Treatment of Osseous Defects after Mandibular Third Molar Removal with a Resorbable Alloplastic Grafting Material: A Case Series with 1- to 2-Year Follow-Up
Abstract
1. Introduction
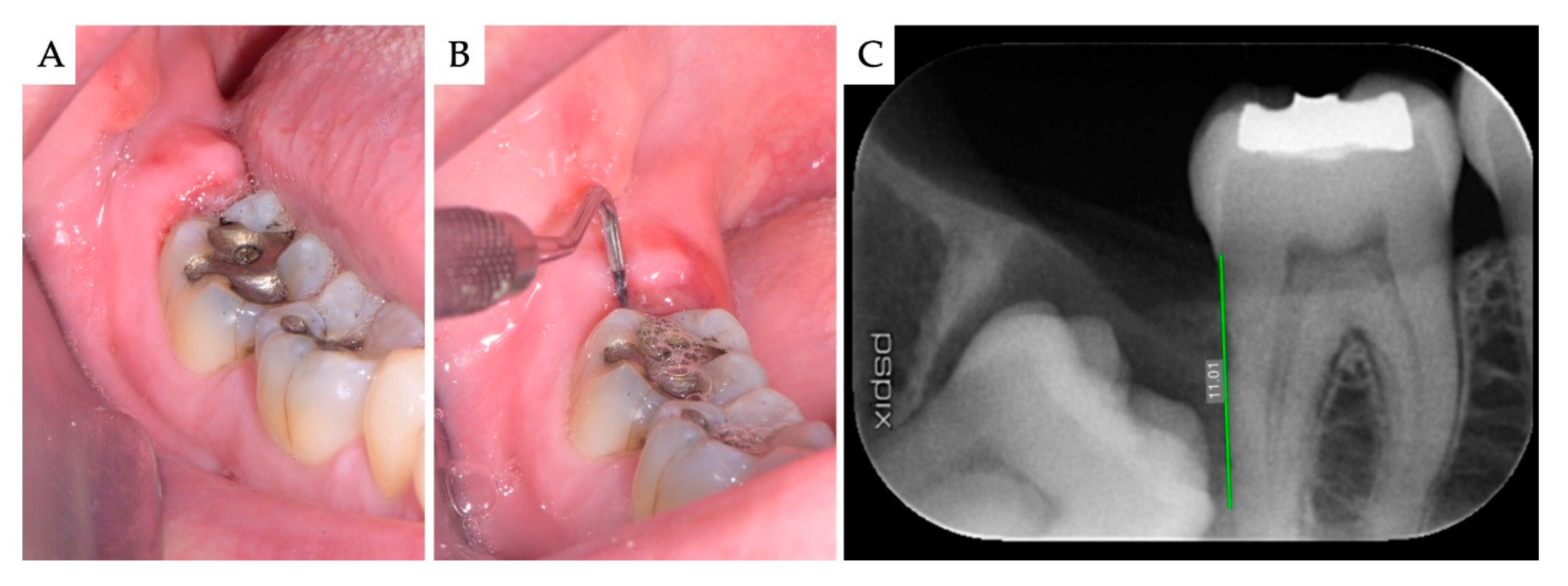
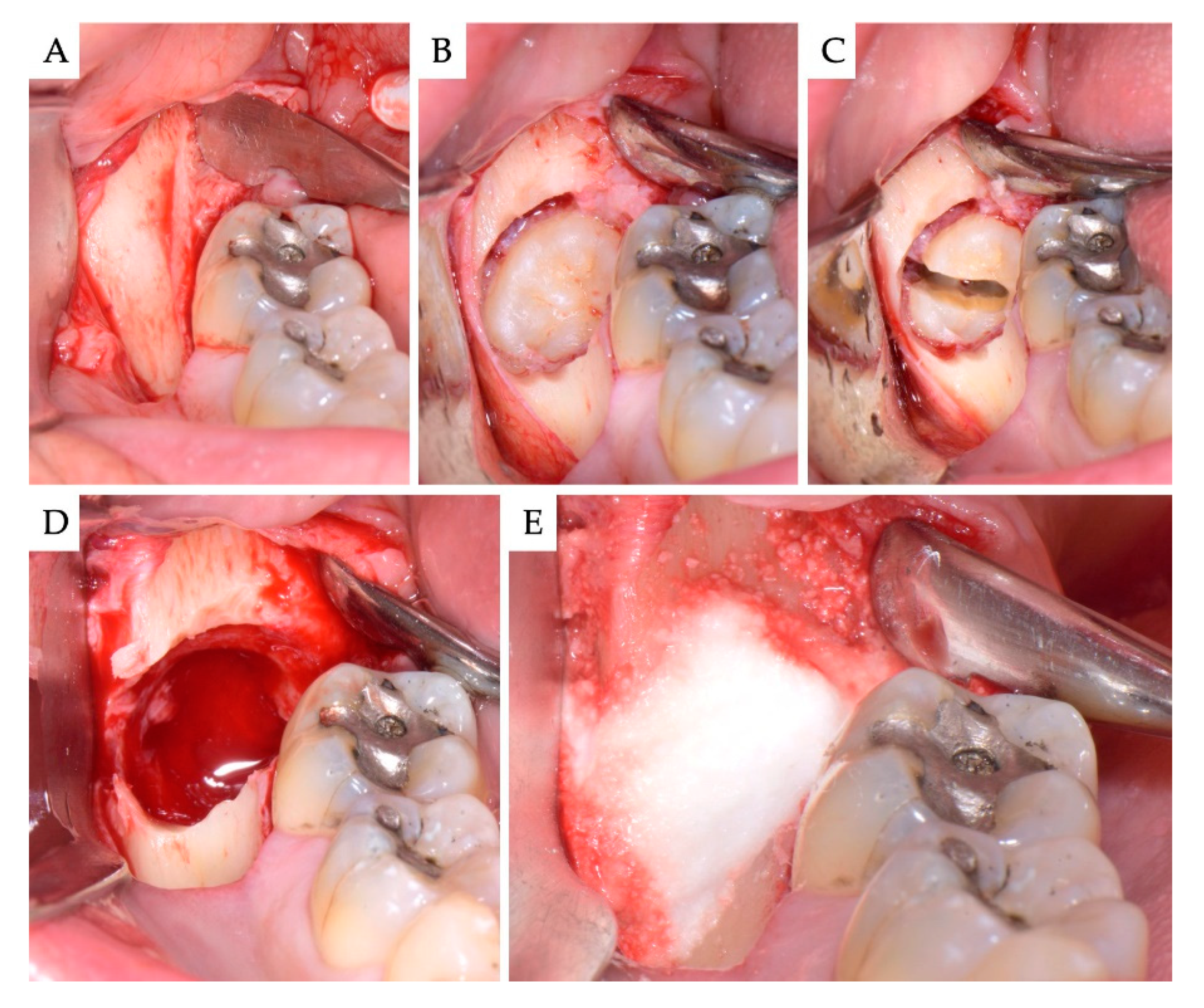
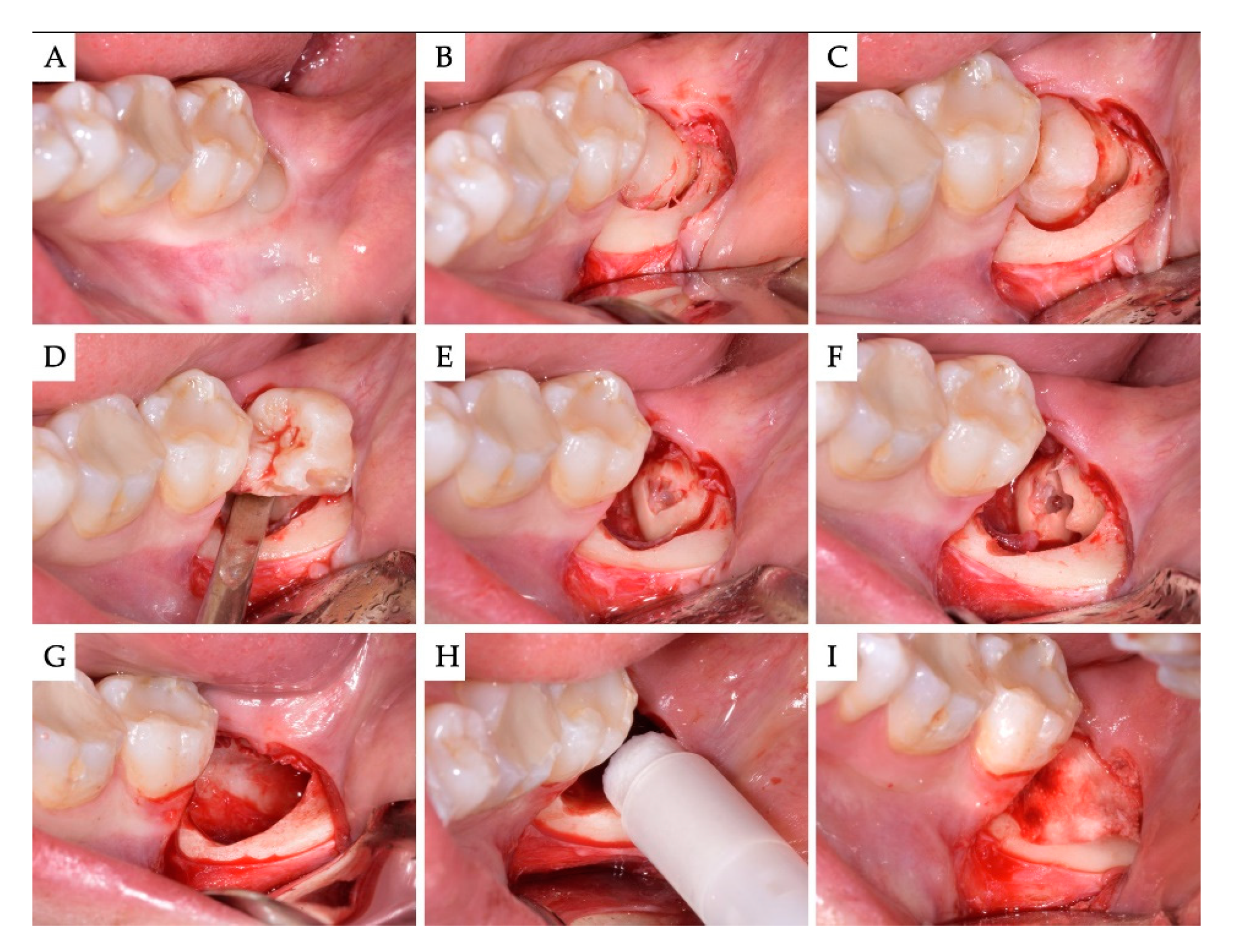
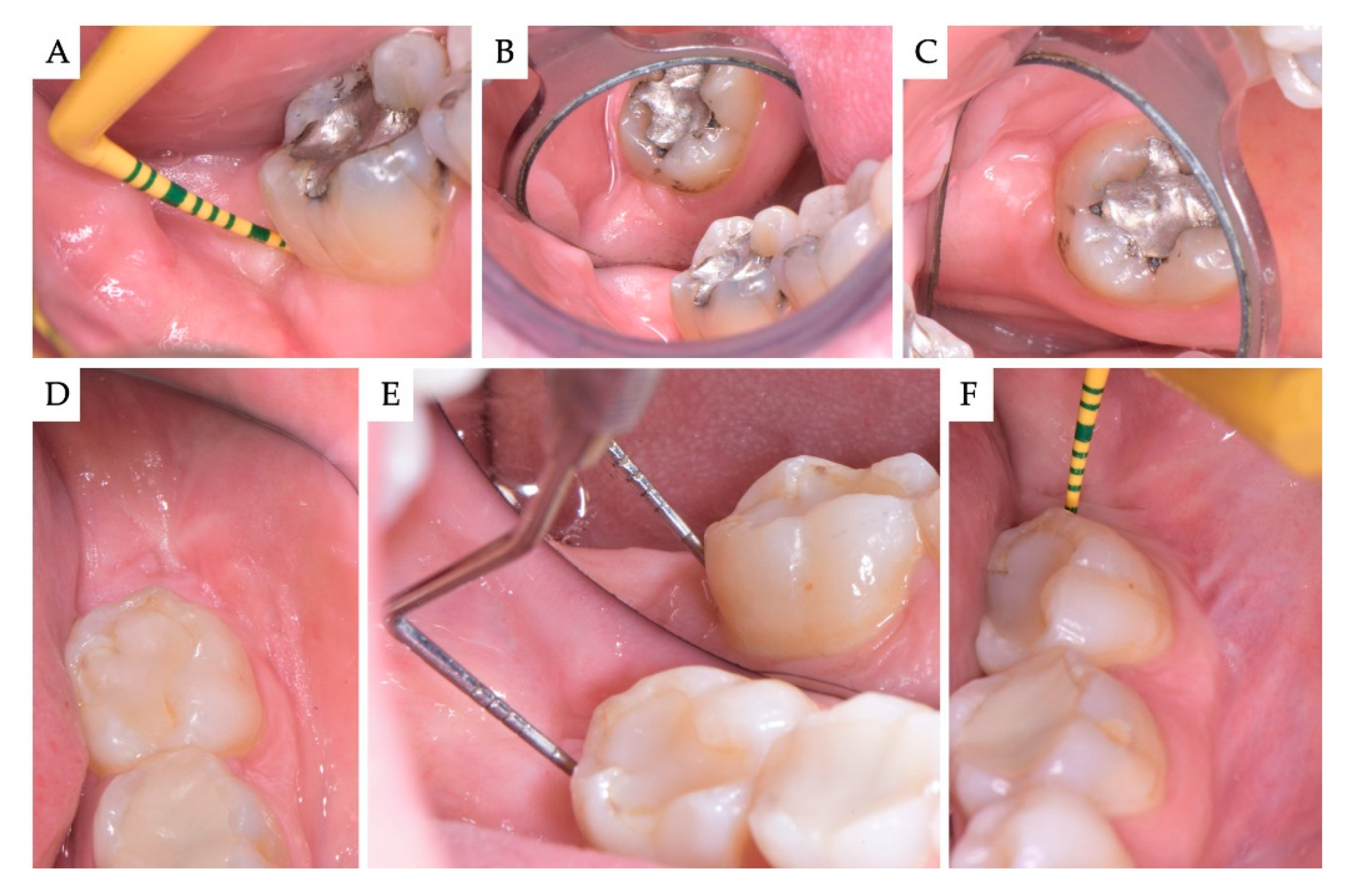
2. Materials and Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Kim, J.W.; Jo, Y.Y.; Kim, J.Y.; Oh, J.H.; Yang, B.E.; Kim, S.G. Retrospective comparative clinical study for silk mat application into extraction socket. Maxillofac. Plast. Reconstr. Surg. 2019, 41, 16. [Google Scholar] [CrossRef] [PubMed]
- Camps-Font, O.; Caro-Bonfill, C.; Sánchez-Garcés, M.À.; Gay-Escoda, C. Periodontal Regenerative Therapy for Preventing Bone Defects Distal to Mandibular Second Molars After Surgical Removal of Impacted Third Molars: A Systematic Review and Meta-Analysis of Randomized Clinical Trials. J. Oral Maxillofac. Surg. 2018, 76, 2482–2514. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.T.; Hum, L.; Chen, Y.W. The effect of regenerative periodontal therapy in preventing periodontal defects after the extraction of third molars: A systematic review and meta-analysis. J. Am. Dent. Assoc. 2016, 147, 709–719. [Google Scholar] [CrossRef] [PubMed]
- Bouloux, G.F.; Busaidy, K.F.; Beirne, O.R.; Chuang, S.K.; Dodson, T.B. What is the risk of future extraction of asymptomatic third molars? A systematic review. J. Oral Maxillofac. Surg. 2015, 73, 806–811. [Google Scholar] [CrossRef]
- Barbato, L.; Kalemaj, Z.; Buti, J.; Baccini, M.; La Marca, M.; Duvina, M.; Tonelli, P. Effect of Surgical Intervention for Removal of Mandibular Third Molar on Periodontal Healing of Adjacent Mandibular Second Molar: A Systematic Review and Bayesian Network Meta-Analysis. J. Periodontol. 2016, 87, 291–302. [Google Scholar] [CrossRef]
- Karapataki, S.; Hugoson, A.; Kugelberg, C.F. Healing following GTR treatment of bone defects distal to mandibular 2nd molars after surgical removal of impacted 3rd molars. J. Clin. Periodontol. 2000, 7, 325–332. [Google Scholar] [CrossRef]
