Utilisation of Carp Skin Post-Production Waste in Binary Films Based on Furcellaran and Chitosan to Obtain Packaging Materials for Storing Blueberries
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Preparation of Furcellaran-Chitosan Films with the Addition of Gelatin Hydrolysate
2.2.2. Film Characterisation
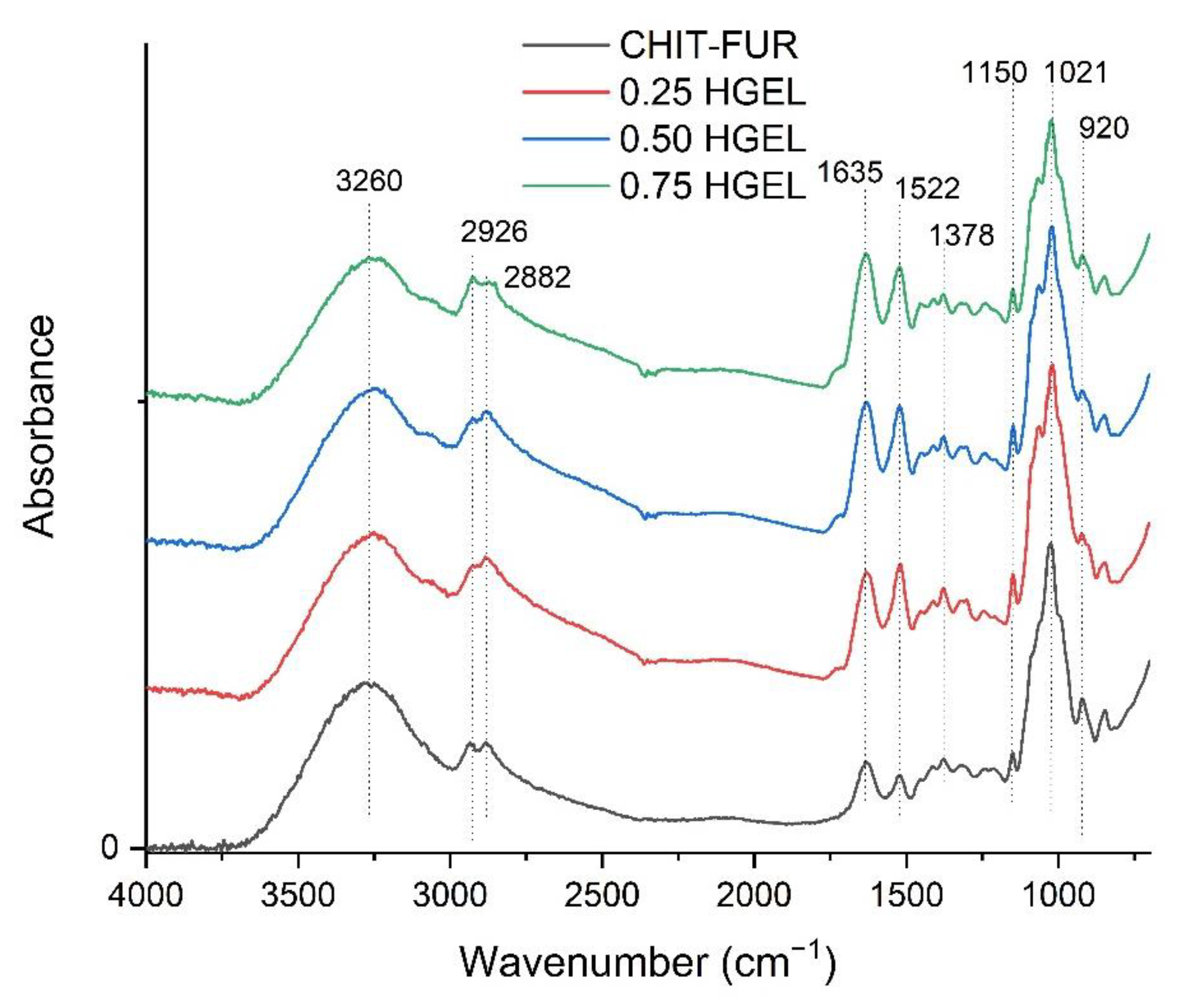
Fourier Transform Infrared Spectroscopy-Attenuated Total Reflection (FTIR-ATR)
Differential Scanning Calorimetry (DSC)
Surface Colour Measurement
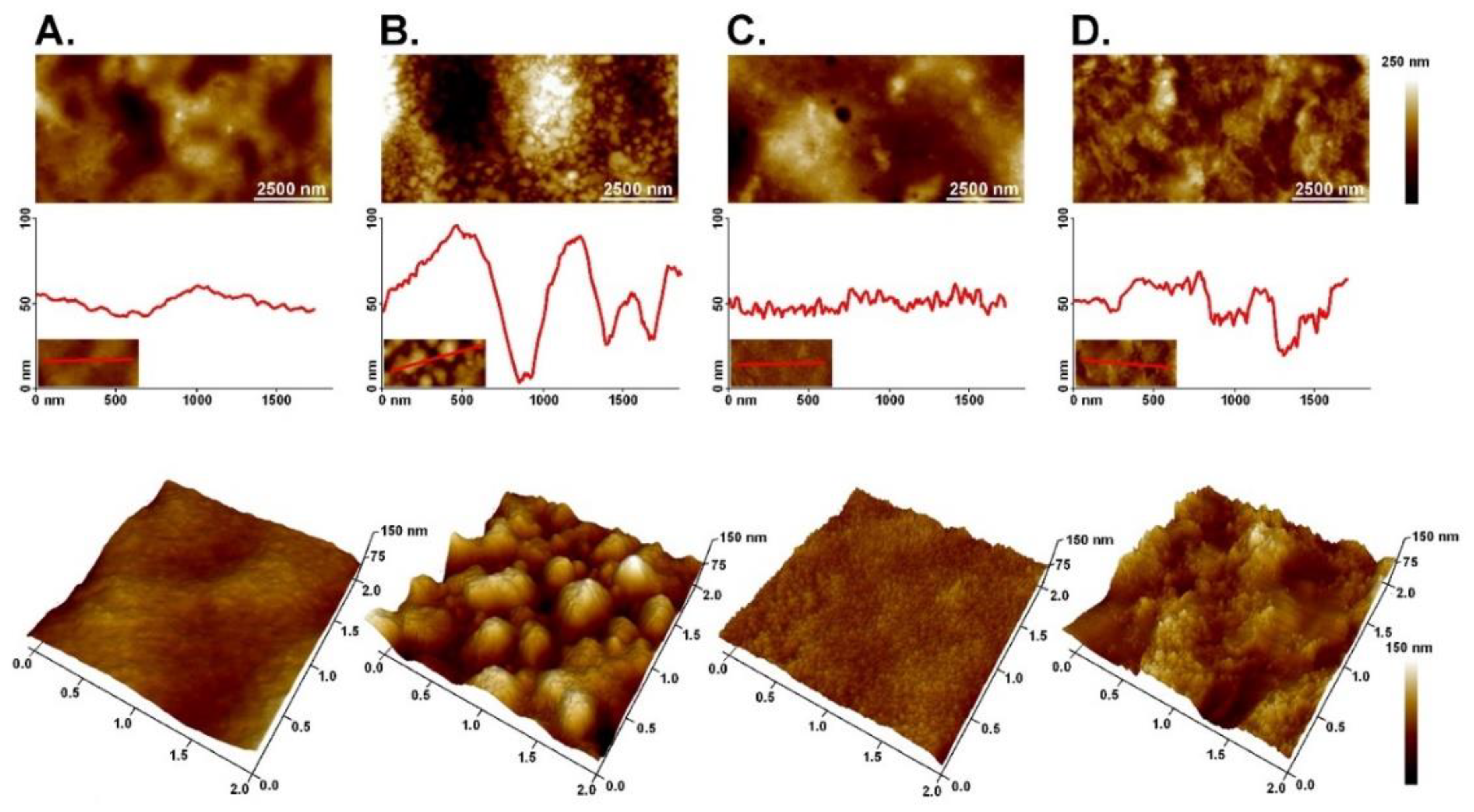
Atomic Force Microscopy (AFM)
Antioxidant Activity
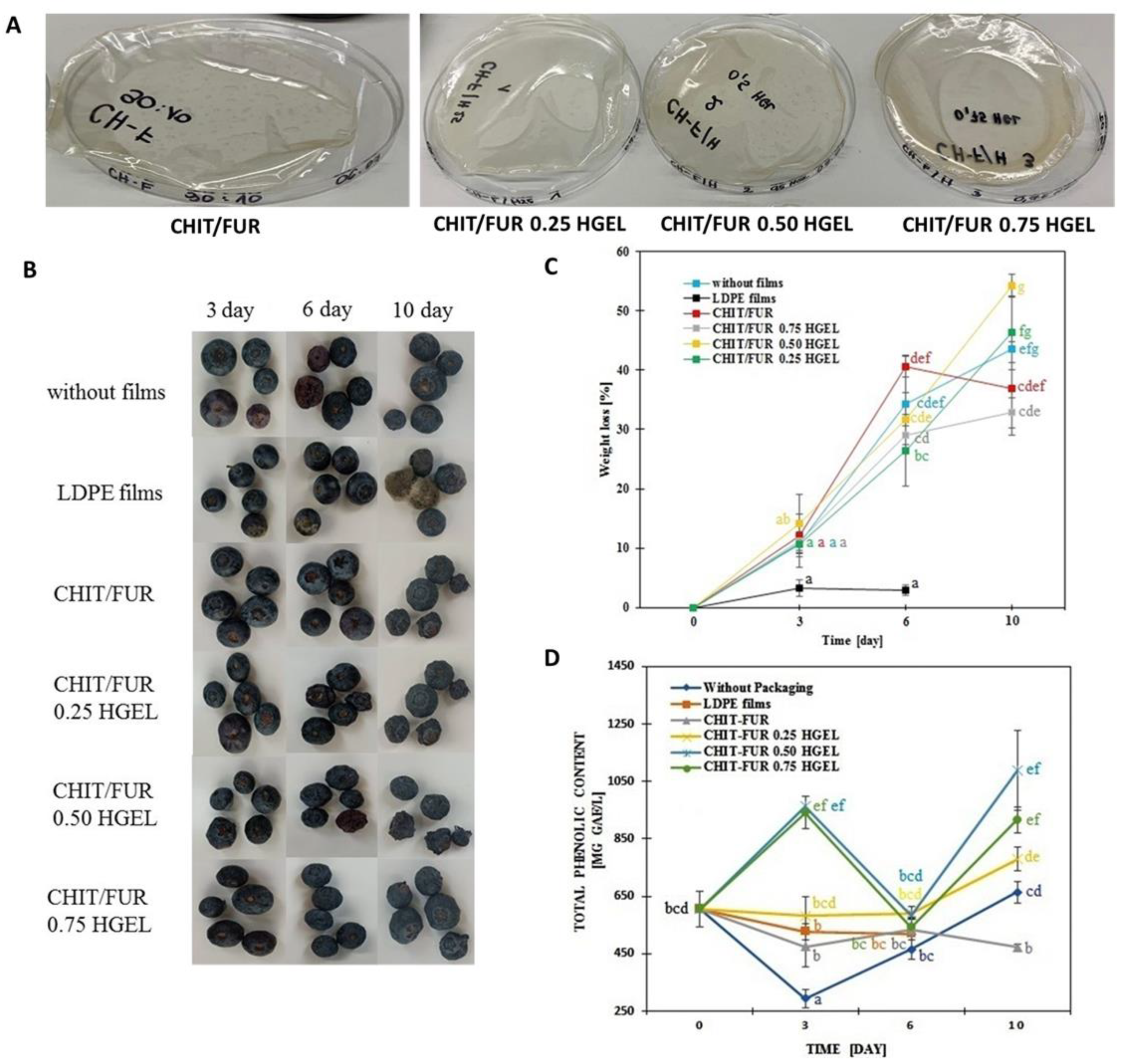
2.2.3. Evaluation of Blueberry Storage Quality
Blueberry Treatments
Weight Loss Ratio
Determination of Total Phenolic Content (TPC)
Colour Determination of Blueberries
2.2.4. Statistical Analysis
3. Results and Discussion
3.1. Characterisation of CHIT, FUR Films and Their Complex with HGEL
3.2. Thermal Properties of CHIT–FUR and CHIT–FUR/HGEL
3.3. Colour Properties of the Tested Films
3.4. Atomic Force Microscopy (AFM)
3.5. Antioxidant Properties
3.6. Application on Model Food—Blueberry
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ivonkovic, A.; Zeljko, K.; Talic, S.; Lasic, M. Biodegradable packaging in the food industry. J. Food Saf. Food Qual 2017, 68, 26–38. [Google Scholar]
- Zhong, Y.; Godwin, P.; Jin, Y.; Xiao, H. Biodegradable polymers and green-based antimicrobial packaging materials: A mini-review. Adv. Ind. Eng. Polym. Res. 2019, 3, 27–35. [Google Scholar] [CrossRef]
- Luangapai, F.; Peanparkdee, M.; Iwamoto, S. Biopolymer films for food industries: Properties, applications, and future aspects based on chitosan. Rev. Agric. Sci. 2019, 7, 59–67. [Google Scholar] [CrossRef] [Green Version]
