Synthesis of Arylene Ether-Type Hyperbranched Poly(triphenylamine) for Lithium Battery Cathodes
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Material Characterization
2.3. Synthesis of the AB2 Type Monomer
2.3.1. Bis(4-methoxyphenyl)amine (1)
2.3.2. Bis(4-methoxyphenyl)-(4-nitro-3-trifluoromethylphenyl)amine (2)
2.4. Synthesis of the A2B Type Monomer
(4-Methoxyphenyl)-bis(4-nitro-3-trifluoromethylphenyl)amine (3)
2.5. Deprotection of Methoxy Groups
2.5.1. Bis(4-hydroxyphenyl)-(4-nitro-3-trifluoromethylphenyl)amine (4)
2.5.2. 4-(Bis(4-nitro-3-trifluoromethylphenyl)amino)phenol (5)
2.6. Potassium Phenoxides Formation of Monomer
2.7. Polymerization
2.8. Electrochemical Characterization
3. Results
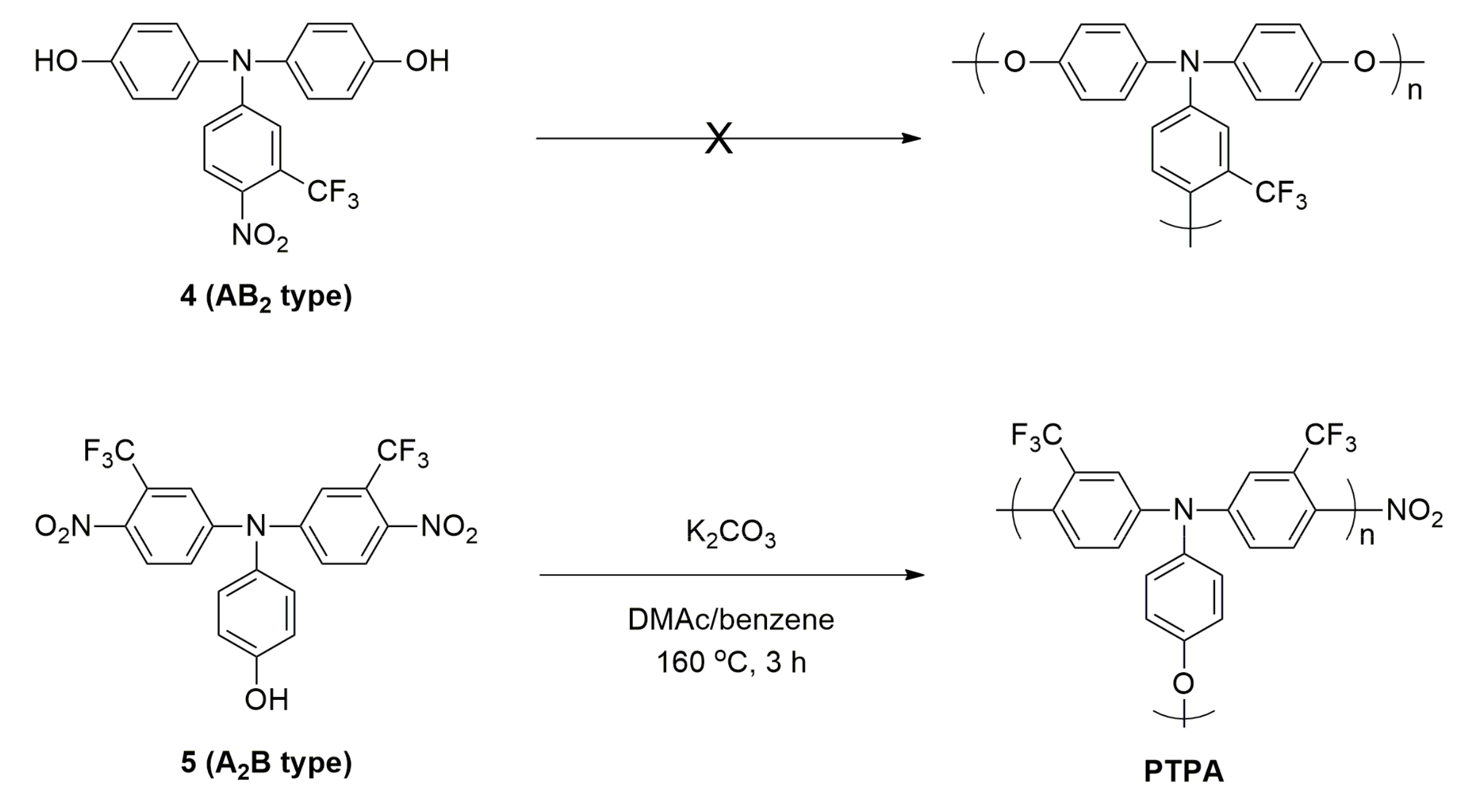
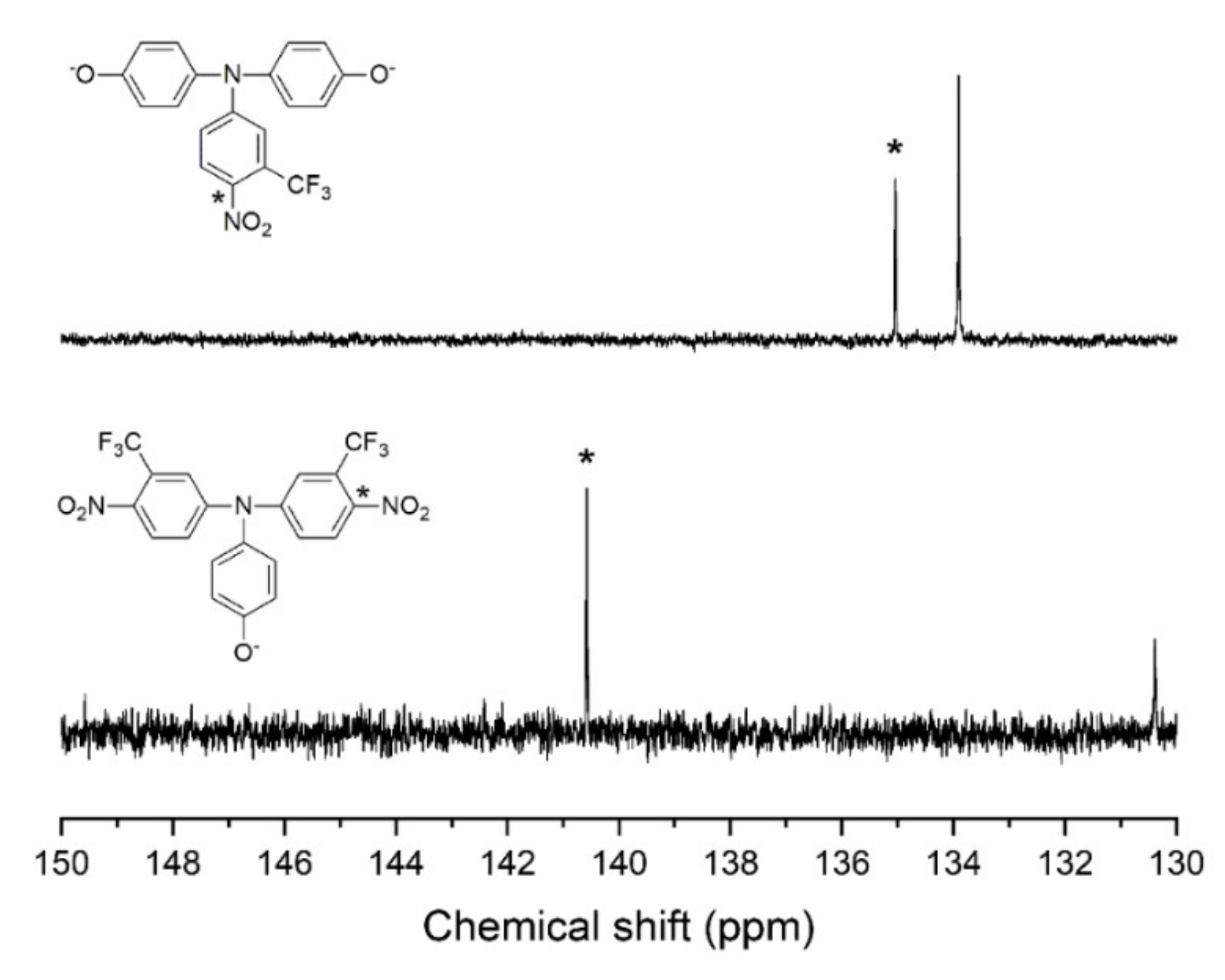
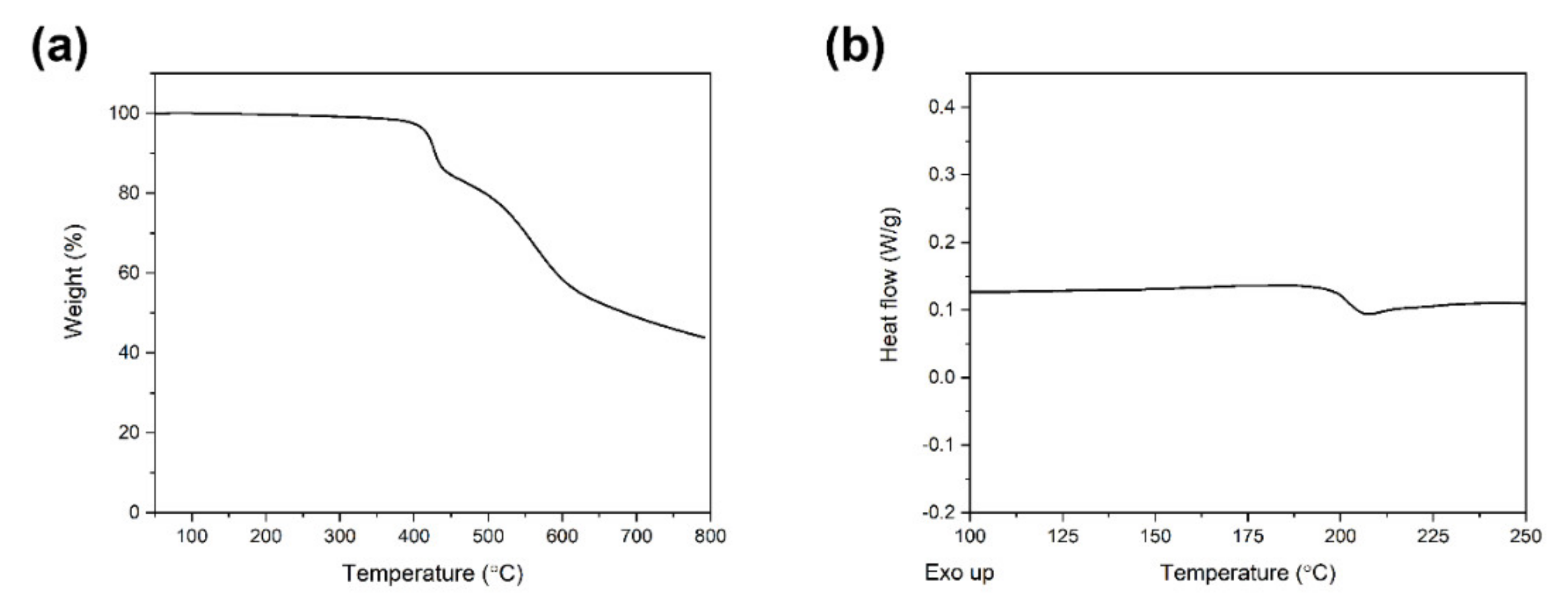
3.1. Synthesis and Characterization
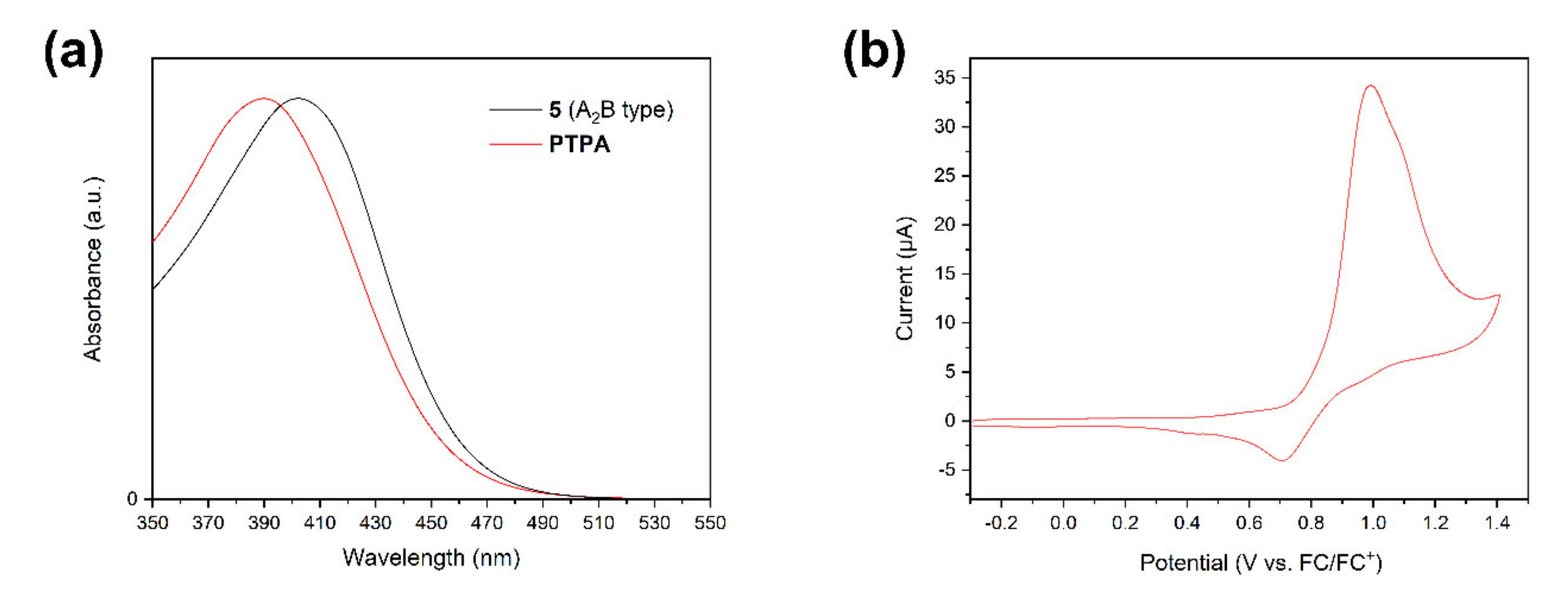
3.2. Electrochemical Investigation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Facchetti, A. π-Conjugated Polymers for Organic Electronics and Photovoltaic Cell Applications. Chem. Mater. 2011, 23, 733–758. [Google Scholar] [CrossRef]
- Ibanez, J.G.; Rincón, M.E.; Gutierrez-Granados, S.; Chahma, M.; Jaramillo-Quintero, O.A.; Frontana-Uribe, B.A. Conducting Polymers in the Fields of Energy, Environmental Remediation, and Chemical–Chiral Sensors. Chem. Rev. 2018, 118, 4731–4816. [Google Scholar] [CrossRef]
- Shirota, Y.; Kageyama, H. Charge Carrier Transporting Molecular Materials and Their Applications in Devices. Chem. Rev. 2007, 107, 953–1010. [Google Scholar] [CrossRef]
- Gibson, H.W. Control of electrical properties of polymers by chemical modification. Polymer 1984, 25, 3–27. [Google Scholar] [CrossRef]
