Pyrolysis of Mixed Plastic Waste: I. Kinetic Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Thermal Decomposition of Mixed Polymer Samples
2.3. Kinetic Theory
- α: is the reaction conversion,
- t: is the time (min),
- β: is the heating rate (K/min),
- T: is the temperature (K),
- Ao: is the pre-exponential factor (min−1),
- E: is the activation energy (J/mol),
- R: is the universal gas constant (8.314 J/mol.K),
- f(α): is the concentration-dependent term
- Tα: Temperature at conversion α,
- T0.5: Temperature at conversion (α) = 0.5,
- (dα/dt)α: Conversion change with time at conversion (α),
- (dα/dt)0.5: Conversion change with time at conversion (α) = 0.5.
3. Results and Discussion
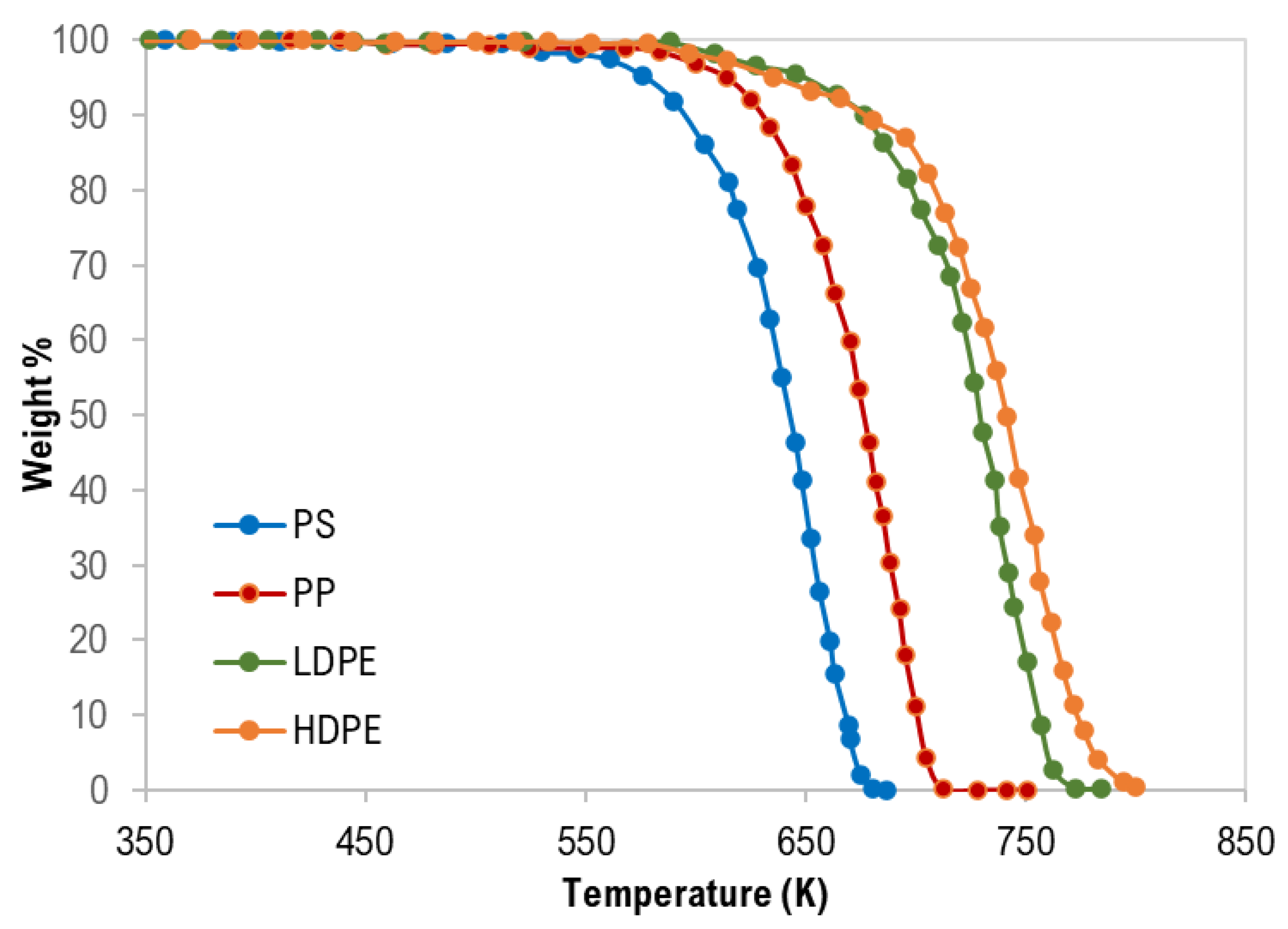
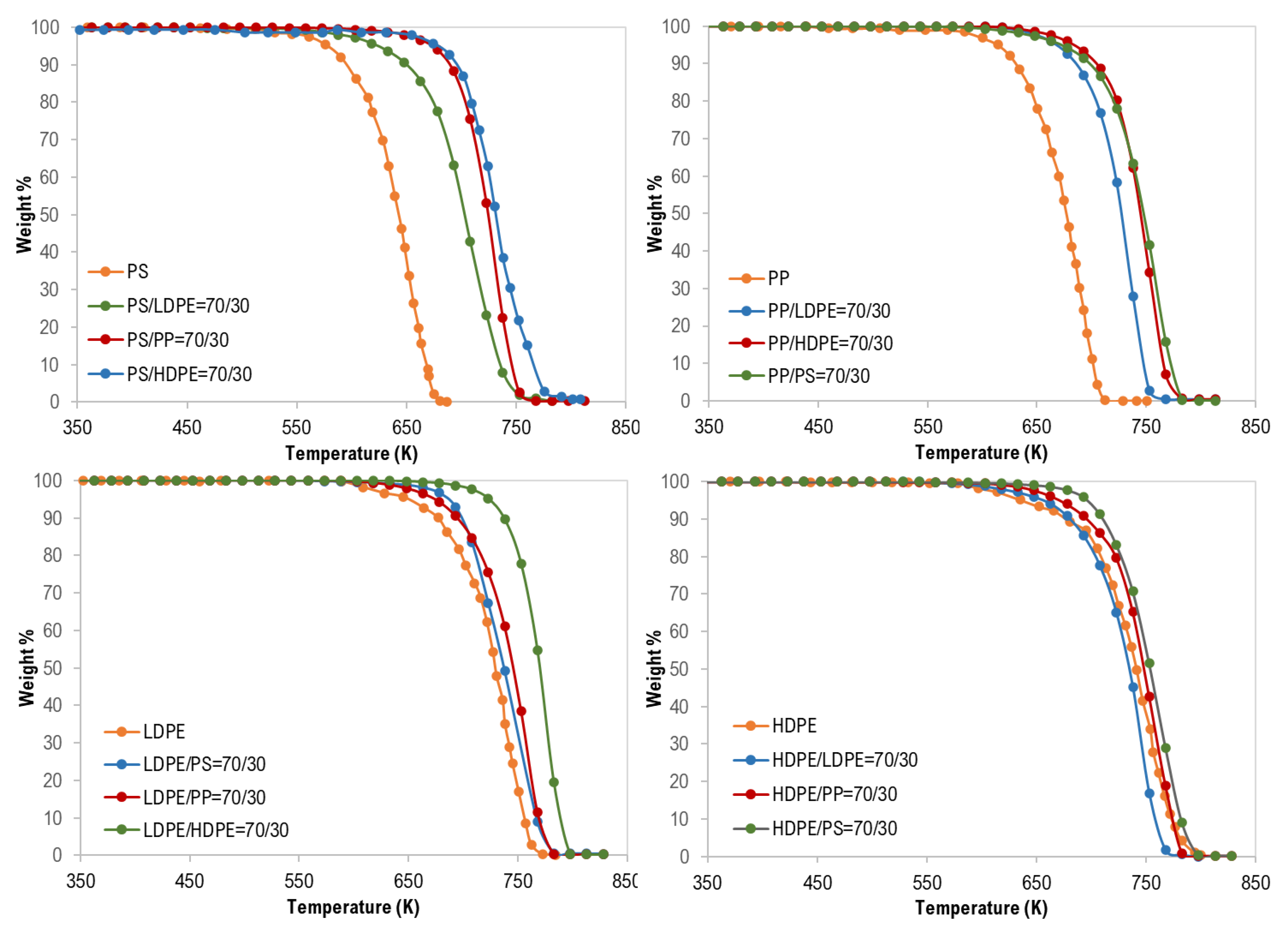
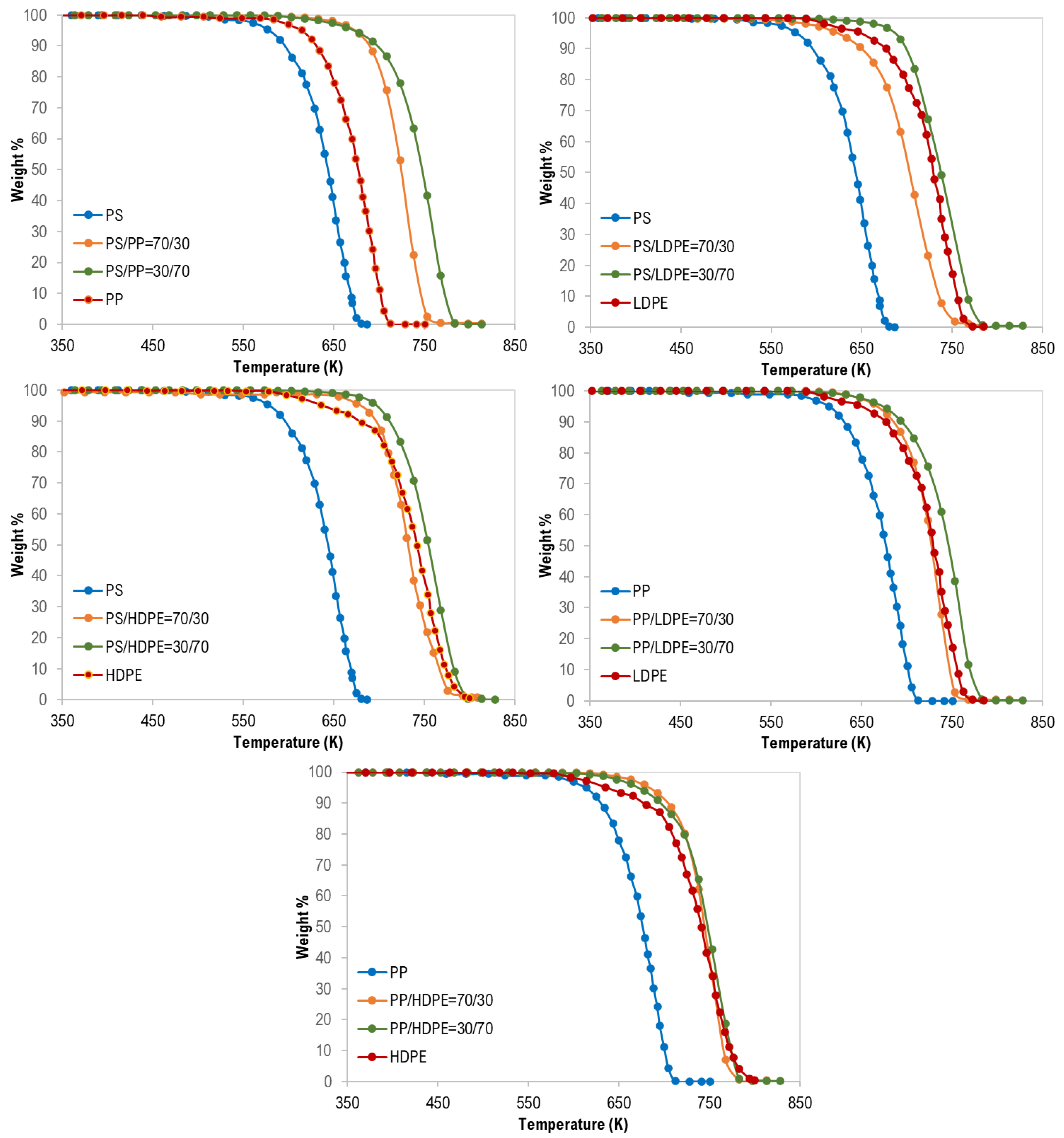
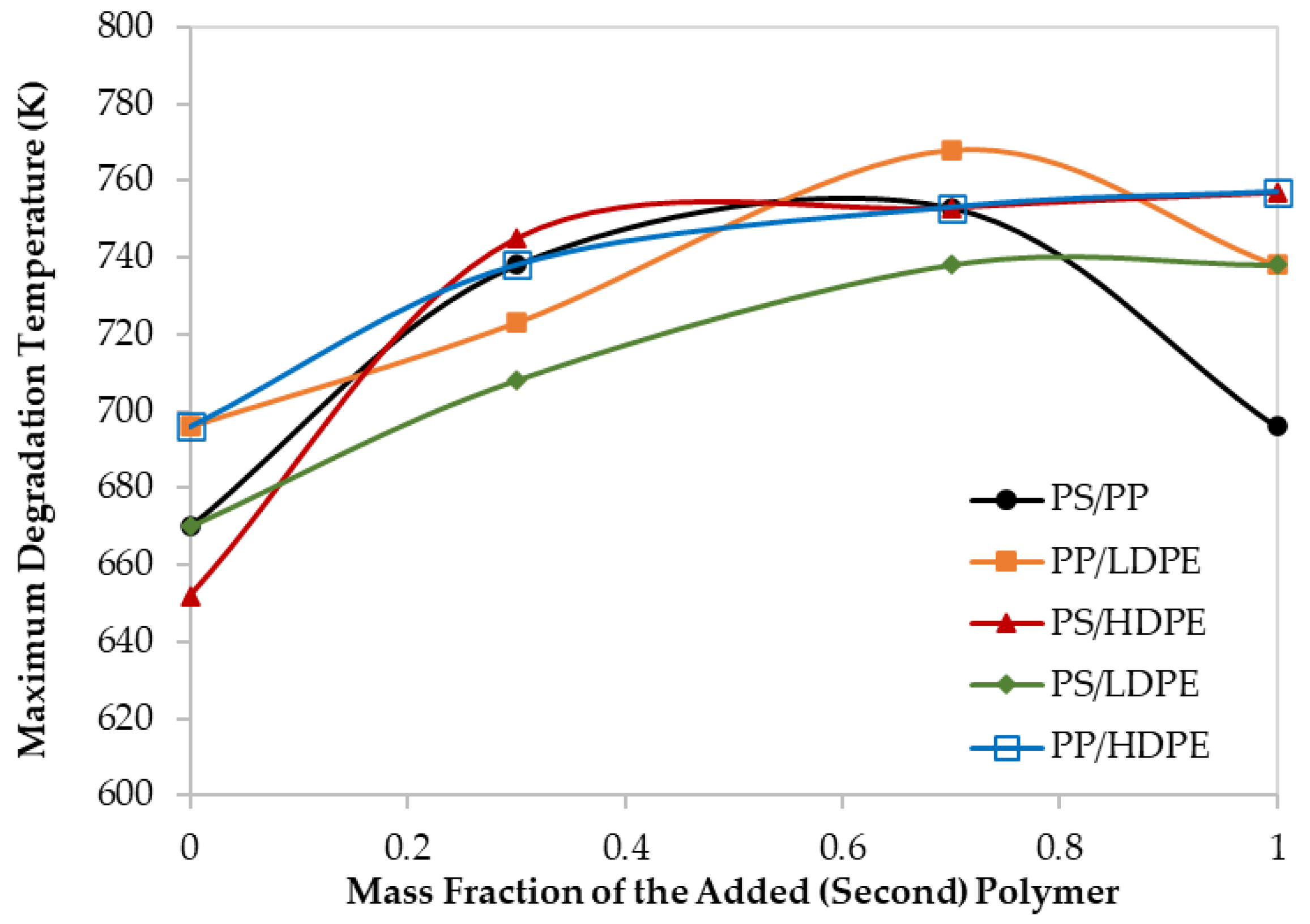
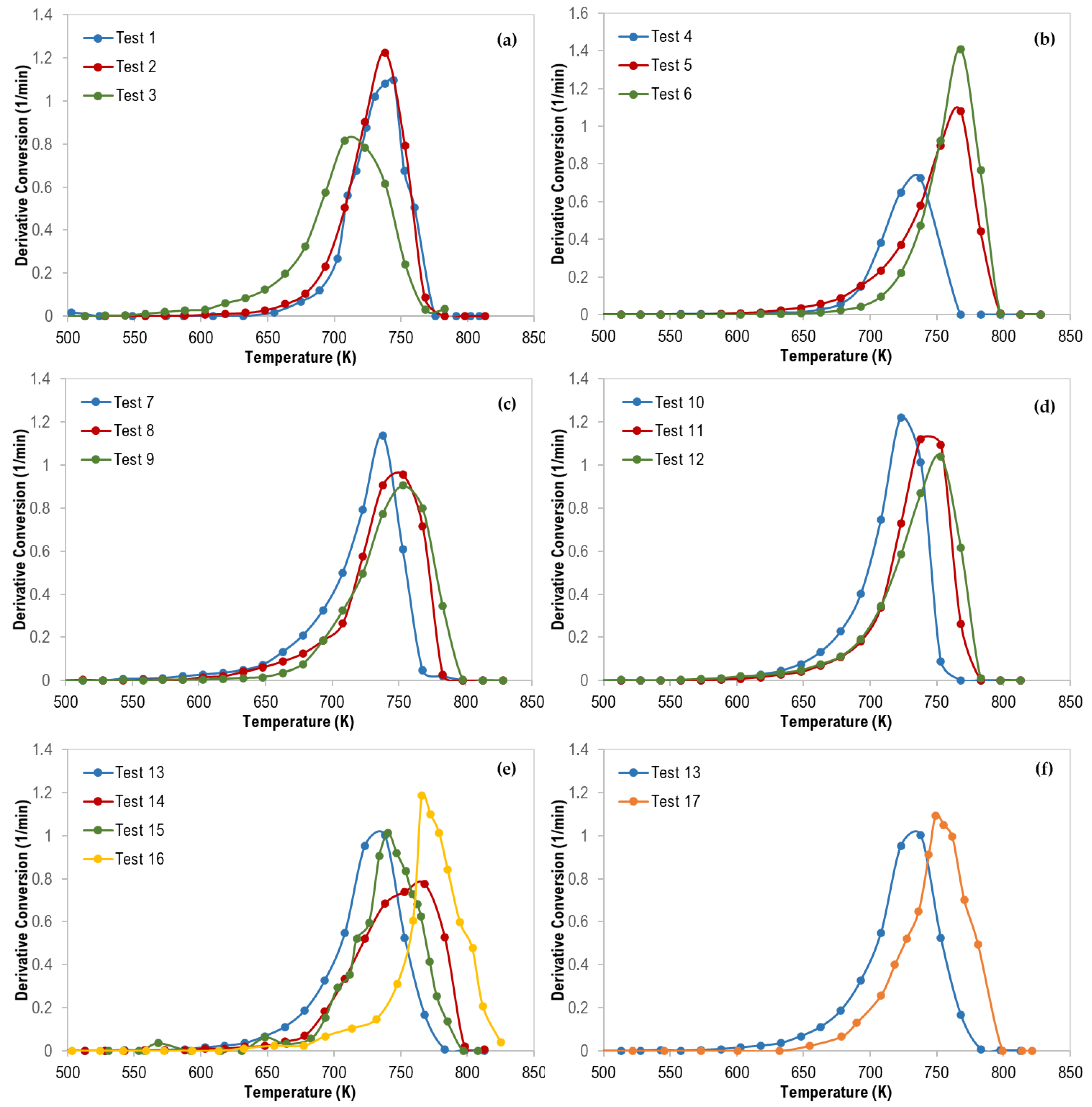
3.1. Thermal Analysis of Mixed Polymers
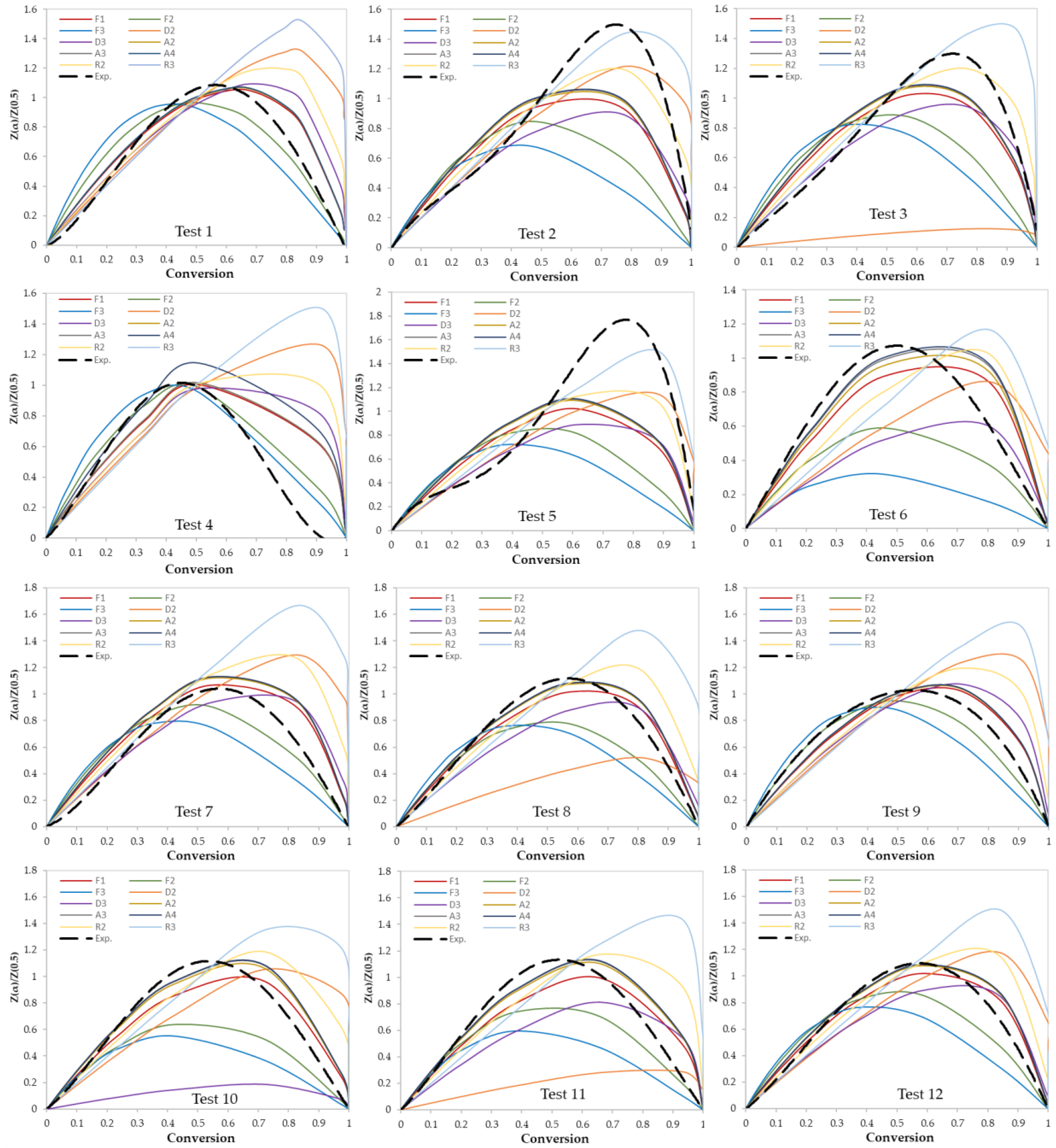
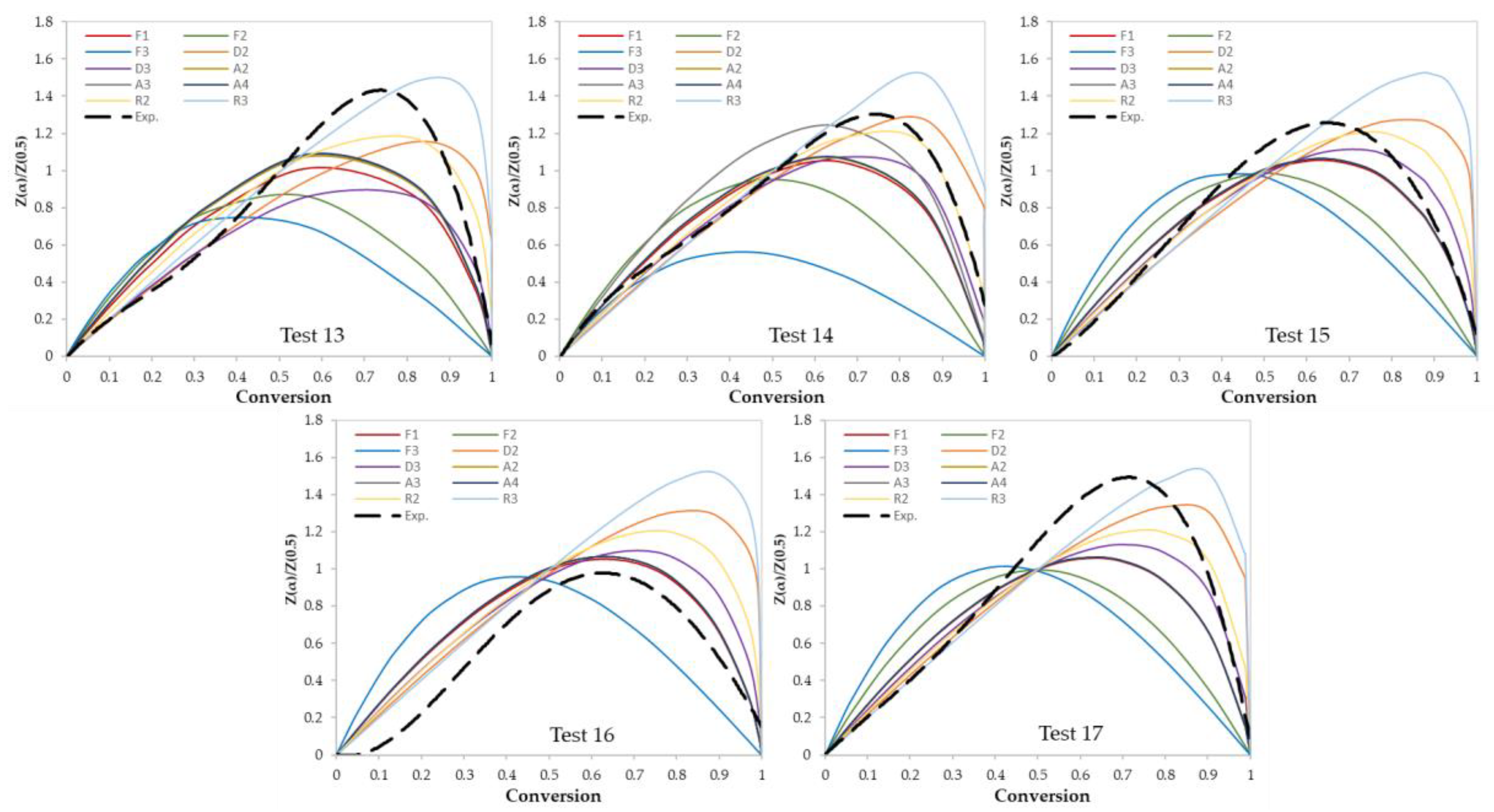
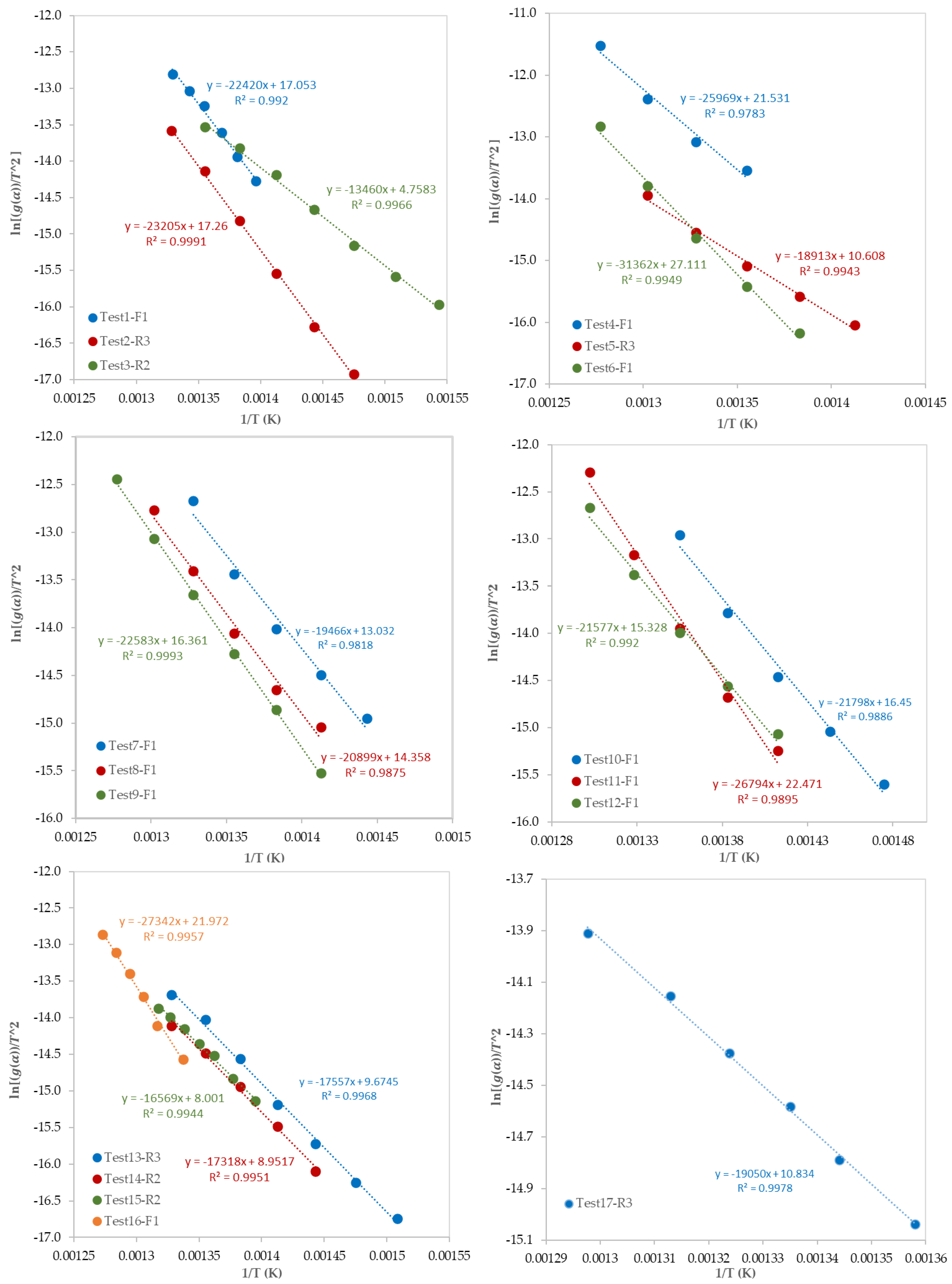
3.2. Determination of Reaction Mechanisms and Kinetic Parameters
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kaza, S.; Yao, L.; Bhada-Tata, P.; VanWoerden, F. What a Waste 2.0: A Global Snapshot of Solid Waste Management to 2050; The World Bank: Washington, DC, USA, 2018. [Google Scholar] [CrossRef]
