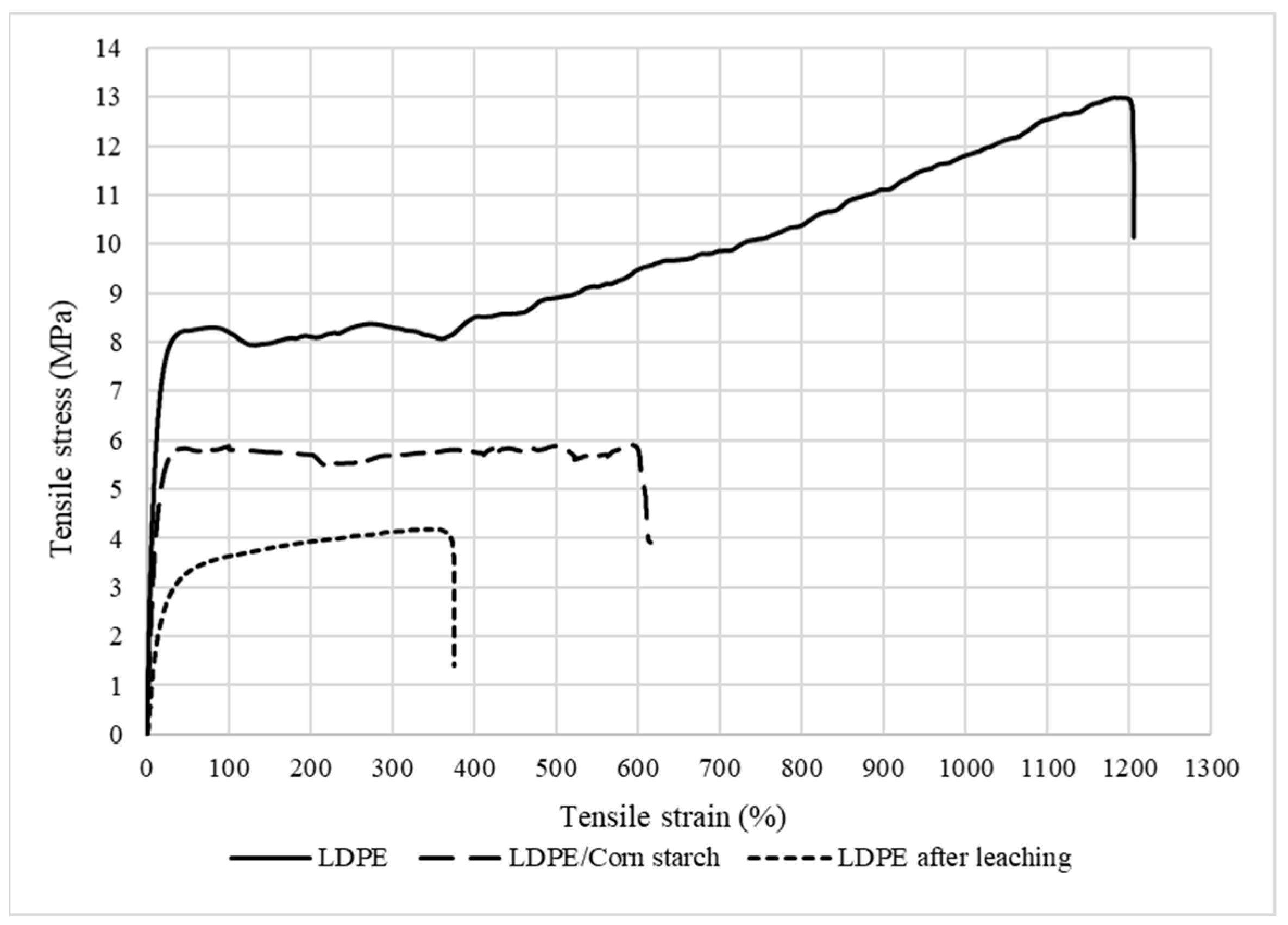
3.5. Tensile Properties
Mechanical Properties of the LDPE Membrane
3.6. Contact Angle
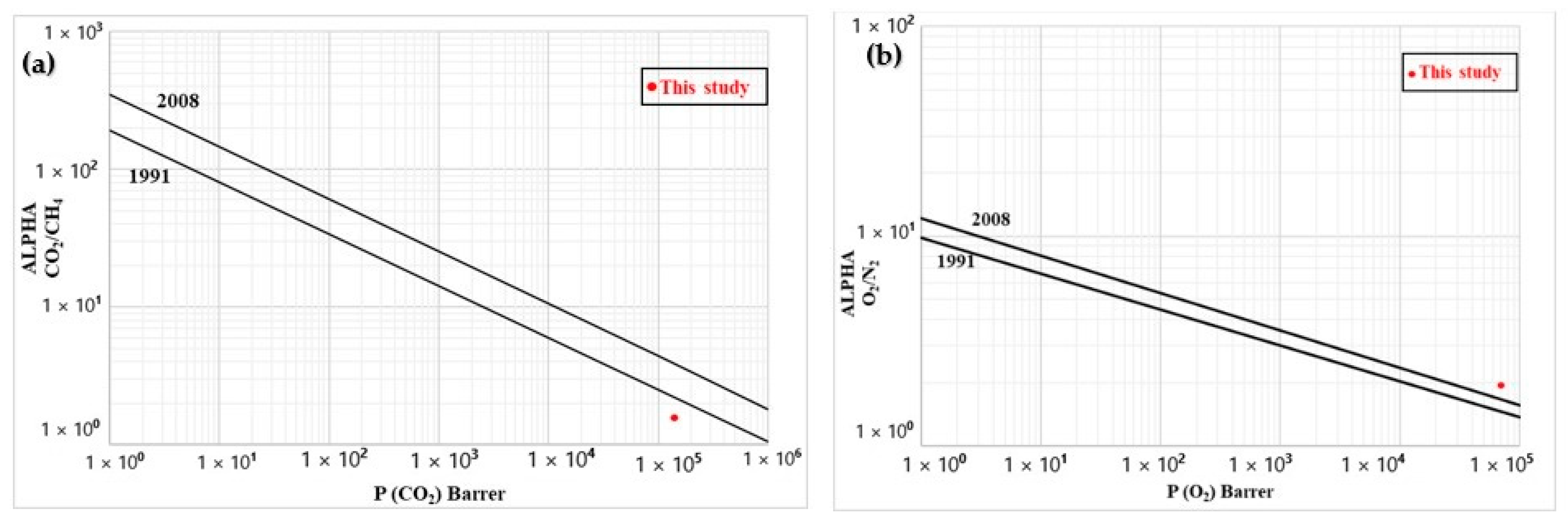
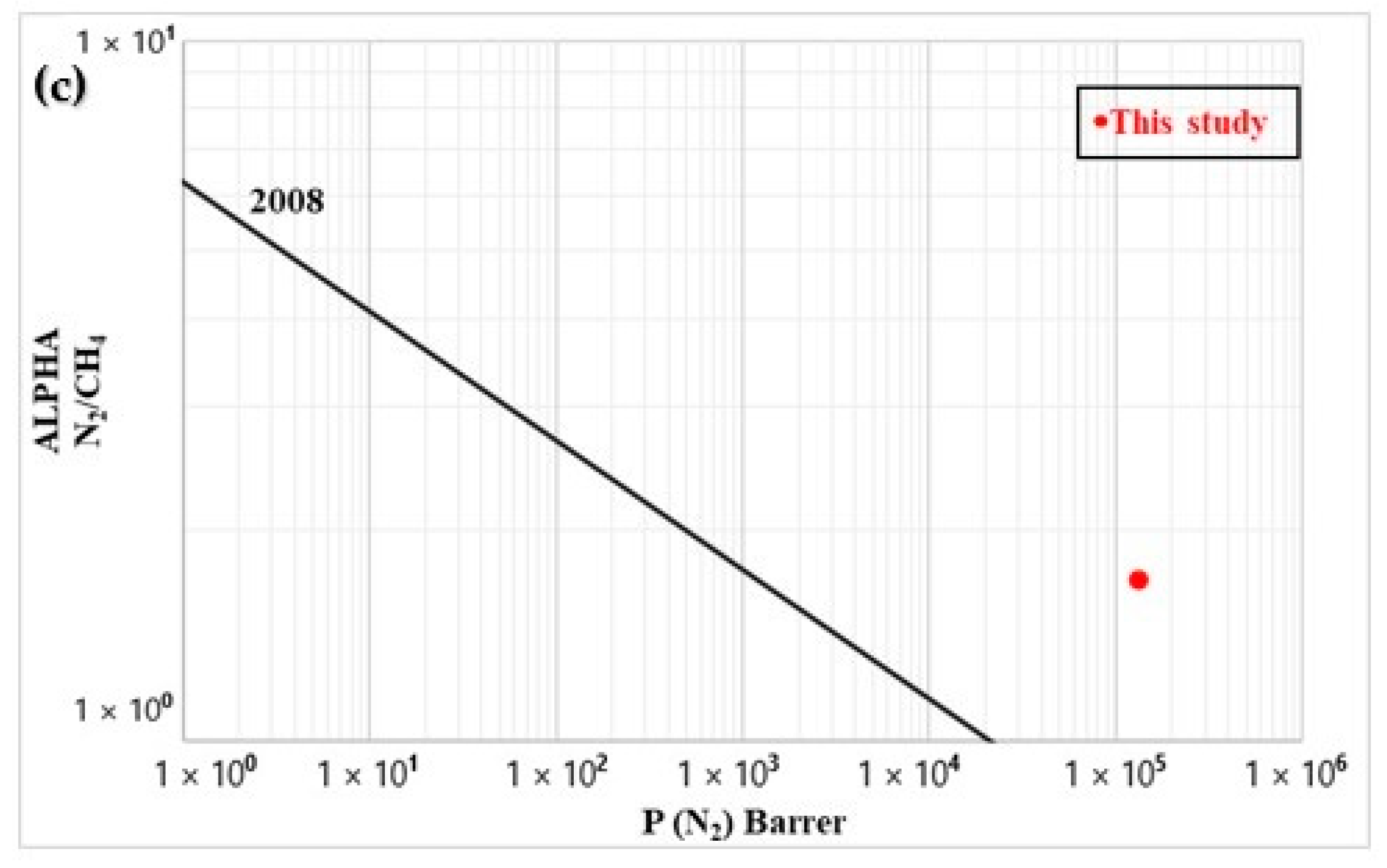
3.7. Permeability
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Correction Statement
References
- Baker, R.W.; Low, B.T. Gas separation membrane materials: A perspective. Macromolecules 2014, 47, 6999–7013. [Google Scholar] [CrossRef]
- Henis, J.M.; Tripodi, M.K. The developing technology of gas separating membranes. Science 1983, 220, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Bernardo, P.; Drioli, E. Membrane gas separation progresses for process intensification strategy in the petrochemical industry. Pet. Chem. 2010, 50, 271–282. [Google Scholar] [CrossRef]
- Akolekar, D.B.; Hind, A.R.; Bhargava, S.K. Synthesis of macro-, meso-, and microporous carbons from natural and synthetic sources, and their application as adsorbents for the removal of quaternary ammonium compounds from aqueous solution. J. Colloid Interface Sci. 1998, 199, 92–98. [Google Scholar] [CrossRef]
- Lin, V.S.Y.; Motesharei, K.; Dancil, K.P.S.; Sailor, M.J.; Ghadiri, M.R. A porous silicon-based optical interferometric biosensor. Science 1997, 278, 840–843. [Google Scholar] [CrossRef]
- Wu, J.; Xu, F.; Li, S.; Ma, P.; Zhang, X.; Liu, Q.; Wu, D. Porous polymers as multifunctional material platforms toward task-specific applications. Adv. Mater. 2019, 31, 1802922. [Google Scholar] [CrossRef]
- Sa-nguanruksa, J.; Rujiravanit, R.; Supaphol, P.; Tokura, S. Porous polyethylene membranes by template-leaching technique: Preparation and characterization. Polym. Test. 2004, 23, 91–99. [Google Scholar] [CrossRef]
