Modified Grape Seeds: A Promising Alternative for Nitrate Removal from Water
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Preparation of MGS
2.3. Characterization of MGS
2.4. Batch Experiments
- (a)
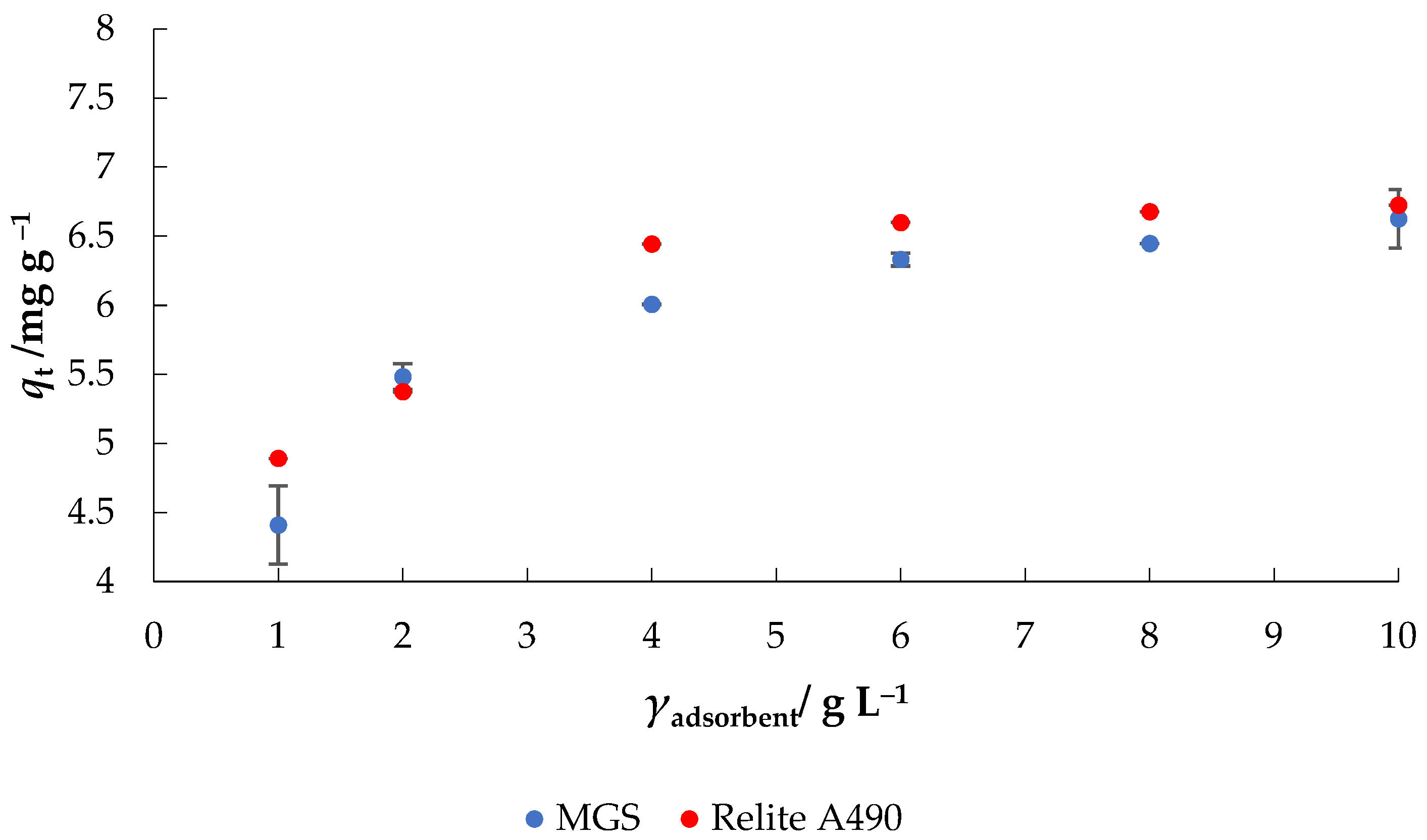
- Effect of initial MGS or Relite A490 concentration: γ0 = 30 mg L−1, γadsorbent = 1–10 g L−1, V = 50 mL, pH = native (6.3), T = 25 °C, t = 120 min, v = 130 rpm.
