The Bad and the Good—Microorganisms in Cultural Heritage Environments—An Update on Biodeterioration and Biotreatment Approaches
Abstract
1. Introduction
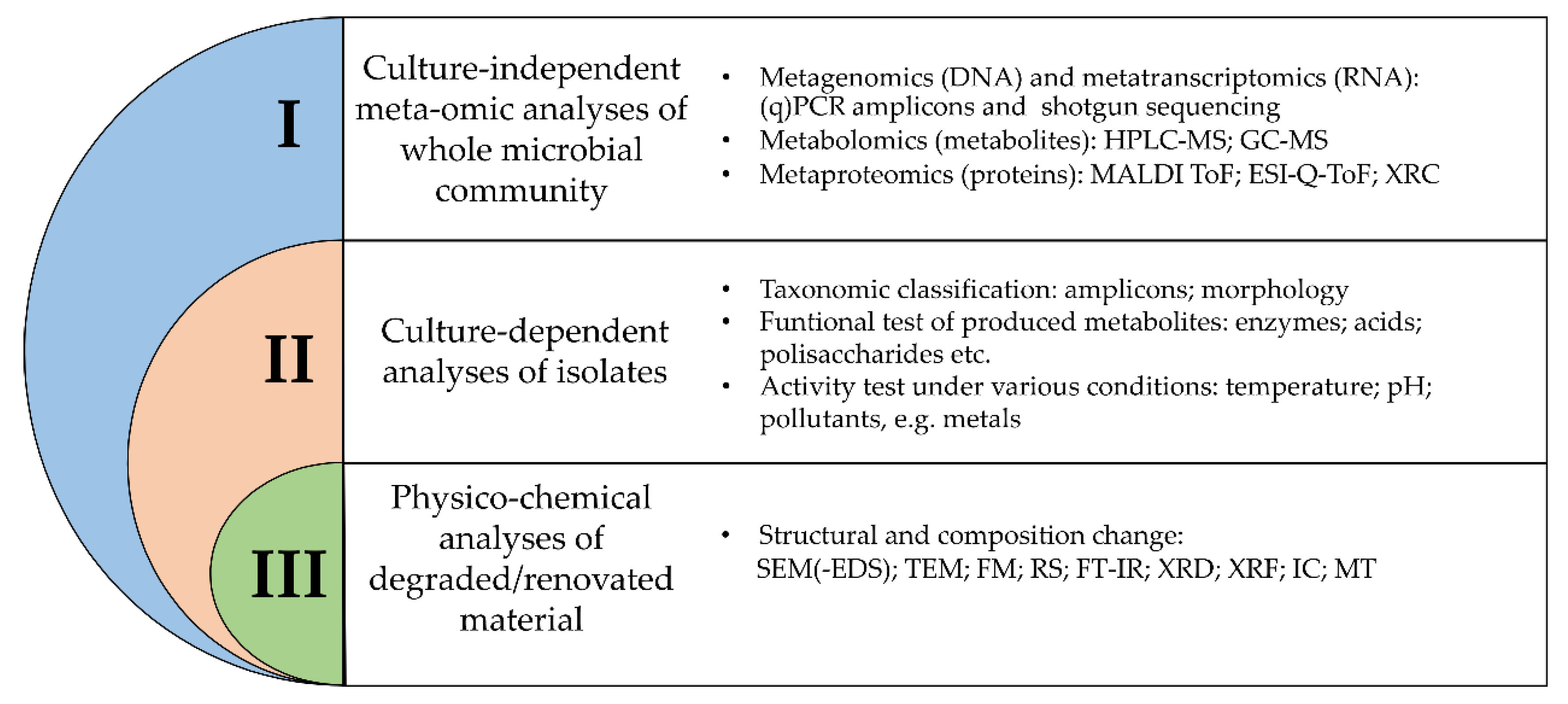
2. Inspection of Methodologies Applied for Identification and Characterization of Microorganisms in Cultural Heritage Objects
2.1. Culture-Independent Meta-Omic Analyses
2.2. Culture-Dependent Analyses
2.3. Physico-Chemical Analyses
3. Microorganism-Driven Deterioration of Historical Artefacts
4. Biotechnological Use of Microorganisms in Cultural Heritage
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Drábková, K.; Ďurovič, M.; Kučerová, I. Influence of gamma radiation on properties of paper and textile fibres during disinfection. Radiat. Phys. Chem. 2018, 152, 75–80. [Google Scholar] [CrossRef]
- Anna, W.; Dorota, R.; Mansur, R.; Sławomir, W. Microorganisms colonising historical cardboard objects from the Auschwitz-Birkenau State Museum in Oświęcim, Poland and their disinfection with vaporised hydrogen peroxide (VHP). Int. Biodeterior. Biodegrad. 2020, 152. [Google Scholar] [CrossRef]
- Turnau, K.; Jędrzejczyk, R.J.; Ważny, R.; Chlebda, D.; Janicka, M.; Pawcenis, D.; Łojewski, T. Microbes of XVI century Arrases of Krakow Royal Castle. Microbiol. Res. 2020, 238. [Google Scholar] [CrossRef] [PubMed]
- Palla, F.; Bruno, M.; Mercurio, F.; Tantillo, A.; Rotolo, V. Essential oils as natural biocides in conservation of cultural heritage. Molecules 2020, 25, 730. [Google Scholar] [CrossRef] [PubMed]
- Fidanza, M.R.; Caneva, G. Natural biocides for the conservation of stone cultural heritage: A review. J. Cult. Herit. 2019, 38, 271–286. [Google Scholar] [CrossRef]
- Kakakhel, M.A.; Wu, F.; Gu, J.D.; Feng, H.; Shah, K.; Wang, W. Controlling biodeterioration of cultural heritage objects with biocides: A review. Int. Biodeterior. Biodegrad. 2019, 143, 104721. [Google Scholar] [CrossRef]
- Reyes-Estebanez, M.; Ortega-Morales, B.O.; Chan-Bacab, M.; Granados-Echegoyen, C.; Camacho-Chab, J.C.; Pereañez-Sacarias, J.E.; Gaylarde, C. Antimicrobial engineered nanoparticles in the built cultural heritage context and their ecotoxicological impact on animals and plants: A brief review. Herit. Sci. 2018, 6, 1–11. [Google Scholar] [CrossRef]
- Marvasi, M.; Mastromei, G.; Perito, B. Bacterial Calcium Carbonate Mineralization in situ Strategies for Conservation of Stone Artworks: From Cell Components to Microbial Community. Front. Microbiol. 2020, 11, 1186. [Google Scholar] [CrossRef]
- Rampazzi, L. Calcium oxalate films on works of art: A review. J. Cult. Herit. 2019, 40, 195–214. [Google Scholar] [CrossRef]
- Soffritti, I.; D’Accolti, M.; Lanzoni, L.; Volta, A.; Bisi, M.; Mazzacane, S.; Caselli, E. The potential use of microorganisms as restorative agents: An update. Sustainability 2019, 11, 3853. [Google Scholar] [CrossRef]
- Troiano, F.; Gulotta, D.; Balloi, A.; Polo, A.; Toniolo, L.; Lombardi, E.; Daffonchio, D.; Sorlini, C.; Cappitelli, F. Successful combination of chemical and biological treatments forthecleaning of stone artworks. Int. Biodeterior. Biodegrad. 2013, 85, 294–304. [Google Scholar] [CrossRef]
- Rampazzo, M.; Manente, S.; Micheluz, A.; Ganzerla, R.; Ravagnan, G. How traces of pollutants in the environment modify bioremediation efficiency performed with Desulfovibrio vulgaris, and the advantage of an optimization protocol using soft chemicals. J. Cult. Herit. 2018, 30. [Google Scholar] [CrossRef]
- Elhagrassy, A.F.; Hakeem, A.; Alhagrassy, A.F. Comparative Study of Biological Cleaning and Laser Techniques for Conservation of Weathered Stone in Failaka Island, Kuwait. Sci. Cult. 2018, 4. [Google Scholar] [CrossRef]
- Rubin, J.; Yin, T.; Au, G.; Chang, B.; Chee, M.; Cheu, L.; Datels, C.; Fischer, L.; Hoffman, M.; Kondaurova, O.; et al. TEA/AECOM 2018 Theme Index and Museum Index: The Global Attractions Attendance Report; Themed Entertainment Association (TEA): Buena Vista, CO, USA, 2019; pp. 1–90. [Google Scholar]
- Kim, K.H.; Kabir, E.; Jahan, S.A. Airborne bioaerosols and their impact on human health. J. Environ. Sci. 2018, 67, 23–35. [Google Scholar] [CrossRef]
- Rascovan, N.; Telke, A.; Raoult, D.; Rolain, J.M.; Desnues, C. Exploring divergent antibiotic resistance genes in ancient metagenomes and discovery of a novel beta-lactamase family. Environ. Microbiol. Rep. 2016, 8. [Google Scholar] [CrossRef]
- Herfst, S.; Böhringer, M.; Karo, B.; Lawrence, P.; Lewis, N.S.; Mina, M.J.; Russell, C.J.; Steel, J.; de Swart, R.L.; Menge, C. Drivers of airborne human-to-human pathogen transmission. Curr. Opin. Virol. 2017, 22, 22–29. [Google Scholar] [CrossRef]
- Adamiak, J.; Bonifay, V.; Otlewska, A.; Sunner, J.A.; Beech, I.B.; Stryszewska, T.; Kanka, S.; Oracz, J.; Zyzelewicz, D.; Gutarowska, B. Untargeted metabolomics approach in halophiles: Understanding the biodeterioration process of building materials. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef]