- Liu, S.; Zhou, C.; Yu, W. Phase separation and structure control in ultra-high molecular weight polyethylene microporous membrane. J. Membr. Sci. 2011, 379, 268–278. [Google Scholar] [CrossRef]
- Mosadegh-Sedghi, S.; Rodrigue, D.; Brisson, J.; Iliuta, M.C. Highly hydrophobic microporous low-density polyethylene hollow fiber membranes by melt-extrusion coupled with salt-leaching technique. Polym. Adv. Technol. 2013, 24, 584–592. [Google Scholar] [CrossRef]
- Shahidi, K.; Rodrigue, D. Production of composite membranes by coupling coating and melt extrusion/salt leaching. Ind. Eng. Chem. Res. 2017, 56, 1306–1315. [Google Scholar] [CrossRef]
- Radakisnin, R.; Abdul Majid, M.S.; Mohd Jamir, M.R.; Mat Tahir, M.F.; Meng, C.E.; al Alshahrani, H. Physical, thermal, and mechanical properties of highly porous polylactic acid/cellulose nanofibre scaffolds prepared by salt leaching technique. Nanotechnol. Rev. 2021, 10, 1469–1483. [Google Scholar] [CrossRef]
- Haouari, S.; Rodrigue, D. Préparation et Caractérisation de Membranes Poreuses à Base de Polyéthylène. Masters’s Thesis, Université Laval, Québec, QC, Canada, 2022. [Google Scholar]
- Gosselin, R.; Rodrigue, D. Cell morphology analysis of high density polymer foams. Polym. Test. 2005, 24, 1027–1035. [Google Scholar] [CrossRef]
- Beg, M.D.H.; Kormin, S.; Bijarimi, M.; Zaman, H.U. Preparation and characterization of low-density Polyethylene/Thermoplastic starch composites. Adv. Polym. Technol. 2016, 35. [Google Scholar] [CrossRef]
