Hydrophobic Thin Films from Sol–Gel Processing: A Critical Review
Abstract
:1. Introduction
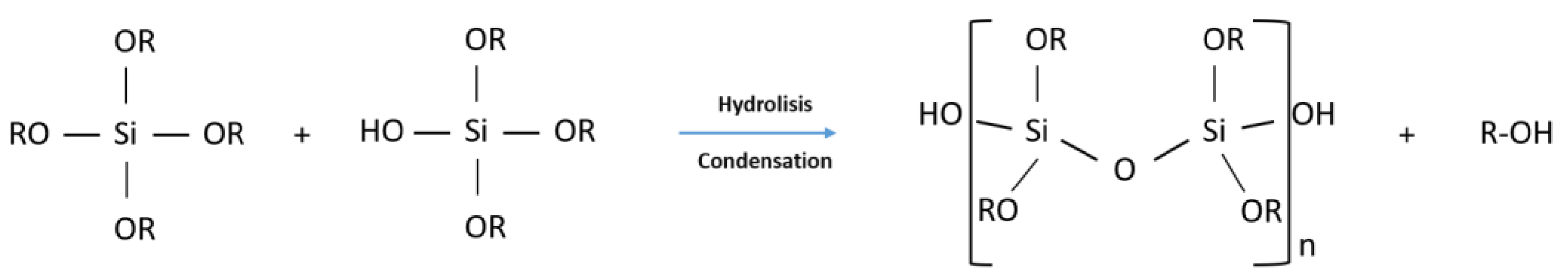
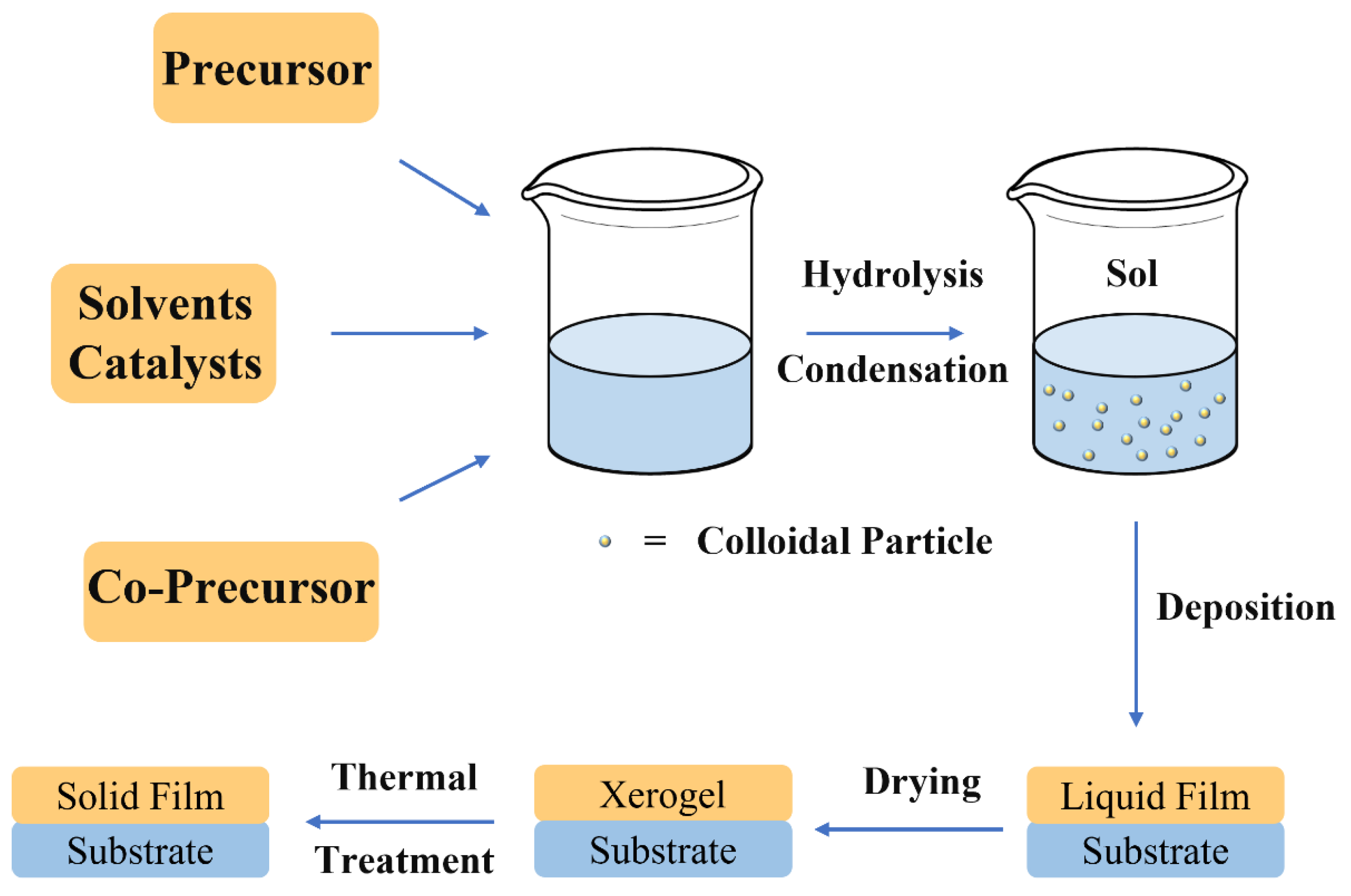
1.1. From Sol–Gel Chemistry to the Functional Coatings
- ➢
- First step: Sol formation. A sol is obtained by mixing the precursors in water, organic solvent (e.g., methanol or ethanol) with a proper catalyst;
- ➢
- Second step: Gel formation. The proceeds of the hydrolysis and condensation reactions allows the sol to gel transition with the formation of two continuous and interconnected phase, one liquid (the sol) and the other solid (the inorganic or hybrid network). This step is used only if a bulk must be obtained. In the case of a film deposition sol of controlled aging are employed. Aging governs the dimensions and structure of inorganic clusters in the precursor sols;
- ➢
- Third step: Film deposition. The precursor sols are used to deposited thin films on different substrates via a specific deposition method (i.e., spin or dip-coating);
- ➢
- Fourth step: Drying. After film deposition the residual solvent is removed through a drying process to obtain a xerogel;
- ➢
- Fifth step: Firing. The film is condensed using a controlled heat-treatment to increase its density and mechanical properties. This step must be carefully controlled to avoid cracking or delamination from the substrate.