- Dyda, M.; Pyzik, A.; Wilkojc, E.; Kwiatkowska-Kopka, B.; Sklodowska, A. Bacterial and fungal diversity inside the medieval building constructed with sandstone plates and lime mortar as an example of the microbial colonization of a nutrient-limited extreme environment (Wawel royal castle, Krakow, Poland). Microorganisms 2019, 7, 416. [Google Scholar] [CrossRef]
- Li, A.; Xiong, J.; Yao, L.; Gou, L.; Zhang, W. Determination of dust and microorganism accumulation in different designs of AHU system in Shaanxi History Museum. Build. Environ. 2016, 104. [Google Scholar] [CrossRef]
- Adamiak, J.; Otlewska, A.; Tafer, H.; Lopandic, K.; Gutarowska, B.; Sterflinger, K.; Piñar, G. First evaluation of the microbiome of built cultural heritage by using the Ion Torrent next generation sequencing platform. Int. Biodeterior. Biodegrad. 2018, 131, 11–18. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, Y.; Zhang, F.; Hu, C.; Liu, G.; Pan, J. Microbial community analyses of the deteriorated storeroom objects in the Tianjin Museum using culture-independent and culture-dependent approaches. Front. Microbiol. 2018, 9. [Google Scholar] [CrossRef]
- Grottoli, A.; Beccaccioli, M.; Zoppis, E.; Fratini, R.S.; Schifano, E.; Santarelli, M.L.; Uccelletti, D.; Reverberi, M. Nanopore Sequencing and Bioinformatics for Rapidly Identifying Cultural Heritage Spoilage Microorganisms. Front. Mater. 2020, 7. [Google Scholar] [CrossRef]
- Ghosh, B.; Lal, H.; Srivastava, A. Review of bioaerosols in indoor environment with special reference to sampling, analysis and control mechanisms. Environ. Int. 2015, 85, 254–272. [Google Scholar] [CrossRef] [PubMed]
- Bøifot, K.O.; Gohli, J.; Moen, L.V.; Dybwad, M. Performance evaluation of a new custom, multi-component DNA isolation method optimized for use in shotgun metagenomic sequencing-based aerosol microbiome research. Environ. Microbiomes 2020, 15. [Google Scholar] [CrossRef]
- Ferguson, R.M.W.; Garcia-Alcega, S.; Coulon, F.; Dumbrell, A.J.; Whitby, C.; Colbeck, I. Bioaerosol biomonitoring: Sampling optimization for molecular microbial ecology. Mol. Ecol. Resour. 2019, 19. [Google Scholar] [CrossRef] [PubMed]
- Piñar, G.; Tafer, H.; Sterflinger, K.; Pinzari, F. Amid the possible causes of a very famous foxing: Molecular and microscopic insight into Leonardo da Vinci’s self-portrait. Environ. Microbiol. Rep. 2015, 7. [Google Scholar] [CrossRef]
- Goyal, P.; Bhola, D.; Varma, A. Introduction to modern tools and techniques to understand microbes. In Modern Tools and Techniques to Understand Microbes; Springer: Cham, Switzerland, 2017; pp. 1–23. [Google Scholar]
- Gutarowska, B.; Pietrzak, K.; Machnowski, W.; Milczarek, J.M. Historical textiles – a review of microbial deterioration analysis and disinfection methods. Text. Res. J. 2017, 87, 2388–2406. [Google Scholar] [CrossRef]
- Sanmartín, P.; DeAraujo, A.; Vasanthakumar, A. Melding the Old with the New: Trends in Methods Used to Identify, Monitor, and Control Microorganisms on Cultural Heritage Materials. Microb. Ecol. 2018, 76, 64–80. [Google Scholar] [CrossRef]
- Gutarowska, B. The use of -omics tools for assessing biodeterioration of cultural heritage: A review. J. Cult. Herit. 2020, 351–361. [Google Scholar] [CrossRef]
- Adams, R.I.; Bateman, A.C.; Bik, H.M.; Meadow, J.F. Microbiota of the indoor environment: A meta-analysis. Microbiome 2015, 3. [Google Scholar] [CrossRef] [PubMed]
- Barb, J.J.; Oler, A.J.; Kim, H.S.; Chalmers, N.; Wallen, G.R.; Cashion, A.; Munson, P.J.; Ames, N.J. Development of an Analysis Pipeline Characterizing Multiple Hypervariable Regions of 16S rRNA Using Mock Samples. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Fouhy, F.; Clooney, A.G.; Stanton, C.; Claesson, M.J.; Cotter, P.D. 16S rRNA gene sequencing of mock microbial populations-impact of DNA extraction method, primer choice and sequencing platform. BMC Microbiol. 2016, 16. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.H.; Su, J.H.; Shang, J.J.; Wu, Y.Y.; Li, Y.; Bao, D.P.; Yao, Y.J. Evaluation of the ribosomal DNA internal transcribed spacer (ITS), specifically ITS1 and ITS2, for the analysis of fungal diversity by deep sequencing. PLoS ONE 2018, 13. [Google Scholar] [CrossRef] [PubMed]
- Mbareche, H.; Veillette, M.; Bilodeau, G.; Duchaine, C. Comparison of the performance of ITS1 and ITS2 as barcodes in amplicon-based sequencing of bioaerosols. PeerJ 2020, 2020. [Google Scholar] [CrossRef] [PubMed]
- Sze, M.A.; Schloss, P.D. The Impact of DNA Polymerase and Number of Rounds of Amplification in PCR on 16S rRNA Gene Sequence Data. mSphere 2019, 4. [Google Scholar] [CrossRef] [PubMed]
- Bolyen, E.; Rideout, J.R.; Dillon, M.R.; Bokulich, N.A.; Abnet, C.C.; Al-Ghalith, G.A.; Alexander, H.; Alm, E.J.; Arumugam, M.; Asnicar, F.; et al. Reproducible, interactive, scalable and extensible microbiome data science using QIIME 2. Nat. Biotechnol. 2019, 37, 852–857. [Google Scholar] [CrossRef]
- Pauvert, C.; Buée, M.; Laval, V.; Edel-Hermann, V.; Fauchery, L.; Gautier, A.; Lesur, I.; Vallance, J.; Vacher, C. Bioinformatics matters: The accuracy of plant and soil fungal community data is highly dependent on the metabarcoding pipeline. Fungal Ecol. 2019, 41. [Google Scholar] [CrossRef]
