Lyophilized Polyvinylpyrrolidone Hydrogel for Culture of Human Oral Mucosa Stem Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of Poly (N-vinylpyrrolidone) Hydrogel
2.2. Infrared Spectroscopic Analysis
2.3. Raman Spectroscopy Analysis
2.4. Rheological Measurement
2.5. Fabrication and Characterization of Porous Samples
2.6. Isolation of Primary Human Oral Mucosa Cells
2.7. Flow Cytometry Analysis of Isolated Human Oral Mucosa Cells
2.8. Immunocytochemical Analysis
2.9. Cytotoxicity Assay
2.10. Statistical Analysis
3. Results
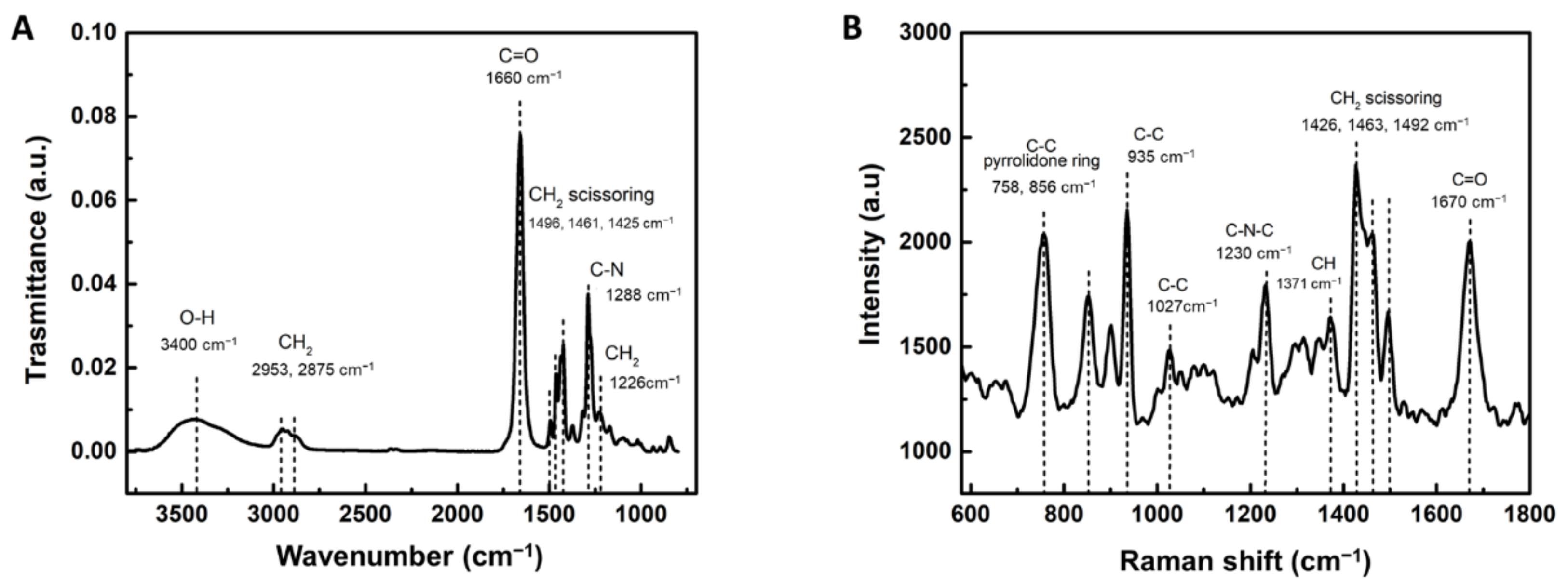
3.1. Chemical Composition of PVP Hydrogel
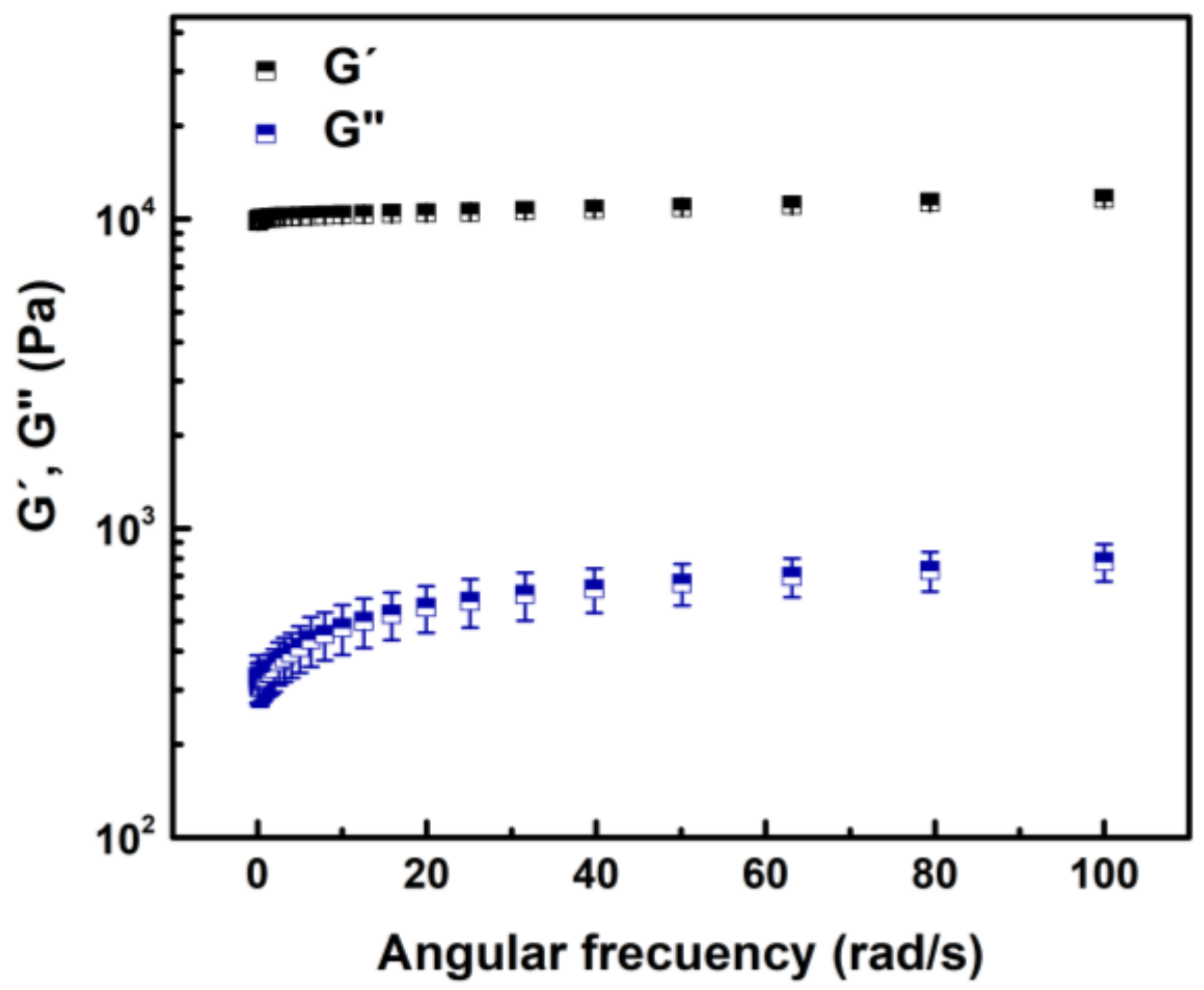
3.2. Oscillatory Frequency Sweep
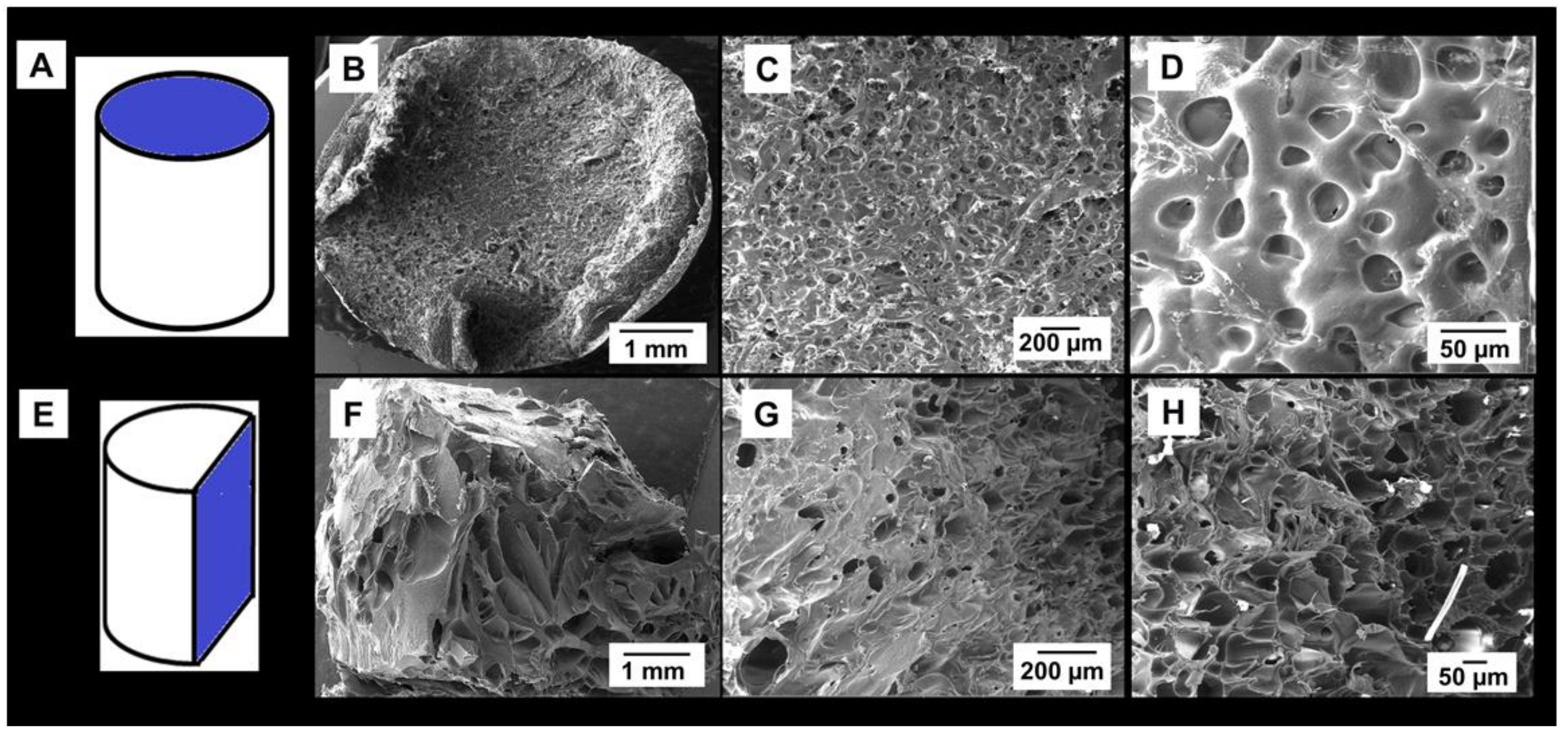
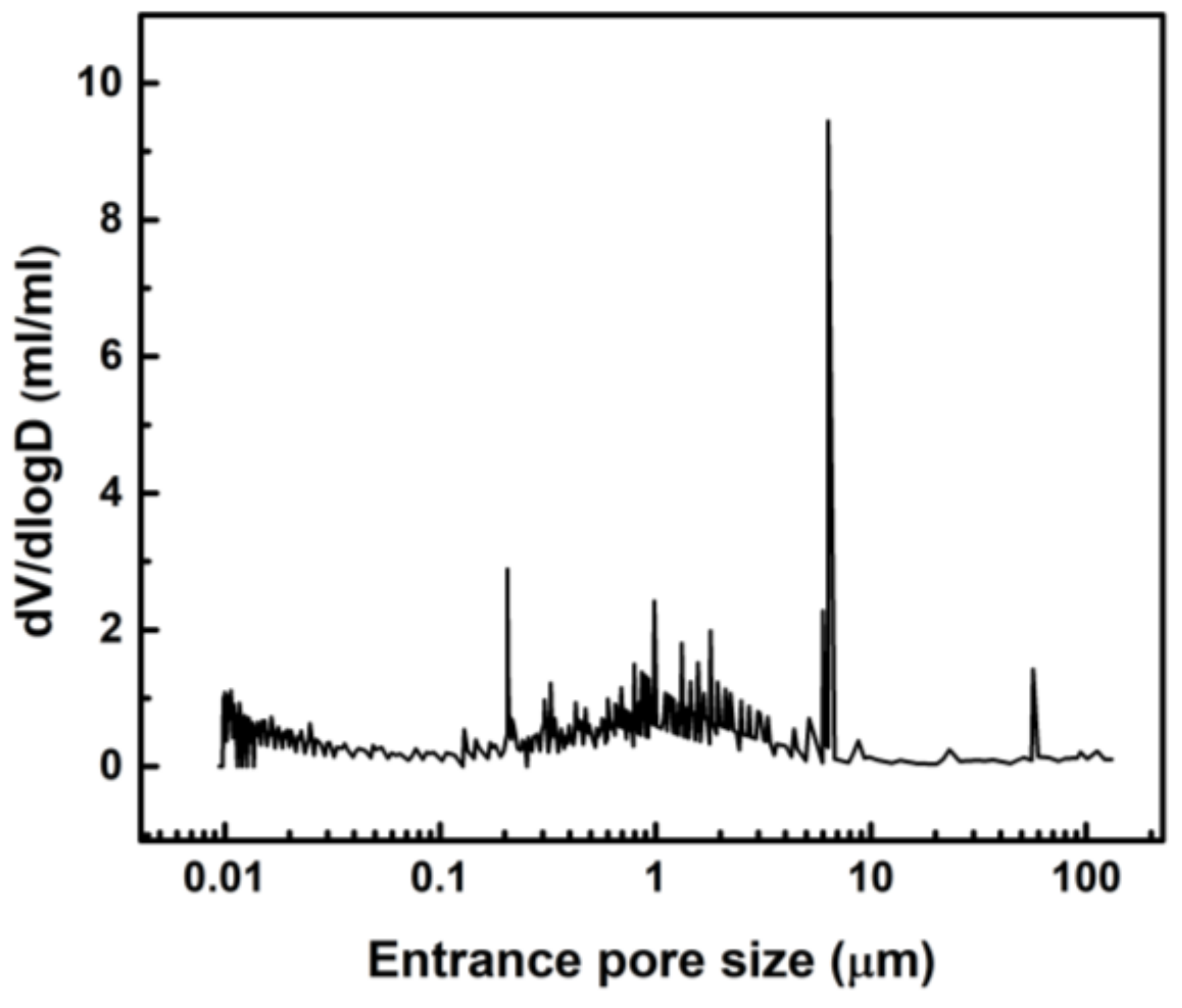
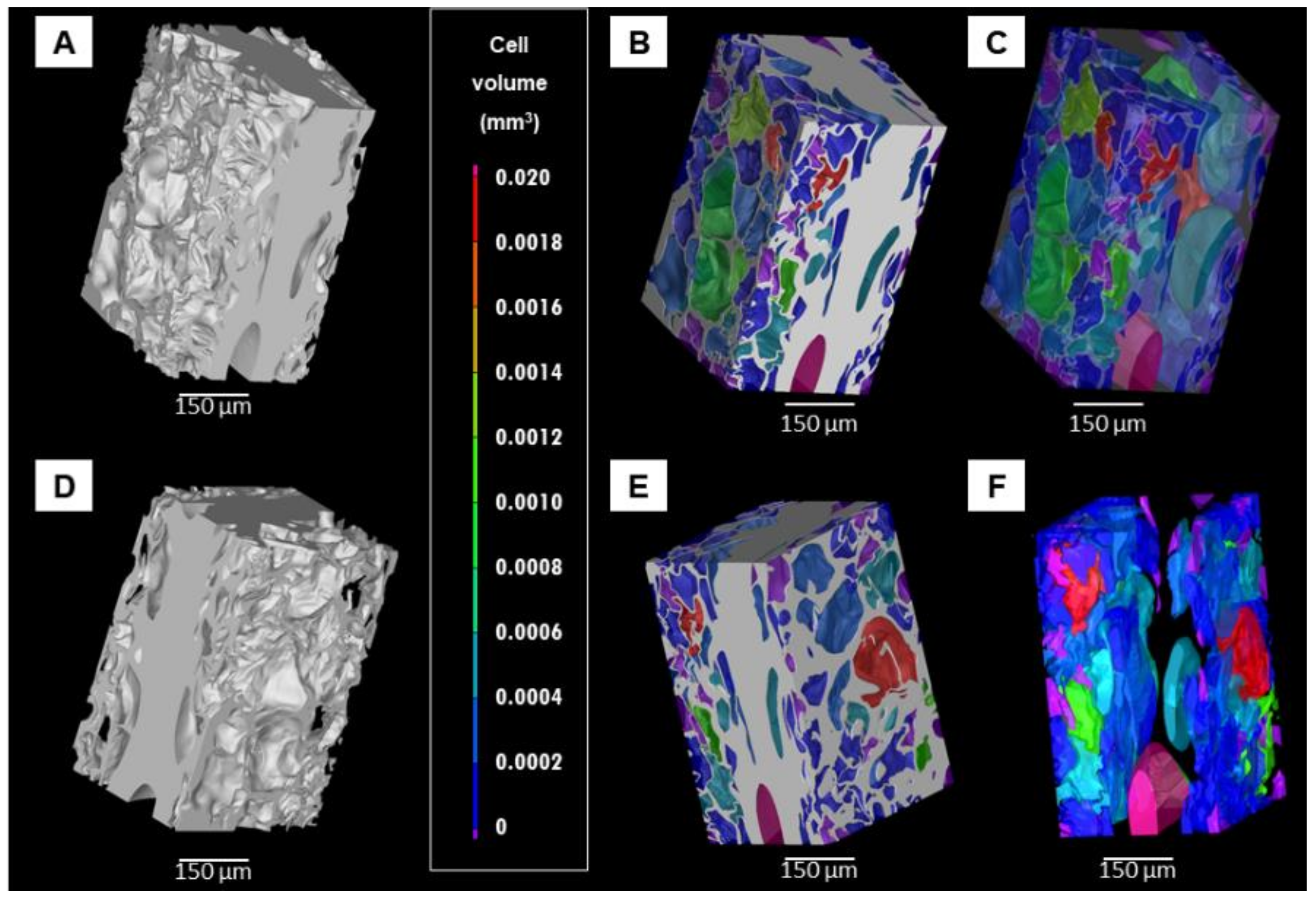
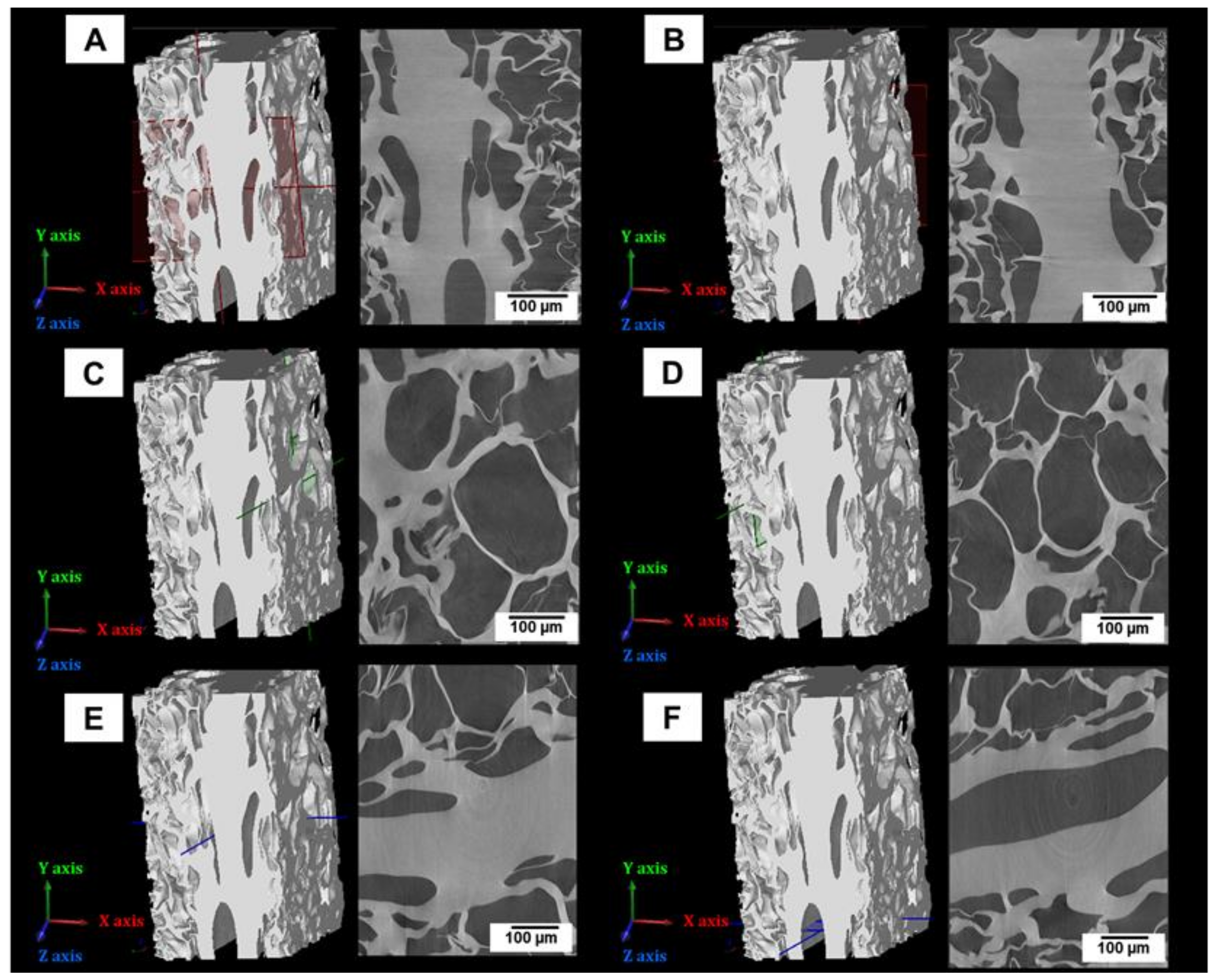
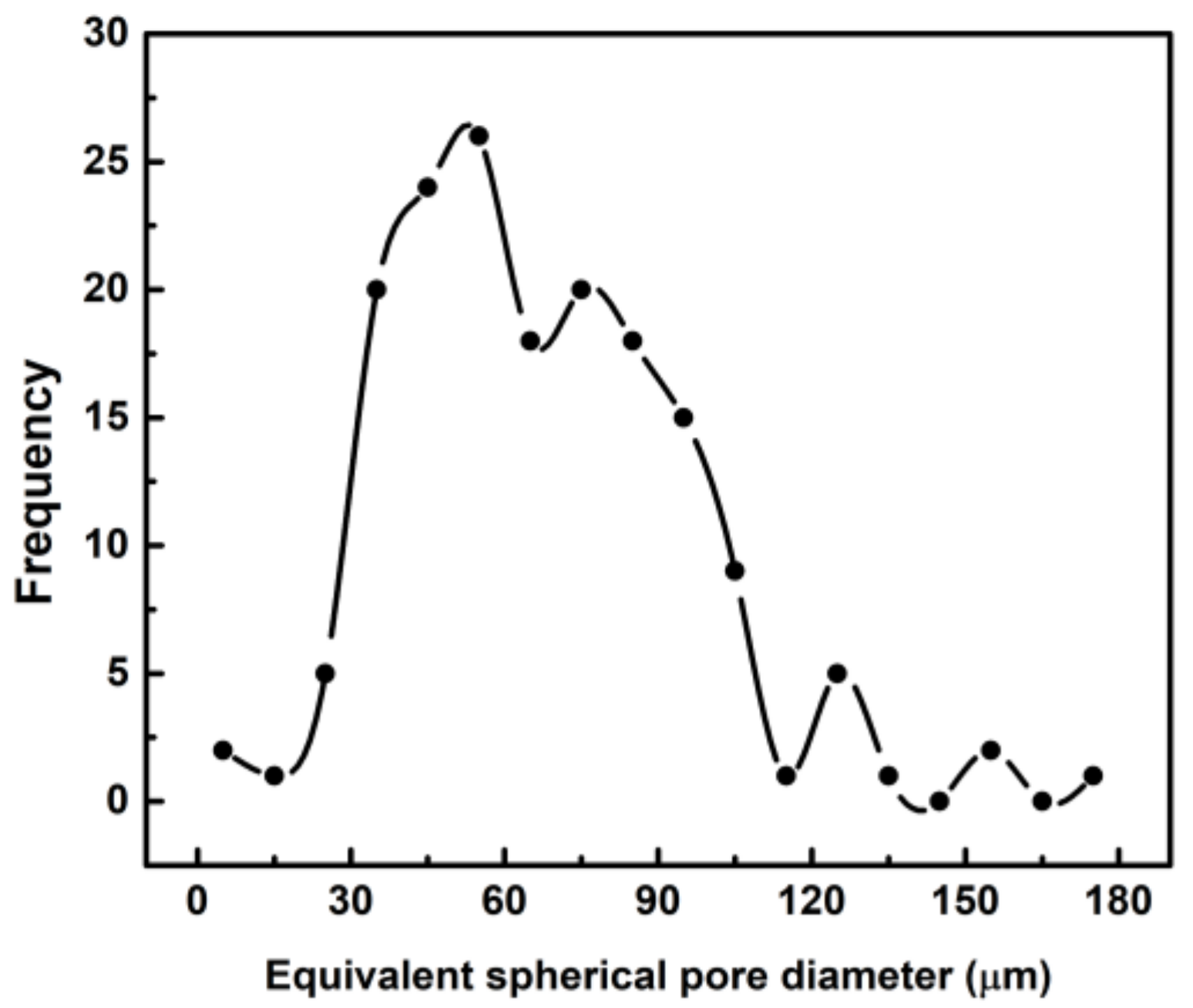
3.3. Porosity Analyses of Lyophilized Hydrogel
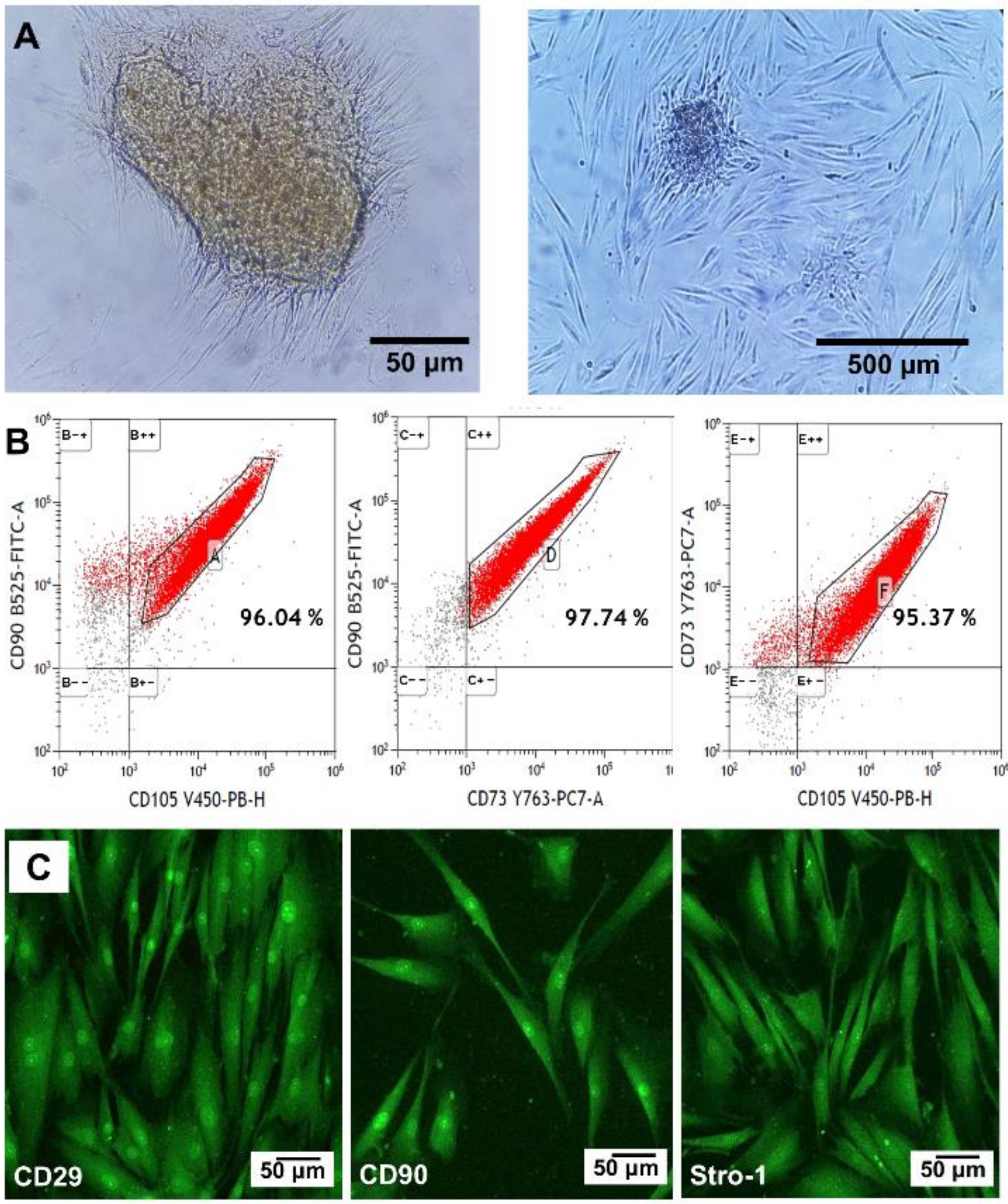
3.4. Primary Human Oral Mucosa Stem Cells
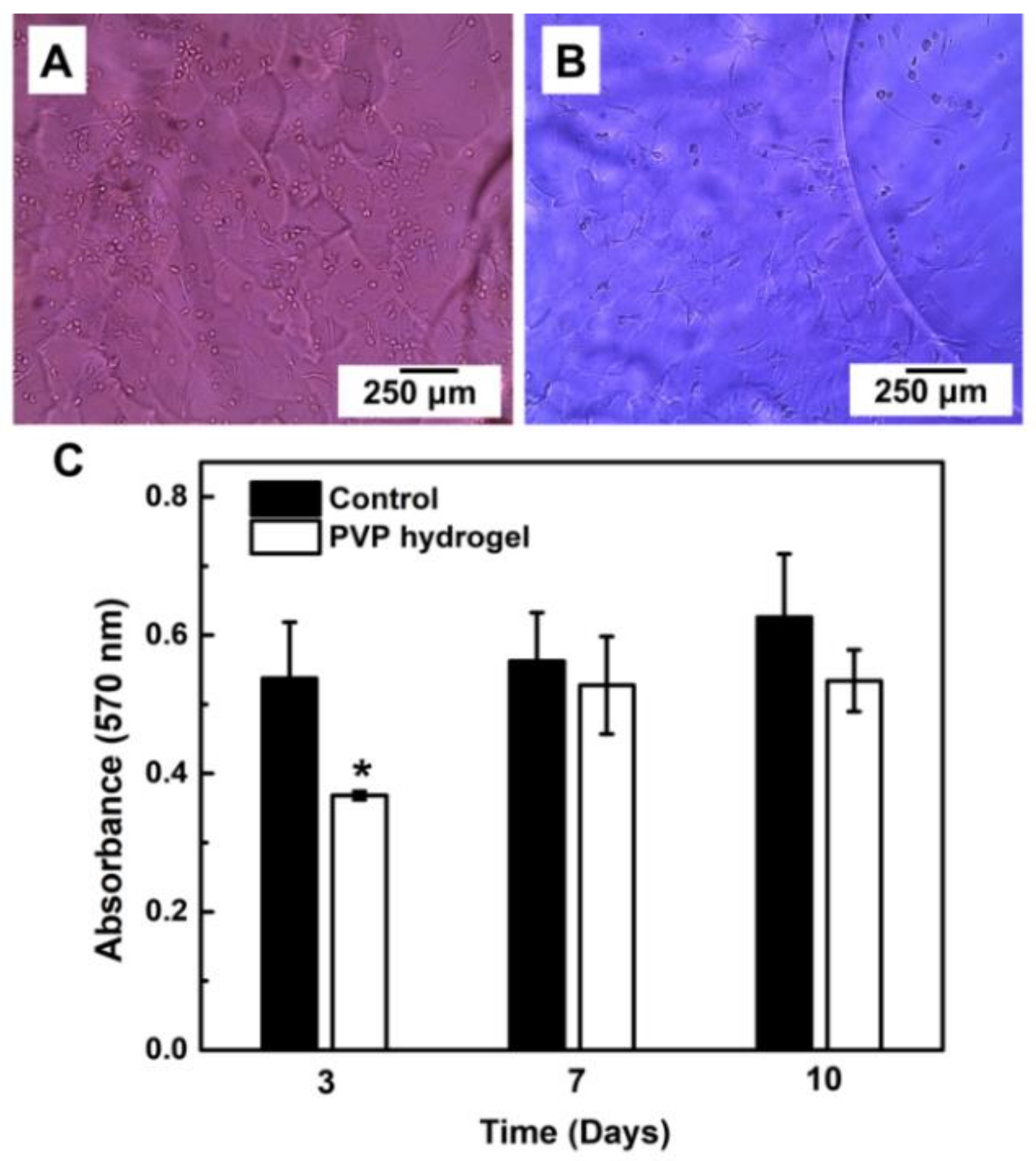
3.5. Hydrogel Cytotoxicity
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Teodorescu, M.; Bercea, M. Poly(vinylpyrrolidone) A Versatile Polymer for Biomedical and Beyond Medical Applications. Polym. Plast. Technol. Eng. 2015, 54, 923–943. [Google Scholar] [CrossRef]
- Lim, J.I.; Im, H.; Lee, W.K. Fabrication of porous chitosan-polyvinyl pyrroldidone scaffolds from a quaternary system via phase separation. J. Biomater. Sci. Polym. Ed. 2015, 26, 32–41. [Google Scholar] [CrossRef]
- Anwar, M.; Pervaiz, F.; Shoukat, H.; Noreen, S.; Shabbir, K.; Majeed, A.; Ijaz, S. Formulation and evaluation of interpenetrating network of xanthan gum and polyvinylpyrrolidone as a hydrophilic matrix for controlled drug delivery system. Polym. Bull. 2020. [Google Scholar] [CrossRef]