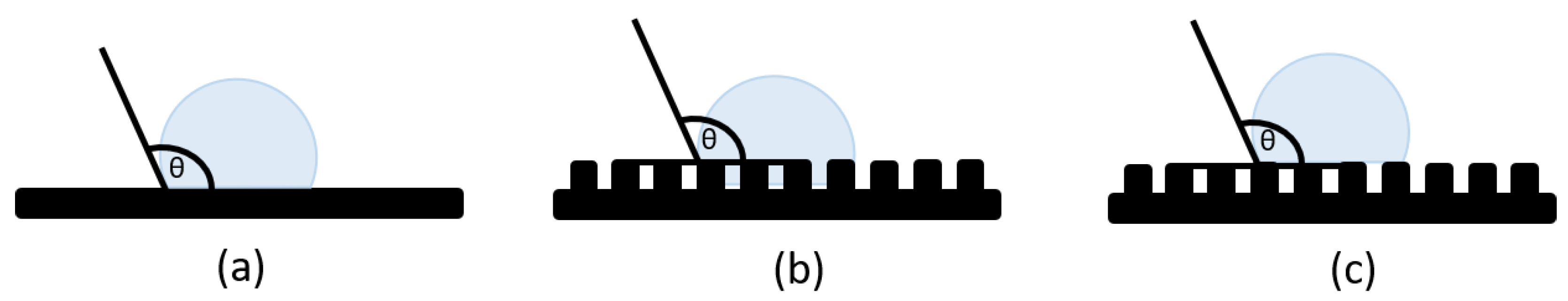
1.2. Properties Design
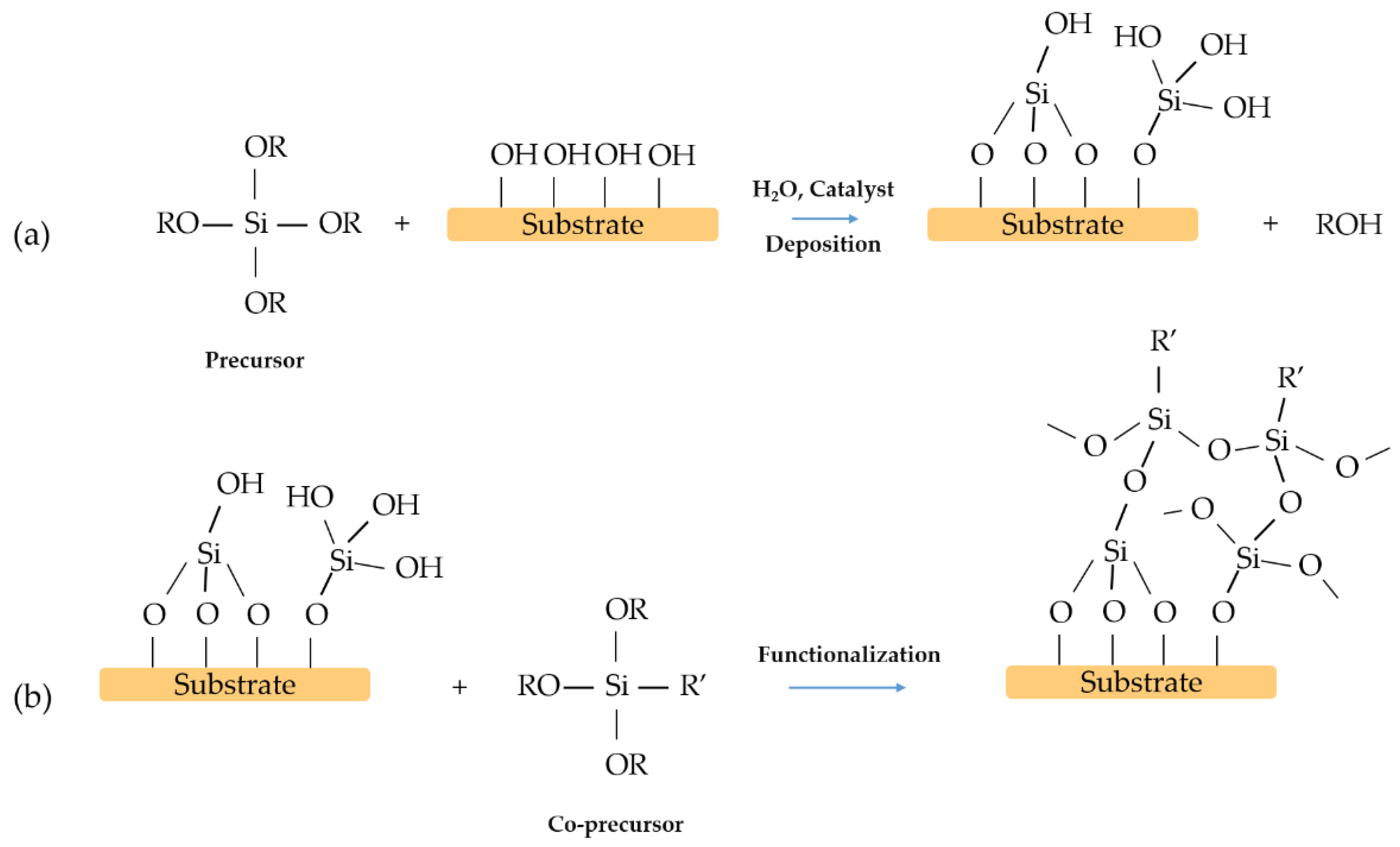
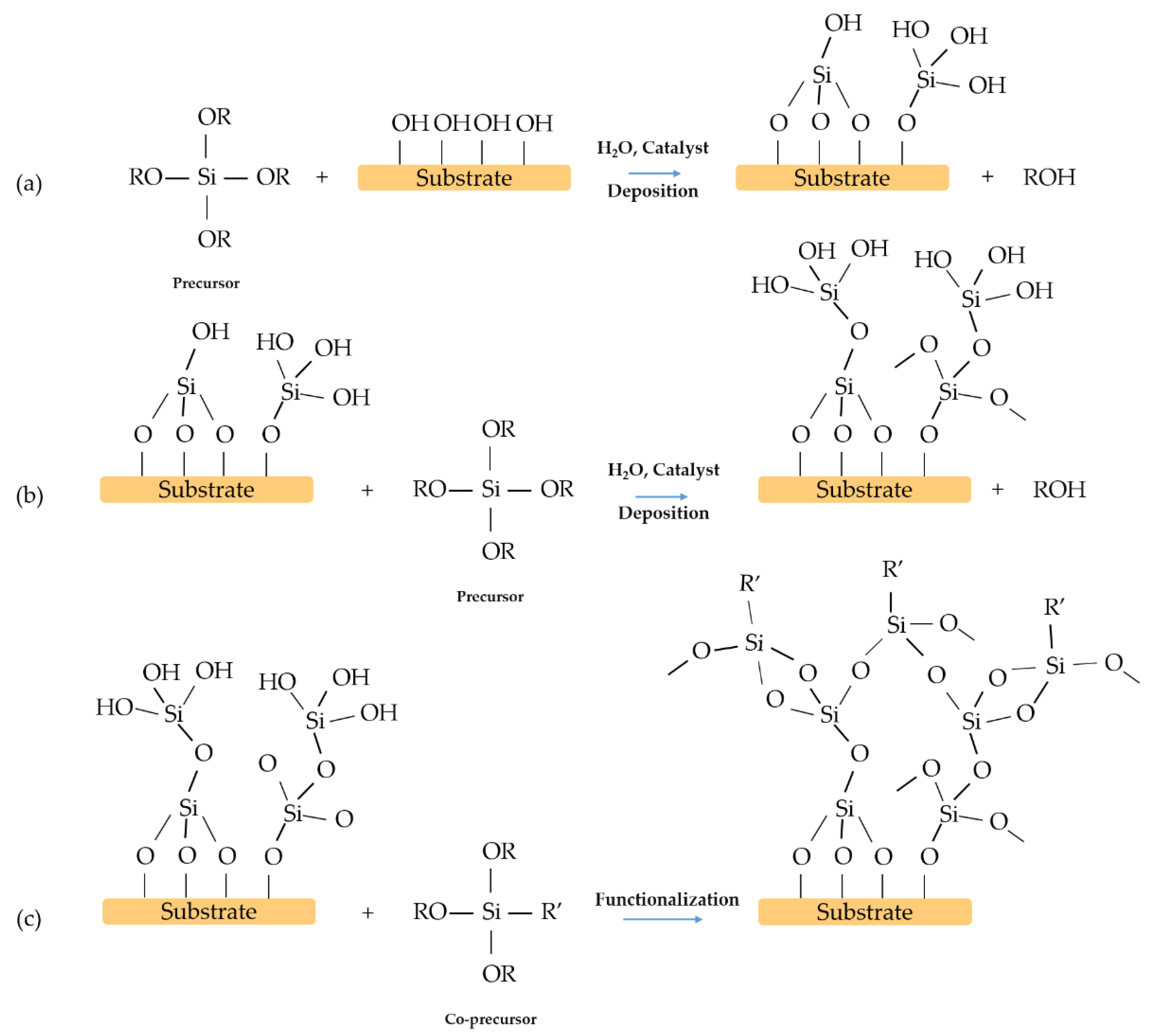
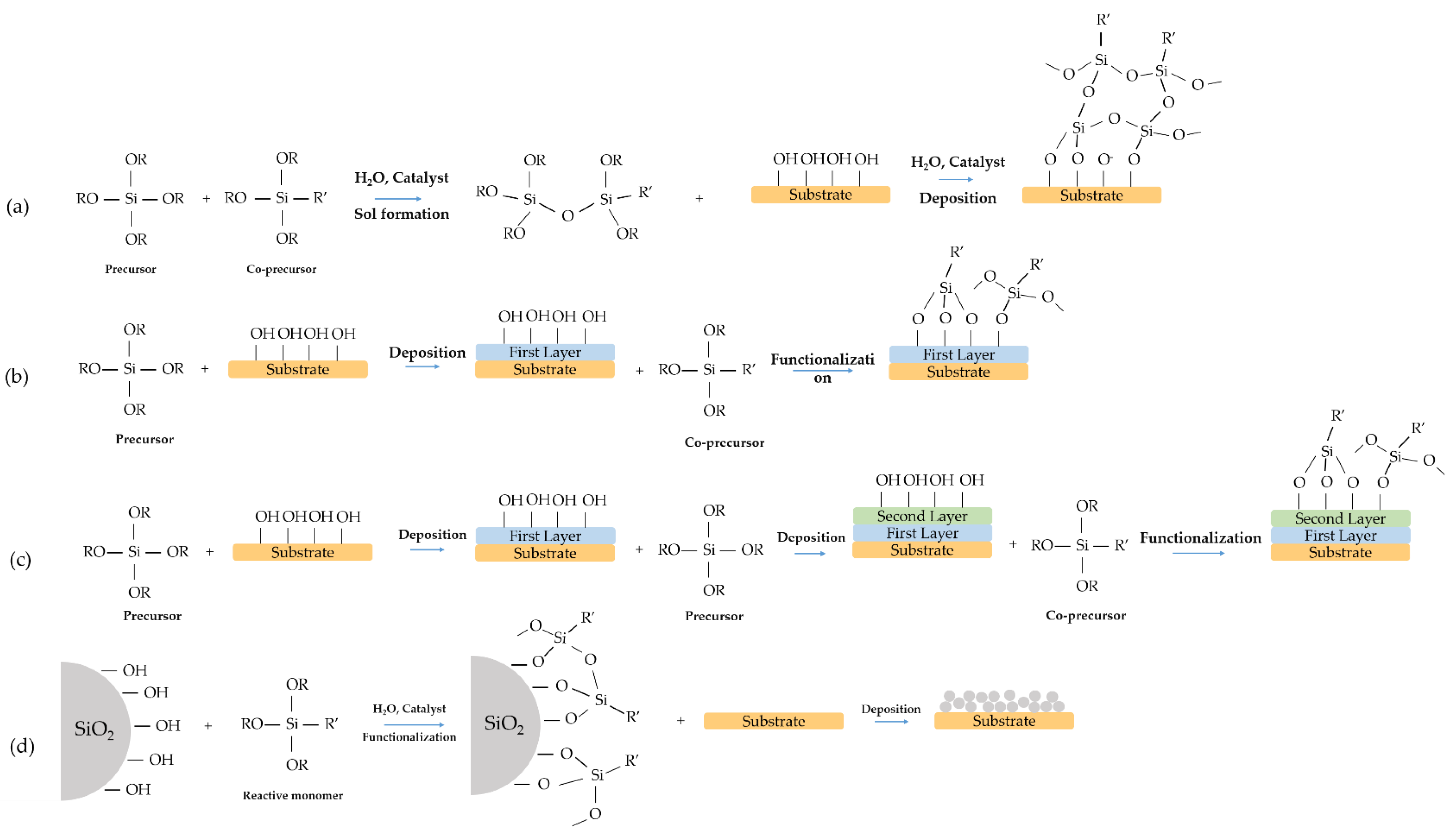
2. One-Step Surface Modification
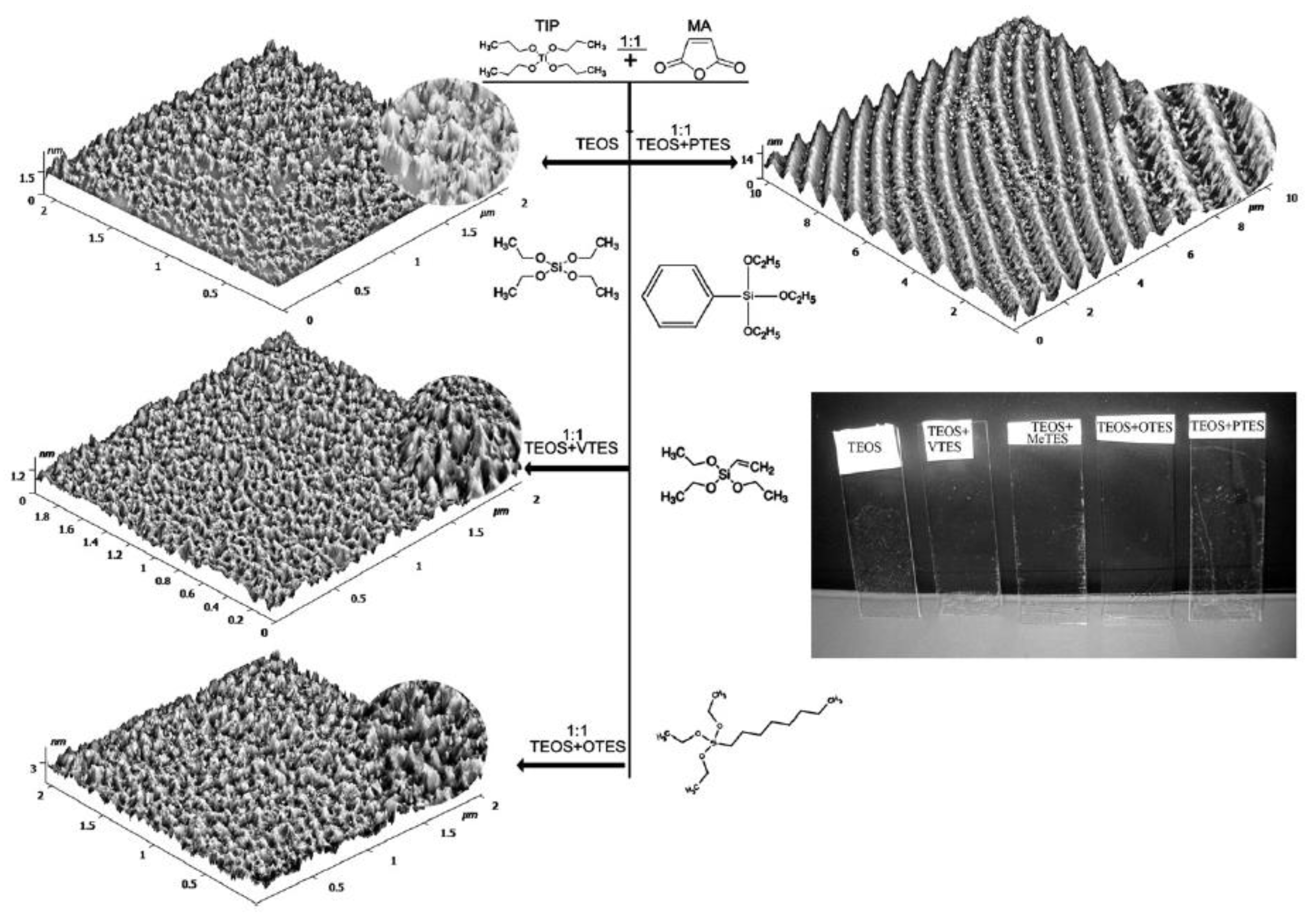
3. Two-Steps Surface Modification
4. Multilayer Films
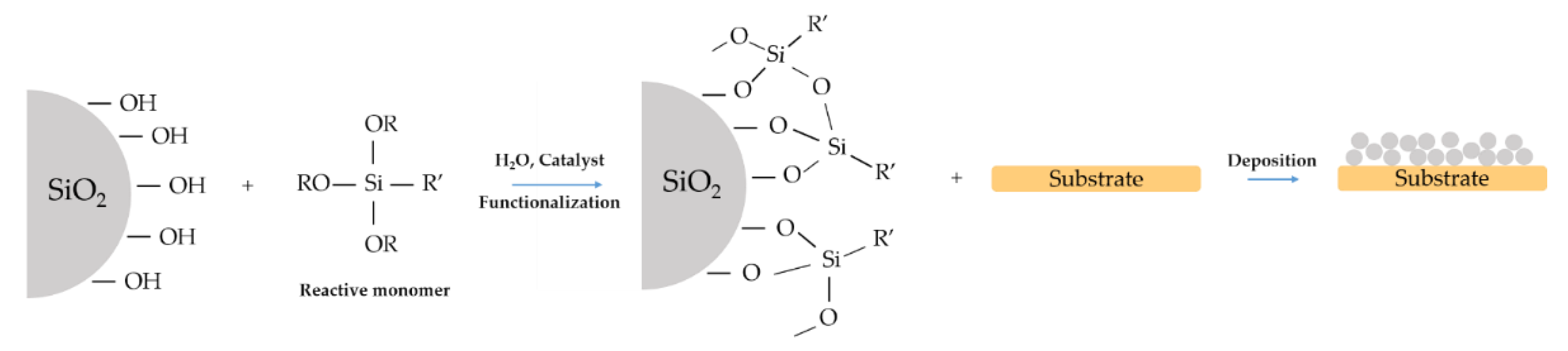
5. Hydrophobic Film with Modified Nanoparticles
6. FAS (Fluoroalkylsilanes)
7. A Brief Comparison of the Different Synthesis Routes
8. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Men, X.; Zhang, Z.; Yang, J.; Zhu, X.; Wanga, K.; Jiang, W. Spray-coated superhydrophobic coatings with regenerability. New J. Chem. 2011, 35, 881–886. [Google Scholar] [CrossRef]
- Manoudis, P.N.; Karapanagiotis, I. Modification of the wettability of polymer surfaces using nanoparticles. Prog. Org. Coat. 2014, 77, 331–338. [Google Scholar] [CrossRef]
- Dhere, S.L.; Latthe, S.S.; Kappenstein, C.; Pajonk, G.M.; Ganesan, V.; Rao, A.V.; Waghe, P.B.; Gupta, S.C. Transparent water repellent silica films by sol–gel process. Appl. Surf. Sci. 2010, 256, 3624–3629. [Google Scholar] [CrossRef]
- Prabhu, K.N.; Fernades, P.; Kumar, G. Effect of substrate surface roughness on wetting behaviour of vegetable oils. Mater. Des. 2009, 30, 297–305. [Google Scholar] [CrossRef]
- Shang, H.M.; Wang, Y.; Limmer, S.J.; Chou, T.P.; Takahashi, K.; Cao, G.Z. Optically transparent superhydrophobic silica-based films. Thin Solid Film. 2005, 472, 37–43. [Google Scholar] [CrossRef]
- Gu, G.; Dang, H.; Zhang, Z.; Wu, Z. Fabrication and characterization of transparent superhydrophobic thin films based on silica nanoparticles. Appl. Phys. 2006, 83, 131–132. [Google Scholar] [CrossRef]
- Liu, H.; Feng, L.; Zhai, J.; Jiang, L.; Zhu, D. Reversible Wettability of a Chemical Vapor Deposition Prepared ZnO Film between Superhydrophobicity and Superhydrophilicity. Langmuir 2004, 20, 5659–5661. [Google Scholar] [CrossRef]
- Luo, B.H.; Shum, P.W.; Zhou, Z.F.; Li, K.Y. Preparation of hydrophobic surface on steel by patterning using laser ablation process. Surf. Coat. Technol. 2018, 204, 1180–1185. [Google Scholar] [CrossRef]
- Woodward, I.; Schofield, W.C.E.; Roucoules, V.; Badyal, J.P.S. Super-hydrophobic Surfaces Produced by Plasma Fluorination of Polybutadiene Films. Langmuir 2003, 19, 3432–3438. [Google Scholar] [CrossRef]
- Zhou, T.; Cheng, X.; Pan, Y.; Li, C.; Gong, L.; Zhang, H. Mechanical performance and thermal stability of glass fiber reinforced silica aerogel composites based on co-precursor method by freeze drying. Appl. Surf. Sci. 2018, 437, 321–328. [Google Scholar] [CrossRef]
- Innocenzi, P. Edited by SpringerBriefs in Materials. In The Sol-to-Gel Transition, 2nd ed.; Springer: Berlin/Heidelberg, Germany, 2016. [Google Scholar]
- Arkles, B. Hydrophobicity, Hydrophilicity and Silanes. Paint Coat. Ind. 2006, 10, 114–135. [Google Scholar]
- Scriven, E.L. Physics and applications of dip coating and spin coating. Mat. Res. Soc. Symp. Proc. 1988, 121, 717–729. [Google Scholar] [CrossRef]
- Aziz, F.; Ismail, A.F. Spray coating methods for polymer solar cells fabrication: A review. Mater. Sci. Semicond. Process. 2015, 39, 416–425. [Google Scholar] [CrossRef] [Green Version]
