Physicochemical and Biological Properties of Mg-Doped Calcium Silicate Endodontic Cement
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of Sol-Gel Calcium Silicate and Mg-Substituted Calcium Silicate
2.2. Characterization of CSC and Mg-CSC
2.3. Setting Time Measurement
2.4. Compressive Strength Test
2.5. Microhardness Test
2.6. Simulated Body Fluid (SBF) Immersion Tests
2.7. In Vitro Cytotoxicity Test
2.8. Alkaline Phosphatase (ALP) Activity Assay
2.9. Alizarin Red S Staining and Quantitative Analysis (ARS)
2.10. Statistical Analysis
3. Results
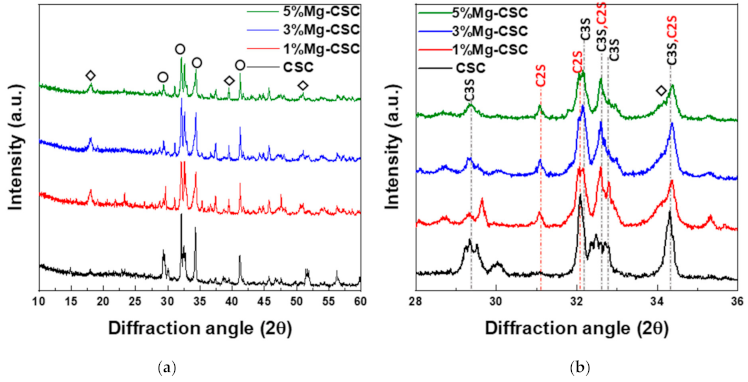
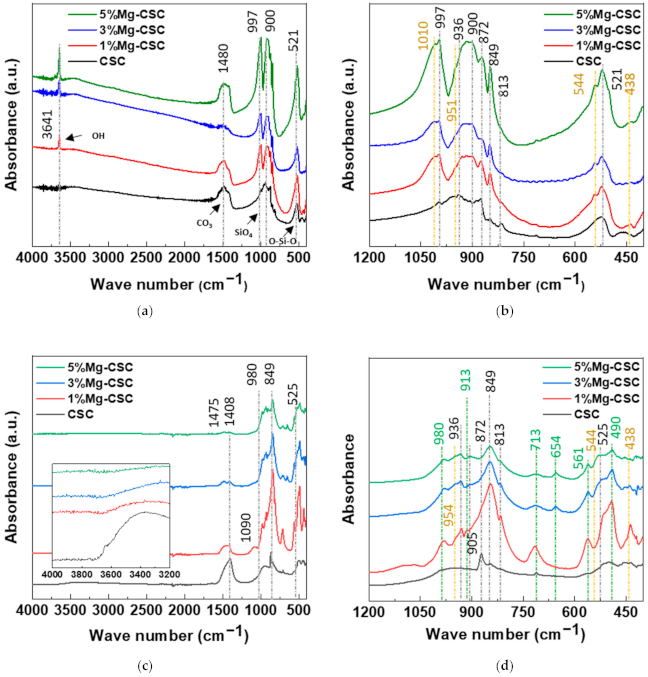
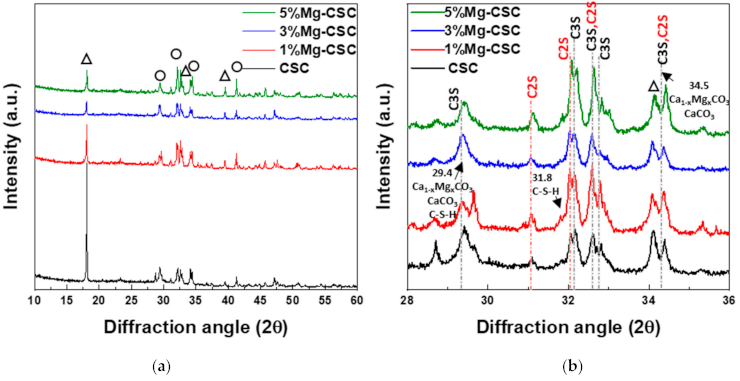
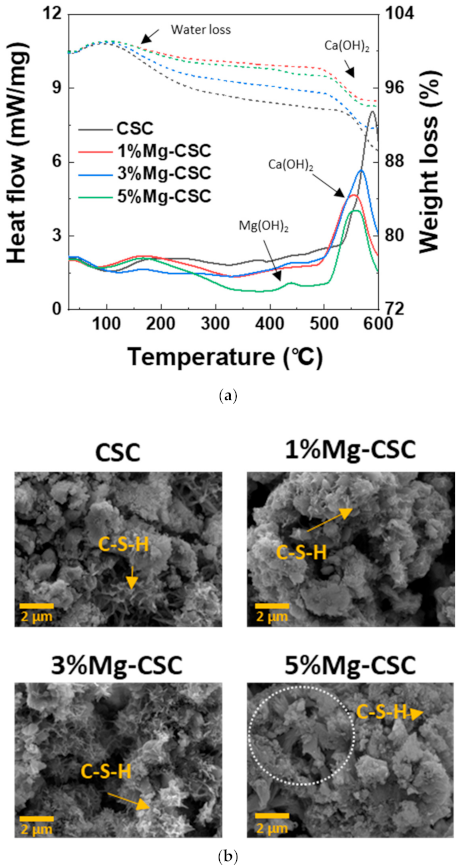
3.1. Characterization of Mg-CSC and Hydrated Mg-CSC (Mg = 0 to 5 mol%)
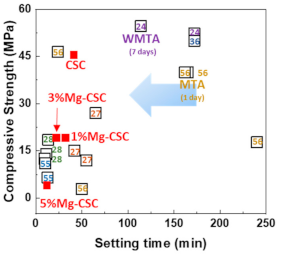
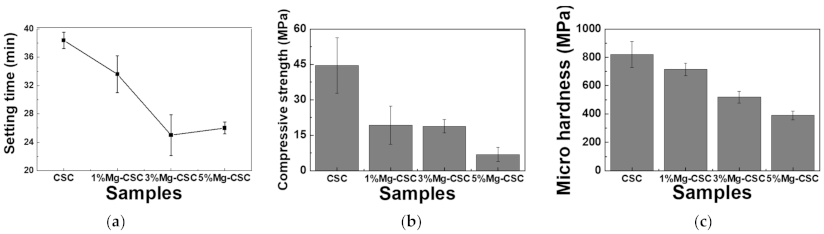
3.2. Setting Time and Mechanical Properties
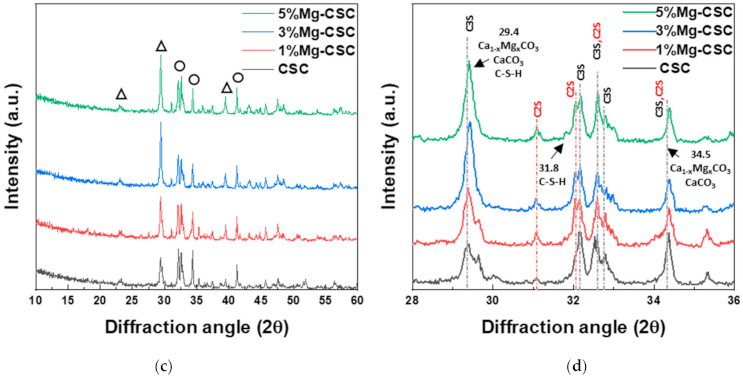
3.3. Characterization of Mg-CSC (Mg = 0 to 5 mol%) after SBF Immersion
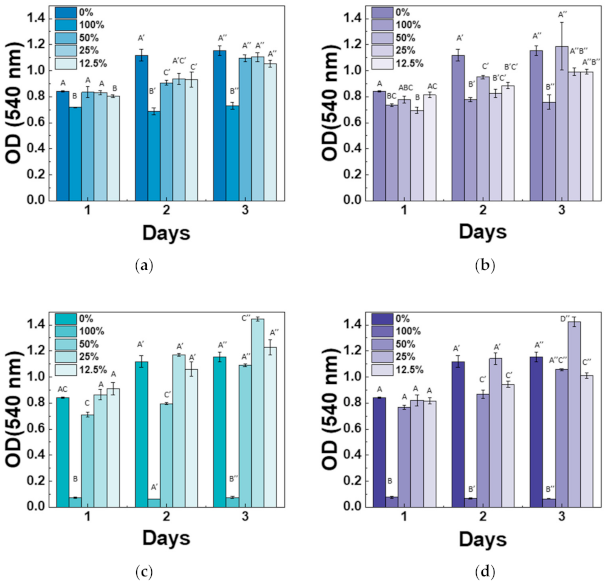
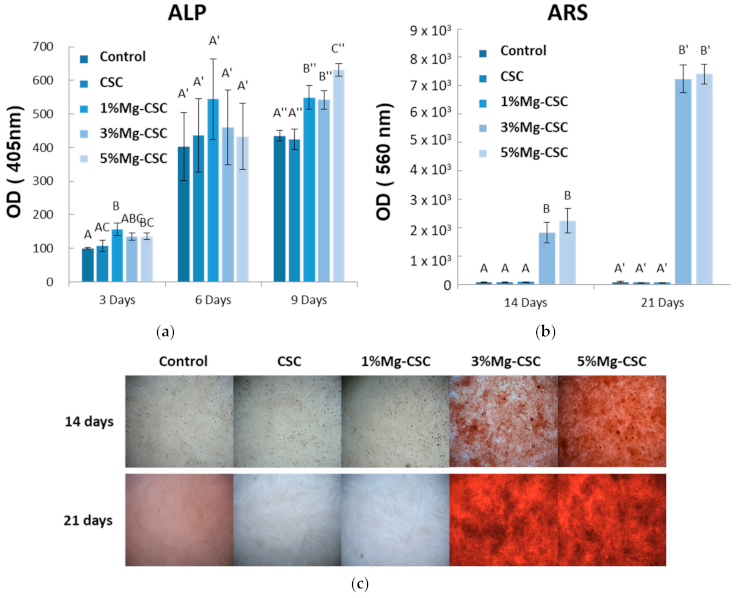
3.4. In Vitro Cytotoxicity and Differential Test
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hilton, T.; Ferracane, J.; Mancl, L.; Baltuck, C.; Barnes, C.; Beaudry, D.; Shao, J.; Lubisich, E.; Gilbert, A.; Lowder, L.; et al. Comparison of CaOH with MTA for direct pulp capping. J. Dent. Res. 2013, 92, S16–S22. [Google Scholar] [CrossRef] [PubMed]
- Mente, J.; Hage, N.; Pfefferle, T.; Koch, M.J.; Geletneky, B.; Dreyhaupt, J.; Martin, N.; Staehle, H.J. Treatment outcome of mineral trioxide aggregate: Repair of root perforations. J. Endod. 2010, 36, 208–213. [Google Scholar] [CrossRef]
- Tang, Y.; Li, X.; Yin, S. Outcomes of MTA as root-end filling in endodontic surgery: A systematic review. Quintessence Int 2010, 41, 41. [Google Scholar]
- Torabinejad, M.; Parirokh, M. Mineral trioxide aggregate: A comprehensive literature review—Part II: Leakage and biocompatibility investigations. J. Endod. 2010, 36, 190–202. [Google Scholar] [CrossRef] [PubMed]
- Parirokh, M.; Torabinejad, M. Mineral trioxide aggregate: A comprehensive literature review—Part I: Chemical, physical, and antibacterial properties. J. Endod. 2010, 36, 16–27. [Google Scholar] [CrossRef]
