Influence of Modified Epoxy Resins on Peroxide Curing, Mechanical Properties and Adhesion of SBR, NBR and XNBR to Silver Wires—Part II: Application of Carboxy-Containing Peroxy Oligomer (CPO)
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
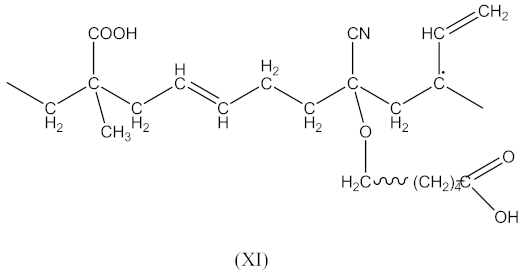
2.1.1. Synthesis of an Oligomeric Modifier
2.1.2. Preparation of Rubber Vulcanizates
2.2. Methods
3. Results
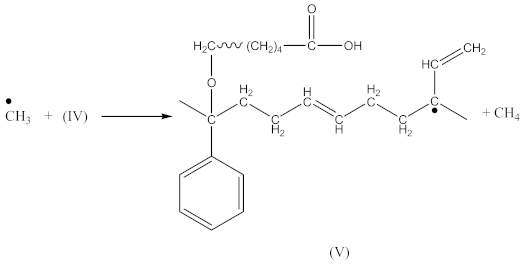
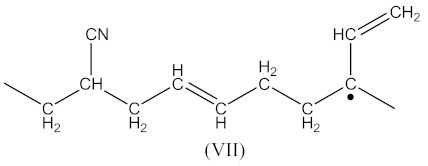
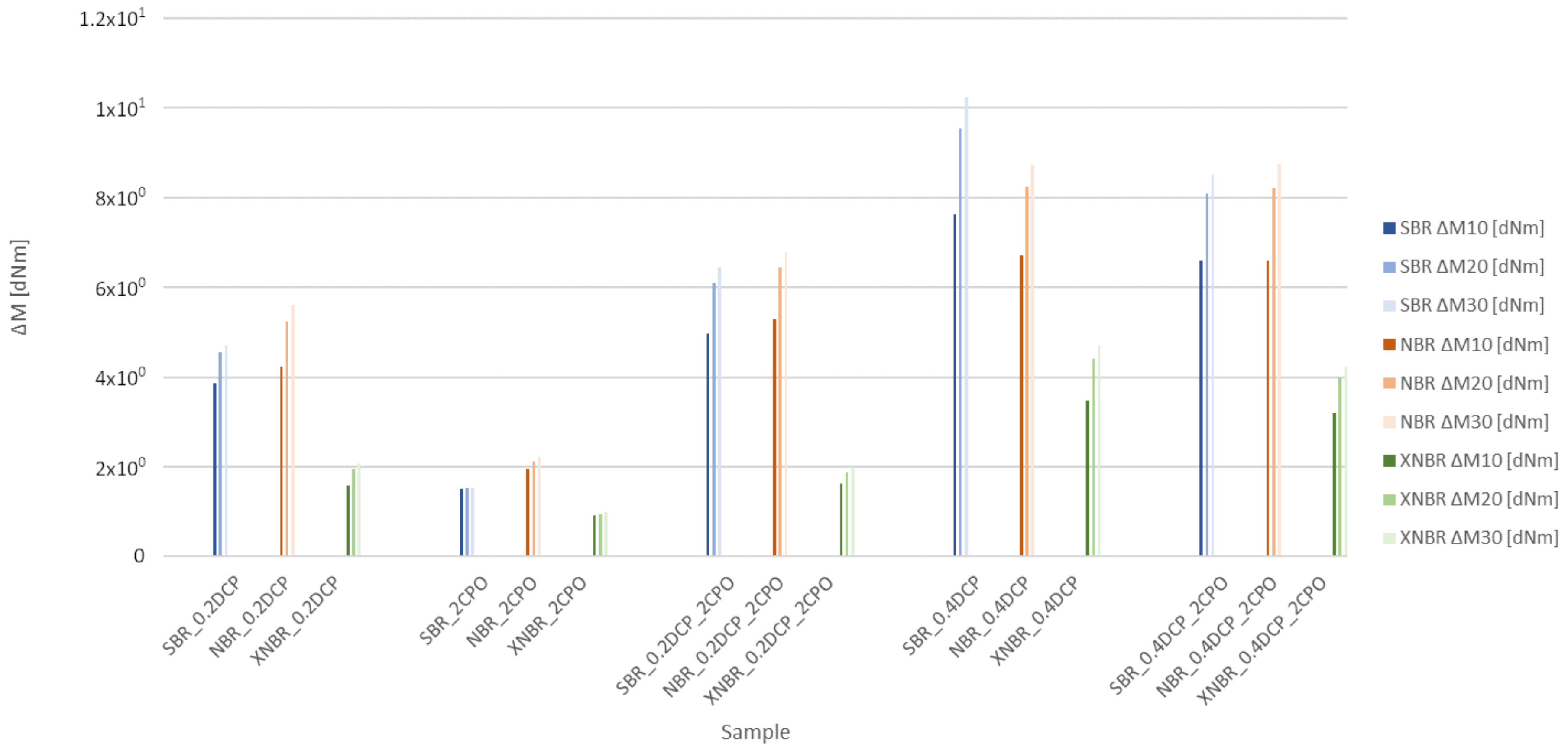
3.1. Kinetics of Crosslinking
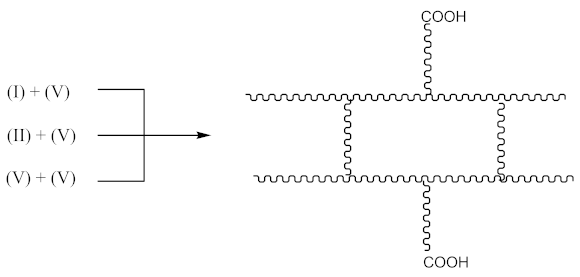
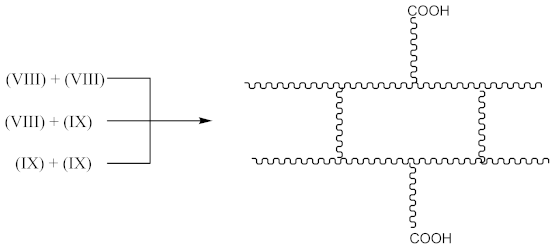
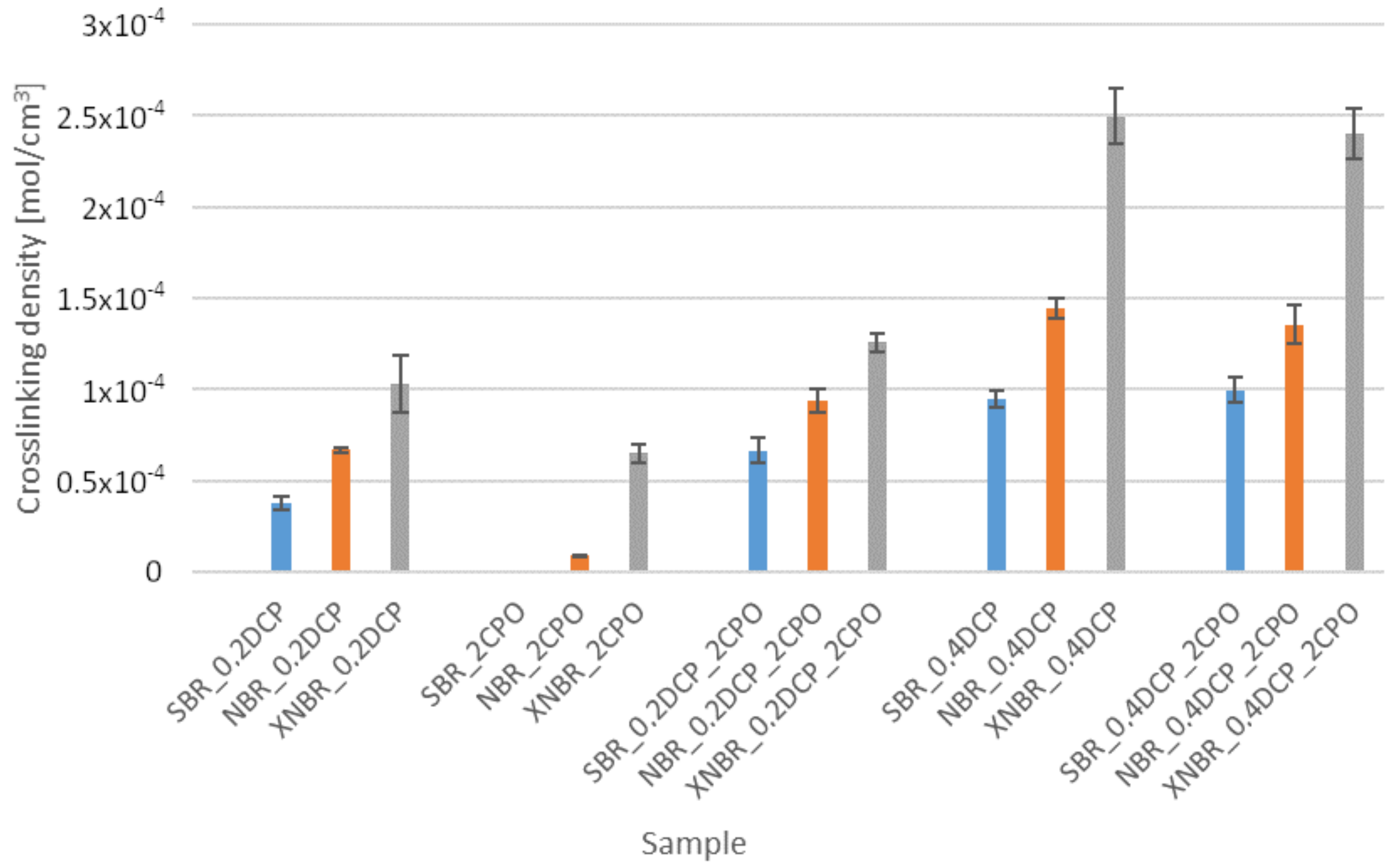
3.2. Crosslink Density of Rubber


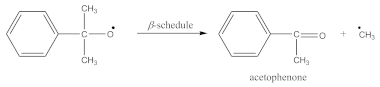

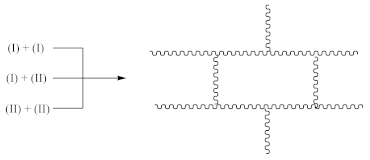
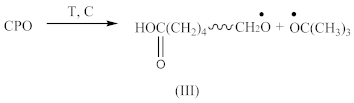



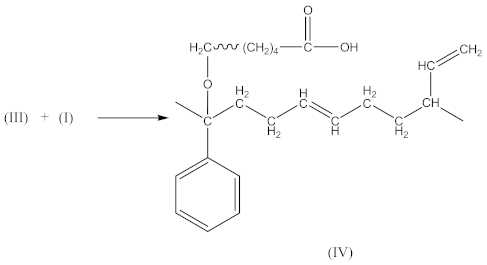
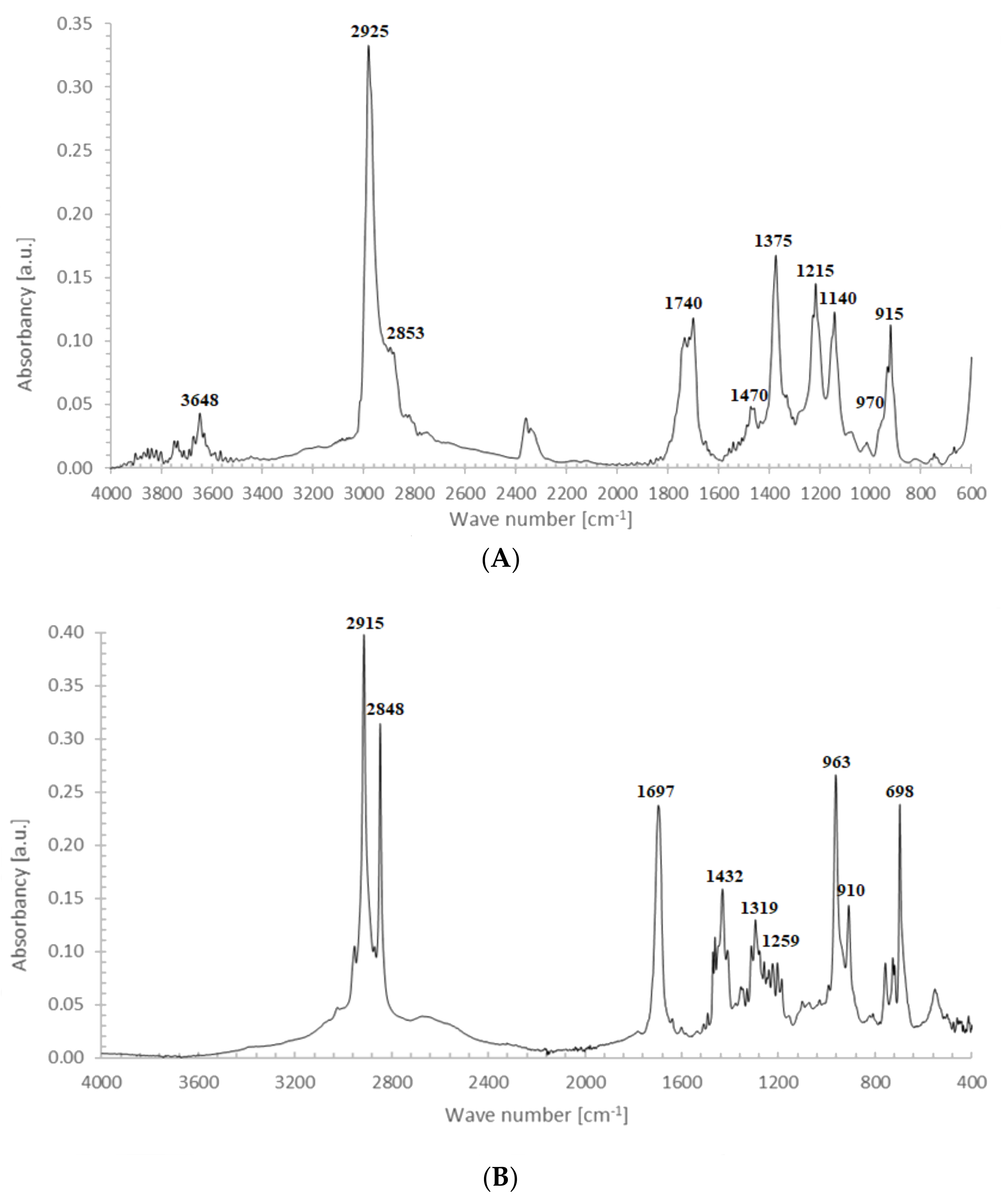
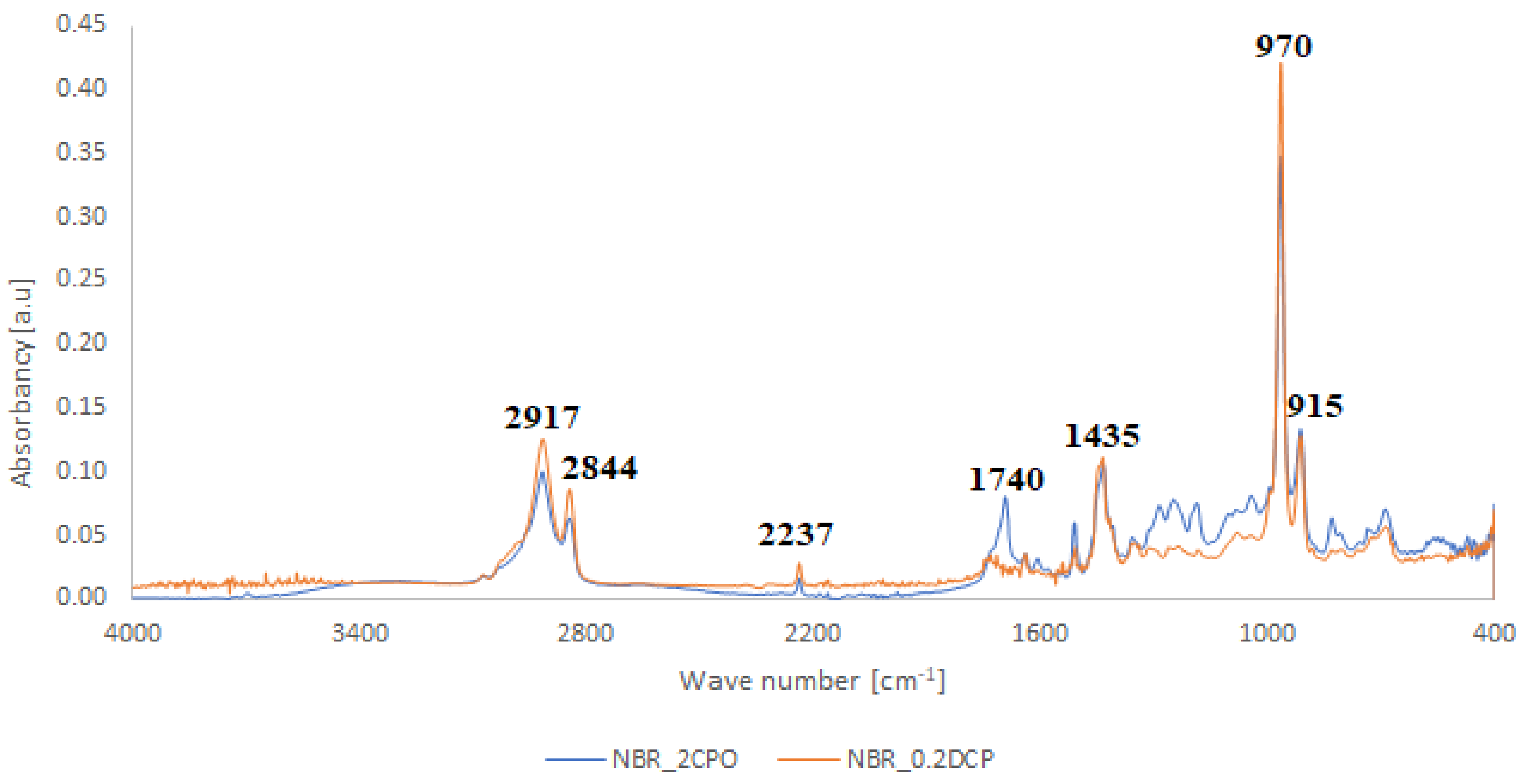
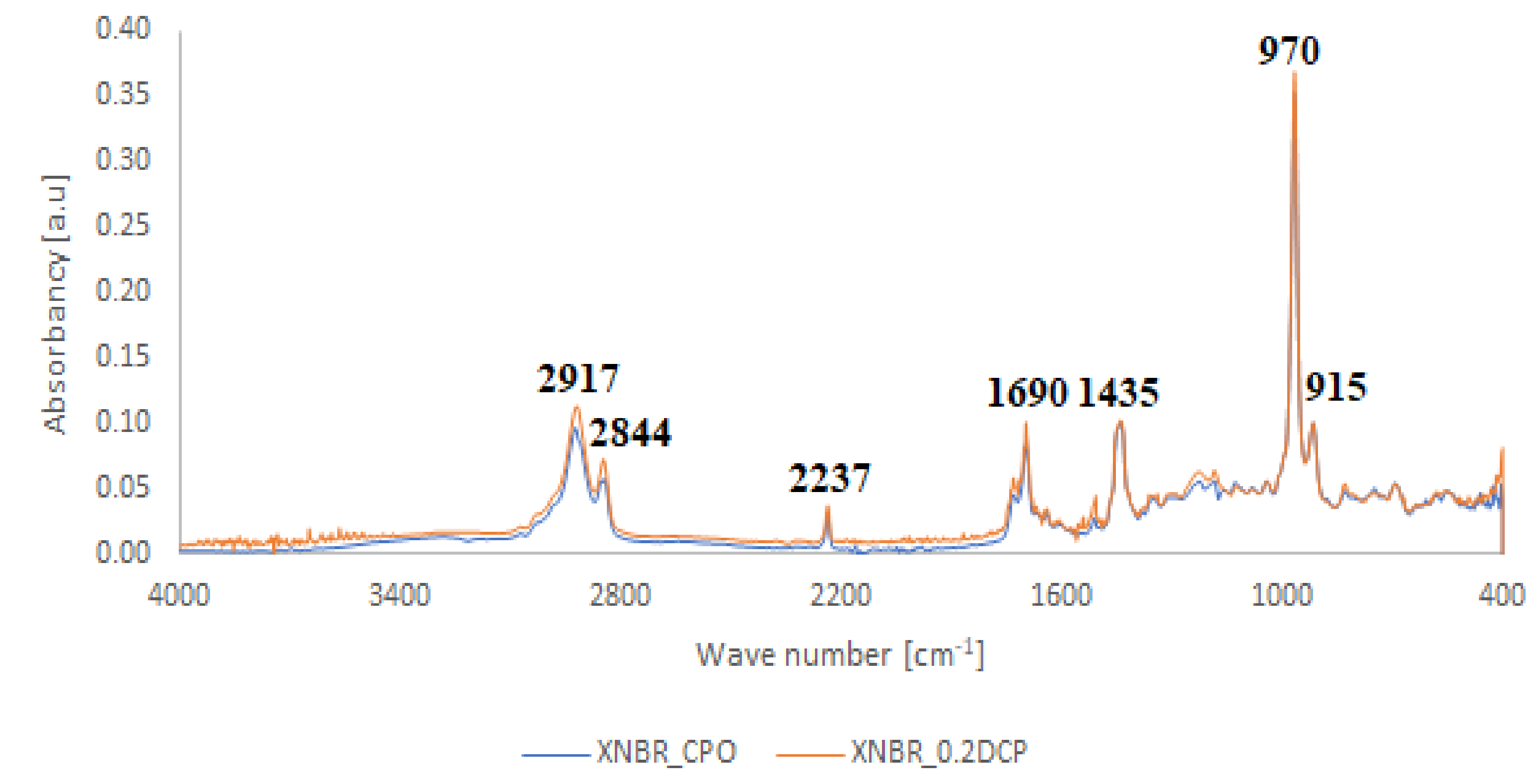
- At 3648 cm−1, vibrations of free OH groups;
- At 2925 and 2853 cm−1, symmetric and asymmetric vibrations of CH2 and CH3 groups;
- At broad absorption at 1740 cm−1, related to C=O vibrations, probably from aldehyde groups;
- At 1470 and 1375 cm−1, stretching vibrations of CH2 and CH3 groups; and
- At 1325, 1215, 1140 and 1050 cm−1, vibrations in alcohol and/or ether fragments.








3.3. Mechanical Properties of Rubber
3.3.1. Tensile Strength (TS)
3.3.2. Tear Resistance (TES)