- Carboni, D.; Pinna, A.; Malfatti, L.; Innocenzi, P. Smart tailoring of the surface chemistry in GPTMS hybrid organic–inorganic films. New J. Chem. 2014, 38, 1635. [Google Scholar] [CrossRef]
- Costacurta, S.; Falcaro, P.; Malfatti, L.; Marongiu, D.; Marmiroli, B.; Cacho-Nerin, F.; Amenitsch, H.; Kirkby, N.; Innocenzi, P. Shaping mesoporous films using dewetting on X-ray prepatterned hydrophilic/hydrophobic layers and pinning effects at the pattern edge. Langmuir 2011, 27, 3898–3905. [Google Scholar] [CrossRef] [PubMed]
- Innocenzi, P.; Malfatti, L.; Falcaro, P. Hard X-rays meet soft matter: When bottom-up and top-down get along well. Soft Matter 2012, 8, 3722–3729. [Google Scholar] [CrossRef]
- Latthe, S.S.; Nadargi, D.Y.; Rao, A.V. TMOS based water repellent silica thin films by co-precursor method using TMES as a hydrophobic agent. Appl. Surf. Sci. 2009, 255, 3600–3604. [Google Scholar] [CrossRef]
- Innocenzi, P.; Esposto, M.; Maddalena, A. Mechanical properties of 3-glycidoxypropyltrimethoxysilane based hybrid organic-inorganic materials. J. Sol-Gel Sci. Technol. 2001, 20, 293. [Google Scholar] [CrossRef]
- Senes, N.; Iacomini, A.; Domingo, N.; Enzo, S.; Mulas, G.; Cuesta-Lopez, S.; Garroni, S. Local Piezoelectric Behavior of Potassium Sodium Niobate Prepared by a Facile Synthesis via Water Soluble Precursors. Phys. Status Solidi 2018, 215, 1700921. [Google Scholar] [CrossRef]
- Gupta, R.; Chaudhury, N.K. Entrapment of biomolecules in sol–gel matrix for applications in biosensors: Problems and future prospects. Biosens. Bioelectron. 2007, 22, 2387–2399. [Google Scholar] [CrossRef]
- Kovtyukhova, N.I.; Buzaneva, E.V.; Waraksa, C.C.; Martin, B.R.; Mallouk, T.E. Surface Sol-Gel Synthesis of Ultrathin Semiconductor Film. Chem. Mater. 2000, 12, 383–389. [Google Scholar] [CrossRef]
- Li, Z.; Houa, B.; Xu, Y.; Wu, D.; Sun, Y.; Hu, W.; Deng, F. Comparative study of sol–gel-hydrothermal and sol–gel synthesis of titania–silica composite nanoparticles. J. Solid State Chem. 2005, 178, 1395–1405. [Google Scholar] [CrossRef]
- Innocenzi, P.; Martucci, A.; Armelao, L. Low temperature preparation of MgxAl2(1-x)Ti(1+x)O5 films by sol-gel processing. J. Eur. Ceram. Soc. 2005, 25, 3587–3591. [Google Scholar] [CrossRef]
- Innocenzi, P. Understanding sol–gel transition through a picture. A short tutorial. J. Sol-Gel Sci. Technol. 2020, 94, 544–550. [Google Scholar] [CrossRef]
- Lebeau, B.; Innocenzi, P. Hybrid materials for optics and photonics. Chem. Soc. Rev. 2011, 40, 886–906. [Google Scholar] [CrossRef] [PubMed]
- Atanacio, A.J.; Latella, B.A.; Barbé, C.J.; Swain, M.V. Mechanical properties and adhesion characteristics of hybrid sol–gel thin films. Surf. Coat. Tech. 2005, 192, 354–364. [Google Scholar] [CrossRef]
- Sharma, P.K.; Rao, K.H. Analysis of different approaches for evaluation of surface energy of microbial cells by contact angle goniometry. Adv. Colloid Interface Sci. 2002, 98, 341–463. [Google Scholar] [CrossRef]
- Owens, D.K. Estimation of the Surface Free Energy of Polymers. J. Appl. Polym. Sci. 1969, 13, 1741–1747. [Google Scholar] [CrossRef]
- Li, D.; Neumann, A.W. Contact Angles on Hydrophobic Solid Surfaces and Their Interpretation. J. Colloid Interface Sci. 1992, 148, 190–200. [Google Scholar] [CrossRef]