- Nizami, A.S.; Ouda, O.K.M.; Rehan, M.; El-Maghraby, A.M.O.; Gardy, J.; Hassanpour, A.; Kumar, S. IMI Ismail, The potential of Saudi Arabian natural zeolites in energy recovery technologies. Energy 2016, 108, 162–171. [Google Scholar] [CrossRef]
- Ouda, O.K.M.; Cekirge, H.M.; Raza, S.A. An assessment of the potential contribution from waste-to-energy facilities to electricity demand in Saudi Arabia. Energy Convers. Manag. 2013, 75, 402–406. [Google Scholar] [CrossRef]
- Silvarrey, L.S.D.; Phan, A.N. Kinetic study of municipal plastic waste. Int. J. Hydrogen Energy 2016, 41, 16352–16364. [Google Scholar] [CrossRef]
- Kaminsky, W.; Schlesselmann, B.; Simon, C.M. Thermal degradation of mixed plastic waste to aromatic and gas. Poly. Degrad. Stab. 1996, 53, 189–197. [Google Scholar] [CrossRef]
- Aguado, J.; Serrano, D.P.; Miguel, G.S.; Castro, M.C.; Madrid, S. Feedstock recycling of polyethylene in a two-step thermo-catalytic reaction system. J. Anal. Appl. Pyrolysis 2007, 79, 415–423. [Google Scholar] [CrossRef]
- Jan, M.R.; Shah, J.; Gulab, H. Catalytic conversion of waste high-density polyethylene into useful hydrocarbons. Fuel 2013, 105, 595–602. [Google Scholar] [CrossRef]
- Al-Yaari, M.; Dubdub, I. Application of Artificial Neural Networks to Predict the Catalytic Pyrolysis of HDPE Using Non-Isothermal TGA Data. Polymers 2020, 12, 1813. [Google Scholar] [CrossRef]
- Matsuzawa, Y.; Ayabe, M.; Nishino, J.; Kubota, N.; Motegi, M. Evaluation of char fuel ratio in municipal pyrolysis waste. Fuel 2004, 83, 1675–1687. [Google Scholar] [CrossRef]
