Synthetic Injectable Biomaterials for Alveolar Bone Regeneration in Animal and Human Studies
Abstract
1. Introduction
1.1. Allografts
1.2. Xenografts
1.3. Autogenous Bone
1.4. Dentin Matrix
1.5. Synthetic Biomaterials
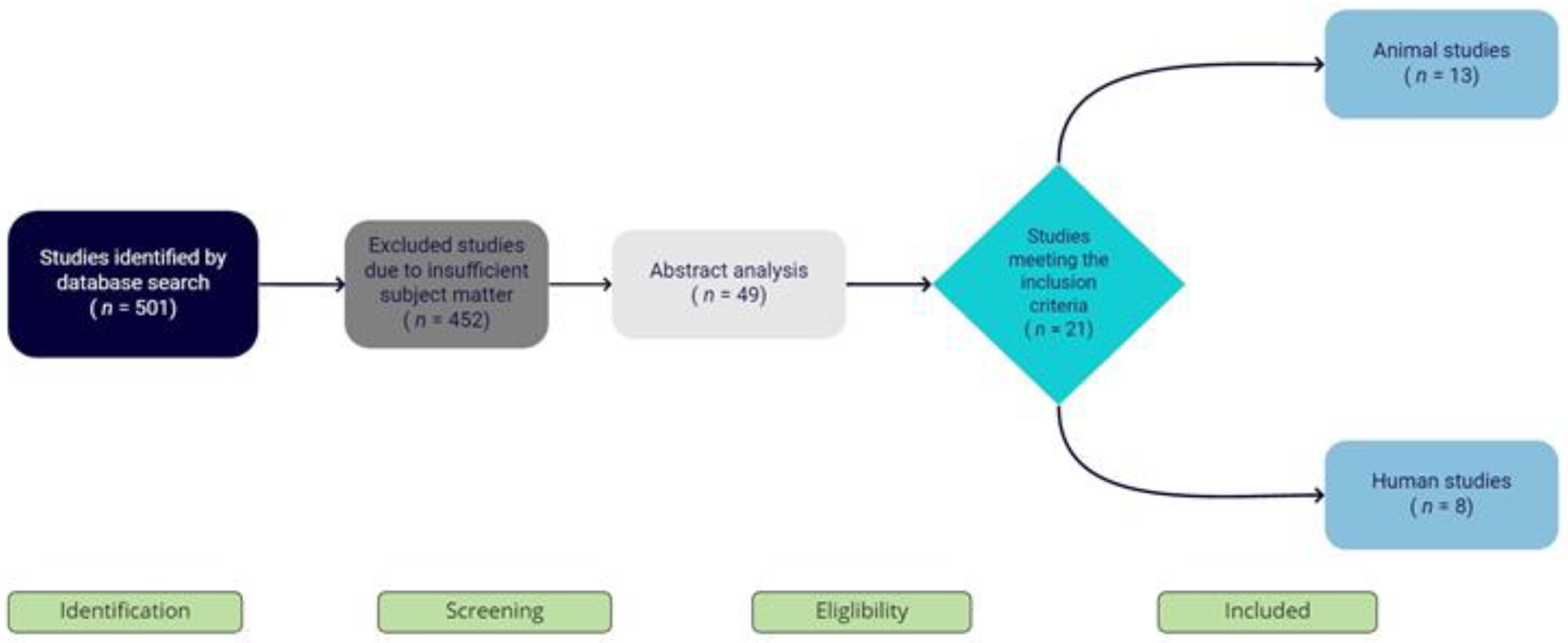
2. Materials and Methods
- -
- human studies;
- -
- animal studies;
- -
- English language studies;
- -
- case reports, clinical cases, experimental pilot studies, randomized clinical trials, and preliminary studies;
- -
- studies limited to the application of synthetic biomaterials in dentistry;
- -
- studies limited to the injectable form of application;
- -
- studies that included biopsy (histomorphological) and radiographic analysis; and
- -
- -
- studies that were not in English;
- -
- studies that were performed on other bones (orthopedic surgery);
- -
- studies that did not use synthetic biomaterial in injectable form; and
- -
- in vitro studies.