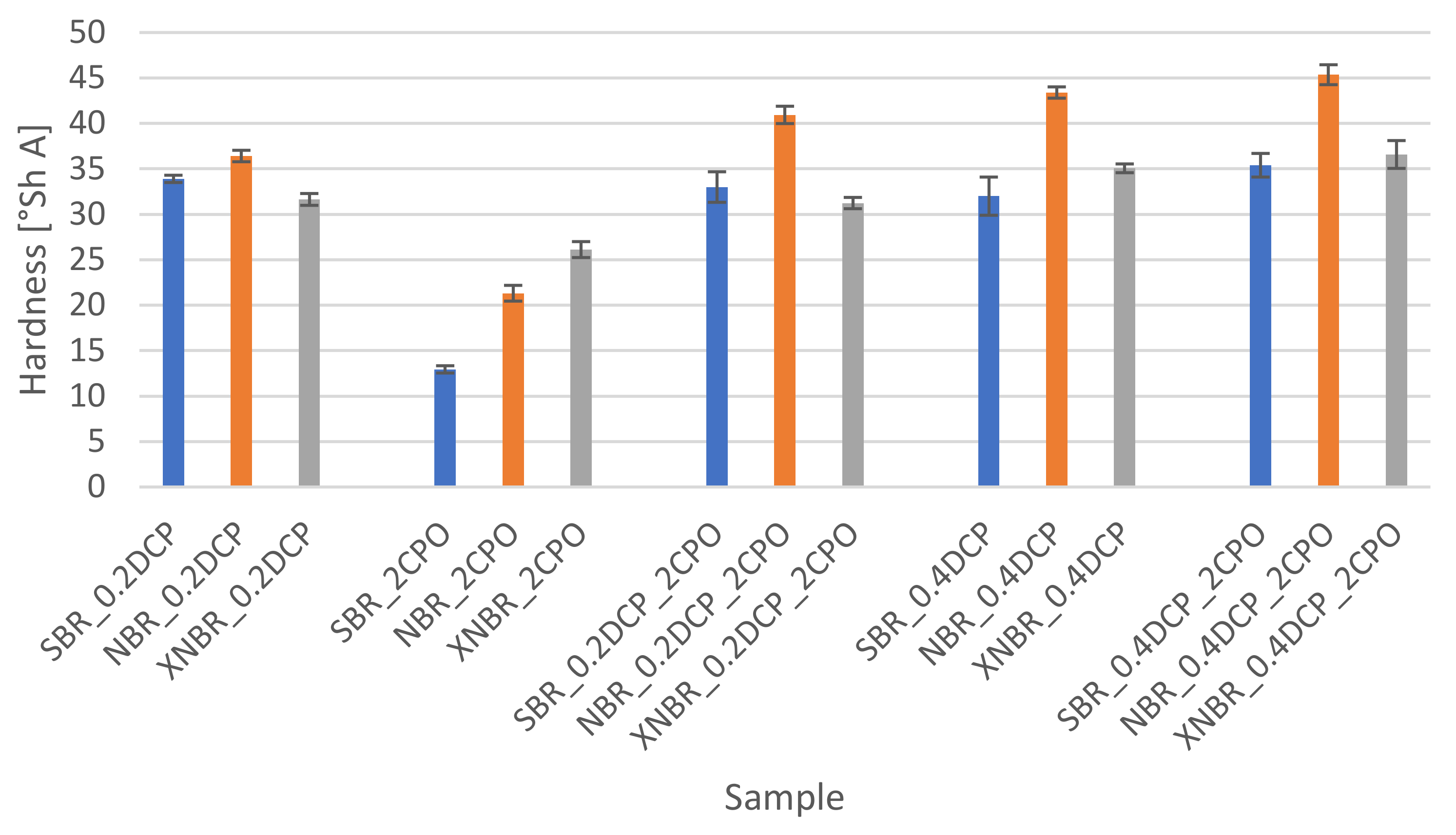
3.3.3. Hardness
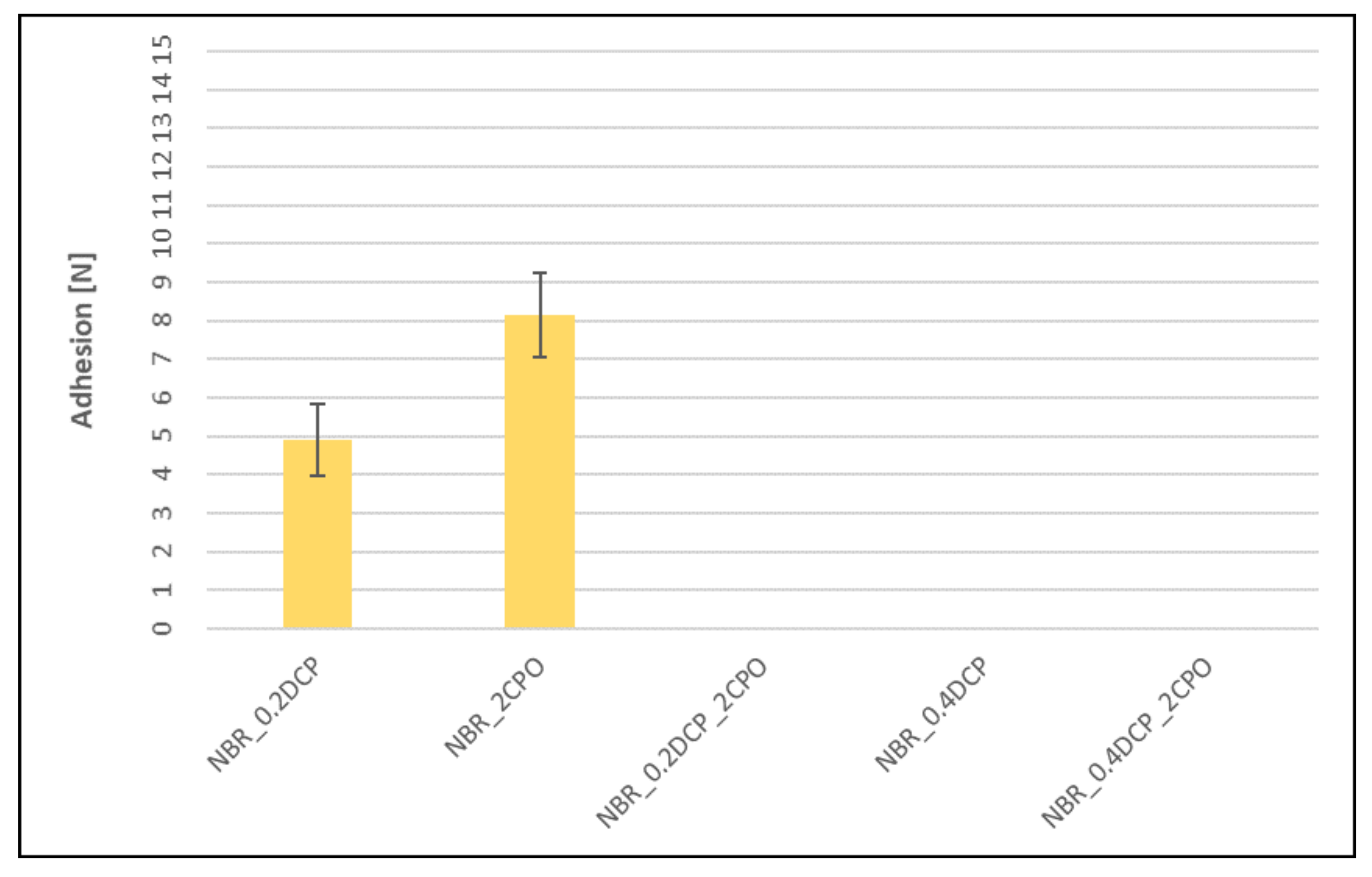
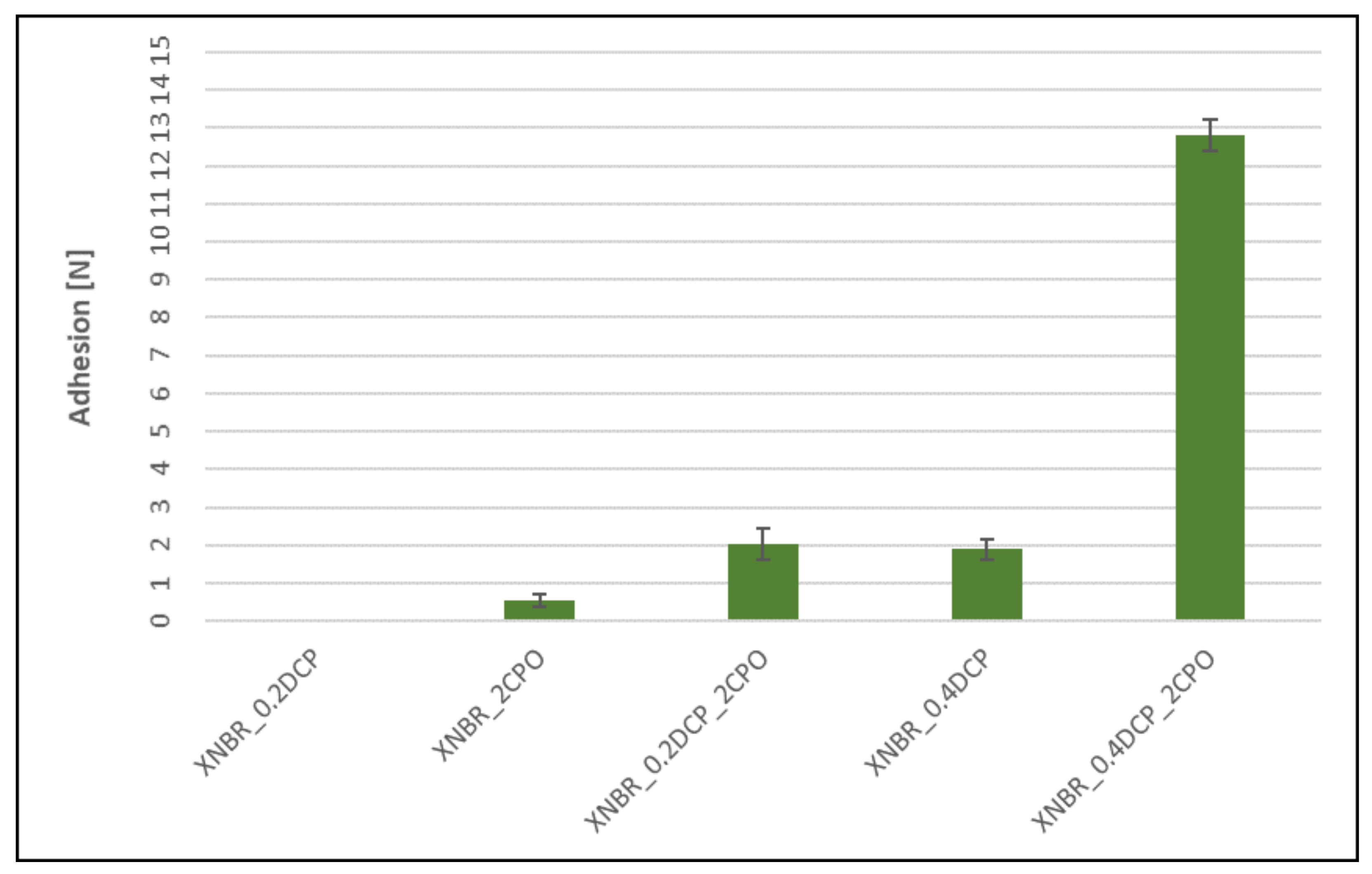
3.4. Adhesion
4. Discussion
5. Conclusions
- The presence of sterically available functional groups that can form strong covalent or covalent–ion metal–polymer bonds, additionally introduced by the modification of the rubber by the CPO resin;
- The optimal degree of rubber crosslinking, also modified by the resin, ensuring the coherence of the polymer matrix; and
- The conditions of vulcanization and the processing of materials which can introduce stresses at the metal–polymer interface.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chudzik, J.; Bieliński, D.M.; Bratychak, M.; Demchuk, Y.; Shyshchak, O.; Jędrzejczyk, M.; Celichowski, G. Influence of Modified Epoxy Resins on Peroxide Curing, Me-chanical Properties and Adhesion of SBR, NBR and XNBR to Silver Wires. Part I: Application of Monoperoxy Derivative of Epoxy Resin (PO). Materials 2021, in press. [Google Scholar]
- Bratychak, M.; Bashta, B.; Astakhova, O.; Shyshchak, O.; Zubal, O. Synthesis Mechanism and Properties of Epoxy Resins Modified with Adipic Acid. Chem. Chem. Technol. 2019, 13, 52–58. [Google Scholar] [CrossRef]
- Sorokin, M.; Lyalushko, K. Praktikum po Khimii i Tehnologii Plenkoobrazuyushih Veshestv; Khimiya: Moscow, Russia, 1971. (In Russian) [Google Scholar]