- Huang, M.; Hou, Y.; Li, Y.; Wang, D.; Zhang, L. High performances of dual network PVA hydrogel modified by PVP using borax as the structure forming accelerator. Des. Monomers Polym. 2017, 20, 505–513. [Google Scholar] [CrossRef]
- Ng, S.L.; Such, G.K.; Johnston, A.P.R.; Antequera-García, G.; Carusoa, F. Controlled release of DNA from poly (vinylpyrrolidone) capsules using cleavable linkers. Biomaterials 2011, 32, 6277–6284. [Google Scholar] [CrossRef]
- Hu, W.; Wang, Z.; Xiao, Y.; Zhang, S.; Wang, J. Advances in crosslinking strategies of biomedical hydrogels. Biomater. Sci. 2019, 7, 843–855. [Google Scholar] [CrossRef]
- Archana, D.; Singh, B.K.; Dutta, J.; Dutta, P.K. Chitosan-PVP-nano silver oxide wound dressing: In vitro and in vivo evaluation. Int. J. Biol. Macromol. 2015, 73, 49–57. [Google Scholar] [CrossRef]
- Akhmanova, M.; Osidak, E.; Domogatsky, S.; Rodin, S.; Domogatskaya, A. Physical, Spatial, and Molecular Aspects of Extracellular Matrix of In Vivo Niches and Artificial Scaffolds Relevant to Stem Cells Research. Stem Cells Int. 2015, 2015, 67025. [Google Scholar] [CrossRef]
- Ghosh, M.; Halperin-Sternfeld, M.; Adler-Abramovich, L. Bio mimicking of extracellular matrix. Adv. Exp. Med. Biol. 2019, 1174, 371–399. [Google Scholar]
- Catoira, M.C.; Fusaro, L.; Di Francesco, D.; Ramella, M.; Boccafoschi, F. Overview of natural hydrogels for regenerative medicine. J. Mater. Sci. Mater. Med. 2019, 30, 115–125. [Google Scholar] [CrossRef]
- Perez, R.A.; Mestres, G. Role of pore size and morphology in musculo-skeletal tissue regeneration. Mater. Sci. Eng. C 2016, 61, 922–939. [Google Scholar] [CrossRef]
- Autissier, A.; Le Visage, C.; Pouzet, C.; Chaubet, F.; Letourneur, D. Fabrication of porous polysaccharide-based scaffolds using a combined freeze-drying/cross-linking process. Acta Biomater. 2010, 6, 3640–3648. [Google Scholar] [CrossRef]
