Photodegradation of Methylene Blue and Rhodamine B Using Laser-Synthesized ZnO Nanoparticles
Abstract
1. Introduction
2. Materials and Methods
2.1. Syntheses of the ZnO Nanoparticles
2.2. Characterization
2.3. Photocatalytic Efficiency
3. Results and Discussion
3.1. Characterization of ZnO Colloidal Solution
3.1.1. ZnO Concentration
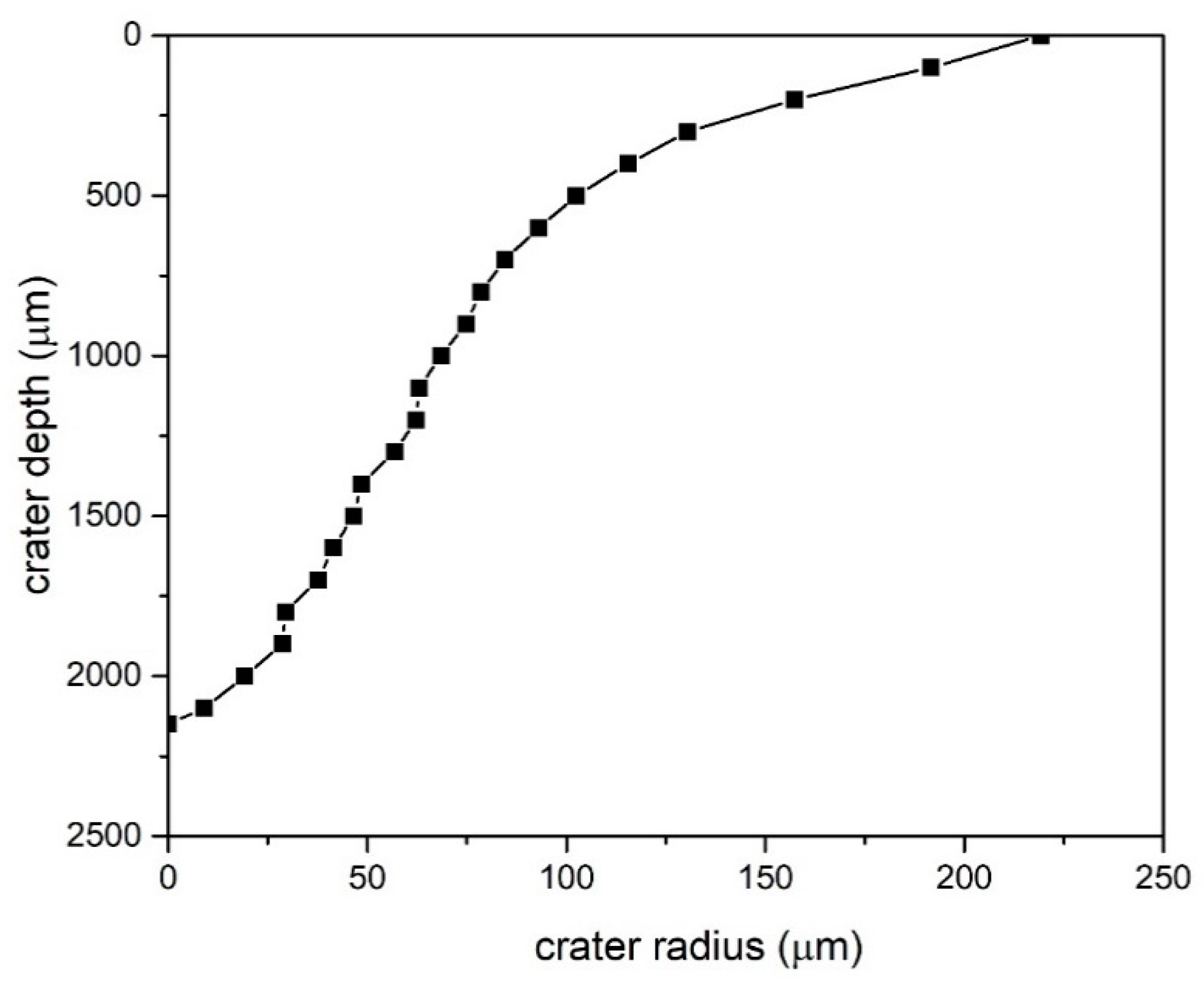
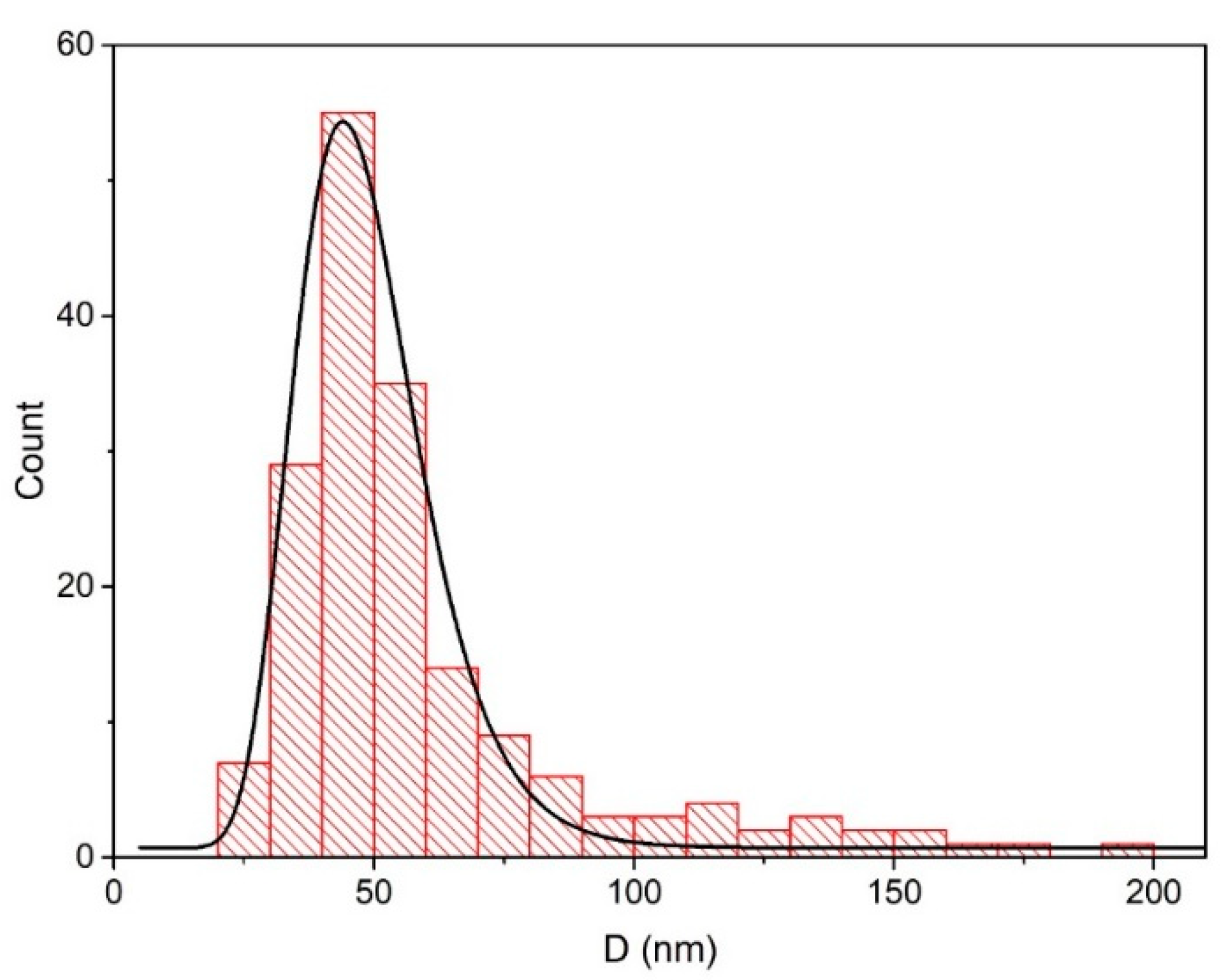
3.1.2. SEM Microscopy Observations
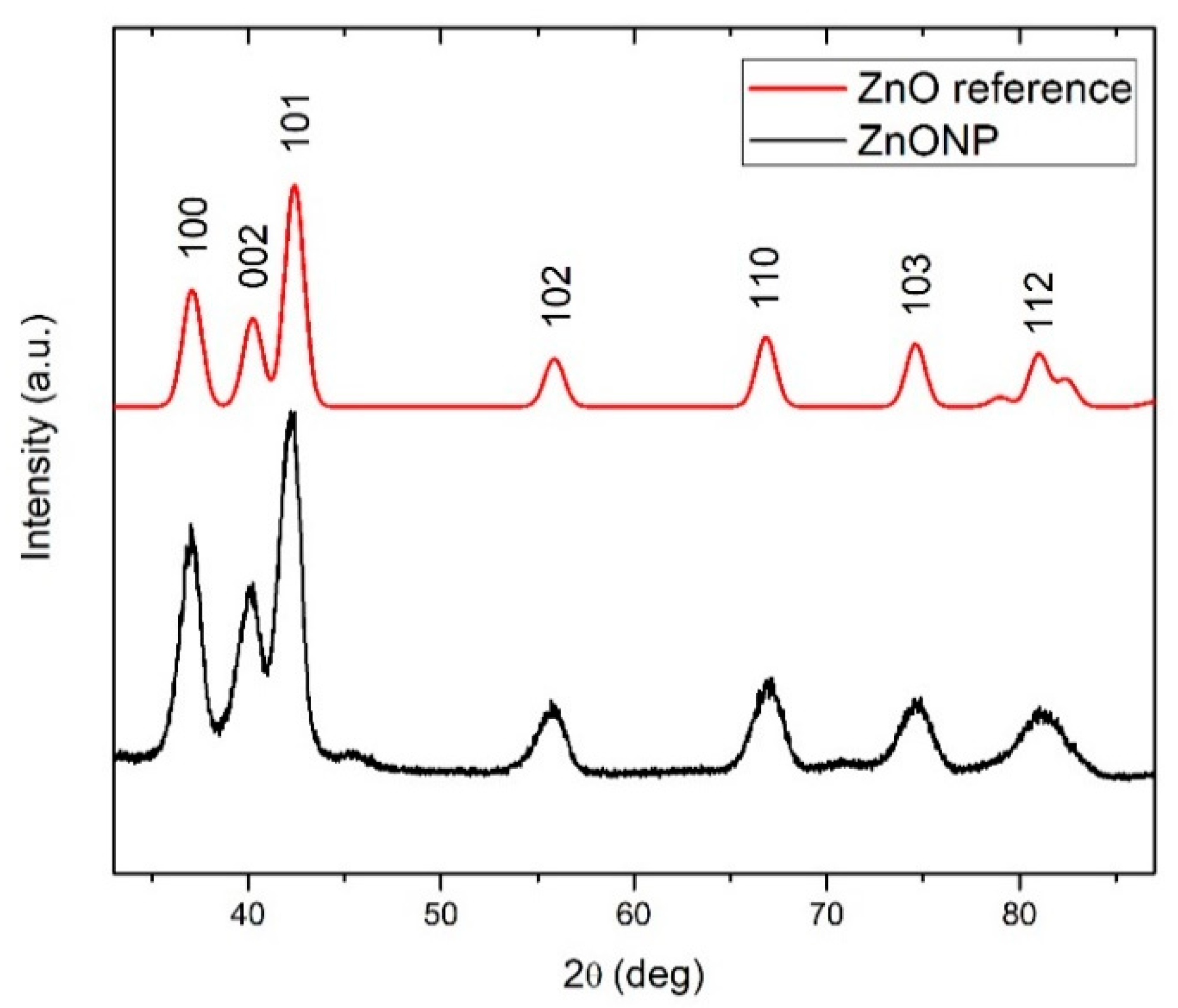
3.1.3. XRD and XPS Analyses
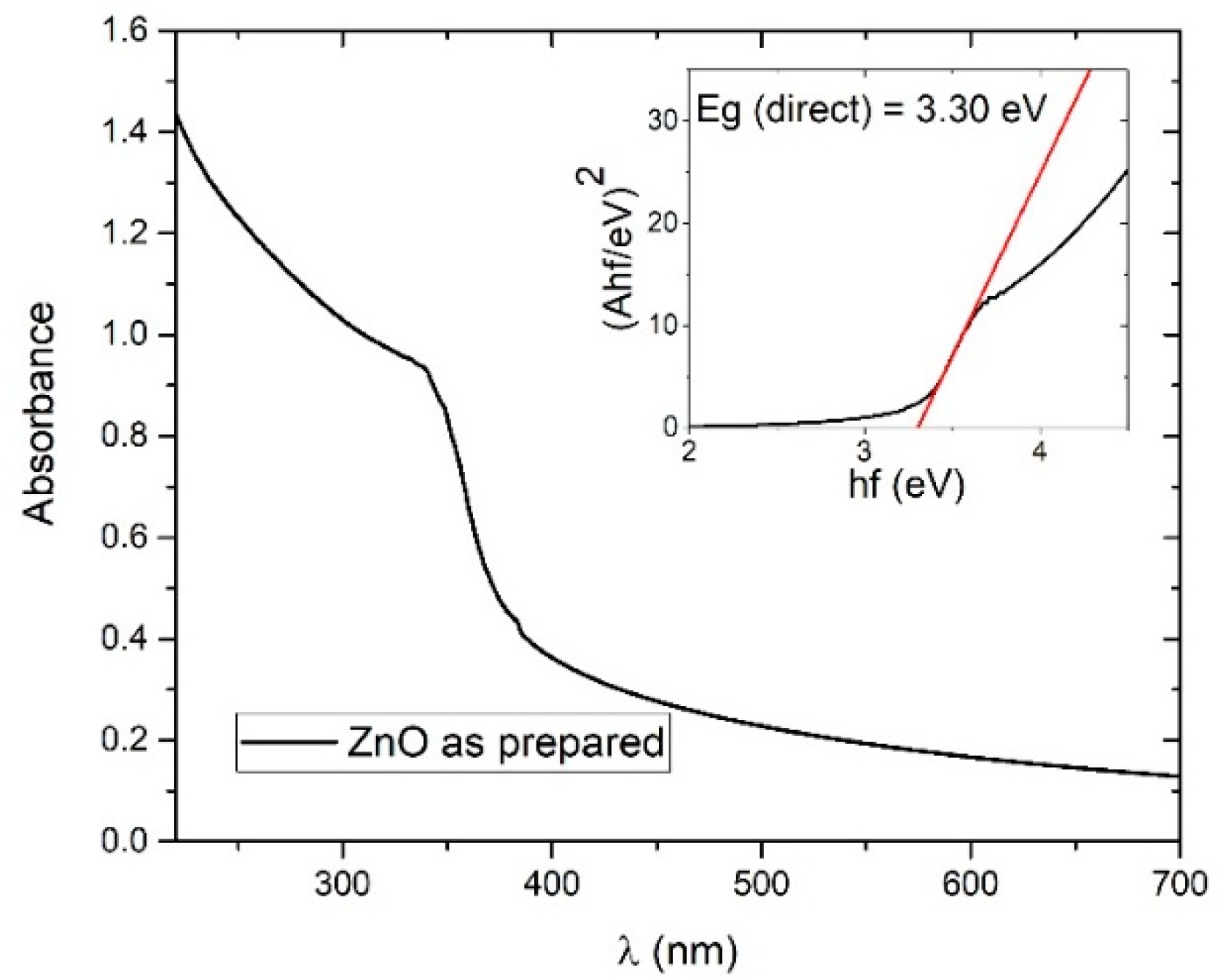
3.1.4. Bandgap Determination
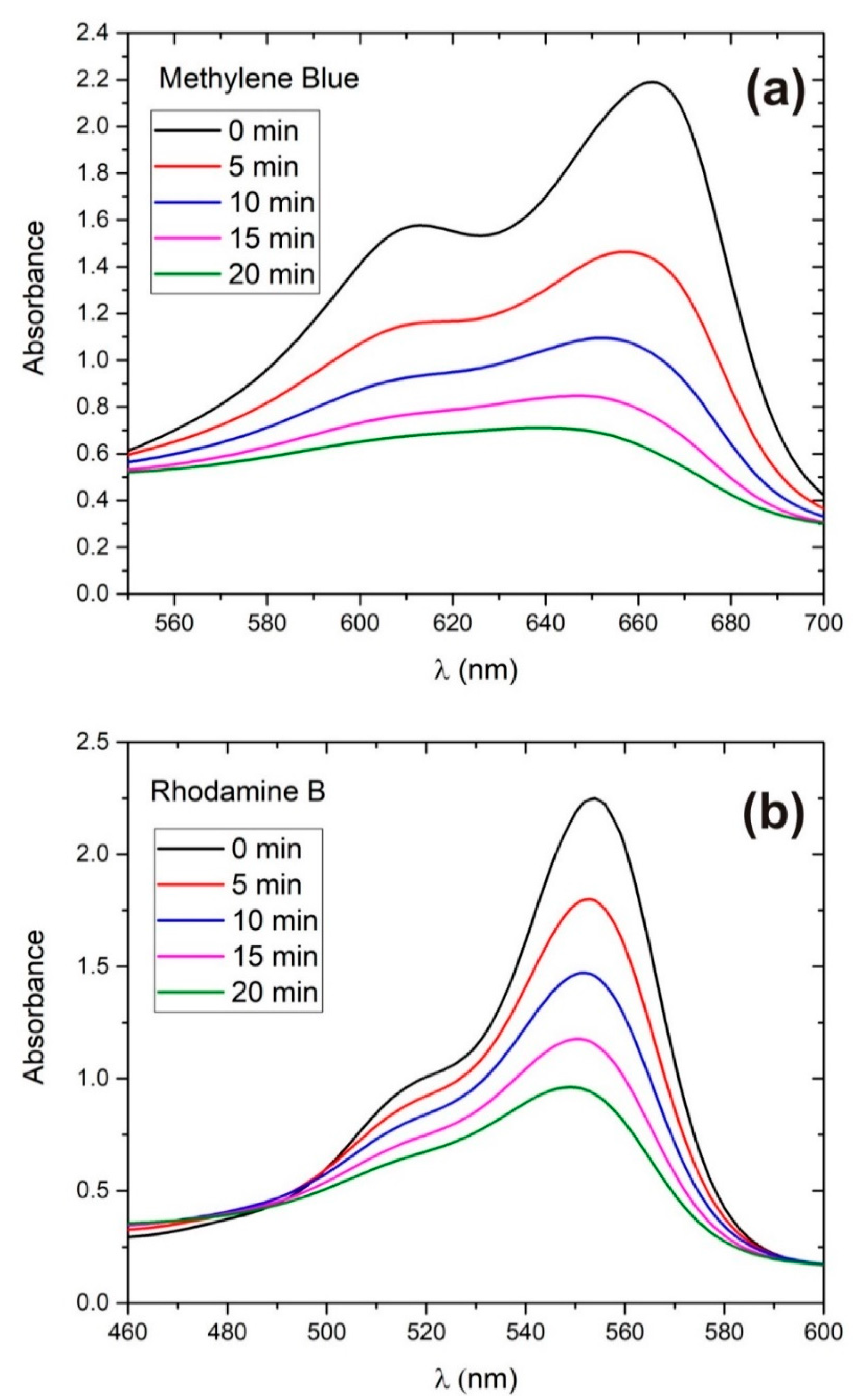
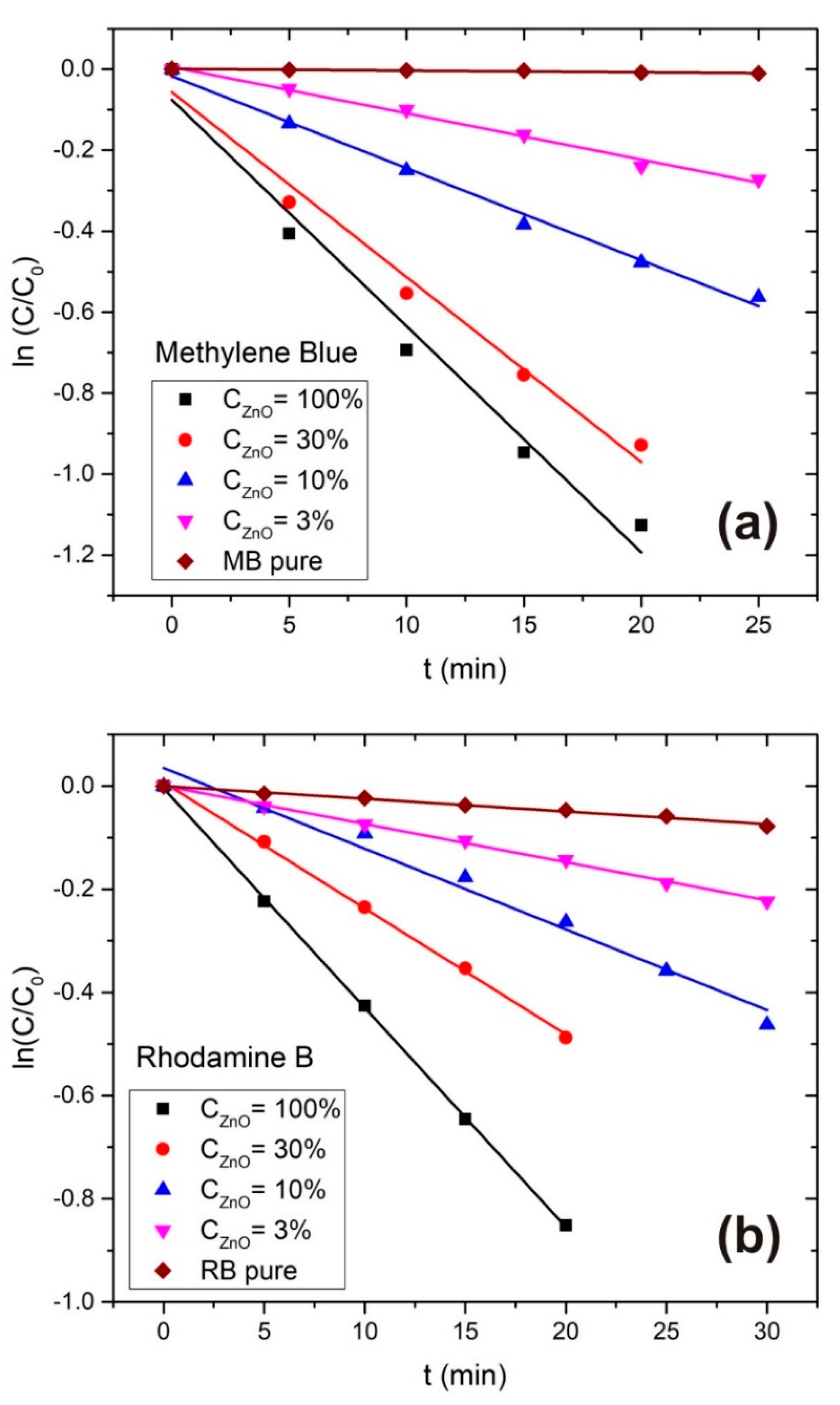
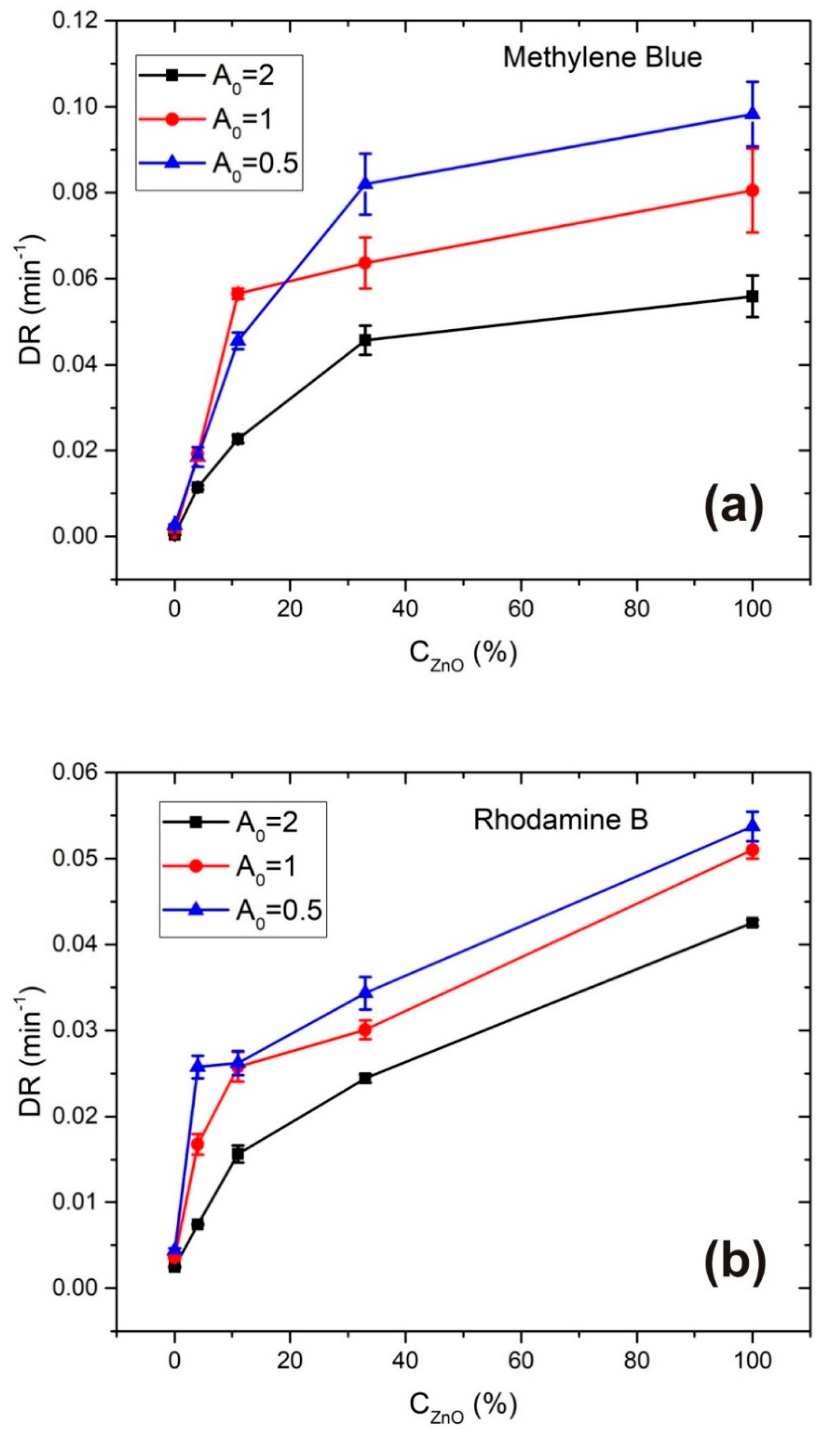
3.2. Photodegradation Rate Calculation
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Pathakoti, K.; Manubolum, M.; MinHwang, H. Nanotechnology applications for environmental industry. In Handbook of Nanomaterials for Industrial Applications; Elsevier: Amsterdam, The Netherlands, 2018; Chapter 48; pp. 894–907. [Google Scholar]
- Chakrabarti, S.; Dutta, B.K. Photocatalytic degradation of model textile dyes in wastewater using ZnO as semiconductor catalyst. J. Hazard. Mater. B 2004, 112, 269–278. [Google Scholar] [CrossRef] [PubMed]
- Qi, K.; Cheng, B.; Yu, J.; Ho, W. Review on the improvement of the photocatalytic and antibacterial activities of ZnO. J. Alloys Compd. 2017, 727, 792–820. [Google Scholar] [CrossRef]
- Song, L.; Wang, Y.; Ma, J.; Zhang, Q.; Shen, Z. Core/shell structured Zn/ZnO Nanoparticles Synthesized by Gaseous Laser Ablation with Enhanced Photocatalysis Efficiency. Appl. Surf. Sci. 2018, 442, 101–105. [Google Scholar] [CrossRef]