- Camilleri, J.; Sorrentino, F.; Damidot, D. Investigation of the hydration and bioactivity of radiopacified tricalcium silicate cement, Biodentine and MTA Angelus. Dent. Mater. 2013, 29, 580–593. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.-H.; Shie, M.-Y.; Kao, C.-T.; Ding, S.-J. The effect of setting accelerator on properties of mineral trioxide aggregate. J. Endod. 2008, 34, 590–593. [Google Scholar] [CrossRef] [PubMed]
- Butt, N.; Bali, A.; Talwar, S.; Chaudhry, S.; Nawal, R.; Yadav, S. Comparison of physical and mechanical properties of mineral trioxide aggregate and Biodentine. Indian J. Dent. Res. 2014, 25, 692. [Google Scholar] [CrossRef]
- Flores-Ledesma, A.; Santana, F.B.; Bucio, L.; Arenas-Alatorre, J.; Faraji, M.; Wintergerst, A. Bioactive materials improve some physical properties of a MTA-like cement. Mater. Sci. Eng. C 2017, 71, 150–155. [Google Scholar] [CrossRef]
- Taylor, H.F.W. Cement Chemistry, 2nd ed.; Thomas Telford: London, UK, 1997. [Google Scholar]
- Liu, W.-C.; Hu, C.-C.; Tseng, Y.-Y.; Sakthivel, R.; Fan, K.-S.; Wang, A.-N.; Wang, Y.-M.; Chung, R.-J. Study on strontium doped tricalcium silicate synthesized through sol-gel process. Mater. Sci. Eng. C 2020, 108, 110431. [Google Scholar] [CrossRef]
- Zhang, Y.; Luan, J.; Zhang, Y.; Sha, S.; Li, S.; Xu, S.; Xu, D. Preparation and characterization of iron-doped tricalcium silicate-based bone cement as a bone repair material. Materials 2020, 13, 3670. [Google Scholar] [CrossRef]
- Yang, L.; Perez-Amodio, S.; Groot, F.Y.B.-D.; Everts, V.; van Blitterswijk, C.A.; Habibovic, P. The effects of inorganic additives to calcium phosphate on in vitro behavior of osteoblasts and osteoclasts. Biomatererials 2010, 31, 2976–2989. [Google Scholar] [CrossRef]
- Hoppe, A.; Güldal, N.S.; Boccaccini, A.R. A review of the biological response to ionic dissolution products from bioactive glasses and glass-ceramics. Biomaterials 2011, 32, 2757–2774. [Google Scholar] [CrossRef]
- Lehmann, G.; Palmero, P.; Cacciotti, I.; Pecci, R.; Campagnolo, L.; Bedini, R.; Siracusa, G.; Bianco, A.; Camaioni, A.; Montanaro, L. Design, production and biocompatibility of nanostructured porous HAp and Si-HAp ceramics as three-dimensional scaffolds for stem cell culture and differentiation. Ceram-Silik 2010, 54, 90–96. [Google Scholar]