- Cazón, P.; Velazquez, G.; Ramírez, J.A.; Vázquez, M. Polysaccharide-based films and coatings for food packaging: A review. Food Hydrocoll. 2016, 68, 136–148. [Google Scholar] [CrossRef]
- Garavand, F.; Rouhi, M.; Razavi, S.H.; Cacciotti, I.; Mohammadi, R. Improving the integrity of natural biopolymer films used in food packaging by crosslinking approach: A review. Int. J. Biol. Macromol. 2017, 104, 687–707. [Google Scholar] [CrossRef]
- Dharmalingam, K.; Anandalakshmi, R. Polysaccharide-Based Films for Food Packaging Applications. In Advances in Sustainable Polymers: Processing and Applications; Katiyar, V., Gupta, R., Ghosh, T., Eds.; Springer Singapore: Singapore, 2019; 9p. [Google Scholar]
- Tang, X.Z.; Kumar, P.; Alavi, S.; Sandeep, K.P. Recent Advances in Biopolymers and Biopolymer-Based Nanocomposites for Food Packaging Materials. Crit. Rev. Food Sci. Nutr. 2012, 52, 426–442. [Google Scholar] [CrossRef]
- Júnior, L.M.; Vieira, R.P.; Jamróz, E.; Anjos, C.A.R. Furcellaran: An innovative biopolymer in the production of films and coatings. Carbohydr. Polym. 2021, 252, 117221. [Google Scholar] [CrossRef] [PubMed]
- Imeson, A.P. 7—Carrageenan and furcellaran. In Handbook of Hydrocolloids, 2nd ed.; Phillips, G.O., Williams, P.A., Eds.; Woodhead Publishing: Amsterdam, The Netherlands, 2009; pp. 164–185. [Google Scholar] [CrossRef]
- Jamróz, E.; Juszczak, L.; Kucharek, M. Development of starch-furcellaran-gelatin films containing tea tree essential oil. J. Appl. Polym. Sci. 2018, 135. [Google Scholar] [CrossRef]
- Hosseinnejad, M.; Jafari, S.M. Evaluation of different factors affecting antimicrobial properties of chitosan. Int. J. Biol. Macromol. 2016, 85, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.I.; Ahmad, R.; Khan, M.S.; Kant, R.; Shahid, S.; Gautam, L.; Hasan, G.M.; Hassan, I. Chitin and its derivatives: Structural properties and biomedical applications. Int. J. Biol. Macromol. 2020, 164, 526–539. [Google Scholar] [CrossRef]
