Biomineralization of Plastic Waste to Improve the Strength of Plastic-Reinforced Cement Mortar
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.1.1. Plastic
2.1.2. Mineralization Solutions
2.2. Batch Testing for Microbial Growth and Plastic Biomineralization
2.2.1. MICP Biomineralization of PET
2.2.2. Types 3–7 Biomineralization Comparisons
2.2.3. Calcium Digests
2.2.4. X-ray Diffraction Spectroscopy
2.3. Preparation and Testing of Plastic-Reinforced Mortar Specimens
2.3.1. Mineralization of Plastic
2.3.2. Cement Mortar Production
2.3.3. Cement Mortar Compressive Testing
2.4. Microscopy
2.5. Characterization of Cement Hydration and Structure
2.6. Data Analysis
3. Results and Discussion
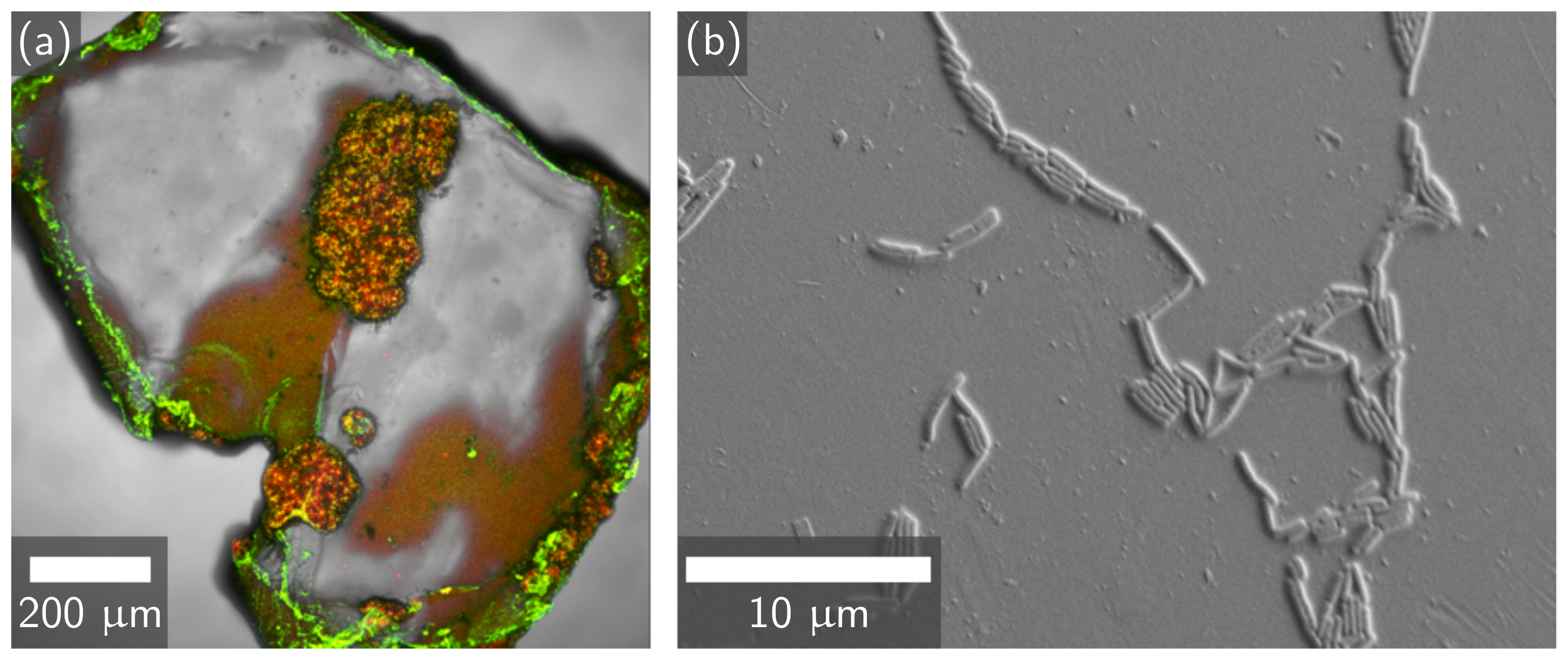
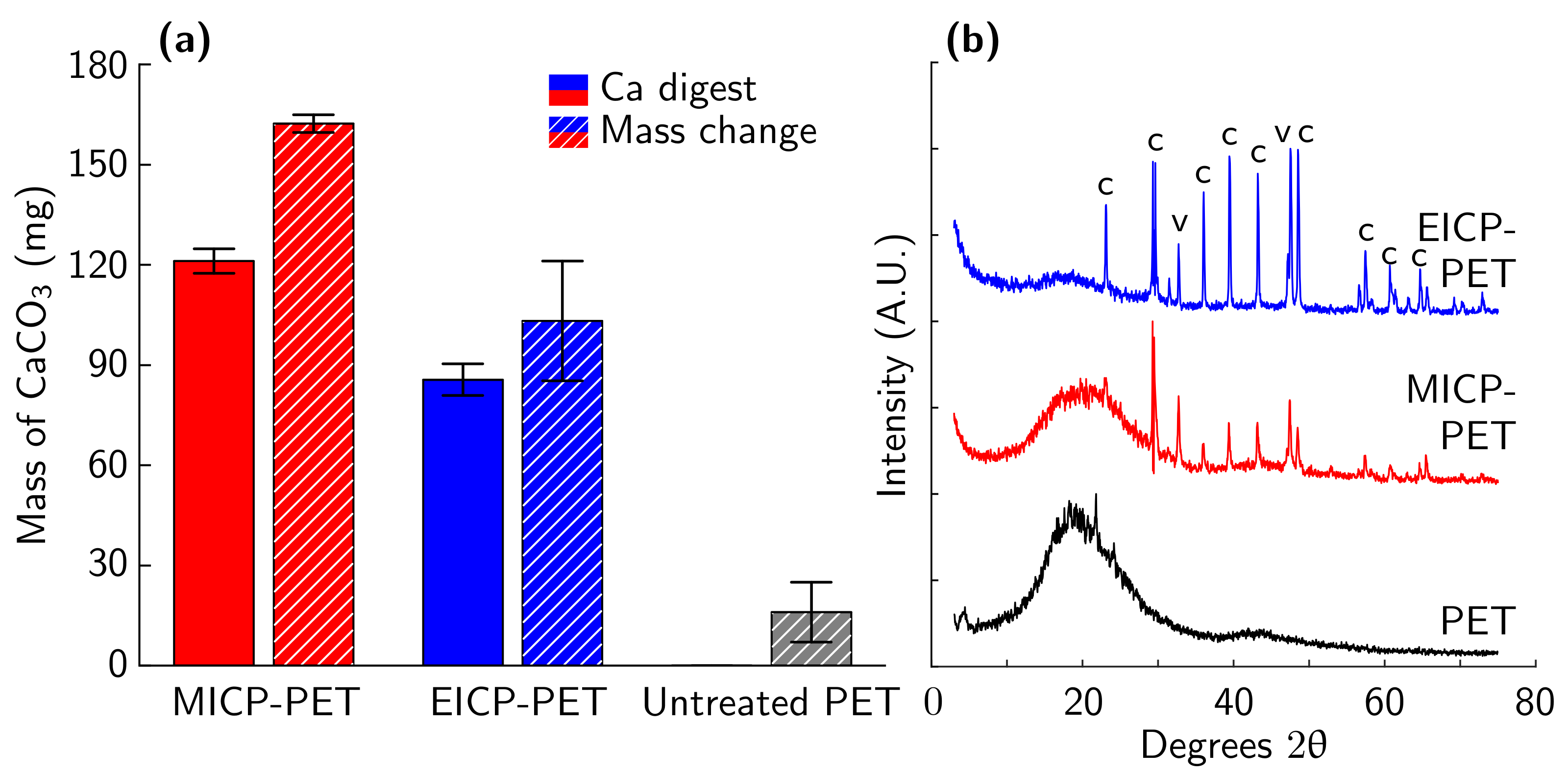
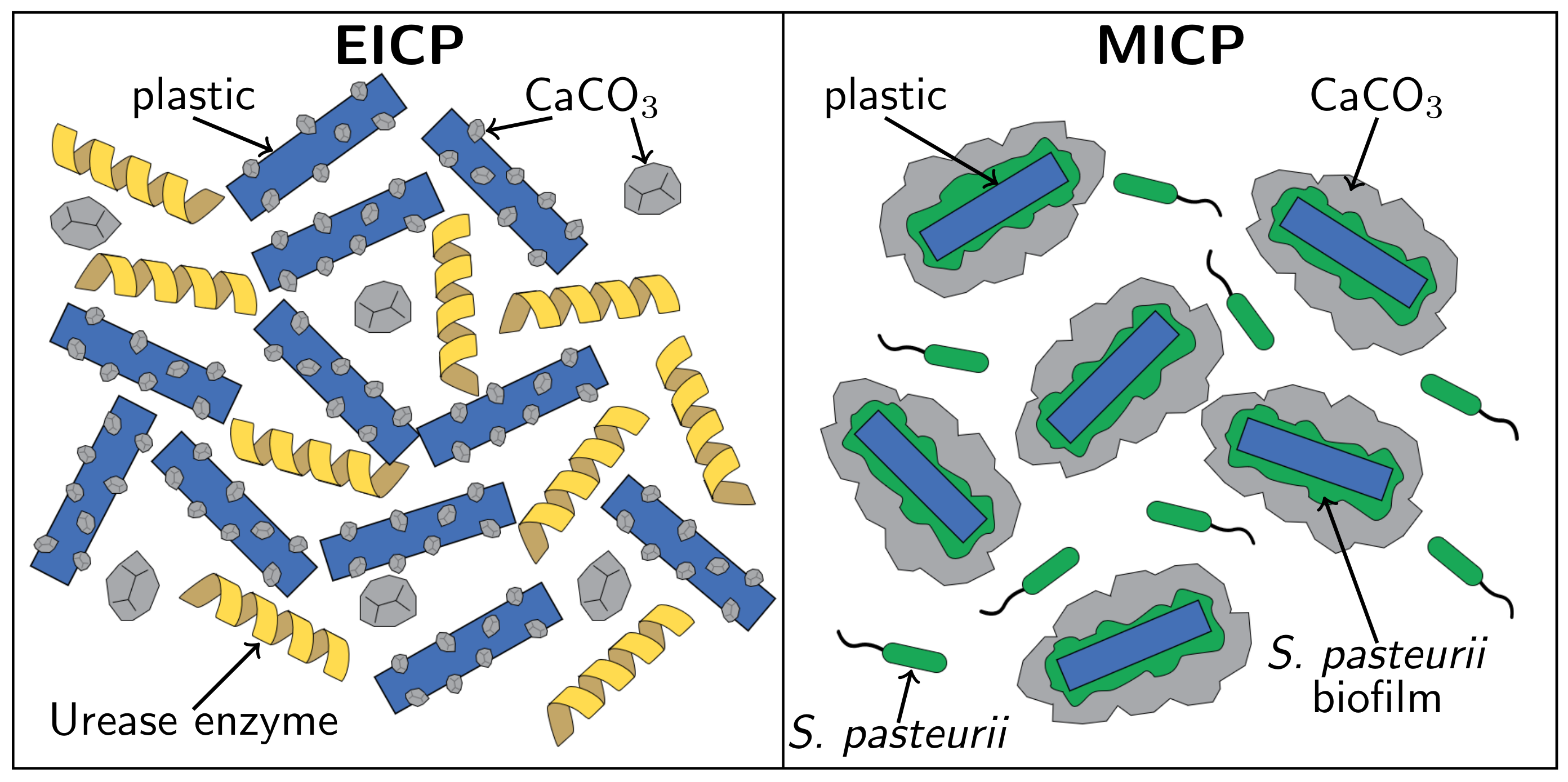
3.1. Comparison of EICP and MICP Treatment of PET
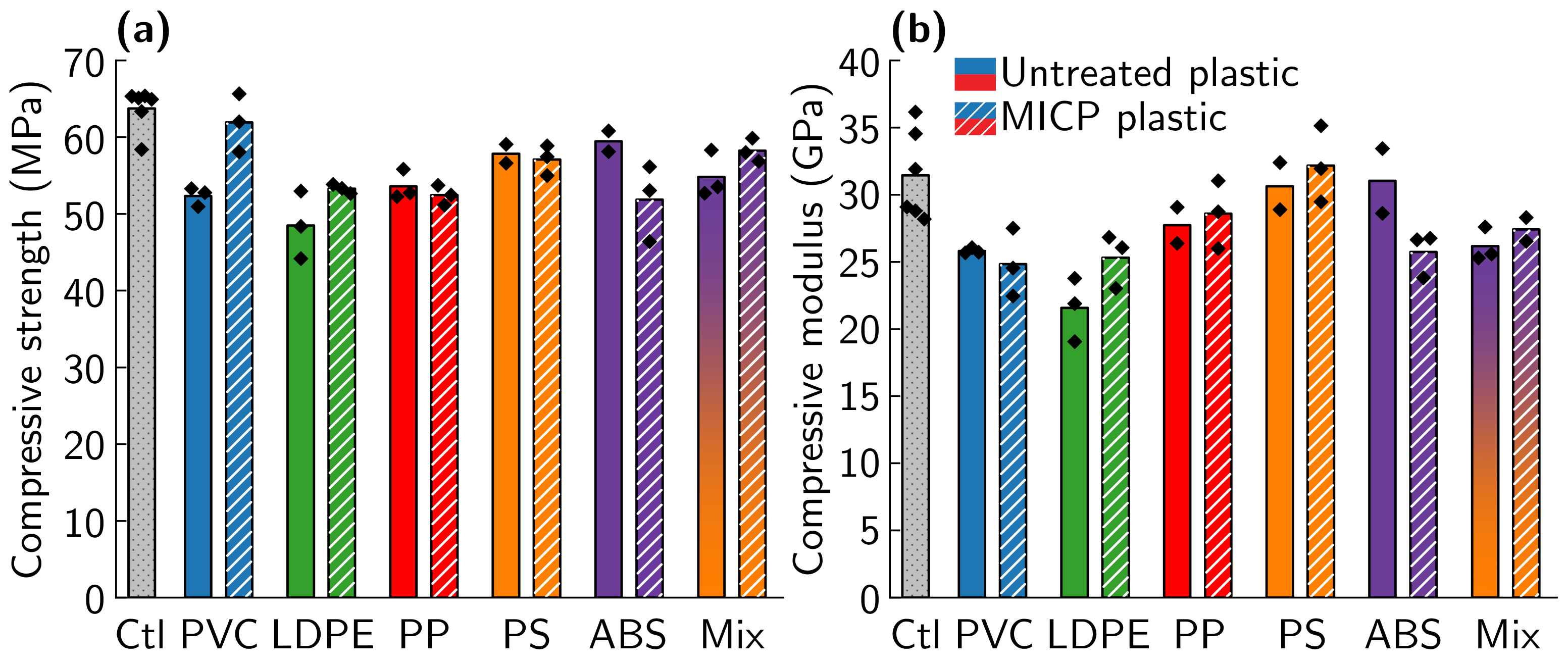
3.2. Comparison of MICP Treatment of Type 3–7 Plastics
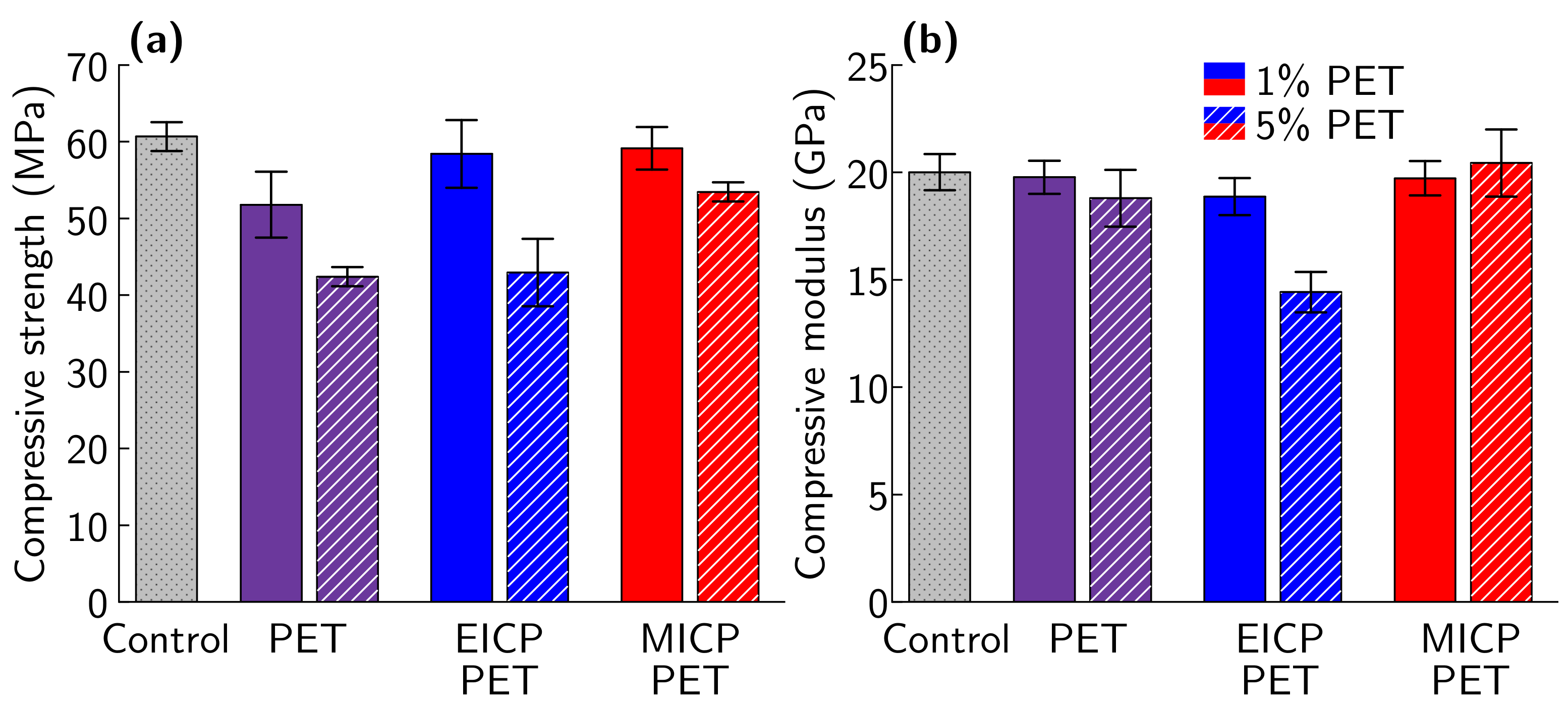
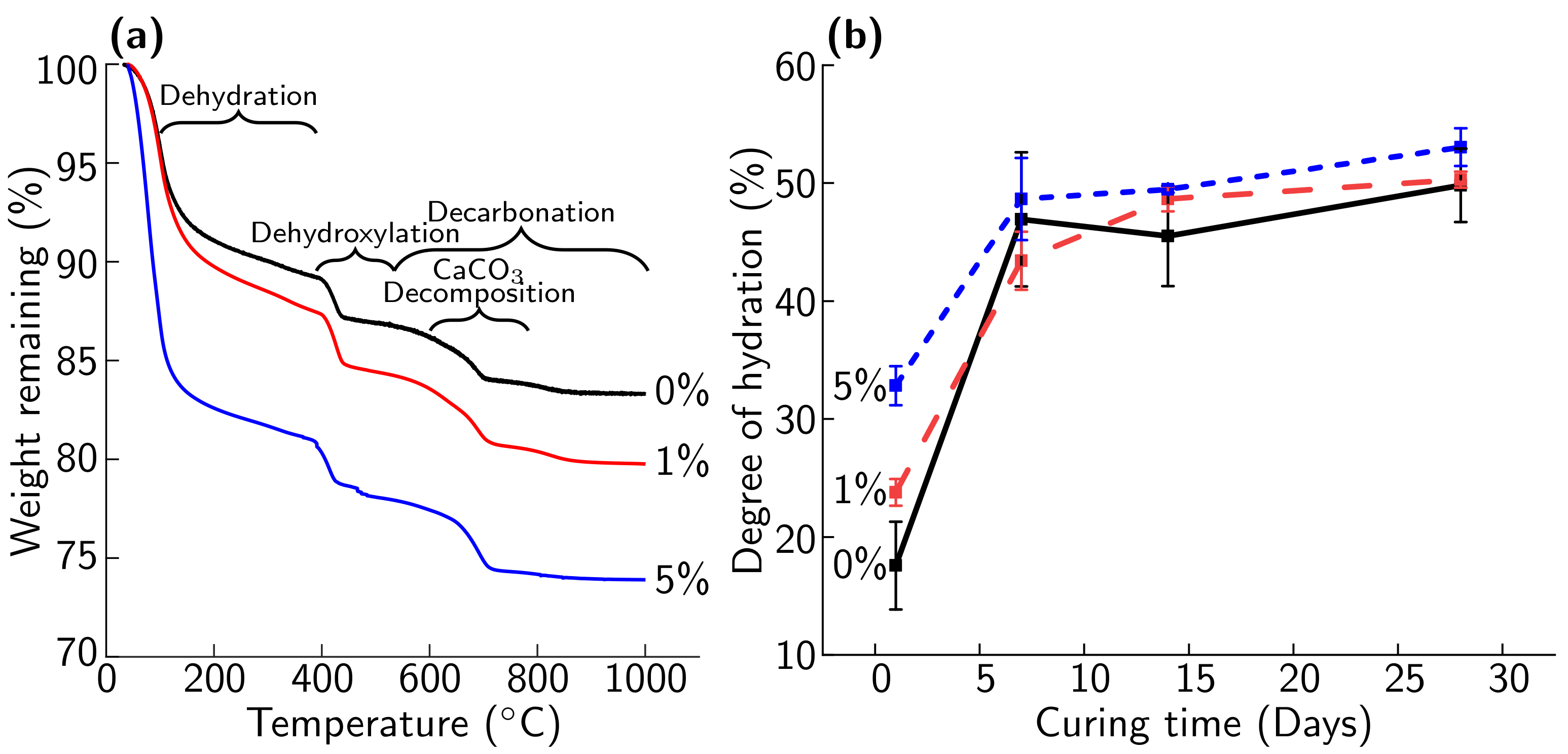
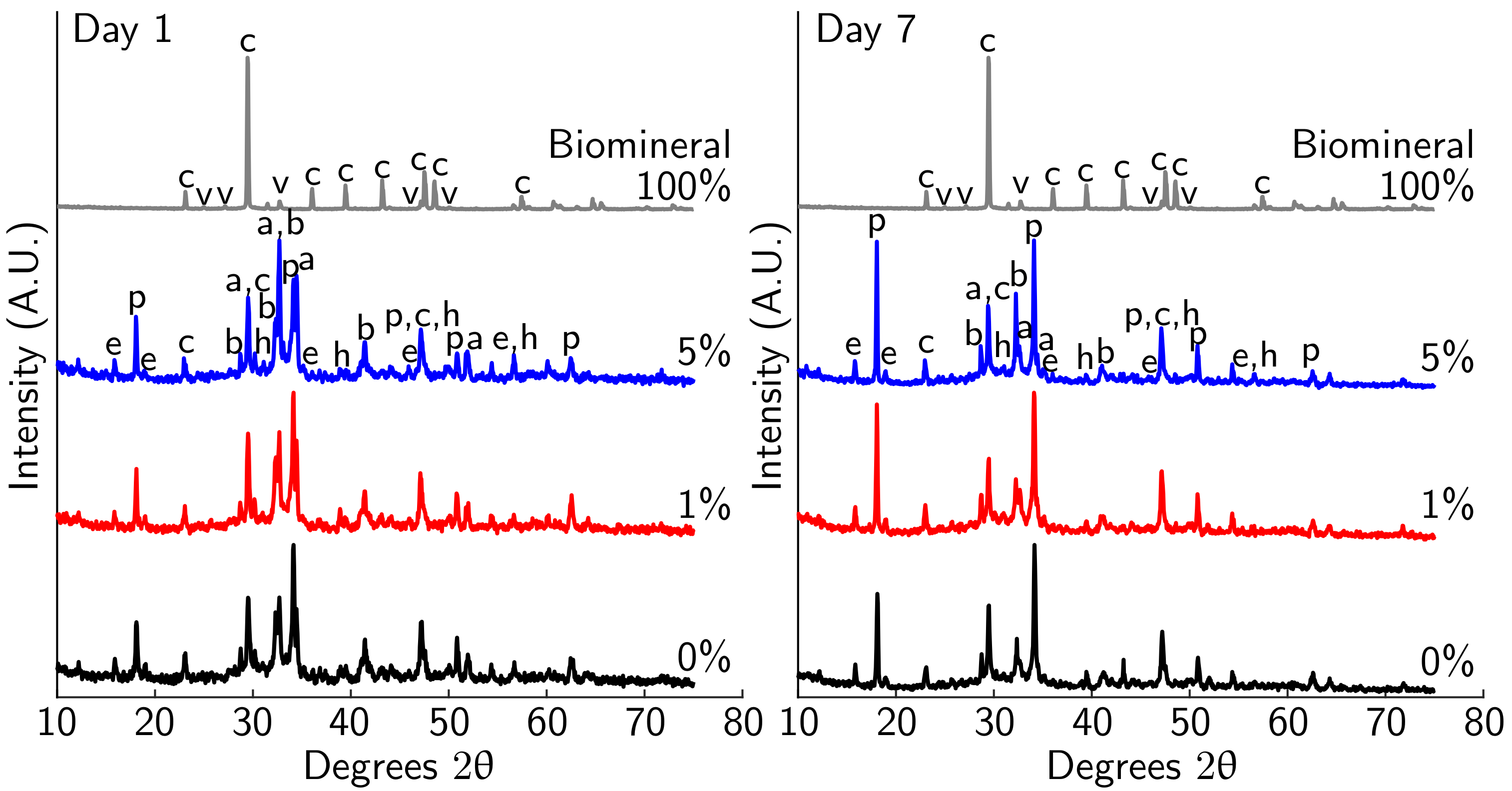
3.3. Effect of Biomineral on Cement Hydration
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Plastics Europe. Plastics—The Facts 2020; Plastics Europe: Brussels, Belgium, 2020. [Google Scholar]
- United Nations Environment Programme. Single-Use Plastics: A Roadmap for Sustainability; United Nations Environment Programme: Washington, DC, USA, 2018. [Google Scholar]
- Babafemi, A.; Šavija, B.; Paul, S.; Anggraini, V. Engineering Properties of Concrete with Waste Recycled Plastic: A Review. Sustainability 2018, 10, 3875. [Google Scholar] [CrossRef]
