Reversible Redox Property of Co(III) in Amorphous Co-doped SiO2/γ-Al2O3 Layered Composites
Abstract
1. Introduction
2. Experimental Procedures
2.1. Sample Synthesis
2.1.1. Co-Doped SiO2/γ-Al2O3 Layered Composite Sample
2.1.2. Co-Doped SiO2/γ-Al2O3 Composite Powder Sample
2.1.3. Homogeneous Co-Doped Aluminosilicate Powder Sample
2.1.4. γ-Al2O3/Co-Doped SiO2/γ-Al2O3 Layered Composite Membrane Sample
2.2. Characterizations
2.2.1. Characterization Techniques
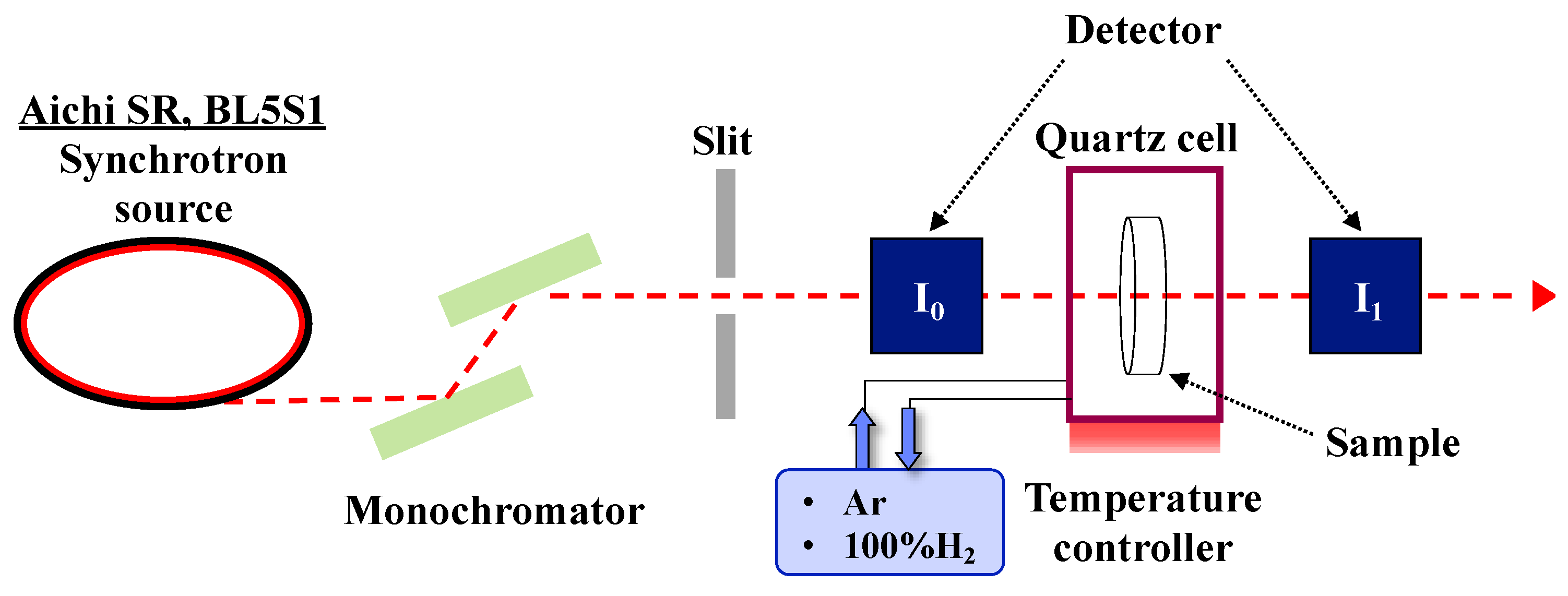
2.2.2. In Situ Characterizations
- heat-treatment to remove adsorbed water at 500 °C for 8 h under a flow of Ar (4 mL·min−1).
- subsequent heat treatment at 500 °C for 0.5 h under a flow of H2 (4 mL·min−1).
- final heat treatment at 500 °C for 8 h under a flow of Ar (4 mL·min−1).