- Fowkes, F.M. Additivity of Intermolecular Forces at Interfaces. I. Determination of the Contribution to Surface and Interfacial Tensions of Dispersion Forces in Various Liquids. J. Phys. Chem. 1962, 66, 1863. [Google Scholar] [CrossRef]
- Young, T. An Essay on the Cohesion of Fluids. Philos. Trans. R. Soc. London 1805, 95, 65. [Google Scholar]
- Wenzel, R.N. Resistance of solid surfaces to wetting by water. J. Ind. Eng. Chem. 1936, 28, 988–994. [Google Scholar] [CrossRef]
- Cassie, A.B.D.; Baxter, S. Wettability of Porous Surfaces. Trans. Faraday Soc. 1944, 40, 546–551. [Google Scholar] [CrossRef]
- Lv, C.; Yang, C.; Hao, P.; He, F.; Zheng, Q. Sliding of Water Droplets on Microstructured Hydrophobic Surfaces. Langmuir 2010, 26, 8704–8708. [Google Scholar] [CrossRef] [PubMed]
- Han, T.; Shr, J.; Wu, C.; Hsieh, C. A modified Wenzel model for hydrophobic behavior of nanostructured surfaces. Thin Solid Film. 2007, 515, 4666–4669. [Google Scholar] [CrossRef]
- Bormashenko, E.; Bormashenko, Y.; Whyman, G.; Pogreb, R.; Stanevsky, O. Micrometrically scaled textured metallic hydrophobic interfaces validate the Cassie–Baxter wetting hypothesis. J. Colloid Interface Sci. 2006, 302, 308–311. [Google Scholar] [CrossRef]
- Yang, C.; Tartaglino, U.; Persson, B.N.J. Influence of Surface Roughness on Superhydrophobicity. PRL 2006, 97, 116103. [Google Scholar] [CrossRef] [Green Version]
- Kijlstra, J.; Reihs, K.; Klamt, A. Roughness and topology of ultra-hydrophobic surfaces. Colloids Surf. A Physicochem. Eng. Asp. 2002, 206, 521–529. [Google Scholar] [CrossRef]
- Marmur, A.; Kojevnikova, S. Super-hydrophobic surfaces: Methodological considerations for physical design. J. Colloid Interface Sci. 2020, 568, 148–154. [Google Scholar] [CrossRef]
- Basu, B.J.; Hariprakash, V.; Aruna, S.T.; Lakshmi, R.V.; Manasa, J.; Shruth, B.S. Effect of microstructure and surface roughness on the wettability of superhydrophobic sol–gel nanocomposite coatings. J. Sol-Gel Sci. Technol. 2010, 56, 278–286. [Google Scholar] [CrossRef]
- Wu, L.Y.L.; Soutar, A.M.; Zeng, X.T. Increasing hydrophobicity of sol–gel hard coatings by chemical and morphological modifications. Surf. Coat. Technol. 2005, 198, 420–424. [Google Scholar] [CrossRef]
- Kino, D.; Okada, K.; Tokudome, Y.; Takahashi, M.; Malfatti, L.; Innocenzi, P. Reactivity of silanol group on siloxane oligomers for designing molecular structure and surface wettability. J. Sol-Gel Sci. Technol. 2021, 97, 734–742. [Google Scholar] [CrossRef]
- Rao, A.V.; Gurav, A.B.; Latthe, S.S.; Vhatkar, R.S.; Imai, H.; Kappenstein, C.; Wagh, P.B.; Gupt, S.C. Water repellent porous silica films by sol–gel dip coating method. J. Colloid Interface Sci. 2010, 352, 30–35. [Google Scholar] [CrossRef]
- Latthe, S.S.; Imai, H.; Ganesan, V.; Rao, A.V. Ultrahydrophobic silica films by sol–gel process. J. Porous Mater. 2010, 17, 565–571. [Google Scholar] [CrossRef]
- Nadargi, D.Y.; Gurav, J.L.; El Hawi, N.; Rao, A.V.; Koebel, M. Synthesis and characterization of transparent hydrophobic silica thin films by single step sol–gel process and dip coating. J. Alloys Compd. 2010, 496, 436–441. [Google Scholar] [CrossRef]