3. Results
4. Discussion
4.1. Animal Studies
4.2. Human Studies
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mayer, Y.; Zigdon-Giladi, H.; Machtei, E.E. Ridge Preservation Using Composite Alloplastic Materials: A Randomized Control Clinical and Histological Study in Humans. Clin. Implant Dent. Relat. Res. 2016, 18, 1163–1170. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.L.; Wong, T.L.T.; Wong, M.C.M.; Lang, N.P. A systematic review of post-extractional alveolar hard and soft tissue dimensional changes in humans. Clin. Oral Implant. Res. 2012, 1–21. [Google Scholar] [CrossRef]
- Karabuda, C.; Ozdemir, O.; Tosun, T.; Anil, A.; Olgaç, V. Histological and clinical evaluation of 3 different grafting materials for sinus lifting procedure based on 8 cases. J. Periodontol. 2001, 72, 1436–1442. [Google Scholar] [CrossRef]
- Allen, F. Bone biology, harvesting and grafting for dental implants: Rationale and clinical applications. Br. Dent. J. 2005, 199, 59. [Google Scholar] [CrossRef][Green Version]
- Kacarevic, Z.P.; Kavehei, F.; Houshmand, A.; Franke, J.; Smeets, R.; Rimashevskiy, D.; Wenisch, S.; Schnettler, R.; Jung, O.; Barbeck, M. Purification processes of xenogeneic bone substitutes and their impact on tissue reactions and regeneration. Int. J. Artif. Organs. 2018, 41, 789–800. [Google Scholar] [CrossRef]
- Torres, J.; Tamimi, F.; Alkhraisat, M.H.; Manchón, Á.; Linares, R.; Prados-Frutos, J.C.; Hernández, G.; López, C.E. Platelet-rich plasma may prevent titanium-mesh exposure in alveolar ridge augmentation with anorganic bovine bone. J. Clin. Periodontol. 2010, 37, 943–951. [Google Scholar] [CrossRef] [PubMed]
- Lindhe, J.; Cecchinato, D.; Donati, M.; Tomasi, C.; Liljenberg, B. Ridge preservation with the use of deproteinized bovine bone mineral. Clin. Oral Implant. Res. 2014, 25, 786–790. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, C.M.; Doering, H.; Schmidt, T.; Lutz, R.; Neukam, F.W.; Schlegel, K.A. Histological results after maxillary sinus augmentation with Straumann® BoneCeramic, Bio-Oss®, Puros®, and autologous bone. A randomized controlled clinical trial. Clin. Oral Implant. Res. 2013, 24, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Helder, M.N.; van Esterik, F.A.S.; Kwehandjaja, M.D.; ten Bruggenkate, C.M.; Klein-Nulend, J.; Schulten, E.A.J.M. Evaluation of a new biphasic calcium phosphate for maxillary sinus floor elevation: Micro-CT and histomorphometrical analyses. Clin. Oral Implant. Res. 2018, 29, 488–498. [Google Scholar] [CrossRef]
- Ohayon, L. Maxillary Sinus Floor Augmentation Using Biphasic Calcium Phosphate: A Histologic and Histomorphometric Study. Int. J. Oral Maxillofac. Implant. 2014, 29, 1143–1148. [Google Scholar] [CrossRef]
- Von Arx, T.; Buser, D. Horizontal ridge augmentation using autogenous block grafts and the guided bone regeneration technique with collagen membranes: A clinical study with 42 patients. Clin. Oral Implant. Res. 2006, 17, 359–366. [Google Scholar] [CrossRef]
- Martinez, A.; Balboa, O.; Gasamans, I.; Otero-Cepeda, X.L.; Guitian, F. Deproteinated bovine bone vs. beta-tricalcium phosphate as bone graft substitutes: Histomorphometric longitudinal study in the rabbit cranial vault. Clin. Oral Implant. Res. 2015, 26, 623–632. [Google Scholar] [CrossRef] [PubMed]
- Jensen, S.S.; Broggini, N.; Hjørting-Hansen, E.; Schenk, R.; Buser, D. Bone healing and graft resorption of autograft, anorganic bovine bone and β-tricalcium phosphate. A histologic and histomorphometric study in the mandibles of minipigs. Clin. Oral Implant. Res. 2006, 17, 237–243. [Google Scholar] [CrossRef] [PubMed]
- Franco, M.; Viscioni, A.; Rigo, L.; Guidi, R.; Strohmenger, L.; Zollino, I.; Avantaggiato, A.; Carinci, F. Wide Diameter Implants Inserted in Jaws Grafted with Homologue Bone. Acta Stomatol. Croat. 2008, 42, 273–282. [Google Scholar]
- Chiapasco, M.; Zaniboni, M.; Boisco, M. Augmentation procedures for the rehabilitation of deficient edentulous ridges with oral implant. Clin. Oral Implant. Res. 2006, 2, 136–159. [Google Scholar] [CrossRef] [PubMed]
