A New Biomimetic Composite Structure with Tunable Stiffness and Superior Toughness via Designed Structure Breakage
Abstract
1. Introduction
2. Mechanical Model and Theoretical Analysis
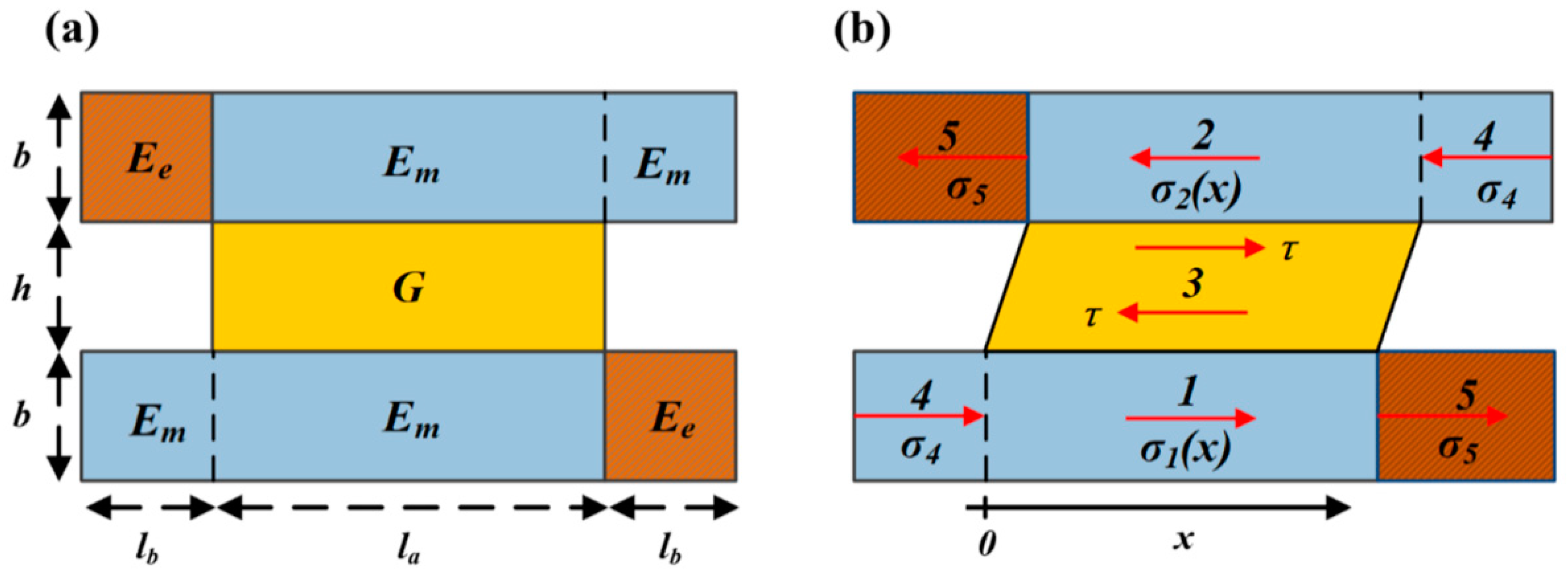
2.1. Mechanical Model of Unit Cell
2.1.1. Unit Cell at “Phase 0”
2.1.2. Unit Cell at “Phase 1”
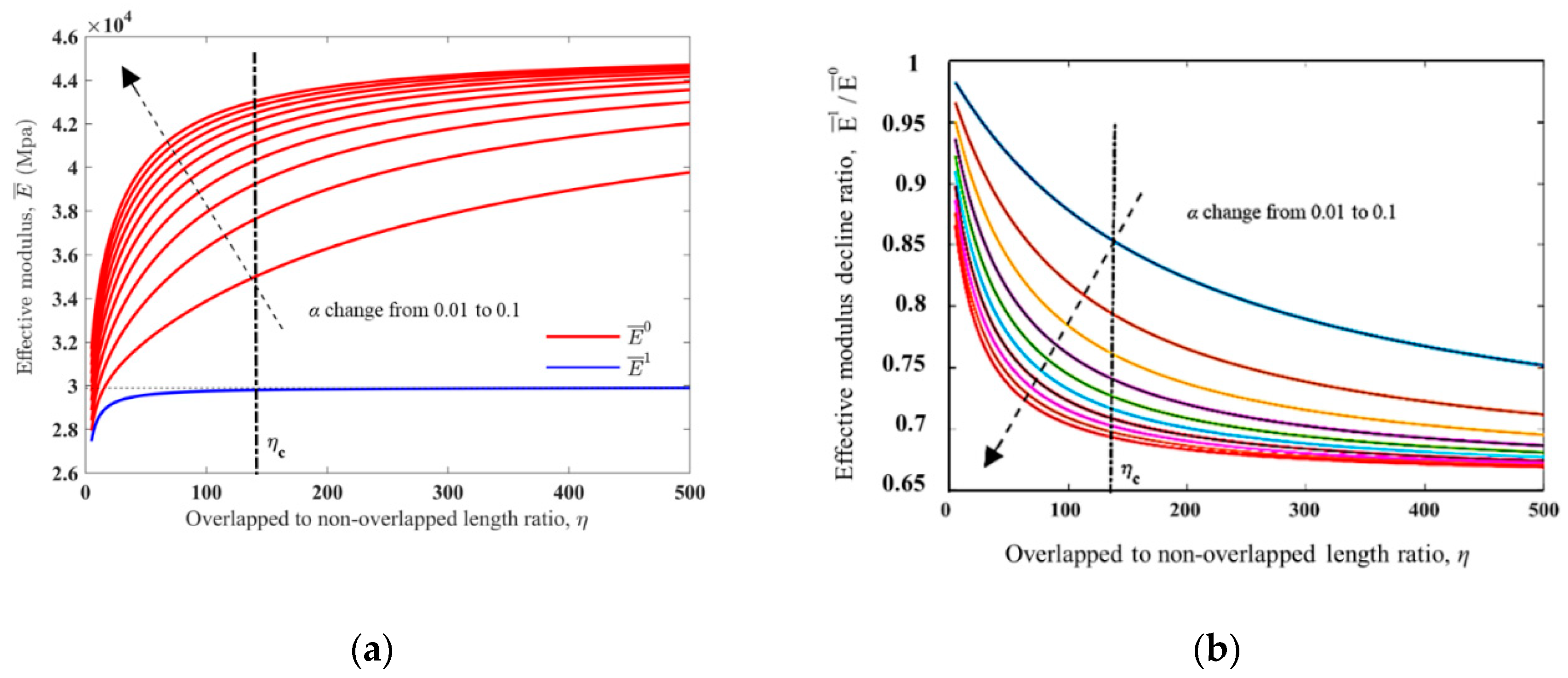
2.2. Unit Cell Elastic Modulus Analysis
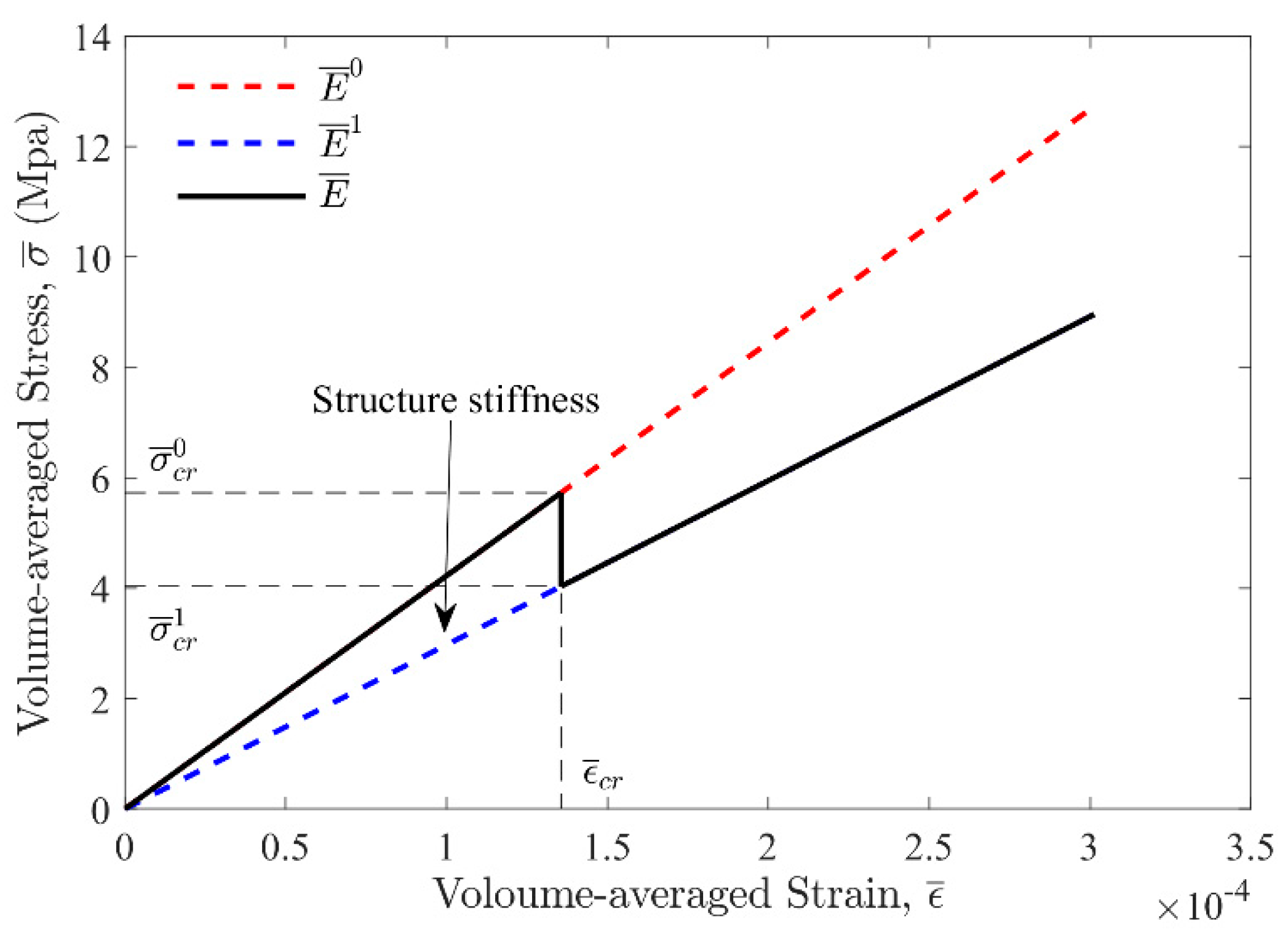
2.3. Unit Cell Phase Changing Process Analysis
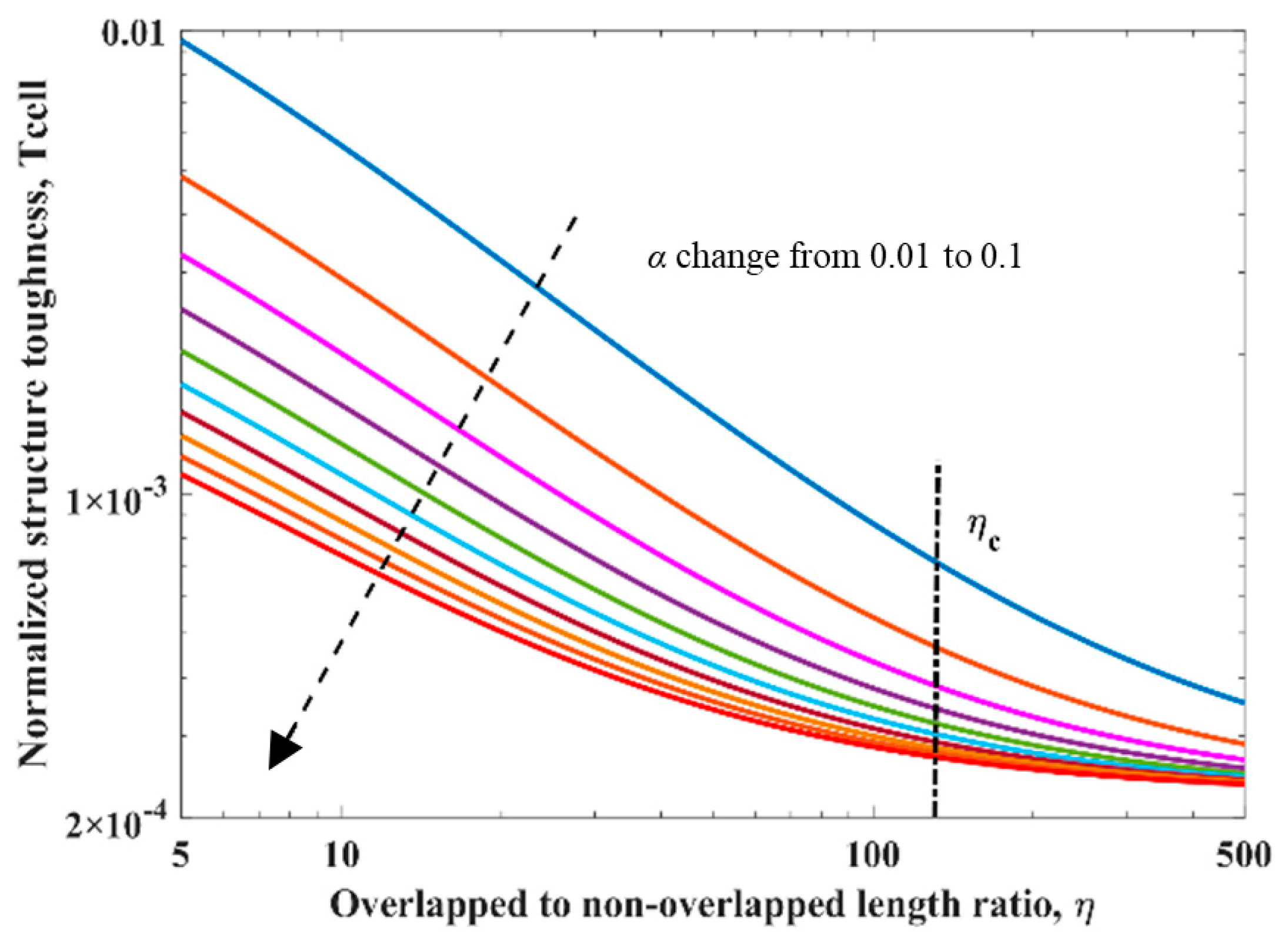
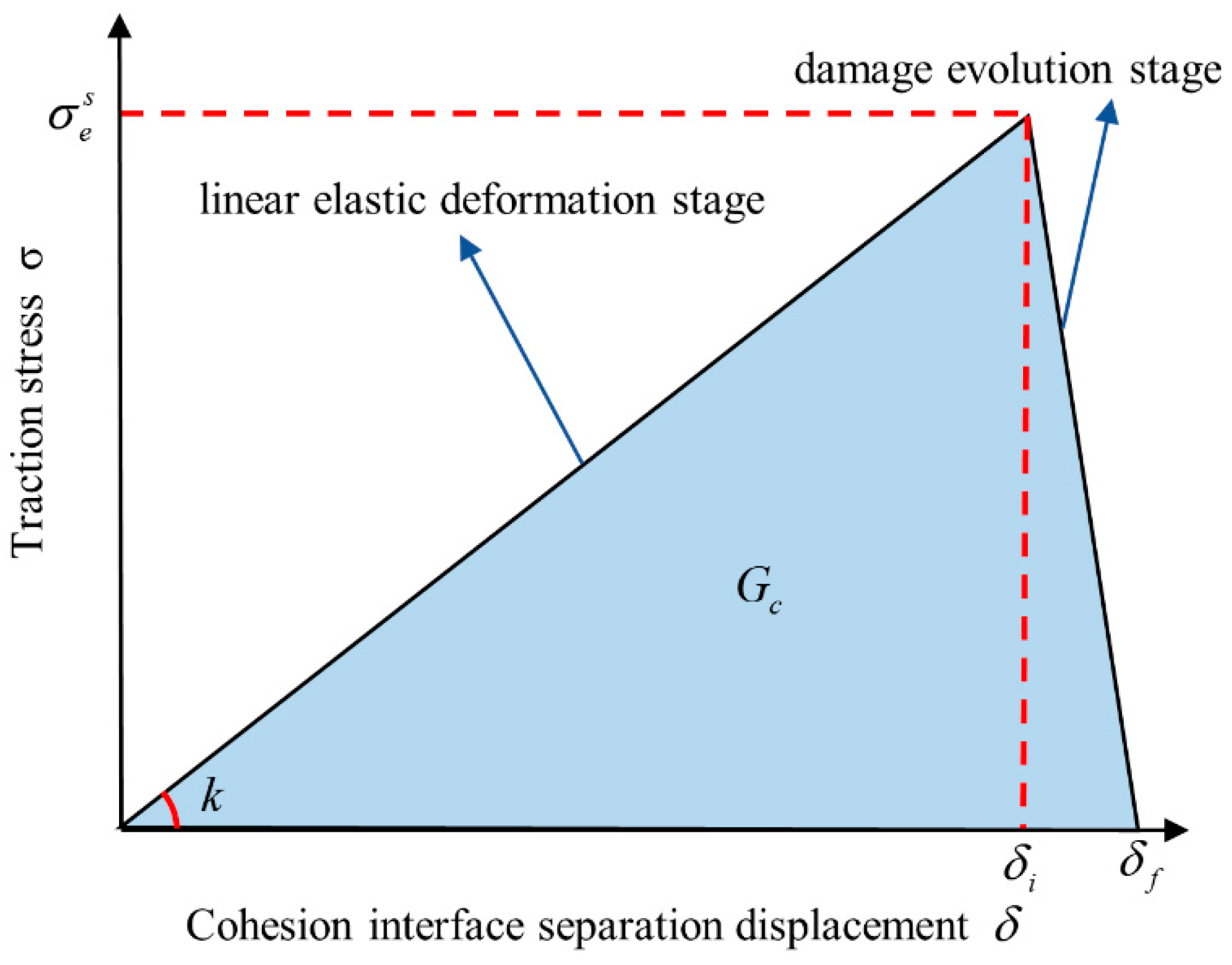
2.4. Structure Toughness Analysis
3. Finite Element Modeling and Simulation
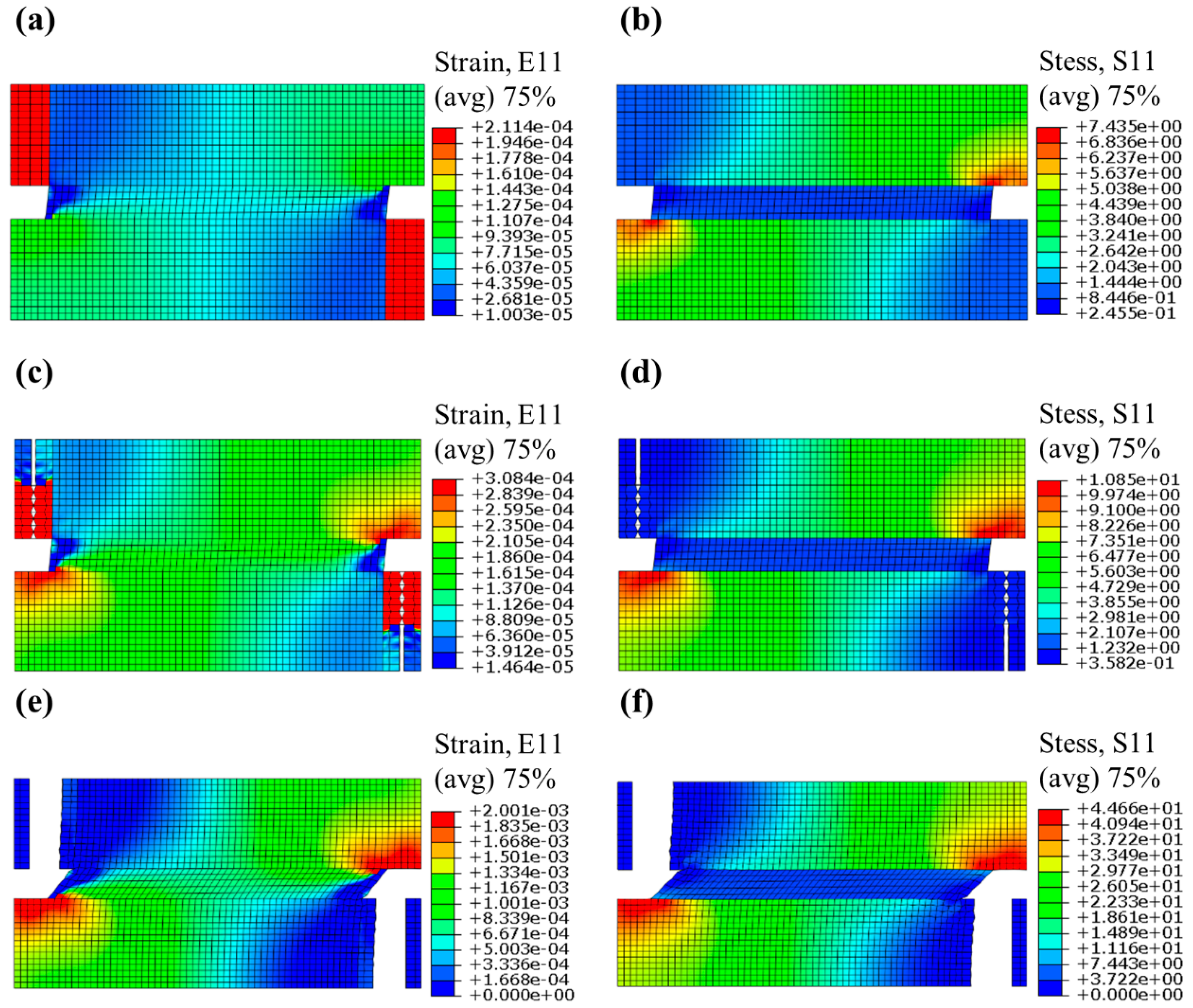
3.1. FEM Simulation Preparation
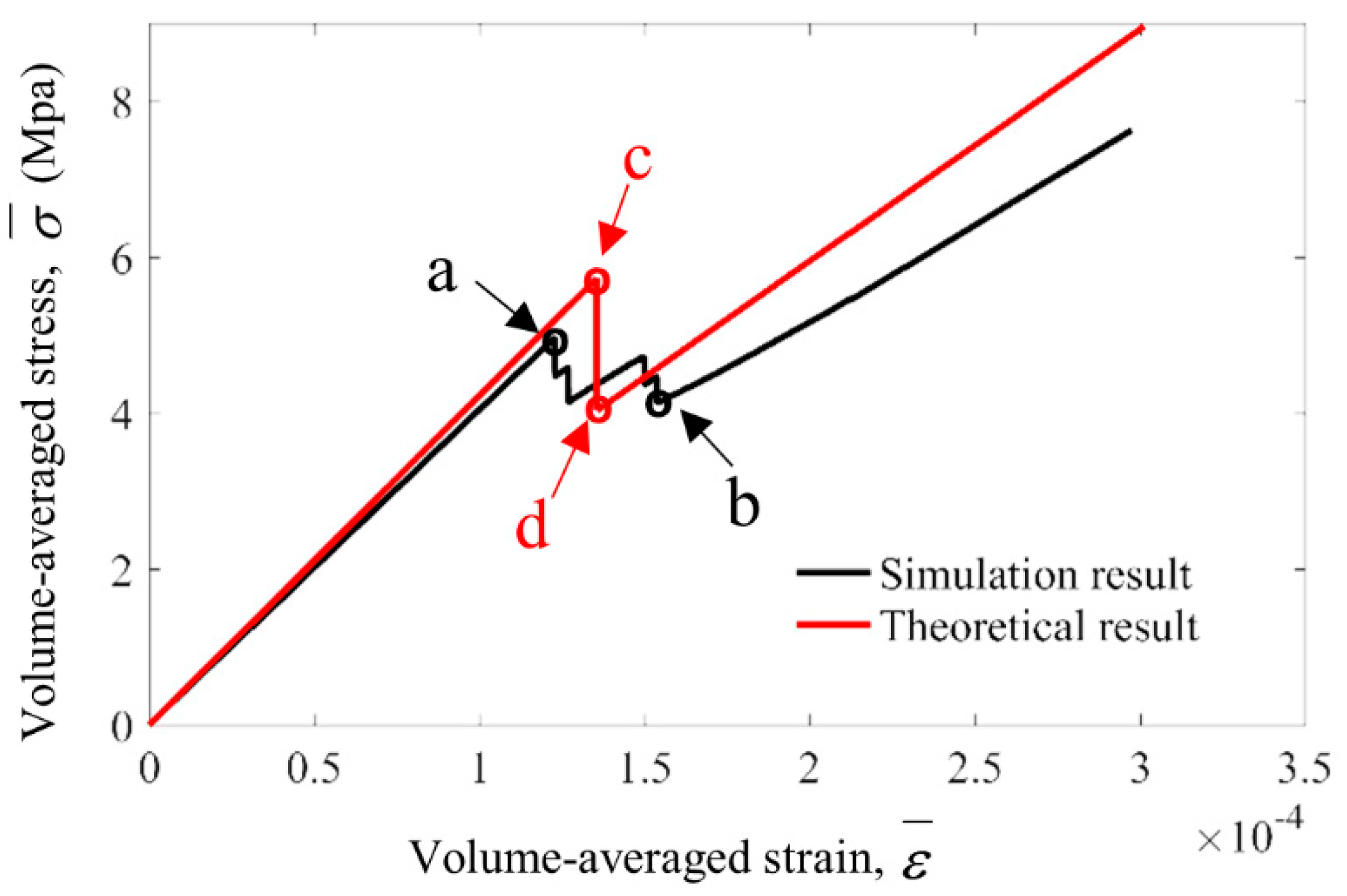
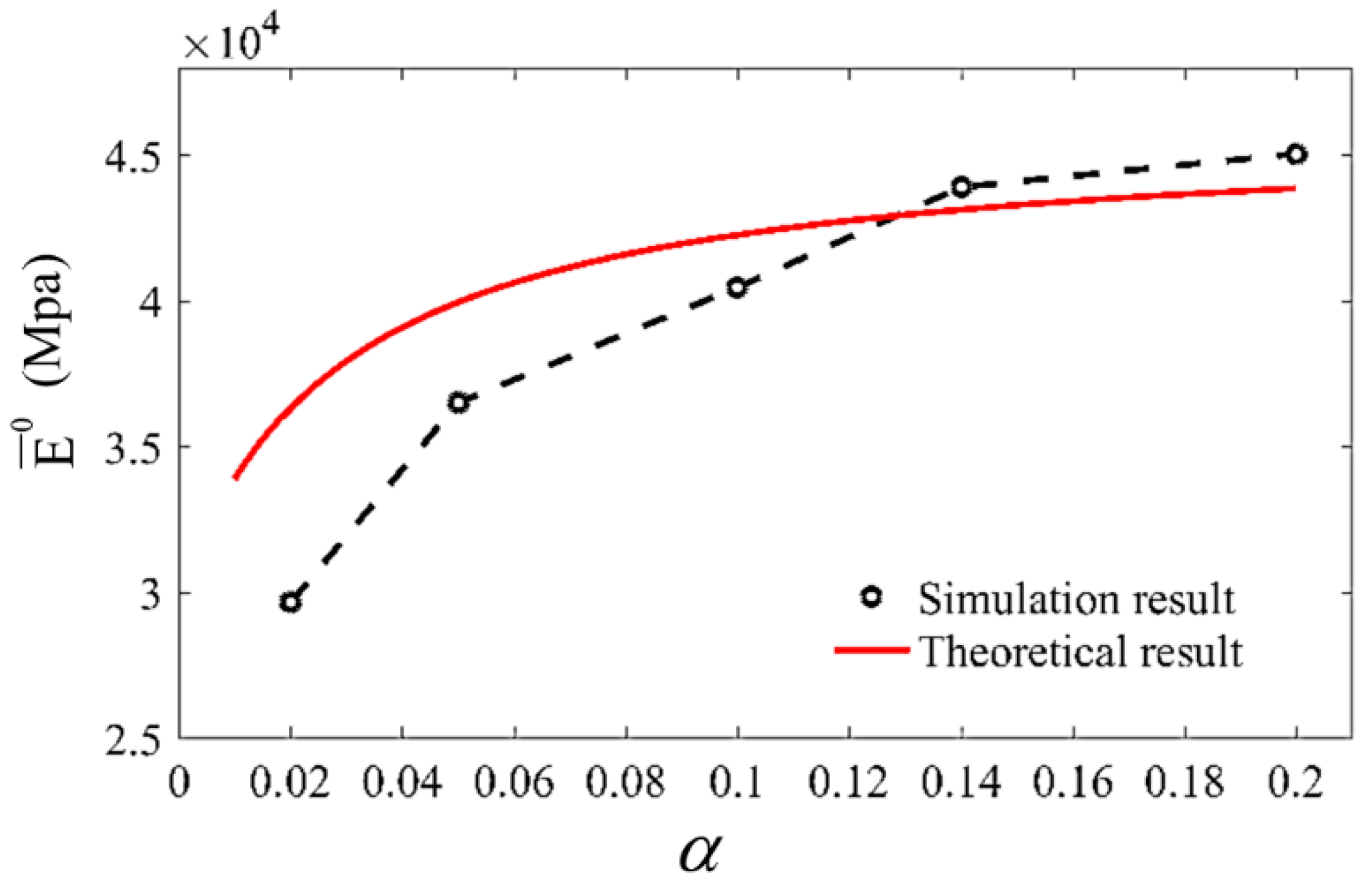
3.2. Simulation Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bechtle, S.; Ang, S.F.; Schneider, G.A. On the mechanical properties of hierarchically structured biological materials. Biomaterials 2010, 31, 6378–6385. [Google Scholar] [CrossRef] [PubMed]