- Latthe, S.S.; Imai, H.; Ganesan, V.; Rao, A.V. Porous superhydrophobic silica films by sol–gel process. Microporous Mesoporous Mater. 2010, 130, 115–121. [Google Scholar] [CrossRef]
- Jeevajothi, K.; Crossiya, D.; Subasri, R. Non-fluorinated, room temperature curable hydrophobic coatings by sol–gel process. Ceram. Int. 2012, 38, 2971–2976. [Google Scholar] [CrossRef]
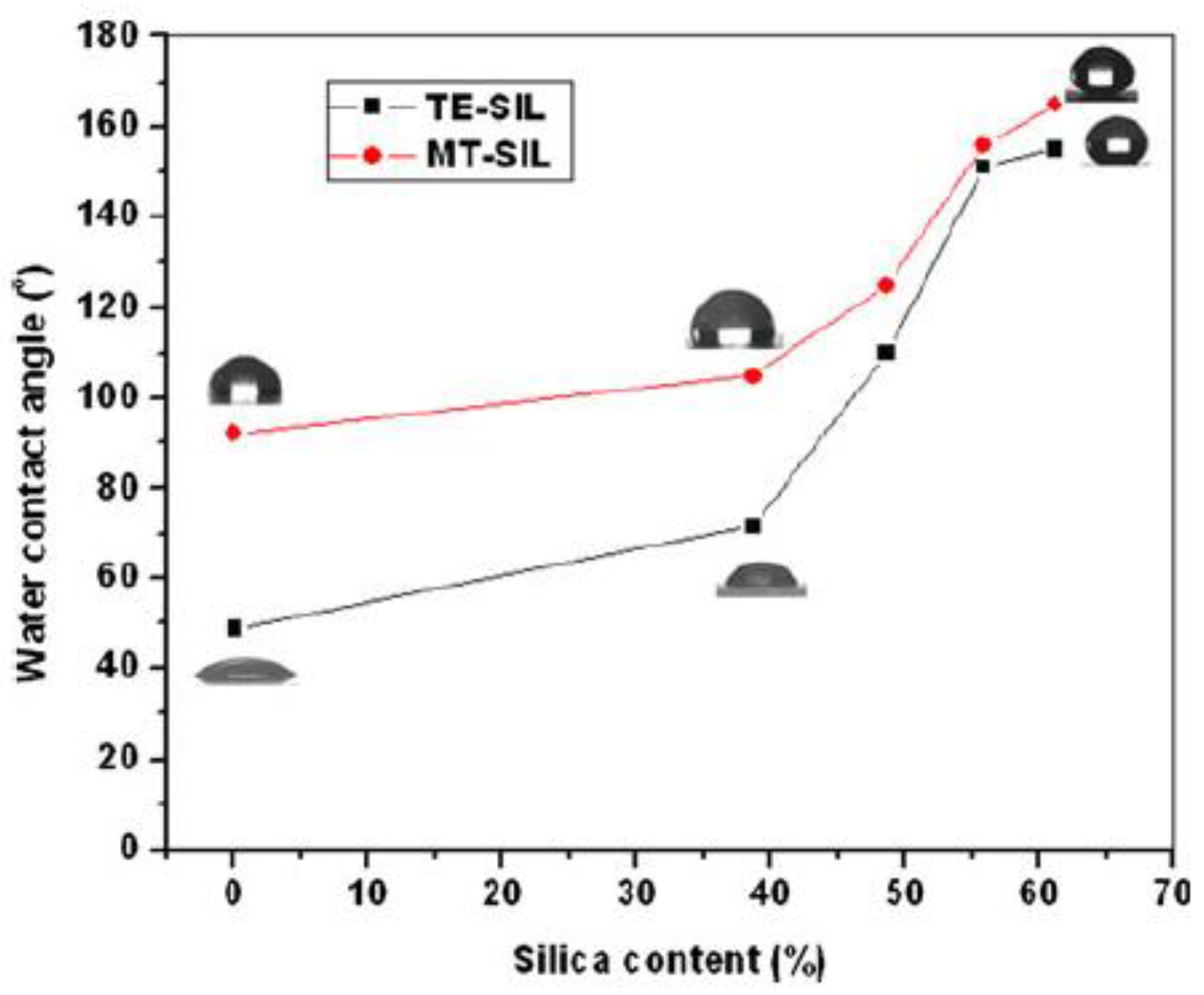
- Ma, Y.; Kanezashi, M.; Tsuru, T. Preparation of organic/inorganic hybrid silica using methyltriethoxysilane and tetraethoxysilane as co-precursors. J. Sol-Gel Sci. Technol. 2010, 53, 93–99. [Google Scholar] [CrossRef]
- Latthe, S.S.; Imai, H.; Ganesan, V.; Rao, A.V. Superhydrophobic silica films by sol–gel co-precursor method. Appl. Surf. Sci. 2009, 256, 217–222. [Google Scholar] [CrossRef]
- Parale, V.G.; Mahadik, D.B.; Kavale, M.S.; Mahadik, S.A.; Rao, A.V.; Mullens, S. Sol–gel preparation of PTMS modified hydrophobic and transparent silica coatings. J. Porous Mater. 2013, 20, 733–739. [Google Scholar] [CrossRef]
- Huang, X.; Yuan, Y.; Liu, S.; Wang, W.; Hong, R. One-step sol-gel preparation of hydrophobic antireflective SiO2 coating on poly(methyl methacrylate) substrate. Mater. Lett. 2017, 208, 62–64. [Google Scholar] [CrossRef]
- Parale, V.G.; Mahadik, D.B.; Mahadik, S.A.; Kavale, M.S.; Wagh, P.B.; Gupta, S.C.; Rao, A.V. OTES modifiedtransparentdipcoatedsilicacoatings. Ceram. Int. 2013, 39, 835–840. [Google Scholar] [CrossRef]
- Bhagat, S.D.; Kim, Y.; Ahn, Y. Room temperature synthesis of water repellent silica coatings by the dip coat technique. Appl. Surf. Sci. 2006, 253, 2217–2221. [Google Scholar] [CrossRef]
- Rao, A.V.; Latthe, S.S.; Nadargi, D.Y.; Hirashima, H.; Ganesan, V. Preparation of MTMS based transparent superhydrophobic silica films by sol–gel method. J. Colloid Interface Sci. 2009, 332, 484–490. [Google Scholar]
- Mahadik, S.A.; Mahadik, D.B.; Parale, V.G.; Wagh, P.B.; Gupta, S.C.; Rao, A.V. Recoverable and thermally stable superhydrophobic silica coating. J. Sol-Gel Sci. Technol. 2012, 62, 490–494. [Google Scholar] [CrossRef]
- Beirami, K.; Baghshahi, S.; Ardestani, M.; Riahi, N. Synthesis and characterization of hydrophobic nano-silica thin coatings for outdoor insulators. Process. Appl. Ceram. 2020, 14, 40–46. [Google Scholar] [CrossRef] [Green Version]
- Purcar, V.; Stamatin, I.; Cinteza, O.; Petcu, C.; Raditoiu, V.; Ghiurea, M.; Miclaus, T.; Andronie, A. Fabrication of hydrophobic and antireflective coatings based on hybrid silica films by sol–gel process. Surf. Coat. Tech. 2012, 206, 4449–4454. [Google Scholar] [CrossRef]
- Mahadik, S.A.; Kavale, M.S.; Mukherjee, S.K.; Rao, A.V. Transparent Superhydrophobic silica coatings on glass by sol–gel method. Appl. Surf. Sci. 2010, 257, 333–339. [Google Scholar] [CrossRef]
- Mahadik, S.A.; Mahadik, D.B.; Kavale, M.S.; Parale, V.G.; Wagh, P.B.; Barshilia, H.C.; Gupta, S.C.; Hegde, N.D.; Rao, A.V. Thermally stable and transparent superhydrophobic sol–gel coatings by spray method. J. Sol-Gel Sci. Technol. 2012, 63, 580–586. [Google Scholar] [CrossRef]
- Latthe, S.S.; Imai, H.; Ganesan, V.; Kappenstein, C.; Rao, A.V. Optically transparent superhydrophobic TEOS-derived silica films by surface silylation method. J. Sol-Gel Sci. Technol. 2010, 53, 208–215. [Google Scholar] [CrossRef]