- Urist, M.R.; Silverman, B.F.; Buring, K.; Dubuc, F.L.; Rosenberg, J.M. The bone induction principle. Clin. Orthop. Relat. Res. 1967, 53, 243–284. [Google Scholar] [CrossRef]
- Mai, Y.; Wu, H.; Mai, Z.; Li, X.; Huang, L.; Liao, H. Preliminary application of injectable calcium phosphate cement/poly (lactic-co-glycolic acid) microspheres for extraction site preservation. Chin. J. Stomatol. 2014, 49, 180–183. [Google Scholar]
- Gauthier, O.; Boix, D.; Grimandi, G.; Aguado, E.; Bouler, J.-M.; Weiss, P.; Daculsi, G. A New Injectable Calcium Phosphate Biomaterial for Immediate Bone Filling of Extraction Sockets: A Preliminary Study in Dogs. J. Periodontol. 1999, 70, 375–383. [Google Scholar] [CrossRef]
- Murata, M.; Akazawa, T.; Mitsugi, M.; Arafat, M.; Um, I.-W.; Minamida, Y.; Kim, K.W.; Kim, Y.K.; Sun, Y.; Qin, C. Autograft of Dentin Materials for Bone Regenera-tion. Adv. Biomater. Sci. Biomed. Appl. 2013. [Google Scholar] [CrossRef]
- Calvo-Guirado, J.L.; Ballester, M.A.; De Aza, P.N.; Fernández-Domínguez, M.; Gehrke, S.A.; Pino, C.D.; Mahesh, L.; Pelegrine, A.A.; Aragoneses, J.M.; Maté-Sánchez de Val, J.E. Particulated, extracted human teeth characterization by SEM-EDX evaluation as a biomaterial for socket preservation: An in vitro study. Materials 2019, 12, 380. [Google Scholar] [CrossRef]
- Lorenz, J.; Barbeck, M.; Kirkpatrick, C.; Sader, R.; Lerner, H.; Ghanaati, S. Injectable Bone Substitute Material on the Basis of ß-TCP and Hyaluronan Achieves Complete Bone Regeneration While Undergoing Nearly Complete Degradation. Int. J. Oral Maxillofac. Implant. 2018, 33. [Google Scholar] [CrossRef]
- Barbeck, M.; Jung, O.; Smeets, R.; Gosau, M.; Schnettler, R.; Rider, P.; Houshmand, A.; Korzinskas, T. Implantation of an injectable bone substitute material enables integration following the principles of guided bone regeneration. Vivo 2020, 34, 557–568. [Google Scholar] [CrossRef]
- Ku, J.K.; Hong, I.; Lee, B.K.; Yun, P.Y.; Lee, J.K. Dental alloplastic bone substitutes currently available in Korea. J. Korean Assoc. Oral Maxillofac. Surg. 2019, 45, 51–67. [Google Scholar] [CrossRef]
- Kakar, A.; Rao, B.H.S.; Hegde, S.; Deshpand, N.; Lindner, A.; Nagursky, H.; Patney, A.; Mahajan, H. Ridge preservation using an in situ hardening biphasic calcium phosphate (β-TCP/HA) bone graft substitute—a clinical, radiological, and histological study. Int. J. Implant Dent. 2017, 3, 1–10. [Google Scholar] [CrossRef]
- Ghanaati, S.; Barbeck, M.; Detsch, R.; Deisinger, U.; Hilbig, U.; Rausch, V.; Sader, R.; Unger, R.E.; Ziegler, G.; Kirkpatrick, C.J. The chemical composition of synthetic bone substitutes influences tissue reactions in vivo: Histological and histomorphometrical analysis of the cellular inflammatory response to hydroxyapatite, beta-tricalcium phosphate and biphasic calcium phosphate ceramics. Biomed. Mater. 2012, 7, 015005. [Google Scholar] [CrossRef]
- Annibali, S.; Iezzi, G.; Sfasciotti, G.L.; Cristalli, M.P.; Vozza, I.; Mangano, C.; La Monaca, G.; Polimeni, A. Histological and Histomorphometric Human Results of HA-Beta-TCP 30/70 Compared to Three Different Biomaterials in Maxillary Sinus Augmentation at 6 Months: A Preliminary Report. BioMed Res. Int. 2015, 2015, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Ghanaati, S.; Barbeck, M.; Hilbig, U.; Hoffmann, C.; Unger, R.; Sader, R.; Peters, F.; Kirkpatrick, C. An injectable bone substitute composed of beta-tricalcium phosphate granules, methylcellulose and hyaluronic acid inhibits connective tissue influx into its implantation bed in vivo. Acta Biomater. 2011, 7, 4018–4028. [Google Scholar] [CrossRef] [PubMed]
- Munerato, M.S.; Biguetti, C.C.; da Silva, R.B.P.; da Silva, A.C.R.; Bacelar, A.C.Z.; da Silva, J.L.; Couto, M.C.R.; Duarte, M.A.H.; Santiago-Junior, J.F.; Bossini, P.S.; et al. Inflammatory response and macrophage polarization using different physicochemical biomaterials for oral and maxillofacial reconstruction. Mater. Sci. Eng. C 2020, 107, 110229. [Google Scholar] [CrossRef] [PubMed]