- Hariharan, C. Photocatalytic degradation of organic contaminants in water by ZnO nanoparticles: Revisited. Appl. Catal. A Gen. 2006, 304, 55–61. [Google Scholar] [CrossRef]
- Lee, K.M.; Lai, C.W.; Ngai, K.S.; Juan, J.C. Recent developments of zinc oxide based photocatalyst in water treatment technology: A review. Water Res. 2016, 88, 428–448. [Google Scholar] [CrossRef]
- Bae, D.; Seger, B.; Vesborg, P.C.K.; Hansen, O.; Chorkendorff, I. Strategies for stable water splitting via protected photoelectrodes. Chem. Soc. Rev. 2017, 46, 1933–1954. [Google Scholar] [CrossRef]
- Zhang, L.; Cheng, H.; Zong, R.; Zhu, Y. Photocorrosion Suppression of ZnO Nanoparticles via Hybridization with Graphite-like Carbon and Enhanced Photocatalytic Activity. J. Phys. Chem. C 2009, 113, 2368–2374. [Google Scholar] [CrossRef]
- Fu, H.; Xu, T.; Zhu, S.; Zhu, Y. Photocorrosion Inhibition and Enhancement of Photocatalytic Activity for ZnO via Hybridization with C60. Environ. Sci. Technol. 2008, 42, 8064–8069. [Google Scholar] [CrossRef]
- Tayebi, M.; Tayebi, A.; Lee, B.K. Photocorrosion suppression and photoelectrochemical (PEC) enhancement of ZnO via hybridization with graphene nanosheets. Appl. Surf. Sci. 2020, 502, 144–189. [Google Scholar] [CrossRef]
- Kim, H.; Yang, B.L. A polyaniline-coated ZnS/ZnO/FTO photoelectrode for improving photocorrosion prevention and visible light absorption. New J. Chem. 2019, 43, 16699–16705. [Google Scholar] [CrossRef]