- (b)
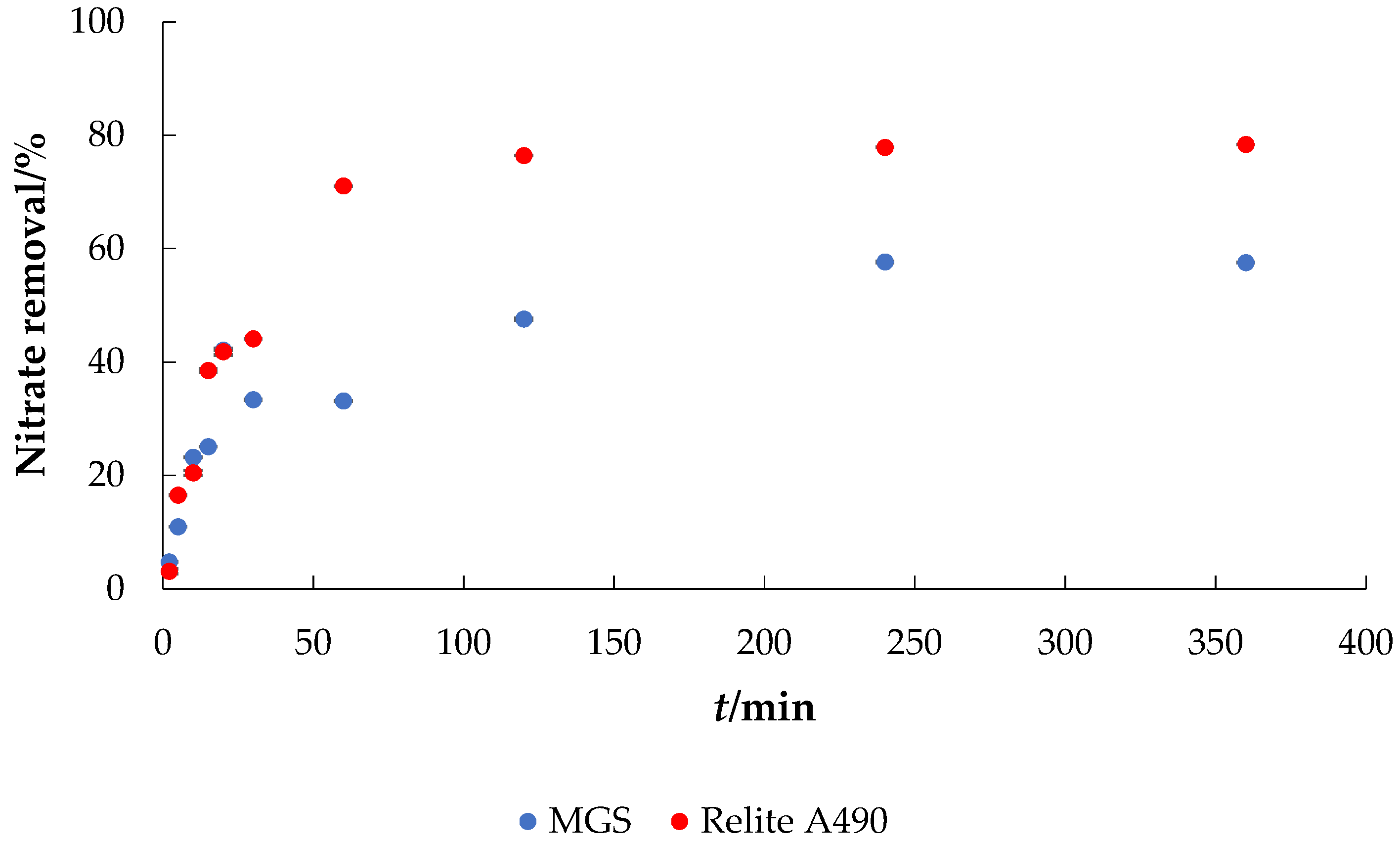
- Effect of contact time: γ0 = 30 mg L−1, γadsorbent = 4 g L−1, V = 50 mL, pH = native (6.3), T = 25 °C, t = 2–360 min, v = 130 rpm.
- (c)
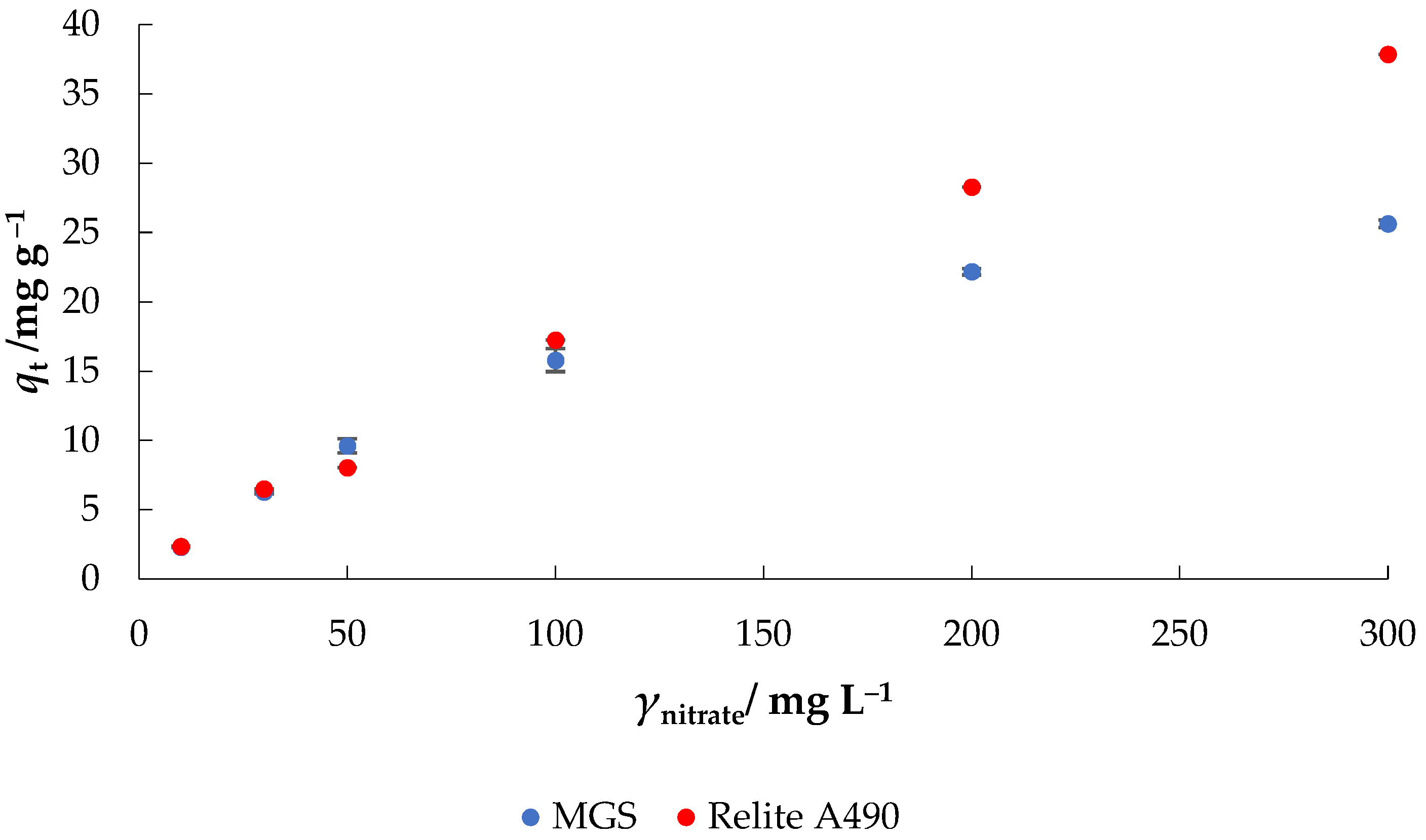
- Effect of initial nitrate concentration: γ0 = 10–300 mg L−1, γadsorbent = 4 g L−1, V = 50 mL, pH = native (6.3), T = 25 °C, t = 120 min, v = 130 rpm.