- Lehmann, G.; Cacciotti, I.; Palmero, P.; Montanaro, L.; Bianco, A.; Campagnolo, L.; Camaioni, A. Differentiation of osteoblast and osteoclast precursors on pure and silicon-substituted synthesized hydroxyapatites. Biomed. Mater. 2012, 7, 055001. [Google Scholar] [CrossRef]
- Cacciotti, I.; Bianco, A.; Lombardi, M.; Montanaro, L. Mg-substituted hydroxyapatite nanopowders: Synthesis, thermal stability and sintering behaviour. J. Eur. Ceram. Soc. 2009, 29, 2969–2978. [Google Scholar] [CrossRef]
- Cacciotti, I.; Bianco, A. High thermally stable Mg-substituted tricalcium phosphate via precipitation. Ceram. Int. 2011, 37, 127–137. [Google Scholar] [CrossRef]
- Knabe, C.; Stiller, M.; Berger, G.; Reif, D.; Gildenhaar, R.; Howlett, C.R.; Zreiqat, H. The effect of bioactive glass ceramics on the ex-pression of bone-related genes and proteins in vitro. Clin. Oral. Implant Res. 2005, 16, 119–127. [Google Scholar] [CrossRef]
- Saboori, A.; Rabiee, M.; Moztarzadeh, F.; Sheikhi, M.; Tahriri, M.; Karimi, M. Synthesis, characterization and in vitro bioactivity of sol-gel-derived SiO2–CaO–P2O5–MgO bioglass. Mater. Sci. Eng. C 2009, 29, 335–340. [Google Scholar] [CrossRef]
- Soulie, J.; Nedelec, J.-M.; Jallot, E. Influence of Mg doping on the early steps of physico-chemical reactivity of sol–gel derived bioactive glasses in biological medium. Phys. Chem. Chem. Phys. 2009, 11, 10473–10483. [Google Scholar] [CrossRef]
- Chen, X.; Liao, X.; Huang, Z.; You, P.; Chen, C.; Kang, Y.; Yin, G. Synthesis and characterization of novel multiphase bioactive glass-ceramics in the CaO-MgO-SiO2system. J. Biomed. Mater. Res. Part B Appl. Biomater. 2010, 93, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.-S.; Lin, H.-P.; Chan, J.C.C.; Wang, W.-C.; Hung, P.-H.; Tsai, Y.-H.; Lee, Y.-L. A novel sol-gel-derived calcium silicate cement with short setting time for application in endodontic repair of perforations. Int. J. Nanomed. 2018, 13, 261–271. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Wang, J.; Zhai, W.; Wang, Z.; Chang, J. The self-setting properties and in vitro bioactivity of tricalcium silicate. Biomaterials 2005, 26, 6113–6121. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Chang, J. Sol–gel synthesis and in vitro bioactivity of tricalcium silicate powders. Mater. Lett. 2004, 58, 2350–2353. [Google Scholar] [CrossRef]