- Dubdub, I.; Al-Yaari, M. Pyrolysis of Low Density Polyethylene: Kinetic Study Using TGA Data and ANN Prediction. Polymers 2020, 12, 891. [Google Scholar] [CrossRef]
- Zhou, R.; Huang, B.; Ding, Y.; Li, W.; Mu, J. Thermal Decomposition Mechanism and Kinetics Study of Plastic Waste Chlorinated Polyvinyl Chloride. Polymers 2019, 11, 2080. [Google Scholar] [CrossRef]
- Xu, F.; Wang, B.; Yang, D.; Hao, J.; Qiao, Y.; Tian, Y. Thermal degradation of typical plastics under high heating rate conditions by TG-FTIR: Pyrolysis behaviors and kinetic analysis. Energy Convers. Manag. 2018, 171, 1106–1115. [Google Scholar] [CrossRef]
- Ma, W.; Rajput, G.; Pan, M.; Lin, F.; Zhong, L.; Chen, G. Pyrolysis of typical MSW components by Py-GC/MS and TG-FTIR. Fuel 2019, 251, 693–708. [Google Scholar] [CrossRef]
- Chan, J.H.; Balke, S.T. The thermal degradation kinetics of polypropylene: Part III. thermogravimetric analyses. Polym. Degrad. Stabil. 1997, 57, 135–149. [Google Scholar] [CrossRef]
- Charde, S.J.; Sonawane, S.S.; Sonawane, S.H.; Shimpi, N.G. Degradation Kinetics of Polycarbonate Composites: Kinetic Parameters and Artificial Neural Network. Chem. Biochem. Eng. Q. 2018, 32, 151–165. [Google Scholar] [CrossRef]
- Ayhan, D. Pyrolysis of municipal plastic wastes for recovery of gasoline-range hydrocarbons. J. Anal. Appl. Pyrolysis 2004, 72, 97–102. [Google Scholar] [CrossRef]