- Tai, K.; Dao, M.; Suresh, S.; Palazoglu, A.; Ortiz, C. Nanoscale heterogeneity promotes energy dissipation in bone. Nat. Mater. 2007, 6, 454. [Google Scholar] [CrossRef] [PubMed]
- Espinosa, H.D.; Rim, J.E.; Barthelat, F.; Buehler, M.J. Merger of structure and material in nacre and bone–Perspectives on de novo biomimetic materials. Prog. Mater. Sci. 2009, 54, 1059–1100. [Google Scholar] [CrossRef]
- Wang, R.; Suo, Z.; Evans, A.; Yao, N.; Aksay, I.A. Deformation mechanisms in nacre. J. Mater. Res. 2001, 16, 2485–2493. [Google Scholar] [CrossRef]
- Evans, A.; Suo, Z.; Wang, R.; Aksay, I.A.; He, M.; Hutchinson, J. Model for the robust mechanical behavior of nacre. J. Mater. Res. 2001, 16, 2475–2484. [Google Scholar] [CrossRef]
- Norman, T.L.; Vashishth, D.; Burr, D. Fracture toughness of human bone under tension. J. Biomech. 1995, 28, 309–320. [Google Scholar] [CrossRef]
- Jäger, I.; Fratzl, P. Mineralized collagen fibrils: A mechanical model with a staggered arrangement of mineral particles. Biophys. J. 2000, 79, 1737–1746. [Google Scholar] [CrossRef]
- Ascenzi, A.; Bonucci, E. The tensile properties of single osteons. Anat. Rec. 1967, 158, 375–386. [Google Scholar] [CrossRef]