- Guo, X.; Baumgarten, M.; Müllen, K. Designing π-conjugated polymers for organic electronics. Prog. Polym. Sci. 2013, 38, 1832–1908. [Google Scholar] [CrossRef]
- Kaloni, T.P.; Schreckenbach, G.; Freund, M.S. Band gap modulation in polythiophene and polypyrrole-based systems. Sci. Rep. 2016, 6, 36554. [Google Scholar] [CrossRef]
- Jia, X.; Ge, Y.; Shao, L.; Wang, C.; Wallace, G.G. Tunable Conducting Polymers: Toward Sustainable and Versatile Batteries. ACS Sustain. Chem. Eng. 2019, 7, 14321–14340. [Google Scholar] [CrossRef]
- Rittmeyer, S.P.; Groß, A. Structural and electronic properties of oligo- and polythiophenes modified by substituents. Beilstein J. Nanotechnol. 2012, 3, 909–919. [Google Scholar] [CrossRef]
- Kumar, D.; Sharma, R.C. Advances in conductive polymers. Eur. Polym. J. 1998, 34, 1053–1060. [Google Scholar] [CrossRef]
- Meng, Q.; Cai, K.; Chen, Y.; Chen, L. Research progress on conducting polymer based supercapacitor electrode materials. Nano Energy 2017, 36, 268–285. [Google Scholar] [CrossRef]
- Li, Z.; Gong, L. Research Progress on Applications of Polyaniline (PANI) for Electrochemical Energy Storage and Conversion. Materials 2020, 13, 548. [Google Scholar] [CrossRef]
- Bhosale, M.E.; Chae, S.; Kim, J.M.; Choi, J.-Y. Organic small molecules and polymers as an electrode material for rechargeable lithium ion batteries. J. Mater. Chem. A 2018, 6, 19885–19911. [Google Scholar] [CrossRef]
- Muench, S.; Wild, A.; Friebe, C.; Häupler, B.; Janoschka, T.; Schubert, U.S. Polymer-Based Organic Batteries. Chem. Rev. 2016, 116, 9438–9484. [Google Scholar] [CrossRef]
- Hong, X.; Liu, Y.; Li, Y.; Wang, X.; Fu, J.; Wang, X. Application Progress of Polyaniline, Polypyrrole and Polythiophene in Lithium-Sulfur Batteries. Polymers 2020, 12, 331. [Google Scholar] [CrossRef]
- Quijada, C. Special Issue: Conductive Polymers: Materials and Applications. Materials 2020, 13, 2344. [Google Scholar] [CrossRef] [PubMed]