- Prodan, A.; Tremaroli, V.; Brolin, H.; Zwinderman, A.H.; Nieuwdorp, M.; Levin, E. Comparing bioinformatic pipelines for microbial 16S rRNA amplicon sequencing. PLoS ONE 2020, 15. [Google Scholar] [CrossRef]
- Ettenauer, J.; Piñar, G.; Tafer, H.; Sterflinger, K. Quantification of fungal abundance on cultural heritage using real time PCR targeting the β-actin gene. Front. Microbiol. 2014, 5. [Google Scholar] [CrossRef]
- Unterwurzacher, V.; Pogner, C.; Berger, H.; Strauss, J.; Strauss-Goller, S.; Gorfer, M. Validation of a quantitative PCR based detection system for indoor mold exposure assessment in bioaerosols. Environ. Sci. Process. Impacts 2018, 20, 1454–1468. [Google Scholar] [CrossRef] [PubMed]
- del Mondo, A.; de Natale, A.; Pinto, G.; Pollio, A. Correction to: Novel qPCR probe systems for the characterization of subaerial biofilms on stone monuments. Ann. Microbiol. 2019, 69. [Google Scholar] [CrossRef]
- Šoltys, K.; Planý, M.; Biocca, P.; Vianello, V.; Bučková, M.; Puškárová, A.; Sclocchi, M.C.; Colaizzi, P.; Bicchieri, M.; Pangallo, D.; et al. Lead soaps formation and biodiversity in a XVIII Century wax seal coloured with minium. Environ. Microbiol. 2020, 22. [Google Scholar] [CrossRef] [PubMed]
- Nygaard, A.B.; Tunsjø, H.S.; Meisal, R.; Charnock, C. A preliminary study on the potential of Nanopore MinION and Illumina MiSeq 16S rRNA gene sequencing to characterize building-dust microbiomes. Sci. Rep. 2020, 10. [Google Scholar] [CrossRef]
- Piñar, G.; Poyntner, C.; Lopandic, K.; Tafer, H.; Sterflinger, K. Rapid diagnosis of biological colonization in cultural artefacts using the MinION nanopore sequencing technology. Int. Biodeterior. Biodegrad. 2020, 148. [Google Scholar] [CrossRef]
- Louati, M.; Ennis, N.J.; Ghodhbane-Gtari, F.; Hezbri, K.; Sevigny, J.L.; Fahnestock, M.F.; Cherif-Silini, H.; Bryce, J.G.; Tisa, L.S.; Gtari, M. Elucidating the ecological networks in stone-dwelling microbiomes. Environ. Microbiol. 2020, 22. [Google Scholar] [CrossRef]
- Bertrand, D.; Shaw, J.; Kalathiyappan, M.; Ng, A.H.Q.; Kumar, M.S.; Li, C.; Dvornicic, M.; Soldo, J.P.; Koh, J.Y.; Tong, C.; et al. Hybrid metagenomic assembly enables high-resolution analysis of resistance determinants and mobile elements in human microbiomes. Nat. Biotechnol. 2019, 37. [Google Scholar] [CrossRef]
- Meng, H.; Zhang, X.; Katayama, Y.; Ge, Q.; Gu, J.D. Microbial diversity and composition of the Preah Vihear temple in Cambodia by high-throughput sequencing based on genomic DNA and RNA. Int. Biodeterior. Biodegrad. 2020, 149. [Google Scholar] [CrossRef]
- Van Vliet, S. Bacterial Dormancy: How to Decide When to Wake Up. Curr. Biol. 2015, 25, R753–R755. [Google Scholar] [CrossRef]
- Villa, F.; Vasanthakumar, A.; Mitchell, R.; Cappitelli, F. RNA-based molecular survey of biodiversity of limestone tombstone microbiota in response to atmospheric sulphur pollution. Lett. Appl. Microbiol. 2015, 60, 92–102. [Google Scholar] [CrossRef]
- Kooli, W.M.; Junier, T.; Shakya, M.; Monachon, M.; Davenport, K.W.; Vaideeswaran, K.; Vernudachi, A.; Marozau, I.; Monrouzeau, T.; Gleasner, C.D.; et al. Remedial treatment of corroded iron objects by environmental Aeromonas isolates. Appl. Environ. Microbiol. 2019, 85. [Google Scholar] [CrossRef]
- Shakya, M.; Lo, C.C.; Chain, P.S.G. Advances and challenges in metatranscriptomic analysis. Front. Genet. 2019, 10, 904. [Google Scholar] [CrossRef] [PubMed]
- Gutarowska, B.; Celikkol-Aydin, S.; Bonifay, V.; Otlewska, A.; Aydin, E.; Oldham, A.L.; Brauer, J.I.; Duncan, K.E.; Adamiak, J.; Sunner, J.A.; et al. Metabolomic and high-throughput sequencing analysis-modern approach for the assessment of biodeterioration of materials from historic buildings. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Szulc, J.; Jablonskaja, I.; Jabłońska, E.; Ruman, T.; Karbowska-Berent, J.; Gutarowska, B. Metabolomics and metagenomics characteristic of historic beeswax seals. Int. Biodeterior. Biodegrad. 2020, 152. [Google Scholar] [CrossRef]
- Szulc, J.; Ruman, T.; Karbowska-Berent, J.; Kozielec, T.; Gutarowska, B. Analyses of microorganisms and metabolites diversity on historic photographs using innovative methods. J. Cult. Herit. 2020. [Google Scholar] [CrossRef]
- Kraková, L.; Šoltys, K.; Otlewska, A.; Pietrzak, K.; Purkrtová, S.; Savická, D.; Puškárová, A.; Bučková, M.; Szemes, T.; Budiš, J.; et al. Comparison of methods for identification of microbial communities in book collections: Culture-dependent (sequencing and MALDI-TOF MS) and culture-independent (Illumina MiSeq). Int. Biodeterior. Biodegrad. 2018, 131. [Google Scholar] [CrossRef]
- Manfredi, M.; Barberis, E.; Gosetti, F.; Conte, E.; Gatti, G.; Mattu, C.; Robotti, E.; Zilberstein, G.; Koman, I.; Zilberstein, S.; et al. Method for Noninvasive Analysis of Proteins and Small Molecules from Ancient Objects. Anal. Chem. 2017, 89. [Google Scholar] [CrossRef]