- Ascenzi, A.; Bonucci, E. The compressive properties of single osteons. Anat. Rec. 1968, 161, 377–391. [Google Scholar] [CrossRef]
- Rho, J.-Y.; Kuhn-Spearing, L.; Zioupos, P. Mechanical properties and the hierarchical structure of bone. Med. Eng. Phys. 1998, 20, 92–102. [Google Scholar] [CrossRef]
- Meyers, M.A.; McKittrick, J.; Chen, P.-Y. Structural biological materials: Critical mechanics-materials connections. Science 2013, 339, 773–779. [Google Scholar] [CrossRef] [PubMed]
- Schwarcz, H.P.; McNally, E.A.; Botton, G.A. Dark-field transmission electron microscopy of cortical bone reveals details of extrafibrillar crystals. J. Struct. Biol. 2014, 188, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Georgiadis, M.; Mueller, R.; Schneider, P. Techniques to assess bone ultrastructure organization: Orientation and arrangement of mineralized collagen fibrils. J. R. Soc. Interface 2016, 13, 20160088. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Li, D.; Hao, R. Experimental Study of Cortical Bone Microstructure and Its Toughening Mechanism. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2019; p. 032052. [Google Scholar]
- Rief, M.; Gautel, M.; Oesterhelt, F.; Fernandez, J.M.; Gaub, H.E. Reversible unfolding of individual titin immunoglobulin domains by AFM. Science 1997, 276, 1109–1112. [Google Scholar] [CrossRef] [PubMed]
- Smith, B.L.; Schäffer, T.E.; Viani, M.; Thompson, J.B.; Frederick, N.A.; Kindt, J.; Belcher, A.; Stucky, G.D.; Morse, D.E.; Hansma, P.K. Molecular mechanistic origin of the toughness of natural adhesives, fibres and composites. Nature 1999, 399, 761. [Google Scholar] [CrossRef]
- Li, H.; Oberhauser, A.F.; Fowler, S.B.; Clarke, J.; Fernandez, J.M. Atomic force microscopy reveals the mechanical design of a modular protein. Proc. Natl. Acad. Sci. USA 2000, 97, 6527–6531. [Google Scholar] [CrossRef]
- Fantner, G.E.; Hassenkam, T.; Kindt, J.H.; Weaver, J.C.; Birkedal, H.; Pechenik, L.; Cutroni, J.A.; Cidade, G.A.; Stucky, G.D.; Morse, D.E. Sacrificial bonds and hidden length dissipate energy as mineralized fibrils separate during bone fracture. Nat. Mater. 2005, 4, 612. [Google Scholar] [CrossRef]