- Kuo, C.-W.; Chang, J.-C.; Huang, Y.-T.; Chang, J.-K.; Lee, L.-T.; Wu, T.-Y. Applications of Copolymers Consisting of 2,6-di(9H-carbazol-9-yl)pyridine and 3,6-di(2-thienyl)carbazole Units as Electrodes in Electrochromic Devices. Materials 2019, 12, 1251. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Du, Y.; Wang, Q.; Xu, J.; Zhou, Y.; Bao, J.; Shen, J.; Zhou, X. A Yolk–Shell-Structured FePO4 Cathode for High-Rate and Long-Cycling Sodium-Ion Batteries. Angew. Chem. Int. Ed. 2020, 59, 17504–17510. [Google Scholar] [CrossRef]
- Yue, L.; Liang, J.; Wu, Z.; Zhong, B.; Luo, Y.; Liu, Q.; Li, T.; Kong, Q.; Liu, Y.; Asiri, A.M.; et al. Progress and perspective of metal phosphide/carbon heterostructure anodes for rechargeable ion batteries. J. Mater. Chem. A 2021, 9, 11879–11907. [Google Scholar] [CrossRef]
- Wijeratne, K.; Ail, U.; Brooke, R.; Vagin, M.; Liu, X.; Fahlman, M.; Crispin, X. Bulk electronic transport impacts on electron transfer at conducting polymer electrode–electrolyte interfaces. Proc. Natl. Acad. Sci. USA 2018, 115, 11899–11904. [Google Scholar] [CrossRef]
- Xie, J.; Zhang, Q. Recent progress in rechargeable lithium batteries with organic materials as promising electrodes. J. Mater. Chem. A 2016, 4, 7091–7106. [Google Scholar] [CrossRef]
- Zhao, W.; Gao, L.; Yue, L.; Wang, X.; Liu, Q.; Luo, Y.; Li, T.; Shi, X.; Asiri, A.M.; Sun, X. Constructing a hollow microflower-like ZnS/CuS@C heterojunction as an effective ion-transport booster for an ultrastable and high-rate sodium storage anode. J. Mater. Chem. A 2021, 9, 6402–6412. [Google Scholar] [CrossRef]
- Janoschka, T.; Hager, M.D.; Schubert, U.S. Powering up the Future: Radical Polymers for Battery Applications. Adv. Mater. 2012, 24, 6397–6409. [Google Scholar] [CrossRef]