- Aloy-Prósper, A.; García-Mira, B.; Larrazabal-Morón, C.; Peñarrocha-Diago, M. Distal probing depth and attachment level of lower second molars following surgical extraction of lower third molars: A literature review. Med. Oral Patol. Oral Cir. Bucal 2010, 15, e755–e759. [Google Scholar] [CrossRef][Green Version]
- Dodson, T.B. Is there a role for reconstructive techniques to prevent periodontal defects after third molar surgery? J. Oral Maxillofac. Surg. 2005, 63, 891–896. [Google Scholar] [CrossRef]
- Kumar, N.; Prasad, K.; Ramanujam, L.; Ranganath, K.; Dexith, J.; Chauhan, A. Evaluation of treatment outcome after impacted mandibular third molar surgery with the use of autologous platelet-rich fibrin: A randomized controlled clinical study. J. Oral Maxillofac. Surg. 2015, 73, 1042–1049. [Google Scholar] [CrossRef]
- Kugelberg, C.F.; Ahlström, U.; Ericson, S.; Hugoson, A.; Thilander, H. The influence of anatomical, pathophysiological and other factors on periodontal healing after impacted lower third molar surgery. A multiple regression analysis. J. Clin. Periodontol. 1991, 18, 37–43. [Google Scholar] [CrossRef]
- Kugelberg, C.F.; Ahlström, U.; Ericson, S.; Hugoson, A.; Kvint, S. Periodontal healing after impacted lower third molar surgery in adolescents and adults. A prospective study. Int. J. Oral Maxillofac. Surg. 1991, 20, 18–24. [Google Scholar] [CrossRef]
- Ge, J.; Yang, C.; Zheng, J.; Hu, Y. Autogenous bone grafting for treatment of osseous defect after impacted mandibular third molar extraction: A randomized controlled trial. Clin. Implant. Dent. Relat. Res. 2017, 19, 572–580. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.W.; Lee, C.T.; Hum, L.; Chuang, S.K. Effect of flap design on periodontal healing after impacted third molar extraction: A systematic review and meta-analysis. Int. J. Oral Maxillofac. Surg. 2017, 46, 363–372. [Google Scholar] [CrossRef] [PubMed]
