Pentavalent Vanadium Species as Potential Corrosion Inhibitors of Al2Cu Intermetallic Phase in the Sulfuric(VI) Acid Solutions
Abstract
1. Introduction
2. Materials and Methods
3. Results and Discussion
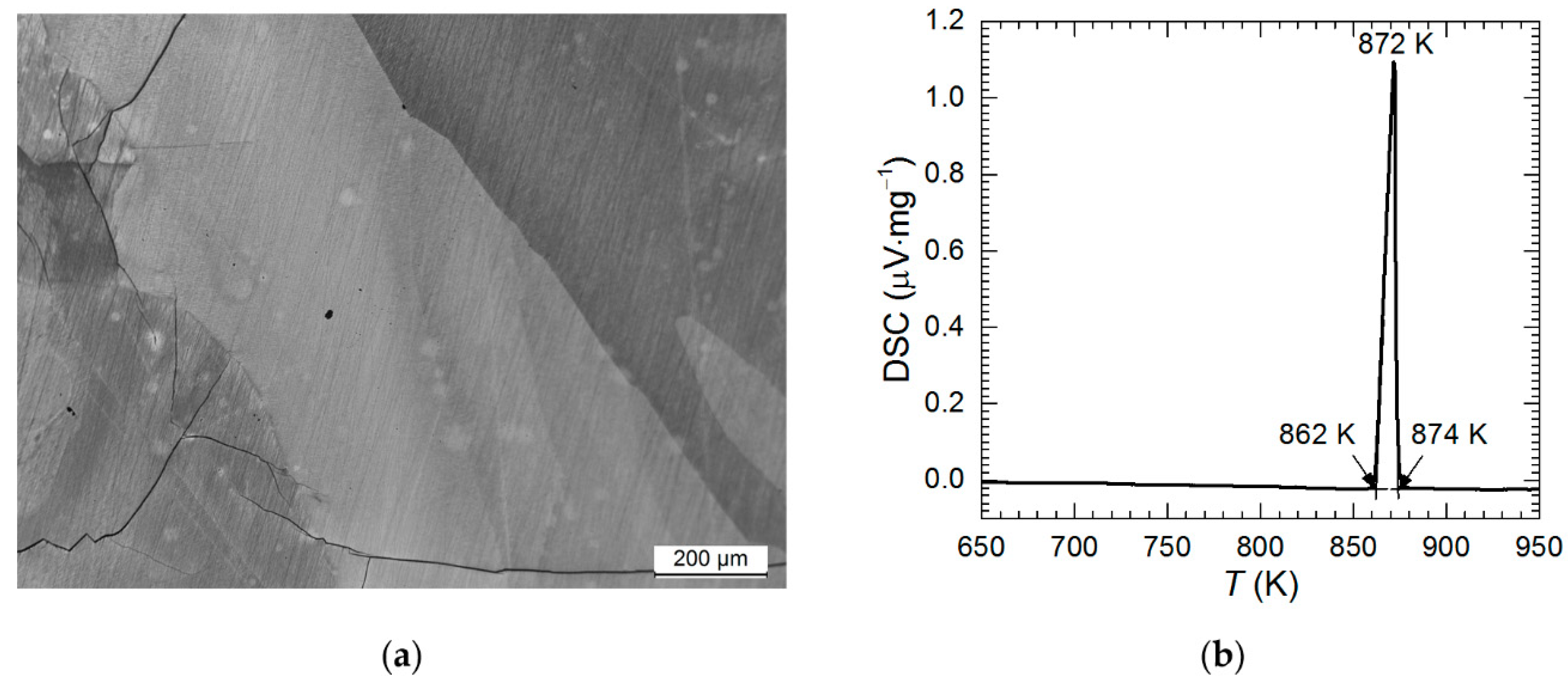
3.1. Characterization of Al2Cu Intermetallic Phase
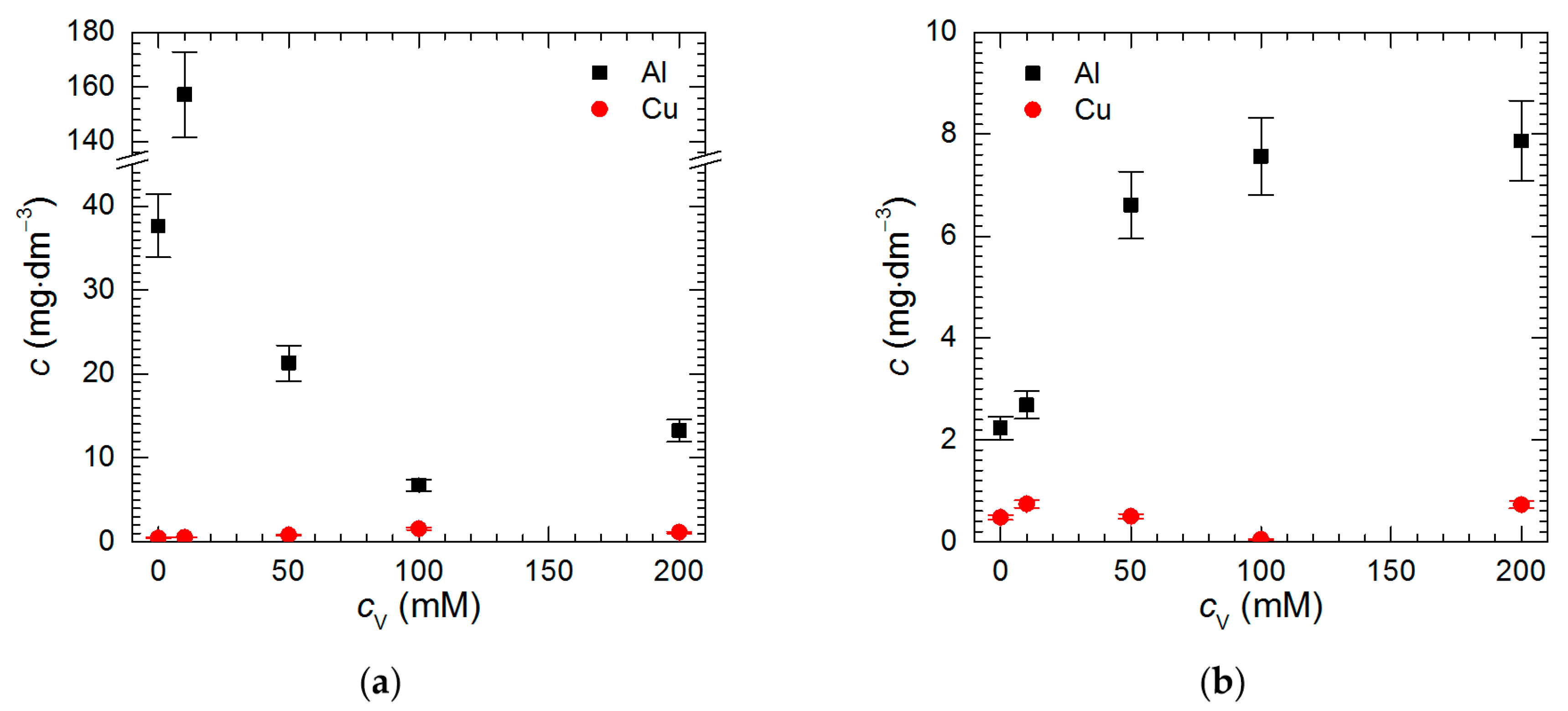
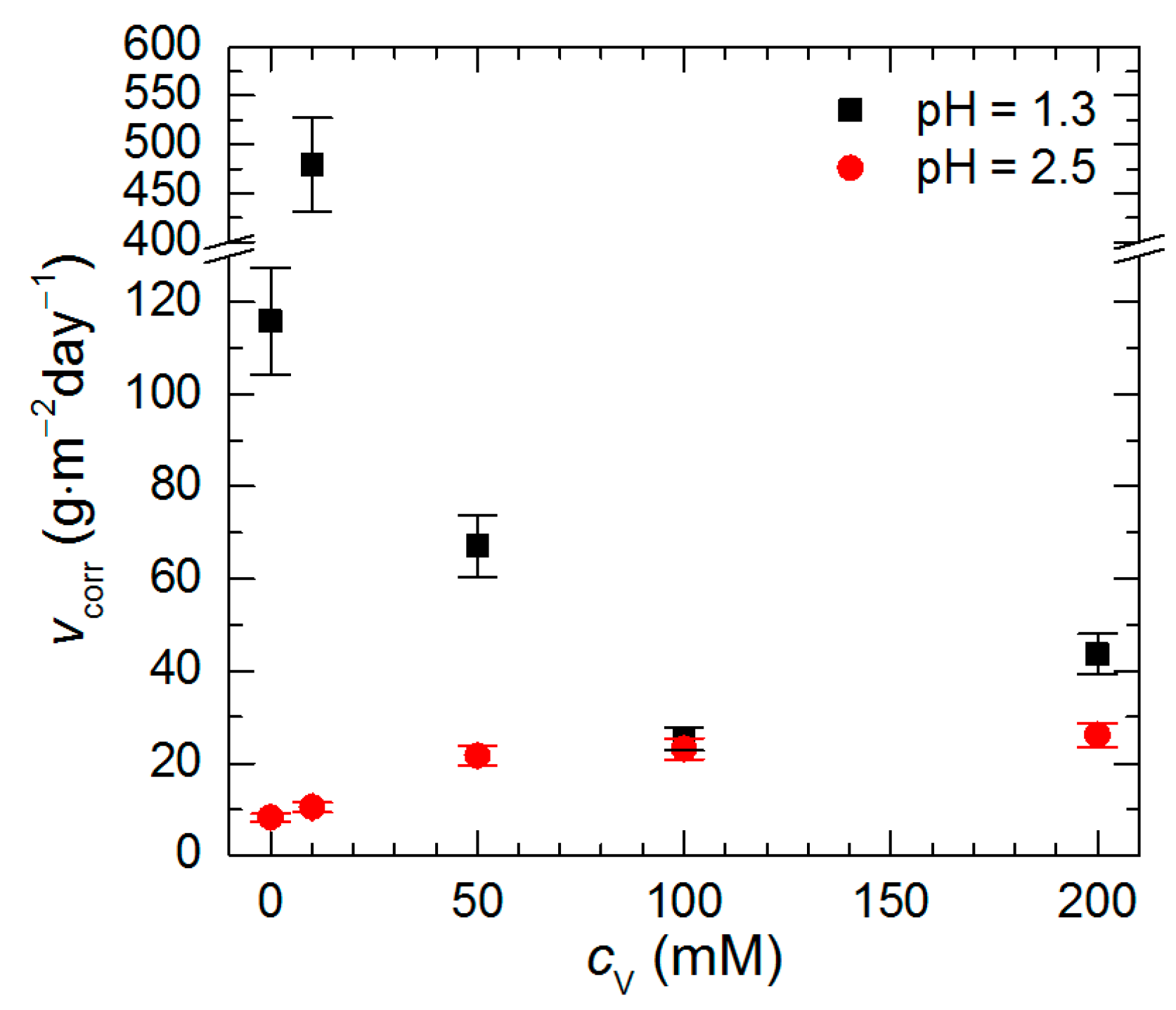
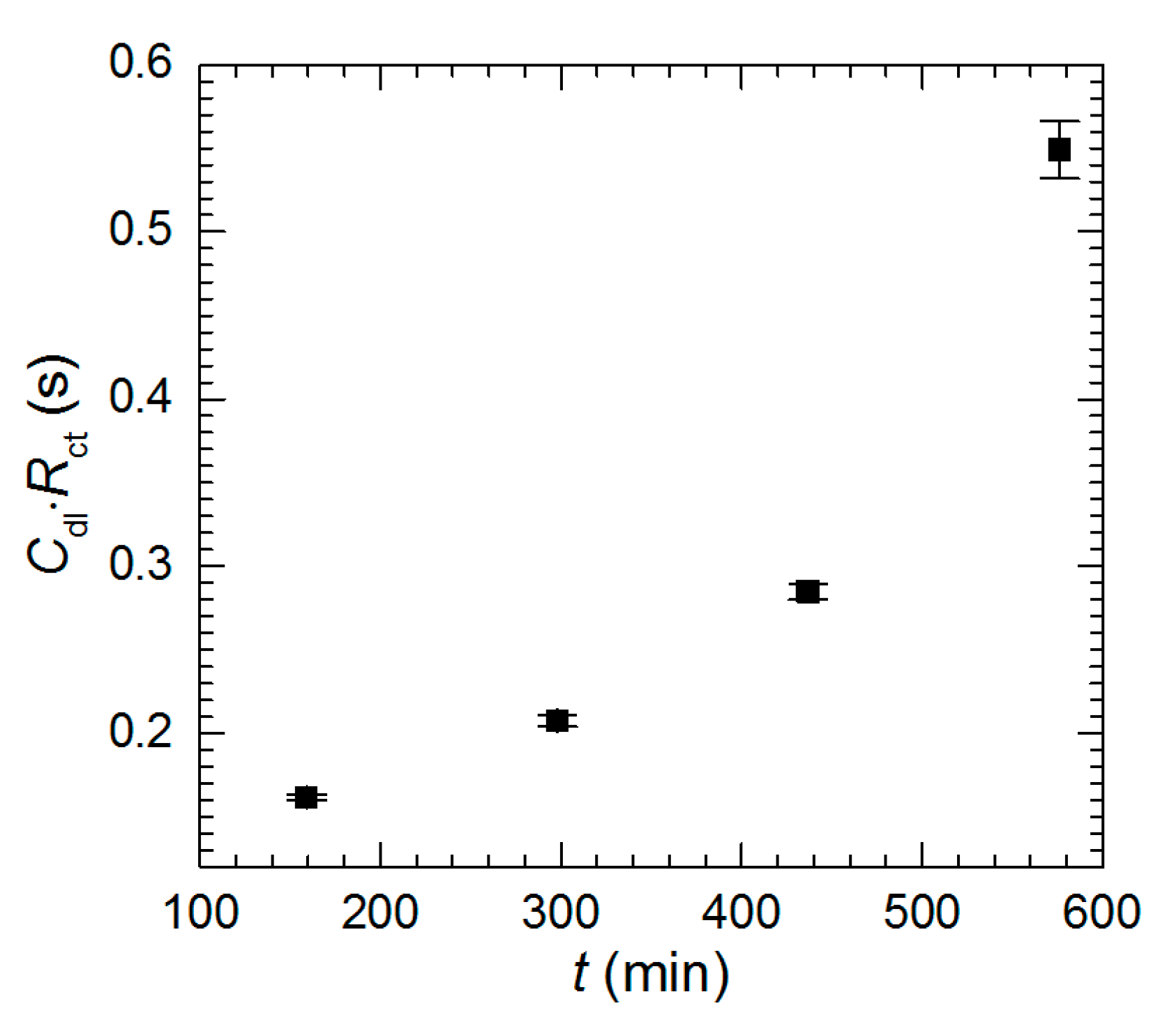
3.2. Corrosion Rate of Al2Cu Intermetallic Phase
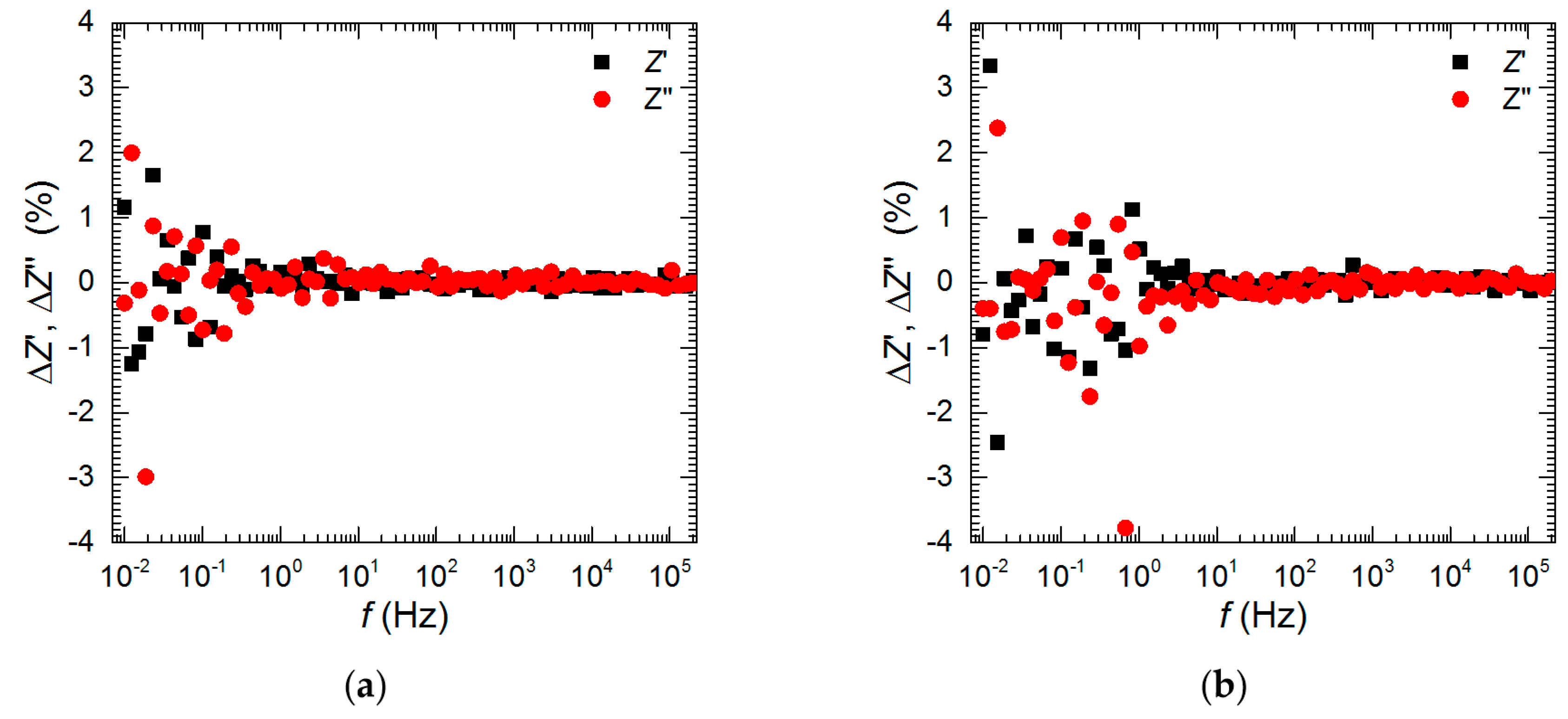
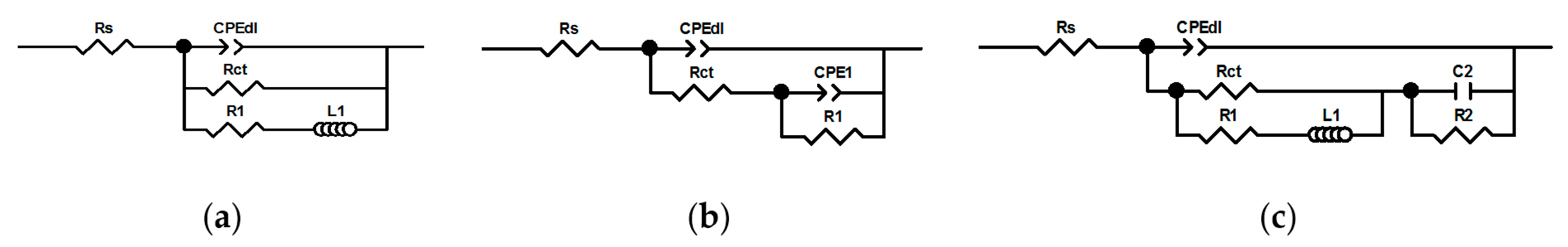
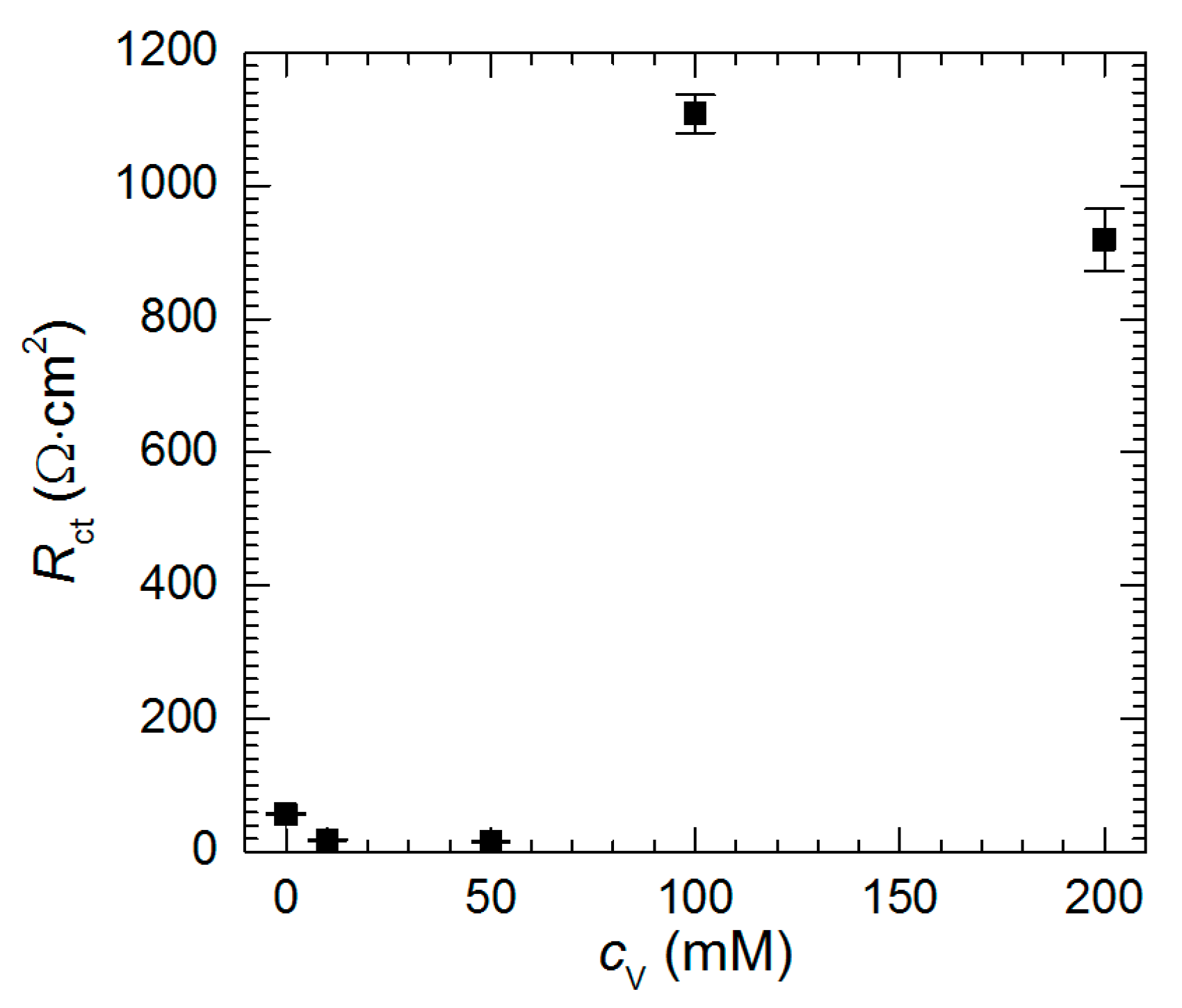
3.3. Electrochemical Analysis—Influence of Vanadium Species
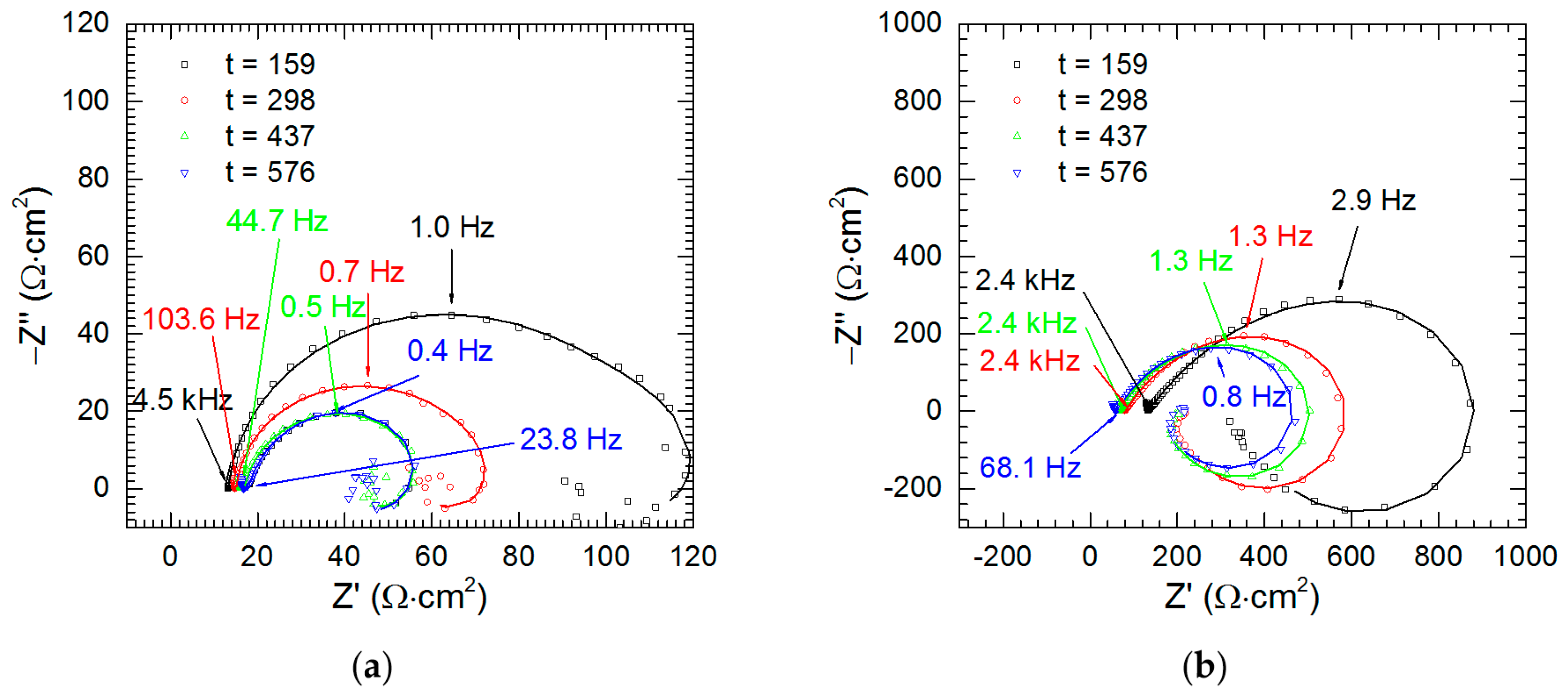
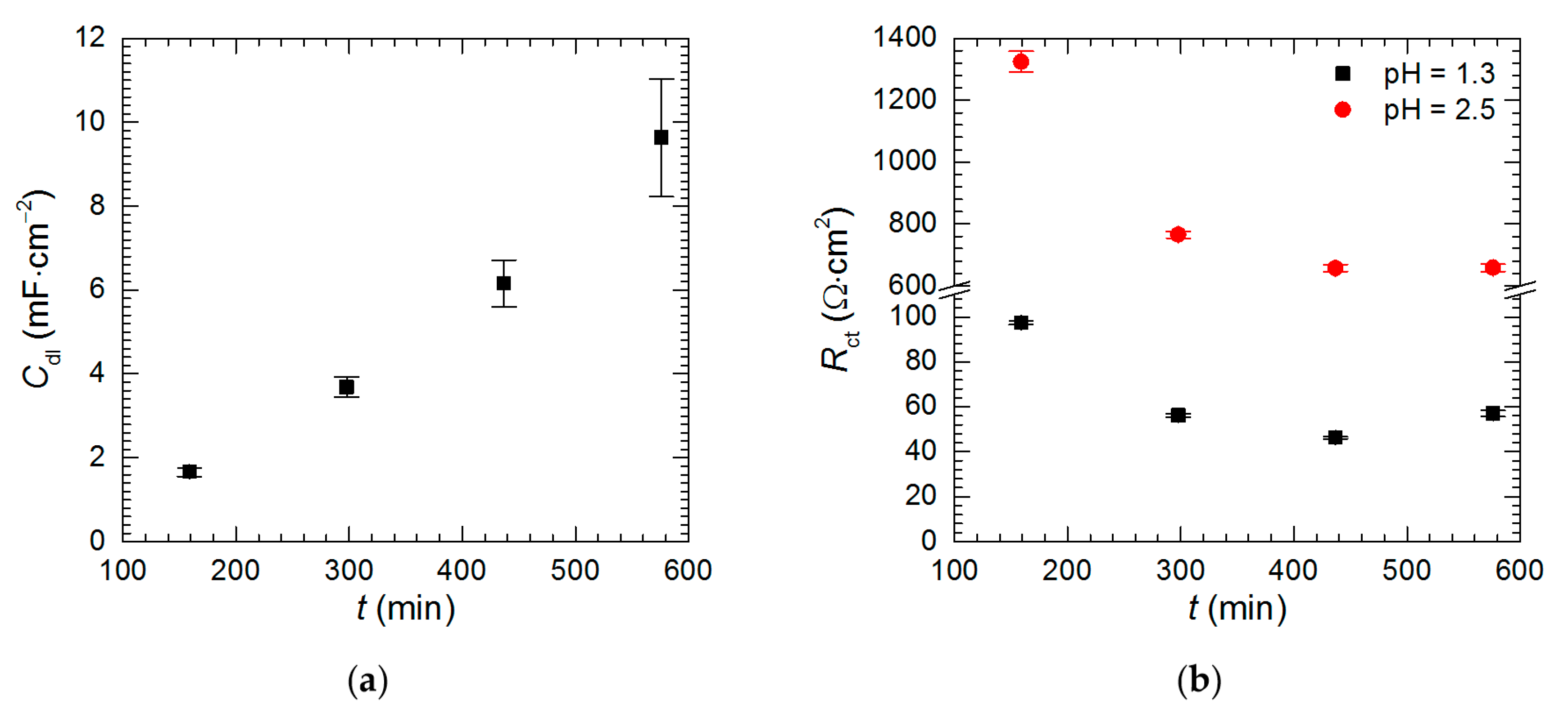
3.4. Electrochemical Analysis—The Kinetics of Selective Corrosion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Belov, N.A.; Eskin, D.G.; Aksenov, A.A. Multicomponent Phase Diagrams: Applications for Commercial Aluminum Alloys; Elsevier: Oxford, UK, 2005; pp. 152–192. [Google Scholar]
- Scully, J.R.; Knight, T.O.; Buchheit, R.G.; Peebles, D.E. Electrochemical characteristics of the Al2Cu, Al3Ta and Al3Zr intermetallic phases and their relevancy to the localized corrosion of Al alloys. Corros. Sci. 1993, 35, 185–195. [Google Scholar] [CrossRef]
- Buchheit, R.G. A compilation of corrosion potentials reported for intermetallic phases in aluminum alloys. J. Electrochem. Soc. 1995, 142, 3994–3996. [Google Scholar] [CrossRef]
