Empirical NOx Removal Analysis of Photocatalytic Construction Materials at Real-Scale
Abstract
:1. Introduction
- (1)
- A photoreactor test was conducted to assess the NO removal performance of construction materials such as coatings, paints and shotcrete using a modified ISO 22197-1 method. The UV irradiation of the outdoor environment was analyzed, and the experiment was conducted in an indoor laboratory under UV irradiation identical to that of the outdoor condition. Subsequently, photoreactor tests were conducted on construction materials applied to actual buildings located in Seoul, South Korea. The NO removal performance at the real scale was demonstrated by confirming that the trends of the indoor and outdoor environments showed comparable results.
- (2)
- In this experiment, the NOx removal performance was analyzed by assessing the amount of NOx ions remaining in the water after washing the surface of the specimen artificially exposed to NOx in the laboratory. The preliminary test used two same-sized specimens according to the specification in the ISO 22197-1 standard; the specimens were enlarged to the size of building materials. The experimental conditions were confirmed when the NOx removal performance was found to increase similarly to the tendency of the specimen to increase in size. The enlarged specimen was used for a field experiment by applying a modified method from the ISO 22197-1 standard.
2. Preliminary Test for Measuring NOx Removal
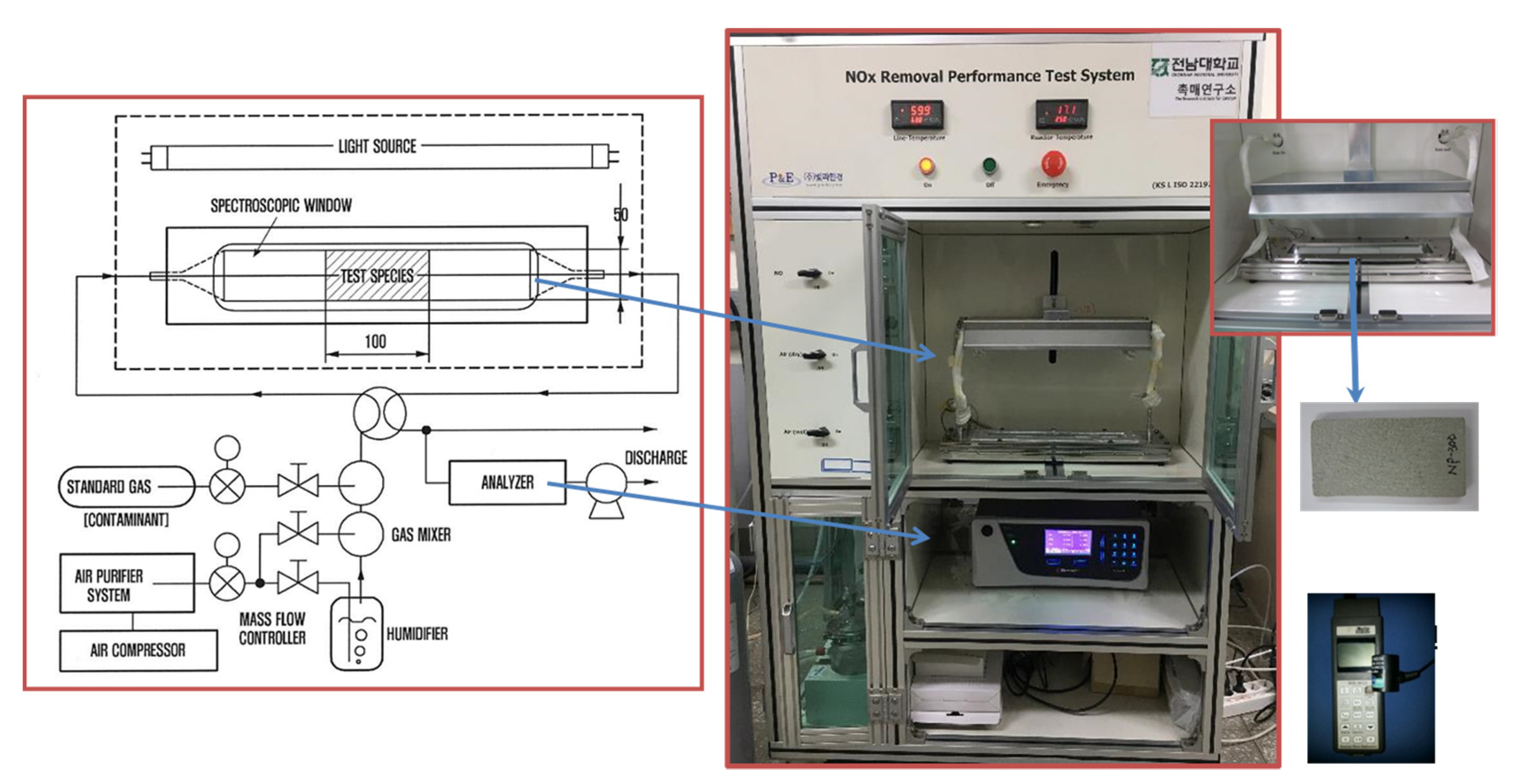
2.1. Experimental Setup
2.2. Photoreactor Test
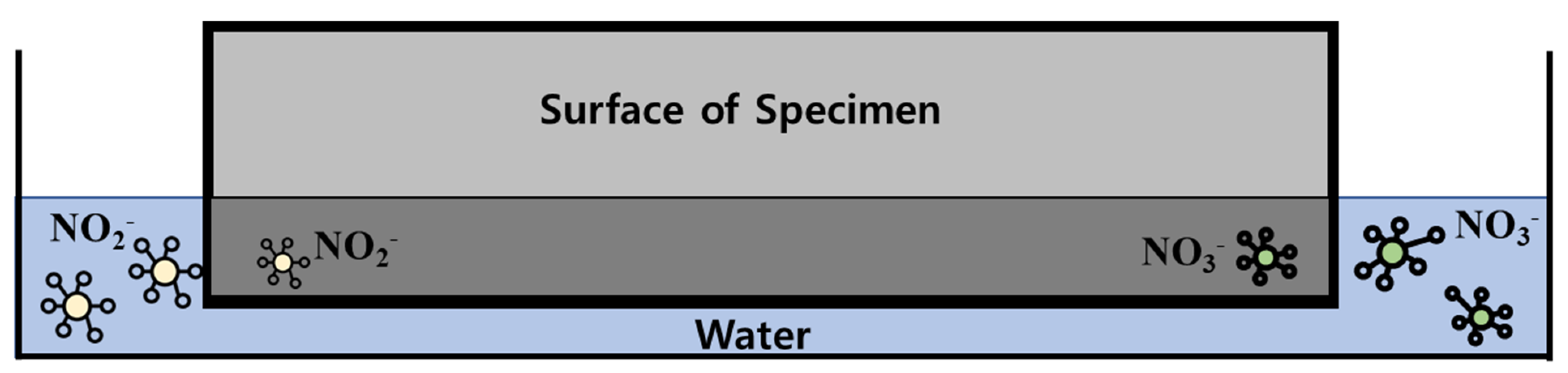
2.3. Washing Water Experiment
- Preparation of specimen
- The base material of the specimen was unified as the CRC board.
- The CRC board used as the base material was washed sufficiently to remove pre-exiting nitrogen oxides.
- The test specimens are prepared with CRC boards; the photocatalytic paint was applied to these boards, and the general aqueous paint specimens were prepared as a control group.
- Methods of analyzing NO removal and washing water.
- NO removal was conducted in accordance with the ISO 22197-1 method.
- Concentrations other than those specified in ISO 22197-1 were not significant in the experiment; hence, the experiments were conducted only at the prescribed concentration of 1 ppm.
