Proven Anti-Wetting Properties of Molybdenum Tested for High-Temperature Corrosion-Resistance with Potential Application in the Aluminum Industry
Abstract
:1. Introduction
- The use of modelling to predict and demonstrate the anti-wetting properties of bulk Mo in contact with molten Al;
- Provide experimental proof of the anti-stick properties of metallic Mo in a molten Al alloy using the ALCAN standard immersion test, thus leading to a potential application in the Al industry;
- The application of radio frequency (RF) suspension plasma-spray (SPS) technology in synthesizing high-density Mo-based coatings, a process that cannot be easily achieved due to Mo’s high melting point at 2896 K (2623 °C).
2. Materials and Methods
2.1. Modelling as a Predictive Tool
2.1.1. Simulation by FactSage™
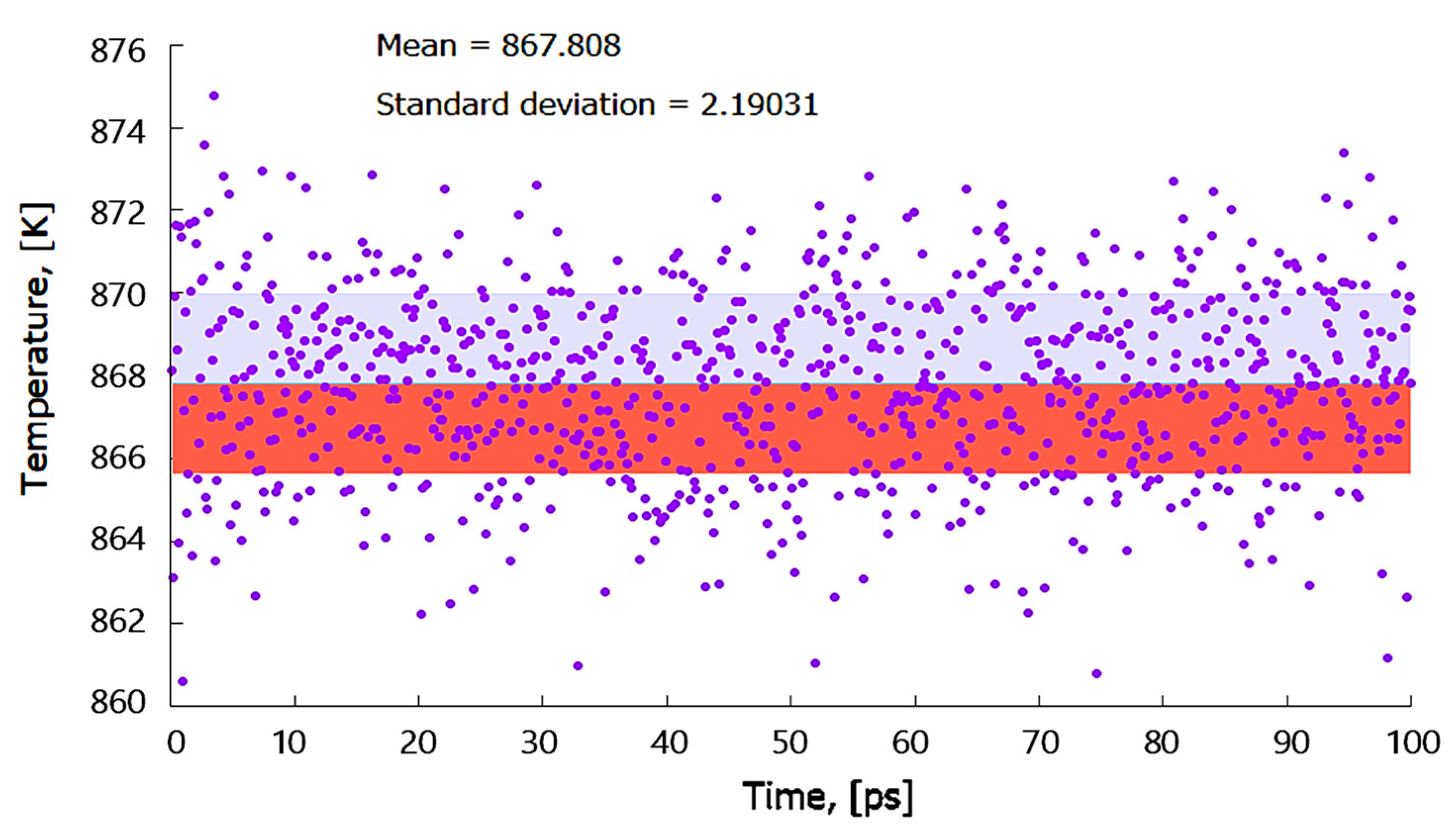
2.1.2. Simulation Using Classical Molecular Dynamics
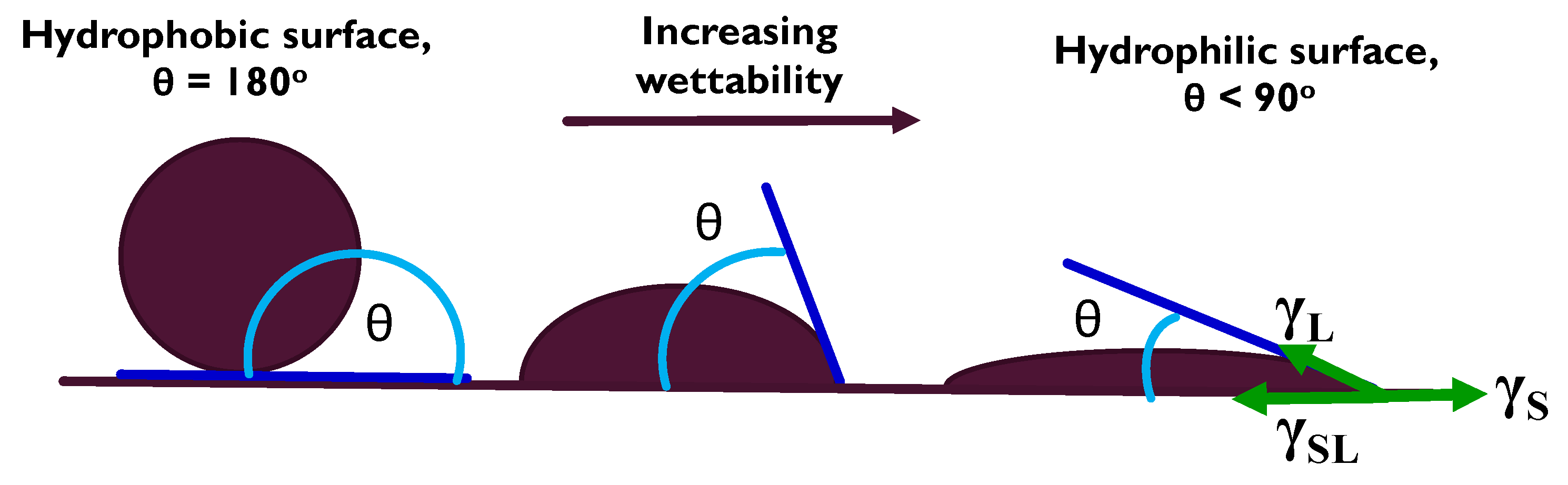
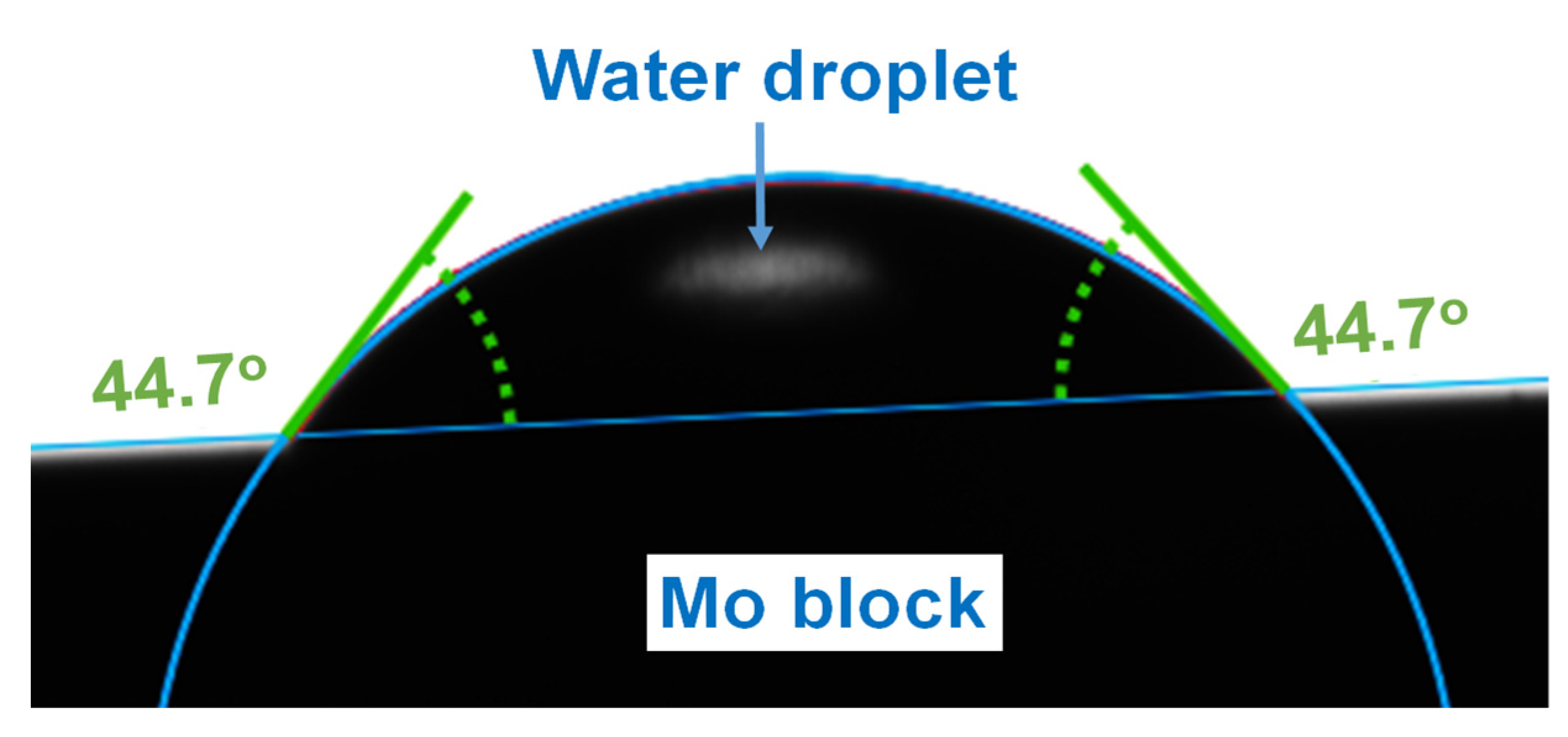
2.2. Surface Energy Determination
2.3. The ALCAN Standard Immersion Test
2.4. Materials Characterization
2.4.1. Optical Microscopy
2.4.2. Elemental Analysis by Optical Emission Spectrometry (OES)
2.4.3. Scanning Electron Microscopy (SEM)
2.4.4. X-ray Diffraction (XRD)
3. Results
- (a)
- Computer simulation results, which indicate that modelling as a predictive tool using both FactSage™ thermochemical software and Classical Molecular Dynamics showed that there would be no reaction and atomic diffusion at the interface between the Mo block and the molten Al alloy. The estimation of the surface energy by the VCG theory using the sessile drop experiments equally predicted a weak surface interaction at the Mo(s)–Al(l) interface.
- (b)
- Experimental data from the static ALCAN immersion test agree with the simulation results, although some traces of Al-Mo alloys were detected on the Al-rich side of the Mo(s)–Al(l) interface. A weak interaction existed between the Al-Mo alloys and the solid Mo block (Mo-rich side), making it easy for Mo to peel off and demonstrate its anti-wetting properties; we suspect that the 20% mass loss on Mo was due to the chemical attack along the grain boundaries leading to intergranular corrosion.