- Laurito, D.; Lollobrigida, M.; Graziani, F.; Guerra, F.; Vestri, A.; De Biase, A. Periodontal effects of a transposed versus a conventional flap in mandibular third molar extractions. J. Craniofac. Surg. 2016, 27, 708–711. [Google Scholar] [CrossRef]
- Sammartino, G.; Tia, M.; Marenzi, G.; di Lauro, A.E.; D’Agostino, E.; Claudio, P.P. Use of autologous platelet-rich plasma (PRP) in periodontal defect treatment after extraction of impacted mandibular third molars. J. Oral Maxillofac. Surg. 2005, 63, 766–770. [Google Scholar] [CrossRef]
- Sammartino, G.; Tia, M.; Gentile, E.; Marenzi, G.; Claudio, P.P. Platelet-rich plasma and resorbable membrane for prevention of periodontal defects after deeply impacted lower third molar extraction. J. Oral Maxillofac. Surg. 2009, 67, 2369–2373. [Google Scholar] [CrossRef] [PubMed]
- Hassan, K.S.; Marei, H.F.; Alagl, A.S. Does grafting of third molar extraction sockets enhance periodontal measures in 30- to 35-year-old patients? J. Oral Maxillofac. Surg. 2012, 70, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Andrade Munhoz, E.; Bodanezi, A.; Ferreira Junior, O.; Mauro Granjeiro, J. Bone crestal height and bone density after third-molar extraction and grafting: A long-term follow-up study. Clin. Oral Investig. 2011, 15, 123–126. [Google Scholar] [CrossRef] [PubMed]
- Chan, H.L.; Lin, G.H.; Fu, J.H.; Wang, H.L. Alterations in bone quality after socket preservation with grafting materials: A systematic review. Int. J. Oral Maxillofac. Implants 2013, 28, 710–720. [Google Scholar] [CrossRef]
- Le, B.Q.; Nurcombe, V.; Cool, S.M.; van Blitterswijk, C.A.; de Boer, J.; La Pointe, V.L.S. The Components of Bone and What They Can Teach Us about Regeneration. Materials 2017, 11, 14. [Google Scholar] [CrossRef]
- Chappuis, V.; Rahman, L.; Buser, R.; Janner, S.F.M.; Belser, U.C.; Buser, D. Effectiveness of contour augmentation with guided bone regeneration: 10-year results. J. Dent. Res. 2018, 97, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Iocca, O.; Farcomeni, A.; Pardiñas Lopez, S.; Talib, H.S. Alveolar ridge preservation after tooth extraction: A Bayesian Network meta-analysis of grafting materials efficacy on prevention of bone height and width reduction. J. Clin. Periodontol. 