- Barnes, R.J.; Molina, R.; Xu, J.; Dobson, P.J.; Thompson, I.P. Comparison of TiO2 and ZnO nanoparticles for photocatalytic degradation of methylene blue and the correlated inactivation of gram-positive and gram-negative bacteria. J. Nanopart. Res. 2013, 15, 1432. [Google Scholar] [CrossRef]
- Ong, C.B.; Ng, L.Y.; Mohammad, A.W. A review of ZnO particles as solar photocatalysts: Synthesis, mechanisms and applications. Renew. Sust. Energy Rev. 2018, 81, 536–551. [Google Scholar] [CrossRef]
- Ma, D.; Zhai, S.; Wang, Y.; Liu, A. Chuncheng Chen TiO2 Photocatalysis for Transfer Hydrogenation. Molecules 2019, 24, 330. [Google Scholar] [CrossRef] [PubMed]
- Mahmodi, G.; Sharifni, S.; Madani, M.; Vatanpour, V. Photoreduction of carbon dioxide in the presence of H2, H2O and CH4 over TiO2 and ZnO photocatalysts. Sol. Energy 2013, 97, 186–194. [Google Scholar] [CrossRef]
- Chang, X.; Wang, T.; Gong, J. CO2 photo-reduction: Insights into CO2 activation and reaction on surfaces of photocatalysts. Energy Environ. Sci. 2016, 9, 2177–2196. [Google Scholar] [CrossRef]
- Bae, D.; Seger, B.; Hansen, O.; Vesborg, P.C.K.; Chorkendorff, I. Durability testing of photoelectrochemical hydrogen production under day/night light cycled conditions. ChemElectroChem 2019, 6, 106–109. [Google Scholar] [CrossRef]
- Zeng, J.; Li, Z.; Peng, H.; Zheng, X. Core-shell Sm2O3@ZnO nano-heterostructure for the visible light driven photocatalytic performance. Colloids Surf. A 2019, 560, 244–251. [Google Scholar] [CrossRef]
- Ullah, R.; Dutta, J. Photocatalytic degradation of organic dyes with manganese-doped ZnO nanoparticles. J. Hazard. Mater. 2008, 156, 194–200. [Google Scholar] [CrossRef]