- Wang, H.; Qian, J.; Ding, F. Emerging Chitosan-Based Films for Food Packaging Applications. J. Agric. Food Chem. 2018, 66, 395–413. [Google Scholar] [CrossRef] [PubMed]
- Feng, X.; Wang, X.; Xing, W.; Yu, B.; Song, L.; Hu, Y. Simultaneous Reduction and Surface Functionalization of Graphene Oxide by Chitosan and Their Synergistic Reinforcing Effects in PVA Films. Ind. Eng. Chem. Res. 2013, 52, 12906–12914. [Google Scholar] [CrossRef]
- Youssef, A.M.; Abou-Yousef, H.; El-Sayed, S.; Kamel, S. Mechanical and antibacterial properties of novel high performance chitosan/nanocomposite films. Int. J. Biol. Macromol. 2015, 76, 25–32. [Google Scholar] [CrossRef]
- Jiang, Y.; Yin, H.; Zhou, X.; Wang, D.; Zhong, Y.; Xia, Q.; Deng, Y.; Zhao, Y. Antimicrobial, antioxidant and physical properties of chitosan film containing Akebia trifoliata (Thunb.) Koidz. peel extract/montmorillonite and its application. Food Chem. 2021, 361, 130111. [Google Scholar] [CrossRef] [PubMed]
- Tkaczewska, J.; Bukowski, M.; Mak, P. Identification of Antioxidant Peptides in Enzymatic Hydrolysates of Carp (Cyprinus Carpio) Skin Gelatin. Molecules 2018, 24, 97. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tkaczewska, J.; Morawska, M.; Kulawik, P.; Zając, M. Characterization of carp (Cyprinus carpio) skin gelatin extracted using different pretreatments method. Food Hydrocoll. 2018, 81, 169–179. [Google Scholar] [CrossRef]
- Jancikova, S.; Jamróz, E.; Kulawik, P.; Tkaczewska, J.; Dordevic, D. Furcellaran/gelatin hydrolysate/rosemary extract composite films as active and intelligent packaging materials. Int. J. Biol. Macromol. 2019, 131, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Jamróz, E.; Janik, M.; Juszczak, L.; Kruk, T.; Kulawik, P.; Szuwarzyński, M.; Kawecka, A.; Khachatryan, K. Composite biopolymer films based on a polyelectrolyte complex of furcellaran and chitosan. Carbohydr. Polym. 2021, 274, 118627. [Google Scholar] [CrossRef]
