One-Pot Synthesis of Alumina-Titanium Diboride Composite Powder at Low Temperature
Abstract
:1. Introduction
2. Materials and Methods
3. Results and Preliminary Discussion
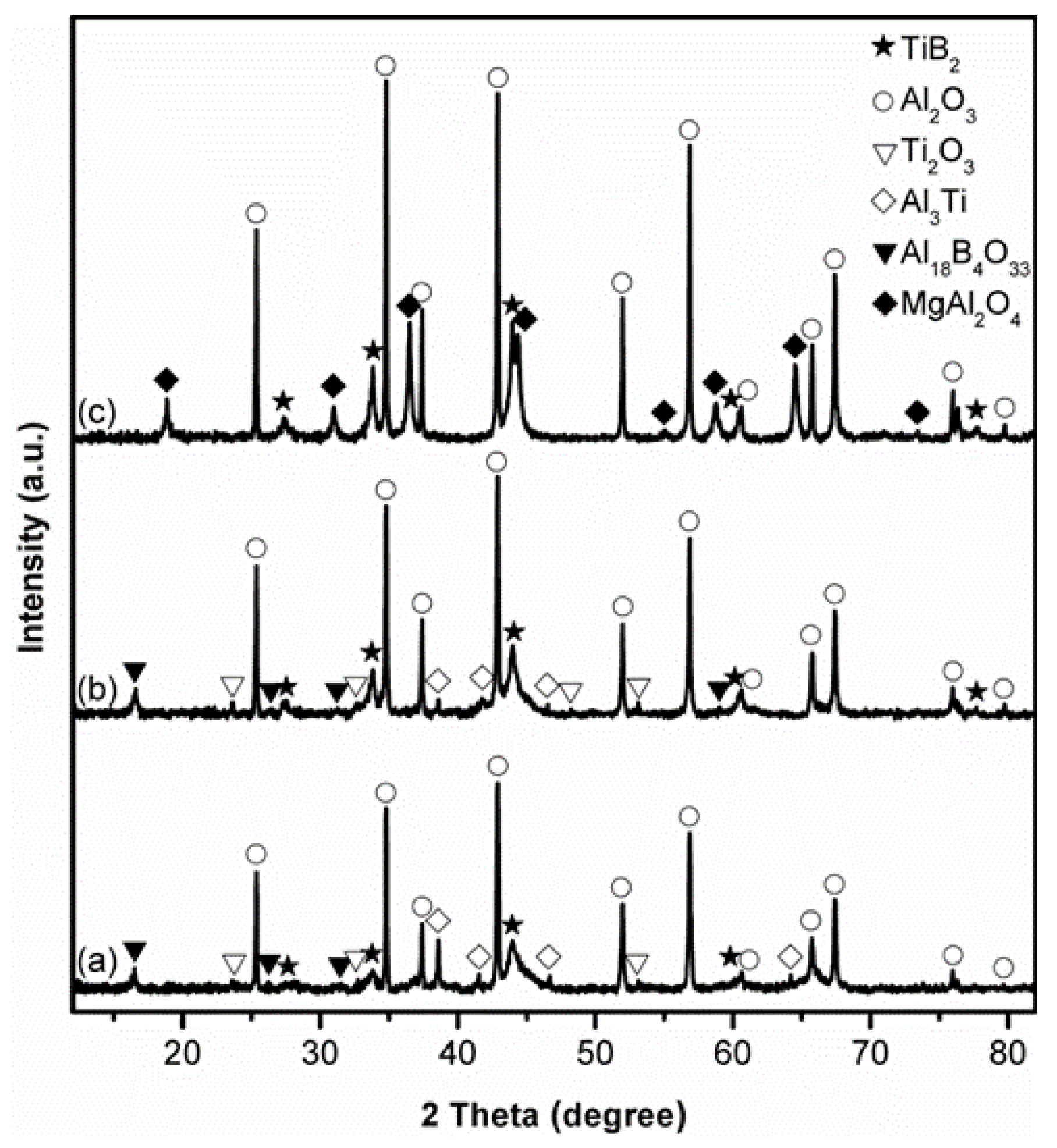
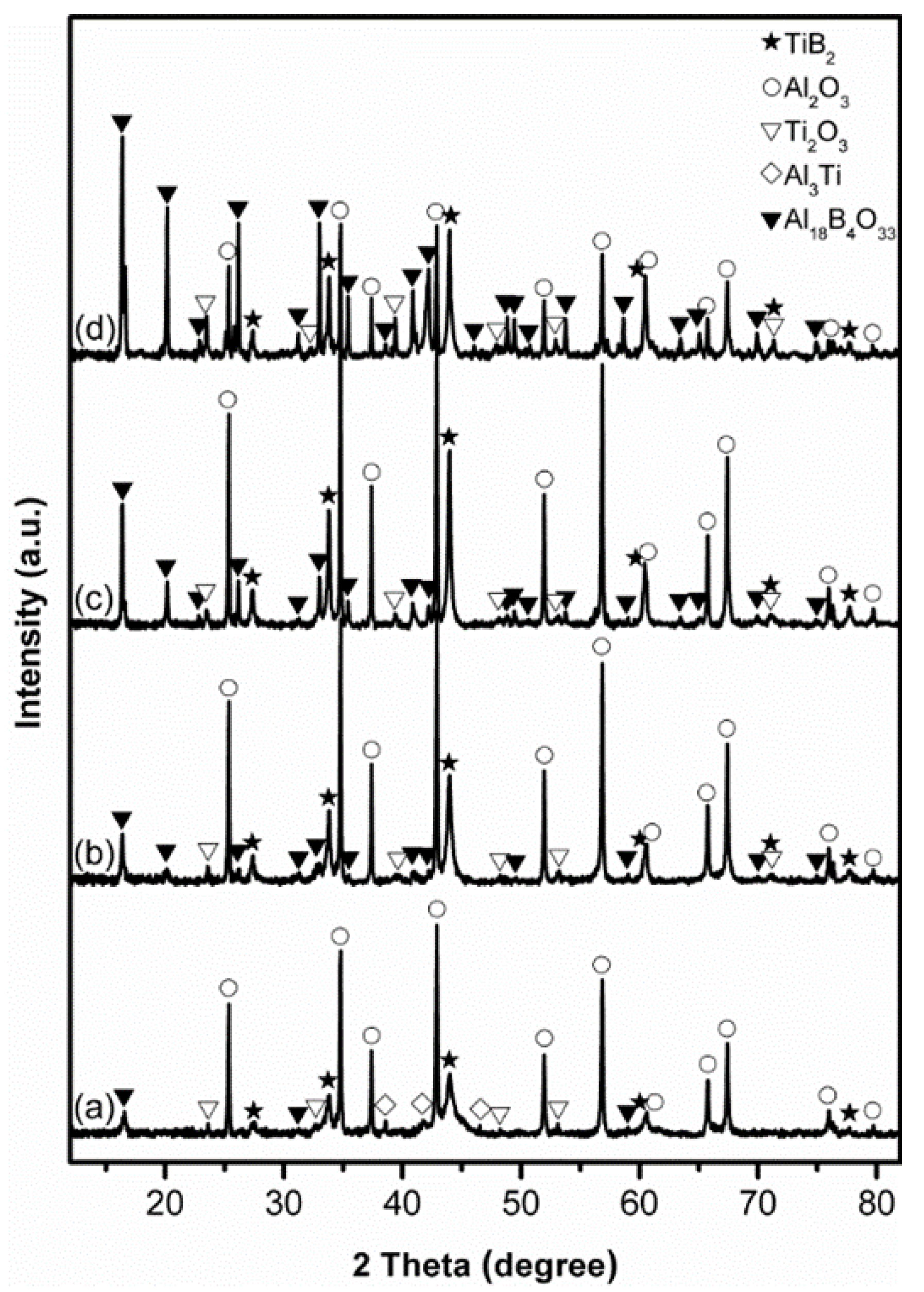
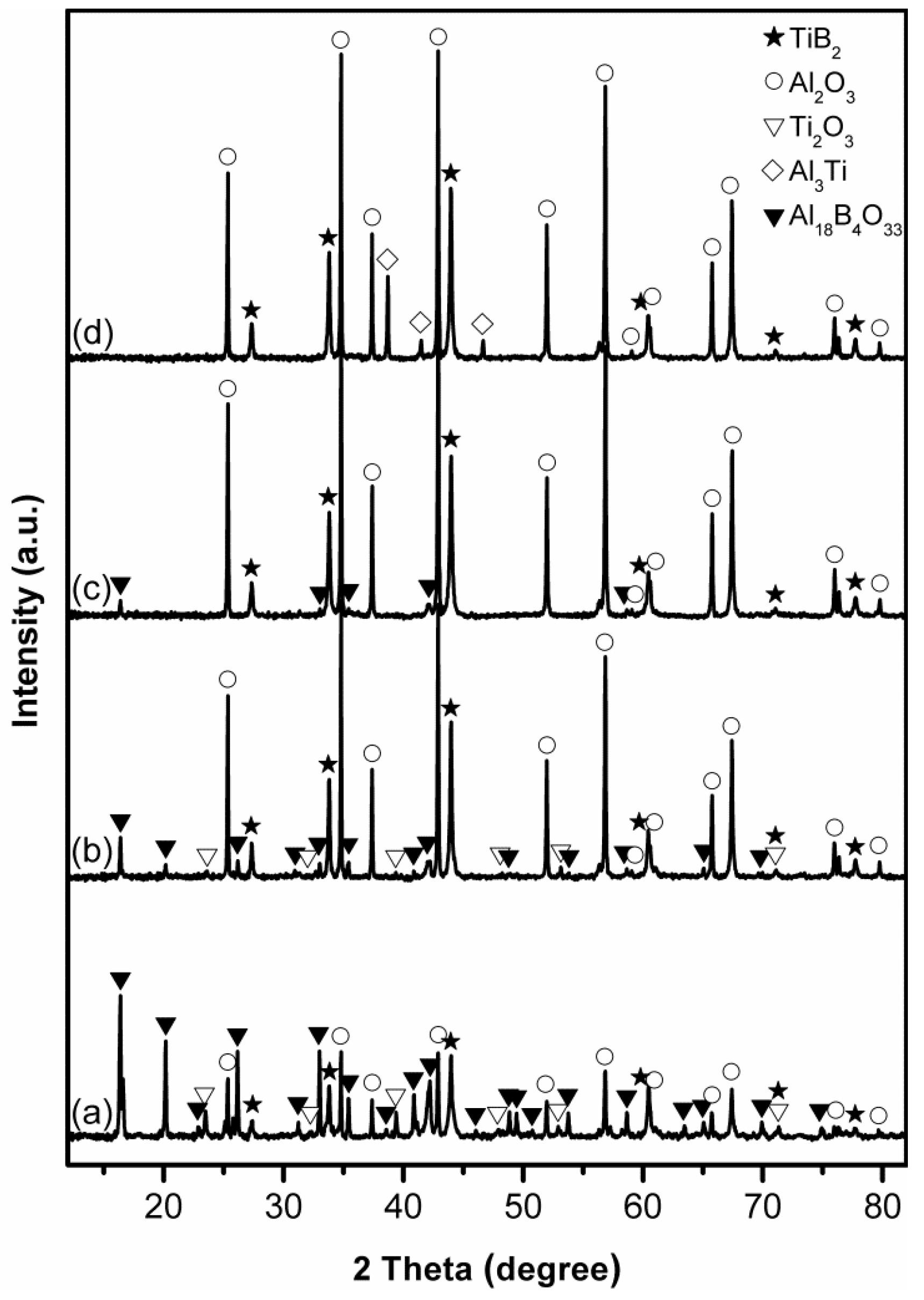
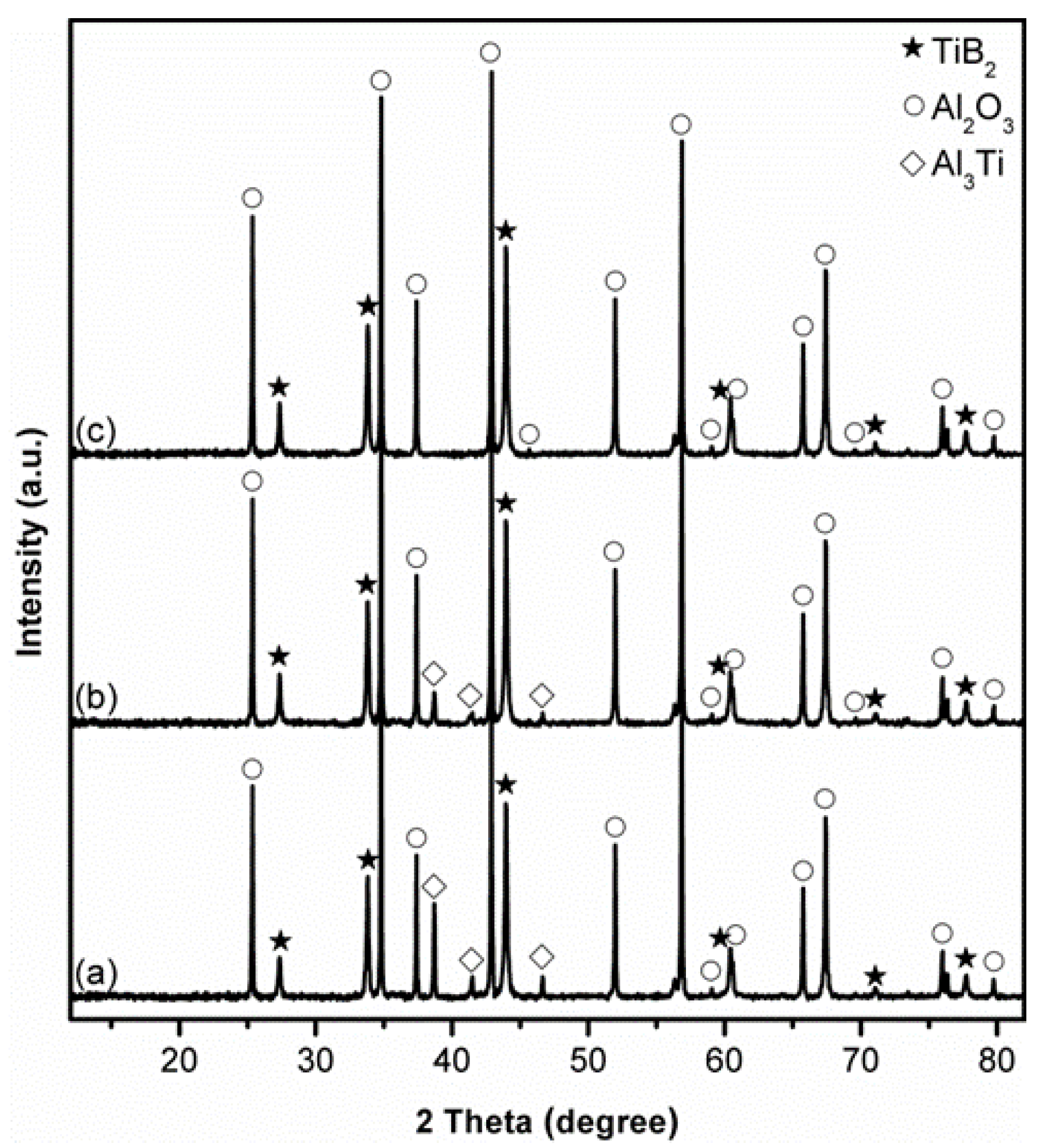
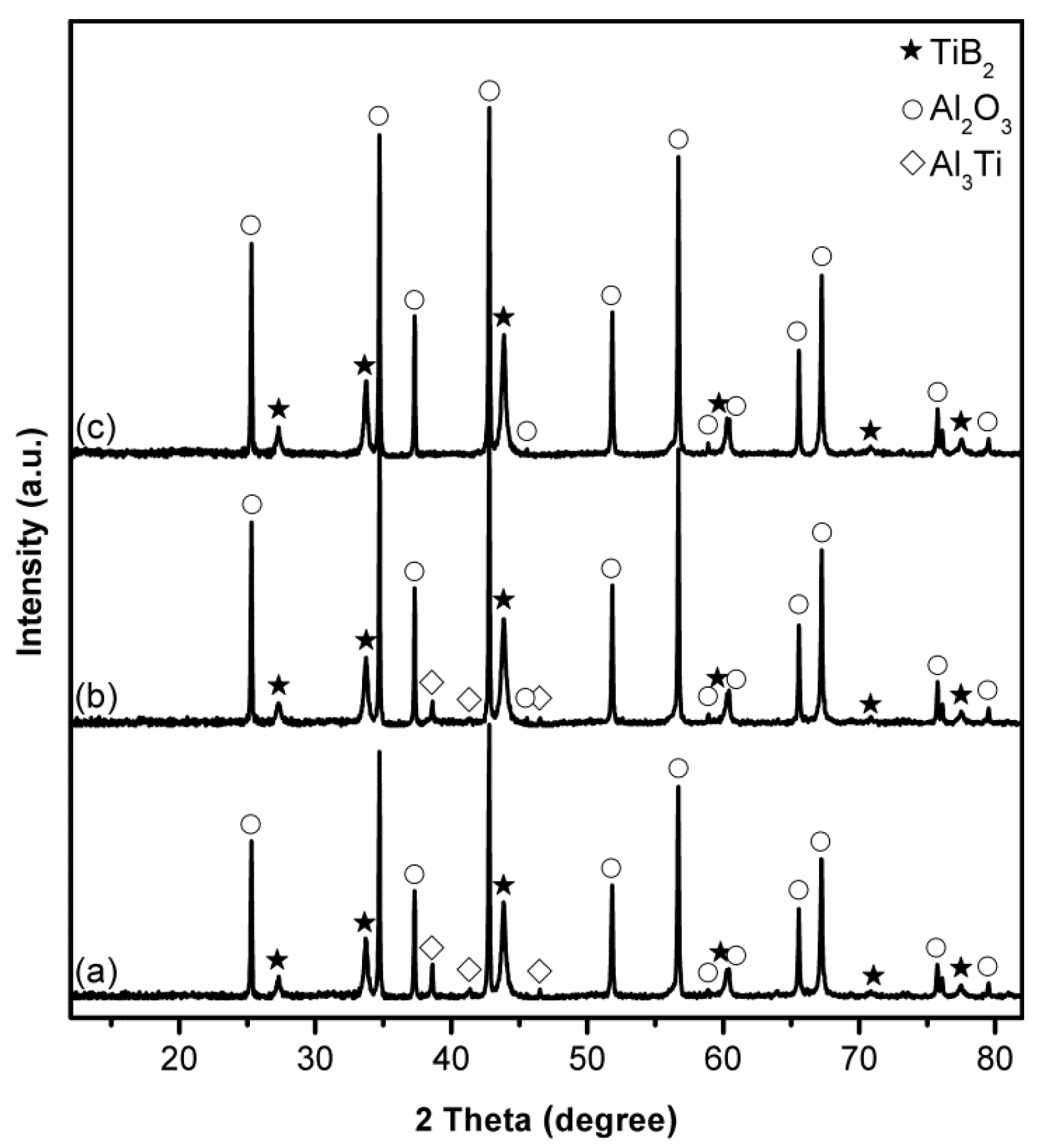
3.1. Influence of Salt Type on Al2O3-TiB2 Formation
3.2. Influence of Firing Temperature on Al2O3-TiB2 Formation
3.3. Influence of Excess Al on Al2O3-TiB2 Formation
3.4. Influence of Excess B2O3 on Al2O3-TiB2 Formation
3.5. Influence of Firing Time on Al2O3-TiB2 Formation and Further Optimisation of Synthesis Condition
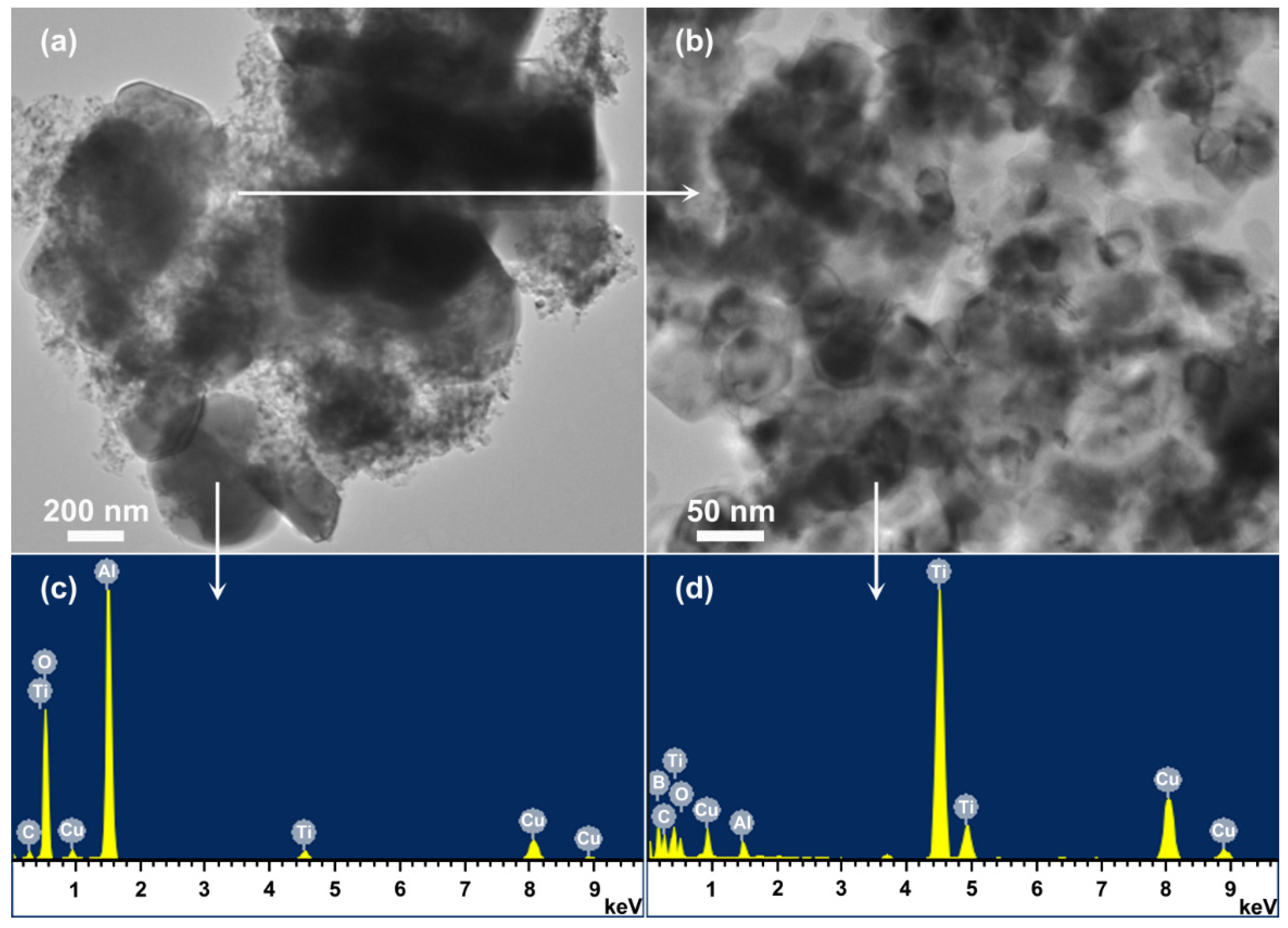
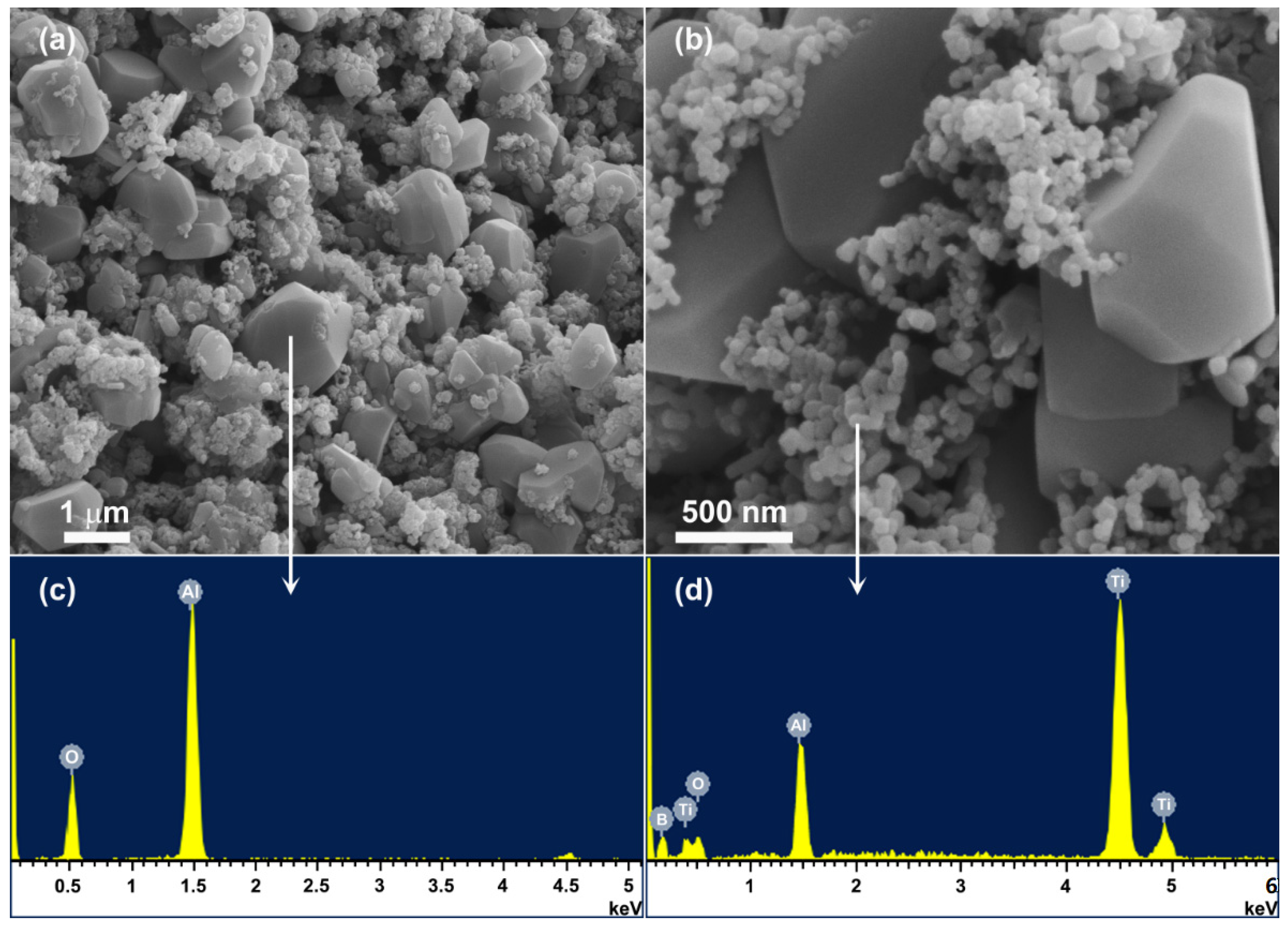
3.6. Microstructure of Al2O3-TiB2 Product Powder
4. Further Discussion and Reaction/Synthesis Mechanism
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Munro, M. Evaluated Material Properties for a Sintered alpha-Alumina. J. Am. Ceram. Soc. 2005, 80, 1919–1928. [Google Scholar] [CrossRef]
- Basu, B.; Balani, K. (Eds.) Bonding, Structure, and Physical Properties. In Advanced Structural Ceramics; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2011; pp. 28–29. [Google Scholar]