3. Results and Discussion
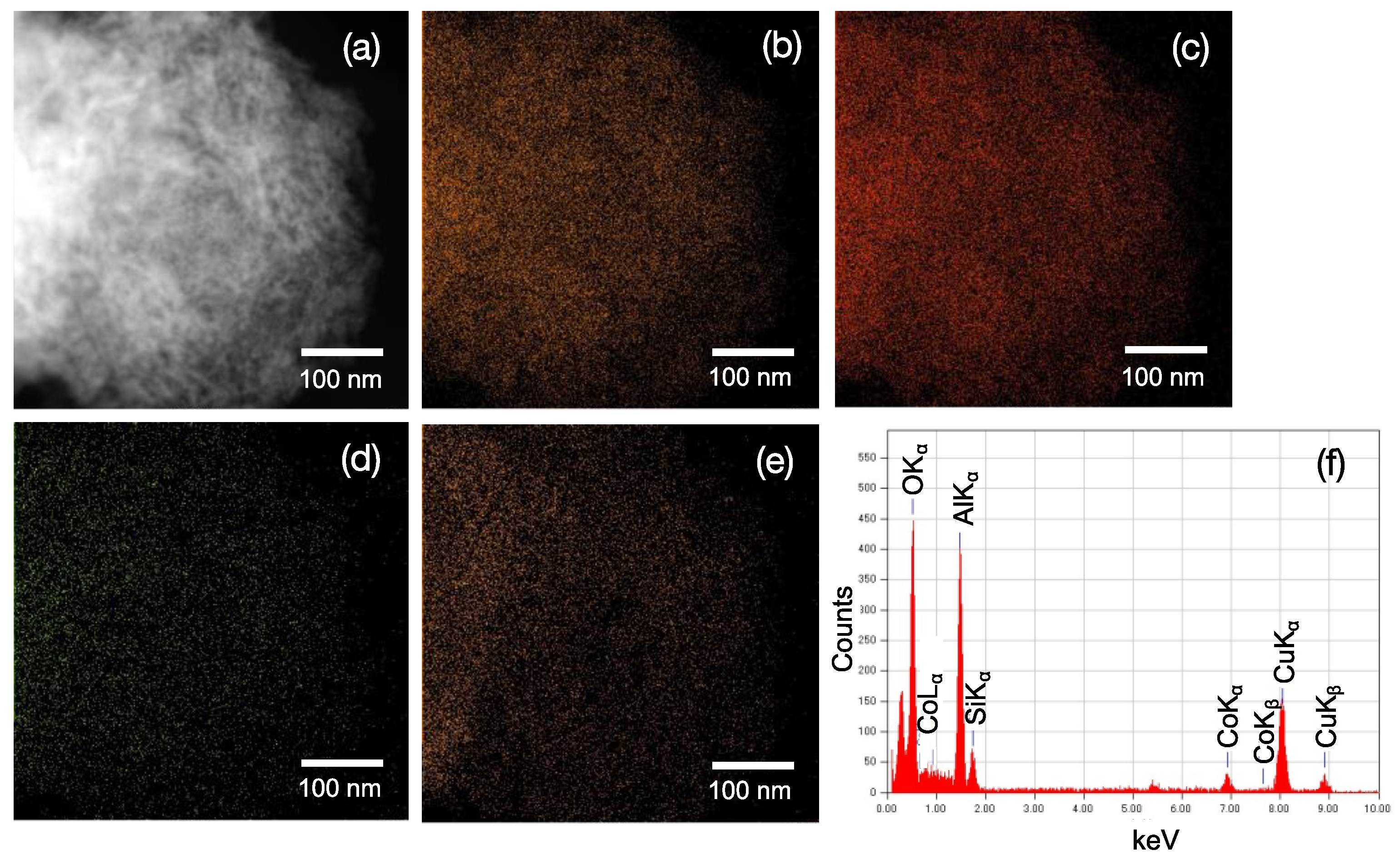
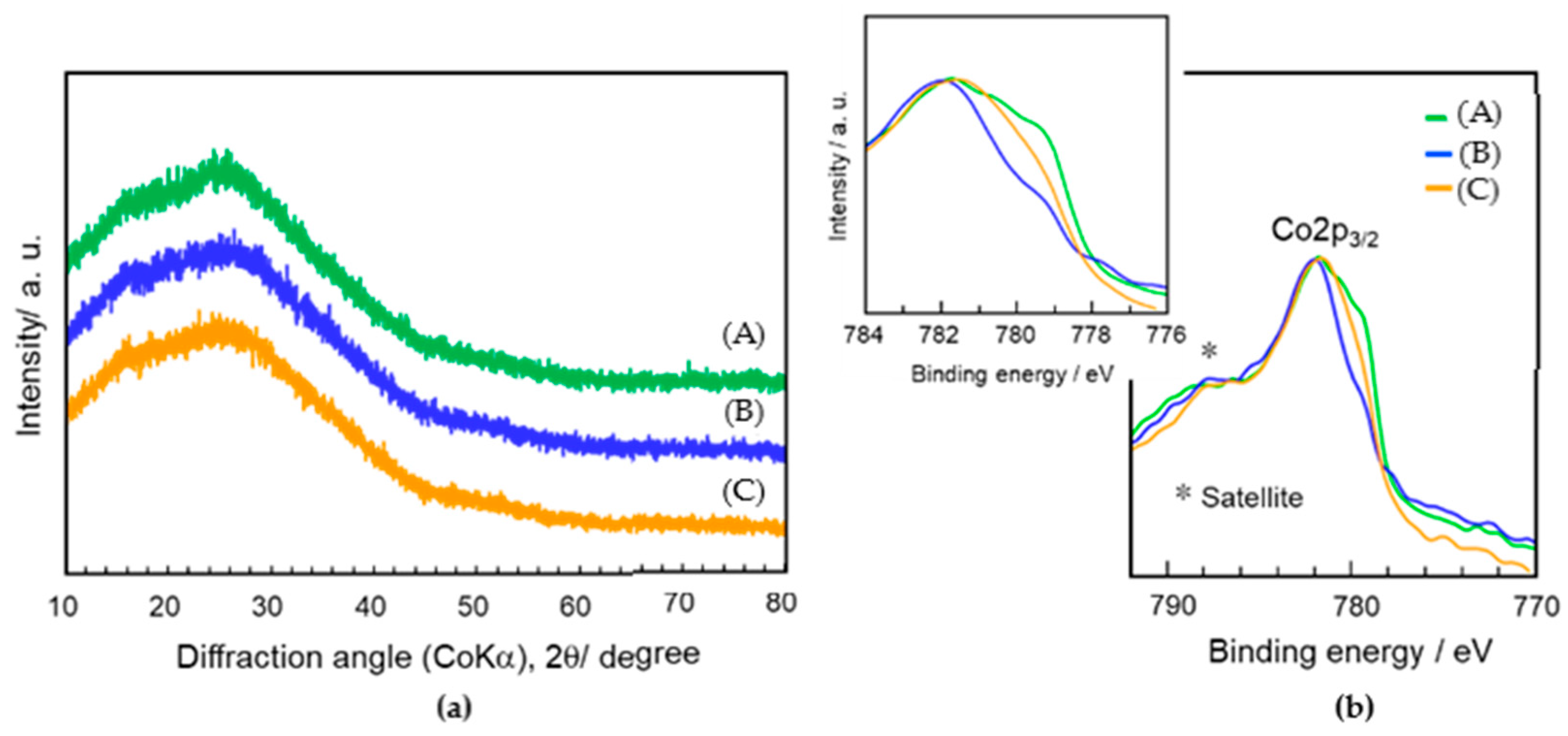
3.1. Distribution of Co Cations within the Co-Doped SiO2/γ-Al2O3 Layered Composite Sample
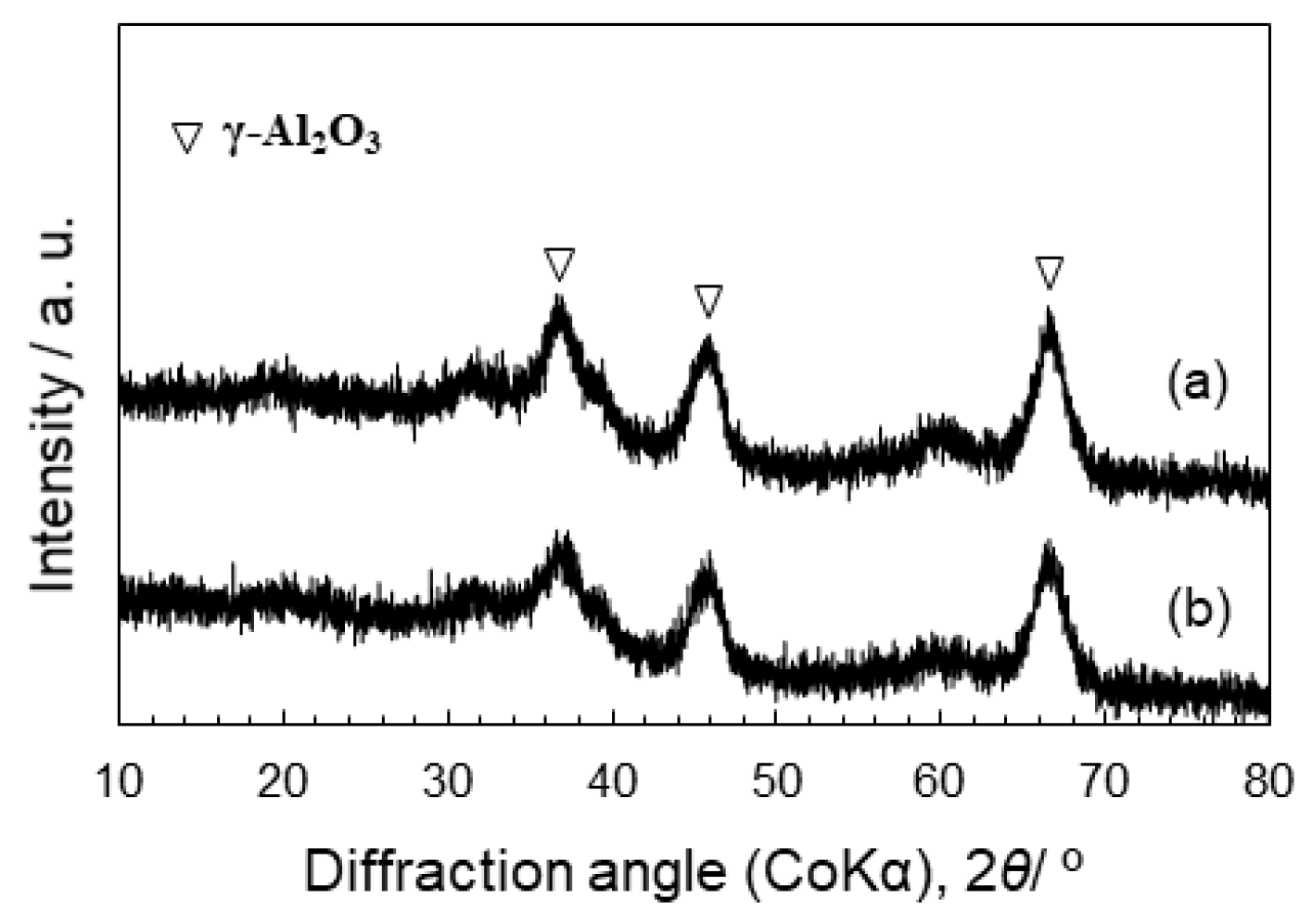