- Buchheit, R.G.; Martinez, M.A.; Montes, L.P. Evidence for Cu Ion Formation by Dissolution and Dealloying the Al2CuMg Intermetallic Compound in Rotating Ring-Disk Collection Experiments. J. Electrochem. Soc. 2000, 147, 119–124. [Google Scholar] [CrossRef]
- Vukmirovic, M.B.; Dimitrov, N.; Sieradzki, K. Dealloying and Corrosion of Al Alloy 2024-T3. J. Electrochem. Soc. 2002, 149, B428. [Google Scholar] [CrossRef]
- Zhang, X.; Hashimoto, T.; Lindsay, J.; Zhou, X. Investigation of the de-alloying behaviour of θ-phase (Al2Cu) in AA2024-T351 aluminium alloy. Corros. Sci. 2016, 108, 85–93. [Google Scholar] [CrossRef]
- Osório, W.R.; Spinelli, J.E.; Ferreira, I.L.; Garcia, A. The roles of macrosegregation and of dendritic array spacings on the electrochemical behavior of an Al–4.5 wt% Cu alloy. Electrochim. Acta 2007, 52, 3265–3273. [Google Scholar] [CrossRef]
- Osório, W.R.; Spinelli, J.E.; Freire, C.M.A.; Cardona, M.B.; Garcia, A. The roles of Al2Cu and of dendritic refinement on surface corrosion resistance of hypoeutectic Al-Cu alloys immersed in H2SO4. J. Alloys Compd. 2007, 443, 87–93. [Google Scholar] [CrossRef]
- Osório, W.R.; Siqueira, C.A.; Santos, C.A.; Garcia, A. The Correlation between Electrochemical Corrosion Resistance and Mechanical Strength of As-Cast Al-Cu and Al-Si Alloys. Int. J. Electrochem. Sci. 2011, 6, 6275–6289. [Google Scholar]
- Obispo, H.M.; Murr, L.E.; Arrowood, R.M.; Trillo, E.A. Copper deposition during the corrosion of aluminum alloy 2024 in sodium chloride solutions. J. Mater. Sci. 2001, 36, 3479–3495. [Google Scholar]
- Lebouil, S.; Tardelli, J.; Rocca, E.; Volovitch, P.; Ogle, K. Dealloying of Al2Cu, Al7Cu2Fe, and Al2CuMg intermetallic phases to form nanoparticulate copper films. Mater. Corros. 2014, 65, 416–424. [Google Scholar] [CrossRef]