2017, 44, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Danesh-Sani, S.A.; Engebretson, S.P.; Janal, M.N. Histomorphometric results of different grafting materials and effect of healing time on bone maturation after sinus floor augmentation: A systematic review and meta-analysis. J. Periodontal Res. 2017, 52, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Cicciù, M.; Cervino, G.; Herford, A.S.; Famà, F.; Bramanti, E.; Fiorillo, L.; Lauritano, F.; Sambataro, S.; Troiano, G.; Laino, L. Facial Bone Reconstruction Using both Marine or Non-Marine Bone Substitutes: Evaluation of Current Outcomes in a Systematic Literature Review. Mar. Drugs 2018, 16, 27. [Google Scholar] [CrossRef] [PubMed]
- Jambhekar, S.; Kernen, F.; Bidra, A.S. Clinical and histologic outcomes of socket grafting after flapless tooth extraction: A systematic review of randomized controlled clinical trials. J. Prosthet. Dent. 2015, 113, 371–382. [Google Scholar] [CrossRef]
- Henkel, J.; Woodruff, M.A.; Epari, D.R.; Steck, R.; Glatt, V.; Dickinson, I.C.; Choong, P.F.; Schuetz, M.A.; Hutmacher, D.W. Bone regeneration based on tissue engineering conceptions—A 21st century perspective. Bone Res. 2013, 1, 216–248. [Google Scholar] [CrossRef]
- Araújo, M.G.; Liljenberg, B.; Lindhe, J. β-tricalcium phosphate in the early phase of socket healing: An experimental study in the dog. Clin. Oral Implants Res. 2010, 21, 445–454. [Google Scholar] [CrossRef]
- Trisi, P.; Rao, W.; Rebaudi, A.; Fiore, P. Histologic effect of pure-phase beta-tricalcium phosphate on bone regeneration in human artificial jawbone defects. Int. J. Periodontics Restorative Dent. 2003, 23, 69–78. [Google Scholar]
- Harel, N.; Moses, O.; Palti, A.; Ormianer, Z. Long-term results of implants immediately placed into extraction sockets grafted with β-tricalcium phosphate: A retrospective study. J. Oral Maxillofac. Surg. 2013, 71, E63–E68. [Google Scholar] [CrossRef]