- Samadi, M.; Zirak, M.; Naseri, A.; Khorashadizade, E.; Moshfegh, A.Z. Recent progress on doped ZnO nanostructures for visible-light photocatalysis. Thin Solid Films 2016, 605, 2–19. [Google Scholar] [CrossRef]
- Whang, T.-J.; Hsieh, M.-T.; Chen, H.-H. Visible-light photocatalytic degradation of methylene blue with laser-induced Ag/ZnO nanoparticles. Appl. Surf. Sci. 2012, 258, 2796–2801. [Google Scholar] [CrossRef]
- Chauhan, A.; Rastogi, M.; Scheier, P.; Bowen, C.; Kumar, R.V.; Vaish, R. Janus nanostructures for heterogenous photocatalysis. Appl. Phys. Rev. 2018, 5, 041111. [Google Scholar] [CrossRef]
- Wang, P.; Huang, B.; Dai, Y. Myung-Hwan Whangbo Plasmonic Photocatalyst: Harvesting Visible Light with Noble Metal Nanoparticles. Phys. Chem. Chem. Phys. 2012, 14, 9813–9825. [Google Scholar] [CrossRef] [PubMed]
- Atabaev, T.S.; Molkenova, A. Upconversion optical nanomaterials applied for photocatalysis and photovoltaics: Recent advances and perspectives. Front. Mater. Sci. 2019, 13, 335–341. [Google Scholar] [CrossRef]
- Chen, C.; Liu, J.; Liu, P.; Yu, B. Investigation of Photocatalytic Degradation of Methyl Orange by Using Nano-Sized ZnO Catalysts. Adv. Chem. Eng. Sci. 2011, 1, 9–14. [Google Scholar] [CrossRef]
- Sirelkhatim, A.; Mahmud, S.; Seeni, A.; Kaus, N.H.M.; Ann, L.C.; Bakhori, S.K.M.; Hasan, H.; Mohamad, D. Review on Zinc Oxide Nanoparticles: Antibacterial Activity and Toxicity Mechanism. Nano Micro Lett. 2015, 7, 219–242. [Google Scholar] [CrossRef]