- Sariibrahimoglu, K.; Wolk, J.G.C.; Leeuwenburgh, S.C.G.; Yubao, L.; Jansen, J.A. Injectable biphasic calcium phosphate cements as a potential bone substitute. J. Biomed. Mater. Res. Part B Appl. Biomater. 2014, 102, 415–422. [Google Scholar] [CrossRef] [PubMed]
- Fujioka-Kobayashi, M.; Schaller, B.; Zhang, Y.; Pippenger, B.E.; Miron, R.J. In vitro evaluation of an injectable biphasic calcium phosphate (BCP) carrier system combined with recombinant human bone morphogenetic protein (rhBMP)-9. Biomed. Mater. Eng. 2017, 28, 293–304. [Google Scholar] [CrossRef]
- Struillou, X.; Boutigny, H.; Badran, Z.; Fellah, B.H.; Gauthier, O.; Sourice, S.; Pilet, P.; Rouillon, T.; Layrolle, P.; Weiss, P.; et al. Treatment of periodontal defects in dogs using an injectable composite hydrogel/biphasic calcium phosphate. J. Mater. Sci. Mater. Med. 2011, 22, 1707–1717. [Google Scholar] [CrossRef]
- Weiss, P.; Layrolle, P.; Clergeau, L.P.; Enckel, B.; Pilet, P.; Amouriq, Y.; Daculsi, G.; Giumelli, B. The safety and efficacy of an injectable bone substitute in dental sockets demonstrated in a human clinical trial. Biomaterial 2007, 28, 3295–3305. [Google Scholar] [CrossRef]
- Liu, W.; Zhang, J.; Rethore, G.; Khairoun, K.; Pilet, P.; Tancret, F.; Bouler, J.-M.; Weiss, P. A novel injectable, cohesive and toughened Si-HPMC (silanized-hydroxypropyl methylcellulose) composite calcium phosphate cement for bone substitution. Acta Biomater. 2014, 10, 3335–3345. [Google Scholar] [CrossRef] [PubMed]
- Hoekstra, J.W.M.; Klijn, R.J.; Meijer, G.J.; Beucken, J.J.J.P.V.D.; Jansen, J.A. Maxillary sinus floor augmentation with injectable calcium phosphate cements: A pre-clinical study in sheep. Clin. Oral Implant. Res. 2013, 24, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Guha, A.K.; Singh, S.; Kumaresan, R.; Nayar, S.; Sinha, A. Mesenchymal cell response to nanosized biphasic calcium phosphate composites. Coll. Surf. B Biointerfaces 2009, 73, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Lanao, R.P.F.; Leeuwenburgh, S.; Wolke, J.G.; Jansen, J.A. Bone response to fast-degrading, injectable calcium phosphate cements containing PLGA microparticles. Biomaterial 2011, 32, 8839–8847. [Google Scholar] [CrossRef] [PubMed]
- Jansen, J.A.; De Ruijter, J.E.; Schaeken, H.G.; Van Der Waerden, J.P.C.M.; Planell, J.A.; Driessens, F.C.M. Evaluation of tricalciumphosphate/ hydroxyapatite cement for tooth replacement: An experimental animal study. J. Mater. Sci. Mater. Med. 1995, 6, 653–657. [Google Scholar] [CrossRef]
- Barbeck, M.; Hoffmann, C.; Sader, R.; Peters, F.; Hübner, W.-D.; Kirkpatrick, C.J.; Ghanaati, S. Injectable Bone Substitute Based on β-TCP Combined With a Hyaluronan-Containing Hydrogel Contributes to Regeneration of a Critical Bone Size Defect Towards Restitutio ad Integrum. J. Oral Implant. 2016, 42, 127–137. [Google Scholar] [CrossRef]
- Boix, D.; Gauthier, O.; Guicheux, J.; Pilet, P.; Weiss, P.; Grimandi, G.; Daculsi, G. Alveolar bone regeneration for immediate implant placement using an injectable bone substitute: An experimental study in dogs. J. Periodontol. 2004, 75, 663–671. [Google Scholar] [CrossRef]
- Aral, A.; Yalçιn, S.; Karabuda, Z.C.; Anιl, A.; Jansen, J.A.; Mutlu, Z. Injectable calcium phosphate cement as a graft material for maxillary sinus augmentation: An experimental pilot study. Clin. Oral Implant. Res. 2008, 19, 612–617. [Google Scholar] [CrossRef]
- Han, X.; Liu, H.; Wang, D.; Su, F.; Zhang, Y.; Zhou, W.; Li, S.; Yang, R. Alveolar Bone Regeneration around Immediate Implants Using an Injectable nHAC/CSH Loaded with Autogenic Blood-Acquired Mesenchymal Progenitor Cells: An Experimental Study in the Dog Mandible. Clin. Implant Dent. Relat. Res. 2011, 15, 390–401. [Google Scholar] [CrossRef]
- Sa, Y.; Yu, N.; Wolke, J.G.; Chanchareonsook, N.; Goh, B.T.; Wang, Y.; Yang, F.; Jansen, J.A. Bone Response to Porous Poly(methyl methacrylate) Cement Loaded with Hydroxyapatite Particles in a Rabbit Mandibular Model. Tissue Eng. Part C Methods 2017, 23, 262–273. [Google Scholar] [CrossRef]
- Kaneko, A.; Marukawa, E.; Harada, H. Hydroxyapatite Nanoparticles as Injectable Bone Substitute Material in a Vertical Bone Augmentation Model. Vivo 2020, 34, 1053–1061. [Google Scholar] [CrossRef]