- (d)
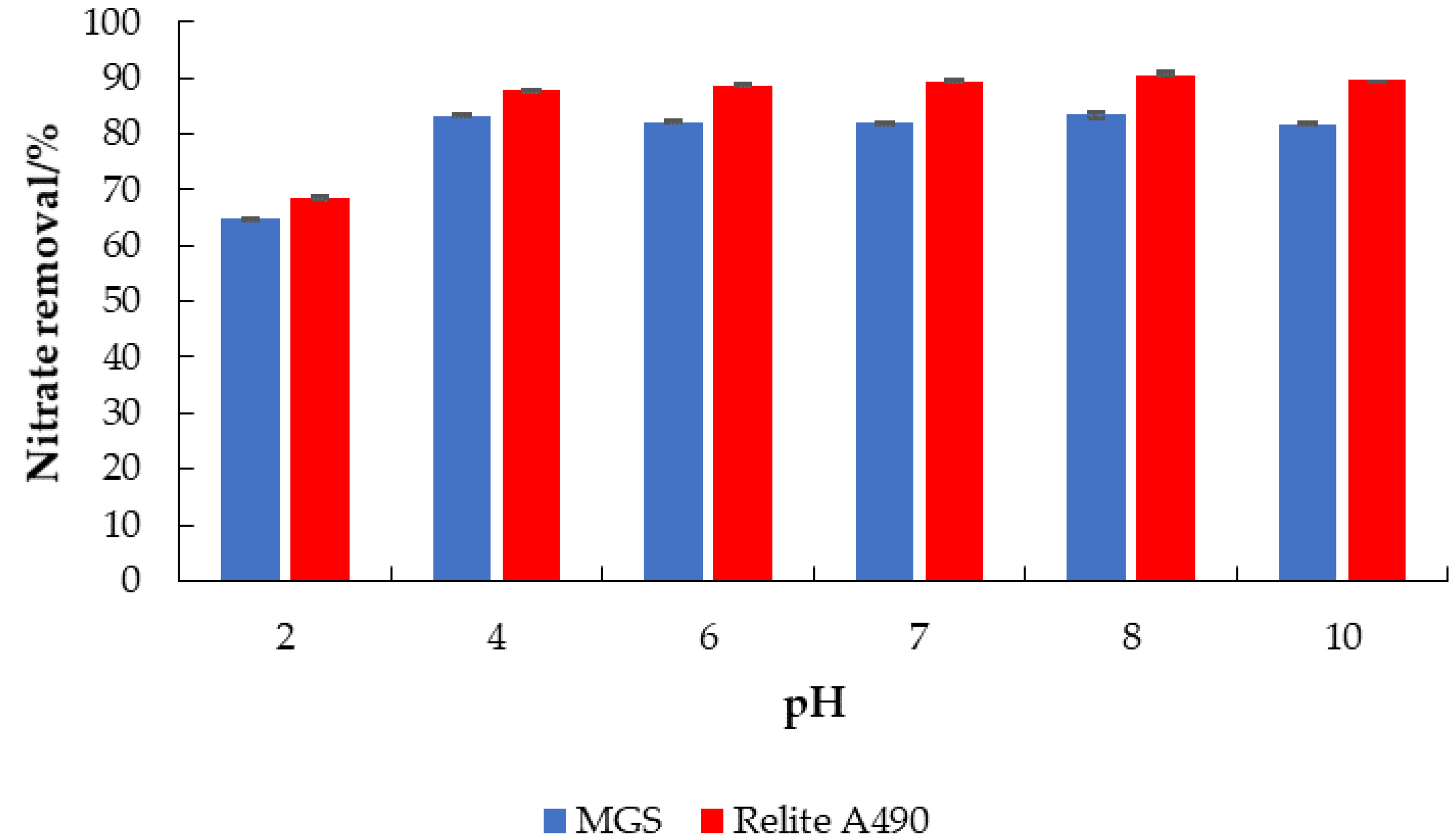
- Effect of initial pH: γ0 = 30 mg L−1, γadsorbent = 4 g L−1, V = 50 mL, pH = 2, 4, 6, 7, 8, 10, T = 25 °C, t = 120 min, v = 130 rpm.
- (e)
- Effect of contact time (synthetic wastewater): γ0 = 30 mg L−1, γadsorbent = 4 g L−1, V = 50 mL, pH = native (7.2), T = 25 °C, t = 2–360 min, v = 130 rpm.