- Chen, X.; Rodrigue, D.; Kaliaguine, S. Diamino-organosilicone APTMDS: A new cross-linking agent for polyimides membranes. Sep. Purif. Technol. 2012, 86, 221–233. [Google Scholar] [CrossRef]
- Nik, O.G.; Chen, X.Y.; Kaliaguine, S. Functionalized metal organic framework-polyimide mixed matrix membranes for CO2/CH4 separation. J. Membr. Sci. 2012, 413, 48–61. [Google Scholar] [CrossRef]
- Malumba Kamba, P.; Janas, S.; Deroanne, C.; Masimango, T.; Béra, F. Structure de l’amidon de maïs et principaux phénomènes impliqués dans sa modification thermique. Biotechnol. Agron. Société Environ. 2011, 15, 315–326. [Google Scholar]
- Biliaderis, C.G.; Page, C.M.; Maurice, T.J.; Juliano, B.O. Thermal characterization of rice starches: A polymeric approach to phase transitions of granular starch. J. Agric. Food Chem. 1986, 34, 6–14. [Google Scholar] [CrossRef]
- Pedroso, A.G.; Rosa, D.D.S. Mechanical, thermal and morphological characterization of recycled LDPE/corn starch blends. Carbohydr. Polym. 2005, 59, 1–9. [Google Scholar] [CrossRef]
- Li, D.; Zhou, L.; Wang, X.; He, L.; Yang, X. Effect of Crystallinity of Polyethylene with Different Densities on Breakdown Strength and Conductance Property. Materials 2019, 12, 1746. [Google Scholar] [CrossRef]
- Pérez, E.; Benavente, R.; Bello, A.; Perena, J.M.; Aguilar, C.; Martínez, M.C. Effect of crystallization conditions on the morphological structure of ethylene-1-butene copolymers obtained by different ziegler-natta catalyst systems. Polym. Eng. Sci. 1991, 31, 1189–1193. [Google Scholar] [CrossRef]
