1. Introduction
Device-associated infections are one of the most common complications in medical practice. Although the development of smaller and continuous-flow devices has improved survival and reduced infection rates, one of the major causes of mortality and morbidity in these patients remains infection [
1]. The treatment of medical device-related infections is challenging. The main reason is that microorganisms can adhere to the surfaces of devices and may form biofilms, i.e., aggregations between microorganisms, leading to differentiated growth, structural change and reduced protection from antimicrobial agents and different host immune mechanisms, when compared to planktonic microorganisms [
2]. Once the biofilm develops it is very difficult to eradicate and the considerable cost of treatments can be due to the frequent need for prolonged hospitalization, surgery and long-term antibacterial therapy. It is reported that at least 1.7 million of annually nosocomial infections in United States are associated with biofilms and the main bacteria causing these infections worldwide are of the
Staphylococcus species and multidrug-resistant Gram-negative bacteria such as
Escherichia coli, Klebsiella pneumoniae,
Acinetobacter baumannii and
Pseudomonas aeruginosa, which is even responsible for catheter-associated urinary tract infections [
2]. However, bacterial adhesion can be affected by surface properties [
3,
4].
Polymers are now widely used for medical device manufacturing: their easy processability, excellent mechanical properties and chemical stability make them the best candidate to be used in several biomedical applications, including catheters, different types of probe, breast implants, heart valves and vascular prostheses [
5,
6,
7]. Polymers have a wide range of properties and the possibility to adapt their surface characteristics to different biological environments has attracted the attention of biomedical device developers. By applying specific surface modification techniques (employing biological, chemical or physical methods), it is possible to properly tailor morphological or chemical surface properties [
8,
9,
10]. It must be noted, however, that some of these procedures, such as chemical modification, etching or roughening, could lead to weakened mechanical properties of the bulk material and/or loss of specific surface functionality.
In this respect, coating technology offers an interesting alternative to manufacturing hygienic surfaces which are capable of limiting bacterial contamination [
11]. The deposition of a thin polymeric top layer starting from an opportune solution/dispersion of antibacterial components in a specific liquid system, is both easy to achieve, from a manufacturing point of view, and ensures that the coating is efficiently charged. This efficiency derives from the possibility to concentrating the antibacterial charge on the surface. Compared to other procedures used to incorporate antibacterial agents (i.e., melt compounding [
12,
13,
14,
15,
16], where antibacterial agents are trapped between layers of the material constituting the device and are not all exposed to the surface) better antibacterial properties can be achieved. This maximized surface/charge ratio is obtained by starting from a lower amount of charged species and by keeping the processing simple, versatile and low-cost. Moreover, an extended life-time of the devices can be achieved due to the polymeric component of the coating which acts as a rate-limiting barrier and heightens performance in different environments by fabricating coatings with multiple antibacterial functions [
17]. However, a key aspect to consider in the formulation is the deposition technique. Spray and dip coating depositions are the main techniques employed for such devices and therefore the dispersion should possess a series of characteristics which would ensure the physical and chemical properties required for their deposition and solidification over the selected substrate [
18,
19]. The formulation of the coating ultimately depends on the chemical compatibility between the polymer matrix and the antibacterial agent, in view of the desired activity and to prevent release contamination. The type of antibacterial activity can be selected choosing from one of these three antibacterial families: antibacterial agent release, contact killing and anti-adhesion/bacteria-repelling [
20,
21,
22,
23].
Preparation of polymeric coatings with chemically anchored contact-killing biocides or the use of a low amount of inorganic compound results in less compatibility problems. For this reason, silver nanoparticles (AgNP) represent an interesting way to prevent the rapid and excessive release of ions by ensuring a high level of antibacterial efficiency [
24]. Although pure metal particles, including silver, have been extensively used for different bacterial strains, a number of disadvantages have been observed. From a processing point of view, metal particles tend to aggregate easily when mixed in a polymer matrix making their production feasible within a specific range of filler amount which, as a consequence, can have a significant impact on antibacterial activity.
Alternatively, coatings dispersions can be formulated to modify the surface energy and promote an antibiofouling effect. For example, an increased hydrophobicity results in a “slippery” surface which is capable of minimizing bacterial adhesion and biofilm formation of several bacterial strains like
P. aeruginosa,
S. aureus, and
E. coli [
25].
Because each antibacterial species/matrix system has unique properties, careful examination of the specific adhesion requirements for targeted application is needed. The performance and lifetimes of the coatings are strongly dependent not only on the intrinsic properties of the materials, but also on adhesion between the coating and substrate. Chemical and physical intermolecular interactions between the substrate and the top coatings help to prevent adhesion failure and ensure long-term durability [
26,
27].
To address these issues and to develop innovative coatings with specific antibacterial functions, we selected silver, chitosan and titanium dioxide. These antibacterials have already used as fillers for thermoplastic polyurethane (TPU)-based composites by us in a previous work [
12]. Here, we use them to prepare TPU-based dispersions/solutions through a simple and versatile preparation method to produce efficient antibacterial coatings. This allows for a direct comparison between alternative preparation techniques (i.e., melt processing and post-processing).