3.1. Computer Modelling
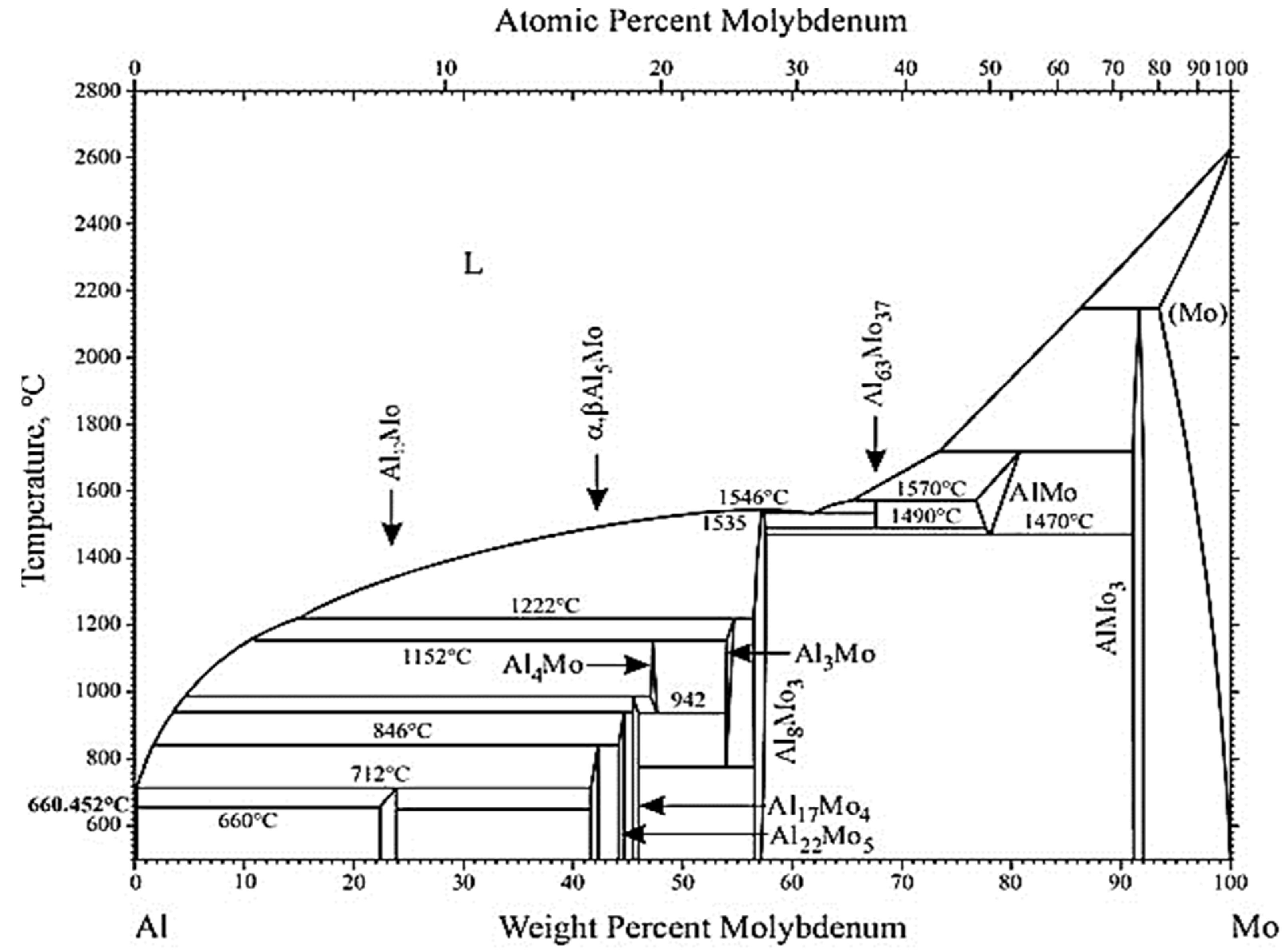
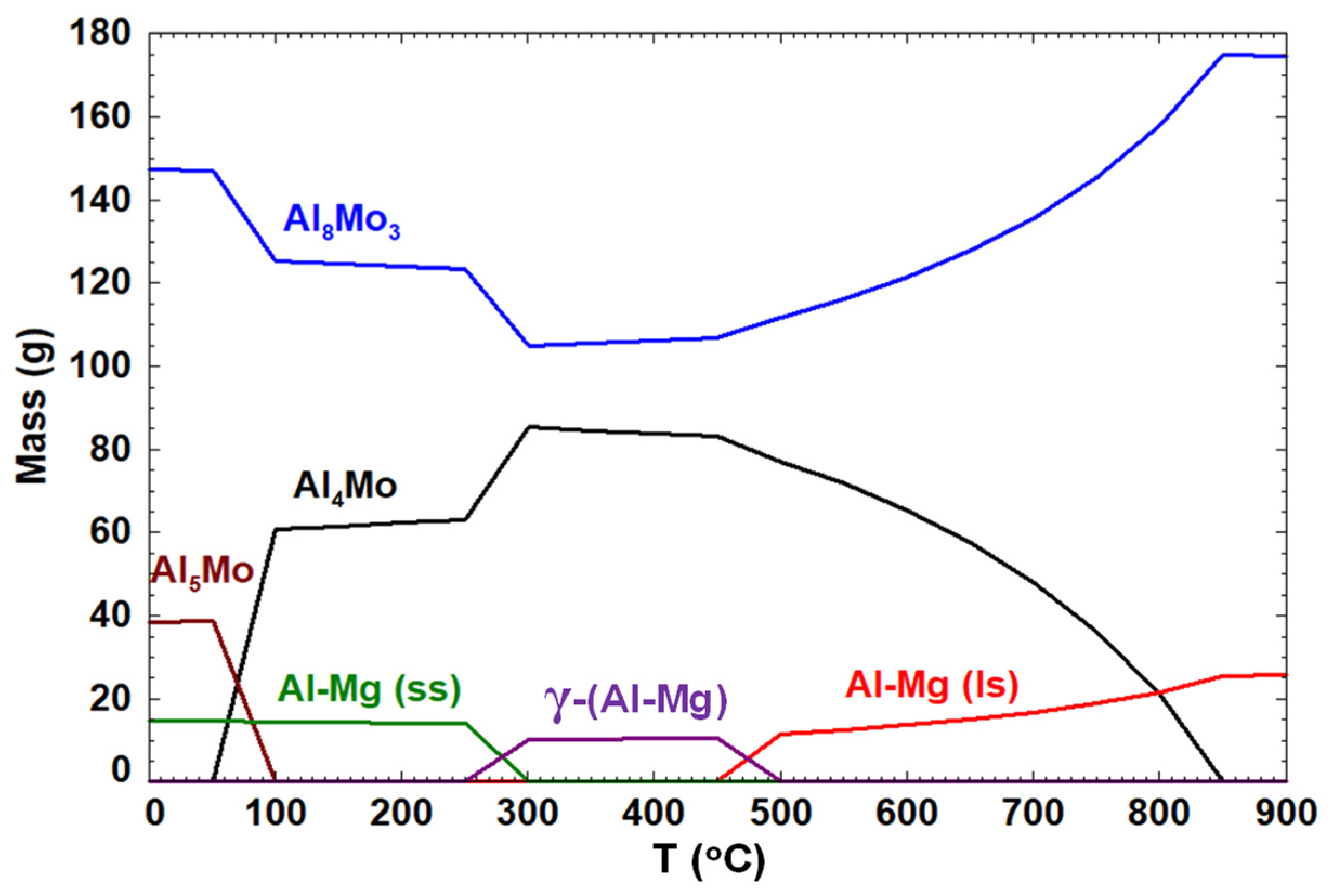
3.1.1. Simulation by FactSage™
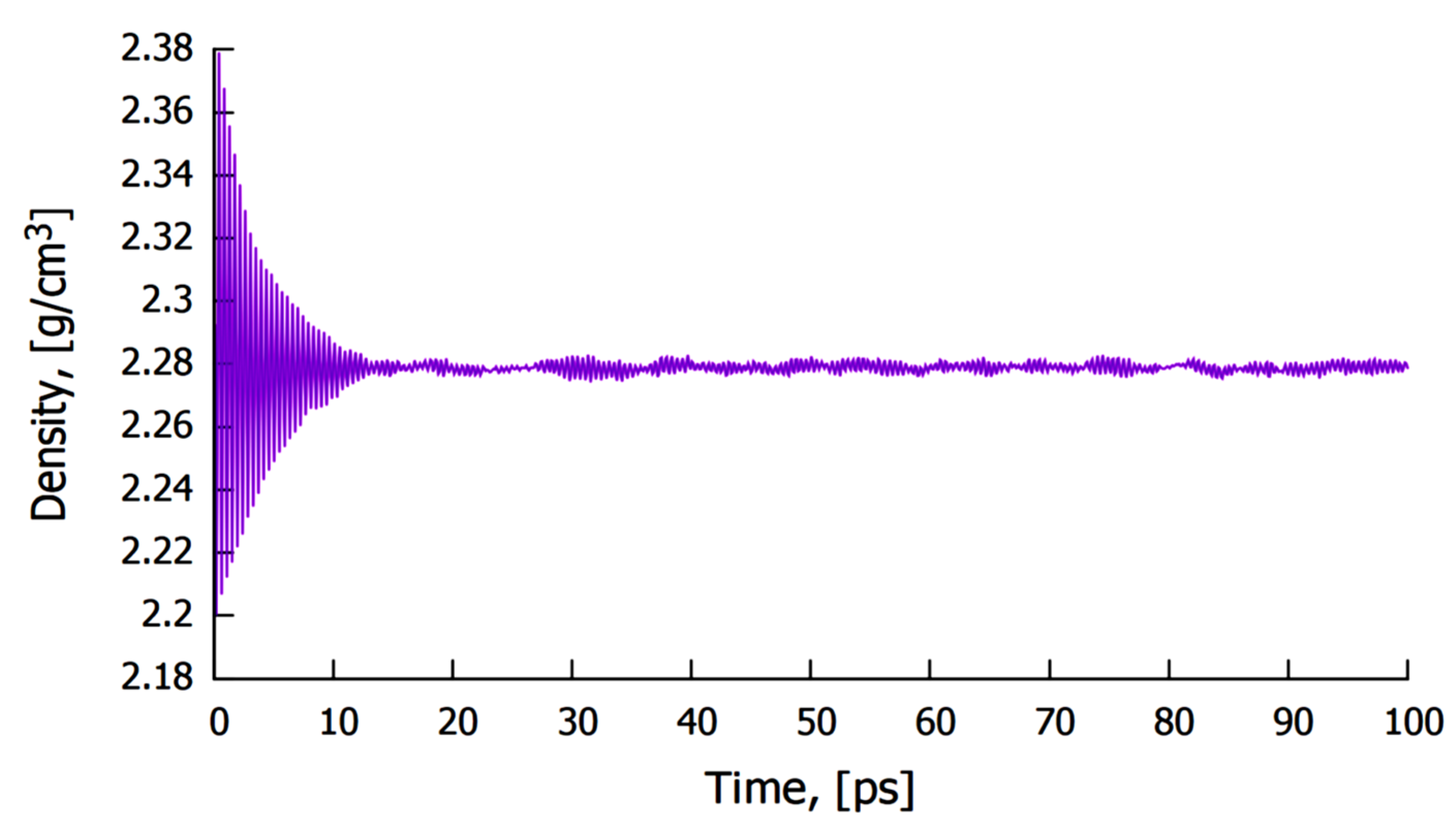
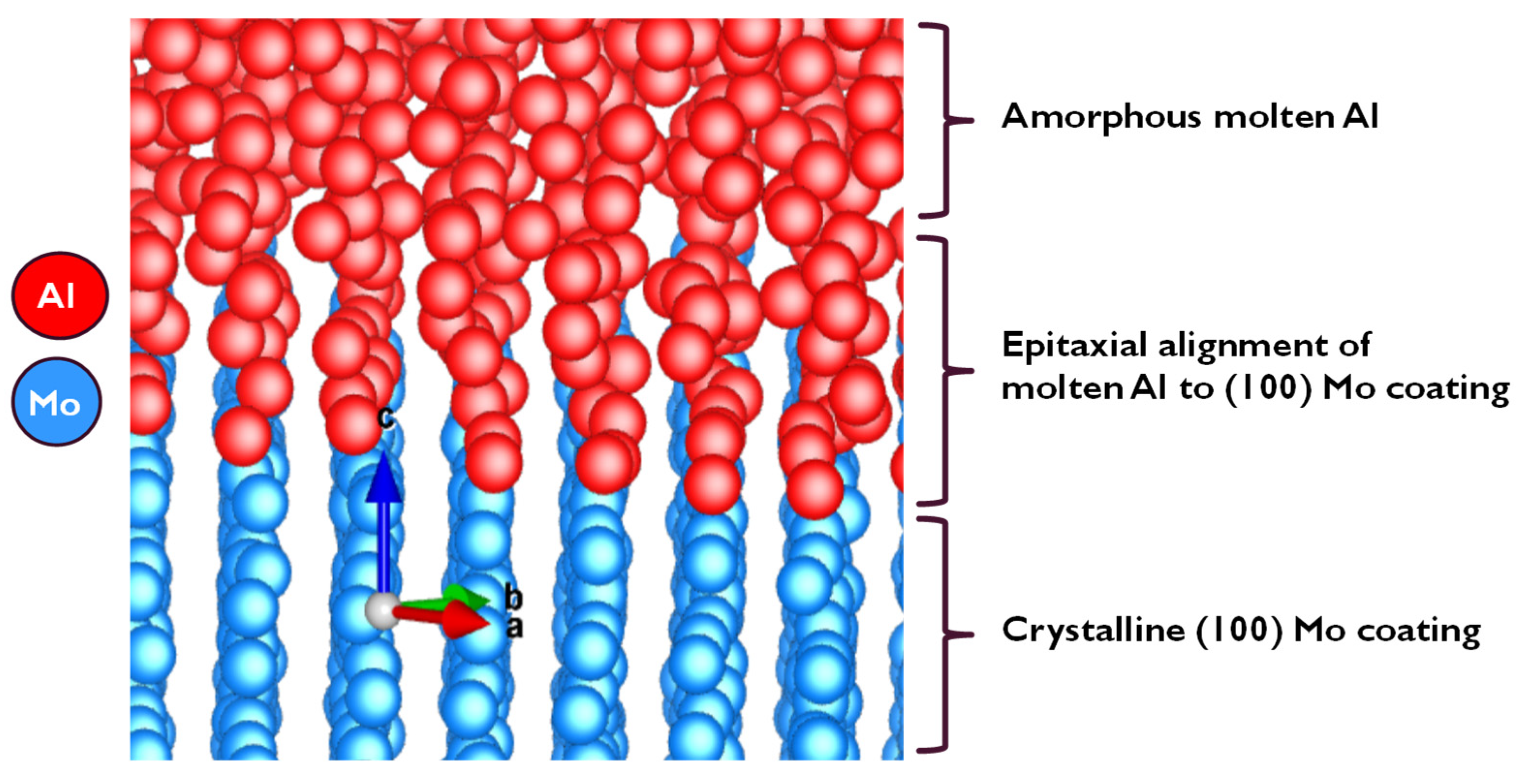
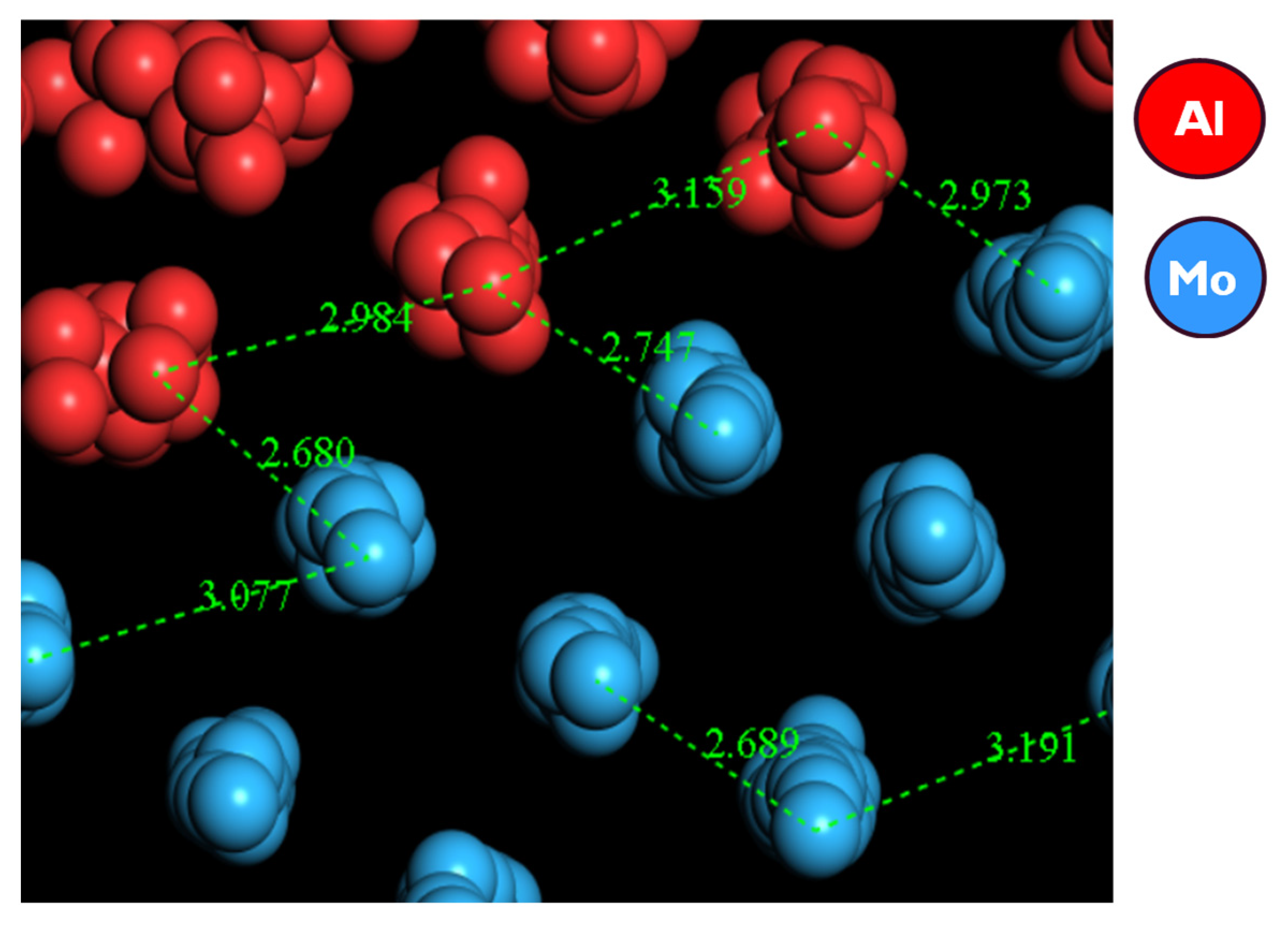
3.1.2. Simulation by Classical Molecular Dynamics
- (i)
- No cross diffusion of atoms at the Al–Mo interface was observed as no atoms of one element moved into the bulk of the other element;
- (ii)
- At the Al–Mo interface, Al formed a non-wetting layer adopting the morphology of the exposed Mo (100) crystal;
- (iii)
- The calculated mean surface (or interaction) energy at the Al–Mo interface was found to be ~203 mJ/m2 (at 1200 K).
3.2. Surface Energy Determination
3.3. The Static ALCAN Immersion Test
3.3.1. Mass Loss Analysis
3.3.2. Analysis of Anti-Wetting Properties through Optical Microscopy
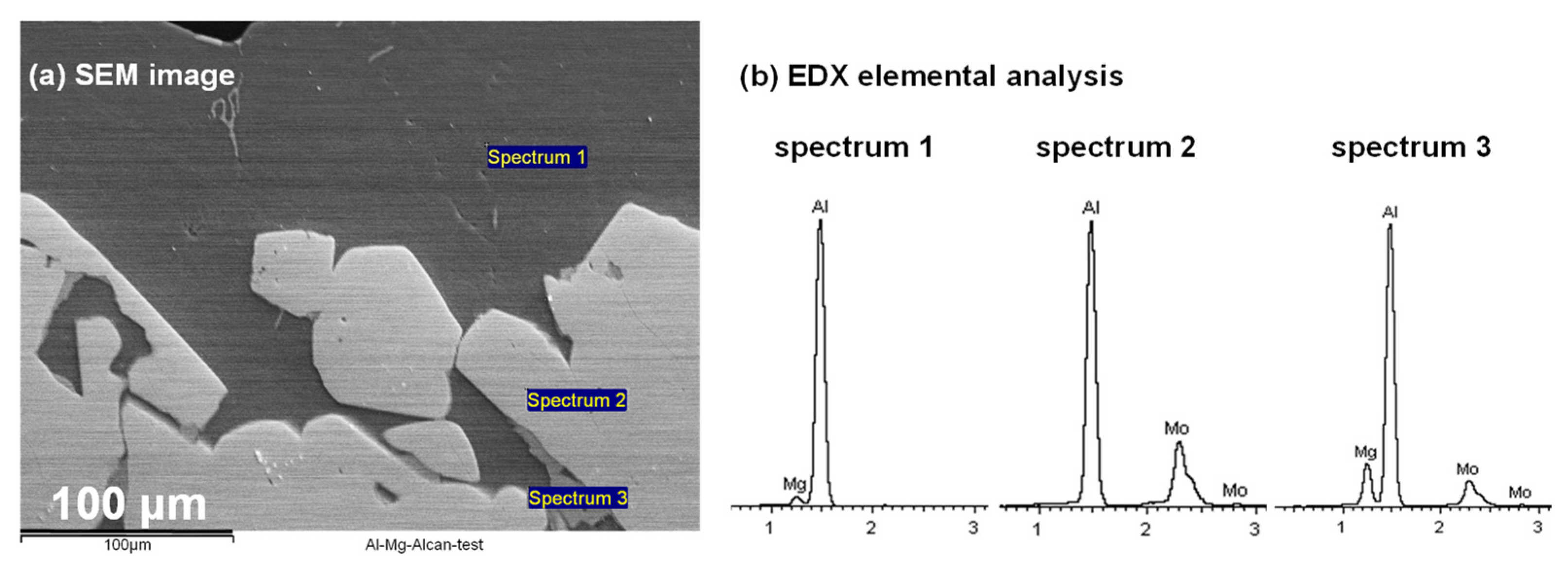
3.3.3. SEM Analysis at the Mo/Al-Mg Interface
3.4. Materials Characterization
3.4.1. Elemental Analysis by OES
3.4.2. SEM Analysis
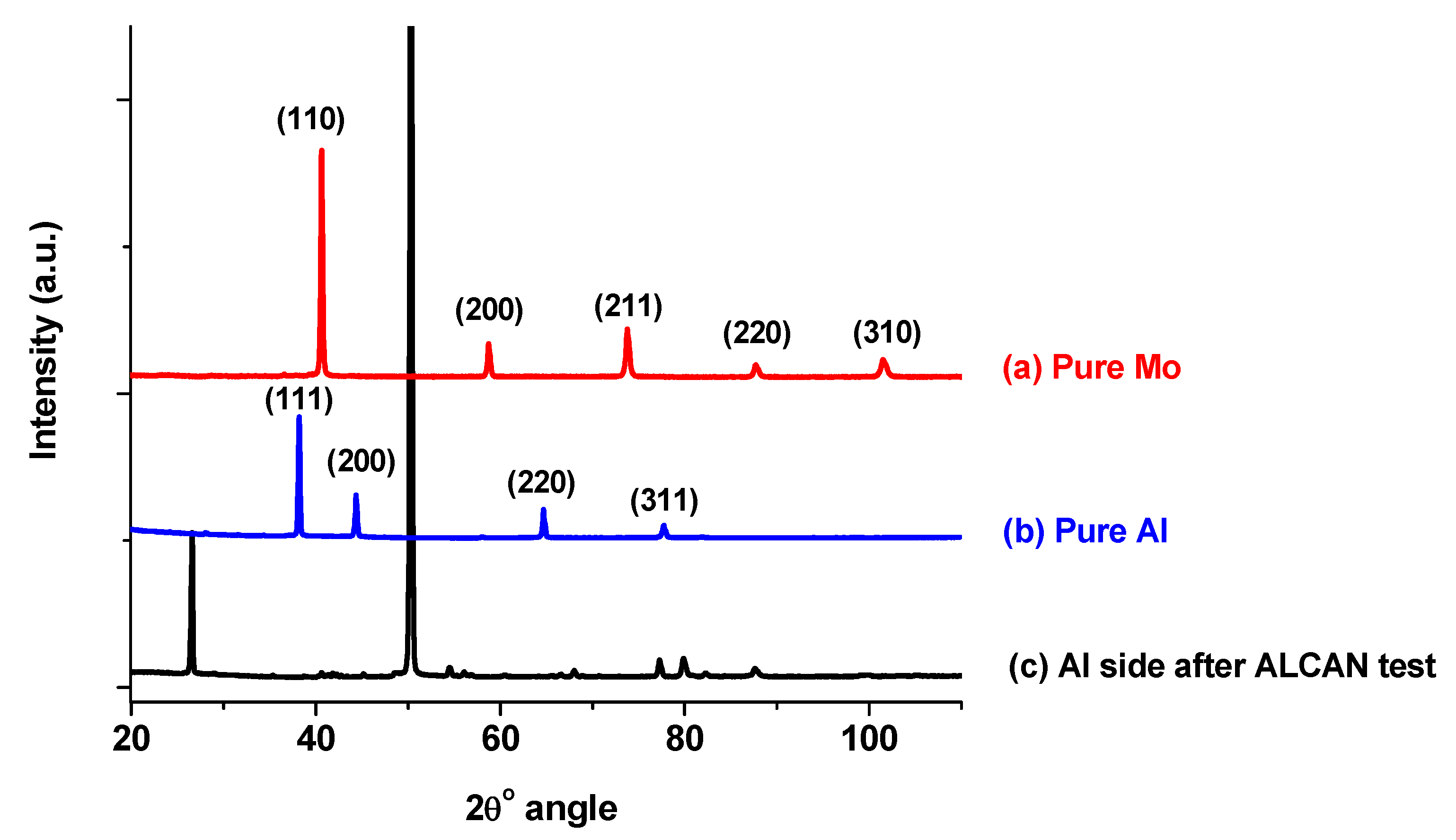
3.4.3. XRD Analysis
4. Discussion
4.1. Modelling Material Properties
4.2. The ALCAN Immersion Test and Application in the Al Industry
- (i)
- While their tests were conducted at 800 °C and below, in this work, the static ALCAN immersion test was performed at 850 °C, with only small quantities of the Al-Mo alloys being observed at the Mo(s)–{Al-Mg}(l) interface;
- (ii)
- Contrary to their case where the Al-Mo alloys formed were known, in the present work, elemental analysis identifying the type of Al-Mo alloys present at the the Mo(s)–{Al-Mg}(l) interface was difficult since the alloys were extremely thin and not cross-sectionally visible at the interface (Figure 13);
- (iii)
- The mass loss of ~20% observed in the Mo block in this work was not principally as a result of the reaction between Mo and the molten Al-Mg alloy, but rather due to intergranular corrosion at the grain boundaries chipping away at the Mo block over time;