- Brinker, C.J.; Scherer, G.W. Sol-Gel Science: The Physics and Chemistry of SOL-Gel Processing, 1st ed.; Academic Press: Boston, MA, USA, 2013. [Google Scholar]
- ISO/WD. ISO 6876. Dentistry—Root Canal Sealing Materials. Filling and Restorative Materials; International Organization for Standardization: Geneva, Switzerland, 2012. [Google Scholar]
- ISO 9917–1. Dentistry—Water Based Cements—Part 1: Powder/Liquid Acid-Base Cements; International Organization for Standardization: Geneva, Switzerland, 2007. [Google Scholar]
- ISO 10993-12: 2012 Biological Evaluation of Medical Devices—Part 12: Specimen Preparation and Reference Materials; International Organization for Standardization: Geneva, Switzerland, 2012.
- Houhua, P.; Liping, H.; Xie, Y.; Pan, H.; Zhao, J.; Huang, L.; Zheng, X. Different response of osteoblastic cells to Mg2+, Zn2+ and Sr2+ doped calcium silicate coatings. J. Mater. Sci. Mater. Med. 2016, 27, 1–13. [Google Scholar] [CrossRef]
- Choi, Y.; Sun, W.; Kim, Y.; Kim, I.-R.; Gong, M.-K.; Yoon, S.-Y.; Bae, M.-K.; Park, B.-S.; Park, S.-B.; Kim, Y.-I. Effects of Zn-doped mesoporous bioactive glass nanoparticles in etch-and-rinse adhesive on the microtensile bond strength. Nanomaterials 2020, 10, 1943. [Google Scholar] [CrossRef]
- Mostafa, N.; Shaltout, A.; Omar, H.; Abo-El-Enein, S. Hydrothermal synthesis and characterization of aluminium and sulfate substituted 1.1nm tobermorites. J. Alloys Compd. 2009, 467, 332–337. [Google Scholar] [CrossRef]
- Wu, M.; Tao, B.; Wang, T.; Zhang, Y.; Wei, W.; Wang, C. Fast-setting and anti-washout tricalcium silicate/disodium hydrogen phosphate composite cement for dental application. Ceram. Int. 2019, 45, 24182–24192. [Google Scholar] [CrossRef]
- Lee, Y.-L.; Wang, W.-H.; Lin, F.-H.; Lin, C.-P. Hydration behaviors of calcium silicate-based biomaterials. J. Formos. Med. Assoc. 2017, 116, 424–431. [Google Scholar] [CrossRef]
- Li, Q.; Hurt, A.P.; Coleman, N.J. The application of 29Si NMR spectroscopy to the analysis of calcium silicate-based cement using BiodentineTM as an example. J. Funct. Biomater. 2019, 10, 25. [Google Scholar] [CrossRef]
- Sembiring, S.; Riyanto, A.; Simanjuntak, W.; Situmeang, R. Effect of MgO-SiO2 ratio on the forsterite (Mg2SiO4) precursors characteristics derived from amorphous rice husk silica. Orient. J. Chem. 2017, 33, 1828–1836. [Google Scholar] [CrossRef]