- Tian, H.; Gao, X.; Yang, T.; Li, D.; Chen, Y. Fabrication and characterization of superhydrophobic silica nanotrees. J. Sol-Gel Sci. Technol. 2008, 48, 277–282. [Google Scholar] [CrossRef]
- Ramezani, M.; Vaezi, M.R.; Kazemzadeh, A. Preparation of silane-functionalized silica films via two-step dipcoating sol–gel and evaluation of their superhydrophobic properties. Appl. Surf. Sci. 2014, 317, 147–153. [Google Scholar] [CrossRef]
- Hou, H.; Chen, Y. Preparation of super-hydrophobic silica films with visible light transmission using phase separation. J. Sol-Gel Sci. Technol. 2007, 43, 53–57. [Google Scholar] [CrossRef]
- Li, X.; Shen, J. A scratch-resistant and hydrophobic broadband antireflective coating by sol–gel method. Thin Solid Film. 2011, 519, 6236–6240. [Google Scholar] [CrossRef]
- Ye, L.; Zhang, Y.; Zhang, X.; Hu, T.; Ji, R.; Ding, B.; Jiang, B. Sol–gel preparationof SiO2/TiO2/SiO2–TiO2 broadband antireflectivecoating for solar cell coverglass. Sol. Energy Mater. Sol. Cells 2013, 111, 160–164. [Google Scholar] [CrossRef]
- Shiu, J.; Kuo, C.; Chen, P.; Mou, C. Fabrication of Tunable Superhydrophobic Surfaces by Nanosphere Lithography. Chem. Mater. 2004, 16, 561–564. [Google Scholar] [CrossRef]
- Xue, L.; Li, J.; Fu, J.; Han, Y. Super-hydrophobicity of silica nanoparticles modified with vinyl groups. Colloids Surf. A Physicochem. Eng. Asp. 2009, 338, 15–19. [Google Scholar] [CrossRef]
- Sutar, R.S.; Gaikwad, S.S.; Latthe, S.S.; Kodag, V.S.; Deshmukh, S.B.; Saptal, L.P.; Kulal, S.R.; Bhosale, A.K. Superhydrophobic Nanocomposite Coatings of Hydrophobic Silica NPs and Poly (methyl methacrylate) with Notable Self-Cleaning Ability. Macromol. Symp. 2020, 393, 2000116. [Google Scholar] [CrossRef]
- Tao, C.; Yang, K.; Zou, X.; Yan, H.; Yuan, X.; Zhang, L.; Jiang, B. Double-layer tri-wavelength hydrophobic antireflective coatings derived from methylated silica nanoparticles and hybrid silica nanoparticles. J. Sol-Gel Sci. Technol. 2018, 86, 285–292. [Google Scholar] [CrossRef]
- Lia, M.; Sua, B.; Zhoua, B.; Wanga, H.; Menga, J. One-pot synthesis and self-assembly of anti-wear octadecyltrichlorosilane/silica nanoparticles composite films on silicon. Appl. Surf. Sci. 2020, 508, 145187. [Google Scholar] [CrossRef]
- Philipavičius, J.; Kazadojev, I.; Beganskienė, A.; Melninkaitis, A.; Sirutkaitis, V.; Kareiva, A. Hydrophobic Antireflective Silica Coatings via Sol-gel Process. Mater. Sci. 2008, 14, 283–287. [Google Scholar]
- Zhang, X.; Cai, S.; You, D.; Yan, L.; Lv, H.; Yuan, X.; Jiang, B. Template-Free Sol-Gel Preparation of Superhydrophobic ORMOSIL Films for Double-Wavelength Broadband Antirefl ective Coatings. Adv. Funct. Mater. 2013, 23, 4361–4365. [Google Scholar] [CrossRef]
- Tao, C.; Zou, X.; Reddy, K.M.; Zhang, L.; Jiang, B. Hydrophobic ultralow refractive-index silica coating towards double-layer broadband antireflective coating with exceptionally high vacuum stability and laser-induced damage threshold. Colloids Surf. 2019, 563, 340–349. [Google Scholar] [CrossRef]
- Li, H.; Li, N.; Zhang, Y.; He, H.; Liu, Z. Anti-reflection OTS-treated SiO2 thin films with superhydrophobic property. J. Sol-Gel Sci. Technol. 2017, 83, 518–526. [Google Scholar] [CrossRef]