- Song, Z.; Zhou, H. Towards sustainable and versatile energy storage devices: An overview of organic electrode materials. Energy Environ. Sci. 2013, 6, 2280–2301. [Google Scholar] [CrossRef]
- Thelakkat, M. Star-Shaped, Dendrimeric and Polymeric Triarylamines as Photoconductors and Hole Transport Materials for Electro-Optical Applications. Macromol. Mater. Eng. 2002, 287, 442–461. [Google Scholar] [CrossRef]
- Iwan, A.; Sek, D. Polymers with triphenylamine units: Photonic and electroactive materials. Prog. Polym. Sci. 2011, 36, 1277–1325. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, B.; Yu, H.; Li, G.; Bao, Y. Synthetic control of network topology and pore structure in microporous polyimides based on triangular triphenylbenzene and triphenylamine units. Soft Matter 2011, 7, 5723–5730. [Google Scholar] [CrossRef]
- Li, G.; Wang, Z. Microporous Polyimides with Uniform Pores for Adsorption and Separation of CO2 Gas and Organic Vapors. Macromolecules 2013, 46, 3058–3066. [Google Scholar] [CrossRef]
- Hu, Y.-C.; Chen, C.-J.; Yen, H.-J.; Lin, K.-Y.; Yeh, J.-M.; Chen, W.-C.; Liou, G.-S. Novel triphenylamine-containing ambipolar polyimides with pendant anthraquinone moiety for polymeric memory device, electrochromic and gas separation applications. J. Mater. Chem. 2012, 22, 20394–20402. [Google Scholar] [CrossRef]
- Bera, D.; Padmanabhan, V.; Banerjee, S. Highly Gas Permeable Polyamides Based on Substituted Triphenylamine. Macromolecules 2015, 48, 4541–4554. [Google Scholar] [CrossRef]
- Liang, Y.; Tao, Z.; Chen, J. Organic Electrode Materials for Rechargeable Lithium Batteries. Adv. Energy Mater. 2012, 2, 742–769. [Google Scholar] [CrossRef]
- Feng, J.K.; Cao, Y.L.; Ai, X.P.; Yang, H.X. Polytriphenylamine: A high power and high capacity cathode material for rechargeable lithium batteries. J. Power Sources 2008, 177, 199–204. [Google Scholar] [CrossRef]