- Bashta, B.; Bruzdziak, P.; Astakhova, O.; Shyshchak, O.; Bratychak, M. Synthesis and Properties of Carboxy-Containing Peroxy Oligomer. Chem. Chem. Technol. 2013, 7, 413–421. [Google Scholar] [CrossRef]
- Yano, S.; Tadano, K.; Nagao, N.; Kutsumizu, S.; Tachino, H.; Hirasawa, E. Dielectric relaxation studies on water absorption of ethylene ionomers. Macromolecules 1992, 25, 7168–7171. [Google Scholar] [CrossRef]
- Mandal, U.K.; Tripathy, D.K.; De, S.K. Effect of silica filler on dynamic mechanical properties of lonic elastomer based on carboxylated nitrile rubber. J. Appl. Polym. Sci. 1995, 55, 1185–1191. [Google Scholar] [CrossRef]
- Vondracek, P.; Pouchelon, A. Ammonia-Induced Tensile Set and Swelling in Silica-Filled Silicone Rubber. Rubber Chem. Technol. 1990, 63, 202–214. [Google Scholar] [CrossRef]
- Akiba, M. Vulcanization and crosslinking in elastomers. Prog. Polym. Sci. 1997, 22, 475–521. [Google Scholar] [CrossRef]
- Ibarra, L.; Alzorriz, M. Effect of temperature on the crosslink densities of nitrile rubber and carboxylated nitrile rubber with zinc peroxide. J. Appl. Polym. Sci. 2002, 86, 335–340. [Google Scholar] [CrossRef]
- Ibarra, L.J. Effect of crosslinking type on the physical properties of carboxylated acrylonitrile butadiene elastomers. J. Appl. Polym. Sci. 1999, 73, 927–933. [Google Scholar] [CrossRef]
- Ibarra, L.; Rodriguez, A.; Mora, I. Ionic nanocomposites based on XNBR-OMg filled with layered nanoclays. Eur. Polym. J. 2007, 43, 753–761. [Google Scholar] [CrossRef]