- Williams, D. Contemporary and Future Biomaterials; Cambridge University Press (CUP): Cambridge, UK, 2018; pp. 475–589. [Google Scholar]
- Yazdani, B.; Xia, Y.; Ahmad, I.; Zhu, Y. Graphene and carbon nanotube (GNT)-reinforced alumina nanocomposites. J. Eur. Ceram. Soc. 2015, 35, 179–186. [Google Scholar] [CrossRef]
- Niihara, K. New Design Concept of Structural Ceramics. J. Ceram. Soc. Jpn. 1991, 99, 974–982. [Google Scholar] [CrossRef] [Green Version]
- Rainforth, W.M. The wear behaviour of oxide ceramics-A Review. J. Mater. Sci. 2004, 39, 6705–6721. [Google Scholar] [CrossRef]
- Chen, H.; Rainforth, W.M.; Lee, W.E. The wear behaviour of Al2O3-SiC ceramic nanocomposites. Scr. Mater. 2000, 42, 555–560. [Google Scholar] [CrossRef]
- Sharifi, E.M.; Karimzadeh, F.; Enayati, M.H. Synthesis of titanium diboride reinforced alumina matrix nanocomposite by mechanochemical reaction of Al-TiO2-B2O3. J. Alloys Compd. 2010, 502, 508–512. [Google Scholar] [CrossRef]
- Sharifi, E.M.; Karimzadeh, F.; Enayati, M.H. Preparation of Al2O3-TiB2 nanocomposite powder by mechanochemical reaction between Al, B2O3 and Ti. Adv. Powder Technol. 2011, 22, 526–531. [Google Scholar] [CrossRef]
- Golla, B.R.; Bhandari, T.; Mukhopadhyay, A.; Basu, B. Titanium Diboride. In Ultra-High Temperature Ceramics: Materials for Extreme Environment Applications; Fahrenholtz, W.G., Wuchina, E.J., Lee, W.E., Zhou, Y., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2014; pp. 316–360. [Google Scholar]