- Tkaczewska, J.; Jamróz, E.; Kulawik, P.; Morawska, M.; Szczurowska, K. Evaluation of the potential use of a carp (Cyprinus carpio) skin gelatine hydrolysate as an antioxidant component. Food Funct. 2019, 10, 1038–1048. [Google Scholar] [CrossRef]
- Katırcı, N.; Işık, N.; Güpür, Ç.; Guler, H.O.; Gursoy, O.; Yilmaz, Y. Differences in antioxidant activity, total phenolic and flavonoid contents of commercial and homemade tomato pastes. J. Saudi Soc. Agric. Sci. 2018, 19, 249–254. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, Z.; Li, Y.; Yang, Y.; Ju, X.; He, R. The preparation and physiochemical characterization of rapeseed protein hydrolysate-chitosan composite films. Food Chem. 2018, 272, 694–701. [Google Scholar] [CrossRef]
- Jamaludin, J.; Adam, F.; Rasid, R.A.; Hassan, Z. Thermal studies on Arabic gum—Carrageenan polysaccharides film. Chem. Eng. Res. Bull. 2017, 19, 80. [Google Scholar] [CrossRef] [Green Version]
- Burfield, D.R.; Loi, P.S.T.; Doi, Y.; Mejzík, J. DSC studies of tactic polypropylenes: The correlation of polymer stereochemistry with thermal properties. J. Appl. Polym. Sci. 1990, 41, 1095–1114. [Google Scholar] [CrossRef]
- Pereda, M.; Dufresne, A.; Aranguren, M.I.; Marcovich, N.E. Polyelectrolyte films based on chitosan/olive oil and reinforced with cellulose nanocrystals. Carbohydr. Polym. 2013, 101, 1018–1026. [Google Scholar] [CrossRef]
- Liu, X.; Xu, Y.; Zhan, X.; Xie, W.; Yang, X.; Cui, S.W.; Xia, W. Development and properties of new kojic acid and chitosan composite biodegradable films for active packaging materials. Int. J. Biol. Macromol. 2019, 144, 483–490. [Google Scholar] [CrossRef] [PubMed]