- Barberis, E.; Baiocco, S.; Conte, E.; Gosetti, F.; Rava, A.; Zilberstein, G.; Righetti, P.G.; Marengo, E.; Manfredi, M. Towards the non-invasive proteomic analysis of cultural heritage objects. Microchem. J. 2018, 139. [Google Scholar] [CrossRef]
- Giuffrida, M.G.; Mazzoli, R.; Pessione, E. Back to the past: Deciphering cultural heritage secrets by protein identification. Appl. Microbiol. Biotechnol. 2018, 102, 5445–5455. [Google Scholar] [CrossRef]
- Atlas, R. Handbook of Microbiological Media, 4th ed.; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar]
- Basu, S.; Bose, C.; Ojha, N.; Das, N.; Das, J.; Pal, M.; Khurana, S. Evolution of Bacterial and Fungal Growth Media. Bioinformation 2015, 11, 182–184. [Google Scholar] [CrossRef]
- Bonnet, M.; Lagier, J.C.; Raoult, D.; Khelaifia, S. Bacterial culture through selective and non-selective conditions: The evolution of culture media in clinical microbiology. New Microbes New Infect. 2020, 34, 100622. [Google Scholar] [CrossRef] [PubMed]
- Laiz, L.; Piñar, G.; Lubitz, W.; Saiz-Jimenez, C. Monitoring the colonization of monuments by bacteria: Cultivation versus molecular methods. Environ. Microbiol. 2003, 5. [Google Scholar] [CrossRef] [PubMed]
- Dziurzynski, M.; Ciuchcinski, K.; Dyda, M.; Szych, A.; Drabik, P.; Laudy, A.; Dziewit, L. Assessment of bacterial contamination of air at the museum of King John III’s palace at wilanow (Warsaw, Poland): Selection of an optimal growth medium for analyzing airborne bacteria diversity. Appl. Sci. 2020, 10, 7128. [Google Scholar] [CrossRef]
- Pangallo, D.; Bučková, M.; Kraková, L.; Puškárová, A.; Šaková, N.; Grivalský, T.; Chovanová, K.; Zemánková, M. Biodeterioration of epoxy resin: A microbial survey through culture-independent and culture-dependent approaches. Environ. Microbiol. 2015, 17. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, D.K.; Khulan, A.; Kim, J. Development of a novel cultivation technique for uncultured soil bacteria. Sci. Rep. 2019, 9. [Google Scholar] [CrossRef] [PubMed]
- Gutleben, J.; Chaib De Mares, M.; van Elsas, J.D.; Smidt, H.; Overmann, J.; Sipkema, D. The multi-omics promise in context: From sequence to microbial isolate. Crit. Rev. Microbiol. 2018, 44, 212–229. [Google Scholar] [CrossRef] [PubMed]
- Savković, Ž.; Stupar, M.; Unković, N.; Ivanović, Ž.; Blagojević, J.; Vukojević, J.; Ljaljević Grbić, M. In vitro biodegradation potential of airborne Aspergilli and Penicillia. Sci. Nat. 2019, 106. [Google Scholar] [CrossRef]
- Pavić, A.; Ilić-Tomić, T.; Pačevski, A.; Nedeljković, T.; Vasiljević, B.; Morić, I. Diversity and biodeteriorative potential of bacterial isolates from deteriorated modern combined-technique canvas painting. Int. Biodeterior. Biodegrad. 2015, 97. [Google Scholar] [CrossRef]
- Lepinay, C.; Mihajlovski, A.; Touron, S.; Seyer, D.; Bousta, F.; Di Martino, P. Bacterial diversity associated with saline efflorescences damaging the walls of a French decorated prehistoric cave registered as a World Cultural Heritage Site. Int. Biodeterior. Biodegrad. 2018, 130. [Google Scholar] [CrossRef]
- Cojoc, L.R.; Enache, M.I.; Neagu, S.E.; Lungulescu, M.; Setnescu, R.; Ruginescu, R.; Gomoiu, I. Carotenoids produced by halophilic bacterial strains on mural paintings and laboratory conditions. FEMS Microbiol. Lett. 2019, 366. [Google Scholar] [CrossRef]
- Kavkler, K.; Gunde Cimerman, N.; Zalar, P.; Demšar, A. FT-Raman analysis of cellulose based museum textiles: Comparison of objects infected and non-infected by fungi. Tekstilec 2018, 61. [Google Scholar] [CrossRef]
- Margariti, C. The application of FTIR microspectroscopy in a non-invasive and non-destructive way to the study and conservation of mineralised excavated textiles. Herit. Sci. 2019, 7. [Google Scholar] [CrossRef]
- Daher, C.; Bellot-Gurlet, L.; Le Hô, A.S.; Paris, C.; Regert, M. Advanced discriminating criteria for natural organic substances of Cultural Heritage interest: Spectral decomposition and multivariate analyses of FT-Raman and FT-IR signatures. Talanta 2013, 115. [Google Scholar] [CrossRef] [PubMed]
- Caneve, L.; Guarneri, M.; Lai, A.; Spizzichino, V.; Ceccarelli, S.; Mazzei, B. Non-destructive laser based techniques for biodegradation analysis in cultural heritage. NDT E Int. 2019, 104. [Google Scholar] [CrossRef]
- Troiano, F.; Polo, A.; Villa, F.; Cappitelli, F. Assessing the microbiological risk to stored sixteenth century parchment manuscripts: A holistic approach based on molecular and environmental studies. Biofouling 2014, 30. [Google Scholar] [CrossRef]
- Unković, N.; Ljaljević Grbić, M.; Stupar, M.; Vukojević, J.; Subakov-Simić, G.; Jelikić, A.; Stanojević, D. ATP bioluminescence method: Tool for rapid screening of organic and microbial contaminants on deteriorated mural paintings. Nat. Prod. Res. 2019, 33. [Google Scholar] [CrossRef]
- Sirt Çıplak, E.; Kiraz, G.A. Enzymatic Activity as a Measure of Total Microbial Activity on Historical Stone. Heritage 2020, 3, 38. [Google Scholar] [CrossRef]
- Negi, A.; Sarethy, I.P. Microbial Biodeterioration of Cultural Heritage: Events, Colonization, and Analyses. Microb. Ecol. 2019, 78, 1014–1029. [Google Scholar] [CrossRef]