- (iv)
- Although modelling by FactSage™ thermochemical software predicted the potential formation of Al4Mo, Al5Mo, and Al8Mo3 alloys when molten Al-Mg alloy is in contact with Mo, kinetically these alloys would form very slowly. In addition, if the alloys formed around the Mo are insoluble in the molten Al-Mg alloy, they would create an impenetrable barrier that stops further reaction with Mo, which is beneficial to the process. Since it was not possible to detect Mo in the molten Al–Mg matrix using OES elemental analysis, it confirmed that, in this work, neither Mo nor the Al-Mo alloys dissolved in the molten Al-Mg alloy as was predicted by computer simulations using Classical Molecular Dynamics in the Materials Studio® packages.
5. Conclusions
- No reaction and no cross diffusion of atoms occurs at the Mo(s)–Al(l) interface;
- Molten Al atoms form a non-wetting layer that adopts an epitaxial orientation in alignment with the exposed solid Mo crystal morphology;
- The calculated mean interfacial energy normalized per unit area of the simulated cell was found to be about 203 mJ/m2, which implies a weak van der Waals interaction between molten Al and solid Mo.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhang, X.M.; Chen, W.P. Review on corrosion-wear resistance performance of materials in molten aluminum and its alloys. Trans. Nonferrous Met. Soc. China 2015, 25, 1715–1731. [Google Scholar] [CrossRef]
- Crisci, J.-P.; Ganser, C.; Damasse, J.-M.; Schmitz, W.; Senk, D.; Stebner, G. Protective Coating Comprising Boron Nitride for Refractory Material Members of an Ingot Mold for Continuous Casting of Metals. U.S. Patent US6051058A, 18 April 2000. Available online: https://patents.google.com/patent/US6051058A/en (accessed on 20 January 2021).
- Laudenklos, M. Mold-Release Layer for Use in the Casting of Nonferrous Metals. U.S. Patent US2010/0237224 A1, 23 September 2010. Available online: https://patents.google.com/patent/US20100237224A1/en (accessed on 20 January 2021).