- Ek, S.; Root, A.; Peussa, M.; Niinistö, L. Determination of the hydroxyl group content in silica by thermogravimetry and a comparison with 1H MAS NMR results. Thermochim. Acta 2001, 379, 201–212. [Google Scholar] [CrossRef]
- Liu, X.; Yu, L.; Liu, H.; Chen, L.; Li, L. Thermal decomposition of corn starch with different amylose/amylopectin ratios in open and sealed systems. Cereal Chem. 2009, 86, 383–385. [Google Scholar] [CrossRef]
- Villalobos, K.; Rojas, H.; González-Paz, R.; Granados, D.B.; González-Masís, J.; Baudrit, J.V.; Corrales-Ureña, Y.R. Production of Starch Films Using Propolis Nanoparticles as Novel Bioplasticizer. J. Renew. Mater. 2017, 5, 189–198. [Google Scholar] [CrossRef]
- Lee, N.; Joo, J.; Lin, K.Y.A.; Lee, J. Waste-to-Fuels: Pyrolysis of Low-Density Polyethylene Waste in the Presence of H-ZSM-11. Polymers 2021, 13, 1198. [Google Scholar] [CrossRef] [PubMed]
- Mani, R.; Bhattacharya, M. Properties of injection moulded starch/synthetic polymer blends—III. Effect of amylopectin to amylose ratio in starch. Eur. Polym. J. 1998, 34, 1467–1475. [Google Scholar] [CrossRef]
- St-Pierre, N.; Favis, B.D.; Ramsay, B.A.; Ramsay, J.A.; Verhoogt, H. Processing and characterization of thermoplastic starch/polyethylene blends. Polymer 1997, 38, 647–655. [Google Scholar] [CrossRef]
- Żołek-Tryznowska, Z.; Kałuża, A. The Influence of Starch Origin on the Properties of Starch Films: Packaging Performance. Materials 2021, 14, 1146. [Google Scholar] [CrossRef]
- Suleman, M.S.; Lau, K.K.; Yeong, Y.F. Plasticization and swelling in polymeric membranes in CO2 removal from natural gas. Chem. Eng. Technol. 2016, 39, 1604–1616. [Google Scholar] [CrossRef]