- Mazzoli, R.; Giuffrida, M.G.; Pessione, E. Back to the past: “Find the guilty bug-microorganisms involved in the biodeterioration of archeological and historical artifacts”. Appl. Microbiol. Biotechnol. 2018, 102, 6393–6407. [Google Scholar] [CrossRef]
- Castrillón Rivera, L.E.; Palma Ramos, A.; Castañeda Sánchez, J.I.; Elisa Drago Serrano, M. Origin and control strategies of biofilms in the cultural heritage. In Antimicrobials, Antibiotic Resistance, Antibiofilm Strategies and Activity Methods; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Poyatos, F.; Morales, F.; Nicholson, A.W.; Giordano, A. Physiology of biodeterioration on canvas paintings. J. Cell. Physiol. 2018, 233, 2741–2751. [Google Scholar] [CrossRef]
- Sharif-Askari, H.; Abu-Hijleh, B. Review of museums’ indoor environment conditions studies and guidelines and their impact on the museums’ artifacts and energy consumption. Build. Environ. 2018, 143. [Google Scholar] [CrossRef]
- Stephens, B. What Have We Learned about the Microbiomes of Indoor Environments? mSystems 2016, 1. [Google Scholar] [CrossRef] [PubMed]
- Meadow, J.F.; Altrichter, A.E.; Kembel, S.W.; Kline, J.; Mhuireach, G.; Moriyama, M.; Northcutt, D.; O’Connor, T.K.; Womack, A.M.; Brown, G.Z.; et al. Indoor airborne bacterial communities are influenced by ventilation, occupancy, and outdoor air source. Indoor Air 2014, 24, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Ii, A.J.P.; Marr, L.C. Sources of airborne microorganisms in the built environment. Microbiome 2015, 1–10. [Google Scholar] [CrossRef]
- Villa, F.; Pitts, B.; Lauchnor, E.; Cappitelli, F.; Stewart, P.S. Development of a laboratory model of a phototroph-heterotroph mixed-species biofilm at the stone/air interface. Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Deveau, A.; Bonito, G.; Uehling, J.; Paoletti, M.; Becker, M.; Bindschedler, S.; Hacquard, S.; Hervé, V.; Labbé, J.; Lastovetsky, O.A.; et al. Bacterial-fungal interactions: Ecology, mechanisms and challenges. FEMS Microbiol. Rev. 2018, 42, 335–352. [Google Scholar] [CrossRef]
- Leplat, J.; Bousta, F.; François, A.; Guiavarc’h, M.; Mertz, J.D.; Brissaud, D. The pink staircase of Sully-sur-Loire castle: Even bacteria like historic stonework. Int. Biodeterior. Biodegrad. 2019, 145. [Google Scholar] [CrossRef]
- Paiva de Carvalho, H.; Sequeira, S.O.; Pinho, D.; Trovão, J.; da Costa, R.M.F.; Egas, C.; Macedo, M.F.; Portugal, A. Combining an innovative non-invasive sampling method and high-throughput sequencing to characterize fungal communities on a canvas painting. Int. Biodeterior. Biodegrad. 2019. [Google Scholar] [CrossRef]
- Kim, Y.S.; Singh, A.P. Wood as cultural heritage material and its deterioration by biotic and abiotic agents. In Secondary Xylem Biology: Origins, Functions, and Applications; Academic Press: Cambridge, MA, USA, 2016. [Google Scholar] [CrossRef]
- Sterflinger, K.; Piñar, G. Microbial deterioration of cultural heritage and works of art—Tilting at windmills? Appl. Microbiol. Biotechnol. 2013, 97, 9637–9646. [Google Scholar] [CrossRef]
- Li, Q.; Zhang, B.; Yang, X.; Ge, Q. Deterioration-associated microbiome of stone monuments: Structure, variation, and assembly. Appl. Environ. Microbiol. 2018, 84. [Google Scholar] [CrossRef]
- Ma, Y.; Zhang, H.; Du, Y.; Tian, T.; Xiang, T.; Liu, X.; Wu, F.; An, L.; Wang, W.; Gu, J.D.; et al. The community distribution of bacteria and fungi on ancient wall paintings of the Mogao Grottoes. Sci. Rep. 2015, 5. [Google Scholar] [CrossRef] [PubMed]
- Ma, W.; Wu, F.; Tian, T.; He, D.; Zhang, Q.; Gu, J.D.; Duan, Y.; Ma, D.; Wang, W.; Feng, H. Fungal diversity and its contribution to the biodeterioration of mural paintings in two 1700-year-old tombs of China. Int. Biodeterior. Biodegrad. 2020, 152. [Google Scholar] [CrossRef]
- Nugari, M.P.; Pietrini, A.M.; Caneva, G.; Imperi, F.; Visca, P. Biodeterioration of mural paintings in a rocky habitat: The Crypt of the Original Sin (Matera, Italy). Int. Biodeterior. Biodegrad. 2009, 63. [Google Scholar] [CrossRef]
- Schütz, M.K.; Schlegel, M.L.; Libert, M.; Bildstein, O. Impact of Iron-Reducing Bacteria on the Corrosion Rate of Carbon Steel under Simulated Geological Disposal Conditions. Environ. Sci. Technol. 2015, 49. [Google Scholar] [CrossRef] [PubMed]
- Beale, D.J.; Karpe, A.V.; Jadhav, S.; Muster, T.H.; Palombo, E.A. Omics-based approaches and their use in the assessment of microbial-influenced corrosion of metals. Corros. Rev. 2016, 34. [Google Scholar] [CrossRef]
- Sakr, A.A.; Ghaly, M.F.; Edwards, H.G.M.; Ali, M.F.; Abdel-Haliem, M.E.F. Involvement of Streptomyces in the Deterioration of Cultural Heritage Materials Through Biomineralization and Bio-Pigment Production Pathways: A Review. Geomicrobiol. J. 2020, 37, 653–662. [Google Scholar] [CrossRef]
- Dakal, T.C.; Cameotra, S.S. Microbially induced deterioration of architectural heritages: Routes and mechanisms involved. Environ. Sci. Eur. 2012, 24. [Google Scholar] [CrossRef]