- Barandehfard, F.; Aluha, J.; Hekmat-ardakan, A.; Gitzhofer, F. Improving corrosion resistance of aluminosilicate refractories towards molten Al-Mg alloy using non-wetting additives: A short review. Materials 2020, 13, 4078. [Google Scholar] [CrossRef] [PubMed]
- Păduraru, L.; Nedeloni, L.; Kazamer, N.; Muntean, R.; Pascal, D.T.; Vălean, P.C.; Nedeloni, M.D. Investigations on dry sliding wear and corrosion resistance of thermal sprayed molybdenum coatings. IOP Conf. Ser. Mater. Sci. Eng. 2018, 416, 012027. [Google Scholar] [CrossRef]
- Popoola, A.; Olorunniwo, O.E.; Ige, O.O. Corrosion resistance through the application of anti-corrosion coatings. In Developments in Corrosion Protection; Aliofkhazraei, M., Ed.; InTechOpen: London, UK, 2014; pp. 241–270. [Google Scholar]
- Yurkov, A. Refractories for Aluminum: Electrolysis and the Cast House; Springer International Publishing: Moscow, Russia, 2017; ISBN 978-3-319-53589-0. [Google Scholar]
- Stansbury, E.E.; Buchanan, R.A. Fundamentals of Electrochemical Corrosion; ASM International: Materials Park, OH, USA, 2000. [Google Scholar]
- Kim, K. Formation of endogenous MgO and MgAl2O4 particles and their possibility of acting as substrate for heterogeneous nucleation of aluminum grains. Surf. Interface Anal. 2015, 47, 429–438. [Google Scholar] [CrossRef] [Green Version]
- Yan, M.; Fan, Z. Durability of materials in molten aluminum alloys. J. Mater. Sci. 2001, 6, 285–295. [Google Scholar] [CrossRef]
- Tunca, N.; Delamore, G.W.; Smith, R.W. Corrosion of Mo, Nb, Cr, and Y in molten aluminum. Metall. Trans. 1990, 21, 2919–2928. [Google Scholar] [CrossRef]
- Ling, Z.; Chen, W.; Xu, W.; Zhang, X.; Lu, T.; Liu, J. The Influence of a Mo Addition on the Interfacial Morphologies and Corrosion Resistances of Novel Fe-Cr-B Alloys Immersed in Molten Aluminum. Materials 2019, 12, 256. [Google Scholar] [CrossRef] [Green Version]
- Takahashi, J.; Ishikawa, K.; Kawakami, K.; Fujioka, M.; Kubota, N. Acta Materialia Atomic-scale study on segregation behavior at austenite grain boundaries in boron- and molybdenum-added steels. Acta Mater. 2017, 133, 41–54. [Google Scholar] [CrossRef]