- Kwolek, P.; Dychtoń, K.; Pytel, M. Orthophosphoric acid solutions of sodium orthovanadate, sodium tungstate, and sodium molybdate as potential corrosion inhibitors of the Al2Cu intermetallic phase. J. Solid State Electrochem. 2019, 23, 3019–3029. [Google Scholar] [CrossRef]
- ASTM B137-95. Standard Test Method for Measurement of Coating Mass Per Unit Area on Anodically Coated Aluminum; ASTM International: West Conshohocken, PA, USA, 2009. [Google Scholar]
- Katz, S.A.; Salem, H. The toxicology of chromium with respect to its chemical speciation: A review. J. Appl. Toxicol. 1993, 13, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Deng, S.; Fu, H. Sodium molybdate as a corrosion inhibitor for aluminium in H3PO4 solution. Corros. Sci. 2011, 53, 2748–2753. [Google Scholar] [CrossRef]
- Kwolek, P.; Kamiński, A.; Dychtoń, K.; Drajewicz, M.; Sieniawski, J. The corrosion rate of aluminium in the orthophosphoric acid solutions in the presence of sodium molybdate. Corros. Sci. 2016, 106, 208–216. [Google Scholar] [CrossRef]
- Dychtoń, K.; Kwolek, P. The replacement of chromate by molybdate in phosphoric acid-based etch solutions for aluminium alloys. Corros. Eng. Sci. Technol. 2018, 53, 234–240. [Google Scholar] [CrossRef]
- Kwolek, P.; Pustuła, A.; Nowak, W.J. Influence of molybdophosphoric acid on the kinetics of the anodic coating dissolution. Surf. Coat. Technol. 2019, 357, 535–542. [Google Scholar] [CrossRef]