- Handa, Y.; Tazato, N.; Nagatsuka, Y.; Koide, T.; Kigawa, R.; Sano, C.; Sugiyama, J. Stenotrophomonas tumulicola sp. nov., a major contaminant of the stone chamber interior in the Takamatsuzuka Tumulus. Int. J. Syst. Evol. Microbiol. 2016, 66. [Google Scholar] [CrossRef]
- Soares, F.; Tiago, I.; Trovão, J.; Coelho, C.; Mesquita, N.; Gil, F.; Catarino, L.; Cardoso, S.M.; Portugal, A. Description of Myxacorys almedinensis sp. nov. (Synechococcales, Cyanobacteria) isolated from the limestone walls of the Old Cathedral of Coimbra, Portugal (UNESCO World Heritage Site). Phytotaxa 2019, 419. [Google Scholar] [CrossRef]
- Ponizovskaya, V.B.; Grum-Grzhimaylo, A.A.; Georgieva, M.L.; Kokaeva, L.Y.; Bilanenko, E.N. Lecanicillium gracile (Cordycipitaceae), a new species isolated from mineral building materials. Phytotaxa 2020, 443. [Google Scholar] [CrossRef]
- Coronado-Ruiz, C.; Avendaño, R.; Escudero-Leyva, E.; Conejo-Barboza, G.; Chaverri, P.; Chavarría, M. Two new cellulolytic fungal species isolated from a 19th-century art collection. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Trovão, J.; Tiago, I.; Soares, F.; Paiva, D.S.; Mesquita, N.; Coelho, C.; Catarino, L.; Gil, F.; Portugal, A. Description of Aeminiaceae fam. Nov., Aeminium gen. Nov. and Aeminium ludgeri sp. Nov. (Capnodiales), isolated from a biodeteriorated art-piece in the Old Cathedral of Coimbra, Portugal. MycoKeys 2019, 45. [Google Scholar] [CrossRef] [PubMed]
- Leplat, J.; François, A.; Bousta, F. Parengyodontium album, a frequently reported fungal species in the cultural heritage environment. Fungal Biol. Rev. 2020. [Google Scholar] [CrossRef]
- Wagner, S.; Lagane, F.; Seguin-Orlando, A.; Schubert, M.; Leroy, T.; Guichoux, E.; Chancerel, E.; Bech-Hebelstrup, I.; Bernard, V.; Billard, C.; et al. High-Throughput DNA sequencing of ancient wood. Mol. Ecol. 2018, 27, 1138–1154. [Google Scholar] [CrossRef] [PubMed]
- Veneranda, M.; Prieto-Taboada, N.; de Vallejuelo, S.F.O.; Maguregui, M.; Morillas, H.; Marcaida, I.; Castro, K.; Madariaga, J.M.; Osanna, M. Biodeterioration of Pompeian mural paintings: Fungal colonization favoured by the presence of volcanic material residues. Environ. Sci. Pollut. Res. 2017, 24, 19599–19608. [Google Scholar] [CrossRef] [PubMed]
- Rosado, T.; Dias, L.; Lança, M.; Nogueira, C.; Santos, R.; Martins, M.R.; Candeias, A.; Mirão, J.; Caldeira, A.T. Assessment of microbiota present on a Portuguese historical stone convent using high-throughput sequencing approaches. Microbiologyopen 2020, 9. [Google Scholar] [CrossRef] [PubMed]
- Brewer, T.E.; Fierer, N. Tales from the tomb: The microbial ecology of exposed rock surfaces. Environ. Microbiol. 2018, 20. [Google Scholar] [CrossRef]
- Meng, H.; Katayama, Y.; Gu, J.D. More wide occurrence and dominance of ammonia-oxidizing archaea than bacteria at three Angkor sandstone temples of Bayon, Phnom Krom and Wat Athvea in Cambodia. Int. Biodeterior. Biodegrad. 2017, 117. [Google Scholar] [CrossRef]
- Mihajlovski, A.; Lepinay, C.; Mirval, A.L.; Touron, S.; Bousta, F.; Di Martino, P. Characterization of the archaeal and fungal diversity associated with gypsum efflorescences on the walls of the decorated Sorcerer’s prehistoric cave. Ann. Microbiol. 2019, 69. [Google Scholar] [CrossRef]
- Perito, B.; Marvasi, M.; Barabesi, C.; Mastromei, G.; Bracci, S.; Vendrell, M.; Tiano, P. A Bacillus subtilis cell fraction (BCF) inducing calcium carbonate precipitation: Biotechnological perspectives for monumental stone reinforcement. J. Cult. Herit. 2014, 15, 345–351. [Google Scholar] [CrossRef]
- Gherardi, F.; Turyanska, L.; Ferrari, E.; Weston, N.; Fay, M.W.; Colston, B.J. Immobilized Enzymes on Gold Nanoparticles: From Enhanced Stability to Cleaning of Heritage Textiles. ACS Appl. Bio Mater. 2019, 2, 5136–5143. [Google Scholar] [CrossRef]
- Mazzoni, M.; Alisi, C.; Tasso, F.; Cecchini, A.; Marconi, P.; Sprocati, A.R. Laponite micro-packs for the selective cleaning of multiple coherent deposits on wall paintings: The case study of Casina Farnese on the Palatine Hill (Rome-Italy). Int. Biodeterior. Biodegrad. 2014, 94, 1–11. [Google Scholar] [CrossRef]
- Fouad ElHagrassy, A. Bio-Restoration of Mural Paintings Using Viable Cells of Pseudomonas stutzeri and Characterization of These Murals. Int. J. Archaeol. 2019, 7, 8. [Google Scholar] [CrossRef]
- Ranalli, G.; Zanardini, E.; Rampazzi, L.; Corti, C.; Andreotti, A.; Colombini, M.P.; Bosch-Roig, P.; Lustrato, G.; Giantomassi, C.; Zari, D.; et al. Onsite advanced biocleaning system for historical wall paintings using new agar-gauze bacteria gel. J. Appl. Microbiol. 2019, 126. [Google Scholar] [CrossRef] [PubMed]
- Andreolli, M.; Lampis, S.; Bernardi, P.; Calò, S.; Vallini, G. Bacteria from black crusts on stone monuments can precipitate CaCO3 allowing the development of a new bio-consolidation protocol for ornamental stone. Int. Biodeterior. Biodegrad. 2020, 153. [Google Scholar] [CrossRef]