- Yamamoto, K.; Suemasa, D.; Masuda, K.; Aita, K.; Endo, T. Hyperbranched Triphenylamine Polymer for UltraFast Battery Cathode. ACS Appl. Mater. Interfaces 2018, 10, 6346–6353. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Yang, X.; Ren, W.; Wang, Y.; Su, F.; Jiang, J.X. Microporous organic polymer-based lithium ion batteries with improved rate performance and energy density. J. Power Sources 2016, 317, 49–56. [Google Scholar] [CrossRef]
- Xu, T.; Xiong, J.; Du, X.; Zhang, Y.; Song, S.; Xiong, C.; Dong, L. Polytriphenylamine Derivative and Carbon Nanotubes as Cathode Materials for High-Performance Polymer-Based Batteries. J. Phys. Chem. C 2018, 122, 20057–20063. [Google Scholar] [CrossRef]
- Friebe, C.; Lex-Balducci, A.; Schubert, U.S. Sustainable Energy Storage: Recent Trends and Developments toward Fully Organic Batteries. ChemSusChem 2019, 12, 4093–4115. [Google Scholar] [CrossRef]
- Su, C.; Yang, F.; Ji, L.; Xu, L.; Zhang, C. Polytriphenylamine derivative with high free radical density as the novel organic cathode for lithium ion batteries. J. Mater. Chem. A 2014, 2, 20083–20088. [Google Scholar] [CrossRef]
- Su, C.; He, H.; Xu, L.; Zhao, K.; Zheng, C.; Zhang, C. A mesoporous conjugated polymer based on a high free radical density polytriphenylamine derivative: Its preparation and electrochemical performance as a cathode material for Li-ion batteries. J. Mater. Chem. A 2017, 5, 2701–2709. [Google Scholar] [CrossRef]
- Xiong, J.; Wei, Z.; Xu, T.; Zhang, Y.; Xiong, C.; Dong, L. Polytriphenylamine derivative with enhanced electrochemical performance as the organic cathode material for rechargeable batteries. Polymer 2017, 130, 135–142. [Google Scholar] [CrossRef]