- Hayashi, C.; Kinoshita, A.; Oda, S.; Mizutani, K.; Shirakata, Y.; Ishikawa, I. Injectable Calcium Phosphate Bone Cement Provides Favorable Space and a Scaffold for Periodontal Regeneration in Dogs. J. Periodontol. 2006, 77, 940–946. [Google Scholar] [CrossRef]
- Shirakata, Y.; Taniyama, K.; Yoshimoto, T.; Takeuchi, N.; Noguchi, K. Effect of bone swaging with calcium phosphate bone cement on periodontal regeneration in dogs. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2012, 114, 35–42. [Google Scholar] [CrossRef]
- Oortgiesen, D.A.W.; Walboomers, X.F.; Bronckers, A.L.J.J.; Meijer, G.J.; Jansen, J.A. Periodontal regeneration using an injectable bone cement combined with BMP-2 or FGF-2. J. Tissue Eng. Regen. Med. 2012, 8, 202–209. [Google Scholar] [CrossRef]
- Oortgiesen, D.A.W.; Meijer, G.J.; Bronckers, A.L.J.J.; Walboomers, X.F.; Jansen, J.A. Regeneration of the periodontium using enamel matrix derivative in combination with an injectable bone cement. Clin. Oral Investig. 2012, 17, 411–421. [Google Scholar] [CrossRef]
- Wolff, K.-D.; Swaid, S.; Nolte, D.; Böckmann, R.A.; Hölzle, F.; Müller-Mai, C. Degradable injectable bone cement in maxillofacial surgery: Indications and clinical experience in 27 patients. J. Cranio-Maxillofac. Surg. 2004, 32, 71–79. [Google Scholar] [CrossRef]
- Stanton, D.C.; Chou, J.C.; Carrasco, L.R. Injectable calcium-phosphate bone cement (Norian) for reconstruction of a large mandibular defect: A case report. J. Oral Maxillofac. Surg. 2004, 62, 235–240. [Google Scholar] [CrossRef]
- Papanchev, G.; Georgiev, T.; Peev, S.; Arnautska, H.; Zgurova, N.; Borisova-Papancheva, T.; Dzhongova, E. Comparison of the rates of bone regeneration in Sinus lift grafting with a Calcium-Phosphate paste between the 6th and the 9th month—A clinical case. Scr. Sci. Med. Dent. 2015, 1, 41. [Google Scholar] [CrossRef][Green Version]
- Khaled, H.; Atef, M.; Hakam, M. Maxillary sinus floor elevation using hydroxyapatite nano particles vs tenting technique with simultaneous implant placement: A randomized clinical trial. Clin. Implant Dent. Relat. Res. 2019, 21, 1241–1252. [Google Scholar] [CrossRef]
- Georgiev, T.; Peev, S.; Arnautska, H.; Gencheva, A.; Gerdzhikov, I. An Evaluation of Three-Dimensional Scans of the Time-Dependent Volume Changes in Bone Grafting Materials. Int. J. Sci. Res. 2017, 6, 562–571. [Google Scholar] [CrossRef]
- Čandrlić, M.; Perić Kačarević, Ž.; Ivanišević, Z.; Tomas, M.; Včev, A.; Faj, D.; Matijević, M. Histological and Radiological Features of a Four-Phase Injectable Synthetic Bone Graft in Guided Bone Regeneration: A Case Report. Int. J. Environ. Res. Public Health 2020, 18, 206. [Google Scholar] [CrossRef]
| Author | Title | Year | Aim of the Study | Biomaterial | Preparation/Properties of Biomaterial | Animal Model | Outcomes | Follow-up Period (Months) | Reference |
|---|---|---|---|---|---|---|---|---|---|
| Mai et al. | Preliminary Application of Injectable Calcium Phosphate Cement/Poly(Lactic-co-Glycolic Acid) Microspheres for Extraction Site Preservation | 2014 | Assess the ability of injectable calcium phosphate cement (CPC) + poly(lactic-co-glycolic acid) (PLGA) microspheres. | Injectable calcium phosphate cement (CPC) + poly(lactic-co-glycolic acid) (PLGA) microspheres | No data | Dog | Newly formed bone 69.2% ± 1.8% 94.7% ± 1.1% 96.0% ± 0.9% | 1 2 3 | [17] |
| Gauthier et al. | A New Injectable Calcium Phosphate Biomaterial for Immediate Bone Filling of Extraction Sockets: A Preliminary Study in Dogs | 1999 | Assess the effects of IBS in bone regeneration. | Injectable CaP: BCP granules with 60/40 HA/β-TCP + cellulose polymer (MHPC 2%) | Application in ready-to-use glass flasks BCP granule diameter 200–500 µm | Dog | Newly formed bone: mandible (64.31% +/− 10.27%); maxilla (48.96% +/− 8.90%) | 3 | [18] |