- Deng, Y.; Cranford, S.W. Tunable toughness of model fibers with bio-inspired progressive uncoiling via sacrificial bonds and hidden length. J. Appl. Mech. 2018, 85, 111001. [Google Scholar] [CrossRef]
- Zhou, X.; Guo, B.; Zhang, L.; Hu, G.-H. Progress in bio-inspired sacrificial bonds in artificial polymeric materials. Chem. Soc. Rev. 2017, 46, 6301–6329. [Google Scholar] [CrossRef]
- Ji, B.; Gao, H. Mechanical properties of nanostructure of biological materials. J. Mech. Phys. Solids 2004, 52, 1963–1990. [Google Scholar] [CrossRef]
- Yao, H.; Gao, H. Multi-scale cohesive laws in hierarchical materials. Int. J. Solids Struct. 2007, 44, 8177–8193. [Google Scholar] [CrossRef]
- Kim, Y.; Kim, Y.; Lee, T.-I.; Kim, T.-S.; Ryu, S. An extended analytic model for the elastic properties of platelet-staggered composites and its application to 3D printed structures. Compos. Struct. 2018, 189, 27–36. [Google Scholar] [CrossRef]
- Hao, R.; Li, D. An inspiration from the microstructure of the cortical bone in goat tibia. In Bioinspired, Biomimetic and Nanobiomaterials; ICE Virtual Library: London, UK, 2019; pp. 1–11. [Google Scholar]