- Su, C.; Zhu, X.; Xu, L.; Zhou, N.; He, H.; Zhang, C. Organic polytriphenylamine derivative-based cathode with tailored potential and its electrochemical performances. Electrochim. Acta 2016, 196, 440–449. [Google Scholar] [CrossRef]
- Zhu, L.; Cao, X. p-Dopable Poly(4-nitro)triphenylamine as Cathode Material with High Discharge Voltage for Lithium Ion Batteries. Int. J. Electrochem. Sci. 2015, 10, 4359–4365. [Google Scholar]
- Zhu, L.; Cao, X. p-Dopable poly(4-cyano)triphenylamine: A high voltage organic cathode for lithium ion batteries. Mater. Lett. 2015, 150, 16–19. [Google Scholar] [CrossRef]
- Xiang, J.; Sato, K.; Tokue, H.; Oyaizu, K.; Ho, C.-L.; Nishide, H.; Wong, W.-Y.; Wei, M. Synthesis and Charge–Discharge Properties of Organometallic Co-polymers of Ferrocene and Triphen-ylamine as Cathode Active Materials for Organic-Battery Applications. Eur. J. Inorg. Chem. 2016, 2016, 1030–1035. [Google Scholar] [CrossRef]
- Su, C.; Yang, F.; Xu, L.; Zhu, X.; He, H.; Zhang, C. Radical Polymer Containing a Polytriphenylamine Backbone: Its Synthesis and Electrochemical Performance as the Cathode of Lithium-Ion Batteries. Chempluschem 2015, 80, 606–611. [Google Scholar] [CrossRef]
- Gao, C.; Yan, D. Hyperbranched polymers: From synthesis to applications. Prog. Polym. Sci. 2004, 29, 183–275. [Google Scholar] [CrossRef]
- Wei, Y.; Li, X.; Xu, Z.; Sun, H.; Zheng, Y.; Peng, L.; Liu, Z.; Gao, C.; Gao, M. Solution processible hyperbranched inverse-vulcanized polymers as new cathode materials in Li–S batteries. Polym. Chem. 2015, 6, 973–982. [Google Scholar] [CrossRef]
- Zeigler, D.F.; Candelaria, S.L.; Mazzio, K.A.; Martin, T.R.; Uchaker, E.; Suraru, S.-L.; Kang, L.J.; Cao, G.; Luscombe, C.K. N-Type Hyperbranched Polymers for Supercapacitor Cathodes with Variable Porosity and Excellent Electrochemical Stability. Macromolecules 2015, 48, 5196–5203. [Google Scholar] [CrossRef]