- Caballé-Serrano, J.; Zhang, S.; Ferrantino, L.; Simion, R.; Chappuis, V.; Bosshardt, D.D. Tissue Response to a Porous Collagen Matrix Used for Soft Tissue Augmentation. Materials 2019, 12, 3721. [Google Scholar] [CrossRef]
- Li, X.; Rombouts, W.; Van der Gucht, J.; De Vries, R.; Dijksam, J.A. Mechanics of composite hydrogels approaching phase separation. PLoS ONE 2019, 14, e0211059. [Google Scholar] [CrossRef]
- Wu, Y.; Chen, Y.X.; Yan, J.; Yang, S.; Dong, P.; Soman, P. Fabrication of conductive polyaniline hydrogel using porogen leaching and projection micro-stereolithography. J. Mater. Chem. B 2015, 3, 5352–5360. [Google Scholar] [CrossRef]
- Tang, Y.; Lin, S.; Yin, S.; Jiang, F.; Zhou, M.; Yang, G.; Sun, N.; Zhang, W.; Jiang, X. In situ gas foaming based on magnesium particle degradation: A novel approach to fabricate injectable macroporous hydrogels. Biomaterials 2020, 232, 119727. [Google Scholar] [CrossRef]
- Hassan, C.M.; Peppas, N.A. Structure and morphology of freeze/thawed PVA hydrogels. Macromolecules 2000, 33, 2472–2479. [Google Scholar] [CrossRef]
- Morariu, S.; Bercea, M.; Teodorescu, M.; Avadanei, M. Tailoring the properties of poly(vinyl alcohol)/poly(vinylpyrrolidone) hydrogels for biomedical applications. Eur. Polym. J. 2016, 84, 313–325. [Google Scholar] [CrossRef]
- Babrnáková, J.; Pavliňáková, V.; Brtníková, J.; Sedláček, P.; Prosecká, E.; Rampichová, M.; Filová, E.; Hearnden, V.; Vojtová, L. Synergistic effect of bovine platelet lysate and various polysaccharides on the biological properties of collagen-based scaffolds for tissue engineering: Scaffold preparation, chemo-physical characterization, in vitro and ex ovo evaluation. Mater. Sci. Eng. C 2019, 100, 236246. [Google Scholar] [CrossRef]
- Savina, I.; Ingavle, G.; Cundy, A.; Mikhalovsky, S.V. A simple method for the production of large volume 3D macroporous hydrogels for advanced biotechnological, medical and environmental applications. Sci. Rep. 2016, 6, 21154. [Google Scholar] [CrossRef]