- Kimura, I.; Hotta, N.; Hiraoka, Y.; Saito, N.; Yokota, Y. Sintering and characterization of Al2O3-TiB2 composites. J. Eur. Ceram. Soc. 1989, 5, 23–27. [Google Scholar] [CrossRef]
- Liu, J.; Ownby, P.D. Enhanced mechanical properties of alumina by dispersed titanium diboride particulate inclusions. J. Am. Ceram. Soc. 1991, 74, 241–243. [Google Scholar] [CrossRef]
- Ranganath, S.; Subrahmanyam, J. Impact response of Al2O3 and Al2O3-TiB2 ceramic composites. J. Mater. Sci. Lett. 1991, 10, 1297–1298. [Google Scholar] [CrossRef]
- Bellosi, A.; De Portu, G.; Guicciardi, S. Preparation and properties of electroconductive Al2O3-based composites. J. Eur. Ceram. Soc. 1992, 10, 307–315. [Google Scholar] [CrossRef]
- Jianxin, D.; Xing, A.; Zhaoqian, L. Friction and wear behavior of Al2O3/TiB2 composite against cemented carbide in various atmospheres at elevated temperature. Wear 1996, 195, 128–132. [Google Scholar] [CrossRef]
- Jianxin, D.; Tongkun, C.; Lili, L. Self-lubricating behaviors of Al2O3/TiB2 ceramic tools in dry high-speed machining of hardened steel. J. Eur. Ceram. Soc. 2005, 25, 1073–1079. [Google Scholar] [CrossRef]