- Chowlu, A.C.K.; Reddy, P.K.; Ghoshal, A.K. Pyrolytic decomposition and model-free kinetics analysis of mixture of polypropylene (PP) and low-density polyethylene (LDPE). Thermochim. Acta 2009, 485, 20–25. [Google Scholar] [CrossRef]
- Anjum, M.; Miandad, R.; Waqas, M.; Tarar, I.A.; Alafif, Z.; Aburiazaiza, A.S.; Barakat, M.A.; Akhtar, T. Solid waste management in Saudi Arabia: A review. J. Appl. Agric. Biotechnol. 2016, 1, 13–26. [Google Scholar]
- Yu, J.; Sun, L.; Ma, C.; Qiao, Y.; Yao, H. Thermal degradation of PVC: A review. Waste Manag. 2016, 48, 300–314. [Google Scholar] [CrossRef]
- Park, C.H.; Jeon, H.S.; Park, J.K. PVC removal from mixed plastics by triboelectrostatic separation. J. Hazard. Mater. 2007, 144, 470–476. [Google Scholar] [CrossRef]
- Aboulkas, A.; El Harfi, K.; Al Bouadili, A. Thermal degradation behaviors of polyethylene and polypropylene. Part I Pyrolysis Kinet. Mech. Energy Convers. Manag. 2010, 51, 1363–1369. [Google Scholar] [CrossRef]
- Anene, A.F.; Fredriksen, S.B.; Saetre, K.A.; Tokheim, L.A. Experimental Study of Thermal and Catalytic Pyrolysis of Plastic Waste Components. Sustainability 2018, 10, 3979. [Google Scholar] [CrossRef]
- Mumbacha, G.D.; Alves, J.L.F.; Silva, J.C.G.D.; Sena, R.F.D.; Marangoni, C.; Machado, R.A.F.; Bolzan, A. Thermal investigation of plastic solid waste pyrolysis via the deconvolution technique using the asymmetric double sigmoidal function: Determination of the kinetic triplet, thermodynamic parameters, thermal lifetime and pyrolytic oil composition for clean energy recovery. Energy Convers. Manag. 2019, 200, 112031. [Google Scholar] [CrossRef]