3.2. Properties of Co-Doped SiO2/γ-Al2O3 Composites
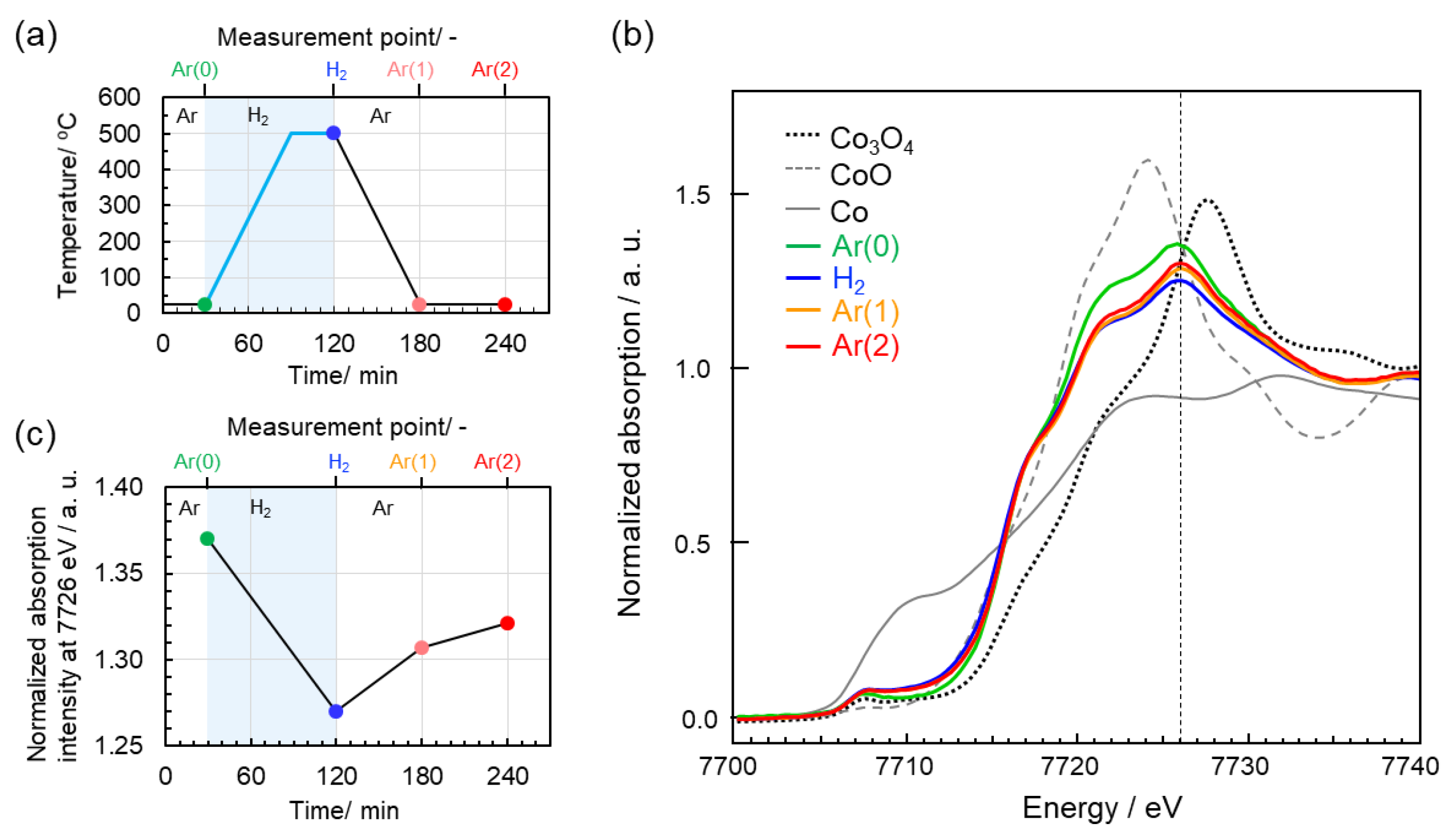
3.3. Redox Behavior of Co Species
- The reduction-oxidation of Co cation species in the CoSiOAlpow sample is associated with the formation-deformation of OH groups.
- The redox reactions begin to proceed at relatively low temperatures around 250–350 °C.
- The redox reactions are reversible and governed by the H2 partial pressure at T ≥ 250 °C.