- Gonzalez, L.; Rodriguez, A.; Marcos-Fernández, Á.; Chamorro, C. Crosslink Reaction Mechanisms of Diene Rubber with Dicumyl Peroxide. Rubber Chem. Technol. 1996, 69, 203–214. [Google Scholar] [CrossRef]
- Kruželák, J.; Sýkora, R.; Hudec, I. Sulphur and peroxide vulcanisation of rubber compounds—Overview. Chem. Pap. 2016, 70, 1533–1555. [Google Scholar] [CrossRef]
- Ibarra, L.; Marcos-Fernandez, A.; Alzorriz, M. Mechanistic approach to the curing of carboxylated nitrile rubber (XNBR) by zinc peroxide/zinc oxide. Polymer 2002, 43, 1649. [Google Scholar] [CrossRef]
- Park, J.; You, I.; Sangbaie, S.; Unyong, J. Material approaches to stretchable strain sensors. ChemPhysChem 2015, 16, 1155. [Google Scholar] [CrossRef] [PubMed]
- Chou, H.L.; Wu, C.M.; Lin, F.D.; Rick, J. Interactions between silver nanoparticles and polyvinyl alcohol nanofibers. AIP Adv. 2014, 4, 087111. [Google Scholar] [CrossRef]
- Friedrich, J. Metal-Polymer Systems, Interface Design and Chemical Bonding; Wiley-VCH Verlag: Boschstraat, Germany, 2018; ISBN 978-3-527-33677-7. [Google Scholar]
- Strunskus, T.; Grunze, M.; Kochendoerfer, G.; Wöll, C.H. Identification of physical and chemical interaction mechanisms for the metals gold, silver, copper, palladium, chromium, and potassium with polyimide surfaces. Langmuir 1996, 12, 2712. [Google Scholar] [CrossRef]
- Kruželák, J.; Sýkora, R.; Hudec, I. Influence of mixed sulfur/peroxide curing system and thermo-oxidative ageing on the properties of rubber magnetic composites. J. Polym. J. Eng. 2015, 35, 21. [Google Scholar] [CrossRef]