| Struillou et al. | Treatment of Periodontal Defects in Dogs Using an Injectable Composite Hydrogel/Biphasic Calcium Phosphate | 2011 | Assess the ability of the hydrogel to promote the cohesion of BCP granules. | Hydrogel/BCP: BCP + Si-HPMC hydrogel; BCP granules (Biomatlante SARL, Vigneux de Bretagne, France)–hydroxypropyl methyl cellulose (HPMC, Colorcon-Dow Chemical, France) | BCP + Si: HPMC mixed in two sterile syringes BCP granule diameter 80–200 µm | Dog | Bone–material contact 61.3% ± 9.2% Bone ingrowth 35.5% ± 13.9% | 3 | [31] |
| Hoekstra et al. | Maxillary Sinus Floor Augmentation with Injectable Calcium Phosphate Cements: A Pre -clinical Study in Sheep | 2013 | Assess the biological performance of two types of injectable CPC: PLGA + PLGA microspheres. | CaP: 85% alpha-tricalcium phosphate (CAM Bioceramics BV, Leiden, the Netherlands) + 10% dicalcium phosphate anhydrous (Baker, Griesheim, Germany) + 5% precipitated hydroxyapatite (Merck, Darmstadt, Germany) PLGA (Purac Biomaterials BV, Gorinchem, the Netherlands) in two types: Purasorb® PDLG 5002A and Purasorb® PDLG 5002 | Powder and liquid mixed in apparatus (Silamat) and shaken for 15 s PLGAL-AT microsphere size 37 ± 11 µm PLGAH-EC microsphere size 41 ± 10 µm | Sheep | Newly formed bone within the ROI CPC-PLGAL-AT (low molecular weight) 26.4% ± 10.5% CPC-PLGAH-EC (high molecular weight) 8.6% ± 3.9% | 3 | [34] |
| Boix et al. | Alveolar Bone Regeneration for Immediate Implant Placement Using an Injectable Bone Substitute: An Experimental Study in Dogs | 2004 | Quantitatively assess the different parameters of bone regeneration with IBS. | IBS: BCP granules with 60/40 HA/β-TCP + polymer cellulose derivative (MHPC) composite material obtaining by mixing 3% MHPC with BCP granules | Application in ready-to-use plastic injectors BCP granule diameter 40–80 µm | Dog | Terms of the number of threads in contact with bone 8.6% -bone–implant contact 11.0% -peri-implant bone density 14.7% | 3 | [39] |
| Aral et al. | Injectable Calcium Phosphate Cement as Graft Material for Maxillary Sinus Augmentation: An Experimental Pilot Study | 2008 | Assess the effectiveness of injectable CaP cement as a graft material. | Injectable calcium phosphate cement (Augmentech AT, Wetzlar, Germany) | Powder and liquid mixed in apparatus (Silamat) and shaken for 15 s Applicattion in ready-to-mix syringe system | Sheep | Bone–implant contact (BIC) 36% ± 5% | 3 | [40] |
| Han et al. | Alveolar immediate implants using around Immediate Implants Using an Injectable nHAC/CSH Loaded with Autogenic Blood -Acquired Mesenchymal Progenitor Cells: An Experimental Study in the Dog Mandible | 2011 | Assess new bone formation using nHAC/CSH + blood mesenchymal progenitor cells (dBMPC). | Injectable bone substitute powder composed of CSH and nHAC mixed with liquid | Application in a 5 mL syringe with a puncture needle | Dog | Bone–implant contact: dBMPC + nHAC/CSH 65.03% +/− 3.13% nHAC/CSH 33.13% +/− 7.29% Bone density: dBMPC + nHAC/CSH 61.74% +/− 3.6% nHAC/CSH 12.12% +/− 3.08% | 3 | [41] |
| Sa et al. | Bone Response to Porous poly(Methyl Methacrylate) Cement Loaded with Hydroxyapatite Particles in a Rabbit Mandibular Model | 2017 | Assess bone formation and the response to porous PMMA with or without (HA). | PMMA powder mixed with 1% dibenzoyl peroxide | Solid and liquid manually mixed HA particle size 0.5–1 mm | Rabbit | Bone ingrowth PMMA vs. PMMA-HA: no statistically significant difference PMMA vs. PMMA-HA: statistically significant difference in benefit of PMMA-HA | 1 3 | [42] |
| Kaneko et al. | Hydroxyapatite Nanoparticles as Injectable Bone Substitute Material in a Vertical Bone Augmentation Model | 2020 | Assess the benefit of bone graft gel containing hydroxyapatite nanoparticles. | Injectable bone substitute: two types of nano-HA gel (high, low viscosity) containing glycerin + carboxymethylcellulose matrix from SofSera (Tokyo, Japan) | Application in a syringe with a 25 G needle Average particle size 40 nm | Mice | New bone area significantly greater in the low-viscosity (35%) group than in the high-viscosity group (26%) | 3 | [43] |