- Pope, M.T. Heteropoly and Isopoly Oxometalates; Springer: Berlin, Germany, 1983; pp. 1–3, 34–40. [Google Scholar]
- Kwolek, P. Effect of Na3VO4 inhibitor on the corrosion resistance of Al2Cu intermetallic phase in H3PO4 aqueous solution. Arch. Metall. Mater. 2020, 65, 175–183. [Google Scholar]
- Ralston, K.D.; Chrisanti, S.; Young, T.L.; Buchheit, R.G. Corrosion Inhibition of Aluminum Alloy 2024-T3 by Aqueous Vanadium Species. J. Electrochem. Soc. 2008, 155, C350–C359. [Google Scholar] [CrossRef]
- Ralston, K.D.; Young, T.L.; Buchheit, R.G. Electrochemical Evaluation of Constituent Intermetallics in Aluminum Alloy 2024-T3 Exposed to Aqueous Vanadate Inhibitors. J. Electrochem. Soc. 2009, 156, C135–C146. [Google Scholar] [CrossRef]
- Iannuzzi, M.; Frankel, G.S. Mechanisms of corrosion inhibition of AA2024-T3 by vanadates. Corros. Sci. 2007, 49, 2371–2391. [Google Scholar] [CrossRef]
- Boukamp, B. A linear Kronig-Kramers transform test for immittance data validation. J. Electrochem. Soc. 1995, 142, 1885–1894. [Google Scholar] [CrossRef]
- Boukamp, B. Electrochemical impedance spectroscopy in solid state ionics: Recent advances. Solid State Ionics 2004, 169, 65–73. [Google Scholar] [CrossRef]
- Mansfeld, F. The Polarization Resistance Technique for Measuring Corrosion Currents. In Advances in Corrosion Science and Technology; Fontana, M.G., Staehle, R., Eds.; Plenum Press: Boston, MA, USA, 1970; pp. 163–262. [Google Scholar]
- Kwolek, P.; Gradzik, A.; Szeliga, D.; Kościelniak, B. Selective corrosion of Al2Cu intermetallic phase in orthophosphoric acid aqueous solutions. Arch. Metall. Mater. 2019, 64, 1223–1229. [Google Scholar]