2. Materials and Methods
2.1. Materials
The Estane® 58887NAT 036, a thermoplastic polyurethane (TPU) of biomedical grade characterized by an excellent resistance to hydrolysis with excellent performance at low temperature and clarity, was supplied by Lubrizol (Wickliffe, OH, USA). Silver Nitrate (AgNO3), titanium dioxide anatase nanopowder (TiO2) with a size distribution ≤25 nm and low molecular weight chitosan (CHIT) with an average molecular weight of 50–190 kg/mol (based on viscosity) and a degree of deacetylation of 75–85%, were purchased by Sigma-Aldrich, Milano, Italy. Waterborne polyurethane (WPU) dispersed in water, with a weight-average molecular weight of 9200 g/mol and polydispersity of 17.3 was used.
2.2. Preparation of the Mother Liquors
Silver nanoparticle (AgNP) dispersions were prepared following these steps: 10 mL of tetrahydrofuran (THF, Sigma-Aldrich, Milano, Italy) was employed to dissolve 600 mg of TPU in order to have a TPU solution (60 mg/mL) while 2 solutions of aqueous AgNO3 were prepared with concentrations of 5 and 25 mM, respectively. Typically, 1 mL of each aqueous AgNO3 solution was added to a TPU solution and left to react under sunlight for 30–60 min in order to promote AgNP formation. TiO2 dispersions were similarly prepared by dispersing 3 mg of this material in TPU/THF solutions (60 mg/mL). Chitosan solutions with a concentration of 10 mg/mL were prepared by dissolving the compound in an aqueous solution (1% v/v) of acetic acid (Sigma-Aldrich, Milano, Italy).
In order to avoid the use of THF, necessary for the dissolution of the TPU, and to develop a more ecofriendly system, some experiments were performed starting with waterborne polyurethane (dispersed in water). The addition of 1 mL of AgNO3 aqueous solution 25 mM to 10 mL of WPU was performed.
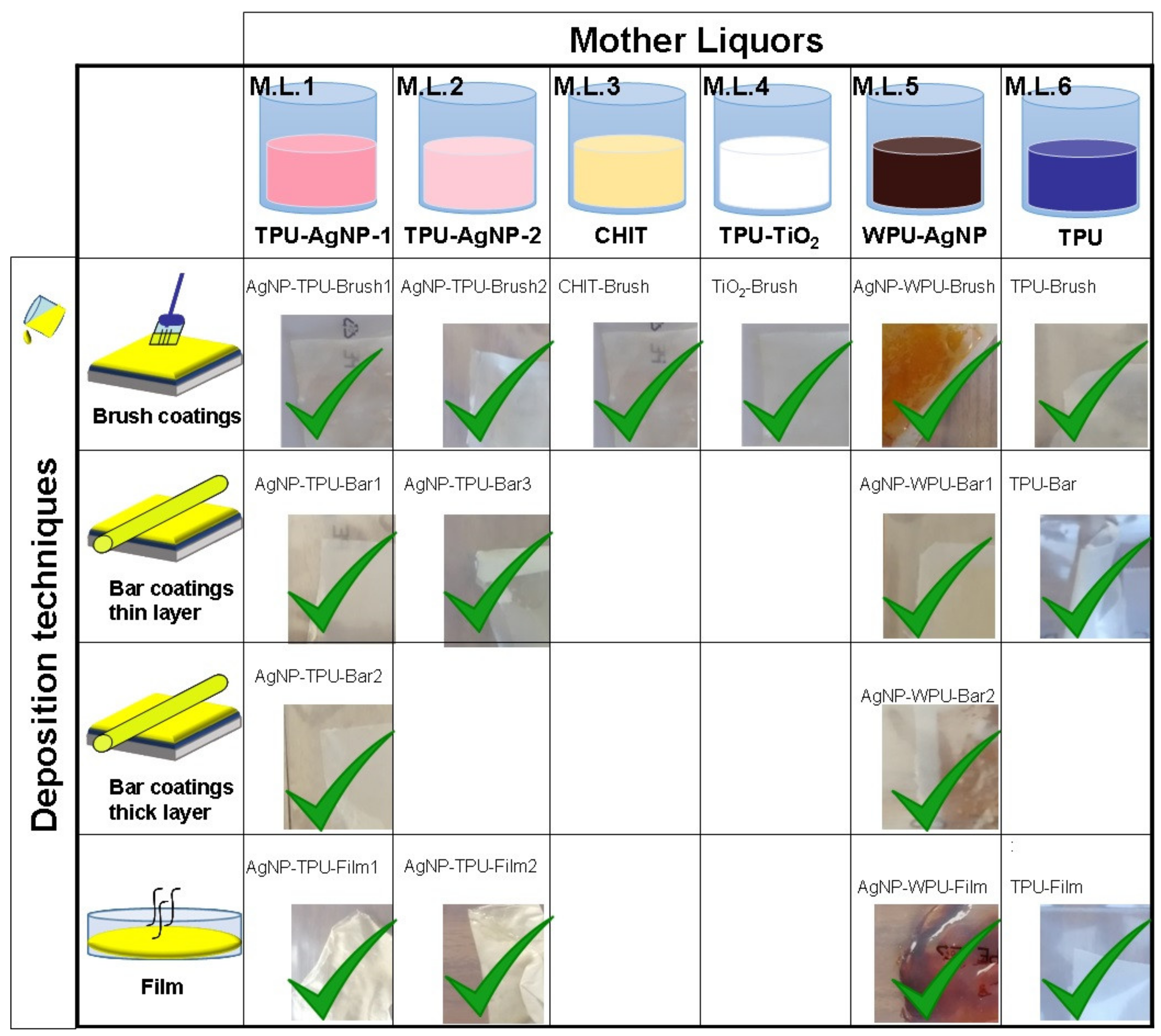
In this way, it was possible to prepare the following antibacterial mother liquors (M.L.) (
Figure 1): M.L.1 = TPU-AgNP-1 0.04 wt %; M.L.2 = TPU-AgNP-2 0.008 wt %; M.L.3 = CHIT 1 wt %; M.L.4 = TPU-TiO
2 0.03 wt %; M.L.5 = WPU-AgNP (all the weight percentages refer to the antibacterial content). A TPU–THF solution was treated under identical conditions for reference purposes (M.L.6). A schematic representation of the general procedure used for the preparation of the M.L. is shown in
Figure S1 of the Supporting Information part.