- Kucera, T.; Sponer, P.; Urban, K.; Kohout, A. Histological assessment of tissue from large human bone defects repaired with β-tricalcium phosphate. Eur. J. Orthop. Surg. Traumatol. 2014, 24, 1357–1365. [Google Scholar] [CrossRef]
- Yuan, H.; Fernandes, H.; Habibovic, P.; de Boer, J.; Barradas, A.M.; de Ruiter, A.; Walsh, W.R.; van Blitterswijk, C.A.; de Bruijn, J.D. Osteoinductive ceramics as a synthetic alternative to autologous bone grafting. Proc. Natl. Acad. Sci. USA 2010, 107, 13614–13619. [Google Scholar] [CrossRef]
- Tang, Z.; Li, X.; Tan, Y.; Fan, H.; Zhang, X. The material and biological characteristics of osteoinductive calcium phosphate ceramics. Regen. Biomater. 2018, 5, 43–59. [Google Scholar] [CrossRef] [PubMed]
- Miron, R.J.; Zhang, Q.; Sculean, A.; Buser, D.; Pippenger, B.E.; Dard, M.; Shirakata, Y.; Chandad, F.; Zhang, Y. Osteoinductive potential of 4 commonly employed bone grafts. Clin. Oral Investig. 2016, 20, 2259–2265. [Google Scholar] [CrossRef] [PubMed]
- Barradas, A.M.; Yuan, H.; van Blitterswijk, C.; Habibovic, P. Osteoinductive biomaterials: Current knowledge of properties, experimental models and biological mechanisms. Eur. Cells Mater. 2010, 21, 407–429. [Google Scholar] [CrossRef] [PubMed]
- Cheng, L.; Shi, Y.; Ye, F.; Bu, H. Osteoinduction of calcium phosphate biomaterials in small animals. Mater. Sci. Eng. C Mater. Biol. Appl. 2013, 33, 1254–1260. [Google Scholar] [CrossRef]
- Malhotra, A.; Habibovic, P. Calcium phosphates and angiogenesis: Implications and advances for bone regeneration. Trends Biotechnol. 2016, 34, 983–992. [Google Scholar] [CrossRef]
- Al Ruhaimi, K.A. Effect of adding resorbable calcium sulfate to grafting materials on early bone regeneration in osseous defects in rabbits. Int. J. Oral Maxillofac. Implants 2000, 15, 859–864. [Google Scholar]
- Pecora, G.; Andreana, S.; Margarone, J.E.; Covani, U.; Sottosanti, J.S. Bone regeneration with a calcium sulfate barrier. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endodontol. 1997, 84, 424–429. [Google Scholar] [CrossRef]