- Giraudo, N.; Bergdolt, S.; Wohlgemuth, J.; Welle, A.; Schuhmann, R.; Königer, F.; Thissen, P.; Koeniger, F. Calcium silicate phases explained by high-temperature-resistant phosphate probe molecules. Langmuir 2016, 32, 13577–13584. [Google Scholar] [CrossRef]
- Balbinot, G.D.S.; Leitune, V.C.B.; Nunes, J.S.; Visioli, F.; Collares, F.M. Synthesis of sol–gel derived calcium silicate particles and development of a bioactive endodontic cement. Dent. Mater. 2020, 36, 135–144. [Google Scholar] [CrossRef]
- Tantawy, M.A.; Shatat, M.R.; El-Roudi, A.M.; Taher, M.A.; Abd-El-Hamed, M. Low temperature synthesis of belite cement based on silica fume and lime. Int. Sch. Res. Not. 2014, 2014, 1–10. [Google Scholar] [CrossRef]
- Bernard, E.; Lothenbach, B.; Le Goff, F.; Pochard, I.; Dauzères, A. Effect of magnesium on calcium silicate hydrate (C-S-H). Cem. Concr. Res. 2017, 97, 61–72. [Google Scholar] [CrossRef]
- Zhang, T.; Zou, J.; Wang, B.; Wu, Z.; Jia, Y.; Cheeseman, C.R. Characterization of magnesium silicate hydrate (MSH) gel formed by reacting MGO and silica fume. Materials 2018, 11, 909. [Google Scholar] [CrossRef]
- Li, L.-Y.; Cui, L.-Y.; Liu, B.; Zeng, R.-C.; Chen, X.-B.; Li, S.-Q.; Wang, Z.-L.; Han, E.-H. Corrosion resistance of glucose-induced hydrothermal calcium phosphate coating on pure magnesium. Appl. Surf. Sci. 2019, 465, 1066–1077. [Google Scholar] [CrossRef]
- Sha, W.; O’Neill, E.; Guo, Z. Differential scanning calorimetry study of ordinary Portland cement. Cem. Concr. Res. 1999, 29, 1487–1489. [Google Scholar] [CrossRef]
- Cuesta, A.; Zea-Garcia, J.D.; Londono-Zuluaga, D.; De La Torre, A.G.; Santacruz, I.; Vallcorba, O.; Dapiaggi, M.; Sanfélix, S.G.; Aranda, M.A.G. Multiscale understanding of tricalcium silicate hydration reactions. Sci. Rep. 2018, 8, 8544. [Google Scholar] [CrossRef]
- Liu, W.; Zhai, D.; Huan, Z.; Wu, C.; Chang, J. Novel tricalcium silicate/magnesium phosphate composite bone cement having high compressive strength, in vitro bioactivity and cytocompatibility. Acta Biomater. 2015, 21, 217–227. [Google Scholar] [CrossRef]
- Hamed, M.M.; Holiel, M.; Ahmed, I.M. Sorption behavior of cesium, cobalt and europium radionuclides onto hydroxyl magnesium silicate. Radiochim. Acta 2016, 104, 873–890. [Google Scholar] [CrossRef]