- Tampieri, A.; Bellosi, A. Oxidation of monolithic TiB2 and of Al2O3-TiB2 composite. J. Mater. Sci. 1993, 28, 649–653. [Google Scholar] [CrossRef]
- Plovnick, R.H.; Richards, E. New combustion synthesis route to TiB2-Al2O3. Mater. Res. Bull. 2001, 36, 1487–1493. [Google Scholar] [CrossRef]
- Stadlbauer, W.; Kladnig, W.; Gritzner, G. Al2O3-TiB2 composite ceramics. J. Mater. Sci. Lett. 1989, 8, 1217–1220. [Google Scholar] [CrossRef]
- Liu, G.; Yan, D.; Zhang, J. Microstructure and mechanical properties of TiB2-Al2O3 composites. J. Wuhan Univ. Technol. Sci. Ed. 2011, 26, 696–699. [Google Scholar] [CrossRef]
- Li, J.; Gao, L.; Guo, J. Mechanical properties and electrical conductivity of TiN-Al2O3 nanocomposites. J. Eur. Ceram. Soc. 2003, 23, 69–74. [Google Scholar] [CrossRef]
- Logan, K.V. Shaped Refractory Products and Method of Making Same. United States Patent No. 4, 891, 337, 2 January 1990. [Google Scholar]
- Ray, S.P. Boride-alumina composites: Synthesis and Fabrication. Metall. Trans. A 1992, 23, 2381–2385. [Google Scholar] [CrossRef]
- Kecskes, L.J.; Niiler, A.; Kottke, T.; Logan, K.V.; Villalobos, G.R. Dynamic consolidation of combustion-synthesized alumina-titanium diboride composite ceramics. J. Am. Ceram. Soc. 2005, 79, 2687–2695. [Google Scholar] [CrossRef]