- Wong, A.C.-Y.; Lam, F. Study of selected thermal characteristics of polypropylene/polyethylene binary blends using DSC and TGA. Polym. Test. 2002, 21, 691–696. [Google Scholar] [CrossRef]
- Himmelblau, D.M. Process Analysis Statistical Methods; John Wiley & Sons, Inc.: New York, NY, USA, 1968. [Google Scholar] [CrossRef]
- Ciliz, N.K.; Ekinci, E.; Snape, C.E. Pyrolysis of virgin and waste polypropylene and its mixtures with waste polyethylene and polystyrene. Waste Manag. 2004, 24, 173–181. [Google Scholar] [CrossRef]
- Yao, Z.; Yu, S.; Su, W.; Wu, W.; Tang, J.; Qi, W. Kinetic studies on the pyrolysis of plastic waste using a combination of model-fitting and model-free methods. Waste Manag. Res. 2020, 38, 77–85. [Google Scholar] [CrossRef]
- Marcilla, A.; García-Quesada, J.C.; Sánchez, S.; Ruiz, R. Study of the catalytic pyrolysis behaviour of polyethylene–polypropylene mixtures. J. Anal. Appl. Pyrolysis 2005, 74, 387–392. [Google Scholar] [CrossRef]
- Hujuri, U.; Ghoshal, A.K.; Gumma, S. Modelling pyrolysis kinetics of plastic mixtures. Polym. Degrad. Stab. 2008, 93, 1832–1837. [Google Scholar] [CrossRef]
- Miranda, R.; Yang, J.; Roy, C.; Vasile, C. Vacuum pyrolysis of commingled plastics containing PVC I. Kinetic study. Polym. Degrad. Stab. 2001, 72, 469–491. [Google Scholar] [CrossRef]
| Plastic | Proximate Analysis, wt% | Ultimate Analysis, wt% | |||||