- Jani, Y.; Hogland, W. Waste glass in the production of cement and concrete—A review. J. Environ. Chem. Eng. 2014, 2, 1767–1775. [Google Scholar] [CrossRef]
- Kishore, K.; Gupta, N. Application of domestic & industrial waste materials in concrete: A review. Mater. Today Proc. 2019, 26, 2926–2931. [Google Scholar] [CrossRef]
- Miller, S.A. Supplementary cementitious materials to mitigate greenhouse gas emissions from concrete: Can there be too much of a good thing? J. Clean. Prod. 2018, 178, 587–598. [Google Scholar] [CrossRef]
- Yina, S.; Tuladhar, R.; Sheehan, M.; Combe, M.; Collister, T. A life cycle assessment of recycled polypropylene fibre in concrete footpaths. J. Clean. Prod. 2016, 112, 2231–2242. [Google Scholar] [CrossRef]
- Kim, S.B.; Yi, N.H.; Kim, H.Y.; Kim, J.H.J.; Song, Y.C. Material and structural performance evaluation of recycled PET fiber reinforced concrete. Cem. Concr. Compos. 2010, 32, 232–240. [Google Scholar] [CrossRef]
- Pešić, N.; Živanović, S.; Garcia, R.; Papastergiou, P. Mechanical properties of concrete reinforced with recycled HDPE plastic fibres. Constr. Build. Mater. 2016, 115, 362–370. [Google Scholar] [CrossRef]
- Sharma, R.; Bansal, P.P. Use of different forms of waste plastic in concrete—A review. J. Clean. Prod. 2016, 112, 473–482. [Google Scholar] [CrossRef]
- Yin, S. Development of Recycled Polypropylene Plastic Fibres to Reinforce Concrete. Ph.D. Thesis, James Cook University, Douglas, Australia, 2017. [Google Scholar] [CrossRef]
- Ochi, T.; Okubo, S.; Fukui, K. Development of recycled PET fiber and its application as concrete-reinforcing fiber. Cem. Concr. Compos. 2007, 29, 448–455. [Google Scholar] [CrossRef]
- Saikia, N.; De Brito, J. Use of plastic waste as aggregate in cement mortar and concrete preparation: A review. Constr. Build. Mater. 2012, 34, 385–401. [Google Scholar] [CrossRef]