2.3. Preparation of the Coatings
TPU films with fixed dimensions (5 × 5 cm
2) were prepared by compression molding following the procedure presented in our previous work [
12] and used as substrates for the coating employed.
Coatings from M.L.1 to 6 (all the mother liquors) were prepared by dropping circa 3 mL of each dispersion on TPU compression molded films. To ensure a uniform thickness for the coatings and in order to distribute them over the entire substrates, we made use of a brush to spread the dispersions (
Figure 1). A similar amount (3 mL) of M.L.1, M.L.2 and M.L.5 was used to cast films of the respective materials, used to compare their antibacterial activity with the AgNP-based coating activities. A TPU film from M.L.6 was also prepared as a reference sample (
Figure 1).
Another set of samples was prepared by using 3 mL of M.L.1, M.L.2, M.L.5 and M.L.6, respectively, and depositing them by a bar coater (TQC Sheen Automatic Film Applicator, Capelle aan den Ijssel, The Netherlands). TPU-based coatings were deposited at a rate of 50 mm/s with a “wet” thickness of 100 μm. A slower deposition rate (15 mm/s) was used for WPU-based coatings imposing a temperature of 60 °C through the use of a heating plate due to the different viscosity of this system. The same amount of M.L.1 and M.L.5 was also bar coated imposing a “wet” thickness of 200 μm. The curing of the coated substrates was carried out at room temperature at least for 4 h (
Figure 1).
2.4. Characterization
The molecular, structural, thermal, mechanical and rheological properties together with surface morphology characteristics of the TPU have been widely reported in our previous work [
12].
UV absorption spectra as a function of exposure time to ultraviolet light (curves taken every 15 s of exposure) were recorded using a PerkinElmer (Waltham, MA, USA) Lambda 900 spectrometer, for two samples at different Ag precursor concentration (25 mM and 50 mM). UV curing was carried out using a BlueWave 200 UV Light-Curing Spot Lamp at 4500 mW/cm2 (Hamamatsu Photonics, Arese, Italy).
UV Absorption spectra as a function of exposure time to sun light were recorded using a PerkinElmer (Waltham, MA, USA) Lambda 900 spectrometer, for a M.L.1 dispersion using a quartz cell and for a casted film prepared by the M.L.5 dispersion.
Contact angle (CA) measurements were performed using a CAM 200 from KSV Instruments Ltd. (Helsinky, Finland). Typically, a 10 μL drop of water was deposited on the surface of each prepared film to determine the static contact angles, respectively.
To capture high-resolution images and obtain elemental maps of the atomic elements of the different film and coatings, a scanning electron microscope (SEM) Zeiss EVO-MA10 (Carl Zeiss, Oberkochen, Germany) coupled to an energy dispersive X-ray spectroscopy (EDS) detector (X-max 50 mm2, Oxford Instruments, Oxford, UK) was utilized. Acceleration voltage used was 20 kV. Samples were only gold sputtered for SEM images acquisition.
2.5. Antibacterial Tests
2.5.1. Bacterial Strains and Culture Conditions
The microorganisms used were
Escherichia coli ATCC 25922 (
E. coli) and
Staphylococcus aureus ATCC 25923 (
S. aureus), kindly supplied by R. Migliavacca (Department of Clinical Surgical, Diagnostic and Pediatric Sciences, University of Pavia, Italy). Bacteria were grown in their appropriate medium, overnight, under aerobic conditions at 37 °C using a shaker incubator (VDRL Stirrer 711/CT, Asal Srl, Italy):
E. coli was inoculated in Luria Bertani broth (LB) (ForMedium
TM, Hunstanton, Norfolk, UK) whereas
S. aureus in BHI (Brain Heart Infusion broth) (Scharlab S.L., Sentmenat, Barcelon, Spain). Both cultures were reduced to a final density of 1 × 10
10 cells/mL as determined by comparing the optical density (OD600) of the sample with a standard curve relating OD600 to cell number [
28].