- Avena-Bustillos, R.; Olsen, C.; Olson, D.; Chiou, B.; Yee, E.; Bechtel, P.; McHugh, T. Water Vapor Permeability of Mammalian and Fish Gelatin Films. J. Food Sci. 2006, 71, E202–E207. [Google Scholar] [CrossRef]
- Marinello, F.; La Storia, A.; Mauriello, G.; Passeri, D. Atomic Force microscopy techniques to investigate activated food packaging materials. Trends Food Sci. Technol. 2018, 87, 84–93. [Google Scholar] [CrossRef]
- El-Hack, M.E.A.; El-Saadony, M.T.; Shafi, M.E.; Zabermawi, N.M.; Arif, M.; Batiha, G.E.; Khafaga, A.F.; El-Hakim, Y.M.A.; Al-Sagheer, A.A. Antimicrobial and antioxidant properties of chitosan and its derivatives and their applications: A review. Int. J. Biol. Macromol. 2020, 164, 2726–2744. [Google Scholar] [CrossRef] [PubMed]
- Jamróz, E.; Kulawik, P.; Tkaczewska, J.; Guzik, P.; Zając, M.; Juszczak, L.; Krzyściak, P.; Turek, K. The effects of active double-layered furcellaran/gelatin hydrolysate film system with Ala-Tyr peptide on fresh Atlantic mackerel stored at −18 °C. Food Chem. 2020, 338, 127867. [Google Scholar] [CrossRef]
- Rabea, E.I.; Badawy, M.E.-T.; Stevens, C.V.; Smagghe, G.; Steurbaut, W. Chitosan as antimicrobial agent: Applications and mode of action. Biomacromolecules 2003, 4, 1457–1465. [Google Scholar] [CrossRef] [PubMed]
- Zekrehiwot, A.; Yetenayet, B.T.; Ali, M.; Abebe, Z.; Tola, Y.B.; Mohammed, A. Effects of edible coating materials and stages of maturity at harvest on storage life and quality of tomato (Lycopersicon Esculentum Mill.) fruits. Afr. J. Agric. Res. 2017, 12, 550–565. [Google Scholar] [CrossRef] [Green Version]
- Yaman, O.; Bayoιndιrlι, L. Effects of an Edible Coating and Cold Storage on Shelf-life and Quality of Cherries. LWT Food Sci. Technol. 2002, 35, 146–150. [Google Scholar] [CrossRef]