- Al-Tulaian, B.S.; Al-Shannag, M.J.; Al-Hozaimy, A.R. Recycled plastic waste fibers for reinforcing Portland cement mortar. Constr. Build. Mater. 2016, 127, 102–110. [Google Scholar] [CrossRef]
- Jacob-Vaillancourt, C.; Sorelli, L. Characterization of concrete composites with recycled plastic aggregates from postconsumer material streams. Constr. Build. Mater. 2018, 182, 561–572. [Google Scholar] [CrossRef]
- Naik, T.R.; Singh, S.S.; Huber, C.O.; Brodersen, B.S. Use of post-consumer waste plastics in cement-based composites. Cem. Concr. Res. 1996, 26, 1489–1492. [Google Scholar] [CrossRef]
- Feng, J.; Yang, F.; Qian, S. Improving the bond between polypropylene fiber and cement matrix by nano calcium carbonate modification. Constr. Build. Mater. 2021, 269, 121249. [Google Scholar] [CrossRef]
- Hao, Y.; Cheng, L.; Hao, H.; Shahin, M.A. Enhancing fiber/matrix bonding in polypropylene fiber reinforced cementitious composites by microbially induced calcite precipitation pre-treatment. Cem. Concr. Compos. 2018, 88, 1–7. [Google Scholar] [CrossRef]
- Phillips, A.J.; Gerlach, R.; Lauchnor, E.; Mitchell, A.C.; Cunningham, A.B.; Spangler, L. Engineered applications of ureolytic biomineralization: A review. Biofouling 2013, 29, 715–733. [Google Scholar] [CrossRef]
- Bachmeier, K.L.; Williams, A.E.; Warmington, J.R.; Bang, S.S. Urease activity in microbiologically-induced calcite precipitation. J. Biotechnol. 2002, 93, 171–181. [Google Scholar] [CrossRef]
- Stocks-Fischer, S.; Galinat, J.K.; Bang, S.S. Microbiological precipitation of CaCO3. Soil Biol. Biochem. 1999, 31, 1563–1571. [Google Scholar] [CrossRef]