- Lü, P.; Qu, Z.; Wang, Q.; Bai, L.; Zhao, S. Nonlinear Conductive Behaviour of Silver Nanowires/Silicone Rubber Composites. IOP Conf. Ser. Mater. Sci. Eng. 2018, 301, 012052. [Google Scholar] [CrossRef]


| Compound | Rubber | DCP | CPO |
|---|---|---|---|
| SBR_0.2DCP | 100 | 0.2 | 0 |
| SBR_2CPO | 100 | 0 | 2 |
| SBR_0.2DCP_2CPO | 100 | 0.2 | 2 |
| SBR_0.4DCP | 100 | 0.4 | 0 |
| SBR_0.4DCP_2CPO | 100 | 0.4 | 2 |
| NBR_0.2DCP | 100 | 0.2 | 0 |
| NBR_2CPO | 100 | 0 | 2 |
| NBR_0.2DCP_2CPO | 100 | 0.2 | 2 |
| NBR_0.4DCP | 100 | 0.4 | 0 |
| NBR_0.4DCP_2CPO | 100 | 0.4 | 2 |
| XNBR_0.2DCP | 100 | 0.2 | 0 |
| XNBR_2CPO | 100 | 0 | 2 |
| XNBR_0.2DCP_2CPO | 100 | 0.2 | 2 |
| XNBR_0.4DCP | 100 | 0.4 | 0 |
| XNBR_0.4DCP_2CPO | 100 | 0.4 | 2 |
| Rubber Mix | t05 (min) | t90 (min) | MH (dNm) | ML (dNM) | ΔM (dNm) |
|---|---|---|---|---|---|
| SBR_0.2DCP | 1.0 | 17 | 5.2 | 1.0 | 4.2 |
| SBR_2CPO | 0.5 | 18 | 2.4 | 1.0 | 1.4 |
| SBR_0.2DCP_2CPO | 1.0 | 20 | 6.8 | 1.0 | 5.8 |
| SBR_0.4DCP | 1.0 | 17 | 10.2 | 1.0 | 9.2 |
| SBR_0.4DCP_2CPO | 1.0 | 18 | 8.7 | 1.0 | 7.7 |
| NBR_0.2DCP | 1.0 | 25 | 6.5 | 1.4 | 5.1 |
| NBR_2CPO | 1.0 | 32 | 3.4 | 1.4 | 2.0 |
| NBR_0.2DCP_2CPO | 1.0 | 24 | 7.5 | 1.4 | 6.1 |
| NBR_0.4DCP | 1.0 | 21 | 9.1 | 1.3 | 7.8 |
| NBR_0.4DCP_2CPO | 1.0 | 22 | 9.2 | 1.3 | 7.9 |
| XNBR_0.2DCP | 1.0 | 25 | 2.4 | 0.5 | 1.9 |
| XNBR_2CPO | 1.0 | 40 | 1.4 | 0.5 | 0.9 |
| XNBR_0.2DCP_2CPO | 1.2 | 25 | 2.4 | 0.5 | 1.9 |