- Krstulović, N.; Umek, P.; Salamon, K.; Capan, I. Synthesis of Al-doped ZnO nanoparticles by laser ablation of ZnO: Al2O3 target in water. Mater. Res. Express 2017, 4, 105003. [Google Scholar] [CrossRef]
- Mintcheva, N.; Aljulaih, A.A.; Wunderlich, W.; Kulinich, S.A.; Iwamori, S. Laser-Ablated ZnO Nanoparticles and Their Photocatalytic Activity Toward Organic Pollutants. Materials 2018, 11, 1127. [Google Scholar] [CrossRef]
- Honda, M.; Goto, T.; Owashi, T.; Rozhin, A.G.; Yamaguchi, S.; Itoc, T.; Kulinich, S.A. ZnO nanorods prepared via ablation of Zn with millisecond laser in liquid media. Phys. Chem. Chem. Phys. 2016, 18, 23628. [Google Scholar] [CrossRef]
- Krstulović, N.; Salamon, K.; Budimlija, O.; Kovač, J.; Dasović, J.; Umek, P.; Capan, I. Parameters optimization for synthesis of Al-doped ZnO nanoparticles by laser ablation in water. Appl. Surf. Sci. 2018, 440, 916–925. [Google Scholar] [CrossRef]
- Zhang, D.; Liu, J.; Li, P.; Tian, Z.; Liang, C. Recent Advances in Surfactant-Free, Surface Charged and Defect-Rich Catalysts Developed by Laser Ablation and Processing in Liquids. ChemNanoMat 2017, 3, 512–533. [Google Scholar] [CrossRef]
- Jang, Y.J.; Simer, C.; Ohm, T. Comparison of zinc oxide nanoparticles and its nano-crystalline particles on the photocatalytic degradation of Methylene Blue. Mater. Res. Bull. 2006, 41, 67–77. [Google Scholar] [CrossRef]
- Rahman, Q.I.; Ahmad, M.; Misra, S.K.; Lohani, M. Effective photocatalytic degradation of Rhodamine B dye by ZnO nanoparticles. Mater. Lett. 2013, 91, 170–174. [Google Scholar] [CrossRef]