- Hommel, J.; Akyel, A.; Frieling, Z.; Phillips, A.J.; Gerlach, R.; Cunningham, A.B.; Class, H. A Numerical Model for Enzymatically Induced Calcium Carbonate Precipitation. Appl. Sci. 2020, 10, 4538. [Google Scholar] [CrossRef]
- De Muynck, W.; De Belie, N.; Verstraete, W. Microbial carbonate precipitation in construction materials: A review. Ecol. Eng. 2010, 36, 118–136. [Google Scholar] [CrossRef]
- Phillips, A.J.; Cunningham, A.B.; Gerlach, R.; Hiebert, R.; Hwang, C.; Lomans, B.P.; Westrich, J.; Mantilla, C.; Kirksey, J.; Esposito, R.; et al. Fracture Sealing with Microbially-Induced Calcium Carbonate Precipitation: A Field Study. Environ. Sci. Technol. 2016, 50, 4111–4117. [Google Scholar] [CrossRef]
- Wang, J.Y.; Soens, H.; Verstraete, W.; De Belie, N. Self-healing concrete by use of microencapsulated bacterial spores. Cem. Concr. Res. 2014, 56, 139–152. [Google Scholar] [CrossRef]
- Lee, Y.S.; Park, W. Current challenges and future directions for bacterial self-healing concrete. Appl. Microbiol. Biotechnol. 2018, 102, 3059–3070. [Google Scholar] [CrossRef]
- Dakhane, A.; Das, S.; Hansen, H.; O’Donnell, S.; Hanoon, F.; Rushton, A.; Perla, C.; Neithalath, N. Crack Healing in Cementitious Mortars Using Enzyme-Induced Carbonate Precipitation: Quantification Based on Fracture Response. J. Mater. Civ. Eng. 2018, 30, 04018035. [Google Scholar] [CrossRef]
- ASTM International. C1609/C1609M-19a Standard Test Method for Flexural Performance of Fiber-Reinforced Concrete (Using Beam with Third-Point Loading); Standard; ASTM International: West Conshohocken, PA, USA, 2019. [Google Scholar] [CrossRef]