2.5.2. Bacterial Viability
Materials were sterilized with ethanol 70%, washed twice in sterile ddH2O and incubated with 1 × 105/sample. The inoculation was carried out for direct contact and indirect contact. The viability was assessed with 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) colorimetric assay (Sigma-Aldrich, St. Louis, SM, USA) on bacteria in direct contact and on bacterial suspension in indirect contact. After 3 h of incubation, at 37 °C, the MTT reaction was stopped by adding solution C (2-propanol, HCl 0.04 N), and further incubated for 15 min at 37 °C. The colorimetric reaction was analyzed at CLARIOstar (BMG Labtech, Ortenberg, Germany) at 570 nm wavelength with 630 nm as reference wavelength. Results firstly normalized to Tissue Culture Plate (TCP) and then to TPU set as 100%. The experiment was carried out in triplicate and repeated 2 times.
2.5.3. SEM Analysis
Bacteria were incubated on TPU polymeric films for 24 h at 37 °C. The films were washed carefully with phosphate buffer solution (PBS) 1X and fixed with 2.5% (v/v) glutaraldehyde (Sigma-Aldrich, St. Louis, SM, USA) in 0.1 M Na-cacodylate buffer (Sigma-Aldrich, St. Louis, SM, USA) (pH 7.2), for 1 h at 4 °C. After two washes with Na-cacodylate, to remove excess of glutaraldehyde, S. aureus samples were dehydrated using increasing concentrations of ethanol (25, 50, 75%) for 5 min and two washes of 96% ethanol for 10 min. E. coli samples were dehydrated just with two washes of 96% ethanol (Merck Life Science S.r.l., Milano, Italy) for 10 min. The samples were lyophilized for 3 h using a K-850 apparatus (Emitech Ltd, Ashford, UK) and placed on a mounting base. Finally, TPU films were sputter coated with gold and investigated using a Zeiss EVO-MA10 scanning electron microscope (Carl Zeiss, Oberkochen, Germany), 20 kV acceleration voltage.
2.5.4. Statistical Analysis
All the statistical calculations related to antibacterial tests were carried out using GraphPad Prism 5.0 (GraphPad Inc., San Diego, CA, USA). Statistical analysis was performed using Student’s unpaired, two-sided t-test (significance level of p ≤ 0.05).
2.6. Adhesion Tests
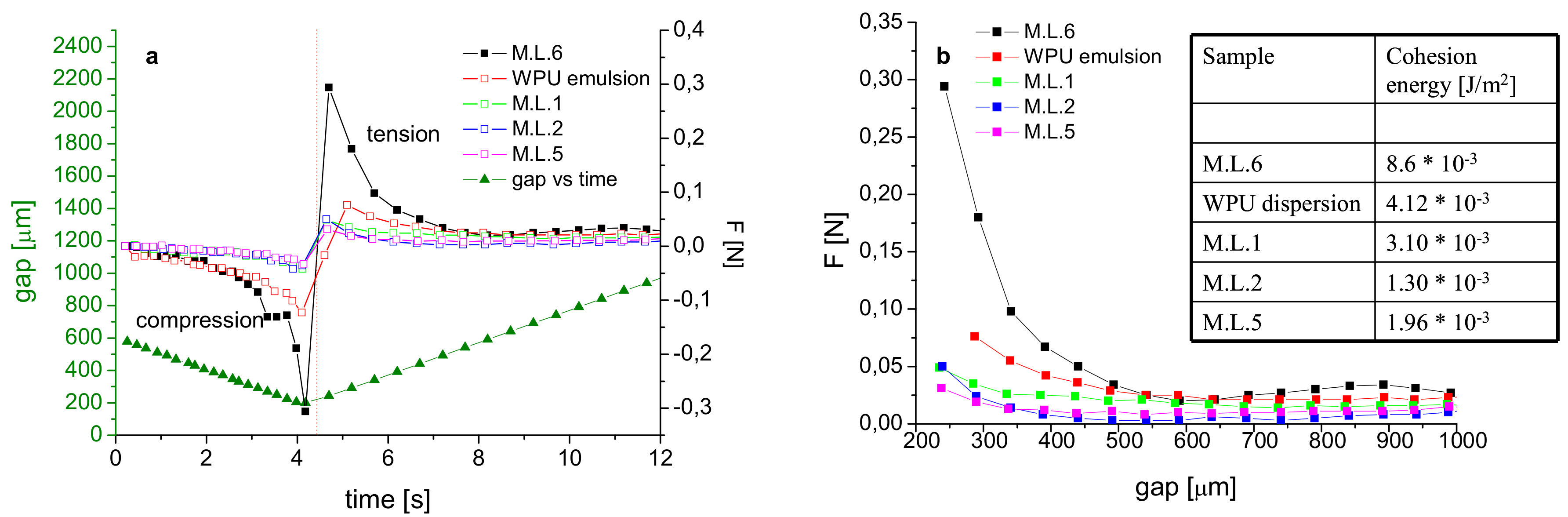
The bond strength measurements were performed using a rotational rheometer AR 2000 from TA Instruments (New Castle, DE, USA) in parallel-plate geometry. The load cell has a force resolution of 1 mN, while the stepper motor can resolve distances as small as 0.001 mm. All the experiments were performed at 10 °C by using a Peltier temperature controller (TA Instruments New Castle, DE, USA). The experiment consisted of two steps: (1) a compression with upper plate moving at a constant velocity (0.1 mm/s) in the downward direction, (2) at the fixed gap (0.4 mm), the motion of the upper plate is reversed at the same speed, leading to a position sufficient to guarantee cohesive failure.