- Zobac, O.; Kroupa, A.; Zemanova, A.; Richter, K.W. Experimental Description of the Al-Cu Binary Phase Diagram. Metall. Mater. Trans. A Phys. Metall. Mater. Sci. 2019, 50, 3805–3815. [Google Scholar] [CrossRef]
- Huang, Y.; Zhang, Y. Influence of the electrochemical properties of vanadium oxides on specific capacitance by molybdenum doping. Bull. Mater. Sci. 2019, 42, 37. [Google Scholar] [CrossRef]
- Zoski, C.G. Handbook of Electrochemistry; Elsevier: Amsterdam, The Netherlands, 2007; pp. 815–817. [Google Scholar]
- De Levie, R. Electrochemical response of porous and rough electrodes. In Advances in Electrochemistry and Electrochemical Engineering; Delahay, P., Ed.; Interscience: New York, NY, USA, 1967; pp. 329–397. [Google Scholar]
- Lasia, A. Electrochemical Impedance Spectroscopy and Its Applications; Springer: New York, NY, USA, 2014; pp. 177–196, 210–214. [Google Scholar]
- Chen, L.; Lasia, A. Ni-Al Powder Electrocatalyst for Hydrogen Evolution. J. Electrochem. Soc. 1993, 140, 2464–2473. [Google Scholar] [CrossRef]
- Chen, L.; Lasia, A. Study of the kinetics of hydrogen evolution reaction on Nickel-Zinc powder electrodes. J. Electrochem. Soc. 1992, 139, 3214–3219. [Google Scholar] [CrossRef]
- Epelboin, I.; Gabrielli, C.; Keddam, M.; Takenouti, H. The Study of the Passivation Process by the Electrode Impedance Analysis. In Electrochemical Materials Science. Comprehensive Treatise of Electrochemistry; Bockris, J.O., Conway, B.E., Yeager, E., White, R.E., Eds.; Springer: Boston, MA, USA, 1981; pp. 151–192. [Google Scholar]