- Phillips, A.J.; Lauchnor, E.; Eldring, J.; Esposito, R.; Mitchell, A.C.; Gerlach, R.; Cunningham, A.B.; Spangler, L.H. Potential CO2 leakage reduction through biofilm-induced calcium carbonate precipitation. Environ. Sci. Technol. 2013, 47, 142–149. [Google Scholar] [CrossRef]
- Thrane, L.W.; Daily, R.L.; Thane, A.; Kirkland, C.M.; Mccarney, E.R.; Dykstra, R.; Codd, S.L.; Phillips, A.J. Detecting Microbially Induced Calcium Carbonate Precipitation in Porous Systems Using Low-Field Nuclear Magnetic Resonance Relaxometry. J. Geotech. Geoenviron. Eng. 2020, 146, 04020012. [Google Scholar] [CrossRef]
- Jung, D.; Biggs, H.; Erikson, J.; Ledyard, P.U. New Colorimetric reaction for end-point, continuous-flow, and kinetic measurement of urea. Clin. Chem. 1975, 21, 1136–1140. [Google Scholar] [CrossRef]
- Phillips, A.J. Biofilm-Induced Calcium Carbonate Precipitation: Application in the Subsurface. Ph.D. Thesis, Montana State University, Bozeman, MT, USA, 2013. [Google Scholar]
- Frieling, Z. Urease Immobilization for Advancing Enzyme-Induced Calcium Carbonate Precipitation Applications. Master’s Thesis, Montana State University, Bozeman, MT, USA, 2019. [Google Scholar]
- ASTM International. C109/C109M-20b Standard Test Method for Compressive Strength of Hydraulic Cement Mortars (Using 2-in. or [50 mm] Cube Specimens); Standard; ASTM International: West Conshohocken, PA, USA, 2020. [Google Scholar] [CrossRef]
- ASTM International. C305-20 Standard Practice for Mechanical Mixing of Hydraulic Cement Pastes and Mortars of Plastic Consistency; Standard; ASTM International: West Conshohocken, PA, USA, 2020. [Google Scholar] [CrossRef]
- ASTM International. ASTM C192: Standard Practice for Making and Curing Concrete Test Specimens in the Laboratory; Standard; ASTM International: West Conshohocken, PA, USA, 2019. [Google Scholar] [CrossRef]