- Duan, J.; Wu, R.; Strik, B.C.; Zhao, Y. Effect of edible coatings on the quality of fresh blueberries (Duke and Elliott) under commercial storage conditions. Postharvest Biol. Technol. 2011, 59, 71–79. [Google Scholar] [CrossRef]
- Lafarga, T.; Aguiló-Aguayo, I.; Bobo, G.; Chung, A.V.; Tiwari, B.K. Effect of storage on total phenolics, antioxidant capacity, and physicochemical properties of blueberry (Vaccinium corymbosum L.) jam. J. Food Process. Preserv. 2018, 42, e13666. [Google Scholar] [CrossRef]
- Campaniello, D.; Bevilacqua, A.; Sinigaglia, M.; Corbo, M.R. Chitosan: Antimicrobial activity and potential applications for preserving minimally processed strawberries. Food Microbiol. 2008, 25, 992–1000. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Yue, J.; Gong, X.; Qian, B.; Wang, H.; Deng, Y.; Zhao, Y. Blueberry leaf extracts incorporated chitosan coatings for preserving postharvest quality of fresh blueberries. Postharvest Biol. Technol. 2014, 92, 46–53. [Google Scholar] [CrossRef]
- Alvarez, M.V.; Ponce, A.G.; Moreira, M.R. Influence of polysaccharide-based edible coatings as carriers of prebiotic fibers on quality attributes of ready-to-eat fresh blueberries. J. Sci. Food Agric. 2017, 98, 2587–2597. [Google Scholar] [CrossRef]
- Bof, M.; Laurent, F.; Massolo, F.; Locaso, D.; Versino, F.; García, M. Bio-Packaging Material Impact on Blueberries Quality Attributes under Transport and Marketing Conditions. Polymers 2021, 13, 481. [Google Scholar] [CrossRef] [PubMed]
- Abugoch, L.; Tapia, C.; Plasencia, D.; Pastor, A.; Castro-Mandujano, O.; López, L.; Escalona, V.H. Shelf-life of fresh blueberries coated with quinoa protein/chitosan/sunflower oil edible film. J. Sci. Food Agric. 2016, 96, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Bambace, M.F.; Alvarez, M.V.; Moreira, M.D.R. Novel functional blueberries: Fructo-oligosaccharides and probiotic lactobacilli incorporated into alginate edible coatings. Food Res. Int. 2019, 122, 653–660. [Google Scholar] [CrossRef]