- Moulder, J.F.; Stickle, W.F.; Sobol, P.E.; Bomben, K.D. Handbook of X-Ray Photoelectron Spectroscopy; Physical Electronics Inc.: Eden Prairie, MN, USA, 1995. [Google Scholar]
- Krstulović, N.; Milošević, S. Drilling enhancement by nanosecond–nanosecond collinear dual-pulse laser ablation of titanium in vacuum. Appl. Surf. Sci. 2010, 256, 4142–4148. [Google Scholar] [CrossRef]
- Tseng, C.-A.; Lin, J.-C.; Weng, W.-H.; Lin, C.-C. Photoelectron spectroscopy and optical properties of Al-doped ZnO films prepared by sputtering with radio frequency power applied to Al target. Jpn. J. Appl. Phys. 2013, 52, 025801. [Google Scholar] [CrossRef]
- Plodinec, M.; Grčić, I.; Willinger, M.G.; Hammud, A.; Huang, X.; Panžić, I.; Gajović, A. Black TiO2 nanotube array decorated with Ag nanoparticles for enhanced visible-light photocatalytic oxidation of salycilic acid. J. Alloys Compd. 2019, 776, 883–896. [Google Scholar] [CrossRef]
- Čižmar, T.; Panžić, T.; Salamon, K.; Grčić, I.; Radetić, L.; Marčec, J.; Gajović, A. Low-Cost synthesis of Cu-Modified Immobilized Nanoporous TiO2 for Photocatalytic Degradation of 1H-Benzotriazole. Catalysts 2020, 10, 19. [Google Scholar] [CrossRef]
- Wu, C.-H.; Chern, J.-M. Kinetics of Photocatalytic Decomposition of Methylene Blue. Ind. Eng. Chem. Res. 2006, 45, 6450–6457. [Google Scholar] [CrossRef]
- Sakthivel, S.; Neppolian, B.; Shankar, M.V.; Arabindoo, B.; Palanichamy, M.; Murugesan, V. Solar photocatalytic degradation of azo dye: Comparison of photocatalytic efficiency of ZnO and TiO2. Sol. Energy Mater. Sol. Cells. 2003, 77, 65–82. [Google Scholar] [CrossRef]