- Psomiadou, E.; Arvanitoyannis, I.; Biliaderis, C.G.; Ogawa, H.; Kawasaki, N. Biodegradable films made from low density polyethylene (LDPE), wheat starch and soluble starch for food packaging applications. Part 2. Carbohydr. Polym. 1997, 33, 227–242. [Google Scholar] [CrossRef]
- Michaels, A.S.; Bixler, H.J. Flow of gases through polyethylene. J. Polym. Sci. 1961, 50, 413–439. [Google Scholar] [CrossRef]
- Rezakazemi, M.; Shahidi, K.; Mohammadi, T. Hydrogen separation and purification using crosslinkable PDMS/zeolite A nanoparticles mixed matrix membranes. Int. J. Hydrog. Energy 2012, 37, 14576–14589. [Google Scholar] [CrossRef]
- Shahidi, K.; Rodrigue, D. Gas transport and mechanical properties of PDMS-TFS/LDPE nanocomposite membranes. J. Polym. Res. 2018, 25, 1–9. [Google Scholar] [CrossRef]
- Robeson, L.M. Correlation of separation factor versus permeability for polymeric membranes. J. Membr. Sci. 1991, 62, 165–185. [Google Scholar] [CrossRef]
- Robeson, L.M. The upper bound revisited. J. Membr. Sci. 2008, 320, 390–400. [Google Scholar] [CrossRef]


| Matrix | Tf (°C) | Hf (J/g) | Tc (°C) | Hc (J/g) | Xc (%) |
|---|---|---|---|---|---|
| LDPE (10 °C/min) | 125 | 23.0 | 108 | 28.0 | 8.2 |
| LDPE (2 °C/min) | 125 | 5.3 | 112 | 16.4 | 5.7 |
| Curves | Td1 1 (°C) | Td2 1 (°C) | Td3 1 (°C) | Td4 1 (°C) | Wd1 2 (%) | Wd2 2 (%) | Wd3 2 (%) | Wd4 2 (%) | Residue (%) |
|---|---|---|---|---|---|---|---|---|---|
| CS | 40–135 | 253–650 | - | - | 5.7 | 82.2 | - | - | 4.4 |
| LDPE | 317–392 | 393–500 | - | - | 11.0 | 88.0 | - | - | 0.5 |
| LDPE/CS | 130–190 | 278–344 | 344–391 | 391–500 | 2.7 | 34.3 | 8.0 | 49.0 | 4.9 |