- Grenier, J.; Duval, H.; Barou, F.; Lv, P.; David, B.; Letourneur, D. Mechanisms of pore formation in hydrogel scaffolds textured by freeze-drying. Acta Biomater. 2019, 94, 195–203. [Google Scholar] [CrossRef]
- Teixeira, M.A.; Amorim, M.; Felgueiras, H.P. Poly(Vinyl Alcohol)-Based Nanofibrous Electrospun Scaffolds for Tissue Engineering Applications. Polymers 2019, 12, 7. [Google Scholar] [CrossRef]
- Risbud, M.V.; Bhonde, M.R.; Bhonde, R.R. Effect of chitosan-polyvinyl pyrrolidone hydrogel on proliferation and cytokine expression of endothelial cells: Implications in islet immunoisolation. J. Biomed. Mater. Res. 2001, 57, 300–305. [Google Scholar] [CrossRef]
- Wang, F.; Yu, M.; Yan, X.; Wen, Y.; Zeng, Q.; Yue, W.; Yang, P.; Pei, X. Gingiva-derived mesenchymal stem cell-mediated therapeutic approach for bone tissue regeneration. Stem Cells Dev. 2011, 20, 2093–2102. [Google Scholar] [CrossRef]
- Han, J.; Menicanin, D.; Marino, V.; Ge, S.; Mrozik, K.; Gronthos, S.; Bartold, P.M. Assessment of the regenerative potential of allogeneic periodontal ligament stem cells in a rodent periodontal defect model. J. Periodontal Res. 2014, 49, 333–345. [Google Scholar] [CrossRef]
- Diomede, F.; Gugliandolo, A.; Cardelli, P. Three-dimensional printed PLA scaffold and human gingival stem cell-derived extracellular vesicles: A new tool for bone defect repair. Stem Cell Res. Ther. 2018, 9, 104. [Google Scholar] [CrossRef]
- Zhang, Q.Z.; Nguyen, A.L.; Yu, W.H.; Le, A.D. Human oral mucosa and gingiva: A unique reservoir for mesenchymal stem cells. J. Dent. Res. 2012, 91, 1011–1018. [Google Scholar] [CrossRef]
- Marynka-Kalmani, K.; Treves, S.; Yafee, M.; Rachima, H.; Gafni, Y.; Cohen, M.A.; Pitaru, S. The Lamina Propria of Adult Human Oral Mucosa Harbors a Novel Stem Cell Population. Stem Cells 2010, 28, 984–995. [Google Scholar] [CrossRef]