- Asl, M.S.; Delbari, S.A.; Azadbeh, M.; Namini, A.S.; Mehrabian, M.; Nguyen, V.-H.; Van-Le, Q.; Shokouhimehr, M.; Mohammadi, M. Nanoindentational and conventional mechanical properties of spark plasma sintered Ti-Mo alloys. J. Mater. Res. Technol. 2020, 9, 10647–10658. [Google Scholar] [CrossRef]
- Raykhtsaum, G. PGM Highlights: Platinum Alloys: A Selective Review of the Available Literature—Johnson Matthey Technology Review. Platin. Met. Rev. 2013, 57, 202–213. [Google Scholar] [CrossRef]
- Eustathopoulos, N. Wetting by liquid metals—Application in materials processing: The contribution of the Grenoble group. Metals 2015, 5, 350–370. [Google Scholar] [CrossRef] [Green Version]
- Nikonova, R.M.; Lad, V. V Contact interaction of metal melts with fullerite. J. Mater. Process. Technol. 2020, 9, 12559–12567. [Google Scholar] [CrossRef]
- Hashim, J.; Looney, L.; Hashmi, M.S.J. The wettability of SiC particles by molten aluminium alloy. J. Mater. Process. Technol. 2001, 119, 324–328. [Google Scholar] [CrossRef]
- Okamoto, H. Al-Mo (Aluminum-Molybdenum). J. Phase Equilibria Diffus. 2010, 31, 492–493. [Google Scholar] [CrossRef]
- Bale, C.W.; Bélisle, E.; Chartrand, P.; Decterov, S.A.; Eriksson, G.; Gheribi, A.E.; Hack, K.; Jung, I.H.; Kang, Y.B.; Melançon, J.; et al. Van FactSage Thermochemical Software and Databases, 2010–2016. Calphad 2016, 54, 35–53. [Google Scholar] [CrossRef] [Green Version]
- BIOVIA Dassault Systèmes, Materials Studio. Forcite (2019); Dassault Systèmes: Vélizy-Villacoublay, France, 2020. [Google Scholar]
- Daw, M.S.; Baskes, M.I. Semiempirical, Quantum Mechanical Calculation of Hydrogen Embrittlement in Metals. Phys. Rev. Lett. 1983, 50, 1285–1288. [Google Scholar] [CrossRef]
- Daw, M.S.; Baskes, M.I. Embedded-atom method: Derivation and application to impurities, surfaces, and other defects in metals. Phys. Rev. B 1984, 29, 6443–6453. [Google Scholar] [CrossRef] [Green Version]
- Zhou, X.W.; Johnson, R.A.; Wadley, H.N.G. Misfit-energy-increasing dislocations in vapor-deposited CoFe/NiFe multilayers. Phys. Rev. B 2004, 69, 144113. [Google Scholar] [CrossRef] [Green Version]