4. Discussion
As in our previous work [
12], we elected to use silver, titanium dioxide and chitosan for their recognized antibacterial properties. In the present study, however, the use of these compounds in the form of coatings is presented with the idea of providing a direct comparison with alternative preparation techniques (i.e., melt processing and post-processing). For this reason, different antibacterial M.L.s were prepared and used for the preparation of film and coatings (
Figure 1). Besides the solvent casted films, which turned out to be very efficient and good reference samples, coatings from the different M.L.s were prepared on TPU substrates by using a brush in the first set of experiments and a film applicator successively. The bar coater was used with the idea of allowing a more homogeneous deposition of the coating. However, coatings with a thickness of 10–20 μm were obtained and only the employment of a 200 μm bar for some selected samples allowed to obtain thicker films comparable with the brushed one (~50 μm). For M.L.3 and 4 we used the same antibacterial materials, simply dissolved or dispersed in an opportune liquid, a different strategy was employed for the silver-based film and coatings.
We used a silver salt precursor to promote nanoparticle formation and create more efficient antibacterial materials with less possible cytotoxicity problems caused by silver release. This comes from the unique properties of metal nanoparticles, due to their high surface/volume ratio. It is in this sense that, silver nanoparticles, in view of their “easy” preparation via economic and sustainable methods, are becoming more interesting especially for use in biomedical applications. Several preparation methods have been reported [
24,
32,
33,
34,
35]. These preparation methods are carried out using a series of components whose control allows to influence the size and shape of the AgNP. Usually, this occurs through silver(I) precursor reduction and consists of two steps: nucleation and growth. The reduction requires the use of a reducing agent capable of promoting silver metallic formation. Since the metal form is not thermodynamically stable, especially in aqueous or alcoholic solution used, a stabilizer is used to prevent a further oxidation of the obtained metal. Stabilizers not only contrast the high reduction potential of silver protecting the nanoparticles but prevent agglomeration and influence their size and shape, playing a determinant role in the nucleation and growth stages. For this reason, several different methods have been developed to examine not only different reducing agents, but also several stabilizers [
24].
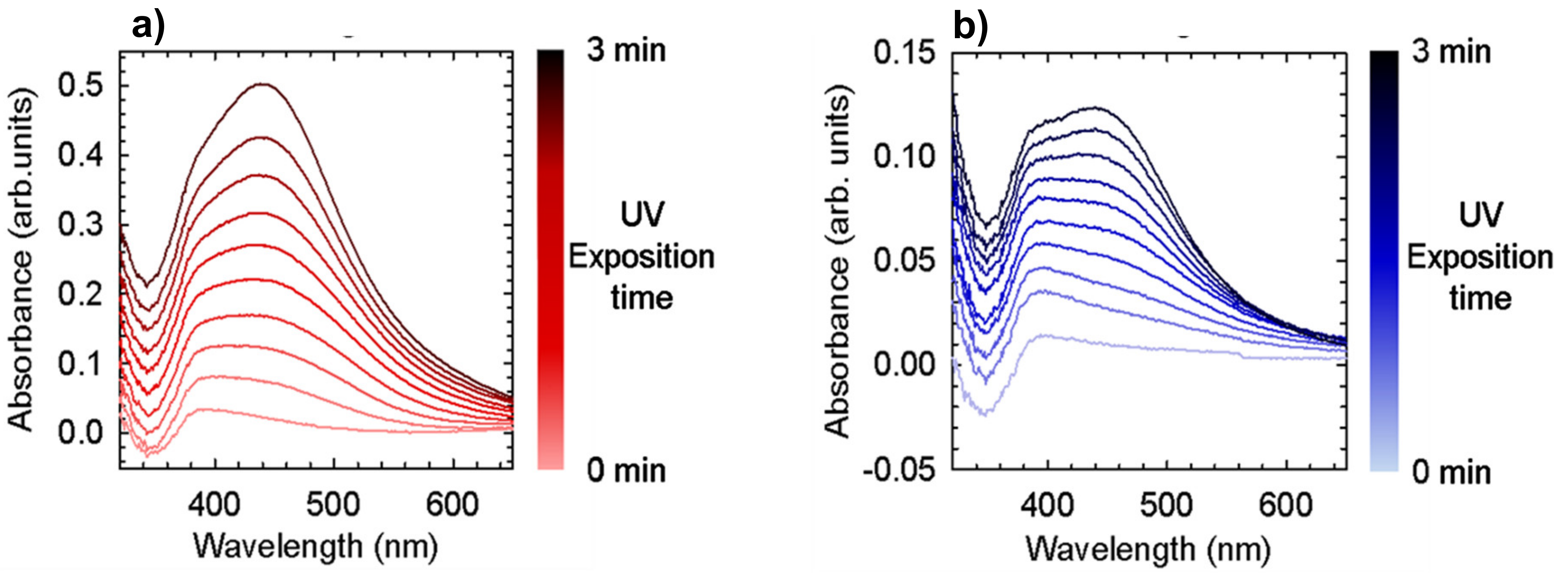