- Bhat, S.I.; Ahmadi, Y.; Ahmad, S. Recent Advances in Structural Modifications of Hyperbranched Polymers and Their Applications. Ind. Eng. Chem. Res. 2018, 57, 10754–10785. [Google Scholar] [CrossRef]
- Cotter, R.J. Engineering Plastics: A Handbook of Polyarylethers; Gordon and Breach Publisher: Amsterdam, The Netherlands, 1995; ISBN 2884491120. [Google Scholar]
- Lee, M.S.; Kim, S.Y. Synthesis of Poly(arylene ether)s Containing Triphenylamine Units via Nitro Displacement Reaction. Macromolecules 2005, 38, 5844–5845. [Google Scholar] [CrossRef]
- Lee, J.; Heo, J.; Park, C.; Kim, B.-K.; Kwak, J.; Seo, M.; Kim, S.Y. Synthesis of triarylamine-containing poly(arylene ether)s by nucleophilic aromatic substitution reaction. J. Polym. Sci. Part A Polym. Chem. 2014, 52, 2692–2702. [Google Scholar] [CrossRef]
- Carter, K.R. Aryl fluoride monomers in nucleophilic aromatic substitution polymerization: Evaluation of monomer reactivity by 19F NMR spectroscopy. Macromolecules 1995, 28, 6462–6470. [Google Scholar] [CrossRef]
- Matsumoto, K.; Komuro, H.; Kai, T.; Jikei, M. Synthesis of poly(ether sulfone)s by self-polycondensation of AB-type monomers. Polym. J. 2013, 45, 909–914. [Google Scholar] [CrossRef]
- Williams, F.J.; Donahue, P.E. Reactions of phenoxides with nitro- and halo-substituted phthalimides. J. Org. Chem. 1977, 42, 3414–3419. [Google Scholar] [CrossRef]
- Markezich, R.L.; Zamek, O.S. Reactions of fluoride and nitrite ions with 4-nitrophthalimides. J. Org. Chem. 1977, 42, 3431–3434. [Google Scholar] [CrossRef]
- White, D.M.; Takekoshi, T.; Williams, F.J.; Relles, H.M.; Donahue, P.E.; Klopfer, H.J.; Loucks, G.R.; Manello, J.S.; Matthews, R.O.; Schluenz, R.W. Polyetherimides via nitro-displacement polymerization: Monomer synthesis and 13C-NMR analysis of monomers and polymers. J. Polym. Sci. Polym. Chem. Ed. 1981, 19, 1635–1658. [Google Scholar] [CrossRef]