- The washing of the specimens that have undergone the ISO 22197-1 experiment are in accordance with the washing method specified in ISO 22197-1.
- The washing of ISO specimens exposed to the outdoor environment should be performed according to the washing method specified in ISO 22197-1.
3. Methodology for the Field Test
3.1. Photoreactor Field Test Method
3.2. ISO-Based Washing Water Field Test Method for Real-Scale Construction Materials
- Panels with photocatalytic paint applied
- The material of the panel was CRC board.
- The paint was applied to the panel after washing and drying the CRC board.
- The control group was prepared with a CRC board with general aqueous paint applied.
- Methods of washing and analyzing the panels
- A total of 2 L of washing water was used for each panel.
- The number of washes was limited to two; the washing intervals were determined by considering the weather conditions.
- The panel was washed by spraying.
- A separate holder and recovery bin was designed to collect rainwater.
- The number of ions in the washing water were measured via ion chromatography.
4. Results of NO Removal Analysis Experiment
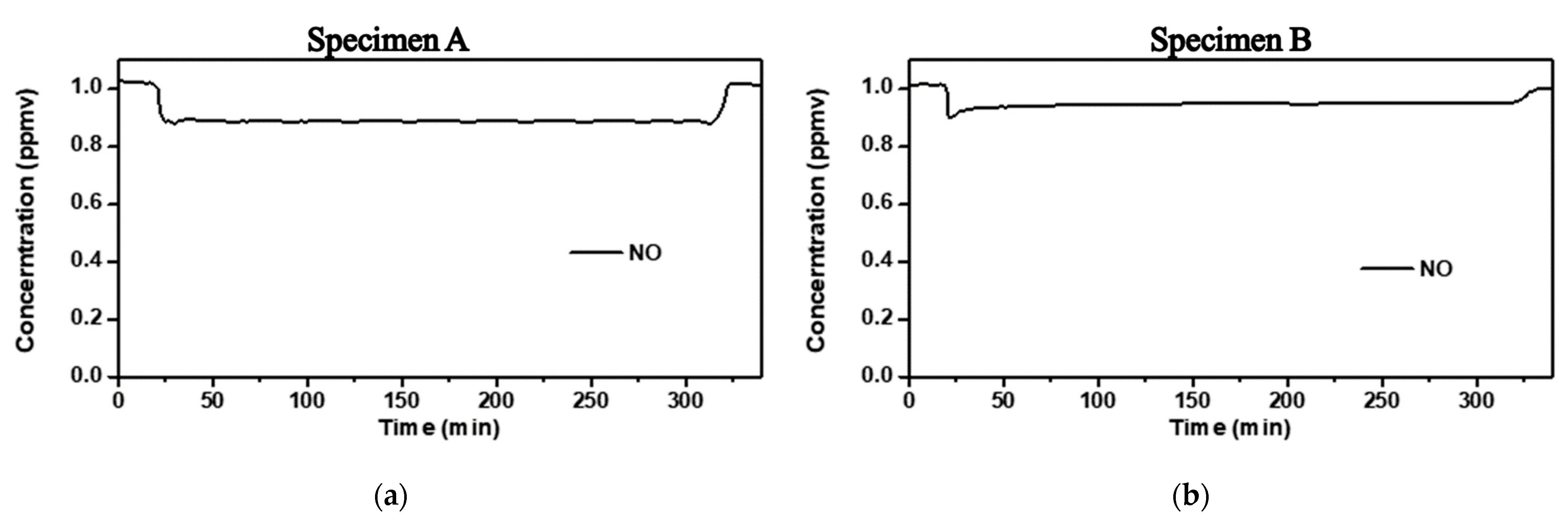
4.1. Photoreactor Test Results
4.2. Washing Water Field Test Results of Specimens
4.3. Washing Water Field Test Results of Photocatalytic Panel
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Sillman, S. The relation between ozone, NOx and hydrocarbons in urban and polluted rural environments. Atmos. Environ. 1999, 33, 1821–1845. [Google Scholar] [CrossRef]
- Sun, Y.; Zwolińska, E.; Chmielewski, A.G. Abatement technologies for high concentrations of NOx and SO2 removal from exhaust gases: A review. Crit. Rev. Environ. Sci. Technol. 2016, 46, 119–142. [Google Scholar] [CrossRef]
- Yoshida, K.; Makino, S.; Sumiya, S.; Muramatsu, G.; Helferich, R. Simultaneous reduction of NOx and particulate emissions from diesel engine exhaust. SAE Trans. 1989, 1994–2005. [Google Scholar] [CrossRef]
- Hodan, W.M.; Barnard, W.R. Evaluating the Contribution of PM2.5 Precursor Gases and Re-Entrained Road Emissions to Mobile Source PM2.5 Particulate Matter Emissions; MACTEC Federal Programs: Research Triangle Park, NC, USA, 2004. [Google Scholar]