2.5. Column Experiments
3. Results and Discussion
3.1. Characterization of MGS
3.2. Applications of MGS for the Removal of Nitrate from Water
3.3. Adsorption of Nitrate onto MGS from Real Wastewater
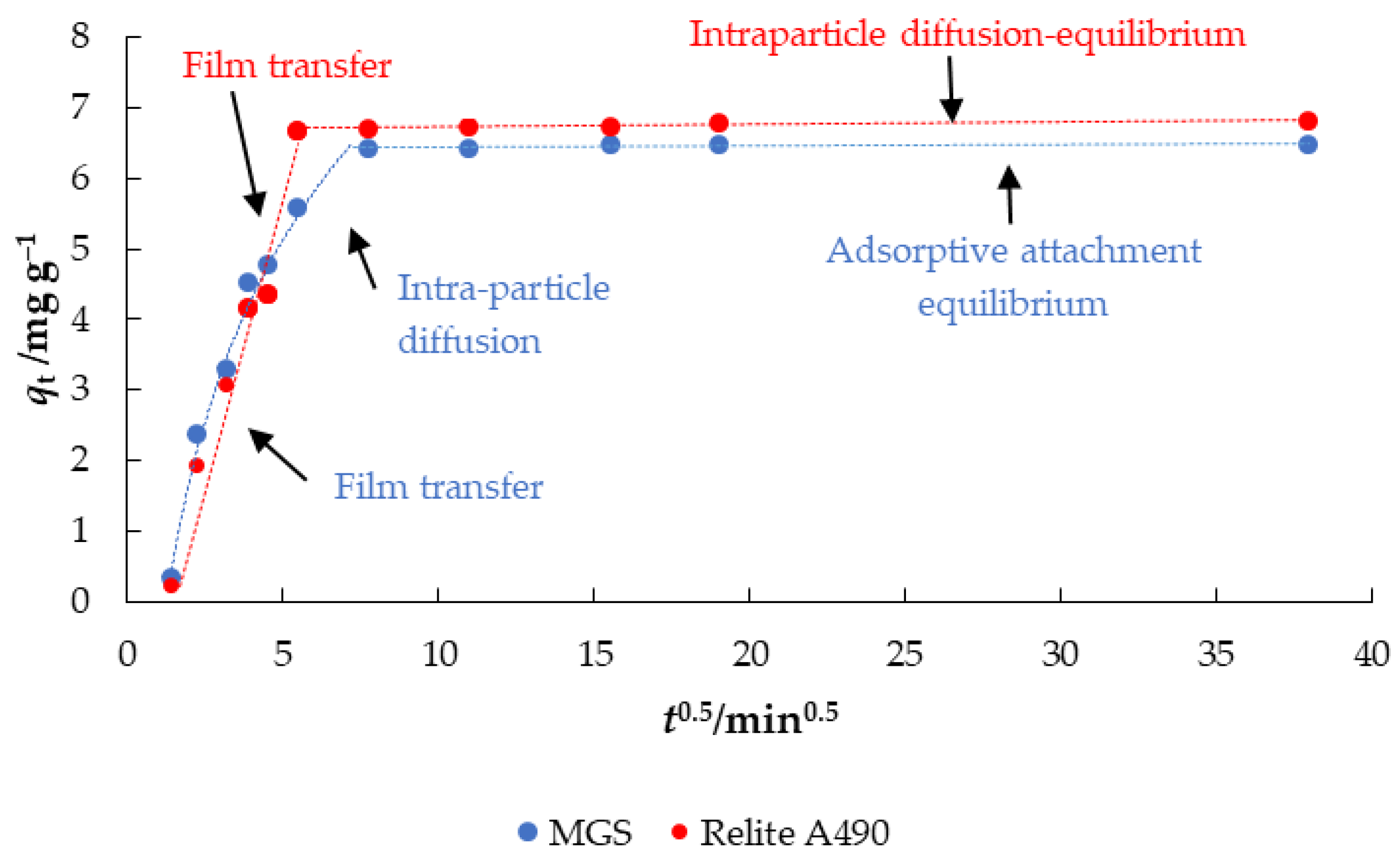
3.4. Adsorption Kinetics
3.5. Adsorption Isotherms
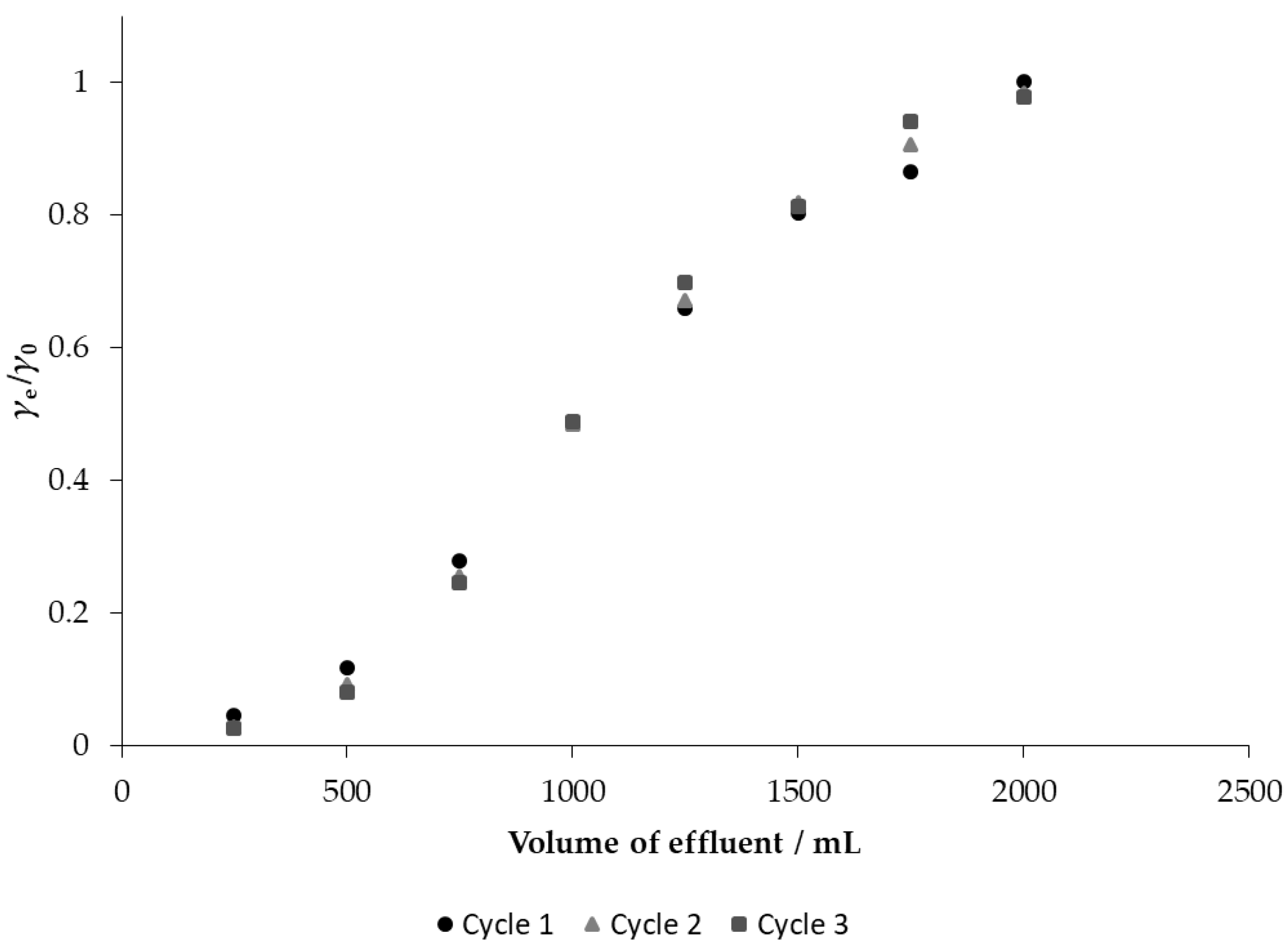
3.6. Breakthrough and Desorption Studies
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chauhan, K.; Kaur, J.; Singh, P.; Sharma, P.; Sharma, P.; Chauhan, G.S. An Efficient and Regenerable Quaternary Starch for Removal of Nitrate from Aqueous Solutions. Ind. Eng. Chem. Res. 2016, 55, 2507–2519. [Google Scholar] [CrossRef]
- Filipović, V.; Petošić, D.; Nakić, Z.; Bubalo, M. Prisutnost nitrata u podzemnim vodama. Hrvat. Vode 2013, 21, 119–128. [Google Scholar]
- Nujić, M.; Habuda-Stanić, M. Nitrates and nitrites, metabolism and toxicity. Food Health Dis. 2017, 6, 63–73. [Google Scholar]
- Olgun, A.; Atar, N.; Wang, S. Batch and column studies of phosphate and nitrate adsorption on waste solids containing boron impurity. Chem. Eng. J. 2013, 222, 108–119. [Google Scholar] [CrossRef] [Green Version]
- Satayeva, A.R.; Howell, C.A.; Korobeinyk, A.V.; Jandosov, J.; Inglezakis, V.J.; Mansurov, Z.A.; Mikhalovsky, S.V. Investigation of rice husk derived activated carbon for removal of nitrate contamination from water. Sci. Total Environ. 2018, 630, 1237–1245. [Google Scholar] [CrossRef]
- Chen, H.; Liu, S.; Liu, T.; Yuan, Z.; Guo, J. Efficient nitrate removal from synthetic groundwater via in situ utilization of short-chain fatty acids from methane bioconversion. Chem. Eng. J. 2020, 393, 124594. [Google Scholar] [CrossRef]
- Schoeman, J.J.; Steyn, A. Nitrate removal with reverse osmosis in a rural area in South Africa. Desalination 2003, 155, 15–26. [Google Scholar] [CrossRef]
- Öztürk, N.; Bektaş, T.E. Nitrate removal from aqueous solution by adsorption onto various materials. J. Hazard. Mater. 2004, 112, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Bi, J.; Peng, C.; Xu, H.; Ahmed, A.-S. Removal of nitrate from groundwater using the technology of electrodialysis and electrodeionization. Desalin. Water Treat. 2011, 34, 394–401. [Google Scholar] [CrossRef]
- Naushad, M.; Khan, M.A.; ALOthman, Z.A.; Khan, M.R. Adsorptive removal of nitrate from synthetic and commercially available bottled water samples using De-Acidite FF-IP resin. J. Ind. Eng. Chem. 2014, 20, 3400–3407. [Google Scholar] [CrossRef]
- Naushad, M.; Ahamad, T.; Al-Maswari, B.M.; Abdullah Alqadami, A.; Alshehri, S.M. Nickel ferrite bearing nitrogen-doped mesoporous carbon as efficient adsorbent for the removal of highly toxic metal ion from aqueous medium. Chem. Eng. J. 2017, 330, 1351–1360. [Google Scholar] [CrossRef]