- Takekoshi, T. Synthesis of High Performance Aromatic Polymers via Nucleophilic Nitro Displacement Reaction. Polym. J. 1987, 19, 191–202. [Google Scholar] [CrossRef][Green Version]
- Lee, H.S.; Kim, J.H. Measurement of Physical Properties of Conducting Polymers. Polym. Sci. Technol. 2007, 18, 488–495. [Google Scholar]
- Peng, Z.; Yi, X.; Liu, Z.; Shang, J.; Wang, D. Triphenylamine-Based Metal–Organic Frameworks as Cathode Materials in Lithium-Ion Batteries with Coexistence of Redox Active Sites, High Working Voltage, and High Rate Stability. ACS Appl. Mater. Interfaces 2016, 8, 14578–14585. [Google Scholar] [CrossRef]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kang, I.; Lee, T.; Yoon, Y.R.; Kim, J.W.; Kim, B.-K.; Lee, J.; Lee, J.H.; Kim, S.Y. Synthesis of Arylene Ether-Type Hyperbranched Poly(triphenylamine) for Lithium Battery Cathodes. Materials 2021, 14, 7885. https://doi.org/10.3390/ma14247885
Kang I, Lee T, Yoon YR, Kim JW, Kim B-K, Lee J, Lee JH, Kim SY. Synthesis of Arylene Ether-Type Hyperbranched Poly(triphenylamine) for Lithium Battery Cathodes. Materials. 2021; 14(24):7885. https://doi.org/10.3390/ma14247885
Chicago/Turabian StyleKang, Inah, Taewoong Lee, Young Rok Yoon, Jee Woo Kim, Byung-Kwon Kim, Jinhee Lee, Jin Hong Lee, and Sang Youl Kim. 2021. "Synthesis of Arylene Ether-Type Hyperbranched Poly(triphenylamine) for Lithium Battery Cathodes" Materials 14, no. 24: 7885. https://doi.org/10.3390/ma14247885
APA StyleKang, I., Lee, T., Yoon, Y. R., Kim, J. W., Kim, B.-K., Lee, J., Lee, J. H., & Kim, S. Y. (2021). Synthesis of Arylene Ether-Type Hyperbranched Poly(triphenylamine) for Lithium Battery Cathodes. Materials, 14(24), 7885. https://doi.org/10.3390/ma14247885