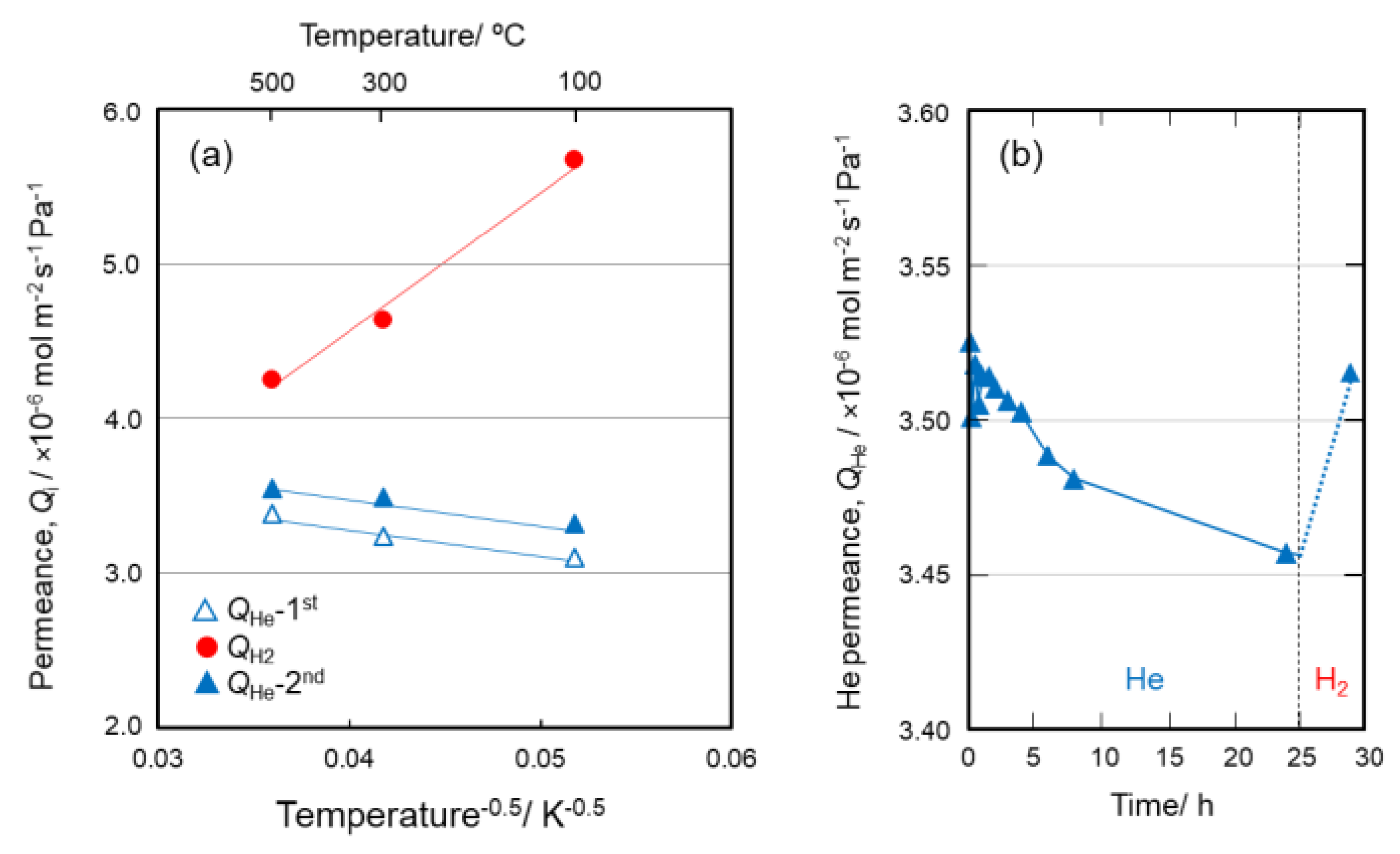
3.4. Gas Permeation Properties of γ-Al2O3/Co-doped SiO2/γ-Al2O3 Layered Composite Membrane
4. Conclusions
- XPS depth profile analyses for the Co-doped SiO2/mesoporous γ-Al2O3 layered composite sample revealed preferential Co diffusion from the SiO2 top-layer matrix to the γ-Al2O3 surface, and the equilibrium chemical composition of the Al, Si, and Co within the mesoporous γ-Al2O3 layer was determined as Al:Si:Co = 85:10:5.
- XRD, HRTEM, and HAADF-STEM analyses revealed that the Co-doped SiO2/γ-Al2O3 composite powder sample with the equilibrium composition of Al:Si:Co = 85:10:5 kept its amorphous state without crystallization of Co oxides after H2 treatment at 500 °C.
- In situ XANES spectroscopic analyses during the 500 °C heat treatment under H2 flow and subsequent cooling to RT under Ar flow exhibited reversible redox properties of Co cations in the Co-doped SiO2/γ-Al2O3 composite powder sample.
- XPS and DRIFT spectroscopic analyses and cyclic TPR/TPD measurements concluded the reversible Co(III)/Co(II) redox reactions were associated with OH formation (hydrogenation)-deformation (dehydrogenation) within amorphous aluminosilicate, which was governed by the H2 partial pressure at 250–500 °C.