- Monteagudo, S.M.; Moragues, A.; Gálvez, J.C.; Casati, M.J.; Reyes, E. The degree of hydration assessment of blended cement pastes by differential thermal and thermogravimetric analysis. Morphological evolution of the solid phases. Thermochim. Acta 2014, 592, 37–51. [Google Scholar] [CrossRef]
- Deboucha, W.; Leklou, N.; Khelidj, A.; Oudjit, M.N. Hydration development of mineral additives blended cement using thermogravimetric analysis (TGA): Methodology of calculating the degree of hydration. Constr. Build. Mater. 2017, 146, 687–701. [Google Scholar] [CrossRef]
- Monteiro, P.J.; Clodic, L.; Battocchio, F.; Kanitpanyacharoen, W.; Chae, S.R.; Ha, J.; Wenk, H.R. Incorporating carbon sequestration materials in civil infrastructure: A micro and nano-structural analysis. Cem. Concr. Compos. 2013, 40, 14–20. [Google Scholar] [CrossRef]
- Stutzman, P.E.; Feng, P.; Bullard, J.W. Phase Analysis of Portland Cements by Combined Quantitative X-ray Powder Diffraction and Scanning Electron Microscopy. NIST J. Res. 2016, 121, 47–107. [Google Scholar] [CrossRef]
- Ipavec, A.; Gabrovšek, R.; Vuk, T.; Kaučič, V.; Maček, J.; Meden, A. Carboaluminate Phases Formation During the Hydration of Calcite-Containing Portland Cement. J. Am. Ceram. Soc. 2011, 94, 1238–1242. [Google Scholar] [CrossRef]
- Jeong, Y.; Hargis, C.W.; Chun, S.; Moon, J. Effect of Calcium Carbonate Fineness on Calcium Sulfoaluminate-Belite Cement. Materials 2017, 10, 900. [Google Scholar] [CrossRef]
- Runčevski, T.; Dinnebier, R.E.; Magdysyuk, O.V.; Pöllmann, H. Crystal structures of calcium hemicarboaluminate and carbonated calcium hemicarboaluminate from synchrotron powder diffraction data. Acta Crystallogr. Sect. B 2012, 68, 493–500. [Google Scholar] [CrossRef]
