Laser Surface Alloying of Austenitic 316L Steel with Boron and Some Metallic Elements: Microstructure
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials and Specimens
2.2. Laser Surface Alloying of 316L Steel
- amorphous boron,
- mixture of amorphous boron and Stellite-6 powders with mass ratio 1:1,
- mixture of amorphous boron and nickel powders with mass ratio 1:1,
- mixture of amorphous boron and Ni–Cr powders with mass ratio 1:1.
2.3. Microstructure Observations
2.4. X-ray Microanalysis and Phase Analysis
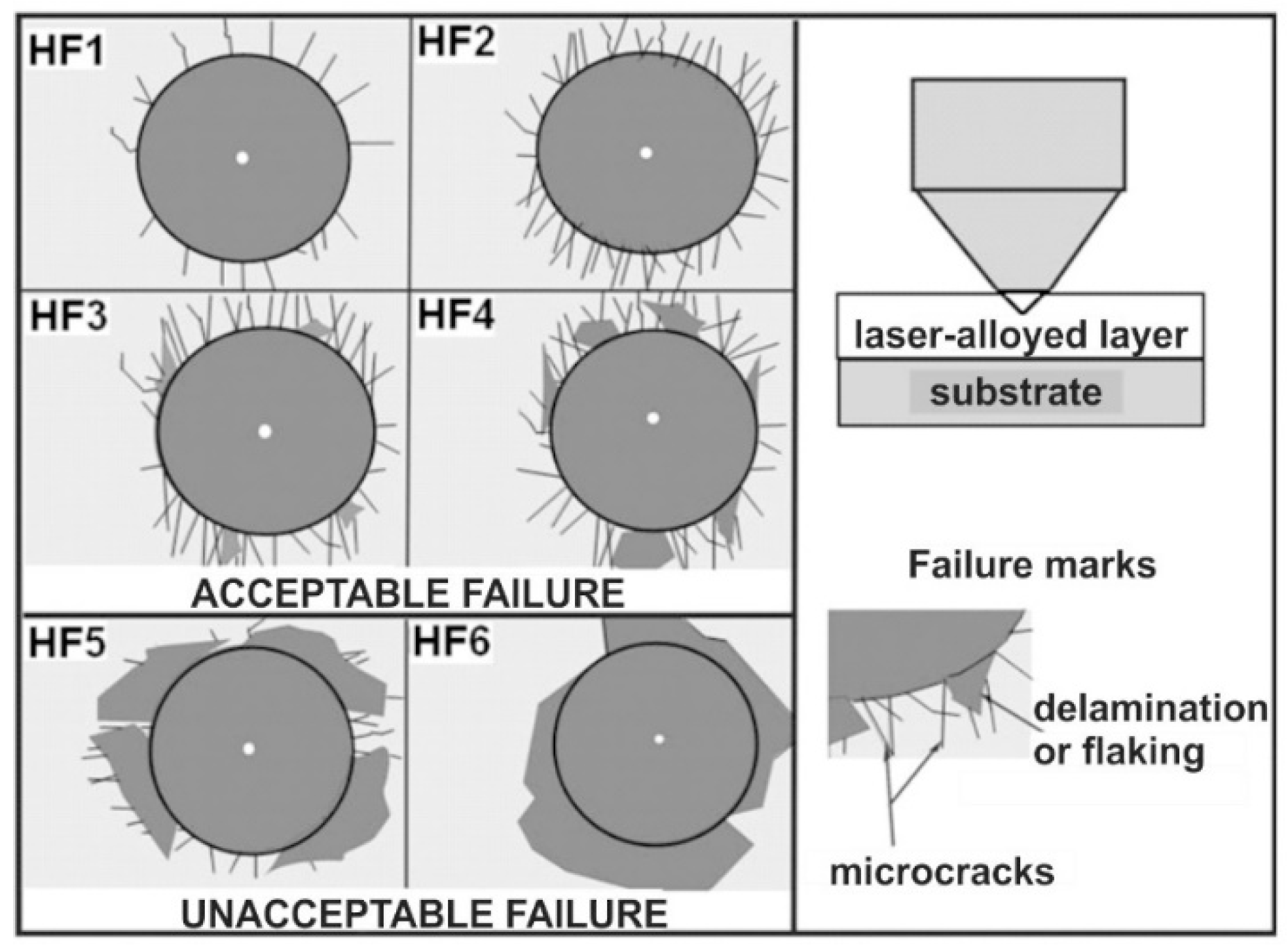
2.5. Cohesion
3. Results and Discussion
3.1. Selection of the Laser Surface Alloying Parameters
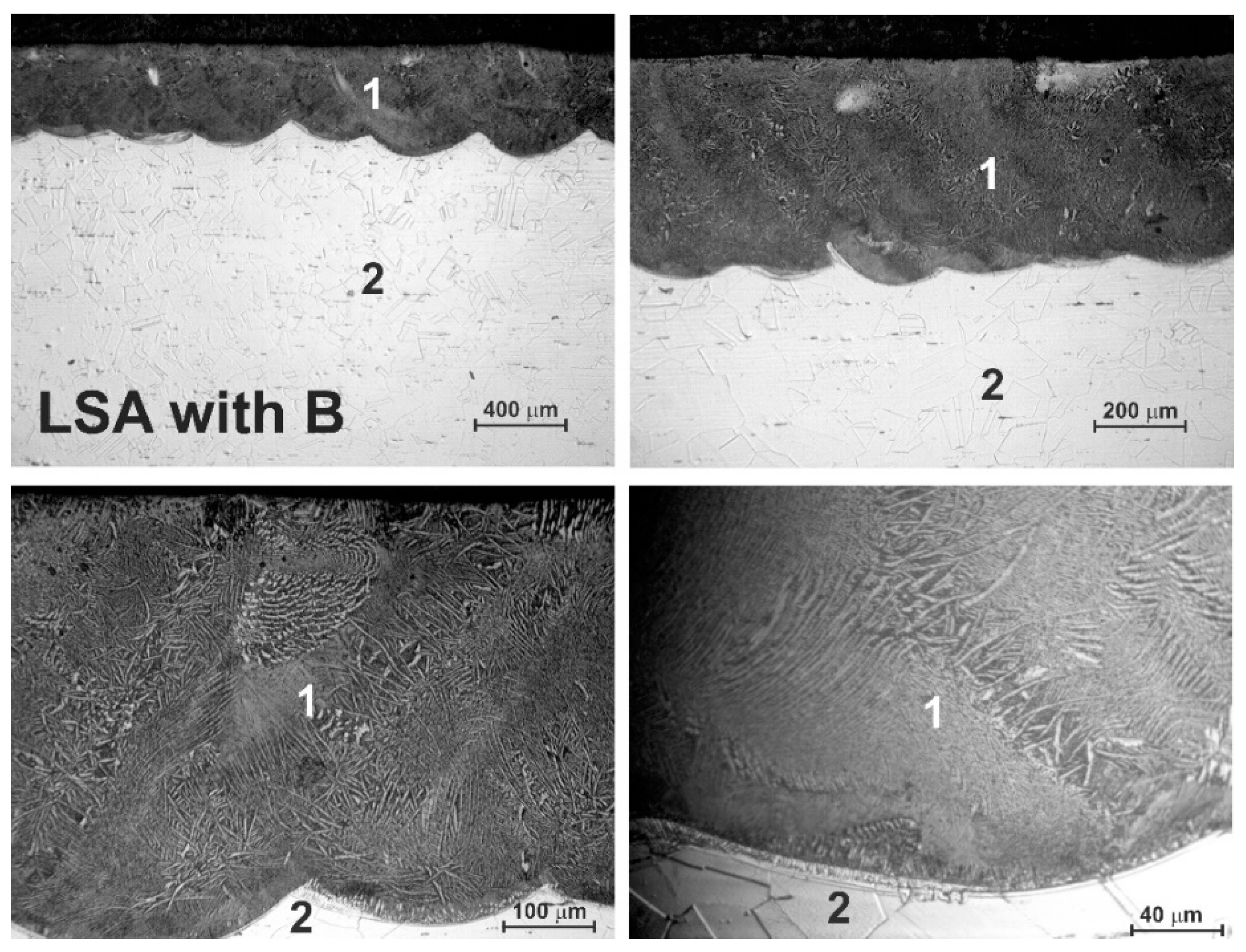
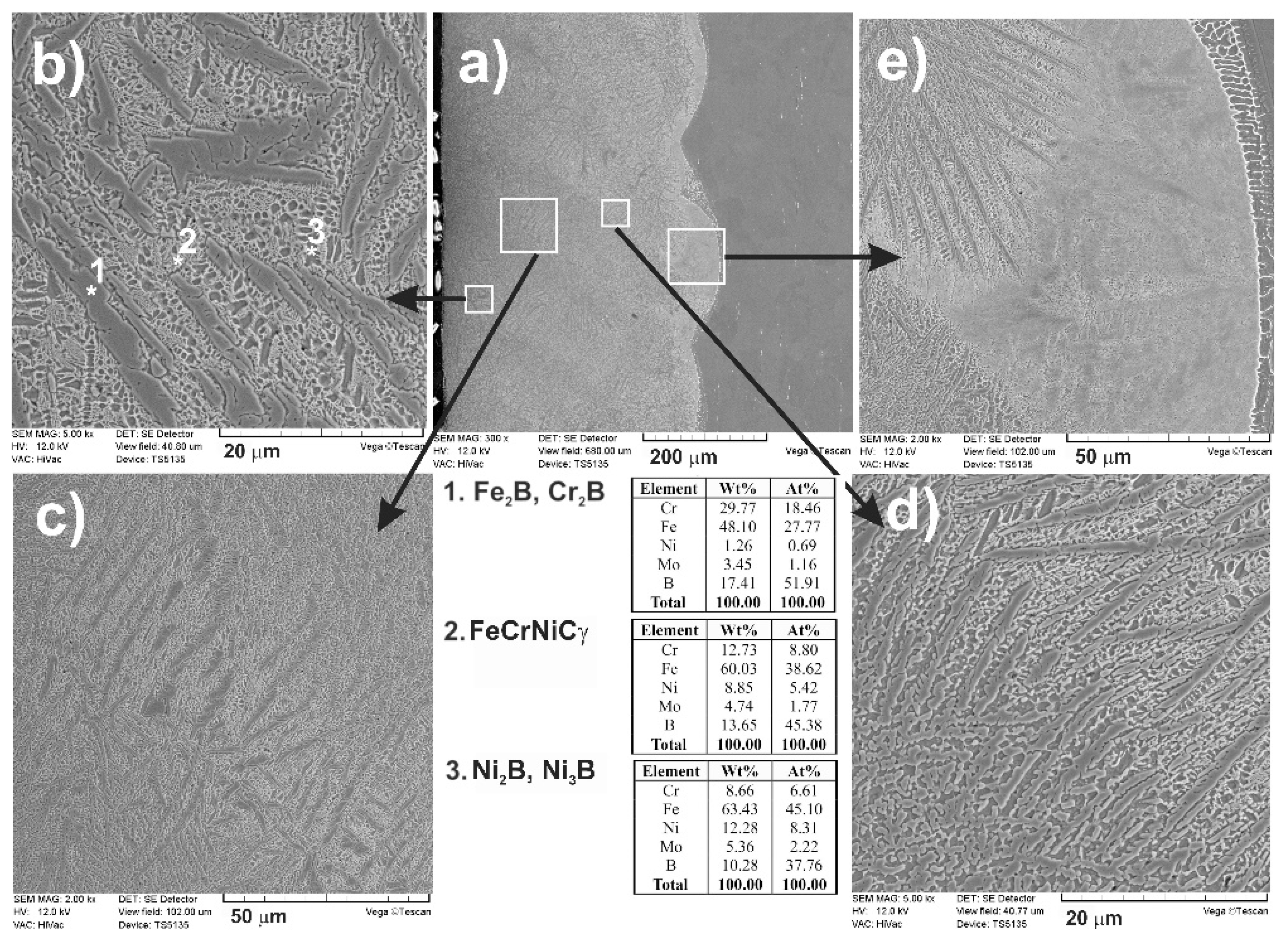
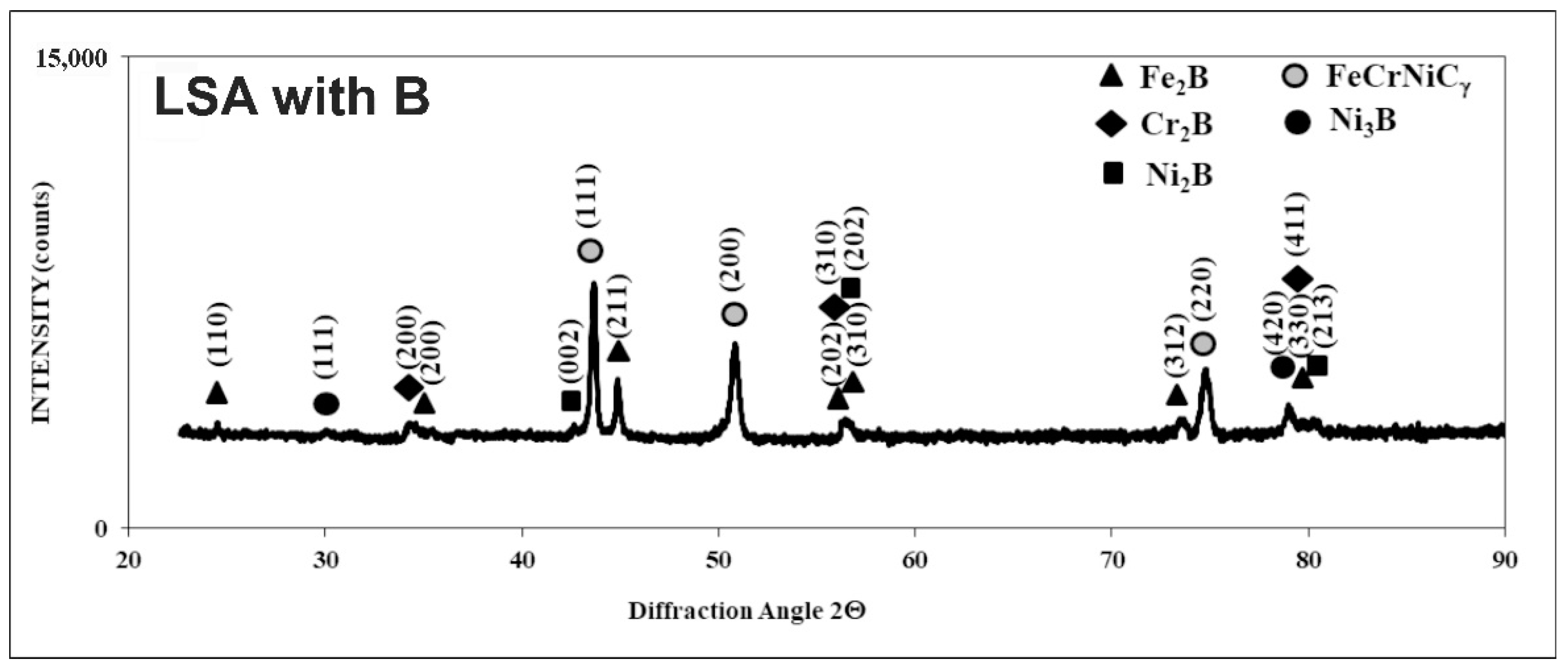
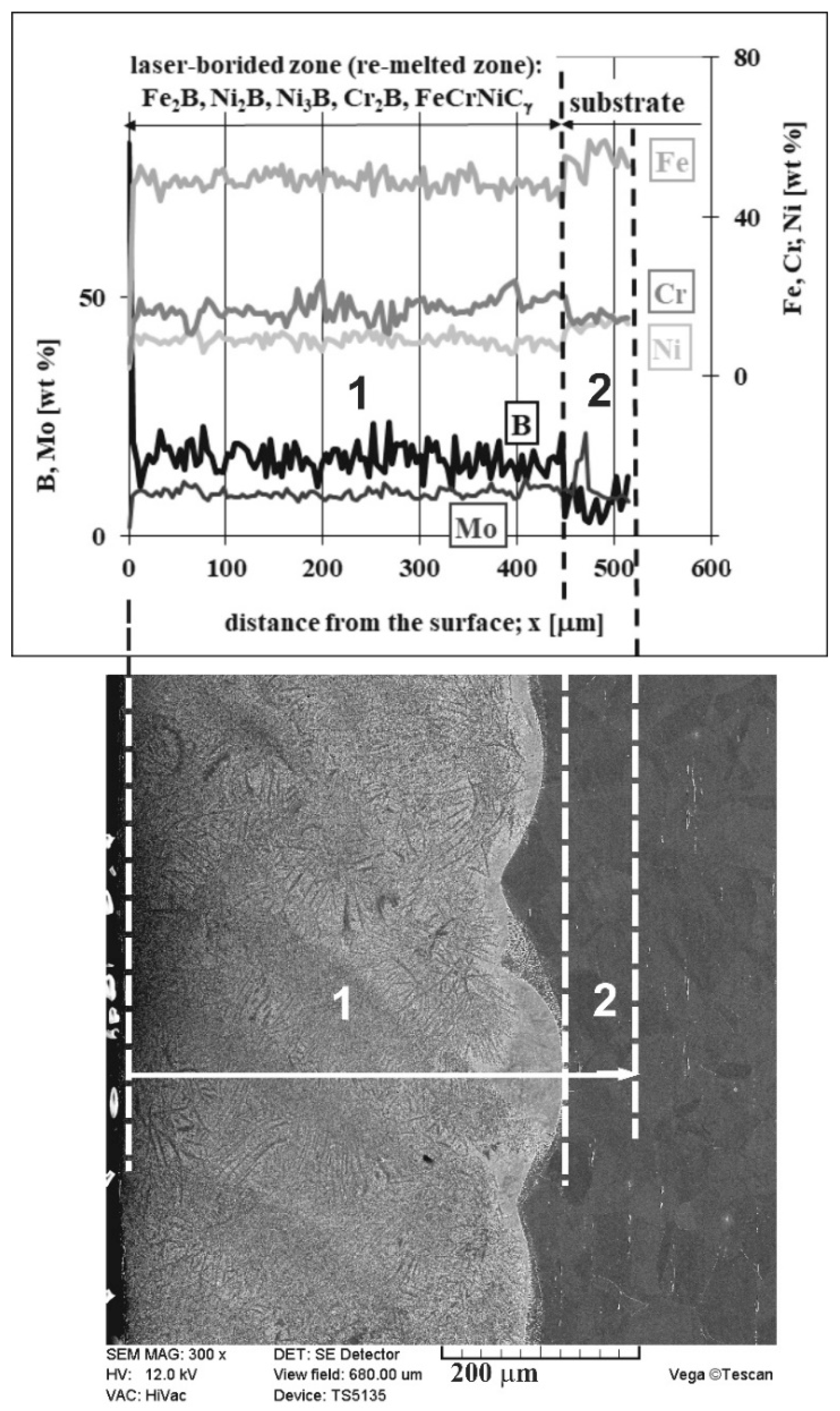
3.2. Microstructure of Laser-Alloyed Layer with Boron
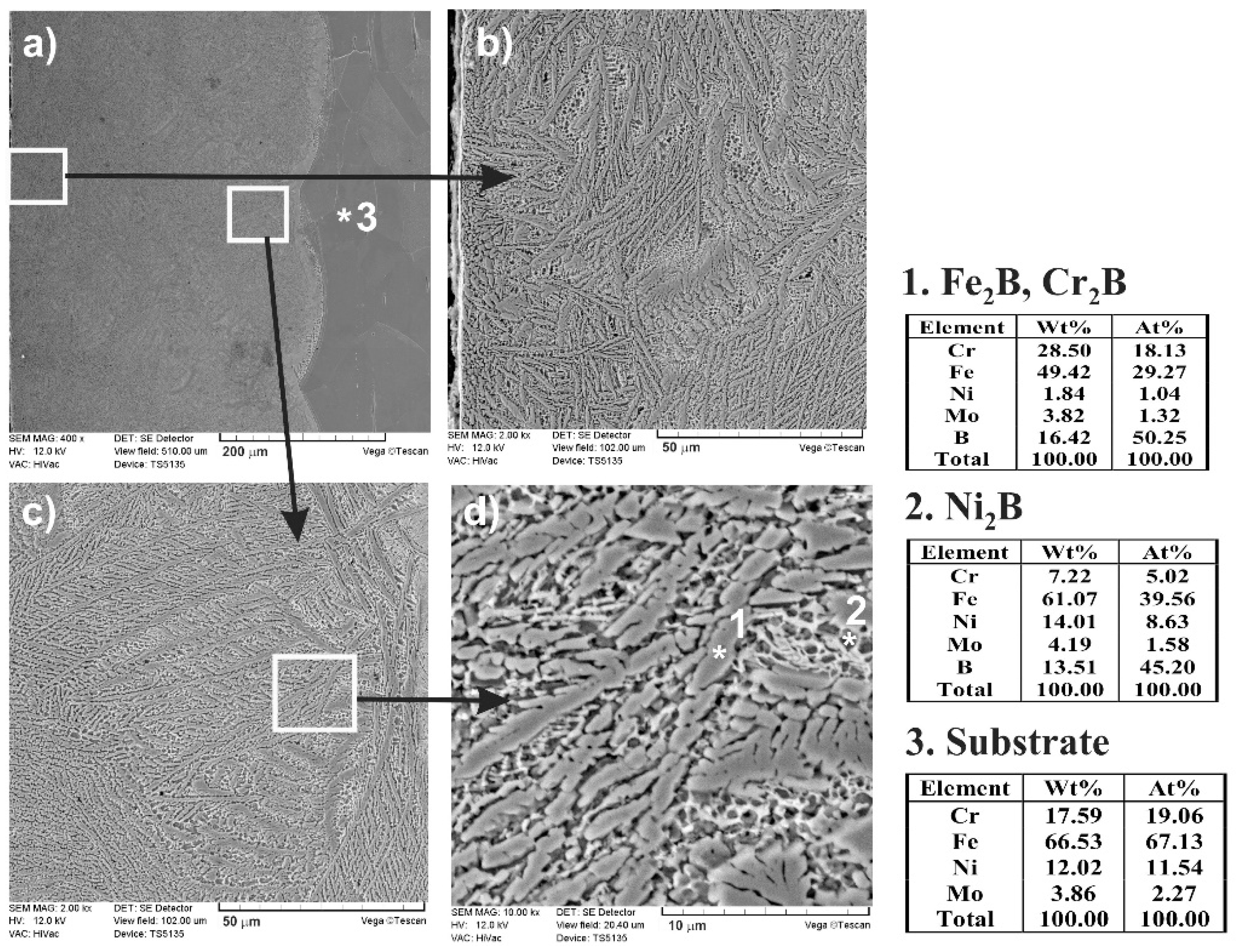
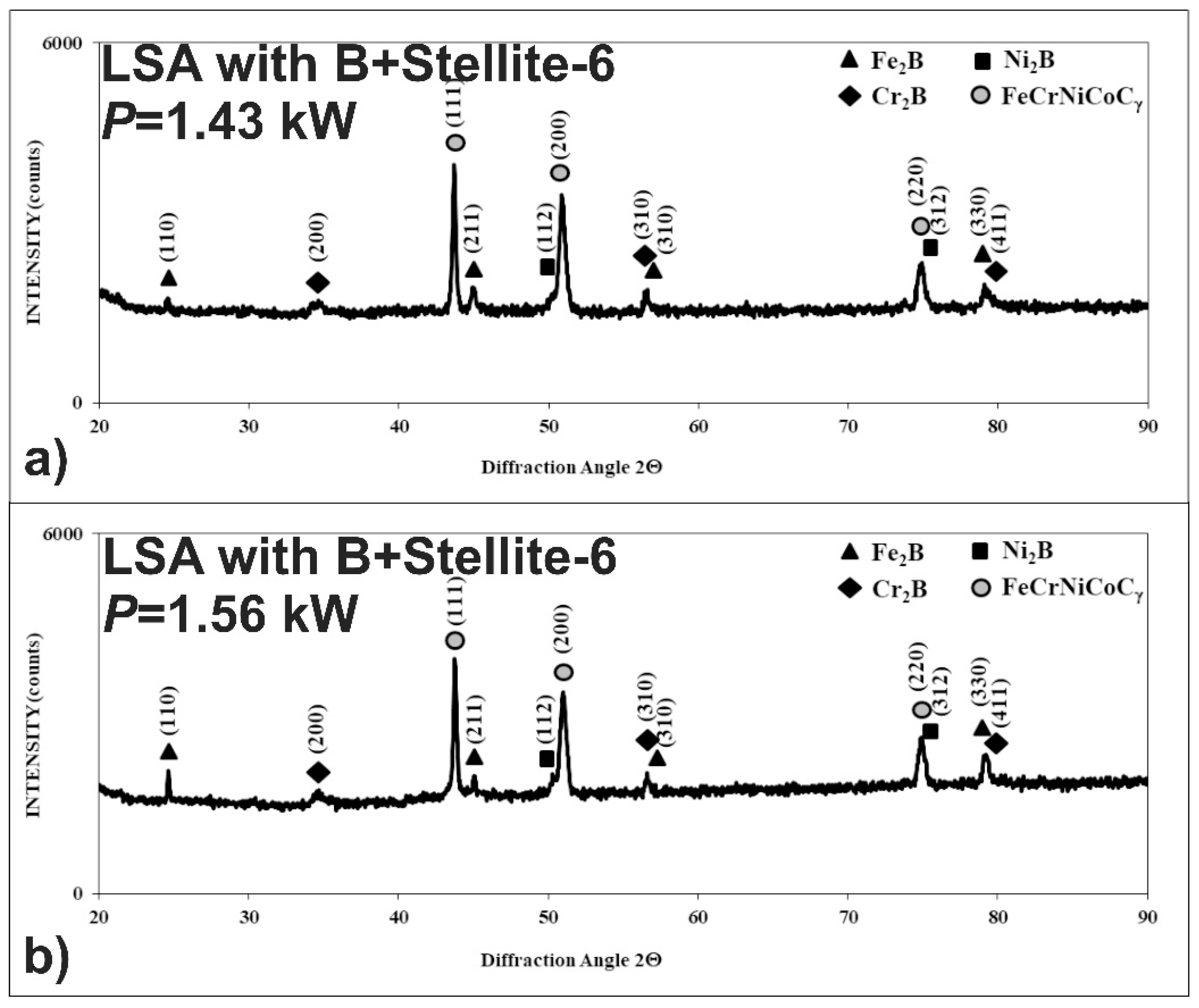
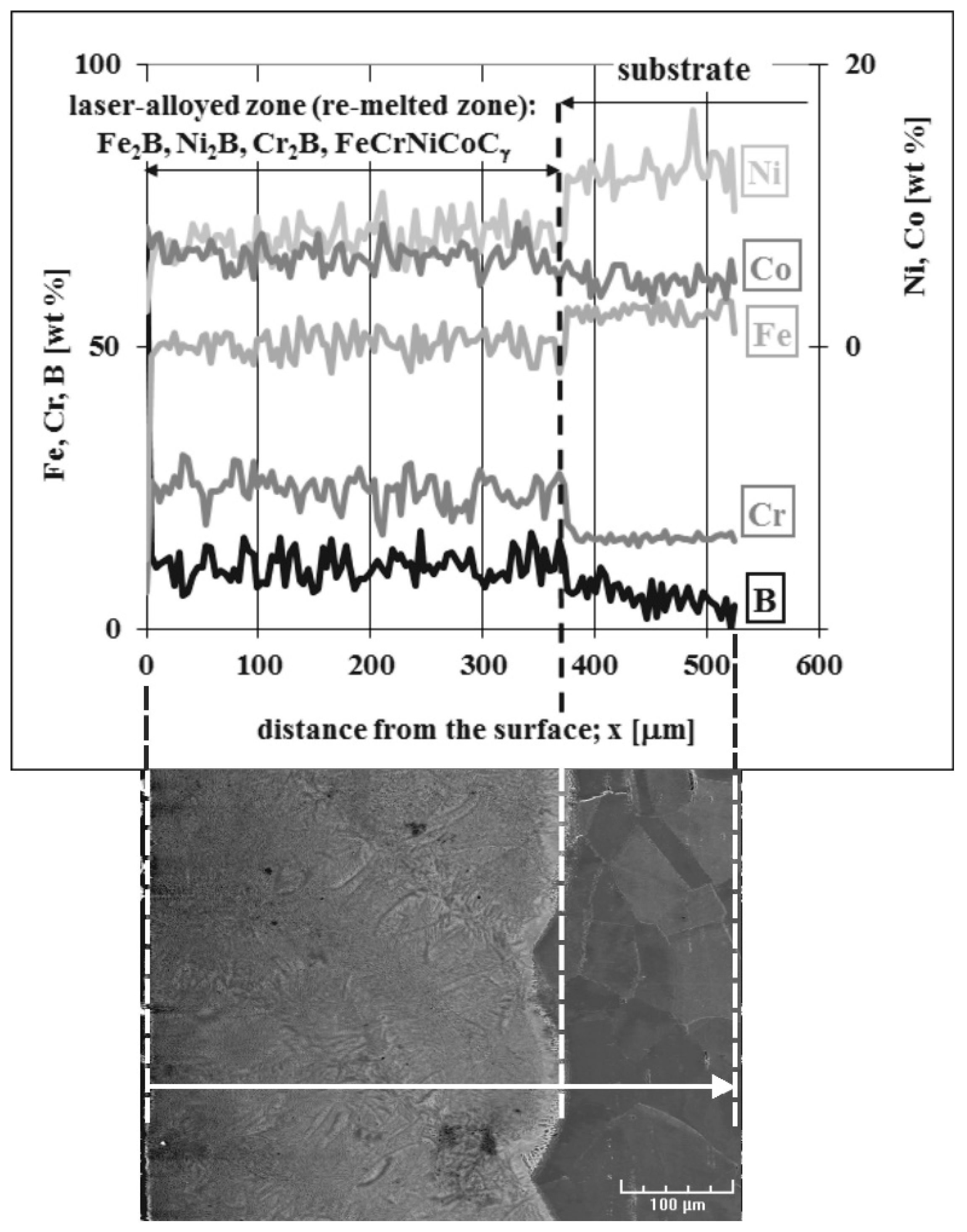
3.3. Microstructure of Laser-Alloyed Layer with Boron and Stellite-6
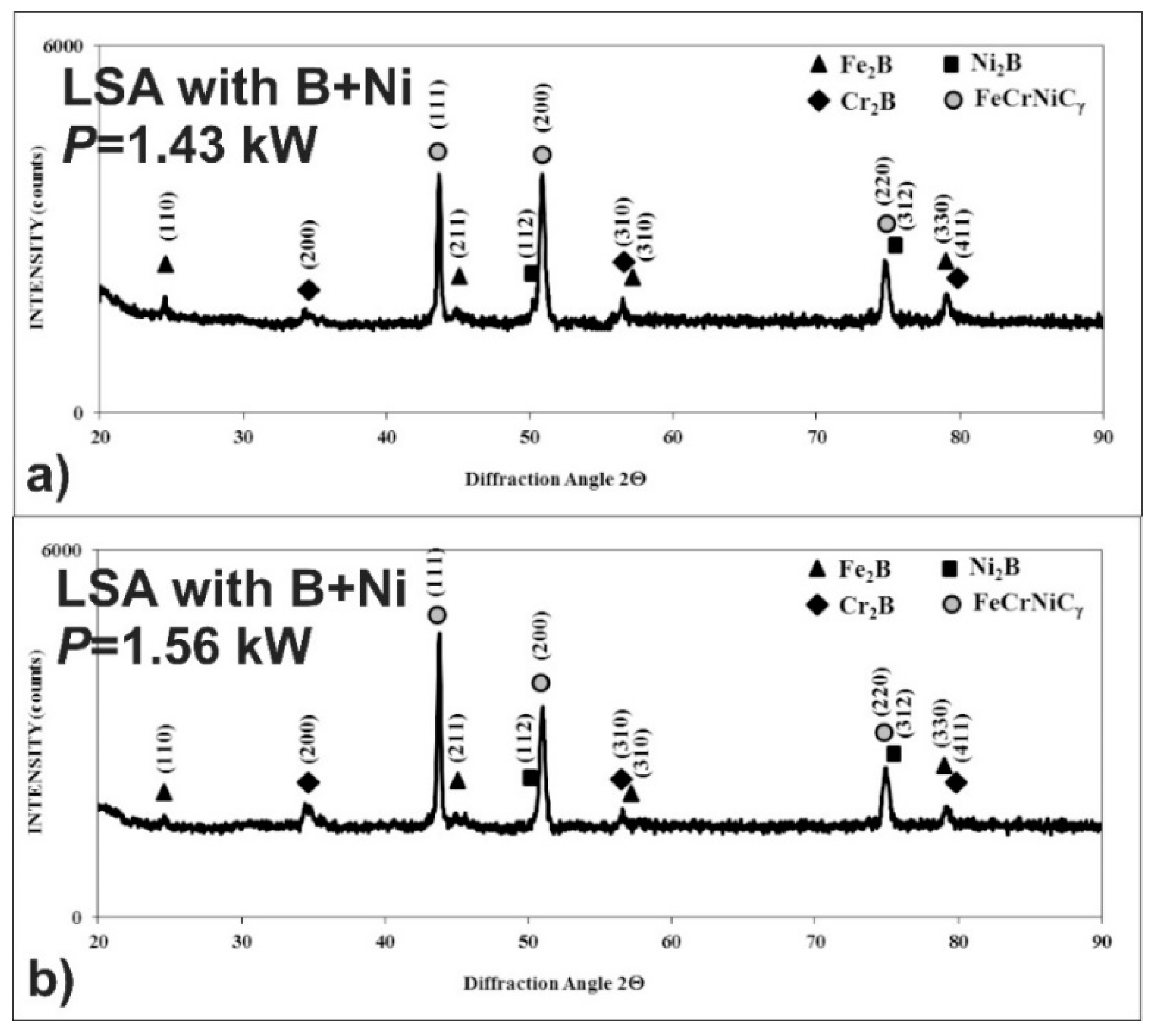
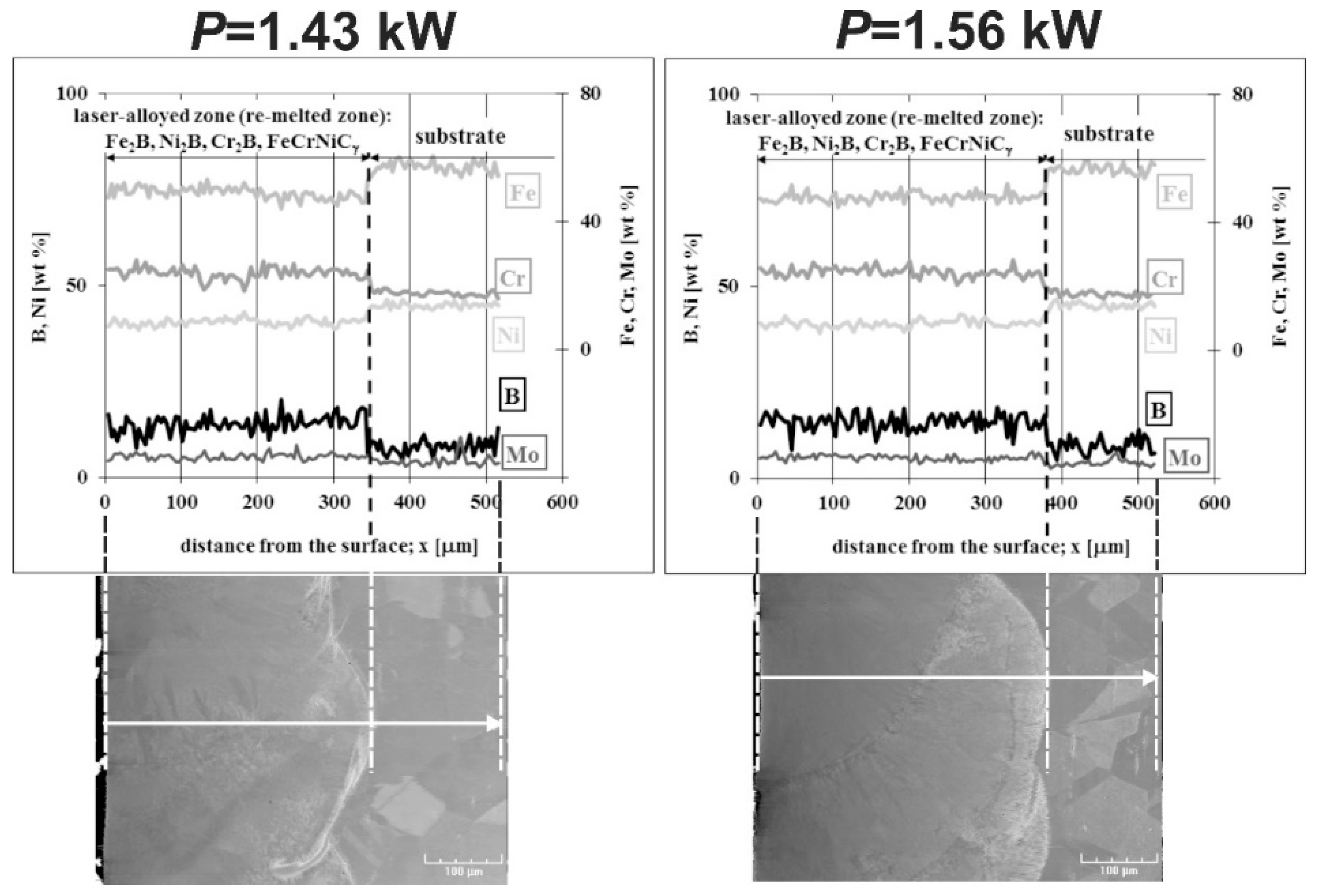
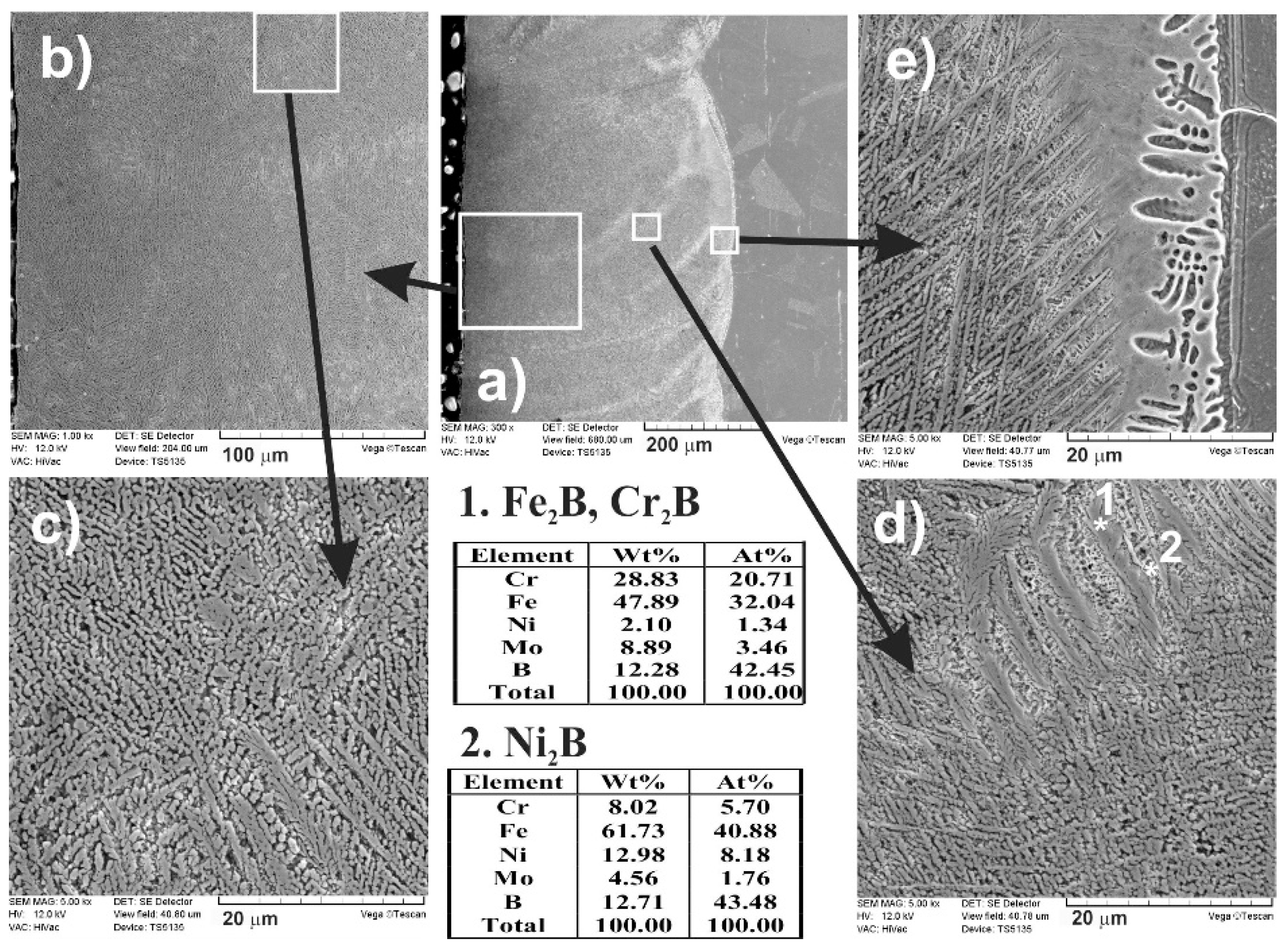
3.4. The Microstructure of Laser-Alloyed Layer with Boron and Nickel
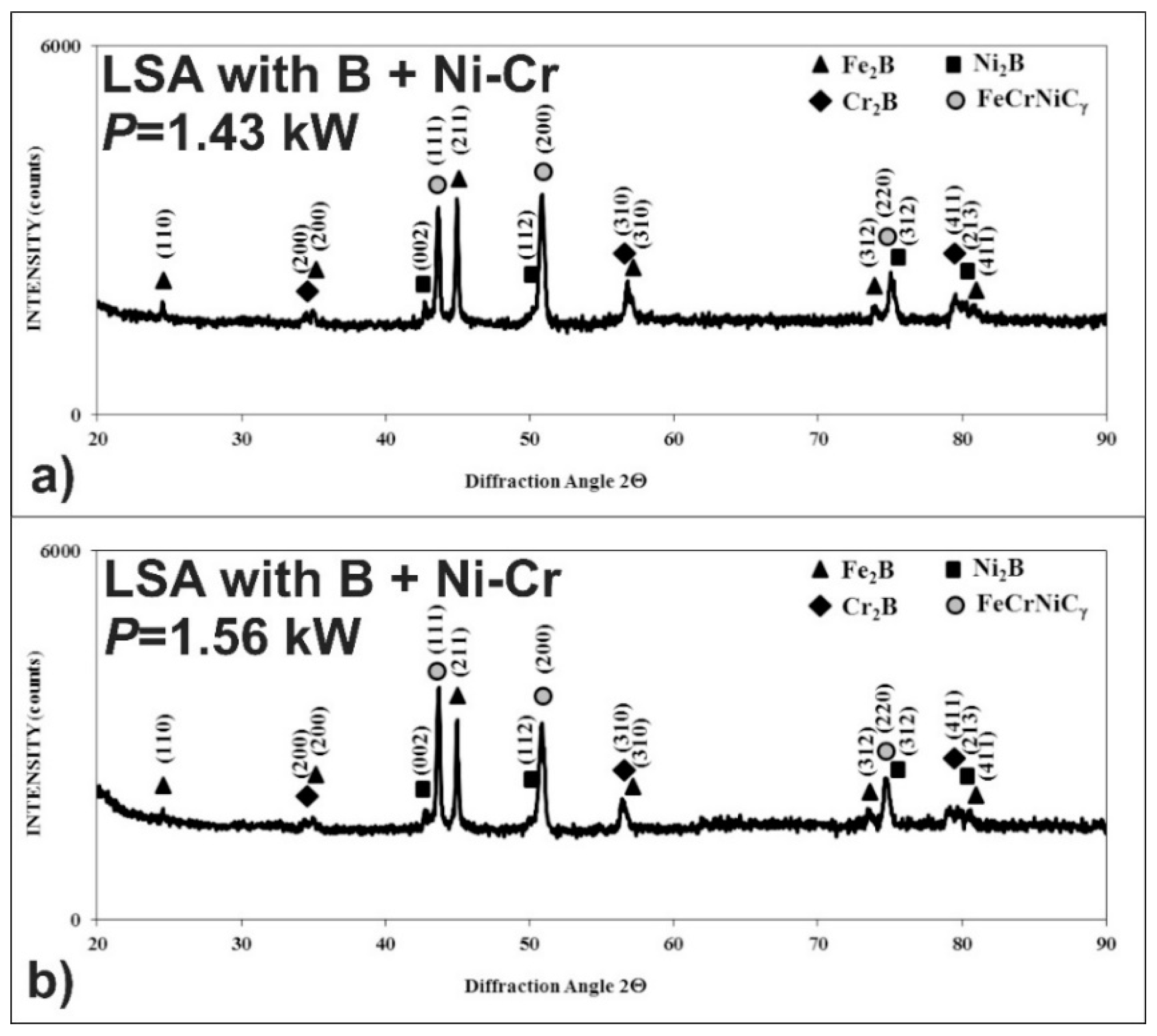
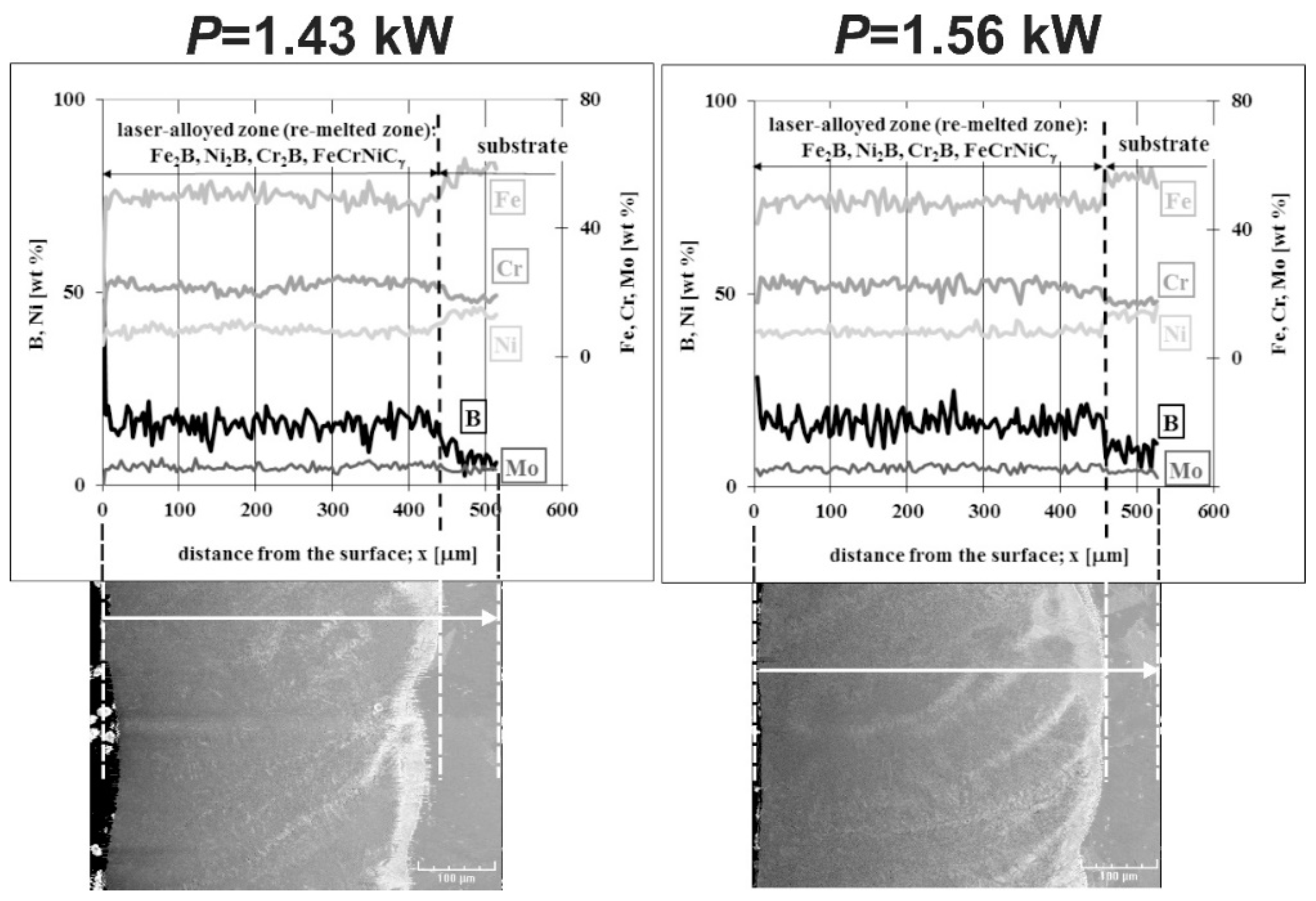
3.5. Microstructure of Laser-Alloyed Layer with Boron, Nickel and Chromium
3.6. Cohesion of Laser-Alloyed Layers
4. Conclusions
- Surface layers produced by laser alloying of 316L steel with boron or boron and selected metallic elements (Ni, Cr, Co) were characterized by a microstructure of acceptable quality, i.e., devoid of defects typical of laser processing (microcracks, gas pores),
- Obtaining a layer without defects required a dilution ratio (DR) of at least 0.37. All the laser-alloyed layers were characterized by the relatively high dilution ratio in the range of 0.41–0.54,
- All the laser-alloyed layers constituted the re-melted zone (MZ) only,
- The effects of laser irradiation weren’t observed in the heat-affected zone (HAZ) due to no possibility of changing the austenitic structure by martensite transformation during fast cooling. Hence, the microstructure of HAZ didn’t differ from the substrate material,
- All the re-melted zones produced were of a composite nature, i.e., the hard ceramic phases, such as Fe2B, Cr2B, Ni2B, or Ni3B borides, occurred in the austenitic matrix,
- The use of powder mixtures of boron and selected metallic elements as alloying materials, instead of only boron powder, made it possible to reduce the laser beam power needed to produce the laser-alloyed layers of acceptable quality,
- The uniform thickness of each laser-alloyed layer, i.e., depth of re-melted zone, was achieved because of the relatively high overlapping (86%),
- The thickness of the laser-alloyed layers ranged from 308 to 432 μm and was much greater than the thickness of the surface layers produced on austenitic 316L steel by diffusion techniques of boriding,
- All the laser-alloyed layers, produced in austenitic 316L steel, were characterized by excellent cohesion,