- Ma, Z.Y.; Tjong, S.C. In Situ ceramic particle-reinforced aluminum matrix composites fabricated by reaction pressing in the TiO2 (Ti)-Al-B (B2O3) systems. Met. Mater. Trans. A 1997, 28, 1931–1942. [Google Scholar] [CrossRef]
- Xia, T.D.; Liu, T.Z.; Zhao, W.J.; Munir, Z.A.; Wang, T.M. Photo- and cathodoluminescence of the combustion-synthesized Al2O3–TiB2 composites. J. Mater. Res. 2000, 15, 1622–1629. [Google Scholar] [CrossRef]
- Meyers, M.; Olevsky, E.; Ma, J.; Jamet, M. Combustion synthesis/densification of an Al2O3-TiB2 composite. Mater. Sci. Eng. A 2001, 311, 83–99. [Google Scholar] [CrossRef]
- Zhu, H.G.; Wang, H.Z.; Ge, L.Q.; Chen, S.; Wu, S.Q. Formation of composites fabricated by exothermic dispersion reaction in Al-TiO2-B2O3 system. Trans. Nonferrous Met. Soc. China 2007, 17, 590–594. [Google Scholar] [CrossRef]
- Zhu, H.; Wang, H.; Ge, L. Wear properties of the composites fabricated by exothermic dispersion reaction synthesis in an Al-TiO2-B2O3 system. Wear 2008, 264, 967–972. [Google Scholar] [CrossRef]
- Yeh, C.; Li, R. Formation of TiB2-Al2O3 and NbB2-Al2O3 composites by combustion synthesis involving thermite reactions. Chem. Eng. J. 2009, 147, 405–411. [Google Scholar] [CrossRef]
- Mousavian, R.T.; Sharafi, S.; Shariat, M.H. Microwave-assisted combustion synthesis in a mechanically activated Al-TiO2-H3BO3 system. Int. J. Refract. Met. Hard Mater. 2011, 29, 281–288. [Google Scholar] [CrossRef]