|---|---|---|---|---|---|---|---|
| Moisture | Volatile | Ash | C | H | N | S | |
| PP | 0.076 | 99.630 | 0.294 | 85.00 | 14.73 | 0.04 | 0.23 |
| PS | 0.235 | 99.590 | 0.175 | 90.47 | 9.43 | 0.00 | 0.08 |
| HDPE | 0.405 | 99.377 | 0.218 | 82.77 | 16.92 | 0.00 | 0.29 |
| LDPE | 0.199 | 99.653 | 0.148 | 83.00 | 16.75 | 0.00 | 0.25 |
| Test No. | Mass Fraction, % | Test No. | Mass Fraction, % | ||||||
|---|---|---|---|---|---|---|---|---|---|
| PS | LDPE | HDPE | PP | PS | LDPE | HDPE | PP | ||
| 1 | 70 | 0 | 30 | 0 | 10 | 0 | 30 | 0 | 70 |
| 2 | 70 | 0 | 0 | 30 | 11 | 0 | 0 | 30 | 70 |
| 3 | 70 | 30 | 0 | 0 | 12 | 30 | 0 | 0 | 70 |
| 4 | 30 | 70 | 0 | 0 | 13 | 33.3 | 0 | 33.3 | 33.3 |
| 5 | 0 | 70 | 0 | 30 | 14 | 48.4 | 19.2 | 32.5 | 0 |
| 6 | 0 | 70 | 30 | 0 | 15 | 48.4 | 34.9 | 0 | 16.7 |
| 7 | 0 | 30 | 70 | 0 | 16 | 0 | 15.5 | 55.0 | 29.5 |
| 8 | 0 | 0 | 70 | 30 | 17 | 25.0 | 25.0 | 25.0 | 25.0 |
| 9 | 30 | 0 | 70 | 0 | |||||
| Reaction Mechanism | f(α) | g(α) |
|---|---|---|
| First-order reaction (F1) | 1−α | −ln (1−α) |
| Second order reaction (F2) | (1−α)2 | [1/(1−α)] −1 |
| Third order reaction (F3) | (1−α)3 | {[1/(1−α)2] −1}/2 |
| One dimensional diffusion (D1) | 1/(2 α) | α2 |
| Two dimensional diffusion (D2) | 1/[−ln (1−α)] | (1−α) ln(1−α) + α |
| Three dimensional diffusion (D3) | 3/{2[1−(1−α)1/3]} | [1−(1−α)1/3]2 |
| Avrami–Erofeev (A2) | 2(1−α)[−ln(1−α)]1/2 | [−ln(1−α)]1/2 |
| Avrami–Erofeev (A3) | 3(1−α)[−ln(1−α)]2/3 | [−ln(1−α)]1/3 |