- Jroundi, F.; Schiro, M.; Ruiz-Agudo, E.; Elert, K.; Martín-Sánchez, I.; González-Muñoz, M.T.; Rodriguez-Navarro, C. Protection and consolidation of stone heritage by self-inoculation with indigenous carbonatogenic bacterial communities. Nat. Commun. 2017, 8, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Navarro, C.; Jroundi, F.; Gonzalez-Muñoz, M.T. Stone Consolidation by Bacterial Carbonatogenesis: Evaluation of in situ Applications. Restor. Build. Monum. 2015, 21. [Google Scholar] [CrossRef]
- Romano, I.; Abbate, M.; Poli, A.; D’Orazio, L. Bio-cleaning of nitrate salt efflorescence on stone samples using extremophilic bacteria. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Bosch-Roig, P.; Allegue, H.; Bosch, I. Granite pavement nitrate desalination: Traditional methods vs. biocleaning methods. Sustainability 2019, 11, 4227. [Google Scholar] [CrossRef]
- Daskalakis, M.I.; Magoulas, A.; Kotoulas, G.; Catsikis, I.; Bakolas, A.; Karageorgis, A.P.; Mavridou, A.; Doulia, D.; Rigas, F. Pseudomonas, Pantoea and Cupriavidus isolates induce calcium carbonate precipitation for biorestoration of ornamental stone. J. Appl. Microbiol. 2013, 115, 409–423. [Google Scholar] [CrossRef]
- Xu, J.; Wang, X. Self-healing of concrete cracks by use of bacteria-containing low alkali cementitious material. Constr. Build. Mater. 2018, 167, 1–14. [Google Scholar] [CrossRef]
- Comensoli, L.; Maillard, J.; Albini, M.; Sandoz, F.; Junier, P.; Joseph, E. Use of bacteria to stabilize archaeological iron. Appl. Environ. Microbiol. 2017, 83. [Google Scholar] [CrossRef] [PubMed]
- Kooli, W.M.; Comensoli, L.; Maillard, J.; Albini, M.; Gelb, A.; Junier, P.; Joseph, E. Bacterial iron reduction and biogenic mineral formation for the stabilisation of corroded iron objects. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Cote, C.; Rosas, O.; Basseguy, R. Geobacter sulfurreducens: An iron reducing bacterium that can protect carbon steel against corrosion? Corros. Sci. 2015, 94. [Google Scholar] [CrossRef]
- Barbabietola, N.; Tasso, F.; Alisi, C.; Marconi, P.; Perito, B.; Pasquariello, G.; Sprocati, A.R. A safe microbe-based procedure for a gentle removal of aged animal glues from ancient paper. Int. Biodeterior. Biodegrad. 2016, 109. [Google Scholar] [CrossRef]
- Jeszeová, L.; Benžová, R.; Gluštíková, M.; Šišková, A.; Kisová, Z.; Planý, M.; Kraková, L.; Bauerová-Hlinková, V.; Pangallo, D. Biocleaning of historical documents: The use and characterization of bacterial enzymatic resources. Int. Biodeterior. Biodegrad. 2019, 140. [Google Scholar] [CrossRef]
- Albelda Berenguer, M.; Monachon, M.; Jacquet, C.; Junier, P.; Rémazeilles, C.; Schofield, E.J.; Joseph, E. Biological oxidation of sulfur compounds in artificially degraded wood. Int. Biodeterior. Biodegrad. 2019, 141. [Google Scholar] [CrossRef]
- Sanmartín, P.; Bosch-Roig, P. Biocleaning to remove graffiti: A real possibility? Advances towards a complete protocol of action. Coatings 2019, 9, 104. [Google Scholar] [CrossRef]
- Mazzoli, R.; Giuffrida, M.G.; Pessione, E. Back to the past—Forever young: Cutting-edge biochemical and microbiological tools for cultural heritage conservation. Appl. Microbiol. Biotechnol. 2018, 102, 6815–6825. [Google Scholar] [CrossRef]
- Seifan, M.; Berenjian, A. Microbially induced calcium carbonate precipitation: A widespread phenomenon in the biological world. Appl. Microbiol. Biotechnol. 2019, 103. [Google Scholar] [CrossRef]
- Castro-Alonso, M.J.; Montañez-Hernandez, L.E.; Sanchez-Muñoz, M.A.; Macias Franco, M.R.; Narayanasamy, R.; Balagurusamy, N. Microbially induced calcium carbonate precipitation (MICP) and its potential in bioconcrete: Microbiological and molecular concepts. Front. Mater. 2019, 6, 1–15. [Google Scholar] [CrossRef]
- Chandra, P.; Singh, E.; Kumar, R.; Ahmad, J. The role of microorganisms in removal of sulfates from artistic stonework. In Microbial Biotechnology Approaches to Monuments of Cultural Heritage; Springer: Singapore, 2020; pp. 103–135. [Google Scholar]
- Albini, M.; Letardi, P.; Mathys, L.; Brambilla, L.; Schröter, J.; Junier, P.; Joseph, E. Comparison of a bio-based corrosion inhibitor versus benzotriazole on corroded copper surfaces. Corros. Sci. 2018, 143. [Google Scholar] [CrossRef]
- Ahmed, H.E.; Kolisis, F.N. An investigation into the removal of starch paste adhesives from historical textiles by using the enzyme α-amylase. J. Cult. Herit. 2011, 12. [Google Scholar] [CrossRef]
- Silva, M.; Rosado, T.; Teixeira, D.; Candeias, A.; Caldeira, A.T. Green mitigation strategy for cultural heritage: Bacterial potential for biocide production. Environ. Sci. Pollut. Res. 2017, 24. [Google Scholar] [CrossRef] [PubMed]
- Caselli, E.; Pancaldi, S.; Baldisserotto, C.; Petrucci, F.; Impallaria, A.; Volpe, L.; D’Accolti, M.; Soffritti, I.; Coccagna, M.; Sassu, G.; et al. Characterization of biodegradation in a 17th century easel painting and potential for a biological approach. PLoS ONE 2018, 13. [Google Scholar] [CrossRef] [PubMed]
| Material | Bacteria | Fungi | Reference |
|---|---|---|---|