| Film type | Peak Temperature (Tm) (°C) | Enthalpy (ΔHm) (J/g) |
|---|---|---|
| CHIT–FUR | 190.3 a ± 8.6 | 154.80 c ± 4.2 |
| HGEL 0.25 | 198.0 a ± 11.6 | 122.57 a ± 2.5 |
| HGEL 0.50 | 202.4 a ± 5.7 | 129.23 ab ± 3.9 |
| HGEL 0.75 | 201.6 a ± 4.5 | 138.57 b ± 4.5 |
| Film Type | L | a* | b* | ΔE |
|---|---|---|---|---|
| 9:1 CHIT–FUR | 91.99 d ± 0.38 | −1.54 b ± 0.02 | 18.81 a ± 0.36 | - |
| HGEL 0.25 | 89.26 c ± 0.51 | −1.64 b ± 0.17 | 30.07 b ± 1.41 | 11.59 |
| HGEL 0.50 | 85.63 b ± 1.0 | −0.93 a ± 0.18 | 32.20 b ± 1.41 | 15.04 |
| HGEL 0.75 | 90.65 a ± 0.70 | −0.94 a ± 0.04 | 18.17 a ± 0.86 | 1.74 |
| Film Type | DPPH (%) | FRAP (μmol Trolox/g of Dried Film) |
|---|---|---|
| CHIT–FUR | 2.53 a ± 0.21 | 0.34 a ± 0.11 |
| HGEL 0.25 | 53.63 b ± 0.44 | 1.11 b ± 0.31 |
| HGEL 0.50 | 61.17 c ± 0.99 | 1.19 b ± 0.25 |
| HGEL 0.75 | 60.32 c ± 0.02 | 1.85 c ± 0.15 |
| Without Packaging | LDPE Films | CHIT–FUR | HGEL 0.25 | HGEL 0.50 | HGEL 0.75 | ||
|---|---|---|---|---|---|---|---|
| Day 0 | L | 32.65 ab ± 1.78 | |||||
| a* | −0.20 ± 0.14 | ||||||
| b* | −5.23 ab ± 0.79 | ||||||
| Day 3 | L | 32.48 ab ± 1.81 | 32.59 ab ± 1.32 | 32.01 ab ± 2.49 | 31.02 ab ± 1.74 | 32.04 ab ± 2.27 | 29.63 ab ± 2.45 |
| a* | 1.48 ± 0.95 | −0.09 ± 0.13 | 0.10 ± 0.44 | 1.01 ± 0.65 | 0.44 ± 0.19 | 0.48 ± 0.48 | |
| b* | −3.43 b ± 1.42 | −3.85 b ± 2.69 | −4.85 ab ± 1.48 | −3.51 b ± 0.70 | −4.21 ab ± 0.93 | −2.95 b ± 1.22 | |
| Day 6 | L | 34.12 b ± 1.35 | 31.04 ab ± 2.12 | 30.74 ab ± 2.03 | 30.22 ab ± 1.94 | 30.79 ab ± 2.65 | 32.62 ab ± 1.06 |
| a* | 1.94 ± 1.65 | 0.07 ± 0.63 | 0.01 ± 0.24 | 0.65 ± 0.60 | 1.17 ± 1.55 | 0.01 ± 0.33 | |