| Avrami–Erofeev (A4) | 4(1−α)[−ln(1−α)]3/4 | [−ln(1−α)]1/4 |
| Phase boundary—one dimension (R1) | 1 | α |
| Contracting cylinder (R2) | 2(1−α)1/2 | 1−(1−α)1/2 |
| Contracting sphere (R3) | 3(1−α)1/3 | 1−(1−α)1/3 |
| Power low (P2) | 2 α1/2 | α1/2 |
| Power low (P3) | 3 α2/3 | α1/3 |
| Power low (P4) | 4 α3/4 | α1/4 |
| Polymer Sample | Tonset (K) | Tpeak (K) | Tendset (K) | ∆T (K) |
|---|---|---|---|---|
| PS | 545 | 652 | 680 | 135 |
| PP | 583 | 696 | 710 | 127 |
| LDPE | 608 | 738 | 770 | 162 |
| HDPE | 614 | 757 | 790 | 176 |
| Test No. | Tpeak (K) | Composition (wt %) |
|---|---|---|
| 1 | 745 | PS/HDPE (70/30) |
| 2 | 738 | PS/PP (70/30) |
| 3 | 708 | PS/LDPE (70/30) |
| 4 | 738 | LDPE/PS (70/30) |
| 5 | 768 | LDPE/PP (70/30) |
| 6 | 768 | LDPE/HDPE (70/30) |
| 7 | 748 | HDPE/LDPE (70/30) |
| 8 | 753 | HDPE/PP (70/30) |
| 9 | 753 | HDPE/PS (70/30) |
| 10 | 723 | PP/LDPE (70/30) |
| 11 | 738 | PP/HDPE (70/30) |
| 12 | 753 | PP/PS (70/30) |
| 13 | 738 | PS/HDPE/PP (33.3/33.3/33.3) |
| 14 | 768 | PS/LDPE/HDPE (48.4/19.2/32.5) |
| 15 | 741 | PS/LDPE/PP (48.4/34.9/16.7) |
| 16 | 766 | LDPE/HDPE/PP (15.5/55/29.5) |
| 17 | 749 | PS/PP/LDPE/HDPE (25/25/25/25) |
| Test No. | Kinetic Parameters | Reaction Mechanism | ||
|---|---|---|---|---|
| E (kJ/mol) | ln (Ao) | R2 | ||
| 1 | 186 | 31.16 | 0.992 | First order reaction (F1) |
| 2 | 193 | 31.41 | 0.9991 | Contracting sphere (R3) |
| 3 | 104 | 16.92 | 0.9966 | Contracting cylinder (R2) |