- Mishra, S.; Gokuul, V.; Paswan, S. Alumina-titanium diboride in situ composite by self-propagating high-temperature synthesis (SHS) dynamic compaction: Effect of compaction pressure during synthesis. Int. J. Refract. Met. Hard Mater. 2014, 43, 19–24. [Google Scholar] [CrossRef]
- Khaghani-Dehaghani, M.A.; Ebrahimi-Kahrizsangi, R.; Setoudeh, N.; Nasiri-Tabrizi, B. Mechanochemical synthesis of Al2O3-TiB2 nanocomposite powder from Al-TiO2-H3BO3 mixture. Int. J. Refract. Met. Hard Mater. 2011, 29, 244–249. [Google Scholar] [CrossRef]
- Abdellahi, M.; Heidari, J.; Sabouhi, R. Influence of B source materials on the synthesis of TiB2-Al2O3 nanocomposite powders by mechanical alloying. Int. J. Miner. Met. Mater. 2013, 20, 1214–1220. [Google Scholar] [CrossRef]
- Yang, W.; Dong, S.; Luo, P.; Yangli, A.; Liu, Q.; Xie, Z. Effect of Ni addition on the preparation of Al2O3-TiB2 composites using high-energy ball milling. J. Asian Ceram. Soc. 2014, 2, 399–402. [Google Scholar] [CrossRef] [Green Version]
- Rabiezadeh, A.; Hadian, A.; Ataie, A. Preparation of alumina/titanium diboride nano-composite powder by milling assisted sol-gel method. Int. J. Refract. Met. Hard Mater. 2012, 31, 121–124. [Google Scholar] [CrossRef]