- Schmelzer, E.; McKeel, D.; Jörg, G. Characterization of Human Mesenchymal Stem Cells from Different Tissues and Their Membrane Encasement for Prospective Transplantation Therapies. BioMed Res. Int. 2019, 5, 1–13. [Google Scholar] [CrossRef]
- Hernández-Monjaraz, B.; Santiago-Osorio, E.; Monroy-García, A.; Ledesma-Martínez, E.; Mendoza-Núñez, V.M. Mesenchymal Stem Cells of Dental Origin for Inducing Tissue Regeneration in Periodontitis: A Mini-Review. Int. J. Mol. Sci. 2018, 19, 944. [Google Scholar] [CrossRef]
- Schop, D.; Janssen, F.W.; van Rijn, L.; Fernandes, H.; Bloem, R.M.; De Bruijn, J.D.; Van Dijkhuizen-Radersma, R. Growth, Metabolism, and Growth Inhibitors of Mesenchymal Stem Cells. Tissue Eng. Part A 2009, 15, 1877–1886. [Google Scholar] [CrossRef]
- Oliver-Urrutia, C.; Rosales-Ibañez, R.; Domínguez-García, M.V.; Flores-Estrada, J.; Flores-Merino, M.V. Synthesis and evaluation of poly acrylic acid/polyvinylpyrrolidone interpenetrating network as a matrix for oral mucosa cells. J. Biomater. Appl. 2020, 34, 998–1008. [Google Scholar] [CrossRef]
- Flores-Merino, M.V.; Chirasatitsin, S.; Lo Presti, C. Nanoscopic mechanical anisotropy in hydrogel surfaces. Soft Matter 2010, 6, 4466–4470. [Google Scholar] [CrossRef]
- Paganin, D.; Mayo, S.C.; Gureyev, T.E.; Miller, P.R.; Wilkins, S.W. Simultaneous phase and amplitude extraction from a single defocused image of a homogeneous object. J. Microsc. 2002, 206, 33–40. [Google Scholar] [CrossRef]
- Van Aarle, W.; Palenstijn, W.J.; Cant, J.; Janssens, E.; Bleichrodt, F.; Dabravolski, A.; De Beenhouwer, J.; Batenburg, K.J.; Sijbers, J. Fast and flexible X-ray tomography using the ASTRA toolbox. Opt. Express 2016, 24, 25129–25147. [Google Scholar] [CrossRef]