- The LSA process with boron and the selected metallic elements was energy- and material-saving as well as environmentally friendly compared to the diffusion processes of boriding,
- The usefulness of the proposed laser surface alloying to increase hardness and wear resistance of 316L steel without sacrificing its corrosion resistance would be confirmed in the next paper.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Glaeser, W.A. Steels. In Materials for Tribology, 1st ed.; Tribology Series; Elsevier: New York, NY, USA, 1992; Volume 20, pp. 1–260. [Google Scholar]
- Skołek-Stefaniszyn, E.; Kaminski, J.; Sobczak, J.; Wierzchoń, T. Modifying the properties of AISI 316L steel by glow discharge assisted low-temperature nitriding and oxynitriding. Vacuum 2010, 85, 164–169. [Google Scholar] [CrossRef]
- Skołek-Stefaniszyn, E.; Burdynska, S.; Mroz, W.; Wierzchoń, T. Structure and wear resistance of the composite layers produced by glow discharge nitriding and PLD method on AISI 316L austenitic stainless steel. Vacuum 2009, 83, 1442–1447. [Google Scholar] [CrossRef]
- Olzon-Dionysio, M.; Olzon-Dionysio, D.; Campos, M.; Takemitsu Shigeyosi, W.; de Souza, S.D.; de Souza, S. Corrosion resistance of AISI 316L plasma nitride at different temperatures and times. Hyperfine Interact. 2019, 240, 26. [Google Scholar] [CrossRef]
- De Araújo Junior, E.; Bandeira, M.; Manfrinatoa, M.D.; Moreto, J.A.; Borges, R.; dos Santos Valese, S.; Suzuki, P.A.; Rossino, L.S. Effect of ionic plasma nitriding process on the corrosion and micro-abrasive wear behavior of AISI 316L austenitic and AISI 470 super-ferritic stainless steels. J. Mater. Res. Technol. 2019, 8, 2180–2191. [Google Scholar]
- De Las Heras, E.; Ybarra, G.; Lamas, D.; Cabo, A.; Dalibon, E.L.; Brühl, S.P. Plasma nitriding of 316L stainless steel in two different N2-H2 atmospheres—Influence on microstructure and corrosion resistance. Surf. Coat. Technol. 2017, 313, 47–54. [Google Scholar] [CrossRef]
- Borowski, T.; Kulikowski, K.; Adamczyk-Cieślak, B.; Rożniatowski, K.; Spychalski, M.; Tarnowski, M. Influence of nitrided and nitrocarburised layers on the functional properties of nitrogen-doped soft carbon-based coatings deposited on 316L steel under DC glow-discharge conditions. Surf. Coat. Technol. 2020, 392, 125705. [Google Scholar] [CrossRef]
- Campos, M.; de Souza, S.; Davim, J.P.; de Souza, S.D.; Olzon-Dionysio, M. Influence of the gas pressure of plasma nitriding on the structural, mechanical and tribological surface properties of AISI 316L. Mater. Res. 2019, 22, e20190302. [Google Scholar] [CrossRef]
- Godec, M.; Donik, Č.; Kocijan, A.; Podgornik, B.; Skobir Balantič, D.A. Effect of post-treated low-temperature plasma nitriding on the wear and corrosion resistance of 316L stainless steel manufactured by laser powder bed fusion. Addit. Manuf. 2020, 32, 101000. [Google Scholar] [CrossRef]
- Frączek, T.; Olejnik, M.; Jasiński, J.; Skuza, Z. Short-term low-temperature glow discharge nitriding of 316L austenitic steel. Metalurgija 2011, 50, 151–154. [Google Scholar]