- Péter, L.; Arai, J.; Akahoshi, H. Impedance of a reaction involving two adsorbed intermediates: Aluminum dissolution in non-aqueous lithium imide solutions. J. Electroanal. Chem. 2000, 482, 125–138. [Google Scholar] [CrossRef]
- Macdonald, D.D.; Lee, K.H.; Moccari, A.; Harrington, D. Evaluation of Alloy Anodes for Aluminum-Air Batteries: Corrosion Studies. Corrosion 1988, 44, 652–657. [Google Scholar] [CrossRef]
- Cao, C. On the impedance plane displays for irreversible electrode reactions based on the stability conditions of the steady state II. Two state variables besides electrode potential. Electrochim. Acta 1990, 35, 837–844. [Google Scholar] [CrossRef]
- Łosiewicz, B.; Jurczakowski, R.; Lasia, A. Kinetics of hydrogen underpotential deposition at iridium in sulfuric and perchloric acids. Electrochim. Acta 2017, 225, 160–167. [Google Scholar] [CrossRef]

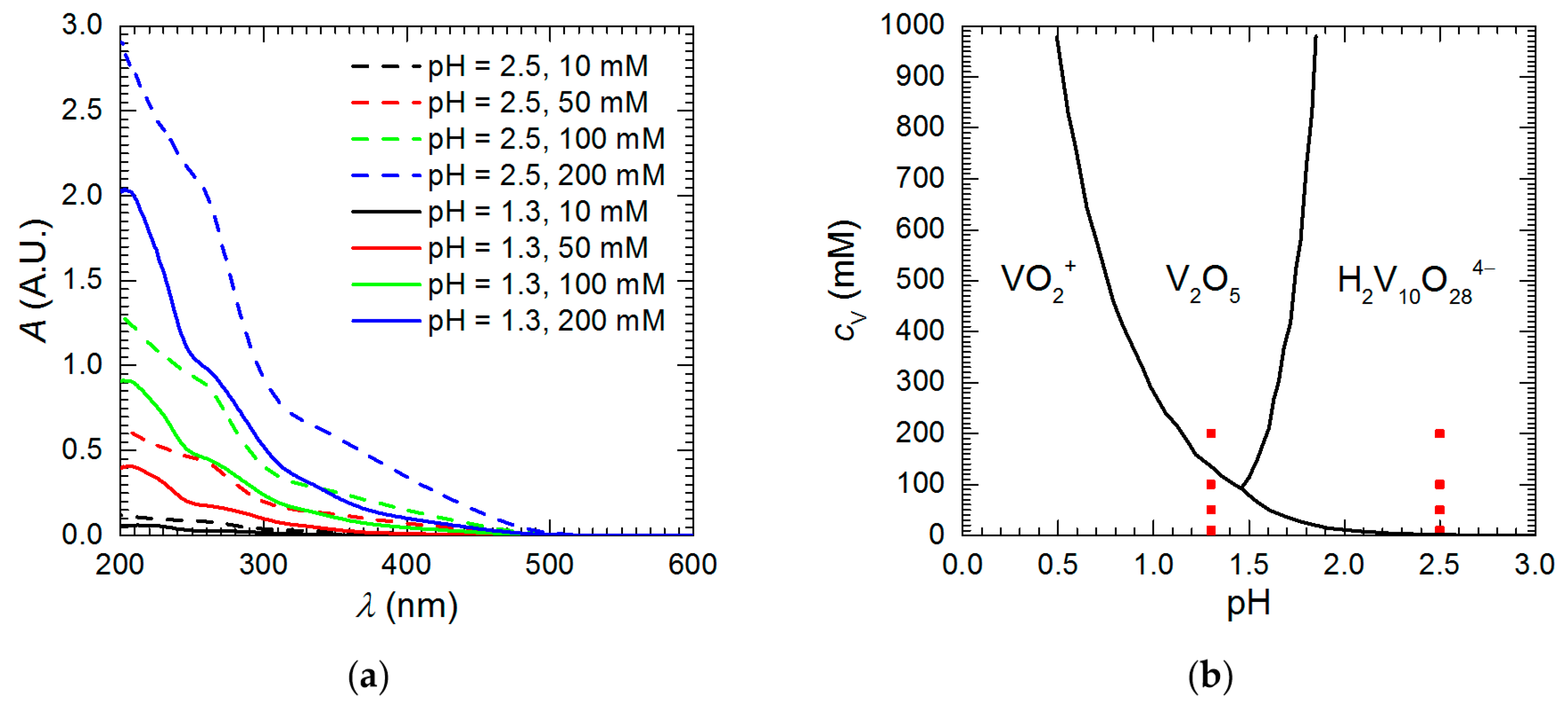

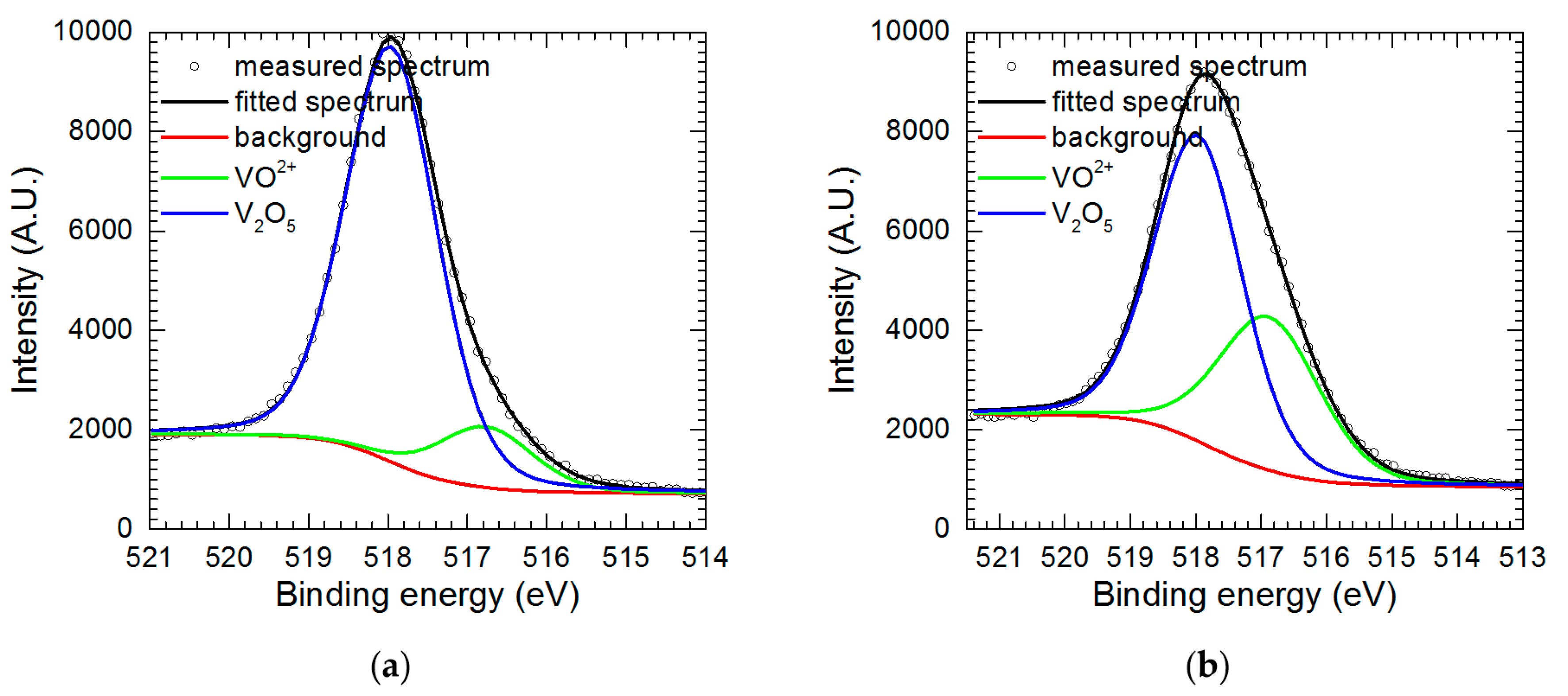
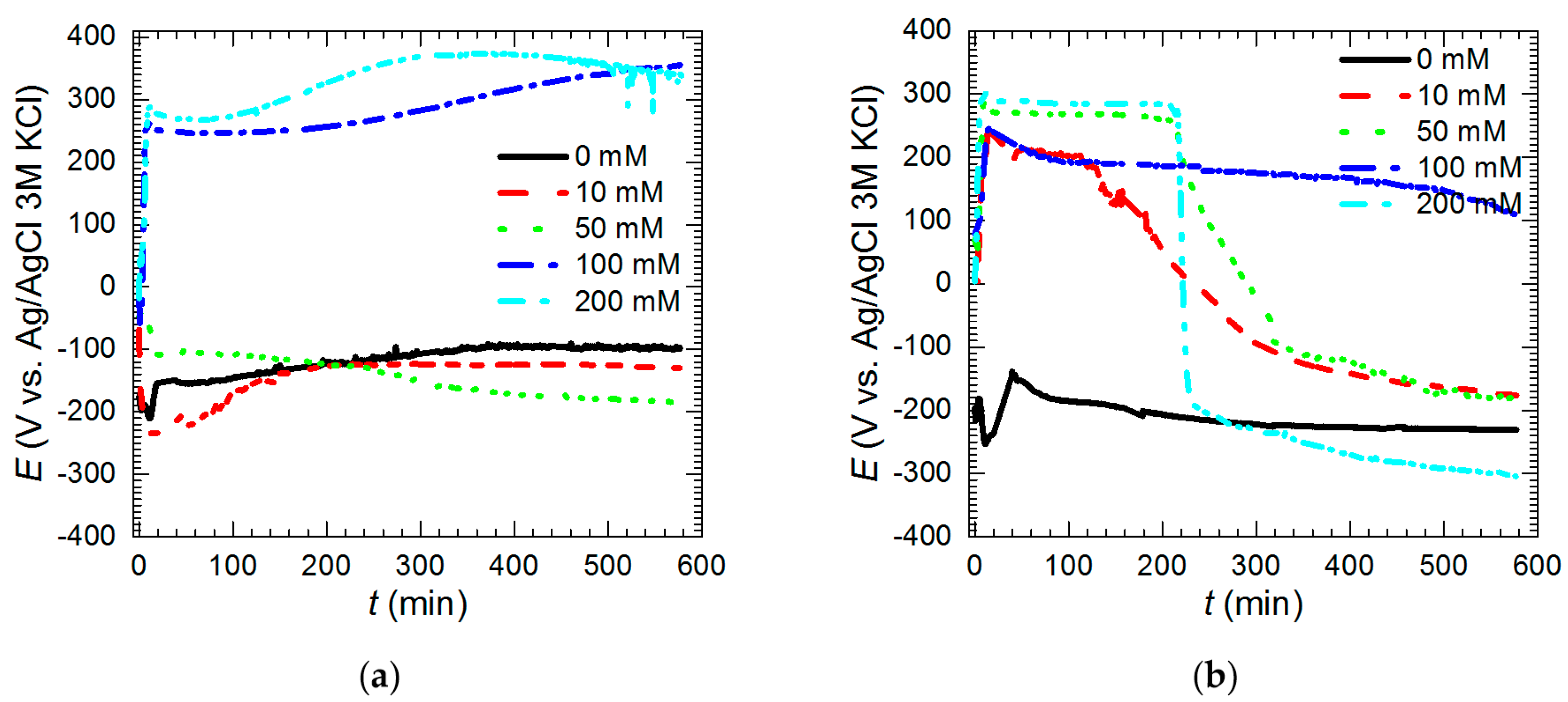
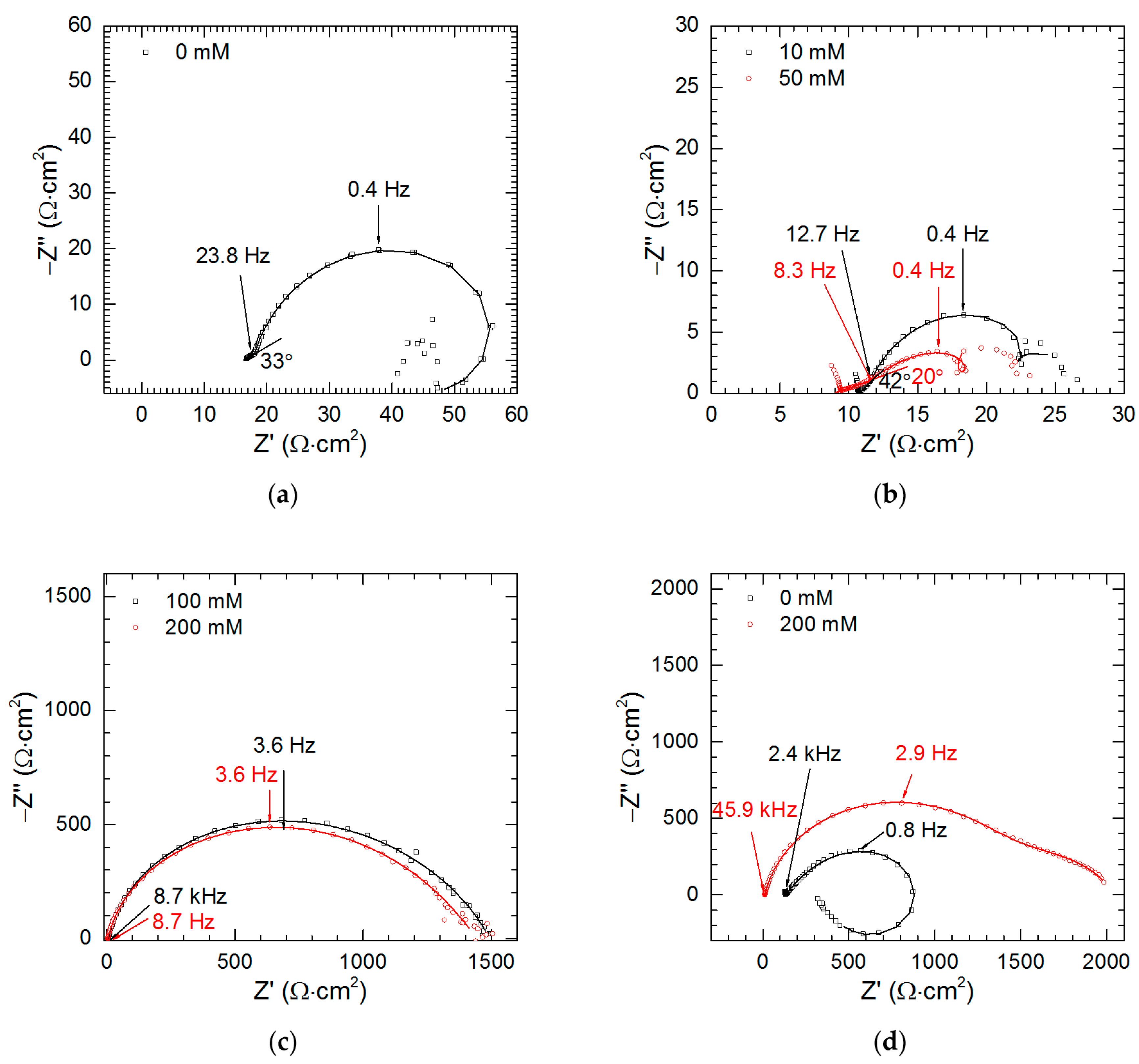
| Measurement | O 1s | V 2p | |
|---|---|---|---|
| O2− | V5+ | V4+ | |
| 1 | 69.8 | 26.3 | 3.9 |
| 2 | 68.5 | 26.3 | 5.2 |
| Average | 69.2 | 26.3 | 4.6 |
| Measurement | O 1s | V 2p | A l2s | Cu 2p | |
|---|---|---|---|---|---|
| O2− | V5+ | V4+ | Al3+ | Cu2+/Cu | |
| 1 | 54.8 | 16.0 | 9.1 | 19.0 | 1.2 |
| 2 | 61.9 | 15.7 | 6.9 | 15.3 | 0.3 |
| Average | 58.3 | 15.8 | 8.0 | 17.1 | 0.7 |
| Experimental Conditions | χ2 | S | Rs (Ω·cm2) | Tdl (mF·sα−1·cm−2) | αdl | Rct (Ω·cm2) | R1 (Ω·cm2) | L1 (H·cm2) | C2 (mF·cm−2) | R2 (Ω·cm2) |