- Kim, J.-Y.; Lee, E.-Y.; Choi, I.; Kim, J.; Cho, K.-H. Effects of the particulate matter2.5 (PM2.5) on lipoprotein metabolism, uptake and degradation, and embryo toxicity. Mol. Cells 2015, 38, 1096. [Google Scholar]
- Skalska, K.; Miller, J.S.; Ledakowicz, S. Trends in NOx abatement: A review. Sci. Total Environ. 2010, 408, 3976–3989. [Google Scholar] [CrossRef]
- Bowman, C.T. Control of combustion-generated nitrogen oxide emissions: Technology driven by regulation. Symp. (Int.) Combust. 1992, 24, 859–878. [Google Scholar] [CrossRef]
- Heck, R.M. Catalytic abatement of nitrogen oxides–stationary applications. Catal. Today 1999, 53, 519–523. [Google Scholar] [CrossRef]
- Chang, M.B.; Lee, H.M.; Wu, F.; Lai, C.R. Simultaneous removal of nitrogen oxide/nitrogen dioxide/sulfur dioxide from gas streams by combined plasma scrubbing technology. J. Air Waste Manag. Assoc. 2004, 54, 941–949. [Google Scholar] [CrossRef]
- Guo, M.-Z.; Ling, T.-C.; Poon, C.-S. TiO2-based self-compacting glass mortar: Comparison of photocatalytic nitrogen oxide removal and bacteria inactivation. Build. Environ. 2012, 53, 1–6. [Google Scholar] [CrossRef]
- Brogren, C.; Karlsson, H.T.; Bjerle, I. Absorption of NO in an alkaline solution of KMnO4. Chem. Eng. Technol. 1997, 20, 396–402. [Google Scholar] [CrossRef]
- Guseva, T.; Potapova, E.; Tichonova, I.; Shchelchkov, K. Nitrogen oxide emissions reducing in cement production. IOP Conf. Ser. Mater. Sci. Eng. 2021, 1083, 012083. [Google Scholar] [CrossRef]
- Rodríguez, M.H.; Melián, E.P.; Díaz, O.G.; Araña, J.; Macías, M.; Orive, A.G.; Rodríguez, J.D. Comparison of supported TiO2 catalysts in the photocatalytic degradation of NOx. J. Mol. Catal. A Chem. 2016, 413, 56–66. [Google Scholar] [CrossRef]
- Bengtsson, N.; Castellote, M. Heterogeneous photocatalysis on construction materials: Effect of catalyst properties on the efficiency for degrading NOx and self cleaning. Mater. Constr. 2014, 64, e013. [Google Scholar] [CrossRef] [Green Version]
- Cárdenas, C.; Tobón, J.I.; García, C.; Vila, J. Functionalized building materials: Photocatalytic abatement of NOx by cement pastes blended with TiO2 nanoparticles. Constr. Build. Mater. 2012, 36, 820–825. [Google Scholar] [CrossRef]