- Shi, Y.; Xiong, D.; Liu, Y.; Wang, N.; Zhao, X. Swelling, mechanical and friction properties of PVA/PVP hydrogels after swelling in osmotic pressure solution. Mater. Sci. Eng. C 2016, 65, 172–180. [Google Scholar] [CrossRef]
- Fanesi, G.; Abrami, M.; Zecchin, F.; Giassi, I.; Dal Ferro, E.; Boisen, A.; Grassi, G.; Bertoncin, P.; Grassi, M.; Marizza, P. Combined Used of Rheology and LF-NMR for the Characterization of PVP-Alginates Gels Containing Liposomes. Pharm. Res. 2018, 35, 171. [Google Scholar] [CrossRef]
- Alsarra, I.A.; Hamed, A.Y.; Alanazi, F.K.; Neau, S.H. Rheological and mucoadhesive characterization of poly (vinylpyrrolidone) hydrogels designed for nasal mucosal drug. Arch. Pharm. Res. 2011, 34, 573–582. [Google Scholar] [CrossRef]
- El-Sayed, K.M.F.; Paris, S.; Becker, S.; Kaseem, N.; Ungefroren, H.; Fändrich, F.; Wiltfang, J.; Dörfer, C. Isolation and characterization of multipotent postnatal stem/progenitor cells from human alveolar bone proper. J. Craniomaxillofac. Surg. 2012, 40, 735–742. [Google Scholar] [CrossRef]
- Lopérgolo, L.C.; Lugao, A.B.; Catalini, L.H. Direct UV photocrosslinking of poly (N-vinyl-2-pyrrolidone) (PVP) to produce hydrogels. Polymer 2003, 44, 6217–6222. [Google Scholar] [CrossRef]
- Cheng, S.; Gandevia, S.C.; Green, M.; Sinkus, R.; Bilston, L.E. Viscoelastic properties of the tongue and soft palate using MR elastography. J. Biomech. 2011, 44, 450–454. [Google Scholar] [CrossRef]
- Chen, J.; Ahmad, R.; Li, W.; Swain, M.; Ki, Q. Biomechanics of oral mucosa. J. R. Soc. Interface 2015, 12, 20150325. [Google Scholar] [CrossRef]
- Cameron, A.R.; Frith, J.E.; Cooper-White, J.J. The influence of substrate creep on mesenchymal stem cell behaviour and phenotype. Biomaterials 2011, 32, 5979–5993. [Google Scholar] [CrossRef]
- Barber-Pérez, N.; Georgiadou, M.; Guzmán, C.; Isomursu, A.; Hamidi, H.; Ivaska, J. Mechano-responsiveness of Fibrillar Adhesions on Stiffness-Gradient Gels. J. Cell Sci. 2020, 133, 242909. [Google Scholar] [CrossRef]