| b* | −5.62 ab ± 0.91 | −5.32 ab ± 1.30 | −5.81 ab ± 0.59 | −5.70 ab ± 0.54 | −5.00 ab ± 1.20 | −7.15 a ± 0.88 | |
| Day 10 | L | 32.76 ab ± 1.58 | 29.85 ab ± 1.23 | 28.06 a ± 3.14 | 29.06 ab ± 1.68 | 32.61 ab ± 1.75 | |
| a* | 0.25 ± 0.53 | −0.27 ± 0.04 | 0.12 ± 0.28 | 0.68 ± 1.06 | 0.06 ± 0.16 | ||
| b* | −4.50 ab ± 1.37 | −4.67 ab ± 0.55 | −3.31 b ± 1.39 | −3.48 b ± 1.45 | −4.50 ab ± 0.49 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janik, M.; Jamróz, E.; Tkaczewska, J.; Juszczak, L.; Kulawik, P.; Szuwarzyński, M.; Khachatryan, K.; Kopel, P. Utilisation of Carp Skin Post-Production Waste in Binary Films Based on Furcellaran and Chitosan to Obtain Packaging Materials for Storing Blueberries. Materials 2021, 14, 7848. https://doi.org/10.3390/ma14247848
Janik M, Jamróz E, Tkaczewska J, Juszczak L, Kulawik P, Szuwarzyński M, Khachatryan K, Kopel P. Utilisation of Carp Skin Post-Production Waste in Binary Films Based on Furcellaran and Chitosan to Obtain Packaging Materials for Storing Blueberries. Materials. 2021; 14(24):7848. https://doi.org/10.3390/ma14247848
Chicago/Turabian StyleJanik, Magdalena, Ewelina Jamróz, Joanna Tkaczewska, Lesław Juszczak, Piotr Kulawik, Michał Szuwarzyński, Karen Khachatryan, and Pavel Kopel. 2021. "Utilisation of Carp Skin Post-Production Waste in Binary Films Based on Furcellaran and Chitosan to Obtain Packaging Materials for Storing Blueberries" Materials 14, no. 24: 7848. https://doi.org/10.3390/ma14247848
APA StyleJanik, M., Jamróz, E., Tkaczewska, J., Juszczak, L., Kulawik, P., Szuwarzyński, M., Khachatryan, K., & Kopel, P. (2021). Utilisation of Carp Skin Post-Production Waste in Binary Films Based on Furcellaran and Chitosan to Obtain Packaging Materials for Storing Blueberries. Materials, 14(24), 7848. https://doi.org/10.3390/ma14247848