- Devahasdin, S.; Fan Jr, C.; Li, K.; Chen, D.H. TiO2 photocatalytic oxidation of nitric oxide: Transient behavior and reaction kinetics. J. Photochem. Photobiol. A Chem. 2003, 156, 161–170. [Google Scholar] [CrossRef]
- Ichiura, H.; Kitaoka, T.; Tanaka, H. Photocatalytic oxidation of NOx using composite sheets containing TiO2 and a metal compound. Chemosphere 2003, 51, 855–860. [Google Scholar] [CrossRef]
- Yu, Q.; Brouwers, H. Indoor air purification using heterogeneous photocatalytic oxidation. Part I: Experimental study. Appl. Catal. B Environ. 2009, 92, 454–461. [Google Scholar] [CrossRef]
- Luévano-Hipólito, E.; Martínez-De La Cruz, A.; López-Cuellar, E.; Yu, Q.; Brouwers, H. Synthesis, characterization and photocatalytic activity of WO3/TiO2 for NO removal under UV and visible light irradiation. Mater. Chem. Phys. 2014, 148, 208–213. [Google Scholar] [CrossRef] [Green Version]
- Ballari, M.; Carballada, J.; Minen, R.I.; Salvadores, F.; Brouwers, H.; Alfano, O.M.; Cassano, A.E. Visible light TiO2 photocatalysts assessment for air decontamination. Process Saf. Environ. Prot. 2016, 101, 124–133. [Google Scholar] [CrossRef]
- Kuo, C.-S.; Tseng, Y.-H.; Huang, C.-H.; Li, Y.-Y. Carbon-containing nano-titania prepared by chemical vapor deposition and its visible-light-responsive photocatalytic activity. J. Mol. Catal. A Chem. 2007, 270, 93–100. [Google Scholar] [CrossRef]
- Maggos, T.; Plassais, A.; Bartzis, J.; Vasilakos, C.; Moussiopoulos, N.; Bonafous, L. Photocatalytic degradation of NOx in a pilot street canyon configuration using TiO2-mortar panels. Environ. Monit. Assess. 2008, 136, 35–44. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Jee, H.; Lim, M.; Kim, J.H.; Kwon, S.J.; Lee, K.M.; Nezhad, E.Z.; Bae, S. Photocatalytic performance evaluation of titanium dioxide nanotube-reinforced cement paste. Materials 2020, 13, 5423. [Google Scholar] [CrossRef] [PubMed]
- Xiang, W.; Ming, C. Implementing extended producer responsibility: Vehicle remanufacturing in China. J. Clean. Prod. 2011, 19, 680–686. [Google Scholar] [CrossRef]