- Frączek, T.; Ogórek, M.; Skuza, Z.; Prusak, R. Mechanism of ion nitriding of 316L austenitic steel by active screen method in a hydrogen-nitrogen atmosphere. Int. J. Adv. Manuf. Technol. 2020, 109, 1357–1368. [Google Scholar] [CrossRef]
- Nishimoto, A.; Fukube, T.; Tanaka, T. Effect of surface deposits on nitriding layer formation of active screen plasma nitriding. Mater. Trans. 2016, 57, 1811–1815. [Google Scholar] [CrossRef]
- Lin, K.; Li, X.; Dong, H.; Guo, P.; Gu, D. Nitrogen mass transfer and surface layer formation during the active screen plasma nitriding of austenitic stainless steels. Vacuum 2018, 148, 224–229. [Google Scholar] [CrossRef]
- Li, Y.; Wang, Z.; Wang, L. Surface properties of nitrided layer on AISI 316L austenitic stainless steel produced by high temperature plasma nitriding in short time. Appl. Surf. Sci. 2014, 298, 243–250. [Google Scholar] [CrossRef]
- Diaz-Guillen, J.C.; Naeem, M.; Acevedo-Davila, J.L.; Hdz-Garcia, H.M.; Iqbal, J.; Khan, M.A.; Mayen, J. Improved Mechanical Properties, Wear and Corrosion Resistance of 316L Steel by Homogeneous Chromium Nitride Layer Synthesis Using Plasma Nitriding. J. Mater. Eng. Perform. 2020, 29, 877–889. [Google Scholar] [CrossRef]
- Zhang, T.; Wu, J.; Jin, L.; Zhang, Z.; Rong, W.; Zhang, B.; Wang, Y.; He, Y.; Liu, W.; Li, X. Enhancing the mechanical and anticorrosion properties of 316L stainless steel via a cathodic plasma electrolytic nitriding treatment with added PEG. J. Mater. Sci. Technol. 2019, 35, 2630–2637. [Google Scholar] [CrossRef]
- Biehler, J.; Hoche, H.; Oechsner, M. Corrosion properties of polished and shot-peened austenitic stainless steel 304L and 316L with and without plasma nitriding. Surf. Coat. Technol. 2017, 313, 40–46. [Google Scholar] [CrossRef]
- Jayalakshmi, M.; Huilgol, P.; Ramachandra Bhat, B.; Udaya Bhat, K. Microstructural characterization of low temperature plasma-nitrided 316L stainless steel surface with prior severe shot peening. Mater. Des. 2016, 108, 448–454. [Google Scholar] [CrossRef]
- Rabelo Menezes, M.; Godoy, C.; Buono, V.T.L.; Schvartzman, M.M.M.; Wilson, J.C. Effect of shot peening and treatment temperature on wear and corrosion resistance of sequentially plasma treated AISI 316L steel. Surf. Coat. Technol. 2017, 309, 651–662. [Google Scholar] [CrossRef]
- Kovaci, H.; Seçer, Y. Improved tribological performance of AISI 316L stainless steel by a combined surface treatment: Surface texturing by selective laser melting and plasma nitriding. Surf. Coat. Technol. 2020, 400, 126178. [Google Scholar] [CrossRef]
- Adachi, S.; Ueda, N. Wear and corrosion properties of cold-sprayed aisi 316l coatings treated by combined plasma carburizing and nitriding at low temperature. Coatings 2018, 8, 456. [Google Scholar] [CrossRef]
- Adachi, S.; Egawa, M.; Yamaguchi, T.; Ueda, N. Low-temperature plasma nitriding for austenitic stainless steel layers with various nickel contents fabricated via direct laser metal deposition. Coatings 2020, 10, 365. [Google Scholar] [CrossRef]
- Li, Y.; Zhu, Y.; Ye, Q.; Zhang, S.; Zhao, J.; He, Y. Effect of hybrid surface treatment composed of plasma nitriding and W-Cr-Ti-Al-N coating on tribological behavior of AISI 316L steel. Tribol. Online 2018, 13, 316–319. [Google Scholar] [CrossRef]