- Sondi, I.; Salopek-Sondi, B. Influence of the primary structure of enzymes on the formation of CaCO3 polymorphs: A comparison of plant (Canavaliaensi formis) and bacterial (Bacilluspasteurii) ureases. Langmuir 2005, 21, 8876–8882. [Google Scholar] [CrossRef]
- Almajed, A.; Tirkolaei, H.K.; Kavazanjian, E.; Hamdan, N. Enzyme Induced Biocementated Sand with High Strength at Low Carbonate Content. Sci. Rep. 2019, 9, 1135. [Google Scholar] [CrossRef]
- Heveran, C.M.; Liang, L.; Nagarajan, A.; Hubler, M.H.; Gill, R.; Cameron, J.C.; Cook, S.M.; Srubar, W.V. Engineered Ureolytic Microorganisms Can Tailor the Morphology and Nanomechanical Properties of Microbial-Precipitated Calcium Carbonate. Sci. Rep. 2019, 9, 14721. [Google Scholar] [CrossRef]
- Dhami, N.K.; Mukherjee, A.; Reddy, M.S. Micrographical, minerological and nano-mechanical characterisation of microbial carbonates from urease and carbonic anhydrase producing bacteria. Ecol. Eng. 2016, 94, 443–454. [Google Scholar] [CrossRef]
- Achal, V.; Pan, X.; Özyurt, N. Improved strength and durability of fly ash-amended concrete by microbial calcite precipitation. Ecol. Eng. 2011, 37, 554–559. [Google Scholar] [CrossRef]
- International Code Council. The Strength of Concrete. In 2015 Concrete Manual; International Code Council: Washington, DC, USA, 2015; Chapter 3; p. 25. [Google Scholar]
- Matschei, T.; Lothenbach, B.; Glasser, F.P. The role of calcium carbonate in cement hydration. Cem. Concr. Res. 2007, 37, 551–558. [Google Scholar] [CrossRef]






| Attachment Phase | Mineralization Phase | |
|---|---|---|
| Test condition | S. pasteurii, plastic, CMM− | Plastic (with biofilm), CMM+ |