- The Co(III) cations in the present composite samples formed via the Fenton reaction in the presence of H2O2 during the sample syntheses.
- Gas permeation measurements for the γ-Al2O3/Co-doped SiO2/γ-Al2O3 layered composite membrane under the cyclic He–H2–He flow suggested that H2 triggered a chemical valve property: micropores under He flow (closure) and mesopores under H2 flow (open), which could be explained by the reversible redox reactions of Co(III)/(II) associated with bond cleavage (hydrogenation)-regeneration (dehydrogenation) of the Si–O–Al amorphous network formed in situ at the hetero interface of the Co-doped SiO2/γ-Al2O3.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Lawal, S.; Kanezashi, M.; Nagasawa, H.; Tsuru, T. Development of an acetylacetonate-modified silica-zirconia composite membrane applicable to gas separation. J. Membr. Sci. 2020, 599, 117844. [Google Scholar] [CrossRef]
- Oyama, S.T.; Aono, H.; Takagaki, A.; Sugawara, T.; Kikuchi, R. Synthesis of Silica Membranes by Chemical Vapor Deposition Using a Dimethyldimethoxysilane Precursor. Membranes 2020, 10, 50. [Google Scholar] [CrossRef]
- Prabhu, A.K.; Oyama, S. Highly hydrogen selective ceramic membranes: Application to the transformation of greenhouse gases. J. Membr. Sci. 2000, 176, 233–248. [Google Scholar] [CrossRef]
- Da Costa, J.D.; Lu, G.; Rudolph, V.; Lin, Y. Novel molecular sieve silica (MSS) membranes: Characterisation and permeation of single-step and two-step sol–gel membranes. J. Membr. Sci. 2002, 198, 9–21. [Google Scholar] [CrossRef]
- Iwamoto, Y. Precursors-Derived Ceramic Membranes for High-Temperature Separation of Hydrogen. J. Ceram. Soc. Jpn. 2007, 115, 947–954. [Google Scholar] [CrossRef]
- Miricioiu, M.G.; Iacob, C.; Nechifor, G.; Niculescu, V.-C. High Selective Mixed Membranes Based on Mesoporous MCM-41 and MCM-41-NH2 Particles in a Polysulfone Matrix. Front. Chem. 2019, 7, 332. [Google Scholar] [CrossRef]
- Ren, X.; Tsuru, T. Organosilica-Based Membranes in Gas and Liquid-Phase Separation. Membranes 2019, 9, 107. [Google Scholar] [CrossRef]
- Brinker, C.J.; Scherer, G.W. Sol-Gel Science, the Physics and Chemistry of Sol-Gel Processing; Academic Press: Boston, MA, USA, 1990; p. 908. ISBN 0-12-134970-5. [Google Scholar]
- Tanaka, K.; Sakata, Y. Present and Future Prospects of Hydrogen Production Process Constructed by the Combination of Photocatalytic H2O Splitting and Membrane Separation Process. Membranes 2011, 36, 113–121. [Google Scholar] [CrossRef]
- Miyajima, K.; Eda, T.; Nair, B.N.; Honda, S.; Iwamoto, Y. Hydrothermal stability of hydrogen permselective amorphous silica membrane synthesized by counter diffusion chemical vapor deposition method. J. Ceram. Soc. Jpn. 2013, 121, 992–998. [Google Scholar] [CrossRef][Green Version]
- Yoshida, K.; Hirano, Y.; Fujii, H.; Tsuru, T.; Asaeda, M. Hydrothermal Stability and Performance of Silica-Zirconia Membranes for Hydrogen Separation in Hydrothermal Conditions. J. Chem. Eng. Jpn. 2001, 34, 523–530. [Google Scholar] [CrossRef]
- Kanezashi, M.; Asaeda, M. Hydrogen permeation characteristics and stability of Ni-doped silica membranes in steam at high temperature. J. Memb. Sci. 2006, 271, 86–93. [Google Scholar] [CrossRef]
- Igi, R.; Yoshioka, T.; Ikuhara, Y.H.; Iwamoto, Y.; Tsuru, T. Characterization of Co-Doped Silica for Improved Hydrothermal Stability and Application to Hydrogen Separation Membranes at High Temperatures. J. Am. Ceram. Soc. 2008, 91, 2975–2981. [Google Scholar] [CrossRef]
- Uhlmann, D.; Liu, S.; Ladewig, B.P.; Da Costa, J.C.D. Cobalt-doped silica membranes for gas separation. J. Membr. Sci. 2009, 326, 316–321. [Google Scholar] [CrossRef]
- Yacou, C.; Smart, S.; Da Costa, J.C.D. Long term performance cobalt oxide silica membrane module for high temperature H2 separation. Energy Environ. Sci. 2012, 5, 5820–5832. [Google Scholar] [CrossRef]