- Zhang, S.; Khangkhamano, M.; Zhang, H.; Yeprem, H.A. Novel Synthesis of ZrB2 powder via molten-salt-mediated magnesiothermic Reduction. J. Am. Ceram. Soc. 2014, 97, 1686–1688. [Google Scholar] [CrossRef]
- Bao, K.; Liu, C.; Yazdani, B.; Damavandi, Y.; Zhang, S. Low-Temperature preparation of lanthanum hexaboride fine powder via magnesiothermic reduction in molten salt. J. Ceram. Sci. Technol. 2016, 7, 403–408. [Google Scholar]
- Bao, K.; Wen, Y.; Khangkhamano, M.; Zhang, S. Low-temperature preparation of titanium diboride fine powder via magnesiothermic reduction in molten salt. J. Am. Ceram. Soc. 2017, 51, 257–2272. [Google Scholar] [CrossRef]
- Li, T.; Chen, J.; Xiao, J.; Wei, Y.; Li, G.; Zhang, S.; Li, N. Formation of liquid-phase isolation layer on the corroded interface of MgO/Al2O3-SiC-C refractory and molten steel: Role of SiC. J. Am. Ceram. Soc. 2021, 104, 2366–2377. [Google Scholar] [CrossRef]
- Hashimoto, S.; Yamaguchi, A. Synthesis of MgAl2O4 (Spinel) Powder Using MgCl2. J. Ceram. Soc. Jpn. 2001, 109, 894–896. [Google Scholar] [CrossRef] [Green Version]
- Horlait, D.; Grasso, S.; Chroneos, A.; Lee, W. Attempts to synthesise quaternary MAX phases (Zr,M)2AlC and Zr2(Al,A)C as a way to approach Zr2AlC. Mater. Res. Lett. 2016, 4, 137–144. [Google Scholar] [CrossRef] [Green Version]