|---|---|---|---|---|---|---|---|---|---|---|
| cV = 0 mM, t = 159 min | 1.05 × 10−4 | 0.11 | 13 (1) | 2.055 (0.022) | 0.95 (0.01) | 97 (1) | 0 | 211 (16) | 17.805 (1.368) | 91 (2) |
| cV = 0 mM, t = 298 min | 5.46 × 10−4 | 0.04 | 15 (1) | 4.201 (0.067) | 0.96 (0.01) | 56 (1) | 0 | 95 (6) | 28.958 (2.534) | 44 (1) |
| cV = 0 mM, t = 437 min | 2.23 × 10−4 | 0.01 | 17 (1) | 7.804 (0.083) | 0.91 (0.01) | 46 (1) | 61 (1) | 121 (4) | ||
| cV = 0 mM, t = 576 min | 1.59 × 10−4 | 0.01 | 18 (1) | 12.856 (0.168) | 0.86 (0.01) | 57 (1) | 36 (1) | 65 (2) | ||
| cV = 10 mM, t = 576 min | 3.76 × 10−4 | 0.02 | 11 (1) | 35.294 (1.202) | 0.85 (0.02) | 18 (1) | 0 | 17 (1) | 99.884 (6.842) | 16 (1) |
| cV = 50 mM, t = 576 min | 2.59 × 10−4 | 0.01 | 11 (1) | 66.191 (2.919) | 0.60 (0.03) | 15 (1) | 0 | 23 (1) | 201.722 (17.043) | 17 (1) |
| Experimental Conditions | χ2 | S | Rs (Ω·cm2) | Tdl (mF·sα−1·cm−2) | αdl | Rct (Ω·cm2) | R1 (Ω·cm2) | T1 (mF·sα−1·cm−2) | α1 | |
|---|---|---|---|---|---|---|---|---|---|---|
| pH = 1.3 | cV = 100 mM, t = 576 min | 2.68 × 10−4 | 0.03 | 7 (1) | 0.043 (0.001) | 0.88 (0.01) | 1108 (29) | 379 (33) | 0.660 (0.083) | 0.64 (0.04) |
| cV = 200 mM, t = 576 min | 2.42 × 10−4 | 0.03 | 5 (1) | 0.044 (0.001) | 0.88 (0.01) | 919 (47) | 521 (52) | 0.437 (0.066) | 0.54 (0.03) | |
| pH = 2.5 | cV = 200 mM, t = 159 min | 2.91 × 10−4 | 0.04 | 11 (1) | 0.048 (0.001) | 0.87 (0.01) | 1450 (11) | 581 (21) | 2.205 (0.117) | 0.68 (0.02) |
| Experimental Conditions | χ2 | S | Rs (Ω·cm2) | Tdl (mF·sα−1·cm−2) | αdl | Rct (Ω·cm2) | R1 (Ω·cm2) | L1 (H·cm2) |