| Hayashi et al. | Injectable Calcium Phosphate Bone Cement Provides Favorable Space and a Scaffold for Periodontal Regeneration in Dogs | 2006 | Assess the influence of injectable calcium phosphate bone cement. | Monocalcium phosphate monohydrate, α-tricalcium phosphate, and calcium carbonate + solution of sodium phosphate | Powder and liquid blended in a capsule for 20 s in amalgam mixer apparatus Application in specially designed applicator | Dog | New bone formation 4.90% ± 0.56% | 3 | [44] |
| Shirakata et al. | Effect of Bone Swaging with Calcium Phosphate Bone Cement on Periodontal Regeneration in Dogs | 2012 | Assess the effects of modified BS + CPC on periodontal healing. | Injectable CPC grafting materials (Norian PDC; Shofu Inc.,Kyoto, Japan) | Powder and liquid blended in a capsule for 20 s in amalgam mixer apparatus Application in specially designed applicator | Dog | Newly formed bone 3.53 ± 0.30 mm | 2 | [45] |
| Oortgiesen et al. | Periodontal Regeneration Using an Injectable Bone Cement Combined with BMP-2 or FGF-2 | 2012 | Histologically assess the healing of CaP cement + BMP-2 or FGF-2. | Injectable CaP gel containing BMP-2 or FGF-2 | Powder and liquid mixed in apparatus (Silamat) and shaken for 15 s Application in a 2 mL syringe | Rat | Bone formation: statistically significant difference in benefit of CaP/BMP group | 3 | [46] |
| Oortgiesen et al. | Regeneration of the Periodontium Using Enamel Matrix Derivative in Combination with an Injectable Bone Cement | 2013 | Histologically assess the healing of CaP + EMD. | Enamel matrix derivate (EMD); PLGA microparticles (Purasorb, Purac, Gorinchem, the Netherlands); CaP (85% alpha-tricalcium phosphate, 10% dicalcium phosphate, 5% precipitated hydroxyapatite) | Powder and liquid mixed in apparatus (Silamat) and shaken for 15 s Application in a 2 mL syringe PLGA microsphere diameter 26 +/− 8 µm | Rat | More bone formation EMD vs. CaP/EMD group (1.9 -> 2.9) 50% | 3 | [47] |
| Author | Title | Year | Aim of the Study | Biomaterial | Preparation/Properties of Biomaterial | Outcomes | Follow-up Period (Months) | Reference |
|---|---|---|---|---|---|---|---|---|
| Lorenz et al. | Injectable Bone Substitute Material on the Basis of β-TCP and Hyaluronan Achieves Complete Bone Regeneration While Undergoing Nearly Complete Degradation | 2018 | Assess the regenerative potential and pathways of injectable bone substitute material. | Β-TCP mixed with an organic substance containing methylcellulose and sodium hyaluronate | Particles size < 63 mm | Newly formed bone 44.92% ± 5.16% Connective tissue 52.49% ± 6.43% Remnants of the IBS 2.59% ± 2.05% | 4 | [21] |
| Weiss et al. | The Safety and Efficacy of an Injectable Bone Substitute in Dental Sockets Demonstrated in a Human Clinical Trial | 2007 | Assess the safety of the filler material and the efficacy of the material for filling human tooth sockets and preventing bone loss. | Injectable calcium phosphate ceramic suspension (CAP ceramic particles suspended in a saline solution containing 2% hydroxylpropylmethyl cellulose) | Application in a 5 mL glass syringe BCP particle diameter 80–200 µm | Radiographic density of alveolar bone crest 76% ± 10% 84% ± 12% | 3 6 | [32] |
| Wolff et al. | Degradable Injectable Bone Cement in Maxillofacial Surgery: Indications and Clinical Experience in 27 Patients | 2004 | Treat the athrophic anterior mandible in combination with the insertion of dental implants. | Injectable CaP-Norian SRS (monocalcium phosphate monohydrate, tricalcium phosphate, calcium carbonate mixed with soidum phospahte solution) | Application of 5–10 mL of biomaterial ICPBC final compressive strength 2.1 MPA | - After 30 months, material completely replaced (only radiologically) - Height of the athropic mandible increased from 13 to 20 mm | 12–30 (mean 29.2 months) -for patients who underwent enodsseous implants—11–26 (mean 15.5 months) | [48] |
| Stanton et al. | Injectable Calcium-Phosphate Bone Cement (Norian) for Reconstruction of a Large Mandibular Defect: A Case Report | 2004 | Reconstruct the large bone defect created by the enucleation of an odotogenic keratocyst. | Norian (Synthes Maxillofacial, West Chester, PA) | ICPBC final compressive strength 2.7 MPA | ¼ of the Norian that was placed sequestered through the mucosa; progressive resorption of Norian and replacement with new bone (only radiologically) | 2 36 | [49] |