| XNBR_0.4DCP | 1.0 | 21 | 4.7 | 0.5 | 4.2 |
| XNBR_0.4DCP_2CPO | 1.1 | 21 | 4.4 | 0.5 | 3.9 |
| Sample | ν (mol/cm3) | νA (mol/cm3) | ν − νA (mol/cm3) |
|---|---|---|---|
| SBR_0.2DCP | 0.41 × 10−4 | 0.41 × 10−4 | 0.00 |
| SBR_2CPO | 0.00 | 0.00 | 0.00 |
| SBR_0.2DCP_2CPO | 1.25 × 10−4 | 1.19 × 10−4 | 0.06 × 10−4 |
| SBR_0.4DCP_2CPO | 0.92 × 10−4 | 0.89 × 10−4 | 0.03 × 10−4 |
| SBR_0.4DCP | 0.91 × 10−4 | 0.91 × 10−4 | 0.00 |
| NBR_0.2DCP | 0.62 × 10−4 | 0.62 × 10−4 | 0.00 |
| NBR_2CPO | 1.85 × 10−5 | 0.91 × 10−5 | 0.09 × 10−4 |
| NBR_0.2DCP_2CPO | 0.99 × 10−4 | 0.93 × 10−4 | 0.06 × 10−4 |
| NBR_0.4DCP_2CPO | 1,20 × 10−4 | 1,04 × 10−4 | 0.16 × 10−4 |
| NBR_0.4DCP | 1,43 × 10−4 | 1,43 × 10−4 | 0.00 |
| XNBR_0.2DCP | 1.07 × 10−4 | 1.03 × 10−4 | 0.04 × 10−4 |
| XNBR_2CPO | 1.04 × 10−4 | 0.65 × 10−4 | 0.39 × 10−4 |
| XNBR_0.2DCP_2CPO | 1.33 × 10−4 | 1.06 × 10−4 | 0.27 × 10−4 |
| XNBR_0.4DCP_2CPO | 2.41 × 10−4 | 2.16 × 10−4 | 0.25 × 10−4 |
| XNBR_0.4DCP | 2.78 × 10−4 | 2.76 × 10−4 | 0.02 × 10−4 |
| Sample | E100 (MPa) | E200 (MPa) | E300 (MPa) | Tensile Strength (MPa) | Elongation at Break (%) |
|---|---|---|---|---|---|
| SBR_0.2DCP | 0.57 ± 0.11 | 0.96 ± 0.08 | 1.16 ± 0.06 | 1.59 ± 0.21 | 510 ± 89 |
| SBR_2CPO | 0.40 ± 0.04 | 0.43 ± 0.04 | 0.45 ± 0.05 | 0.50 ± 0.05 | 595 ± 165 |
| SBR_0.2DCP_2CPO | 0.78 ± 0.20 | 1.14 ± 0.29 | 1.62 ± 0.49 | 2.43 ± 0.90 | 374 ± 81 |
| SBR_0.4DCP | 0.72 ± 0.08 | 1.16 ± 0.00 | 1.43 ± 0.08 | 1.51 ± 0.16 | 313 ± 14 |