- Kumar, M.; Sharma, S.; Kumar, D. Removal of methylene blue and rhodamine B from water by zirconium oxide/graphene. Water Sci. 2016, 30, 51–60. [Google Scholar]
- Balcha, A.; Yadav, P.O.; Dey, T. Photocatalytic degradation of methylene blue dye by zinc oxide nanoparticles obtained from precipitation and sol-gel methods. Environ. Sci. Pollut. Res. Int. 2016, 23, 25485–25493. [Google Scholar] [CrossRef]
- Abdellah, M.H.; Nosier, S.A.; El-Shazly, A.H.; Mubarak, A.A. Photocatalytic decolorization of methylene blue using TiO2/UV system enhanced by air sparkling. Alex. Eng. J. 2018, 57, 3727–3735. [Google Scholar] [CrossRef]



| A0 | CZnO | MB | RB | ||||
|---|---|---|---|---|---|---|---|
| DR (min−1) | R2 | t1/2 (min) | DR (min−1) | R2 | t1/2 (min) | ||
| 2 | C100% | 0.0559 | 0.9784 | 12.4 | 0.0425 | 0.9998 | 16.3 |
| C30% | 0.0457 | 0.9838 | 15.2 | 0.0244 | 0.9988 | 28.4 | |
| C10% | 0.0227 | 0.9934 | 30.5 | 0.0156 | 0.9793 | 44.3 | |
| C3% | 0.0114 | 0.9916 | 60.8 | 0.0074 | 0.9983 | 93.5 | |
| pure dye | 0.0004 | 0.9350 | 1733 | 0.0025 | 0.9899 | 281 | |
| 1 | C100% | 0.0805 | 0.9855 | 8.6 | 0.0510 | 0.9855 | 13.6 |
| C30% | 0.0636 | 0.9914 | 10.9 | 0.0301 | 0.9914 | 23.1 | |
| C10% | 0.0565 | 0.9991 | 12.3 | 0.0258 | 0.9991 | 26.9 | |
| C3% | 0.0192 | 0.9872 | 36.1 | 0.0168 | 0.9872 | 41.4 | |
| pure dye | 0.0010 | 0.9844 | 673 | 0.0036 | 0.9863 | 193 | |
| 0.5 | C100% | 0.0983 | 0.9942 | 7.1 | 0.0538 | 0.9979 | 12.9 |
| C30% | 0.0820 | 0.9925 | 8.5 | 0.0343 | 0.9942 | 20.2 | |
| C10% | 0.0456 | 0.9964 | 15.2 | 0.0262 | 0.9914 | 26.4 | |