| Papanchev et al. | Comparison of the Rates of Bone Regeneration in Sinus Lift Grafting with Calcium Phosphate Paste between the 6th and 9th Month—A Clinical Case | 2015 | Find out whether there are significant differences in bone formation between the 6- and 9-month period after sinus lift grafting. | Maxresorb inject (Botiss Dental, Berlin, Germany) | Syringe | 1. Operation: right sinus lift; 15% of newly formed bone 2. Operation: left sinus lift; 21% of newly formed bone | 9 6 | [50] |
| Khaled et al. | Maxillary Sinus Floor Elevation Using Hydroxyapatite Nanoparticles vs. Tenting Technique with Simultaneous Implant Placement: A Randomized Clinical Trial | 2018 | Assess the amount of bone height gain, density values, and implant stability after sinus augmentation with hydroxyapatite. | Nano-hydroxyapatite bone substitute (Nanostreams, HA nanoparticles (calcium phosphate nanoparticles, Nanostreams MC, Derby, Derbyshire, United Kingdom) | Disposabe syringe | - Bone height Nano group (7.0 ± 0.8 mm) Tent group (5.0 ± 1.5 mm) - Mean bone density Nano group (548 ± 25 HU) Tent group (420 ± 23 HU) - Mean ISQ value Nano group (78 ± 5) Tent group (77 ± 5) | 6 | [51] |
| Georgiev et al. | An Evaluation of Three-Dimensional Scans of the Time-Dependent Volume Changes in Bone Grafting Materials | 2015 | Compare volume loss between bone grafting materials. | Maxresorb inject (calcium phosphate paste composed of 80% nano-hydroxyapatite aquagel and 20% biphasic calcium phosphate granules) | Syringe | Bone graft volume loss 0.5256 cm³ | 36 | [52] |
| Čandrlić et al. | Histological and Radiological Features of Four-Phase Injectable Synthetic Bone Graft in Guided Bone Regeneration—A Case Report | 2021 | Assess the efficacy of ISBG in the managment of bucal fenestration. | ISBG (Maxresorb inject, Botiss Biomaterials GmbH, Berlin, Germany) + native collagen membrane (Collprotect, Botiss Biomaterials GmbH, Berlin, Germany) | Syringe | ROI gray level 138.5 ROI gray level 454 Mineralized tissue 24.76% ISBG 12.56% Soft tissue 62.68% | 10 days 6 months | [53] |
| Patient and Population (P) | Human and Animal Studies |
|---|---|
| Intervention (I) | Application of injectable synthetic bone grafting materials in dentistry |
| Comparator or control group | Application of other types of bone grafting materials |
| Outcomes (O) | Newly formed bone, bone–implant contact, bone–material contact, bone ingrowth, bone density, remnants of IBS, connective tissue, radiographic density of alveolar bone crest, bone graft volume loss |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomas, M.; Čandrlić, M.; Juzbašić, M.; Ivanišević, Z.; Matijević, N.; Včev, A.; Cvijanović Peloza, O.; Matijević, M.; Perić Kačarević, Ž. Synthetic Injectable Biomaterials for Alveolar Bone Regeneration in Animal and Human Studies. Materials 2021, 14, 2858. https://doi.org/10.3390/ma14112858
Tomas M, Čandrlić M, Juzbašić M, Ivanišević Z, Matijević N, Včev A, Cvijanović Peloza O, Matijević M, Perić Kačarević Ž. Synthetic Injectable Biomaterials for Alveolar Bone Regeneration in Animal and Human Studies. Materials. 2021; 14(11):2858. https://doi.org/10.3390/ma14112858
Chicago/Turabian StyleTomas, Matej, Marija Čandrlić, Martina Juzbašić, Zrinka Ivanišević, Nikola Matijević, Aleksandar Včev, Olga Cvijanović Peloza, Marko Matijević, and Željka Perić Kačarević. 2021. "Synthetic Injectable Biomaterials for Alveolar Bone Regeneration in Animal and Human Studies" Materials 14, no. 11: 2858. https://doi.org/10.3390/ma14112858
APA StyleTomas, M., Čandrlić, M., Juzbašić, M., Ivanišević, Z., Matijević, N., Včev, A., Cvijanović Peloza, O., Matijević, M., & Perić Kačarević, Ž. (2021). Synthetic Injectable Biomaterials for Alveolar Bone Regeneration in Animal and Human Studies. Materials, 14(11), 2858. https://doi.org/10.3390/ma14112858