- Li, Y.; Taggart, M.A.; McKenzie, C.; Zhang, Z.; Lu, Y.; Pap, S.; Gibb, S. Utilizing low-cost natural waste for the removal of pharmaceuticals from water: Mechanisms, isotherms and kinetics at low concentrations. J. Clean. Prod. 2019, 227, 88–97. [Google Scholar] [CrossRef] [Green Version]
- Keränen, A.; Leiviskä, T.; Hormi, O.; Tanskanen, J. Preparation of cationized pine sawdust for nitrate removal: Optimization of reaction conditions. J. Environ. Manag. 2015, 160, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Abdolali, A.; Guo, W.S.; Ngo, H.H.; Chen, S.S.; Nguyen, N.C.; Tung, K.L. Typical lignocellulosic wastes and by-products for biosorption process in water and wastewater treatment: A critical review. Bioresour. Technol. 2014, 160, 57–66. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.Y.; Yue, Q.Y.; Xu, X.; Gao, B.Y.; Zhang, J.; Li, Q.; Xu, J.T. Optimized conditions in preparation of giant reed quaternary amino anion exchanger for phosphate removal. Chem. Eng. J. 2010, 157, 161–167. [Google Scholar] [CrossRef]
- Yang, W.; Wang, J.; Shi, X.; Tang, H.; Wang, X.; Wang, S.; Zhang, W.; Lu, J. Preferential Nitrate Removal from Water Using a New Recyclable Polystyrene Adsorbent Functionalized with Triethylamine Groups. Ind. Eng. Chem. Res. 2020, 59, 5194–5201. [Google Scholar] [CrossRef]
- Gu, B.; Ku, Y.K.; Jardine, P.M. Sorption and binary exchange of nitrate, sulfate, and uranium on an anion-exchange resin. Environ. Sci. Technol. 2004, 38, 3184–3188. [Google Scholar] [CrossRef]
- OECD. Guideline for Testing of Chemicals; Test Guidline 302 B; OECD: Paris, France, 1992. [Google Scholar]
- Stjepanović, M.; Velić, N.; Lončarić, A.; Gašo-Sokač, D.; Bušić, V.; Habuda-Stanić, M. Adsorptive removal of nitrate from wastewater using modified lignocellulosic waste material. J. Mol. Liq. 2019, 285, 535–544. [Google Scholar] [CrossRef]
- Keränen, A.; Leiviskä, T.; Gao, B.Y.; Hormi, O.; Tanskanen, J. Preparation of novel anion exchangers from pine sawdust and bark, spruce bark, birch bark and peat for the removal of nitrate. Chem. Eng. Sci. 2013, 98, 59–68. [Google Scholar] [CrossRef]
- Naushad, M.; Sharma, G.; Kumar, A.; Sharma, S.; Ghfar, A.A.; Bhatnagar, A.; Stadler, F.J.; Khan, M.R. Efficient removal of toxic phosphate anions from aqueous environment using pectin based quaternary amino anion exchanger. Int. J. Biol. Macromol. 2018, 106, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karachalios, A.; Wazne, M. Nitrate removal from water by quaternized pine bark using choline based ionic liquid analogue. J. Chem. Technol. Biotechnol. 2013, 88, 664–671. [Google Scholar] [CrossRef] [Green Version]