- Das, P.; Salerno, S.; Remigy, J.C.; Lahitte, J.F.; Bacchin, P.; De Bartolo, L. Double porous poly (Ɛ-caprolactone)/chitosan membrane scaffolds as niches for human mesenchymal stem cells. Colloids Surf. B 2019, 184, 110493. [Google Scholar] [CrossRef]
- Bonartsev, A.P.; Zharkova, I.I.; Voinova, V.V.; Kuznetsova, E.S.; Zhuikov, V.A.; Makhina, T.K.; Myshkina, V.L.; Potashnikova, D.M.; Chesnokova, D.V.; Khaydapova, D.D.; et al. Poly (3-hydroxybutyrate)/poly(ethylene glycol) scaffolds with different microstructure: The effect on growth of mesenchymal stem cells. 3 Biotech 2018, 8, 328. [Google Scholar] [CrossRef]
- Annabi, N.; Nichol, J.W.; Zhong, X.; Ji, C.; Koshy, S.; Khademhosseini, A.; Dehghani, F. Controlling the porosity and microarchitecture of hydrogels for tissue engineering. Tissue Eng. Part B 2010, 16, 371–383. [Google Scholar] [CrossRef]
- Romero, E.; Simms, P.H. Microstructure investigation in unsaturated soils: A Review with special attention to contribution of mercury intrusion porosimetry and environmental scanning electron microscopy. Geotech. Geol. Eng. 2008, 26, 705–727. [Google Scholar] [CrossRef]
- Lawyer, T.; McIntosh, K.; Clavijo, C.; Potekhina, L.; Mann, B.K. Formulation Changes Affect Material Properties and Cell Behavior in HA-Based Hydrogels. Int. J. Cell Biol. 2012, 2012, 1–9. [Google Scholar] [CrossRef]
- Izgordu, M.S.; Uzgur, E.I.; Ulag, S.; Sahin, A.; Yilmaz, B.K.; Kilic, B.; Ekren, N.; Oktar, F.N.; Gunduz, O. Investigation of 3D-Printed Polycaprolactone-/Polyvinylpyrrolidone-Based Constructs. Cartilage 2019, 1, 1–10. [Google Scholar] [CrossRef]
- Xiong, S.; Wang, J.; Zhu, W.; Yang, K.; Ding, G.; Li, X.; Eun, D.D. Onlay repair technique for the management of ureteral strictures: A comprehensive review. Biomed Res. Int. 2020, 27, 6178286. [Google Scholar] [CrossRef]