In our study, we introduced TPU as reducing agent and stabilizer. The electron rich groups (i.e., carbonilic, carbossilic, amminic and aromatic groups) constituting the TPU polymer chain serve as reducing agents to promote silver precursor reduction. The polymeric nature of TPU ensures the stabilizing effect for nanoparticle stability. The observed colour variation of M.L.1, 2 and 5 after sunlight exposition (
Figure 2) and the characteristic surface plasmon resonance band of Ag nanoparticles (
Figure 3), whose intensity depends on silver precursor concentration and UV exposition, confirm the nanoparticle formation. To the best of our knowledge, this is the first time that silver nanoparticle formation has occurred through the use of a single component which is able to guarantee both functions. We have only found a work where a similar strategy was utilized, in particular N,N-dimethylformamide (DMF) was used as a solvent and reducing agent, and polyurethane as a structure-directing agent [
36]. However, we successfully used this strategy with other polymeric systems having similar required characteristics but dispersed or dissolved in different solvents (i.e., WPU and chitosan, see also
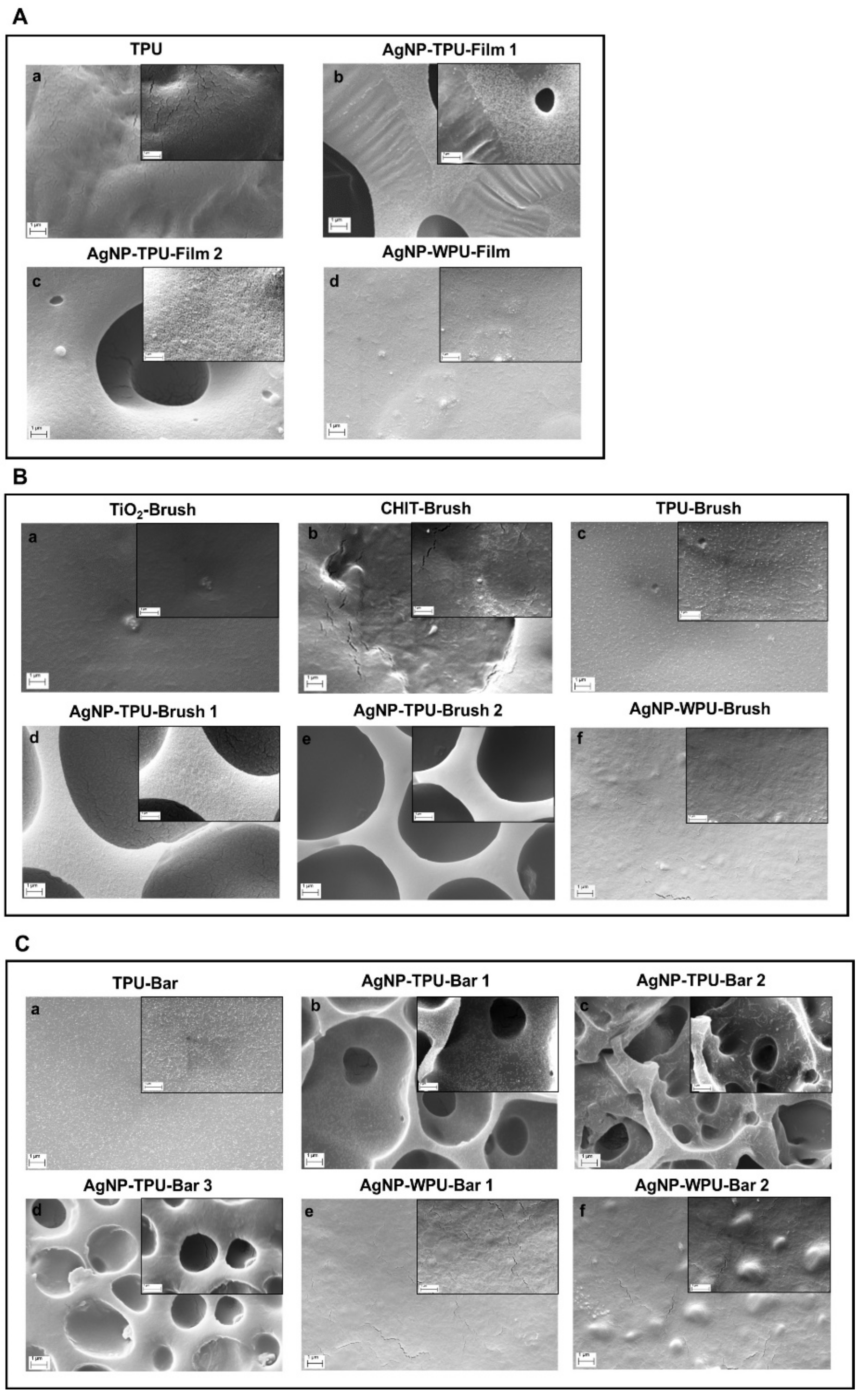
Figure S11), showing how AgNP formation is independent from the solvent employed. Here, we only report a comparison with a WPU, highlighting the potentiality sustainability of this method. Furthermore, even in this process where a single component is used for AgNP formation, the shape and size of the nanoparticles can be triggered and controlled by modulating the exposure to light or temperature. Silver nanoparticles, with an average dimension of ~20–50 nm, have been observed for the different systems considered. As observed in the SEM images reported in this paper, their presence and dispersion can be affected by the precursor concentration and the deposition technique. EDS maps of the samples confirm these data, highlighting the role played by the porous morphology in AgNP detection (
Figure 6). The low concentration of the silver precursor allows to detect AgNP presence only for porous materials, where the amount of detectable silver is inevitably higher. AgNP presence, also expected for WPU-Film is not always observed. In particular, we manage to highlight, by SEM (
Figure 4b), the presence of silver nanoparticles over the film prepared by M.L.5, avoiding the sterilization, whereas the low concentration of the silver precursor prevents their detection by EDS (
Figure 6b), in the absence of porous morphology. Finally, the versatility of this preparation method could promote the use of AgNP in many fields of applications, especially those for which a well dispersed and low concentrated amount of nanoparticles is requested [
37].