- Andersen, H.C. Molecular dynamics simulations at constant pressure and/or temperature. J. Chem. Phys. 1980, 72, 2384–2393. [Google Scholar] [CrossRef] [Green Version]
- Leimkuhler, B.; Noorizadeh, E.; Theil, F. A Gentle Stochastic Thermostat for Molecular Dynamics. J. Stat. Phys. 2009, 135, 261–277. [Google Scholar] [CrossRef] [Green Version]
- Asadi, E.; Zaeem, M.A.; Nouranian, S.; Baskes, M.I. Two-phase solid—Liquid coexistence of Ni, Cu, and Al by molecular dynamics simulations using the modified embedded-atom method. Acta Mater. 2015, 86, 169–181. [Google Scholar] [CrossRef]
- Karavaev, A.V.; Dremov, V.V.; Pravishkina, T.A. Precise calculation of melting curves by molecular dynamics. Comput. Mater. Sci. 2016, 124, 335–343. [Google Scholar] [CrossRef]
- Sun, C.; Lee, S.; Hwang, W.; Hwang, J.; Tang, I.; Fu, Y. Surface Free Energy of Alloy Nitride Coatings Deposited Using Closed Field Unbalanced Magnetron Sputter Ion Plating Magnetron Sample. Mater. Trans. 2006, 47, 2533–2539. [Google Scholar] [CrossRef] [Green Version]
- Good, R.J.; Chaudhury, M.K. Theory of Adhesive Forces Across Interfaces. In Fundamentals of Adhesion; Lee, L.-H., Ed.; Springer: New York, NY, USA, 1991; pp. 153–172. [Google Scholar]
- Amama, P.B.; Putnam, S.A.; Barron, R.; Maruyama, B. Wetting behavior and activity of catalyst supports in carbon nanotube carpet growth. Nanoscale 2013, 2642–2646. [Google Scholar] [CrossRef]
- Fokin, V.M.; Zanotto, E.D. Crystal nucleation in silicate glasses: The temperature and size dependence of crystal/liquid surface energy. J. Non. Cryst. Solids 2000, 265, 105–112. [Google Scholar] [CrossRef] [Green Version]
- Krüss-Gmbh. So You Want to Measure Surface Energy? Krüss-Gmbh: Hamburg, Germany, 1999; pp. 1–8. [Google Scholar]
- Koshy, P. Effect of Chemical Additives on the Interfacial Phenomena of High Alumina Refractories with Al-Alloys. Ph.D. Thesis, The University of New South Wales, Sydney, Australia, 2009; p. 284. [Google Scholar]