- Hao, R.; Li, D.X. A New Structure Model of Biomimetic Composites Considering the Sub-Microscale Porosity of Bone. Adv. Eng. Mater. 2019, 21, 1900095. [Google Scholar] [CrossRef]
- Wei, X.; Naraghi, M.; Espinosa, H.D. Optimal Length Scales Emerging from Shear Load Transfer in Natural Materials: Application to Carbon-Based Nanocomposite Design. ACS Nano 2012, 6, 2333–2344. [Google Scholar] [CrossRef] [PubMed]
- O’brien, F.J.; Hardiman, D.A.; Hazenberg, J.; Mercy, M.V.; Mohsin, S.; Taylor, D.; Lee, T.C. The behaviour of microcracks in compact bone. Eur. J. Morphol. 2005, 42, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Liu, X.; Yan, Z.; Cai, J.; Kang, F.; Shan, S.; Wang, P.; Zhai, M.; Guo, X.E.; Luo, E. Spatiotemporal characterization of microdamage accumulation in rat ulnae in response to uniaxial compressive fatigue loading. Bone 2018, 108, 156–164. [Google Scholar] [CrossRef]
- Katsamenis, O.L.; Jenkins, T.; Thurner, P.J. Toughness and damage susceptibility in human cortical bone is proportional to mechanical inhomogeneity at the osteonal-level. Bone 2015, 76, 158–168. [Google Scholar] [CrossRef]