- Rasskazova, L.A.; Zhuk, I.V.; Korotchenko, N.M.; Brichkov, A.S.; Chen, Y.-W.; Paukshtis, E.A.; Ivanov, V.K.; Kurzina, I.A.; Kozik, V.V. Synthesis of magnesium-and silicon-modified hydroxyapatites by microwave-assisted method. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef]
- Liu, X.; Lin, W.; Chen, B.; Zhang, F.; Zhao, P.; Parsons, A.; Rau, C.; Robinson, I. Coherent diffraction study of calcite crystallization during the hydration of tricalcium silicate. Mater. Des. 2018, 157, 251–257. [Google Scholar] [CrossRef]
- Sharma, U.; Ali, D.; Singh, L.P. Formation of C–S–H nuclei using silica nanoparticles during early age hydration of cementitious system. Eur. J. Environ. Civ. Eng. 2019, 1–12. [Google Scholar] [CrossRef]
- Durgun, E.; Manzano, H.; Pellenq, R.J.M.; Grossman, J.C. Understanding and controlling the reactivity of the calcium silicate phases from first principles. Chem. Mater. 2012, 24, 1262–1267. [Google Scholar] [CrossRef]
- Mestres, G.; Ginebra, M.-P. Novel magnesium phosphate cements with high early strength and antibacterial properties. Acta Biomater. 2011, 7, 1853–1861. [Google Scholar] [CrossRef]
- Abdalla, M.M.; Lung, C.Y.K.; Neelakantan, P.; Matinlinna, J.P. A novel, doped calcium silicate bioceramic synthesized by sol–gel method: Investigation of setting time and biological properties. J. Biomed. Mater. Res. Part B Appl. Biomater. 2020, 108, 56–66. [Google Scholar] [CrossRef]
- Stephan, D.; Dikoundou, S.N.; Raudaschl-Sieber, G. Hydration characteristics and hydration products of tricalcium silicate doped with a combination of MgO, Al2O3 and Fe2O3. Thermochim. Acta 2008, 472, 64–73. [Google Scholar] [CrossRef]
- Walling, S.A.; Provis, J.L. Magnesia-based cements: A journey of 150 years, and cements for the future? Chem. Rev. 2016, 116, 4170–4204. [Google Scholar] [CrossRef]
- Reddy, M.; Nancollas, G. The crystallization of calcium carbonate: IV. The effect of magnesium, strontium and sulfate ions. J. Cryst. Growth 1976, 35, 33–38. [Google Scholar] [CrossRef]
- Myszka, B.; Schüßler, M.; Hurle, K.; Demmert, B.; Detsch, R.; Boccaccini, A.R.; Wolf, S.E. Phase-specific bioactivity and altered Ostwald ripening pathways of calcium carbonate polymorphs in simulated body fluid. RSC Adv. 2019, 9, 18232–18244. [Google Scholar] [CrossRef]