The film morphology and the observed porosity derive from the choice of the polymer and the solvents used for M.L. preparation. Many methods for the preparation of porous structures have been used and reported in the literature, from electrostatic spinning to laser ablation techniques or gas flow [
38,
39,
40]. However, one of the most used and interesting is the phase-inversion process, a method where a homogeneous polymer-solution is put into contact with a nonsolvent, resulting in phase separation responsible of the porous formation [
29,
41]. The separation into a polymer-rich phase and polymer-poor phase is guaranteed by the exchange of solvent/nonsolvent at the interface. The final morphology of the film obtained is regulated by several parameters correlated to the kinetics and the thermodynamics of the phase separation and therefore many equilibrium phase diagrams for different relevant systems were built as a useful tool for morphology control [
29,
42].
Two different phase separation processes have been observed and are competitive during membrane formation: solid–liquid (S–L) demixing and liquid–liquid (L–L) demixing. (S–L) demixing, or crystallization, leads to membranes with a spherulitic or axialitic morphology, whereas (L–L) demixing to porous structure [
29]. At lower polymer concentrations the typical cellular morphology is observed which is a result of a diffusion-induced phase separation process typical of (L–L) demixing [
29,
42]. Similarly in our AgNP-TPU systems, being at low polymer concentration (5.6 wt %) with about 9 wt % of water and neglecting AgNO
3 content (0.04 wt %), a porous morphology was obtained for our “ternary system” independently from the casting or deposition technique employed. Differently from what is reported in the literature, where an immersion precipitation process through the use of a coagulant bath is used until the exchange of solvent and nonsolvent is completed, our system consists of a one pot preparation and only after the film is casted or deposited [
42,
43,
44]. Moreover, the homogeneous and abundant AgNP presence observed after plasma treatment on the film obtained from M.L.1 and deposited by spin-coating (
Figure S5), confirms that the AgNO
3 aggregates solvated in the polymer matrix during the solidification and react with the proper TPU functional groups to be reduced in metallic silver while the volatile THF rapidly evaporated [
43,
45]. Where this porosity is not required, it is always possible to limit the use of the nonsolvent as in the case of WPU (only water used) or to simply dissolve the silver precursor directly in THF, the solvent employed for TPU dissolution.
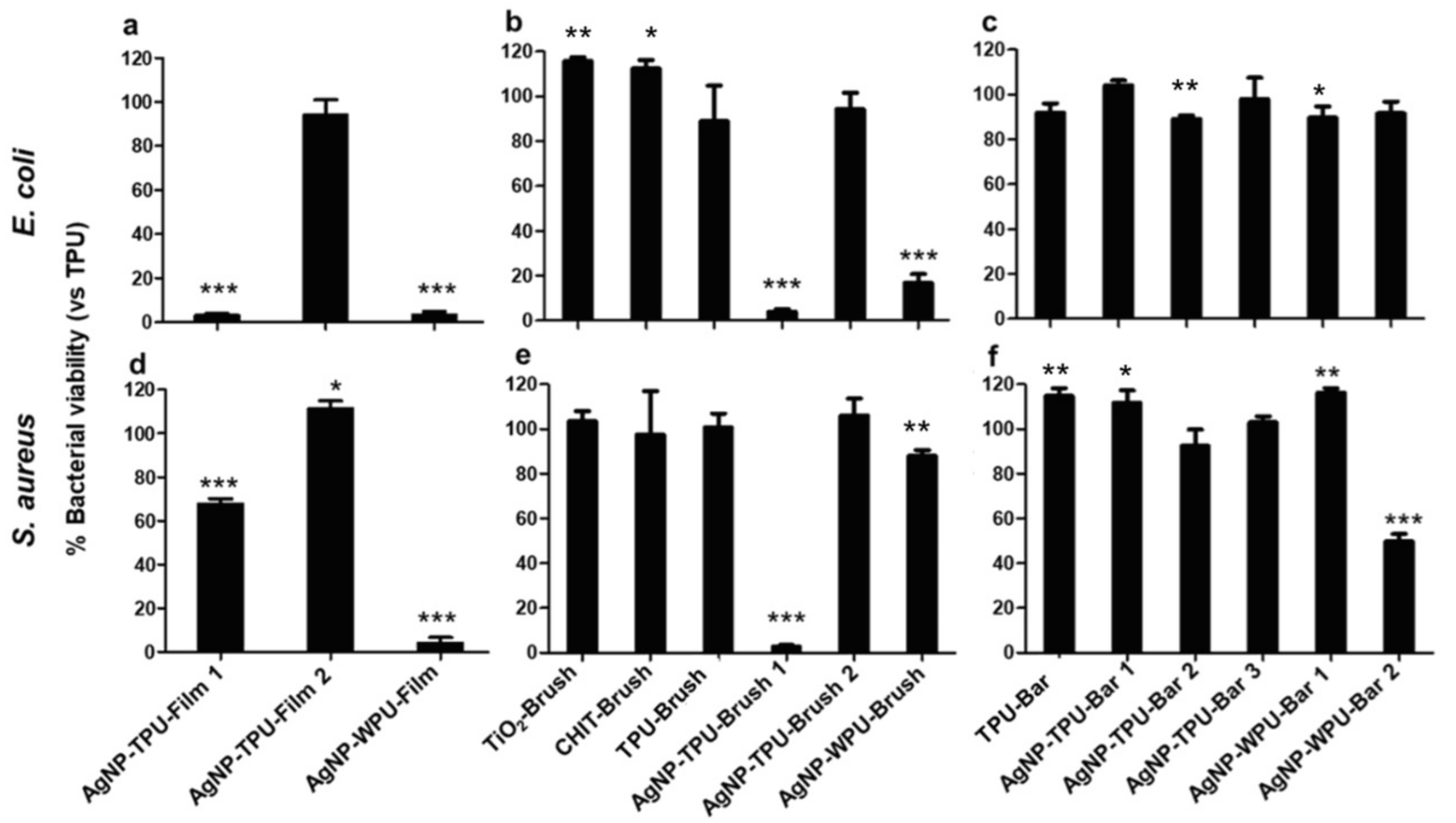
Several antibacterial tests were carried out through direct contact and indirect contact, giving information on the viability of planktonic culture and adhesion of bacteria at two different times of incubation, 6 h and 24 h, respectively (
Figures S8, S9,
Figure 7and
Figure 8). Bacterial planktonic culture tests at 24 h, show better performances for the AgNP casted films, with the highest precursor concentration versus both bacterial strains. A similar result is also observed for the brushed coating (AgNP-TPU-Brush1), clearly demonstrating the role played by the initial precursor concentration. Bar coated samples, even those characterized by the highest amount of silver, do not present similar activities. This suggests a probable loss or a lower transferred amount of the antibacterial during this deposition procedure and for this reason, only at shorter times (6 h) did their capability resist against