- Begley, M.R.; Philips, N.R.; Compton, B.G.; Wilbrink, D.V.; Ritchie, R.O.; Utz, M. Micromechanical models to guide the development of synthetic ‘brick and mortar’ composites. J. Mech. Phys. Solids 2012, 60, 1545–1560. [Google Scholar] [CrossRef]
- Sakhavand, N.; Shahsavari, R. Universal composition-structure-property maps for natural and biomimetic platelet-matrix composites and stacked heterostructures. Nat. Commun. 2015, 6, 6523. [Google Scholar] [CrossRef]
- Hill, R. Elastic properties of reinforced solids: Some theoretical principles. J. Mech. Phys. Solids 1963, 11, 357–372. [Google Scholar] [CrossRef]



| Non-Dimensional Parameters | A | ||||||
|---|---|---|---|---|---|---|---|
| Value | 5–500 | 0.01–0.1 | 0.4 | 2 | 10 | 0.9091 | 1.928 |
| Geometric Parameter | ||||
|---|---|---|---|---|
| (mm) | 3 | 1 | 10 | 1 |
| Part | Element Type | (MPA) | Poisson’s Ratio | ||
|---|---|---|---|---|---|
| Hard plate | CPE4R | 50000 | 0.3 | ||
| Shear part | CPE4R | 5600 | 0.4 | ||
| Joint part | CPE4R | 5000 | 0.3 | ||