| C3% | 0.0185 | 0.9709 | 37.5 | 0.0258 | 0.9927 | 26.9 | |
| pure dye | 0.0026 | 0.9688 | 266 | 0.0044 | 0.9800 | 159 | |
| Dye Abs. Maxima | C100% | C30% | ||
|---|---|---|---|---|
| A0 | DR (MB)/min−1 | DR (RB)/min−1 | DR (MB)/min−1 | DR (RB)/min−1 |
| 2 | 0.056 | 0.043 | 0.046 | 0.024 |
| 1 | 0.081 | 0.051 | 0.064 | 0.030 |
| 0.5 | 0.098 | 0.054 | 0.082 | 0.034 |
| DR(A0 = 0.5)/DR (A0 = 2) | 1.75 | 1.26 | 1.78 | 1.42 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blažeka, D.; Car, J.; Klobučar, N.; Jurov, A.; Zavašnik, J.; Jagodar, A.; Kovačević, E.; Krstulović, N. Photodegradation of Methylene Blue and Rhodamine B Using Laser-Synthesized ZnO Nanoparticles. Materials 2020, 13, 4357. https://doi.org/10.3390/ma13194357
Blažeka D, Car J, Klobučar N, Jurov A, Zavašnik J, Jagodar A, Kovačević E, Krstulović N. Photodegradation of Methylene Blue and Rhodamine B Using Laser-Synthesized ZnO Nanoparticles. Materials. 2020; 13(19):4357. https://doi.org/10.3390/ma13194357
Chicago/Turabian StyleBlažeka, Damjan, Julio Car, Nikola Klobučar, Andrea Jurov, Janez Zavašnik, Andrea Jagodar, Eva Kovačević, and Nikša Krstulović. 2020. "Photodegradation of Methylene Blue and Rhodamine B Using Laser-Synthesized ZnO Nanoparticles" Materials 13, no. 19: 4357. https://doi.org/10.3390/ma13194357
APA StyleBlažeka, D., Car, J., Klobučar, N., Jurov, A., Zavašnik, J., Jagodar, A., Kovačević, E., & Krstulović, N. (2020). Photodegradation of Methylene Blue and Rhodamine B Using Laser-Synthesized ZnO Nanoparticles. Materials, 13(19), 4357. https://doi.org/10.3390/ma13194357