- Ifang, S.; Gallus, M.; Liedtke, S.; Kurtenbach, R.; Wiesen, P.; Kleffmann, J. Standardization methods for testing photo-catalytic air remediation materials: Problems and solution. Atmos. Environ. 2014, 91, 154–161. [Google Scholar] [CrossRef]
- Chen, J.; Poon, C.-S. Photocatalytic construction and building materials: From fundamentals to applications. Build. Environ. 2009, 44, 1899–1906. [Google Scholar] [CrossRef]
- Seo, D.; Yun, T.S. NOx removal rate of photocatalytic cementitious materials with TiO2 in wet condition. Build. Environ. 2017, 112, 233–240. [Google Scholar] [CrossRef]
- Song, Y.W.; Kim, M.Y.; Chung, M.H.; Yang, Y.K.; Park, J.C. NOx-Reduction Performance Test for TiO2 Paint. Molecules 2020, 25, 4087. [Google Scholar] [CrossRef]
- Gallus, M.; Akylas, V.; Barmpas, F.; Beeldens, A.; Boonen, E.; Boréave, A.; Cazaunau, M.; Chen, H.; Daële, V.; Doussin, J. Photocatalytic de-pollution in the Leopold II tunnel in Brussels: NOx abatement results. Build. Environ. 2015, 84, 125–133. [Google Scholar] [CrossRef]
- Ballari, M.; Hunger, M.; Hüsken, G.; Brouwers, H. NOx photocatalytic degradation employing concrete pavement containing titanium dioxide. Appl. Catal. B Environ. 2010, 95, 245–254. [Google Scholar] [CrossRef]
- Folli, A.; Strøm, M.; Madsen, T.P.; Henriksen, T.; Lang, J.; Emenius, J.; Klevebrant, T.; Nilsson, Å. Field study of air purifying paving elements containing TiO2. Atmos. Environ. 2015, 107, 44–51. [Google Scholar] [CrossRef]
- Boonen, E.; Beeldens, A. Photocatalytic roads: From lab tests to real scale applications. Eur. Transp. Res. Rev. 2013, 5, 79–89. [Google Scholar] [CrossRef] [Green Version]
- Boonen, E.; Beeldens, A. Recent photocatalytic applications for air purification in Belgium. Coatings 2014, 4, 553–573. [Google Scholar] [CrossRef] [Green Version]
- Cassar, L. Photocatalysis of cementitious materials: Clean buildings and clean air. MRS Bull. 2004, 29, 328–331. [Google Scholar] [CrossRef]
- Guerrini, G.L. Photocatalytic performances in a city tunnel in Rome: NOx monitoring results. Constr. Build. Mater. 2012, 27, 165–175. [Google Scholar] [CrossRef]
- Yu, Q.; Hendrix, Y.; Lorencik, S.; Brouwers, H. Field study of NOx degradation by a mineral-based air purifying paint. Build. Environ. 2018, 142, 70–82. [Google Scholar] [CrossRef]