| Joint part | COH2D4 | 5000 | 0.3 | 5 | 2 × 10−2 |
| Theoretical result | 4.226 × 104 | 2.975 × 104 | 0.704 | 1.420 × 10−4 |
| Simulation result | 4.046 × 104 | 2.565 × 104 | 0.6341 | 1.446 × 10−4 |
| Relative error | 4.27% | 13.77% | 9.92% | 1.84% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Li, D. A New Biomimetic Composite Structure with Tunable Stiffness and Superior Toughness via Designed Structure Breakage. Materials 2020, 13, 636. https://doi.org/10.3390/ma13030636
Wang X, Li D. A New Biomimetic Composite Structure with Tunable Stiffness and Superior Toughness via Designed Structure Breakage. Materials. 2020; 13(3):636. https://doi.org/10.3390/ma13030636
Chicago/Turabian StyleWang, Xiaohan, and Dongxu Li. 2020. "A New Biomimetic Composite Structure with Tunable Stiffness and Superior Toughness via Designed Structure Breakage" Materials 13, no. 3: 636. https://doi.org/10.3390/ma13030636
APA StyleWang, X., & Li, D. (2020). A New Biomimetic Composite Structure with Tunable Stiffness and Superior Toughness via Designed Structure Breakage. Materials, 13(3), 636. https://doi.org/10.3390/ma13030636