| SBR_0.4DCP_2CPO | 0.74 ± 0.07 | 1.31 ± 0.24 | - | 1.65 ± 0.19 | 201 ± 21 |
| NBR_0.2DCP | 0.81 ± 0.07 | 1.26 ± 0.15 | 1.55 ± 0.11 | 1.68 ± 0.12 | 287 ± 49 |
| NBR_2CPO | 0.41 ± 0.01 | 0.52 ± 0.04 | 0.61 ± 0.07 | 1.02 ± 0.11 | 643 ± 186 |
| NBR_0.2DCP_2CPO | 1.19 ± 0.25 | - | - | 1.74 ± 0.29 | 179 ± 59 |
| NBR_0.4DCP | 1.04 ± 0.14 | - | - | 1.49 ± 0.26 | 169 ± 20 |
| NBR_0.4DCP_2CPO | 0.96 ± 0.02 | - | - | 1.24 ± 0.10 | 157 ± 29 |
| XNBR_0.2DCP | 0.67 ± 0.03 | 0.77 ± 0.03 | 0.83 ± 0.03 | 1.86 ± 0.17 | 1070 ± 76 |
| XNBR_2CPO * | 0.55 ± 0.01 | 0.55 ± 0.01 | 0.52 ± 0.06 | >0.80 ± 0.06 | * |
| XNBR_0.2DCP_2CPO * | 0.67 ± 0.04 | 0.85 ± 0.07 | 0.99 ± 0.10 | >2.19 ± 0.44 | * |
| XNBR_0.4DCP | 0.76 ± 0.03 | 0.92 ± 0.05 | 1.06 ± 0.08 | 2.51 ± 0.29 | 653 ± 82 |
| XNBR_0.4DCP_2CPO | 0.74 ± 0.03 | 0.96 ± 0.05 | 1.18 ± 0.36 | 2.05 ± 0.36 | 529 ± 92 |
| Sample | Fmax (N/mm) | Fmin (N/mm) |
|---|---|---|
| SBR_0.2DCP | 6.05 | 5.78 |
| SBR_2CPO | No tearing | |
| SBR_0.2DCP_2CPO | 3.68 | 2.85 |
| SBR_0.4DCP | 3.5 | 2.3 |
| SBR_0.4DCP_2CPO | 3.6 | 2.41 |
| NBR_0.2DCP | 5.96 | 3.18 |
| NBR_2CPO | No tearing | |
| NBR_0.2DCP_2CPO | 4.08 | 2.96 |
| NBR_0.4DCP | 2.66 | 1.72 |
| NBR_0.4DCP_2CPO | 2.43 | 1.78 |
| XNBR_0.2DCP | No tearing | |
| XNBR_2CPO | No tearing | |
| XNBR_0.2DCP_2CPO | No tearing | |
| XNBR_0.4DCP | 7.71 | 5.75 |