|---|---|---|---|---|---|---|---|---|
| cV = 0 mM, t = 159 min | 7.13 × 10−4 | 0.07 | 133 (1) | 0.557 (0.009) | 0.64 (0.01) | 1325 (33) | 294 (6) | 772 (11) |
| cV = 0 mM, t = 298 min | 4.49 × 10−4 | 0.04 | 80 (1) | 0.587 (0.008) | 0.68 (0.01) | 766 (11) | 150 (3) | 398 (4) |
| cV = 0 mM, t = 437 min | 8.39 × 10−4 | 0.08 | 65 (1) | 0.722 (0.011) | 0.70 (0.01) | 657 (12) | 133 (2) | 368 (4) |
| cV = 0 mM, t = 576 min | 1.97 × 10−4 | 0.01 | 61 (1) | 0.966 (0.012) | 0.71 (0.01) | 658 (12) | 138 (2) | 356 (3) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kwolek, P.; Kościelniak, B.; Wytrwal-Sarna, M. Pentavalent Vanadium Species as Potential Corrosion Inhibitors of Al2Cu Intermetallic Phase in the Sulfuric(VI) Acid Solutions. Materials 2020, 13, 1946. https://doi.org/10.3390/ma13081946
Kwolek P, Kościelniak B, Wytrwal-Sarna M. Pentavalent Vanadium Species as Potential Corrosion Inhibitors of Al2Cu Intermetallic Phase in the Sulfuric(VI) Acid Solutions. Materials. 2020; 13(8):1946. https://doi.org/10.3390/ma13081946
Chicago/Turabian StyleKwolek, Przemysław, Barbara Kościelniak, and Magdalena Wytrwal-Sarna. 2020. "Pentavalent Vanadium Species as Potential Corrosion Inhibitors of Al2Cu Intermetallic Phase in the Sulfuric(VI) Acid Solutions" Materials 13, no. 8: 1946. https://doi.org/10.3390/ma13081946
APA StyleKwolek, P., Kościelniak, B., & Wytrwal-Sarna, M. (2020). Pentavalent Vanadium Species as Potential Corrosion Inhibitors of Al2Cu Intermetallic Phase in the Sulfuric(VI) Acid Solutions. Materials, 13(8), 1946. https://doi.org/10.3390/ma13081946