- Ye, Q.W.; Li, Y.; Zhang, M.Y.; Zhang, S.Z.; Bi, Y.J.; Gao, X.P.; He, Y.Y. Electrochemical behavior of (Cr, W, Al, Ti, Si) N multilayer coating on nitrided AISI 316L steel in natural seawater. Ceram. Int. 2020, 46, 22404–22418. [Google Scholar] [CrossRef]
- Sun, Y.; Li, X.; Bell, T. Structural characteristics of low temperature plasma carburised austenitic stainless steel. Mater. Sci. Technol. 1999, 15, 1171–1178. [Google Scholar] [CrossRef]
- García Molleja, J.; Nosei, L.; Ferrón, J.; Bemporad, E.; Lesage, J.; Chicot, D.; Feugeas, J. Characterization of expanded austenite developed on AISI 316L stainless steel by plasma carburization. Surf. Coat. Technol. 2010, 204, 3750–3759. [Google Scholar] [CrossRef]
- Sun, Y. Tribocorrosion behavior of low temperature plasma carburized stainless steel. Surf. Coat. Technol. 2013, 228, 342–348. [Google Scholar] [CrossRef]
- Gobbi, S.J.; Gobbi, V.J.; Reinke, G. Improvement of mechanical properties and corrosion resistance of 316L and 304 stainless steel by low temperature plasma cementation. Rev. Mater. 2020, 25, 1–10. [Google Scholar] [CrossRef]
- Ballinger, J.; Catledge, S.A. Metal-boride interlayers for chemical vapor deposited nanostructured NSD films on 316 and 440C stainless steel. Surf. Coat. Technol. 2015, 261, 244–252. [Google Scholar] [CrossRef]
- Chegroune, R.; Keddam, M.; Abdellah, Z.N.; Ulker, S.; Taktak, S.; Günes, I. Characterization and kinetics of plasma-paste-borided AISI 316 steel. Mater. Tehnol. 2016, 50, 263–268. [Google Scholar] [CrossRef]
- Keddam, M.; Chegroune, R.; Kulka, M.; Makuch, N.; Panfil, D.; Siwak, P.; Taktak, S. Characterization, tribological and mechanical properties of plasma paste borided AISI 316 steel. Trans. Indian Inst. Met. 2018, 71, 79–90. [Google Scholar] [CrossRef]
- Kulka, M. Current Trends in Boriding: Techniques; Engineerings Materials Series; Springer International Publishing: Cham, Switzerland, 2019; pp. 1–282. ISBN 978-3-030-06781-6. [Google Scholar]
- Ozdemir, O.; Omar, M.A.; Usta, M.; Zeytin, S.; Bindal, C.; Ucisik, A.H. An investigation on boriding kinetics of AISI 316 stainless steel. Vacuum 2009, 83, 175–179. [Google Scholar] [CrossRef]
- Balusamy, T.; Sankara Narayanan, T.S.N.; Ravichandran, K.; Park, I.S.; Lee, M.H. Effect of surface mechanical attrition treatment (SMAT) on pack boronizing of AISI 304 stainless steel. Surf. Coat. Technol. 2013, 232, 60–67. [Google Scholar] [CrossRef]
- Kayali, Y.; Büyüksagis, A.; Yalçin, Y. Corrosion and wear behaviors of boronized AISI 316L stainless steel. Met. Mater. Int. 2013, 19, 1053–1061. [Google Scholar] [CrossRef]
- Kayali, Y.; Büyüksagis, A.; Günes, I.; Yalçin, Y. Investigation of corrosion behaviors at different solutions of boronized AISI 316L stainless steel. Prot. Met. Phys. Chem. Surf. 2013, 49, 348–358. [Google Scholar] [CrossRef]
- Campos-Silva, I.; Bernabé -Molina, S.; Brávo-Barcenas, D.; Martínez-Trinidad, J.; Rodríguez-Castro, G.; Meneses-Amador, A. Improving the adhesion resistance of the boride coatings to AISI 316L steel substrate by diffusion annealing. J. Mater. Eng. Perform. 2016, 25, 3852–3862. [Google Scholar] [CrossRef]