- Mousavian, R.T.; Sharafi, S.; Roshan, M.R.; Shariat, M.H. Effect of mechanical activation of reagents’ mixture on the high-temperature synthesis of Al2O3-TiB2 composite powder. J. Therm. Anal. Calorim. 2011, 104, 1063–1070. [Google Scholar] [CrossRef]
- Jacobson, N.S.; Myers, D.L. High-temperature vaporization of B2O3(l) under reducing conditions. J. Phys. Chem. B 2011, 115, 13253–13260. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, X.; Bao, K.; Chen, J.; Jia, Q.; Zhang, S. One-Pot Synthesis of Alumina-Titanium Diboride Composite Powder at Low Temperature. Materials 2021, 14, 4742. https://doi.org/10.3390/ma14164742
Liu X, Bao K, Chen J, Jia Q, Zhang S. One-Pot Synthesis of Alumina-Titanium Diboride Composite Powder at Low Temperature. Materials. 2021; 14(16):4742. https://doi.org/10.3390/ma14164742
Chicago/Turabian StyleLiu, Xueyin, Ke Bao, Junfeng Chen, Quanli Jia, and Shaowei Zhang. 2021. "One-Pot Synthesis of Alumina-Titanium Diboride Composite Powder at Low Temperature" Materials 14, no. 16: 4742. https://doi.org/10.3390/ma14164742
APA StyleLiu, X., Bao, K., Chen, J., Jia, Q., & Zhang, S. (2021). One-Pot Synthesis of Alumina-Titanium Diboride Composite Powder at Low Temperature. Materials, 14(16), 4742. https://doi.org/10.3390/ma14164742