- Petcu, C.; Alexandrescu, E.; Balan, A.; Tanase, M.A.; Cinteza, L.O. Synthesis and Characterisation of Organo-Modified Silica Nanostructured Films for theWater-Repellent Treatment of Historic Stone Buildings. Coatings 2020, 10, 1010. [Google Scholar] [CrossRef]
- Yang, H.; Pi, P.; Cai, Z.; Wen, X.; Wang, X.; Cheng, J.; Yang, Z. Facile preparation of super-hydrophobic and super-oleophilic silica film on stainless steel mesh via sol–gel process. Appl. Surf. Sci. 2010, 256, 4095–4102. [Google Scholar] [CrossRef]
- Daoud, W.A.; Xin, J.H.; Tao, X. Synthesis and characterization of hydrophobic silica nanocomposites. Appl. Surf. Sci. 2006, 252, 5368–5371. [Google Scholar] [CrossRef]
- Yuan, Y.; Yan, G.H.; Huang, S.H.; Hong, R.J. Preparation of hydrophobic SiO2/PMHS sol and ORMOSIL antireflective films for solar glass cover. Sol. Energy 2016, 130, 1–9. [Google Scholar] [CrossRef]
- Tao, C.; Yan, H.; Yuan, X.; Yin, Q.; Zhu, J.; Ni, W.; Yan, L.; Zhang, L. Hydrophobic antireflective coatings with ultralow refractive index synthesized by deposition of methylated hollow silica nanoparticles. Mater. Lett. 2016, 183, 374–377. [Google Scholar] [CrossRef]
- Yang, X.; Zhu, L.; Chen, Y.; Bao, B.; Xu, J.; Zhou, W. Controlled hydrophilic/hydrophobic property of silica films by manipulating the hydrolysis and condensation of tetraethoxysilane. Appl. Surf. Sci. 2016, 376, 1–9. [Google Scholar] [CrossRef]
- Wu, L.Y.L.; Tan, G.H.; Zeng, X.T.; Li, T.H.; Chen, Z. Synthesis and Characterization of Transparent Hydrophobic Sol-Gel Hard Coatings. J. Sol-Gel Sci. Technol. 2006, 38, 85–89. [Google Scholar] [CrossRef]
- Wu, Y.L.; Chen, Z.; Zeng, X.T. Nanoscale morphology for high hydrophobicity of a hard sol–gel thin film. Appl. Surf. Sci. 2008, 254, 6952–6958. [Google Scholar] [CrossRef]
- Chia, F.; Liu, D.; Wu, H.; Lei, J. Mechanically robust and self-cleaning antireflection coatings from nanoscale binding of hydrophobic silica nanoparticles. Sol. Energy Mater. Sol. Cells 2019, 200, 109939. [Google Scholar] [CrossRef]
- Raza, M.A.; Zandvliet, H.J.W.; Poelsema, B.; Kooij, E.S. Hydrophobic surfaces with tunable dynamic wetting properties via colloidal assembly of silica microspheres and gold nanoparticles. J. Sol-Gel Sci. Technol. 2015, 74, 357–367. [Google Scholar] [CrossRef] [Green Version]
- Xue, C.; Tian, Q.; Ji, S.; Zhao, L.; Ding, Y.; Li, H.; An, Q. The fabrication of mechanically durable and stretchable superhydrophobic PDMS/SiO2 composite film. RSC Adv. 2020, 10, 19466. [Google Scholar] [CrossRef]
- Ambrohewicz, D.; Ciesielczyk, F.; Nowacka, M.; Karasiewicz, J.; Piasecki, A.; Maciejewski, H.; Jesionowski, T. Fluoroalkylsilane versus alkylsilane as hydrophobic agents for silica and silicates. J. Nanomater. 2013, 631938. [Google Scholar]
- ECHA. Committee for Risk Assessment (RAC) Committee for Socio-Economic Analysis (SEAC). Available online: https://echa.europa.eu/documents/10162/8930d3e6-54c5-024f-a012-2b3dc77f6a2d (accessed on 7 November 2021).
- Evans, G. Summary of Evidence—Solvent-Based Hydrophobic Coatings and Risks for Acute Respiratory Toxicity. Health Saf. Exec. 2017. Available online: https://www.hse.gov.uk/research/rrpdf/rr1112.pdf (accessed on 7 November 2021).