- Cao, W.; Dang, Z.; Zhou, X.Q.; Yi, X.Y.; Wu, P.X.; Zhu, N.W.; Lu, G.N. Removal of sulphate from aqueous solution using modified rice straw: Preparation, characterization and adsorption performance. Carbohydr. Polym. 2011, 85, 571–577. [Google Scholar] [CrossRef]
- Gao, B.-Y.; Xu, X.; Wang, Y.; Yue, Q.-Y.; Xu, X.-M. Preparation and characteristics of quaternary amino anion exchanger from wheat residue. J. Hazard. Mater. 2009, 165, 461–468. [Google Scholar] [CrossRef] [PubMed]
- Kalaruban, M.; Loganathan, P.; Shim, W.G.; Kandasamy, J.; Naidu, G.; Nguyen, T.V.; Vigneswaran, S. Removing nitrate from water using iron-modified Dowex 21K XLT ion exchange resin: Batch and fluidised-bed adsorption studies. Sep. Purif. Technol. 2016, 158, 62–70. [Google Scholar] [CrossRef]
- Kalaruban, M.; Loganathan, P.; Shim, W.G.; Kandasamy, J.; Ngo, H.H.; Vigneswaran, S. Enhanced removal of nitrate from water using amine-grafted agricultural wastes. Sci. Total Environ. 2016, 565, 503–510. [Google Scholar] [CrossRef] [PubMed]
- Hekmatzadeh, A.A.; Karimi-Jashni, A.; Talebbeydokhti, N.; Kløve, B. Adsorption kinetics of nitrate ions on ion exchange resin. Desalination 2013, 326, 125–134. [Google Scholar] [CrossRef]
- Sowmya, A.; Meenakshi, S. A novel quaternized chitosan–melamine–glutaraldehyde resin for the removal of nitrate and phosphate anions. Int. J. Biol. Macromol. 2014, 64, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Namasivayam, C.; Sangeetha, D. Removal and recovery of nitrate from water by ZnCl2 activated carbon from coconut coir pith, an agricultural solid waste. Indian J. Chem. Technol. 2005, 12, 513–521. [Google Scholar] [CrossRef]
- Sowmya, A.; Meenakshi, S. An efficient and regenerable quaternary amine modified chitosan beads for the removal of nitrate and phosphate anions. J. Environ. Chem. Eng. 2013, 1, 906–915. [Google Scholar] [CrossRef]
- Shojaipour, M.; Ghaemy, M.; Amininasab, S.M. Removal of NO3− ions from water using bioadsorbent based on gum tragacanth carbohydrate biopolymer. Carbohydr. Polym. 2020, 227, 115367. [Google Scholar] [CrossRef]
- Kim, J.; Benjamin, M.M. Modeling a novel ion exchange process for arsenic and nitrate removal. Water Res. 2004, 38, 2053–2062. [Google Scholar] [CrossRef]
- Banu, H.T.; Meenakshi, S. Synthesis of a novel quaternized form of melamine–formaldehyde resin for the removal of nitrate from water. J. Water Process Eng. 2017, 16, 81–89. [Google Scholar] [CrossRef]
- Hamoudi, S.; Belkacemi, K. Adsorption of nitrate and phosphate ions from aqueous solutions using organically-functionalized silica materials: Kinetic modeling. Fuel 2013, 110, 107–113. [Google Scholar] [CrossRef]