| Positive control | S. pasteurii, CMM− | None |
| Negative control | Plastic, CMM− | Plastic (no biofilm), CMM+ |
| Plastic Type (s) | Mineralization Method | wt.% Replacement |
|---|---|---|
| PET | EICP | 1%, 5% |
| PET | MICP | 1%, 5% |
| PET | None | 1%, 5% |
| Types 3–7 | MICP | 5% |
| Types 3–7 | None | 5% |
| None (control) | None | 0% |
| Change in Compressive Strength Relative to Control | |||
|---|---|---|---|
| Plastic Replacement | Untreated PET | EICP-PET | MICP-PET |
| 1% | −14.6% | −3.74% | −2.54% |
| 5% | −30.1% | −29.2% | −11.9% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kane, S.; Thane, A.; Espinal, M.; Lunday, K.; Armağan, H.; Phillips, A.; Heveran, C.; Ryan, C. Biomineralization of Plastic Waste to Improve the Strength of Plastic-Reinforced Cement Mortar. Materials 2021, 14, 1949. https://doi.org/10.3390/ma14081949
Kane S, Thane A, Espinal M, Lunday K, Armağan H, Phillips A, Heveran C, Ryan C. Biomineralization of Plastic Waste to Improve the Strength of Plastic-Reinforced Cement Mortar. Materials. 2021; 14(8):1949. https://doi.org/10.3390/ma14081949
Chicago/Turabian StyleKane, Seth, Abby Thane, Michael Espinal, Kendra Lunday, Hakan Armağan, Adrienne Phillips, Chelsea Heveran, and Cecily Ryan. 2021. "Biomineralization of Plastic Waste to Improve the Strength of Plastic-Reinforced Cement Mortar" Materials 14, no. 8: 1949. https://doi.org/10.3390/ma14081949
APA StyleKane, S., Thane, A., Espinal, M., Lunday, K., Armağan, H., Phillips, A., Heveran, C., & Ryan, C. (2021). Biomineralization of Plastic Waste to Improve the Strength of Plastic-Reinforced Cement Mortar. Materials, 14(8), 1949. https://doi.org/10.3390/ma14081949