- Cordero, J.; Hingorani, R.; Jiménez-Relinque, E.; Grande, M.; Borge, R.; Narros, A.; Castellote, M. NOx removal efficiency of urban photocatalytic pavements at pilot scale. Sci. Total Environ. 2020, 719, 137459. [Google Scholar] [CrossRef]
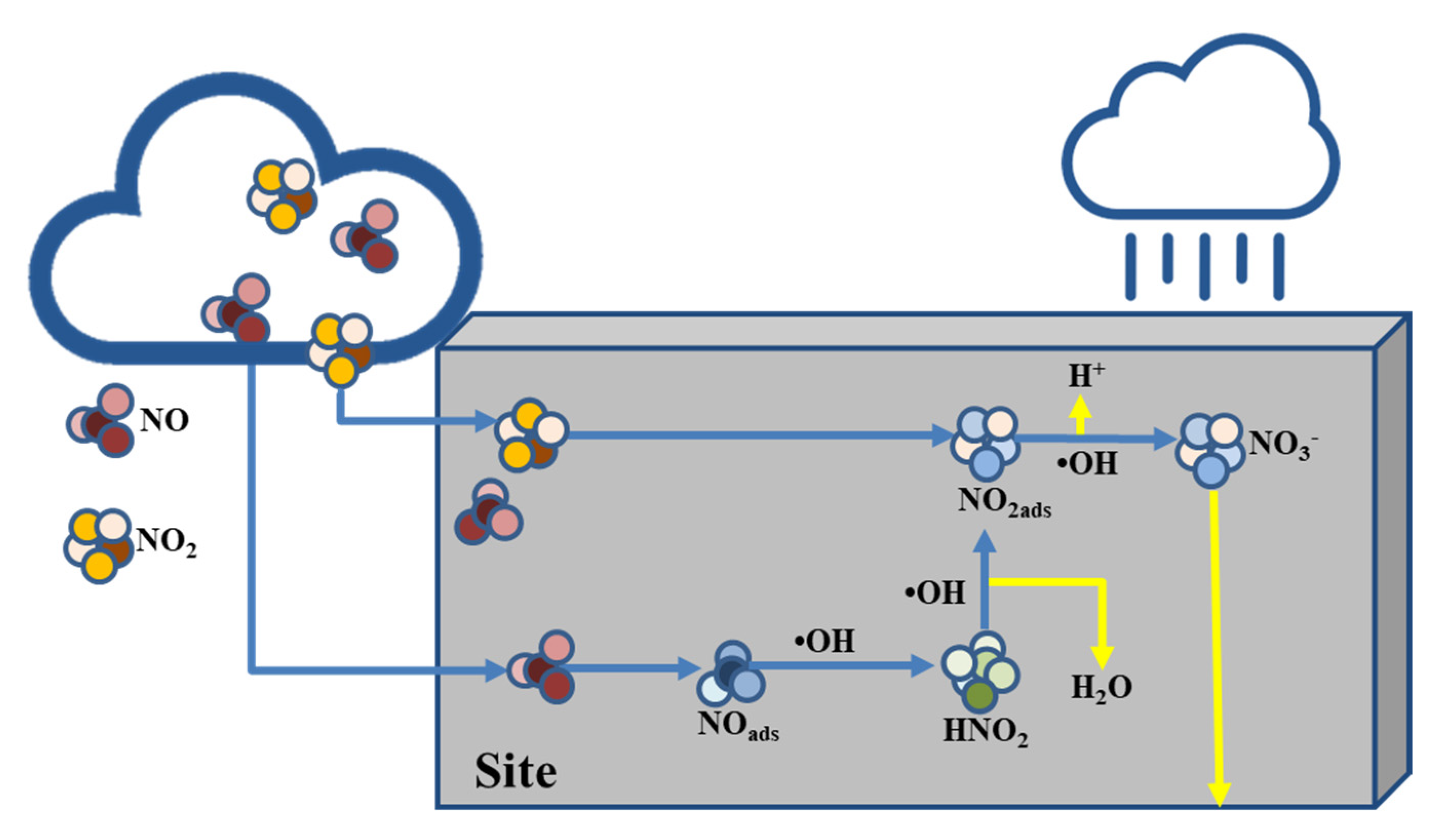








| Process | Formula |
|---|---|
| Activation | |
| Hole trapping | |
| Electron trapping | |
| Hydroxyl attack |
| Material no. | Photocatalytic Coating | Photocatalytic Paint | Photocatalytic Shotcrete | |||
|---|---|---|---|---|---|---|
| Contained Chemicals | Proportion (%) | Contained Chemicals | Proportion (%) | Contained Chemicals | Proportion (%) | |
| 1 | TiO2 | 1.75 | TiO2 | 3.5 | TiO2 | 3.5 |
| 2 | Silicone compound | 5.6 | Fluid ceramic binder | 7.2 | Synthetic resins emulsions | 2.2 |
| 3 | Water | 51.0 | Others | 89.3 | Cements | 17.2 |
| 4 | Others | 41.65 | - | - | Others (Aggregates and water, etc.) | 77.1 |
| Materials | NO Injection (μmol) | NO Removal (μmol/50 cm2) | Measuring Time (Hour) | UV Irradiation (W/m2) |
|---|---|---|---|---|
| Photocatalytic coating | 38.57 | 7.29 | 5 | 10 |
| Photocatalytic paint | 37.78 | 5.76 | 5 | 10 |
| Photocatalytic shotcrete | 8.61 | 11.28 | 1 | 10 |
| Model | 930 Compact IC Flex |
|---|---|
| Column | Metrosep A Supp 5, 250 × 4 mm |
| Eluent | 3.2 mM Sodium carbonate, 1.0 M Sodium bicarbonate |
| Flow rate | 0.7 mL/min |
| Inj. Volume | 20 μL |
| Detection | Conductivity Detector |
| Division | Removed NO (μmol) | Nitric Acid Ionic Weight (μmol) | Ratio (%) |
|---|---|---|---|
| Specimen A | 4.74 | 0.96 | 48 |
| Specimen B | 2.03 | 0.34 | 41 |
| Division | NO Amount with Washing Once (μmol) | Accumulated Removed NO Amount (μmol) |
|---|---|---|
| CRC-A | 0.81 | 3.57 |
| CRC-B | 1.11 | 2.84 |
| Materials | NO Injection Rate (µmol) | NO Removal (µmol/50 cm2) | NO Removal (µmol/50 cm2) | UV Irradiation (W/m2) | Measurement Date (Weather) | |
|---|---|---|---|---|---|---|
| Building A: Photocatalytic coating | 118.97 | 11.06 | 3.68 | 2–13 | 25 October 2018 (Sunny) | |
| Building B: Photocatalytic paint | 1st | 107.82 | 10.03 | 3.34 | 6–14 | 14 November 2018 (Sunny) |
| 2nd | 122.79 | 4.87 | 1.62 | 2–16 | 5 December 2018 (Cloudy) | |