S. aureus in almost all the samples, while the casted films are also active versus
E. coli. An analogous trend is also observed in our analysis of the bacterial adhesion tests at 6 h (
Figure S9), where all the samples show good properties mainly versus
S. aureus, with the exception of the casted films which were once again active against both strains. On the contrary, adhesion tests at 24 h show a general improvement of antibacterial properties compared to the viability on planktonic culture at 24 h, especially against
S. aureus, with these activities being directly measured on the film surfaces (
Figure 8). These data are supported by the SEM analysis performed to evaluate bacterial adhesion of
E. coli and
S. aureus on TPU and casted films (A); on brush films (B) and bar coated films (C) (
Figure 9).
To assess whether these materials could present some release of the AgNP, bacterial viability tests after incubation with Ag released in LB were performed. Since all these materials exhibit very high viability values, associated with low antibacterial activity, we conclude that these systems do not release nanoparticles easily.
By playing with the solvent/polymer system and concentrations, important processing and deposition parameters, such as viscosity and wettability, can be influenced, allowing the employment of several deposition techniques, not least the spray one. Some of the formulated antibacterial M.L.s, in particular AgNP-TPU and AgNP-WPU, have been successfully deposited via dip coating on parts of medical probes (
Figure S12). Moreover, several substrates according to the final purpose can be used. For convenience, we deposited such coatings on TPU substrate in order to promote adhesion and aware of substrate surface deformations and stresses arising from the use of the same solvent. However, attempts were made on polyethylene (PE) substrates (Supporting Information,
Figure S13) without success, since we could not properly functionalize the substrate surface [
26]. This weakened the adhesion between the coating and substrate, denying the possibility of performing antibacterial test. However, a simple corona treatment on PE surfaces could enhance the adhesion and allows the preparation of antibacterial coatings over other interesting polymer substrates with commercial relevance.
In our experience, the adhesion of PU-based coatings can be affected by chemical structure modifications associated with reduced hydrogen bonding efficiency responsible for a limited capability of microphase separation [
26]. Moreover, we recently reported on how the TPU hydrogen bonding efficiency can be perturbed by the use of silver, titanium dioxide and chitosan fillers with a consequent effect on the complex microphase separation of this material [
12]. Since the same antibacterials were used for film and coatings preparation, we believe that the observed trend of tack test experiments is a result of the lower molecular ordering introduced. The significant reduction of the cohesion energies observed from M.L.6 to M.L.1 and M.L.2, and from WPU dispersion to M.L.5, respectively, supports this thesis and allows us to associate the reduced adhesion properties and the observed cohesive failures with the lower molecular ordering introduced by the presence of the antibacterial agents.