- Strocchi, R.; Orsini, G.; Iezzi, G.; Scarano, A.; Rubini, C.; Pecora, G.; Piattelli, A. Bone regeneration with calcium sulfate: Evidence for increased angiogenesis in rabbits. J. Oral Implant. 2002, 28, 273–278. [Google Scholar] [CrossRef]
- Ruga, E.; Gallesio, C.; Chiusa, L.; Boffano, P. Clinical and histologic outcomes of calcium sulfate in the treatment of postextraction sockets. J. Craniofac. Surg. 2011, 22, 494–498. [Google Scholar] [CrossRef]
- Anson, D. Using calcium sulfate in guided tissue regeneration: A recipe for success. Compend. Contin. Educ. Dent. 2000, 21, 365–370. [Google Scholar] [PubMed]
- Mazor, Z.; Mamidwar, S.; Ricci, J.L.; Tovar, N.M. Bone repair in periodontal defect using a composite of allograft and calcium sulfate (DentoGen) and a calcium sulfate barrier. J. Oral Implant. 2011, 37, 287–292. [Google Scholar] [CrossRef] [PubMed]
- Leventis, M.; Fairbairn, P.; Mangham, C.; Galanos, A.; Vasiliadis, O.; Papavasileiou, D.; Horowitz, R. Bone Healing in Rabbit Calvaria Defects Using a Synthetic Bone Substitute: A Histological and Micro-CT Comparative Study. Materials 2018, 11, 2004. [Google Scholar] [CrossRef] [PubMed]
- Eleftheriadis, E.; Leventis, M.D.; Tosios, K.I.; Faratzis, G.; Titsinidis, S.; Eleftheriadi, I.; Dontas, I. Osteogenic activity of β-tricalcium phosphate in a hydroxyl sulphate matrix and demineralized bone matrix: A histological study in rabbit mandible. J. Oral Sci. 2010, 52, 377–384. [Google Scholar] [CrossRef]
- Podaropoulos, L.; Veis, A.A.; Papadimitriou, S.; Alexandridis, C.; Kalyvas, D. Bone regeneration using b-tricalcium phosphate in a calcium sulfate matrix. J. Oral Implant. 2009, 35, 28–36. [Google Scholar] [CrossRef]
- Leventis, M.D.; Fairbairn, P.; Dontas, I.; Faratzis, G.; Valavanis, K.D.; Khaldi, L.; Kostakis, G.; Eleftheriadis, E. Biological response to β-tricalcium phosphate/calcium sulfate synthetic graft material: An experimental study. Implant. Dent. 2014, 23, 37–43. [Google Scholar] [CrossRef]
- Fairbairn, P.; Leventis, M. Protocol for Bone Augmentation with Simultaneous Early Implant Placement: A Retrospective Multicenter Clinical Study. Int. J. Dent. 2015, 2015, 589135. [Google Scholar] [CrossRef]