| Building C: Photocatalytic shotcrete | 1st | 95.84 | 6.78 | 2.26 | 1 | 13 November 2018 (Sunny) |
| 2nd | 114.52 | 2.62 | 0.87 | 2–9 | 6 December 2018 (Cloudy) | |
| Specimen Type | NOx Removal Amount (μmol) | Eluted NOx Amount (μmol) |
|---|---|---|
| Aqueous paint specimen | 0.08 | 0.53 |
| Photocatalytic paint specimen A | 7.90 | 7.86 |
| Photocatalytic paint specimen B | 10.80 | 10.38 |
| Photocatalytic paint specimen C | 2.40 | 3.62 |
| Photocatalytic paint specimen D | 2.45 | 3.25 |
| Specimen Type | ISO NOx Removal Performance (µmol/50 cm²·5 h) | Elution Amount after ISO Experiment (µmol) | Elution Amount after Outdoor Exposure for Five Days (µmol) |
|---|---|---|---|
| Aqueous paint | 0.08 | 0.53 | 3.88 |
| Photocatalytic paint sample C | 2.40 | 3.62 | 14.13 |
| Photocatalytic Panels | Eluted NO2−, NO3− Ions (μmol) |
|---|---|
| Aqueous paint | 210 |
| Photocatalytic panel (S1) | 1469 |
| Photocatalytic panel (S2) | 475 |
| Photocatalytic panel (S3) | 504 |
| Division | Daily Average NOx Removal Amount of Panel (1) (µmol) | Estimated Annual NOx Removal Amount of Panel (2) (µmol) | Estimated Annual NOx Removal Amount (3) (g/m2) |
|---|---|---|---|
| Aqueous paint | 42.00 | 10,920 | 0.46 |
| Photocatalytic panel (S1) | 293.80 | 76,388 | 3.19 |
| Photocatalytic panel (S2) | 95.00 | 24,700 | 1.03 |
| Photocatalytic panel (S3) | 100.80 | 26,208 | 1.09 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, M.; Kim, H.; Park, J. Empirical NOx Removal Analysis of Photocatalytic Construction Materials at Real-Scale. Materials 2021, 14, 5717. https://doi.org/10.3390/ma14195717
Kim M, Kim H, Park J. Empirical NOx Removal Analysis of Photocatalytic Construction Materials at Real-Scale. Materials. 2021; 14(19):5717. https://doi.org/10.3390/ma14195717
Chicago/Turabian StyleKim, Miyeon, Hyunggeun Kim, and Jinchul Park. 2021. "Empirical NOx Removal Analysis of Photocatalytic Construction Materials at Real-Scale" Materials 14, no. 19: 5717. https://doi.org/10.3390/ma14195717
APA StyleKim, M., Kim, H., & Park, J. (2021). Empirical NOx Removal Analysis of Photocatalytic Construction Materials at Real-Scale. Materials, 14(19), 5717. https://doi.org/10.3390/ma14195717