- Afshar, S. CIREP-ALCAN: Corrosion Tests on Refractories for Aluminium Treatment Furnaces, Internal Report; CIREP Research Group, École Polytechnique: Montreal, QC, Canada, 2010; pp. 1–12. [Google Scholar]
- Allahevrdi, M.; Afshar, S.; Allaire, C. Additives and the Corrosion Resistance of Aluminosilicate Refractories in Molten Al-5Mg. J. Met. 1998, 50, 30–34. [Google Scholar] [CrossRef]
- Draining, W.D. Thermal expansion and density measurements of molten and solid materials at high temperatures by the gamma attenuation technique. AIP Rev. Sci. Instrum. 1979, 50, 1–57. [Google Scholar]
- Poirier, D.R. Density, viscosity, and diffusion coefficients in hypoeutectic Al-Si Liquid alloys: An assessment of available data. Metall. Mater. Trans. B 2014, 45, 1345–1354. [Google Scholar] [CrossRef]
- Forsyth, J.B.; Gran, G. The structure of the intermetallic phase γ(Mo-Al)-Mo3Al8. Acta Cryst. 1962, 15, 100–104. [Google Scholar] [CrossRef]
- Guisbiers, G.; José-Yacaman, M. Use of Chemical Functionalities to Control Stability of Nanoparticles. Encycl. Interfacial Chem. Surf. Sci. Electrochem. 2018, 875–885. [Google Scholar] [CrossRef]
- Che, J.G.; Chan, C.T.; Jian, W.-E.; Leung, T.C. Surface atomic structures, surface energies, and equilibrium crystal shape of molybdenum. Phys. Rev. B 1998, 57, 1875–1880. [Google Scholar] [CrossRef] [Green Version]
- Wang, T.; Tian, X.; Yang, Y.; Li, Y.; Wang, J. Structures of seven molybdenum surfaces and their coverage dependent hydrogen adsorption. Phys. Chem. Chem. Phys. 2016, 18, 6005–6012. [Google Scholar] [CrossRef]
- Hodkin, E.N.; Nicholas, M.G.; Poole, D.M. The surface energies of solid molybdenum, niobium, tantalum and tungsten. J. Less Common Met. 1970, 20, 93–103. [Google Scholar] [CrossRef]
- Allen, B.C. The surface free energy of solid molybdenum. J. Less Common Met. 1969, 17, 403–410. [Google Scholar] [CrossRef]
- Sperry, F.S. The ALCAN Standard Immersion Test. Choosing the Best Refractory Material for an Aluminum Furnace. Available online: http://fssperry.com/articles/best-refractory-material-for-an-aluminum-furnace/ (accessed on 20 January 2021).