- Fairbairn, P.; Leventis, M.; Mangham, C.; Horowitz, R. Alveolar Ridge Preservation Using a Novel Synthetic Grafting Material: A Case with Two-Year Follow-Up. Case Rep. Dent. 2018, 2018, 6412806. [Google Scholar] [CrossRef]
- Yang, H.L.; Zhu, X.S.; Chen, L.; Chen, C.M.; Mangham, D.C.; Coulton, L.A.; Aiken, S.S. Bone healing response to a synthetic calcium sulfate/β-tricalcium phosphate graft material in a sheep vertebral body defect model. J. Biomed. Mater. Res. B Appl. Biomater. 2012, 100, 1911–1921. [Google Scholar] [CrossRef]
- Artzi, Z.; Weinreb, M.; Givol, N.; Rohrer, M.D.; Nemcovsky, C.E.; Prasad, H.S.; Tal, H. Biomaterial Resorption Rate and Healing Site Morphology of Inorganic Bovine Bone and β-Tricalcium Phosphate in the Canine: A 24-month Longitudinal Histologic Study and Morphometric Analysis. Int. J. Oral Maxillofac. Implants 2004, 19, 357–368. [Google Scholar]
- Palti, A.; Hoch, T. A concept for the treatment of various dental bone defects. Implant. Dent. 2002, 11, 73–78. [Google Scholar] [CrossRef]
- Kugelberg, C.F. Periodontal healing two and four years after impacted lower third molar surgery. A comparative retrospective study. Int. J. Oral Maxillofac. Surg. 1990, 19, 341–345. [Google Scholar] [CrossRef]
- De Biase, A.; Mazzucchi, G.; Di Nardo, D.; Lollobrigida, M.; Serafini, G.; Testarelli, L. Prevention of Periodontal Pocket Formation after Mandibular Third Molar Extraction Using Dentin Autologous Graft: A Split Mouth Case Report. Case Rep. Dent. 2020, 2020, 1762862. [Google Scholar] [CrossRef]
- Corinaldesi, G.; Lizio, G.; Badiali, G.; Morselli-Labate, A.M.; Marchetti, C. Treatment of intrabony defects after impacted mandibular third molar removal with bioabsorbable and non-resorbable membranes. J. Periodontol. 2011, 82, 1404–1413. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, A. Periosteum: A highly underrated tool in dentistry. Int. J. Dent. 2012, 2012, 717816. [Google Scholar] [CrossRef] [PubMed]