- García-Léon, R.A.; Martínez-Trinidad, J.; Campos-Silva, I.; Wong-Angel, W. Mechanical characterization of the AISI 316L alloy exposed to boriding process. DYNA 2020, 87, 34–41. [Google Scholar] [CrossRef]
- Reséndiz-Calderon, C.D.; Rodríguez-Castro, G.A.; Meneses-Amador, A.; Campos-Silva, I.E.; Andraca-Adame, J.; Palomar-Pardavé, M.E.; Gallardo-Hernández, E.A. Micro-abrasion wear resistance of borided 316L stainless steel and AISI 1018 steel. J. Mater. Eng. Perform. 2017, 26, 5599–5609. [Google Scholar] [CrossRef]
- Hernández-Sánchez, E.; Velázquez, J.C.; Castrejón-Flores, J.L.; Chino-Ulloa, A.; Torres Avila, I.P.; Carrera-Espinoza, R.; Yescas-Hernández, J.A.; Orozco-Alvarez, C. Tribological behavior of borided AISI 316L steel with reduced friction coefficient and enhanced wear resistance. Mater. Trans. 2019, 60, 156–164. [Google Scholar] [CrossRef]
- Kheyrodin, M.; Habibolahzadeh, A.; Babak Mousavi, S. Wear and corrosion behaviors of duplex surface treated 316L austenitic stainless steel via combination of boriding and chromizing. Prot. Met. Phys. Chem. Surf. 2017, 53, 105–111. [Google Scholar] [CrossRef]
- Ozbek, I.; Konduk, B.A.; Bindal, C.; Ucisik, A.H. Characterization of borided AISI 316L stainless steel implant. Vacuum 2002, 65, 521–525. [Google Scholar] [CrossRef]
- Mebarek, B.; Keddam, M. Prediction model for studying the growth kinetics of fe2b boride layers during boronizing. Ing. Syst. d’Information 2019, 24, 201–205. [Google Scholar] [CrossRef]
- Haruman, E.; Sun, Y.; Adenan, M.S. A comparative study of the tribocorrosion behaviour of low temperature nitrided austenitic and duplex stainless steels in NaCl solution. Tribol. Int. 2020, 151, 106412. [Google Scholar] [CrossRef]
- Kutschmann, P.; Lindner, T.; Börner, K.; Reese, U.; Lampke, T. Effect of Adjusted Gas Nitriding Parameters on Microstructure and Wear Resistance of HVOF-Sprayed AISI 316L Coatings. Materials 2019, 12, 1760. [Google Scholar] [CrossRef] [PubMed]
- Boes, J.; Röttger, A.; Becker, L.; Theisen, W. Processing of gas-nitrided AISI 316L steel powder by laser powder bed fusion—Microstructure and properties. Addit. Manuf. 2019, 30, 100836. [Google Scholar] [CrossRef]
- Hussain, P.; Mahmoud, H.; Basha, S.N.; Mohamad, A.I. Correlation between microstructure and micro-hardness of 316L nitrided austenitic stainless steel. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2020; Volume 863, p. 012025. [Google Scholar]
- Kim, S.G.; Kim, J.N.; Wang, J.P.; Kang, C.Y. Microstructure and nanosize precipitate of nitrided 316L stainless steel. Met. Mater. Int. 2019, 25, 127–134. [Google Scholar] [CrossRef]
- Fernández-Valdés, D.; Meneses-Amador, A.; Rodríguez-Castro, G.A.; Arzate-Vázquez, I.; Campos-Silva, I.; Nava-Sánchez, J.L. Standing contact fatigue behavior of nitrided AISI 316L steels. Surf. Coat. Technol. 2019, 377, 124871. [Google Scholar] [CrossRef]
- Istiroyah; Pamungkas, M.A.; Saroja, G.; Ghufron, M.; Juwono, A.M. Characteristic of Low Temperature Carburized Austenitic Stainless Steel. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2018; Volume 299, p. 012048. [Google Scholar]