- Ghanim, A.N.; Ajjam, S.K. Kinetic Modelling of Nitrate Removal from Aqueous Solution during Electrocoagulation. Civ. Environ. Res. 2013, 3, 64–73. [Google Scholar]
- Alahabadi, A.; Hosseini-Bandegharaei, A.; Moussavi, G.; Amin, B.; Rastegar, A.; Karimi-Sani, H.; Fattahi, M.; Miri, M. Comparing adsorption properties of NH4Cl-modified activated carbon towards chlortetracycline antibiotic with those of commercial activated carbon. J. Mol. Liq. 2017, 232, 367–381. [Google Scholar] [CrossRef]
- Simonin, J. On the comparison of pseudo-first order and pseudo-second order rate laws in the modeling of adsorption kinetics. Chem. Eng. J. 2016, 300, 254–263. [Google Scholar] [CrossRef] [Green Version]
- Ho, Y.S.; McKay, G. Pseudo-second order model for sorption processes. Process Biochem. 1999, 34, 451–465. [Google Scholar] [CrossRef]
- Weber, W.J., Jr. Evolution of a Technology. J. Environ. Eng. 1984, 110, 899–917. [Google Scholar] [CrossRef]
- Weber, W.J.; Morris, J.C. Kinetics of adsorption on carbon from solution. J. Sanit. Eng. Div. 1963, 2, 31–60. [Google Scholar] [CrossRef]
- Stjepanović, M.; Velić, N.; Galić, A.; Kosović, I.; Jakovljević, T.; Habuda-Stanić, M. From waste to biosorbent: Removal of congo red from water by waste wood biomass. Water (Switzerland) 2021, 13, 279. [Google Scholar] [CrossRef]
- Foo, K.Y.; Hameed, B.H. Insights into the modeling of adsorption isotherm systems. Chem. Eng. J. 2010, 156, 2–10. [Google Scholar] [CrossRef]
- Ogata, F.; Imai, D.; Kawasaki, N. Adsorption of nitrate and nitrite ions onto carbonaceous material produced from soybean in a binary solution system. J. Environ. Chem. Eng. 2015, 3, 155–161. [Google Scholar] [CrossRef]
- Naushad, M.; Ahamad, T.; Sharma, G.; Al-Muhtaseb, A.H.; Albadarin, A.B.; Alam, M.M.; ALOthman, Z.A.; Alshehri, S.M.; Ghfar, A.A. Synthesis and characterization of a new starch/SnO2 nanocomposite for efficient adsorption of toxic Hg2+ metal ion. Chem. Eng. J. 2016, 300, 306–316. [Google Scholar] [CrossRef]


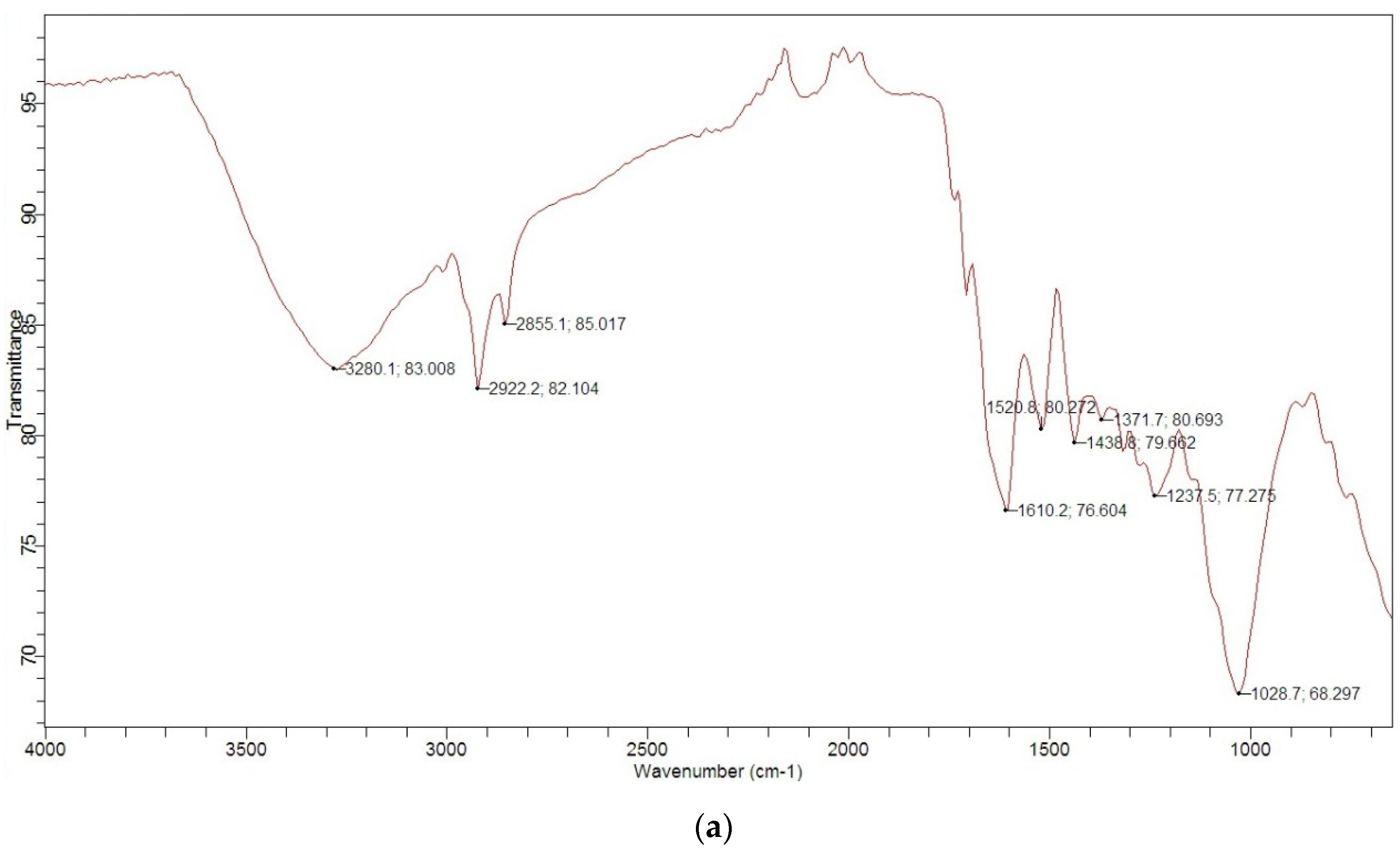
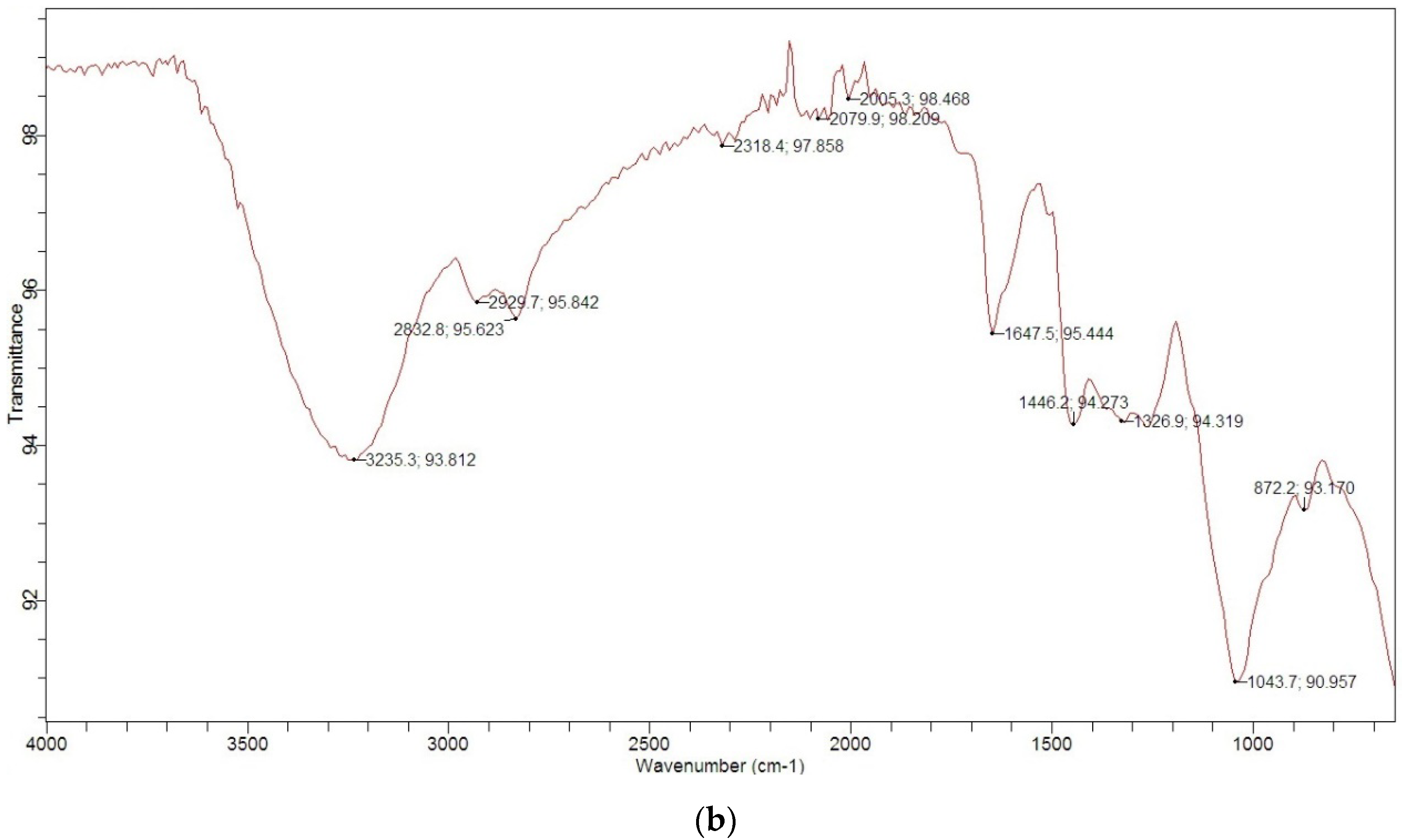


| Typical Characteristics | |
|---|---|
| Matrix | Porous copolymer styrene-DVB |
| Functional group | Quaternary ammonium group |
| Color and physical form | Light yellowish opaque beads |
| Particle size range | 0.3–1.18 mm |
| Effective size | 0.4 mm |
| Uniformity coefficient | 1.7 max |
| Ionic form | Cl− |
| Total exchange capacity | 0.9 min eq/L |
| Water retention | 46–54% |
| Chemical stability | Stable in the whole pH range |
| Thermal stability | 100 °C max (Cl− form) |
| Parameter % Mass | GS | MGS |
|---|---|---|
| C | 48.61 | 45.10 |
| H | 6.11 | 9.01 |