| XNBR_0.4DCP_2CPO | 7.89 | 6.75 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chudzik, J.; Bieliński, D.M.; Bratychak, M.; Demchuk, Y.; Astakhova, O.; Jędrzejczyk, M.; Celichowski, G. Influence of Modified Epoxy Resins on Peroxide Curing, Mechanical Properties and Adhesion of SBR, NBR and XNBR to Silver Wires—Part II: Application of Carboxy-Containing Peroxy Oligomer (CPO). Materials 2021, 14, 1285. https://doi.org/10.3390/ma14051285
Chudzik J, Bieliński DM, Bratychak M, Demchuk Y, Astakhova O, Jędrzejczyk M, Celichowski G. Influence of Modified Epoxy Resins on Peroxide Curing, Mechanical Properties and Adhesion of SBR, NBR and XNBR to Silver Wires—Part II: Application of Carboxy-Containing Peroxy Oligomer (CPO). Materials. 2021; 14(5):1285. https://doi.org/10.3390/ma14051285
Chicago/Turabian StyleChudzik, Joanna, Dariusz M. Bieliński, Michael Bratychak, Yuriy Demchuk, Olena Astakhova, Marcin Jędrzejczyk, and Grzegorz Celichowski. 2021. "Influence of Modified Epoxy Resins on Peroxide Curing, Mechanical Properties and Adhesion of SBR, NBR and XNBR to Silver Wires—Part II: Application of Carboxy-Containing Peroxy Oligomer (CPO)" Materials 14, no. 5: 1285. https://doi.org/10.3390/ma14051285
APA StyleChudzik, J., Bieliński, D. M., Bratychak, M., Demchuk, Y., Astakhova, O., Jędrzejczyk, M., & Celichowski, G. (2021). Influence of Modified Epoxy Resins on Peroxide Curing, Mechanical Properties and Adhesion of SBR, NBR and XNBR to Silver Wires—Part II: Application of Carboxy-Containing Peroxy Oligomer (CPO). Materials, 14(5), 1285. https://doi.org/10.3390/ma14051285