| Membrane after leaching | 178–303 | 303–389 | 389–496 | - | 2.4 | 13.0 | 83 | - | 1.3 |
| Materials | ρ (g/cm3) |
|---|---|
| CS | 1.504 |
| LDPE | 0.932 |
| LDPE/CS | 1.125 |
| Membrane after leaching | 0.915 |
| (MPa) | E (MPa) | |||||
|---|---|---|---|---|---|---|
| LDPE | 8.3 (±0.2) | 79 (±5) | 12.3 (±0.6) | 1204 (±11) | 3.03 (±0.27) | 84.9 (±2.4) |
| LDPE/CS | 5.8 (±1.0) | 46 (±8) | 4.6 (±2.0) | 628 (±9) | 1.28 (±0.49) | 63.5 (±10.0) |
| LDPE after leaching | 4.2 (±0.2) | 334 (±9) | 2.6 (±0.4) | 375 (±9) | 0.49 (±0.11) | 21.1 (±1.1) |
| Face | 1 | 2 | 3 | Standard Deviation |
|---|---|---|---|---|
| Top | 95.6 | 95.8 | 98.1 | 1.0 |
| Bottom | 93.8 | 94.9 | 97.6 | 1.4 |
| Membrane | Permeability (105 Barrer) | Ideal Selectivity (-) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| CO2 | CH4 | O2 | N2 | CO2/N2 | CO2/CH4 | CO2/O2 | CH4/O2 | CH4/N2 | O2/N2 | |
| LDPE after leaching | 1.37 | 2.20 | 0.71 | 1.33 | 1.0 | 1.6 | 1.9 | 3.1 | 1.7 | 1.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Haouari, S.; Rodrigue, D. A Low-Cost Porous Polymer Membrane for Gas Permeation. Materials 2022, 15, 3537. https://doi.org/10.3390/ma15103537
Haouari S, Rodrigue D. A Low-Cost Porous Polymer Membrane for Gas Permeation. Materials. 2022; 15(10):3537. https://doi.org/10.3390/ma15103537
Chicago/Turabian StyleHaouari, Selim, and Denis Rodrigue. 2022. "A Low-Cost Porous Polymer Membrane for Gas Permeation" Materials 15, no. 10: 3537. https://doi.org/10.3390/ma15103537
APA StyleHaouari, S., & Rodrigue, D. (2022). A Low-Cost Porous Polymer Membrane for Gas Permeation. Materials, 15(10), 3537. https://doi.org/10.3390/ma15103537