| 4 | 216 | 35.79 | 0.9783 | First order reaction (F1) |
| 5 | 157 | 24.55 | 0.9943 | Contracting sphere (R3) |
| 6 | 260 | 41.56 | 0.9949 | First order reaction (F1) |
| 7 | 158 | 26.98 | 0.9818 | First order reaction (F1) |
| 8 | 174 | 28.4 | 0.9875 | First order reaction (F1) |
| 9 | 188 | 30.48 | 0.9993 | First order reaction (F1) |
| 10 | 181 | 30.53 | 0.9886 | First order reaction (F1) |
| 11 | 223 | 36.76 | 0.9895 | First order reaction (F1) |
| 12 | 179 | 29.4 | 0.992 | First order reaction (F1) |
| 13 | 146 | 23.54 | 0.9968 | Contracting sphere (R3) |
| 14 | 144 | 22.81 | 0.9951 | Contracting cylinder (R2) |
| 15 | 138 | 21.81 | 0.9944 | Contracting cylinder (R2) |
| 16 | 227 | 36.28 | 0.9957 | First order reaction (F1) |
| 17 | 158 | 24.78 | 0.9978 | Contracting sphere (R3) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dubdub, I.; Al-Yaari, M. Pyrolysis of Mixed Plastic Waste: I. Kinetic Study. Materials 2020, 13, 4912. https://doi.org/10.3390/ma13214912
Dubdub I, Al-Yaari M. Pyrolysis of Mixed Plastic Waste: I. Kinetic Study. Materials. 2020; 13(21):4912. https://doi.org/10.3390/ma13214912
Chicago/Turabian StyleDubdub, Ibrahim, and Mohammed Al-Yaari. 2020. "Pyrolysis of Mixed Plastic Waste: I. Kinetic Study" Materials 13, no. 21: 4912. https://doi.org/10.3390/ma13214912
APA StyleDubdub, I., & Al-Yaari, M. (2020). Pyrolysis of Mixed Plastic Waste: I. Kinetic Study. Materials, 13(21), 4912. https://doi.org/10.3390/ma13214912