- Fujisaki, S.; Hataya, K.; Saito, T.; Arai, S.; Iwamoto, Y.; Kuroda, K. Nanostructural characterizations of hydrogen-permselective Si–Co–O membranes by transmission electron microscopy. J. Mater. Res. 2009, 24, 372–378. [Google Scholar] [CrossRef]
- Miller, C.R.; Wang, D.K.; Smart, S.; Da Costa, J.C.D. Reversible Redox Effect on Gas Permeation of Cobalt Doped Ethoxy Polysiloxane (ES40) Membranes. Sci. Rep. 2013, 3, srep01648. [Google Scholar] [CrossRef]
- Estournès, C.; Lutz, T.; Guille, J. Reduction of nickel in soda—lime—silicate glass by hydrogen. J. Non-Cryst. Solids 1996, 197, 192–196. [Google Scholar] [CrossRef]
- Johnston, W.D.; Chelko, A.J. Reduction of Ions in Glass by Hydrogen. J. Am. Ceram. Soc. 1970, 53, 295–301. [Google Scholar] [CrossRef]
- Smedskjaer, M.M.; Deubener, J.; Yue, Y. Inward Cationic Diffusion and Formation of Silica-Rich Surface Nanolayer of Glass. Chem. Mater. 2009, 21, 1242–1247. [Google Scholar] [CrossRef]
- Chahadih, A.; El Hamzaoui, H.; Cristini, O.; Bigot, L.; Bernard, R.; Kinowski, C.; Bouazaoui, M.; Capoen, B. H2-induced copper and silver nanoparticle precipitation inside sol-gel silica optical fiber preforms. Nanoscale Res. Lett. 2012, 7, 487. [Google Scholar] [CrossRef]
- Bates, S.A.; Verma, A.A.; Paolucci, C.; Parekh, A.A.; Anggara, T.; Yezerets, A.; Schneider, W.F.; Miller, J.T.; Delgass, W.N.; Ribeiro, F.H. Identification of the active Cu site in standard selective catalytic reduction with ammonia on Cu-SSZ-13. J. Catal. 2014, 312, 87–97. [Google Scholar] [CrossRef]
- Nogami, M. Reduction Mechanism for Eu Ions in Al2O3-Containing Glasses by Heat Treatment in H2 Gas. J. Phys. Chem. B 2015, 119, 1778–1784. [Google Scholar] [CrossRef]
- Nogami, M.; Koiwai, A.; Nonaka, T. Control of Oxidation State of Eu Ions in Na2O-Al2O3–SiO2 Glasses. J. Am. Ceram. Soc. 2016, 99, 1248–1254. [Google Scholar] [CrossRef]
- Van Tuyen, H.; Nonaka, T.; Yamanaka, K.-I.; Chau, P.M.; Hai, N.T.Q.; Quang, V.X.; Nogami, M. Fluorescence properties of valence-controlled Eu2+ and Mn2+ ions in aluminosilicate glasses. J. Lumin. 2017, 184, 83–88. [Google Scholar] [CrossRef]
- Van Tuyen, H.; Nogami, M.; Hung, L.X. Reduction of Sm3+ and Eu3+ ions-co-doped Al2O3–SiO2 glasses and photoluminescence properties. Opt. Mater. 2020, 100, 109639. [Google Scholar] [CrossRef]
- Chau, P.T.M.; Hai, N.T.Q.; Quang, V.X.; Van Tuyen, H.; Nogami, M. Control Valence and Luminescence Properties of Cerium Ions in Al2O3–SiO2 Glasses Prepared by Sol–Gel Method. J. Electron. Mater. 2019, 48, 6972–6977. [Google Scholar] [CrossRef]
- Kubo, M.; Kojima, M.; Mano, R.; Daiko, Y.; Honda, S.; Iwamoto, Y. A hydrostable mesoporous γ-Al2O3 membrane modified with Si–C–H organic-inorganic hybrid derived from polycarbosilane. J. Membr. Sci. 2020, 598, 117799. [Google Scholar] [CrossRef]
- Sing, K.S.W. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity (Recommendations 1984). Pure Appl. Chem. 1985, 57, 603–619. [Google Scholar] [CrossRef]
- Barrett, E.P.; Joyner, L.G.; Halenda, P.P. The Determination of Pore Volume and Area Distributions in Porous Substances. I. Computations from Nitrogen Isotherms. J. Am. Chem. Soc. 1951, 73, 373–380. [Google Scholar] [CrossRef]
- Barrer, R.M.; Rideal, E.K. Permeation, diffusion and solution of gases in organic polymers. Trans. Faraday Soc. 1939, 35, 628–643. [Google Scholar] [CrossRef]
- Iwamoto, Y.; Sato, K.; Kato, T.; Inada, T.; Kubo, Y. A hydrogen-permselective amorphous silica membrane derived from polysilazane. J. Eur. Ceram. Soc. 2005, 25, 257–264. [Google Scholar] [CrossRef]
- Inada, T.; Uno, N.; Kato, T.; Iwamoto, Y. Meso-Porous Alumina Capillary Tube as a Support for High-Temperature Gas Separation Membranes by Novel Pulse Sequential Anodic Oxidation Technique. J. Mater. Res. 2005, 20, 114–120. [Google Scholar] [CrossRef]