- Lin, Z.; Fateh, A.; Salem, D.M.; Intini, G. Periosteum: Biology and applications in craniofacial bone regeneration. J. Dent. Res. 2014, 93, 109–116. [Google Scholar] [CrossRef] [PubMed]
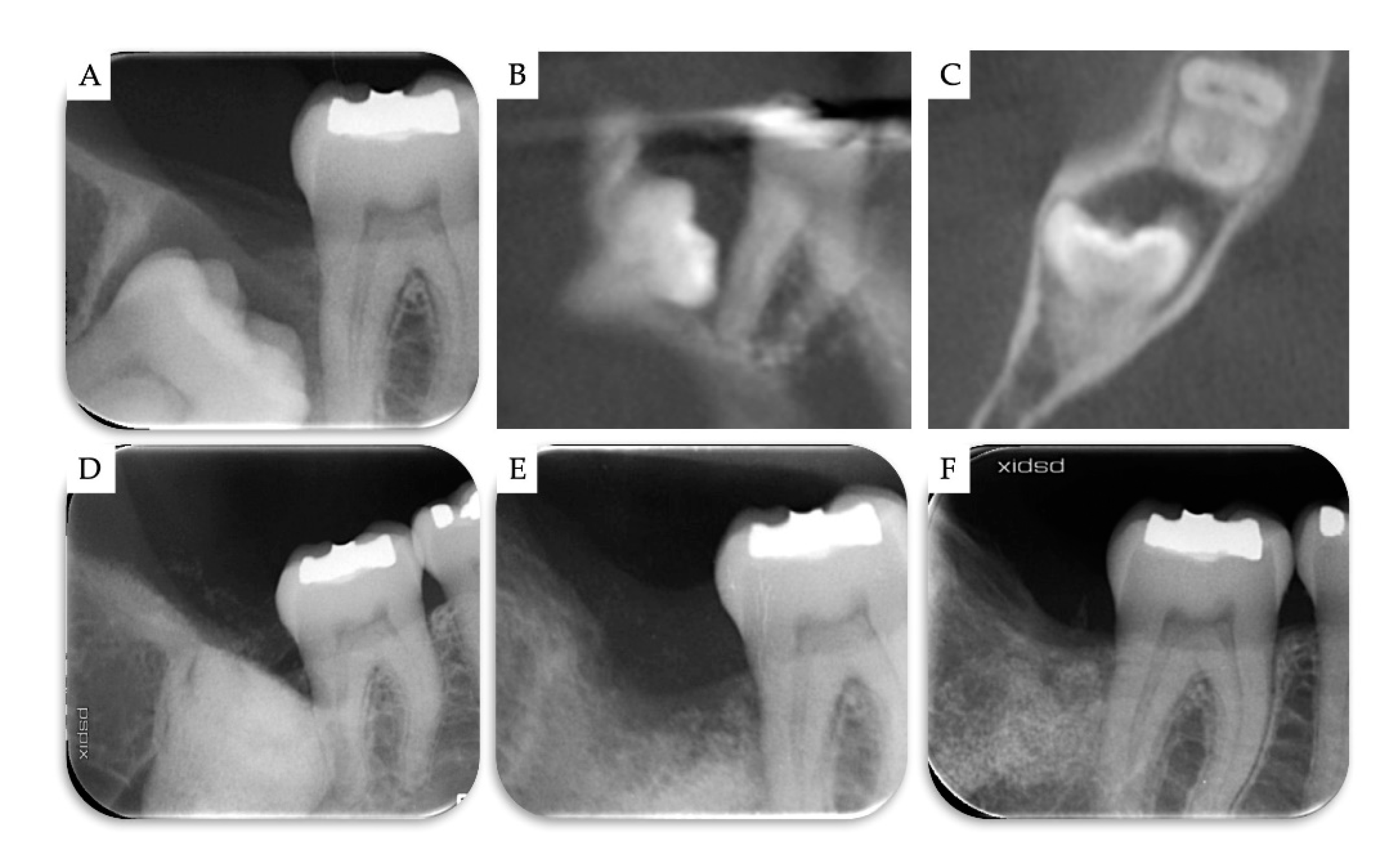
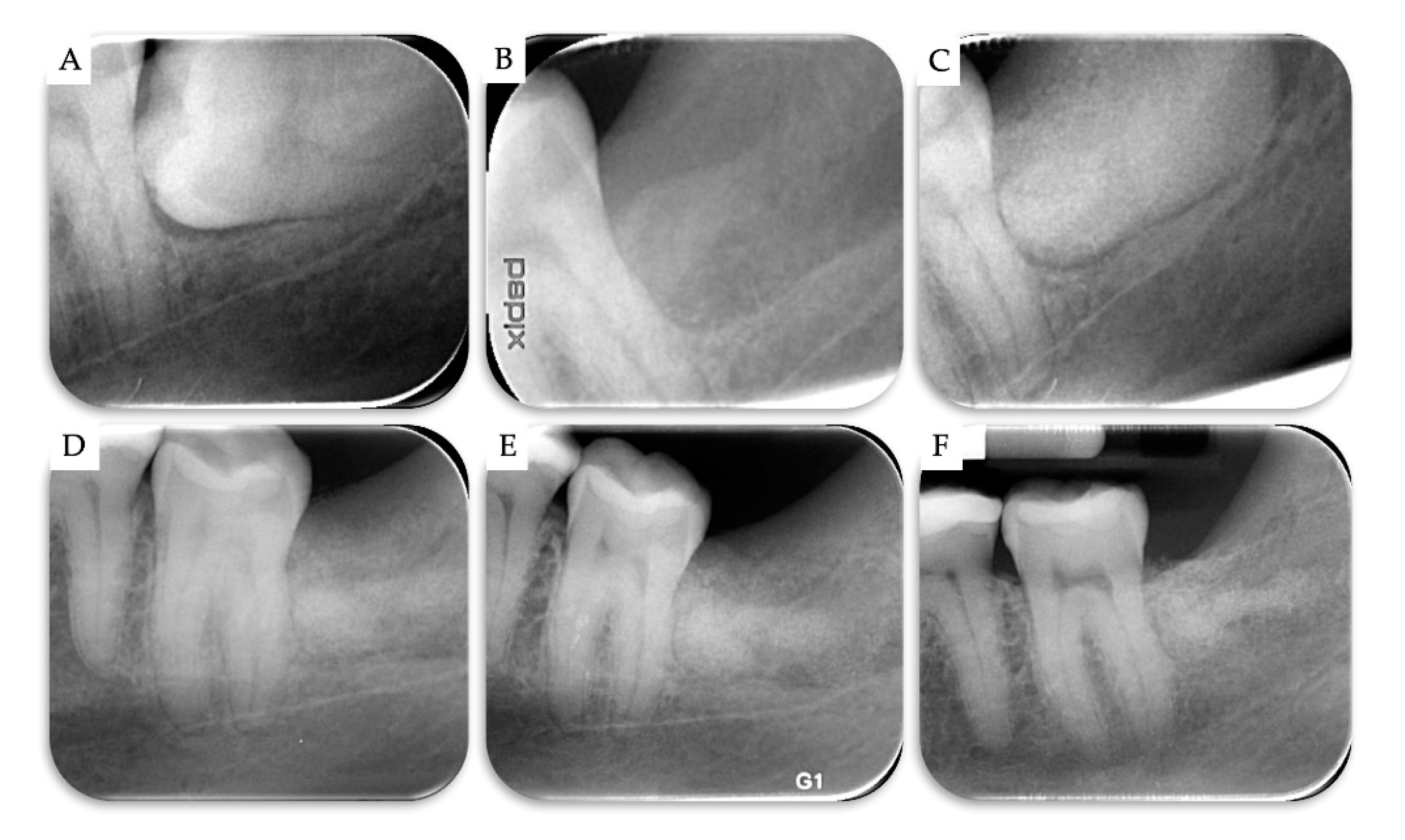
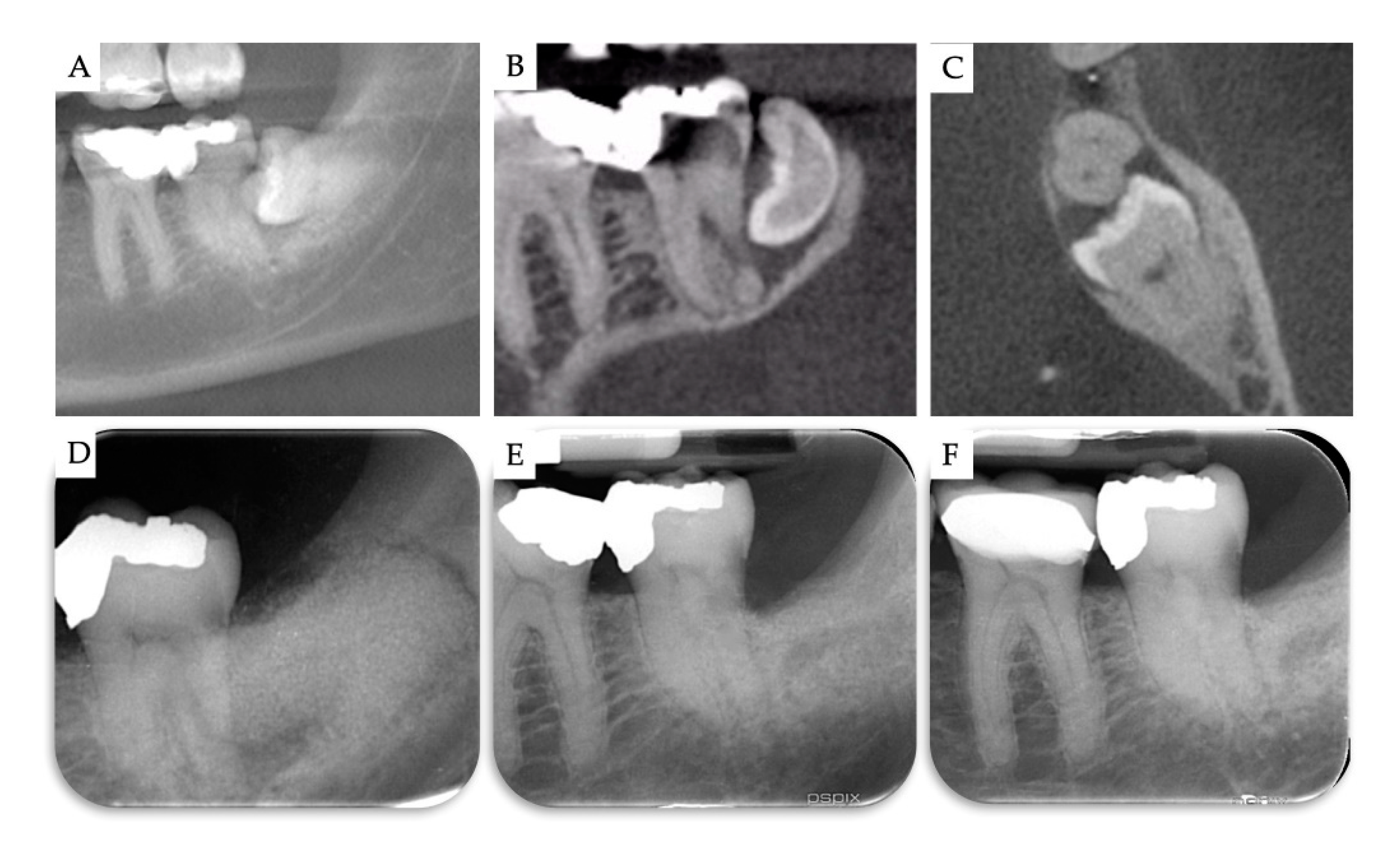
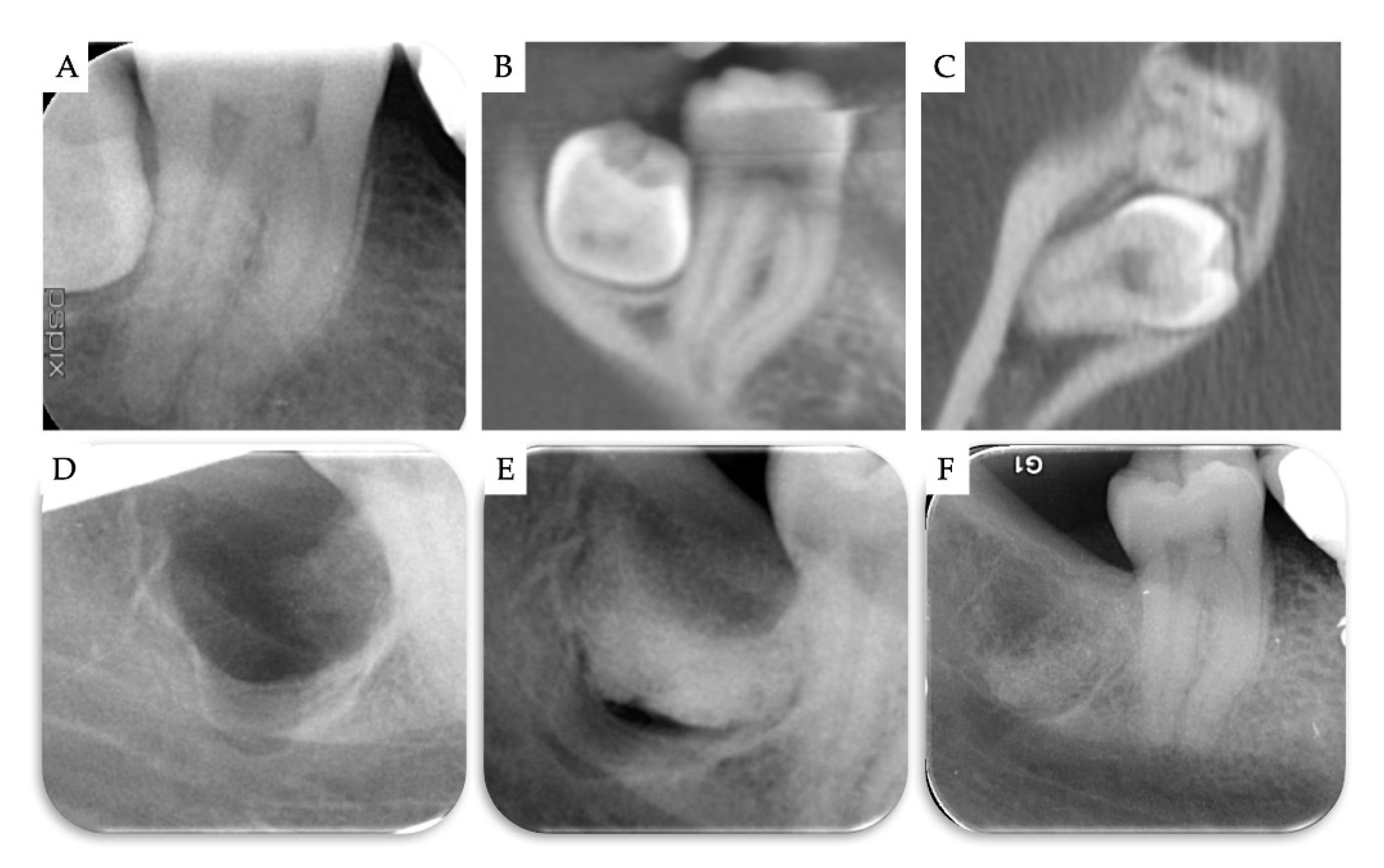
| Case | Gender | Age | Smoker | M3 | Impaction | Follow-Up (years) | PD T0 (mm) | PD T1 (mm) | BD T0 (mm) | BD T1 (mm) | BG (mm) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | F | 51 | No | 48 | Mesio-angular | 2 | 12 | 3 | 11.1 | 5.2 | 5.86 |
| 2 | F | 36 | No | 38 | Horizontal | 2 | - | 1 | 7.4 | 1.3 | 6.1 |
| 3 | M | 42 | No | 38 | Horizontal | 1 | - | 2 | 10.2 | 4.4 | 5.79 |
| 4 | M | 34 | No | 48 | Horizontal | 1 | - | 2 | 8.6 | 2.1 | 6.51 |
| Mean | 40.75 | 1.5 | 2.00 | 9.33 | 3.25 | 6.07 | |||||
| SD | 6.61 | 0.5 | 0.71 | 1.43 | 1.6 | 0.28 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leventis, M.; Tsetsenekou, E.; Kalyvas, D. Treatment of Osseous Defects after Mandibular Third Molar Removal with a Resorbable Alloplastic Grafting Material: A Case Series with 1- to 2-Year Follow-Up. Materials 2020, 13, 4688. https://doi.org/10.3390/ma13204688
Leventis M, Tsetsenekou E, Kalyvas D. Treatment of Osseous Defects after Mandibular Third Molar Removal with a Resorbable Alloplastic Grafting Material: A Case Series with 1- to 2-Year Follow-Up. Materials. 2020; 13(20):4688. https://doi.org/10.3390/ma13204688
Chicago/Turabian StyleLeventis, Minas, Efstathia Tsetsenekou, and Demos Kalyvas. 2020. "Treatment of Osseous Defects after Mandibular Third Molar Removal with a Resorbable Alloplastic Grafting Material: A Case Series with 1- to 2-Year Follow-Up" Materials 13, no. 20: 4688. https://doi.org/10.3390/ma13204688
APA StyleLeventis, M., Tsetsenekou, E., & Kalyvas, D. (2020). Treatment of Osseous Defects after Mandibular Third Molar Removal with a Resorbable Alloplastic Grafting Material: A Case Series with 1- to 2-Year Follow-Up. Materials, 13(20), 4688. https://doi.org/10.3390/ma13204688