| N | 2.03 | 9.83 |
| Kinetic Model | Formula | Reference |
|---|---|---|
| Pseudo-first order | . | [37] |
| where qe is the amount of dye adsorbed at equilibrium, qt (mg g−1) is the amount of nitrate adsorbed at time t (min) and k1 (min−1) is the pseudo-first-order rate constant. | ||
| Pseudo-second order | . | [38] |
| where k2 (g mg−1 min−1) is the pseudo-second-order rate constant | ||
| Elovich | [36] | |
| where α (mg g−1 min−1) is Elovich constant indicative of the initial adsorption rate, β (mg g−1) is Elovich constant indicative of the desorption constant |
| Kinetic model | Parameter | Adsorbent | |
|---|---|---|---|
| MGS | Relite A490 | ||
| qe exp (mg g−1) | 6.488 | 6.805 | |
| Pseudo-first order | qe cal (mg g−1) | 6.451 | 6.847 |
| k1 (min−1) | 0.073 | 0.062 | |
| R2 | 0.987 | 0.974 | |
| Pseudo-second order | qe cal (mg g−1) | 6.863 | 7.304 |
| k1 (g mg−1 min−1) | 0.016 | 0.012 | |
| R2 | 0.969 | 0.935 | |
| Elovich | α (mg g−1 min−1) | 4.618 | 3.115 |
| β (mg g−1) | 1.114 | 0.990 | |
| R2 | 0.763 | 0.745 | |
| Intraparticle Diffusion Model | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Parameter | |||||||||
| Adsorbent | ki1 * | C1 | R12 | ki2 * | C2 | R22 | ki3 * | C3 | R32 |
| MGS | 1.6787 | <0 | 0.9431 | 0.6744 | 1.855 | 0.9758 | 2·10−3 | 6.4193 | 0.6496 |
| Relite A490 | 1.4737 | <0 | 0.9776 | 3.3·10−3 | 6.6936 | 0.7043 | N/A | N/A | N/A |
| Isotherm Model | MGS | Relite A490 |
|---|---|---|
| qm exp./mg g−1 | 25.626 | 37.910 |
| Langmuir | ||
| qm cal./mg g−1 | 27.468 | 54.183 |
| KL/L mg−1 | 0.043 | 0.015 |
| RL | 0.074 | 0.137 |
| R2 | 0.981 | 0.971 |
| MSE | 1.353 | 4.679 |
| RMSE | 1.163 | 2.163 |
| Freundlich | ||
| Kf/(mg g−1 (L/mg)1/n) | 3.756 | 2.752 |
| n | 2.688 | 1.885 |
| R2 | 0.991 | 0.988 |
| MSE | 0.661 | 1.919 |
| RMSE | 0.813 | 1.385 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stjepanović, M.; Velić, N.; Habuda-Stanić, M. Modified Grape Seeds: A Promising Alternative for Nitrate Removal from Water. Materials 2021, 14, 4791. https://doi.org/10.3390/ma14174791
Stjepanović M, Velić N, Habuda-Stanić M. Modified Grape Seeds: A Promising Alternative for Nitrate Removal from Water. Materials. 2021; 14(17):4791. https://doi.org/10.3390/ma14174791
Chicago/Turabian StyleStjepanović, Marija, Natalija Velić, and Mirna Habuda-Stanić. 2021. "Modified Grape Seeds: A Promising Alternative for Nitrate Removal from Water" Materials 14, no. 17: 4791. https://doi.org/10.3390/ma14174791
APA StyleStjepanović, M., Velić, N., & Habuda-Stanić, M. (2021). Modified Grape Seeds: A Promising Alternative for Nitrate Removal from Water. Materials, 14(17), 4791. https://doi.org/10.3390/ma14174791