- House, W.A. Inhibition of calcite crystal growth by inorganic phosphate. J. Colloid Interface Sci. 1987, 119, 505–511. [Google Scholar] [CrossRef]
- Ciraldo, F.E.; Boccardi, E.; Melli, V.; Westhauser, F.; Boccaccini, A.R. Tackling bioactive glass excessive in vitro bioreactivity: Preconditioning approaches for cell culture tests. Acta Biomater. 2018, 75, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Khedmat, S.; Dehghan, S.; Hadjati, J.; Masoumi, F.; Nekoofar, M.H.; Dummer, P.M.H. In vitro cytotoxicity of four calcium sili-cate-based endodontic cements on human monocytes, a colorimetric MTT assay. Restor. Dentist. Endodont. 2014, 39, 149. [Google Scholar] [CrossRef]
- Zhang, X.; Zu, H.; Zhao, D.; Yang, K.; Tian, S.; Yu, X.; Lu, F.; Liu, B.; Wang, B.; Wang, W.; et al. Ion channel functional protein kinase TRPM7 regulates Mg ions to promote the osteoinduction of human osteoblast via PI3K pathway: In vitro simulation of the bone-repairing effect of Mg-based alloy implant. Acta Biomater. 2017, 63, 369–382. [Google Scholar] [CrossRef]
- D’Onofrio, A.; Kent, N.; Shahdad, S.; Hill, R. Development of novel strontium containing bioactive glass based calcium phosphate cement. Dent. Mater. 2016, 32, 703–712. [Google Scholar] [CrossRef]
- Dawood, A.E.; Parashos, P.; Wong, R.H.; Reynolds, E.C.; Manton, D.J. Calcium silicate-based cements: Composition, properties, and clinical applications. J. Investig. Clin. Dent. 2017, 8, e12195. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yoo, K.-H.; Kim, Y.-I.; Yoon, S.-Y. Physicochemical and Biological Properties of Mg-Doped Calcium Silicate Endodontic Cement. Materials 2021, 14, 1843. https://doi.org/10.3390/ma14081843
Yoo K-H, Kim Y-I, Yoon S-Y. Physicochemical and Biological Properties of Mg-Doped Calcium Silicate Endodontic Cement. Materials. 2021; 14(8):1843. https://doi.org/10.3390/ma14081843
Chicago/Turabian StyleYoo, Kyung-Hyeon, Yong-Il Kim, and Seog-Young Yoon. 2021. "Physicochemical and Biological Properties of Mg-Doped Calcium Silicate Endodontic Cement" Materials 14, no. 8: 1843. https://doi.org/10.3390/ma14081843
APA StyleYoo, K.-H., Kim, Y.-I., & Yoon, S.-Y. (2021). Physicochemical and Biological Properties of Mg-Doped Calcium Silicate Endodontic Cement. Materials, 14(8), 1843. https://doi.org/10.3390/ma14081843