| Textile and canvas paintings | Achromobacter sp., Alcaligenes sp., Arthrobacter sp., Bacillus sp., Brevibacterium sp., Cellulomonas sp., Cellvibrio sp., Cellfalcicula sp., Clostridium sp., Corynebacterium sp., Cytophaga sp., Halobacillus sp., Kocuria sp., Micrococcus sp., Microspora sp., Microbispora sp., Myxococcoides sp., Nocardia sp., Oceanobacillus sp., Paracoccus sp., Paenisporosarcina sp., Pseudomonas sp., Proteus sp., Rhodococcus sp., Sporocytophaga sp., Staphylococcus sp., Streptomyces sp. | Acremonium sp., Alternaria sp., Aspergillus sp., Aureobasidium sp., Candida sp., Cephalothecium sp., Chaetomium sp., Chrysosporium sp., Cladosporium sp., Epidermophyton sp., Fusarium sp., Microsporum sp., Mucor sp., Myrothecium sp., Neurospora sp., Penicillium sp., Rhizopus sp., Scopulariopsis sp., Stachybotrys sp., Stemphylium sp., Talaromyces sp., Tolypocladium sp., Trichoderma sp., Trichophyton sp., Uloclodium sp., Verticillium sp. | [29,81,82,83,91] |
| Wood, paper, parchment | Bacillus sp., Cellulomonas sp., Cellvibrio sp., Cellfacicula sp., Clostridium sp., Cytophaga sp., Marinobacter sp., Micromonospora sp., Xanthomonas sp., Pseudomonas sp., Sphingomonas sp., Staphylococcus sp., Virgibacillus sp. | Acremonium sp., Alternaria sp., Antrodia sp., Aspergillus sp., Aureobasidium sp., Chaetomium sp., Chrysosporium sp., Cladosporium sp., Coniophora sp., Coriolellus sp., Donkioporia sp., Epicoccum sp., Fibuloporia sp., Fusarium sp., Gloeophyllum sp., Gymnoascus sp., Mucor sp., Paecilomyces sp., Penicillium sp., Rhizopus sp., Serpula sp., Trichoderma sp., Verticillium sp. | [81,82,92,93] |
| Stone and wall paintings | Alcaligenes sp., Arthrobacter sp., Bacillus sp., Blastococcus sp., Blastocatella sp., Bryobacter sp., Chroococcidiopsis sp., Eucapsis sp., Flavobacterium sp., Kocuria sp., Leptolyngbya sp., Micrococcus sp., Modestobacter sp., Mycobacterium sp., Nitrobacter sp., Nocardia sp., Paenibacillus sp., Pseudomonas sp., Pseudonocardia sp. Rubrobacter sp., Sarcina sp., Scytonema sp., Sphingomonas sp., Staphylococcus sp., Streptomyces sp., Thiobacillus sp. | Alternaria sp., Aspergillus sp., Capnobotryella sp., Cladosporium sp., Coniosporium sp., Exophiala sp. Hortea sp., Knufia sp., Mucor sp., Trimmatostroma sp., Sarcinomyces sp., Penicillium sp., Rhizopus sp., Serpula sp., Trichoderma sp. | [80,81,93,94,95,96,97] |
| Metal | Acinetobacter sp., Chryseobacterium sp., Desulfomicrobium sp, Desulfosarcina sp., Desulfovibrio sp., Methanococcus sp., Shewanella sp., Sphingomonas sp., Stenotrophomonas sp., Thiobacillus sp, Vibrio sp. | Alternaria sp., Antrodia sp., Arthrinium sp., Aspergillus sp., Candida sp., Cladosporium sp., Clonostachys sp., Cryptococcus sp., Chrysosporium sp., Debaryomyces sp., Exophiala sp., Fusarium sp., Paecilomyces sp., Penicillium sp., Pichia sp., Poria sp., Rhodotorula sp. | [98,99] |
| Photographs | Burkholderia sp., Delftia sp., Enhydrobacter sp., Mesorhizobium sp., Neisseria sp., Olsenella sp., Paenibacillus sp., Pseudomonas sp., Saccharopolyspora sp. | Alternaria sp., Aspergillus sp., Chaetomium sp., Fusarium sp., Penicillium sp., Talaromyces sp., Trichoderma sp. | [56,81] |
| Material | Bacteria | References |
|---|---|---|
| Stone and wall paintings | The indigenous community of carbonatogenic bacteria, Acinetobacter sp., Bacillus sp., Brevibacterium sp., Cupriavidus metalidurans, Cellulosimicrobium cellulans, Desulfovibrio desulfuricans, Desulfovibrio vulgaris, Halomonas campaniensis, Micrococcus sp., Myxococcus xanthus, Pantonea sp., Pseudomonas aeruginosa, Pseudomonas chlororaphis, Pseudomonas koreensis, Pseudomonas stutzeri, Sporosarcina pasteurii, Stenotrophomonas maltophilia | [116,117,118,119,120,121,122,123,124,125] |
| Metal | Aeromonas sp., Desulfitobacterium hafniense, Geobacter sulfurreducens, Shewanella loihica, Sporosarcina pasteurii | [52,126,127,128] |
| Paper/glue | Bacillus flexus, Exiguobacterium undae, Ochrobactrum sp., Paracoccus sp. | [129,130] |
| Textile | Bacillus sp. | [115] |
| Wood | Thiobacillus denitrificans | [131] |
| Graffiti | Aerobacter aerogenes, Comamonas sp., Pseudomonas stutzeri | [132] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pyzik, A.; Ciuchcinski, K.; Dziurzynski, M.; Dziewit, L. The Bad and the Good—Microorganisms in Cultural Heritage Environments—An Update on Biodeterioration and Biotreatment Approaches. Materials 2021, 14, 177. https://doi.org/10.3390/ma14010177
Pyzik A, Ciuchcinski K, Dziurzynski M, Dziewit L. The Bad and the Good—Microorganisms in Cultural Heritage Environments—An Update on Biodeterioration and Biotreatment Approaches. Materials. 2021; 14(1):177. https://doi.org/10.3390/ma14010177
Chicago/Turabian StylePyzik, Adam, Karol Ciuchcinski, Mikolaj Dziurzynski, and Lukasz Dziewit. 2021. "The Bad and the Good—Microorganisms in Cultural Heritage Environments—An Update on Biodeterioration and Biotreatment Approaches" Materials 14, no. 1: 177. https://doi.org/10.3390/ma14010177
APA StylePyzik, A., Ciuchcinski, K., Dziurzynski, M., & Dziewit, L. (2021). The Bad and the Good—Microorganisms in Cultural Heritage Environments—An Update on Biodeterioration and Biotreatment Approaches. Materials, 14(1), 177. https://doi.org/10.3390/ma14010177