- Istiroyah; Septi, D.W. The Effect of Quenching Media on Hardness and Carbon Content in Carburized Steel. In IOP Conference Series: Materials Science and Engineering; IOP Publishing: Bristol, UK, 2019; Volume 546, p. 042014. [Google Scholar]
- Ceschini, L.; Chiavari, C.; Lanzoni, E.; Martini, C. Low-temperature carburised AISI 316L austenitic stainless steel: Wear and corrosion behavior. Mater. Des. 2012, 38, 154–160. [Google Scholar] [CrossRef]
- Hsu, C.H.; Huang, K.H.; Lin, M.R. Annealing effect on tribological property of arc-deposited TiN film on 316L austenitic stainless steel. Surf. Coat. Technol. 2014, 259, 167–171. [Google Scholar] [CrossRef]
- Zhang, L.; Yang, H.; Pang, X.; Gao, K.; Tran, H.T.; Volinsky, A.A. TiN-coating effects on stainless steel tribological behavior under dry and lubricated conditions. J. Mater. Eng. Perform. 2014, 23, 1263–1269. [Google Scholar] [CrossRef][Green Version]
- Major, B. Laser processing for surface modification by remelting and alloying of metallic systems. In Materials Surface Processing by Directed Energy Techniques; Paleau, Y., Ed.; Elsevier: New York, NY, USA, 2006. [Google Scholar]
- Goły, M.; Kusiński, J. Microstructure and properties of the laser treated 30CrMnMo16–8 chromium steel. In Problems of Modern Techniques in Aspect of Engineering and Education; Paweł, K., Ed.; Monography Cracow Institute of Technology: Cracow, Poland, 2006; pp. 183–188. [Google Scholar]
- Dewi, H.S.; Volpp, J.; Kaplan, A.F.H. Short thermal cycle treatment with laser of vanadium microalloyed steels. J. Manuf. Proc. 2020, 57, 543–551. [Google Scholar] [CrossRef]
- Bendoumi, A.; Makuch, N.; Chegroune, R.; Kulka, M.; Keddam, M.; Dziarski, P.; Przestacki, D. The effect of temperature distribution and cooling rate on microstructure and microhardness of laser re-melted and laser-borided carbon steels with various carbon concentrations. Surf. Coat. Technol. 2020, 387, 125541. [Google Scholar] [CrossRef]
- Li, Z.; Tong, B.; Zhang, Q.; Yao, J.; Kovalenko, V.; Li, Z. Influence of initial microstructure on the microstructure evolution and mechanical properties of 1.0C-1.5Cr steel in the laser surface quenching. Mater. Sci. Eng. A 2020, 788, 139490. [Google Scholar] [CrossRef]
- Siddiqui, A.A.; Dubey, A.K. Recent trends in laser cladding and surface alloying. Opt. Laser Technol. 2020, 134, 106619. [Google Scholar] [CrossRef]
- Kulka, M.; Makuch, N.; Pertek, A. Microstructure and properties of laser-borided 41Cr4 steel. Opt. Laser Technol. 2013, 45, 308–318. [Google Scholar] [CrossRef]
- Piasecki, A.; Kulka, M.; Kotkowiak, M. Wear resistance improvement of 100CrMnSi6–4 bearing steel by laser boriding using CaF2 self-lubricating addition. Tribol. Int. 2016, 97, 173–191. [Google Scholar] [CrossRef]
- Paczkowska, M.; Ratuszek, W.; Waligóra, W. Microstructure of laser boronized nodular iron. Surf. Coat. Technol. 2010, 205, 2542–2545. [Google Scholar] [CrossRef]
- Filip, R.; Sieniawski, J.; Pleszakov, E. Formation of surface layers on Ti–6Al–4V titanium alloy by laser alloying. Surf. Eng. 2006, 22, 53–57. [Google Scholar] [CrossRef]