- Saunders, N.; Thermotech Ltd. The Al-Mo system (aluminum-molybdenum). J. Phase Equilib. 1997, 18, 370–378. [Google Scholar] [CrossRef]
- Rizzoli, C.; Salamakha, P.S.; Sologub, O.L.; Bocelli, G. X-ray investigation of the Al–B–N ternary system: Isothermal section at 1500 °C: Crystal structure of the Al0.185B6CN0.256 compound. J. Alloys Compd. 2002, 343, 135–141. [Google Scholar] [CrossRef]
- Swanson, H.E.; Tatge, E. Standard X-ray Diffraction Powder Patterns. Natl. Bur. Stand. 1953, I, 43. [Google Scholar]
- Yuan, S.; Lin, N.; Zou, J.; Liu, Z.; Yu, Y.; Ma, Y. Manipulation tribological behavior of Ti6Al4V alloy via a duplex treatment of double glow plasma surface molybdenizing-laser surface texturing (LST). J. Mater. Res. Technol. 2020, 9, 6360–6375. [Google Scholar] [CrossRef]
- Ayadi, S.; Hadji, A.; Hakan, K.; Selman, D. Microstructure and wear behavior of a Cr-Mo-Nb alloyed manganese steel. J. Mater. Res. Technol. 2020, 9, 11545–11562. [Google Scholar] [CrossRef]
- Zhou, H.; Zhang, Y.; Hua, X.; Yang, Z. High-temperature anti-wear behavior of alumina-reinforced Ti-Zr-Mo alloy composites. Wear 2014, 319, 184–190. [Google Scholar] [CrossRef]
- Kalin, M.; Polajnar, M. The wetting of steel, DLC coatings, ceramics and polymers with oils and water: The importance and correlations of surface energy, surface tension, contact angle and spreading. Appl. Surf. Sci. 2014, 293, 97–108. [Google Scholar] [CrossRef]
- Rieke, P.C. Application of Van Oss-Chaudhury-Good theory of wettability to interpretation of interracial free energies of heterogeneous nucleation. J. Cryst. Growth 1997, 182, 472–484. [Google Scholar] [CrossRef]
- Gaur, A.P.S.; Sahoo, S.; Ahmadi, M.; Dash, S.P.; Guinel, M.J.; Katiyar, R.S. Surface energy engineering for tunable wettability through controlled synthesis of MoS2. Nano Lett. 2014, 14, 4314–4321. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rouquerol, J.; Rouquerol, F.; Sing, K.S.W.; Llewellyn, P.; Maurin, G. Adsorption by Powders and Porous Solids: Principles, Methodology and Applications, 2nd ed.; Academic Press: Oxford, UK, 2014; ISBN 0080970362/9780080970363. [Google Scholar]
- Wang, K.; Chang, B.; Chen, J.; Fu, H.; Lin, Y.; Lei, Y. Effect of molybdenum on the microstructures and properties of stainless steel coatings by laser cladding. Appl. Sci. 2017, 7, 1065. [Google Scholar] [CrossRef]
- Alekseeva, E.; Karasev, A.; Jönsson, P.G.; Alkhimenko, A. Effect of inclusions on the corrosion properties of the nickel-based alloys 718 and ep718. Metals 2020, 10, 1177. [Google Scholar] [CrossRef]
- Luo, H.; Wu, J.; Liu, Z.; Chen, W. Corrosion behavior of Molybdenum in molten aluminum. Appl. Mech. Mater. 2012, 155–156, 969–973. [Google Scholar] [CrossRef]






| Lattice Parameters | Before Melting | After Melting |
|---|---|---|
| a | 80.99 Å | 64.19 Å |
| b | 80.99 Å | 62.00 Å |
| c | 161.98 Å | 291.28 Å |
| α | 90° | 89.29° |
| β | 90° | 100.02° |
| γ | 90° | 84.09° |
| Metal Bonds | Bond Lengths (Å) | |
|---|---|---|
| Simulated Values (This Work) | Actual Data Values | |
| Al-Al | 3.0 | 2.79 |
| Al-Mo | 2.7 | 2.76 |
| Mo-Mo | 2.7–3.1 | 2.09–3.27 |
| Sample | Before Test | After Test | Loss (%) |
|---|---|---|---|
| Diameter (mm) | 44.28 | 41.27 | 6.80% |
| Height (mm) | 25.44 | 24.78 | 2.59% |
| Mass (g) | 396.30 | 315.13 | 20.48% |
| Sampling | Al | Mg | Mo | Mn | Si | Fe | Total * |
|---|---|---|---|---|---|---|---|
| Before ALCAN test | 94.2 | 4.60 | 0.005 | 0.320 | 0.220 | 0.160 | 99.545 |
| After ALCAN test | 95.700 | 2.890 | 0.012 | 0.370 | 0.360 | 0.160 | 99.492 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gitzhofer, F.; Aluha, J.; Langlois, P.-O.; Barandehfard, F.; Ntho, T.A.; Abatzoglou, N. Proven Anti-Wetting Properties of Molybdenum Tested for High-Temperature Corrosion-Resistance with Potential Application in the Aluminum Industry. Materials 2021, 14, 5355. https://doi.org/10.3390/ma14185355
Gitzhofer F, Aluha J, Langlois P-O, Barandehfard F, Ntho TA, Abatzoglou N. Proven Anti-Wetting Properties of Molybdenum Tested for High-Temperature Corrosion-Resistance with Potential Application in the Aluminum Industry. Materials. 2021; 14(18):5355. https://doi.org/10.3390/ma14185355
Chicago/Turabian StyleGitzhofer, François, James Aluha, Pierre-Olivier Langlois, Faranak Barandehfard, Thabang A. Ntho, and Nicolas Abatzoglou. 2021. "Proven Anti-Wetting Properties of Molybdenum Tested for High-Temperature Corrosion-Resistance with Potential Application in the Aluminum Industry" Materials 14, no. 18: 5355. https://doi.org/10.3390/ma14185355
APA StyleGitzhofer, F., Aluha, J., Langlois, P.-O., Barandehfard, F., Ntho, T. A., & Abatzoglou, N. (2021). Proven Anti-Wetting Properties of Molybdenum Tested for High-Temperature Corrosion-Resistance with Potential Application in the Aluminum Industry. Materials, 14(18), 5355. https://doi.org/10.3390/ma14185355