- Ganz, J.; Shor, E.; Guo, S.; Sheinin, A.; Arie, I.; Michaelevski, I.; Pitaru, S.; Offen, D.; Levenberg, S. Implantation of 3D Constructs Embedded with Oral Mucosa-Derived Cells Induces Functional Recovery in Rats with Complete Spinal Cord Transection. Front. Neurosci. 2017, 11, 589. [Google Scholar] [CrossRef]
- Egusa, H.; Sonoyama, W.; Nishimura, M.; Atsuta, I.; Akiyama, K. Stem cells in dentistry-part I: Stem cell sources. J. Prosthodont Res. 2012, 56, 151–165. [Google Scholar] [CrossRef]
- Abou-Neel, E.A.; Chrzanowski, W.; Salih, V.M.; Kim, H.W.; Knowles, J.C. Tissue engineering in dentistry. J. Dent. 2014, 42, 915–928. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliver-Urrutia, C.; Rosales Ibañez, R.; Flores-Merino, M.V.; Vojtova, L.; Salplachta, J.; Čelko, L.; Kaiser, J.; Montufar, E.B. Lyophilized Polyvinylpyrrolidone Hydrogel for Culture of Human Oral Mucosa Stem Cells. Materials 2021, 14, 227. https://doi.org/10.3390/ma14010227
Oliver-Urrutia C, Rosales Ibañez R, Flores-Merino MV, Vojtova L, Salplachta J, Čelko L, Kaiser J, Montufar EB. Lyophilized Polyvinylpyrrolidone Hydrogel for Culture of Human Oral Mucosa Stem Cells. Materials. 2021; 14(1):227. https://doi.org/10.3390/ma14010227
Chicago/Turabian StyleOliver-Urrutia, Carolina, Raúl Rosales Ibañez, Miriam V. Flores-Merino, Lucy Vojtova, Jakub Salplachta, Ladislav Čelko, Jozef Kaiser, and Edgar B. Montufar. 2021. "Lyophilized Polyvinylpyrrolidone Hydrogel for Culture of Human Oral Mucosa Stem Cells" Materials 14, no. 1: 227. https://doi.org/10.3390/ma14010227
APA StyleOliver-Urrutia, C., Rosales Ibañez, R., Flores-Merino, M. V., Vojtova, L., Salplachta, J., Čelko, L., Kaiser, J., & Montufar, E. B. (2021). Lyophilized Polyvinylpyrrolidone Hydrogel for Culture of Human Oral Mucosa Stem Cells. Materials, 14(1), 227. https://doi.org/10.3390/ma14010227