- Guo, C.; Zhou, J.; Zhao, J.; Guo, B.; Yu, Y.; Zhou, H.; Chen, J. Microstructure and friction and wear behavior of laser boronizing composite coatings on titanium substrate. Appl. Surf. Sci. 2011, 257, 4398–4405. [Google Scholar] [CrossRef]
- Kulka, M.; Makuch, N.; Dziarski, P.; Piasecki, A.; Miklaszewski, A. Microstructure and properties of laser-borided composite layers formed on commercially pure titanium. Opt. Laser Technol. 2014, 56, 409–424. [Google Scholar] [CrossRef]
- Makuch, N.; Kulka, M.; Dziarski, P.; Przestacki, D. Laser surface alloying of commercially pure titanium with boron and carbon. Opt. Lasers Eng. 2014, 57, 64–81. [Google Scholar] [CrossRef]
- Kulka, M.; Dziarski, P.; Makuch, N.; Piasecki, A.; Miklaszewski, A. Microstructure and properties of laser-borided Inconel 600-alloy. Appl. Surf. Sci. 2013, 284, 757–771. [Google Scholar] [CrossRef]
- Kulka, M.; Makuch, N.; Dziarski, P.; Piasecki, A. A study of nanoindentation for mechanical characterization of chromium and nickel borides’ mixtures formed by laser boriding. Ceram. Int. 2014, 40, 6083–6094. [Google Scholar] [CrossRef]
- Ayers, J.D.; Tucker, T.R. Particulate-TiC-hardened steel surfaces by laser melt injection. Thin Solid Films 1980, 73, 201–207. [Google Scholar] [CrossRef]
- Kim, T.H.; Kim, B.C. Chromium carbide laser-beam surface-alloying treatment on stainless steel. J. Mater. Sci. 1992, 27, 2967–2973. [Google Scholar] [CrossRef]
- Tassin, C.; Laroudie, F.; Pons, M.; Lelait, L. Improvement of the wear resistance of 316L stainless steel by laser surface alloying. Surf. Coat. Technol. 1996, 80, 207–210. [Google Scholar] [CrossRef]
- Rieker, C.; Morris, D.G.; Steffen, J. Formation of hard microcrystalline layers on stainless steel by laser alloying. Mater. Sci. Technol. 1989, 5, 590–594. [Google Scholar] [CrossRef]
- Kwok, C.T.; Cheng, F.T.; Man, H.C. Laser-fabricated Fe-Ni-Co-Cr-B austenitic alloy on steels. Part, I. Microstructures and cavitation erosion behavior. Surf. Coat. Technol. 2001, 145, 194–205. [Google Scholar] [CrossRef]
- Kulka, M.; Mikolajczak, D.; Makuch, N.; Dziarski, P.; Miklaszewski, A. Wear resistance improvement of austenitic 316L steel by laser alloying with boron. Surf. Coat. Technol. 2016, 291, 292–313. [Google Scholar] [CrossRef]
- Kubashevsky, O. Iron—Binary Phase Diagrams; Springer: Berlin/Heidelberg, Germany, 1982. [Google Scholar]
- Verein Deutscher Ingenieure Normen. VDI 3198; VDI-Verlag Düsseldorf: Düsseldorf, Germany, 1991. [Google Scholar]
- Tassin, C.; Laroudie, F.; Pons, M.; Lelait, L. Carbide-reinforced coatings on AISI 316 L stainless steel by laser surface alloying. Surf. Coat. Technol. 1995, 76, 450–455. [Google Scholar] [CrossRef]
- Sun, G.F.; Zhang, Y.K.; Zhang, M.K.; Zhou, R.; Wang, K.; Liu, C.S.; Luo, K.Y. Microstructure and corrosion characteristics of 304 stainless steel laser-alloyed with Cr–CrB2. Appl. Surf. Sci. 2014, 295, 94–107. [Google Scholar] [CrossRef]
- Kwok, C.T.; Lo, K.H.; Chan, W.K.; Cheng, F.T.; Man, H.C. Effect of laser surface melting on intergranular corrosion behaviour of aged austenitic and duplex stainless steels. Corros. Sci. 2011, 53, 1581–1591. [Google Scholar] [CrossRef]