- Xu, J.; Yu, Q.; Liu, J.; Yin, Y.; Han, Y.; Li, B. Preparation and characterization of polyfluoroaniline/organosiloxane hybrid films. J. Sol-Gel Sci. Technol. 2014, 69, 580–585. [Google Scholar] [CrossRef]
- Sermon, P.A.; Leadley, J.G. Fluoroalkylsilane Modification of Sol–Gel SiO2-TiO2 Coatings. J. Sol-Gel Sci. Technol. 2004, 32, 293–296. [Google Scholar] [CrossRef]






| Silicon Alkoxide | Organically Modified Alkoxide | Substrate | Coating Method | Contact Angle/° | Data from Ref. |
|---|---|---|---|---|---|
| TEOS | methyl (MTES) | Glass | Dip-coating | 160 | [47] |
| TEOS | HDMS | Glass | Dip- and Spray-coating | 166 | [48] |
| TEOS | VTMS | Glass | Dip-coating | 145 | [49] |
| TEOS | TMES | Glass | Dip-coating | 151 | [50] |
| TEOS | phenyl | Glass | Dip-coating | 133 | [51] |
| TEOS | PMHS | PMMA | Dip-coating | 125 | [52] |
| TEOS | OTES | Glass | Dip-coating | 125 | [53] |
| TMOS | HMDS | Stainless steel plate | Dip-coating | 145 | [54] |
| Silicon Alkoxide | Organically Modified Alkoxide | Substrate | Coating Method | Contact Angle /° | Data from Ref. |
|---|---|---|---|---|---|
| MTES/TMMS | TMCS | Glass | Dip-coating | 172 | [59] |
| MTES | TMCS | Glass | Spray-coating | 167 | [60] |
| TEOS | DMCS/TMCS | Glass | Dip-coating | 162 | [61] |
| TEOS | TMCS | Glass | Dip-coating | 153 | [62] |
| ETES | iso-OTMS | Glass | Dip-coating | 160 | [63] |
| Silicon Alkoxide | Organically Modified Alkoxide | Substrate | Coating Method | Contact Angle/° | Data from Ref. |
|---|---|---|---|---|---|
| TEOS | HDMS/MTMS | Glass | Spin-coating | 165 | [72] |
| TEOS | HMDS | Glass | Dip-coating | 160 | [73] |
| TEOS | HMDS | Glass and Silicon wafer | Dip-coating | 126 | [74] |
| TEOS | OTS | Glass | Spin-coating | 150 | [75] |
| TEOS | MTES/VTES/OTES/OTS | Glass | Brushing | 146 | [76] |
| TEOS | MTES | Stainless steel mesh | Dip-coating | 142 | [77] |
| TEOS | GPTMS/HDTMS | Silicon, glass, and cellulosic cotton | Spin-coating | 141 | [78] |
| TEOS | PMHS | Glass | Dip-coating | 130 | [79] |
| TEOS | MTES | Glass | Dip-coating | 122 | [80] |
| TEOS | TEOS | Glass | Spin-coating | 121 | [81] |
| MTES/TEOS | PDMS | Glass | Spray-coating | 133 | [82] |
| MTES/TEOS | PDMS | Glass | Spray-coating | 120 | [83] |
| HMDS | MTMS | Glass | Dip-coating | 161 | [84] |
| SiO2-NPs | MPTMS | Silicon wafer | Spray-coating | 162 | [85] |
| SiO2-NPs | PDMS/OTMS | Shrink film | Spray-coating | 155 | [86] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poddighe, M.; Innocenzi, P. Hydrophobic Thin Films from Sol–Gel Processing: A Critical Review. Materials 2021, 14, 6799. https://doi.org/10.3390/ma14226799
Poddighe M, Innocenzi P. Hydrophobic Thin Films from Sol–Gel Processing: A Critical Review. Materials. 2021; 14(22):6799. https://doi.org/10.3390/ma14226799
Chicago/Turabian StylePoddighe, Matteo, and Plinio Innocenzi. 2021. "Hydrophobic Thin Films from Sol–Gel Processing: A Critical Review" Materials 14, no. 22: 6799. https://doi.org/10.3390/ma14226799
APA StylePoddighe, M., & Innocenzi, P. (2021). Hydrophobic Thin Films from Sol–Gel Processing: A Critical Review. Materials, 14(22), 6799. https://doi.org/10.3390/ma14226799