- Ravel, B.; Newville, M. ATHENA, ARTEMIS, HEPHAESTUS: Data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Radiat. 2005, 12, 537–541. [Google Scholar] [CrossRef] [PubMed]
- Barrow, B.N.S.; Scullard, A.; Collis, N. Surface Selective 1H and 27Al MAS NMR Observations of Strontium Oxide Doped γ-Alumina. Johns. Matthey Technol. Rev. 2016, 60, 90–97. [Google Scholar] [CrossRef]
- Silverstein, R.M.; Bassler, G.C.; Morrill, T.C. Spectrometric Identification of Organic Compounds; John Wiley & Sons: New York, NY, USA, 1991; pp. 135–139. ISBN 4-8079-0506-6. [Google Scholar]
- Mouline, Z.; Asai, K.; Daiko, Y.; Honda, S.; Bernard, S.; Iwamoto, Y. Amine-functionalized polycarbosilane hybrids for CO2-selective membranes. J. Eur. Ceram. Soc. 2017, 37, 5213–5221. [Google Scholar] [CrossRef]
- Chakarova, K.; Drenchev, N.; Mihaylov, M.; Nikolov, P.; Hadjiivanov, K. OH/OD Isotopic Shift Factors of Isolated and H-Bonded Surface Silanol Groups. J. Phys. Chem. C 2013, 117, 5242–5248. [Google Scholar] [CrossRef]
- Kengne, B.-A.F.; Alayat, A.M.; Luo, G.; McDonald, A.G.; Brown, J.D.; Smotherman, H.; McIlroy, D.N. Preparation, surface characterization and performance of a Fischer-Tropsch catalyst of cobalt supported on silica nanosprings. Appl. Surf. Sci. 2015, 359, 508–514. [Google Scholar] [CrossRef]
- Tada, S.; Ando, S.; Asaka, T.; Daiko, Y.; Honda, S.; Bernard, S.; Iwamoto, Y. Hydrogen transport property of polymer-derived cobalt cation-doped amorphous silica. Inorg. Chem. Front. 2020. [Google Scholar] [CrossRef]
- Fenton, H.J.H. LXXIII.—Oxidation of tartaric acid in presence of iron. J. Chem. Soc. Trans. 1894, 65, 899–910. [Google Scholar] [CrossRef]
- Puppo, A.; Halliwell, B. Formation of hydroxyl radicals from hydrogen peroxide in the presence of iron. Is haemoglobin a biological Fenton reagent? Biochem. J. 1988, 249, 185–190. [Google Scholar] [CrossRef]
- Ling, S.K.; Wang, S.; Peng, Y. Oxidative degradation of dyes in water using Co2+/H2O2 and Co2+/peroxymonosulfate. J. Hazard. Mater. 2010, 178, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Breck, D.W. Zeolite Molecular Sieves: Structure, Chemistry, and Use; John Wiley & Sons: New York, NY, USA, 1974; pp. 634–636. ISBN 978-0471099857. [Google Scholar]
- Yoshioka, T.; Tsuru, T.; Asaeda, M. Molecular dynamics study of gas permeation through amorphous silica network and inter-particle pores on microporous silica membranes. Mol. Phys. 2004, 102, 191–202. [Google Scholar] [CrossRef]
- Yoshioka, T.; Kanezashi, M.; Tsuru, T. Micropore size estimation on gas separation membranes: A study in experimental and molecular dynamics. AIChE J. 2013, 59, 2179–2194. [Google Scholar] [CrossRef]
- Malek, K.; Coppens, M.-O. Knudsen self- and Fickian diffusion in rough nanoporous media. J. Chem. Phys. 2003, 119, 2801–2811. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tada, S.; Saito, S.; Mori, A.; Mizuno, H.; Ando, S.; Asaka, T.; Daiko, Y.; Honda, S.; Bernard, S.; Iwamoto, Y. Reversible Redox Property of Co(III) in Amorphous Co-doped SiO2/γ-Al2O3 Layered Composites. Materials 2020, 13, 5345. https://doi.org/10.3390/ma13235345
Tada S, Saito S, Mori A, Mizuno H, Ando S, Asaka T, Daiko Y, Honda S, Bernard S, Iwamoto Y. Reversible Redox Property of Co(III) in Amorphous Co-doped SiO2/γ-Al2O3 Layered Composites. Materials. 2020; 13(23):5345. https://doi.org/10.3390/ma13235345
Chicago/Turabian StyleTada, Shotaro, Shota Saito, Akito Mori, Hideki Mizuno, Shiori Ando, Toru Asaka, Yusuke Daiko, Sawao Honda, Samuel Bernard, and Yuji Iwamoto. 2020. "Reversible Redox Property of Co(III) in Amorphous Co-doped SiO2/γ-Al2O3 Layered Composites" Materials 13, no. 23: 5345. https://doi.org/10.3390/ma13235345
APA StyleTada, S., Saito, S., Mori, A., Mizuno, H., Ando, S., Asaka, T., Daiko, Y., Honda, S., Bernard, S., & Iwamoto, Y. (2020). Reversible Redox Property of Co(III) in Amorphous Co-doped SiO2/γ-Al2O3 Layered Composites. Materials, 13(23), 5345. https://doi.org/10.3390/ma13235345