- Hirsch, T.; Hoffmann, F.; Mayr, P. Röntgenographische untersuchungen mikrostruktureller kenngrößen von verbindungsschichten gasnitrierter stähle. Haerteri Tech. Mitt. 1996, 51, 390–398. [Google Scholar]
- Hirsch, T.; Hoffmann, F.; Mayr, P. Effect of different compound layer and base material microstructures on microstrain and domain size of nitrided steel. Surf. Eng. 1998, 14, 481–488. [Google Scholar] [CrossRef]
- Kraus, I.; Ganev, N.; Gosmanova, G.; Tietz, H.D.; Pfeiffer, L.; Böhm, S. Residual stress measurement in alumina coatings. Mater. Sci. Eng. A 1995, 199, L15–L17. [Google Scholar] [CrossRef]
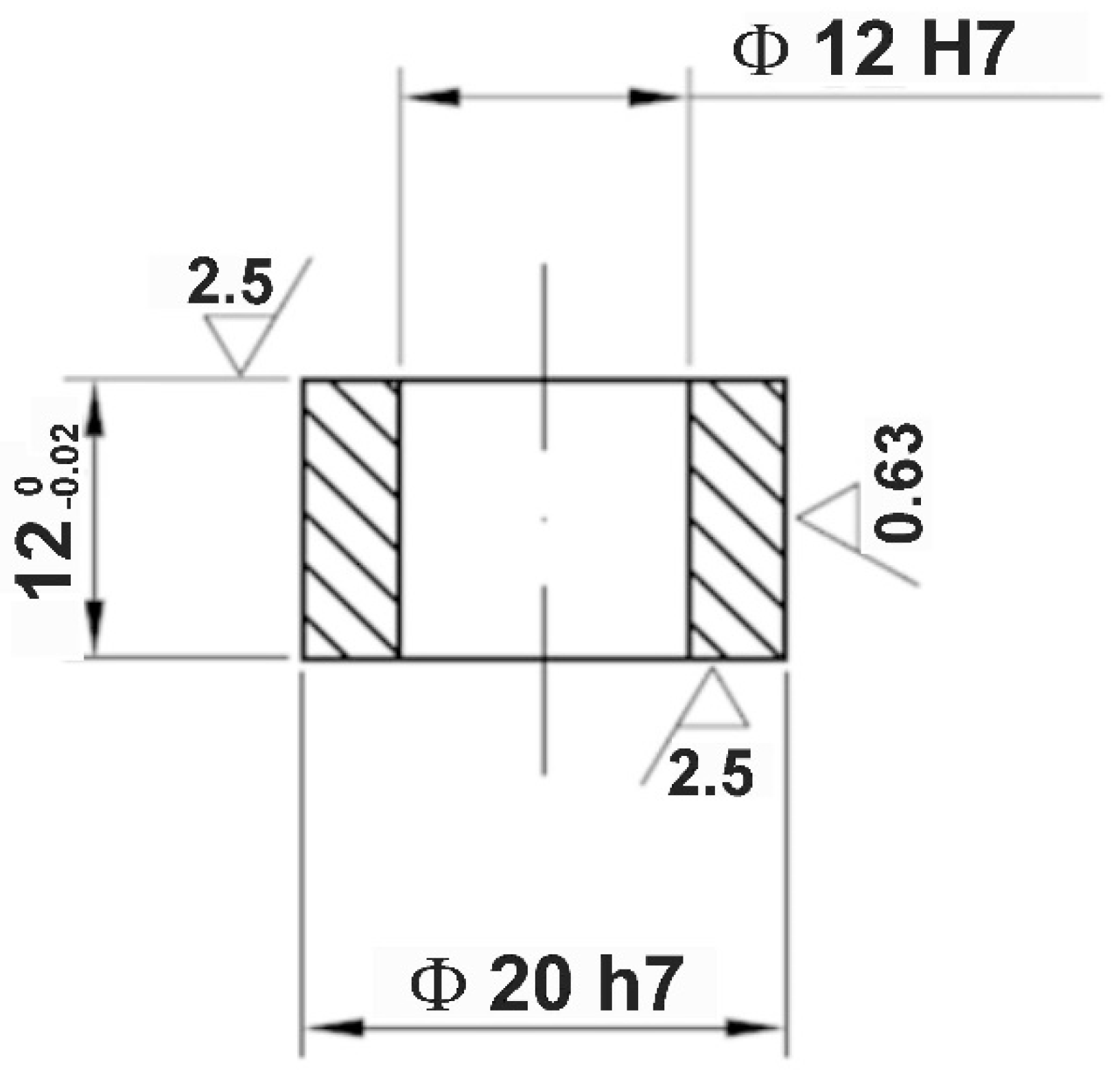
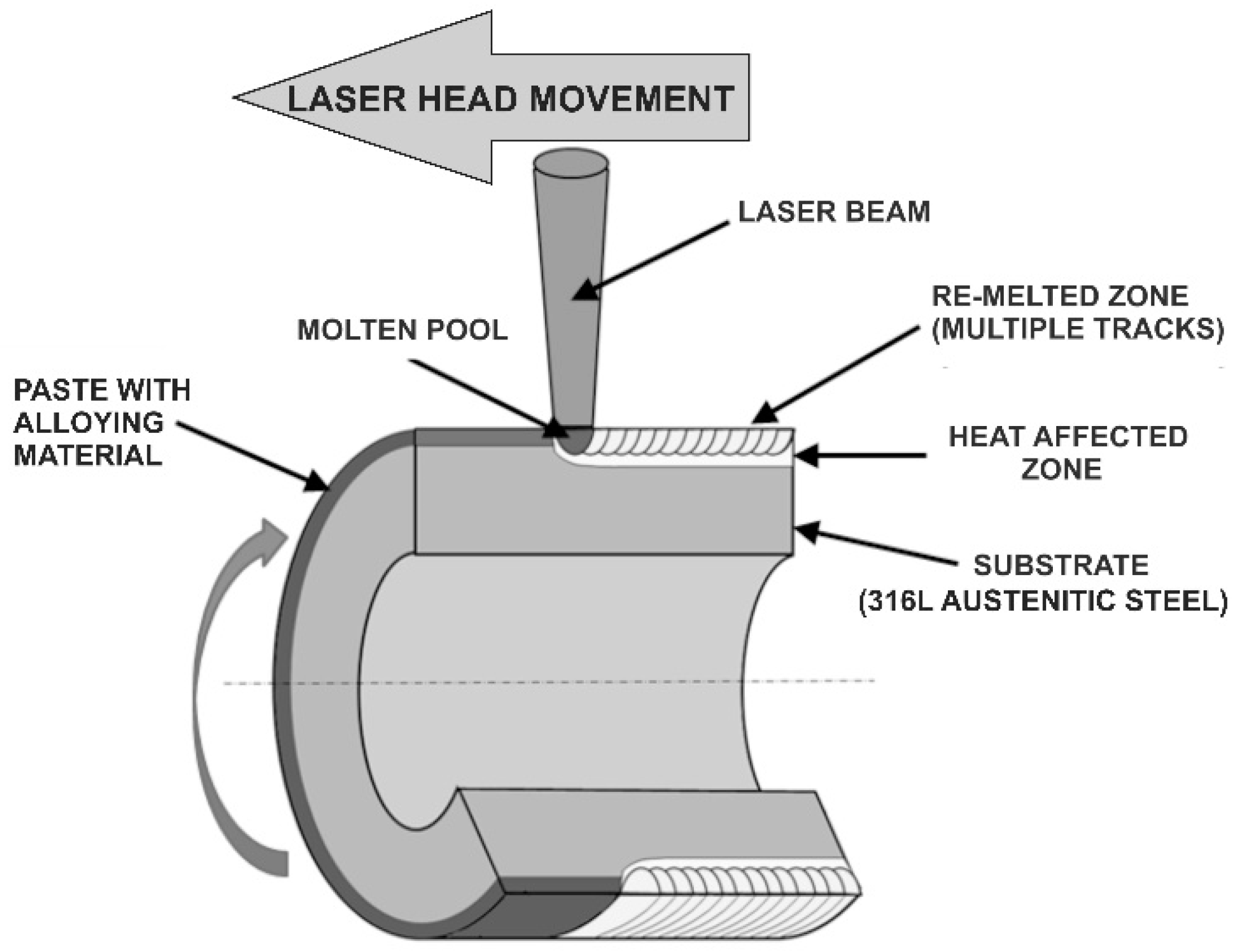

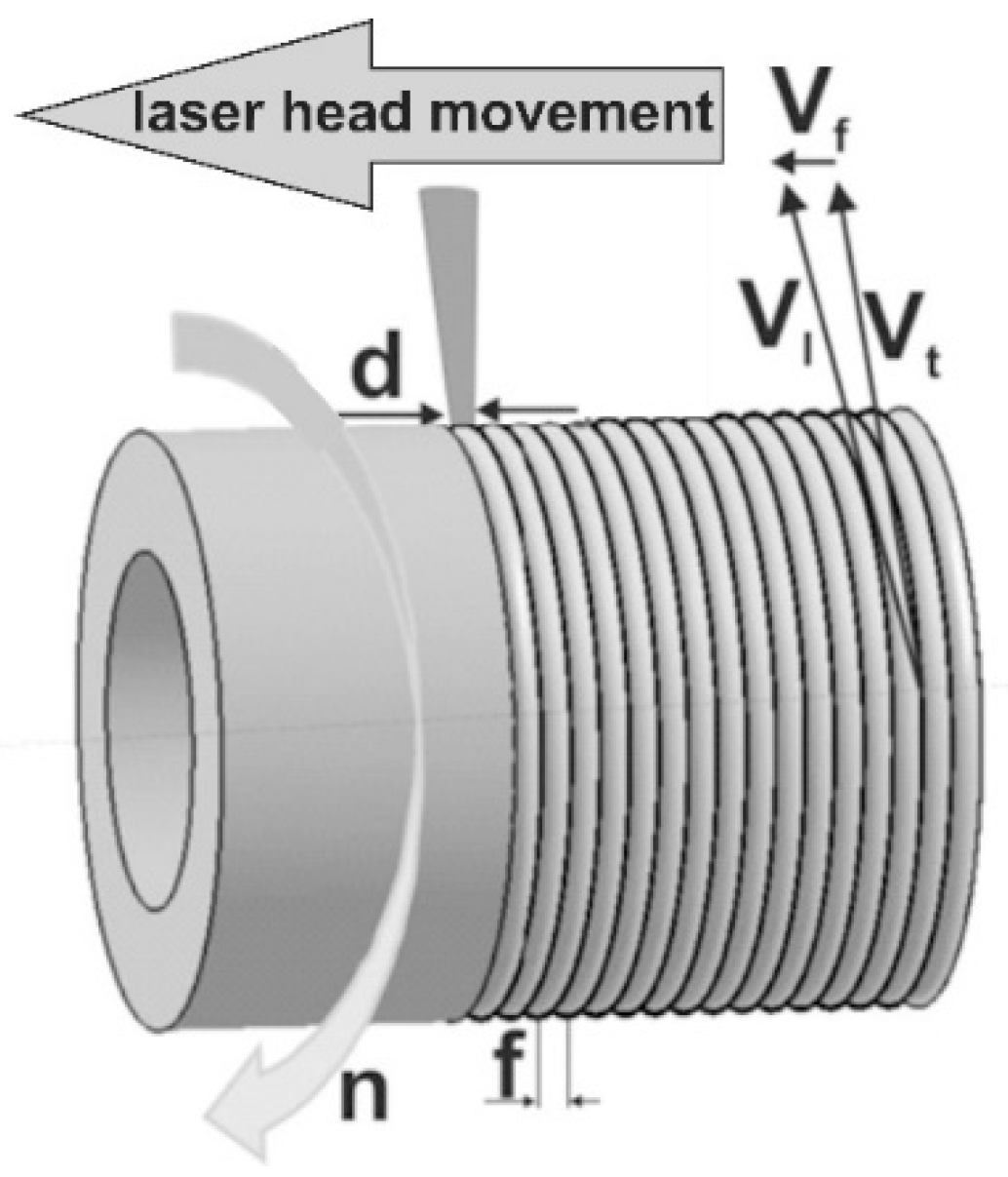
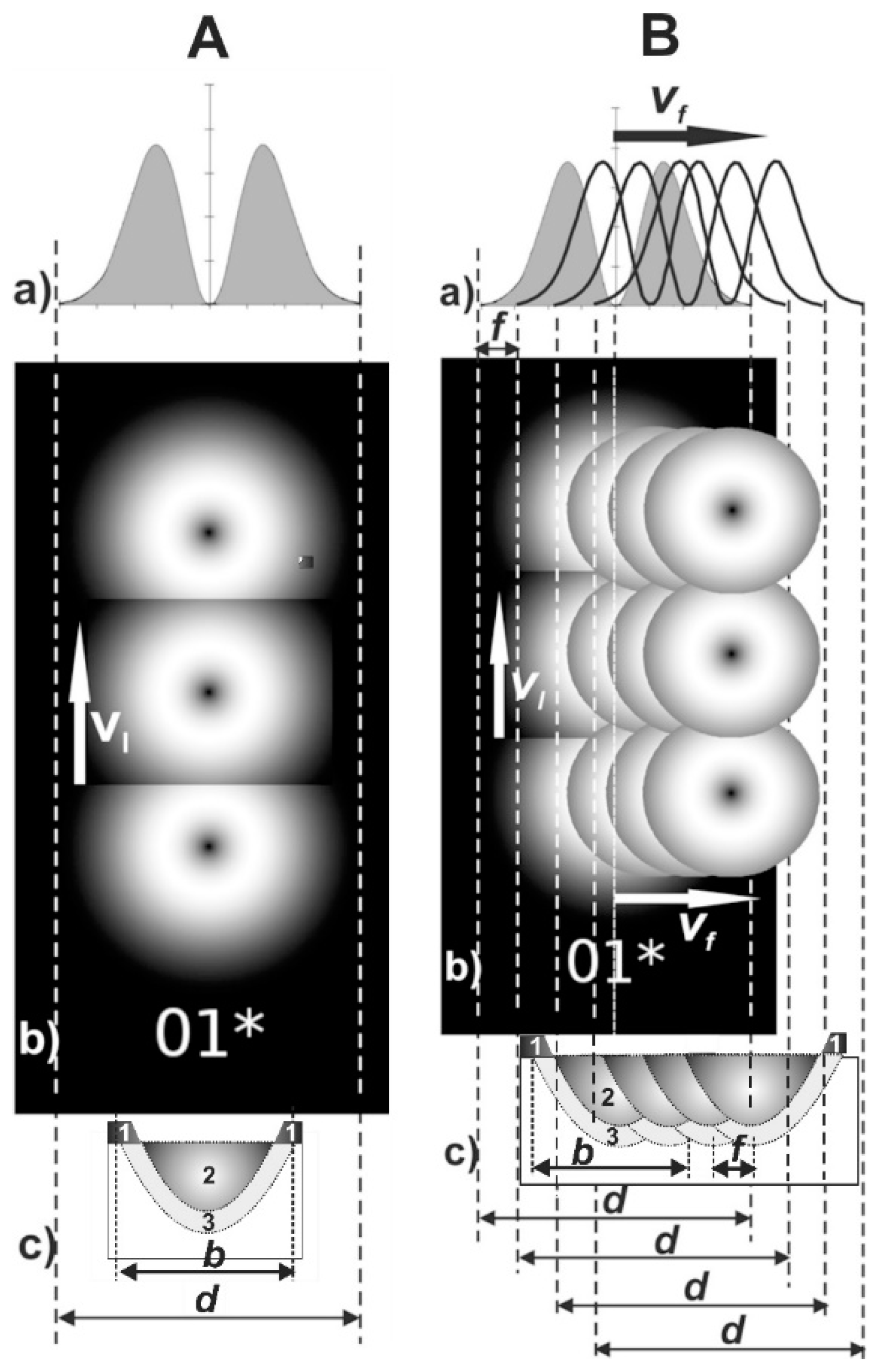










| Element | C | Cr | Ni | Mo | Mn | Si | Fe |
|---|---|---|---|---|---|---|---|
| (wt%) | 0.023 | 17.45 | 12.92 | 2.88 | 0.56 | 0.45 | balance |
| Element | C | Cr | Ni | W | Mn | Si | Fe | Co |
|---|---|---|---|---|---|---|---|---|
| (wt%) | 1.2 | 28.0 | <3.0 | 4.5 | 0.56 | 1.1 | <3.0 | balance |
| Type of Alloying Material | Thickness of Paste Coating tC (mm) | Laser Beam Diameter d (mm) | Scanning Rate vl (m·min−1) | Overlapping O (%) | Laser Beam Power P (kW) | Averaging Irradiance E (kW/cm2) |
|---|---|---|---|---|---|---|
| B | 200 | 2 | 2.88 | 86 | 1.82 | 59.73 |
| B + Stellite-6, mass ratio 1:1 | 1.43 | 45.52 | ||||
| 1.56 | 49.66 | |||||
| B + Ni, mass ratio 1:1 | 1.43 | 45.52 | ||||
| 1.56 | 49.66 | |||||
| B + Ni–Cr (4:1), mass ratio 1:1 | 1.43 | 45.52 | ||||
| 1.56 | 49.66 |
| Type of Alloying Material | Laser Beam Power P (kW) | Averaging Depth of MZ dMZ (μm) | Dilution Ratio DR |
|---|---|---|---|
| B | 1.82 | 432 | 0.54 |
| B + Stellite-6, mass ratio 1:1 | 1.43 | 338 | 0.41 |
| 1.56 | 384 | 0.48 | |
| B + Ni, mass ratio 1:1 | 1.43 | 345 | 0.42 |
| 1.56 | 383 | 0.48 | |
| B + Ni–Cr (4:1), mass ratio 1:1 | 1.43 | 352 | 0.43 |
| 1.56 | 395 | 0.49 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kulka, M.; Mikołajczak, D.; Makuch, N.; Dziarski, P.; Przestacki, D.; Panfil-Pryka, D.; Piasecki, A.; Miklaszewski, A. Laser Surface Alloying of Austenitic 316L Steel with Boron and Some Metallic Elements: Microstructure. Materials 2020, 13, 4852. https://doi.org/10.3390/ma13214852
Kulka M, Mikołajczak D, Makuch N, Dziarski P, Przestacki D, Panfil-Pryka D, Piasecki A, Miklaszewski A. Laser Surface Alloying of Austenitic 316L Steel with Boron and Some Metallic Elements: Microstructure. Materials. 2020; 13(21):4852. https://doi.org/10.3390/ma13214852
Chicago/Turabian StyleKulka, Michał, Daria Mikołajczak, Natalia Makuch, Piotr Dziarski, Damian Przestacki, Dominika Panfil-Pryka, Adam Piasecki, and Andrzej Miklaszewski. 2020. "Laser Surface Alloying of Austenitic 316L Steel with Boron and Some Metallic Elements: Microstructure" Materials 13, no. 21: 4852. https://doi.org/10.3390/ma13214852
APA StyleKulka, M., Mikołajczak, D., Makuch, N., Dziarski, P., Przestacki, D., Panfil-Pryka, D., Piasecki, A., & Miklaszewski, A. (2020). Laser Surface Alloying of Austenitic 316L Steel with Boron and Some Metallic Elements: Microstructure. Materials, 13(21), 4852. https://doi.org/10.3390/ma13214852









